Headache
OVERVIEW
Headache is a common complaint in the history of clients receiving therapy. It has been determined in one study that only 4% of the total population has never experienced a headache.79 Headache can be the major complaint that brings the client to therapy but is often a comorbidity that can limit participation in therapy. Individuals with chronic headaches report lifestyle changes to varying degrees. The ability to perform daily tasks is limited by feelings of fatigue, the need to stop activity in order to limit external impact such as light and sound, and the inability to concentrate on task at hand. Engaging in leisure activity is also decreased during headache periods.14 Days of lost work have been carefully studied and compared to that in individuals who do not have chronic headaches and have been noted to be on average almost 6 days/yr for an individual with significant headache such as migraine. Tools designed to measure the impact of headache are the Migraine Disability Assessment (MIDAS) and the Headache Impact Test (HIT). The HIT is an Internet-based scale and can be found at www.headachetest.com. A shorter paper version is also available. These tools help to define aspects of the headache to increase communication to practitioners, describe severity, and record change over time as an outcome measure.9
Disability related to headache has been studied using the SF-36 Health Status Profile, with lower scores representing more disability than in the normal population. Scores are also lower than the population with osteoarthritis. Scores are particularly low in the physical and emotional areas, with low vitality scores, and follow the pattern of the individuals with depression. Depression occurs in 30% of individuals with headache and is strongly associated with being on disability or welfare, unemployment, and age under 50 years.45
The International Headache Society (IHS) classification system is the standard system used for both clinical and research purposes. Box 37-1 shows the major categories as defined by the IHS. According to this classification, primary chronic headaches are episodic in nature with headache-free intervals. Primary headaches are not caused by other diseases. Examples of primary headaches are migraine headaches, tension-type headaches, and cluster headaches. Fig. 37-1 illustrates the pain patterns associated with typical complaints of headache and head pain.15
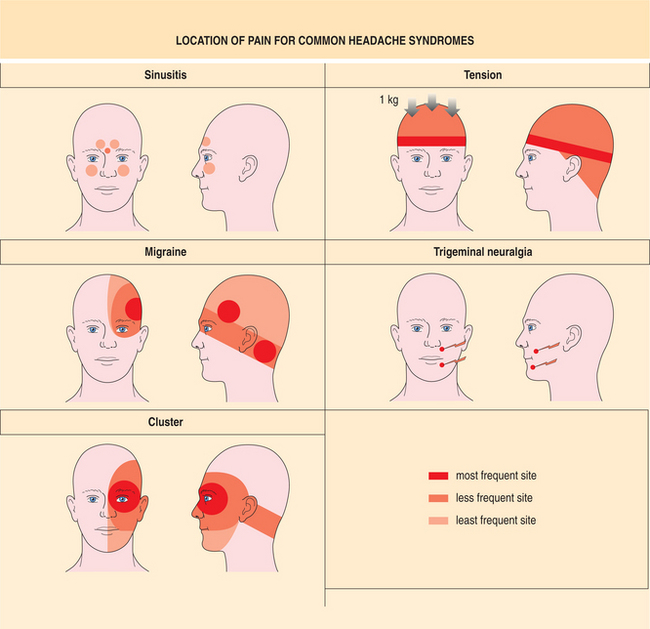
Figure 37-1 Typical headache patterns. Primary headaches include tension-type, migraine, and cluster. Sinus headache, related to infection, is a common secondary headache. Trigeminal neuralgia is an example of cranial neuralgia. (See Chapter 40). (From Weinstein JM: Headache and facial pain. In Yanoff M, Duker JS, Augsburger JJ, et al, eds: Ophthalmology, ed 2, St Louis, 2004, Mosby.)
The IHS classification is based on a descriptive system that categorizes primary headaches based on their symptom profiles. Alternatives for classification have been proposed that would consider the primary headaches as a continuum. For example, tension-type headaches can also have some of the symptoms ascribed to migraine when the headache is severe, and these headaches are often described as borderline migraine. Individuals with chronic migraine often also report headaches that are tension-type, and these have been called interval headaches. These headaches are not as severe in nature as the migraine. On the other hand, excessive muscle contraction does not play a critical role in tension-type headache, whereas muscle tenderness and pain are prominent in migraine. If the continuum concept is valid, the tension-type headache may be at one end of the distribution of severity and migraine headache may be at the other end. Many people have described “mixed” headache disorders in which tension headaches, posttraumatic headaches, or secondary headaches appear to trigger migraine headaches.74
Secondary headaches are caused by associated disease and are categorized by etiology. Secondary headaches have diverse causes, ranging from serious and lifethreatening conditions such as brain tumors, strokes, meningitis, and subarachnoid hemorrhages to less serious but common conditions such as withdrawal from caffeine and discontinuation of analgesics. Secondary headaches can be associated with systemic illness, fever, and increased blood pressure. Although headache with hypertension is rare except with grossly high blood pressure, episodic hypertension can give rise to throbbing headache in the occipital region. Sinusitis and other general medical disorders can have headache as a symptom. Headaches caused by illness resolve when the underlying cause is successfully treated.2
Posttraumatic headache, another common but less specific headache, begins after flexion-extension injuries and is associated with brain injury. This headache may be localized to the site of injury, or it may be generalized with varying intensity. Common complaints in addition to head pain are irritability, lack of concentration, and memory loss. These symptoms are consistent with both chronic headache and mild or moderate head injury. When dizziness is associated, it can further restrict activity. Posttraumatic headache is difficult to treat because of its multifactorial cause of onset and the mechanical damage associated with the trauma.
A sudden onset of severe headache is usually related to an intracranial disorder such as subarachnoid hemorrhage, brain tumor, or meningitis (see Chapter 29). These headaches will most often be accompanied by other neurologic signs, such as weakness, visual disturbances, and possibly altered mental status or coma. A structural, or space-occupying, lesion is suspected in headaches that disrupt sleep, are triggered by exertion, or cause excessive drowsiness. Headaches secondary to brain tumors, endocrinopathies, and other medical problems are only a very small percentage of all headaches.44 Even though pathologic conditions account for only a few cases of headache pain, it is critical for the therapist to understand what mechanisms may be responsible and when the client may need medical or emergent care. Table 37-1 lists some of the causes of headache that may require immediate medical attention.
Table 37-1
Pathologic Conditions Causing Headache
| Pathologic Condition | Signs and Symptoms |
| Subdural hematoma | Mild to severe, intermittent headache; neurologic symptoms include fluctuating consciousness |
| Subarachnoid hemorrhage | Sudden onset, severe and constant headache; elevated blood pressure; can cause change in consciousness |
| Increased cranial pressure | Mild to severe headache; neurologic symptoms include hemiparesis, visual changes, and brainstem symptoms, such as vomiting, altered consciousness |
| Meningitis, viral and bacterial | Severe headache with radiation down neck; acute illness and fever; positive Kernig’s sign |
| Brain abscess | Mild to severe headache; local or distant infection; fever may not be present; neurologic signs consistent with local site of infection |
| Central nervous system | Localized headache and focal neurologic symptoms; cranial nerve symptoms often seen |
| Central nervous system neoplasm | Localized headache and focal neurologic symptoms; cranial nerve symptoms often seen |
| Toxicity | Generalized headache, pulsating; other signs of toxicity may be present |
| Sinusitis | Frontal or dull headache, usually worse in morning; increased pain in cold damp air; nasal discharge |
| Otitis media, mastoiditis | Feeling of fullness in ear, stabbing pains in head, vertigo, and tinnitus |
Headache can be difficult to evaluate; however, the intensity, quality, and site of pain may provide clues. Because the diagnosis of headache is essentially a clinical diagnosis, the physical therapist is often involved in the diagnosis. Awareness of the variety of manifestations can direct the intervention both medically and physically. Current understanding of the pathology and physiology of the headache puts it into the realm of the neurologic evaluation. The therapist needs to understand the cause, precipitating factors, and typical course of chronic headache pain. Understanding headache is a challenging task given the large number of headache syndromes and the possibility of a continuum within one diagnosis.31,68
PRIMARY HEADACHES
Overview, Definition, and Incidence
Tension-type headaches are the most common type of primary headache; as many as 90% of adults have had or will have tension headaches, representing 25 million people in the United States. Tension-type headaches occur primarily as a response to stress. Tension headaches are more common among women than men, especially with advancing age.8
The IHS divides tension-type headaches into subcategories that are believed to be varieties of the same disorder. Infrequent episodic headaches are those that occur less than 12 days/yr; frequent headaches occur from 12 to 180 days/yr. Chronic headaches are those reported more than 180 days/yr. The headaches are further defined by whether or not they are associated with pericranial pain. Prevalence appears to peak in 30-to 40-year-olds in both men and women, increasing with level of education in both genders.86 The prevalence is between 16% and 39% in most studies. The prevalence of infrequent episodic headaches among 40-year-olds in the general population is 53%.92
There appears to be an increase in headaches that are regarded as chronic (chronic tension-type headache). The headache is recurrent, lasting for 30 minutes to 7 days. The headache can be moderate to severe in intensity with minimal aggravation by physical activity. It is chronic, bilateral, constricting, and nonpulsatile, unlike the migraine, which is unilateral and throbbing.
Distinguishing chronic tension-type headache from migraine and from medication-induced headache is a diagnostic challenge, and the management is different. In general, individuals with tension-type headache seek less medical attention than migraineurs.72
Etiologic and Risk Factors
Pain and psychologic factors clearly affect one another. Individuals with tension-type headache tend to report greater use of nonoptimal coping strategies, including withdrawal, avoidance, and self-criticism when coping with negative life events. Tension-type headache sufferers appear to have a high level of fearfulness about pain and may have an attentional bias toward bodily function versus environmental awareness. There is some evidence in females with tension-type headache of a component of catastrophizing an event that leads to a feeling of helplessness. No clear link has been established between these behavioral findings and the findings of central sensitization, the role of the limbic system, and reticular disinhibition, although there is interest in that area. Some day, the relationship, or the cause versus effect, may be better understood and the neurologic construct may be better defined.
Some biochemical changes are common to both migraine and tension-type headache. Blood levels of magnesium are low in both conditions, and there is increased blood flow in anterior, middle, and posterior cerebral arteries.
Pathogenesis
The pain input to the brain may be increased because of activation or sensitization of peripheral sensory afferent neurons, possibly by endogenous substances such as serotonin and bradykinin. Increased hardness of muscle tissue in the upper neck is found in the absence of muscle firing.
Sensitization of second-order neurons and neurons at the level of the trigeminal nucleus alters nonpainful input so that it is perceived as noxious at the level of the cortex. Stimuli to skin, tendons, and muscle cause pain in the area around the head and, at the same time, in areas distant from the head. This may be consistent with referred hyperalgesia, relating to the convergence of multiple peripheral sensory afferents onto sensitized spinal cord neurons, which project to central structures that have another level of sensitization. Serotonin levels are likely to play an important role, as the headache pain can be modified by administration of drugs that work at the level of the serotonin receptor.3
Lack of normal inhibitory control in the central nervous system may also play a role in tension-type headache. Pain that can normally be down-regulated by interneurons may lose that function so that there is a loss of inhibitory control, or disinhibition. The neurons responsible probably belong to the bulbar reticular formation. This area receives afferents from the periphery but also from limbic structures, the orbitofrontal cortex, the nucleus raphe magnus, and the periaqueductal grey matter.
Electromyographic (EMG) analysis to determine if there is increased muscle activity or increased resting tone during or between headache episodes has been inconclusive. EMG recordings do not appear to have diagnostic usefulness, and the increases that have been reported may have to do with protective adaptation rather that the cause of pain.
Clinical Manifestations
There is increased tenderness to palpation of the tissues around the head. Both muscles and tendon insertions have been found to be excessively tender. The level of tenderness correlates to the intensity of the headache. The pain is usually in the whole head or in the neck and is reported as tightness or pressure. The pain is often described as dull and nonthrobbing. The severity of tension-type headache increases with increasing frequency. Nausea is not associated with tension headache, but there are reports of anorexia, mild photophobia, and phonophobia.56
Sleep disorders are commonly reported with tension headache, but studies do not show evidence of a relationship. Lack of sleep can precipitate the headache. Stress, fatigue and not eating on time are among the most common headache precipitants. Other triggers, consistent with migraine, are smoke, alcohol, smells, and weather changes.
MEDICAL MANAGEMENT
The diagnosis of tension headache requires the exclusion of other causative disorders. The medical history should include the evolution of the headache. A general physical and neurologic examination should be performed to rule out a disease process, including palpation of the pericranial muscles to identify tenderness and trigger points. Referred pain patterns should also be recorded. The temporal, lateral pterygoid, masseter, sternocliedomastoid, and trapezius muscles should be looked at specifically. It should be noted whether the palpation is done during the headache or nonheadache phase, as there can be up to a 25% increase in pain perception during headache.
Except for their frequency and intensity, chronic tension-type headaches are similar to frequent episodic tension-type headaches. Chronic tension-type headaches occur most often in women over 50 years of age and have usually evolved from episodic tension headaches. Chronic tension-type headache may be linked with medication overuse (see later), and the diagnosis should be made only after there have been 15 days free of medication. It is important to recognize that individuals with confirmed migraine are prone to increased incidence of chronic tension-type headaches between migraine attacks. The individual will usually describe a difference between the headache types.
Tension-type headaches, because they occur with greater frequency and duration, and moderate intensity, causes a broader spectrum of fatigue and depression than a migraine might. Current studies have found that individuals with chronic tension-type headache report a lower quality of life.
TREATMENT.
The first step in treatment should be a written record of the frequency and severity of attacks and of the medication currently consumed for them; often there is more medication used than the patient had been aware of. Overuse of analgesics and nonsteroidal antiinflammatory drugs (NSAIDs) may increase the frequency of tension-type headaches (see later). Estrogenic hormones, including oral contraception or hormonal replacement therapy, may worsen tension-type headaches.
Analgesics and NSAIDs are the most typical medications used for control of tension-type headaches, and often the individual will self-select the dosage. Most often, the doses taken are not adequate to control the pain but are taken more often than is effective, leading to rebound as a trigger for headache. Ibuprofen appears to be the fastest acting and is superior to aspirin for relief of headache pain; it has a lower risk of gastrointestinal bleeding compared to other NSAIDs. Combination therapy using analgesics can enhance the effect and allow lower dosages and lower risk of side effects. Caffeine has long been used as an analgesic adjuvant, often combined with aspirin or ibuprofen. It has been common to include codeine and butalbital, but dependency on the drug may develop. When migraine headache and tension-type headache are occurring at the same time, use of triptans shows effectiveness for both headache types. As the role of serotonin becomes better understood in tension-type headaches, medication directed at modulation of serotonin will become more common.
Tricyclic antidepressants such as amitriptyline are useful in chronic tension-type headache. If depression is comorbid, tricyclics are generally regarded as more effective than selective serotonin reuptake inhibitors. Stress management strategies are effective, as are cognitive-behavioral therapies. A combination been shown to be better than either strategy alone.62
PROGNOSIS.
Over time there is an increased risk of the episodic tension-type headache developing into a chronic tension-type headache.
Migraine
The definition of the World Federation of Neurology contains the following language regarding migraine: “a familial disorder characterized by recurrent attacks of headache widely variable in intensity, frequency and duration. Attacks are commonly unilateral and are usually associated with anorexia, nausea and vomiting. In some cases they are preceded by neurologic and mood disturbances.”104
Migraine syndrome is difficult to define due to the fact that the attacks vary widely between individuals. Within one individual, each attack may have different characteristics. The prodrome or aura, the nature of the attack, and the type of pain may represent separate pathologic processes that happen at the same time or within the cycle of the headache. The trigger that causes one type of migraine may be different from the one that causes another type. Most authors agree that stressful periods are linked to the onset of the headache, although the manifestations may be quite different.
In the United States, migraine headaches often go undiagnosed or are misdiagnosed as tension-type or sinus headaches. There also seems to be two types of migraine sufferers, those who seek medical intervention and those who self-medicate without medical consultation. The ability to determine the appropriate link between pathologic processes and intervention remains a medical challenge. As a result, many migraine sufferers may not receive effective treatment.16
Incidence
Migraine headaches are the second most common type of primary headache. An estimated 28 million people in the United States (about 12% of the population) will experience migraine headaches at some point.78 An estimated 6% of men and up to 18% of women have migraine headaches. In 90% of migraineurs, the first attack generally develops before the age of 40 years.53 Clinical and epidemiologic investigations indicate that adult women are at greater risk for the development of migraine than are adult men. In women the frequency of headaches is highest during their reproductive years, when estrogen levels are higher, and decreases to some extent after menopause. Frequency in adult men does not appreciably change between ages 20 and 65 years.54,77
About 45% of cases of migraine emerge during childhood or adolescence. Migraine headaches are estimated to appear earlier in males than in females, and in both genders migraine with aura is more likely to develop at an earlier age than migraine without aura.17,77 Young boys have a frequency and intensity slightly higher than young girls.
Childhood headaches include complaints of bitemporal pain and a shorter duration (less than 2 hours). Headaches appear more frequently in children than in adults. Episodic vertigo, abdominal pain, and autonomic responses are more common during an attack in a child than is headache. Cyclic vomiting syndrome (CVS) appears to be a common precursor to migraine in children.
Etiologic and Risk Factors
It has been determined that there is not a “migraine personality” but rather that those individuals who are susceptible to migraine may also be at risk for other alterations of the nervous system. Anxiety disorders, especially panic attacks and depression, are characterized by disturbances in the same neurochemical systems, and therefore the syndromes may overlap or could even be considered one syndrome with a subset of conditions. There may also be a central serotonin demodulation that is seen within the context of psychologic disorders. Chronic pain related to migraine may also lead to changes in behavior.22,26,64
Comorbidity with possible association to other diseases has been studied. A relationship between migraine and stroke has been found in many studies, but the mechanism is not clear, and there seem to be different risk factors with age and type of migraine attack. In young women 20 to 45 years, there is an increased incidence of stroke in the migraine population. Smoking or using oral contraceptives increases this risk.66 Common elements such as abnormal platelet aggregation, a well-known component of migraine and a possible stroke risk factor, may explain the correlation. Migraine is common in the young population; stroke is not. There appears to be a higher incidence of stroke in between migraine attacks in individuals with aura. The activation of brainstem structures as the mechanism for migraine may predispose individuals to posterior fossa stroke, the most common type found in young stroke victims with migraine. Posterior fossa strokes are also more common in the general younger population. In migraine-induced stroke, the symptoms of aura last longer than 1 hour, and neuroimaging confirms infarction.65
Despite an apparent reported connection with heart disease and mitral valve prolapse, the link does not appear to be strong. Hypertension may be one of the factors that changes episodic migraine into a chronic daily headache. Allergy, Ménière’s disease, lupus, and epilepsy have all been reported often as comorbidities, and the changes in migraine status as a result of interventions directed toward these conditions may help to describe the link.85
For many migraineurs, certain foods must be avoided. The foods most likely to be linked to migraine are milk, eggs, corn, and wheat.76 Environmental factors such as food additives and colorings could also be a trigger for migraine. Coffee, alcohol (especially red wine), and sugar can set off an episode in vulnerable individuals.
Often the headaches are associated with hormonal changes of menstruation and use of oral contraceptives. Pure menstrual migraines appear to be more frequently migraine without aura. It appears that these migraines may be related to the abrupt decrease in estradiol that occurs immediately before menstruation after several days of exposure to high levels of estrogens. It also appears that there may be an intrinsic estrogen receptor sensitivity that differs among women. This may therefore create an individual response to the hormone level. Attempts to find differences in ovarian hormone levels between women with menstrual migraine and controls have not yielded consistent results.61
When the headaches follow a pattern related to hormone changes, pregnancy brings periods of both exacerbation and relief. In the first trimester of pregnancy, the number and severity of migraines rise. The headaches abate during the second trimester and then increase again in the third trimester. Migraine can be triggered during delivery and may be more prevalent in the weeks and months following childbirth.15
There is a positive family history of migraine in about 60% of cases, which suggests a hereditary factor. However, the patterns of migraine inheritance are complex, both in the mode of inheritance of migraine and in the role of genetic factors in the pathogenesis. Studies show an increased risk of migraine with aura among first-degree relatives. Twin studies show that migraine with aura is caused by a combination of genetic and environmental factors.63,97
Multiple genes probably contribute to the genetic susceptibility for migraine, with different genes associated with the different types of migraine. The failure to identify a simple genetic mechanism does not rule out genetic determinants of migraine, but it suggests either that not all migraine syndromes are genetically equivalent or that environmental factors also play a significant role.
In familial hemiplegic migraine, mutations of genes have been found for at least two separate chromosomes, and there is overlap with the gene mutations found in a benign form of epilepsy that is only briefly active in children. These mutations may make the brain more susceptible to prolonged cortical spreading depression via excessive glutamate release or decreased removal of glutamate and potassium from the synaptic cleft. It is believed that there is another gene that may be responsible that has yet to be identified.33
Study of mutations in the motor nerve terminals that contain P-type calcium channels in mice may shed some light on the mutations that would contribute to changes in acetylcholine release identified with a variety of movement disorders and may also identify part of the mechanism of the pain of migraine. Susceptibility to migraine during periods of recovery from prolonged stress suggests that the mechanism might involve a switch in autonomic balance from sympathetic to parasympathetic dominance.22
Pathogenesis
The components of migraine are complex, and it appears that the mechanisms vary among individuals and among attacks in the same individual. The more that is known about migraine, the more levels of dysfunction are identified.71 The mechanism that causes pain may be different from the mechanism that causes the neurologic symptoms. The trigger for the migraine may give the clue to the nature of the symptoms. Fig. 37-2 illustrates the possible mechanisms related to migraine headache.36
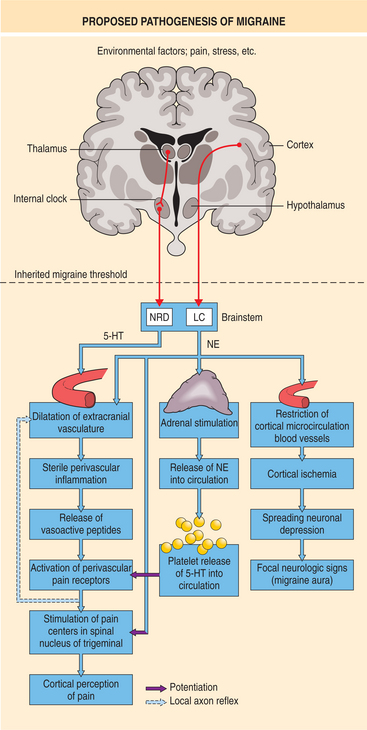
Figure 37-2 Mechanisms that are related to the pathogenesis of migraine headache based on current proposals. (From Weinstein JM: Headache and facial pain. In Yanoff M, Duker JS, Augsburger JJ, et al, eds: Ophthalmology, ed 2, St Louis, 2004, Mosby.)
Pain is the most common complaint in migraine and may relate to several causal factors. The trigeminal complex, including cranial nerve V, is a key component in the distribution of the pain within the head and neck associated with migraine. Surrounding the large cerebral vessels, pial vessels, large venous sinuses, and dura mater is a plexus of largely unmyelinated fibers that arise from the ophthalmic division of the trigeminal ganglion and the upper cervical dorsal roots. The dural nerves that innervate the cranial vessels consist largely of smalldiameter myelinated and unmyelinated fibers that almost are nociceptive in function, so that stimulation of the cranial vessels causes pain. The meninges that cover the brain parenchyma, and the arteries of the cortex are richly supplied by nerve fibers from the trigeminal ganglion and the upper cervical dorsal roots. These fibers contain substance P and calcitonin gene–related peptide that can be released when the trigeminal ganglion is stimulated, resulting in nociception, or pain. Sensitization of the mechanoreceptors in these structures enhances the responses to mechanical stimuli.67 Sensitization of the menigial primary afferents creates a painful response to traction and pressure. This is thought to be responsible for the diffuse pain often noted in one area of the head and the throbbing pain that seems to be associated with the heartbeat.15 Routine physical activities such as coughing, sneezing, bending over, or performing the Valsalva maneuver can cause pain through this mechanism. Sterile neuroinflammation is another possible result of the peptide release; however, there does not appear to be the level of tissue damage found in other inflammatory processes. As in other inflammatory disorders, inflammation in migraine is neither the original nor the leading event, but one of the mechanisms involved in its pathogenesis, helping to explain findings such as sensitization.
The central modulation of pain is related to a complex neurochemical and neurophysiologic system in the brainstem. The central processes of the trigeminal afferents play a role in migraine pain. Long-lasting central sensitization may prolong the effects of a pain stimulus related to the trigeminal vascular system. Common to the complaints of migraine is the phenomenon of allydonia, or pain responses to common activities such as hair combing, shaving, or wearing earrings or a necklace. This is the result of sensitization of the neurons of the medullary dorsal horn that receive sensory input from the dura and skin. This abnormal input is sent to the thalamus and then relayed to the cortex.
Results obtained by positron emission tomographic (PET) scan during a spontaneous migraine attack suggest that the pathologic activity in brainstem nuclei regulating antinociception and vascular control may be related to the pathogenesis of migraine. The brainstem contains networks that modulate pain transmission. In the medulla are so-called off-cells that inhibit pain and on-cells that facilitate pain. It is believed that these cells modulate the activity of the trigeminal nucleus ceruleus and neurons of the dorsal horn. Increased on-cell firing or decreased off-cells firing could be responsible for the increased sensitivity to both painful and nonpainful stimuli. Both pain and stress can turn on the system and may account for some of the association between pain and stress. Pain tolerance decreases in individuals who develops headache. Normally, stimulation of sensory fibers excites γ-aminobutyric acid (GABA) receptors, and the inhibition reduces the excitability of pain neurons in the dorsal horn of the spine in order to modulate pain response to stimulation. This mechanism may be aberrant in migraine sufferers, and so there is less inhibition. It has been found that tenderness in the neck, not muscle contraction, correlates with headache development.73
Neurovascular transmitters and receptors play a role in headache through their influence on the regulation of tone in cranial blood vessels. Acetylcholine and vasoactive intestinal polypeptide in the cranial arteries cause relaxation of the vessels. Vascular changes occur before and during the migraine headache. In the headache with aura, cerebral blood flow is reduced by about 20% before the headache, resulting in cerebral hypoxia. This cerebral hypoxia is thought to be responsible for the neurologic defects that characterize the aura. Blood flow increases during the headache, and there appears to be some relationship to the arteries, to referred pain patterns of the veins, and to the site of pain. Dilation of the middle cerebral artery and the superficial temporal artery on the pain side during migraine has been demonstrated.
Endothelial cells found in the vessel wall synthesize and release substances that produce vasodilation and vasoconstriction of the cerebral vessels. Cerebral blood vessels are very responsive to chemical stimuli such as changes in carbon dioxide tension and oxygen level. Bradykinin, adenosine, and histamine are released not only by neurons but also by endothelium, platelets, and mast cells. These substances have been proposed as local regulators of cerebral blood flow.18
The neurotransmitter 5-hydroxytryptamine (5-HT), known as serotonin, appears to be a part of the pathogenesis of migraine. Chronically low availability of serotonin is found in individuals with migraine, so a hypersensitivity to the presence of serotonin develops. Platelets contain virtually all the serotonin present in the blood and release serotonin during aggregation. Platelets play an important role in the acute inflammatory response. Many persons with migraine show hyperaggregability of platelets when free from headache. Aggregated platelets release catecholamines and serotonin that may cause the initial stage of vasoconstriction. Platelet aggregation is increased during the prodromal stage of migraine. This could be due to an increase in epinephrine, thrombin, or arachidonic acid in the circulation in response to anxiety. There appears to be a decrease in aggregation during the actual headache. Reported changes in plasma serotonin may be due to changes in platelet function. Serotonin also excites pain nerve endings and increases the pain response. The serotonergic neurons also have a close association with cortical astroglial cells that help to maintain homeostasis in the brain.71
The brainstem is considered to be implicated in the changes in cerebral blood flow by stimulation of the locus ceruleus or the nuclei raphes. The nuclei raphes appear to increase blood flow in the brain. Although there is no direct anatomic connection between raphe neurons and the meningeal and cerebral blood vessels, these vessels can respond to changes in central serotonin neurotransmission. Serotonergic neurons located in brainstem nuclei raphes are involved in the regulation of multiple other physiologic functions, including stress, pain, appetite, mood, and sleep. The neurons change their firing rate during the sleep-wake cycle. This may explain why sleep is often the best antidote to a migraine.
Cortical spreading depression is believed to be part of the neuronal abnormality leading to migraine. It is a mechanism that starts with a small excitatory response that begins to spread through the brain and then causes a suppression of electroencephalographic (EEG) activity that moves through the cortex. Cortical spreading depression involves gross disturbances of the brain’s extracellular environment. The potassium level increases, the extracellular glutamate level increases considerably, and the extracellular calcium level decreases. Calcium ion channels control and maintain electrical potential across cell membranes and generation of electrical signals (see Chapter 28). Abnormal ion channel function is believed to be the mechanism in the rare form of familial hemiplegic migraine.37
Cortical spreading depression may be responsible for the reflex activation of sensory pathways that induce inflammation within the meninges as described earlier. Cortical spreading depression is accompanied by local vascular responsiveness. Spread of first aura symptoms and spread of cortical depression proceed at the same speed. This may explain the initial hypoperfusion that is related to the aura of headache. Cortical hyperemia may be responsible for the flashing jagged light that sometimes occurs just before the pain begins. Once the headache is underway, it appears that the vascularity changes and hyperperfusion are not related to the degree or area of the pain. Nitric oxide is produced both immediately after cortical spreading depression and in a second prolonged wave lasting several hours. Nitric oxide has a key role in regulation of cardiovascular tone and control of arterial blood pressure. Nitric oxide may also have actions at both the primary and second-order neurons of the trigeminal pain pathways.
It appears that additional pain input from muscles and other cranial tissues may contribute to migraine. Modest additional input from pericranial muscles can fire the neurons and cause pain. There is often tenderness of pericranial structures during migraine. It is believed that perhaps cortical and brainstem control may enhance or diminish the pain caused by a certain pain input from these structures. See Fig. 37-2, which demonstrates the mode of dysfunction that may take place in response to the triggers and the area targeted by treatments.
In those individuals with menstrual migraine, there is a link to falling blood levels of estrogen, and the migraine may be due to hormone-related pain modulation. Estrogen has a significant effect on the synaptic morphology of the neurons in the periaqueductal grey area. Afferents from the trigeminal nucleus complex and periaqueductal grey areas that project to them contain estrogen receptors. The pariaqueductal grey area produces a number of behavioral and physiologic responses, including autonomic changes, sexual behavior, fear and rage reactions, and anxiety. The role of sex hormones associated with migraine may be through dysfunction of the inhibitory circuits from the locus ceruleus to the trigeminal nucleus complex. Higher levels of estrogen are correlated with activity in the locus ceruleus and inhibition of the trigeminal nucleus complex in response to painful stimulation of inflammation, so this is consistent with the typical nature of the headache, which is triggered during the time that the estrogen levels have fallen prior to the onset of menstrual bleeding.60
Prostaglandins could also cause the severe systemic upset and nausea and vomiting that occur. Levels of prostaglandins are elevated during migraine in the plasma, saliva, and venous blood. Prostaglandins are also implicated in the bradykinin-induced excitation of nociceptors.31 Proinflammatory cytokines can cause sensory hypersensitivity and are expressed in the central nervous system by neurons.
Migraine associated with food with containing tyramine may occur in persons who have a deficiency of tyramine-o-sulfatase. The excess tyramine could be responsible for the release of catecholamines and could initiate the vasoconstrictive stages of migraine.
Clinical Manifestations
Migraine Without Aura.: Migraine headaches may be dull or throbbing. They usually build up gradually and may last for 4 to 72 hours. Headache is aggravated by routine physical activity, and there is an association with nausea. Photophobia, or increased sensitivity to light, is common, and the migraineur will often seek a dark, quiet place. There are often various combinations of fatigue, difficulty in concentrating, neck stiffness, blurred vision, yawning, and pallor.
When the headache resolves, there is commonly a feeling of heaviness and aching in the head, the scalp may be tender, and there may be considerable fatigue. At the termination of the headache there is often marked diuresis, or increased urination.17,30
The trigeminal brainstem nuclear complex has a somatotopic representation of the trigeminal dermatome that is continuous with the representation of the posterior head and neck region in the upper cervical dorsal horn. Pain patterns representing increased neuronal activity in the trigeminal nucleus caudalis and dorsal horn include upper cervical pain, usually on one side only.
Migraine with Aura.: The headache will frequently start with a period of depression, irritability, and loss of appetite. This is often the beginning of what is known as the aura. These symptoms may be a result of spreading depression.
Paresthesias can also be a part of an aura and are second in frequency to visual symptoms. Paresthesias of the hand and face are most common, with less involvement of the leg and trunk. The paresthesia often involves the tongue, something that is rarely seen in cases of stroke or transient ischemic attack (TIA). There is usually tingling of the hands with numbness.50 The attacks are predictable using the sensory homunculus. Speech difficulty during an aura reflects the involvement of the dominant hemisphere.30 Vertigo and dizziness may be related to brainstem activity or changes in blood flow around the vestibular mechanism (see the section on Vestibular Migraine later). For some individuals there is a combination of aura symptoms, and the type of aura may change with each episode of headache. Neglect, spatial and geographic disorientation, anxiety, and amnesia have all been associated with the aura of migraine.
Autonomic changes can also result in paresthesia of the face or extremities. The aura consists of fully reversible symptoms that precede or accompany the headache. When a migraine is preceded by visual symptoms, it is known as a visual aura. The aura is usually described as changes in the visual field. Visual images change, and there can be loss of focus, spots of darkness, and zigzag flashing lights. It often begins with a hazy spot close to the center of vision; initially vision is unclear or it seems difficult to focus. The hazy spot will form into a star shape that further develops into a shape known as a fortification because it is semicircular with angles like those of fortifications. This scintillating vision consists of luminous, bright, flickering colors of the spectrum, much like a prism catching light. It can be combined with a scotoma, or an area of vision that appears to be obstructed, or missing. The visual image fades as the headache begins. The headache is intense, throbbing, and usually contralateral to the visual field changes.17 Fig. 37-3 shows the image as it would appear during a migraine.

Figure 37-3 Scintillating scotoma in migraine with aura. The leading edge of the scotoma is “positive” (i.e., it consists of bright flickering imagery that obscures or replaces the normal visual field), whereas the trailing edge of the scotoma often is “negative” (i.e., it displays a relatively dark area that fully or partially obscures the visual surround). The illustration depicts a typical fortification scotoma with sharply angulated borders; many other variants of the migraine scotoma may occur. (From Weinstein JM: Headache and facial pain. In Yanoff M, Duker JS, Augsburger JJ, et al, eds: Ophthalmology, ed 2, St Louis, 2004, Mosby.)
Typical Aura Without Headache.: In some cases, the aura symptoms are not followed by headache. When recurrent aura symptoms occur in the absence of headaches, the distinction from mimicking conditions becomes more important. This is especially important when the symptoms are very short or very long, or they begin after the age of 40. Aura without headache is seen primarily in men and in advancing age.30
Sporadic and Familial Hemiplegic Migraine.: In sporadic and familial hemiplegic migraine, the presentation is migraine with aura, including motor weakness. Paresis can be associated with impaired coordination. The person experiences symptoms of numbness and weakness preceding the onset of the headache. The numbness in the face and arm may spread to involve one side of the body. Basilar symptoms may be present with dysphasia or aphasia causing difficulty with speech. Comprehension is preserved. This type of migraine may alternate with headaches without hemiplegic symptoms. Pain may be either contralateral or ipsilateral to symptoms, and on rare occasions there is loss of consciousness. Although the symptoms appear to resemble a TIA or mild stroke, migrainous infarction is rare. Age of onset for the hemiparetic migraine is 10 to 15 years, and there is usually a history within the family. Familial hemiplegic migraine appears to be related to genes on chromosome 19 or 1.25 The migraine is considered sporadic if there is no first-or second-degree relative with hemiplegic migraine. There is high variability of the attacks, symptoms, and the disease course among individuals with familial hemiplegic migraine.98
Basilar-Type Migraine.: Basilar migraine can occur throughout life in both sexes. Basilar migraines have a symptom profile that suggests posterior fossa involvement localizing to the vascular territory of the basilar artery—the brainstem, cerebellum, and occipital lobes. The prodromal symptoms reflect brainstem dysfunction. There are complaints of altered level of consciousness, dysarthria, tinnitus, ataxia, diplopia, symptoms in both the temporal and nasal fields of both eyes, and peripheral dysesthesias, followed by occipital headache.5 Given how often vertigo, fluctuating hearing, and tinnitus occur together, basilar migraine may be difficult to distinguish from peripheral disorders such as hydrops (see Chapter39 ) and from vertebrobasilar TIAs (see Chapter 32). The syndrome is most common in adolescent girls. The symptoms are bilateral, reflecting the bihemispheric dysfunction associated with the basilar blood flow.7
Vestibular Migraine.: Dizziness with complaints of true vertigo is extremely common in migraine, and migraineurs often have a lifelong or childhood history of motion sensitivity, with symptoms provoked by amusement park rides, riding in the back seat of a car, or reading in the car. Most migraine sufferers will report some motion sensitivity during a severe headache and will often prefer to lie still in a dark, quiet room, although these symptoms alone are not sufficient to be considered vestibular migraine.64
When dizziness is the primary complaint or is the predominant component of the aura, then it may be considered as vestibular migraine. Vertigo occurring as an aura may arise from the same transient inhibition of neuronal function responsible for the visual aura. Episodic vertigo from vestibular migraine can be thought of as a subset of basilar migraine. Spells usually last approximately an hour but can last several hours or days in patients who have severe symptoms.12 Nausea, vomiting, hypersensitivity to motion, and postural instability are cardinal signs. Some persons complain of vertigo, a swimming sensation, or imbalance without a complaint of headache. Dizziness does not always accompany the headache but may intermittently coincide with the headache. Changes are rarely seen on electronystagmography (ENG), indicating a peripheral deficit that is transient. The vestibulocochlear symptoms occur most often as a part of the headache. Auditory symptoms also occur in migraine. Phonophobia is the most common, with complaints of tinnitus more frequent than in the general population. Hearing loss alone is rarely associated with migraine.48 Box 37-2 defines both definite and probable vestibular migraine.75
The mechanisms are still not clearly understood, but it may be that neuropeptides released during the migrainous event could increase the firing of the peripheral vestibular apparatus, or the vestibular nuclei.75 This may result in a transient increase in the firing on one side of the system, causing the subjective sensation of moving. Changes in cerebral blood flow during the migraine may contribute to the dizziness. The inner ear is supplied by branches of the anterior inferior cerebellar artery arising from the basilar artery, and the anterior inferior cerebellar artery also supplies the anterolateral pons, middle cerebellar peduncle, and flocculus. Prolonged vasoconstriction of vertebral and basilar arteries may be responsible, or the cause could be the direct mechanical pressure of the dilated vessel during the vasodilating period of the headache. The dizziness can be associated with the position of the head, indicating an association with benign paroxysmal positional vertigo (see Chapter 39).
Acute attacks can be treated with various analgesics. Preventive treatment is most frequently accomplished with amitryptiline, β-blockers, calcium channel blockers such as verapamil, and acetazolamide. Migraine may be associated with many vestibular symptoms.51
Ophthalmoplegic Migraine.: Ophthalmoplegic migraine results in pain around the eye and paralysis in the distribution of the third, fourth, or sixth cranial nerve and can produce diplopia, or double vision. One of the features of ophthalmoplegic migraine is that the headache always precedes the oculomotor deficit by several hours or days. The paralysis can progress from being transient to lasting several days, and in some persons it becomes permanent. People with ophthalmoplegic migraine typically give a history of many years’ duration of migraine without oculomotor involvement before the ophthalmoplegia develops.7,101
Retinal Migraine.: Retinal migraine is repeated attacks of monocular visual disturbance, including scintillations, scotomata, or blindness, associated with migraine headache. Visual changes are strictly unilateral. There may be neuronal spreading depression or involvement of the posterior ciliary vasculature. TIA must be ruled out, as emboli from the carotid artery can cause similar symptoms.5
Status Migrainosus.: The headache of status migrainosus (SM) lasts more than 72 hours, and a usual trigger will set it off. SM typically resembles the individual’s usual severe migraine, but there may be a spread of pain to other areas as the headache persists, and the spread of allodynia occurs. The premenstrual part of the female cycle is a time of particular risk for SM, and changes in hormone status, pregnancy, miscarriage, or change in birth control pills can be factors. Upper respiratory or urinary tract infections can also be the trigger for SM. Overuse of analgesics and rebound withdrawal-type headache, the headache that comes when not taking medication, can trigger SM. Ruling out other possible causes is critical.
Prolonged vomiting to the point of dehydration is common. Severe pain and fatigue limit activity, and hospital admission may be appropriate. Ergots and antiemetics are effective to some degree. Corticosteroids may be helpful, and opioids are sometimes used. Comorbid depression is frequently seen.10
Migraine Aura Status.: Migraine aura status is a rare subtype with persistent aura without infarction. One or more symptoms that the individual usually experiences as a part of his or her typical migraine aura persist for more than 1 week. Visual symptoms are most common, but it can be any symptom. Most typical treatment is ineffective, and the problem often must run its course. Valproate has been helpful in a small number of patients.
Chronic Migraine.: Most cases of chronic migraine start as migraine without aura. The criterion for chronic migraine is headache for 15 or more days a month for more than 3 months. Medication overuse is the most common cause of chronic migraine, with rebound or withdrawal of medications as a trigger. Women predominantly report chronic migraine, and they report a change in symptoms over time, with decreasing photophobia, and nausea and a headache that resembles a mixture of tension-type headache and migraine.87
MEDICAL MANAGEMENT
In most cases the diagnosis of migraine can be established by the history alone. The neurologic examination is normal. Diagnostic procedures are unnecessary and may lead to confusion. An EEG may show focal slowing if taken during an attack of migraine and may create the impression of a space-occupying lesion or infarction. Magnetic resonance imaging (MRI) scans show diffuse white matter changes in the frontal subcortical and deep white matter at the level of the basal ganglia. This may be due to platelet aggregation with microemoboli, abnormal cerebrovascular regulation, and repeated attacks of hypoperfusion during the aura.
TREATMENT.
It is clear that the causes of migraine are multifactorial, and therefore the most successful treatment is directed at the system that is most likely implicated. Avoidance of precipitating factors becomes a natural component of controlling frequency. Rest in a quiet, dark room is often necessary during the attack. Pharmacologic treatment can be abortive or preventative by reducing frequency and severity. Because the migraine type may vary in one individual in different attacks, prevention is sometimes difficult. Timing of medication related to migraine type may be critical. There may be a change to the blood-brain barrier during migraine that allows inhibitory substances to pass into the brain, which changes pain perception; however, the gastric stasis that can be part of the aura can limit or slow the absorption of medication.38
Vasoconstricting drugs such as Cafergot, a combination of ergotamine tartrate and caffeine, are often helpful. Caffeine has several properties that may be of benefit in the treatment of migraine. Concurrent oral administration of caffeine with aspirin increases the peak plasma concentration of aspirin. It has mood-altering properties, so that the increased mental alertness, lessened fatigue, and feeling of well-being that follow caffeine ingestion counterbalance some of the symptoms of migraine. Caffeine causes cerebral vasoconstriction and may also be antiinflammatory.88 Caffeine use is a major risk factor for development of chronic daily headache and can cause increases nervousness, sleeplessness, and anxiety.
NSAIDs are useful analgesics for migraine-type pain. They also play a role in the inhibition of prostaglandin synthesis in brain neurons. NSAIDs are particularly effective when inflammation has caused sensitization of pain receptors to mechanical or chemical stimuli. They appear to prolong the catecholamine and serotonin turnover in brain neurons and block the release of serotonin. Antinflammatories inhibit platelet formation that becomes abnormal during a headache phase. Aspirin and other NSAIDs are quickly absorbed. Contraindications for use are ulcers and bleeding disorders.18 Increasing the dosage of these medications does not significantly increase their effectiveness, and there is a dose-dependent increase in the side effects, so use of aspirin and other NSAIDs at a low dose should be tried before going on to higher doses. There is no evidence that trying another type of NSAID after a failure of one type is helpful.
Ergot alkaloids provide vasoconstriction and are more active on large arteries. There is a temporary increase in blood pressure. These drugs may decrease activity in the central trigeminal neurons. Ergot alkaloid can be used to abort a headache. There is some concern about prescribing a drug that induces vasospasm in individuals with aura or those with hemiplegic migraine.95 A short-term increase in blood pressure may limit use in some patients.
Triptans are effective in controlling the symptoms associated with migraine. The drug action is at the level of the serotonin receptor agonist. Typically, the sensation of throbbing is diminished with use of triptans. There is evidence suggesting that triptans may also activate descending pain-modulating pathways that affect the trigeminal system. The most common triptan, sumatriptan, is a rapidly effective agent for aborting attacks when given subcutaneously by an autoinjection device. In less severe cases, sumatriptan is given orally. Other triptans now in use are zolmitriptan, rizatriptan, naratriptan, eletriptan, and frovatriptan. Headache recurrence is a problem with all triptans, and they do not appear to be effective during aura. Triptans primarily cause constriction in the cranial arteries with an effect in the coronary arteries as well. These drugs therefore should be used with caution in individuals with coronary artery disease.84 The nausea and vomiting induced by migraine can be controlled by drugs such as metoclopramide, which works as an antiemetic, counteracting gastric stasis by increasing intestinal motility and increasing the absorption of other migraine drugs.
Prophylactic treatment may be necessary if migraine occurs more than three times per month. The mode of action may involve both an effect on extracerebral vasculature and the stabilization of serotonergic neurotransmission. The β-blocking drugs affect the central catecholaminergic system, relating to the central mechanism of the headache. In this class of drugs are propranolol, metoprolol, timolol, nadolol, and atenolol.17 Side effects are fatigue, gastrointestinal symptoms, and dizziness. Vivid dreams, nightmares, insomnia, depression, and memory disturbances will cause termination of use.94
Calcium channel antagonist drugs such as verapamil may reduce the frequency of attacks after an interval of several weeks, but the severity and duration of attacks are not influenced.1,96 Flunarizine can be used for migraine prophylaxis if β-blockers are ineffective or contraindicated. Box 37-3 summarizes the acute and prophylactic treatment of migraine.
With the current understanding of the mechanism of migraine, new treatments are being developed. Drugs directed at inhibiting spreading depression, reducing glutamate levels, and controling nitric oxide biosynthesis show promise. Antiemetics combined with other drugs are being studied, and the use of neuroleptics with an effect on serotonergic, adrenergic, and cholinergic neurotransmitter systems are in clinical trials.9
Antiepileptic drugs such as gabapentin, valproate, and topiramate can be effective for migraine prophylaxis with an effect on levels and action of GABA in the central nervous system and influence on the activity of sodium and calcium channels. Gabapentin is effective in both episodic and chronic migraine. Valproate blocks neurogenic inflammation. Topiramate inhibits the activation of trigeminocervical neurons. Paresthesias, cognitive effects, and dizziness are side effects that may limit use. Valproate should not be used during pregnancy, due to possible neural tube defects.89,91
Botulinum toxin (Botox) has been used for migraine prophylaxis but has not been well studied. Central effects may be in the hippocampal neurons and astrocytes, leading to a decreased release of neuropeptides and possible blockade of glutamate.
Herbal therapies have also been used for many years with reported beneficial outcomes. Feverfew (Tanacetum parthenium) appears to decrease headache intensity and nonheadache symptoms of nausea, vomiting, photophobia, and phonophobia. Feverfew is rich in sesquiterpenelactoned, which may be a nonspecific serotonin, bradykinin, prostaglandin, and acetylcholine antagonist. Long-term use has not been carefully evaluated.
Studies indicate that magnesium given as trimagnesium dicitrate appears to have some effect on the intensity and duration of menstrual migraine, and shows promise in use in children. Riboflavin may improve oxygen metabolism at the level of the mitochondria. It has been effective in reducing attack frequency, intensity and duration, but there are few studies to support this. Coenzyme Q10, also working at the level of the mitochondria, has been proven to decrease the frequency of migraine.
Acupuncture has been used with limited results, and the effects are temporary. Its effect is most likely related to changes in stress levels and control of pain responses. Hypnosis has also been used for a long time; however, it has been difficult to determine its effects in current studies. Behavioral treatment has targeted coping with pain and relaxation and examining unrealistic beliefs and their role in creating stress. It includes distraction, imagery, assertiveness training, and problem solving.26
PROGNOSIS.
Men have a 48% persistence rate, whereas women have a 79% rate. Menopause and the resultant decrease in estrogen usually result in a decrease in the frequency and severity of migraine. However, there have been reports of migraine attacks beginning after the onset of menopause.
There has been some question about the relationship of migraine to increased chance of stroke. Migrainous cerebral infarction is associated with an exceptionally severe migraine with changes seen on computed tomographic (CT) scan.11
Cluster Headache
Cluster headaches (CHs) are rare but are the most painful of the primary headaches. There are two types of CH. The episodic type is the most common, constituting 80% of all cases. Episodic CH is defined by periods of susceptibility to headache, called cluster periods, alternating with periods of remission. The headache can occur daily or several times per day for a period of several weeks, known as the cluster period. The headache will go away for several months and recur as another series or cluster.69,90
Chronic cluster headache is a term used when the headaches have gone on for at least 12 months and remissions last less than 14 days. Chronic CH evolves from episodic CH.49
Incidence
CHs affect approximately 1% to 4% of the population. CHs occur mostly in men between the ages of 20 and 50 years and predominantly between ages 27 and 30 years, approximately 10 years later than the typical onset of migraine. Black males appear to have a higher incidence than males of other racial backgrounds. The ratio favoring men over women is getting smaller over time.102
There is a correlation between CH and prior head trauma. There is question as to whether this may be related to venous vasculitis, since there is evidence of inflammation.102
Studies of genetic risk are beginning to show some prevalence of CH in certain families, with a positive history in more than 7% of individuals with CH. The possibility of gene mutation is strong with a high concordance found in monozygotic twin studies.83
Pathogenesis
Like migraine, CH appears to have multifaceted components and dysfunction at many levels. There is vasodilation of the ophthalmic and internal carotid arteries, ipsilateral to the pain. The vascular changes may be a result of stimulation of the trigeminal complex, rather than a casual factor. Stimulation of the trigeminal ganglion causes a cerebral vasodilator response. There is an increase in the level of calcitonin gene–related peptide and substance P. The cerebrospinal fluid shows changes consistent with both parasympathetic and sympathetic and central involvement in CH.
Platelet levels of histamine and serotonin increase during the episode of headache and decrease in the absence of headache. Fig. 37-4 shows the relationship among histamine, gastric secretions, and pain. Blood pressure typically increases; heart rate decreases during attacks of CH. The CH period may be characterized by disturbances in neuroendocrine substances based on the circadian rhythm, which may explain its cyclic nature.23,46
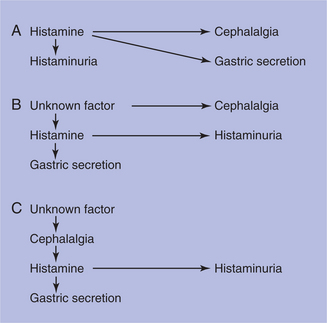
Figure 37-4 Possible relationship between histamine headache and gastric secretion in cluster headache. A, Histamine as the causative agent. B, Histamine level as an independent phenomenon. C, Increased histamine as a secondary product. (From Sjaastad O: Cluster headache syndrome, London, 1992, Saunders, p 150.)
The carotid body may play a major role in the pathogenesis of CHs by its receptor mechanism associated with blood pressure and heart rate. Throughout the course of the cluster period, it may be that the chemoreceptor activity is affected by central control of sympathetic and parasympathetic pathways.39
Hypothalamic regulation of the endocrine system may explain the cyclical nature of the headaches. There is decreased plasma testosterone during the CH period and alterations of production of hormones and melatonin. Melatonin levels are generally low during the day and increase during the hours of darkness and sleep. The nature of the hypothalamic disturbance is not clear.
Clinical Manifestations
The onset of CH is sudden, with excruciating pain. In the majority of CH cases, the headache remains on one side of the head in all recurrences throughout life. The headache is usually localized to one eye and the frontotemporal region (see Fig. 37-1). The pain is boring and nonthrobbing. Autonomic symptoms, which are often on the opposite side from the pain, include photophobia, tearing, and nasal congestion. Autonomic changes may be bilateral. Occasionally Horner’s syndrome (constricted pupil, droopy eyelid) or forehead sweating will appear on the uninvolved side of the face.6 Fig. 37-5 shows the interrelationship of pain and autonomic symptoms in CH. Persons will occasionally complain of blurred vision on the painful side. Persons with CH generally prefer to assume an erect rather than a reclining posture during attacks.29
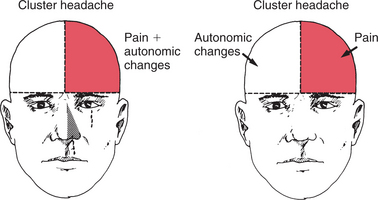
Figure 37-5 The interrelationship of pain and autonomic symptoms and signs in cluster headache. The usual pattern is pain and autonomic changes on the same slide (left). A less common pattern is pain on one side and autonomic changes on the opposite side (right). (From Sjaastad O: Cluster headache syndrome, London, 1992, Saunders, p 49.)
Vasodilators such as nitroglycerin will induce cluster attacks. Often, but not always, alcohol induces an acute cluster attack while the person is in an active cluster period.
Attacks are commonly induced on awakening from an afternoon nap or from sleep during the night, most commonly 90 minutes after falling asleep. Melatonin levels are low during the day and increase during the hours of darkness and sleep; in individuals with CH, the 24-hour production of melatonin is reduced, and the acrophase (the time from midnight to the moment of peak hormone level) is moved forward. Low serotonin level may be responsible for the low melatonin production. Sleep apnea and hypoxemia are associated with CHs.99
CHs follow a circadian rhythm and occur often during the same time of year. They occur most during the summer or winter solstice, appearing shortly after either the longest or shortest day of the year. Even the setting of the clock for daylight saving time will affect the headache cycle, often bringing an end to an episode.102
Persons with CH have some similar physical characteristics, such as a hypermasculine appearance that includes increased and asymmetric facial wrinkles and thick orange peel skin, giving them the appearance of people with alcoholism. Men with CH are on average 3 inches taller than age-matched controls. The typical client with CH smokes cigarettes and drinks alcohol. Personality inventories show a relationship to anxiety, compulsivity, and hypochondria.19
MEDICAL MANAGEMENT
Diagnosis is based on the symptoms and history. Paroxysmal hemicrania, trigeminal neuralgia, and temporal arteritis may have similar symptoms, but they are not episodic. Diagnostic criteria are strict unilaterality, severe intensity, orbital localization, and short duration. Differential diagnosis includes migraine, trigeminal neuralgia, chronic paroxysmal hemicrania, pericarotid syndrome, sinusitis, and glaucoma.13
TREATMENT.
Education should be provided regarding precipitating factors, including alcohol, abrupt changes in sleeping patterns related to travel, and work shift changes. Lack of sleep or afternoon naps may precipitate a headache. Bursts of anger or prolonged anticipatory anxiety can provoke a headache during the cluster period. Altitude hypemia can increase incidence at levels above 5000 feet, including on airplane flights. Avoiding these situations may reduce the frequency of CH during a cluster period. Upright activities, including walking, appear to be of some relief. Few people choose to lie down during an attack.19
Treatment is similar to that for migraine. Sumatriptan and ergotamine-containing drugs are fast acting and effective for controlling acute symptoms. For prevention of headache during cluster periods, verapamil can be combined with prednisone. Methysergide is effective, but side effects include fibrotic complications, and it appears to lose its effectiveness with repeated use for some. Valproic acid is effective but needs to be carefully monitored for liver, pancreas, and blood count changes. Lithium carbonate leads to long-term remission, but has a narrow therapeutic window and side effects that include tremor and renal and thyroid dysfunction.70 Combinations of these drugs are often used rather than one drug in maximal dosages.
Melatonin therapies can be helpful, with remission coming within 3 to 5 days. Oxygen inhalation gives symptomatic relief of headache but does not change the cluster period.
Surgical procedures, including radiosurgeries directed toward the sensory trigeminal nerve, have been the most successful. Hypothalamic implants have been used in individuals with intractable chronic CH. The results are encouraging, with notable pain reduction. All deep-brain electrode implantation procedures carry a small risk of mortality due to intracerebral hemorrhage. Future studies are directed toward other areas of the pain matrix that may be suitable targets for neuromodulation in patients with CH who do not respond to hypothalamic modulation.52
PROGNOSIS.
In the natural course of CH, around 10% of those affected will transition from episodic CH to chronic CH, and that same number may change from chronic CH to episodic CH. One third will suffer for around 20 years and then experience a complete remission. In another one third the attacks will be mild and no longer require medical intervention. The final one third continue to have attacks in the same pattern and to the same degree. CHs do not begin in old age.
SECONDARY HEADACHES
In cervicogenic headache, pain is localized to the neck and occipital regions and may project to the forehead, orbital region, temples, vertex, or ears. Pain is precipitated or aggravated by special neck movements or sustained neck posture. There can be resistance to or limitation of passive neck movements, and texture, tone, or pain response to active and passive stretching and contraction. Radiologic examination reveals at least one of the following: movement abnormalities in flexion/extension, abnormal posture, fractures, congenital abnormalities, bone tumors, rheumatoid arthritis, or other distinct pathology (but not spondylosis or osteochondrosis, because these are conditions that are common incidental findings in asymptomatic individuals). The criteria do not insist that the lesion be located within the neural territory of C1 to C4 but that it be in the neck.81 The mechanism, which may be related to disk disease in the lower neck, may involve compensatory increase in movement, especially in the facet joints in the upper cervical spine, thus causing pain to travel along the C1 to C4 nerve to the interface of the trigeminal complex. It is believed that nociceptive impulses and other sensory impulses can be involved in sensitization of the brainstem and thus be involved in the spread of pain into the head as described earlier.
Disorders of the cervical spine and atlantooccipital junction may create localized pain and referred pain to the head. Muscles innervated by C1 to C3 all attach to the head and are tender near their attachment sites. Spasm of the posterior neck muscles and pericranial muscles is believed to be a source of pain, but the evidence is mixed. Abnormal posture of the trunk and neck may trigger headache. Alignment of the head in relationship to the cervical spine will affect the length and tension of the muscles and ligaments. When the upper back is rounded, the position of the head is adjusted to maintain the eyes in the vertical position. This often results in shortening of the suboccipital muscles and abnormal positioning of the cervical spine.12 Trigger point pathology of the upper cervical muscles, innervated by C1 to C3, appears to be associated with headache and needs to be distinguished from upper cervical joint pain.
Occipital neuralgia can be related, or a single-point complaint that is isolated to the greater or lesser occipital nerve distribution. It includes the presence of a constant deeper burning pain with occasional shooting pains. Paresthesia and numbness over the occipital scalp are usually present. Occipital neuralgia is believed to arise from trauma or entrapment of the occipital nerve as it goes through the suboccipital muscles, or it may be related to the C2 spinal root.
Temporomandibular joint (TMJ) dysfunction should be ruled out as a cause of tension headache. Damage to alar ligaments can cause headache and can be ruled out with use of functional CT.
The C2-C3 apophyseal joint is suspected of producing cervicogenic headache related to its biomechanical vulnerability as a transitional joint between the C1 and C2 vertebra, which rotate the head, and the C3 to C7 vertebra, which flex and extend the neck. It bears both vectors of stress and the result is a mechanical irritation of C3; this joint and the third occipital nerve appear most vulnerable to trauma from acceleration-deceleration or whiplash injuries. If that is the case, blockades of C3 can relieve headache pain.1
It is difficult to diagnosis cervicogenic headache, because there is overlap in the symptoms, including unilateral, occipital pain radiating to the upper cervical area. Head movement can increase migraine pain related to allodynia but may not be the primary trigger.21 There is no consensus on what constitutes an adequate physical examination of the neck or the techniques for performing the examination. It is not even clear what findings are significant.27 Passive neck positioning in extension and rotation toward the side of pain or digital pressure to the involved facet regions or occipital nerve are typically reported as painful.40 Muscular trigger points are usually found in the suboccipital, cervical, and shoulder musculature and may refer to the head. These findings are in the absence of neurologic findings of cervical radiculopathy, although the patient might report scalp paresthesia or dysesthesia.100,103
MEDICAL MANAGEMENT
The failure to demonstrate conclusively a specific disease or dysfunction of the neck is an impediment to specific treatment for the individual with possible cervicogenic headache.82
Cervical epidural steroid injections may be indicated for multilevel disk or spine degeneration. Nerve blocks, trigger point injections, or radiofrequency thermal neurolyis may block the cascade of sensitization to the central mechanisms. Surgical interventions such as neurotomy, dorsal rhizotomy, and microvascular decompression of nerve roots to perform blocks or reduce pressure on a nerve often also provide only temporary relief, with the possibility of longer intensification of pain. Nerve stimulators have been used successfully in migraine and may be of some benefit in cervicogenic headache.7
Tricyclic antidepressants have been used at a lower dosage than that required for the treatment of patients with depression. Simple analgesics may be used to manage pain but have long-term side effects that should be considered.
Muscle relaxants may be of benefit as they relate to the central nervous system.42
Studies using botulinum toxin have been mixed, and this agent may be more effective if the “follow the pain” strategy is used, injecting the muscles that are reported as tender.41
Physical and manual therapies are important modalities for treatment of cervicogenic headache. Long-term prevention and control of headaches appears greatest in individuals who are involved in ongoing exercise and physical conditioning programs.8,47
Posttraumatic Headache Syndromes
Overview, Definition, and Risk Factors
Posttraumatic headache syndrome is not a single pathology but a group of traumatically induced disorders with overlapping symptoms. The headache is a manifestation of the brain dysfunction and can have contribution from the musculoskeletal injuries. The headache can last longer after the trauma than the musculoskeletal problems.59 When the headache develops within 7 days of the trauma and resolves within 3 months, it is considered acute. The headache can begin weeks or months after the trauma and can persist for years. This headache is considered chronic. The risk of developing chronic posttraumatic headache is greater for mild or moderate head injury than for severe head injury.43
Pathogenesis
The mechanisms of posttraumatic headache mirror those of primary headaches. Both central and peripheral sensitization may play a role in posttraumatic headache. Neurochemical changes similar to those found in migraine include excessive release of excitatory amino acids, especially glutamate; an increase in extracellular potassium, intracellular sodium, or calcium; and an abnormal accumulation of serotonin. There is a decline in magnesium levels, abnormalities in nitric oxide formation, and changes in catecholamines and endogenous opioids. Chronic local inflammation can release substance P, bradykinin, and other pain-producing peptides. The nerve endings responding to these neurochemicals can trigger spasm and pain.32
Diffuse axonal injury resulting from the shear forces to the brain and brainstem remain difficult to image. With newer MRI techniques, PET, and single-photon emission computed tomography (SPECT), the parenchymal delayed cerebral atrophy and axonal damage related to membrane injury may be identified. The ability to detect metabolic brain changes using magnetic resonance spectroscopy will help to determine the level of brain damage sustained. Xenon inhalation techniques provide additional insight into brain perfusion abnormalities.
The connection between the cervical nerves (C1 to C3) and the cervical portion of the trigeminal nucleus forming the trigeminocervical nucleus may play a role in a similar manner as in both tension-type headache and migraine. Pain from the upper cervical nerves is then referred to the ophthalmic division of the trigeminal nerve, and the pain is felt in the orbital, frontal, and temporal regions. Although there is no direct head trauma with some whiplash injury, studies show that these accidents can cause cerebral hemorrhages on the surface of the brain and upper cervical spinal cord.
Clinical Manifestations
Posttraumatic tension-type headaches are generally mild to moderate with dull, aching, pressing, tightening feelings that are usually not pulsating. They are often bilateral and bandlike. These headaches do not involve nausea or vomiting and are not aggravated by bright lights.
Posttraumatic migrainelike headaches can become incapacitating when combined with other sequelae of brain injury. They are described as pulsating or throbbing. They trigger nausea and vomiting, and they are triggered by bright lights, noise, and odor. Increased activity can make the headache worse, and often the challenge of daily tasks can be overwhelming.
Posttraumatic dysautonomic cephalalgia is a result of neck injury and is similar to migraine. The pain is unilateral and throbbing, and there are pupillary dilation and excessive sweating on the side of the headache during the headache phase. Miosis can occur between headaches.93
Posttraumatic clusterlike headaches appear as CHs do. Trauma may trigger a cluster period during what had been a remission.
Cervicogenic headache related to cervical acceleration/deceleration syndrome is associated with an injury to the neck without a direct strike to the head. Usually there is both shear damage to the brain and musculoskeletal damage to the soft tissues in the neck, jaw, and scalp. There are complaints of neck pain, headache, visual disturbances, dizziness, weakness, paresthesias, and concentration and memory disturbances as well as psychologic symptoms. Complaints of memory and concentration disturbances after whiplash injury is common.
Dizziness and vertigo are often reported along with headache. When the dizziness is triggered by activity, the headache can be worse. In an effort to control the sensation of dizziness that comes with head position changes or when there is excessive visual stimulation, the individual will co-contract the muscles of the neck to limit stimulation of the vestibular system resulting in a cervicogenic headache. See Chapter 38 for further description of the mechanism of cervicogenic dizziness. Box 37-4 lists the typical complaints associated with posttraumatic headaches.
Headache status before trauma does not make one more or less likely to develop headache after trauma. But the incidence of headache after trauma is high regardless of prior experience with headache. Mechanical impact factors such as an abnormal position of the head (i.e. rotation) increase the risk of posttraumatic headache.
MEDICAL MANAGEMENT
The treatment of posttraumatic headaches proceeds in the manner described earlier for the type of headache that is manifested. Early and intensive treatment may be the main factors in stopping headache and preventing a chronic condition. Recognizing this type of headache is critical in the course of treatment of an individual with brain injury related to a blow to the head or other causes of whiplash. These headaches are often not recognized by the medical practitioners who provide the follow-up care. Individuals with persistent complaints of headache are often assigned the label of malinger or symptom magnifier.
Medication Overuse Headaches
Frequent intake of medications used for headache can lead to chronic headache. It appears that all drugs used for the treatment of headache may lead to chronic headache. Triptan overuse is common, and the time of onset of chronicity is shorter compared to ergots and analgesics. Women appear to be more prone to overuse than men. Migraineurs are most susceptible to headache related to excessive medication. If a migraineur is taking an analgesic for arthritis, for example, there can be an increase in migraine; this is not seen as significantly with CHs, and an individual taking medication for arthritis who has no history of headache does not begin to have headaches because of the medication.105 Sensitization of receptors and changes in receptor thresholds may explain the phenomenon. Circumstances in which there is more consequence to the headache, as before an important date or during a period of work that should not be interrupted, and the instruction to take the medication as early as possible at the onset of a headache may lead to excessive use of medications.80
Overuse of analgesic medications resulting in analgesic-rebound phenomenon is common when the headache type has not been identified appropriately.106 Intervention by therapists to relieve the mechanical contribution to cervical pain is critical, in addition to pharmacologic treatment.107
Headaches in the Elderly
Primary headaches continue into the later decades of life, with a decrease noted in migraine, cluster headache, and hemicrania. New-onset headaches in the elderly that are related to another disorder are more common than in the young. Increased use of nitrates for control of angina can cause headache. Chronic daily headache often persists into old age. Hypertension and atherosclerotic and hemorrhagic cerebrovascular disease can produce new-onset headache in the elderly.28 Diseases of the aged such as chronic obstructive lung disease can cause dull, throbbing headaches. Anemia, the hypercalcemia related to malignancy, and chronic renal failure can all be a cause of headache. There is a dull headache that can be a part of the pathology of Parkinson’s disease and can clear with the use of antiparkinson medication. The incidence of intracranial disease increases with age, with tumors and subdural hematomas creating space-occupying lesions (see Chapters 30 and 32). Cervical spondylosis and TMJ related to loss of teeth increase with age and may be a cause of headache pain. Depression, which is often comorbid with headache, may be underreported in the aged, and when the headache is treated with antidepressants, both conditions are improved.
Temporal Arteritis
Headache is the most common symptom in temporal arteritis, and this diagnosis should be considered in all adults older than 50 years who have headache or facial pain. Symptoms of vasculitis and vascular insufficiency are common relating to extracranial carotid circulation. Box 37-5 gives the typical complaints related to temporal arteritis.
Temporal arteritis may be related to widespread rheumatologic involvement such as polymyalgia rheumatica. These symptoms may be rather nonspecific and include malaise and easy fatigability, weight loss, anorexia, and unexplained fevers, as well as proximal myalgias.
The headache of temporal arteritis classically is located over a branch of the superficial temporal artery and is described as a dull ache that persists throughout the day. It may be accompanied by tenderness of the artery and overlying scalp. If severely affected, the artery may be nonpulsatile.
Sedimentation rate should be measured to rule out temporal arteritis in individuals older than 60 years who have headache or facial pain, unless the pain obviously results from another cause. Visual loss may result and can be avoided with early diagnosis and aggressive treatment. Findings from a temporal artery biopsy can confirm the diagnosis. Unless otherwise contraindicated, corticosteroid therapy should be instituted, pending results of the biopsy.
Sinus Headaches
The diagnosis of pain that results from acute sinus inflammation is rarely difficult. Often a prior history of sinus inflammation or respiratory allergies is elicited. In general, the pain is of low to moderate intensity and is present on a daily basis. The pain usually is localized to the frontal or maxillary area, and there is tenderness to tapping over the affected sinus. The pain is often worsened by bending forward and may be triggered by blowing the nose or sneezing. Symptoms of nasal stuffiness are usually present, and mucopurulent drainage from the nostrils may be seen. If the nasal passages are blocked, use of a nasal decongestant can be useful diagnostically and often results in discharge. In doubtful cases, a simple plain film of the sinuses or an opinion from an otolaryngologist should be obtained.
Postdural Puncture Headaches
Another disabling headache can occur after lumbar puncture or with epidural anesthesia. If a myelogram is performed, it can trigger this type of headache. The headache is due to leakage of cerebrospinal fluid and changes in the pressure within the skull. The headache is usually self-limiting but can last 3 to 5 days. An individual with this type of headache must lie still, because movement, including Valsalva maneuver, increases pain. Hydration helps, in combination with analgesics and antiemetics. Intravenous caffeine sodium benzoate constricts cerebral blood vessels. An epidural blood patch may be necessary. Blood taken from the peripheral vein and put into the epidural space clots and forms a patch. There is increased risk of pain, infection, and spinal cord compression. An epidural blood patch cannot be used in persons taking anticoagulants. Aspirin is not used as an analgesic due to its anticoagulant properties.11
References
1. Alexander, MP. In the pursuit of proof of brain damage after whiplash injury. Neurology. 1998;51:336–340.
2. Aminoff, MJ. Nervous system. In: Tierney LM, McPhee SJ, Papadakis MA, eds. Current medical diagnosis and treatment. ed 3. Norwalk, CT: Appleton & Lange; 1994:799–854.
3. Bendtsen, L, Treed, RD. Sensitization of myofascial pain pathways in tension-type headaches. In: Olesen J, Goadsby PJ, Ramadan NM, et al, eds. The headaches. ed 3. Philadelphia: Lippincott Williams & Wilkins; 2006:637–643.
4. Reference deleted in pages.
5. Bigel, M, Welch, KMA. Basilar migraines and retinal migraines. In: Olesen J, Goadsby PJ, Ramadan NM, et al, eds. The headaches. ed 3. Philadelphia: Lippincott Williams & Wilkins; 2006:589–598.
6. Black, DF, Bordini, CA, Russell, D. Symptomatology of cluster headache. In: Olesen J, Goadsby PJ, Ramadan NM, et al, eds. The headaches. ed 3. Philadelphia: Lippincott Williams & Wilkins; 2006:789–797.
7. Biondi, DM. Physical treatments for headache: a structured review. Headache. 2005;45:1–9.
8. Biondi, DM. Cervicogenic headache: a review of diagnostic and treatment strategies. J Am Osteopath Assoc. 2005;105(4 suppl):16–22.
9. Berg, J, Ramadan, NM. Societal burden of the headache. In: Olesen J, Goadsby PJ, Ramadan NM, et al, eds. The headaches. ed 3. Philadelphia: Lippincott Williams & Wilkins; 2006:35–42.
10. Campbell, JK, Zagami, AB. Status migrainosus. In: Olesen J, Tfelt-Hansen P, Welch KM, eds. The headaches. ed 2. Philadelphia: Lippincott Williams & Wilkins; 2000:525–528.
11. Connolly, MA. Postdural puncture headache. Am J Nurs. 2000;99(11):48–49.
12. Cutrer, FM, Baloh, RW. Migraine-associated dizziness. Headache. 1992;32(6):300–304.
13. Dahlof, CG, Sazena, PR. Novel compounds in development for acute treatment of migraine. In: Olesen J, Tfelt-Hansen P, Welch KM, eds. The headaches. ed 2. Philadelphia: Lippincott Williams & Wilkins; 2000:439–443.
14. Dahlof, CG, Solomon, GD. Impact of the headache on the individual and family. In: Olesen J, Goadsby PJ, Ramadan NM, et al, eds. The headaches. ed 3. Philadelphia: Lippincott Williams & Wilkins; 2006:27–34.
15. Dalessio, DJ, Silberstein, SD. Diagnosis and classification of headache. In: Dalessio DJ, Silberstein SD, eds. Wolff’s headache and other head pain. ed 6. New York: Oxford University Press; 1993:3–18.
16. Davidoff, RA. Migraine: manifestations, pathogenesis, and management, ed 2. New York: Oxford University Press, 2002.
17. Diamond, S. Tension-type headaches. In: Dalessio DJ, Silberstein SD, eds. Wolff’s headache and other head pain. ed 6. New York: Oxford University Press; 1993:235–254.
18. Diamond, S, Dalessio, DJ. Migraine headache. In: Diamond S, Dalessio DJ, eds. The practicing physician’s approach to headache. ed 5. Baltimore: Williams & Wilkins; 1992:51–79.
19. Dodick, DW, Campbell, JK. Cluster headache: diagnosis, management, and treatment. In: Silberstein SD, Dalessio DJ, Lipton RB, eds. Wolff’s headache and other head pain. ed 7. New York: Oxford University Press; 2001:283–309.
20. Reference deleted in pages.
21. Drottning, M, Staff, PH, Sjaastad, O. Cervicogenic headache after whiplash injury. Cephalalgia. 1997;17:228–289.
22. Drummond, PD. Psychological mechanisms of migraine. In: Olesen J, Tfelt-Hansen P, Welch KM, eds. The headaches. ed 2. Philadelphia: Lippincott Williams & Wilkins; 2000:313–317.
23. Drummond, PD, Alessandri, M. Neurophysiology and autonomic dysfunction in cluster headaches. In: Olesen J, Goadsby PJ, Ramadan NM, et al, eds. The headaches. ed 3. Philadelphia: Lippincott Williams & Wilkins; 2006:767–774.
24. Reference deleted in pages.
25. Ducros, A, Campbell, JK. Familial hemiplegic migraine. In: Olesen J, Tfelt-Hansen P, Welch KM, eds. The headaches. ed 2. Philadelphia: Lippincott Williams & Wilkins; 2000:501–505.
26. Edeling, J. Manual therapy for chronic headache, ed 2. Oxford: Butterworth-Heinemann, 1994.
27. Edmeads, JG. Disorders of the neck: cervicogenic headache. In: Silberstein SD, Dalessio DJ, Lipton RB, eds. Wolff’s headache and other head pain. ed 7. New York: Oxford University Press; 2001:447–459.
28. Edmeads, JG, Wang, SJ. Headaches in the elderly. In: Olesen J, Goadsby PJ, Ramadan NM, et al, eds. The headaches. ed 3. Philadelphia: Lippincott Williams & Wilkins; 2006:1105–1110.
29. Ekbom, K, Nappi, G. Diagnosis, differential diagnosis and prognosis of cluster headache. In: Olesen J, Tfelt-Hansen P, Welch KM, eds. The headaches. ed 2. Philadelphia: Lippincott Williams & Wilkins; 2000:725–729.
30. Ekbom, K, Solomon, S. Management of cluster headache. In: Olesen J, Tfelt-Hansen P, Welch KM, eds. The headaches. ed 2. Philadelphia: Lippincott Williams & Wilkins; 2000:731–740.
31. Evans, RW, Rozen, T, Adelman. Neuroimaging and other diagnostic testing in headache. In: Silberstein SD, Dalessio DJ, Lipton RB, eds. Wolff’s headache and other head pain. ed 7. New York: Oxford University Press; 2001:27–49.
32. Evans, RW. The postconcussion syndrome and whiplash injuries: a question-and-answer review for primary care physicians. Prim Care. 2004;31(1):1–17.
33. Ferrari, MD, Kraig, RP. Spreading depression. In: Olesen J, Goadsby PJ, Ramadan NM, et al, eds. The headaches. ed 3. Philadelphia: Lippincott Williams & Wilkins; 2006:269–275.
34. Ferrari, MD. Genetics and migraine. In: Olesen J, Goadsby PJ, Ramadan NM, et al, eds. The headaches. ed 3. Philadelphia: Lippincott Williams & Wilkins; 2006:251–261.
35. Garcia, JH. Pathology. In: Olesen J, Tfelt-Hansen P, Welch KM, eds. The headaches. ed 1. New York: Raven Press; 1993:175–177.
36. Goadsby, PJ, Lipton, RB, Ferrari, MD. Migraine current understanding and treatment. N Engl J Med. 2002;346:257–270.
37. Goadsby, PJ. Migraine, aura and cortical spreading depression: why are we still talking about it? Ann Neurol. 2001;49:4–6.
38. Goadsby, PJ, Ramadan, NM. Potential new drugs for the acute and prophylactic treatment of migraines. In: Olesen J, Goadsby PJ, Ramadan NM, et al, eds. The headaches. ed 3. Philadelphia: Lippincott Williams & Wilkins; 2006:569–575.
39. Gotlib, A, Theil, H. A selected annotated bibliography of the core biomedical literature pertaining to stroke, cervical spine manipulation and head-neck movement. J Can Chiropractic Assoc. 1985;29:80–89.
40. Hall, T, Robinson, K. The flexion-rotation test and active cervical mobility a comparative measurement study in cervicogenic headache. Man Ther. 2004;9:197–202.
41. Hobson, DE, Gladish, DF. Botulinum toxin injection for cervicogenic headache. Headache. 1997;37:253–255.
42. Kash, H, Bach, FW, Stengaard-Pedersin, K, et al. Development of pain and neurological complaints after whiplash: a 1-year prospective study. Neurology. 2003;60:743–748.
43. Keidel, M, Ramadan, NM. Acute posttraumatic headaches. In: Olesen J, Goadsby PJ, Ramadan NM, et al, eds. The headaches. ed 3. Philadelphia: Lippincott Williams & Wilkins; 2006:863–872.
44. Jay, GW. The headache handbook: diagnosis and treatment. Boca Raton, FL: CRC Press, 1999.
45. Jelinski, SE. Factors associated with depression in patients referred to headache specialists. Neurology. 2007;68(7):489–495.
46. Jensen, R. Pathophysiological mechanisms of tension-type headache: a review of epidemiological and experimental studies. Cephalalgia. 1999;19(6):602–621.
47. Jull, G, Stanton, WR. Predictors of responsiveness to physiotherapy management of cervicogenic headache. Cephalagia. 2005;25:101–108.
48. Kayan, A, Hood, JD. Neuro-otological manifestations of migraine. Brain. 1984;107:1123–1142.
49. Kudrow, L. Cluster headache: diagnosis, management, and treatment. In: Dalessio DJ, Silberstein SD, eds. Wolff’s headache and other head pain. ed 6. New York: Oxford University Press; 1993:171–197.
50. Lance, JW. The pathophysiology of migraine. In: Dalessio DJ, Silberstein SD, eds. Wolff’s headache and other head pain. ed 6. New York: Oxford University Press; 1993:59–95.
51. Lempert, T. Migrainous vertigo. Neurol Clin. 2005;23(3):715–730.
52. Leone, M. Deep brain stimulation in headache. Lancet Neurol. 2006;5(10):873–877.
53. Lipton, RB, Stewart, WF, Celentano, DD, et al. Migraine diagnosis and treatment: results from the American Migraine Study II. Headache. 2001;41:638–645.
54. Lipton, RB, Hamelsky, SW, Stewart, WF. Epidemiology and impact of headache. In: Silberstein SD, Dalessio DJ, Lipton RB, eds. Wolff’s headache and other head pain. ed 7. New York: Oxford University Press; 2001:85–107.
55. Reference deleted in pages.
56. Jensen, R, Becker, WJ. Symptomatology of episodic tension-type headaches. In: Olesen J, Goadsby PJ, Ramadan NM, et al, eds. The headaches. ed 3. Philadelphia: Lippincott Williams & Wilkins; 2006:685–693.
57. Magee, DJ. Orthopedic physical assessment, ed 2. Philadelphia: Saunders, 1992.
58. Maitland, GD. Vertebral manipulation, ed 5. Oxford: Butterworth-Heinemann, 1986.
59. Marcus, E, Scharff, L, Mercer, MA, et al. Musculoskeletal abnormalities in chronic headache: a controlled comparison of headache diagnostic groups. Headache. 1999;39:21–27.
60. Massiou, H, MacGregor, EA. Influence of female hormones on migraines. In: Olesen J, Goadsby PJ, Ramadan NM, et al, eds. The headaches. ed 3. Philadelphia: Lippincott Williams & Wilkins; 2006:331–342.
61. Massoiou, H, Bousser, MG. Influence of female hormones on migraine. In: Olesen J, Tfelt-Hansen P, Welch KM, eds. The headaches. ed 2. Philadelphia: Lippincott Williams & Wilkins; 2000:261–267.
62. Matthew, NT, Ashina, M. Acute pharmacology of tension type headaches. In: Olesen J, Goadsby PJ, Ramadan NM, et al, eds. The headaches. ed 3. Philadelphia: Lippincott Williams & Wilkins; 2006:727–735.
63. Matthew, NT, Bendtsen, L. Prophylactic pharmacotherapy of tension-type headache. In: Olesen J, Tfelt-Hansen P, Welch KM, eds. The headaches. ed 2. Philadelphia: Lippincott Williams & Wilkins; 2000:667–673.
64. McGrath, PJ, Holroyd, KA, Sorbi, MJ. Psychological and behavioral treatments of migraine. In: Olesen J, Tfelt-Hansen P, Welch KM, eds. The headaches. ed 2. Philadelphia: Lippincott Williams & Wilkins; 2000:371–378.
65. Merikangas, KR, Low, NCD, Rassmussen, BK. Migraine comorbidity. In: Olesen J, Goadsby PJ, Ramadan NM, et al, eds. The headaches. ed 3. Philadelphia: Lippincott Williams & Wilkins; 2006:243–250.
66. Merikangas, KR, Rasmussen, BK. Migraine comorbidity. In: Olesen J, Tfelt-Hansen P, Welch KM, eds. The headaches. ed 2. Philadelphia: Lippincott Williams & Wilkins; 2000:235–240.
67. Messlinger, K, Burstein, R. Anatomy of central nervous system pathways related to head pain. In: Olesen J, Tfelt-Hansen P, Welch KM, eds. The headaches. ed 2. Philadelphia: Lippincott Williams & Wilkins; 2000:77–86.
68. Messlinger, K, Dostrovsky, JO, Strassman, AM. Anatomy and physiology of head pain. In: Olesen J, Goadsby PJ, Ramadan NM, et al, eds. The headaches. ed 3. Philadelphia: Lippincott Williams & Wilkins; 2006:95–110.
69. Nappi, G, Russell, D. Symptomatology of cluster headache. In: Olesen J, Tfelt-Hansen P, Welch KM, eds. The headaches. ed 2. Philadelphia: Lippincott Williams & Wilkins; 2000:717–723.
70. Olesen, J, Cutrer, FM. Migraine with aura and its subforms. In: Olesen J, Tfelt-Hansen P, Welch KM, eds. The headaches. ed 2. Philadelphia: Lippincott Williams & Wilkins; 2000:345–357.
71. Olesen, J, Goadsby, PJ. Synthesis of migraine mechanisms. In: Olesen J, Tfelt-Hansen P, Welch KM, eds. The headaches. ed 2. Philadelphia: Lippincott Williams & Wilkins; 2000:331–336.
72. Olesen, J, Welch, KMA. Tension-type headache: introduction. In: Olesen J, Goadsby PJ, Ramadan NM, et al, eds. The headaches. ed 3. Philadelphia: Lippincott Williams & Wilkins; 2006:619–621.
73. Olesen, J, Goadsby, PJ. Synthesis of migraine mechanisms. In: Olesen J, Goadsby PJ, Ramadan NM, et al, eds. The headaches. ed 3. Philadelphia: Lippincott Williams & Wilkins; 2006:393–399.
74. Olesen, J, Lipton, RB. Classification of headache. In: Olesen J, Goadsby PJ, Ramadan NM, et al, eds. The headaches. ed 3. Philadelphia: Lippincott Williams & Wilkins; 2006:9–16.
75. Parker, W. Migraine and the vestibular system in adults. Am J Otol. 1991;12:25–34.
76. Pradalier, LA. Immunologic aspects of migraine. In: Olesen J, Tfelt-Hansen P, Welch KM, eds. The headaches. ed 2. Philadelphia: Lippincott Williams & Wilkins; 2000:277–281.
77. Rasmussen, BK, Stewart, WF. Epidemiology of migraine. In: Olesen J, Tfelt-Hansen P, Welch KM, eds. The headaches. ed 2. Philadelphia: Lippincott Williams & Wilkins; 2000:227–233.
78. Rassmussen, BK. Epidemiology of migraine. In: Olesen J, Goadsby PJ, Ramadan NM, et al, eds. The headaches. ed 3. Philadelphia: Lippincott Williams & Wilkins; 2006:235–242.
79. Rasmussen, BK, Jensen, R, Shroll, M, et al. Epidemiology of headache in a general population a prevalence study. J Clin Epidemiol. 1991;44:1147–1157.
80. Relja, G, Granato, A, Atonello, RM, et al. Headache induced by chronic substance use: analysis of medication overused and minimum dose required to induce headache. Headache. 2004;44:148–153.
81. Ronnen, HR, deKorte, PJ, Brink, PR, et al. Acute whiplash injury: is there a role for MR imaging? A prospective study of 100 patients. Radiology. 1996;201:93–96.
82. Rosenfeld, M, Gunnarsson, R, Borenstein, P. Early intervention in whiplash-associated disorders. Spine. 2000;25:1782–1787.
83. Russell, MB. Genetics of cluster headache. In: Olesen J, Tfelt-Hansen P, Welch KM, eds. The headaches. ed 2. Philadelphia: Lippincott Williams & Wilkins; 2000:679–682.
84. Saxena, PR, Tfelt-Hansen, P. Triptans, 5 HT 1B/1D receptor agonists in the acute treatment of migraine. In: Olesen J, Tfelt-Hansen P, Welch KM, eds. The headaches. ed 2. Philadelphia: Lippincott Williams & Wilkins; 2000:411–433.
85. Shechter, AL, Lipton, RB, Silberstein, SD. Migraine comorbidity. In: Silberstein SD, Dalessio DJ, Lipton RB, eds. Wolff’s headache and other head pain. ed 7. New York: Oxford University Press; 2001:108–120.
86. Schwarts, B, Stewart, WF, Simon, D, et al. Epidemiology of tension-type headache. JAMA. 1998;279(5):381–383.
87. Silberstien, SD, Lipton, RB. Chronic daily headache, including transformed migraine, chronic tension-type headache and medication overuse. In: Silberstein SD, Dalessio DJ, Lipton RB, eds. Wolff’s headache and other head pain. ed 7. New York: Oxford University Press; 2001:247–283.
88. Silberstein, SD, Saper, JR. Migraine: diagnosis and treatment. In: Dalessio DJ, Silberstein SD, eds. Wolff’s headache and other head pain. ed 6. New York: Oxford University Press; 1993:96–170.
89. Silberstein, SD, Tfelt-Hansen. Antiepileptic drugs in migraine prophylaxis. In: Olesen J, Goadsby PJ, Ramadan NM, et al, eds. The headaches. ed 3. Philadelphia: Lippincott Williams & Wilkins; 2006:545–552.
90. Sjaastad, O. Cluster headache syndrome. London: Saunders, 1992.
91. Steiner, TJ, Tfelt-Hansen, P. Antiepileptic drugs in migraine prophylaxis. In: Olesen J, Tfelt-Hansen P, Welch KM, eds. The headaches. ed 2. Philadelphia: Lippincott Williams & Wilkins; 2000:483–487.
92. Swerdlow, B. Whiplash and related headaches. Boca Raton, FL: CRC Press, 1999.
93. Tfelt-Hansen, P. Antiemetic, prokinetic, neuroleptic and miscellaneous drugs in the acute treatment of migraine. In: Olesen J, Tfelt-Hansen P, Welch KM, eds. The headaches. ed 2. Philadelphia: Lippincott Williams & Wilkins; 2000:445–451.
94. Tfelt-Hansen, P, Saxena, PR. Ergot alkaloids in the acute treatment of migraine. In: Olesen J, Tfelt-Hansen P, Welch KM, eds. The headaches. ed 2. Philadelphia: Lippincott Williams & Wilkins; 2000:399–410.
95. The International Classification of Headache disorders. Cephalagia. 2004;24(suppl 1):1–160. [2nd edition].
96. Toda, N, Tfelt-Hansen, P. Calcium antagonists in migraine prophylaxis. In: Olesen J, Tfelt-Hansen P, Welch KM, eds. The headaches. ed 2. Philadelphia: Lippincott Williams & Wilkins; 2000:477–482.
97. Thomsen, LL, Eriksen, MK, Roemer, SF, et al. A population-based study of familial hemiplegic migraine suggests revised diagnostic criteria. Brain. 2002;125:1379–1391.
98. Thomsen, LL, Ostergaard, E, Olesen, J, et al. Evidence for a separate type of migraine with aura: sporadic hemiplegic migraine. Neurology. 2003;60:595–601.
99. Torrelli, P, Manzoni, GC. Pain and behavior in cluster headache: a prospective study and review of the literature. Funct Neurol. 2003;18:205–210.
100. Travell, JG, Simons, DG. Myofascial pain and dysfunction: the trigger point manual. Baltimore: Williams & Wilkins, 1983.
101. Troost, T, Zagami, AS. Ophthalmoplegic migraine and retinal migraine. In: Olesen J, Tfelt-Hansen P, Welch KM, eds. The Headaches. Ed 2. Philadelphia: Lippincott Williams & Wilkins; 2000:511–516.
102. Waldenlind, E, Bussone, G. Biochemistry, cicannual and circadian rhythms, endocrinology and immunology of cluster headache. In: Olesen J, Tfelt-Hansen P, Welch KM, eds. The headaches. ed 2. Philadelphia: Lippincott Williams & Wilkins; 2000:687–696.
103. Winkelstein, B, Nightengale, R, Richardson, WM. The cervical facet capsule and its role in whiplash injury. Spine. 2000;25:1238–1246.
104. World Federation of Neurology. Definition of migraine. In: Cochrane AL, ed. Background to migraine: Third Migraine Symposium. London: Heinemann; 1970:181–182.
105. Zeeberg, P. Probable medication-overuse headache: the effect of a 2-month drug-free period. Neurology. 2006;66(12):1894–1898.
106. Zwart, J, Dyb, G, Hagen, K, et al. Analgesic use: a predictor of chronic pain and medication overuse headache: the Head-HUNT Study. Neurology. 2003;61:160–164.
107. Zwart, J, Dyb, G, Hagen, K, et al. Analgesic overuse among subjects with headache, neck and low back pain. Neurology. 2004;62:1540–1544.