Correlation of physical signs and cardiovascular disease
Cardiac failure
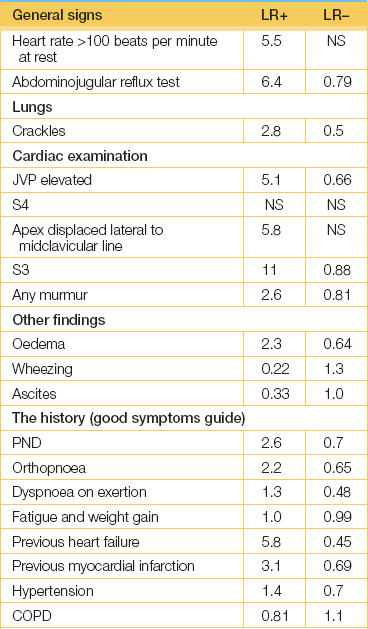
Cardiac failure is one of the most common syndromes: the signs of cardiac failure should be sought in all patients admitted to hospital, especially if there is a complaint of dyspnoea (see Questions box 9.2, page 132).1 Cardiac failure has been defined as a reduction in cardiac function such that cardiac output is reduced relative to the metabolic demands of the body and compensating mechanisms have occurred. The specific signs depend on whether the left, right or both ventricles are involved. It is important to note that the absence of definite signs of cardiac failure may not exclude the diagnosis. Patients with compensated, chronic cardiac failure may be normal on cardiac examination.
LEFT VENTRICULAR FAILURE (LVF)
• Symptoms: exertional dyspnoea, orthopnoea, paroxysmal nocturnal dyspnoea.
• General signs: tachypnoea, due to raised pulmonary pressures; central cyanosis, due to pulmonary oedema; Cheyne-Stokes breathing (see Table 9.4, page 134), especially in sedated elderly patients; peripheral cyanosis, due to low cardiac output; hypotension, due to low cardiac output; cardiac cachexia (see List 7.1).
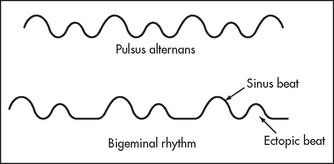
• Arterial pulse: sinus tachycardia, due to increased sympathetic tone; low pulse pressure (low cardiac output); pulsus alternans (alternate strong and weak beats; it is unlike a bigeminal rhythm caused by regular ectopic beats, in that the beats are regular; see Figure 7.1)—this is a rare but specific sign of unknown aetiology.
• Apex beat: displaced, with dilation of the left ventricle; dyskinetic in anterior myocardial infarction or dilated cardiomyopathy; palpable gallop rhythm. The absence of these signs does not exclude left ventricular failure.
• Auscultation: left ventricular S3 (an important sign); functional mitral regurgitation (secondary to valve ring dilation).
• Lung fields: signs of pulmonary congestion (basal inspiratory crackles) or pulmonary oedema (crackles and wheezes throughout the lung fields), due to raised venous pressures (increased preload). The typical middle-to-late inspiratory crackles at the lung bases may be absent in chronic, compensated heart failure, and there are many other causes of basal inspiratory crackles. This makes crackles a rather nonspecific and insensitive sign of heart failure.
• Other signs: abnormal Valsalva response; positive abdominojugular reflux test; right ventricular failure may complicate left ventricular failure, especially if this is severe and chronic.
• Signs of the underlying or precipitating cause:
– Causes of LVF: (1) myocardial disease (ischaemic heart disease, cardiomyopathy); (2) volume overload (aortic regurgitation, mitral regurgitation, patent ductus arteriosus); (3) pressure overload (systolic hypertension, aortic stenosis).
– Signs of a precipitating cause: anaemia, thyrotoxicosis (page 358), rapid arrhythmia (usually atrial fibrillation). (See Good signs guide 7.1.)
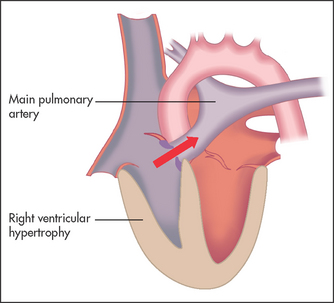
RIGHT VENTRICULAR FAILURE (RVF)
• Symptoms: ankle, sacral or abdominal swelling, anorexia, nausea.
• General signs: peripheral cyanosis, due to low cardiac output.
• Arterial pulse: low volume, due to low cardiac output.
• Jugular venous pressure: raised, due to the raised venous pressure (right heart preload); Kussmaul’s sign, due to poor right ventricular compliance (e.g. right ventricular myocardial infarction); large v waves (functional tricuspid regurgitation secondary to valve ring dilation).
• Praecordial palpation; parasternal impulse (right ventricular heave).
• Auscultation: right ventricular S3; pansystolic murmur of functional tricuspid regurgitation (absence of a murmur does not exclude tricuspid regurgitation).
• Abdomen: tender hepatomegaly, due to increased venous pressure transmitted via the hepatic veins; pulsatile liver (a useful sign), if tricuspid regurgitation is present.
• Oedema: due to sodium and water retention plus raised venous pressure; may be manifested by pitting ankle and sacral oedema, ascites or pleural effusions (small).
• Signs of the underlying cause:
– Causes of RVF: (1) chronic obstructive pulmonary disease (most common cause of cor pulmonale); (2) left ventricular failure (severe chronic LVF causes raised pulmonary pressures resulting in secondary right ventricular failure); (3) volume overload (atrial septal defect, primary tricuspid regurgitation); (4) other causes of pressure overload (pulmonary stenosis, idiopathic pulmonary hypertension); (5) myocardial disease (right ventricular myocardial infarction, cardiomyopathy).
Chest pain
Many of the causes of chest pain represent a medical (or surgical) emergency. The appropriate diagnosis or differential diagnosis is often suggested by the history, and urgent investigations (e.g. ECG, chest X-ray, lung scan or CT pulmonary angiogram) may be indicated. However, a careful and rapid physical examination may add important information in many cases. In all cases the general inspection and measurement of the vital signs will help with the assessment of the severity and urgency of the problem. Certain specific signs may help with the diagnosis.2,3
MYOCARDIAL INFARCTION
• General signs: there are few specific signs of myocardial infarction but many patients appear obviously unwell and in distress from their chest pain. Sweating (often called diaphoresis by accident and emergency staff), an appearance of anxiety (angor animi or sense of impending doom) and restlessness may be obvious. It is important that all of this information be recorded so that changes to the patient’s condition can be assessed as the infarct evolves.
• Pulse and blood pressure (BP): tachycardia and/or hypotension (25% with anterior infarction from sympathetic hyperactivity); bradycardia and/or hypotension (up to 50% with inferior infarction from parasympathetic hyperactivity). Other arrhythmias including atrial fibrillation (due to atrial infarction), ventricular tachycardia and heart block may be present.
• Jugular venous pressure (JVP): increased with right ventricular infarction; Kussmaul’s sign is a specific and sensitive sign of right ventricular infarction in patients with a recent inferior infarct.
• Apex beat: dyskinetic in patients with large anterior infarction.
• Auscultation: S4; S3; decreased intensity of heart sounds; transient apical midsystolic or late-systolic murmur (in 25% from mitral regurgitation secondary to papillary muscle dysfunction), or a pericardial friction rub (usually only occurs some days later).
• Complications: arrhythmias (ventricular tachycardia, atrial fibrillation, ventricular fibrillation or heart block); heart failure; cardiogenic shock; rupture of a papillary muscle; perforation of the ventricular septum; ventricular aneurysm; thromboembolism or cardiac rupture. Signs of these complications (which do not usually occur for a few days after the infarct) include the development of a new murmur, recurrent chest pain, dyspnoea, sudden hypotension or sudden death.
The Killipa Class can be calculated from the examination. It gives considerable prognostic information:2
| Killip Class I | No evidence of heart failure. |
| Killip Class II | Mild heart failure; crackles over lower third or less of the lungs; systolic BP > 90 mmHg. |
| Killip Class III | Pulmonary oedema, crackles more than one-third of chest; systolic BP > 90 mmHg. |
| Killip Class IV | Cardiogenic shock, pulmonary oedema, crackles more than one-third of chest, systolic BP < 90 mmHg. |
Killip Class III or IV is associated with a greater than five-fold mortality risk and Class II with a greater than three-fold risk compared with Class I.
PULMONARY EMBOLISM
There may be no physical signs of this condition, but dyspnoea (which may be profound and make the patient exhausted) is often the most obvious indication of a large pulmonary embolism. There is usually a resting tachycardia. Signs of shock—hypotension and cyanosis—indicate a very large and life-threatening embolus. There may be signs of deep vein thrombosis (DVT) in the legs but absence of these by no means excludes the diagnosis.
ACUTE AORTIC DISSECTION
Acute aortic dissection is a difficult diagnosis that cannot usually be excluded on clinical grounds. A tear in the intima leads to blood surging into the aortic media, separating the intima and adventitia; this may present acutely or chronically. There are three different types: type I begins in the ascending aorta and extends proximally and distally; type II is limited to the ascending aorta and aortic arch (this is particularly associated with Marfan’s syndrome); and type III begins distal to the left subclavian artery and has the best prognosis.
• Symptoms: chest pain (typically very severe, it radiates to the back and is maximal in intensity at the time of onset due to either the aortic tear or associated myocardial infarction); stroke; syncope (associated with tamponade); symptoms of left ventricular failure; and, rarely, limb pain (ischaemia), paraplegia (spinal cord ischaemia) or abdominal pain (mesenteric ischaemia).
• Signs: the examination can reveal signs that make the diagnosis very likely (specific but not sensitive). There may be signs of a body habitus associated with dissection (e.g. Marfan’s, Ehlers-Danlos syndromes). The pulse and blood pressure in both arms must be assessed. A diminution of the radial pulse on one side or a difference in blood pressure of 20 mmHg or more between the arms is significant and suggests dissection has progressed to involve the origin of the arm vessels. Examine the patient for signs of pericardial tamponade (page 96), which occurs if the aorta ruptures into the pericardial sac. Examine the heart for signs of aortic regurgitation caused by disruption of the aortic valve annulus. A neurological examination may reveal signs of hemiplegia, due to dissection of one of the carotid arteries. Rare signs that have been described include a pulsatile sternoclavicular joint, hoarseness (recurrent laryngeal nerve compression) and dysphagia (oesophageal compression). A tracheal tug (page 140; Oliver’s sign) may be present if there is already an aneurysm of the aortic arch.b The dilated vessel crosses the left main bronchus and pulls the trachea down during systole.
Pericardial disease
ACUTE PERICARDITIS
• Signs: fever; dyspnoea; pericardial friction rub—sit the patient up and listen to the heart with the patient holding his or her breath in deep expiration.
• Causes of acute pericarditis: (1) viral infection (coxsackie virus A or B, influenza); (2) after myocardial infarction—early, or late (10–14 days, termed Dressler’s syndromec); (3) after pericardiotomy (cardiac surgery); (4) uraemia; (5) neoplasia—tumour invasion (e.g. bronchus, breast, lymphoma) or after irradiation for tumour; (6) connective tissue disease (e.g. systemic lupus erythematosus, rheumatoid arthritis); (7) hypothyroidism; (8) other infections (e.g. tuberculosis, pyogenic pneumonia or septicaemia); (9) acute rheumatic fever.
CHRONIC CONSTRICTIVE PERICARDITIS
• Pulse and blood pressure: pulsus paradoxus (more than the normal 10 mmHg fall in the arterial pulse pressure on inspiration, because increased right ventricular filling compresses the left ventricle); low blood pressure.
• JVP: raised; Kussmaul’s sign—lack of a fall or even increased distension on inspiration (50%); prominent x and y descents (brisk collapse during diastole).
• Auscultation: heart sounds distant, early S3; early pericardial knock (rapid ventricular filling abruptly halted).
• Abdomen: hepatomegaly, due to raised venous pressure; splenomegaly, due to raised venous pressure; ascites.
• Causes of chronic constrictive pericarditis: (1) cardiac operation or trauma; (2) tuberculosis, histoplasmosis or pyogenic infection; (3) neoplastic disease; (4) mediastinal irradiation; (5) connective tissue disease (especially rheumatoid arthritis); (6) chronic renal failure.
ACUTE CARDIAC TAMPONADE
• General signs: tachypnoea; anxiety and restlessness; syncope. Patients look very ill.
• Pulse and blood pressure: rapid pulse rate; pulsus paradoxus; hypotension.
• JVP: raised; prominent x but an absent y descent.
• Auscultation: soft heart sounds.
• Lungs: dullness and bronchial breathing at the left base, due to lung compression by the distended pericardial sac.
INFECTIVE ENDOCARDITIS
• General signs: fever; weight loss; pallor (anaemia).
• Hands: splinter haemorrhages; clubbing (within six weeks of onset); Osler’s nodes (rare); Janeway lesions (very rare).
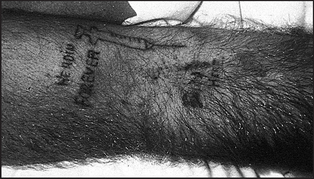
• Arms: evidence of intravenous drug use (see Figure 7.2)—right (and left) heart endocarditis can result from this.
• Eyes: pale conjunctivae (anaemia); retinal or conjunctival haemorrhages—Roth’s spotsd are fundal vasculitic lesions with a yellow centre surrounded by a red ring (see Figure 7.3).
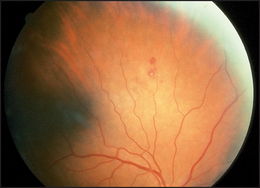
Figure 7.3 Roth’s spot on fundoscopy (Dr Chris Kennedy & Professor Ian Constable © Lion’s Eye Institute, Perth.)
• Heart—signs of underlying heart disease: (1) acquired (mitral regurgitation, mitral stenosis, aortic stenosis, aortic regurgitation); (2) congenital (patent ductus arteriosus, ventricular septal defect, coarctation of the aorta); (3) prosthetic valves.
• Peripheral evidence of embolisation to limbs or central nervous system.
• Urinalysis: haematuria (a fresh urine specimen will then show dysmorphic red cells and red cell casts on microscopy).
Systemic hypertension
It is important to have in mind a method for the examination of a patient with systemic hypertension. The examination aims to measure the blood pressure level, determine whether there is an under lying cause present and assess the severity as determined by signs of end-organ damage. It is a common clinical problem.
On general inspection the signs of the rare causes of secondary hypertension must be sought: for example, Cushing’se syndrome, acromegaly, polycythaemia (page 284) or chronic renal failure.
Take the blood pressure, with the patient lying and standing, using an appropriately sized cuff. A rise in diastolic pressure on standing occurs typically in essential hypertension; a fall on standing may suggest a secondary cause, but is usually an effect of antihypertensive medications. Palpate for radiofemoral delay, and check the blood pressure in the legs if coarctation of the aorta is suspected or if severe hypertension is discovered before 30 years of age.
Next examine the fundi for retinal changes of hypertension (see Figures 7.4 and 7.5), which can be classified from grades 1 to 4:
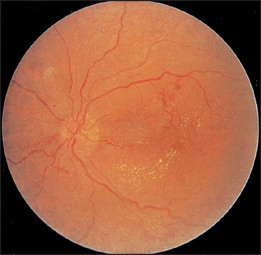
Figure 7.4 Hypertensive retinopathy grade 3. Note the flame-shaped haemorrhages and cottonwool spots. (Dr Chris Kennedy & Professor Ian Constable © Lion’s Eye Institute, Perth.)
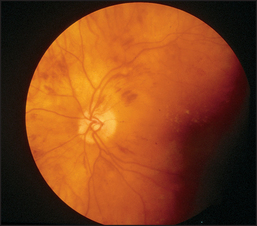
Figure 7.5 Hypertensive retinopathy grade 4. Note AV nipping, silver wiring and papilloedema. (Dr Chris Kennedy & Professor Ian Constable © Lion’s Eye Institute, Perth.)
| Grade 1 | ’Silver wiring’ of the arteries only (sclerosis of the vessel wall reduces its transparency so that the central light streak becomes broader and shinier). |
| Grade 2 | Grade 1 plus arteriovenous nipping or nicking (indentation or deflection of the veins where they are crossed by the arteries). |
| Grade 3 | Grade 2 plus haemorrhages (flame-shaped) and exudates (soft-cottonwool spots due to ischaemia; or hard—lipid residues from leaking vessels). |
| Grade 4 | Grade 3 plus papilloedema. |
It is important to describe the changes present rather than just give a grade.
Now examine the rest of the cardiovascular system for signs of left ventricular failure secondary to hypertension and for coarctation of the aorta. A fourth heart sound is frequently detectable if the blood pressure is greater than 180/110 mmHg.
Then go to the abdomen to palpate for renal or adrenal masses (possible causes) and for the presence of an abdominal aortic aneurysm (a possible complication). Auscultate for a renal bruit (page 208) due to renal artery stenosis.4 Remember that most left-sided abdominal bruits arise from the splenic artery and are of no significance. A bruit is less likely to be significant if it is short, soft and midsystolic. A loud systolic–diastolic bruit that is prominent in the epigastrium is more likely to be associated with renal artery stenosis.
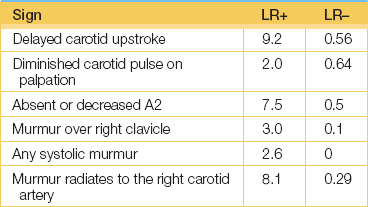
Examine the central nervous system for signs of previous cerebrovascular accidents, and palpate and auscultate the carotid arteries for bruits (however, see pages 83 and 428 for discussion of the accuracy of this examination). Stenosis may be a manifestation of vascular disease, and may be associated with renal artery stenosis. Urinalysis should also be performed to look for evidence of renal disease.
CAUSES OF SYSTEMIC HYPERTENSION
Hypertension may be essential or idiopathic (more than 95% of cases), or secondary (less than 5%). Immoderate alcohol and salt consumption and obesity (see Questions box 4.5, page 54) are associated with hypertension. Obstructive sleep apnoea is also an association.
Secondary causes include: (1) renal disease—renal artery stenosis, chronic pyelonephritis, analgesic nephropathy, connective tissue disease, glomerulonephritis, polycystic disease, diabetic nephropathy, reflux nephropathy; (2) endocrine disorders—Cushing’s syndrome, Conn’s syndrome (primary aldosteronism), phaeochromocytoma, acromegaly, thyrotoxicosis, hypothyroidism, hyperparathyroidism; (3) coarctation of the aorta; and (4) other, such as the contraceptive pill, polycythaemia rubra vera, toxaemia of pregnancy, neurogenic causes (increased intracranial pressure, lead poisoning, acute porphyria) or hypercalcaemia.
COMPLICATIONS OF HYPERTENSION
These include left ventricular failure, cerebrovascular ischaemic events (strokes), renal failure and eye disease (blindness). Hypertension is also a risk factor for ischaemic heart disease and peripheral vascular disease, including abdominal aortic aneurysm and arterial dissections.
Pulmonary hypertension
Systolic pulmonary artery pressures higher than 30 mmHg are abnormal and constitute pulmonary hypertension. Symptoms of pulmonary hypertension do not usually occur until the pressures are about twice normal (i.e. >50 mmHg). Exertional dyspnoea and fatigue are then common, and chest pain probably due to right ventricular ischaemia occurs in up to 50% of patients. It is important to know what signs to look for in a patient who may have pulmonary hypertension.
• General signs (usually only in patients with severe hypertension): tachypnoea; peripheral cyanosis and cold extremities, due to low cardiac output; hoarseness (very rare, due to pulmonary artery compression of the left recurrent laryngeal nerve).
• Pulse: usually of small volume, due to the low cardiac output (only in severe disease).
• JVP: prominent a wave, due to forceful right atrial contraction.
• Apex beat/praecordium: right ventricular heave; palpable P2.
• Auscultation: systolic ejection click, due to dilation of the pulmonary artery; loud P2,f due to forceful valve closure because of high pulmonary artery pressures; S4; pulmonary ejection murmur, due to dilation of the pulmonary artery resulting in turbulent blood flow; murmur of pulmonary regurgitation if dilation of the pulmonary artery occurs.
• Signs of right ventricular failure (late: termed cor pulmonale).
CAUSES OF PULMONARY HYPERTENSION
Pulmonary hypertension may be idiopathic (primary) or secondary. Secondary causes include: (1) pulmonary emboli—for example, blood clots, tumour particles, fat globules; (2) lung disease—COPD (page 139), obstructive sleep apnoea, interstitial lung disease (e.g. pulmonary fibrosis); (3) left ventricular failure resulting in back-pressure into the pulmonary circulation; (4) congenital heart disease causing a left-to-right shunt—atrial septal defect, ventricular septal defect, patent ductus arteriosus; and (5) severe kyphoscoliosis.
Innocent murmurs
The detection of a systolic murmur on routine examination is a common problem. It can cause considerable alarm to both the patient and the examining clinician. These murmurs in asymptomatic people are often the result of normal turbulence within the heart and great vessels. When no structural abnormality of the heart or great vessels is present these are called innocent, functional or organic murmurs. They probably arise from vibrations within the aortic arch near the origins of the head and neck vessels or from the right ventricular outflow tract. They are more common in children and young adults. They are louder just after exercise and during febrile illnesses (a common time for them to be detected).
Innocent murmurs are always systolic. (A venous hum, which is not really a murmur, has both systolic and diastolic components.) They are usually soft and ejection-systolic in character. Those arising from the aortic arch may radiate to the carotids and be heard in the neck. Those arising from the right ventricular outflow tract are loudest in the pulmonary area and may have a scratchy quality. These outflow tract murmurs must be distinguished from the pulmonary flow murmur of an atrial septal defect. Therefore. it is important to listen carefully for wide or fixed splitting of the second heart sound before pronouncing a murmur innocent (see Questions box 7.1).
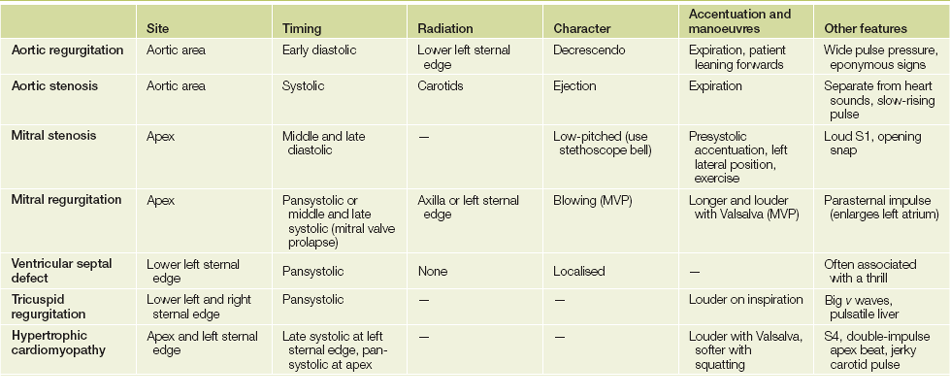
Valve diseases of the left heart (see Table 7.1)
MITRAL STENOSIS
The normal area of the mitral valve is 4 to 6 cm2. Reduction of the valve area to half normal or less causes significant obstruction to left ventricular filling, and blood will flow from the left atrium to the left ventricle only if the left atrial pressure is raised.
• Symptoms: dyspnoea, orthopnoea, paroxysmal nocturnal dyspnoea (increased left atrial pressure); haemoptysis (ruptured bronchial veins); ascites, oedema, fatigue (pulmonary hypertension).
• General signs: tachypnoea; ‘mitral facies’; peripheral cyanosis (severe mitral stenosis).
• Pulse and blood pressure: normal or reduced in volume, due to a reduced cardiac output; atrial fibrillation may be present because of left atrial enlargement.
• JVP: normal; prominent a wave if pulmonary hypertension is present; loss of the a wave if the patient is in atrial fibrillation.
• Palpation: tapping quality of the apex beat (palpable S1); right ventricular heave and palpable P2 if pulmonary hypertension is present; diastolic thrill rarely (lay patient on the left side).
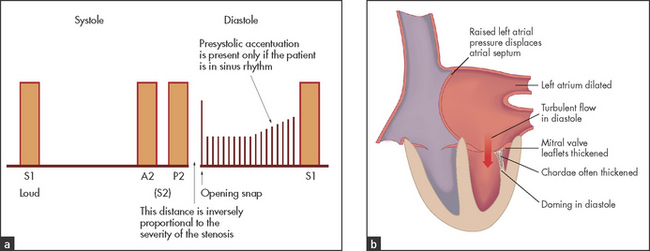
• Auscultation (see Figure 7.6): loud S1 (valve cusps widely apart at the onset of systole)—this also indicates that the valve cusps remain mobile; loud or perhaps rather palpable P2 if pulmonary hypertension is present; opening snap (high left atrial pressure forces the valve cusps apart, but the valve cone is halted abruptly); low-pitched rumbling diastolic murmur (best heard with the bell of the stethoscope with the patient in the left lateral position, and quite different in quality and timing from the murmur of aortic regurgitation); a late diastolic accentuation of the diastolic murmur may occur if the patient is in sinus rhythm, but is usually absent if atrial fibrillation has supervened—this is best heard in the left lateral position; exercise accentuates the murmur (ask the patient to sit up and down quickly in bed several times).g
• Signs indicating severe mitral stenosis (valve area less than 1 cm2): small pulse pressure; soft first heart sound (immobile valve cusps); early opening snap (due to increased left atrial pressure); long diastolic murmur (persists as long as there is a gradient); diastolic thrill at the apex; signs of pulmonary hypertension.
• Causes of mitral stenosis: (1) rheumatic (following acute rheumatic fever); (2) congenital parachute valve (all chordae insert into one papillary muscle—rare).
MITRAL REGURGITATION (CHRONIC)
A regurgitant mitral valve allows part of the left ventricular stroke volume to regurgitate into the left atrium, imposing a volume load on both the left atrium and the left ventricle.
• Symptoms: dyspnoea (increased left atrial pressure); fatigue (decreased cardiac output).
• Pulse: normal, or sharp upstroke due to rapid left ventricular decompression; atrial fibrillation is relatively common.
• Palpation: the apex beat is displaced, diffuse and hyperdynamic; a pansystolic thrill is occasionally present at the apex; a parasternal impulse may be present (due to left atrial enlargement behind the right ventricle—the left atrium is often larger in mitral regurgitation than in mitral stenosis and can be enormous).
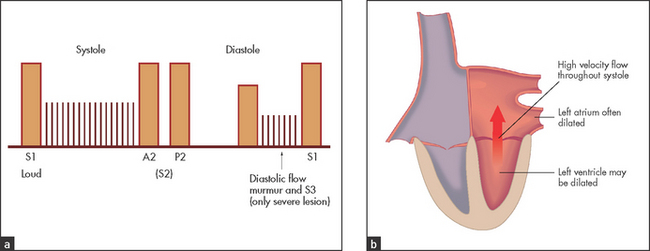
• Auscultation (see Figure 7.7): soft or absent S1 (by the end of diastole, atrial and ventricular pressures have equalised and the valve cusps have drifted back together); left ventricular S3, which is due to rapid left ventricular filling in early diastole and, when soft, does not imply severe regurgitation; pansystolic murmur maximal at the apex and usually radiating towards the axilla.
• Signs indicating severe chronic mitral regurgitation: small volume pulse; enlarged left ventricle; loud S3; soft S1; A2 is early, because rapid left ventricular decompression into the left atrium causes the aortic valve to close early; early diastolic rumble; signs of pulmonary hypertension; signs of left ventricular failure.
• Causes of chronic mitral regurgitation: (1) mitral valve prolapse; (2) ‘degenerative’—associated with ageing; (3) rheumatic; (4) papillary muscle dysfunction, due to left ventricular failure or ischaemia; (5) cardiomyopathy—hypertrophic, dilated or restrictive cardiomyopathy; (6) connective tissue disease (e.g. Marfan’s syndrome, rheumatoid arthritis, ankylosing spondylitis); (7) congenital (e.g. atrioventricular canal defect).
ACUTE MITRAL REGURGITATION
In this case patients can present with pulmonary oedema and cardiovascular collapse. The murmur may be softer and lower pitched than that of severe chronic mitral regurgitation. It tends to be short and may be decrescendo (i.e. declines in intensity towards the end of systole) because atrial pressure is increased.
With anterior leaflet chordae rupture the murmur radiates to the axilla and back; with posterior leaflet rupture the murmur radiates to the cardiac base and carotids.
MITRAL VALVE PROLAPSE (SYSTOLIC-CLICK MURMUR SYNDROME)
This syndrome can cause a systolic murmur or click, or both, at the apex. The presence of the murmur indicates that there is some mitral regurgitation present.
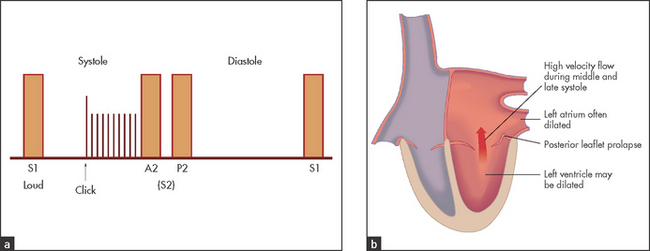
• Auscultation (see Figure 7.8): typically there is a midsystolic click followed by a middle and late systolic murmur that extends to the second heart sound. It often has a blowing quality. There may, however, be a click and no murmur (suggests little or no regurgitation) or a typical murmur without an audible click.
• Dynamic auscultation: murmur and click occur earlier and may become louder with the Valsalva manoeuvre and with standing (unlike the ejection click of aortic or pulmonary stenosis), but with squatting and isometric exercise both murmur and click occur later and may become softer.
• Causes of mitral valve prolapse: (1) myxomatous degeneration of the mitral valve tissue—it is very common, especially in women, and the severity may increase with age, particularly in men, so that significant mitral regurgitation may supervene; (2) may be associated with atrial septal defect (secundum), hypertrophic cardiomyopathy or Marfan’s syndrome.
AORTIC STENOSIS
The normal area of the aortic valve is more than 2 cm2. Significant narrowing of this valve restricts left ventricular outflow and imposes a pressure load on the left ventricle.
• Symptoms: exertional chest pain (50% do not have coronary artery disease), exertional dyspnoea and exertional syncope.
• General signs: usually there is nothing remarkable about the general appearance.
• Pulse: there may be a plateau or anacrotic pulse, or the pulse may be late peaking (tardus) and of small volume (parvus).5
• Palpation: the apex beat is hyperdynamic and may be slightly displaced; systolic thrill at the base of the heart (aortic area).
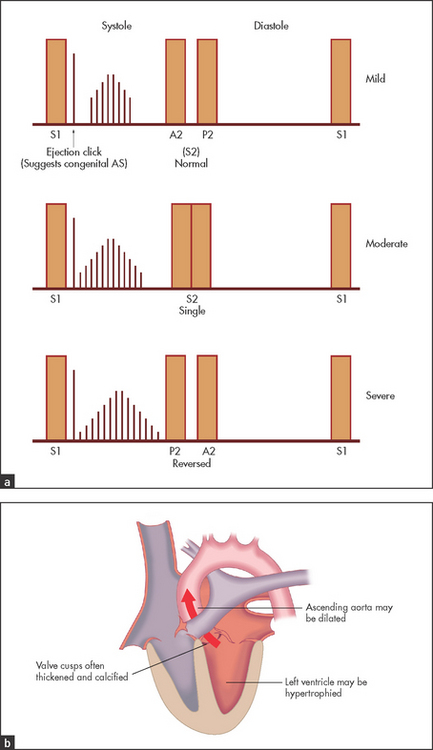
• Auscultation (see Figure 7.9): a narrowly split or reversed S2 because of delayed left ventricular ejection; a harsh midsystolic ejection murmur, maximal over the aortic area and extending into the carotid arteries (see Figure 7.10), is characteristic. However, it may be heard widely over the praecordium and extend to the apex. The murmur is loudest with the patient sitting up and in full expiration; associated aortic regurgitation is common; in congenital aortic stenosis where the valve cusps remain mobile and the dome of the valve comes to a sudden halt, an ejection click may precede the murmur—the ejection click is absent if the valve is calcified or if the stenosis is not at the valve level but above or below it (supra or subvalvular stenosis).
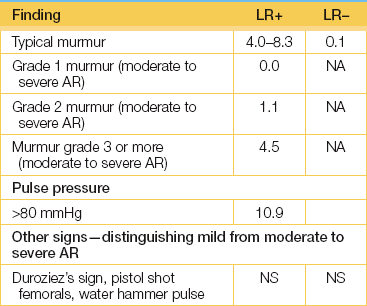
• Signs indicating severe aortic stenosis (see Good signs guide 7.2; valve area less than 1 cm2, or valve gradient greater than 50 mmHg): plateau pulse, carotid pulse reduced in force; thrill in the aortic area; length of the murmur and lateness of the peak of the systolic murmur; soft or absent A2; left ventricular failure (very late sign); pressure-loaded apex beat. These signs are not reliable for distinguishing between moderate and severe disease. It is important to remember that the signs of severity of aortic stenosis are less reliable in the elderly.6
• Causes of aortic stenosis: (1) degenerative calcific aortic stenosis, particularly in elderly patients; (2) calcific in younger patients, usually on a congenital bicuspid valve; (3) rheumatic.
• Other types of aortic outflow obstruction are also possible: (1) supravalvular obstruction, where there is narrowing of the ascending aorta or a fibrous diaphragm just above the aortic valve—this is rare and may be associated with a characteristic facies (a broad forehead, widely set eyes and a pointed chin); there is a loud A2 and often a thrill in the area of the sternal notch; (2) subvalvular obstruction, where there is a membranous diaphragm or fibrous ridge just below the aortic valve—aortic regurgitation is associated and is due to a jet lesion affecting the coronary cusp of the valve; (3) dynamic left ventricular outflow tract obstruction may occur in hypertrophic cardiomyopathy—here there may be a double apical impulse. Atrial contraction into a stiff left ventricle may be palpable before the left ventricular impulse (only in the presence of sinus rhythm, of course).
Aortic sclerosis presents in the elderly; there are none of the peripheral signs of aortic stenosis. The diagnosis implies the absence of a gradient across the aortic valve despite some thickening and a murmur.
AORTIC REGURGITATION
The incompetent aortic valve allows regurgitation of blood from the aorta to the left ventricle during diastole for as long as the aortic diastolic pressure exceeds the left ventricular diastolic pressure.7,8
• Symptoms: occur in the late stages of disease and include exertional dyspnoea, fatigue, palpitations (hyperdynamic circulation) and exertional angina.
• General signs: Marfan’s syndrome, ankylosing spondylitis or one of the other seronegative arthropathies or, rarely, Argyll Robertson pupils may be obvious.
• Pulse and blood pressure: the pulse is characteristically collapsing, a ‘water hammer’h pulse (see Table 7.2); there may be a wide pulse pressure. This sign is most obvious if the clinician raises the patient’s arm while feeling the radial pulse with the web spaces of the lifting hand. A bisferiens pulse (from the Latin, to beat twice) may be a sign of severe aortic regurgitation or of combined aortic regurgitation and aortic stenosis. It is best assessed at the carotid artery, where two beats can be felt in each cardiac cycle. It is probably caused by a Venturi effect in the aorta related to rapid ejection of blood and brief in-drawing of the aortic wall, leading to a diminution of the pulse followed by a rebound increase. It was a particular favourite of Galen’s.i
TABLE 7.2
Eponymous signs of aortic regurgitation
| Quincke’s sign | Capillary pulsation in the nail beds—it is of no value, as this sign occurs normally. |
| Corrigan’s sign | Prominent carotid pulsations; the Corrigan water hammer pulse sign is present when the patient lies supine with the arms beside the body; the radial pulse is compressed until it disappears, the arm is then lifted perpendicular to the body and the pulse becomes palpable again even though the same pressure has been maintained on the radial artery. |
| De Musset’s sign | Head nodding in time with the heartbeat. |
| Hill’s sign | Increased blood pressure (>20 mmHg) in the legs compared with the arms. |
| Mueller’s sign | Pulsation of the uvula in time with the heartbeat. |
| Duroziez’s sign | Systolic and diastolic murmurs over the femoral artery on gradual compression of the vessel, the vessel is compressed with the diaphragm of the stethoscope. A systolic murmur will always be heard. As the compression is increased a diastolic murmur will be heard in patients with significant aortic regurgitation and is due to retrograde flow of blood back towards the heart in diastole. Tilting the diaphragm towards the patient’s head will make the diastolic bruit softer if the patient has aortic regurgitation but louder if the bruit is due to an increased cardiac output (e.g. due to thyrotoxicosis). |
| Traube’s sign | A double sound heard over the femoral artery on compressing the vessel distally; this is not a ’pistol shot’ sound that may be heard over the femoral artery with very severe aortic regurgitation. |
| Mayne’s sign | A decrease in diastolic pressure of 15 mmHg when the patient’s arm is held above the head compared with that when the arm is at the level of the heart. |
| Rosenbach’s liver pulsation sign | Pulsation of the liver in time with the heartbeat (in the absence of tricuspid regurgitation). |
| Austin Flint murmur | Short rumbling diastolic murmur, thought by Flint to be due to functional mitral stenosis caused by impinging of the aortic regurgitant jet on the anterior mitral valve leaflet. |
| Becker’s sign | Accentuated retinal artery pulsations. |
| Gerhard’s sign | Pulsatile spleen. |
| Landolfi’s sign | Prominent alternating constriction and dilation of the pupils (hippus, from the Greek hippos — ’horse’—and its rhythmical galloping). |
| Lincoln’s sign (see Figure 7.11(c)) | Exaggerated movement of the ankle when one leg is crossed over the other; said to have been described by Abraham Lincoln from a photograph of himself (he did not know the cause). |
| Sherman’s sign | An easily palpable dorsalis pedis pulse in a patient over the age of 75 years. |
| Watson’s water hammer pulse | See page 105. |
| Ashrafian’s sign | Pulsatile pseudo proptosis. |
Note: These signs are amusing, but not often helpful. The signs were named after the following people: Heinrich Quincke (1842-1922), German neurologist; Dominic Corrigan (1802-80), Edinburgh graduate who worked in Dublin and is credited with discovering aortic regurgitation; Alfred de Musset, 19th-century French poet who suffered from aortic regurgitation (the sign was noticed by his brother, a physician); Sir Leonard Hill (1866-1952), English physiologist who also described the physiology of the cerebral circulation; Frederick Von Mueller (1858-1941), German physician who also noted an increase in metabolism in exophthalmic goitre; Paul Duroziez (1826-97), French physician; Ludwig Traube (1818-76), Hungarian physician who worked in Germany; Otto Heinrich Becker (1828-1890), professor of ophthalmology, University of Heidelberg, who also described this sign in patients with Graves’ disease; Lincoln’s sign is like de Musset’s sign in being named after the patient with the condition; Thomas Watson, English physician, who described this sign in 1844; Hutan Ashrafian, cardiothoracic surgeon, St Mary’s Hospital, London, who described this sign in 2006—proof that the hunt for more signs of aortic regurgitation goes on.
(Babu AN, Kymes SM, Carpenter Fryer SM. Eponyms and the diagnosis of aortic regurgitation: What says the evidence? Ann Intern Med 2003: 138:736-745.)
• Neck: prominent carotid pulsations (Corrigan’s sign).
• Palpation: the apex beat is characteristically displaced and hyperkinetic. A diastolic thrill may be felt at the left sternal edge when the patient sits up and breathes out.
• Auscultation (see Figure 7.11): A2 (the aortic component of the second heart sound) may be soft; there is a decrescendo high-pitched diastolic murmur beginning immediately after the second heart sound and extending for a variable time into diastole—it is loudest at the third and fourth left intercostal spaces; a systolic ejection murmur is usually present (due to associated aortic stenosis or to torrential flow across a normal diameter aortic valve). Aortic stenosis is distinguished from an aortic flow murmur by the presence of the peripheral signs of significant aortic stenosis, such as a plateau pulse. However, the harsher and louder the murmur (and especially if there is a thrill), the more likely it is aortic stenosis. An Austin Flint murmurj should also be listened for. This is a low-pitched rumbling mid-diastolic and presystolic murmur audible at the apex (the regurgitant jet from the aortic valve causes the anterior mitral valve leaflet to shudder). It can be distinguished from mitral stenosis because S1 (the first heart sound) is not loud and there is no opening snap. Many other signs have been described, but they are interesting rather than helpful (see Table 7.2 and Good signs guide 7.3).
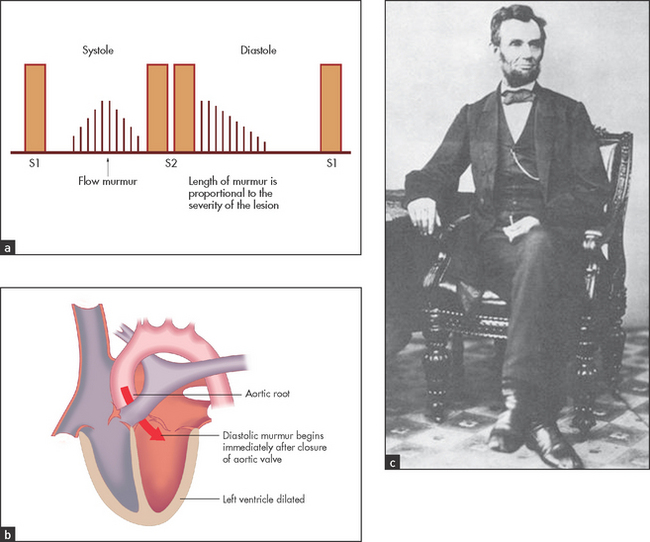
Figure 7.11 Aortic regurgitation: (a) murmur, at the left sternal edge; (b) anatomy; (c) Lincoln’s sign: left foot is blurred by motion
• Signs indicating severe chronic aortic regurgitation: collapsing pulse; wide pulse pressure (systolic pressure 80 mmHg more than the diastolic); long decrescendo diastolic murmur; left ventricular S3 (third heart sound); soft A2; Austin Flint murmur; signs of left ventricular failure.
• Causes of aortic regurgitation: disease may affect the valvular area or aortic root, and may be acute or chronic.
• Causes of chronic aortic regurgitation: (1) valvular—rheumatic (rarely the only murmur in this case), congenital (e.g. bicuspid valve; ventricular septal defect—an associated prolapse of the aortic cusp is not uncommon), seronegative arthropathy, especially ankylosing spondylitis; (2) aortic root dilation (murmur may be maximal at the right sternal border)—Marfan’s syndrome, aortitis (e.g. seronegative arthropathies, rheumatoid arthritis, tertiary syphilis), dissecting aneurysm.
• Acute aortic regurgitation: presents differently—there is no collapsing pulse (blood pressure is low) and the diastolic murmur is short.
• Causes of acute aortic regurgitation: (1) valvular—infective endocarditis; (2) aortic root—Marfan’s syndrome, dissecting aneurysm of the aortic root.
Valve diseases of the right heart
TRICUSPID STENOSIS
• JVP: raised; giant a waves with a slow y descent may be seen.
• Auscultation: a diastolic murmur audible at the left sternal edge, accentuated by inspiration, very similar to the murmur of mitral stenosis except for the site of maximal intensity and the effect of respiration (louder on inspiration); tricuspid regurgitation and mitral stenosis are often present as well; no signs of pulmonary hypertension.
• Abdomen: presystolic pulsation of the liver, caused by forceful atrial systole.
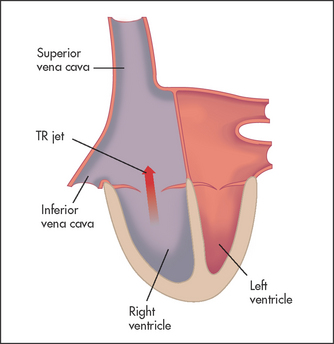
TRICUSPID REGURGITATION (See Figure 7.12)
• JVP: large v waves; the JVP is elevated if right ventricular failure has occurred.
• Palpation: right ventricular heave (parasternal impulse).
• Auscultation: there may be a pansystolic murmur maximal at the lower end of the sternum that increases on inspiration, but the diagnosis can be made on the basis of the peripheral signs alone.
• Abdomen: a pulsatile, large and tender liver is usually present and may cause the right nipple to dance in time with the heart beat; ascites, oedema and pleural effusions may also be present.
• Legs: dilated, pulsatile veins.
• Causes of tricuspid regurgitation:k (1) functional (no disease of the valve leaflets)—right ventricular failure; (2) rheumatic—only very rarely does rheumatic tricuspid regurgitation occur alone, usually mitral valve disease is also present; (3) infective endocarditis (right-sided endocarditis in intravenous drug addicts); (4) tricuspid valve prolapse; (5) right ventricular papillary muscle infarction; (6) trauma (usually caused by a steering wheel injury to the sternum); (7) congenital—Ebstein’s anomaly.l
PULMONARY STENOSIS (IN ADULTS) (See Figure 7.13)
• General signs: peripheral cyanosis, due to a low cardiac output, but only in severe cases.
• Pulse: normal or reduced if cardiac output is low.
• JVP: giant a waves because of right atrial hypertrophy; the JVP may be elevated.
• Palpation: right ventricular heave; thrill over the pulmonary area.
• Auscultation: the murmur may be preceded by an ejection click; a harsh and usually loud ejection systolic murmur, heard best in the pulmonary area and with inspiration, is typically present; right ventricular S4 may be present (due to right atrial hypertrophy). It is not well heard over the carotid arteries.
• Abdomen: presystolic pulsation of the liver may be present.
• Signs of severe pulmonary stenosis: an ejection systolic murmur peaking late in systole; absence of an ejection click (also absent when the pulmonary stenosis is infundibular i.e. below the valve level); presence of S4; signs of right ventricular failure.
• Causes of pulmonary stenosis: (1) congenital; (2) carcinoid syndrome (rare).
PULMONARY REGURGITATION
This is an uncommon pathological condition; trivial pulmonary regurgitation is often found at echocardiography and is considered physiological.
• Auscultation: a decrescendo diastolic murmur that is high-pitched and audible at the left sternal edge is characteristic—this typically but not always increases on inspiration (unlike the murmur of aortic regurgitation). It is called the Graham Steell murmurm when it occurs secondary to pulmonary artery dilation caused by pulmonary hypertension. (Note: If there are no signs of pulmonary hypertension, a decrescendo diastolic murmur at the left sternal edge is more likely to be due to aortic regurgitation than to pulmonary regurgitation.)
• Causes of pulmonary regurgitation: (1) pulmonary hypertension; (2) infective endocarditis; (3) following balloon valvotomy for pulmonary stenosis or surgery for pulmonary atresia; (4) congenital absence of the pulmonary valve.
PROSTHETIC HEART VALVES
The physical signs with common types of valves are presented in Table 7.3. Mechanical prosthetic valves should have a crisp sound. Muffling of the mechanical sounds may be a sign of thrombotic obstruction of the valve or chronic tissue ingrowth (pannus). After replacement of the aortic valve the presence of audible aortic regurgitation may indicate a paravalvular leak, often through a stitching hole in the valve sewing ring. As tissue valves age and degenerate they may develop signs of regurgitation or stenosis, or both.
TABLE 7.3
Prosthetic heart valves: physical signs
| Type | Mitral | Aortic |
| Ball valve (e.g. Starr-Edwards)* | Sharp mitral opening sound after S2, sharp closing sound at S1 Systolic ejection murmur, no diastolic murmur | Sharp aortic opening sound Systolic ejection murmur (harsh), no diastolic murmur unless a paravalvular leak has occurred, early diastolic murmur indicates AR usually due to a paravalvular leak** |
| Disc valve (e.g. Bjork-Shiley)† | Sharp closing sound at S1, soft systolic ejection murmur and diastolic rumble (diastolic murmur occasionally) | Sharp closing sound at S2, systolic ejection murmur (soft) |
| Porcine or bovine pericardial valve‡ | Usually sound normal, diastolic rumble mitral opening sound occasionally | Closing sound usually heard, systolic ejection murmur (soft), no diastolic murmur |
| Bileaflet valve (e.g. St Jude) | − | Aortic valve opening and closing sounds common, soft systolic ejection murmur common |
| Homograft (human) valve | Normal heart sounds, occasional soft systolic murmur; early diastolic murmur if AR has occurred | – |
Less often, a mitral regurgitation murmur suggests the same problem with a prosthetic mitral valve.
*Modern mechanical valves (e.g. St Jude) make softer opening and closing sounds than older valves. The Starr-Edwards valve is often very noisy and sounds like a ball rattling around in a cage (which is what it is). The valve sounds may be audible to everyone in the room when things are quiet.
**An aortic regurgitation murmur present after aortic valve replacement suggests regurgitation of the valve ring. It is not uncommon.
†Severe prosthetic dysfunction causes absence of the opening or closing sounds. Ball and cage valves cause more haemolysis than other types and make the most noise, while disc valves are more thrombogenic.
‡Bioprosthetic obstruction or patient-prosthetic mismatch causes diastolic rumbling. These valves are used less often in the mitral position because they often have a very limited life there. A degenerated bioprosthetic valve may cause murmurs of regurgitation or stenosis, or both.
(Modified from Smith ND, Raizada V, Abrams J. Auscultation of the normally functioning prosthetic valve. Ann Intern Med 1981; 95:594.)
Cardiomyopathy
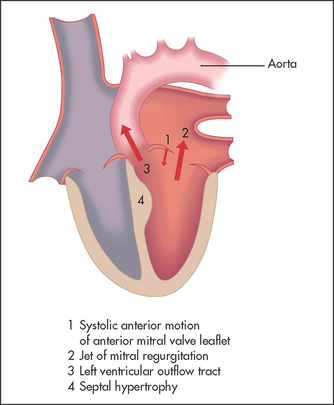
HYPERTROPHIC CARDIOMYOPATHY (See Figure 7.14)
This is abnormal hypertrophy of the muscle in the left ventricular or right ventricular outflow tract, or both. It can obstruct outflow from the left ventricle late in systole when the hypertrophied area contracts. Systolic displacement of the mitral valve apparatus into the left ventricular outflow tract also occurs, causing mitral regurgitation and contributing to the outflow obstruction. Although the outflow tract is narrowed by the hypertrophied septum, the major contribution to the dynamic increase in obstruction comes from the systolic movement of the mitral valve. Variants of hypertrophic cardiomyopathy may involve the mid-ventricle or apex with varying degrees of obstruction.
• Symptoms: dyspnoea (increased left ventricular end-diastolic pressure due to abnormal diastolic compliance), angina, syncope or sudden death (secondary to ventricular fibrillation or a sudden increase in outflow obstruction).
• Pulse: sharp rising and jerky or double (bisferiens). Rapid ejection by the hypertrophied ventricle early in systole is followed by obstruction caused by the displacement of the mitral valve into the outflow tract. This is quite different from the pulse of aortic stenosis.
• JVP: there is usually a prominent a wave, due to forceful atrial contraction against a noncompliant right ventricle.
• Palpation: double or triple apical impulse, due to presystolic expansion of the ventricle caused by atrial contraction.
• Auscultation: late systolic murmur at the lower left sternal edge and apex (due to the obstruction) and a pansystolic murmur at the apex (due to mitral regurgitation); S4.
• Dynamic manoeuvres: the outflow murmur is increased by the Valsalva manoeuvre, by standing and by isotonic exercise; it is decreased by squatting and isometric exercise.
• Causes of hypertrophic cardiomyopathy: (1) autosomal dominant (sarcomeric heavy chain or troponin gene mutation) with variable expressivity; (2) idiopathic; (3) Friedreich’s ataxian (page 475).
DILATED CARDIOMYOPATHY
This heart muscle abnormality results in a global reduction in cardiac function. Coronary artery disease is excluded as a cause by definition. (Ischaemic cardiomyopathy is a term often used to describe severe myocardial dysfunction secondary to recurrent ischaemic events.) The signs are those of congestive cardiac failure, including those of mitral and tricuspid regurgitation. The heart sounds themselves may be very quiet. Ventricular arrhythmias are common. It is a common indication for cardiac transplantation.
Acyanotic congenital heart disease
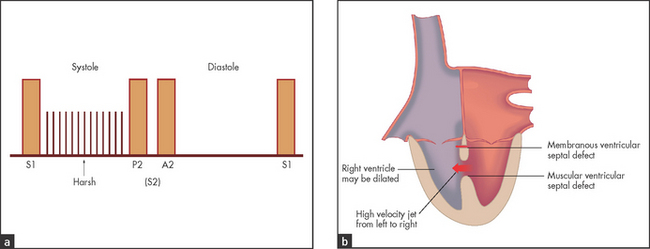
VENTRICULAR SEPTAL DEFECT
In this condition one or more holes are present in the membranous or muscular ventricular septum.
• Palpation: hyperkinetic displaced apex if the defect is large; and a thrill at the left sternal edge.
• Auscultation (see Figure 7.15): a harsh pansystolic murmur maximal at, and almost confined to, the lower left sternal edge with a third or fourth heart sound—the murmur is louder on expiration; sometimes a mitral regurgitation murmur is associated. There is often a palpable systolic thrill. The murmur is often louder and harsher when the defect is small.
• Causes of ventricular septal defect: (1) congenital; (2) acquired (e.g. myocardial infarction involving the septum).
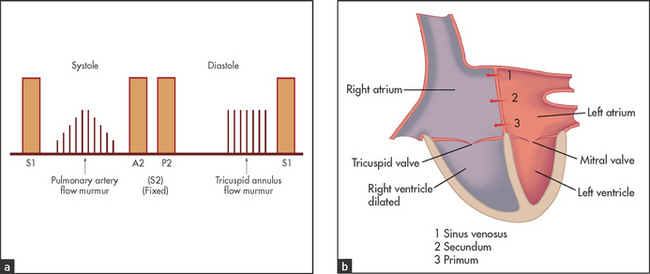
ATRIAL SEPTAL DEFECT
There are two main types: ostium secundum (90%), where there is a defect in the part of the septum that does not involve the atrioventricular valves, and ostium primum, where the defect does involve the atrioventricular valves.
• Palpation: normal or right ventricular enlargement.
• Auscultation (see Figure 7.16): fixed splitting of S2; the defect produces no murmur directly, but increased flow through the right side of the heart can produce a low-pitched diastolic tricuspid flow murmur and more often a pulmonary systolic ejection murmur—these are both louder on inspiration.
• Signs: the signs of an ostium primum defect are the same as for an ostium secundum defect, but associated mitral regurgitation, tricuspid regurgitation or a ventricular septal defect may be present. The left ventricular impulse is often impalpable.
PATENT DUCTUS ARTERIOSUS (See Figure 7.17)
This is a persistent embryonic vessel that connects the pulmonary artery and the aorta. The shunt is from the aorta to the pulmonary artery unless pulmonary hypertension has supervened.
• Pulse and blood pressure: a collapsing pulse with a sharp upstroke (due to ejection of a large volume of blood into the empty aorta with systole); low diastolic blood pressure (due to rapid decompression of the aorta).
• Palpation: often there is a hyperkinetic apex beat.
• Auscultation: if the shunt is of moderate size a single second heart sound is heard, but if the shunt is of significant size reversed splitting of the second heart sound occurs (due to a delayed A2 because of an increased volume load in the left ventricle); a continuous loud ‘machinery’ murmur maximal at the first left intercostal space is usually present; flow murmurs through the left side of the heart, including a mitral mid-diastolic murmur, may be heard.
COARCTATION OF THE AORTA (See Figure 7.18)
This is congenital narrowing of the aorta usually just distal to the origin of the left subclavian artery. It is more common in males. The underlying cause is uncertain but seems related to abnormal placement of tissue involved in the closing of the ductus arteriosus. There is an association with bicuspid aortic valve and Turner’s syndrome.
• Signs: the upper body may be better developed than the lower; radiofemoral delay is present, and the femoral pulses are weak; hypertension occurs in the arms but not in the legs; a midsystolic murmur is usually audible over the praecordium and the back, due to blood flow through collateral chest vessels and across the coarct itself.
EBSTEIN’S ANOMALY
This is a very rare lesion. The abnormality is a downward displacement of the tricuspid valve apparatus into the right ventricle so that the right atrium becomes very large and consists partly of ventricular muscle, while the right ventricle becomes small. An atrial septal defect is commonly associated. Characteristically, multiple clicks occur due to asynchronous closure of the tricuspid valve. Tricuspid regurgitation is usually present.
Cyanotic congenital heart disease
This is a difficult area. The causes of congenital heart disease are listed in List 7.2. The important point to determine is whether or not signs of pulmonary hypertension are present. Congenital heart disease in which a shunt from the left to the right side of the circulation occurs leads to an increase in pulmonary blood flow. This can cause reactive pulmonary hypertension so that pulmonary pressures eventually exceed systemic pressures. When that happens, the systemic to pulmonary (left to right) shunt will reverse. This right-to-left shunt leads to deoxygenated blood being mixed in the systemic circulation, resulting in cyanosis. This is called Eisenmenger’s syndrome.o
EISENMENGER’S SYNDROME (PULMONARY HYPERTENSION AND A RIGHT-TO-LEFT SHUNT)
It may be possible to decide at what level the shunt occurs by listening to the second heart sound (S2). If there is wide fixed splitting, this suggests an atrial septal defect. If a single second heart sound is present, this suggests truncus arteriosus or a ventricular septal defect. A normal or reversed S2 suggests a patent ductus arteriosus.
TETRALOGY OF FALLOTp
There are four features that are due to a single developmental abnormality: (1) ventricular septal defect; (2) right ventricular outflow obstruction, which determines the severity of the condition, and can be at the pulmonary valve or infundibular level; (3) an aorta that overrides the ventricular septal defect and is responsible for the cyanosis; and (4) right ventricular hypertrophy secondary to outflow obstruction.
• Signs: central cyanosis—this occurs without pulmonary hypertension because venous mixing is possible at the ventricular level, where pressures are balanced. The aorta overrides both ventricles and so receives right and left ventricular blood. Clubbing and polycythaemia are usually present. There may be evidence of right ventricular enlargement—a parasternal impulse at the left sternal edge. A systolic thrill caused by pulmonary valve or right ventricular outflow obstruction may be present. There is no overall cardiomegaly. On auscultation the second heart sound is single and there are no signs of pulmonary hypertension; a pulmonary systolic ejection murmur is present.
‘Grown-up’ congenital heart disease
Patients who have been treated for serious congenital cardiac conditions now frequently survive into adult life. Many of the surgical procedures undertaken for these conditions, especially 20 years ago, were palliative rather than curative. The patients present with specific symptoms and signs.
TETRALOGY OF FALLOT
Patients who have had repair of this condition in infancy may present with particular problems. Repair of the right ventricular outflow obstruction and enlargement of the pulmonary valve annulus may leave severe pulmonary regurgitation. This may lead eventually to exertional dyspnoea. The surgery itself has, until recently, required a right ventriculotomy (cutting into the right ventricle). This leaves a scar that can be associated with cardiac rhythm abnormalities in later life. Patients may present with palpitations or syncope.
TRANSPOSITION OF THE GREAT ARTERIES
Most adults who have had surgery for this abnormality have had a palliative operation called a Mustard procedure. In this abnormality, the pulmonary artery is connected to the left ventricle and the aorta to the right ventricle. Thus the systemic and pulmonary circulations are in parallel. This is not compatible with life unless some connection between the two circulations is present. Neonates with the condition will have an atrial septal defect created soon after birth with a catheter-based balloon (balloon septostomy). This allows mixing of the circulations. Later, ‘baffles’ are created surgically in the atria to direct blood returning from the body into the right atrium across the atrial septal defect and into the left atrium, where it is pumped into the pulmonary artery and into the lungs. Blood returning from the lungs into the left atrium is directed across into the right atrium and into the morphological right ventricle and on into the aorta. This means that the morphological right ventricle is working as the systemic ventricle. This arrangement works very well, but there are long-term concerns about the ability of the right ventricle to cope with systemic workloads.
• Symptoms: symptoms that commonly occur include palpitations caused by supraventricular arrhythmias, dizziness caused by bradycardias and breathlessness related to failure of the systemic ventricle. Occasionally, obstruction of the baffles may occur. The most common problem is with the superior vena caval baffle, which leads to facial swelling and flushing.
• Signs: include the usual scar, facial flushing and oedema, cyanosis, peripheral oedema from inferior caval baffle obstruction and signs of tricuspid regurgitation. On auscultation there may be a gallop rhythm and the murmurs of mitral and tricuspid regurgitation.
References
1. Stevenson, LW, Perluff, JK. The limited reliability of physical signs for estimating hemodynamics in chronic heart failure. JAMA. 1989; 261:884–888. [Physical signs poorly predict haemodynamic changes in heart failure. However, some signs are useful.].
2. Khot, U, Jia, G, Moliterno, DJ, et al. Prognostic value of physical examination for heart failure in non-ST elevation acute coronary syndromes. JAMA. 2003; 290:2174–2181. [This analysis of the Killip classification for patients with acute coronary syndromes expands the relevance of the classification from its original use for patients with ST elevation infarction in the pre-thrombolytic era.].
3. Klompas, M. Does this patient have an acute thoracic dissection. JAMA. 2002; 287(17):2262–2272.
4. Turnbull, JM. Is listening for abdominal bruits useful in the evaluation of hypertension? The rational clinical examination. JAMA. 1995; 274:16. [If an abdominal bruit extends into diastole, this has a high predictive value for a clinically important bruit. The pitch and intensity are not helpful.].
5. Etchells, E, Bell, C, Robb, K. Does this patient have an abnormal systolic murmur. JAMA. 1997; 277:564–571. [The most useful positive predictive features for aortic stenosis appear to be a slow rate of rise of the carotid pulse, a mid-to-late peak intensity of the murmur and a decreased second heart sound; absence of radiation to the right carotid helps rule it out.].
6. Aronow, WS. Prevalence and severity of valvular aortic stenosis determined by Doppler echocardiography, and its association with echocardiographic and electrocardiographic left ventricular hypertrophy and physical signs of aortic stenosis in elderly patients. Am J Cardiol. 1991; 67:776–777. [Analysis of the signs of severity of aortic stenosis in elderly patients shows that they are less reliable than in younger patients.].
7. Choudhry, NK, Etchells, EE. Does this patient have aortic regurgitation. JAMA. 1999; 281:2231–2238. [Rational Clinical Examination Series). It is easy if you listen carefully: the presence of an early diastolic murmur at the left sternal edge best rules in AR; the absence of an early diastolic murmur essentially rules out AR.].
8. Aronow, WS, Kronzon, I. Correlation of prevalence and severity of aortic regurgitation detected by pulsed Doppler echocardiography with the murmur of aortic regurgitation in elderly patients in a long-term health care facility. Am J Cardiol. 1989; 63:128–129.
aT. Killip, a New Zealand cardiologist, published his classification in 1967.
bAlthough described now chiefly in association with severe asthma and COPD, this sign was originally described by William Oliver (1836–1908), a Canadian military surgeon, in 1878 as a sign of a thoracic aortic aneurysm; it has been taken over by the chest doctors and paediatricians.
cWilliam Dressler (1890–1969), a New York cardiologist, described this syndrome in 1956.
dMoritz von Roth (1839–1914), Swiss physician and pathologist, described these changes in 1872.
eHarvey Cushing (1869–1939), professor of surgery, Harvard University, and ‘founder of neurosurgery’. Friend of Osler and prize-winning writer of Osler’s biography in 1925.
fThis traditional sign is not very helpful. A loud P2 is more likely to mean the patient is thin rather than there is pulmonary hypertension; a palpable P2 may be more significant.
gThis murmur is famously difficult to identify. Fewer than 10% of medical students identified the murmur on a standardised audiotape.
hThis Victorian children’s toy consisted of a sealed tube half-filled with fluid, with the other half being a vacuum. Inversion of the tube caused the fluid to fall rapidly without air resistance and strike the other end with a noise like a hammer blow. It is not easy to imagine a child today being entertained by this for very long.
iClaudius Galen (130–200 AD). Born in Pergamum, he worked as a gladiator’s surgeon but moved to Rome in 164 AD to become the city’s most famous physician. He was the first to describe the cranial nerves. He never performed dissection on human bodies, but his often erroneous anatomical teachings were regarded as infallible for 15 centuries.
jAustin Flint (1812–1886), New York physician and professor of medicine at the New Orleans Medical School, described this murmur in 1862. Author of The principles and practice of medicine. He was very much opposed to the naming of signs after people.
kDoppler echocardiography has shown that trivial tricuspid regurgitation is very common and is then considered physiological. Christian Doppler (1803–1853) was an Austrian physicist and mathematician.
lWilhelm Ebstein (1836–1912), professor of medicine at Göttingen in Germany, who invented and developed palpation.
mGraham Steell (1851–1942), Manchester physician, described this murmur in 1888.
nNikolaus Friedreich (1825–82), German physician, described this disease in 1863. He succeeded Virchow as professor of pathological anatomy at Würzburg at the age of 31.
oVictor Eisenmenger (1864–1932), German physician. He described this syndrome in 1897.
pEtienne-Louis Fallot (1850–1911), professor of hygiene, Marseilles, described this in 1888.