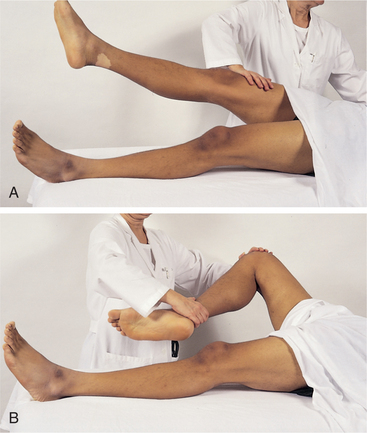
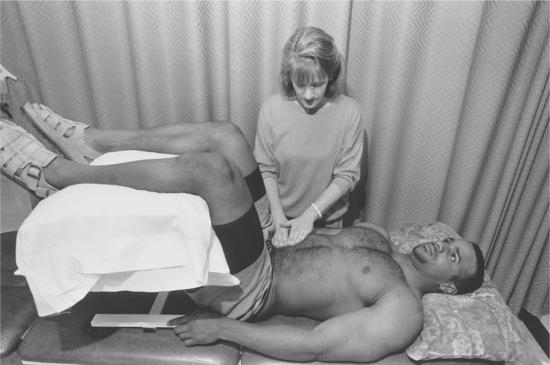
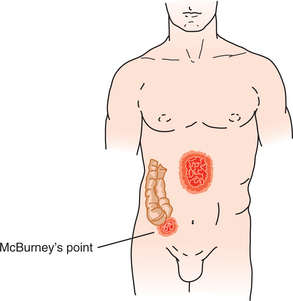
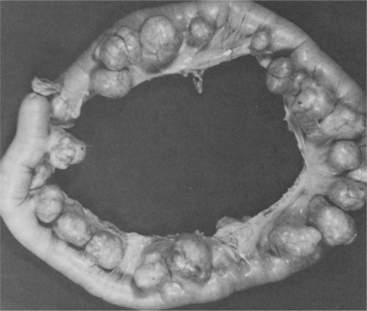
1. Adams, BD. Pinch-an-inch test for appendicitis. South Med J. 2005;98(12):1207–1209.
2. Agency for Health Care Policy and Research (AHCPR) Colorectal cancer screening: summary, 2001. Available on-line at http://www.ahcpr.gov Accessed April 26, 2008.
3. Ahuja, A, Brent, L. Revisting the spondyloarthropathies: a new era of treatment. J Musculoskelet Med. 2006;23(9):654–666.
4. Aldoori, WH, Giovannucci, EL, Rimm, EB, et al. Use of acetaminophen and nonsteroidal antiinflammatory drugs: a prospective study and the risk of symptomatic diverticular disease in men. Arch Fam Med. 1998;7(3):262–263.
5. Ali, A, Toner, BB, Stuckless, N, et al. Emotional abuse, self-blame, and self-silencing in women with irritable bowel syndrome. Psychosom Med. 2000;62(1):76–82.
6. American Cancer Society (ACS) Statistics for 2007: cancer facts and figures for African Americans 2007-2008. Available on-line at www.cancer.org Accessed May 28, 2007.
7. American Physical Therapy Association (APTA). Guidelines for recognizing and providing care for victims of domestic violence. Alexandria, VA: APTA, 1997. [No. P-138].
8. Anderson, M, Robinson, M. Watching for-and managing-joint problems in inflammatory bowel disease. J Musculoskelet Med. 1996;13(11):28–34.
9. Aviles, A. The role of surgery in primary gastric lymphoma: results of a controlled clinical trial. Ann Surg. 2004;240(1):44–50.
10. Aviles, A. Surgery and chemotherapy versus chemotherapy as treatment of high-grade MALT gastric lymphoma. Med Oncol. 2006;23(2):295–300.
11. Befus, AD, Mathison, R, Davison, J. Integration of neuro-endocrine immune responses in defense of mucosal surfaces. Am J Trop Med Hyg. 1999;60(4):26–34.
12. Best, WR. Development of a Crohn’s disease activity index. Gastroenterology. 1976;70(3):439–444.
13. Blomhoff, S, Spetalen, S, Jacobsen, MB, et al. Intestinal reactivity to words with emotional content and brain information processing in irritable bowel syndrome. Dig Dis Sci. 2000;45(6):1160–1165.
14. Blomhoff, S, Spetalen, S, Jacobsen, MB, et al. Rectal tone and brain information processing in irritable bowel syndrome. Dig Dis Sci. 2000;45(6):1153–1159.
15. Breusch, SJ. Acute pseudo-obstruction of the colon following left-sided total hip replacement. Int J Clin Pract. 1997;51(5):327–329.
16. Brown, C. Electrical stimulation for fecal incontinence following bowel resection. Rehab Oncol. 2001;19(3):20.
17. Brown, LM. Helicobacter pylori: epidemiology and routes of transmission. Epidemiol Rev. 2000;22(2):283–297.
18. Burdick, JS. Esophageal cancer prevention, cure, and palliation. Semin Gastrointest Dis. 2000;11(3):124–133.
19. Camilleri, M, Lee, JS, Viramontes, B, et al. Insights into the pathophysiology and mechanisms of constipation, irritable bowel syndrome, and diverticulosis in older people. J Am Geriatr Soc. 2000;48(9):1142–1150.
20. Camilleri, M, Spiller, R. Irritable bowel syndrome: diagnosis and treatment. Edinburgh: Saunders, 2002.
21. Campbell-Thompson, M, Lynch, IJ, Bhardwaj, B. Expression of estrogen (ER) subtypes and ERbeta isoforms in colon cancer. Cancer Res. 2001;61(2):632–640.
22. Cassara, JE, Shaheen, NJ. Endoscopic anti-reflux devices: a year of challenges and change. Curr Opin Gastroenterol. 2006;22(4):423–428.
23. Centers for Disease Control and Prevention (CDC). Hypertrophic pyloric stenosis in infants following pertussis prophylaxis with erythromycin. JAMA. 2000;283(4):471–472.
24. Chao, A. Meat consumption and risk of colorectal cancer. JAMA. 2005;293(2):233–234.
25. Cheng, Y, Macera, CA, Davis, DR, et al. Does physical activity reduce the risk of developing peptic ulcers? Br J Sports Med. 2000;34(2):116–121.
26. Chermesh, I, Eliakim, R. Probiotics and gastrointestinal tract: where are we in 2005? World J Gastroenterol. 2006;12(6):853–857.
27. Christensen, J. Hypothesis: how might oesophagitis cause hiatus hernia? Neurogastroenterol Motil. 2003;15(5):567–569.
28. Christensen, J, Miftakhov, R. Hiatus hernia: a review of evidence for its origin in esophageal longitudinal muscle dysfunction. Am J Med. 2000;108(suppl 4a):3S–7S.
29. Clark, TJ, McKenna, LS, Jewell, MJ. Physical therapists’ recognition of battered women in clinical settings. Phys Ther. 1996;76(1):12–19.
30. Clarke, HD. Acute pseudo-obstruction of the colon as a postoperative complication of hip arthroplasty. J Bone Joint Surg Am. 1997;79:1642–1647.
31. Colbert, LH, Hartman, TJ, Malila, N, et al. Physical activity in relation to cancer of the colon and rectum in a cohort of male smokers. Cancer Epidemiol Biomarkers Prev. 2001;10(3):265–268.
32. Cole, RP. Functional recovery in cancer rehabilitation. Arch Phys Med Rehabil. 2000;81(5):623–627.
33. Compton, CC, Greene, FL. The staging of colorectal cancer: 2004 and beyond. CA Cancer J Clin. 2004;54(6):295–308.
34. Cremonini, F. Irritable bowel syndrome: epidemiology, natural history, health care seeking and emerging risk factors. Gastroenterol Clin North Am. 2005;34(2):189–204.
35. Crew, KD. Epidemiology of upper gastrointestinal malignancies. Semin Oncol. 2004;31:450–464.
36. Crocker, JA, Gudas, SA. Rehabilitation referral patterns in colorectal carcinoma. Rehab Oncol. 2005;23(3):17–21.
37. Crohn’s and Colitis Foundation of America (CCFA) Questions and answers about Crohn’s disease and ulcerative colitis, 2001. Available on-line at http://www.ccfa.org Accessed April 26, 2008.
38. Crowell, MD. The role of serotonin in the pathophysiology of irritable bowel syndrome. Am J Manag Care. 2001;7(suppl 8):S252–S260.
39. Cunningham-Rundles, S, Lin, DH. Nutrition and the immune system of the gut. Nutrition. 1998;14(7-8):573–579.
40. Dahshan, A. Helicobacter pylori and infantile hypertrophic pyloric stenosis: is there a possible relationship? J Pediatr Gastroenterol Nutr. 2006;42(3):262–264.
41. Danese, S. Etiopathogenesis of inflammatory bowel disease. World J Gastroenterol. 2006;12(30):4807–4812.
42. de la Chapelle, A. Genetic predisposition to colorectal cancer. Nat Rev Cancer. 2004;4(10):769–780.
43. De Lillo, AR, Rose, S. Functional bowel disorders in the geriatric patient: constipation, fecal impaction, and fecal incontinence. Am J Gastroenterol. 2000;95(4):901–905.
44. Dean, E. Oxygen transport deficits in systemic diseases and implications for physical therapy. Phys Ther. 1997;77(2):187–202.
45. Duffy, MJ. Carcinoembryonic antigen as a marker for colorectal cancer: is it clinically useful? Clin Chem. 2001;47(4):624–630.
46. Duggan, JM. Systematic review: the liver in coeliac disease. Aliment Pharmacol Ther. 2005;21(5):515–518.
47. Easterling, C. Attaining and maintaining isometric and isokinetic goals of the Shaker exercise. Dysphagia. 2005;20(2):133–138.
48. Elliott, DE. Helminths as governors of immune-mediated inflammation. Int J Parasitol. 2007;37(5):457–464.
49. elMaraghy, AW. Ogilvie’s syndrome after lower extremity arthroplasty. Can J Surg. 1999;42(2):133–137.
50. Falk, GW. Barrett’s esophagus. Gastroenterology. 2002;122:1569–1591.
51. Fasano, A. Prevalence of celiac disease in at-risk and not-at-risk groups in the United States: a large multicenter study. Arch Intern Med. 2003;163(3):286–292.
52. Feldman, M, Peptic ulcer disease. ACP Medicine. New York: WebMD; 2004. Available on-line at www.medscape.com/viewarticle/494030 Accessed May 25, 2007.
53. Ferdjallah, M, Wertsch, JJ, Shaker, R. Spectral analysis of surface electromyography (EMG) of upper esophageal sphincter-opening muscles during head lift exercise. J Rehabil Res Dev. 2000;37(3):335–340.
54. Fitzgibbons, RJ, Jr. Watchful waiting vs. repair of inguinal hernia in minimally symptomatic men: a randomized clinical trial. JAMA. 2006;295(3):285–292.
55. Food and Drug Administration (FDA). FDA approves drugs for colorectal, lung cancer. FDA Consum. 2007;41(1):5.
56. Gaspar, LE, Winter, K, Kocha, WI, et al. A phase I/II study of external beam radiation, brachytherapy, and concurrent chemotherapy for patients with localized carcinoma of the esophagus (Radiation Therapy Oncology Study Group 9207): final report. Cancer. 2000;88(5):988–995.
57. Gelbmann, CM. Prediction of treatment refractoriness in ulcerative colitis and Crohn’s disease-do we have reliable markers? Inflamm Bowel Dis. 2000;6(2):123–131.
58. Gersh, MR. Physical therapy implications for clients with colorectal cancer. Rehab Oncol. 2004;22(3):15–22.
59. Gershon, MD. The second brain: the scientific basis of gut instinct and a groundbreaking new understanding of nervous disorders of the stomach and small intestine. New York: HarperCollins, 1998.
60. Goodman, CC, Snyder, TE. Differential diagnosis in physical therapy, ed 3. Philadelphia: Saunders, 2000.
61. Goodwin, RD, Stein, MB. Generalized anxiety disorder and peptic ulcer disease among adults in the United States. Psychosom Med. 2002;64(6):862–866.
62. Gow, PJ, Chapman, RW. Modern management of oesophageal varices. Postgrad Med J. 2001;77(904):75–81.
63. Green, JA, Amaro, R, Barkin, JS. Symptomatic gastroesophageal reflux as a risk factor for esophageal adenocarcinoma. Dig Dis Sci. 2000;45(12):2367–2368.
64. Harrington, KL, Haskvitz, EM. Managing a patient’s constipation with physical therapy. Phys Ther. 2006;86(11):1511–1519.
65. Hawk, E, Lubet, R, Limburg, P. Chemoprevention in hereditary colorectal cancer syndromes. Cancer. 1999;86(11 suppl):2551–2563.
66. Hegab, AM, Luketic, VA. Bleeding esophageal varices: how to treat this dreaded complication of portal hypertension. Postgrad Med. 2001;109(2):75–76. [81-86, 89].
67. Heitkemper, M. Overlapping conditions in women with irritable bowel syndrome. Urol Nurs. 2005;25(1):25–31.
68. Higgins, PDR. Epidemiology of constipation in North America: a systematic review. Am J Gastroenterol. 2004;99:750–759.
69. Hong, JJ. A prospective randomized study of clinical assessment versus computed tomography for the diagnosis of acute appendicitis. Surg Infect (Larchmt). 2003;4(3):231–239.
70. Humes, DJ. Acute appendicitis. BMJ. 2006;333(7567):530–534.
71. Inadomi, JM. Screening and surveillance for Barrett esophagus in high-risk groups: a cost-utility analysis. Ann Intern Med. 2003;138:176–186.
72. Iqbal, A. Endoscopic therapies of gastroesophageal reflux disease. World J Gastroenterol. 2006;12(17):2641–2655.
73. Jemal, A. Cancer statistics, 2007. CA Cancer J Clin. 2007;57(1):43–66.
74. Johns Hopkins Medical Letter. Do you need a B12 boost? Johns Hopkins Med Lett. 1998;10(4):6.
75. Johnson, C. Handling the hurt: physical therapy and domestic violence. Phys Ther. 1997;5(1):52–64.
76. Kang, W, Kudsk, KA. Is there evidence that the gut contributes to mucosal immunity in humans? JPEN J Parenter Enteral Nutr. 2007;31(3):246–258.
77. Kauffman, D. Postoperative outcomes for patients with colon cancer: laparoscopy-assisted vs. open colostomy. Rehab Oncol. 2003;21(2):18–20.
78. Kemp, S, Batt, ME, The “sports hernia”: a common cause of groin pain. Phys Sports Med. 1998;26(1). Available on-line at http://www.physsportsmed.com/issues/1998/01jan/batt.htm. Accessed May 21, 2007.
79. Khoury, RM, Camacho-Lobato, L, Katz, PO, et al. Influence of spontaneous sleep positions on nighttime recumbent reflux in patients with gastroesophageal reflux disease. Am J Gastroenterol. 1999;94(8):2069–2073.
80. Kluin, J. Endoscopic evaluation and treatment of groin pain in the athlete. Am J Sports Med. 2004;32(4):944–949.
81. Koffler, KH. Strength training accelerates gastrointestinal transit in middle-aged and older men. Med Sci Sports Exerc. 1992;24:415–419.
82. Koloski, NA. Predictors of health care seeking for irritable bowel syndrome: a critical review of the literature on symptom and psychosocial factors. Am J Gastroenterol. 2001;96(5):1340–1349.
83. Kountouras, J, Boura, P, Lygidakis, NJ. New concepts of molecular biology for colon carcinogenesis. Hepatogastroenterology. 2000;35:1291–1297.
84. Krok, KL. Colorectal cancer in inflammatory bowel disease. Curr Opin Gastroenterol. 2004;20(1):43–48.
85. Kumar, A. Results of inguinal canal repair in athletes with sports hernia. J R Coll Surg Edinb. 2002;47(3):561–565.
86. Kushi, LH. American Cancer Society guidelines on nutrition and physical activity for cancer prevention: reducing the risk of cancer with healthy food choices and physical activity. CA Cancer J Clin. 2006;56(5):254–281.
87. Labianca, R. Development and clinical indications of cetuximab. Int J Biol Markers. 2007;22(suppl 4):S40–46.
88. Lanza, E. High dry bean intake and reduced risk of advanced colorectal adenoma recurrence among participants in the polyp prevention trial. J Nutr. 2006;136(7):1896–1903.
89. Lembo, A. Current concepts: chronic constipation. N Engl J Med. 2003;349:1360–1368.
90. Lin, OS. Screening colonoscopy in very elderly patients: prevalence of neoplasia and estimated impact on life-expectancy. JAMA. 2006;295(20):2357–2365.
91. Lucak, S, Diagnosis irritable bowel syndrome. MedGenMed. 2004;6(1). Available on-line at www.medscape.com/viewarticle/465760 Accessed May 26, 2007.
92. Lynch, PM. COX-2 inhibition in clinical cancer prevention. Oncology. 2001;15(3 suppl 5):21–26.
93. MacDonald, TT. Immunopathogenesis of Crohn’s disease. J Parenter Enteral Nutr. 2005;29(4 suppl):S118–S124.
94. Maheshwai, N. Are young infants treated with erythromycin at risk for developing hypertrophic pyloric stenosis? Arch Dis Child. 2007;92(3):271–273.
95. Marks, DJB. Defective acute inflammation in Crohn’s disease: a clinical investigation. Lancet. 2006;367(9511):668–678.
96. Marshall, JL. Bevacizumab in the treatment of colorectal cancer. Clin Adv Hematol Oncol. 2007;5(1 suppl 1):8–9.
97. Mayne, ST. Diet, obesity, and reflux in the etiology of adenocarcinomas of the esophagus and gastric cardia in humans. J Nutr. 2002;132:3467S–3470S.
98. Meshkinpour, H. Effects of regular exercise in management of chronic idiopathic constipation. Dig Dis Sci. 1998;43:2379–2383.
99. Montalto, M. Management and treatment of lactose malabsorption. World J Gastroenterol. 2006;12(2):187–191.
100. Morbidity and Mortality Weekly Report (MMWR). Diagnosis and management of foodborne illnesses. MMWR Recomm Rep. 2001;50(RR-2):1–69.
101. Morbidity and Mortality Weekly Review (MMWR). Diagnosis and management of foodborne illnesses: a primer for physicians and other health care professionals. MMWR Recomm Rep. 2004;53(RR-4):1–33.
102. Morbidity and Mortality Weekly Report (MMWR), Preliminary FoodNet data on the incidence of foodborne illnesses-selected sites, United States, 2000. MMWR Morb Mortal Wkly Rep. 2001;50(13):241–246. Available on-line at http://www.cdc.gov/mmwr/preview/mmwrhtml/rr5304a1.htm Accessed May 22, 2007.
103. Moreels, TG, Pelckmans, PA. Gastrointestinal parasites: potential for refractory inflammatory bowel diseases. Inflamm Bowel Dis. 2005;11(2):178–184.
104. Morgner, A, Bayerdorffer, E, Neubauer, A, et al. Gastric MALT lymphoma and its relationship to Helicobacter pylori infection: management and pathogenesis of the disease. Microsc Res Tech. 2000;48(6):349–356.
105. Mort, JR. Interaction between selective serotonin reuptake inhibitors and nonsteroidal antiinflammatory drugs: review of the literature. Pharmacotherapy. 2006;26(9):1307–1313.
106. Murphy, TV, Gargiullo, PM, Massoudi, MS, et al. Intussusception among infants given an oral rotavirus vaccine. N Engl J Med. 2001;344(8):564–572.
107. Nagura, H, Ohtani, H, Sasano, H, et al. The immuno-inflammatory mechanism for tissue injury in inflammatory bowel disease and Helicobacter pylori-infected chronic active gastritis: roles of the mucosal immune system. Digestion. 2001;63(suppl 1):12–21.
108. Nakatsuchit, T. The necessity of chest physical therapy for thorascopic esophagectomy. J Int Med Res. 2005;33(4):434–441.
109. Nelson, JD. Acute colonic pseudo-obstruction (Ogilvie syndrome) after arthroplasty in the lower extremity. J Bone Joint Surg Am. 2006;88(3):604–610.
110. Oliveria, SA, Christos, PJ, Talley, NJ, et al. Heartburn risk factors, knowledge, and prevention strategies: a population-based survey of individuals with heartburn. Arch Intern Med. 1999;159(14):1592–1598.
111. Overmier, JB, Murison, R. Anxiety and helplessness in the face of stress predisposes, precipitates, and sustains gastric ulceration. Behav Brain Res. 2000;110(1-2):161–174.
112. Pahor, M, Guralnik, JM, Salive, ME, et al. Physical activity and risk of severe gastrointestinal hemorrhage in older persons. JAMA. 1994;272:595–599.
113. Paulozzi, LJ. Is Helicobacter pylori a cause of infantile hypertrophic pyloric stenosis? Med Hypotheses. 2000;55(2):119–125.
114. Pickhardt, PJ. CT colonography (virtual colonoscopy): a practical approach for population screening. Radiol Clin North Am. 2007;45(2):361–375.
115. Pilotto, A. Recent advances in the treatment of GERD in the elderly: focus on proton pump inhibitors. Int J Clin Pract. 2005;59(10):1204–1209.
116. Pothoulakis, C, Lamont, JT. Microbes and microbial toxins: paradigms for microbial-mucosal interactions: II. The integrated response of the intestine to Clostridium difficile toxins. Am J Physiol Gastrointest Liver Physiol. 2001;280(2):G178–G183.
117. Potosky, AL. Age, sex, and racial differences in the use of standard adjuvant therapy for colorectal cancer. J Clin Oncol. 2002;20:1192–1202.
118. Quigley, EM, Quera, R. Small intestinal bacterial overgrowth: roles of antibiotics, prebiotics, and probiotics. Gastroenterology. 2006;130(2 suppl 1):S78–S90.
119. Reddy, A, Fried, B. The use of Trichuris suis and other helminth therapies to treat Crohn’s disease. Parasitol Res. 2007;100(5):921–927.
120. Reilly, J, Baker, GA, Rhodes, J, et al. The association of sexual and physical abuse with somatization: characteristics of patients presenting with irritable bowel syndrome and non-epileptic attack disorder. Psychol Med. 1999;29(2):399–406.
121. Reis, LAG. The annual report to the nation on the status of cancer with a special section on colorectal cancer. Cancer. 2000;88:2398–2424.
122. Romano, M, Cuomo, A, Eradication of Helicobacter pylori: a clinical update. MedGenMed. 2004;6(1). Available on-line at http://www.medscape.com/viewarticle/468006. Accessed May 26, 2007.
123. Rose, SJ, Rothstein, JM. Muscle mutability: general concepts and adaptations to altered patterns of use. Phys Ther. 1982;62:1773.
124. Rubin, DT, Dachman, AH. Virtual colonoscopy: a novel imaging modality for colorectal cancer. Curr Oncol Rep. 2001;3(2):88–93.
125. Saito, YA. The epidemiology of irritable bowel syndrome in North America: a systematic review. Am J Gastroenterol. 2002;97(8):1910–1915.
126. Sandler, RS, Halabi, S, Kaplan, EB, et al. Use of vitamins, minerals, and nutritional supplements by participants in a chemoprevention trial. Cancer. 2001;91(5):1040–1045.
127. Sartor, RB. Mechanisms of disease: pathogenesis of Crohn’s disease and ulcerative colitis. Nat Clin Pract Gastroenterol Hepatol. 2006;3(7):390–407.
128. Schatzkin, A. Dietary fiber and whole-grain consumption in relation to colorectal cancer in the NIH-AARP Diet and Health Study. Am J Clin Nutr. 2007;85(5):1353–1360.
129. Schatzkin, A, Lanza, E, Corle, D, et al. Lack of effect of a low-fat, high-fiber diet on the recurrence of colorectal adenomas, Polyp Prevention Trial Study Group. N Eng J Med. 2000;342(16):1149–1155.
130. Schoon, EJ, Blok, BM, Geerling, BJ, et al. Bone mineral density in patients with recently diagnosed inflammatory bowel disease. Gastroenterology. 2000;119(5):1203–1208.
131. Schoon, EJ, Muller, MC, Vermeer, C, et al. Low serum and bone vitamin K status in patients with longstanding Crohn’s disease: another pathogenetic factor of osteoporosis in Crohn’s disease? Gut. 2001;48(4):448.
132. Schulte, CM, Dignass, AU, Goebell, H, et al. Genetic factors determine extent of bone loss in inflammatory bowel disease. Gastroenterology. 2000;119(4):909–920.
133. Semrad, CE. Bone mass and gastrointestinal disease. Ann N Y Acad Sci. 2000;904:564–570.
134. Serebruany, VL. Selective serotonin reuptake inhibitors and increased bleeding risk. Am J Med. 2006;119(2):113–116.
135. Shanahan, F. Inflammatory bowel disease: immunodiagnostics, immunotherapeutics, and ecotherapeutics. Gastroenterology. 2001;120(3):622–635.
136. Sherwood, W. Infantile hypertrophic pyloric stenosis: an infectious cause? Pediatr Surg Int. 2007;23(1):61–63.
137. Silano, M. Delayed diagnosis of coeliac disease increases cancer risk. BMC Gastroenterol. 2007;7:8.
138. Slattery, ML. Physical activity and colon cancer: a public health perspective. Ann Epidemiol. 1997;7:137–145.
139. Smith, RA. American Cancer Society guidelines for the early detection of cancer, 2006. CA Cancer J Clin. 2006;56(1):11–15.
140. Souza, RF, Spechler, SJ. Concepts in the prevention of adenocarcinoma of the distal esophagus and proximal stomach. CA Cancer J Clin. 2005;55(6):334–351.
141. Spinzi, G, Belloni, G, Martegani, A, et al. Computed tomographic colonography and conventional colonoscopy for colon diseases: a prospective blinded study. Am J Gastroenterol. 2001;96(2):394–400.
142. Stephen, AE. Shortened length of stay and hospital cost reduction with implementation of an accelerated clinical care pathway after elective colon resection. Surgery. 2003;133:277–282.
143. Storm-Dickerson, TL. What have we learned over the past 20 years about appendicitis in the elderly? Am J Surg. 2003;185(3):198–201.
144. LStrate, Can diverticular disease patients eat nuts, corn, and popcorn? Presentation at Digestive Diseases Week Annual Meeting, Washington, DC, May 22, 2007.
145. Streitz, JM, Jr. Endoscopic surveillance of Barrett’s esophagus: does it help? J Thorac Cardiovasc Surg. 1993;105:383–387.
146. Stroupe, KT. Tension-free repair versus watchful waiting for men with asymptomatic or minimally symptomatic inguinal hernias. J Am Coll Surg. 2006;203(4):458–468.
147. Sutton, P. Helicobacter pylori vaccines and mechanisms of effective immunity: is mucus the key? Immunol Cell Biol. 2001;79(1):67–73.
148. Swan, KG, Wolcott, M. The athletic hernia. Clin Orthop Rel Res. 2006;455:78–87.
149. Tam, WC. Impact of endoscopic suturing of the gastroesophageal junction on lower esophageal sphincter function and gastroesophageal reflux in patients with reflux disease. Am J Gastroenterol. 2004;99(2):195–202.
150. Vainio, H, Bianchini, F. Prevention of disease with pharmaceuticals. Pharmacol Toxicol. 2001;88(3):111–118.
151. Van Veen, RN. Successful endoscopic treatment of chronic groin pain in athletes. Surg Endosc. 2007;21(2):189–193.
152. Vanagunas, A. Managing gastrointestinal problems in athletes. J Musculoskel Med. 1999;16(7):405–415.
153. Ward, EM. Barrett’s esophagus is common in older men and women undergoing screening colonoscopy regardless of reflux symptoms. Am J Gastroenterol. 2006;101(1):12–17.
154. Weinryb, RM. Psychological factors in irritable bowel syndrome: a population-based study of patients, non-patients and controls. Scand J Gastroenterol. 2003;38(5):503–510.
155. Weinstock, JV. Helminths and mucosal immune modulation. Ann N Y Acad Sci. 2006;1072:356–364.
156. Wessinger, S. Increased use of selective serotonin uptake inhibitors in patients admitted with gastrointestinal hemorrhage: a multicenter retrospective analysis. Aliment Pharmacol Ther. 2006;23(7):937–944.
157. Whitehead, WE. Systematic review of the comorbidity of irritable bowel syndrome with other disorders: what are the causes and implications? Gastroenterology. 2002;122(4):1140–1156.
158. Wijnhoven, BP, Tilanus, HW, Dinjens, WN. Molecular biology and Barrett’s adenocarcinoma. Ann Surg. 2001;233(3):322–337.
159. Wilson, S. Systematic review: the effectiveness of hypnotherapy in the management of irritable bowel syndrome. Aliment Pharmacol Ther. 2006;24(5):769–780.
159a. Wolpin, BM. Adjuvant treatment of colorectal cancer. CA Cancer J Clin. 2007;57(3):168–185.
160. Yang, YX, Lichtenstein, GR. Methotrexate for the maintenance of remission in Crohn’s disease. Gastroenterology. 2001;120(6):1553–1555.
161. Yuan, Y. Peptic ulcer disease today. Nat Clin Pract Gastroenterol Hepatol. 2006;3(2):80–89.