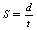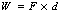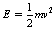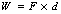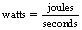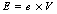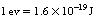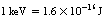Chapter 4 Units of measurement
Chapter contents
4.1 Aim
This chapter introduces the reader to the main units that are used in measurement in radiographic science.
4.2 Units of measurement
Science has three fundamental tools that are used in its attempts to understand the external world:
Problems arise when making experimental measurements as to what quantities to measure and how to measure them. In particular, the units in which these quantities are expressed must be defined so that when two people make the same measurement they get the same results. Also, there are obvious advantages if one set of units is universally adopted as the basis for all measurements.
Each of the base units discussed in the next section relies on the appropriate standard to which each measurement is compared. Thus, there are units of standard length, standard mass, standard time interval and so on. Without such standards, no accurate measurements can be made. This in turn retards the development of adequate theories, or models, of the world.
4.3 SI base units
There are a plethora of units of measurement used throughout the world. The International System of Units (SI) attempts to replace this with seven standard units. These standards are termed SI units (see Table 4.1) and represent the fundamental measurements that we might wish to make of a body:
• What is its size? (unit of length–metre)
• How massive is it? (unit of mass–kilogram)
• How bright is it? (unit of luminous intensity–candela)
• How much electrical current flows through it? (unit of electrical current–ampere)
• How many elementary particles does it contain? (unit of amount of substance–mole)
• How hot is it? (unit of temperature–Kelvin)
• How do all the quantities vary with time? (unit of time–second)
| QUANTITY | UNIT OF MEASUREMENT | SYMBOL |
|---|---|---|
| Length | Metre | m |
| Mass | Kilogram | kg |
| Luminous intensity | Candela | cd |
| Electric current | Ampere | A |
| Amount of a substance | Mole | mol |
| Temperature | Kelvin | K |
| Time | Second | s |
The very precise definitions of the units are not required for the rest of this text, but if you wish to see them, they are given in Appendix C.
The base units of mass, length and time are termed fundamental or base units since one or more of them is always involved in the measurement of any other quantity.
These seven SI base units may be combined to give derived units, as described in the next section.
4.3.1 Derived SI units
A number of derived SI units can be formed by the combination of the seven base units. Some of these are sufficiently important to be given their own names and they are listed in Table 4.2 and discussed in the rest of this chapter.
Table 4.2 Derived SI units and their definitions
| Quantity | DEFINITION | SI UNIT | SCALAR/VECTOR |
|---|---|---|---|
| Speed | Distance travelled in unit time | Metre per second (m.s−1) | Scalar |
| Velocity | Distance travelled in unit time in a given direction | Metre per second (m.s−1) | Vector |
| Acceleration | Change of velocity in unit time | Metre per second (m.s−2) | Vector |
| Force | The application of unit force to unit mass produces unit acceleration | Newton (N) (kg.m.s−2) | Vector |
| Pressure | Force applied per unit area | Pascal (Pa) (N.m−2) | Vector |
| Weight | Force acting on a body due to gravity | Newton (N) (kg.m.s−2) | Scalar |
| Work | Product of the force acting on a body times the distance the body moves | Joule (J) (n.m) | Scalar |
| Energy | Kinetic energy: work which can be done by a system because of its velocity | Joule (J) | Scalar |
| Potential energy: work which can be performed because of the position or state of a system | Joule (J) | ||
| Power | Rate of doing work | Watt (W) (J.s−1) | Scalar |
| Momentum | Product of mass and the velocity of the body | (kg.m.s−1) | Vector |
Further derived units are of a more specialized nature (e.g. absorbed radiation dose) and will be discussed in the specific chapters which require such measurement.
For some quantities, we can consider a body moving between two points. For certain measurements, it is important to know how far the body has travelled between the two points. In other cases, we wish to know not only how far it has travelled but also the direction it has travelled. Measurements where the direction is important are termed vector quantities whereas those where the direction is not important are known as scalar quantities.
4.3.2 Speed and velocity
The speedometer in a car is calibrated in terms of kilometres per hour (kph) or miles per hour (mph). Either of these shows that speed means distance travelled in unit time.
In SI units, speed (S), distance (d) and time (t) are related by the equation:
where d is in metres, t is in seconds and S, therefore, is in metres per second (m.s−l). The speedometer in a car gives no indication of the direction in which the car is moving so we can see that speed is a scalar quantity. Velocity is measured in the same units as speed (m.s−1) but this time the direction of movement is also measured. Thus, a car travelling at a constant speed around a roundabout is continuously changing its velocity.
4.3.3 Acceleration
Acceleration implies a change in velocity and is defined as the change in velocity per unit time (a vector quantity).
For example, the acceleration due to gravity is approximately 9.8 meters per second per second (9.8 m.s−2). This means that for a free-falling body, the velocity increases by 9.8 m.s−1 after each second. Thus, if a body is dropped, its downward velocity is 9.8 m.s−1 after the first second, 19.6 m.s−1 after the next second and so on.
The acceleration due to gravity causes a free-falling body to increase its velocity, but if the accelerating force is in the opposite direction to the direction of movement of the body, then it will cause it to lose velocity. This force causes a negative acceleration or a deceleration.
Also, notice that acceleration is a vector quantity as the acceleration has direction (a vector measurement).
4.3.4 Force
Newton’s second law of motion (see Ch. 3) shows that the net force acting on a body is proportional to the mass of the body multiplied by the acceleration produced on the body. The units of force are therefore kg.m.s−2 in SI units. However, this quantity is sufficiently important to be given its own special name and is known as the newton. This can be defined as follows:
Force is a vector quantity as it has direction. The acceleration produced by the action of the force (also a vector quantity) is in the same direction as the force.
4.3.5 Pressure
Pressure is defined as the force exerted per unit area. The units of pressure would therefore be N.m−2 Again, it is sufficiently important to merit its own unit, known as the pascal, and is defined as follows:
The pressure acting on a body is 1 pascal if 1 newton of force is applied per square metre of body surface.
The difference between force and pressure can be readily appreciated if one considers crossing some snow wearing either shoes or skis. In both cases, the force is the same but this force is applied to a smaller area in the case of shoes (the pressure on the snow is greater) so they tend to sink into the snow.
4.3.6 Weight and mass
As we have already seen, mass is a base SI unit, is defined as the amount of matter in a body and is defined against the standard kilogram. This is sometimes confused with the mole (the amount of substance in a body) and so it is possibly easier to understand the concept of mass if we consider it in terms of inertia. We know that a force acting on a body will produce an acceleration and that the force, the mass and the acceleration are linked by the equation F=m×a (see Equation 3.1) so that inertia can be defined as the body’s resistance to acceleration. From the equation, it can be seen that as the mass of the body increases, so the force required to produce a given acceleration also increases, i.e. the inertia of the body increases with mass.
The weight of a body is the downward force on the body due to the gravitational attraction of Earth. Hence, the weight of a body is expressed in newtons, not in kilograms. An equation similar to Equation 3.1 links weight (w), mass (m) and gravity (g):
From the above discussion and equation, we can see that a body always has mass but it only has weight in the presence of a gravitational field. Hence, a body in deep space has no weight (because of the zero gravitational field) but it has mass as it still requires a force to cause it to change its velocity (i.e. it has inertia).
4.3.7 Work and energy
Both work and energy are measured using the same units. A force is said to do work if it moves its point of application in the same direction as the applied force. This can be expressed as:
where W is work, F is force and d is distance.
Thus, the units of work are newtons and meters (N.m). Again, the concept of work is sufficiently important to merit its own unit, which is the joule.
1 joule of work is performed when a force of 1 newton moves its point of application through a distance of 1 metre.
Energy can be considered as the capacity of a body to do work. There are two types of energy that we need to consider separately:
4.3.7.1 Kinetic energy
Kinetic energy is energy that a body possesses by virtue of its motion. This motion may be translational (movement along a path) or rotational, or a combination of both types. The kinetic energy is simply the work that must be done in the process of bringing the body to rest. We have already looked at this when we considered Newton’s laws of motion and established an equation (Equation 3.4) for the kinetic energy of a body of mass m having a velocity of v.
Consider an electron, which has been accelerated across the X-ray tube and is travelling with a velocity v at the point when it starts to collide with the atoms of the target.
It has a kinetic energy (=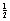 mν2) and then starts to liberate some of that energy in the form of X-ray photons. The energy of X-ray photons is measured in electron-volts (eV).
mν2) and then starts to liberate some of that energy in the form of X-ray photons. The energy of X-ray photons is measured in electron-volts (eV).
4.3.7.2 Potential energy
Potential energy is energy possessed by a body (or a system) by virtue of its condition or state. Thus, a stationary body has potential energy if it is in a condition that allows it to release its stored energy. The potential energy of the system can be thought of as the work that the system will perform in bringing its potential energy to zero. If we consider a body of mass m, which is at a height h above the ground, then we can apply Equation 4.3 to this situation.
The force at work here is the weight of the body (mg) and the distance it can move its height above the ground (h), so we now have the equation for the potential energy (PE):
Consider the case of a hospital lift sitting at the ground floor. Attached to this lift, over a pulley system, is a counterweight. This counterweight has potential energy because of its position. If the brakes on the lift are released, it will assist in moving the lift up to the top floor (i.e. the counterweight performs work).
Work and energy are both scalar quantities, as they do not have direction.
4.3.8 Power
A particular car may reach a speed of 60 mph in 6 s while another car of the same mass does the same speed in 20 s. The first car is said to be more powerful than the second. Assuming that the cars are travelling in the same direction, then their velocity is the same and so their kinetic energy (=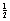 mν2) is the same, but the first car reached that energy more quickly. Hence, power can be expressed as the rate at which energy is expended and, since energy and work are basically the same thing, we can say that:
mν2) is the same, but the first car reached that energy more quickly. Hence, power can be expressed as the rate at which energy is expended and, since energy and work are basically the same thing, we can say that:
Thus, power is measured in joules per second (J.s−1) but is again an important enough concept to merit its own unit, the watt. Thus:
4.3.9 Momentum
In everyday speech, momentum expresses the ability of a moving body to ‘keep going’. This depends on the mass of the body and its velocity and so momentum is defined:
We have already, briefly, come across the concept of momentum in the law of conservation of momentum (Ch. 3).
4.4 Units used in radiography
Many derived SI units (such as the joule, coulomb, etc.) are used in radiographic science. However, other units that do not strictly adhere to the SI system are especially useful to radiography and are unlikely to be discontinued, because of their practical convenience. These are shown and defined in Table 4.3.
Table 4.3 Units used in radiography
| UNIT | DEFINITION |
|---|---|
| mA | The average electrical current passing through an X-ray tube during an exposure (measured in milliamperes) |
| mAs | The average current passing through the tube during an exposure multiplied by the exposure time in seconds (1 mAs=1 millicoulomb) |
| keV | The energy imparted to an electron when passing through a potential difference of 1 kV in a vacuum |
4.4.1 mA and mAs
X-rays are produced in an X-ray tube when electrons from the cathode, with high kinetic energy, strike the anode (see Chs 21 and 30). If we assume that each electron has a chance of producing X-rays, then the intensity of X-ray production is proportional to the number of electrons striking the anode per second. The number of electrons flowing per second is related to the current flowing through the tube (see Ch. 8). The SI unit of current is the ampere (A), as we saw earlier in this chapter, and it is equivalent to a current of 6×1018 electrons per second. This unit of current is too large a unit for radiography so current is measured in milliamperes (1 mA=10−3 A).
We only wish the tube to produce X-rays in sufficient quantity to produce the image on the recording medium (e.g. film) and so an exposure time is selected by the operator. It can be seen that, all other factors remaining constant, the amount of blackening (or optical density) of the film will be determined by the number of X-rays leaving the tube. From the above arguments, this is determined by the total number of electrons striking the anode of the tube. The total number of electrons striking the target (and hence the X-ray output) is determined by the number of electrons flowing in unit time (related to the mA) and the length of time for which the current flows (the exposure time in seconds, s). The X-ray output from a tube (if all other factors remain unaltered) is determined by the mAs.
Any combination of mA and time which produces a given mAs will result in the same quantity of X-rays being emitted by the tube. If 60 mAs was required to produce an acceptable image, this could be delivered in the ways listed in Table 4.4.
Table 4.4 Different combinations of mA and time to produce a given mAs
| CURRENT (mA) | EXPOSURE TIME (S) | mAs |
|---|---|---|
| 10 | 6.0 | 60 |
| 20 | 3.0 | 60 |
| 100 | 0.6 | 60 |
| 200 | 0.3 | 60 |
| 300 | 0.2 | 60 |
| 600 | 0.1 | 60 |
The reasons why we might wish to use the different combinations of mA and time will be discussed in Chapter 25.
As we shall see in Chapter 7, the mAs is equivalent to the millicoulomb (10−3 C), which is the unit of electrical charge. The mAs is, however, used in preference to the millicoulomb as it makes it more obvious that this can be altered by altering the tube current (mA) or the exposure time (s).
4.4.2 keV
As we have already seen in this chapter, energy is measured in joules. When we come to consider the energies involved in the atom or the energies of the photons of the X-ray beam, the coulomb is an extremely large unit. The electron-volt (eV) and the kiloelectron-volt (keV=103 eV) are much more convenient units of measurement for such energies.
If an electron is accelerated from rest across a potential difference of 1 volt in a vacuum, it gains a kinetic energy of 1 electron-volt.
Similarly, an electron accelerated from rest across a potential difference of 1 kilovolt in a vacuum gains a kinetic energy of 1 kiloelectron-volt. The energy (E) in joules can be calculated from the equation:
In this equation, e is the charge on the electron (1.6×10−19 C) and V is the potential difference measured in volts. Thus:
If 75 kVp is selected by an operator for a specific exposure, then some of the electrons travelling across the X-ray tube will have a kinetic energy of 75 keV when they strike the anode. If we assume that some of these electrons give up all their energy as a single X-ray photon, then the energy of this photon will be 75 keV and it will represent the maximum photon energy in this beam. Thus, by altering the kVp, the operator can alter the maximum photon energy of the beam.
In this chapter, we considered the following factors related to units of measurement:
• The SI base units for length, mass, luminous intensity, electric current, amount of substance, temperature and time.
• The SI derived units for speed, velocity, acceleration, force, pressure, weight, work, energy, power and momentum.
• Units of measurement used in radiography in the form of mA, mAs and keV.