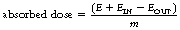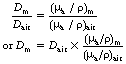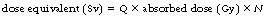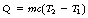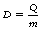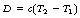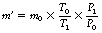Chapter 27 Principles of radiation dosimetry
Chapter contents
27.1 Aim
The aim of this chapter is to introduce the reader to the concepts of exposure, absorbed dose and dose equivalent. It then goes on to consider methods of absolute measurement of radiation dose and different relative methods of dose measurement.
27.2 Introduction
As we saw in Chapter 1 of this book, we live in an environment where we are continuously subjected to ionizing radiation from natural causes such as cosmic rays and naturally occurring radionuclides. In fact, about 90% of the average UK radiation dose comes from natural sources. In addition, there are artificial contributions to the radiation dose because of fallout from weapons testing, leakage from nuclear power plants, manufacture of radionuclides and medical exposure to radiation. All ionizing radiations, whether natural or artificial, constitute a hazard. It is assumed that the greater the radiation dose to which the population is exposed, the greater the hazard. The accurate measurement of radiation dose received by the population is therefore important in trying to quantify the hazard. As can be seen from Figure 1.4, medical radiation constitutes the largest single contribution of the artificial radiation exposure to the population in the UK and so it is important to minimize this radiation dose and hence the total population dose. However, the hazards associated with medical irradiation must be considered against the benefits of diagnosis and treatment. This risk–benefit concept will be discussed in Chapter 44.
27.3 Units of exposure and dose
When an X-ray beam passes through air, it produces excitation and ionization of the air molecules. The electrons ejected in this first interaction (e.g. during photoelectric absorption) can have sufficient energy to ionize other atoms and so produce more electrons – the delta rays. Delta rays are responsible for the great majority of ionizations, often referred to as secondary ionizations. The net effect on the air is:
• the formation of electrical charges in the air by ionization
• the absorption of energy by the air as the electrical charges are slowed down by collision with the air molecules (thus producing further ionization)
• the consequent production of heat energy because of the transfer of energy to the air molecules.
The traditional measure of exposure concerns the first of these effects only and is a measure of the amount of ionization that occurs in air. The unit of exposure is defined as:
The exposure at a particular point in a beam of X or gamma radiation is the ratio Q/m, where Q is the total electrical charge of one sign produced in a small volume of air of mass m.
The units of exposure are coulombs per kilogram (C.kg−1) of air. It is important to remember that exposure can only be defined for air and only for X or gamma radiation.
Exposure rate (C.kg−1.s−1) is a measure of the intensity of a beam of given quality since the greater the number of photons at a given energy passing through unit area, the greater the amount of ionization of air in unit time.
In air, the proportions of ionization and heat produced by the absorption of radiation are approximately constant and therefore do not depend on the energy of the radiation. The total amount of ionization produced in air is proportional to the energy absorbed from the beam, e.g. the average energy required to produce ionization in air is about 33 eV, so an X-ray photon of energy 33 keV which is fully absorbed in air produces about 1000 primary ionizations.
The atomic number of air is 7.64, which is close to that of muscle at 7.42. For this reason, the mass absorption coefficients of air and muscle are very similar. This means that the energy absorbed from an X-ray beam by a given mass of air is very similar to the energy absorbed from the beam by the same mass of muscle. The energy absorbed by both air and muscle is thus proportional to the exposure measured in air. This is the main reason for the importance of air as a medium in radiation dosimetry as it allows the dose in tissue to be calculated from knowledge of the air exposure.
27.3.1 Exposure and air kerma
In recent years, the term ‘exposure’ has fallen out of common usage and has been replaced by absorbed dose in air or air kerma (the initials of kerma stand for kinetic energy released per unit mass of absorber); for instance, the maximum permissible radiation leakage rate from the X-ray tube is now quoted in air kerma. The main reason for this is that it is much easier to calculate the absorbed dose in a structure from the air kerma.
27.3.2 Absorbed dose and kerma
The measurement of the quantity of electrical charge produced in air by ionization is not the same as the measurement of the energy actually absorbed, although the two quantities are proportional to each other. The energy absorbed by unit mass of the medium is stated as the absorbed dose and is defined thus:
The absorbed dose in a medium is in the ratio E/m, where E is the energy absorbed by the medium due to a beam of ionizing radiation being directed at a small mass m.
The unit of absorbed dose is the gray (Gy) and so we can say that l gray=1 joule per kilogram (1 Gy=1 J.kg−1).
Note that exposure is defined in terms of X or gamma radiation only, while absorbed dose is defined in terms of any ionizing radiation. Therefore, the absorbed dose from alpha-particles, beta-particles and neutrons are all measured in grays. However, ultraviolet radiation is only capable of excitation rather than ionization of the atoms of the medium and so is outside the scope of the definition of absorbed dose.
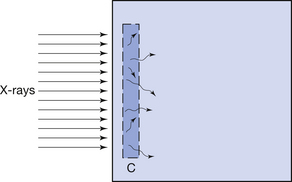
If all the electrons produced by the primary and secondary ionizations within a medium are stopped within it, then it can be seen that the energy removed from the beam of ionizing radiation is the same as the energy absorbed by the medium (this makes the assumption that all the fluorescent or characteristic radiation is absorbed, as is the case in body tissues). This does not necessarily apply to a very small volume within the medium – such a volume may be removing energy from the beam but the absorbed energy may be deposited outside the volume (but still within the body) due to the distance travelled by the electrons before coming to rest. Electrons with an energy of 1 MeV travel for about 5 mm in tissue before coming to rest.
This effect is illustrated in Figure 27.1 (See page 199). Here an incoming X-ray beam of high energy interacts with a volume element V within the medium. Because of the high energy of the beam, the electrons produced by Compton scatter are scattered in a forward direction, so much of their energy is absorbed outside the volume V. There will also be secondary ionizations resulting in the production of delta rays, but for simplicity these are not shown in the figure. In general, if the secondary electrons produced within the volume deposit a total energy E within the medium, and EIN and EOUT are the total energies of the electrons entering and escaping from the volume, then the absorbed dose in grays is given by:
where m is the mass of the particular small volume considered. If a larger volume is considered, then this formula can be used to calculate the average absorbed dose in that volume.
Electronic equilibrium is said to occur if EIN=EOUT, since there is no net loss or gain of the electrons over the small volume being considered. If EIN=EOUT is a constant value not equal to zero, there is said to be quasielectronic equilibrium. If the intensity of the radiation is varied, the net loss or gain of electrons will vary in proportion. An example of electronic equilibrium occurs in the free-air ionization chamber, which will be discussed later in Section 27.5.2.
The absorbed dose expresses the quantity of energy absorbed in the medium due to a beam of ionizing radiation passing through it. As stated at the beginning of this section, the site of the attenuating events (e.g. photoelectric absorption) may be at some distance from the absorption process because of the distance travelled by the ejected electrons before coming to rest. The quantity which measures the amount of attenuation in a small volume is called the kerma (see Sect. 27.3.1).
Kerma is also measured in grays and may differ significantly from the absorbed dose at any particular position within the medium.
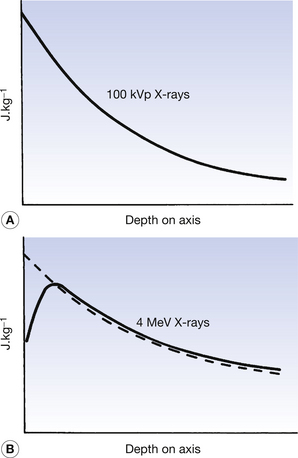
The absorbed dose and kerma along the axis of a beam of X radiation are shown in Figure 27.2. Figure 27.2A shows the case where an X-ray beam generated at 100 kVp is incident upon soft tissue: this type of situation might occur in diagnostic radiography. The electrons released in the primary and secondary ionizations are of relatively low energy and so are absorbed close to the site of the initial attenuating interactions. The kerma and absorbed dose at any particular point along the beam axis are essentially the same and the curves are coincident in the figure. This is not the case if the X-ray beam has high photon energy, since electrons produced by the initial ionization have considerable energy and so deposit their energy some distance from the point of the original attenuation process. As can be seen in Figure 27.2B, the kerma and the absorbed dose due to 4 MeV X-rays interacting with tissue are not the same. It may be easier to understand these curves if it is remembered that:
27.3.3 Effects of different media
Instruments that are used to measure absorbed dose or absorbed dose rate are called dosimeters and dose-rate meters respectively. Some of these instruments are described in more detail in later sections of this chapter (see Sect. 27.5 onwards). It is common practice to calibrate these meters to read the absorbed dose or dose rate in air through which the X- or gamma-rays are passing. Such a dosimeter may read 0.5 mGy as the total absorbed dose in air at a point within an X-ray beam. It must not be inferred, however, that this is the absorbed dose that would bereceived by any other medium if placed in the same position. For two media to receive the same absorbed dose, each must absorb the same energy from the beam per unit mass (remember, 1 Gy=1 J.kg−1). This is the same as saying that the mass absorption coefficient of the two media must be equal. Thus, if Dair is the absorbed dose in air and Dm the absorbed dose in a medium when both are irradiated with the same beam of X-rays, it follows that if the mass absorption coefficients are not equal, this equation may by drawn up:
If the mass absorption coefficient of air and the given medium are known at the energy of the X-ray quanta, the absorbed dose in the medium may be calculated using Equation 27.2. In practice, this allows us to measure the absorbed dose in air at a certain point and then calculate the absorbed dose in the patient at the same point without subjecting the patient to a great degree of discomfort.
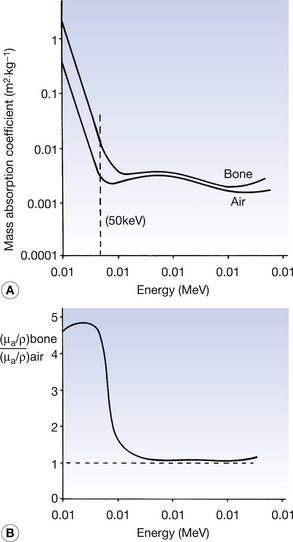
The mass absorption coefficients of both air and bone vary with photon energy. These variations of the two coefficients are shown in Figure 27.3A and the variations in the ratio of the two coefficients are shown in Figure 27 3B. As can be seen from the graphs, at low photon energies (50 keV is shown with the broken line in Figure 27.3A), the mass absorption coefficient of bone is considerably higher than that of air. This is because at low energies the photoelectric effect predominates (τ/ρ ∝ Z3/E3) and the atomic number of bone (Z=14) is approximately double that of air (Z=7.64). For this reason and because of the large difference in density, we get a high level of contrast between bone and air on a radiograph (this can be seen on the chest radiograph or on radiographs of the paranasal sinuses). At an energy of about 1 MeV, however, the two graphs are very close and the ratio of the two coefficients approaches 1. This is because of the dominance of Compton scatter in this region (σ/ρ ∝ electron density and the electron density for bone and air is approximately the same). This means that there would be a low level of contrast between the two if they were radiographed using 1-MeV photons. At above about 10 MeV the curves again diverge owing to the greater amount of pair production in bone compared to air ((π/ρ ∝ Z). Thus, it is clear that an instrument calibrated to read absorbed dose in air must be used with caution when calculating the absorbed dose in another medium as the relationships between the absorption coefficients vary with the photon energies. This is particularly the case in the diagnostic range of energies where absorption is principally by the photoelectric effect, which is very sensitive to both atomic number and photon energy.
27.4 Quality factor and dose equivalent
As described in the previous section, the absorbed dose measures the energy absorbed per unit mass of the medium when it is subjected to any type of ionizing radiation. The biological effects of the radiation on tissue, for instance, do not depend solely on the absorbed dose, but also on the type of radiation and on the absorbed dose rate. It is found that alpha-particles will cause considerably more damage (about 20 times as much) in a biological specimen compared to the same absorbed dose of X-rays. It is also found that radiation delivered as a single large dose will generally cause more biological damage than the same dose fractionated into multiple small doses and delivered over a period of time.
The differences in biological effects of different types of ionizing radiations are due to the different densities of ionizations they produce in a sample. Radiation which causes large numbers of ionization per unit length of track through a material will cause large amounts of biological damage. You may remember, when an atom is ionized (e.g. by a photoelectric interaction), an electron (negative ion) is released and the atom now becomes a positive ion – an ion pair has been formed. X-rays and beta-particles do not produce ion pairs as close together as do the more massive protons or alpha-particles of the same energy. Protons or alpha-particles are brought to rest quickly within the medium by losing their kinetic energy in the production of many ions over a short distance – an alpha particle of energy 1 MeV will only travel 5×10−3 mm in tissue, protons will travel 3×10−2 mm and beta-particles will travel 5 mm before being brought to rest. Because of this, the larger particles break chemical bonds, which are very close together, and so the chance of repair is reduced. This means that they have a greater biological effect on the specimen. It is also found that neutrons will produce dense ionization by the ejection of protons from the nuclei or by nuclear recoil. The absorbed dose in grays is thus not an accurate measure of the biological effects of different types of radiation owing to the very different patterns of ionization produced. The unit used to measure the overall biological effects of different types of radiation is called the unit of dose equivalent and is measured in sieverts (Sv). The absorbed dose in grays and the dose equivalent in sieverts are related to each other, as shown in Equation 27.3:
where Q is known as the quality factor for the radiation and is related to the number of ion pairs produced per unit length by the radiation. N includes other factors that may affect the biological process, such as the dose rate. In many cases, the value of N is 1 and so the equation is frequently quoted without the factor N appearing.
Table 27.1 shows the value of the quality factor for different types of radiation. Note that Q is unity for X-rays and gamma-rays so the absorbed dose is the same as the dose equivalent for these radiations. The biological effect of particulate radiations is therefore compared to that of X-rays or gamma-rays by means of the value of Q. As can be seen from Table 27.1, electrons also have a quality factor of unity. This is because an external beam of electrons will produce secondary electrons with the same election density as X-rays and gamma-rays. However, alpha-particles have a Q of 20, indicating that the same absorbed dose will produce 20 times as much biological damage as the same absorbed dose of X-rays.
Table 27.1 Quality factors for different ionizing radiations
| TYPE OF IONIZING RADIATION | QUALITY FACTOR |
|---|---|
| X-rays or gamma-rays | 1 |
| Electrons or beta-particles | 1 |
| Thermal neutrons | 2.3 |
| Fast neutrons (or neutrons of high energy) | 10 |
| Protons | 10 |
| Alpha-particles | 20 |
| Recoil nuclei (e.g. in alpha decay) | 20 |
| Fission fragments | 20 |
Since Q is a comparative number, the sievert has the same units as the gray (J.kg−1). The quality factor may be considered a scaling factor relating the biological effect of absorbed dose to the same dose of X-rays or gamma-rays.
Dose equivalent in sieverts has a vital role to play in radiation protection, where it is required to consider the sum of the effects of exposure to different types of radiation.
The dose equivalent is too crude a unit for use in radiobiology as it considers the average effect(s) on a group of cells. In radiobiology, we wish to look more precisely at individual effects on cells, e.g. impairment of cell reproduction. For this, we use a more precise scaling factor – the relative biological effectiveness (RBE). The RBE compares the absorbed doses of different ionizing radiations required to produce the same biological effect. As with the quality factor (Q), these are usually compared to the same dose of X or gamma radiation.
The remainder of this chapter is concerned with a brief overview of some of the methods used to measure exposure and absorbed doses.
27.5 Absolute measurement of absorbed dose
The absolute measurement of absorbed dose in air due to a beam of X-rays requires very careful techniques and very specialized equipment. It is more suited to a specialized laboratory than to a hospital or university department environment. In the UK, the National Physics Laboratory and in the USA, the National Bureau of Standards calibrates and checks specialized dosimeters under carefully controlled conditions. Such dosimeters are termed absolute standards. Dosimeters used in hospitals and universities are sent to such centres on a regular basis to be calibrated against the absolute standards: such dosimeters are then known as secondary standards. Further dosimeters are calibrated against these secondary standards. Such dosimeters are known as substandards. This initial section considers the manner in which an absolute measurement of absorbed dose may be made and the following section (Sect. 27.6) is an overview of the most common of the relative methods of assessing absorbed dose.
27.5.1 Calorimetry
A beam of X-rays or gamma-rays will be attenuated as it passes through a medium and the attenuation processes (see Ch. 23) will produce many ionizations within the medium. The atoms of the medium eventually absorb the kinetic energy of the electron ejected from their atoms. This results in these atoms having an increase in their kinetic energy – heat will be produced in the medium. The medium will experience a temperature rise that is proportional to the heat energy absorbed by the medium and therefore the absorbed dose. In Section 5.3.2, we have shown that:
where Q is the heat energy, m is the mass of the body, c is its specific heat capacity and (T2 − T1) is the temperature rise experienced by the body.
We also know from the earlier sections of this chapter (see Sect. 27.3.2) that absorbed dose (D) is energy per unit mass of the medium and so:
Thus, we can produce the equation:
Using the above equation, if we know the specific heat capacity of the medium, the absorbed dose may be calculated from the temperature rise produced in an irradiated medium. This is known as the calorimetric method of absorbed dose measurement. However, the temperature rise produced is very small: 1 Gy will produce a temperature rise of about 2×10−4°C and so the process needs very controlled conditions and is most appropriate when measuring very large absorbed doses of radiation. A more sensitive method is to collect the charge produced in an ionization chamber: this is described below.
27.5.2 The free-air ionization chamber
The free-air ionization chamber shown in Figure 27.4 uses ions produced by the absorption of an X-ray beam in air collected by oppositely charged plates situated in the air. The liberated electrons are attracted towards the positive plate and the positive ions are attracted towards the negative plate. Thus charge, whose magnitude is proportional to the exposure in coulombs per kilogram and the absorbed dose in grays, flows through the chamber. Certain precautions are necessary, however, to achieve accurate results:
• As shown in Figure 27.4 below, the central lower disc is surrounded by an annulus which is at earth potential. As Figure 27.4 is a vertical section through such a chamber, the annulus appears as if it were two separate plates. This construction enables an accurate estimation of the volume of air from which the ion pairs are collected, as it ensures that the lines of electrical force are at right angles to both the collecting plates. Note that this is not the case at the outer edge of the annulus, where the lines of electrical force are bowed outwards, thus including an unknown quantity of air beyond the edge of the plates. The volume of air from which the ion pairs are collected may be calculated by knowing the geometry of ABCDI. Ion pairs produced outside this volume are still collected by the annulus but do not pass through the meter M and its associated electronic amplifier and do not contribute to the current indicated by the meter.
• Some electrons produced by ionization in the region ABCD will escape and produce further ionizations over the annulus rather than the central disc. This suggests that the current measured by the meter, M, is too low, but this is not the case because on average the same number of electrons are gained by the volume under consideration. This is a case of electron equilibrium, described earlier (Sect. 27.3.2).
• The potential difference across the plates must be sufficiently high to collect all the ion pairs produced in the air. If we irradiate a free-air ionization chamber with a steady beam of radiation, the current flowing through the chamber will vary with the potential difference across the plates, as shown in Figure 27.5.
• Below the saturation voltage, some of the positive and negative ions recombine by mutual attraction (or germinal recombination) and so not all ions are collected. Above saturation voltage, the electrical field strength between the plates is large enough to ensure that all the ions move to the appropriately charged plate for collection. The actual voltage depends on the separation on the plates, but typically this is in the region of a few hundred volts.
• The separation of the plates, d, shown in Figure 27.4, must be sufficiently large to enable the production of all the secondary ionizations in air. No electron produced during the ionization process must reach the plates before it produces all the ion pairs of which it is capable. If d is too small, then the current measured by the meter, M, will be too low since there are too few ion pairs produced in the air volume and hence the estimate of the exposure or absorbed dose will be too low. The required plate separation depends on the energy of the X- or gamma-ray beam, since photons with high energies will produce electrons with correspondingly high energies that will travel further in air. Typical values vary from about 20 cm for photon energies up to 250 keV to several metres for photons of energy above 1 MeV. From this it can be seen that the greater the energy of the beam, the more cumbersome the measurement, due to the necessity for greater separation of the plates.
• The total charge in coulombs measured by the free-air ionization chamber is a direct measure of the exposure in C.kg−1 and is proportional to the absorbed dose in grays (J.kg−1). The mass of the air irradiated depends on the temperature and the pressure of air and must be corrected for the effects of these variations. If ρ0 is the density of air at a known temperature and pressure, then the mass of the air m0 in the irradiated volume, v can be calculated as m0=ρ0v At a new temperature and pressure (Tl and P1), the density changes to ρ0T0Pl/T1P0 so the mass of the air, m′, being irradiated is given by:
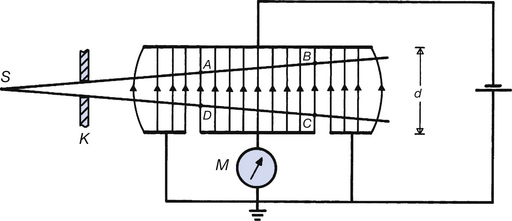
Figure 27.4 A diagrammatic vertical section through a free-air ionization chamber. S is the radiation source and K is a collimator. The electrons resulting from the ionization that takes place in the area ABCD are collected on the collecting plate CD which is connected to the meter M. This measure the negative charge produced as a result of the radiation.
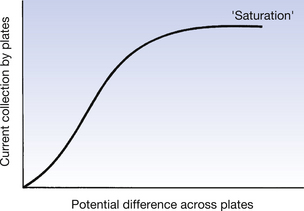
Figure 27.5 The variation of current flowing through the meter M in Figure 27.4 with the applied potential across the plates.
The exposure (in C.kg−1) is the ratio of the total charge collected to the mass of air irradiated (m′). The absorbed dose (in grays) is calculated from the energy absorbed divided by m′ – the energy absorbed can be calculated from the charge collected since it takes about 33 eV to produce one ion pair in air.
The ionization method is not suitable for use with liquids owing to the very rapid germinal recombination of the ions and the relatively high current which flows through many liquids, even when they are not being irradiated. A semiconductor (see Sect. 27.6.6) may, however, be used to collect the ion pairs produced by irradiation.
From the above it can be seen that the free-air ionization chamber is suitable for the absolute measurement of exposure and absorbed dose in air. The absorbed dose, which would have occurred in another medium, placed in the same beam of radiation may be calculated from the mass absorption coefficients, as explained earlier in Section 27.3.3.
27.5.3 Chemical methods of dose measurement
We have already established that radiation affects the chemical bonds between atoms of a material through which it passes by both ionization and excitation of electrons. Research by Fricke has shown that ionizing radiation is able to transform a dilute solution of ferrous sulphate, FeSO4, to ferric sulphate, Fe2(SO4)3, by rearrangement of the chemical bonds. The number of ferric ions so produced is proportional to the absorbed dose – 100 eV of absorbed dose will produce about 15 ferric ions. A chemical measurement of the concentration of the ferric ions produced at a given energy of a given radiation beam may used as a measure of the absorbed dose in water.
Such a chemical dosimeter may be calibrated against either of the two preceding methods of absorbed dose measure. It is included in this section on absolute methods of dose measurement because once the conversion factor between quantity of ferric ions is known, no further calibration is necessary. The process of calibration is similar to the calculation of absorbed dose in the free-air ionization chamber from the knowledge of the energy required to produce an ion pair in air.
The above method of dose measurement is known as the Fricke dosimeter, but it is only suitable for the estimation of very large doses, in excess of 20 Gy. This is because of chemical impurities present in the solution, the rapid rate of germinal recombination of the ions produced and relatively insensitive methods of chemical estimation of the quantity of ferric produced. However, it is particularly suitable for use with high-energy radiation beams and for irregular shapes of irradiated volumes The advent of conformational radiotherapy treatment and the requirement under the Ionizing Radiations (Medical Exposure) Regulations (IR(ME)R: see Ch. 44) to optimize radiation dose to the patient have resulted in the development of a number of polymer gels. These are tissue equivalent with a density of 0.99 g.cm−1 and show a linear response to high-energy photons from 300 keV to in excess of 8 MeV. Ionizing radiation also has a polymerization effect on the gel supporting the ferrous sulphate atoms; this reduces germinal recombination and prevents migration of the ferric sulphate atoms formed outside the beam area. Magnetic resonance spectrometry is used to estimate the number of ferric sulphate atoms present. If placed (in a suitable container) in an amorphic phantom, such dosimeters may be used to confirm the steep dose gradients that are an essential feature of conformational therapy treatments.
27.6 Types of detectors and dosimeters
So far in this chapter we have considered the measurement of absorbed dose by absolute methods. These form a standard against which other types of dosimeter can be compared or calibrated. There are many such relative methods by which absorbed dose may be estimated, each with some advantages and disadvantages. The most common of these methods are briefly outlined in the following pages.
27.6.1 The thimble ionization chamber
The size and configuration of the free-air ionization chamber discussed so far make it a suitable instrument for the standardization of radiation dose measurement but totally unsuitable for routine dose measurement in a hospital environment – a plate separation of 5 metres would be required if we needed to measure the dose rate at a patient’s skin from a cobalt-60 source!
The thimble ionization chamber shown in Figure 27.6 circumvents some of these difficulties by, as it were, ‘condensing’ the air into a solid medium surrounding the central electrode. The cap of the thimble chamber is said to be air equivalent, i.e. it is made of a material that has the same atomic number as air (e.g. graphite, bakelite, plastic) and so its absorption properties are the same as the same mass of air. The central aluminium electrode has a fixed amount of positive charge put on it from an external source. When the chamber is irradiated, some of the more energetic electrons liberated in the cap will penetrate into the air of the chamber and be attracted to the central electrode. Thus, the central electrode will lose some of its positive charge. By the choice of suitable materials, the thimble chamber can be made to have the same absorbing properties as the same mass of air. Such a device is calibrated for several photon energies against a standard chamber, such as the free-air chamber described earlier in this chapter, and a correction factor is used to convert the indicated loss of charge from the central electrode to true absorbed dose. The choice of wall thickness of the cap is one of the factors that influence the applied correction, since a thin ‘cap’ may not produce sufficient electrons entering the chamber, while too thick a cap will absorb more radiation than it needs to. Note that the vast majority of the electrons used to measure the change in charge or the current through the chamber are produced in the wall of the chamber and not in the air cavity of the chamber, but it is the passage of such electrons into the air cavity that enables the change in charge or the current to be measured. Corrections for variations in the temperature and pressure of the air must be made, as is the case for the free-air chamber.
Thimble-type chambers are still extensively used in radiation measurements in hospitals. For example, the calibration of the radiation output from a teletherapy machine is usually accomplished by the use of a thimble chamber connected to an electronic amplification system, which measures and displays the charge produced in the chamber during irradiation. However, in order to relate the reading obtained because of a given exposure to the radiation output of the machine (usually expressed in cGy.min−1), certain correction factors need to be applied. These include the following:
• The reading must be corrected for temperature and pressure.
• The reading must be corrected by a factor which relates the reading of this substandard unit to a secondary standard unit calibrated by a national body (e.g. the Health Protection Agency) – this correction factor depends on the energy of the radiation.
• The correction which requires to be applied to the secondary standard to compare it with the absolute standard – this is again related to the energy of the radiation and a factor to convert exposure to absorbed dose at the appropriate radiation energy.
In addition, there may be some machine-dependent correction factors, such as correction for ‘switch-on’ and ‘switch-off’ errors.
27.6.2 The geiger–müller counter
The thimble chamber described in the previous section is an example of an ionization chamber where the charge collected on the electrodes is proportional to the energy absorbed from the X-ray beam. The Geiger–Müller counter works on the principle of gas multiplication and gives the same magnitude of electrical pulse per absorption event whatever the energy of the absorbed radiation. The structure of a typical Geiger–Müller tube is shown in Figure 27.7A below.
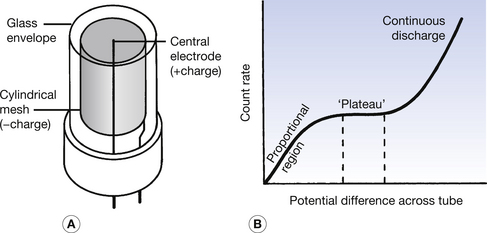
Figure 27.7 (A) Basic construction of the Geiger–Müller tube. (B) A graph of the operating characteristics of such a tube, which is operated on its plateau region.
The glass envelope contains an inert gas (argon) at low pressure and two electrodes – a positively charged central electrode and a negatively charged mesh cylinder. Ionization is caused in the gas by the entry of a photon or by the entry of particulate radiation (if the window is sufficiently thin to allow particles to enter the envelope). The ions are attracted to the appropriate electrode and, as they pass through the gas, they gain sufficient energy to eject electrons from the gas atoms if the potential difference between the electrodes is sufficiently great. The electrons so produced continue this process and rapid gas multiplication takes place, especially near the central electrode, since the field strength is great in this region. The effect of gas multiplication is such that well in excess of 1 million electrons are collected by the central electrode for every single ion produced in the primary absorption process. These ‘electron avalanches’ form the pulses, which allow the system to count the number of initial ionization events. The presence of a small quantity of alcohol vapour in the gas helps to quench the gas multiplication process so that it does not become continuous. It does this by absorbing the kinetic energy of the positive ions in the gas so that they are prevented from striking the mesh with sufficient energy to release further electrons and so keep the process going indefinitely. Alternatively, the potential difference between the electrodes may be momentarily reduced after an electron avalanche, thus terminating the gas multiplication. In either case, there is a dead time after each pulse, where another absorption event, if present, is not recorded. A typical dead time is 5 μs and so the differences between the observed count rates and the real count rates are negligible except at high count rates – at an observed count rate of 1000 per second, the true count rate is 1005, whereas at an observed count rate of 100 000 per second, the true count rate is 200 000! The correct potential difference to be applied to a Geiger–Müller tube is determined in practice by plotting a graph of the count rate obtained when a small radioactive source is placed near the tube against the applied voltage. Such a graph is shown in Figure 27.7B. Three distinct regions of such a graph exist:
1. The proportional region, where some gas multiplication takes place and the sizes of the electrical pulses are proportional to the energy deposited in the gas by the radiation.
2. The plateau region, where maximum gas amplification takes place and all electrical pulses have the same size, irrespective of the energy of the radiation.
3. The continuous-discharge region, where the electrical field strength is sufficient to ionize the gas atoms and so produce continuous unwanted gas multiplication. The plateau is usually between 100 and 1500 V, depending on the size of the Geiger–Müller tube.
As can be seen, the Geiger–Müller tube is suitable for detecting the presence of radiation rather than for an accurate estimation of absorbed doses, since the pulses bear no relationship to the energy of the radiation causing them. For this reason it is often used as a contamination monitor for radioactive spillage or as a method of determining whether radiation is present in a specific area.
27.6.3 Scintillation detectors
The operation of a scintillation detector employing a sodium iodide crystal and a photomultiplier tube is described in Chapter 37, which considers radionuclide imaging and nuclear medicine in more detail.
Any suitable scintillating material can be used, whether solid or liquid, and the principle of operation is that the size of the electrical pulse produced by the photomultiplier is proportional to the energy deposited in the scintillator. Scintillation plastics have been produced which have an atomic number close to that of air and tissue. These can be termed as being air equivalent and so have similar variations of absorption to air, with variations in photon energy. They are useful in estimating absorbed dose in air. Sodium iodide has a much higher atomic number than air and will show a marked variation in absorption with photon energy, especially near its absorption edges (see Sect. 23.5). This requires correction factors to be applied for different photon energies if an accurate estimation of the absorbed dose in air is to be made. This is particularly so for thin crystals, which show a more marked variation in absorption with photon energy compared with thick crystals.
Scintillation counters are very sensitive devices and are used in many applications in radiography and radionuclide imaging, e.g. for the detection of radioactive contamination, for estimation of in-vitro radioactivity, as radiation detectors on computed tomography and osteoporosis scanners and as the detection mechanism in gamma cameras.
27.6.4 Thermoluminescent dosimetry (TLD)
The basic physics of thermoluminesce has already been described (see Sect. 24.5). Thermoluminescence may be used to estimate radiation doses by the use of lithium fluoride in the form of powder, extruded chips or impregnated Teflon discs or rods. The impurities in the lithium fluoride generate electron traps and the number of electrons which are ‘stuck’ in these traps is proportional to the absorbed dose in the lithium fluoride. The average atomic number of lithium fluoride is 8.2 so it is close to soft tissue (Z=7.5). Both have similar absorption variations with photon energy. The small discs do not show up on radiographs and so may be strapped to the part of the body where we wish to measure the absorbed dose. After irradiation, the discs are heated and the amount of light emitted is compared to a standard dosimeter to which a known dose has been given. The dose to the disc can then be calculated by direct proportion. The discs are then annealed and may be reused.
The fact that the discs have radiolucency similar to tissue allows us to use them to estimate radiation dose without interfering with the radiograph or radiotherapy treatment. The discs are small, measuring only a few millimetres across, and so may be used to estimate the dose to different structures in the body during a diagnostic or therapeutic procedure. An additional advantage is that they can be used to monitor radiation dose ranging from 1 μGy to 1 KGy.
The role of TLD in personnel monitoring will be considered in Chapter 44.
27.6.5 Photographic film
Photographic film will produce an increase in optical density when it is irradiated and processed. The response, however, is not in linear proportion to the absorbed dose of the emulsion. A calibration graph for the particular film at known radiation doses and specific processing conditions must be produced. This is a major disadvantage if it is to be used as a method of dose estimation. It is also true that the film emulsion has a higher atomic number than tissue (AgBr has an average atomic number of 41) and so has significantly higher photoelectric absorption at low energies. For these reasons, the use of photographic film has largely been replaced by TLD dosimetry.
27.6.6 Semiconductor detectors
As we saw in Chapter 15, electrons in a semiconductor can readily have their energy raised to that of the conduction band and so can take part in electrical conduction. The absorption of energy from an X-ray photon (by either photoelectric absorption or Compton scattering) can raise an electron to the conduction band energies. This electron causes secondary electrons from the atoms of the material to be raised to the conduction band, by imparting some of its energy to them. If a potential difference is placed across the semiconductor, then these electrons are collected before they have time to recombine. Thus there is a current pulse whose magnitude is proportional to the number of electrons and hence the absorbed dose within the semiconductor. This is similar to the current through an irradiated ionization chamber and the semiconductor detector can be thought of as a solid-state ionization chamber. It has the great advantage over the air ionization chamber that it produces 10 times as many ion pairs for a given dose of radiation and so is much more sensitive to small doses. This is because only 3 eV is required to produce an ion pair in a semiconductor, compared to 33 eV to produce an ion pair in air. The electrical signal obtained from the semiconductor device is more accurate – it has a smaller statistical uncertainty – and may, for example, be used to produce very accurate gamma-ray spectra.
Semiconductor detectors may be calibrated against a thimble chamber, for example, for a given energy of radiation.
Semiconductor detectors tend to be used for more specialized forms of radiation detection, e.g. a small semiconductor detector may be inserted into the rectum to measure rectal dose.
In this chapter, you should have learnt the following:
• The definition of exposure and the relationship between exposure and absorbed dose (see Sect. 27.3).
• The definition of and the relationship between absorbed dose and kerma (see Sect. 27.2).
• The effects of different media on the absorbed dose (see Sect. 27.3.3).
• The meaning of the terms quality factor and dose equivalent and how their values vary for different types of radiation (see Sect. 27.4).
• Measurement of radiation exposure and absorbed dose by the free-air ionization chamber (see Sect. 27.5.2).
• Measurement of radiation exposure and absorbed dose by chemical dosimeters (see Sect. 27.5).
• Measurement of radiation exposure and absorbed dose by the thimble ionization chamber (see Sect. 27.6.1).
• Detection of the presence of radiation by the Geiger–Müller counter (see Sect. 27.6.2),
• Measurement of radiation exposure and absorbed dose by scintillation detectors (see Sect. 27.6.3),
• Measurement of radiation exposure and absorbed dose by thermoluminescent dosimetry (see Sect. 27.6.4).
• The use of photographic film in dosimetry (see Sect. 27.6.5).
• Measurement of radiation exposure and absorbed dose by semiconductor detectors (see Sect. 27.6.6).