The Gastrointestinal System
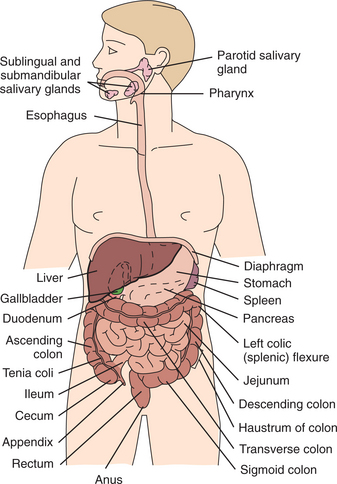
The gastrointestinal (GI) tract consists of upper and lower segments with separate functions. The upper GI tract includes the mouth, esophagus, stomach, and duodenum and aids in the ingestion and digestion of food. The lower GI tract includes the small and large intestines (Fig. 16-1). The small intestine accomplishes digestion and absorption of nutrients, whereas the large intestine absorbs water and electrolytes, storing waste products of digestion until elimination.
The enteric nervous system has become the focus of new research and discoveries in a new area of study referred to as psychoneuroimmunology with a subspecialty of clinical gastroenterology called neurogastroenterology. There are as many nerve cells in the human small intestine as there are in the human spinal cord, and the enteric nervous system can function completely independently of the central nervous system. The enteric nervous system is sometimes referred to as the “brain in the bowel.”
Insight into the connections among emotions, brain function, and GI function have revolutionized our thinking about the so-called mind-body connection. New information is being discovered about the sensory functions of the intestine and how neural, hormonal, and immune signals interact. More than 30 chemicals of different classes (neuropeptides, neurotransmitters) transmit instructions to the brain, and all these chemicals are represented in the enteric nervous system.
Representatives of all the major categories of immune cells are found in the gut or can be recruited rapidly from the circulation in response to an inflammatory stimulus. The constant presence of these neurotransmitters and neuromodulators in the bowel suggests that emotional expression or active coping generates a balance in the neuropeptide-receptor network and physiologic healing beginning in the GI system. In fact, it has been suggested that since the enteric nervous system can function on its own, it is possible that the brain in the bowel can have its own psychoneuroses such as the functional bowel syndromes discussed later in this chapter.59
Scientists continue to study influences of the nervous system on immune and inflammatory responses in the mucosal surfaces of the intestines along with the innervation of the immune system and the molecular communication pathways as these relate to emotions and thoughts and the GI system.76
The gut immune system has 70% to 80% of the body’s immune cells, and the protective blocking action of the secretory response in the gut is crucial to the integrity of the GI tract immune function and host defense. Studies suggest that the development and expression of the regional immune system of the GI tract is independent of systemic immunity. Nutrients have fundamental and regulatory influences on the immune response of the GI tract and therefore on host defense.
Reduction of normal bacteria in the gut after antibiotic treatment or in the presence of infection may interfere with the nutrients available for immune function in the GI tract. New understanding of intestinal disorders and new approaches to the management of these disorders are expected in the next decade.
SIGNS AND SYMPTOMS OF GASTROINTESTINAL DISEASE
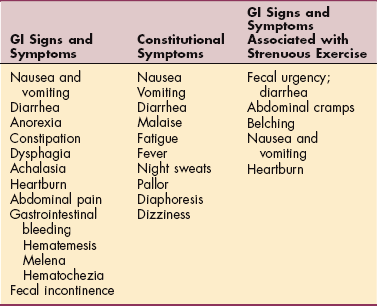
Clinical manifestations of GI disease can be caused by a variety of underlying conditions or disorders. The primary condition may be of GI origin, but some GI symptoms are part of a collection of systemic symptoms called constitutional symptoms and may be associated with any systemic condition (Table 16-1).
Nausea occurs when nerve endings in the stomach and other parts of the body are irritated and usually precedes vomiting. Intense pain in any part of the body can produce nausea as a result of the nausea-vomiting mechanism of the involuntary autonomic nervous system. Nausea can be caused by strong emotions and may accompany psychologic disorders, a variety of systemic disorders (e.g., acute myocardial infarction, diabetic acidosis, migraine, hepatobiliary and pancreatic disorders, Ménière’s syndrome, and GI disorders), and drugs such as morphine, codeine, excess alcohol, anesthetics, and anticancer drugs.
Vomiting may be caused by anything that precipitates nausea. Complications of vomiting include fluid and electrolyte imbalances, pulmonary aspiration of vomitus, gastroesophageal mucosal tear (Mallory-Weiss syndrome), malnutrition, and rupture of the esophagus.
Diarrhea (abnormal frequency or volume of stools) results in poor absorption of water and nutritive elements and electrolytes, fluid volume deficit, and acidosis as a result of bicarbonate depletion (see Chapter 5). Other systemic effects of prolonged diarrhea are dehydration, electrolyte imbalance, and weight loss. The causes of diarrhea are many and varied (Table 16-2). Drug-induced diarrhea, most commonly associated with antibiotics, may not develop until 2 to 3 weeks after first ingestion of an antibiotic, but if the onset of diarrhea coincides with the use of drugs, it may resolve when the drug is discontinued.
Table 16-2
Causes of Diarrhea and Constipation
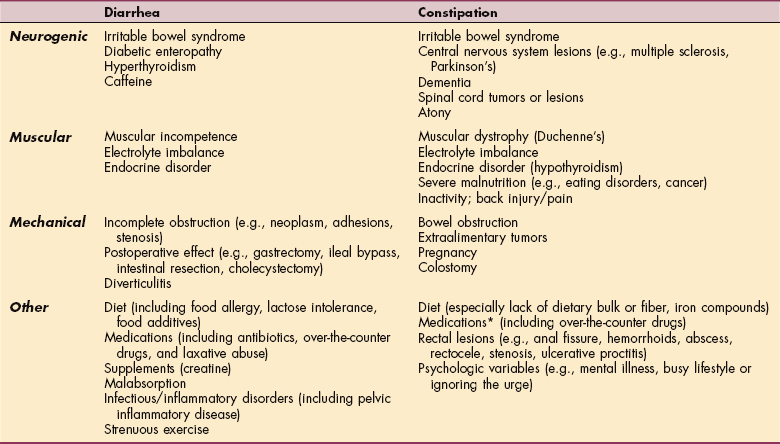
*Many drugs commonly prescribed, especially for older adults, can cause constipation, including α-adrenergic blocking agents, angiotensin-converting enzyme inhibitors, antiarrhythmics, anticholinergic antiseizure drugs, antihistamines, antilipidemics, antiparkinson drugs, antipsychotics, benzodiazepines, calcium channel blockers, nonsteroidal antiinflammatory agents, opioids, and antidepressants.
Anorexia, diminished appetite or aversion to food, is a nonspecific symptom often associated with nausea, vomiting, and sometimes diarrhea. It may be associated with disorders of other organ systems, including cancer, heart disease, and renal disease. It is the major component of eating disorders such as anorexia nervosa.
Anorexia-cachexia, a systemic response to cancer, occurs as a result of increased metabolic rate caused by the tumor cells and metabolites produced and released by tumor cells into the bloodstream. These effects of tumor cells stimulate the satiety center in the hypothalamus and produce appetite loss, gross alterations of metabolic patterns, and a profound systemic condition referred to as anorexia-cachexia. A downward spiral of symptoms occurs with appetite loss leading to malnutrition, weight loss, muscular weakness, and a negative nitrogen balance that contributes to the development of cachectic wasting.
Constipation is a common condition affecting up to one quarter of the American population, especially prevalent in women and people over the age of 65 years.68 Constipation occurs when fecal matter is too hard to pass easily or when bowel movements are so infrequent that discomfort and other symptoms interfere with daily activities.
Constipation may occur as a result of many factors such as diet, dehydration (including lack of fluid intake), side effects of medication, acute or chronic diseases of the digestive system, inactivity or prolonged bed rest, emotional stress, personality, and lack of exercise (see Table 16-2).
Although constipation often is described as a condition of old age, it is probably multifactorial, caused more often by lifestyle factors than by physiologic decline. Lifelong bowel habits, current diet, lack of fluid intake, and immobility are likely causes of constipation in the older adult (older than 65 years).43 Constipation may be the result of underlying organic disease or may be caused by lesions or structural abnormalities within the colon that narrow the intestines and/or rectum, slow-transit alimentary canal, and defecatory disorders.64
People with mechanical low back pain may develop constipation as a result of muscle guarding and splinting that causes reduced bowel motility. Pressure on sacral nerves from stored fecal content may cause an aching discomfort in the sacrum, buttocks, or thighs.60
Constipation associated with defecatory disorders can be the result of pelvic floor or anal sphincter dysfunction, including pelvic floor dyssynergia, spastic pelvic floor syndrome, and anismus. In these disorders, the external anal sphincter contracts and tightens rather than relaxing and opening during defecation. Individuals with this type of constipation often strain to defecate and experience incomplete bowel emptying.89
Dysphagia (difficulty swallowing) produces the sensation that food is stuck somewhere in the throat or chest (esophagus). Dysphagia may be a symptom of many other disorders, including neurologic conditions (e.g., stroke, Alzheimer’s disease, Parkinson’s disease), local trauma and muscle damage (including physical assault), or mechanical obstruction. Obstruction may be intrinsic, originating in the wall of the esophageal lumen (e.g., tumors, strictures, outpouchings called diverticula), or extrinsic, outside the esophageal lumen, such as a tumor or swelling that prevents the passage of food. Dysphagia caused by swelling can occur as a side effect of certain types of drugs such as antidepressants, antihypertensives, and asthma medications.
Achalasia is a failure to relax the smooth muscle fibers of the GI tract. This especially occurs as a result of failure of the lower esophageal sphincter (LES) to relax normally with swallowing. The affected person reports a feeling of fullness in the sternal region and progressive dysphagia.
Although the cause of achalasia is not known, the loss or absence of ganglion cells in the myenteric plexus of the esophagus appears to be a part of the cause. The myenteric plexus is the nerve plexus lying in the muscular layers of the esophagus, stomach, and intestines. Anxiety and emotional tension aggravate the condition and precipitate the attacks. Progression of the condition results in dilation of the esophagus and loss of peristalsis in the lower two thirds of the esophagus.
Heartburn, dyspepsia, pyrosis, or indigestion, a burning sensation in the esophagus usually felt in the midline below the sternum in the region of the heart, is often a symptom of gastroesophageal reflux and occurs when acidic contents of the stomach move backward or regurgitate into the esophagus. The presence of a hiatal hernia, drugs such as alcohol and aspirin, and movements such as lifting, stooping, or bending over after a large meal may bring on heartburn. Indigestion also may be a potential manifestation of angina associated with coronary artery disease.
Certain foods act as muscle relaxants and can also bring on heartburn. For example, chocolate contains four substances that can relax the LES: caffeine, theobromine, theophylline, and fat. Fat-rich foods lower sphincter muscle pressure by release of cholecystokinin from the upper intestinal mucosa. Fat also delays emptying of the stomach, giving more opportunity for this effect to occur. Other implicated foods include spicy and highly seasoned foods, onions, alcohol, peppermint, and spearmint.
Emotional stress can stimulate the vagus nerve, which controls the secretory and motility functions of the stomach. Stimulation of this cranial nerve causes the stomach to churn, increases the flow of various gastric juices, and causes contraction and spasm of the pylorus (opening of the stomach into the duodenum). Heartburn can occur if some of the stomach contents are displaced into the esophagus during this nervous activity.
Abdominal pain accompanies a large number of GI diseases and may be mechanical, inflammatory, ischemic, or referred. Mechanical pain occurs because of stretching of the wall of a hollow organ or the capsule of a solid organ. Inflammatory pain occurs via the release of mediators such as prostaglandins, histamine, and serotonin or bradykinin that stimulate sensory nerve endings.
Ischemic pain occurs as tissue metabolites are released in an area of diminished blood flow. Referred pain usually is well localized and may be associated with hyperalgesia and muscle guarding. Pain from the spine also can be referred to the abdomen, usually as a result of nerve root irritation or compression. This type of neuromusculoskeletal pain referred to the abdomen is characteristically associated with hyperesthesia over the involved spinal dermatomes and is intensified by motions such as coughing, sneezing, or straining.
GI bleeding may be characterized by coffee-ground emesis (vomiting of blood that has been in contact with gastric acid), hematemesis (vomiting of bright red blood), melena (black, tarry stools), or hematochezia (bleeding from the rectum, or maroon-colored stools), depending on the location of the lesion. Bleeding may not be clinically obvious to the client and may be diagnosed only by further testing.
The major causes of upper GI bleeding in the therapy population are erosive gastritis common in (1) severely ill people with major trauma or systemic illness, burns, or head injury; (2) peptic ulcers; (3) use of nonsteroidal antiinflammatory drugs (NSAIDs) such as aspirin or ibuprofen; and (4) chronic alcohol use. Drugs such as warfarin, heparin, and aspirin used as anticoagulants in the treatment of pulmonary emboli, venous thrombus, or valvular abnormalities can contribute to or exacerbate gastric erosion and subsequent bleeding.
Accumulation of blood in the GI tract is irritating and increases peristalsis, causing nausea, vomiting, or diarrhea with possible referred pain to the shoulder or back. The digestion of proteins originating from massive upper GI bleeding is reflected by an increase in blood urea nitrogen (BUN) (see Table 40-2). Other complications include fatigue, postural hypotension, tachycardia, weakness, or shortness of breath on exertion. Slow, chronic blood loss may result in iron deficiency anemia.
Fecal incontinence (inability to control bowel movements) has both psychologic and physiologic contributing factors. Psychologic factors include anxiety, confusion, disorientation, and depression. The most commonly observed physiologic causes seen in a therapy practice are neurologic sensory and motor impairment (e.g., stroke and spinal cord injury); anal distortion secondary to traumatic childbirth, sexual assault, hemorrhoids, and hemorrhoidal surgery; altered levels of consciousness; and severe diarrhea.
AGING AND THE GASTROINTESTINAL SYSTEM
Age-related changes in GI function begin before the age of 50 years. Constipation, incontinence, and diverticular disease are the GI problems most commonly seen in older adults, but each of these disorders has many different underlying causes, and the specific pathogenesis dictates treatment.
Oral changes may include tooth enamel and dentin wear and increasing tooth decay, causing periodontal disease and subsequent tooth loss. Sensory changes may include decreased taste buds and diminished sense of smell resulting in altered sense of taste. These oral and sensory changes eventually depress the appetite and make eating less pleasurable. Salivary secretion decreases, contributing to dry mouth, and when it is complicated by tooth decay or loss, chewing food and swallowing become more difficult.
The alimentary organs (esophagus, stomach, small intestine, and colon), like all muscular structures, lose some tone with age but still manage to perform almost as well in age as in youth. Changes within the alimentary tract include decreases in gastric motility, blood flow, nutrient absorption, and volume and acid content of gastric juice. These changes slow gastric digestion and emptying but usually not enough to be noticeable. Proteins, fats, minerals including iron and calcium, and vitamins are absorbed more slowly and in lesser amounts, and carbohydrates are absorbed more slowly.
A decline in the production of intrinsic factor (IF), a substance that promotes vitamin B12 absorption in the stomach, frequently occurs after middle age. B12 is required to produce blood cells and to maintain the integrity of the nervous and GI systems. B12 deficiency can lead to pernicious anemia with its hematologic and neurologic manifestations and GI symptoms (e.g., diarrhea, constipation, weight loss).
In advanced age, the prevalence of such problems associated with B12 deficiency increases to 90% for those aged 90 and older. Other causes of B12 deficiency with aging include low dietary intake of B12, gastric atrophy (gradual loss of stomach lining with decreased hydrochloric acid), and atrophic gastritis. As these conditions progress, IF is lost and, with it, the ability to extract B12 from food.
Atrophic gastritis may be the result of many possible variables, including normal aging, nutritional deficiency (e.g., iron, folate, ascorbate), autoimmune mechanisms, endocrine insufficiency (e.g., thyroid, adrenal, pancreatic), or infection (usually with Helicobacter pylori).74 H. pylori is a gram-negative spiral bacterium that lives in the gastric mucosal layer of humans and induces a chronic inflammatory response that can result in both peptic ulceration and gastric neoplasms.
Further research is needed regarding the basic mechanisms in neuromuscular dysfunction with aging, including studies of physical characteristics of the colonic wall, pelvic floor function, and neurohormonal control of motility and sensation. Insights on the pathophysiology and mechanisms of neural injury and aging may lead to more specific treatments in the future (e.g., serotonergic agents and neurotrophins).19
In addition to the effects of aging on the GI system, changes in other organ systems (e.g., endocrine, cardiovascular, and nervous systems) also can affect GI structure and function, producing many variations in presentation of illness. Extraintestinal disorders, such as diabetes and the neurologic and vascular changes that occur with age, have a greater effect on the GI tract than the natural process of aging.
THE ESOPHAGUS
Definition and Incidence
A hiatal or diaphragmatic hernia occurs when the cardiac (lower esophageal) sphincter becomes enlarged, allowing the stomach to pass through the diaphragm into the thoracic cavity (Fig. 16-3). Hernias are either congenital, resulting from a failure of formation or fusion of the multiple developmental components of the diaphragm, or acquired, as a result of penetrating wounds, particularly stab wounds and gunshot wounds; blunt trauma, as occurs in motor vehicle accidents; and less commonly as a result of surgical trauma, empyema, and subphrenic abscess.
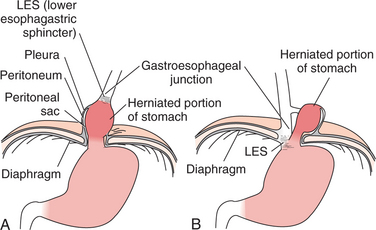
Figure 16-3 Hiatal hernia. A, Sliding hiatal hernia. Approximately 90% of esophageal hiatal hernias are sliding hernias. The stomach and gastroesophageal junction are displaced upward into the thorax (i.e., the stomach and gastroesophageal junction slide up into the thoracic cavity, following the usual path of the esophagus through an enlarged hiatal opening in the diaphragm). B, Rolling hiatal hernia. The remaining hiatal hernias are rolling or paraesophageal hernias. The gastroesophageal junction stays below the diaphragm, but all or part of the stomach pushes through into the thorax.
Hiatal hernia (symptomatic or asymptomatic) is common, and the incidence has been estimated as 5 per 1000. The incidence increases with age and may be as high as 60% in people over 60 years of age. Women are affected more often than men, and children may have the sliding type but do not usually exhibit symptoms until they reach middle age.
Etiologic and Risk Factors
As an acquired condition, multiple causes and risk factors exist for the development of hiatal hernia. Anything that weakens the diaphragm muscle or alters the hiatus (the opening in the diaphragm for the passage of the esophagus) and increases intraabdominal pressure can predispose a person to hiatal hernia. Muscle weakness can be congenital or caused by aging, trauma, surgery, or anything that increases intraabdominal pressure (Box 16-1).
Pathogenesis and Clinical Manifestations
As part of the stomach herniates through a weakness in the diaphragm, regurgitation and motor impairment cause the major clinical manifestations associated with this type of hernia. There is evidence for its origin in esophageal longitudinal muscle dysfunction, indicating that this condition could originate from alterations in nerve innervation, alteration in the viscoelastic properties of distal esophagus, or increased strength of unopposed longitudinal muscle layers.27,28
Symptoms vary depending on the type of hernia present and increase in the presence of tight, constrictive clothing or if the person is in a recumbent position. A sliding hernia may produce heartburn 30 to 60 minutes after a meal, especially if the person is lying down or sleeping in the supine position. Large sliding hernias with reflux may be associated with substernal pain. Rolling hernias are not subject to altered pressure or the resulting reflux, but the person may complain of difficult and painful swallowing.
MEDICAL MANAGEMENT
DIAGNOSIS, TREATMENT, AND PROGNOSIS.
Hiatal hernias may be diagnosed by ultrasonography or barium swallow with fluoroscopy showing the position of the stomach in relation to the diaphragm. These tests may both be necessary to confirm the diagnosis, because the hernia may slide down when the person is placed in the upright position for the radiograph.
The primary treatment remains symptomatic control through the use of antacids and elevating the head of the bed. Treatment is essentially the same for gastroesophageal reflux disease (see the section on Gastroesophageal Reflux Disease in this chapter). The prognosis is good overall with recurrences expected.
Gastroesophageal Reflux Disease
Gastroesophageal reflux disease (GERD), or esophagitis, may be defined as an inflammation of the esophagus, which may be the result of reflux (backward flow) of infectious agents, chemical irritants, physical agents such as radiation and nasogastric intubation, or gastric juices. Heartburn or acid indigestion is not just another term for GERD; it is the most common symptom of GERD, a complex disease with potentially serious complications.
Reflux esophagitis is the most common type of esophagitis, with backward or return flow of the stomach and duodenal contents into the esophagus. Other types of esophagitis, such as infectious esophagitis, may occur with immunosuppression resulting from viral, bacterial, fungal, or parasitic organisms. Chemical esophagitis is usually a result of accidental poisoning in children or attempted suicide in adults. External irradiation for the treatment of thoracic cancers may include portions of the esophagus and lead to esophagitis and stricture.
Incidence and Etiologic Factors
Although any age can be affected, this condition has an increasing incidence with increasing age; older people are more likely to develop severe disease.115
It is estimated that 15% or more of the population has daily symptoms of GERD and as much as one third of the population has monthly symptoms. A reduction in the pressure of the LES, increased gastric pressure, or gastric contents located near the gastroesophageal junction can contribute to the development of esophageal reflux. A wide range of foods and lifestyle factors can contribute to GERD110 (Table 16-3).
Reflux is often associated with a sliding hiatal hernia, also called a diaphragmatic hernia (see Fig. 16-3). Smooth muscle relaxants used for cardiac conditions, such as β-adrenergics, aminophylline, nitrates, and calcium channel blockers, may contribute to incompetence of the LES.
Pathogenesis
Normally, a high-pressure zone exists around the gastroesophageal sphincter, which permits the passage of food and liquids but prevents reflux. Any of the predisposing factors listed in Table 16-3 may alter the pressure around the LES, resulting in reflux.
Hydrochloric acid or gastric and duodenal contents containing bile acid and pancreatic juice coming in contact with the walls of the esophagus cause inflammation and mucosal ulcerations that may bleed. Subsequent granulation tissue causes scarring that frequently develops into esophageal strictures that narrow the esophagus, making swallowing difficult.
Clinical Manifestations
Heartburn, reflux, belching, dysphagia, and painful swallowing are the primary symptoms of esophagitis in younger adults. Pain usually is described as a burning sensation that moves up and down and may radiate to the back, neck, or jaw. Heartburn most often occurs 30 to 60 minutes after a meal and is produced by contact of regurgitated contents with the inflamed esophageal mucosa.
Children can be affected by GERD. Symptoms are similar to those experienced by adults. Persistent GERD is linked to developmental problems. Neurologic disorders and GERD can have overlapping symptoms, such as irritability associated with arching, neck extension, and abnormal muscle tone with spastic movements.
Asthma may be associated with GERD in both children and adults, although the relationship is not well understood at this time. Reflux exacerbates asthma, potentially by the acid’s triggering a vagal response. Aspiration of gastric acid contents can cause bronchorestriction; acid reflux medications can actually help reduce asthma symptoms.
Older adults (older than 70 years) are more likely to have atypical symptoms such as dysphagia, vomiting, respiratory difficulties, weight loss, anemia, and anorexia with or without heartburn or acid regurgitation. Dysphagia is an indication of narrowing of the lumen, usually as a result of edema, spasm, or esophageal strictures. Pathophysiologic changes in esophageal function associated with age are also contributing factors in the development of dysphagia.115 Aggravating factors include recumbency, bending, and meals; relief is obtained with antacids or baking soda, standing and walking, fluids, and avoidance of predisposing factors.
Although GERD is more serious and potentially more damaging than simple heartburn, its symptoms are not necessarily more severe. Reflux in the absence of esophagitis may be asymptomatic or accompanied by a sour taste in the mouth; severe reflux may reach the pharynx and mouth and result in laryngitis and morning hoarseness. Pulmonary aspiration can occur in people who are incapacitated (e.g., neurologically impaired); recurrent pulmonary aspiration can cause aspiration pneumonia, pulmonary fibrosis, or chronic asthma.
Complications of chronic heartburn include reflux esophagitis, strictures, and Barrett’s esophagus. Reflux esophagitis is an inflammation of the lining of the esophagus caused by chronic exposure to stomach acid. Esophageal ulcers that can bleed and cause severe pain can develop. When the ulcers heal, scar tissue forms, creating fibrous strictures that can narrow the lumen of the esophagus.
Persistent GERD may cause a more serious complication known as Barrett’s esophagus. Barrett’s esophagus is a condition that occurs when a damaged esophagus heals abnormally, so that the lining develops a type of cell normally found in the intestine rather than in the esophagus. The intestine-type cells are more resistant to acid but can become cancerous.
MEDICAL MANAGEMENT
Diagnostic tools include history, endoscopy, barium radiography, and H. pylori and esophageal pH testing. A full diagnostic evaluation is not always required when history and current symptoms clearly point to esophagitis; a therapeutic trial of treatment in mild cases may be diagnostic in itself.
Ironically, some individuals who think they have excess stomach acid may actually suffer from indigestion from deficient stomach acid. Taking antacids when stomach acid is already chronically deficient can lead to nutritional deficiencies such as vitamin B12 deficiency. The end result can be even more complications from anemia and dementia. Proper medical evaluation is always advised before self-treating for what may seem like indigestion.
Response to nitroglycerin may help the physician differentiate between esophagitis and angina pectoris, but the response is not always diagnostic because nitroglycerin also can relieve esophageal spasm and some women with angina improve with antacids rather than nitroglycerin.
TREATMENT.
The goals of treatment are to alleviate symptoms, heal esophagitis if present, maintain remission of the disease, and manage any complications.
MEDICATIONS.
The first-line treatment is acid-suppressing inhibitors called proton pump inhibitors (PPIs) (e.g., Prilosec, Aciphex, Prevacid, Nexium) for the healing of erosive GERD and maintenance of healed erosive GERD (Table 16-4).
These medications shut off the chemical pump that transports acid into the stomach, cutting acid production to zero. This usually brings fast relief and healing of burns and erosions in the esophagus. The individual treated with these drugs must digest foods without the benefit of stomach acid.
Long-term suppression of stomach acid is not advised unless recurrence cannot be controlled or esophageal healing cannot be maintained. Doctors usually administer PPIs for a limited time to allow healing but avoid adverse effects of chronic use. The long-term effects of PPIs remain unknown. Researchers are looking for a way to restore the natural mechanisms that prevent gastric acid reflux.
Uncomplicated cases of esophagitis and typical symptoms of heartburn and regurgitation may be treated with two types of over-the-counter drugs: histamine 2 blockers (H2-blockers) such as Tagamet, Zantac, or Pepcid AC and antacids (Tums, Rolaids, Mylanta, Maalox). Antacids neutralize stomach acid, whereas histamine blockers prevent acid secretion. The goal of antacids is quick symptom relief, whereas the goal of histamine blockers is long-term cure. Drinking fluids between meals (not with meals) helps reduce the likelihood of reflux.
Individuals should avoid prolonged and regular use of antacids as this can reduce the body’s phosphate levels, with resultant fatigue and loss of appetite. Aluminum hydroxide tends to produce constipation, whereas magnesium hydroxide can cause loose stools or diarrhea in some individuals.
LIFESTYLE MODIFICATIONS.
Lifestyle modifications also may be recommended, including wearing loose clothing; avoiding caffeine, nicotine, alcohol, salicylates, and NSAIDs; remaining upright at least 3 hours after meals; avoiding meals near bedtime or nap time; losing weight, if obese (this last recommendation has not been confirmed by clinical studies).
Elevation of the head of the bed at least 6 inches to reduce nocturnal reflux and enhance esophageal acid clearance may help. Some people find it helpful to sleep on their left side. This may help keep the acidic contents of the stomach below the juncture of the lower esophagus.
Some people find it helpful to avoid vigorous activities for 1 to 3 hours after eating. Avoiding large meals that can distend the stomach and avoiding foods that decrease pressure in the LES (e.g., chocolate, peppermint, alcohol, caffeine, fried and fatty foods), thereby causing reflux, may be helpful. Acidic foods such as orange juice, wine, and tomatoes may irritate an already inflamed esophagus. Keeping a food diary and recording any symptoms may help each individual determine personal triggers.
Chewing sugarless gum after a meal helps promote salivation and aids in neutralizing acid. Greater saliva production also soothes the esophagus by washing acid back down to the stomach. Avoiding peppermint flavors and quitting the use of tobacco products will also increase saliva production.
SURGERY.
Minimally invasive endoscopic surgical procedures (e.g., antireflux therapy) to tighten up the LES are being developed. These procedures may eventually allow the majority of people to stop taking all GERD medications, including PPIs.72
Endoscopic or luminally delivered techniques for the treatment of GERD include endoscopic radiofrequency therapy, endoscopic injection or implantation of a liquid polymer directly into the weakened esophageal sphincter, and plication. Data to establish long-term efficacy of these techniques have not been reported yet, but the results of short-term studies have been favorable for some individuals. Not everyone is a good candidate for surgical intervention.149
Suturing or gastroplication is done endoscopically so that the surgeon can see where to place the stitches. A special suturing device called an EndoCinch is inserted into the esophagus, allowing the surgeon to place a stitch in the upper stomach about 1 cm below the LES. Another stitch is placed next to the first and the two stitches are tied or clipped together to form a pleat. More than one pleat may be needed. Fundoplication is another antireflux surgical procedure. It involves wrapping the fundus of the stomach around the lower esophagus, creating either a 360-degree wrap or an incomplete wrap.
An implantable product called Enteryx has been removed from the market due to reported complications. Enteryx was a liquid injected into the lower esophagus through an endoscope, which then solidified into a spongy material to reinforce the sphincter.22
PROGNOSIS.
The prognosis is good for reflux esophagitis with complete symptom resolution, but often variable for the chemical type and poor for infectious esophagitis. GERD can contribute to asthma and vocal cord inflammation, and people who have uncontrolled acid reflux are at increased risk of developing esophageal cancer, which has a very poor prognosis.50,63
Mallory-Weiss Syndrome
Mallory-Weiss syndrome is mucosal laceration of the lower end of the esophagus accompanied by bleeding. The most common cause is severe retching and vomiting as a result of alcohol abuse, eating disorders such as bulimia, or in the case of a viral syndrome.
Other conditions such as pregnancy, migraine, hiatal hernia, gastric ulcer, biliary disease, and various medications have been associated with Mallory-Weiss syndrome. Any event that suddenly raises transabdominal pressure in exercise or lifting can cause such a tear. Diagnosis is made on endoscopy, and when treatment is necessary, fluid replacement and blood transfusion and H2-receptor antagonists may be administered. Endoscopic ligation may be required if bleeding cannot be brought under control.
Scleroderma Esophagus
Esophageal involvement is common in people with progressive systemic sclerosis caused by the CREST syndrome (calcinosis, Raynaud’s phenomenon, esophageal motility disorder, sclerodactyly, and telangiectasia; see the section on Systemic Sclerosis in Chapter 10).
The esophageal lesions in systemic sclerosis consist of muscular atrophy of the smooth muscle portion, with weakness of contraction in the lower two thirds of the esophagus and incompetence of the LES.
Symptoms include dysphagia to solids and to liquids in the recumbent position. Heartburn and regurgitation occur in the presence of gastroesophageal reflux and esophagitis. Currently, no effective treatment exists for the motor difficulty, but reflux esophagitis and its complications are treated aggressively as described earlier.
Neoplasm
Histologically, two types of esophageal cancer exist: squamous cell and adenocarcinoma. Worldwide, more than 90% of all esophageal cancers are squamous cell carcinomas. In the West, however, there has been a decline in the frequency of squamous cell carcinoma and a dramatic rise in the frequency of adenocarcinoma of the esophagus.
Esophageal cancer is relatively uncommon, but the incidence of adenocarcinoma is rising, possibly as a result of H. pylori eradication in GERD. Evidently, although H. pylori predisposes individuals to peptic ulceration and gastric neoplasms, it also has a protective effect against esophageal cancer. The mechanism by which this works remains unclear.
Barrett’s esophagus, which is a precursor of esophageal adenocarcinoma, may be more common in older adults (over age 65) than previously thought. Screening for Barrett’s esophagus in patients undergoing a colonoscopy has revealed the presence of Barrett’s esophagus in up to 22% of men and 11% of women.153
Risk Factors
Esophageal cancer is known for its marked variation by geographic region, ethnic background, and gender. In the United States, adenocarcinoma of the esophagus most frequently affects middle-aged white men, whereas squamous cell cancer is much more common in blacks and is associated with alcohol and tobacco use.18,35
The presence of other esophageal disease such as hiatal hernia, reflux, Barrett’s esophagus, rings, webs, diverticula, stricture from lye ingestion, achalasia, and other head and neck cancers increases the risk of developing esophageal cancer. Gastroesophageal reflux is highly correlated with an increased risk of esophageal adenocarcinoma (but not squamous cell carcinoma of the esophagus).63
Etiologic Factors
Chronic inadequate nutrition can impair both the structure and function of the esophagus. Nutritional deprivation, particularly deficiencies of vitamins C, E, and B6, niacin, selenium, and zinc, results in mucosal changes, increasing the vulnerability of esophageal mucosa to neoplastic changes. Obesity has been established as a strong risk factor for esophageal adenocarcinoma.140
Any change in esophageal function that permits food and drink to remain in the esophagus for prolonged periods of time can result in ulceration and metaplasia. Chronic exposure to irritants such as alcohol and tobacco (inhaled or chewed) also can cause neoplastic transformation. Nitrosamines are powerful carcinogens involved as causative agents of cancers of the lung, oral cavity, esophagus, and pancreas associated with the use of tobacco products. Chronic stimulation of nicotinic receptors by nicotine and nitrosamines in smokers is one of the molecular events responsible for stimulation of cell proliferation and ultimately neoplasms.
Tumors may be obstructive, causing circumferential compression or ulceration with bleeding. Distant metastases also may occur, most commonly involving the liver and lung, but almost any organ can be involved.
Barrett’s esophagus is a premalignant condition in which the normal squamous epithelium of the esophagus that is damaged by GERD is replaced by metaplastic epithelium, predisposing the individual to esophageal adenocarcinoma.
The progression of Barrett’s metaplasia to adenocarcinoma is associated with several changes in gene structure, gene expression, and protein structure. Researchers are identifying some of the molecular alterations in hopes of finding markers for early cancer detection or prognostication.158
Pathogenesis
Adenocarcinoma of the esophagus is thought to develop through a stepwise process termed the metaplasia-dysplasia-carcinoma sequence. The replacement of reflux-damaged esophageal epithelium by metaplastic intestine-type epithelium results in Barrett’s esophagus. The metaplastic epithelia may be predisposed to deoxyribonucleic acid (DNA) damage that causes dysplasia and carcinoma.140
Underlying these changes in cell phenotype are sequential genetic alterations that bring the normal epithelial cells closer to malignancy. Many of the molecular alterations that occur during carcinogenesis of the upper GI tract affect genes whose protein products regulate the cell cycle (see Fig. 9-3) clock apparatus, the key nuclear mechanism that controls whether a cell will proliferate, differentiate, or die. There is a molecular switch that helps the cell “decide” which phase it will enter. The exact sequence of molecular changes necessary to alter this switching and produce adenocarcinoma remains obscure.140
Clinical Manifestations
Dysphagia with or without pain is the predominant symptom of this condition and may not occur until the diameter of the lumen of the esophagus is reduced 30% to 50%. Pain associated with dysphagia usually is described as pressurelike and may radiate posteriorly between the scapulae.
Heartburn initiated by lying down is the most common type of pain. Constant retrosternal chest pain that radiates to the back may occur in the presence of mediastinal extension or spinal nerve compression. Other signs and symptoms include anorexia and weight loss, hoarseness resulting from laryngeal nerve compression, and tracheoesophageal fistula causing cough and recurrent pneumonia.
MEDICAL MANAGEMENT
Preventing GERD and Barrett’s esophagus is the major method of reducing rates of esophageal adenocarcinoma. Despite this knowledge, the rates of esophageal adenocarcinoma remain unchanged.153 There are many possible reasons for this, but the exact reason(s) remains unclear. It is possible that GERD is underreported and undertreated in many adults, especially those over age 65. Many people with GERD may have few or no symptoms, so that progression of the condition to Barrett’s disease and transformation to adenocarcinoma goes unnoticed until it is too late.153
Whereas early screening and detection can reveal dysplasia (a potentially curable form of neoplasia), there is no proof that such screening programs actually reduce morbidity or mortality associated with this condition.140
Studies suggest that 40% of individuals with esophageal adenocarcinoma have no history of GERD symptoms.71 This means that screening only individuals with known GERD will leave out a significant portion of adults who may be affected. And there are reports of incurable malignancies developing despite endoscopic surveillance programs.145
Researchers are continuing to study the strategy that early treatment of individuals with GERD might prevent the first step in the metaplasia-dysplasia-carcinoma sequence. Some day it may be possible to use a biomarker to identify individuals with GERD who are predisposed to develop Barrett’s esophagus. Aggressive acid suppression and screening might be appropriate for this group of people.
DIAGNOSIS.
Diagnosis is made by endoscopy with cytology and biopsy. After diagnosis, staging of the disease is performed with chest and abdominal computed tomographic (CT) scanning or, if available, endoscopic ultrasonography to determine the most appropriate treatment.
TREATMENT.
Neoplasms are classified as resectable with curative intent, resectable but not curable, and not resectable and not curable. The presence of distant metastases, invasion of the mediastinal muscularis or pleural invasion, or distant lymph node involvement excludes a curative resection. Curative surgery is esophageal reconstruction, which may improve the ability to eat and may prevent local tumor complications.
The use of preoperative chemotherapy with radiation is under investigation. Patients with unresectable disease or poor operative candidates may receive radiation therapy, which provides short-term relief of symptoms. Combinations of esophageal brachytherapy, external beam radiation, and multidrug combination chemotherapy are under clinical investigation; the use of brachytherapy appears to be associated with severe toxicity and the development of fistulas.56
PROGNOSIS.
Endoscopic surveillance can detect esophageal adenocarcinomas when they are early and curable, but most of these neoplasms are detected at an advanced stage. Carcinoma of the esophagus has one of the lowest possibilities of cure, with 5-year survival rates estimated to be approximately 10% overall and a median survival of less than 10 months.
The first symptoms of esophageal cancer are not usually apparent until the tumor involves the entire esophageal circumference. More importantly, the tumor by that time has often invaded the deeper layers of the esophagus and adjacent structures and is unresectable. Esophageal cancer metastasizes rapidly, and given the continuous nature of lymphatic vessels in the area, removal of lymph nodes with the tumor is impossible, contributing to the poor prognosis.
Esophageal Varices
Esophageal varices are dilated veins in the lower third of the esophagus immediately beneath the mucosa. Dilation occurs in the presence of portal hypertension, usually secondary to cirrhosis of the liver. All the blood from the intestine drains via the portal vein to the liver before passing into the general circulation. Therefore, any disease of the liver or portal vein that obstructs the flow of blood will cause expanding force pressure.
The normal anatomic reaction to this condition is to decompress the portal venous system by opening up bypass veins (collaterals), most commonly around the lower esophagus and stomach. When blood flow can no longer be counterbalanced by the variceal wall tension, the dilated veins (varices) rupture and bleed. Rupture and hemorrhage are common when portal pressure causes the varices to reach a size greater than 5 mm in diameter.
Variceal bleeding usually presents with painless but massive hematemesis with or without melena. Associated signs range from mild postural tachycardia to profound shock, depending on the extent of blood loss and degree of hypovolemia (decreased amount of blood in the body). The clinical picture is frequently consistent with chronic liver disease.
Diagnosis requires differentiation from peptic ulcer, gastritis, and other bleeding sources, often concurrent conditions in people with cirrhosis secondary to alcoholism. Diagnosis is made by fiberoptic endoscopy. Bleeding varices constitute one of the most common causes of death in people with cirrhosis and other disorders associated with portal hypertension; therefore, prevention and treatment are very important to prevent and replace blood loss and maintain intravascular volume.
About half of all episodes of variceal hemorrhage cease without intervention, although a high risk of rebleeding exists. Various prophylactic and therapeutic approaches to management include pharmacologic agents and endoscopic interventions such as band ligation or sclerotherapy (the injection of hardening agents).
For bleeding not controlled with these methods, a stent may be placed between the hepatic vein and the intrahepatic portion of the portal vein (transjugular intrahepatic portosystemic shunt [TIPS]).66 This procedure provides a means of lowering portal pressure. Liver transplantation may be considered in cases unresponsive to treatment.62
Congenital Conditions
Overview.: Tracheoesophageal fistula (TEF) is the most common esophageal anomaly and one of the most common congenital defects, occurring in approximately 1 in 4000 live births with equal gender distribution. In this disorder, the esophagus fails to develop as a continuous passage and abnormal communication between the lower portion of the esophagus and trachea occurs, often combined with some form of esophageal atresia, a condition in which the esophagus ends in a blind pouch (Fig. 16-4). Other associated conditions include congenital heart disease, prematurity, and the VATER complex (vertebral defects, imperforate anus, tracheoesophageal fistula, and radial and renal dysplasia).
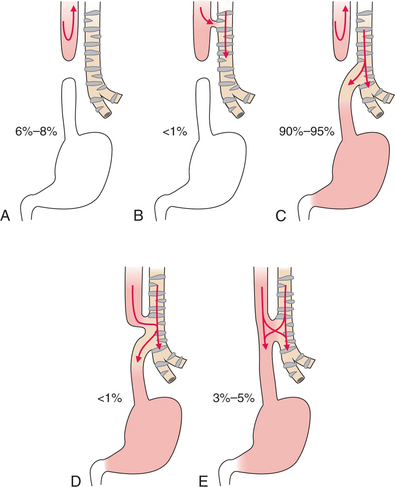
Figure 16-4 Five types of esophageal atresia and tracheoesophageal fistula. A, Simple esophageal atresia. Proximal and distal esophagus end in blind pouches. Nothing enters the stomach; regurgitated food and fluid may enter the lungs. B, Proximal and distal esophageal segments end in blind pouches, and a fistula connects the proximal esophagus to the trachea. Nothing enters the stomach; food and fluid enter the lungs. C, Proximal esophagus ends in a blind pouch, and a fistula connects the trachea to the distal esophagus. Air enters the stomach; regurgitated gastric secretions enter the lungs through the fistula. D, Fistula connects both proximal and distal esophageal segments to the trachea. Air, food, and fluid enter the stomach and lungs. E, Simple tracheoesophageal fistula between otherwise normal esophagus and trachea. Air, food, and fluid enter the stomach and lungs. Between 90% and 95% of esophageal anomalies are type C; 6% to 8% are type A; 3% to 5% are type E; and less than 1% are type B or D.
Etiologic Factors and Pathogenesis.: Because TEF is a congenital malformation, the cause is unknown, but abnormalities are postulated to arise from defective differentiation as the trachea separates from the esophagus during the fourth to sixth weeks of embryonic development. Defective growth of endodermal cells leads to atresia (closure or absence of a normal body opening or tubular structure). In 90% of cases, the esophagus ends in a blind pouch with communication between the distal esophagus and the trachea. Less often, the proximal esophagus communicates with the trachea, or the esophagus is continuous in an H-type fistula.
Clinical Manifestations.: The blind end of the proximal esophagus has a capacity of only a few milliliters, so as the infant with esophageal atresia swallows oral secretions, the pouch fills and overflows into the pharynx, resulting in excessive drooling and, occasionally, aspiration.
If a fistula connects the trachea with the distal esophagus, the abdomen fills with air and becomes distended, which may interfere with breathing. If the fistula connects the proximal esophagus to the trachea, the first feeding after birth will signal a problem.
As the infant swallows, the blind end of the esophagus and the mouth fill with fluid that is aspirated into the lungs when the infant tries to take a breath. This triggers a protective cough and the choke reflex with intermittent cyanosis. Coughing, choking, and cyanosis are called the three Cs of TEF and may occur especially with the H-type fistula, which may not be diagnosed for weeks to months after birth.
MEDICAL MANAGEMENT
DIAGNOSIS, TREATMENT, AND PROGNOSIS.
Esophageal anomalies are usually diagnosed at birth on the basis of clinical manifestations, but new technology is making in utero diagnosis more readily available. Occasionally this condition escapes detection until adulthood, when recurrent pulmonary infections call attention to it. Confirmation of the non–H type is made by passing a catheter into the esophagus with radiographs of the chest and abdomen taken with the tube in place to show the level of the blind pouch. Fluoroscopy using radiopaque fluid also may be used to establish the diagnosis.
Surgical treatment to restore esophageal continuity and eliminate the fistula usually is performed shortly after birth. Surgical procedures may be performed in stages for infants who are premature, have multiple anomalies, or are in poor health. Antibiotics are instituted early owing to the certainty of aspiration pneumonia.
Without early diagnosis and treatment this condition is rapidly fatal. Early detection prevents feedings until the problem is corrected; feeding can cause aspiration and its complications. The survival rate is nearly 100% in full-term infants without severe respiratory distress or other anomalies. In premature, low-birth-weight infants with associated anomalies, the incidence of complications is high.
THE STOMACH
Definition and Incidence
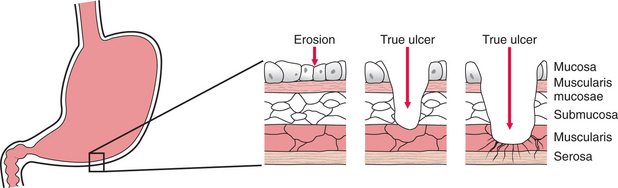
Gastritis, inflammation of the lining of the stomach (gastric mucosa), is not a single disease but represents a group of the most common stomach disorders. Gastric erosions by definition are limited to the mucosa and do not extend beneath the muscularis mucosae. Based on clinical features, gastritis can be classified as acute or chronic.
Other classifications may be made according to clinical, endoscopic, radiographic, or pathologic criteria. Acute gastritis may be hemorrhagic or acute erosive, reflecting the presence of bleeding from the gastric mucosa. Gastric erosions and sites of hemorrhage may be distributed diffusely throughout the gastric mucosa or localized to the body of the stomach.
Chronic gastritis has two forms classified as types A and B. Type A gastritis is the less common form of chronic gastritis, associated with pernicious anemia, and is possibly an autoimmune disorder. The most severe type of chronic gastritis, chronic fundal gastritis, occurs in association with autoimmune diseases such as diabetes, Addison’s disease, and thyroid disease, also suggesting an autoimmune mechanism.
Type B, the more common form of chronic gastritis, is caused by chronic bacterial infection by H. pylori. H. pylori is a gram-negative bacterium infecting half the world’s population and causing chronic active gastritis in virtually all infected individuals.122
Etiologic Factors
Acute erosive gastritis may develop without apparent cause but is more likely to occur in association with serious illness or with various medications such as aspirin or other NSAIDs that can produce acute gastric mucosal erosions.
GI complications occur only in a small percentage of people taking NSAIDs, but the widespread use of these agents results in a substantial number of people affected. Most susceptible persons are those 65 years of age or older, especially those who have a history of ulcer disease.
Other risk factors include taking NSAIDs longer than 3 months, taking high-dose or multiple NSAIDs, and concurrent corticosteroid or anticoagulation therapy. Use of NSAIDs combined with selective serotonin uptake inhibitors is also linked with upper GI bleeding.105,134,156
Acute erosive gastritis associated with physiologic stress, often referred to as stress-induced gastritis, is associated with hospitalization for severe life-threatening disease, central nervous system injury, or trauma (particularly burns but also renal failure, mechanical ventilation, sepsis, and hepatic failure).
No persuasive evidence exists that acute gastric mucosal injury associated with stress, alcohol, aspirin, or other NSAIDs progresses to chronic gastritis. H. pylori is a primary risk factor in the development of chronic gastritis and the gastritis-associated diseases (e.g., gastric ulcer, duodenal ulcer, gastric cancer, gastric B-cell lymphoma/ mucosa-associated lymphoid tissue [MALT] lymphoma), but other risk factors include aging, vitamin deficiencies, abnormalities of the gastric juice, hiatal hernia, or a combination of any of these.
Pathogenesis
Agents known to injure the gastric mucosa (e.g., H. pylori, aspirin or other NSAIDs, bile acids, pancreatic enzymes, alcohol) alter the mucosal defense mechanism, leading to acute gastritis. The mechanism of mucosal injury is unclear and probably multifactorial. The most commonly accepted theory for agent-induced mucosal injury is the suppression of endogenous prostaglandins that normally stimulate the protective secretion of mucus.
The progression of chronic gastritis has three phases. Superficial gastritis is the initial stage with inflammation limited to the upper epithelial half of the gastric mucosa. Atrophic gastritis, the second stage, takes place as the inflammatory process extends to the deep portions of the mucosa with progressive distortion and destruction of the gastric glands.
Pepsinogen, hydrochloric acid, and IF are diminished, and the feedback mechanism that normally inhibits gastrin secretions is impaired, causing elevated plasma levels of gastrin. As mentioned under Aging and the Gastrointestinal System, IF is a glycoprotein secreted by the gastric glands that plays an important role in the absorption of vitamin B12. IF deficiency resulting in vitamin B12 deficiency may lead to pernicious anemia. The final stage, gastric atrophy, involves a profound loss of the glandular structures with thinning of the mucosa.
Clinical Manifestations
The most noticeable symptom of acute gastritis is epigastric pain with a feeling of abdominal distention, loss of appetite, and nausea. Pain is much less common with erosive gastritis than with ulcer disease; painless GI hemorrhage is frequently the only clinical manifestation.
Additional symptoms may include heartburn, low-grade fever, and vomiting. Occult (no visible evidence) GI bleeding commonly occurs, especially in cases of trauma and in people taking aspirin or other NSAIDs. Chronic gastritis may be asymptomatic, or pain may occur after eating accompanied by indigestion.
MEDICAL MANAGEMENT
DIAGNOSIS, TREATMENT, AND PROGNOSIS.
The diagnosis of gastritis may be made by a careful history, but confirmation is made by upper endoscopic examination, possibly including biopsy, because epigastric pain may be due to peptic ulcer, gastroesophageal reflux, gastric cancer, biliary tract disease, food poisoning, and viral gastroenteritis.
Noninvasive tests such as the urea breath test or stool antigen test can be used to confirm the presence of H. pylori within the stomach. Antimicrobial therapy along with antisecretory drugs are used to eradicate H. pylori.
Management of gastritis requires avoidance of identified irritating substances (e.g., caffeine, nicotine, alcohol) combined with the use of PPIs, antacids, and/or H2-blocking agents to block or reduce gastric acid secretion and minimize stomach acidity.
Vitamin B12 is administered to correct pernicious anemia when it develops secondary to chronic gastritis. Because people taking NSAIDs and those in intensive care units have a high incidence of erosive gastritis, preventive therapy may be used to reduce mucosal injury.
Prognosis is good for both acute and chronic gastritis, especially with removal of the predisposing factors for acute gastritis. The risk of gastric cancer is known to be high in people with chronic gastritis and particularly in those with atrophic gastritis.
Peptic Ulcer Disease
An ulcer is a break in the protective mucosal lining exposing submucosal areas to gastric secretions. The word peptic refers to pepsin, a proteolytic enzyme, the principal digestive component of gastric juice, which acts as a catalyst in the chemical breakdown of protein.
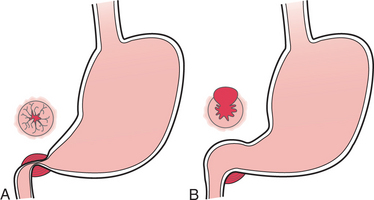
Acute lesions of the mucosa that do not extend through the muscularis mucosae are referred to as erosions. Chronic ulcers involve the muscular coat, destroying the musculature and replacing it with permanent scar tissue at the site of healing. Ulcers extending to the muscularis mucosae damage blood vessels, causing hemorrhage (Fig. 16-5).
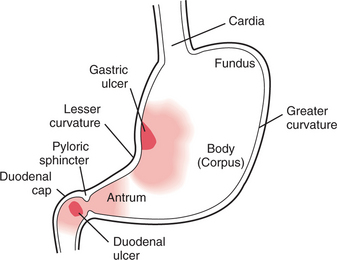
Two kinds of peptic ulcer exist: the gastric ulcer (GU), which affects the lining of the stomach, and the duodenal ulcer (DU), which occurs in the duodenum. DUs are two to three times more common than GUs, although these ulcers can coexist. About 95% of DUs occur in the duodenal bulb or cap. About 60% of benign GUs are located at or near the lesser curvature and most frequently on the posterior wall (Fig. 16-6).
Stress ulcers, or secondary ulcers, occur in response to prolonged psychologic or physiologic stress (e.g., severe trauma, surgery, extensive burns, brain injury), causing an upset in the aggressive-defensive balance. For example, gastric mucosal changes develop within 72 hours in 80% of clients with burns over more than 35% of the body. The mechanism causing stress ulcers is unknown but probably involves ischemia of the gastric mucosa, which has large oxygen requirements and low gastric pH (high acidity). Stress ulcers differ pathologically and clinically from peptic ulcers with very few symptoms and are painless until perforation and hemorrhage occur.
Incidence
In the United States there are about 500,000 new cases of peptic ulcer and 4 million ulcer recurrences per year. Up to 10% of the American population will develop an ulcer at some time in their lives.
Older age groups, especially middle-aged and older adults, are more likely to develop GUs; the peak incidence for GUs is in the sixth decade. Approximately 10% to 20% of people with GUs also have DUs.
DUs are the most common, with an average age at onset in the mid-thirties, although DUs can occur at any time (including infancy). In the past, men were more likely than women to develop gastric and duodenal ulcers, but now equal distribution exists between the genders. The overall frequency of DU has been decreasing in the United States, especially in males.
Etiologic and Risk Factors
In 90% of all ulcers (as well as for chronic gastritis), H. pylori bacterial infection is an important risk factor for and cause of the condition. Although H. pylori has been identified as a major cause of ulcer disease, not all infected people develop ulcers. This may be because certain H. pylori strains are more virulent than others. Some people naturally produce more stomach acid than others, which may explain some of the differences.
The majority of recent studies have not found tobacco use or alcohol consumption to be risk factors for H. pylori infection, although adequate nutritional status, especially frequent consumption of fruits and vegetables and vitamin C, appears to be protective.17
Lifestyle and psychologic stress are still considered as potential factors in the development of ulcers in some people. The presence of recurrent ulcers in individuals with multiple stressors, poor coping skills, and persistent anxiety and depression lends support to the hypothesis that physiologic changes leading to ulcer can occur secondary to psychologic stress. The mechanism of this association remains unknown.61,111
The long-term use of NSAIDs has deleterious effects on the entire GI tract, from the esophagus to the colon, although the most obvious clinical effect is on the gastroduodenal mucosa. The exact mechanism remains unknown, but theoretically these drugs break down the mucous membrane that protects the GI tract by inhibiting the synthesis of gastric mucosal prostaglandins.
Prostaglandins have two types of actions: inhibition of acid secretion and enhancement of mucosal resistance to injury by mechanisms independent of acid-secretion inhibition, the latter phenomenon being called cytoprotection. This interference with normal mucosal protective mechanisms leads to local injury by allowing stomach acids to dissolve the intestine (for further discussion of the systemic effects of NSAIDs, see Chapter 5; see also Table 5-1 for a list of commonly used NSAIDs).
Pathogenesis
The mechanisms of injury differ between duodenal and gastric ulcers. DU is essentially an H. pylori–related disease and is caused mainly by an increase in acid and pepsin load with gastric metaplasia occurring in the duodenal cap.
GU is most commonly associated with NSAID ingestion, although H. pylori might also be present. In both conditions, ulcer is associated with an imbalance between protective and aggressive factors, with inflammation being a major cause in this imbalance.161 Cholinergic hypersensitivity and parasympathetic dominance are related to the stimulation of hydrochloric acid and pepsin, which is a cofactor in the development of erosive injury to the gastric mucosa.
Psychologic stress, cigarette smoking, alcohol consumption, use of NSAIDs (including aspirin), oral bisphosphonates, potassium chloride, immunosuppressive medications, and an age-related decline in prostaglandin levels have all been shown to contribute to peptic ulcer disease. It was, however, the discovery that H. pylori is a significant factor in the development of peptic ulcer disease that led to the understanding of the role that inflammation and its associated cytokine cascade plays in gastric acid secretion.161
The presence of the H. pylori infection induces various humoral and cellular immunities in the gastric mucosa, but the exact mechanism of H. pylori–induced gastroduodenal diseases remains unknown. Prolonged impaired and excessive immunoinflammatory responses with destruction of the mucosal barrier system appear to be the underlying mechanism for tissue injury.107 H. pylori evades attack by the host immune system and causes chronic inflammation by several mechanisms (e.g., reducing the thickness of the mucus gel layer, decreasing blood flow to the mucosal layer).161
In the case of emotional stress, an increase in gastric secretion, blood supply, and gastric motility by irritation of the vagal nerve stimulates the thalamus. The sympathetic nervous system then causes the blood vessels in the duodenum to constrict, making the mucosa more vulnerable to trauma from gastric acid and pepsin secretion. Multiple chemical, neural, and hormonal factors participate in regulation of gastric acid secretion, making ulcer development a multifactorial process in such cases.
Clinical Manifestations
No specific symptom differentiates H. pylori–induced ulcers from NSAID-associated ulcers. The classic symptom of peptic ulcer is epigastric pain described as burning, gnawing, cramping, or aching near the xiphoid, coming in waves that last several minutes. Distention of the duodenal bulb produces epigastric pain, which may radiate to the back.
Perforation of the posterior duodenal wall causes steady midline pain in the thoracic spine from T6 to T10 with radiation to the right upper quadrant. The daily pattern of pain is related to the secretion of acid and the presence of food in the stomach (e.g., the presence of food may cause GU pain, whereas pain occurs 1 to 3 hours after meals with DUs).
Other symptoms include nausea, loss of appetite, and sometimes weight loss. Symptoms may occur for 3 or 4 days or weeks, subsiding only to reappear weeks or months later.
Many people report symptoms outside the classic presentation of DU. Some people are asymptomatic until complications occur; this is especially typical in the older adult as weakened abdominal muscles and diminished pain perception mask early symptoms or present as nonspecific indicators such as mental confusion.
Complications may include hemorrhage with resultant anemia, perforation, and obstruction accompanied by unremitting pain. Symptoms depend on the severity of the hemorrhage. In mild bleeding, slight weakness and diaphoresis may be the only symptoms. Signs of bleeding include bright red blood in the vomitus, coffee-ground vomitus, and melena (black, tarry stools).
MEDICAL MANAGEMENT
Ulcers are diagnosed on the basis of symptoms and history, although the history is not as characteristic for GU as it is for DU. Appropriate use of one of the many tests to diagnose H. pylori (e.g., serology, urease breath testing, saliva test, biopsy, and culture) can identify those individuals likely to benefit from antimicrobial treatment.
Breath testing and serology have comparable sensitivity and specificity, but serology provides faster results and is preferred for the initial diagnosis. Breath testing is more useful than serology in diagnosing failure of H. pylori eradication or reinfection in people who were previously treated for H. pylori infection, because serologic results will remain positive for several months even after successful treatment.52
Other tests may include barium radiographic examination and gastroscopy (endoscope passed into the stomach) used to determine the site of bleeding and to differentiate between benign and malignant ulcerations.
PREVENTION AND TREATMENT.
The primary goals of medical treatment of peptic ulcers are (1) relief of symptoms, (2) promotion of healing, (3) prevention of complications, and (4) prevention of recurrences. Each person responds differently to different treatment modalities, requiring individual treatment planning. In general, GUs tend to heal more slowly than DUs.
Antimicrobials (antibiotics) are 85% effective in the treatment of H. pylori, along with antisecretory drugs such as PPIs, antacids, and H2-blocking agents (stomach acid suppressors or blockers; see Table 16-4) to allow the ulcer to heal completely.
Antibiotic resistance with increasing eradication failure rates is becoming increasingly common and has led to studies of new regimens for primary therapy of GERD to eliminate the acid-hypersecretory state that predisposes the person to peptic ulcer disease. Issues of dosage and timing remain under investigation.
There does not appear to be lifelong immunity from H. pylori once infected; researchers are working on vaccine development.147 Researchers are actively seeking ways to prevent ulcer development and ulcer recurrence in NSAID users and the best means to treat non-NSAID, non–H. pylori-associated peptic ulcers.161
NSAID-induced injury with resultant ulceration is a separate issue from H. pylori–induced ulcers. Whether or not anyone who has had an ulcer or chronic indigestion should be tested and treated for H. pylori before starting long-term NSAID therapy remains a point of controversy. Using a protective agent with the NSAID may be recommended, especially if long-term use of the NSAID is needed. The smallest effective dose of NSAIDs is recommended, and clients must be warned against adding over-the-counter NSAIDs to a prescription dose.
No substantial evidence supports dietary modifications as a treatment approach to peptic ulcers, although adequate nutrition may be preventive.17 Bland diets, soft diets, milk and cream diets, and diets free of spices or fruit juices have no known effect in reducing gastric acid secretion, relieving symptoms, or promoting ulcer healing.
Foods that seem to aggravate a person’s symptoms most likely should be avoided. Coffee, caffeinated or not, stimulates gastric acid secretion and should be avoided. Researchers have reported that exercise at least three times a week greatly reduces the risk of GI bleeding. More strenuous forms of exercise such as swimming and bicycling do not provide greater protection from GI bleeding than do more moderate exercises such as walking.25,112
Surgical intervention is required for perforation, because gastric and intestinal contents spilling into the peritoneal cavity can cause chemical peritonitis, bacterial septicemia, and hypovolemic shock. Peristalsis diminishes, and paralytic ileus can develop. Outlet obstruction caused by scarring of the duodenum or caudal portion of the stomach may also require surgery.
PROGNOSIS.
Prognosis is usually good and medical management can adequately control ulcers unless massive hemorrhage or perforation occurs, which carries a high mortality. Both duodenal and gastric ulcers tend to have a chronic course with remissions and exacerbations.
Benign GUs should heal completely within 3 months of treatment. Well-controlled, double-blind studies have shown that curing H. pylori usually results in curing DU disease; however, antimicrobial resistance is largely responsible for treatment failure.
Gastric Cancer
The GI system, including the stomach, is a common site for extranodal disease associated with lymphoma; primary lymphoma of the stomach is relatively uncommon. Occurring most often during the sixth decade of life, it is characterized by epigastric pain, early satiety, and fatigue.
Clinically, primary gastric lymphoma does not differ significantly in its presentation from adenocarcinoma. Management of primary gastric lymphoma remains somewhat controversial; controlled clinical trials to evaluate different therapeutic methods, schedules, and prognostic factors have not been done.
Treatment is based on the staging of disease and may include surgery alone, surgery with radiotherapy, surgery with chemotherapy, and chemotherapy alone. Some experts recommend chemotherapy rather than surgery as the first treatment of choice for primary gastric lymphoma.9
Gastric Adenocarcinoma
Definition and Incidence.: Adenocarcinomas, malignant neoplasms arising from the gastric mucosa, constitute more than 90% of the malignant tumors of the stomach. In 2007, 21,260 new cases of stomach cancer were diagnosed in the United States and 11,210 Americans died from this disease.73
For a long time there has been a downward trend in incidence, most likely attributable to improved sanitation and drinking water that has decreased transmission of the H. pylori bacteria. However, a recent increase in cancer of the gastric cardia has been reported in the United States and European countries. The reason(s) for this are under investigation.97
Men over the age of 40 years are most likely to develop this disease, with a sharp increase in incidence after 50 years.
Etiologic and Risk Factors.: Chronic gastritis with intestinal metaplasia, possibly secondary to chronic H. pylori infection, is a strong risk factor for gastric cancer, especially when combined with family history of gastric cancer.
Other nonenvironmental risk factors include individual susceptibility; pernicious anemia, which causes atrophy of the gastric mucosa in the same locations where gastric tumors arise; type A blood; gastrectomy; gastric polyps; dietary factors such as consumption of smoked fish and meat containing benzopyrene; and nitrosamines produced endogenously in chronic gastritis.
Pathogenesis.: Development of gastric carcinoma is a multistep and multifactorial process beginning with H. pylori in most cases, but the underlying mechanisms remain to be defined. The most common site for adenocarcinoma appears to be the glands of the stomach mucosa located in the distal portion of the stomach on the lesser curvature of the prepyloric antrum (see Fig. 16-6).
Duodenal reflux and insufficient acid secretion may contribute to intestinal metaplasia. The reflux contains caustic bile salts that destroy the normally protective mucosal barrier in the stomach. Insufficient acid secretion by the atrophic mucosa creates an alkaline environment that permits bacteria to multiply and act on nitrates. The resulting increase in nitrosamines damages the DNA of mucosal cells further, promoting metaplasia and neoplasia.
Clinical Manifestations.: The clinical presentation of gastric carcinoma depends on a variety of factors, including the morphologic characteristics of the tumor (e.g., infiltrating versus ulcerating), size of the tumor, presence of gastric outlet obstruction, and metastatic versus nonmetastatic disease. Early stages of gastric cancer may be asymptomatic or present with vague symptoms of indigestion, anorexia, and weight loss similar to peptic ulcer, because ulceration can occur with gastric carcinoma.
MEDICAL MANAGEMENT
At the present time, the best advice for reducing the risk of stomach cancer is to eat at least five ½-cup servings of fruits and vegetables daily, combined with regular physical activity, maintaining a healthy weight, and reducing salt-preserved foods.86
DIAGNOSIS.
Diagnosis may be delayed by the fact that symptomatic relief can be obtained from early GI symptoms using over-the-counter medications. The choice of diagnostic tests depends on the clinical manifestation at the time of presentation. Endoscopy with cytologic brushings and biopsies of suspicious lesions are highly sensitive for detecting gastric carcinoma.
In areas of high incidence, screening upper endoscopy is performed to detect early gastric carcinoma. Once the diagnosis has been made, staging to determine the local extent of disease and the presence of nodal or distant metastases must be done. Staging is accomplished through the use of liver chemistry tests, abdominal imaging studies (e.g., CT scan), and biopsy of suspected lymph nodes.
TREATMENT.
Surgical therapy is still the treatment of choice for primary gastric adenocarcinoma. Despite many attempts, the postoperative strategies of adjuvant chemotherapy have been ineffective. Multimodality treatment consisting of preoperative chemotherapy and surgery may provide improved results if endoscopic ultrasonography and staging laparoscopy provide early identification of locally advanced tumors. Prevention through eradication of H. pylori is recommended only in individuals with high risk of cancer at this time.
PROGNOSIS.
Prognosis depends on the degree of gastric wall penetration, the presence of lymph node metastases, and the location of the primary site. Screening programs in other countries detect approximately 40% of tumors early with a 5-year survival rate of more than 60%.
Without screening, the prognosis is poor, because symptoms do not occur until the tumor has penetrated muscle layers of the stomach, spread to local tissue by direct extension in the abdominal cavity, metastasized via the lymphatic system, or created a paraneoplastic manifestation.
Paraneoplastic syndromes are discussed in greater depth in Chapter 9. When these syndromes are associated with stomach cancer, affected individuals may present with any of the following:
• Trousseau’s syndrome (spontaneous peripheral venous thrombosis of the upper and lower extremities that occurs in association with visceral carcinoma)
Acanthosis nigricans is a skin condition associated with an internal carcinoma characterized by diffuse thickening of the skin with grey, brown, or black pigmentation, usually in body folds such as the axillae (Fig. 16-7).
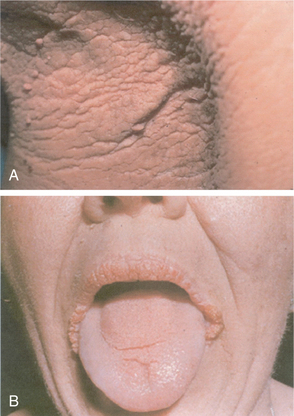
Figure 16-7 A, Paraneoplastic acanthosis nigricans with a velvety hyperpigmented rash in the axilla in an individual with gastric cancer. B, Acanthosis nigricans of the oral mucosa. This pattern of involvement is almost always associated with cancer. (From Goldman L: Cecil textbook of medicine, ed 22, Philadelphia, 2004, Saunders. Courtesy of Dr. Timothy Berger, Professor of Clinical Dermatology, University of California, San Francisco.)
Metastatic gastric carcinoma is currently incurable. Surgical resection is only possible in one third of gastric cancers. Of those people in whom surgical resection is a possibility, 20% survive 10 years.
Congenital Conditions
Definition and Overview.: Pyloric stenosis (PS) is an obstruction at the pyloric sphincter (the sphincter at the distal opening of the stomach into the duodenum). The pyloric sphincter is a ring of muscles that serve to close the opening from the stomach into the intestine (Fig. 16-8). Obstruction occurs as a congenital condition, or in adults the most common cause is ulcer disease. When present as a congenital condition, it is known as hypertrophic PS caused by hypertrophy of the sphincter and is one of the most common surgical disorders of early infancy.
Incidence and Etiologic Factors.: The cause of congenital hypertrophy of the pyloric sphincter is unknown. White males are affected more commonly than females in a 4: 1 ratio. It is more likely to occur in a full-term infant than in a premature infant, especially the first-born child. Siblings, offspring of affected persons, and fathers and sons are at increased risk of developing PS (genetic predisposition). Prophylactic administration of erythromycin to newborns exposed to neonatal pertussis has been reported to have a possible causal role in infantile PS.23,94 The role of H. pylori as a cause of infantile hypertrophic PS is under investigation.40,113,136
Increased third-trimester maternal gastric secretion associated with maternal stress-related factors increases the likelihood of PS in the infant. PS may also be associated with other congenital conditions such as Turner’s syndrome, trisomy 18, intestinal malrotation, esophageal and duodenal atresia, and anorectal anomalies.
The incidence of adult idiopathic PS is unknown, but it is considered rare. Although many physicians believe this condition is secondary to local disease, others think the condition in adults is the same entity as that observed in infants and children, but in a milder form and later in appearance.
Pathogenesis.: The histologic and anatomic abnormalities in adult PS are indistinguishable from those in the infantile form. Individual fibers of the pyloric sphincter thicken or hypertrophy, so the entire sphincter is grossly enlarged and inelastic.
Hyperplasia of the pyloric muscle occurs because of the extra peristaltic effort required to force the gastric contents through the narrow opening into the duodenum. This hypertrophy and hyperplasia form a palpable nodule severely narrowing the pyloric canal between the stomach and the duodenum, causing partial obstruction.
Over time, inflammation and edema further reduce the size of the lumen, progressing to complete obstruction preventing food from passing from the stomach to the small intestine. Progressive obstruction results in complications of malnutrition and fluid and electrolyte abnormalities.
Clinical Manifestations.: Projectile vomiting is the most common and dramatic early symptom and may occur at birth. Projectile vomiting describes forcible vomiting that ejects vomitus 1 foot or more when in a supine position and 3 to 4 feet when in an upright or side-lying position.
Overall, the age of onset and pattern of vomiting vary, but usually regurgitation or occasional projectile vomiting develops around the second to fourth week after birth. Projectile vomiting quickly leads to dehydration and lethargy with rapid progression to complete obstruction and the accompanying complications of malnutrition, weakness, wasting, weight loss, and fluid and electrolyte imbalances.
The palpable nodule is firm, movable, about the size of an olive, and felt in the right upper quadrant in approximately 80% of all infants with PS. Persistent or episodic symptoms in some adults may extend from infancy with nausea and vomiting, epigastric pain, weight loss, and anorexia most commonly present. In contrast to congenital PS in the infant, the abdominal mass that occurs in adult PS is too small to be palpable.
MEDICAL MANAGEMENT
DIAGNOSIS, TREATMENT, AND PROGNOSIS.
In infancy, diagnosis is usually by history and recognition of the clinical presentation. Ultrasound imaging is so accurate for the diagnosis of PS that it has replaced the upper GI series in most institutions.
Some infants are treated with antispasmodic drugs to relax the pylorospasm and nutritional management, including refeeding the infant after vomiting, waiting to see if the pylorus spontaneously opens by 6 to 8 months of age. Surgical repair, including endoscopic balloon dilatations, pyloromyotomy (local resection of the involved region of the pylorus), and pyloroplasty, is the standard medical treatment with a very high success rate for the infant and the adult. Placement of a stent at the site of obstruction is gaining popularity as an alternative intervention procedure.
Postoperative vomiting is not uncommon in the pediatric population, especially during the first 24 to 48 hours. Complications include persistent pyloric obstruction; partial, superficial, or total wound separation (dehiscence); or in the case of stent implantation, stent migration.
THE INTESTINES
Definition and Overview
Malabsorption syndrome is a group of disorders (celiac disease, cystic fibrosis, Crohn’s disease, chronic pancreatitis, pancreatic carcinoma, pernicious anemia, short-gut syndrome) characterized by reduced intestinal absorption of dietary components and excessive loss of nutrients in the stool.
Traditionally, malabsorption disorders have been classified as maldigestion (failure of the chemical processes of digestion) or malabsorption (failure of the intestinal mucosa to absorb the digested nutrients). Nutrients most commonly malabsorbed include fat, fatty acids or bile salts, calories (fat, protein, carbohydrates), iron, vitamin B12, folic acid, calcium, vitamin D, magnesium, potassium, vitamin K, water, and lactose (sugar in milk).
The conditions can occur separately or together simultaneously. Digestive defects include cystic fibrosis, in which pancreatic enzymes are absent; biliary or liver diseases with altered bile flow; and lactase deficiency, in which congenital or secondary lactose intolerance occurs with lactose malabsorption.
Absorptive defects may be primary, such as celiac disease, or secondary to inflammatory disease of the bowel with the concomitant accelerated bowel motility and impaired absorption (e.g., ulcerative colitis, Crohn’s disease).
Recent studies suggest that the prevalence of celiac disease, in particular, is much higher in the United States than previously thought, with rates equal to those in Europe. In fact, celiac disease may be more common than other better-known GI problems such as Crohn’s disease and ulcerative colitis. Risk is highest for individuals of European ancestry; about 70% of reported cases occur in women.51
Celiac disease is an immune-mediated enteropathic condition triggered in genetically susceptible individuals by the ingestion of gluten, a protein found in wheat, rye, barley, and oats, that prompts an inflammatory response in the small intestine.
Etiologic Factors and Pathogenesis
Clients in a therapy practice affected by malabsorption most often include people who develop gastroenteritis secondary to NSAID use, fibrosis caused by progressive systemic sclerosis or radiation injury, drug-induced malabsorption, exocrine deficiency of the pancreas caused by diabetes mellitus, and short-gut syndrome.
Short-gut syndrome (short-bowel syndrome) is the malabsorptive state that often follows extensive resection of the small intestine or, more rarely, congenital shortening of the bowel structures. Hyperabsorption of substances (e.g., vitamin D, calcium) also can occur with the intake of excessive amounts of calcium carbonate (e.g., Tums) for acid indigestion.
Generally, maldigestion is caused by deficiencies of enzymes (e.g., pancreatic lipase) and specific defects (e.g., poor digestion, lactose intolerance). Inadequate secretion of bile salts (e.g., advanced liver disease, obstruction of the common bile duct) and inadequate reabsorption of bile in the ileum also contribute to maldigestion.
In the case of inadequate absorption, food is fully digested but not adequately absorbed. This situation occurs when the absorptive surface is normal but inadequate, or adequate but not functioning normally. This problem occurs within the mucosal cell and may be highly specific owing to a gene defect. Malabsorption syndrome also can be caused by a digestive defect, a mucosal abnormality, or lymphatic obstruction and can be a generalized malabsorption or an isolated malabsorption of a particular nutrient.
In the case of celiac disease, human leukocyte antigen (HLA) appears to promote an inflammatory response to gliadin, a protein fragment of gluten. This autoimmune response attacks and destroys the small, fingerlike projections in the intestinal wall known as villi, where most nutrient absorption takes place. Impaired nutrient absorption is the outcome.
Clinical Manifestations
Early manifestations of malabsorption are progressive and not easily noticed by the person affected. Although symptoms often start in childhood, they may not appear or be recognized until much later in life. In the case of celiac disease, stress, surgery, infection, emotional crises, or some other event can precipitate the sensitivity reaction.
Weight loss, fatigue, depression, and abdominal bloating are early symptoms. A change in bowel habits may occur with production of bulky, malodorous oil–covered stools (steatorrhea) that are difficult to flush. Excessive nocturnal reabsorption of intestinal fluids may cause nocturia. A gluten-related skin disorder, dermatitis herpetiformis (Fig. 16-9), also may be present. Other gluten-induced non-GI symptoms include joint pain, fatigue, depression, and weight loss (or gain from abdominal bloating).
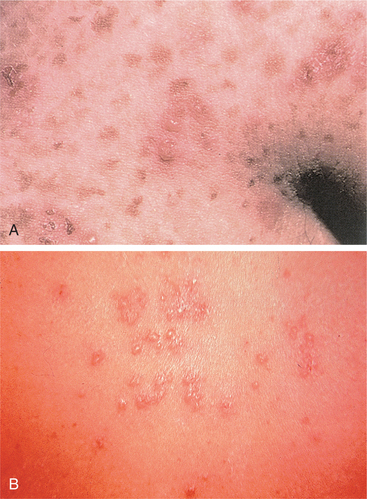
Figure 16-9 A and B, Dermatitis herpetiformis. Typical grouped pruritic papulovesicles associated with skin reaction from gluten sensitivity. This skin manifestation usually appears on the buttocks, elbows, or knees. It can affect children but is more common in adults between the ages of 30 and 40. It is almost always a sign of celiac disease and will usually resolve in 3 to 6 months with a gluten-free diet. (A, from Feldman M: Sleisenger and Fordtran’s gastrointestinal and liver disease, ed 8, Philadelphia, 2006, Saunders. Courtesy of Dr. Timothy Berger, San Francisco. B, from Noble J: Textbook of primary care medicine, ed 3, St Louis, 2001, Mosby. Courtesy James C. Shaw, MD.)
Other common signs and symptoms include explosive diarrhea, chronic diarrhea, abdominal cramps and bloating, indigestion, and flatulence. Late manifestations caused by nutritional deficiencies secondary to the malabsorption may include muscle wasting owing to diminished muscle mass; changes in bone mineral density with the development of osteoporosis secondary to impaired absorption of calcium, phosphate, and/or vitamin D; low blood pressure; infertility; and abdominal distention with active bowel sounds.
Any cause of decreased IF can result in decreased absorption of vitamin B12, resulting in pernicious anemia. Other clinical findings are dependent on the particular condition or specific nutrient involved. The therapist is most likely to see the symptoms listed in Table 16-5.
Table 16-5
Symptoms Associated with Malabsorption
| Symptoms | Malabsorbed Nutrients |
| Muscle weakness, muscle wasting, paresthesias | Generalized malnutrition; fat, protein, carbohydrates |
| Osteomalacia | Fat, protein, carbohydrates, iron, water; vitamins A, D, K |
| Tetany, paresthesias, Trousseau’s sign, Chvostek’s sign | Calcium, vitamin D, magnesium, potassium |
| Numbness and tingling; neurologic damage | Vitamin B12, vitamin B complex |
| Bone pain, fractures, skeletal deformities | Calcium, vitamin D, protein |
| Muscle spasms | Electrolyte imbalance, calcium, pregnancy |
| Easy bleeding or bruising | Vitamin K |
| Generalized swelling | Protein |
| Dermatitis herpetiformis | Gluten |
Food-induced symptoms may appear within a few minutes of ingesting foods interpreted by the body as allergens. Or it may be several hours or even days before noticeable symptoms occur. In some cases it depends on the amount consumed, the other foods eaten with it, and the way it was prepared (fresh, cooked, peeled, unpeeled).
MEDICAL MANAGEMENT
Differentiation among the various causes of malabsorption is important in determination of the specific treatment. A large number of diagnostic tests are available (e.g., blood tests for antibodies characteristic in celiac disease, such as tissue transglutaminase, antiendomysial antibodies, and immunoglobulin A; fecal fat analysis for fat malabsorption; oral tolerance tests and measurement of breath hydrogen for lactose intolerance; other breath tests to detect the presence of compounds produced by intraluminal bacteria).
Specific tests are also available to assess pancreatic insufficiency. Biopsy of the small intestinal mucosa may be necessary. Sometimes clinical trials for treatable conditions are diagnostic (e.g., gluten-free diet for celiac disease, antibiotics for bacterial overgrowth, use of over-the-counter preparations of lactase enzyme for lactose intolerance).
Anyone with a relative who has celiac disease should be screened for the condition. Screening should also be considered for anyone with a personal history of thyroid disease, type 1 diabetes, osteoporosis, liver dysfunction, or unexplained GI symptoms.
TREATMENT.
Therapy depends on the underlying condition, such as avoidance of gluten for celiac disease, exclusion of milk and dairy products for lactose intolerance,99 or replacement of pancreatic enzymes for pancreatic insufficiency (cystic fibrosis).
Probiotics, live microbial food supplements that beneficially affect the host by improving intestinal microbial balance, alleviating lactose intolerance, and enhancing immune function, may be beneficial in the treatment of malabsorption syndromes (and other GI-related conditions).26,118
Muscular twitching and tetany are treated with calcium phosphate or gluconate administered orally or intravenously. In severe cases total parenteral nutrition (TPN) may be the only treatment option, as in the case of reduced absorptive surface (e.g., short-gut syndrome). Parenteral nutrition (sometimes called hyperalimentation) is a technique for meeting a person’s nutritional needs by means of intravenous feedings, allowing bowel rest. Nutrition by intravenous feeding is administered via a central venous catheter, usually inserted into the superior vena cava, and may be TPN or supplemental. TPN also may be given peripherally for short periods of time.
PROGNOSIS.
The prognosis is good for any of these conditions if the underlying defect can be corrected. About 95% of people with celiac disease or other food-related GI dysfunction who follow a corrective (usually gluten-free) diet recover completely. Full recovery can take up to 6 months if treatment begins before permanent damage occurs; otherwise, recovery is much longer for longstanding cases. In some cases, symptoms can be managed with proper diet and probiotics but complete recovery does not occur.
A higher incidence of non-Hodgkin’s lymphoma in adult life may occur with untreated gluten-sensitive enteropathy. Anyone who develops GI symptoms while in remission on a gluten-free diet should be evaluated for cancer.137
People with celiac disease have a greater risk of developing other related immune system disorders. Genes that make a person more susceptible to celiac disease also are known to be connected with autoimmune disorders, including type 1 diabetes, systemic lupus erythematosus, Sjögren’s syndrome, scleroderma, autoimmune chronic active hepatitis, Graves’ disease, Addison’s disease, and myasthenia gravis.
There is also a possible link between celiac disease and liver dysfunction, which may explain the lack of response to a gluten-free diet that some individuals experience. Evidence of increased incidence of mild, asymptomatic hepatitis and primary biliary cirrhosis in association with celiac disease has been reported.46
Vascular Diseases
Blood is supplied to the bowel by the celiac and superior and inferior mesenteric arteries. These arteries have anastomotic intercommunications at the head of the pancreas and along the transverse bowel. Obstruction of blood flow can occur as a result of atherosclerotic occlusive lesions or embolism.
Intestinal Ischemia
Acute intestinal ischemia results from embolic occlusions of the visceral branches of the abdominal aorta, generally in people with valvular heart disease, atrial fibrillation, or left ventricular thrombus. Other causes include thrombosis of one or more visceral vessels involved with arteriosclerotic occlusive changes or nonocclusive mesenteric vascular insufficiency in people with congestive heart failure receiving recently instituted digitalis therapy.
Symptoms of acute intestinal ischemia include rapid onset of crampy or steady epigastric or periumbilical abdominal pain combined with minimal or no findings on abdominal examination and a high leukocyte count. Angiography of the superior mesenteric artery is essential to early diagnosis. If occlusion is present, laparotomy is performed to reestablish blood flow to the intestine if possible and to remove necrotic bowel if some viable bowel exists.
Atherosclerotic plaque of the superior mesenteric, celiac, and inferior mesenteric arteries causes a significant decrease in blood flow to the intestines, resulting in chronic intestinal ischemia. Symptoms of epigastric or periumbilical pains lasting for 1 to 3 hours in a person over 45 years of age who has peripheral vascular disease points to a diagnosis of intestinal ischemia. Arteriography may be performed if the person is a good candidate for surgery.
Bacterial Infections
Foodborne Illnesses (Botulism)
Overview and Incidence.: Foodborne illnesses (considered to be any illness that is related to food ingestion) and, in particular, botulism were once considered rare diseases. Today foodborne illness has become a serious public health problem. The Centers for Disease Control and Prevention reports that each year 76 million people get sick, more than 300,000 are hospitalized, and 5000 deaths occur as a result of foodborne illnesses, primarily among the very young, the older adult, and the immunocompromised.101 It is estimated that only a fraction of the people who experience GI tract symptoms from foodborne illnesses actually seek medical care.
Recent changes in human demographics, food preferences, changes in food production and distribution systems, microbial adaptation, and lack of support for public health resources have led to the emergence of novel in addition to traditional foodborne diseases. Increasing travel and trade opportunities have increased the risk of contracting and spreading foodborne illness locally, regionally, and globally.102
Etiology.: Most cases of foodborne illness or death are caused by bacterial agents such as Escherichia coli and Campylobacter, Listeria, Shigella, and Salmonella species that are linked to milk, shellfish, unpasteurized apple cider, eggs, fish, poultry, meats, and raw produce.100
Clostridium botulinum is the specific bacterium responsible for foodborne botulism, often referred to as “food poisoning.” C. botulinum spores from soil or dust can also cause wound botulism in open wounds or injured tissue. Only foodborne botulism is discussed here; see Chapter 39 for a complete discussion of the effects of botulism on the peripheral nervous system.
Pathogenesis.: Once they are ingested these neurotoxins resist gastric digestion and proteolytic enzymes and are readily absorbed into the blood from the proximal small intestine. Minute amounts of circulating toxin reach the cholinergic nerve endings at the myoneural junction and bind to the presynaptic nerve terminals. The toxin abolishes transmission at skeletal neuromuscular junctions. Inhibition of acetylcholine release from cholinergic terminals at the motor endplate results in flaccid paralysis. A predilection exists for the cranial nerves, with progression caudally and symmetrically to the trunk and the extremities.
Clinical Manifestations.: Many, but not all foodborne illnesses have GI tract symptoms. Symptoms can be from mild to severe; generally, the faster the onset of symptoms, the more severe the disease.
In addition to GI symptoms of prolonged bloody diarrhea leading to dehydration, weight loss, fever, nausea, and severe abdominal pain, early neurologic involvement includes motor weakness, paresthesias, and cranial nerve palsies with visual changes (double vision or diplopia, and intolerance of light or photophobia), ptosis, dysarthria or dysphagia, difficulty breathing, and impaired swallowing.
No sensory changes occur. Motor weakness of the face and neck muscles can progress to involve the diaphragm, accessory muscles of breathing, and extremities. Vomiting, abdominal pain, back pain, headache, and constipation may occur before or after the onset of paralysis. The usual secondary effects of flaccid motor paralysis can occur, such as severe muscle wasting, pressure ulcers, and aspiration pneumonia.
MEDICAL MANAGEMENT
DIAGNOSIS, TREATMENT, AND PROGNOSIS.
Toxin identification is made by stool or serum analysis or cultures of the organism. Electromyography is confirmatory and necessary to differentiate this condition from myasthenia gravis or Guillain-Barré syndrome. Nerve conduction velocities are normal, but action potentials are decreased with a supramaximal stimulus. Facilitation is found after repetitive stimulation at high frequency.
Selection of appropriate treatment depends on identification of the responsible pathogen if possible and determining if specific therapy is available. Immediate administration of antitoxin prevents further binding of free botulinum toxin to the presynaptic endings. Many episodes of acute gastroenteritis are self-limiting and require fluid replacement and supportive care.
Untreated food botulism can be fatal within 24 hours of toxin ingestion. Intubation and mechanical ventilation may be needed. Respiratory failure caused by rapid onset of respiratory muscle weakness is a complication that is often fatal.
Inflammatory Bowel Disease
Inflammatory bowel disease (IBD) refers to two inflammatory conditions: Crohn’s disease and ulcerative colitis (Table 16-6). Crohn’s disease (CD) is a chronic lifelong inflammatory disorder that can affect any segment of the intestinal tract (intestine or ileum, large intestine or colon) and even tissues in other organs. It can affect all layers of the intestine and is characterized by diseased areas of intestine with normal intestine between (“skip” areas) with periods of exacerbation and remission. CD is also referred to as regional enteritis, regional ileitis, terminal ileitis, or granulomatous colitis, depending on the location of the inflammation.
Table 16-6
Comparison of the Characteristics of Crohn’s Disease and Ulcerative Colitis
| Characteristics | Crohn’s Disease | Ulcerative Colitis |
| Incidence | ||
| Age at onset | Any age; 10-30 yr most common | Any age; 10-40 yr most common |
| Family history | 20% to 25% | 20% |
| Gender (prevalence) | Equal in women and men | Equal in women and men |
| Cancer risk | Increased; early detection best means of prevention | Increased; preventable with bowel resection |
| Pathogenesis | ||
| Location of lesions | Any segment; usually small or large intestine | Large intestine; rectum |
| Skip lesions | Common | Absent |
| Inflammation and ulceration | Entire intestinal wall involved | Mucosal/submucosal layers involved |
| Granulomas | Typical | Uncommon |
| Thickened bowel wall | Typical | Uncommon |
| Narrowed lumen and obstruction | Typical | Uncommon |
| Fissures and fistulas | Common | Absent |
| Clinical Manifestations | ||
| Abdominal pain | Mild to severe; common | Mild to severe; less frequent |
| Diarrhea | May be absent; moderate | Typical; often severe; chronic |
| Bloody stools | Uncommon | Typical |
| Abdominal mass | Common; right lower quadrant | Uncommon |
| Anorexia | Can be severe | Mild or moderate |
| Weight loss | Can be severe | Mild to moderate |
| Skin rashes | Common, mild | Common, mild |
| Joint pain | Common, mild to moderate | Common, mild to moderate |
| Growth retardation (pediatric) | Often marked | Usually mild |
| Clinical course | Remissions and exacerbations | Remissions and exacerbations |
Ulcerative colitis (UC) is a chronic inflammatory disorder of the mucosa and submucosa of the colon in a continuous manner without skips. It is characterized by chronic diarrhea and rectal bleeding with ulceration. It may be referred to as ulcerative proctitis (involving the rectum only) or pancolitis (involving the entire colon).
Incidence
Both CD and UC are bowel disorders of unknown cause involving genetic and immunologic influences on the GI tract’s ability to distinguish foreign antigens from self-antigens. These two conditions can occur in all age groups and affect both genders equally; incidence peaks between the ages of 15 and 35, but CD does occur in later decades, usually after age 50. An estimated 1 million Americans are affected by IBD, with equal distribution between these two conditions.37