The respiratory examination
Examination anatomy
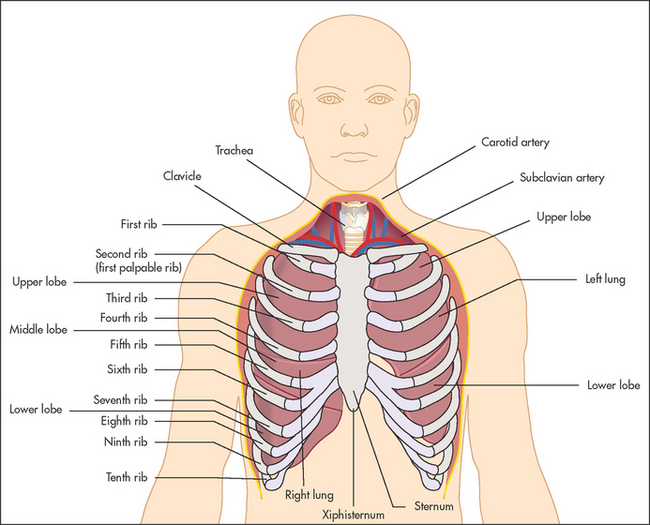
The lungs are paired asymmetrical organs protected by the cylinder composed of the ribs, vertebrae and diaphragm. The surface of the lungs is covered by the visceral pleura, a thin membrane, and a similar outer layer (the parietal pleura) lines the rib cage. These membranes are separated by a thin layer of fluid and enable the lungs to move freely during breathing. Various diseases of the lungs and of the pleura themselves, including infection and malignancy, can cause accumulation of fluid within the pleural cavity (a pleural effusion).
The heart, trachea, oesophagus and the great blood vessels and nerves sit between the lungs and make up the structure called the mediastinum. The left and right pulmonary arteries supply their respective lung. Gas exchange occurs in the pulmonary capillaries that surround the alveoli, the tiny air sacs which lie beyond the terminal bronchioles. Oxygenated blood is returned via the pulmonary veins to the left atrium. Abnormalities of the pulmonary circulation such as raised pulmonary venous pressure resulting from heart failure or pulmonary hypertension can interfere with gas exchange.
The position of the heart, whose apex points to the left, means that the left lung is smaller than the right and has only two lobes, which are separated by the oblique fissure. The right lung has both horizontal (upper) and oblique (lower) fissures dividing it into three lobes (see Figure 10.1).
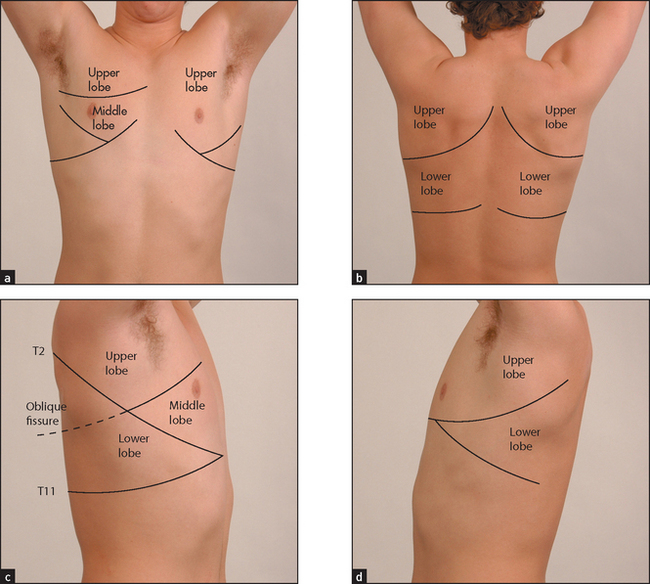
Figure 10.1 Lobes of the lung (a) Anterior; (b) posterior; (c) lobes of the right lung; (d) lobes of the left lung. Refer to Figure 12.4, page 165, for a list of the segments in each lobe. (Courtesy of Glenn McCulloch)
The muscles of respiration are the diaphragm upon which the bases of the lungs rest and the intercostal muscles. During inspiration, the diaphragm flattens and the intercostal muscles contract to elevate the ribs. Intrathoracic pressure falls as air is forced under atmospheric pressure into the lungs. Expiration is a passive process resulting from elastic recoil of the muscles. Abnormalities of lung function or structure may change the normal anatomy and physiology of respiration, for example as a result of over-inflation of the lungs (chronic obstructive pulmonary disease [COPD]. Muscle and neurological diseases can also affect muscle function adversely, and abnormalities of the control of breathing in the respiratory centres of the brain in the pons and medulla can interfere with normal breathing patterns.
During the respiratory examination, keep in mind the surface anatomy (see Figure 10.2) of the lungs and try to decide which lobes are affected.
Positioning the patient
The patient should be undressed to the waist.1 Women should wear a gown or have a towel or some clothing to cover their breasts when the front of the chest is not being examined. If the patient is not acutely ill, the examination is easiest to perform with him or her sitting over the edge of the bed or on a chair.
General appearance
If the patient is an inpatient in hospital, look around the bed for oxygen masks, metered dose inhalers (puffers) and other medications, and the presence of a sputum mug. Then make a deliberate point of looking for the following signs before beginning the detailed examination.
DYSPNOEA
Watch the patient for signs of dyspnoea at rest. Count the respiratory rate; the normal rate at rest should not exceed 25 breaths per minute (range 16–25). The frequently quoted normal value of 14 breaths per minute is probably too low; normal people can have a respiratory rate of up to 25, and the average is 20 breaths per minute. It is traditional to count the respiratory rate surreptitiously while affecting to count the pulse. The respiratory rate is the only vital sign that is under direct voluntary control. Tachypnoea refers to a rapid respiratory rate of greater than 25. Bradypnoea is defined as a rate below 8, a level associated with sedation and adverse prognosis. In normal relaxed breathing, the diaphragm is the only active muscle and is active only in inspiration; expiration is a passive process.
CHARACTERISTIC SIGNS OF COPD
Look to see whether the accessory muscles of respiration are being used. This is a sign of an increase in the work of breathing, and COPDa is an important cause. These muscles include the sternocleidomastoids, the platysma and the strap muscles of the neck. Characteristically, the accessory muscles cause elevation of the shoulders with inspiration, and aid respiration by increasing chest expansion. Contraction of the abdominal muscles may occur in expiration in patients with obstructed airways. Patients with severe COPD often have in-drawing of the intercostal and supraclavicular spaces during inspiration. This is due to a delayed increase in lung volume despite the generation of large negative pleural pressures.
In some cases, the pattern of breathing is diagnostically helpful (see Table 9.4, page 134). Look for pursed-lips breathing, which is characteristic of patients with severe COPD. This manoeuvre reduces the patient’s breathlessness, possibly by providing continuous positive airways pressure and helping to prevent airways collapse during expiration. Patients with severe COPD may feel more comfortable leaning forwards with their arms on their knees. This position compresses the abdomen and pushes the diaphragm upwards. This partly restores its normal domed shape and improves its effectiveness during inspiration. Increased diaphragmatic movements may cause downward displacement of the trachea during inspiration—tracheal tug (this is also a sign of severe asthma, especially in children).b
CYANOSIS
Central cyanosis is best detected by inspecting the tongue. Examination of the tongue differentiates central from peripheral cyanosis. Lung disease severe enough to result in significant ventilation–perfusion imbalance (such as pneumonia, COPD and pulmonary embolism), may cause reduced arterial oxygen saturation and central cyanosis. Cyanosis becomes evident when the absolute concentration of deoxygenated haemoglobin is 50 g/L of capillary blood. Cyanosis is usually obvious when the arterial oxygen saturation falls below 90% in a person with a normal haemoglobin level. Central cyanosis is therefore a sign of severe hypoxaemia. In patients with anaemia, cyanosis does not occur until even greater levels of arterial desaturation are reached. The absence of obvious cyanosis does not exclude hypoxia. The detection of cyanosis is much easier in good (especially fluorescent) lighting conditions and is said to be more difficult if the patient’s bed is surrounded by cheerful pink curtains.
CHARACTER OF THE COUGH
Coughing is a protective response to irritation of sensory receptors in the submucosa of the upper airways or bronchi. Ask the patient to cough several times. Lack of the usual explosive beginning may indicate vocal cord paralysis (the ‘bovine’ cough). A muffled, wheezy, ineffective cough suggests obstructive pulmonary disease. A very loose productive cough suggests excessive bronchial secretions due to chronic bronchitis, pneumonia or bronchiectasis. A dry, irritating cough may occur with chest infection, asthma or carcinoma of the bronchus and sometimes with left ventricular failure or interstitial lung disease (ILD). It is also typical of the cough produced by ACE inhibitor drugs. A barking or croupy cough may suggest a problem with the upper airway—the pharynx and larynx, or pertussis infection.
SPUTUM
Sputum should be inspected. Careful study of the sputum is an essential part of the physical examination. The colour, volume and type (purulent, mucoid or mucopurulent), and the presence or absence of blood, should be recorded.
STRIDOR
Obstruction of the larynx or trachea (the extra-thoracic airways) may cause stridor, a rasping or croaking noise loudest on inspiration. This can be due to a foreign body, a tumour, infection (e.g. epiglottitis) or inflammation (see List 10.1). It is a sign that requires urgent attention.
HOARSENESS
Listen to the patient’s voice for hoarseness (dysphonia), as this may indicate recurrent laryngeal nerve palsy associated with carcinoma of the lung (usually left-sided), or laryngeal carcinoma. However, the most common cause is laryngitis and the use of inhaled corticosteroids for asthma. Non-respiratory causes include hypothyroidism.
Hands
As usual, examination in detail begins with the hands.
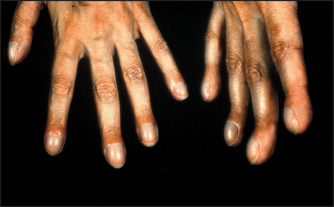
CLUBBING
Look for clubbing, which is due to respiratory disease in up to 80% of cases (see Figure 10.3 and List 5.1 on page 62). An uncommon but important association with clubbing is hypertrophic pulmonary osteoarthropathy (HPO). HPO is characterised by the presence of periosteal inflammation at the distal ends of long bones, the wrists, the ankles and the metacarpal and the metatarsal bones. There is swelling and tenderness over the wrists and other involved areas. Rarely HPO may occur without clubbing. The causes of HPO include primary lung carcinoma and pleural fibromas. It is important to note that clubbing does not occur as a result of COPD.
STAINING
Look for staining of the fingers (actually caused by tar, as nicotine is colourless); a sign of cigarette smoking (see Figure 3.12 on page 36). The density of staining does not indicate the number of cigarettes smoked, but depends rather on the way the cigarette is held in the hand.
WASTING AND WEAKNESS
Compression and infiltration by a peripheral lung tumour of a lower trunk of the T1 nerve root results in wasting of the small muscles of the hand and weakness of finger abduction.
PULSE RATE
Tachycardia and pulsus paradoxus are important signs of severe asthma. Tachycardia is a common side effect of the treatment of asthma with β-agonist drugs, and accompanies dyspnoea or hypoxia of any cause.
FLAPPING TREMOR (ASTERIXIS)
Ask the patient to dorsiflex the wrists with the arms outstretched and to spread out the fingers. A flapping tremor with a 2- to 3-second cycle may occur with severe CO2 retention, usually due to severe COPD.2 The problem is an inability to maintain a posture. Asterixisc can also be demonstrated by asking the patient to protrude the tongue or lift the leg and keep the foot dorsiflexed. However, this is a late and unreliable sign and can also occur in patients with liver or renal failure. Patients with severe CO2 retention may be confused, and typically have warm peripheries and a bounding pulse.
Face
The nose is sited conveniently in the centre of the face. In this position it may readily be inspected inside and out. Ask the patient to tilt the head back. It may be necessary to use a nasal speculum to open the nostrils, and a torch. Look for polyps (associated with asthma), engorged turbinates (various allergic conditions) and a deviated septum (nasal obstruction).
As already discussed, look at the tongue for central cyanosis. Look in the mouth for evidence of an upper respiratory tract infection (a reddened pharynx and tonsillar enlargement, with or without a coating of pus). A broken tooth or a rotten tooth stump may predispose to lung abscess or pneumonia. Patients with sleep apnoea may have ‘crowding’ of the pharynx. This means a reduction in the size of the velopharyngeal lumen, which is the space between the soft palate, the tonsils and the back of the tongue. Those who use a sleep apnoea mask at night often have marks from the mask on the face and puffiness around the eyes. They tend to be obese and have a short thick neck and a small pharynx; sometimes the maxilla and mandible appear retracted (receding chin).
Sinusitis is suggested by tenderness over the sinuses on palpation. If acute sinusitis is suspected, a torch can be used to transilluminate the frontal and maxillary sinuses.3 Place the torch in the patient’s mouth and examine the sinuses in a dark room. Normal transillumination generally excludes sinusitis. Complete opacification suggests sinusitis but partial opacification is less helpful. The torch should then be used to look for purulent discharge in the pharynx.
Look at the patient’s face for the red, leathery, wrinkled skin of the smoker.4 There may be facial plethora or cyanosis if superior vena caval obstruction is present. Look for the characteristics of obstructive sleep apnoea (see above).
Inspect the eyes for evidence of the rare Horner’sd syndrome (a constricted pupil, partial ptosis and loss of sweating), which can be due to an apical lung carcinoma (Pancoast’se tumour) compressing the sympathetic nerves in the neck. There may be skin changes on the face that suggest scleroderma or connective tissue disease.
Trachea
The position of the trachea is most important, and time should be spent establishing it accurately. This examination is uncomfortable for the patient, so you must be gentle. From in front of the patient push the forefinger of your right hand up and backwards from the suprasternal notch until the trachea is felt (see Figure 10.4). If the trachea is displaced to one side, its edge rather than its middle will be felt and a larger space will be present on one side than the other. Slight displacement to the right is fairly common in normal people. Significant displacement of the trachea suggests, but is not specific for, disease of the upper lobes of the lung (see List 10.2).
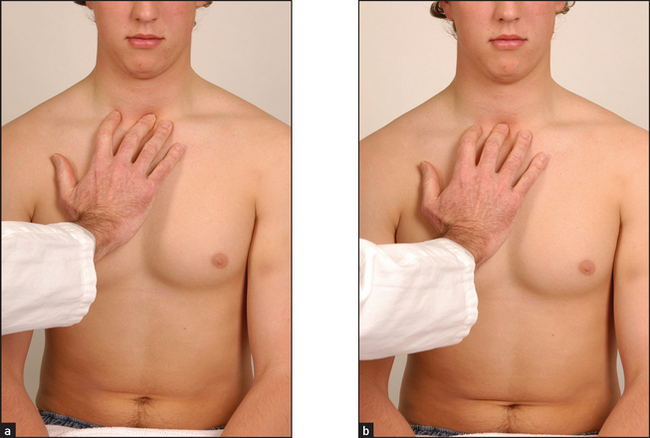
Figure 10.4 Feeling for the position of the trachea—a similar gap should be palpable on each side (Courtesy of Glenn McCulloch)
A tracheal tug is demonstrated when the finger resting on the trachea feels it move inferiorly with each inspiration. This is a sign of gross overexpansion of the chest because of airflow obstruction. This movement of the trachea may be visible, and it is worth spending time inspecting the trachea when COPD is suspected.
If the patient appears dyspnoeic and use of the accessory muscles of respiration is suspected, place your fingers in the supraclavicular fossae. When the scalene muscles are recruited, they can be felt to contract under the fingers. Even more severe dyspnoea will result in use of the sternomastoid muscles. Their contraction is also easily felt with inspiration. Use of these muscles for long periods is exhausting and a sign of impending respiratory failure.
Chest
The chest should be examined anteriorly and posteriorly by inspection, palpation, percussion and auscultation.1 Compare the right and left sides during each part of the examination.
INSPECTION
Shape and symmetry of chest
When the anteroposterior (AP) diameter is increased compared with the lateral diameter, the chest is described as barrel-shaped (see Figure 10.5). An increase in the AP diameter compared with the lateral diameter (the thoracic ratio) beyond 0.9 is abnormal and is often seen in patients with severe asthma or emphysema. It is not always a reliable guide to the severity of the underlying lung disease and may be present in normal elderly people. It is sometimes an illusion when the abdomen is relatively small in thin people. Severe kyphoscoliosis is a cause of asymmetrical chest deformity.
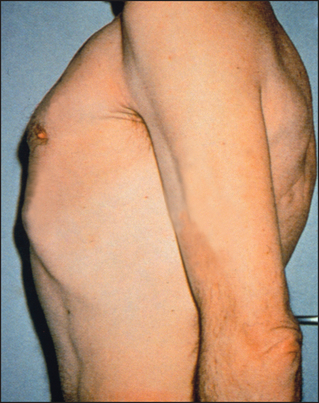
Figure 10.5 Barrel chest (From McDonald FS, ed. Mayo Clinic images in internal medicine, with permission. © Mayo Clinic Scientific Press and CRC Press.)
A pigeon chest (pectus carinatum) is a localised prominence (an outward bowing of the sternum and costal cartilages; see Figure 10.6(b)). It may be a manifestation of chronic childhood respiratory illness, in which case it is thought to result from repeated strong contractions of the diaphragm while the thorax is still pliable. It also occurs in rickets.f
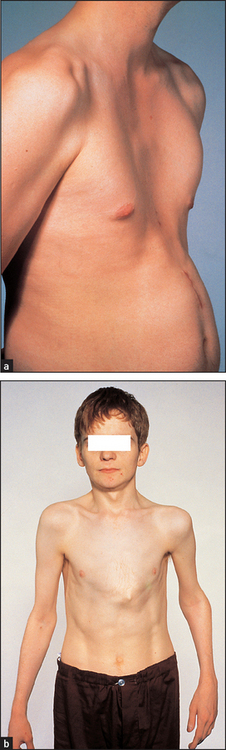
Figure 10.6 (a) Funnel chest (pectus excavatum); (b) Pigeon chest (pectus carinatum) (From Mir MA. Atlas of clinical diagnosis, 2nd edn. Edinburgh: Saunders, 2003, with permission.)
A funnel chest (pectus excavatum) is a developmental defect involving a localised depression of the lower end of the sternum (see Figure 10.6(a)). The problem is usually an aesthetic one, but in severe cases lung capacity may be restricted.
Harrison’sg sulcus is a linear depression of the lower ribs just above the costal margins at the site of attachment of the diaphragm. It can result from severe asthma in childhood, or rickets.
Kyphosis refers to an exaggerated forward curvature of the spine, while scoliosis is lateral bowing. Kyphoscoliosis may be idiopathic (80%), secondary to poliomyelitis or associated with Marfan’s syndrome. Severe thoracic kyphoscoliosis may reduce the lung capacity and increase the work of breathing.
Lesions of the chest wall may be obvious. Look for scars from previous thoracic operations or from chest drains for a previous pneumothorax or pleural effusion. Surgical removal of a lung (pneumonectomy) or of the lobe of a lung (lobectomy) leaves a long diagonal posterior scar on the thorax. The presence of three 2–3 centimetre scars suggests previous video-assisted thoracoscopic surgery, which can be performed to biopsy lymph nodes or carry out lung reduction surgery or pleurodesis. Thoracoplasty causes severe chest deformity; this operation was performed for tuberculosis and involved removal of a large number of ribs on one side of the chest to achieve permanent collapse of the affected lung. It is no longer performed because of the availability of effective anti-tuberculous chemotherapy.
Radiotherapy may cause erythema and thickening of the skin over the irradiated area. There is sharp demarcation between abnormal and normal skin. There may be small tattoo marks indicating the limits of the irradiated area. Signs of radiotherapy usually indicate that the patient has been treated for carcinoma of the lung or breast or, less often, for lymphoma.
Subcutaneous emphysema is a crackling sensation felt on palpating the skin of the chest or neck. On inspection, there is often diffuse swelling of the chest wall and neck. It is caused by air tracking from the lungs and is usually due to a pneumothorax; less commonly it can follow rupture of the oesophagus or a pneumomediastinum (air in the mediastinal space).
Prominent veins may be seen in patients with superior vena caval obstruction. It is important to determine the direction of blood flow (page 158).
Movement of the chest wall should be noted. Look for asymmetry of chest wall movement anteriorly and posteriorly. Assessment of expansion of the upper lobes is best achieved by inspection from behind the patient, looking down at the clavicles during moderate respiration (see Figure 10.7). Diminished movement indicates underlying lung disease. The affected side will show delayed or decreased movement. For assessment of lower lobe expansion, the chest should be inspected posteriorly.
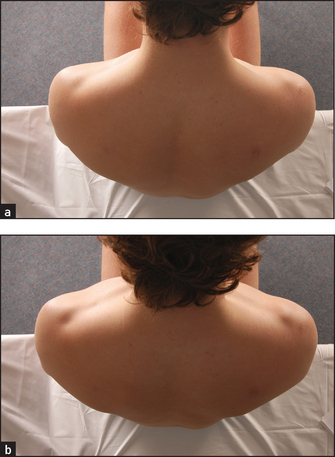
Figure 10.7 Inspecting upper lobe expansion: (a) expiration; (b) inspiration—note symmetrical elevation of the clavicles (Courtesy of Glenn McCulloch)
Reduced chest wall movement on one side may be due to localised lung fibrosis, consolidation, collapse, pleural effusion or pneumothorax.
Bilateral reduction of chest wall movement indicates a diffuse abnormality such as COPD or diffuse interstitial lung disease. Unilateral reduced chest excursion or splinting may be present when patients have pleuritic chest pain or injuries such as rib fractures.
Look for paradoxical inward motion of the abdomen during inspiration when the patient is supine (indicating diaphragmatic paralysis).
PALPATION
Chest expansion
Place your hands firmly on the patient’s chest wall with your fingers extending around the sides of the chest. Your thumbs should almost meet in the middle line and should be lifted slightly off the chest so that they are free to move with respiration (see Figure 10.8). As the patient takes a big breath in, your thumbs should move symmetrically apart at least 5 centimetres. Reduced expansion on one side indicates a lesion on that side. The causes have already been discussed.
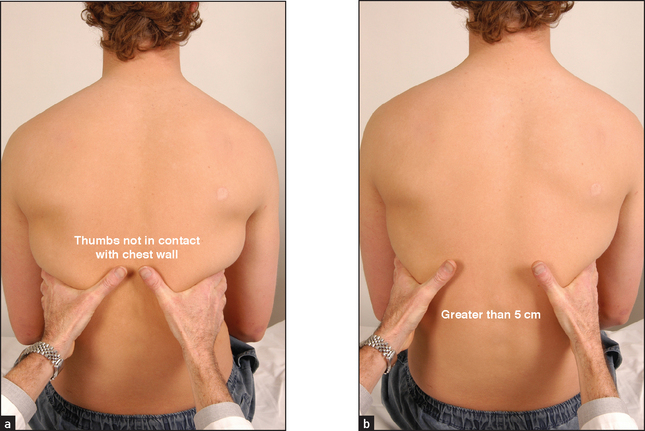
Figure 10.8 Palpation for lower lobe expansion: (a) expiration; (b) inspiration (Courtesy of Glenn McCulloch)
If COPD is suspected, Hoover’sh sign may be sought (see Figure 10.9). Place your hands along the costal margins with your thumbs close to the xiphisternum. Normally inspiration causes them to separate, but the overinflated chest of the COPD patient cannot expand in this way and the diaphragm pulls the ribs and your thumbs closer together5 (LR+ 4.2).6
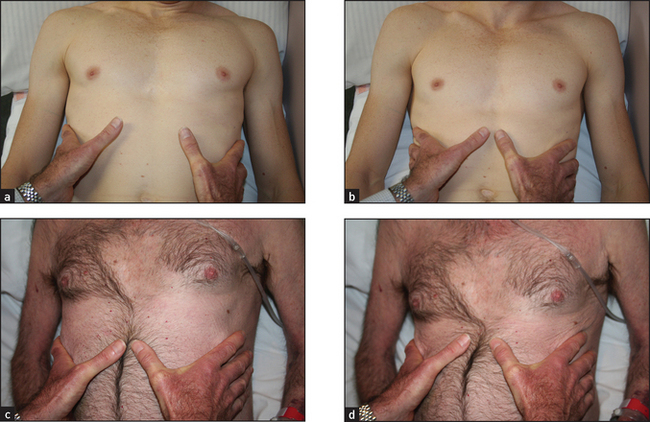
Figure 10.9 (a) Normal inspiration; (b) normal expiration; (c) Hoover’s inspiration; Hoover’s expiration (d).
Lower lobe expansion is assessed from the back in this way. Some idea of upper and middle lobe expansion is possible when the manoeuvre is repeated on the front of the chest, but this is better gauged by inspection.
Apex beat
When the patient is lying down, establishing the position of the apex beat may be helpful, as displacement towards the side of the lesion can be caused by collapse of the lower lobe or by localised interstitial lung disease. Movement of the apex beat away from the side of the lung lesion can be caused by pleural effusion or tension pneumothorax. The apex beat is often impalpable in a chest that is hyperexpanded secondary to COPD.
Vocal (tactile) fremitus
This is a palpable vibration felt by the examiner’s hands on a patient’s chest wall when the patient is speaking (or singingi). Palpate the chest wall with the palm of the hand while the patient repeats ‘ninety-nine’. The front and back of the chest are each palpated in two comparable positions, with the palm of one hand on each side of the chest. In this way differences in vibration on the chest wall can be detected. This can be a difficult sign to interpret, with considerable inter-observer variability, and it is no longer a routine part of the examination. It depends on the recognition of changes in vibration conducted to the examiner’s hands while the patient speaks. Practice is needed to appreciate the difference between normal and abnormal. Vocal fremitus is more obvious in men because of their lower-pitched voices. It may be absent in normal people (high-pitched voice or thick chest wall). It is only abnormal if different on one side from the other. The causes of change in vocal fremitus are the same as those for vocal resonance (page 150).
Ribs
Gently compress the chest wall anteroposteriorly and laterally. Localised pain suggests a rib fracture, which may be secondary to trauma or may be spontaneous as a result of tumour deposition, bone disease or sometimes the result of severe and prolonged coughing. Tenderness over the costochondral junctions suggests the diagnosis of costochondritis as the cause of chest pain.
Regional lymph nodes
The axillary and cervical and supraclavicular nodes must be examined (Chapter 21); they may be enlarged in lung malignancies and some infections.
PERCUSSION
With your left hand on the patient’s chest wall and fingers slightly separated and aligned with the patient’s ribs, press your middle finger firmly against the patient’s chest. Use the pad of your right middle finger (the plexor) to strike firmly the middle phalanx of the middle finger of your left hand (the pleximeter).j Remove the percussing finger quickly so that the note generated is not dampened (this may be less important if the pleximeter finger is held firmly on the chest wall, as it should be). The percussing finger must be held partly flexed and a loose swinging movement should come from the wrist and not from the forearm. Medical students soon learn to keep the right middle fingernail short.
Percussion of symmetrical areas of the anterior, posterior and axillary regions is necessary (see Figure 10.10). Percussion in the supraclavicular fossa over the apex of the lung and direct percussion of the clavicle with the percussing finger are a traditional part of the examination. For percussion posteriorly, the scapulae should be moved out of the way by asking the patient to move the elbows forwards across the front of the chest; this rotates the scapulae anteriorly.
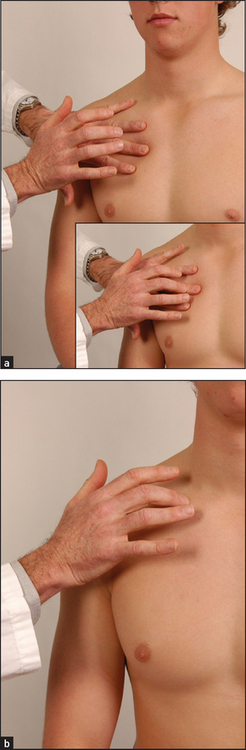
Figure 10.10 Percussion of the chest (a) Percussing (plexor) finger poised. Inset: plexor finger strikes pleximeter finger. (b) Direct percussion of the clavicle for upper lobe resonance. (Courtesy of Glenn McCulloch)
The feel of the percussion note is as important as its sound. The note is affected by the thickness of the chest wall, as well as by underlying structures. Percussion over a solid structure, such as the liver or a consolidated or collapsed area of lung, produces a dull note. Percussion over a fluid-filled area, such as a pleural effusion, produces an extremely dull (stony dull) note. Percussion over the normal lung produces a resonant note and percussion over hollow structures, such as the bowel or a pneumothorax, produces a hyperresonant note.
Considerable practice is required before expert percussion can be performed, particularly in front of an audience. The ability to percuss well is usually obvious in clinical examinations and counts in a student’s favour, as it indicates a reasonable amount of experience in the wards.
Liver dullness
The upper level of liver dullness is determined by percussing down the anterior chest in the midclavicular line. Normally, the upper level of liver dullness is the sixth intercostal space in the right midclavicular line. If the chest is resonant below this level, it is a sign of hyperinflation, usually due to emphysema or asthma. This is a sign with considerable inter-observer variability.
AUSCULTATION
Breath sounds
Using the diaphragm of the stethoscope, listen front and back (Figure 10.11).7–9 It is important to compare each side with the other. Remember to listen high up into the axillae and, using the bell of the stethoscope applied above the clavicles, to listen to the lung apices. A number of observations must be made while auscultating and, as with auscultation of the heart, different parts of the cycle must be considered. Listen for the quality of the breath sounds, the intensity of the breath sounds and the presence of additional (adventitious) sounds.
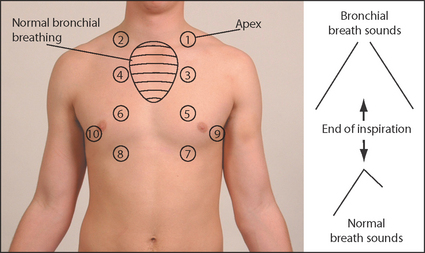
Figure 10.11 Normal and bronchial breath sounds Auscultate in each area shown (the numbers represent a suggested order). Distinguish normal breath sounds from bronchial breathing. (Courtesy of Glenn McCulloch)
Quality of breath sounds
Normal breath sounds are heard with the stethoscope over nearly all parts of the chest. The patient should be asked to breathe in and out through the mouth so that added sounds from the nasopharynx do not interfere.k During expiration these sounds are produced in the large airways rather than in the alveoli. However, inspiratory sounds are probably produced close to where they are heard, in the peripheral parts of the lungs. Since both sounds were once thought to arise in the alveoli, or vesicles of the lungs, they are called vesicular sounds. They have rather fancifully been compared by Laënnec to the sound of wind rustling in leaves. Their intensity is related to total airflow at the mouth and to regional airflow. Normal (vesicular) breath sounds are louder and longer on inspiration than on expiration and there is no gap between the inspiratory and expiratory sounds.
Bronchial breath sounds are present when turbulence in the large airways is heard without being filtered by the alveoli. Bronchial breath sounds have a hollow, blowing quality. They are audible throughout expiration and there is often a gap between inspiration and expiration. The expiratory sound has a higher intensity and pitch than the inspiratory sound. Bronchial breath sounds are more easily remembered than described. They are audible in normal people, posteriorly over the right upper chest where the trachea is contiguous with the right upper bronchus. They are heard over areas of consolidation, as solid lung conducts the sound of turbulence in main airways to peripheral areas without filtering. Causes of bronchial breath sounds are shown in List 10.3.
Occasionally, breath sounds over a large cavity have an exaggerated bronchial quality. This very hollow or amphoricl sound has been likened to that heard when air passes over the top of a hollow jar.
Intensity of the breath sounds
It is better to describe breath sounds as being of normal or reduced intensity than to speak about air entry. The entry of air into parts of the lung cannot be directly gauged from the breath sounds. Asymmetrical reduction of breath sounds is a sign of bronchial obstruction, for example by carcinoma or a foreign body on the side where breath sounds are reduced. Causes of reduced breath sounds include COPD (especially emphysema), pleural effusion, pneumothorax, pneumonia, a large neoplasm and pulmonary collapse. Breath sounds are generally louder if a patient breathes more deeply, for example after exercise.
Added (adventitious) sounds
There are two types of added sounds: continuous (wheezes) and interrupted (crackles).
Continuous sounds are called wheezes. They are abnormal findings and have a musical quality. The wheezes must be timed in relation to the respiratory cycle. They may be heard in expiration or inspiration, or both. Wheezes are due to continuous oscillation of opposing airway walls and imply significant airway narrowing. Wheezes tend to be louder on expiration. This is because the airways normally dilate during inspiration and are narrower during expiration. An inspiratory wheeze implies severe airway narrowing.
The pitch (frequency) of wheezes varies. It is determined only by the velocity of the air jet and is not related to the length of the airway. High-pitched wheezes are produced in the smaller bronchi and have a whistling quality, whereas low-pitched wheezes (sometimes called rhonchi) arise from the larger bronchi.
Wheezes are usually the result of acute or chronic airflow obstruction due to asthma (often high-pitched) or COPD (often low-pitched), secondary to a combination of bronchial muscle spasm, mucosal oedema and excessive secretions. Wheezes are a poor guide to the severity of airflow obstruction. In severe airway obstruction, wheeze can be absent because ventilation is so reduced that the velocity of the air jet is reduced below a critical level necessary to produce the sound.
A fixed bronchial obstruction, usually due to a carcinoma of the lung, tends to cause a localised wheeze, which has a single musical note (monophonic) and does not clear with coughing.
Wheezes must be distinguished from stridor (page 141), which sounds very similar to wheeze but is louder over the trachea and is always inspiratory (wheezes usually occur in expiration but can occur in both inspiration and expiration).
Interrupted non-musical sounds are best called crackles.10,11 There is a lot of confusion about the naming of these sounds, perhaps as a result of mistranslations of Laënnec. Some authors describe low-pitched crackles as râles and high-pitched ones as crepitations, but others do not make this distinction. The simplest approach is to call all these sounds crackles, but also to describe their timing and pitch. Crackles are sometimes present in normal people but these crackles will always clear with coughing.
Crackles are probably the result of loss of stability of peripheral airways that collapse on expiration. With high inspiratory pressures, there is rapid air entry into the distal airways. This causes the abrupt opening of alveoli and of small- or medium-sized bronchi containing secretions in regions of the lung deflated to residual volume. More compliant (distensible) areas open up first, followed by the increasingly stiff areas. Fine- and medium-pitched crackles are not caused by air moving through secretions as was once thought, but by the opening and closing of small airways.
The timing of crackles is of great importance. Early inspiratory crackles (which cease before the middle of inspiration) suggest disease of the small airways and are characteristic of COPD.10 The crackles are heard only in early inspiration and are of medium coarseness. They are different from those heard in left ventricular failure, which occur later in the respiratory cycle.
The timing of crackles is of great importance. Early inspiratory crackles (which cease before the middle of inspiration) suggest disease of the small airways and are characteristic of COPD.10 The crackles are heard only in early inspiration and are of medium coarseness. They are different from those heard in left ventricular failure, which occur later in the respiratory cycle.m Medium crackles are usually due to left ventricular failure. Here the presence of alveolar fluid disrupts the function of the normally secreted surfactant. Coarse crackles are characteristic of pools of retained secretions and have an unpleasant gurgling quality. They tend to change with coughing, which also has an unpleasant gurgling quality. Bronchiectasis is a common cause, but any disease that leads to retention of secretions may produce these features.
Pleural friction rub: when thickened, roughened pleural surfaces rub together as the lungs expand and contract, a continuous or intermittent grating sound may be audible. A pleural rub indicates pleurisy, which may be secondary to pulmonary infarction or pneumonia. Rarely, malignant involvement of the pleura, a spontaneous pneumothorax or pleurodynia may cause a rub.
Vocal resonance
Auscultation over the chest while a patient speaks gives further information about the lungs’ ability to transmit sounds. Over normal lung, the low-pitched components of speech are heard with a booming quality and high-pitched components are attenuated. Consolidated lung, however, tends to transmit high frequencies so that speech heard through the stethoscope takes on a bleating quality (called aegophonyn by Laënnec12). When a patient with aegophony says ‘e’ as in ‘bee’ it sounds like ‘a’ as in ‘bay’.
Increased vocal resonance is a helpful sign in confirming consolidation but may not be necessary as a routine. Ask the patient to say ‘ninety-nine’ while you listen over each part of the chest. Over consolidated lung the numbers will become clearly audible, while over normal lung the sound is muffled. If vocal resonance is present, bronchial breathing is likely to be heard (see List 10.3). Sometimes vocal resonance is increased to such an extent that whispered speech is distinctly heard; this is called whispering pectoriloquy.
If a very localised abnormality is found at auscultation, try to determine the lobe and approximately which segment or segments are involved (see Figure 10.1).
The heart
Cardiac examination is an essential part of the respiratory assessment and vice versa. These two systems are intimately related.
Lay the patient down at 45° and measure the jugular venous pressure (JVP) for evidence of right heart failure (page 94). Next examine the praecordium. It is important to pay close attention to the pulmonary component of the second heart sound (P2). This is best heard at the second intercostal space on the left. It should not be louder than the aortic component, best heard at the right second intercostal space. If the P2 is louder (and especially if it is palpable), pulmonary hypertension should be strongly suspected. There may be signs of right ventricular failure or hypertension. Pulmonary hypertensive heart disease (cor pulmonale) may be due to COPD, ILD, pulmonary thromboembolism, marked obesity, sleep apnoea or severe kyphoscoliosis.
The abdomen
Palpate the liver for ptosis,o due to emphysema, or for enlargement from secondary deposits of tumour in cases of lung carcinoma.
Other
PEMBERTON’S p SIGN
Ask the patient to lift the arms over the head and wait for one minute.13 Note the development of facial plethora, cyanosis, inspiratory stridor and non-pulsatile elevation of the JVP. This occurs in superior vena caval obstruction.
LEGS
Inspect the patient’s legs for swelling (oedema) or cyanosis, which may be clues to cor pulmonale, and look for evidence of deep venous thrombosis.
RESPIRATORY RATE ON EXERCISE
Patients complaining of dyspnoea should have their respiratory rate measured at rest, at maximal tolerated exertion (e.g. after climbing one or two flights of stairs or during a treadmill exercise test) and supine. If dyspnoea is not accompanied by tachypnoea when the patient climbs the stairs, consider the possibility of anxiety or malingering.
References
1. Mulrow, CD, Dolmatch, BL, Delong, ER, et al. Observer variability in the pulmonary examination. J Gen Intern Med. 1986; 1:364–367. [Documents the poor reliability of many respiratory signs.].
2. Conn, HO. Asterixis: its occurrence in chronic obstructive pulmonary disease, with a commentary on its general mechanism. N Engl J Med. 1958; 259:564–569.
3. Williams, JW, Jnr., Simel, DL, Roberts, LR, Samsa, GP. Clinical evaluation for sinusitis; making the diagnosis by history and physical examination. Ann Intern Med. 1992; 117:705–710. [The doctor’s impression of the likelihood of sinusitis was superior to findings of a purulent nasal discharge, history of maxillary toothache, poor response to nasal decongestants and abnormal transillumination.].
4. Model, D. Smokers’ face: an underrated clinical sign. BMJ. 1985; 291:1760–1762. [A red face with leathery skin and excessive wrinkling, associated with a gaunt look, may help identify up to half of chronic smokers.].
5. Garcia-Pachon, E. Paradoxical movement of the lateral rib margin (Hoover sign) for detecting obstructive airway disease. Chest. 2002; 12:651–655.
6. McGee, S. Evidence-based physical diagnosis, 3rd edn. St Louis: Saunders, 2012.
7. Kraman, SS. Lung sounds for the clinician. Arch Intern Med. 1986; 146:411–412. [Describes the physiological evidence for many auscultatory findings.].
8. Earis, J. Lung sounds. Thorax. 1992; 47:671–672.
9. Forgacs, P. The functional basis of lung sounds. Chest. 1978; 73:399–405.
10. Nath, AR, Caple, LH. Respiratory crackles: early and late. Thorax. 1974; 29:223–227. [Severe airways obstruction causes crackles in the first half of inspiration. In contrast, late crackles are not associated with airways obstruction.].
11. Walshaw, MJ, Nissa, M, Pearson, MG, et al. Expiratory lung crackles in patients with fibrosing alveolitis. Chest. 1990; 97:407–409. [Inspiratory crackles are usually considered more important, but this report suggests that expiratory crackles occur intermittently in fibrosing alveolitis, usually in mid-expiration.].
12. Shapira, JD. About egophony. Chest. 1995; 108:865–867.
13. Wallace, C, Siminoski, K. The Pemberton sign. Ann Intern Med. 1996; 125:568–569. [Discusses the mechanism of this useful sign, which is present when a large retrosternal goitre compresses the thoracic inlet.].
aThis condition has undergone many changes in nomenclature, and it is pleasing to think that chest doctors have something to keep them occupied. The term COPD encompasses emphysema, chronic bronchitis, chronic obstructive lung disease (COLD) and chronic airflow limitation (CAL). This term seems quite firmly established (at least for now). The diagnosis of COPD depends on clinical, radiographic and lung function assessment. There may be components of what used to be called chronic bronchitis and emphysema.
bWhen the trachea moves in time with the pulse, the sign suggests an aneurysm of the thoracic aorta—this is the original meaning of tracheal tug. Its appropriation by the chest doctors annoys those aware of its original meaning.
cThe word is derived from the Greek word sterigma, which means to support, and refers to a flapping tremor.
dJohann Horner (1831–1886), professor of ophthalmology in Zurich, described this syndrome in 1869.
eHenry Khunrath Pancoast (1875–1939), professor of roentgenology, University of Pennsylvania, described this in 1932.
fBone disease caused by vitamin D deficiency in childhood.
gEdward Harrison (1766–1838), British general practitioner in Lincolnshire, described this deformity in rickets in 1798. The sign has also been ascribed to Edwin Harrison (1779–1847), a London physician.
hCharles Hoover (1865–1927), professor of medicine in Cleveland from 1907. He also described Hoover’s test for non-organic limb weakness.
iProbably not to be encouraged during an OSCE exam.
jThis was often a piece of wood, ivory or a coin in the 19th century but is now always the examiner’s finger.
kMany people when asked to take big breaths, breathe in, and then, for some reason, stop. Although it is tempting to amuse oneself by waiting patiently for the patient to realise that expiration is also a part of breathing, this tends to waste time.
lFrom the Greek amphiphoreus meaning a ‘vase with a narrow neck’.
mExpiratory crackles may also occur with lung fibrosis.
nFrom the Greek aix meaning ‘goat’ and phone meaning ‘voice’.
oFrom the Greek word for falling, this was once mostly applied to the eyelid but now seems accepted as a description of the displacement of any organ.
pHugh Pemberton (1891–1956), physician, Liverpool, UK.