Chapter 8 Intensive care for the critically ill adult
INTRODUCTION
There remains a perception by some medical disciplines that physiotherapy patient management in the intensive care unit (ICU) is focused solely on the maintenance and improvement of a patient’s cardiopulmonary status. However, the role of the physiotherapist also includes maintenance of musculoskeletal function, optimization of neurological status, and is extending to areas such as extubation/decannulation, ventilator weaning, troubleshooting mechanical ventilation problems and therapeutic fibre optic bronchoscopy (Jones 2001, Norrenberg & Vincent 2000) and involvement in ‘patient at risk’ and medical emergency teams.
Advances in technology and pharmacology have contributed to the increasing survival of patients from critical illness. Dowdy et al (2005) suggest that ICU survivors have a significantly lower quality of life, both before and after ICU stay, compared with the general population. The impact of post-ICU discharge rehabilitation on a patient’s functional capacity and quality of life is as yet unclear.
Physiotherapists may have a role in enhancing functional reserve before patients are admitted to ICU (prehabilitation). This should enable patients to better withstand the stress of ICU procedures and inactivity (Topp et al 2002). It follows that the place of physiotherapy in the management of patients in the ICU comprises a three-segment continuum of care:
Irrespective of the evolving direction of the place of physiotherapy in ICU, optimization of the cardiorespi ratory status of the patient remains a central objective. It is therefore essential that the physiotherapist has:
 an understanding of the optimal means of monitoring and supporting major organ function (cardiac, pulmonary, renal)
an understanding of the optimal means of monitoring and supporting major organ function (cardiac, pulmonary, renal)Physiotherapists should not only acquire competency in techniques necessary for treatment intervention but must also demonstrate the ability tomanage complications that might arise as a consequence of their actions.
Furthermore, a greater understanding by the physiotherapist of the relationship between fluid titration, pharmacology and ventilatory management will not only provide the physiotherapist with an opportunity to optimize the quality of patient care but also should entail an extension of the physiotherapist’s scope of practice, in liaison with a multidisciplinary team.
This chapter will discuss the optimal means of monitoring and supporting the major organ systems of the body and the implications for physiotherapy intervention. The second section of the chapter adopts a problem-based approach and will focus on the place of physiotherapy in the ICU.
MONITORING AND MECHANICAL SUPPORT
Mechanical ventilation
Mechanical ventilation is essential to maintain life for some patients and is often used for patients with respiratory failure (Box 8.1).
Box 8.1 Types of respiratory failure
| Type 1: | |
| Type 2: | |
| Hypoxaemia = PaO2 <60 mmHg with FiO2 >0.5 | |
| Abbreviations: CO2, carbon dioxide; PaO2, partial pressure of oxygen in arterial blood; FiO2, fractional inspired oxygen concentration | |
Hypoxaemia = PaO2 <60 mmHg with FiO2 >0.5
Abbreviations: CO2, carbon dioxide; PaO2, partial pressure of oxygen in arterial blood; FiO2, fractional inspired oxygen concentration
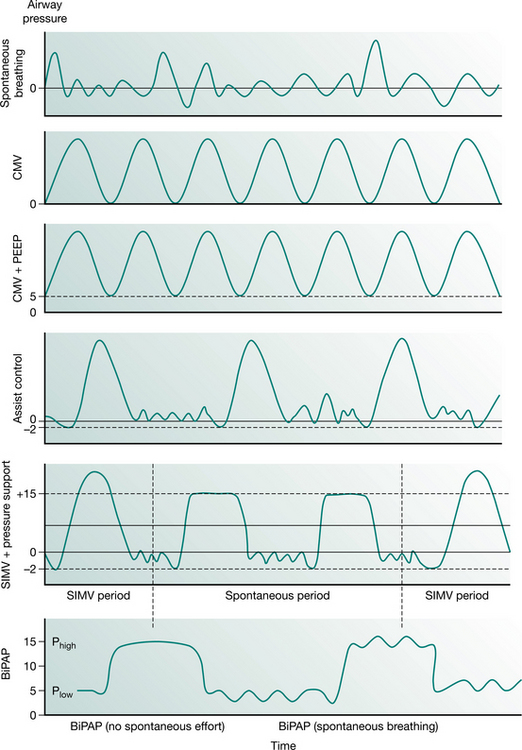
Full ventilatory support should maintain or improve alveolar ventilation (Pilbeam 1992) and reduce the work of breathing. Work of breathing is an important consideration during the provision of mechanical support and can be reduced with various types of inspiratory support. This may be achieved by supporting respiration in various modes such as control, assist-control, synchronized intermittent mandatory ventilation (SIMV) with or without pressure support and bilevel positive airway pressure ventilation (BiPAP). The ventilator pressure waveforms of some common modes of ventilatory support are illustrated in Figure 8.1.
Less conventional methods of ventilation have been introduced with the aim of limiting lung damage and preserving spontaneous breathing. These modes are:
The following descriptions are summarized from the review by Weavind & Wenker 2000.
Pressure control–inverse ratio ventilation (PC–IRV)
This mode of ventilation aims to maintain a constant pressure during ventilation. The physiological basis for adopting this mode of ventilation is that the ‘prolonged’ inspiration promotes alveolar expansion through alveolar recruitment. The inspiratory : expiratory (I : E) ratio is higher (e.g. 1 : 1 or 2 : 1) compared with the traditional 1 : 2; thus complete exhalation from the alveoli with slower time constants is prevented by the short expiratory time, expansion of the alveoli is maintained by auto-peep (generated by the longer inspiratory time). The combination of decelerating flow and maintenance of airway pressure over time results in the inflation of stiff (non-compliant) lung units with long time constants, for example, in patients with acute respiratory distress syndrome (ARDS). However, there have been no demonstrable benefits in terms of patient outcome using this mode.
Airway pressure release ventilation (APRV)
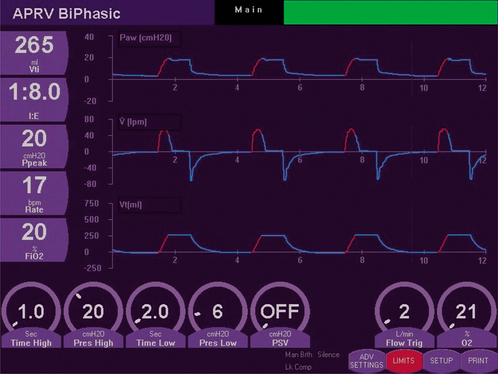
This mode of ventilation maintains continuous positive pressure in the airway (CPAP) with intermittent release of the pressure (essentially an inverse ratio form of bilevel ventilation). The duration of release is rather short, and similar to PC–IRV, and results in an inverse I : E ratio mode of ventilation. Patients who are able to breathe spontaneously with a relatively low work of breathing can utilize APRV, thereby minimizing barotrauma and circulatory compromise. This mode of ventilation may not be suitable for patients with asthma or severe COPD because they tend to find difficulty in emptying their lungs during the short release.
Biphasic positive airway pressure (BiPAP) and bilevel ventilation
BiPAP is pressure-controlled ventilation with two levels of continuous positive airway pressure (CPAP) (Phigh and Plow), with a set breathing rate. The I : E ratio can be adjusted. Bilevel ventilation is a combination of APRV and BiPAP. The mandatory breaths are pressure-controlled and the spontaneous breaths can be pressure supported (often only at Plow). Thus, bilevel ventilation can be used as pressure-controlled ventilation initially in sedated or paralysed patients, then weaned to CPAP and PS (to allow spontaneous breathing) and then to CPAP. Figure 8.2 shows the biphasic waveform on a monitor screen.
High-frequency ventilation
High-frequency ventilation can be jet ventilation or oscillation. In jet ventilation, a high pressure (30–300 kPa) of air with supplementary oxygen is delivered to the airway via a small-bore catheter at frequencies between 60 and 300 Hz. Expiration is passive. This form of ventilation is mainly used during surgery when the airway is disrupted or placement of a tracheal tube is not possible. High-frequency oscillatory ventilation (HFOV) (Fig. 8.3) is oscillation of a continuous distending pressure at rates of 100–1000/min with active inspiration and expiration. The advantage of high-frequency oscillatory ventilation is a stable continuous positive airway pressure, with control of ventilation at high breath rates and small tidal volumes (50–100 ml). The patient needs to be very heavily sedated and/or paralysed to minimize or prevent spontaneous respiration, often resulting in the cough reflex being abated. Humidification is often inadequate with this form of ventilatory support.
Other forms of ventilation and adjuncts
Liquid ventilation involves filling the lungs with a solution that dissolves oxygen. This form of ventilation is largely experimental at present. Extracorporeal membrane oxygenation (ECMO) and extracorporeal CO2 removal (ECCO2R) are other forms of non-conventional ventilation, often used when all other forms of ventilation have failed.
Inhaled nitric oxide can be used as a selective pulmonary vasodilator. It has been used for many decades for the management of severe arterial hypoxaemia and pulmonary hypertension in both adults and children. Recent guidelines (Germann et al 2005) recommend inhaled nitric oxide as useful rescue therapy for the management of severe pulmonary arterial hypertension and severe refractory arterial hypoxaemia, but do not confer any survival benefits in adults.
‘Protective’ and ‘open lung’ ventilation are currently used for such conditions as acute lung injury and acute respiratory distress syndrome. This consists of using low tidal volumes (6–8 ml/kg), allowing the partial pressure of carbon dioxide in arterial blood (PaCO2) to rise and ensuring the alveoli remain inflated with optimal positive end-expiratory pressure (PEEP) (Levitt & Matthay 2006). There is Level 1 evidence (Petrie et al 1995) for low tidal volumes causing a decrease in mortality (Petrucci & Iacovelli 2004).
Monitoring of the body systems
Rapid, potentially lethal physiological and pathological changes can occur in an acutely ill patient; hence the performance of the body must be adequately monitored and supported to optimize patient care. While monitors and equipment are essential for primary measurement and support, the interpretation of the data in concert with astute patient observation goes a long way to ensuring that patients will be safely and effectively cared for.
Monitoring of patients on mechanical ventilation
The primary aim of mechanical ventilation is to provide adequate gas exchange. Different forms of mechanical support have been explained above. Careful observation of the patient data and the pressure, flow and volume waveforms will assist the physiotherapist in identifying the level of synchrony between the patient and the mechanical ventilator, including any potential causes of increased work of breathing or changes in lung/thorax compliance or airway resistance in a mechanically ventilated patient.
Monitoring respiratory muscle function.
Respiratory muscle weakness is often associated with prolonged mechanical ventilation and the cause of failure or delay in weaning. Diaphragmatic strength can be determined non-invasively in mechanically ventilated patients and can be assessed from the twitch gastric, twitch oesophageal and twitch transdiaphragmatic pressures in response to phrenic nerve stimulation (Mills et al 2001). Maximal static inspiratory and expiratory efforts, electromyography of respiratory muscles, respiratory rate, carbon dioxide (CO2) level, pressure–time product, vital capacity and maximum voluntary ventilation are all variables which reflect respiratory muscle function. Respiratory rate and tidal volume presently remain the most convenient and frequently used indices of respiratory muscle function (e.g. respiratory rate/tidal volume provides a rapid shallow breathing index) (Spicer et al 1997). These measures may be used to assess the readiness of the patient to breathe spontaneously and to be weaned from ventilatory support.
Lung mechanics.
Improvement in lung volume is associated with improvement in the elastic properties of the lung. The relationship between changes in lung volume and transpulmonary pressure is referred to as static lung compliance (l/cmH2O). Static lung compliance should be measured during ‘cessation of air flow’, when elastic recoil is independent of airway resistance. Dynamic lung compliance can be measured during ‘uninterrupted’ respiration.
The change in lung volume and pressure are measured at end-inspiration (compliance) and end-expiration (auto-peep); this is when airflow ‘momentarily’ ceases during the ‘normal’ respiratory cycle. Most mechanical ventilators in ICU display/calculate dynamic lung compliance, auto-peep as well as airway resistance.
Assessment of work of breathing.
A patient’s work of breathing may be described as the amount of muscle activity required to overcome the elastic (lung tissue, chest wall and abdominal compartment) and resistive (airways, flow rate) elements of the respiratory system. It includes any additional loads imposed by the mode of ventilator support, artificial airways and humidification devices (which may contribute additional resistive work, particularly through a heat and moisture exchange filter as compared with a heated humidifier circuit). The work of breathing of the intubated and mechanically ventilated patient depends on factors that are either patient- or ventilator-related. Patient-related factors include the type of pulmonary disease (airway, lung parenchyma, pleural space), cardiovascular dysfunction, altered respiratory drive (fever, pain, anxiety, delirium, acid–base disturbance), level of spontaneous respiratory activity and level of sedation/paralysis. Ventilator-related issues include the mode of mechanical ventilation, settings and, most importantly, the level of synchrony between ‘man and machine’.
To optimize mechanical ventilation, the mechanical ventilator must promptly respond to the patient’s demand during inspiration and allow an unimpeded expiration. Mechanical ventilation practices vary internationally and hence patient management varies, depending on institutionally driven, airway and ventilator strategies (Esteban et al 2000).
Most modern mechanical ventilators display real-time ventilator waveforms such as pressure, flow and volume across time or as loops (pressure/volume, flow/volume). Waveform/loop monitoring allows the clinician to interpret the interaction between the patient and the mechanical ventilator. Early detection of unto-ward changes in waveforms may allow the clinician to optimize the ventilator settings (by altering PEEP, trigger sensitivity, pressure support) to minimize any disruption to delivered ventilation (tidal volume, minute volume) and reduce work of breathing during physiotherapy treatment. For adequate synchrony between the patient and the mechanical ventilator, the clinician must ensure that the ventilator responds promptly to the patient inspiratory demand (trigger), provides adequate flow requirements during the inspiratory phase, cycles from inspiration to expiration when the patient starts exhalation and allows full and complete exhalation, before the next ventilator breath delivery. Detailed bedside waveform analysis of various patient work of breathing scenarios throughout the respiratory phases and suggestions for patient clinical management are illustrated later in this chapter.
Monitoring of the respiratory system
The definitive function of the respiratory system is to ensure adequate gas exchange. Respiratory function is best assessed by analysis of measures of oxygenation and ventilation, such as oxygen saturation and blood gases. An understanding of the mechanics of breathing is essential to determine the work of breathing required to achieve a certain level of gas exchange.
Monitoring oxygenation
Blood gases. Arterial blood gases (partial pressure of oxygen and carbon dioxide, and pH) provide essential information about a patient’s metabolic as well as respiratory status. Continuous measurement of arterial blood gases is possible but this technique is not widely practised because of cost, calibration drift and clot formation (Shapiro et al 1993, Zimmerman & Dellinger 1993). Interpretation of arterial blood gases is discussed in Chapter 3.
Transcutaneous gas measurement. This method of measurement is based on the principle that blood flow and oxygen exchange are dependent on skin temperature (Baumburger & Goodfriend 1951). Transcutaneous electrodes include a heating element to maximize local blood flow. When the skin is heated the capillary blood becomes ‘arterialized’ and the arterialized gases diffuse through the skin to the electrodes (Whitehead et al 1980). More recently, a new sensor (TOSCA monitor) for combined continuous transcutaneous monitoring of arterial oxygen saturation and carbon dioxide tension has been shown to be an accurate and valuable tool for respiratory monitoring (Bernet-Buettiker et al 2005, Senn et al 2005). At present this is used more commonly in neonates.
Oximetry. Non-invasive assessment of oxygen saturation by a pulse oximeter (SpO2) was introduced clinically in 1975 (Kendrick 2001). Pulse oximetry is now an expected monitoring component for assessment of hypoxaemia. The oxygen saturation of arterial blood [with partial pressure of oxygen (PO2) of 100 mmHg or 12–13 kPa] is about 97.5% and that of mixed venous blood (with PO2 of 40 mmHg or 5–5.5 kPa) is about 75% (West 2005). The absorption of light energy by blood varies, depending on the wavelength. Red (660 nm) and infrared (940 nm) light result in the greatest separation between deoxyhaemoglobin and oxyhaemoglobin absorption spectra. Pulse oximetry uses light-emitting diodes set at these wavelengths, and as the emitted light passes through the finger (or earlobe), the light energy is variably absorbed by the arterial and venous blood. The absorption ratio of the red/infrared light by the blood is proportional to the amount of desaturated haemoglobin, and from these data the pulse oximeter calculates and displays the SpO2 (Kendrick 2001).
The accuracy of commercial pulse oximeters is about ±2% within the clinical oxygen saturation range of 70–100% (Jensen et al 1998). Pulse oximeter readings may be inaccurate in patients with severe or rapid desaturation, hypotension, hypothermia and low perfusion states. A further limitation of oximetry is that it provides information only on oxygen status, and not on ventilation (PaCO2).
Monitoring carbon dioxide (capnography).
The concentration of carbon dioxide expired during different phases of the respiratory cycle provides information on the effectiveness of alveolar ventilation (Fig. 8.4), not only the end-tidal carbon dioxide value but also tube position and breathing circuit integrity.
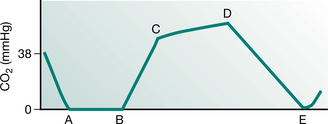
Figure 8.4 A typical capnograph showing normal end-tidal carbon dioxide level. (A–B) Dead space (CO2-free gas); (B–C) mixed dead space and alveolar gas; (C–D) mostly alveolar gas; (D) end-tidal CO2; (D–E) inhaled gas (CO2-free gas).
Detailed ‘indications’ for capnography can be found in the American Association for Respiratory Care (AARC) Clinical Practice Guidelines on Capnography/Capnometry during Mechanical Ventilation (AARC 1995).
Monitoring of the cardiovascular system
Non-invasive monitoring
Blood pressure. The use of automated non-invasive blood pressure (NIBP) devices is common. The mean arterial pressure (MAP) most closely approximates capillary perfusion pressure and is thus a useful measurement. NIBP devices can give inaccurate results in patients with arrhythmias, cardiac valvular lesions, where there is improper cuff application and when the blood pressure is low (Box 8.2).
Box 8.2 Normal values for blood pressure (BP)
Normal value of systolic/diastolic pressure
Electrocardiogram. The electrocardiogram provides information on the rate and rhythm of the heart; it also assists in the diagnosis, and identification of the possible site of myocardial infarction. The pathological feature of myocardial infarction is necrosis of myocardial muscle. Absolute evidence of myocardial necrosis is the pathological Q wave. Q waves, ST elevation and T wave inversion are all associated with transmural infarction (involvement of whole thickness of the myocardium). For sub-endocardial infarction (involvement of the inner layer of the myocardium, adjacent to the endocardium), Q waves do not appear and changes are confined to ST segments and T waves. Thus subendocardial infarction may be difficult to differentiate from ischaemia of the myocardium. Interpretation of the electrocardiogram is discussed in Chapter 3.
Thoracic electrical bioimpedance and impedance cardiography. Thoracic electrical bioimpedance (TEB) relies on measurement of bidirectional blood flow within the aorta by a laser Doppler velocimeter and an impedance measurement unit which determines the cross-sectional area of the vessel. TEB allows the clinician to view beat-to-beat cardiac output. (Newman and Callister 1999, Tjin et al 2001) and provides information on haemodynamic indices such as systolic time interval, left cardiac work index and end diastolic index (Belott 1999, Weiss et al 1997).
Non-invasive measurement of stroke volume and cardiac output by impedance cardiography (ICG) has been shown to be accurate and significantly correlated to conventional thermodilution method (see Thermodilution cardiac output, below) (Scherhag et al 2005). ICG measures synchronized pulse changes in TEB via simple surface electrodes together with a conventional electrocardiogram.
Partial CO2rebreathing cardiac output and NICO. Rather than applying the oxygen Fick method for cardiac output monitoring, the non-invasive CO2 Fick methods for estimation of cardiac output are receiving increased clinical interest. Adopting the CO2 version of the Fick equation has the advantage that CO2 elimination is easier to measure accurately compared with oxygen uptake (Jaffe 1999). The differential Fick partial rebreathing method computes a cardiac output value based on the changes in carbon dioxide elimination and end tidal CO2 in response to a change in ventilation. The non-invasive cardiac output (NICO system) is the first commercially available cardiac output system making use of the principle of partial rebreathing of CO2 (Jaffe 1999).
Echocardiography and Doppler. This uses ultrasound to examine the performance of the heart and great vessels. Information from echocardio-graphy can be presented one- (M-mode), two- or three-dimensionally. M-mode is often used in conjunction with a two-dimensional echo to provide a clear illustration of the structures being investigated (Young & Sanderson 1997).
Doppler echocardiography uses ultrasound to measure blood flow velocity and is able to determine pressure gradients, stenotic valve areas, cardiac output, left ventricular contractility, and diastolic function.
Transoesophageal echocardiography can be considered an invasive technique as it involves the introduction of an ultrasound probe (attached to the end of a flexible endoscope) into the oesophagus. This technique avoids image obstructions caused by the lungs and ribs and allows better views of the valves, septa and thoracic aorta.
Urine output. Urine output is an index of renal perfusion and is a guide to adequacy of cardiac output. With normal renal perfusion, the urine output should be at least 0.5 ml/kg/hour.
Invasive monitoring
Arterial pressure. Intra-arterial measurement should normally be considered accurate, but the systolic pressure may be overestimated due to systolic ‘overshoot’ (a property of the fluid-pressure transducer monitoring system). The ‘area’ under the arterial tracing can provide a rough estimate of the cardiac output (Gomersall & Oh 1997).
Central venous pressure. Central venous pressure (CVP) reflects right ventricular filling and is usually monitored by a catheter inserted via the internal jugular or subclavian vein, and less frequently via the femoral vein. A quick way to confirm correct placement of the catheter is observed pressure change with respiration (Gomersall & Oh 1997). As the right ventricular preload is determined by the volume and not the pressure, the absolute value of CVP is less meaningful. A high CVP value, however, may be associated with conditions that cause a rise in the right atrial pressure (for example right heart failure, reduced right ventricular diastolic compliance, hypervolaemia and pulmonary hypertension) (Gray et al 2002), whereas a low CVP value may suggest hypovolaemia. Changes in CVP may provide useful guidance for fluid management in patients: for example, a minimal rise of CVP despite fluid loading may suggest the loading volume is insufficient; but a rise of CVP of more than 9.5 cmH2O (7 mmHg) may indicate maximal loading. A raised CVP in response to fluid loading is expected to return to its original value within 10 minutes – an indication that the risk of pulmonary oedema is only moderate (Gomersall & Oh 1997) (Box 8.3).
Pulmonary artery catheter (Swan–Ganz). Pulmonary artery catheters are often used in patients with impaired right or left ventricular function, pulmonary hypertension, septic shock and when measurements of cardiac output or mixed venous saturation are indicated. It can also be used to assist in the diagnosis of an intracardiac shunt, such as a ventricular septal defect. Apart from cardiac output, cardiac index and systemic vascular resistance, a pulmonary artery catheter can provide a measure of mean right atrial pressure, systolic and diastolic right ventricular pressure, systolic, diastolic and mean pulmonary artery pressure, and pulmonary artery occlusion pressure.
Pulmonary artery occlusion pressure (PAOP) (previously referred to as pulmonary capillary wedge pressure (PCWP)) provides an estimation of the left atrial pressure (LAP). PAOP is obtained with the balloon at the catheter tip wedged in a pulmonary capillary. PAOP increases in poor left ventricular function, fluid overload and mitral valve disease (and is low in hypovolaemia). A high pulmonary artery pressure (PAP) may indicate high pulmonary vascular resistance.
 Clinical implication
Clinical implication
The physiotherapist should check the pulmonary artery waveform before and after positioning a patient for physiotherapy treatment. The catheter, even though not inflated, may be ‘pushed’ further into the pulmonary capillary and assume a wedged position.
Thermodilution cardiac output. Non-invasive monitoring of cardiac output has been discussed above. Traditional measurement of cardiac output by the thermodilution method requires the use of a pulmonary artery catheter. Cardiac output can be computed from the decrease in blood temperature in the pulmonary artery after injection of a known volume of cold saline into the right atrium. Computation of cardiac output is based on the principle that the decrease in blood temperature is inversely proportional to the extent of dilution of the cold saline (i.e. the higher the cardiac output the less the change in temperature between injection and measurement points).
Continuous thermodilution is possible by monitors that use infusion of heat from a filament in the right atrium rather than injection of cold saline. The monitor displays cardiac output averaged over the previous 3 to 6 minutes (Boldt et al 1994, Yelderman et al 1992).
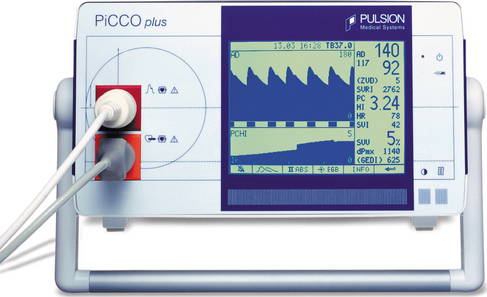
The most commonly used method of continuous cardiac output monitoring is by PiCCO® technology. This technology is less invasive. It requires the insertion of a thermodilution catheter in the femoral or axillary artery and a central venous catheter. (The use of a right heart catheter is not necessary.) This technology is based on the transpulmonary thermodilution technique and arterial pulse contour analysis and provides specific and quantitative parameters including arterial blood pressure, heart rate, cardiac output, global end-diastolic volume [an indicator of cardiac volume, the normal range of global end-diastolic volume index (GEDVI) is 680–800 ml/m2], intrathoracic blood volume [an indicator of thoracic blood volume, the intrathoracic blood volume index (ITBVI) is 850–1000 ml/m2], extravascular lung water [an indicator of pulmonary oedema, extravascular lung water index (EVLWI) is 3.0–7.0 ml/kg], cardiac function index, global ejection fraction, stroke volume, stroke volume variation SVV (an indicator of the potential for a response to intravascular filling, SVV should be less than 10%), pulse pressure variation (also an indicator of the potential for a positive response to intravascular filling) and systemic vascular resistance (an indicator of left ventricular afterload). (Pulsion Medical Systems) (Fig 8.5).
Mixed venous oxygen saturation. Normal oxygen saturation in the venous blood is 75% and mixed venous oxygen saturation (SvO2) reflects the adequacy of tissue perfusion. SvO2 falls when oxygen demand increases (such as stressful procedures, shivering, nursing care) and/or when oxygen delivery is inadequate (poor cardiac output as a result of heart failure). Increased SvO2, however, suggests failure of tissue cells to take up or utilize oxygen from the blood. An SvO2 of 30% or less suggests that oxygen delivery is insufficient to meet tissue oxygen demands and anaerobic metabolism and lactic acidosis will be likely accompaniments in such circumstances.
Monitoring of the neurological system
Level of consciousness
The Glasgow Coma Scale (Chapter 1) is the most common way to objectively index the level of consciousness. Pupil size and level of reactivity to light provides an index of neurological integrity (pupils equal and reactive to light (PERL)). A fixed dilated unilateral pupil indicates pressure on the oculomotor nerve and urgent investigation is necessary. Physiotherapy intervention should be delayed. Fixed dilated pupils indicate severe neurological impairment, which may be made worse by hypoxia or biochemical abnormalities and are often a sign of brainstem death.
Cranial computed tomography scan.
Computed tomography (CT) of the head provides information about the brain and skull. A plain skull radiograph may identify fractures of the skull and CT with contrast is used for investigation of intracranial space-occupying lesions (haemorrhage, tumour or abscess). Cranial CT permits visualization of the following (Kumar 1997):
Magnetic resonance imaging (MRI) is now more commonly used when scanning neurological patients.
Intracranial pressure.
Intracranial pressure (ICP) is measured by insertion of a catheter through the skull into the lateral ventricle or by means of an extradural or subarachnoid bolt (Turner 2002). ICP is often monitored in patients with head injuries, post-brain surgery and for patients with intracranial and subarachnoid haemorrhage or cerebral oedema. The intraventricular catheter has the advantage of allowing drainage of cerebrospinal fluid when the intracranial pressure is high but because it penetrates the dura, there is a greater accompanying risk of intracranial infection. Any change in the intracranial pressure is dependent upon the relative amounts of blood, brain and CSF within the adult skull. ICP allows determination of global cerebral perfusion pressure (CPP), which relates closely to cerebral blood flow (CBF) (Box 8.4).
Box 8.4 Calculation of cerebral perfusion pressure (CPP)
CPP = cerebral perfusion pressure; MAP = mean arterial pressure; ICP = intracranial pressure
Raised PCO2 results in an increase in cerebral blood flow, which will cause a rise in ICP and a lowering of CPP. Hyperventilation may lower the PCO2, thus reducing cerebral vasodilatation and CBF, thereby lowering the ICP. Normal ICP is 10–15 mmHg, but baseline levels are often higher in neurosurgical patients. In order to provide adequate perfusion to the brain, it is generally recommended that CPP should be maintained at a level greater than 60 mmHg (Huang et al 2006).
Jugular bulb oxygen saturation.
Jugular bulb oxygen saturation (SjO2) reflects the adequacy of global cerebral oxygen delivery, although monitoring of SjO2 is now rarely used in neurosurgical ICU. Monitoring of SjO2 is based on the principle that cerebral arterial and mixed venous oxygen difference (A–VDO2) is directly proportional to cerebral metabolic rate (CMRO2) but inversely proportional to cerebral blood flow (CBF). The normal values of SjO2 are between 55–71%. Less than 55% indicates an increase in cerebral oxygen extraction, often due to hypotension or systemic hypoxia. Over 70% indicates hyperaemia. The results have to be interpreted along with other information such as ICP (White & Baker 2002).
Measurement and monitoring of cerebral blood flow
Transcranial Doppler. Transcranial Doppler (TCD) is a non-invasive diagnostic tool that uses sound waves to measure the velocity of blood flow in the basal cranial arteries (Miller 2005). Blood flow velocity, however, is variable and dependent on the diameter of the cerebral arteries. Thus a dimensionless variable, the pulsatility index (PI) is used which is derived from the difference between systolic and diastolic flow velocities divided by the mean velocity. PI is a reflection of the cerebrovascular resistance and a high PI is associated with a low cerebral perfusion pressure (CPP) (Lindegaard 1992). A high correlation between PI and ICP (Voulgaris et al 2005) and CPP (Bellner et al 2004) has been reported. PI has a high predictive value for detecting a CPP of less than 70 mmHg (Voulgaris et al 2005).
Bispectral index (electroencephalographic analysis). The bispectral index (BIS) is a parameter that was developed to measure a patient’s level of awareness during sedation (Haug et al 2004) and to determine the probability of recovery of consciousness in patients in coma (Fabregas et al 2004). BIS is derived from an electroencephalogramic parameter, which includes time and frequency domains and higher-order spectral information. With electrode sensors placed on the forehead of the healthiest brain hemisphere, identified by computed tomography scan, a BIS recording can be quantified on a scale from 0 to 10. BIS correlates with clinical signs of hypnosis (Billard et al 1997, Rosow and Manberg 2001) and is reported to be predictive of traumatic brain injury and neurological outcome at discharge (Haug et al 2004).
PROBLEM IDENTIFICATION AND PHYSIOTHERAPEUTIC INTERVENTIONS IN ICU
Quality of patient care depends on appropriate patient assessment and identification of problems associated with presenting symptoms. Appropriate intervention involves complex decision-making processes. This section discusses broad problems encountered by patients in the ICU and the rationale for interventions to be undertaken. Case studies or common patient scenarios are presented to illustrate the principles of intervention.
Broad problems
Chapter 6 has comprehensively detailed potential problems that may occur in the respiratory patient, mechanisms, medical and physiotherapy management. Apart from the presenting problem, for example, severe pneumonia or multiple trauma, patient’s previous comorbidities, immobility, problems associated with intubation, ventilation and impaired nutrition should be considered. Problems particularly relevant to critically ill patients include:
Decreased lung volumes, compliance and gas exchange
Intubation, mechanical ventilation and the accompanying sedation can result in a number of adverse effects on the respiratory and cardiovascular system. Ventilation/perfusion mismatching may occur due to preferential ventilation of the non-dependent areas (increased dead space) of the lung while the poorly ventilated dependent areas still receive preferential perfusion (increased shunt) especially in the supine position. The monotonous pattern of positive pressure ventilation without spontaneous respiration may impair gas exchange (Hedenstierna et al 1985) and the absence of sighs during mechanical ventilation will lead to decreased surfactant release, decreased lung compliance and progressive pulmonary atelectasis (Antonaglia et al 2006). Decreased functional residual capacity (FRC) also occurs because of cephalad displacement of the diaphragm and loss of lung volumes, both of which occur predominantly in the dependent zones. Alveoli may develop different levels of resistance, those with high resistance taking a longer time to inflate. The different mechanical properties of alveoli may be interpreted as having varying time constants (the product of alveolar compliance × resistance). A long time constant indicates an alveolus that opens slowly during tidal inflation.
In the immobilized ventilated patient, progressive atelectasis will result in a further decrease in lung compliance and gas exchange. The patient may also have a diffusion defect due to factors such as pneumonia, alveolar thickening or acute respiratory distress syndrome (ARDS).
Decreased mucociliary/secretion clearance
Normal mucociliary clearance depends on a complex interaction between ciliated columnar cells in the tracheobronchial tree and special viscoelastic properties of the bronchial secretions. As well as the presence of an invasive airway, immobility and decreased conscious level, the intensive care patient may have a number of factors that specifically impair mucociliary clearance, which include:
Premorbid factors such as a history of smoking, chronic respiratory disease and/or severe neuromuscular disorders other than impaired respiratory muscle strength may also further impair mucociliary/secretion clearance.
Intubation and mechanical ventilation can inhibit the normal mucociliary clearance and be associated with secretion retention and pneumonia (Konrad et al 1994). A patient intubated and ventilated for longer than 48 hours has been shown to be heavily colonized with anaerobic bacteria (Agvald-Ohman et al 2003). The colonization of bacteria may be partly due to suction-induced lesions of mucous membranes. These bacteria are then capable of synthesizing and releasing factors capable of further impairing ciliary mobility and causing a loss in epithelial integrity. It is important that the physiotherapist understands the mechanisms of impaired mucociliary clearance in the intubated ventilated patient and appreciates which methods of intervention are effective.
Mucociliary clearance in the healthy, non-intubated patients includes the cough mechanism. In the intubated patient, mucociliary clearance may be facilitated by the mechanism of annular two-phase gas liquid transport. This is a non-ciliary dependent phasic flow with energy transmitted from moving air to static liquid, resulting in shearing of the secretions.
Weakness of respiratory and peripheral muscles
Mechanical ventilation for as little as 48 hours has been demonstrated to decrease diaphragm strength (Sassoon et al 2002) and endurance of respiratory muscles (Chang et al 2005).
A combination of the catabolic effects of the major illness, stress response, hospital-acquired infections and certain pharmacological agents can result in the loss of large amounts of muscle mass attributed to a proteolytic or protein degradation process or specific critical care weakness syndromes (Latronico et al 2005). General immobility also results in clinically significant bone demineralization and general impairment of orthostatic reflexes. This can result in increased time on mechanical ventilation, longer hospital stay and decreased quality of life on discharge.
Increased work of breathing
The primary reasons for mechanical ventilation are to decrease the work of breathing and optimize gas exchange. Mechanical ventilation can be applied to patients who are or are not making spontaneous respiratory efforts (Georgopoulos et al 2006). The patient’s respiratory system may either be passively ventilated (mandatory modes) or the patient may interact with the ventilator and trigger machine- supported breaths (in synchronized modes with set tidal volume or set pressure), or the patient may spontaneously breathe throughout the respiratory cycle interspersed with positive pressure breaths (bilevel ventilation).
There are many means of assessing the lung/thorax mechanics, work of breathing and metabolic cost of breathing in an intubated and mechanically ventilated patient (Table 8.1).
Table 8.1 Measures of work of breathing, lung thorax/mechanics and metabolic consumption
P0.1 = effort in first 100 msec in inspiration, NIF = negative inspiratory force, MIP = maximal inspiratory pressure, PEEP = positive end-expiratory pressure, f/Vt = rapid shallow breathing index, bpm = breaths per minute
Work of breathing in the mechanically ventilated patient will increase if there is asynchrony between the patient and the ventilator: that is, the ability of the mechanical ventilator to respond promptly to patient demand for flow during inspiration, to cycle from inspiration to expiration and to allow an unimpeded expiration.
An understanding of the basic waveform of ventilatory pattern allows the physiotherapists to obtain much information associated with the patient’s work of breathing during mechanical ventilation. This is covered later in this chapter.
Interventions
This section aims to describe interventions that have been specifically developed for critically ill and/or ventilated patients, including the rationale, indications, modifications and precautions of current respiratory techniques in these patients.
Positive pressure
Hyperinflation techniques.
The ventilated, critically ill patient often has an underlying problem associated with progressive atelectasis and loss of compliance combined with impaired mucociliary clearance. Hyperinflation techniques, manual hyperinflation (MHI) and ventilator hyperinflation (VHI) have been introduced in an effort to improve ventilation and secretion mobilization in a patient whose normal defensive mechanism is lost.
The aims of hyperinflation techniques are to:
 improve lung thoracic compliance (by increased tidal volume/inspiratory pressure, altered inspiratory : expiratory ratio or by increased positive end-expiratory pressure)
improve lung thoracic compliance (by increased tidal volume/inspiratory pressure, altered inspiratory : expiratory ratio or by increased positive end-expiratory pressure)Manual hyperinflation (MHI) is a technique whereby the patient is disconnected from the ventilator and given an altered breathing pattern via a valve circuit and reservoir bag (Fig. 8.6). To reduce the risk of barotrauma, it is recommended that a pressure manometer be incorporated into the circuit so that the peak airway pressure can be monitored during the MHI procedure.
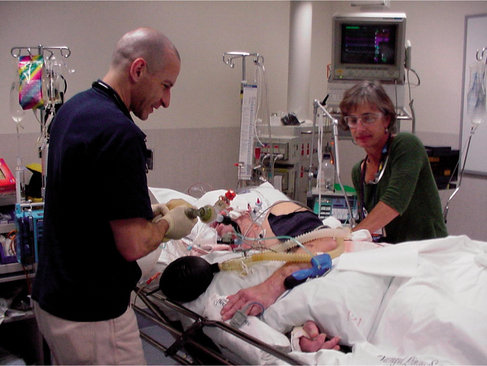
Figure 8.6 An intubated adult receiving manual hyperinflation and chest wall shaking/vibration (note blue pressure manometer, Laerdal MHI circuit with orange spring-loaded PEEP valve)
The ideal pattern includes a slow inspiration, an inspiratory hold and a fast release. This pattern hypothetically has beneficial effects on volume restoration, compliance and removal of secretions. Evidence and rationale for these mechanisms will be discussed below.
Various types of circuits are utilized. Expiratory flow rate and volume produced by the more pliable Mapleson and Magill circuits and the less pliable Air Viva and Laerdal circuits have been compared, both in laboratory and clinical studies (Jones et al 1991, 1992a, 1992b, Maxwell & Ellis 2003, McCarren & Chow 1998, Rusterholz & Ellis 1998). The Mapleson and Magill circuits provide greater tidal volume and faster expiratory flow. When a pressure manometer is not in use, the latter circuits may be safer.
Ventilator hyperinflation (VHI) has been used as an alternative to manual hyperinflation in ventilated patients. The method consists of leaving the patient attached to the ventilator and altering either the volume, pressure or flow/time characteristics of the breath delivered. This method of ventilation has been shown to result in similar levels of secretions removed and improvements in compliance as manual hyperinflation (Berney & Denehy 2002, Savian et al 2005).
Evidence for hyperinflation techniques
Effect on decreased volumes, compliance and oxygenation. As discussed, the critically ill patient may have low lung volumes and decreased lung compliance due to a number of factors. In order to reverse the process of decreased lung compliance, the inspiratory pressure must exceed a critical opening level to expand collapsed alveoli. A pressure of 40 cmH2O has been stated to be a minimum (Rothen et al 1999); however, other studies have resolved atelectasis using lower pressures (Maa et al 2005).
A slow inspiration procedure used in both MHI and VHI results in laminar flows, which encourage alveoli with prolonged time constants to reinflate. A larger tidal volume and inspiratory plateau promotes release of surfactant, which will reduce the surface tension of alveoli and assist re-expansion. The inspiratory hold also facilitates alveolar expansion via collateral ventilation.
MHI has been demonstrated to result in reversal of atelectasis (Stiller et al 1990), improvement in tidal volumes, improvement in chest radiograph scores (Maa et al 2005), improvements in static and dynamic compliance (Hodgson et al 2000, Jones et al 1992b, Paratz et al 2002, Patman et al 2000) and increased yield of secretions (Choi & Jones 2005, Hodgson et al 2000, Jones 2002).
Effect on mucociliary clearance. The fast release technique during the expiration phase of MHI encourages movement of the secretions in a cephalad direction. Patients on mechanical ventilation are normally sedated, with a consequence of reduced ability to cough. The movement of airway secretions in these patients is impaired and may be improved by an increased expiratory to inspiratory flow ratio (the ‘two-phase gas liquid transport’), which is proposed to occur during MHI or VHI (Savian et al 2005).
Laboratory and clinical studies have demonstrated that manual hyperinflation compared with a control manoeuvre results in higher peak expiratory flows, improved lung/thorax compliance and increased removal of secretions (Jones 2002). It appears that a critical expiratory flow must be reached during manual hyperinflation in order for sputum movement to occur and this is linked to the method of delivery of manual hyperinflation, including the type of circuit employed (Maxwell & Ellis 2003, Savian et al 2005); tidal volume; and rapid release of the valve and bag. Research on MHI to date has not established a link between the flow rates generated and the volume of secretions removed.
Inclusion of a positive end-expiratory pressure (PEEP) valve in the manual hyperinflation circuit has been shown to decrease expiratory flow. When the PEEP exceeds 10 cmH2O, MHI may be ineffective as a secretion clearance technique (Savian et al 2005).
Laboratory evidence has suggested that MHI may not be effective in mobilization of thin (low viscosity) secretions and an alternative technique such as gravity-assisted drainage may be more effective (Jones 2002).
A further method of hyperinflation that intensivists and some physiotherapists utilize are ‘recruitment manoeuvres’ (Mols et al 2006). Lung-protective strategies using low tidal volume ventilation are beneficial and improve survival in patients with acute respiratory distress syndrome. However, the low tidal volumes can cause tidal alveolar de-recruitment and atelectasis. A recruitment manoeuvre is a sustained increase in airway pressure with the goal to recruit atelectatic lung tissue. A number of methods are used, including increases in positive end-expiratory pressure (PEEP), sustained increased inspiratory pressure (e.g. 40 cmH2O PEEP for 40 seconds duration), or sigh breaths. Using these techniques, short-term increases in oxygenation and reversal of atelectasis have been reported (Barbas et al 2005).
Precautions for hyperinflation techniques
Haemodynamic instability. Haemodynamic instability is a vague term and can cover a multitude of events. Each intervention will have a different effect depending on the pathophysiology of the patient. The relationship between hyperinflation techniques and the haemodynamic status highlights the need to consider the following factors:
 an intervention such as manual hyperinflation or ventilator hyperinflation (which causes an increase in intrathoracic pressure) may lead to a decrease in preload and subsequently cardiac output and blood pressure. This is more likely in a patient who is hypovolaemic, i.e. has a decrease in overall circulating blood volume either absolutely (haemorrhage) or relatively (sepsis, opiates)
an intervention such as manual hyperinflation or ventilator hyperinflation (which causes an increase in intrathoracic pressure) may lead to a decrease in preload and subsequently cardiac output and blood pressure. This is more likely in a patient who is hypovolaemic, i.e. has a decrease in overall circulating blood volume either absolutely (haemorrhage) or relatively (sepsis, opiates) if the systemic blood pressure is below normal values and supported by inotropes or vasopressors – hyperinflation techniques could further decrease the blood pressure
if the systemic blood pressure is below normal values and supported by inotropes or vasopressors – hyperinflation techniques could further decrease the blood pressure a significant drift in the baseline of the arterial blood pressure waveforms before hyperinflation techniques may indicate that the patient may require increased filling (fluids) and may not tolerate hyperinflation techniques.
a significant drift in the baseline of the arterial blood pressure waveforms before hyperinflation techniques may indicate that the patient may require increased filling (fluids) and may not tolerate hyperinflation techniques.Undrained pneumothorax. This is usually an absolute contraindication. If an underwater drain is in situ and a large air leak is present, hyperinflation methods may be ineffective and may worsen the air leak. The presence of bullae on a plain chest radiograph may not be an absolute contraindication, but limiting peak inspiratory pressure with the aid of a manometer may ensure safety.
Severe bronchospasm and asthma. If the patient has acute asthma, an increase in positive pressure and tidal volume or the delivery of dry unhumidified gas may further increase intrinsic PEEP and aggravate hyperinflation and bronchospasm, thereby increasing the potential risk of barotrauma.
As a general rule, the manual hyperinflation technique must always be preceded, and followed, by auscultation. Clinicians should always be mindful of pneumothorax as a potential risk of the MHI technique.
High PEEP, nitric oxide, heat and moisture exchanger (HME) and hypoxic pulmonary vasoconstriction (HPV). If the patient is receiving high PEEP (>10 cmH2O) and/or nitric oxide as a ventilatory adjunct, it is advisable not to disconnect the ventilator circuit for manual hyperinflation techniques. Interruption to high levels of PEEP can cause de-recruitment and atelectrauma, especially in conditions such as ARDS (Mols et al 2006). Interruption of nitric oxide can cause sudden increases in pulmonary artery pressure and severe strain on the right side of the heart as well as potential severe hypoxaemia. Ventilator hyperinflation may be an alternative in these situations, but may dilute the amount of nitric oxide the patient is receiving, unless the patient is on an automated nitric oxide delivery device.
The use of a heat and moisture exchanger (passive humidifier) in the MHI circuit may optimize humidification and reduce airway irritation. Inhaled bronchodilators before, during or after MHI may be useful if severe bronchospasm is present.
Resolution of severe atelectasis as a consequence of MHI may, however, induce sudden hypoxemia. This is because the blood flow diverted from an atelectatic area due to hypoxic pulmonary vasoconstriction (HPV) may not respond efficiently to re-perfuse the newly re-inflated alveoli. Mismatch of ventilation and perfusion thus occur, leading to sudden hypoxaemia. The patient may need to be ventilated at a higher FiO2 until the pulmonary circulation to the recently re-inflated lung improves.
Non-invasive ventilation (NIV)
Biphasic positive airway pressure (BiPAP), continuous positive airway pressure (CPAP) and intermittent positive pressure breathing (IPPB) have been covered earlier and in Chapters 5 & 10. These modes of ventilation are of particular use in the critically ill patient in attempting to prevent intubation in respiratory failure or in weaning and extubation. Patients with chronic obstructive airways disease, chronic heart failure, obesity and renal failure are often at risk of needing reintubation and ventilation following extubation. These patients may benefit from some form of NIV post-extubation.
Manual techniques
Percussion and vibration.
Although manual techniques such as percussion of the chest wall and vibration during the expiratory phase are commonly used in intensive care patients, often in conjunction with hyperinflation techniques and positioning, individual studies of their effectiveness in this setting are currently lacking. Vibration has been shown to increase expiratory flow rate (MacLean et al 1989), but there are no clinical studies that demonstrate whether this increases removal of secretions. A series of animal and human studies by Unoki et al (2004) demonstrated that chest wall compression moved secretions in a cephalad direction.
Precautions.
Precautions applied to manual techniques have been discussed in Chapter 5. In intensive care patients, precautions also include decreased platelet levels, skin wounds and chest trauma.
Secretion removal techniques
Suction – open/closed.
As critically ill patients are usually intubated, regular pulmonary toilet must be applied. Formerly this was always via the open suction technique: that is disconnection of the endotracheal tube, instillation of a sterile catheter and application of a negative pressure. As the patient did not receive ventilation during this period, an efficient technique in less than 15 seconds was necessary. Most intensive care units now utilize the ‘in-line’ suction technique (closed-suctioning), whereby a sealed catheter is connected to the endotracheal tube and suction is possible without disconnection from the ventilator. This technique has been associated with less risk of desaturation and reduction in lung volume (Cereda et al 2001), fewer arrhythmias, less cardiovascular changes (Lee et al 2001) and less reduction of PEEP (Maggiore et al 2003). However, in pressure-controlled modes of mechanical ventilation, the negative pressure from the suction catheter may trigger ventilator breaths, and the inspiratory flow from the ventilator may force the secretions away from the catheter tip, resulting in fewer secretions being aspirated (Lasocki et al 2006). After suctioning, a lung recruitment technique such as MHI or VHI may be required to minimize the risk of atelectasis induced by the negative pressure suctioning generated by either the open or closed system.
Nasopharyngeal/oropharyngeal suction and mini-tracheotomy.
Nasopharyngeal and oropharyngeal suction have been discussed in detail in Chapter 5. These techniques are often necessary before and during extubation, as well as in attempt to prevent intubation in patients with inefficient coughing efforts or increased secretions.
Minitracheotomy is often utilized in intensive care and is invaluable for patients with secretion retention, weak cough and contraindications to or intolerance of oral/nasopharyngeal airways. However, as only size 10 French gauge suction catheters can be used, this may limit suction effectiveness in some patients. Also a mini-tracheostomy is an uncuffed tube, and hence will not prevent the patient from aspirating oropharyngeal secretions.
Increased moisture to airways
Humidification.
Humidification has been discussed in Chapter 5. Humidification is mandatory for patients on mechanical ventilation to reverse some of the adverse effects of intubation such as reduced tracheal mucus velocity and cilial impairment.
A critically ill patient on high concentration of inspired oxygen will also benefit from heated humidification. A heat and moisture exchanger (HME) can be used as an alternative but has been associated with increased circuit dead space and resistance to airflow. It may also be associated with an increased work of breathing in spontaneously breathing patients who are on low levels of respiratory support (Boots et al 2006). The use of HMEs may increase PaCO2 in patients with acute lung injury/acute respiratory distress syndromes (Moran et al 2006). Nebulization with normal saline via the ventilator circuit has been reported to increase the yield of airway secretions (O’ Riordan et al 2006).
Saline instillation.
Direct instillation of normal saline to the endotracheal tube during or prior to suction in an attempt to decrease viscosity of secretions is a frequently used (and yet sometimes controversial) technique. A number of studies have found that this practice results in decreased SaO2 and/or mixed venous saturation (Ackerman & Mick 1998), no increase in secretion yield (Lerga et al 1997) and possible dislodgement/dispersion of microorganisms into the lower respiratory tract (Hagler & Traver 1994). Schreuder & Jones (2004), however, demonstrated increased sputum wet weight and stable arterial oxygen saturation following use of saline when it was combined with chest physiotherapy. It is recommended that instillation of saline should be reserved for patients with excessively tenacious secretions. In addition, the clinician should anticipate a short-term drop in arterial saturation that may require a temporary increase in FiO2.
Positioning
The physiological effects and rationale of positioning have been covered in detail in Chapter 4. Altering the position of a critically ill patient is a powerful tool and may result in both beneficial and adverse effects. Cardiovascular changes associated with positional changes, especially in critically ill patients, should be closely monitored during physiotherapy. An adequate understanding of the pathophysiology of positioning and its predicted effects is essential.
Gravity-assisted positioning.
Traditional gravity-assisted positions (Chapter 5) are often not utilized in intensive care patients as full positioning is often hindered by cardiovascular instability, equipment and lack of patient cooperation. However, evidence suggests that specifically positioning the patient for the affected lobe results in increased expiratory flow rate, better oxygenation, increased sputum clearance and faster resolution without adverse effects on haemodynamic stability (Berney et al 2004, Krause et al 2000).
Prone positioning.
Specific prone positioning for extended periods of time has been advocated as a method to improve oxygenation and lung mechanics in patients with acute lung injury and acute respiratory distress syndrome. There is strong evidence that this method results in improved lung mechanics and oxygenation due to expansion of the collapsed dorsal regions of the lung (Messerole et al 2002). This technique is most useful if used early in the disease process and may also result in increased secretion clearance due to drainage of the collapsed dorsal regions of the lung. Reduction in carbon dioxide with prone positioning is indicative of improved alveolar ventilation and is predictive of better survival (Gattinoni et al 2003).
Lateral positioning.
The effects of lateral positioning will depend on pathology of the lung, whether unilateral or bilateral and type of ventilation. To maximize alveolar expansion, lung segments to be expanded are often placed in the upper most (non-dependent) lateral position for facilitation of aeration, especially with positive pressure ventilation. However, blood flow will preferentially move to the dependent lung (even more so during positive pressure ventilation); hence, there may be potent effects on gas exchange dependent upon the extent of pulmonary disease (unilateral or bilateral).
In patients with unilateral lung disease, gas exchange may improve by laying the patient on the non-diseased lung (Ibanez et al 1981, Stiller 2000). This may also facilitate secretion drainage. In adopting lateral positioning to optimize gas exchange, the physiotherapist should be aware of the mode of ventilation, monitored variables (tidal volume, airway pressures), inotropic and vasoactive requirements and cardiovascular status (blood pressure, heart rate). For example re-positioning a heavily sedated intubated patient who is receiving a pressure-controlled mode of ventilation (such as pressure support) and who has copious secretions and a poor cough, may lead to severe reductions in tidal volume (and hence minute ventilation) due to the movement of secretions in the major airways which may alter airway resistance.
Continuous lateral rotation therapy or kinetic therapy.
Continuous lateral rotation therapy or kinetic therapy is a relatively new innovation in intensive care and consists of continually changing the position of the patient (to extreme lateral position) in specially designed hydraulic beds. The beds are costly but have been proposed to increase the clearance of airway secretions (Davis et al 2001), reduce the rate of development of ventilator-associated pneumonia (VAP) (Dodek et al 2004, Kirschenbaum et al 2002) and resolve atelectasis if combined with percussion (Raoof et al 1999). However, these beds have not been shown to result in improved patient outcomes such as time on mechanical ventilation or time in the intensive care unit and there are reports of high rates of patient intolerance of the beds.
Precautions in positioning.
Precautions in positioning are similar to those of postural drainage (Chapter 5). Each patient should be assessed individually, especially for the presence of severe cardiac disease, as indiscriminate head-down tilt for extended periods in ventilated patients with cardiac failure has been shown to result in major arrhythmias (Artucio & Pereira 1990) and right side lying has been shown to reduce blood pressure in critically ill patients with cardiac decompensation (Bein et al 1996).
Mobilization
Passive/active exercises.
While passive movement is commonly employed by physiotherapists in maintenance of joint range and muscle length for unconscious and semi-conscious patients in the ICU, there is minimal evidence to support its use. There is emerging evidence that continuous passive movement or electrical stimulation may prevent protein degradation and/or induce induction of messenger ribonucleic acid and c-fos in immobilized subjects (Griffiths et al 1995, Zador et al 1999). When a patient can actively move his limbs, passive limb mobilization exercise is usually no longer indicated.
Sit out of bed.
Sitting up in bed, sitting over the edge of the bed, and sitting the patient out of bed, allows the diaphragm to descend, thus increasing functional residual capacity and facilitating efficient gas exchange. A sitting position also has the advantage of increasing wakefulness and alertness and therapist–patient communication is promoted.
Tilt table.
If a patient is unable to stand due to either decreased power in the hip and knee extensors, placing the patient on a tilt table is an option. Tilt tables have been shown to be in common use in the majority of intensive care units in Europe and Australia (Chang et al 2004a, Norrenberg & Vincent 2000). Increased minute ventilation without adverse haemodynamic changes has been demonstrated when patients are placed on a tilt table (Chang et al 2004b). However, at present there is no evidence for long-term benefit from use of this equipment although weight bearing stimuli have been proven to result in prevention and reversal of osteopenia in medical patients (Jorgensen et al 2000).
Stand, walking.
As soon as a patient is deemed capable (Box 8.5), standing or mobilizing is ideal for the intensive care patient. This is often initially supported by equipment such as a walking frame, oxygen or portable ventilation. Orthostatic reflexes may be impaired by immobilization and therefore monitoring of haemodynamic changes is essential. Short-term improvements in tidal volume, inspiratory flow rate and minute volume with mobilization to standing were demonstrated in intubated, ventilated, abdominal surgical patients (Zafiropoulos et al 2004).
Absence of active mobilization (tilt table, walking or sit out of bed) in intensive care has been shown to be associated with an increased risk of readmission to ICU (Paratz et al 2005). Chiang et al (2006) have demonstrated that daily rehabilitation assisted in reduced time on mechanical ventilation and improved functional status in long-term mechanical ventilator-dependent patients.
Precautions in mobilization.
All intensive care patients are potentially unstable and an important aspect of exercise or mobilization is to achieve a fine balance between progressing the patient and not causing any deterioration in cardiovascular status. While these techniques are relatively simple, the ability to judge when critically ill patients are sufficiently stable to begin such rehabilitation requires considerable expertise. The safety aspects of mobilizing intensive care patients have been extensively reviewed by Stiller et al (2004).
Combined management
For optimal patient management, the above methods of intervention are often combined (e.g. positioning, manual hyperinflation, expiratory vibrations and suction of the endotracheal tube). A combination of techniques has been shown to be effective in increasing secretion yield and reducing airway resistance (Choi & Jones 2005). A controversial point in physiotherapy management of the patient in ICU is whether mechanically ventilated patients should be treated prophylactically, i.e. before problems actually arise. Ntoumeno-poulos et al (2002) demonstrated that prophylactic physiotherapy treatment could result in a decrease in ventilator-associated pneumonia by as much as 31%, demonstrating that preventative treatment is of value.
Conditions in ICU
This section describes the management of some conditions commonly encountered in the ICU. Common problems associated with the conditions are illustrated as case studies.
Acute lung injury (ALI)/acute respiratory distress syndrome (ARDS)
Acute respiratory distress syndrome refers to ‘a clinical syndrome caused by a wide variety of events and characterized by acute onset, refractory hypoxemia, decreased compliance and bilateral diffuse infiltrates on chest radiograph’ (Wilson et al 2001). The term acute lung injury (ALI) was often used and confused with acute respiratory distress syndrome (ARDS). The American-European Consensus Conference (AECC) in 1992 recommended that the term ALI should be used in a ‘broader’ sense and ARDS used for the more severe illness with poor oxygen status. Thus all patients with ARDS suffer ALI but not all patients with ALI will have ARDS. The criteria for diagnosis of ALI and ARDS as set by the AECC are:
Thoracic imaging (both plain radiographs and computed tomography) is one of the essential components in diagnosis and assessment of ARDS.
Pathogenesis of acute respiratory distress syndrome.
Acute respiratory distress syndrome (ARDS) carries a mortality rate of 50–80% (Metnitz et al 1999) and is often associated with sepsis, further increasing the likelihood of mortality. The exact mechanisms involved in the pathogenesis of ARDS are unknown, although infiltrating leukocytes and widespread endothelial injury are typical. Alveolar and pulmonary microcirculatory endothelial injury leads to normal inflammatory responses characterized by the release of cytokines and recruitment of neutrophils to the area of inflammation (Zimmerman et al 1999). This initiates a number of reactions in the lungs, which lead to hypoxaemia.
Patients with ARDS are often administered high concentrations of oxygen, which may further exacerbate the primary lung injury. The release of reactive oxygen species (ROS) causes damage to the alveolar surfactant system and decreases the ability of cells to transport sodium actively across epithelial membranes – an important process in the removal of alveolar fluid. These detrimental effects on lung function, as a consequence of prolonged mechanical ventilation and oxygen therapy, are further aggravated by other risk factors such as old age and sepsis (Wilson et al 2001).
These conditions cause a general inflammatory response with damage to the alveolar–capillary interface, leading to leakage of fluid into the interstitial space/alveoli and resulting in reduced compliance and shunting. Patients are often dyspnoeic, tachypnoeic and severely hypoxaemic. Management revolves around ‘protective ventilation’; that is low tidal volumes and the maintenance of optimal PEEP (MacIntyre 2005). If high tidal volumes are given and the ventilator is frequently disconnected, barotrauma, volutrauma and and/or atelectrauma may result. These syndromes are basically results of damage from high pressure, high volume and repeated deflation and inflation of alveoli. Biotrauma may also result, where high pressure, volume or shearing of alveoli may result in leaking of inflammatory substances from the lung causing a multi-organ system failure.
ARDS can act as one of two distinct pathological types of disease according to the initial type of insult (Gattinoni et al 1998). Those associated with intrapulmonary causes, for example respiratory burns and aspiration pneumonia, tend to have a non-compliant lung with pathology typical of consolidation. If patients have had an extrapulmonary insult, for example pancreatitis or head injury, the patient will have a stiff thoracoabdominal cage and compliant lung with pathology similar to atelectasis. Studies have shown that the latter type (extrapulmonary) is more amenable to hyperinflation manoeuvres such as manual or ventilator hyperinflation (Paratz et al 2002, Pelosi et al 2003). Prone positioning may be beneficial. In the past physiotherapists were advised that any intervention should occur when the patient was in the subacute stage of ARDS. However it has been found that certain interventions such as recruitment of the lung and prone positioning are more successful when introduced early in the disease (Pelosi et al 2002).
How would you manage the two patients in Case studies 8.1 and 8.2?
CASE STUDY 8.1
A 35-year-old male with multiple fractures requires replacement of 5 litres of blood following a motor vehicle accident (MVA). On Day 2 after admission, he develops tachypnoea and bilateral pulmonary infiltrates. His PaO2/FiO2 decreases to 220 and his pulmonary arterial occlusion pressure (PAOP) is 14 cmH2O. He is ventilated on SIMV (16 breaths/minute × 500 ml tidal volume, PEEP 7.5 cmH2O, pressure support 10 cmH2O, FiO2 0.4). His peak pressures on a breath of 500 ml are 27 cmH2O.
CASE STUDY 8.2
A 40-year-old male develops community-acquired pneumonia (right lower lobe) and is admitted to a medical ward with an FiO2 of 0.3 and a PaO2 of 95mmHg. He is expectorating sputum and receives secretion mobilization techniques for 3 days. By Day 4 he is non-productive, increasingly tachypneoic and hypoxaemic (FiO2 0.4, PaO2 75) with bilateral infiltrates on chest radiograph. He is ventilated in intensive care and deteriorates further. On examination, he is on pressure-controlled ventilation (inspiratory pressure 32 cmH2O), respiratory rate 15 breaths /minute, FiO2 0.6, PEEP 10 cmH2O. His tidal volumes on this inspiratory pressure are only 300 ml.
What is your clinical reasoning for the difference in management between these two patients?
Discussion of Case studies 8.1 and 8.2
These two patients differ with respect to the aetiology of their ARDS as well as lung compliance and ventilation method. The patient in Case study 8.2 has lower lung compliance, the ventilation method is aiming to control the pressure and only small volumes are produced by this pressure. As his cause of ARDS was ‘intrapulmonary’, the pathology of his condition is similar to that of ‘consolidation’. If hyperinflation methods are used, very little increase in tidal volume will occur without exceeding a safe pressure. He is also on a high PEEP and, if disconnected, will lose the recruitment gained and be predisposed to atelectrauma. If there was evidence of retained secretions, interventions to mobilize secretions such as percussion or vibrations, while maintaining ventilation and closed suction, could assist.
In contrast, the patient in Case study 8.1 has a less severe form of the disease (ALI P/F ratio, i.e. PaO2/FiO2 is >200) and has reasonable lung compliance with reserve to provide hyperinflation without exceeding recommended peak inspiratory pressures. His type of lung injury is ‘extrapulmonary’ and more amenable to further recruitment. He is not on high PEEP, but a PEEP valve should still be included in the manual hyperinflation circuit. This patient may also benefit from prone positioning, especially as his injury is extrapulmonary and he is in the early stage of the syndrome.
Ventilator-associated pneumonia (VAP)
Ventilator-associated pneumonia (VAP) is an infection that can occur more than 48 hours after starting ventilation and has been quoted as occurring in 30–50% of intensive care patients, with a mortality between 20–70%. The condition extends length of ventilation and length of stay in ICU.
Diagnosis of VAP.
Clinical criteria for the diagnosis of VAP are new or progressive pulmonary infiltrates and at least two of the following (Chytra 2002):
Blood cultures are considered low in both specificity and sensitivity, as critically ill patients in ICU often have multiple potential sources of infection. Cultures of the lower respiratory tract and histological examination of lung tissue are considered a more reliable process for diagnosing VAP. Proximal airway sampling (tracheal tube aspirates) and distal airway sampling (aspiration, brushing and bronchoalveolar lavage) can be achieved using both bronchoscopic and non-bronchoscopic techniques. Both techniques, however, may be subject to error owing to contamination during passage through the upper airway, which is invariably colonized by organisms. Thus ‘quantitative cultures’ and bronchoscopic ‘protected catheter brushing’ are often used. Quantitative culture can differentiate ‘contaminants’ from ‘pathogens’. Often a growth threshold of 103 cfu/ml is used to define organism growth. This ‘quantity’ is equivalent to 105−106 organisms/ml in the secretions.
Treatment of VAP.
The mortality attributable to VAP is significant and therefore prompt administration of appropriate empiric antibiotic therapy directed at the most prevalent and virulent pathogens is essential. As the most common pathogens are Pseudomonas, Enterobacter, Acinetobacter, as well as gram-positive organisms, multi-drug therapy is often required (Bowton 1999), although the use of monotherapy versus combination therapy remains controversial (Chytra 2002). Chastre (2006) has undertaken a review on antimicrobial management in VAP.
There is strong evidence (Minei et al 2006) that measures such as semi-recumbent positioning, continuous turning, handwashing, aspiration of subglottic secretions, selective digestive contamination and early tracheotomy all result in a decreased incidence of VAP. Physiotherapists have a definite role in the prevention of VAP. Ntoumenopoulos and colleagues (2002) found a decreased incidence of VAP (39% vs 8%) as a consequence of physiotherapy intervention.
Burns
Patients with thermal injury are frequently admitted to intensive care. Even if there is no direct respiratory burn, these patients often require mechanical ventilation, due to haemodynamic problems, decreased immune response, severe infections and secondary respiratory compromise (Monafo 1996). Respiratory burns are indicated by a history of burns in an enclosed space, facial burns, loss of consciousness before evacuation from the fire, stridor or wheezing, carbonaceous sputum, oedema and erythema on bronchoscopy. Damage to the mucosal barrier and release of inflammatory mediators are the major pathophysiological events (Bargues et al 2005). Mucosal sloughing often occurs and along with carbon must be cleared from the lungs. There is therefore a strong indication for respiratory physiotherapy (Sheridan 2000).
Patients with respiratory burns require expert respiratory care in addition to musculoskeletal rehabilitation. A number of considerations apply to the respiratory management:
 The patient may be haemodynamically unstable – in the first 2 days fluid resuscitation is an issue and measurements such as urine output, blood pressure and CVP will indicate the overall fluid status. Myocardial ‘stunning’ often occurs in the first 3 days leading to decreased contractility. This is followed by an ongoing hypermetabolic and catabolic state. The patient has a markedly depressed immunological state and is at increased risk of sepsis, especially from a pulmonary source.
The patient may be haemodynamically unstable – in the first 2 days fluid resuscitation is an issue and measurements such as urine output, blood pressure and CVP will indicate the overall fluid status. Myocardial ‘stunning’ often occurs in the first 3 days leading to decreased contractility. This is followed by an ongoing hypermetabolic and catabolic state. The patient has a markedly depressed immunological state and is at increased risk of sepsis, especially from a pulmonary source. Chest wall burns and/or grafting may be a precaution for manual techniques such as percussion or vibration. Consideration should be given to the depth of burn, adequacy of pain relief, and type, timing and viability of the graft. Good communication should occur with the burn surgeons regarding the graft. Vibrations have a larger shearing force than percussion and should be used as an intervention at a later stage of graft healing.
Chest wall burns and/or grafting may be a precaution for manual techniques such as percussion or vibration. Consideration should be given to the depth of burn, adequacy of pain relief, and type, timing and viability of the graft. Good communication should occur with the burn surgeons regarding the graft. Vibrations have a larger shearing force than percussion and should be used as an intervention at a later stage of graft healing.Sepsis and systemic inflammatory response syndrome (SIRS)
Patients admitted with, or acquiring an infection in ICU often develop sepsis, that is a systemic response to infection. Measurements such as respiratory rate, heart rate and temperature will change to defined levels. If this condition worsens, sepsis syndrome, that is sepsis with evidence of organ dysfunction, for example hypoxaemia or renal failure, may develop.
Septic shock is the most extreme manifestation of this condition and refers to sepsis syndrome with hypotension despite adequate fluid resuscitation. There is widespread fluid leakage, peripheral vasodilatation and often an inadequate circulating volume. Patients require inotropic support to maintain an adequate blood pressure and are often monitored with a pulmonary artery catheter.
An inflammatory reaction may also develop to a non-infectious insult such as pancreatitis, burns or post organ transplantation. This is termed systemic inflammatory response syndrome (SIRS). Criteria for the diagnosis of SIRS include two or more of the following: altered temperature >38.0°C or <36.0°C; heart rate >90 beats /minute; respiratory rate >20/min; PaCO2 <32 mmHg; white cell count >12 × 109/l (Worthley 2000). This syndrome can overlap with syndromes of sepsis, but the important difference is that patients with SIRS are more haemodynamically stable than those patients with sepsis and can tolerate most physiotherapy interventions.
In 2002, at the European Society of Intensive Care Annual Congress, the ‘Surviving Sepsis Campaign’ was launched, leading to publication of a document for critical care providers and health agencies to reduce the sepsis mortality rate by 25% in 5 years. The recommendations include a ‘sepsis care bundle’ to optimize patient care. Some of the measures in the ‘bundle’ are the early provision of oxygen therapy, intravenous access, blood cultures, serum lactates, immediate intravenous fluids, blood glucose control, nursed 30-degrees head-up and protective lung ventilatory strategy (to minimize airway pressures/tidal volumes). It has been demonstrated that lack of adherence to ‘sepsis care bundles’ in the first 24 hours of sepsis, results in worse patient outcome (Gao et al 2005).
The scenarios in Case studies 8.3 and 8.4 are common in patients with SIRS and/or sepsis.
CASE STUDY 8.3
A 24-year-old male received second- and third-degree burns to 40% of his arms and legs and respiratory burns, in an industrial accident 2 weeks ago. He remains intubated on spontaneous ventilation PEEP 5 cmH2O and pressure support (PS) 10 cmH2O, FiO2 0.3. He has had grafting to the burns and these have healed. However his vital signs are: temperature 39.5°C, heart rate 135 beats/min (regular) and BP 120/80 mmHg (unsupported). The high heart rate and temperature have persisted for 1 week with no evidence of an infective site despite multiple testing.
These vital signs above and time period described are typical of SIRS. Note the stable blood pressure. This patient could be mobilized, tilted, sat out of bed or given MHI if required. It would be advisable to monitor the heart rate and blood pressure, but his circulation should remain stable during these procedures.
CASE STUDY 8.4
A 46-year-old female is admitted in respiratory failure following diagnosis of severe community-acquired pneumonia and ventilated on synchronized intermittent mandatory ventilation (SIMV) 12 breaths × 600 ml, PEEP 7.5 cmH2O, FiO2 0.4 and pressure support 10 cmH2O. On Day 6 in ICU, her blood pressure de-creases to 85/60 mmHg and heart rate increases to 110/minute. A pulmonary artery catheter is inserted and the following values are noted: cardiac index (CI) = 4.3 l/min/m2 (normal: 2.5–4.2 l/min/m2), systemic vascular resistance index (SVRI) = 1800 dyn ¨ sec/cm−5 ¨ m2 (normal: 1970–2390 dyne ¨ sec/cm−5 ¨ m2) and pulmonary artery occlusion pressure (PAOP) = 6 mmHg (normal: 4–12 mmHg). She is given a large volume of fluid resuscitation and the blood pressure increases to 95/60mmHg. Noradrenaline 5 µg/min increasing to 9 µg/min over 2 hours is administered.
This patient obviously has developed septic shock and she is in a ‘hyperdynamic state.’ The low SVRI indicates that the peripheral circulation is widely vasodilated and the patient does not have adequate circulating fluid. If an intervention of either increased positive pressure (MHI or VHI) or mobility against gravity was given at this stage, there would be inadequate compensatory reflexes and the circulation would fail. The physiotherapist needs to delay these procedures until fluid resuscitation is complete; inotrope/vasopressors levels are decreasing; and blood pressure and SVRI values are in the normal range (Paratz & Lipman 2006, Paratz et al 2002).
Chest trauma
Chest trauma can range from a single rib fracture to multiple rib fractures with a ‘flail’ segment and underlying contusions. Accompanying injuries may also include haemothoraces, pneumothoraces and solid organ injury (for example to the liver). Patients are admitted to intensive care based on whether they can effectively maintain ventilation, but other criteria depend on whether there are other injuries such as head trauma or laceration to organs such as the liver or spleen. Further risk factors for deterioration following chest trauma include age greater than 65 years old and pre-existing respiratory disease. Elderly patients with three or more rib fractures have been shown to have a 5 times greater increase in mortality and a 4 times greater increase in the incidence of pneumonia (Stawicki et al 2004).
The current management of chest trauma is directed towards effective pain relief, avoidance of fluid overloading, early mobilization and avoidance of invasive ventilation if possible. If a flail segment is present, an adequate end-expiratory pressure is required to ‘splint’ the flail segment in order for the patient to ventilate effectively.
Figures 8.7A and 8.7B describe the pathophysiological changes present in a severe chest trauma with a lung contusion and the physiological consequences.
Figure 8.7 (A) Pathophysiological changes following chest trauma with lung contusion. (B) Physiological consequences of severe chest trauma.
These consequences often do not occur until Day 2 or 3, leading to a late deterioration. Management of the chest trauma patient must therefore be proactive and aimed at restoring effective ventilation, reversing atelectasis and mobilizing secretions, using a combination of management techniques. Transcutaneous electrical stimulation (TENS) can be an effective adjunct for pain relief even with an epidural in situ.
Critical illness weakness syndromes
Critical illness weakness syndromes often occur following multisystem organ failure and result in the patient surviving the initial acute problem, but having a major peripheral weakness (including cranial nerves) which often prevents weaning and mobilization. Both critical illness polyneuropathy and myopathy may occur (Latronico et al 2005). The syndrome appears to be related to use of neuromuscular blocking agents, steroids and sedation especially following ventilation for severe asthma. These patients usually require admission to a rehabilitation unit; however, there is no evidence as to whether this accelerates recovery or to the optimal method of rehabilitation. The syndrome can affect function and quality of life for 12 months post-ICU discharge. There is some preliminary evidence that proactive treatment during the paralysis stage, for example continuous passive movement, may prevent this problem (Griffiths et al 1995). With the use of spontaneous modes of mechanical ventilation, and decreased use of steroids or neuromuscular blocking agents, this syndrome may prove less of a problem in intensive care.
Haematological problems
Patients with haematological conditions (e.g. leukaemia) often develop secondary respiratory problems following bone marrow transplants and require admission to intensive care as well as mechanical ventilation. This is more likely in patients with decreased lung volumes due to pulmonary fibrosis caused by pre-transplant irra diation (Shankar & Cohen 2001). It has been demonstrated that non-invasive ventilation applied proactively to haematological patients with lung infiltrates and hypoxaemia results in decreased mortality (Hilbert et al 2001). The benefits of non-invasive ventilation in the management of patients with haematological conditions warrant further investigation.
It is essential for the platelet count of these patients to be checked before interventions. Patients with a low platelet count (<20 ×109/l) may bleed spontaneously and manual techniques such as percussion, vibration and resisted exercise should be avoided. Soft-tipped catheters should be used for suction in patients with low platelet counts. Alternative methods of secretion removal such as the active cycle of breathing techniques, positive expiratory pressure (PEP), oscillatory PEP and autogenic drainage should be considered if the patient is able to cooperate.
Brain injury
Injury to the brain may occur due to trauma (local haematoma or diffuse brain injury) or post subarachnoid haemorrhage. It is important to remember that while primary damage is irreversible, the outcome can also be affected by indirect or secondary brain damage to the brain due to such events as hypoxaemia, hypercarbia, cerebral oedema or hypotension. The brain injury patient therefore needs to be expertly managed. Normal management of the head-injured patient involves the following principles (Dutton & McCunn 2003):
This is achieved by the following management strategies which may address either one or all three of the above aims:
 sedation and paralysis in the early stage (days 1–3) in order to limit increases in ICP from coughing and struggling and also to decrease brain metabolism
sedation and paralysis in the early stage (days 1–3) in order to limit increases in ICP from coughing and struggling and also to decrease brain metabolismFactors such as ICP, CPP, brain CT scan, changes on chest radiograph and arterial blood gases need to be noted in order to balance the significance of injury of one system over the other. For example a patient with chest X-ray changes, borderline arterial blood gases and low to medium ICP may be given active intervention but a patient with a clear chest radiograph, normal arterial blood gases and high ICP may be treated with position changes only.
When planning intervention in the acute severe head injury patient, consideration should be given to the following:
 Keep neck strictly in midline – rotation will block CSF and venous blood and increase ICP. Nurse with head-up 30 degrees.
Keep neck strictly in midline – rotation will block CSF and venous blood and increase ICP. Nurse with head-up 30 degrees. Suction will increase ICP; severe increases in ICP following suction may be prevented by intravenous or topical lidocaine (lignocaine) (Brucia et al 1992).
Suction will increase ICP; severe increases in ICP following suction may be prevented by intravenous or topical lidocaine (lignocaine) (Brucia et al 1992). ICP is likely to increase during a combined physiotherapy intervention (MHI, suction, manual techniques) as a rate of time. Short treatment sessions (<8 minutes) are preferable to a long session (Paratz & Burns 1993, Rudy et al 1991).
ICP is likely to increase during a combined physiotherapy intervention (MHI, suction, manual techniques) as a rate of time. Short treatment sessions (<8 minutes) are preferable to a long session (Paratz & Burns 1993, Rudy et al 1991). Coordinate management – do not allow patient to receive too many interventions e.g. chest X-ray, turning, physiotherapy in quick succession. This is more likely to cause increased ICP.
Coordinate management – do not allow patient to receive too many interventions e.g. chest X-ray, turning, physiotherapy in quick succession. This is more likely to cause increased ICP. During manual hyperinflation an end tidal CO2 monitor should be used in order to control the PaCO2 between 30–35 mmHg or baseline value.
During manual hyperinflation an end tidal CO2 monitor should be used in order to control the PaCO2 between 30–35 mmHg or baseline value. Head-down postural drainage or prone positions are likely to increase ICP. Research has shown that prone positioning may be well tolerated (in terms of ICP) in head-injured patients (Nekludov et al 2006, Thelandersson et al 2006).
Head-down postural drainage or prone positions are likely to increase ICP. Research has shown that prone positioning may be well tolerated (in terms of ICP) in head-injured patients (Nekludov et al 2006, Thelandersson et al 2006).Severe neuromuscular disease
Acute chest infection with an increase in respiratory secretions may precipitate an acute or chronic episode of respiratory failure in patients with chest wall and or neuromuscular disease (i.e. motor neuron disease, Duchenne muscular dystrophy, amyotrophic lateral sclerosis). Mechanical ventilation further impairs secretion clearance and increases the risk of ventilator-associated pneumonia (Konrad et al 1994) and patients with neuromuscular disease often require prolonged weaning from mechanical ventilation (Bach 1993). Improved secretion clearance may be important in the recovery process, but has yet to be proven. Boitano (2006) provides a comprehensive review of the factors necessary for, and optimal means of, enhancing secretion clearance in the neuromuscular diseased patient.
Manual or ventilator hyperinflation, combined with patient positioning and airway suctioning, have been demonstrated to produce significant improvements in static lung/thorax compliance and wet weight of sputum (Berney et al 2004, Hodgson et al 2000) compared with patient positioning and airway suctioning alone, in general medical surgical intubated and ventilated patients. These techniques alone may not be sufficient in the patient with severe neuromuscular weakness.
Patients with severe neuromuscular dysfunction often have a combination of problems, including reduced inspiratory and expiratory muscle strength, increased volume and tenacity of airway secretions, bulbar dysfunction (non-intubated), increased airway resistance and reduced lung/thorax compliance (Boitano 2006). For these patients, conventional chest physiotherapy techniques may be less effective. Hence, newer mechanical devices such as the mechanical in/exsufflator (JH Emerson Co, Mass, USA) have been introduced. This technique combines two approaches: a positive pressure (usually 40 cmH2O) is first applied to produce a maximum inspiration capacity; then, once the patient is ‘fully inflated,’ a sudden negative pressure (usually −40 cmH2O) is employed to assist the patient cough. The in/exsufflation device has been reported to improve peak cough expiratory flow rates in non-intubated patients with severe respiratory muscle weakness due to various neuromuscular disorders (Bach & Saporito 1996, Chatwin et al 2003) but the technique has been poorly investigated to date in intubated patients (Pillastrini et al 2006, Sancho et al 2003). However, one retrospective study (Garstang et al 2000) investigated tracheostomized spinal cord injury (C1-T3) patients’ preference for in/exsufflation for secretion management. In a direct comparison, the results indicated that 89% of patients preferred in/exsufflation to suctioning. In addition, 89% of patients found in/exsufflation faster, 78% found in/exsufflation more convenient and 72% found in/exsufflation more effective than suctioning. Manual-assisted cough (abdominal compression during the expiration phase following a maximal inspiration) (Chapter 15) is another effective means of enhancing expiratory flow and hence secretion clearance (Boitano 2006).
PHYSIOTHERAPY MANAGEMENT OF WORK OF BREATHING AND CONCEPTS OF WEANING FROM MECHANICAL VENTILATION
The primary reasons for mechanical ventilation are to decrease the work of breathing and optimize gas exchange. Synchronized intermittent mandatory ventilation (SIMV) and assist control were the traditional ventilatory modes (Esteban et al 2000). Dual modes (combination of pressure and volume-controlled) of ventilation are becoming more commonly used and it appears that evolution of mechanical ventilation is in the direction of computer-driven protocolized weaning (Lellouche et al 2006).
While increased sedative and narcotic use can achieve greater patient–ventilator synchrony, in the short term (Richman et al 2006) these agents, as well as neuromuscular blocking agents, have been shown to be associated with an increased duration of mechanical ventilation, weaning time and time in the ICU (Arroliga et al 2005). During activities when the patient’s demand of ventilation may increase (e.g. physiotherapy or situations such as anxiety and sepsis) a simple manoeuvre such as increasing the inspiratory flow rate or a change over to a pressure-controlled mode may ensure patient comfort and assist in the reduction of sedative requirement. A better understanding of the interaction between the ventilator and the patient will thus facilitate the role of the physiotherapist in the management of patients under mechanical ventilation. This section will discuss the concepts of work of breathing in patients under mechanical ventilation and issues that facilitate the weaning of patients from mechanical ventilation.
Work of breathing
Work of breathing may include the work undertaken by the patient as well as the work by the ventilator. The active contraction of the respiratory muscles causes the thoracic compartment to expand, inducing a negative pleural pressure. This negative pressure, generated by the respiratory muscles, produces lung expansion through a decrease in alveolar pressure, causing air to flow into the lungs (Cabello & Mancebo 2006). This driving pressure can be generated in three ways:
While minimizing the patient’s work of breathing is the main interest for most clinicians, the optimal balance between the level of work from the patient and the level of support from the ventilator is still unknown. The patient’s work of breathing is dependent on the type of pulmonary disease, respiratory muscle strength, airway (and/or tracheal tube) diameter, airway secretions, anxiety, sedatives, narcotics, neuromuscular blocking agents, the mode of mechanical ventilation/settings and the level of synchrony between the patient and the mechanical ventilator.
A patient’s work of breathing may be described as the amount of muscle activity required to overcome the elastic (lung tissue, chest wall and abdominal compartments) and resistive (airways, flow rate) elements of the respiratory system.
The waveform of the patient’s ventilatory pattern can provide much information about the lung/thoracic compliance and airway resistance. Figure 8.8 illustrates a typical volume-delivered breath with an inspiratory pause (generated by closure of the ventilator valves at the end of inspiration).
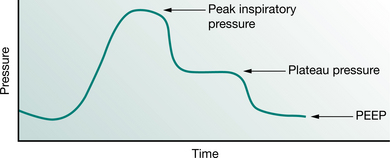
Figure 8.8 The waveform of a typical volume-controlled breath, illustrating a reliable plateau pressure that allows measurement of dynamic lung/thorax compliance (= TV / [PIP − PEEP]) and static lung/thorax compliance (= TV / [Plateau pressure − PEEP]).
The main cause of high peak airway pressure (in volume-controlled mode) or low tidal volume (in pressure-controlled mode) in an intubated patient is often related to problems with airways resistance: for example, a small endotracheal tube (ETT), high flow rates, secretions, bronchospasm. In patients with volume- controlled or dual mode ventilation, if both the peak and plateau pressures are high relative to the tidal volume delivered (5–6 ml/kg), this may indicate diffuse parenchymal disease such as ALI/ARDS or pneumonia.
A more accurate measure of static lung/thorax compliance requires the delivery of a volume-controlled breath with a known plateau/pause pressure using the formula:
Tidal volume/(Plateau pressure − PEEP)
An accurate plateau pressure exists only if a long enough inspiratory pause (zero flow) of 1–3 seconds is incorporated in the ventilatory pattern (Barberis et al 2003). Patient respiratory effort should be minimal during this measurement, and the patient usually needs to be heavily sedated.
Abdominal distension can severely impair diaphragmatic excursion and reduce FRC and may also lead to reduced lung/thorax compliance, which would increase the work of breathing.
In order to optimize mechanical ventilation, it is important to ensure patient–ventilator synchrony (ability of the mechanical ventilator to promptly respond to patient demand for flow during inspiration and to allow an unimpeded expiration), which may reduce the requirements for sedation.
Waveform analysis – assessment of patient–ventilator synchrony
Considering that most mechanical ventilators now display real-time ventilator waveforms such as pressure, flow and volume across time or as loops (pressure/volume, flow/volume), it is imperative that physiotherapists familiarize themselves with basic waveform analysis and the expected changes with therapy or alterations to mechanical ventilatory support.
Specific patient interventions, such as patient re-positioning and secretion movement, may adversely affect ventilation (reduced tidal volume, minute volume), particularly in pressure-controlled modes such as PCV or BiPAP. The early detection of untoward changes in waveforms allows the clinician to optimize the ventilator settings (by altering the PEEP or pressure support settings), modify treatment (increase the head-up tilt) and provide intervention (airway suctioning) to minimize any disruptions to the ventilation delivered.
Bedside waveform analysis (Tables 8.2 and 8.3) can be used to determine the presence of specific clinical problems such as excessive patient trigger, inadequate inspiratory flow, asynchrony with cycling from inspiration to expiration, bronchospasm/gas trapping and airway secretions.
Table 8.2 Pressure waveforms of common clinical problems, causes and clinical signs (volume-controlled)
| Abnormal ventilator waveform appearance | Description and potential causes |
|---|---|
| A Pressure curve (triggering) Excessive trigger 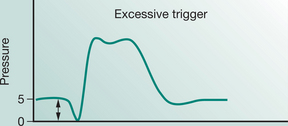 |
Pressure curve At the start of pressure curve there is a negative deflection greater than 1–2 cmH2O, followed by positive pressure breath delivery |
| B Pressure curve (breath attempts) Missed breath attempts 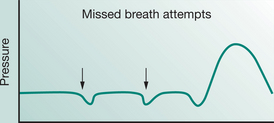 |
Pressure curve Negative deflection(s) in the pressure curve (arrows), below PEEP level without inspiratory flow or positive pressure being delivered |
| C Pressure curve (flow rate) Inadequate flow rate 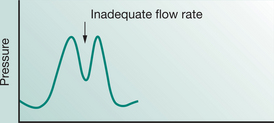 |
Pressure curve Negative inflection in the pressure curve during inspiratory phase (arrow), due to patient inspiratory effort exceeding set ventilator flow rate, often coined ‘bunny ears’ in appearance with no change in inspiratory flow rate |
| D Pressure curve (inspiratory time) Prolonged inspiration 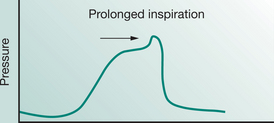 |
Pressure curve A positive inflection at the end of inspiration on the pressure/time curve (arrow), indicating the patient is attempting to exhale while the ventilator is still delivering inspiratory flow. |
Table 8.3 Flow waveforms of common clinical problems, causes and clinical signs (volume-controlled)
| Abnormal ventilator waveform appearance | Description and potential causes |
|---|---|
| E Flow curve (expiratory flow) Gas trapping 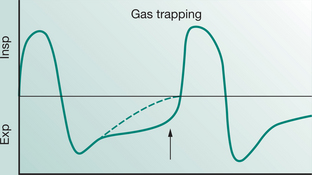 |
Expiratory flow curve (end curve) Expiratory gas trapping (arrow indicates that expiratory flow is still occurring before next positive pressure breath is delivered). Normal exhalation May result in reduced blood pressure due to raised intrathoracic pressure (Note: some intensivists disconnect the patient from mechanical ventilator at end of expiration to assess a rebound increase in blood pressure) |
| F Flow curve (expiratory flow) Missed breath attempts 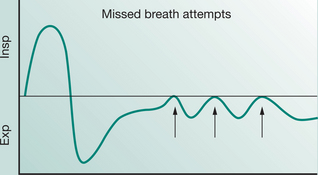 |
Expiratory flow curve Missed breath attempts (arrows) – patient attempts to trigger inspiratory flow |
| G Flow curve (expiratory flow) Saw-tooth pattern 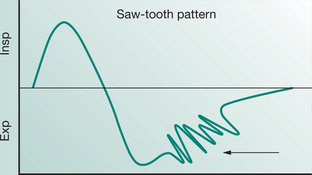 |
Expiratory flow curve Saw-tooth pattern (arrow) on expiration |
The following section will discuss the commonly encountered clinical problems in the intubated and mechanically ventilated patient, and how these problems can be detected through bedside waveform analysis. For this section it will be assumed that the patient is able to breathe in an assisted mode of ventilation (e.g. SIMV with pressure support). The normal pressure waveform is shown in Figure 8.1.
The three variables that determine how effectively a mechanical ventilator delivers a breath are:
 the transition from inspiration to expiration phase – the ‘cycling off’ variable (Georgopoulos et al 2006).
the transition from inspiration to expiration phase – the ‘cycling off’ variable (Georgopoulos et al 2006).Troubleshooting for ventilator triggering.
The clinician should suspect a problem with triggering in the patient who displays excessive accessory muscle use, especially at the start of inspiration. Analysis of the start of the breath delivery on the pressure/time waveforms will detect the amount of negative pressure deflection (Table 8.2 Curve A). If the trigger effort required from the patient is excessive, then ‘missed breath’ attempts may be the result (Table 8.2 Curve B), Case study 8.5.
CASE STUDY 8.5
Troubleshooting for ventilator triggering (Curve A)
A 60-year-old male patient following upper abdominal surgery, orally intubated and mechanically ventilated in SIMV volume-controlled ventilation, set respiratory rate 10 bpm, flow rate 50 lpm, TV 500 ml, PEEP 5 and FiO2 0.4. The nursing staff reported that the patient was restless and agitated overnight, and appeared to be intolerant of the endotracheal tube; regular boluses of intravenous sedation were required throughout the evening. The physiotherapist examined the patient and noticed increased accessory muscle use at the start of inspiration (Table 8.2 Curve A). The pressure trigger sensitivity was noted to be at −4 cmH2O and on inspection, the pressure/time waveform was as illustrated in Curve A. The large negative deflection suggested that a relatively high level of work of breathing was required to initiate inspiratory flow. The physiotherapist then reduced the trigger ‘sensitivity’ from −4 to −1 cmH2O, which resulted in reduced accessory muscle use during inspiration and a reduced negative deflection on the pressure/time curve.
Analysis
This case illustrates that the trigger sensitivity was set too high. Reducing the trigger sensitivity reduced the demand on the work of breathing from the patient and resulted in a lower negative pressure deflection (as shown under Curve A). If these adjustments had not improved the situation, other clinical causes of the increased work of breathing such as intrinsic/auto PEEP should be investigated. Other means of achieving this could be through the use of increased sedation (after discussion with the medical team and dependent on local intensive care unit policy).
Troubleshooting inadequate inspiratory flow delivery (Curve C)
The patient is now being treated by the physiotherapist and has been positioned with the head of the bed flat, in preparation for re-positioning into right side-lying for treatment of his left lower lobe collapse. The physiotherapist notices the patient becoming agitated during repositioning, with increased accessory muscle use and paradoxical chest wall movements. There are also further changes in the pressure/time waveforms (Table 8.2 Curve C). Following a bolus of sedation, the pressure/time curve improved with reduced negative deflection during the inspiratory breath.
Analysis
In patients receiving set volume-controlled modes (SIMV, assist control) with fixed flow rate delivery and with spontaneous breathing effort, the work of breathing will increase if the demand for flow exceeds the set ventilator flow. This may occur during nursing care, exercise or physiotherapy treatments. To reduce the patient inspiratory effort/drive and allow the patient to remain in the same mode of ventilation, the set inspiratory flow rate and sedation could be increased. An alternative way to reduce the demand on the patient’s inspiratory effort would be to change to a pressure-controlled mode (variable flow delivery) or dual mode of ventilation (set tidal volume but with variable flow delivery). These decisions, however, should be multidisciplinary, and dependent on the local intensive care unit policy.
Troubleshooting – cycling from inspiration to expiration.
The factors that determine the ventilator cycling from inspiration to expiration normally depend on the mode of ventilation (pressure, volume or dual mode). The cycling parameters are either:
 percentage reduction of peak inspiratory flow rate (e.g. when inspiratory flow is reduced to 25% of peak value) in pressure support mode of ventilation.
percentage reduction of peak inspiratory flow rate (e.g. when inspiratory flow is reduced to 25% of peak value) in pressure support mode of ventilation.If a patient attempts to exhale while the ventilator is still delivering inspiratory flow, the contraction of the muscles of expiration will lead to an increase in the work of breathing. Bedside detection of the pressure/time waveforms may demonstrate a positive pressure deflection (pressure/time waveform) at the end of the inspiratory cycle (Table 8.2 Curve D). Reduction of the set inspiratory time, tidal volume or inspiratory pressure should facilitate transition to expiration. However, care should be taken as a reduction in set inspiratory time or inspiratory pressure may lead to significant reductions in tidal volume, which must be monitored.
Troubleshooting – gas trapping/intrinsic–PEEP.
The full relaxed respiratory cycle must include enough time for complete exhalation to occur, with zero flow at the end of expiration before the next inspiration is delivered. However, in the mechanically ventilated patient, there is often inadequate time for complete exhalation (Blanch et al 2005); hence ‘gas trapping’ tends to occur (Table 8.3 Curve E).
Gas trapping results in retained gas in the thorax above FRC (causing intrinsic PEEP) which may lead to several cardiac and pulmonary effects:
 The increase in intrathoracic pressure may lead to reduced cardiac filling pressures and compromise blood pressure and cardiac output.
The increase in intrathoracic pressure may lead to reduced cardiac filling pressures and compromise blood pressure and cardiac output. An increase in intrinsic PEEP may increase spontaneous work of breathing, with increased use of expiratory muscles in an attempt to facilitate expiration, and increased use of inspiratory muscles at the start of inspiration due to the generation of increased intrinsic PEEP.
An increase in intrinsic PEEP may increase spontaneous work of breathing, with increased use of expiratory muscles in an attempt to facilitate expiration, and increased use of inspiratory muscles at the start of inspiration due to the generation of increased intrinsic PEEP.Examination of the expiratory flow waveforms will allow the clinician to determine whether there is near full exhalation before the next inspiration (zero flow, at baseline). A sound understanding of the patient’s disease pathology, for example asthma or COPD, will assist the clinician in determining the optimal way of managing gas trapping. For example, if gas trapping is occurring due to inadequate expiratory time, then dependent on the patient status (e.g. normal arterial pH) the set respiratory rate or set tidal volume may be reduced to allow adequate time for full exhalation. On the other hand, gas trapping due to airway narrowing during expiration in patients with asthma, COPD or ARDS may be reduced or avoided by ensuring adequate bronchodilator therapy (again, these decisions should be multidisciplinary and dependent on the local intensive care unit policy).
Gas trapping due to intrinsic PEEP may lead to ventilator asynchrony (Dhand 2005). Patients with intrinsic PEEP who are triggering breaths have to generate a large negative intrapleural pressure that is at least equal to the level of intrinsic PEEP plus the trigger sensitivity level of the ventilator, before a breath can be triggered and delivered. Thus if the PEEP is set at 5 cmH2O, and intrinsic PEEP is 5 cmH2O, the total PEEP is in fact 10 cmH2O; i.e. although the set inspiratory trigger is −1 cmH2O (below PEEP), the patient has to generate a negative intrapleural pressure of at least 6 cmH2O to trigger a breath. This may result in ineffective or wasted patient effort to trigger a ventilator breath, resulting in patient–ventilator asynchrony.
The expiratory flow waveforms should also be checked to ensure complete exhalation before the next breath delivery (Table 8.3 Curve E). Adequate exhalation before the next inspiration should occur, not only during mandatory breath delivery but also during spontaneous ventilation modes such as CPAP and pressure support. Incomplete exhalation (to zero flow or baseline) puts the patient at risk of attempting to trigger inspiratory gas flow before expiration is complete (Table 8.3 Curves E and F). This may also potentially lead to ineffective triggering efforts and patient–ventilator asynchrony (Table 8.3 Curve F), with missed breath attempts.
Measurement of intrinsic PEEP
The measurement of intrinsic PEEP determines the effect of passive elastic recoil on expiratory flow and hence the patient should be under a mandatory ventilator mode and adequately sedated to minimize any respiratory muscle effort. Most modern mechanical ventilators have the facility to measure intrinsic PEEP either through a manual ‘expiratory-hold’ or an intrinsic-PEEP diagnostic function. Ventilator waveforms (pressure and flow) must be monitored during the expiratory hold manoeuvre (Fig. 8.9) to ensure the patient is not actively breathing during the pause (normally 2–5 seconds).
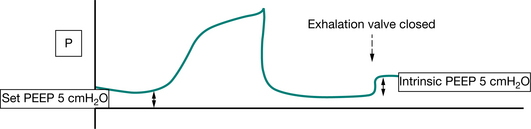
Figure 8.9 Auto-PEEP manoeuvre. The procedure entails closure of the exhalation valve at end exhalation (for up to 3 seconds) to measure the level of intrinsic PEEP (often an automated function in most modern ventilators). The patient should be in a mandatory mode of mechanical ventilation, heavily sedated and/or paralysed to prevent any active respiratory effort. P, pressure; PEEP, positive end-expiratory pressure.
Airway secretions.
Airway secretions in the mechanically ventilated patient may be difficult to detect through conventional means (auscultation, chest palpation), for the following reasons:
 inadequate inspiratory and expiratory flow rates to create turbulent flow (e.g. due to low levels of CPAP and pressure support).
inadequate inspiratory and expiratory flow rates to create turbulent flow (e.g. due to low levels of CPAP and pressure support).Ventilator flow waveform analysis can assist in the detection of airway secretions. The presence of airway secretions or condensate in the ventilator tubing could induce a ‘saw-tooth’ pattern or jagged waveform during the expiratory flow (Table 8.3 Curve G and Fig. 8.10). The ‘saw-tooth’ pattern should disappear with airway suctioning and clearance of secretions or removal of the condensate from the ventilator circuit.
Patient positioning and work of breathing
Supine patient positioning in intensive care is often required when patients are cardiovascularly unstable, have a suspected spinal injury and/or when supported by devices such as an intra-aortic balloon pump. The supine patient position combined with intubation and mechanical ventilation may lead to increased  /
/ mismatch resulting in hypoxaemia. Upright positioning can be used to improve gas exchange and end-expiratory lung volumes even in patients with severe hypoxaemia due to ALI/ARDS (Richard et al 2006), reducing the risk of development of VAP (Drakulovic et al 1999). Head-down positioning, however, is often utilized by physiotherapists for secretion clearance or by medical staff for procedures such as insertion of central venous catheters. Head-down posture may cause reductions in tidal volume and FRC and may be associated with increases in total respiratory resistance and reductions in lung/thorax compliance (Fahy et al 1996). Hence, with the patients on low levels of ventilator support (e.g. 5 cmH2O CPAP and 5 cmH2O pressure support), the recumbent position (supine or head down) may result in increased patient work of breathing. The increase in work of breathing may also depend on factors such as the level of sedation, diameter of endotracheal tube, muscle strength, lung/thorax compliance (abdominal distension, pulmonary oedema) and airway resistance (airway secretions, bronchospasm).
mismatch resulting in hypoxaemia. Upright positioning can be used to improve gas exchange and end-expiratory lung volumes even in patients with severe hypoxaemia due to ALI/ARDS (Richard et al 2006), reducing the risk of development of VAP (Drakulovic et al 1999). Head-down positioning, however, is often utilized by physiotherapists for secretion clearance or by medical staff for procedures such as insertion of central venous catheters. Head-down posture may cause reductions in tidal volume and FRC and may be associated with increases in total respiratory resistance and reductions in lung/thorax compliance (Fahy et al 1996). Hence, with the patients on low levels of ventilator support (e.g. 5 cmH2O CPAP and 5 cmH2O pressure support), the recumbent position (supine or head down) may result in increased patient work of breathing. The increase in work of breathing may also depend on factors such as the level of sedation, diameter of endotracheal tube, muscle strength, lung/thorax compliance (abdominal distension, pulmonary oedema) and airway resistance (airway secretions, bronchospasm).
Appropriately performed physiotherapy is well tolerated from a haemodynamic and metabolic perspective (Berney & Denehy 2003). However, any adverse changes in haemodynamics (blood pressure, heart and pulmonary artery pressure) can be attenuated through increased sedation (Harding et al 1993) or increased pressure support (Harding et al 1995). The increases in metabolic consumption may only be attenuated with neuromuscular blocking agents (Horiuchi et al 1997). But clinicians should be aware that sedation boluses in a pressure-controlled mode of ventilation (e.g. PCV or CPAP and pressure support) may significantly reduce the patient’s spontaneous respiratory effort (work of breathing), which may lead to a reduction in tidal volume/minute volume, and additional inspiratory support may be required.
Lung/thoracoabdominal compliance and airway resistance
Reduced lung/thoracoabdominal compliance or increased airway resistance are common causes of increased work of breathing. The physiotherapist’s assessment of the patient should identify the problem in this clinical scenario. During repositioning to head down or flat for secretion clearance, the functional residual capacity may be reduced, secretions mobilized and lung/thorax compliance (dynamic) further reduced. This may lead either to reductions in tidal volume delivered (pressure-controlled modes) or increased peak airway pressures (volume- or dual-controlled modes) and may increase the patient’s work of breathing. The mode of ventilation (volume-controlled vs pressure-controlled) may impact on the physiological effects of physiotherapy treatment.
Case study 8.6 examines some issues on ventilation and positioning.
CASE STUDY 8.6
A 70-year-old female patient following abdominal surgery, complicated by sepsis and ALI due to an infected peritoneum. This patient was ventilated in BiPAP mode, set rate 20 bpm, Phigh 30 cmH2O, Plow 10 cmH2O, FiO2 0.8, with exhaled tidal volume of 400 ml. The morning chest radiograph demonstrated bilateral diffuse pulmonary infiltrates (alveolar shadowing). The physiotherapist examined the patient and reported bilateral crackles on auscultation with palpable fremitus throughout the left hemi-thorax. The physiotherapist repositioned the patient into right sidelying with the head of the bed flat. The tidal volume reduced from 400 ml to 250 ml, with the respiratory rate unchanged.
What are the implications of the treatment decisions of this physiotherapist?
Analysis
This scenario may commonly occur during physiotherapy treatment and may often be undetected. The reductions in tidal volume may relate to the reduced FRC as a result of altered head-down posturing (from head up to head flat) in combination with a pressure-controlled mode of ventilation. In addition, as the patient had palpable fremitus unilaterally, this is indicative of airway secretions, and repositioning into sidelying may have caused the movement of secretions to the more central airways or caused direct aspiration of secretions into the dependent lung. The set inspiratory pressure in a pressure-controlled mode (such as bilevel ventilation) does not ensure constant tidal volume with changing airways resistance and lung/thorax compliance. The clinician at the bedside must pre-empt and monitor these changes by understanding the disease process, the mode of mechanical ventilation and potential impact of therapy interventions. Inspection of the flow waveforms (reduced inspiratory flow rate and reduced area under the curve) and exhaled tidal volumes (reduced exhaled tidal volume) should alert the clinician to the changing patient status and early recognition/intervention should prevent any untoward changes in gas exchange. Before repositioning the patient head-down, the therapist should have suctioned the airway to clear secretions from central airways and assess cough response. Reassessment of exhaled lung volumes may have demonstrated an increase in tidal volumes with this intervention alone and determined whether there really was a need for repositioning.
The assessment of flow waveforms (saw-tooth) pattern on expiratory flow waveforms, Table 8.3 Curve G) may also have indicated the presence of upper airway secretions, which may have resolved with suctioning alone.
Reduced lung/thorax compliance may be improved with the upright sitting position (Behrakis et al 1983). This, however, does not mean that patients cannot and should not be turned for pressure area care or physiotherapy for secretion clearance. The therapist should, however, be aware of the potent effects of patient repositioning and that changes in mechanical ventilation may be required to minimize any adverse effects (e.g. increased PEEP, FiO2, increased tidal volume or increased inspiratory pressure dependent on the mode of ventilation).
Respiratory muscle strength and weaning
The critically ill patient is predisposed to develop muscle dysfunction/wasting due mainly to inactivity and sepsis, with the consequence of poor activity tolerance, reduced strength and prolonged time on mechanical ventilation in intensive care (Winkelman 2004). The evidence supporting inspiratory muscle training to date has focused on the long-term, difficult to wean patient (Aldrich et al 1989, Martin et al 2002). However, inspiratory muscle training (through reduced trigger sensitivity) in acute critically ill patients from the beginning of mechanical ventilation may not improve respiratory muscle strength or shorten weaning time (Caruso et al 2005).
Interventions aimed at enhancing respiratory muscle strength include:
 partially resting the patient overnight (Vassilakopoulos et al 2006) with increased respiratory support (pressure support)
partially resting the patient overnight (Vassilakopoulos et al 2006) with increased respiratory support (pressure support)  general exercise training including activities of daily living such as sitting over the edge of the bed, standing and ambulation (Chiang et al 2006, Zafiropoulos et al 2004).
general exercise training including activities of daily living such as sitting over the edge of the bed, standing and ambulation (Chiang et al 2006, Zafiropoulos et al 2004).The rehabilitation of the critically ill patient is attracting more and more interest in the literature as a means of minimizing or preventing muscle dysfunction. Activity for the critically ill may be considered along a spectrum of care from passive repositioning into upright or sidelying, to passive range of motion exercises, active assisted exercises, self-care, sitting over the edge of the bed, standing, sitting out of bed and ambulation. The metabolic cost of the activity may therefore be expected to increase as the level of muscle use and participation in activity increases (Weissman & Kemper 1991). However, little is known about the typical level of activity among intensive care unit patients (Winkelman 2004) and there is limited information about the place of activity in preventing complications or improving outcome in ICU (Stiller 2000). The only consistent activity performed by nursing staff may be patient turning for pressure care (Winkelman 2004) with minimal other activities undertaken. Mobilization of an intubated and ventilated patient out of bed (Zafiropoulos et al 2004) can be a useful means of effecting short-term improvements in tidal volume and minute ventilation. Daily rehabilitation/mobilization can significantly improve peripheral and respiratory muscle strength, activities of daily living and reduce time on mechanical ventilation (Chiang et al 2006). Increased pressure support during exercise/physiotherapy may be useful to minimize haemodynamic stress responses.
Evaluation of respiratory muscle strength
The evaluation of respiratory muscle strength in the intubated and mechanically ventilated patient is often complicated by factors such as sedation and lack of cooperation (Man et al 2004).
A basic respiratory strength assessment can be undertaken by the use of a pressure manometer in the circuit or by a diagnostic function on most modern mechanical ventilators. The assessment of maximal inspiratory pressure (MIP), negative pressure generated in the first 100 milliseconds of inspiration (P0.1) and forced vital capacity (FVC) manoeuvres are some of the most commonly reported procedures. However, patient cooperation and understanding of the assessment procedures (MIP and FVC) are required and this is not always feasible due to sedation required for the tolerance of the tracheal tube. With the development of magnetic stimulation, it has become possible to non-volitionally assess respiratory muscles in a clinical setting (Man et al 2004). There have not been any reported links between measures of respiratory muscle strength and extubation or weaning outcomes.
Weaning from mechanical ventilation
Although mechanical ventilation may be lifesaving, it is associated with numerous complications such as ventilator-associated pneumonia, cardiovascular compromise, barotrauma and ventilator-induced lung injury (Epstein 2002). Mechanical ventilation may be associated with diaphragmatic dysfunction (Jubran 2006) and reduced inspiratory muscle endurance (Chang et al 2005). Once clinical improvement has occurred, emphasis is placed on weaning or liberating the patient from mechanical ventilation (Epstein 2002). However, the value of weaning the patient from the ventilator as soon as possible must be balanced against the risks of premature withdrawal, which may be associated with re-intubation, in turn associated with increased mortality (MacIntyre 2004). The McMaster Evidence Based Practice Centre (Cook et al 2000) provides a more detailed overview of weaning from mechanical ventilation. The imbalance between increased respiratory workload, decreased respiratory muscle strength and endurance may be important factors associated with ventilator dependence (Caruso et al 2005). Shock on admission, increased APACHE II score
[= (acute physiology score) + (age points) + (chronic health points)]
ARDS and multiple organ dysfunction are variables significantly associated with prolonged time (>21 days) on mechanical ventilation. These patients also suffer from a high rate of failed extubations, unsuccessful weaning, malnutrition and infection (Estenssoro et al 2006).
Key issues in the management of the ventilated patient are outlined in Box 8.6. Suggested guidelines for extubation are given in Box 8.7.
Box 8.6 Key issues that must be addressed in the overall management of the mechanically ventilated patient
When it has been determined that the disease process or processes have begun to stabilize or reverse, clinicians should:
Box 8.7 Suggested guidelines for extubation
A simple screening procedure at the bedside to identify a patient’s readiness for weaning:
The patient may then undergo a short spontaneous breathing trial of CPAP alone or breathing via a T-piece for 30–90 minutes, with the aim to then extubate the patient if he tolerates this trial (Tonnelier et al 2005).
Weaning strategies
When the patient’s medical condition has been stabilized, weaning off mechanical ventilation is often started. Various approaches to weaning have been adopted by centres around the world (Esteban et al 2000). Bedside measurements of respiratory function (Chapter 3) are often used to assess the suitability of the patient for weaning from mechanical ventilation to extubation, but the predictive success of these measures for the general intensive care population is poor (Conti et al 2004). Recent work by Kuo et al (2006) demonstrated improved predictive accuracy of the measurement of the rapid shallow breathing index at the end of a 2-hour period of spontaneous breathing compared with the traditional method when the measurement is taken at the beginning of the trial. The optimal means to assess appropriateness for weaning and extubation is still evolving.
The requirements for continuing ventilatory support are determined by the balance between the ventilatory capability of the patient and his ventilatory demands (e.g. atelectasis, disease, surgery, sepsis). As the imbalance between ventilatory capability and ventilatory demand begin to resolve (Fig. 8.11), the focus of patient care then concentrates on removing the patient from the ventilator as quickly as possible (MacIntyre 2004). Rehabilitation may play a key role in facilitating weaning from ventilation in long-term patients (Chiang et al 2006).
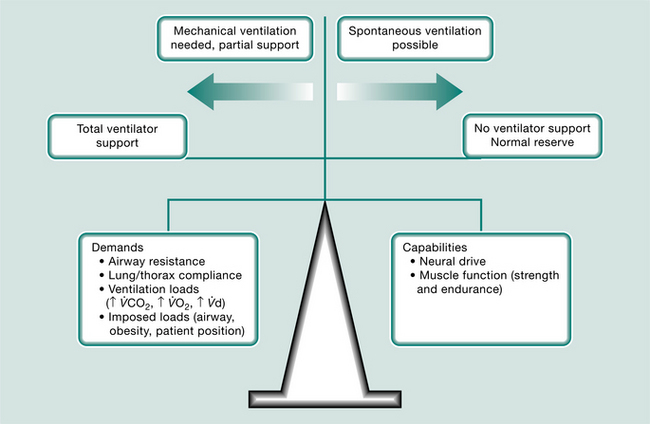
Figure 8.11 The balance between ventilatory loads and demands that determine the need for mechanical ventilator support.
Adapted from MacIntyre 2004
Controversy exists regarding the most appropriate weaning strategy, the indicators used to assess readiness for weaning/extubation and the use of weaning protocols. Early work (Kollef et al 1997) demonstrated a significantly lower weaning time, when weaning was protocol-led by nurse and therapist compared with physician-led. Weaning protocols may have minimal impact in an intensive care environment where there is a high number of qualified nurses, high physician input, good collaboration between the team members and autonomous nursing decision-making in relation to weaning practices (Rose & Nelson 2006). Other weaning strategies include daily T-piece trials, pressure support and extubation to non-invasive ventilation to facilitate the process (Girault et al 1999).
Tracheostomy
A tracheostomy is often performed in an intensive care patient when long-term ventilation (>7–10 days) is anticipated. Tracheostomy is also performed if there is concurrent upper airway obstruction, e.g. tumour, trauma, or if equipment dead space poses an unacceptable hindrance to weaning. A tracheostomy may be instituted either percutaneously or surgically, depending on the urgency and physical features of the patient, such as body mass index, condition and head/neck anatomy. Percutaneous tracheostomy is performed at the bedside and uses a Seldinger dilation technique, which usually leaves only a small residual scar. Surgical tracheostomy requires patient transfer to the operating room, a surgical incision and dissection through to the trachea. In some cases, removal of the cricoid cartilage is necessary but the cosmetic result is less acceptable (Friedman 2006). Tracheostomy assists weaning from mechanical ventilation by reducing dead space, decreasing airway resistance, improving secretion clearance and decreasing the need for sedation (Pierson 2005).
Physiotherapists are usually required to optimize lung function in tracheostomized patients and therefore it is important that they understand the functional characteristics of the tracheostomy tube design and the implications for patient care. Tracheostomy tubes are available in different sizes and styles (Hess 2005). They can be of different diameters and lengths, angled or curved, cuffed or uncuffed, with single or dual tubes (dual-cannula tracheostomy tubes). Fenestrated tracheostomy tubes and tubes which allow subglottic suction are also available (Portex Blue Line Ultra Suctionaid). These latter have a suction port situated above the cuff.
Dimensions of a tracheostomy tube refer to the internal and external diameter, length and curvature of the tube. A patient with a large neck may require a tracheostomy tube with extra proximal length. Too small an internal diameter will increase the resistance to airflow through the tube.
With the cuff deflated, a standard tracheostomy tube allows use of the upper airway and speech (but protection from aspiration is sacrificed). Efficiency of upper airway engagement will be decreased if the outer diameter of the tube is too large.
Dual-cannula tracheostomy tubes have an inner cannula that is disposable or reusable. The use of an inner cannula facilitates regular changing or cleaning and is believed to reduce biofilm formation and the incidence of ventilator-associated pneumonia (Burns et al 1998).
A fenestrated tracheostomy tube is similar to a standard tracheostomy tube except that there is an opening (window) in the posterior portion of the tube above the cuff and the tube is provided with an inner cannula and a plastic plug (decannulation cap). With the cuff deflated, removal of the inner cannula allows the use of the upper airway and permits air to pass through the vocal cords, which facilitates phonation, as does digital occlusion of the tube. A speaking valve can also be placed in the tube for speech training. The decannulation cap allows the patient to breathe through the fenestrations and around the tube and is used to facilitate weaning of the patient from the tracheostomy tube (decannulation). The inner cannula may or may not have a window in situ. Insertion of a non-fenestrated inner cannula allows the tubes to function as an ordinary tracheostomy tube and to be used for suctioning. It should be noted that suction should never be performed with a fenestrated inner cannula in situ, as this will cause damage to the trachea. Fenestrated tracheostomy tubes have also been shown to reduce the work of breathing (Hussey & Bishop 1996). Fenestrated tracheostomy tubes are often used during extended weaning of patients from long-term mechanical ventilation.
Suggested guidelines for decannulation
Pre-weaning criteria have been specified (Heffner 2005, Ladyshewsky & Gousseau 1996) and are summarized in Table 8.4.
Table 8.4 Guidelines for weaning from a tracheostomy
| Indications for weaning | Contraindications to weaning |
|---|---|
While the management of each patient should be individualized, it has been shown that a multidisciplinary approach with a standardized decisional protocol can result in increased success in weaning (Ceriana et al 2003, Ladyshewsky & Gousseau 1996). The usual progression (although this can vary from centre to centre) of weaning a tracheostomy is:
During each stage, there should be close monitoring of respiratory rate, respiratory muscle work, oxygen saturation and cardiovascular parameters. Presence of wheeze, stridor, drooling and respiratory distress indicate that the patient should not progress to the next stage. A mini-tracheostomy (cuffless tracheal tube) may be appropriate following decannulation, if the patient requires assistance with secretion clearance.
Criteria employed to assess each aspect and stage of weaning are controversial. The ‘blue dye’ test used to assess swallowing is cited in the major international guidelines (Heffner & Hess 2001) but has been shown to have low sensitivity and may give a false-negative result, especially when compared with video-fluoroscopy (Ceriana et al 2003). Actual cough strength and ability to clear secretions can be subjective; therefore maximal expiratory pressure (MEP) and/or peak flow measurements have been used to predict the likelihood of being weaned from tracheotomy. During mechanical ventilation, in a stable patient, cuff deflation can be used to facilitate verbal communication (termed ‘leak speech’ without the use of a speaking valve).
The physiotherapist’s role in weaning from mechanical ventilation
Non-physician and nurse-led weaning may significantly improve weaning outcomes, but the impact may depend on the medical staffing levels (Krishnan et al 2004). The physiotherapist’s role in weaning could be directed towards:
 early assessment of patient rehabilitation poten-tial (strength, endurance, bed mobility, transfer training)
early assessment of patient rehabilitation poten-tial (strength, endurance, bed mobility, transfer training) identify readiness for extubation (e.g. minimal secretions, effective cough, airway reflexes present, neurological status)
identify readiness for extubation (e.g. minimal secretions, effective cough, airway reflexes present, neurological status) assisting with tracheostomy weaning (e.g. periods of spontaneous breathing interspersed with periods of respiratory muscle rest on mechanical ventilation)
assisting with tracheostomy weaning (e.g. periods of spontaneous breathing interspersed with periods of respiratory muscle rest on mechanical ventilation)CONCLUSION
The extended role of the physiotherapist in intensive care is currently topical (McPherson et al 2006). While the role and responsibilities of the physiotherapist vary from country to country and even hospital to hospital in the same city, over recent years physiotherapists have successfully gained greater autonomy and their role within the ICU in weaning, extubation, ventilator and tracheostomy management, fibre optic bronchoscopy, ICU outreach, post ICU clinics, and bedside thoracic ultrasound have all received varying amounts of endorsement at different centres.
This chapter has described various means of monitoring and supporting the major organ systems of the body and the implications for physiotherapy intervention, and adopted a problem-based discussion of various physiotherapeutic interventions in the intensive care unit. The inclusion of waveform analysis associated with mechanical ventilation, concepts of weaning from the ventilator and the role of the physiotherapist in optimization of work of breathing, all aim to encourage a greater awareness of the extended scope of their role in intensive care. Tables 8.5 and 8.6 provide a quick reference table for ventilator troubleshooting and management.
Table 8.5 Troubleshooting – management of patients under mechanical ventilation
| Events | Possible causes | Possible action |
|---|---|---|
| High-pressure alarm signals (volume- or dual-controlled modes) | Patient is restless and/with asynchronous breathing | |
| Low-pressure alarm signals | ||
| Malfunction of the ventilator | • Check function of ventilator | |
| Slight drop (1 to 2 cmH2O) in airway pressure in volume-controlled mode is normal after physiotherapy (no alarm signal) | The patient’s lung compliance and/or airway resistance has improved (e.g. after secretion clearance) | • No action required |
| Hypoxaemia | Incorrect settings | |
| Circuit/airway disconnection | ||
| Secretions | ||
| Malposition of the tracheal tube (e.g. down right main bronchus) | ||
| Pneumothorax/pleural effusion | ||
| Onset of new medical problem (e.g. sputum plugging, atelectasis, pulmonary oedema) | ||
| Medications (vasodilators) | • Increase FiO2 |
CPAP, continuous positive airway pressure; ETT, endotracheal tube; FiO2, fractional inspired oxygen concentration; PEEP, positive endexpiratory pressure
| If | Action |
|---|---|
| Cardiopulmonary arrest | Stop intervention and follow basic life support or advanced life support protocols |
| Sudden drop in blood pressure (possibly associated with manual hyperinflation or patient repositioning) | |
| Accidental removal of chest drain | |
| Accidental removal or dislodging of central venous line or pulmonary artery catheter | |
| Sudden onset of cardiac arrhythmias | |
| Sudden desaturation | |
| Sudden rise in intracranial pressure | |
| Blocked tracheal tube | |
| Dislodged tracheal tube (signs of loss of tidal volume or airway pressure, with evident cuff leak) | |
| Sudden onset of ‘absent breath sound’ over one lung | |
| Self-extubation | |
| Sudden reduction in level of consciousness |
CPAP, continuous positive airway pressure; FiO2, fractional inspired oxygen concentration; PEEP, positive end-expiratory pressure; ETT, endotracheal tube; SpO2, oxygen saturation; K+, potassium; Mg2+, magnesium; CO2, carbon dioxide; ICP, intracranial pressure
In addition to clinical research, it is essential that documentary evidence of the cost-effectiveness of physiotherapy in intensive care is collected, to provide outcomes and to direct clinical practice in the future. Opportunities include engagement in collaborative approaches and large observational databases.
If the following happens, alert help, and the recommended actions for consideration are:
Ackerman MH, Mick DJ. Instillation of normal saline before suctioning in patients with pulmonary infections: a prospective randomized controlled trial. American Journal of Critical Care. 1998;7(4):261-266.
Agvald-Ohman C, Wernerman J, Nord CE, Edlund C. Anaerobic bacteria commonly colonize the lower airways of intubated ICU patients. Clinical Microbiology and Infection. 2003;9(5):397-405.
Aldrich TK, Karpel JP, Uhrlass RM, et al. Weaning from mechanical ventilation: adjunctive use of inspiratory muscle resistive training. Critical Care Medicine. 1989;17(2):143-147.
American Association for Respiratory Care (AARC). Clinical practice guidelines. Capnography/capnometry during mechanical ventilation. Respiratory Care. 1995;40(12):1321-1324.
Antonaglia V, Pascotto S, Simoni LD, Zin WA. Effects of a sigh on the respiratory mechanical properties in ALI patients. J Clin Monit Comput. 2006;20(4):243-249.
Arroliga A, Frutos-Vivar F, Hall J, et al. Use of sedatives and neuromuscular blockers in a cohort of patients receiving mechanical ventilation. Chest. 2005;128(2):496-506.
Artucio H, Pereira M. Cardiac arrhythmias in critically ill patients: epidemiologic study. Critical Care Medicine. 1990;18(12):1383-1388.
Bach JR. Mechanical insufflation-exsufflation. Comparison of peak expiratory flows with manually assisted and unassisted coughing techniques. Chest. 1993;104(5):1553-1562.
Bach JR, Saporito LR. Criteria for extubation and tracheostomy tube removal for patients with ventilatory failure. A different approach to weaning. Chest. 1996;110(6):1566-1571.
Barbas CS, de Matos GF, Pincelli MP, et al. Mechanical ventilation in acute respiratory failure: recruitment and high positive end-expiratory pressure are necessary. Current Opinion in Critical Care. 2005;11(1):18-28.
Barberis L, Manno E, Guerin C. Effect of end-inspiratory pause duration on plateau pressure in mechanically ventilated patients. Intensive Care Medicine. 2003;29(1):130-134.
Bargues L, Vaylet F, Le Bever H, L’Her P, Carsin H. Respiratory dysfunction in burned patients. Revue des Maladies Respiratoires.. 2005;22(3):449-460.
Baumburger JP, Goodfriend RB. Determination of arterial oxygen tension in man by equilibration through intact skin. Federation Proceedings. 1951;10:10-21.
Behrakis PK, Baydur A, Jaeger MJ, Milic-Emili J. Lung mechanics in sitting and horizontal body positions. Chest. 1983;83(4):643-646.
Bein T, Metz C, Keyl C, Pfeifer M, Taeger K. Effects of extreme lateral posture on hemodynamics and plasma atrial natriuretic peptide levels in critically ill patients. Intensive Care Medicine. 1996;22(7):651-655.
Bellner J, Romner B, Reinstrup P, et al. Transcranial Doppler sonography pulsatility index (PI) reflects intracranial pressure (ICP). Surgical Neurology. 2004;62(1):45-51.
Belott P. Bioimpedance in the pacemaker clinic. AACN Clinical Issues. 1999;10(3):414-418.
Bernard GR, Artigas A, Brigham KL, et al. The American-European Consensus Conference on ARDS. Definitions, mechanisms, relevant outcomes, and clinical trial coordination. American Journal of Respiratory and Critical Care Medicine. 1994;149(3 Pt 1):818-824.
Bernet-Buettiker V, Ugarte MJ, Frey B, et al. Evaluation of a new combined transcutaneous measurement of PCO2/pulse oximetry oxygen saturation ear sensor in newborn patients. Pediatrics. 2005;115(1):e64-68.
Berney S, Denehy L. A comparison of the effects of manual and ventilator hyperinflation on static lung compliance and sputum production in intubated and ventilated intensive care patients. Physiotherapy Research International. 2002;7(2):100-108.
Berney S, Denehy L. The effect of physiotherapy treatment on oxygen consumption and haemodynamics in patients who are critically ill. Australian Journal of Physiotherapy. 2003;49(2):99-105.
Berney S, Denehy L, Pretto J. Head-down tilt and manual hyperinflation enhance sputum clearance in patients who are intubated and ventilated. Australian Journal of Physiotherapy. 2004;50(1):9-14.
Billard V, Gambus PL, Chamoun N, Stanski DR, Shafer SL. A comparison of spectral edge, delta power, and bispectral index as EEG measures of alfentanil, propofol, and midazolam drug effect. Clinical Pharmacology and Therapeutics. 1997;61(1):45-58.
Blanch L, Bernabe F, Lucangelo U. Measurement of air trapping, intrinsic positive end-expiratory pressure, and dynamic hyperinflation in mechanically ventilated patients. Respiratory Care. 2005;50(1):110-123. ; discussion 23-4
Boitano LJ. Management of airway clearance in neuromuscular disease. Respiratory Care. 2006;51(8):913-922.
Boldt J, Menges T, Wollbruck M, Hammermann H, Hempelmann G. Is continuous cardiac output measurement using thermodilution reliable in the critically ill patient? Critical Care Medicine. 1994;22(12):1913-1918.
Boots RJ, George N, Faoagali JL, et al. Double-heater-wire circuits and heat-and-moisture exchangers and the risk of ventilator-associated pneumonia. Critical Care Medicine. 2006;34(3):687-693.
Bowton DL. Nosocomial pneumonia in the ICU - year 2000 and beyond. Chest. 1999;115(3 Suppl):28S-33S.
Brucia JJ, Owen DC, Rudy EB. The effects of lidocaine on intracranial hypertension. Journal of Neuroscience Nursing. 1992;24(4):205-214.
Burns SM, Spilman S, Wilmoth D, et al. Are frequent inner cannula changes necessary? A pilot study. Heart and Lung. 1998;27:58-62.
Cabello B, Mancebo J. Work of breathing. Intensive Care Medicine. 2006;32(9):1311-1314.
Caruso P, Denari SD, Ruiz SA, et al. Inspiratory muscle training is ineffective in mechanically ventilated critically ill patients. Clinics. 2005;60(6):479-484.
Cereda M, Villa F, Colombo E, et al. Closed system endotracheal suctioning maintains lung volume during volume-controlled mechanical ventilation. Intensive Care Medicine. 2001;27(4):648-654.
Ceriana P, Carlucci A, Navalesi P, et al. Weaning from tracheotomy in long-term mechanically ventilated patients: feasibility of a decisional flow chart and clinical outcome. Intensive Care Medicine. 2003;29:845-848.
Chang A, Hodges P, Boots R, Paratz J. Standing with assistance of a tilt table in intensive care: a survey of Australian physiotherapy practice. Australian Journal of Physiotherapy. 2004;50(1):51-54.
Chang A, Hodges P, Paratz J, Boots R. Using a tilt table improves minute ventilation in critically ill patients. Archives of Rehabilitation Medicine. 2004;85(12):1972-1976.
Chang AT, Boots RJ, Brown MG, Paratz J, Hodges PW. Reduced inspiratory muscle endurance following successful weaning from prolonged mechanical ventilation. Chest. 2005;128(2):553-559.
Chastre J. Ventilator-associated pneumonia: what is new? Surgical Infection. 2006;7(Suppl 2):S81-85.
Chatwin M, Ross E, Hart N, et al. Cough augmentation with mechanical insufflation/exsufflation in patients with neuromuscular weakness. European Respiratory Journal. 2003;21(3):502-508.
Chiang L-L, Wang L-Y, Wu C-P, Wu H-D, Wu Y-T. Effects of physical training on functional status in patients with prolonged mechanical ventilation. Physical Therapy. 2006;86:1271-1281.
Choi JS, Jones AY. Effects of manual hyperinflation and suctioning in respiratory mechanics in mechanically ventilated patients with ventilator-associated pneumonia. Australian Journal of Physiotherapy. 2005;51(1):25-30.
Chytra I. Ventilator-associated pneumonia. RT International. 2002;Fall:30-42.
Conti G, Montini L, Pennisi MA, et al. A prospective, blinded evaluation of indexes proposed to predict weaning from mechanical ventilation. Intensive Care Medicine. 2004;30(5):830-836.
Cook D, Meade M, Guyatt G, Griffith L, Booker L, 2000 Criteria for weaning from mechanical ventilation. Evidence Report/Technology Assessment No. 23 (Prepared by McMaster University under Contract No. 290-97-0017). AHRQ Publication No. 01-E010. Rockville MD: Agency for Healthcare Research and Quality
Davis K, Johannigman JA, Campbell RS, et al. The acute effects of body position strategies and respiratory therapy in paralyzed patients with acute lung injury. Critical Care. 2001;5(2):81-87.
Dhand R. Ventilator graphics and respiratory mechanics in the patient with obstructive lung disease. Respiratory Care. 2005;50(2):246-261. ; discussion 59-61
Dodek P, Keenan S, Cook D, et al. Evidence-based clinical practice guideline for the prevention of ventilator-associated pneumonia. Annals of Internal Medicine. 2004;141(4):305-313.
Dowdy DW, Eid MP, Sedrakyan A, et al. Quality of life in adult survivors of critical illness: a systematic review of the literature. Intensive Care Medicine. 2005;31(5):611-620.
Drakulovic MB, Torres A, Bauer TT, et al. Supine body position as a risk factor for nosocomial pneumonia in mechanically ventilated patients: a randomised trial. Lancet. 1999;354(9193):1851-1858.
Dutton RP, McCunn M. Traumatic brain injury. Current Opinion in Critical Care. 2003;9(6):503-509.
Epstein SK. Weaning from mechanical ventilation. Respiratory Care. 2002;47(4):454-466.
Esteban A, Anzueto A, Alia I, et al. How is mechanical ventilation employed in the intensive care unit? An international utilization review. American Journal of Respiratory and Critical Care Medicine. 2000;161(5):1450-1458.
Estenssoro E, Reina R, Canales HS, et al. The distinct clinical profile of chronically critically ill patients: a cohort study. Critical Care. 2006;10(3):R89.
Fabregas N, Gambus PL, Valero R, et al. Can bispectral index monitoring predict recovery of consciousness in patients with severe brain injury? Anesthesiology. 2004;101(1):43-51.
Fahy BG, Barnas GM, Nagle SE, et al. Effects of Trendelenburg and reverse Trendelenburg postures on lung and chest wall mechanics. Journal of Clinical Anesthesia. 1996;8(3):236-244.
Friedman Y. Percutaneous versus surgical tracheostomy: the continuing saga. Critical Care Medicine. 2006;34(8):2250-2325.
Gao F, Melody T, Daniels DF, Giles S, Fox S. The impact of compliance with 6-hour and 24-hour sepsis bundles on hospital mortality in patients with severe sepsis: a prospective observational study. Critical Care. 2005;9(6):R764-770.
Garstang SV, Kirshblum SC, Wood KE. Patient preference for in-exsufflation for secretion management with spinal cord injury. Journal of Spinal Cord Medicine. 2000;23(2):80-85.
Gattinoni L, Pelosi P, Suter PM, et al. Acute respiratory distress syndrome caused by pulmonary and extrapulmonary disease. Different syndromes? American Journal of Respiratory and Critical Care Medicine. 1998;158(1):3-11.
Gattinoni L, Vagginelli F, Carlesso E, Taccone P, Conte V, Chiumello D, Valenza F, Caironi P, Pesenti A, Prone-Supine Study Group. Decrease in PaCO2 with prone position is predictive of improved outcome in acute respiratory distress syndrome. Critical Care Medicine. 2003;31(12):2727-2733.
Georgopoulos D, Prinianakis G, Kondili E. Bedside waveforms interpretation as a tool to identify patient-ventilator asynchronies. Intensive Care Medicine. 2006;32(1):34-47.
Germann P, Braschi A, Della Rocca G, et al. Inhaled nitric oxide therapy in adults: European expert recommendations. Intensive Care Medicine. 2005;31(8):1029-1041.
Girault C, Daudenthun I, Chevron V, et al. Non-invasive ventilation as a systematic extubation and weaning technique in acute-on-chronic respiratory failure: a prospective, randomized controlled study. American Journal of Respiratory and Critical Care Medicine. 1999;160(1):86-92.
Gomersall CD, Oh TE. Haemodynamic monitoring. In: Oh TE, editor. Intensive care manual. 4th edn. Oxford: Butterworth-Heinemann; 1997:831-838.
Gray HH, Dawkins KD, Morgan JM, Simpson IA. Examination of the cardiovascular system. In Lecture notes on cardiology, 4th edn., Denmark: Blackwell Publishing; 2002:12.
Griffiths RD, Palmer TE, Helliwell T, MacLennan P, MacMillan RR. Effect of passive stretching on the wasting of muscle in the critically ill. Nutrition. 1995;11(5):428-432.
Hagler DA, Traver GA. Endotracheal saline and suction catheters: sources of lower airway contamination. American Journal of Critical Care. 1994;3(6):444-447.
Harding J, Kemper M, Weissman C. Alfentanil attenuates the cardiopulmonary response of critically ill patients to an acute increase in oxygen demand induced by chest physiotherapy. Anesthesia and Analgesia. 1993;77(6):1122-1129.
Harding J, Kemper M, Weissman C. Pressure support ventilation attenuates the cardiopulmonary response to an acute increase in oxygen demand. Chest. 1995;107(6):1665-1672.
Haug E, Miner J, Dannehy M, Seigel T, Biros M. Bispectral electroencephalographic analysis of head-injured patients in the emergency department. Academic Emergency Medicine. 2004;11(4):349-352.
Hedenstierna G, Brismar B, Strandberg A, Lundquist H, Tokics L. New aspects on atelectasis during anaesthesia. Clinical Physiology. 1985;5(Suppl 3):127-131.
Heffner JE. Management of the chronically ventilated patient with a tracheostomy. Chronic Respiratory Disease. 2005;2(3):151-161.
Heffner JE, Hess D. Tracheostomy management in the critically ill patient. Clinical Chest Medicine. 2001;22:55-69.
Hess DE. Tracheostomy tubes and related appliances. Respiratory Care. 2005;50:497-510.
Hilbert G, Gruson D, Vargas F, et al. Noninvasive ventilation in immunosuppressed patients with pulmonary infiltrates, fever, and acute respiratory failure. New England Journal of Medicine. 2001;344(7):481-487.
Hodgson C, Denehy L, Ntoumenopoulos G, Santamaria J, Carroll S. An investigation of the early effects of manual lung hyperinflation in critically ill patients. Anaesthesia and Intensive Care. 2000;28(3):255-261.
Horiuchi K, Jordan D, Cohen D, Kemper MC, Weissman C. Insights into the increased oxygen demand during chest physiotherapy. Critical Care Medicine. 1997;25(8):1347-1351.
Huang SJ, Hong WC, Han YY, et al. Clinical outcome of severe head injury using three different ICP and CPP protocol-driven therapies. Journal of Clinical Neuroscience. 2006;13(8):818-822.
Hussey JD, Bishop MJ. Pressures required to move gas through the native airway in the presence of a fenestrated vs a nonfenestrated tracheostomy tube. Chest. 1996;110:494-497.
Ibanez J, Raurich JM, Abizanda R, et al. The effect of lateral positions on gas exchange in patients with unilateral lung disease during mechanical ventilation. Intensive Care Medicine. 1981;7(5):231-234.
Jaffe MB. Partial CO2 rebreathing cardiac output - operating principles of the NICO system. Journal of Clinical Monitoring and Computing. 1999;15(6):387-401.
Jensen LA, Onyskiw JE, Prasad NG. Meta-analysis of arterial oxygen saturation monitoring by pulse oximetry in adults. Heart and Lung. 1998;27(6):387-408.
Jones AYM. Intensive care physiotherapy - medical staff perceptions. Hong Kong Physiotherapy Journal. 2001;19:9-16.
Jones AYM. Secretion movement during manual lung inflation and mechanical ventilation. Respiratory Physiology and Neurobiology. 2002;132(3):321-327.
Jones AYM, Hutchinson RC, Oh TE. comparison of the peak expiratory flow rates produced by two different circuits in intubated patients. Australian Journal of Physiotherapy. 1992;38:211-215.
Jones AYM, Hutchinson RC, Oh TE. Effects of bagging and percussion on total static compliance of the respiratory system. Physiotherapy. 1992;78:661-666.
Jones AYM, Jones RDM, Bacon-Shone J. A comparison of the expiratory flow rates in two breathing circuits used for manual inflation of the lungs. Physiotherapy. 1991;77(9):593-597.
Jorgensen L, Jacobsen BK, Wilsgaard T, Magnus JH. Walking after stroke: does it matter? Changes in bone mineral density within the first 12 months after stroke. A longitudinal study. Osteoporosis International. 2000;11(5):381-387.
Jubran A. Critical illness and mechanical ventilation: effects on the diaphragm. Respiratory Care. 2006;51(9):1054-1061. ; discussion 1062-1064
Kendrick AH. Non-invasive blood gas measurement. 1. Pulse Oximetry. In: The buyer’s guide. European Respiratory Society; 2001:15-23.
Kirschenbaum L, Azzi E, Sfeir T, Tietjen P, Astiz M. Effect of continuous lateral rotational therapy on the prevalence of ventilator-associated pneumonia in patients requiring long-term ventilatory care. Critical Care Medicine. 2002;30(9):1983-1986.
Kollef MH, Shapiro SD, Silver P, et al. A randomized, controlled trial of protocol-directed versus physician-directed weaning from mechanical ventilation. Critical Care Medicine. 1997;25(4):567-574.
Konrad F, Schreiber T, Brecht-Kraus D, Georgieff M. Mucociliary transport in ICU patients. Chest. 1994;105(1):237-241.
Krause MW, van Aswegen H, de Wet EH. Postural drainage in intubated patients with acute lobar atelectasis: a pilot study. South African Journal of Physiotherapy. 2000;56(3):29-32.
Krishnan JA, Moore D, Robeson C, Rand CS, Fessler HE. A prospective, controlled trial of a protocol-based strategy to discontinue mechanical ventilation. American Journal of Respiratory and Critical Care Medicine. 2004;169(6):673-678.
Kumar AB. Imaging techniques in intensive care. In: Oh TE, editor. Intensive care manual. 4th edn. Oxford: Butterworth-Heinemann; 1997:855-859.
Kuo PH, Wu HD, Lu BY, et al. Predictive value of rapid shallow breathing index measured at initiation and termination of a 2-hour spontaneous breathing trial for weaning outcome in ICU patients. Journal of the Formosan Medical Association. 2006;105(5):390-398.
Ladyshewsky A, Gousseau A. Successful tracheal weaning. Canadian Nurse. 1996;92(2):35-38.
Lasocki S, Lu Q, Sartorius A, et al. Open and closed-circuit endotracheal suctioning in acute lung injury: efficiency and effects on gas exchange. Anesthesiology. 2006;104(1):39-47.
Latronico N, Shehu I, Seghelini E. Neuromuscular sequelae of critical illness. Current Opinion in Critical Care. 2005;11(4):381-390.
Lee CK, Ng KS, Tan SG, Ang R. Effect of different endotracheal suctioning systems on cardiorespiratory parameters of ventilated patients. Annals of the Academy of Medicine, Singapore. 2001;30(3):239-244.
Lellouche F, Mancebo J, Jolliet P, et al. A multicenter randomized trial of computer-driven protocolized weaning from mechanical ventilation. American Journal of Respiratory and Critical Care Medicine. 2006;174(8):894-900.
Lerga C, Zapata MA, Herce A, et al. [Endotracheal suctioning of secretions: effects of instillation of normal serum]. Enfermería Intensiva. 1997;8(3):129-137.
Levitt JE, Matthay MA. Treatment of acute lung injury: historical perspective and potential future therapies. Semin Respiratory Critical Care Medicine. 2006;27(4):426-437.
Lindegaard K-F. Indices of pulsatility. In: Newell DW, Aaslid R, editors. Transcranial Doppler. New York: Raven Press; 1992:67-82.
Maa SH, Hung TJ, Hsu KH, et al. Manual hyperinflation improves alveolar recruitment in difficult-to-wean patients. Chest. 2005;128(4):2714-2721.
MacIntyre NR. Evidence-based ventilator weaning and discontinuation. Respiratory Care. 2004;49(7):830-836.
MacIntyre NR. Current issues in mechanical ventilation for respiratory failure. Chest. 2005;128(5 Suppl 2):561S-7S.
MacLean D, Drummond G, Macpherson C, McLaren G, Prescott R. Maximum expiratory airflow during chest physiotherapy on ventilated patients before and after the application of an abdominal binder. Intensive Care Medicine. 1989;15(6):396-399.
Maggiore SM, Lellouche F, Pigeot J, et al. Prevention of endotracheal suctioning-induced alveolar derecruitment in acute lung injury. American Journal of Respiratory and Critical Care Medicine. 2003;167(9):1215-1224.
Man WD, Moxham J, Polkey MI. Magnetic stimulation for the measurement of respiratory and skeletal muscle function. European Respiratory Journal. 2004;24(5):846-860.
Martin AD, Davenport PD, Franceschi AC, Harman E. Use of inspiratory muscle strength training to facilitate ventilator weaning: a series of 10 consecutive patients. Chest. 2002;122(1):192-196.
Matthay MA, Zimmerman GA, Esmon C, et al. Future research directions in acute lung injury: summary of a National Heart, Lung, and Blood Institute working group. American Journal of Respiratory and Critical Care Medicine. 2003;167(7):1027-1035.
Maxwell LJ, Ellis ER. The effect of circuit type, volume delivered and ‘rapid release’ on flow rates during manual hyperinflation. Australian Journal of Physiotherapy. 2003;49(1):31-38.
McCarren B, Chow CM. Description of manual hyperinflation in intubated patients with atelectasis. Physiotherapy Theory and Practice. 1998;14:199-210.
McPherson K, Kersten P, George S, et al. A systematic review of evidence about extended roles for allied health professionals. Journal of Health Services Research and Policy. 2006;11(4):240-247.
Messerole E, Peine P, Wittkopp S, Marini JJ, Albert RK. The pragmatics of prone positioning. American Journal of Respiratory and Critical Care Medicine. 2002;165(10):1359-1363.
Metnitz PG, Bartens C, Fischer M, et al. Antioxidant status in patients with acute respiratory distress syndrome. Intensive Care Medicine. 1999;25(2):180-185.
Miller RD. Monitors of cerebral blood flow. In Miller’s anesthesia, 6th edn., Philadelphia, PA: Elsevier Churchill Livingstone; 2005:1540-1542. [Edinburgh]
Mills GH, Ponte J, Hamnegard CH, et al. Tracheal tube pressure change during magnetic stimulation of the phrenic nerves as an indicator of diaphragm strength on the intensive care unit. British Journal of Anaesthesia. 2001;87(6):876-884.
Minei JP, Nathens AB, West M, et al. Inflammation and the host response to injury, a large-scale collaborative project: patient-oriented research core-standard operating procedures for clinical care. II. Guidelines for prevention, diagnosis and treatment of ventilator-associated pneumonia (VAP) in the trauma patient. Journal of Trauma. 2006;60(5):1106-1113.
Mols G, Priebe HJ, Guttmann J. Alveolar recruitment in acute lung injury. British Journal of Anaesthesia. 2006;96(2):156-166.
Monafo WW. Initial management of burns. New England Journal of Medicine. 1996;335(21):1581-1586.
Moran T, Bellapart J, Vari A, Mancebo J. Heat and moisture exchangers and heated humidifiers in acute lung injury/acute respiratory distress syndrome patients. Effects on respiratory mechanics and gas exchange. Intensive Care Medicine. 2006;32(4):524-531.
Nekludov M, Bellander B, Mure M. Oxygenation and cerebral perfusion pressure improved in prone position. Acta Anaesthesiologica Scandinavica. 2006;50(8):932-936.
Newman DG, Callister R. The non-invasive assessment of stroke volume and cardiac output by impedance cardiography: a review. Aviation, Space and Environmental Medicine. 1999;70(8):780-789.
Norrenberg M, Vincent JL. A profile of European intensive care unit physiotherapists. European Society of Intensive Care Medicine. Intensive Care Medicine. 2000;26(7):988-994.
Ntoumenopoulos G, Presneill JJ, McElholum M, Cade JF. Chest physiotherapy for the prevention of ventilator-associated pneumonia. Intensive Care Medicine. 2002;28(7):850-856.
O’Riordan TG, Mao W, Palmer LB, Chen JJ. Assessing the effects of racemic and single-enantiomer albuterol on airway secretions in long-term intubated patients. Chest. 2006;129(1):124-132.
Paratz J, Burns Y. The effect of respiratory physiotherapy on intracranial pressure, mean arterial pressure, cerebral perfusion pressure, and end tidal carbon dioxide in ventilated neurosurgical patients. Physiotherapy Theory and Practice. 1993;9:3-11.
Paratz J, Lipman J. Manual hyperinflation causes norepinephrine release. Heart and Lung. 2006;35(4):262-268.
Paratz J, Lipman J, McAuliffe M. Effect of manual hyperinflation on hemodynamics, gas exchange, and respiratory mechanics in ventilated patients. Journal of Intensive Care Medicine. 2002;17(6):317-324.
Paratz J, Zeppos L, Patman S, et al. Adverse physiological events in intensive care - is it an issue? Adelaide: Australian and New Zealand Society Intensive Care Medicine, 2005.
Patman S, Jenkins S, Stiller K. Manual hyperinflation - effects on respiratory parameters. Physiotherapy Research International. 2000;5(3):157-171.
Pelosi P, Brazzi L, Gattinoni L. Prone position in acute respiratory distress syndrome. European Respiratory Journal. 2002;20(4):1017-1028.
Pelosi P, D’Onofrio D, Chiumello D, et al. Pulmonary and extrapulmonary acute respiratory distress syndrome are different. European Respiratory Journal Suppl. 2003;42:48s-56s.
Petrie GJ, Barnwell E, Grimshaw J, On behalf of the Scottish Intercolegiate Guidelines Network. Clinical guidelines: criteria for appraisal for national use. Edinburgh: Royal College of Physicians, 1995.
Petrucci N, Iacovelli W. Ventilation with lower tidal volumes versus traditional tidal volumes in adults for acute lung injury and acute respiratory distress syndrome. Cochrane Database of Systematic Reviews. (2):2004. ): CD003844
Pierson DJ. Tracheostomy and weaning. Respiratory Care. 2005;50(4):526-533.
Pilbeam SP. Mechanical ventilation: physiological and clinical applications, 2nd edn., St. Louis: Mosby Year Book; 1992:169-170.
Pillastrini P, Bordini S, Bazzocchi G, Belloni G, Menarini M. Study of the effectiveness of bronchial clearance in subjects with upper spinal cord injuries: examination of a rehabilitation programme involving mechanical insufflation and exsufflation. Spinal Cord. 2006;44(10):614-616.
PULSION Medical Systems AG: PiCCO technology www.pulsion.com/index.php?id=39 (accessed 13 July 2007)
Raoof S, Chowdhrey N, Raoof S, et al. Effect of combined kinetic therapy and percussion therapy on the resolution of atelectasis in critically ill patients. Chest. 1999;115(6):1658-1666.
Richard JC, Maggiore SM, Mancebo J, et al. Effects of vertical positioning on gas exchange and lung volumes in acute respiratory distress syndrome. Intensive Care Medicine. 2006;32(10):1623-1626.
Richman PS, Baram D, Varela M, Glass PS. Sedation during mechanical ventilation: a trial of benzodiazepine and opiate in combination. Critical Care Medicine. 2006;34(5):1395-1401.
Rose L, Nelson S. Issues in weaning from mechanical ventilation: literature review. Journal of Advanced Nursing. 2006;54(1):73-85.
Rosow C, Manberg PJ. Bispectral index monitoring. Anesthesiology Clinics of North America. 2001;19(4):947-966. xi
Rothen HU, Neumann P, Berglund JE, et al. Dynamics of re-expansion of atelectasis during general anaesthesia. British Journal of Anaesthesia. 1999;82(4):551-556.
Rudy EB, Turner BS, Baun M, Stone KS, Brucia J. Endotracheal suctioning in adults with head injury. Heart and Lung. 1991;20(6):667-674.
Rusterholz B, Ellis E. The effect of lung compliance and experience on manual hyperinflation. Australian Journal of Physiotherapy. 1998;44(1):23-28.
Sancho J, Servera E, Vergara P, Marin J. Mechanical insufflation-exsufflation vs. tracheal suctioning via tracheostomy tubes for patients with amyotrophic lateral sclerosis: a pilot study. America Journal of Physical Medicine and Rehabilitation. 2003;82(10):750-753.
Sassoon CS, Caiozzo VJ, Manka A, Sieck GC. Altered diaphragm contractile properties with controlled mechanical ventilation. Journal of Applied Physiology. 2002;92(6):2585-2595.
Savian C, Chan P, Paratz J. The effect of positive end-expiratory pressure level on peak expiratory flow during manual hyperinflation. Anesthesia and Analgesia. 2005;100(4):1112-1116.
Scherhag A, Kaden JJ, Kentschke E, Sueselbeck T, Borggrefe M. Comparison of impedance cardiography and thermodilution-derived measurements of stroke volume and cardiac output at rest and during exercise testing. Cardiovascular Drugs and Therapy. 2005;19(2):141-147.
Schreuder FM, Jones UF, 2004 The effect of saline installation on sputum yield and oxygen saturation measurement in adult intubated patients. Abstract, Thoracic Society Australia and New Zealand, Sydney, Australia
Senn O, Clarenbach CF, Kaplan V, Maggiorini M, Bloch KE. Monitoring carbon dioxide tension and arterial oxygen saturation by a single earlobe sensor in patients with critical illness or sleep apnea. Chest. 2005;128(3):1291-1296.
Shankar G, Cohen DA. Ideopathic pneumonia syndrome after bone marrow transplantation: the role of pre-transplant radiation conditioning and local cytokine dysregulation in promoting inflammation and fibrosis. International Journal of Experimental Pathology. 2001;82(2):101-113.
Shapiro BA, Mahutte CK, Cane RD, Gilmour IJ. Clinical performance of a blood gas monitor: a prospective, multicenter trial. Critical Care Medicine. 1993;21(4):487-494.
Sheridan RL. Airway management and respiratory care of the burn patient. Internatonal Anesthesiology Clinics. 2000;38(3):129-145.
Spicer M, Hughes P, Green M. A non-invasive system to evaluate diaphragmatic strength in ventilated patients. Physiological Measurement. 1997;18(4):355-361.
Stawicki SP, Grossman MD, Hoey BA, Miller DL, Reed JF3rd. Rib fractures in the elderly: a marker of injury severity. Journal of the American Geriatrics Society. 2004;52(5):805-808.
Stiller K. Physiotherapy in intensive care: towards an evidence-based practice. Chest. 2000;118(6):1801-1813.
Stiller K, Geake T, Taylor J, Grant R, Hall B. Acute lobar atelectasis: a comparison of two chest physiotherapy regimens. Chest. 1990;98(6):1336-1340.
Stiller K, Phillips A, Lambert P. The safety of mobilization and its effects on haemodynamic and respiratory status of intensive care patients. Physiotherapy Theory and Practice. 2004;20:175-185.
Thelandersson A, Cider A, Nellgard B. Prone position in mechanically ventilated patients with reduced intracranial compliance. Acta Anaesthesiologica Scandinavica. 2006;50(8):937-941.
Tjin SC, Ho YC, Lam YZ, Hao J, Ng BK. Continuous cardiac output monitoring system. Medical and Biological Engineering and Computing. 2001;39(1):101-104.
Tonnelier JM, Prat G, Le Gal G, et al. Impact of a nurses’ protocol-directed weaning procedure on outcomes in patients undergoing mechanical ventilation for longer than 48 hours: a prospective cohort study with a matched historical control group. Critical Care. 2005;9(2):R83-89.
Topp R, Ditmyer M, King K, Doherty K, Hornyak J. The effect of bed rest and potential of prehabilitation on patients in the intensive care unit. AACN Clinical Issues. 2002;13(2):263-276.
Turner JS. Monitoring and interpreting medical investigations. In: Pryor JA, Prasad SA, editors. Physiotherapy for respiratory and cardiac problems: adults and paediatrics. 3rd edn. Edinburgh: Churchill Livingstone; 2002:132-134.
Ulatowski J. Cerebral protection. In: Oh TE, editor. Intensive care manual. 4th edn. Oxford: Butterworth-Heinemann; 1997:403-411.
Unoki T, Mizutani T, Toyooka H. Effects of expiratory rib cage compression combined with endotracheal suctioning on gas exchange in mechanically ventilated rabbits with induced atelectasis. Respiratory Care. 2004;49(8):896-901.
Vassilakopoulos T, Zakynthinos S, Roussos C. Bench-to-bedside review: Weaning failure - should we rest the respiratory muscles with controlled mechanical ventilation? Critical Care 2006. 2006;10(1):204.
Voulgaris SG, Partheni M, Kaliora H, et al. Early cerebral monitoring using the transcranial Doppler pulsatility index in patients with severe brain trauma. Medical Science Monitor. 2005;11(2):49-52. CR
Weavind L, Wenker OC. Newer modes of ventilation: an overview. The Internet Journal of Anesthesiology. 2000;4:4.
Weiss SJ, Kulik JP, Calloway E. Bioimpedance cardiac output measurements in patients with presumed congestive heart failure. Academic Emergency Medicine. 1997;4(6):568-573.
Weissman C, Kemper M. The oxygen uptake-oxygen delivery relationship during ICU interventions. Chest. 1991;99(2):430-435.
West JB. Gas transport by the blood - how gases are moved to the peripheral tissues. In Respiratory physiology: the essentials, 7th edn., Philadelphia, PA: Lippincott Williams & Wilkins; 2005:77.
White H, Baker A. Continuous jugular venous oximetry in the neurointensive care unit - a brief review. Canadian Journal of Anaesthesia. 2002;49(6):623-629.
Whitehead MD, Halsall D, Pollitzer MJ, et al. Transcutaneous estimation of arterial PO2 and PCO2 in newborn infants with a single electrochemical sensor. Lancet. 1980;1(8178):111-114.
Wilson JN, Pierce JD, Clancy RL. Reactive oxygen species in acute respiratory distress syndrome. Heart and Lung. 2001;30(5):370-375.
Winkelman C. Inactivity and inflammation: selected cytokines as biologic mediators in muscle dysfunction during critical illness. AACN Clinical Issues. 2004;15(1):74-82.
Worthley LI. Shock: a review of pathophysiology and management. Part II. Critical Care and Resuscitation. 2000;2(1):66-84.
Yelderman ML, Ramsay MA, Quinn MD, et al. Continuous thermodilution cardiac output measurement in intensive care unit patients. Journal of Cardiothoracic and Vascular Anesthesia. 1992;6(3):270-274.
Young RJ, Sanderson JE. Echocardiography in intensive care. In: Oh TE, editor. Intensive care manual. 4th edn. Oxford: Butterworth-Heinemann; 1997:197-205.
Zador E, Dux L, Wuytack F. Prolonged passive stretch of rat soleus muscle provokes an increase in the mRNA levels of the muscle regulatory factors distributed along the entire length of the fibers. Journal of Muscle Research and Cell Motility. 1999;20(4):395-402.
Zafiropoulos B, Alison JA, McCarren B. Physiological responses to the early mobilisation of the intubated, ventilated abdominal surgery patient. Australian Journal of Physiotherapy. 2004;50(2):95-100.
Zimmerman GA, Albertine KH, Carveth HJ, et al. Endothelial activation in ARDS. Chest. 1999;116(1 Suppl):18S-24S.
Zimmerman JL, Dellinger RP. Initial evaluation of a new intra-arterial blood gas system in humans. Critical Care Medicine. 1993;21(4):495-500.