Oncology
Cancer is a term that refers to a large group of diseases characterized by uncontrolled cell proliferation and spread of abnormal cells. Other terms used interchangeably for cancer are malignant neoplasm, tumor, malignancy, and carcinoma. According to the American Cancer Society (ACS), about 5% of cancer is genetic, whereas 95% is related to other (often modifiable) factors. Only oncologic concepts are presented in this chapter; individual cancers are discussed in the chapters devoted to the affected system. Since cancer and cancer treatment can affect multiple systems, the reader is encouraged to read this chapter along with Chapter 5 for a more complete understanding of its potentially wide-ranging systemic effects.
DEFINITIONS
Normal tissue contains cells of uniform size, shape, maturity, and nuclear structure. Differentiation is the process by which normal cells undergo physical and structural changes as they develop to form different tissues of the body. Differentiated cells specialize in different physiologic functions.
In malignant cells, differentiation is altered and may be lost completely so that the malignant cell may not be recognizable in relationship to its parent cell. When a tumor has completely lost identity with the parent tissue, it is considered to be undifferentiated (anaplastic). In this case, it may become difficult or impossible to identify the malignant cell’s tissue of origin. In general, the less differentiated a tumor becomes, the faster the metastasis (spread) and the worse the prognosis.
Dysplasia
A variety of other tissue changes can occur in the body. Some of these changes are benign, whereas others denote a malignant or premalignant state. Dysplasia is a general category that indicates a disorganization of cells in which an adult cell varies from its normal size, shape, or organization. This is often caused by chronic irritation such as is seen with changes in cervical (uterine) epithelium as a result of long-standing irritation of the cervix. Dysplasia may reverse itself or may progress to cancer.
Metaplasia
Metaplasia is the first level of dysplasia (early dysplasia). It is a reversible and benign but abnormal change in which one adult cell changes from one type to another. For example, the most common type of epithelial metaplasia is the change of columnar epithelium of the respiratory tract to squamous epithelium.
Another example of metaplasia is Barrett’s esophagus (also called Barrett’s epithelium) in which squamous epithelium of the esophagus is replaced by the glandular epithelium of the stomach. Although metaplasia usually gives rise to an orderly arrangement of cells, it may sometimes produce disorderly cellular patterns (i.e., cells varying in size, shape, and orientation to one another). Anaplasia (loss of cellular differentiation) is the most advanced form of metaplasia and is a characteristic of malignant cells only.
Hyperplasia
Hyperplasia refers to an increase in the number of cells in tissue, resulting in increased tissue mass. This type of change can be a normal consequence of physiologic alterations (physiologic hyperplasia) such as increased breast mass during pregnancy, wound healing, or bone callus formation. Neoplastic hyperplasia, however, is the increase in cell mass because of tumor formation and is an abnormal process. The presence of these types of hyperplastic breast tissue increases the risk of later development of breast cancer.141
Tumors
Tumors, or neoplasms, are defined as abnormal growths of new tissue that serve no useful purpose and may harm the host organism by competing for vital blood supply and nutrients. These new growths may be benign or malignant (see the next discussion) and primary or secondary.
A primary tumor arises from cells that are normally local to the given structure, whereas a secondary tumor arises from cells that have metastasized from another part of the body. For example, a primary neoplasm of bone arises from within the bone structure itself, whereas a secondary neoplasm occurs in bone as a result of metastasized cancer cells from another (primary) site.
Carcinoma in situ refers to preinvasive, premalignant epithelial tumors of glandular or squamous cell origin. These tumors have not broken through basement membranes of the squamous cells and occur in the cervix, skin, oral cavity, esophagus, bronchus, and breast. Carcinoma in situ that affects glandular epithelium occurs most commonly in the cervix, breast, stomach, endometrium, large bowel, and prostate gland (prostate intraepithelial neoplasia). How long the characteristic cell disorganization and atypical changes last before becoming invasive is variable for different cancers.
CLASSIFICATIONS OF NEOPLASM
A neoplasm can be classified on the basis of cell type, tissue of origin, degree of differentiation, anatomic site, or whether it is benign or malignant. A benign growth is usually considered harmless and does not spread to or invade other tissue. Certain benign growths, recognized clinically as tumors, are not truly neoplastic but rather represent overgrowth of normal tissue elements (e.g., vocal cord polyps, skin tags, hyperplastic polyps of the colon). However, benign growths can become large enough to distend, compress, or obstruct normal tissues and to impair normal body functions, as in the case of benign central nervous system (CNS) tumors. These tumors can cause disability and even death.
When tumors (benign or malignant) are classified by cell type, they are named according to the tissue from which they arise (Table 9-1). The five major classifications of normal body tissue are epithelial, connective and muscle, nerve, lymphoid, and hematopoietic tissue. Not all tissue types fit into one of these five categories, thus requiring a miscellaneous category for other tissues (not included in Table 9-1) such as the tissues of the reproductive glands, placenta, and thymus.
Table 9-1
Classification of Neoplasms by Cell Type of Origin
| Tissue of Origin | Benign | Malignant |
| Epithelial Tissue | ||
| Surface epithelium (skin) and mucous membrane | Papilloma | Squamous cell, basal cell, and transitional cell carcinoma |
| Epithelial lining of glands or ducts | Adenoma | Adenocarcinoma |
| Pigmented cells (melanocytes of basal layer) | Nevus (mole) | Malignant melanoma |
| Connective Tissue and Muscle | ||
| Fibrous tissue | Fibroma | Fibrosarcoma |
| Adipose | Lipoma | Liposarcoma |
| Cartilage | Chondroma | Chondrosarcoma |
| Bone | Osteoma | Osteosarcoma |
| Blood vessels | Hemangioma | Hemangiosarcoma |
| Smooth muscle | Leiomyoma | Leiomyosarcoma |
| Striated muscle | Rhabdomyoma | Rhabdomyosarcoma |
| Nerve Tissue | ||
| Nerve cells | Neuroma | — |
| Glia | — | Glioma or neuroglioma |
| Ganglion cells | Ganglioneuroma | Neuroblastoma |
| Nerve sheaths | Neurilemoma | Neurilemma sarcoma |
| Meninges | Meningioma | Meningeal sarcoma |
| Retina | — | Retinoblastoma |
| Lymphoid Tissue | ||
| Lymph nodes | — | Lymphoma |
| Spleen | — | — |
| Intestinal lining | — | — |
| Hematopoietic Tissue | ||
| Bone marrow | — | Leukemias, myelodysplasia, and myeloproliferative syndromes |
| Plasma cells | — | Multiple myeloma |
Epithelium covers all external body surfaces and lines all internal spaces and cavities. The skin, mucous membranes, gastrointestinal tract, and lining of the bladder are examples of epithelial tissue. The functions of epithelial tissues are to protect, excrete, and absorb. Cancer originating in any of these epithelial tissues is called a carcinoma. Tumors derived from glandular tissues are called adenocarcinomas.
Connective tissue consists of elastic, fibrous, and collagenous tissues such as bone, cartilage, and fat. Cancers originating in connective tissue and muscle (mesenchymal origin) are called sarcomas. Nerve tissue includes the brain, spinal cord, and nerves and consists of neurons, nerve fibers, dendrites, and a supporting tissue composed of glial cells.
Tumors arising in nerve tissue are named for the type of cell involved. For example, tumors arising from astrocytes, a type of glial cell thought to form the blood-brain barrier, are called astrocytomas. Tumors arising in nerve tissue are often benign, but because of their critical location they are more likely to be harmful than benign tumors in other sites.
Malignancies originating in lymphoid tissues are called lymphomas. Lymphomas can arise in many parts of the body, wherever lymphoid tissue is present (see Fig. 7-8). The most common sites to find lymphoid malignancies are the lymph nodes and spleen. However, lymphomas can appear in other parts of the body such as the skin, CNS, stomach, small bowel, bone, and tonsils.30
Hematopoietic malignancies include leukemias, multiple myeloma, myelodysplasia, and the myeloproliferative syndromes.
Staging and Grading
Staging is the process of describing the extent of disease at the time of diagnosis to aid treatment planning, predict clinical outcome (prognosis), and compare the results of different treatment approaches. The stage of disease at the time of diagnosis reflects the rate of growth, the extent of the neoplasm, and the prognosis. A simplified way to stage cancer is as follows:
Stage 0: Carcinoma in situ (premalignant, preinvasive)
Stage I: Early stage, local cancer.
Stage II: Increased risk of spread because of tumor size.
Stage III: Local cancer has spread but may not be disseminated to distant regions.
Stage IV: Cancer has spread and disseminated to distant sites.
In some cases, cancer may be staged as II or III depending on the spread of the specific type of cancer. For example, in Hodgkin’s disease, stage II indicates lymph nodes are affected on one side of the diaphragm. Stage III indicates affected lymph nodes above and below the diaphragm.208
Systems of Staging
Staging systems are specific for each type of cancer. For example, cervical cancer is staged using the International Federation of Gynecology and Obstetrics (FIGO) System of Staging, which is based on clinical examination, rather than surgical findings (see Box 20-3). Lymphomas are staged using the Ann Arbor staging (Box 9-1).
The TNM (tumor, node, metastases) system is used most often for solid tumors and has been adapted for other types of tumor. Some cancers do not have a staging system (e.g., brain cancer) and some can be staged using more than one system. The International Union Against Cancer (UICC) is the universally accepted staging system, which incorporated the TNM classification of malignant tumors the American Joint Committee on Cancer (AJCC) system previously used. The National Cancer Institute provides more detailed information about staging.130
It is important to realize the TNM staging system is simply an anatomic staging system that describes the anatomic extent of the primary tumor, as well as the involvement of regional lymph nodes and distant metastases. Revisions to the TNM staging system are made as the understanding of the natural history of tumors at various sites improves with advancing technology. In the TNM classification scheme, tumors are staged according to the following basic components (Box 9-2):
• Tumor (T) refers to the primary tumor and carries a number from 0 to 4.
• Regional lymph nodes (N) represents regional lymph node involvement; also ranked from 0 to 4.
• Metastasis (M) is zero (0) if no metastasis has occurred or 1 if metastases are present.
Numbers are used with each component to denote extent of involvement; for example, T0 indicates undetectable, and T1, T2, T3, and T4 indicate a progressive increase in size or involvement.155
In the TNM system, clinical stage is denoted by a small “c” before the stage (e.g., cT2N1M0) or by a small “p” to indicate the pathologic stage (e.g., pT2N0). Clinical staging is made indirectly by observation of the person before the tumor is treated or removed. The pathologic stage is determined by direct examination of the tumor by the pathologist once it has been removed and is considered a more accurate reflection of the tumor and its spread. Not all tumors are resected or excised so pathologic staging is not always available. Pathologic staging may underestimate the true stage for individuals who received adjuvant treatment (radiation or chemotherapy) before surgery.
Pathologic staging may also be in error if the slice of tissue examined is normal; oversight of tumor burden can lead to misstaging and delayed diagnosis and treatment. Staging continues to improve with new, more precise levels of screening sensitivity. For example, molecular screening for the presence of markers characteristic of some diseases nearly eliminates the possibility of true disease stage.208
Grading
Grading 1 through 4 is another way to define a tumor and classifies the degree of malignancy and differentiation of malignant cells. For example, a low-grade tumor typically has cells more closely resembling normal cells and tends to remain localized, whereas a high-grade tumor has poorly differentiated cells that tend to metastasize early. Staging is more predictive than grading.
INCIDENCE
Estimates of worldwide incidence, mortality, and prevalence of 26 cancers are available from the International Agency for Research on Cancer (IARC). Geographic variations between 20 large areas of the world are presented. Most of the international variation is due to exposure to known or suspected risk factors related to lifestyle or environment. The IARC has been researching and providing a database of global cancer estimates (GLOBOCAN) for the last 30 years.55
The most commonly diagnosed cancers are lung, breast, and colorectal; the most prevalent cancer in the world is breast cancer (4.4 million survivors up to 5 years after diagnosis). Lung cancer accounts for the most number of cancer deaths worldwide.143 The ACS publishes annual cancer statistics and estimates cancer trends (Fig. 9-1). Each year the ACS calculates estimates of the number of new cancer cases and expected cancer deaths in the United States and compiles the most recent data on cancer incidence, mortality, and survival.96
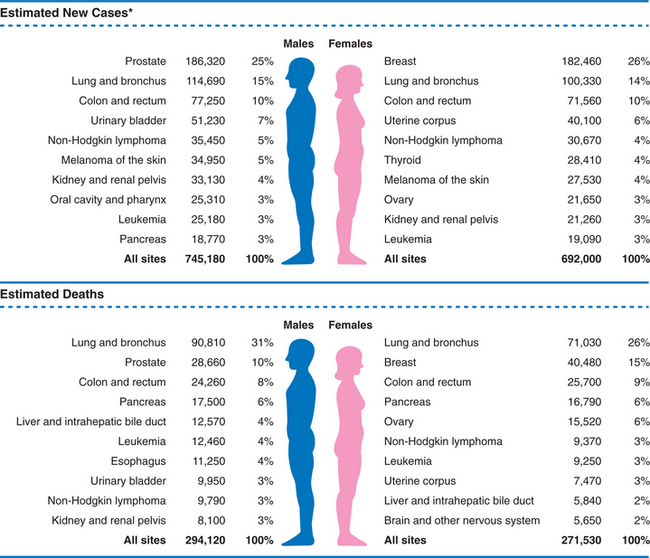
Figure 9-1 Estimated new cancer cases (top) and cancer deaths (bottom), 2008, in the United States (percent distribution of sites by gender). (From Jemal A, Siegel R, Ward E, et al: Cancer statistics, 2008, CA Cancer J Clin 58[2]:71-91, 2008.)
Based on statistical estimates, in the year 2007, the ACS predicts about 1.4 million new cases of invasive cancer in the United States and approximately 565,000 cancer-related deaths. This figure does not include most skin cancers, which are expected to affect 1 million people per year.96
It is estimated that at least one in three people will be diagnosed with some form of invasive cancer in their lifetime and 3 of 5 people will be cured and/or survive 5 years after cancer treatment. However, cancer is still the second leading cause of death in the United States, exceeded only by heart disease. Poor health and nutrition habits, continued smoking, ozone destruction, and a long-term lack of exercise among many people continue to be discussed as contributors to the overall rise of this disease.178
The National Cancer Institute (NCI) established the Surveillance, Epidemiology, and End Results (SEER) program in 1973 as a way to report population-based data of site-specific incidences and outcomes of cancer. Because the United States does not have a nationwide cancer registry, the precise number of new cases of cancer diagnosed each year in the United States is not fully known. As a result of this, the number of new cancer cases occurring annually is estimated using complex statistical measures.157
Trends in Cancer Incidence and Survival
Overall incidence of cancer peaked in 1990 and has declined in the last decade by an average of 1.1% annually, with a 1.4% decline in cancer death rates. In 2003 and 2004, the rate doubled to 2% per year, largely attributed to smoking cessation among men, which peaked in the mid1960s. The latest drop in cancer deaths occurred across all four major cancer types (lung, colon and rectal, prostate, and breast; lung cancer deaths in women stayed relatively constant).
Survival rates for cancer are on the rise, increasing from 50% to 64% over the last 30 years. There are nearly 10 million cancer survivors in the United States today. Targeted cancer therapies promise to improve these statistics in the coming years. Cancer prevention strategies may reduce the incidence of cancer occurrence and recurrence.82
Gender-Based Incidence
Among men, the most common cancers are predicted to be cancers of the prostate, lung and bronchus, and colon/rectum. Among women, the three most commonly diagnosed cancers are expected to be cancers of the breast, lung and bronchus, and colon/rectum.
The largest decreases in deaths occurred among men (especially among black men) who bear the heaviest overall cancer burden and colorectal cancer in particular. Officials have attributed the steady downward trends to improved vigilance among Americans, who are benefiting from early screening and advances in treatment, as well as smoking less, improving their diets, and exercising more.
The decline in rates of breast cancer deaths has been attributed in part to increased mammography but also to more aggressive therapy; overall decline in deaths among women may also be the result of the recent falloff in hormone replacement therapy.
Although death rates from breast cancer continue to decline, the incidence of the disease climbed 1.2% annually between 1992 and 2000. Breast cancer alone accounted for approximately 178,400 new cancer cases in women in 2007.96
Likewise, improved screening, detection, and treatment of prostate cancer have resulted in a decline in the death rate associated with this type of cancer. About a dozen cancers continue to rise in incidence or mortality, including melanoma, non-Hodgkin’s lymphoma, thyroid, esophageal cancer, breast (increased incidence but decreased mortality), and female lung cancer.
ETIOLOGY
The cause of cancer varies, and causative agents are generally subdivided into two categories: those of endogenous (genetic) origin and those of exogenous (environmental or external) origin. It is likely that most cancers develop as a result of multiple environmental, viral, and genetic factors working together to disrupt the immune system along with failure of an aging immune system to recognize and scavenge cells that have become less differentiated.
Certain cancers show a familial pattern, giving people a hereditary predisposition to cancer. The most common cancers showing a familial pattern include prostate, breast, ovarian, and colon cancers. Research efforts have been directed at finding genes associated with various cancers that could identify high-risk individuals for screening and early detection.
The ACS estimates that 50% of all cancers are caused by one or more of nearly 500 different cancer-causing agents (e.g., tobacco use, viruses, chemical agents, physical agents, drugs, alcohol, hormones).178 Etiologic agents capable of initiating the malignant transformation of a cell (i.e., carcinogenesis) are called carcinogens. The study of viruses as carcinogens is one of the most rapidly advancing areas in cancer research today.
Researchers now have evidence that viruses play a role in the pathogenesis of cervical carcinomas, some hepatomas, Burkitt’s lymphomas, nasopharyngeal carcinomas, adult T-cell leukemias, and indirectly, many Kaposi’s sarcomas.40 Viruses, such as the human immunodeficiency virus (HIV), the causative agent of acquired immunodeficiency syndrome (AIDS), weaken cell-mediated immunity, resulting in malignancies.
Chemical agents (e.g., tar, soot, asphalt, dyes, hydrocarbons, oils, nickel, or arsenics) and physical agents (e.g., radiation or asbestos) may cause cancer after close and prolonged contact with these agents. Most people affected by chemical agents are industrial workers. Radiation exposure is usually from natural sources, especially ultraviolet radiation from the sun, which can cause changes in deoxyribonucleic acid (DNA) structure that lead to malignant transformation. Notable exceptions include past history of radiation treatment for acne, thymus, or thyroid conditions. Basal and squamous cell carcinomas and malignant melanoma are all linked to ultraviolet exposure. See further discussion of chemical and physical agents in Chapter 4.
Some drugs, such as cancer chemotherapeutic agents, are in themselves carcinogenic. Cytotoxic drugs, including steroids, decrease antibody production and destroy circulating lymphocytes. Cancer clients treated with chemotherapy are at risk for future development of leukemia and other cancers.
Hormones have been linked to tumor development and growth, such as estrogen stimulating the growth of the endometrial lining, which over time becomes anaplastic. Other types of cancer occurring in target or hormone-responsive tissues include ovary and prostate cancers.
Excessive alcohol consumption is associated with cancer of the mouth, pharynx, larynx, esophagus, and pancreas. It can also indirectly contribute to liver cancer (i.e., alcohol causes liver cirrhosis, which is associated with cancer). The possible association between alcohol and breast cancer is under investigation. The pathophysiologic link remains unclear, but researchers postulate that alcohol influences the metabolism of estrogen, and increased estrogen exposure is a known risk factor for breast cancer (see the section on Breast Cancer in Chapter 20).
RISK FACTORS
Advancing age is one of the most significant risk factors for cancer. In addition to age and the carcinogens described earlier in the section on Etiologic Factors, predisposing factors also influence the host’s susceptibility to various etiologic agents (Box 9-3), which are discussed further in the next section of this chapter.
Nine modifiable risk factors are responsible for more than one-third of cancer deaths worldwide (tobacco, alcohol, obesity, inactivity, diet/nutrition, unsafe sex, urban air pollution, indoor smoke from household fuels, or contaminated injections in health care settings). Of these, smoking and alcohol consumption are the most damaging. This means that even without the potential benefits of early detection and treatment, at least one-third of cancer deaths are preventable.39,200
Experimental and epidemiologic evidence has established an association between at least eight viruses and various cancer sites. At least 10% of cancer worldwide is caused by viruses, tobacco, and diet. Some risk factors are interactive and become exponential rather than additive (e.g., alcohol and smoking for oral pharyngeal cancers).
There is no evidence to support popular theories that some people are more likely to develop cancer because of specific personality traits such as anger, hostility, frustration, sexual repression, or conflicted parent-child relationship. Additionally, cancer survival is not predicted by personality type, including neuroticism, extroversion, or low self-esteem.75,128
Aging
Age over 50 years (some experts say age over 40 years) is a significant risk factor for the development of cancer. The association of cancer and aging is becoming more common because of the aging of the general population. The median age for all cancer is approximately 70 years and will become even older over the next several decades.52 The risk of multiple diseases (comorbidity) also increases with age, creating limitations in the life expectancy of individual aging adults and enhancing the likelihood of treatment complications.
Older people may be more susceptible to cancer simply because they have been exposed to carcinogens longer than younger people. The effects of age on immune function and host defense are being studied to determine what the association is between cancer and age (see the section on Aging and the Immune System in Chapter 7).
Factors such as accumulated nonlethal damage to DNA by free radicals, increased proinflammatory factors, and age-associated declines in DNA repair are important.52 Studies on mutations in cancer-causing structures, such as telomeres (a region of DNA at the end of chromosomes), DNA repair aberrations, and dysregulation of important hormonal and immune modulators, are all being reported as potential reasons for the increasing incidence of cancer in older adults.13,183
Clues about the lifespan of a cell and about aging in general are emerging from recent research on telomeres. In normal cells, the telomere shortens each time a cell divides. Telomeres provide chromosomal stability by protecting the ends from degradation and recombination. A minimal telomere length is needed to maintain tissue homeostasis.126
The cell dies when the telomere becomes so short that it can no longer divide. An important enzyme, telomerase, helps keep normally dividing cells healthy by rebuilding the telomeres. Telomerase normally shuts down when cells are mature, but in cancer, the enzyme enables cancer cells to grow with unlimited cell divisions. Telomerase is active in up to 85% of all human cancers.25,26,145 See also the section on Cellular Aging in Chapter 6. This understanding has led to discoveries regarding the lifespan of human cells, their relationship to aging, and the development of many illnesses associated with aging such as cancer. It has been reported that normal cells do not divide indefinitely during the lifespan of a human because of a phenomenon called the Hayflick limit and a stopping process or permanent growth arrest called cellular senescence.
The Hayflick limit was discovered by Leonard Hayflick in 1965.79 Hayflick observed that cells dividing in cell culture divided about 50 times before dying. As cells approach this limit, they show more signs of old age. For most differentiated cells the limit to the number of times a cell divides has been determined in all cell types. The human limit is around 52, but there is some variation from cell type to cell type and more significantly, from organism type to organism type (e.g., mice and humans). This limit has been linked to the shortening of telomeres and is believed to be one of the causes of aging. Telomeres may act as cellular clocks that control aging. This is called the telomere theory of aging. If the shortening of telomeres can be slowed or prevented, life expectancy may be extended but perhaps at a higher risk of tumorigenesis.83
Many in vitro and in vivo studies support the idea that telomere length is strongly correlated with lifespan. Longer telomeres have been associated with shorter lifespan (e.g., mice have very long telomeres and a short lifespan; the opposite is true for humans). Telomeres shorten progressively with each cell division; when a critical telomere length (Hayflick limit) is reached, the cells undergo senescence and subsequently apoptosis (programmed cell death).83
An alternate view is the theory of dysfunctional senescence. Some researchers have proposed that failure of cells or tissues to enter into cellular senescence occurs as a result of defects in genomic maintenance mechanisms after years of mutation and leads to cancer.23,204 In other words, cellular senescence may reduce cancer mortality rather than promote it late in life, thus positively contributing to longevity in organisms with renewable tissues.112
Cancer cells constitute one exception to the limits on cell division. It is believed that the Hayflick limit exists principally to help prevent cancer. If a cell becomes cancerous and the Hayflick limit is approaching, the cell will only be able to divide a limited number of times before it dies. Once it reaches this limiting number of divisions, the formed tumor will no longer be able to reproduce and the cells will die off.
Cancer cells that have found ways around the Hayflick limit are referred to as “immortal.” Such immortal cells may still die, but the group of immortalized cells produced from cell division of an immortal cell has no limit as to how many times cell division might take place. Telomeres in immortal cells are maintained by telomerase. Irrespective of telomere length, if telomerase is active, telomeres can be maintained at a sufficient length to ensure cell survival.83 This presents a unique challenge in preventing and killing cancer cells. Creating telomerase inhibitors may possibly produce a means of supporting anticancer activity.
People age 65 years and older have a risk of cancer development much greater than younger persons, and some cancers in the older adult population seem to be biologically different from those in younger people. For example, the poor prognosis for older adults with acute leukemia is not just due to poor tolerance of aggressive chemotherapy but is more likely associated with cytogenetic resistance to chemotherapy.90
All the highest-incidence cancers affect older adults in larger numbers. In both men and women over 65 years, cancers of the colon/rectum, stomach, pancreas, and bladder accounted for two-thirds to three-fourths of the total number of these malignancies. More than 65% of lung cancers and 50% of non-Hodgkin’s lymphomas occur in older men and women; 77% of the cases of prostate cancer occurred in men older than 65 years; and 48% of breast cancer and 46% of ovarian cancer occurred in women over 65 years.218
As previously mentioned, malignancies of the lung, colon/rectum, breast, and prostate account for the highest number of cancer deaths in the United States. Malignancies of the pancreas, stomach, ovary, and bladder and non-Hodgkin’s lymphomas are also a major cause of cancer deaths. For each of these cancers, more than one-half of the cancer deaths occur in persons older than 65 years.218
Lifestyle
Lifestyle or personal behaviors, such as tobacco use, diet and nutrition, alcohol use, and sexual and reproductive behavior, are cited as risk factors for the development of cancer. Lifestyle-related risk factors for cancer combined with cancer-causing substances in the environment and the presence of genes that increase the risk of cancer account for 70% of the total risk for developing cancer.
Tobacco
Both epidemiologic and experimental data support the conclusion that tobacco (including smokeless tobacco) is carcinogenic and remains the most important cause of cancer. Tobacco use accounts for approximately 30% of cancer deaths, with lung cancer now the leading cancer causing deaths in both genders.42 Cigarette smoking is related to nearly 90% of all lung cancers, and accumulating evidence suggests that cigarette smoking increases the incidence of cancer of the bladder, pancreas, and to a lesser extent, the kidney, larynx, oral cavity, and esophagus.
Diet and Nutrition
The major role of diet and nutrition in affecting cancer risk is well established.3a,6,19 Consumption of a poor diet may blunt the immune system’s natural defense mechanisms against genetic damage caused by long-term exposure to an environmental carcinogen. Diet and nutrition can directly influence various hormonal factors affecting growth and differentiation in the carcinogenic process. A healthful diet is thought to act, at least in part, to detoxify carcinogens and to inhibit certain processes in carcinogenesis, particularly at the stage of growth and spread. Diet and nutrition can influence these processes (positively or negatively) by providing bioactive compounds to specific tissues via the circulatory system or by modulating hormone levels.
Differences in certain dietary patterns among populations explain a proportion of cancers. These dietary patterns in combination with physical inactivity contribute to obesity and metabolic consequences such as increased levels of growth factor, insulin, estrogen, and possibly testosterone. These hormones tend to promote cellular growth.68
A history of obesity and/or type 2 diabetes are risk factors for breast, prostate, and colorectal cancer. Excess weight also contributes to cancers of the uterus, kidney, esophagus, pancreas, and gallbladder.21,144 Fat (adipose tissue) as an endocrine gland and its role in cancer is discussed in Chapter 11.
It is estimated that approximately one-third of cancer mortality in developing countries may result from dietary causes.7,210 The intake of cured, pickled, smoked, salted, and preserved food has been conclusively linked to stomach cancer, and it is suspected that there is a correlation between the amount of fat in the diet and the incidence of colorectal cancer in the United States.21 There is a similar correlation between excessive red meat consumption (defined as beef, pork, or lamb) and prostate, colon, and/or rectal cancer.103 Epidemiologic data also suggest links between fat intake and prostate and ovarian cancers, although not all findings are consistent.80
Reduction of cancer risks for most epithelial tumors, especially colorectal cancer, has been demonstrated repeatedly in the presence of increased dietary intake of fresh fruits and vegetables and fiber. High cruciferous vegetable consumption may reduce bladder cancer risk and the risk of non-Hodgkin’s lymphoma.120,220 Although the role of fruits and vegetables and their antioxidant qualities in reducing free radicals that contribute to cancer of various sites has been proved, a recent report suggests the recommendation to eat an abundance of fruits and vegetables to reduce cancer risk may be overstated. Cardiovascular benefits were much more evident; the 30% to 50% reduction in cancer that research suggested previously was not supported.209 Experts reviewing the results of this study questioned the methodology used and pointed out that choosing healthy foods, such as fruits and vegetables, to meet caloric and nutritional needs is more protective than a diet laden with sugar, fat, or low-nutrient foods.
Additionally, cancer survivors are often highly motivated to improve nutrition and begin an exercise program after receiving a diagnosis of cancer. The ACS continues to publish and update best clinical practices related to optimal nutrition and physical activity during and after cancer treatment. In the absence of scientific evidence that diet, nutrition, and physical activity can prevent cancer recurrence, reasonable conclusions are offered.47,103
Cancer and cancer treatment can cause profound metabolic and physiologic alterations affecting the body’s needs for adequate nutritional intake. Gastrointestinal side effects of treatment can lead to loss of appetite and weight loss accompanied by malnutrition. All the major treatment modalities (e.g., surgery, chemotherapy, or radiation) can adversely impact how the body digests, absorbs, and uses food. Preserving lean body mass is an important goal of nutritional care for survivors, especially during active cancer treatment.174
The use of nutritional supplements and antioxidants remains controversial. Until more evidence is available that suggests more benefit than harm, the Institute of Medicine suggests it is prudent for cancer survivors receiving chemotherapy or radiation therapy to avoid exceeding more than 100% of the daily value for antioxidant-type vitamins during the treatment phase.92
Alcohol
Alcohol consumption has been linked to increased rates of cancer of the mouth, pharynx, larynx, esophagus, liver, breast, and probably colon. In people who have been diagnosed with cancer, alcohol intake could also affect the risk for new primary cancers of these sites. Alcohol intake can increase the circulating levels of estrogens, theoretically increasing the risk for breast cancer recurrence.166,187
With tobacco use, alcohol interacts with smoke synergistically, increasing the risk of malignant tumors by acting as a solvent for the carcinogenic smoke products and thus increasing the absorption of carcinogens. Evidence is suggestive (but not yet convincing) in associating alcohol consumption and cancers of the colon, pancreas, breast, bladder, and head and neck.80
Sexual and Reproductive Behaviors
Sexual and reproductive behaviors are linked to the risk of developing various cancers. For example, the risk of developing cervical cancer is linked with early sexual intercourse and multiple partners. Pregnancy and childbearing seem to be protective against cancers of the endometrium, ovary, and breast. Prolonged lactation may also have a significant impact in the reduction of breast cancer risk by reducing the cumulative exposure of breast tissue to estrogen.80 Other risk factors for breast cancer are discussed in Chapter 20.
Hormonal Exposure
Hormonal exposure is a factor for women. For example, prolonged exposure to estrogen (e.g., early onset of menses, menopause after age 50, nulliparity or no children, first child after age 30, never breastfed children, or use of first-generation oral contraceptives before 1975) is a risk factor for estrogen-sensitive breast cancer.
Prolonged use of estrogen hormone replacement therapy for relief of menopausal symptoms has been linked with increased rates of breast cancer. Data from the Women’s Health Initiative (WHI) resulted in a halt to the routine use of estrogen and progestin in combination (Prempro) in 2002 and estrogen alone (Premarin) in 2004. When compared with a placebo group, it was clear that hormone users were experiencing more breast cancer, heart disease, stroke, and blood clots. Estrogen showed some benefit, but it was not enough to outweigh the risks.215
Growth factors, such as insulin growth factor 1 (IGF-1), and hormones, such as estrogen and testosterone, are considered risk factors linked with cancers other than breast and prostate (e.g., lung, endometrial, and colon). A growing body of recent literature indicates that besides its essential role in growth and development, growth hormone may play a role in the development and progression of cancer.147
The insulin-cancer hypothesis postulates that complex links between excess body weight, the insulin–IGF axis, and cancer suggest molecular mechanisms are present that lead to increased availability of IGF-1, providing a cellular environment that favors tumor formation.162 Growth factor signaling pathways appear to be up-regulated in hormone-resistant tumors and interact with estrogen receptor signaling, which remains functional even after long-term endocrine deprivation. Intensive research efforts to develop antitumor agents that inhibit IGF signaling in human tumors are underway.74
Geographic Location and Environmental Variables
The incidence of different types of cancer varies geographically. People living in rural areas are less likely to use preventive screening services or to exercise regularly. Colon cancer is more prevalent in urban than in rural areas, but in rural areas, especially among farmers, skin cancer is more common. Availability of specialty care is a possible contributing issue for this group of people.
The greater susceptibility of certain geographic areas within the United States is probably related to exposure to different carcinogens.35 The increased incidence of cancer found in urban areas may be related to the increased pool of minorities, increased poverty represented in this group, local smoking ordinances, and diet (e.g., cost and availability of fresh fruits and vegetables).150
Occupational or environmental exposure to chemicals (e.g., herbicides, insecticides, dyes), fibers (e.g., asbestos), radon, and air pollution is a risk factor for lung and hematologic cancers. Researchers are investigating the possible causal relationship between environmental exposure and the increased incidence of childhood cancers. The U.S. Environmental Protection Agency (EPA) has identified the carcinogenic effects from hazardous exposures. Heritable genetic and chromosomal mutations caused by environmental or occupational exposures to agents (e.g., chemicals, radiation) can be passed on to the next generation. For further discussion, see Chapter 4.
According to the seventh report on the Biological Effects of Ionizing Radiation (BEIR VII) issued by the National Academy of Science, exposure to even low-dose imaging radiology (including computed tomography [CT] scans) can result in the development of malignancy. Exposure to medical x-rays is linked with leukemia, thyroid cancer, and breast cancer. There is a 1 in 1000 chance of developing cancer from a single CT scan of the chest, abdomen, or pelvis. The latency period for leukemias is 2 to 5 years and 10 to 30 years for solid tumors.129
Ethnicity
Despite advances in cancer diagnosis, treatment, and survival, racial and ethnic minorities suffer disproportionately from cancer. Poverty has emerged as a significant factor influencing poor cancer outcomes for all races, especially among minorities.156 Inequities in insurance status adversely affect low income families, preventing individual members from obtaining screening, access to quality care, or the entire range of cancer care available.185
In particular, racial disparities exist between Caucasians and other groups, especially African Americans. Overall, incidence and mortality from cancer is 10% higher in African Americans compared to Caucasians.18,185 Studies have shown that equal treatment yields equal outcomes among individuals with equal disease.12,93 At present, this increased incidence is attributed to preventable risk factors such as the absence of early screening, delayed diagnosis, and smoking and diet. The number of African American men who smoke is decreasing, but the incidence of lung cancer and other smoking-related diseases remains high, possibly because black men tend to smoke cigarettes with a higher tar and nicotine content. The incidence rates of prostate cancer among black men are at least 50% higher than rates for men of other ethnic groups.96
Lung cancer is the leading cause of cancer death among African American women. The number of African American women (aged 45 to 54 years) who have died from lung cancer has increased 30% over the last 2 decades.212 The number of African American women of all ages who have died from breast cancer has risen nearly 20% over the last 25 years. Breast cancer is the second leading cause of death for African American women. Colorectal cancer has increased in both African American men and women; black women are twice as likely to develop cervical cancer and nearly three times as likely to die from it as other women; African American men have the world’s highest rate of prostate cancer.98
Some specific forms of cancer affect other ethnic groups at rates higher than the national average (e.g., stomach and liver cancers among Asian-American populations and colorectal cancer among Alaska Natives). African Americans have a lower incidence of bladder cancer but higher mortality rates compared to Caucasians.201 The incidence of and mortality rates for esophageal cancer are twice as high for African Americans compared to Caucasians.14
Differences among ethnic groups represent a challenge to understand the reasons and an opportunity to reduce illness and death while improving survival rates. Hispanic people originate from 23 different countries with a wide range of diversity. Racial variations exist in tumor growth, susceptibility, and treatment response. For example, Latino populations have different drug resistance gene expression than non-Latino whites.138
Hispanics are the poorest minority group with the highest uninsured rate of all groups.138 They have higher rates of cervical, esophageal, gallbladder, and stomach cancers. New cases of female breast and lung cancers are increasing among Hispanics, who are diagnosed at later stages and have lower survival rates than whites.
Asian Americans have a unique situation in that they are the only racial/ethnic group to experience cancer as the leading cause of death with proportionately more cancer of infectious origin (e.g., human papillomavirus–induced cervical cancer, hepatitis B virus–induced liver cancer, and stomach cancer) than any other minority group. Cultural barriers to intervention exist such as overcoming resistance to physician visits, reducing tobacco use, and increasing exercise.28
Precancerous Lesions
Precancerous lesions and some benign tumors may undergo later transformation into cancerous lesions and tumors. Common precancerous lesions include pigmented moles, burn scars, senile keratosis, leukoplakia, and benign adenomas or polyps of the colon or stomach. All such lesions need to be examined periodically for signs of changes.
Stress
Recent research suggests a strong link between stress and cancer. Chronic physical or emotional stress can cause hormonal or immunologic changes or both, which in turn can facilitate the growth and proliferation of cancer cells. There is substantial evidence from both healthy populations and people with cancer that links psychologic stress with immune down-regulation. Distress and depression are associated with two important processes for carcinogenesis: poorer repair of damaged DNA and alterations in apoptosis.
Both aging processes and psychologic stress affect the immune system; in fact, the effects of stress and age are interactive. Psychologic stress can both mimic and exacerbate the effects of aging. Older adults often show greater immunologic impairment to stress compared to younger adults.72 Conversely, the possibility that psychologic interventions and social support may enhance immune function and survival is under further investigation.100 Psychologic modulation of immune function is now a well-established phenomenon. Psychoneuroimmunology and psychoneuroendocrinology research focuses on how the brain and body communicate with each other in a multidirectional flow of information that consists of hormones, neurotransmitter/neuropeptides, and cytokines.205
Proponents of psycho-oncology (psychoneuroimmunology and cancer) suggest that advances in mind-body medicine research combined with healthy nutrition and lifestyle choices can have a significant impact on health, health maintenance, disease, and disease prevention, including cancer.99,205 See the section on Psychoneuroimmunology Theory in Chapter 1; see also the section on Stress in Chapter 3.
PATHOGENESIS
Early in the study of cancer the concept that neoplasia originates in a single cell by acquired genetic change was proposed and remains today the view of cancer pathogenesis most supported by experimental evidence. This hypothesis, called the somatic mutation theory, was first substantiated when investigations of tumors confirmed that tumor cells are characterized by chromosomal abnormalities, numerical and structural.
The discovery that chromosomal aberration is one of the basic mechanisms of tumor cell proliferation laid the foundation of modern cancer cytogenetics (study of chromosomes in cancer). Chromosomal changes can include addition or deletion of entire chromosomes (numerical changes) or translocations, deletions, inversions, and insertions of parts of chromosomes (structural changes). Translocations occur when two or more chromosomes exchange material and are common in leukemias and sarcomas. Deletions or losses of chromosomal material are common in epithelial adenocarcinomas of the large bowel, lung, breast, and prostate. Chromosomal deletions may lead to neoplastic development when a tumor suppressor gene is lost. Chromosomal inversions and insertions are less common but still cause abnormal juxtaposition (side-by-side placement) of genetic material.164
At first the question arose: are acquired chromosomal abnormalities the cause of the neoplastic changes in cells or merely the result of the neoplastic state? Chromosomal banding techniques developed in the 1970s have allowed precise identification of chromosomal changes. This information, along with molecular genetic techniques developed during the 1980s, has enabled researchers to investigate this question by examining tumor cells at the level of individual genes.
From these studies, two functionally different classes of cancer-relevant genes have been detected: (1) the dominant oncogenes and (2) the recessive tumor suppressor genes. Both gene classes have been detected at just those chromosomal sites that are visibly involved in cancer-associated rearrangements. To date, over 100 genes have been found to be structurally or functionally altered after neoplasia-associated chromosomal abnormalities.
Exactly how these chromosomal changes contribute to the malignant process remains unclear. Chromosomal rearrangements may lead to oncogene activation, either by a regulatory change causing increased production of normal oncogene-encoded peptides or by creating a deranged oncogene template that codes for an abnormal protein product.
Another proposed mechanism suggests that chromosomal changes inactivate a tumor suppressor gene through chromosomal deletion. Loss of tumor suppressor genes is suspected because chromosomal regions found to be consistently missing in tumor cells have been observed in carcinomas of the lung, breast, bladder, and kidney.
An important tumor suppressor gene currently under study is p53. The p53 gene appears to trigger programmed cell death (apoptosis) as a way of regulating uncontrolled cellular proliferation. Mutations in the p53 tumor suppressor gene result in loss of the ability of the gene protein to bind with DNA and act as a suppressor for the division of that cell.139 Mutations in p53 are associated with resistance to chemotherapy and radiation therapy.
Additionally, researchers have demonstrated that cancer cells develop multiple mechanisms of their own to evade apoptosis. Cancer cells can inactivate proapoptotic factors or up-regulate antiapoptotic factors. Strategies to induce apoptosis specifically in tumor cells are currently under investigation. One proapoptotic protein that has been shown to induce apoptosis in a wide variety of tumor cells is a member of the tumor necrosis factor (TNF) superfamily called TNF–related apoptosis-inducing ligand (TRAIL).27,167
Another genetic suppressor of cell growth and division also plays a part in the aging process. As cells divide and grow older, there is continuous progressive shortening of the end portions of the chromosomes or telomeres of those cells. Studies of human fibroblasts and other human tissues have shown a very close association between the development of cancer and the overproduction of the enzyme telomerase. When this enzyme is present, it prevents the telomeres from shortening, thus lengthening the lifespan of the cell indefinitely. Telomerase has been found to be present in more than 85% of human cancer cells but is absent in most normal human tissues.145,168,202
Recurrent or persistent inflammation may induce, promote, or influence susceptibility to carcinogenesis by causing DNA damage, inciting tissue reparative proliferation, and/or creating an environment (soil) rich with cytokines and growth factors. Chronic inflammation and the metabolic products of phagocytosis are often accompanied by the excessive formation of reactive oxygen and nitrogen that are potentially damaging to DNA, lipoproteins, and cell membranes.176
Although much remains to be learned about the cascade of genetic changes for every kind of cancer, increasing understanding may suggest a means for interrupting the genetic events leading to cancer and for diagnosing the early stages of tumorigenesis. Current research continues to focus on the following major areas of biologic study53:
• Regulation of cellular proliferation and expression of oncogenes and tumor suppressor genes
• Telomere length and telomerase
• Free radical–induced DNA damage (see Fig. 5-2) and regulation of apoptosis
• Immune function and response (e.g., senescence, surveillance, enhancement)
• Cellular and humoral factors associated with the chronic inflammatory process
Current Theory of Oncogenesis
The study of viruses in tumors has led researchers to discover small segments of genetic DNA called oncogenes. Oncogenes, also called cancer-causing genes or protooncogenes, have the ability to transform normal cells into malignant cells, independently or incorporated with a virus. Oncogenes are thought to be the abnormal counterparts of protooncogenes, which aid in regulating biologic functions, such as cell division, in normal cells.
Oncogenes may be activated by carcinogens, at which point they alter the regulation of growth in the cell. Oncogenes force a cell to grow even when its surroundings contain none of the cues that normally provoke growth. Oncogenes are hyperactivated versions of normal cellular growth-promoting genes. By releasing strong, unrelenting growth-stimulating signals into a cell, oncogenes can drive cell growth ceaselessly.
Researchers have also discovered a group of regulatory genes, called antioncogenes and now called tumor suppressor genes, that have the opposite effect of oncogenes. When activated, tumor suppressor genes can regulate growth and inhibit carcinogenesis. Tumor suppressor genes are the “brakes” to the “stuck accelerator” of the activated oncogene (e.g., p53 or telomeres). When defects in the oncogene occur simultaneously with inactivation of growth-suppressing genes, aggressive cell proliferation takes place with the creation of certain types of tumor cells.
Tumor Biochemistry and Pathogenesis
Carcinogenesis is the process by which a normal cell undergoes malignant transformation. Usually, it is a multistep process, involving progressive changes after genetic damage to or alteration of cellular DNA through the development of hyperplasia, metaplasia, dysplasia, carcinoma in situ, invasive carcinoma, and metastatic carcinoma in that order.123 These discrete stages in tumor development suggest that a single altered gene only suffices to push a cell part of the way down the path to actual malignancy. The process is completed when multiple, successive changes occur in distinct cellular genes, including activation or overexpression of oncogenes and loss or mutation of tumor suppressor genes.
The number of genetic events required for conversion of normal cells to malignant cells is still debated, but, at least in the case of many solid tumors (e.g., colon carcinomas), this number may be as great as seven or eight. This high number of genetic events may imply that genetic instability occurs during cancer progression.123 This requirement for multiple changes creates an important protective mechanism against cancer. If a small number of genetic changes sufficed to transform a normal cell into a malignant one, multiple tumors would develop easily. These multiple barriers, along with the normal circuitry inside cells, ensure that only the rare cell will sustain the requisite number of changes for making a cancer cell. On the other hand, cancer has developed multiple methods centering on genetic mutation to promote self-survival and perpetuation. The pliability of cancer cells to mutate in several different phenotypes in an attempt to find one that will survive and colonize at a metastatic site makes finding effective treatment difficult at best.
INVASION AND METASTASES
Malignant tumors differ from benign tumors in their ability to metastasize or spread from the primary site to other locations in the body. Metastasis occurs when cells break away from the primary tumor, travel through the body via the blood or lymphatic system, and become trapped in the capillaries of organs. From there, they infiltrate the organ tissue and grow into new tumor deposits. Cancer can also spread to adjacent structures and penetrate body cavities by direct extension. For example, ovarian tumors frequently shed cells into the peritoneal cavity where they grow to cover the surface of abdominal organs and cause ascites.
Patterns of metastasis differ from cancer to cancer. Although there is no clear explanation of the exact mechanism of metastasis, certain cancers tend to spread to specific organs or sites in the body in a predictable manner (Table 9-2). The five most common sites of metastasis are the lymph nodes, liver, lung, bone, and brain. The spread of cancer may be influenced by a variety of host factors such as the aging or dysfunctional immune system, increasing age, hormonal environment, pregnancy, and stress. Factors that may slow the spread of metastasis include radiation, chemotherapy, anticoagulants, steroids, and other antiinflammatory agents.
Table 9-2
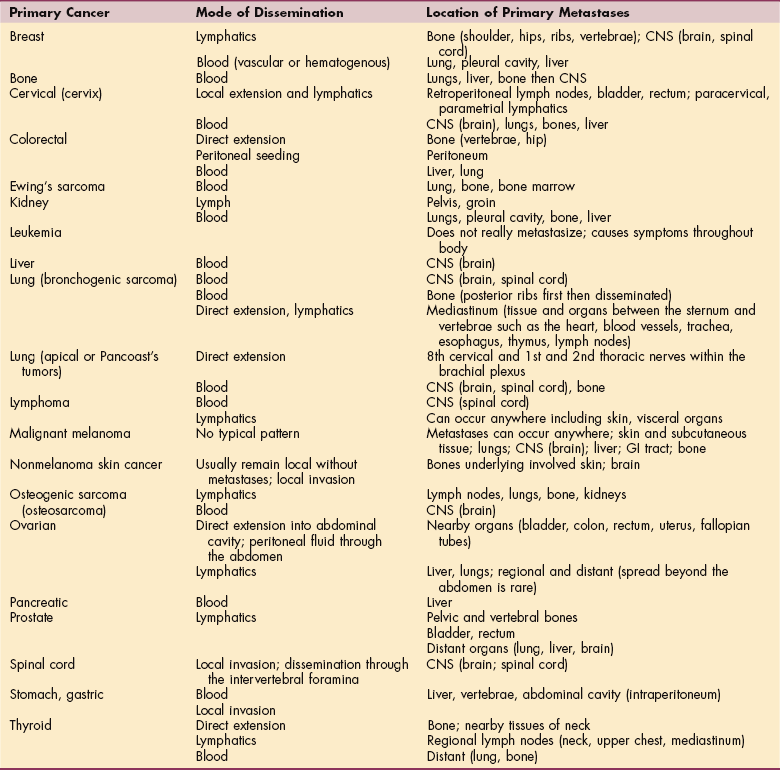
Adapted from Goodman CC, Snyder TE: Differential diagnosis for physical therapists: screening for referral, ed 4, Philadelphia, 2007, WB Saunders. CNS, Central nervous system; GI, gastrointestinal.
Seed Versus Soil Theory of Metastasis
Some cancers favor certain sites of metastasis over others so that metastases only occur if the cancer cell (the seed) finds a favorable microenvironment at the site of the host (the soil). Certain tumor cells seem to have specific affinity for certain organs. The idea that metastasis is organ specific was first proposed in 1889 by Stephen Paget, an English surgeon who first published the seed versus soil hypothesis to explain the pattern of metastasis.56
Studies in the 1990s showed that there is “cross-talk” between metastatic cells and the organ microenvironment. Host cells secrete growth factors that prompt tumor cell replication and allow the tumor to take over the homeostatic mechanisms of the host. Angiogenesis, the process by which blood vessels from preexisting vessels grow into the solid tumor, is one way that tumor cells take over homeostatic mechanism for their own gain.56
Traditional cancer treatment targets the seed, whereas today’s research is focused on approaches that target the soil, making the sites of metastasis unsuitable for the growth of cancer cells. There are many challenges in preventing metastasis because the microenvironments of metastasis sites can be very different. For example, lung cancer that spreads to the femur can behave very differently from lung cancer that spreads to the spine. Treatment that is optimal in the primary organ may not work in the metastatic sites.140
Animal studies show that the surgical removal of a primary tumor can result in the rapid growth of previously dormant metastatic cells. Additional challenges to preventing metastases are possible if this phenomenon occurs in humans.22
Incidence of Metastasis
Approximately 30% of clients with newly diagnosed cancers have clinically detectable metastases. At least 30% to 40% of the remaining clients who are clinically free of metastases harbor occult (hidden) metastases. Unfortunately, most people have multiple sites of metastatic disease, not all of which present at any one time. The formation of metastatic colonies is a continuous process, commencing early in the growth of the primary tumor and increasing with time.
Even metastases have the potential to metastasize; the presence of large, identifiable metastases in a given organ can be accompanied by a greater number of micrometastases that have been disseminated more recently from the primary tumor or the metastasis. The size variation in metastases and the dispersed anatomic location of metastases can make complete surgical removal of disease impossible, limiting the effective concentration of anticancer drugs that can only be delivered to tumor cells in metastatic colonies.
Mechanisms of Metastasis
For rapidly growing tumors, millions of tumor cells are shed into the vascular system each day. Only a very small percentage of circulating tumor cells initiate metastatic colonies because most cells that have invaded the bloodstream are quickly eliminated. Classic isotope studies have shown that 99% of circulating potentially tumorigenic cells are killed by blood vessel turbulence within 24 hours.56,57,69 Metastases of the remaining 1% require a good deal of coordination between the cancer cells and the body (Fig. 9-2).
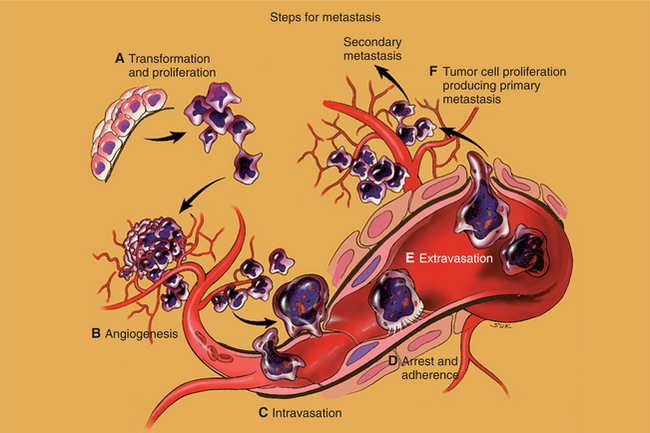
Figure 9-2 Major mechanisms of metastases. To metastasize, tumor cells must gain several unique biologic properties such as invasive growth (A), induction of vascular growth (B), vascular invasion (C), adherence to endothelial cells or thrombosis of peripheral sinusoids (D), continuation of invasive growth with extravasation (E), and formation of primary and secondary metastatic foci (F). Not all tumor cells develop all the abilities shown here; some cell clones may subspecialize and just create angiogenesis; others may invade and move on. (From Dorfman HD, Czerniak B: Bone tumors, St. Louis, 1998, Mosby.)
The greater the number of invasive tumor cells in the bloodstream, the greater the probability that some cells will survive to form metastases. Metastasis is more likely to occur via the veins as opposed to the arteries because the cancer cannot break through the arterial wall. The major challenge in treating cancer is not eradicating the primary tumor because surgery or radiation is effective in these early cases. Eradicating metastases, often already present at the time of diagnosis is the key factor to cancer cure.
A complicated series of tumor-host interactions resulting in a metastatic colony is called the metastatic cascade and is similar for all tumor cells. Once a primary tumor is initiated and starts to move by local invasion, then blood vessels from preexisting vessels grow into the solid tumor, a process called tumor angiogenesis (see Fig. 9-2). As a normal physiologic process, angiogenesis is crucial to tissue growth, repair, and maintenance.
The ability of a tumor to grow beyond a very small mass (1 to 2 mm) depends on its ability to gain access to an adequate supply of blood and in some cases (e.g., breast and prostate) the presence of hormonal factors. The supply of blood allows the tumor to obtain essential nutrients, such as oxygen, and to eliminate metabolic waste products, such as carbon dioxide and acids. The blood supply to tumors is provided by growth of new capillaries and larger vessels into the tumor mass from the blood supply of adjacent normal tissues.
The normal process of angiogenesis begins with the formation of endothelial cell sprouts followed by proliferation and migration of neighboring endothelial cells along the preformed extensions. The initiating event and mechanism of sprouting are unknown; in apoptotic cells, endothelial cell sprouting occurs via electrostatic signaling. Negatively charged membrane surfaces in apoptotic cells initiate the formation of directional endothelial cell sprouts that extend toward the dying cells.207
Tumor-derived proteins called angiogenesis factors also facilitate the use of nearby blood vessels from normal tissues and promote the growth of new blood vessels into the malignant tissue.164 The actual factors involved in tumor angiogenesis are very complex, but two cytokines have been identified as primary stimulators of vascular proliferation.
Both vascular endothelial growth factor (VEGF) and fibroblast growth factor stimulate proliferation of vascular cells and even allow the newly formed blood vessels to be easily invaded by the cancer cells that are closely adjacent to them.51 Increased tumor contact with the circulatory system provides tumors with a mechanism to enter the general circulation and colonize at distant sites. Antiangiogenic therapy shows promise as a strategy for cancer treatment (see the section on Treatment in this chapter).
Tumors generally lack a well-formed lymphatic network, so communication of tumor cells with lymphatic channels occurs only at the tumor periphery and not within the tumor mass. Lymphatic dissemination and hematogenous dissemination occur in parallel. Tumors excrete acidlike enzymes that dissolve the basement membrane and break through to the lymphatics. Cancer cells can enter the bloodstream where lymph nodes drain into veins (e.g., lymphatic intersection with the subclavian vein).
Clinical Manifestations of Metastasis
Metastatic spread usually occurs within 3 to 5 years after initial diagnosis and treatment of malignancy, although some low-grade lesions can reappear as much as 15 to 20 years later. It is therefore very important to conduct a thorough past medical history as part of any client interview. Metastases occur most commonly to areas of the body that provide an environment rich in nutrition to the colonized tumor cells, such as the lung, brain, liver, and bone; metastases can be found in other areas as well (e.g., the lymph nodes, skin, ovaries, and adrenal glands).
Pulmonary System (Lungs)
Pulmonary metastases are the most common of all metastatic tumors because venous drainage of most areas of the body is through the superior and inferior venae cavae into the heart, making the lungs the first organ to filter malignant cells. Parenchymal metastases are asymptomatic until tumor cells have obstructed bronchi, resulting in pulmonary symptoms, or until tumor cells have expanded and reached the parietal pleura where pain fibers are stimulated.
A dry, persistent cough is often the first symptom of pulmonary metastases. Pleural pain can indicate pleural invasion, and shortness of breath (dyspnea) usually occurs in the presence of a malignant pleural effusion. If hemoptysis occurs, there is usually bronchial tissue invasion either by a primary lung malignancy or metastatic disease.199
Hepatic System (Liver)
Liver metastases are among the most ominous signs of advanced cancer. The liver filters blood coming in from the gastrointestinal tract, making it a primary metastatic site for tumors of the stomach, colorectum, and pancreas. Symptoms include abdominal and/or right upper quadrant pain, general malaise and fatigue, anorexia, early satiety and weight loss, and sometimes low-grade fevers.
Skeletal System (Bone)
Primary bone tumors, such as osteogenic sarcoma, metastasize initially to the lungs, whereas a large proportion of cancer cases metastasize first to the bone, often with a poor prognosis (see also Chapter 26). Bone is one of the three most favored sites of solid tumor metastasis, indicating that the bone microenvironment provides fertile ground for the growth of many tumors. Although lung, breast, and prostate are the three primary sites responsible for most metastatic bone disease, tumors of the thyroid and kidney, lymphoma, and melanoma can also metastasize to the skeletal system.
Bone metastases may be the osteolytic type, marked by areas of decreased bone density, or osteoblastic, appearing as areas of dense scarring and increased bone density. Osteolytic metastases predominate in lung, kidney, and thyroid cancer; breast is primarily osteolytic but can be osteoblastic and prostate is usually but not always osteoblastic. The axial skeleton is most commonly involved with spread to the spine, pelvis, ribs, proximal femora, proximal humeri, and skull.125
The primary symptom associated with bone metastases is pain. Pain is usually deep and worsened by activity, especially weight bearing. Disabling pathologic fractures, especially of the vertebral bodies and proximal ends of the long bones, may occur in up to one-half of people with osteolytic metastases and are sometimes one of the first signs of a malignant process.73 Blastic lesions often result in an elevated serum alkaline phosphatase, whereas lytic lesions may not.118
Hypercalcemia (abnormally high concentration of blood calcium) is a frequent complication of neoplastic disease and is associated with bony metastases, particularly osteolytic lesions as a result of increased bone resorption. The presence of tumor cells in the bone disturbs the balance between new bone formation and bone resorption, resulting in abnormal bone remodeling.
Carcinoma cells secrete a variety of factors that stimulate tumor growth, as well as osteoclast recruitment and activation. Osteoclasts are bone cells that break down bone tissue. Although more than 80% of people with hypercalcemia have bony metastasis, the severity of the hypercalcemia does not correlate with the extent of the bony disease.
Hydration and administration of oral or intravenous bisphosphonates (e.g., alendronate [Fosamax], risedronate [Actonel], ibandronate [Boniva], zoledronate [Zometa]) are the mainstays of current treatment and have contributed to the decrease in the frequency of hypercalcemia and to minimizing or preventing bone loss. Bisphosphonates prevent thinning of the bone by inhibiting osteoclast function, thereby inhibiting bone resorption. There is no life-prolonging benefit with bisphosphonates, but there is a substantial reduction of morbidity (e.g., decreased pain, prevention of fracture and/or deformity) associated with bone metastases and improved quality of life (QOL).17,32,40,85
Central Nervous System
Brain.: Many primary tumors may lead to CNS metastases. Lung carcinomas account for approximately one-half of all metastatic brain lesions. Breast carcinoma and malignant melanoma also commonly metastasize to the brain. Metastatic disease in the brain is life-threatening and emotionally debilitating. Metastatic brain tumors can increase intracranial pressure, obstruct the normal flow of cerebrospinal fluid, change mentation and contribute to cognitive impairment, and reduce sensory and motor function. The management of cognitive impairments is important, and the therapist can use the same strategies known for people with traumatic brain injury (see Chapter 33).
Clinical manifestations of brain metastases depend on the location, either in the brain or outside the brain in the bony cranium exerting compression externally. The therapist can look at CT scan results, see the location of pathologic conditions, and correlate these to signs and symptoms observed clinically (see Table 30-3). Primary tumors of the CNS rarely develop metastases outside the CNS despite the highly invasive capacity of these tumors. Microscopically, some CNS (brain) tumor cells (astrocytomas) may spread widely within the CNS but rarely metastasize outside it.
Tumor cells traveling from the lung via the pulmonary veins and carotid artery can result in metastases to the CNS. Lung cancer is the most common primary tumor to metastasize to the brain. Any neurologic sign may be the presentation of a silent lung tumor.73
Spinal Cord.: Metastatic involvement of the vertebrae may result in epidural spinal (usually anterior) cord compression. In addition, severe, destructive osteolytic lesions can lead to fracture and fragility of one or more vertebral bodies. In such cases, compression of the cord occurs as a result of the subsequent deformity.124 Spinal cord and nerve root compression cause either insidious or rapid loss of neurologic function. This compression phenomenon occurs in approximately 5% of people with systemic cancer and is most often caused by carcinoma of the lung, breast, prostate, or kidney. Lymphoma and multiple myeloma may also result in spinal cord and nerve root compression.
The earliest neurologic symptoms include gradual onset of distal weakness and sensory changes, including numbness, paresthesias, and coldness. The incidence of permanent motor dysfunction has markedly decreased in the past 2 decades because of earlier diagnosis and treatment.40 The client who presents with spinal cord symptoms caused by metastatic epidural disease and resultant compression may have only transient symptoms with proper medical treatment. More than 95% of people with spinal cord compression complain of progressive central or radicular back pain often aggravated by recumbency, weight bearing, sneezing, coughing, or Valsalva’s maneuver. Sitting often relieves it.
Lymphatic System
Cancer-related surgery or radiation treatment affecting the lymph nodes may result in dysfunction of the lymphatic system presenting as lymphedema. It has a wide range of onset from weeks to years from the initial insult to the lymphatic system. The therapist may be the first health care professional to assess for abnormalities or changes in the extremities. See further discussion in Chapter 13.
Diagnosis of Metastasis
Metastases usually reproduce the cellular structure of the primary growth well enough to enable a pathologist to determine the site of the primary tumor. For example, bone metastases from a carcinoma of the thyroid not only exhibit a microscopic structure similar to the original tumor but also may produce thyroid hormone. Sometimes, symptoms of a cancer will present in the metastatic site rather than the site of origin (primary site). If the primary tumor cannot be found, the malignant tissue is called carcinoma of unknown primary. Special histologic stains can be done on the unknown tissue and compared to slides of a previous malignancy to determine similarity.
Cancer Recurrence
Disease-free survival describes the time between diagnosis and recurrence or relapse. Recurrences may be local, regional, disseminated, or a combination of these. The most important predictors of recurrent cancer are the stage at the time of initial therapy and histologic findings. Recurrence of cancer may be first recognized by the return of systemic symptoms associated with paraneoplastic phenomena. The metabolic or toxic effects of the syndrome (e.g., hypercalcemia or hyponatremia) may constitute a more urgent hazard to life than the underlying cancer.
CLINICAL MANIFESTATIONS
Most cancers in their earliest stages are asymptomatic but treatable if found. Most primary site cancers cause certain symptoms that are recognizable causes for suspicion or concern. For example, endometrial cancer causes abnormal bleeding so often that it is usually detected in its early stages. Laryngeal cancer causes hoarseness, which is also an early sign. However, lung cancer is usually quite extensive before it causes enough symptoms to warrant investigation, as is true with breast cancer if it is a deeply buried tumor that is difficult to palpate. Most cancer is detected early and can be cured or successfully treated.
As the cancer progresses, symptoms characteristic of the involved organ or tissue may start to develop. With advanced cancer, nausea, vomiting, and retching (NVR) accompanied by anorexia and subsequent weight loss are common as a result of the malignant process and its treatment. NVR is especially prevalent in association with lung carcinoma, hypernephroma, and pancreatic carcinoma.
Anorexia has been attributed to tumor production of TNF, which is a protein (a cytokine) also called cachectin. TNFs are believed to play an important role in mediating inflammation and cytotoxic reactions (along with interleukins). TNFs produce necrosis of tumor cells by eliminating the blood supply to these growths. Small amounts of TNF are beneficial in promoting wound healing and preventing tumors, but uncontrolled production is accompanied by symptoms of fever, weight loss, and tissue damage that can cause more problems than the benefits provided.
Cancer-related anorexia/cachexia (CAC) is a complex phenomenon in which metabolic abnormalities, proinflammatory cytokines produced by the host immune system, circulating tumor-derived catabolic factors, decreased food intake, and possibly other unknown factors all contribute. Profound muscle loss is prominent in CAC syndrome as a result of decreased protein synthesis and abnormal muscle proteolysis.
Later, the rapid growth of the tumor encroaches on healthy tissue, causing destruction, necrosis, ulceration, and hemorrhage and producing many local and systemic effects. Pain may occur as a late symptom caused by infiltration, compression, or destruction of nerves. With advanced or stage IV cancer, the host presents systemically with muscular weakness, anemia, and coagulation disorders such as granulocyte and platelet abnormalities. See the section on Disorders of Hemostasis and Thrombocytopenia in Chapter 14.
Fever may be seen with cancer in the absence of infection and is produced either by white blood cells inducing a pyrogen (an agent that causes fever) or by direct tumor production of a pyrogen. Continued spread of the cancer may lead to gastrointestinal, pulmonary, or vascular obstruction. Secondary infections frequently occur as a result of the host’s decreased immunity and can lead to death.
Other vital organs may be affected, such as the brain, in which increased intracranial pressure by tumor cells can cause strokelike symptoms. In addition to the local effects of tumor growth, cancer can produce systemic signs and symptoms that are not direct effects of either the tumor or its metastases (e.g., paraneoplastic syndromes or fever with renal cancer).
Cancer Pain
One of the most common symptoms of cancer is pain, affecting between 50% and 70% of clients in its early stages and 60% to 90% of clients in late stages of the disease. It is estimated that 1.1 million Americans experience cancer-related pain annually.29 Alternately stated, pain occurs in approximately one-quarter of adults with newly diagnosed malignancies, one-third of individuals undergoing treatment, and three-quarters of all people with advanced disease.71,195
Depression and anxiety may increase the person’s perception of pain or may be the result of the cancer pain. Symptoms often go unreported or underreported because clients are reluctant to take the pain medication prescribed. An unfounded fear of tolerance, addiction, or adverse effects from pain medication may result in underreporting of painful symptoms with subsequent inadequate cancer pain control and unnecessary pain-induced loss of function. Likewise, physicians may hesitate to provide adequate pain medications based on this misconception of client addiction.
Etiology and Pathogenesis
The cause of cancer pain is multifaceted, and the characteristics of the pain depend on the tissue structure, as well as on the mechanisms involved (Table 9-3). Some pain is caused by pressure on nerves or by the displacement of nerves. Microscopic infiltration of nerves by tumor cells can result in continuous, sharp, stabbing pain generally following the pattern of nerve distribution. Ischemic pain (throbbing) may also result from interference with blood supply or from blockage within hollow organs.
Table 9-3
Common Patterns of Pain Referral
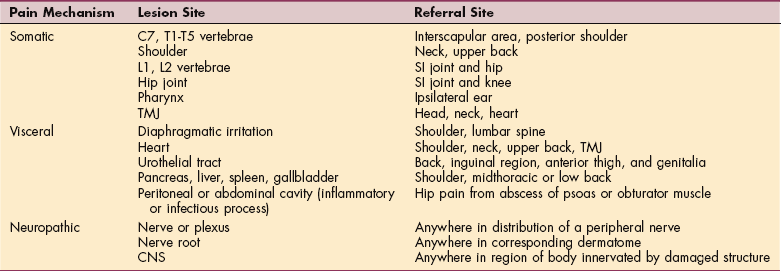
From Goodman CC, Snyder TE: Differential diagnosis for the physical therapist: screening for referral, ed 4, Philadelphia, 2007, WB Saunders. SI, Sacroiliac; TMJ, temporomandibular joint; CNS, central nervous system.
A common cause of cancer pain is metastasis of cancer to bone. Lung, breast, prostate, thyroid, and the lymphatics are the primary sites responsible for most metastatic bone disease. Bone metastasis results in increased release of prostaglandins and cytokines and subsequent bone destruction caused by breakdown and resorption. Bone pain may be mild to intense. Movement, weight bearing, and ambulation exacerbate painful symptoms from bone destruction. Pathologic fractures with resultant muscle spasms can develop; in the case of vertebral involvement, nerve pain may also occur.
Pain may also result from diagnostic or therapeutic procedures such as surgery, radiation therapy, or chemotherapy (e.g., mucositis, stomatitis, esophageal inflammation, localized skin burns; see Table 9-8).
Clinical Manifestations
Signs and symptoms accompanying mild-to-moderate superficial pain may include hypertension, tachycardia, and tachypnea (rapid, shallow breathing) as the result of a sympathetic nervous system response. In severe or visceral pain, a parasympathetic nervous system response is more characteristic, with hypotension, bradycardia, nausea, vomiting, tachypnea, weakness, or fainting.
Spinal cord compression from metastases may present as radicular back pain, leg weakness, and unilateral loss of bowel or bladder control. Back pain may precede the development of neurologic signs and symptoms. The presence of jaundice in association with an atypical presentation of back pain may indicate hepatobiliary obstruction and/or liver metastasis. Signs of nerve root compression may be the first indication of a cancer, in particular lymphoma, multiple myeloma, or cancer of the lung, breast, prostate, or kidney. Other neurologic or musculoskeletal manifestations of neoplasm are discussed in the section on Paraneoplastic Syndromes in this chapter.
Immobility and inflammation can lead to pain. Inflammation with its accompanying symptoms of redness, edema, pain, heat, and loss of function may progress to infection, necrosis, and sloughing of tissue. If the inflammatory process alone is present, the pain is characterized by tenderness. Pain may be excruciating in the presence of tissue necrosis and sloughing.
Pain Control
Pain management and control may depend on its underlying etiology. For example, epidural metastases with impending spinal cord compression require treatment with steroids, radiation, chemotherapy, or neurosurgery. Abdominal pain caused by obstruction of the hollow organs requires evaluation for surgical intervention.41
Treatment approaches depend on whether the individual is experiencing acute or chronic pain. The hope is to begin by gaining control of the pain during the acute phase and then to sustain that pain relief while minimizing side effects.
Pain should be screened, assessed, and managed according to clinical practice guidelines (CPGs). CPGs for the treatment of cancer-related pain in adults outlining the process of screening, evaluation, and intervention have been published by the National Comprehensive Cancer Network (NCCN).131 A similar clinical practice guideline is available from the NCCN for pediatric cancer pain.135
Before starting therapy, the physician determines the underlying pain mechanism and diagnoses the pain syndrome. Pain control measures used include narcotic and nonnarcotic analgesics; chemotherapy or radiation therapy or both; surgery; nerve blocks; or other more invasive pain control measures, such as intraspinal opioids, rhizotomy, or cordotomy. Newer drugs, such as the bisphosphonates, have been useful in the treatment of refractory bone pain.29
Appropriate opioid selection may be difficult and depends on the individual’s pain intensity and any current analgesic therapy. Morphine, hydromorphone, fentanyl, and oxycodone are the opioids commonly used in the United States. A balance between analgesia and side effects might be achieved by changing to an equivalent dose of an alternative opioid. This approach, known as opioid rotation, is now a widely accepted technique used to address poorly responsive pain.131
Several methods of continuous infusion that are widely used in clinical practice include “around the clock,” “as needed,” and “patient-controlled analgesia” (PCA). Around the clock dosing is provided to chronic pain patients for continuous pain relief. A “rescue dose” should be provided as a subsequent treatment for patients receiving these controlled-release medications. Rescue doses of short-acting opioids should be provided for pain that is not relieved by sustained/controlled release opioids.
Opioids administered on an as needed basis are for patients who have intermittent pain with pain-free intervals. The as needed method is also used when rapid dose escalation is required.
The PCA technique allows a person to control a device that delivers a bolus of analgesic “on demand” (according to and limited by parameters set by a physician). This system permits the person to self-administer a premeasured dose of analgesic by pressing a button that activates a pump syringe containing the analgesic. Small intermittent doses of the analgesic administered via intravenous (IV) line maintain blood levels that ensure comfort and minimize the risk of oversedation. Clinical studies report that people using PCA effectively maintain comfort without oversedation and use less drug than the amount normally given by intramuscular (IM) injection.
Nonpharmacologic modalities, such as massage, acupuncture, imagery/hypnosis, reflexology, relaxation training, and other forms of complementary therapies, are based on client preference and clinical judgment of what is best when practicing integrative medicine or health care. Complementary therapies can lessen procedural pain and distress even among children, especially when fear, anxiety, and tension heighten pain perception.6,41,114
Whereas severe cancer pain is treated pharmaceutically, mild-to-moderate joint and muscle pain can be addressed by the rehabilitation professional. Pain elimination through the use of medication may not be possible without accompanying severe loss of function, which is an undesirable outcome.
Noninvasive physical agents, such as cryotherapy, thermotherapy, electrical stimulation, immobilization, exercise, massage, biofeedback, and relaxation techniques, may be effective in pain management. Much debate exists about the safety and efficacy of massage therapy for individuals with cancer, especially anyone with lymphedema or at risk for developing lymphedema.34 A review of data included in the Cochrane Database of Systematic Reviews suggests that conventional care for people with cancer can safely incorporate massage therapy, although individuals with cancer may be at higher risk for adverse events. There is no evidence that massage therapy can spread cancer, although direct pressure over a tumor is usually discouraged.34
The strongest evidence for the benefits of massage is stress and anxiety reduction. Research regarding the use of massage for pain control and management of other symptoms is promising. Massage therapists may advocate the use of massage to reduce constipation, improve immune system function, help promote postoperative wound healing, and reduce scar tissue formation, as well as to help release metabolic waste by improving circulation. Published trials for these indications have not been reported.34
Modifications to massage (e.g., lighten pressure or avoid deep tissue massage) may be necessary to prevent potential harm such as bleeding, fracture, or increased pain when individuals with cancer receiving massage have a coagulation disorder (low platelet count or when receiving warfarin/heparin/aspirin therapy). Similar precautions are required for anyone with cancer metastases to the bones. Massage should be avoided over open or healing wounds or radiation dermatitis.34
People with cancer may also experience pain because of nerve damage. This damage can be caused directly by tumor invasion or indirectly as a side effect of cytotoxic drug therapy (e.g., taxanes or platinum agents). The treatment of neuropathic pain remains a dilemma since conventional analgesic drugs do not always provide relief.
Recommended treatment for neuropathic pain includes infrared (Anodyne), antidepressant drugs (e.g., amitriptyline); antiepileptics (e.g., carbamazepine and gabapentin); and steroids (e.g., methylprednisolone and dexamethasone). Pain relief is usually not immediate, and the drugs used must be taken continuously, thus side effects, such as sedation and bone marrow depression, can be additional problems.29,84
Management of pain in people with cancer who live in long-term care facilities remains an ongoing concern. Consistent, daily pain is prevalent among nursing home residents with cancer and is frequently untreated, particularly among older and minority clients.16 For individuals with difficult-to-control chronic pain, complementary therapies can help even if it is only to reduce the level of analgesics required to maintain pain control.
Cancer-Related Fatigue
Much has been written about cancer-related fatigue (CRF) and its impact on clients. CRF is a distressing, persistent, and subjective sense of tiredness or exhaustion related to cancer or cancer treatment that is not proportional to recent activity and interferes with usual functioning.133 CRF syndrome is a collection of symptoms with multiple characteristics and problems. Fatigue is a nearly universal symptom in all people receiving chemotherapy, radiotherapy, and treatment with biologic response modifiers; reduced physical performance and fatigue are universal after bone marrow transplantation.
Up to 30% of cancer survivors report a loss of energy for years after cessation of treatment. For many people with cancer, fatigue is severe and imposes limitations on normal daily activities.25 Many people’s perceptions are that fatigue is more distressing than pain or nausea and vomiting, which can be managed with medication for most clients.
Fatigue should be screened, assessed, and managed according to CPGs, which have been published for CRF.133 All individuals should be screened for fatigue at their initial visit, at regular intervals during and after cancer treatment, and as clinically indicated. Fatigue should be recognized, evaluated, monitored, documented, and treated promptly for all age groups, at all stages of disease, during and after treatment. Clients and their families should be informed that management of fatigue is an integral part of total health care.133
Using a numeric rating scale, fatigue can be rated as mild (1 to 3), moderate (4 to 6), or severe (7 to 10).158,179 Children can be asked if they are “tired” or “not tired.” Fatigue that causes distress or interferes with daily activities or functioning should be treated according to its severity and the presence of other treatable factors known to contribute to fatigue (e.g., pain, emotional distress, sleep disturbance, anemia, nutritional deficits, deconditioning, and comorbidities).
Clients should be reassured that treatment-related fatigue is not necessarily an indicator of disease progression. It may be the result of anemia, deconditioning, or the presence of certain cytokines (e.g., interleukin [IL]-1, IL-6, or TNF-α). There may be contributing psychosocial factors such as anxiety, depression, and disrupted sleep pattern.
Despite the prevalence of CRF, the exact mechanisms involved in its pathophysiology are unknown. Abnormal accumulation of muscle metabolites, production of cytokines, changes in neuromuscular function, abnormalities in adenosine triphosphate (ATP) synthesis, serotonin dysregulation, and vagal afferent activation are just a few more of the proposed theories. Comorbidities, such as cardiopulmonary impairments, may put an individual at greater risk for fatigue.133
Likewise, the cause of fatigue in posttreatment disease-free individuals is unclear and likely multifactorial. Higher serum markers, such as interleukin-1 receptor antagonist (IL-1ra) and soluble TNF type II (sTNF-RII), and lower cortisol levels have been observed in fatigued cancer survivors compared with nonfatigued survivors. Significantly higher levels of circulating T lymphocytes have also been observed in fatigued survivors. These findings together point to a chronic inflammatory process involving T cells as a possible fatigue-inducing mechanism.133
Paraneoplastic Syndromes
In addition to the local effects of tumor growth, cancer can produce systemic signs and symptoms that are not direct effects of either the tumor or its metastases. When tumors produce signs and symptoms at a site distant from the tumor or its metastasized sites, these remote effects of malignancy are collectively referred to as paraneoplastic syndromes.
Although malignant cells frequently lose the function, appearance, and properties associated with the normal cells of the tissue of origin, they can acquire in some cases new cellular functions uncharacteristic of the originating tissue. Many of these syndromes involve ectopic hormone production by tumor cells or the secretion of biochemically active substances that cause metabolic abnormalities. For example, tumors in nonendocrine tissues sometimes acquire the ability to produce and secrete hormones that are distributed by the circulation and act on target organs at a site other than the location of the tumor. The most common cancer associated with paraneoplastic syndromes is small cell cancer of the lung, which can produce adrenocorticotropic hormone (ACTH) in amounts sufficient to cause Cushing’s syndrome.
Malignancy is often associated with a wide variety of musculoskeletal disorders, which may be the presenting symptoms of an occult tumor. Although musculoskeletal symptoms often result from direct invasion by the malignancy or its metastases into bone, joints, or soft tissue, they may also occur without invasion as a result of the paraneoplastic disorders, including well-recognized syndromes, as well as less well-defined disorders referred to as cancer arthritis.193
Incidence
Previously, paraneoplastic syndromes occurred in 10% to 20% of all cancer clients, but this figure may be increasing because of greater physician awareness and the availability of serodiagnostic tests for some syndromes. Neurologic paraneoplastic syndromes are more rare, occurring in 1% to 2% of people with malignancy.
Etiology and Pathogenesis
The causes of paraneoplastic syndromes are not well understood, but the following four groups of mechanisms have been identified:
• Effects initiated by a variety of vasoactive tumor products (e.g., serotonin, histamine, catecholamines, prostaglandins, and vasoactive peptides), which usually occur in the small bowel and less commonly in the lung or stomach.
• Effects caused by the destruction of normal tissues by tumor, such as occurs when osteolytic skeletal metastases cause hypercalcemia.
• Effects caused by unknown mechanisms, such as unidentified tumor products or circulating immune complexes stimulated by the tumor (e.g., osteoarthropathy as a result of bronchogenic carcinoma).
• Effects caused by autoantibodies (antibodies directed against the host’s tissue), for example, cancer cells produce antibodies that impair presynaptic calcium channel activity, hindering the release of the neurotransmitter acetylcholine, resulting in muscle weakness.
There is increasing evidence that many of the neurologic paraneoplastic syndromes appear to be an immune reaction against antigens shared by the cancer and the nervous system. The immune response is triggered by and directed against the tumor, which then cross-reacts with protein expressed by the peripheral or CNS. Consequently, any part of the nervous system can be affected.61,113
Clinical Manifestations
The paraneoplastic syndromes are of considerable clinical importance because they may accompany relatively limited neoplastic growth and provide an early clue to the presence of certain types of cancer. Nonspecific symptoms, such as neurologic changes, anorexia, malaise, diarrhea, weight loss, and fever, may be the first clinical manifestations of a paraneoplastic syndrome. Even these types of nonspecific symptoms occur as a result of the production of specific biochemical products by the tumor itself.
Paraneoplastic syndromes with musculoskeletal manifestations are listed in Table 9-4. Gradual, progressive muscle weakness may develop over a period of weeks to months. The proximal muscles (especially of the pelvic girdle) are most likely to be involved; the weakness does stabilize. Reflexes of the involved extremities are present but diminished. Proximal leg weakness (Lambert-Eaton myasthenic syndrome) is most often associated with small cell carcinoma of the lung.
Table 9-4
Paraneoplastic Syndromes: Rheumatologic Associations and Clinical Features
| Malignancy | Rheumatologic Associations | Clinical Features |
| Lung cancer | Myasthenic syndrome | Proximal leg weakness |
| Lymphoproliferative disease (leukemia) | Vasculitis | Necrotizing vasculitis |
| Plasma cell dyscrasia | Cryoglobulinemia | Vasculitis, Raynaud’s disease, arthralgia, neurologic symptoms |
| Hodgkin’s disease | Immune complex disease | Nephrotic syndrome |
| Ovarian cancer | Complex regional pain syndrome | Palmar fasciitis and polyarthritis |
| Carcinoid syndrome | Scleroderma | Scleroderma-like changes: anterior tibia |
| Colon cancer | Pyogenic arthritis | Enteric bacteria cultured from joint |
| Mesenchymal tumors | Osteogenic osteomalacia | Bone pain, stress fractures |
| Renal cell cancer (and other tumors) | Severe Raynaud’s phenomenon | Digital necrosis |
| Pancreatic cancer | Panniculitis | Subcutaneous nodules, especially males |
| Lung cancer, small cell | Eaton-Lambert syndrome |
Muscular and cutaneous disorders associated with malignancy are presented in Table 9-5. Myositis may precede, follow, or arise concurrently with the malignancy and tends to occur most often in older people. Cutaneous paraneoplastic syndromes are a large group of dermatoses that may be associated with an internal malignancy.
Table 9-5
Muscular and Cutaneous Disorders Associated with Malignancy
| Muscular | Cutaneous |
| Amyloidosis | Acanthosis (diffuse thickening) |
| Amyotrophic lateral sclerosis | Dermatomyositis |
| Polymyositis | Extramammary Paget’s disease |
| Lambert-Eaton myasthenic syndrome (LEMS) | Nigricans (blackish discoloration; changes in skin pigmentation) |
| Myasthenia gravis | Pemphigus vulgaris (water blisters) |
| Metabolic myopathies | Pruritus (itching) |
| Primary neuropathic diseases | Pyoderma gangrenosum (eruption of skin ulcers) |
| Type II muscle atrophy | Reactive erythemas (skin redness) |
Data from Gilkeson GS, Caldwell DS: Rheumatologic associations with malignancy, J Musculoskel Med 7(1):70, 1990; Cohen PR: Cutaneous paraneoplastic syndromes, Am Fam Physician 50:1273-1282, 1994.
There may be rheumatologic symptoms referred to as carcinoma polyarthritis. This condition can be differentiated from rheumatoid arthritis by the presence of asymmetric joint involvement, involvement of primarily the lower extremity (although symmetric involvement of the hands has been reported),193 explosive onset, late age of onset, and the presence of malignancy and arthritis together. Neurologic paraneoplastic syndromes are of unknown cause and include subacute cerebellar degeneration, amyotrophic lateral sclerosis, sensory or sensorimotor peripheral neuropathy, Guillain-Barré syndrome, myasthenia gravis, and the Lambert-Eaton myasthenic syndrome (LEMS).
Isolated cases of paraneoplastic stiff-person syndrome have been reported in association with small cell lung cancer, thymoma (thymus), melanoma, and breast adenocarcinoma.95,136,159 Women are affected twice as often as men, most likely because of the association with breast cancer. Paraneoplastic stiff-person syndrome is characterized by progressive symptoms of neuropathy or myelopathy with increased muscle tone and rigidity in the spine and lower extremities, especially the ankle dorsiflexors with loss of ankle motion.127
The disease may take years to manifest but in some individuals, symptoms develop over a period of weeks. The first symptom may be a persistent progressive stiffening of the back or leg that is triggered by sudden noise, touch, fatigue, or made worse by stress such as time pressure (e.g., hurrying to cross a street). Stiffness progresses to hypertonia, spasm, and then severe, painful rigidity. Walking becomes difficult, and the individual is at increased risk for unprotected falls (i.e., they fall like a tin soldier). Symptoms are greatly relieved during sleep.
Although rare, this condition has been encountered by therapists in an oncology practice. Stiff-person syndrome is normally associated with a viral cause, such as meningitis or encephalitis, but in the case of cancer-induced stiff-person syndrome, it is chemically induced by the tumor, classifying it as a paraneoplastic syndrome.95 It is seen more often in clients with a history of diabetes and other autoimmune diseases (e.g., hyperthyroidism, hypothyroidism, or anemia).127
MEDICAL MANAGEMENT
DIAGNOSIS, TREATMENT, AND PROGNOSIS.
Serodiagnostic tests are available for some paraneoplastic syndromes, and characteristic abnormalities in magnetic resonance imaging (MRI) have been identified in association with neurologic paraneoplastic syndromes. Biochemical markers in urine provide specific monitoring of the response of bone metastases to treatment. This early diagnosis of paraneoplastic syndromes provides for prevention of tumor progression and subsequent problems such as bone pain, fracture, and hypercalcemia. In the case of cancer polyarthritis, the absence of rheumatoid nodules, absence of rheumatoid factor, and absence of family history of rheumatoid disease help in the diagnostic process.
Interestingly, paraneoplastic syndromes do not necessarily parallel the course of the disease but can be more related to the amount of antibody present rather than the amount of tumor volume. Some of the cutaneous paraneoplastic syndromes will respond to specific measures, such as systemic corticosteroid therapy, but for the most part, successful resolution requires eradication of the underlying malignancy.
Diagnosis of stiff-man syndrome is made by physical examination and immunocytochemistry methods demonstrating the presence of anti-GAD (glutamic acid decarboxylase) autoantibodies in the blood. Diagnosis may be confirmed by a positive response to medications used to treat this condition (e.g., benzodiazepines).