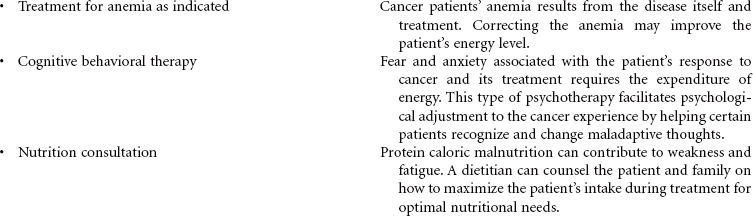Hematolymphatic, Immunological, and Oncological Care Plans
![]() For additional care plans, go to http://evolve.elsevier.com/Gulanick/.
For additional care plans, go to http://evolve.elsevier.com/Gulanick/.
Anemia
Iron Deficiency; Cobalamin (B12) Deficiency; Pernicious Anemia; Aplastic Anemia
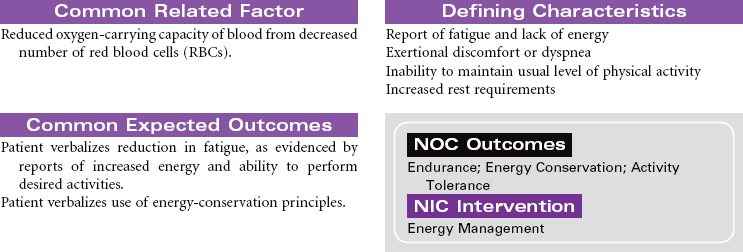
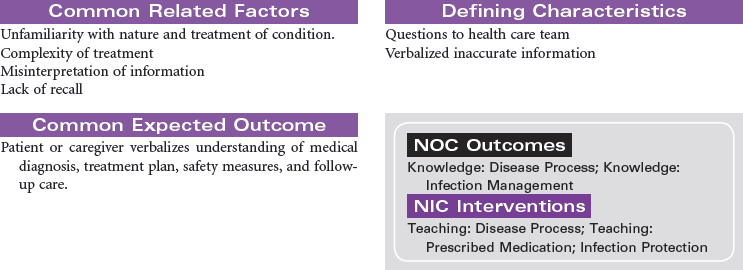
Anemia is a general diagnostic term referring to a decrease in number or derangement in function of erythrocytes (red blood cells [RBCs]), and is the most common hematological disorder. Classification of the type of anemia begins with the complete blood count, with subsequent stepwise testing providing the actual diagnosis that guides treatment and prognosis. Three anemia classification strategies exist: cytometric, which measures the RBC mass and hemoglobin concentration; erythrokinetic, which measures RBC destruction and production; and biochemical, which looks at DNA. Cytometric measurements are easily measured and begin with evaluation of the mean corpuscular hemoglobin concentration (MCHC) and mean corpuscular volume (MCV). MCHC may be normocytic or hypochromic. MCV may be normocytic, macrocytic, or microcytic. The pattern of combination of these indexes classifies the anemia, directing the next sequence of blood work to assist in diagnosis and etiology of the anemia.

Iron deficiency anemia is a hypochromic, microcytic anemia usually occurring over time, resulting from dietary deficiencies, poor absorption, chronic blood loss as seen in younger women with heavy menses or older persons with gastrointestinal (GI) loss from ulcer, use of nonsteroidal medications, or GI malignancy. Cobalamin, or B12, deficiency and pernicious anemia are both caused by deficiency of B12. Pernicious anemia impedes B12 absorption by lack of intrinsic factor in the stomach. Poor diet intake, certain medications, alcoholism, and some bowel disorders can cause cobalamin deficiency, resulting in this macrocytic anemia.
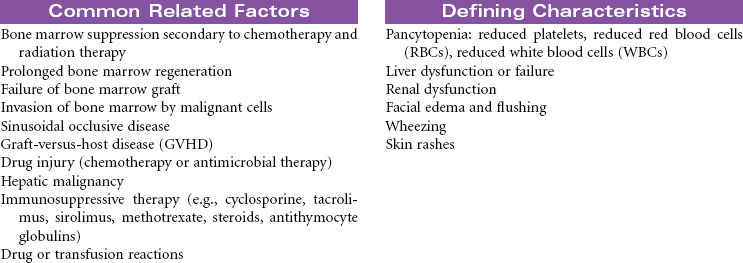
Aplastic anemia is a disease of diverse causes characterized by a decrease in precursor cells in the bone marrow and replacement of the marrow with fat. Aplastic anemia is characterized by pancytopenia, depression of all blood elements: white blood cells (leukopenia), RBCs (anemia), and platelets (thrombocytopenia). The underlying cause of aplastic anemia remains unknown. Possible pathophysiological mechanisms include certain infections, toxic dosages of chemicals and drugs, radiation damage, and impairment of cellular interactions necessary to sustain hematopoiesis. Advances in bone marrow transplantation and immunosuppressive therapy have significantly improved outcomes. This care plan focuses on ongoing care in the ambulatory care setting.
Cancer Chemotherapy
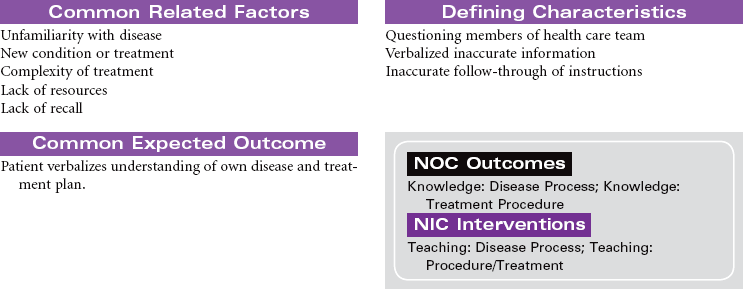
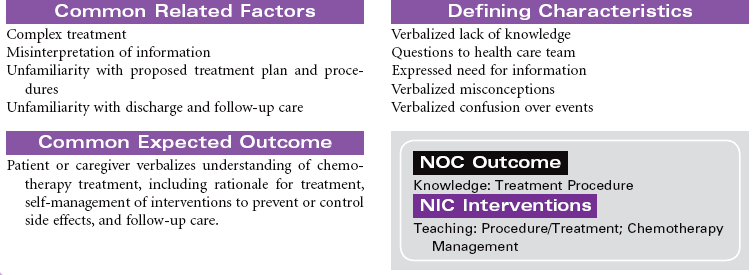
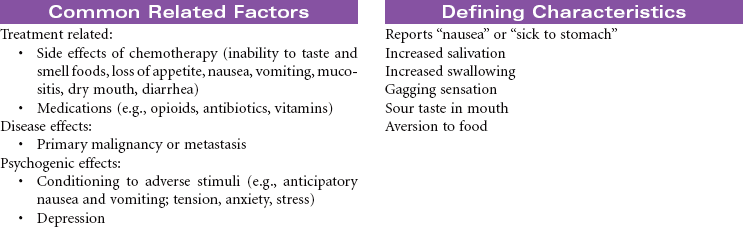
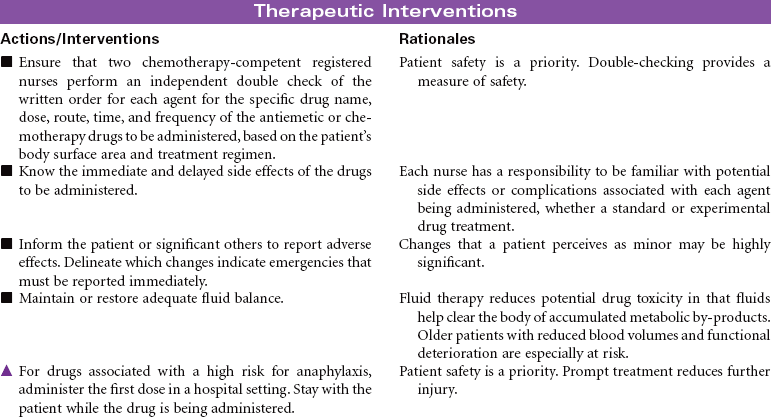
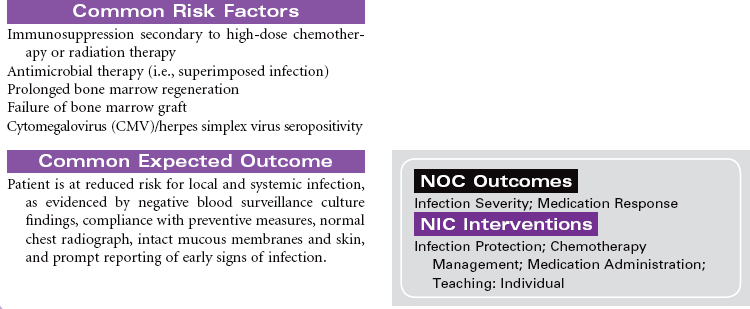
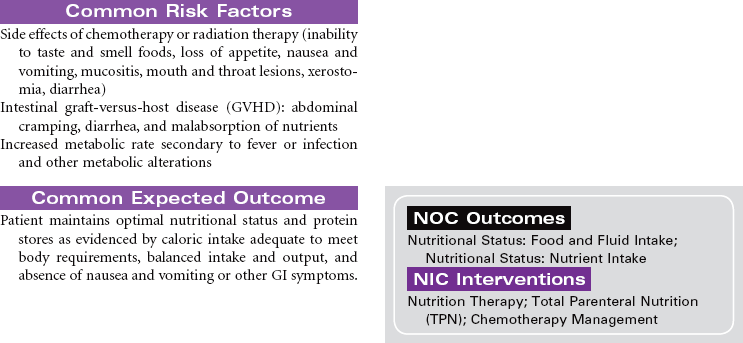
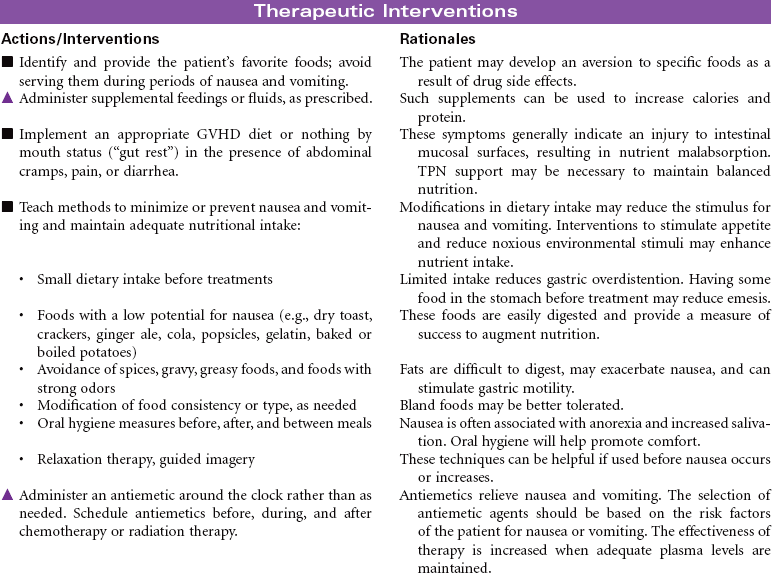
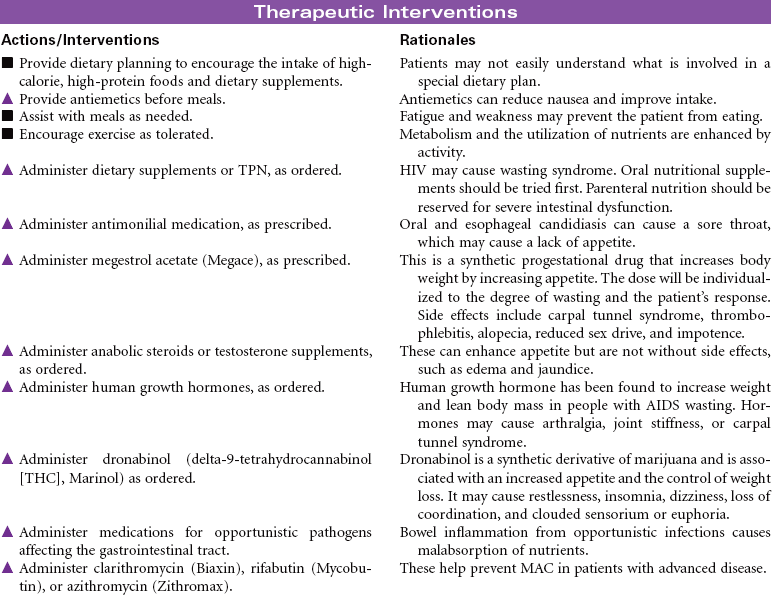
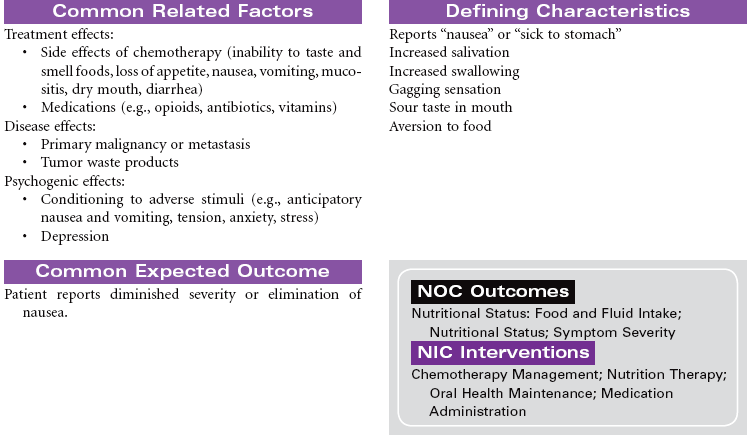
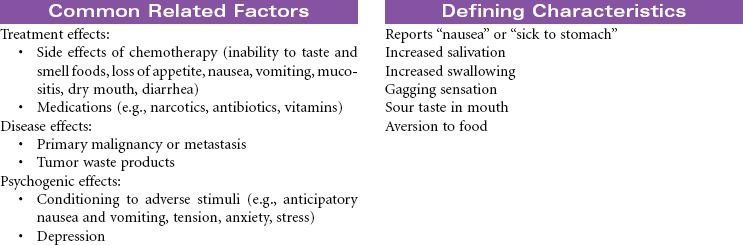
Cancer chemotherapy is the administration of cytotoxic drugs by various routes for the purpose of destroying malignant cells. Chemotherapeutic drugs are commonly classified according to their antineoplastic action: alkylating agents, antitumor antibiotics, antimetabolites, vinca alkaloids, and hormonal agents. Another way of classifying cancer chemotherapeutic agents is based on where in the cancer cell’s life cycle the drug has its effect. Cell cycle–specific drugs exert their cytotoxic effect at a specific point in the cell cycle. Drugs that affect the cancer cell at any point in its cycle are called cell cycle–nonspecific drugs. These drugs are dose dependent in their therapeutic effect. Typically a combination of chemotherapeutic agents is administered to destroy the greatest number of tumor cells at different stages of cell replication. Cancer chemotherapy may be administered in the hospital, ambulatory care, or even home setting. It is recommended by the Oncology Nursing Society that chemotherapy, biotherapy, and targeted therapies be administered by a qualified chemotherapy-competent nurse. Depending on the specific cancer, cell type, cellular mutations, and stage of disease, newer targeted therapies or biotherapies may be administered along with chemotherapy. The goal of systemic treatment is cure, control, or symptom relief. It is often used as an adjunct to surgery and radiation. Because chemotherapy drugs are highly toxic and are given systemically, they affect normal cells as well as cancer cells. Most of the side effects of cancer chemotherapy are the result of the drugs’ effects on rapidly dividing normal cells in the hair follicles, the gastrointestinal tract, and the bone marrow. The Oncology Nursing Society has developed evidence-based resources for patients experiencing chemotherapy-related side effects.
Cancer Radiation Therapy
External Beam; Brachytherapy; Teletherapy
Radiation therapy is the use of ionizing radiation delivered in prescribed doses to a malignancy. Ionizing radiation interacts with the atoms and molecules of malignant cells, interfering with mitotic activity, thereby causing DNA damage. This damage interferes with the malignant cell’s ability to reproduce. Adjacent healthy cells experience the same detrimental effects, however, resulting in untoward side effects from radiation therapy. Radiation therapy may be curative of some cancers, or it may be used as a palliative treatment to reduce the pain and pressure from large tumors. Radiation may be used alone or in combination with other treatment modalities such as surgery, chemotherapy, and/or biotherapy.
Radiation therapy can be divided into two broad categories: external radiation, also known as teletherapy, and internal radiation, commonly known as brachytherapy. Teletherapy administers a prescribed dosage of radiation at a distance from the patient using a machine, such as a linear accelerator. Brachytherapy is the implantation of either sealed (solid) or unsealed (fluid) radioactive sources. The sealed radioactive implant may be contained within an applicator, needle, or seed, and is placed in or near the malignancy. The unsealed radioactive isotope can be administered through the intravenous or oral route or by instillation into a specific body cavity.
The radiation oncologist prescribes the treatment modality and amount of treatment necessary. This treatment plan is based on the location, size, and biological characteristics of the malignancy. The patient’s health history, current health status, and previous cancer treatments are taken into consideration in treatment planning. All health care providers need to implement principles of radiation safety when caring for patients undergoing radiation therapy.
Disseminated Intravascular Coagulation
Coagulopathy; Defibrination Syndrome; DIC
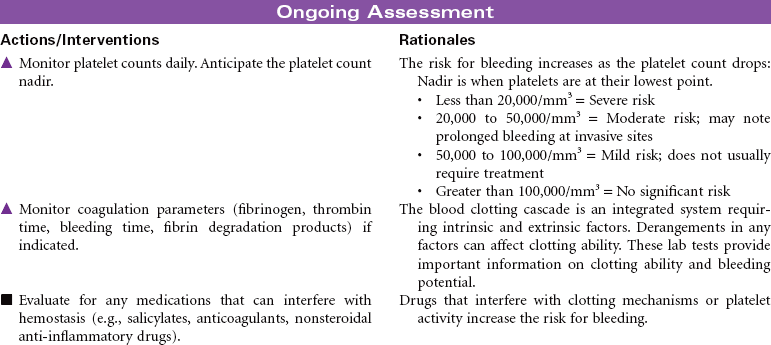
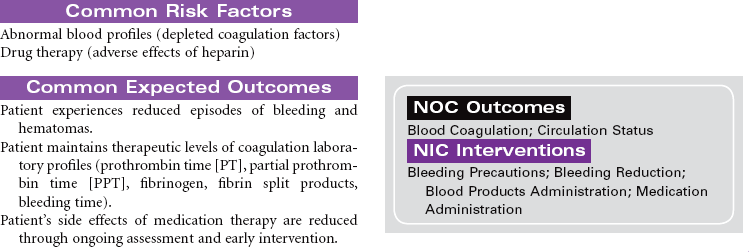
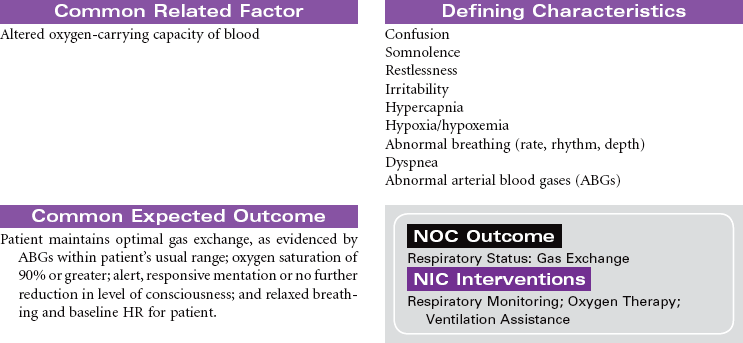
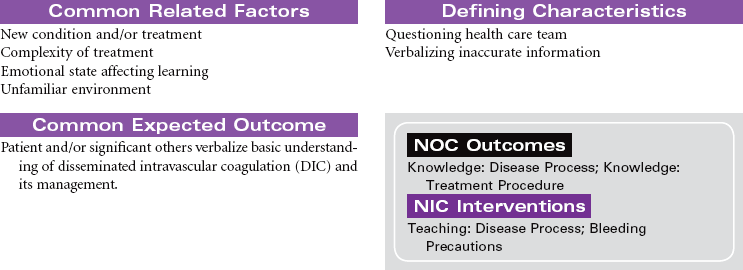
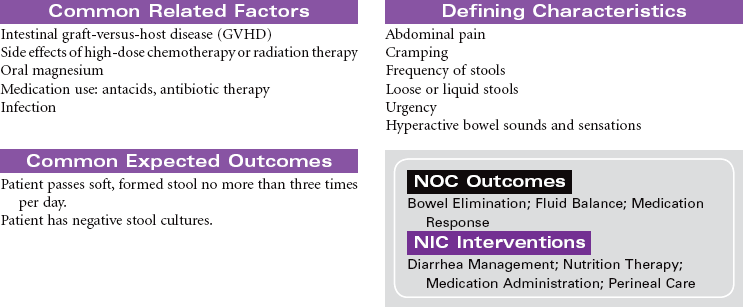
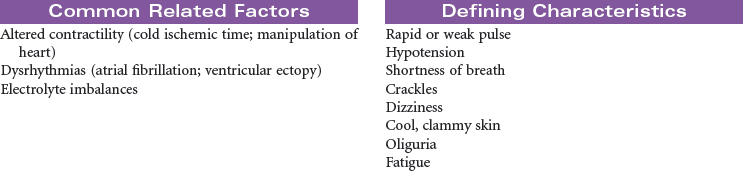
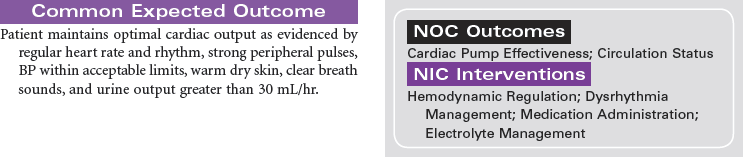
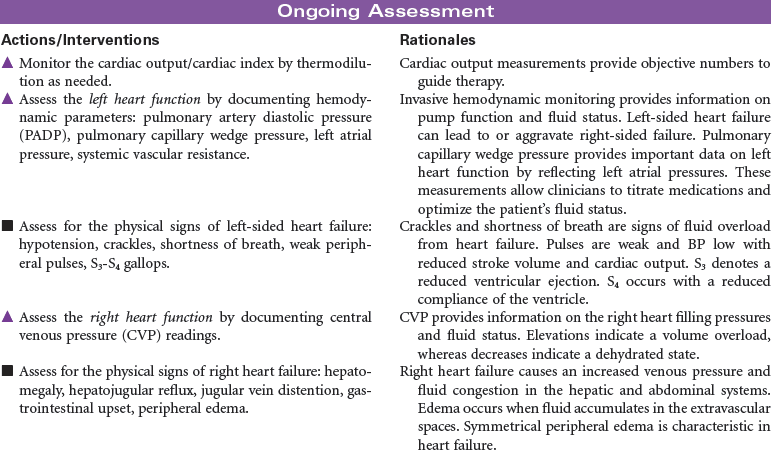
Disseminated intravascular coagulation (DIC) is a coagulation disorder that prompts overstimulation of the normal clotting cascade and results in simultaneous thrombosis and hemorrhage. The formation of microclots affects tissue perfusion in the major organs, causing hypoxia, ischemia, and tissue damage. Coagulation occurs in two different pathways: intrinsic and extrinsic. These pathways are responsible for formation of fibrin clots and blood clotting, which maintains hemostasis. In the intrinsic pathway, endothelial cell damage commonly occurs because of sepsis or infection. The extrinsic pathway is initiated by tissue injury such as from malignancy, trauma, or obstetrical complications. DIC may present as an acute or chronic condition. The medical management of DIC is primarily aimed at: (1) treating the underlying cause, (2) managing complications from both primary and secondary causes, (3) supporting organ function, and (4) stopping abnormal coagulation and controlling bleeding. Morbidity and mortality depend on underlying cause and severity of the coagulopathy.
Hematopoietic Stem Cell Transplantation
Bone Marrow Transplant; Peripheral Blood Stem Cell Transplant
Bone marrow transplantation (BMT) and peripheral blood stem cell transplantation (PBSCT) are terms that now more commonly fall under the umbrella term of hematopoietic stem cell transplantation (HSCT). The indications for HSCT are expanding; it is used as both a curative and investigational treatment for both malignant and nonmalignant conditions. HSCT should not be confused with the controversial field of embryonic stem cells. Embryonic stem cells, derived from fertilized embryos, are undifferentiated cells that have the ability to form any adult cell. Hematopoietic stem cells are the “mother” cells that differentiate only into the cells of the blood system (e.g., white blood cells [WBCs], red blood cells [RBCs], platelets).
HSCT is used to replace diseased bone marrow, as a hematopoietic rescue after high-dose therapy (radiation or chemotherapy), as a form of immunotherapy, and as a vehicle for gene therapy.
There are three major types of transplants:
• Syngeneic: Donor from an identical twin
• Allogeneic: Can be related (from a matched sibling) or unrelated (from a volunteer in the Be The Match Registry). This is also referred to as a matched unrelated donor (MUD) transplant.
There are three sources of hematopoietic stem cells:
• Peripheral blood: The stem cells that normally reside in the bone marrow can be moved or mobilized into the bloodstream (peripheral circulation) and collected in an outpatient procedure via a cell separator or apheresis machine. This procedure does not require anesthesia. The majority of all transplants performed today use peripheral blood stem cells rather than bone marrow stem cells.
• Bone marrow: These cells are collected from the pelvic bones through a series of aspirations. Bone marrow harvesting is a surgical procedure done under general anesthesia.
• Umbilical cord, placental: This is a rich source of stem cells that are collected at the time of delivery from tissue that is normally discarded.
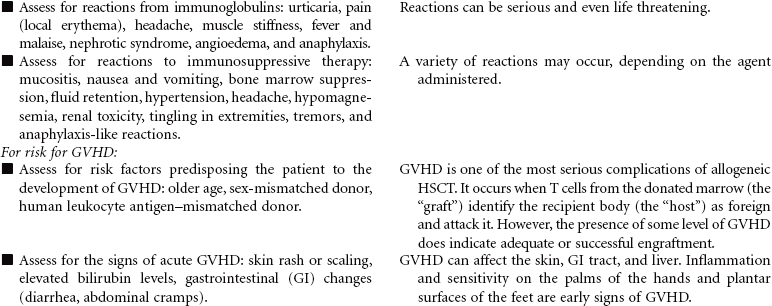
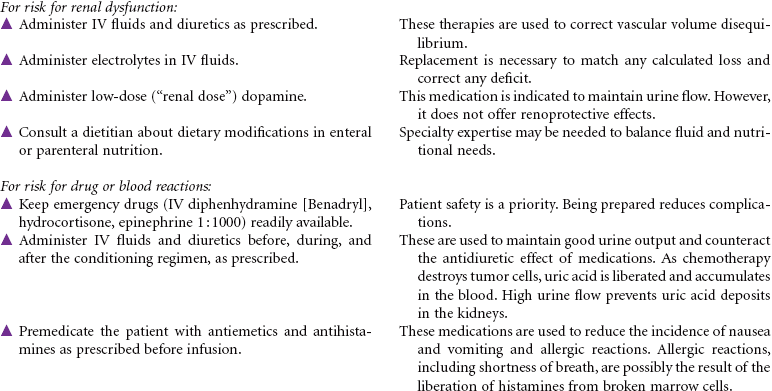
There is one other classification of transplant based on the amount and type of pretransplant therapy that is administered. Standard transplants use strong treatment (chemotherapy and/or radiation therapy) administered before transplantation to destroy the host’s diseased cells and suppress the host’s immune system. This therapy is referred to as ablative therapy, because it eliminates all host blood and immune cells. Reduced-intensity transplants—also called nonmyeloablative transplants or minitransplants—are transplants that use less intense treatment to prepare for transplantation than a standard transplant does. Thus the doses of chemotherapy given before transplantation are much lower and do not necessarily eliminate all diseased cells. This type of transplant is only used in the allogeneic setting, because this method relies on the donor’s immune cells to fight disease. This care plan focuses on inpatient care. Emotional issues related to HSCT are not addressed here.
Human Immunodeficiency Virus
Acquired Immunodeficiency Syndrome (AIDS)
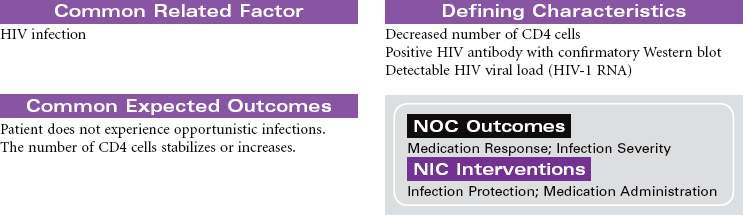
Human immunodeficiency virus (HIV) causes acquired immunodeficiency syndrome (AIDS). Transmission of HIV occurs in situations that allow contact with body fluids that are infected with the virus. The primary body fluids associated with transmission are blood, vaginal secretions, semen, and breast milk. Transmission of HIV can occur during sexual intercourse with an infected partner. Transmission through blood and blood product administration occurred early in the history of HIV in the United States. With current methods for screening blood donors and testing donated blood before transfusion, this is no longer considered a route of infection transmission. However, contact with infected blood through shared IV equipment and accidental needle sticks is still possible. Perinatal transmission of the virus from mother to baby is thought to occur during pregnancy, during delivery, or through breast-feeding. Most of the early victims of the syndrome were homosexual men; however, in many cities today, infected IV drug users, their sexual partners, and their children outnumber infected homosexual men. Despite efforts to increase routine, voluntary testing and counseling for HIV, many patients first learn that they are infected after their disease is advanced.
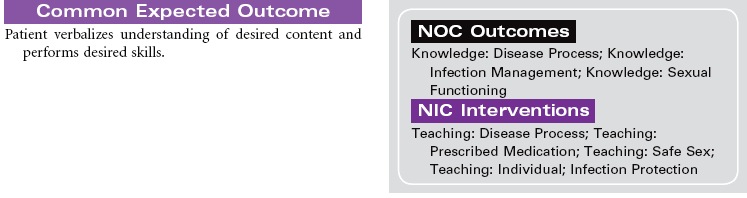
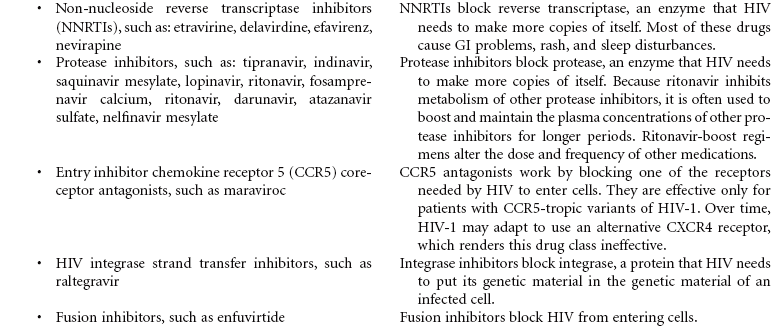
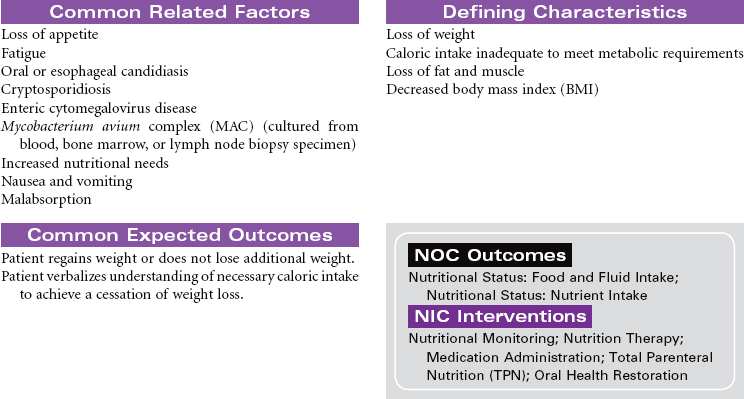
The first signs of HIV infection occur when the body produces HIV antibodies. Flulike signs and symptoms that may last 1 to 2 weeks characterize this stage of the infection. After this stage, the patient may be asymptomatic for acute infection, depending on his or her general state of health. This asymptomatic stage can last 10 years or longer. When the immune system begins to fail, the patient exhibits signs of immune system incompetence. The patient begins to develop clinical conditions such as cancers and opportunistic infections. When the patient’s CD4 lymphocyte count falls below 200, AIDS is diagnosed. Patients present at various stages of the disease. Treatment regimens are changing rapidly. Patients are treated in hospital, ambulatory care, and home care settings. People may receive prophylactic antiretroviral therapy following high-risk, unprotected sex or injection drug exposures. The nursing diagnosis list of problems for various stages of HIV infection and AIDS is extensive. Some are highlighted here.
Leukemia
Acute Lymphocytic Leukemia; Acute Myelocytic Leukemia; Chronic Lymphocytic Leukemia; Lymphocytic Leukemia; Chronic Myelocytic Leukemia; Nonlymphocytic Leukemia; Myelogenous Leukemia; Granulocytic Leukemia
Leukemia is a malignant disorder of the blood-forming system, including the bone marrow and spleen. The proliferation of immature white blood cells (WBCs) interferes with the production and function of the red blood cells (RBCs) and platelets. Leukemia can be characterized by identification of the type of leukocyte involved: myelogenous or lymphocytic. In acute lymphocytic leukemia there is a proliferation of lymphoblasts (most commonly seen in children); in acute myelocytic leukemia (most common after 60 years of age), there is a proliferation of myeloblasts. In chronic lymphocytic leukemia, there are increased lymphocytes (more common in men, especially after 50 years of age); in chronic myelocytic leukemia, granulocytes are increased (common in middle age).
Depending on the type of leukemia, therapeutic management may consist of combined chemotherapeutic agents, radiation therapy, and/or stem cell transplantation. Chemotherapeutic treatment consists of several stages: induction therapy, intensification, consolidation therapy, and maintenance therapy. The goals of nursing care are to prevent complications and provide educational and emotional support. This care plan addresses ongoing care of a patient in an ambulatory setting receiving maintenance therapy.
Lymphoma: Hodgkin Lymphoma; Non-Hodgkin Lymphoma
Lymphoma is a malignant disorder of the lymph nodes, spleen, and other lymphoid tissue. Lymphomas include related diseases with a variety of symptoms, treatment options, and outcomes depending on the lymphocyte type and stage of disease. Lymphomas are classified as either Hodgkin lymphoma or non-Hodgkin lymphoma. A specific cause has not been identified, although associations with viral disease such as Epstein-Barr and mononucleosis and environmental exposure to toxins have been noted. The Centers for Disease Control and Prevention has included lymphoma in the list of clinical conditions that are part of the case definition for AIDS.
Hodgkin lymphoma is a disorder of the lymph nodes, usually presenting with node enlargement. It is seen more frequently in men than women, first between the ages of 20 and 40, and then again after 55 years of age. Non-Hodgkin lymphoma is a disorder of the lymphocytes that involves many different histological variations. It is seen more frequently in middle-age men.
Depending on the type of lymphoma, therapeutic management may consist of combination chemotherapy, radiation therapy, and/or stem cell transplantation. The prognosis is usually poorer for non-Hodgkin lymphoma because of its later stage at diagnosis.
The goals of nursing care are to provide educational and emotional support and to prevent complications. This care plan addresses ongoing care of a patient in an ambulatory setting receiving maintenance therapy.
Multiple Myeloma
Myelomatosis; Plasma Cell Myeloma; Kahler Disease
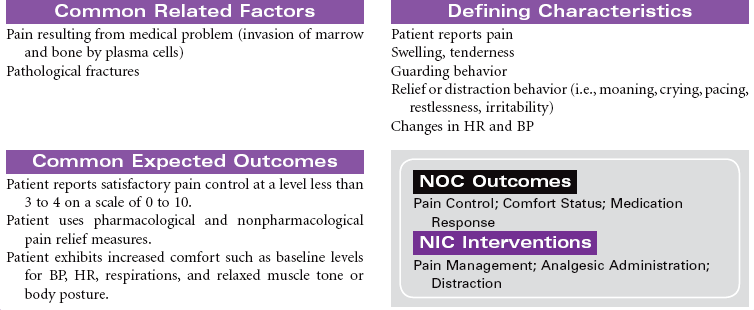
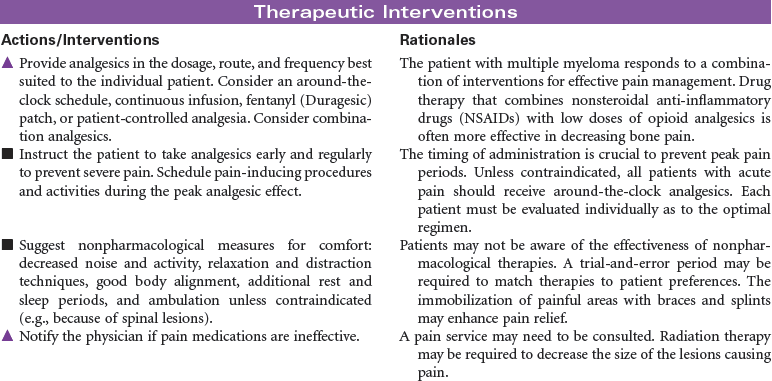
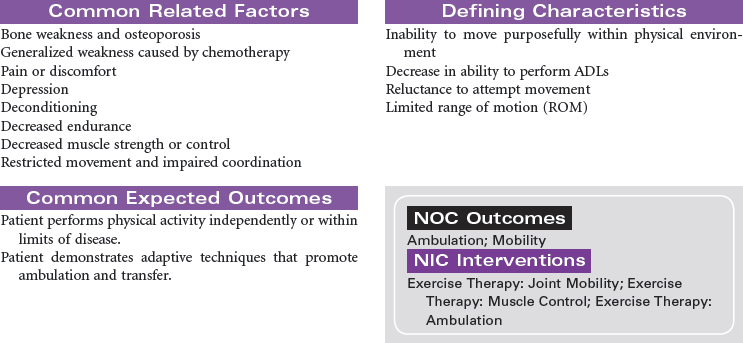
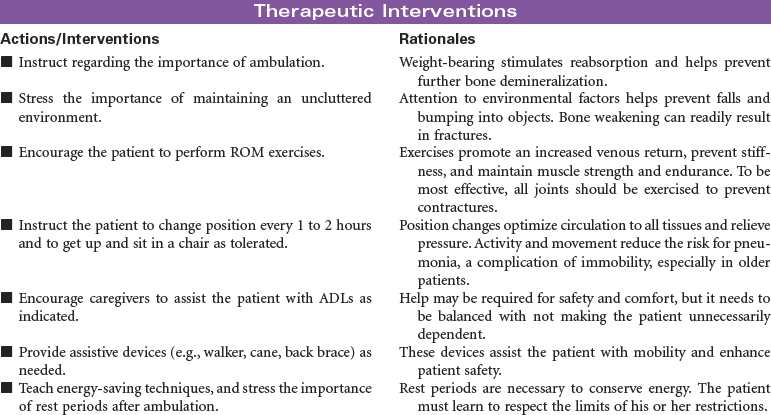
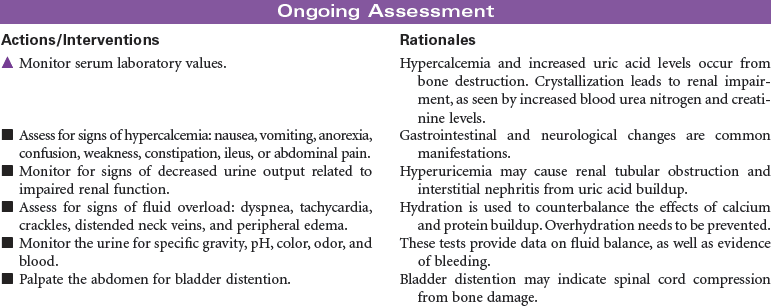
Multiple myeloma is the second most common hematological malignancy. It is a plasma B-cell malignancy that is characterized by the overproduction of immunoglobulins. This pathophysiology results in disruption of normal red blood cells, leukocytes, and platelets, resulting in anemia, infection, and bleeding problems. The cause of multiple myeloma is unknown, with no clear risk factors identified beyond age. There are no specific symptoms with multiple myeloma. Clinically, it may present itself as destruction of bone, infiltration of bone marrow, the presence of immunoglobulins in the urine or serum, recurrent infections, pain, fatigue, or symptoms of renal failure. This disease affects older adults, with a median age at diagnosis of 62 years. It is more common in African Americans than in Caucasian and in men than in women.
Neutropenia
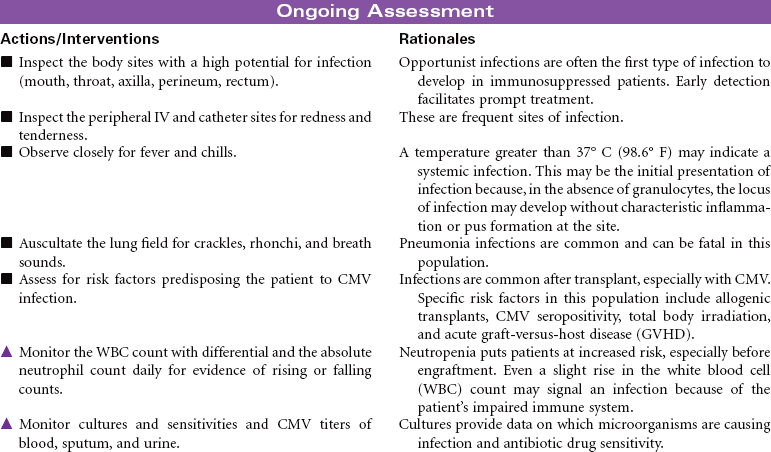
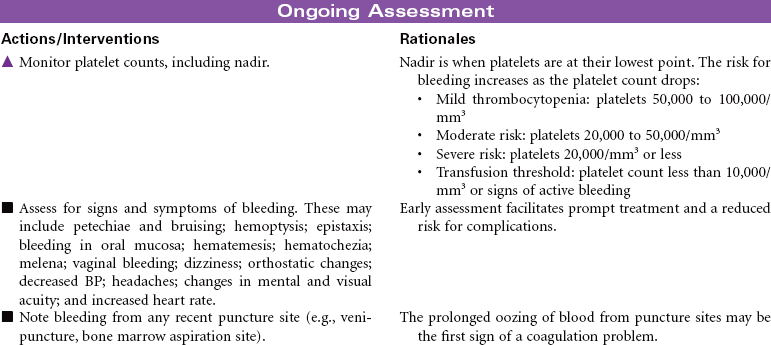
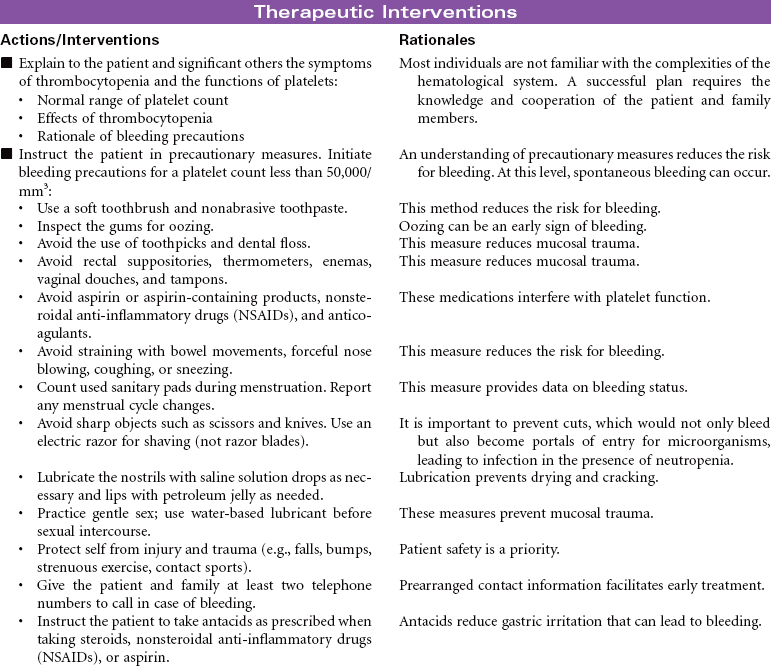
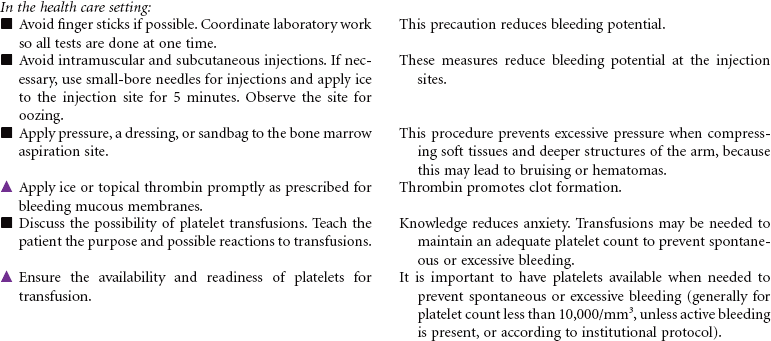
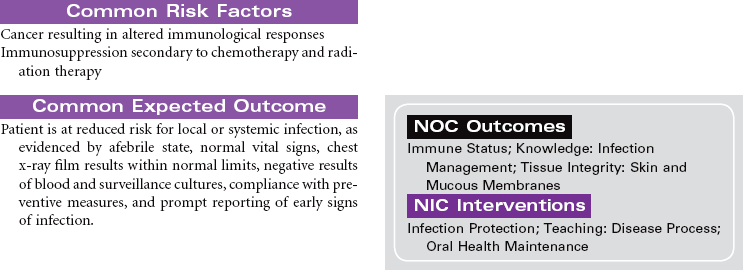
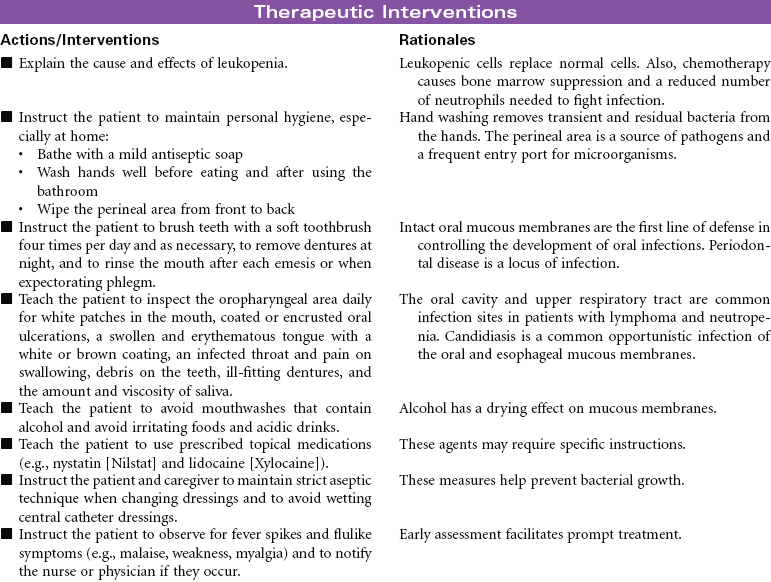
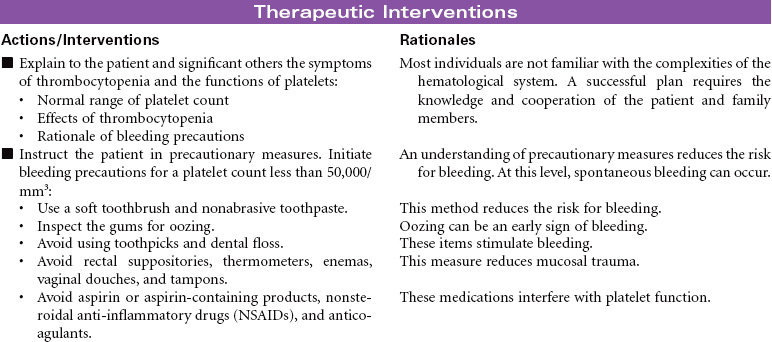
Neutropenia is a deficiency in granulocytes, a type of white blood cell (WBC). There are three types of granulocytes: basophils, eosinophils, and neutrophils. Neutropenia and its complications center around the neutrophilic granulocyte. Neutropenia is a below-normal number of circulating neutrophils that may result in overwhelming, potentially life-threatening infection. Neutrophils constitute 45% to 75% of all WBCs. Their primary function is phagocytosis, the digestion and subsequent destruction of microorganisms; as such, they are one of the body’s most powerful first lines of defense against infection. The chance of developing a serious infection is related not only to the absolute level of circulating neutrophils but also to the length of time the patient is neutropenic. Prolonged duration predisposes the patient to a higher risk for infection. Neutropenia not only predisposes one to infection but also causes it to be more severe when an infection occurs. Neutropenia is usually associated with another medical condition being treated. It can be acute or chronic. Primary diagnosis is by complete blood cell count and bone marrow aspiration. Treatment depends on cause and severity. This care plan focuses on outpatient management.
Organ Transplantation, Solid
Heart Transplantation; Lung Transplantation; Liver Transplantation; Kidney Transplantation; Living Kidney Donors
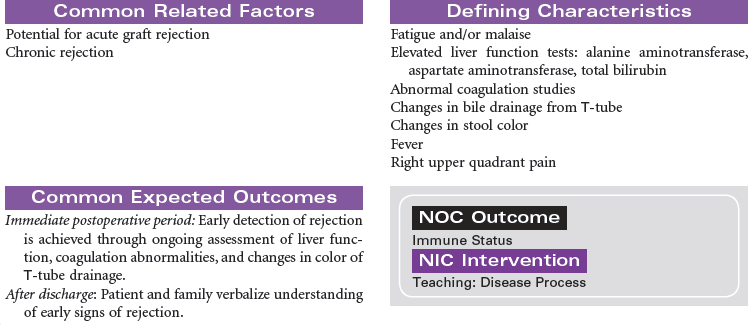
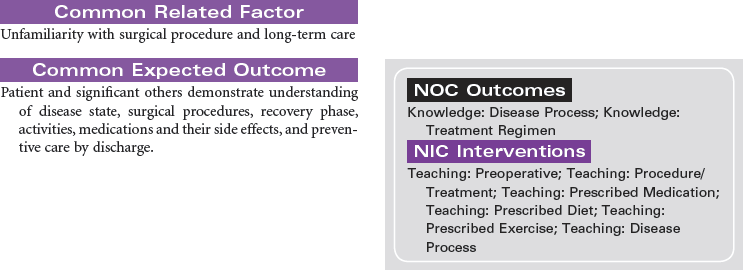
Solid organ transplantation is a treatment option for patients with end organ damage that has not responded to optimal medical therapy or alternative surgical strategies. Because of a limited supply of suitable organs for transplantation, candidates are carefully screened to ensure they meet selection criteria established by each transplant program. Comorbid conditions such as diabetes, vascular diseases, and malignancies are carefully evaluated to determine the added risk potential to an individual. Social support, psychosocial issues, and financial means are also carefully evaluated to maximize outcomes for patients and families. This care plan will present the nursing diagnoses, interventions, and outcomes across four types of solid organ transplantation: heart, lung, liver, and kidney. The organ-specific problems and interventions are presented first, followed at the end by presentation of three problems that relate to all types of transplantation.
Heart Transplantation
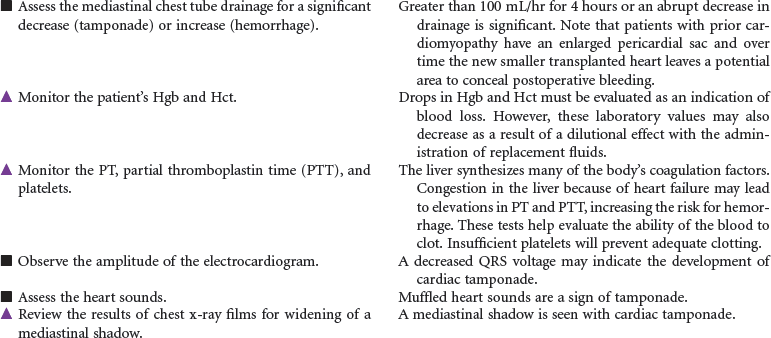
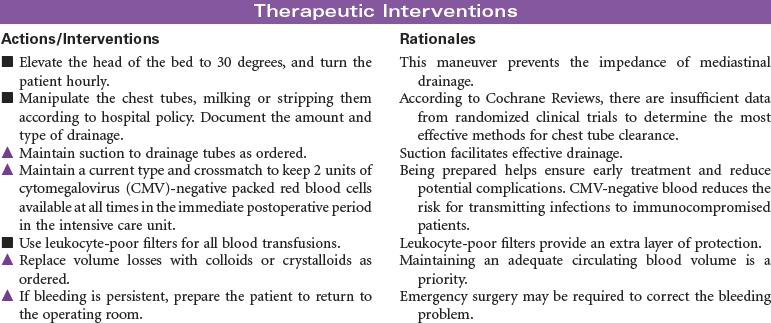
Candidate selection for heart transplantation is based on the consensus from the International Society for Heart and Lung Transplantation (ISHLT). The most common diagnoses of patients referred for heart transplantation include ischemic cardiomyopathy and idiopathic dilated cardiomyopathy. Patients with New York Heart Association class 3 or 4 heart failure are often at risk for rhythm disturbances as well as for the development of biventricular failure. Many may have pacemakers or automatic internal cardiac defibrillators implanted to protect them from sudden death. Because of the shortage of suitable donor hearts, patients must be carefully screened to ensure that heart transplantation is the best and only option. Patients who have failed optimal medical therapy to control their heart failure are often candidates to be evaluated for transplantation. Mechanical circulatory assist devices can be used as a bridge to transplantation or as destination therapy in select cases.
In the United States there are approximately 2000 deceased donor heart transplants performed each year. Nursing care of patients during the immediate post-transplant period requires intravascular pressure monitoring, excellent clinical assessment skills, and a plan of care that addresses prevention of rejection, infection, and bleeding complications. Patient and family education is important in preparing recipients for self-care and for optimizing long-term outcomes.
Lung Transplantation
The first successful human single lung transplant was performed in 1983, followed by a successful double lung transplant in 1986. Fewer than 1500 lung transplants are performed annually in the United States, with the majority of lungs being procured from deceased donors. Living lung transplants were performed for children with cystic fibrosis in the late 1990s and early 2000s. However, because of the risk to living donors, there were only three living donor lung transplants performed in 2007 and none in 2008. Lung transplantation is often complicated by infection.
The major diagnoses of patients requiring lung transplantation include emphysema, alpha-1-antitrypsin deficiency, interstitial pulmonary fibrosis, cystic fibrosis, and primary pulmonary hypertension. Because of the shortage of potential lungs for transplantation, patients are selected according to criteria described by the International Society of Heart and Lung Transplantation. Testing to determine candidacy for lung transplantation includes chest x-ray film, ventilation-perfusion (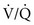 ) scan, 6-minute walk, pulmonary function tests, arterial blood gases, echocardiogram, and cardiac catheterization for patients over the age of 40.
) scan, 6-minute walk, pulmonary function tests, arterial blood gases, echocardiogram, and cardiac catheterization for patients over the age of 40.
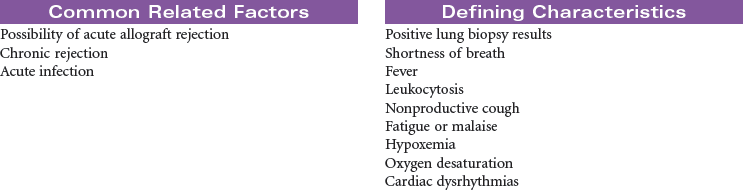
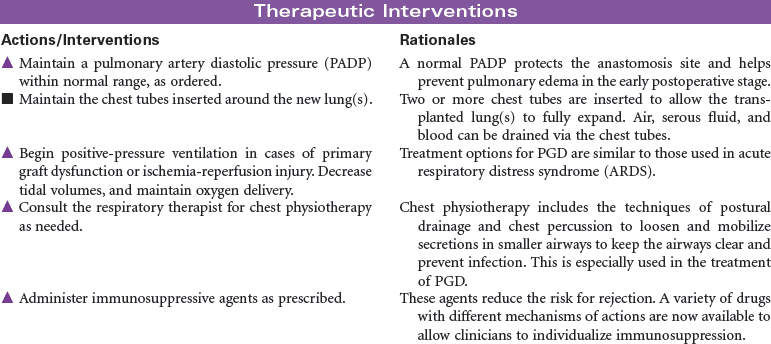
Lung transplant recipients usually require intensive nursing care for the first 4 to 7 days immediately postoperatively. In this period, complications include reperfusion injury, hemothorax, pneumothorax, pleural effusion, rejection, and infection.
Liver Transplantation
Liver transplantation is a treatment option for persons with end-stage liver disease for whom all possible modes of surgical and medical treatment have been exhausted.
The indications for liver transplantation can be either acute (fulminant) liver failure or chronic liver disease. The most common causes of chronic liver disease and cirrhosis are hepatitis C, alcohol, nonalcoholic steatohepatitis, primary biliary cirrhosis, and primary sclerosing cholangitis. Less-common causes are hepatitis B and metabolic diseases such as hemochromatosis and Wilson’s disease. Patients with hepatocellular carcinoma must meet certain criteria, called the Milan criteria, to be considered for liver transplant. Milan criteria state that there must be one single tumor less than 5 cm or no greater than three tumors each measuring less than 3 cm.
The absolute contraindications for liver transplant are active alcohol or substance abuse, severe cardiopulmonary or other comorbid conditions that would preclude meaningful recovery after transplant, active extrahepatic malignancy, hepatic malignancy with macrovascular or diffuse tumor invasion, active and uncontrolled infection, technical or anatomical barriers, psychological factors that would likely preclude recovery after liver transplantation, and brain death. Relative contraindications for liver transplant include advanced age, cholangiocarcinoma (can be considered under strict protocols), chronic or refractory infections, HIV, previous malignancy within 5 years, portal vein thrombosis, active psychiatric illness, and poor social support.
A liver transplant evaluation is performed by a comprehensive team that reviews each individual’s need and examines data from several imaging studies and a battery of laboratory tests. Patients are listed on the waiting list according to their blood type and Model of End-Stage Liver Disease (MELD) score, which is calculated by using the patient’s international normalized ratio, creatinine, and total bilirubin. This score ranges from as low as 6 to as high as 40. The surgical procedure entails the excision of both donor and recipient livers and transplantation of the donor liver into the recipient (orthotopically transplanted). Live donor liver transplantation involves removing a portion of the liver from a healthy donor who underwent extensive workup for suitability before the surgery.
Because of the chronic systemic problems resulting from end-stage liver disease and the technical complexity of the transplant procedure, postoperative recovery can be complicated. However, with ongoing compliance with medical therapy and adherence to lifestyle changes, the transplant patient can live an active and productive life.
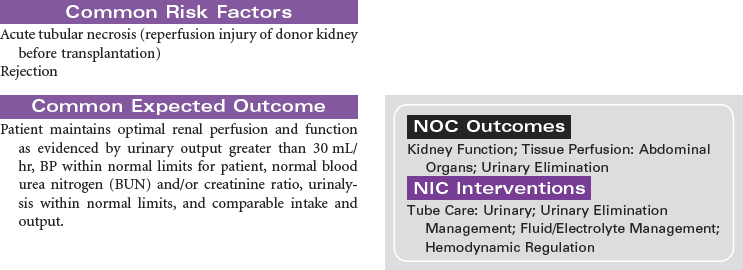
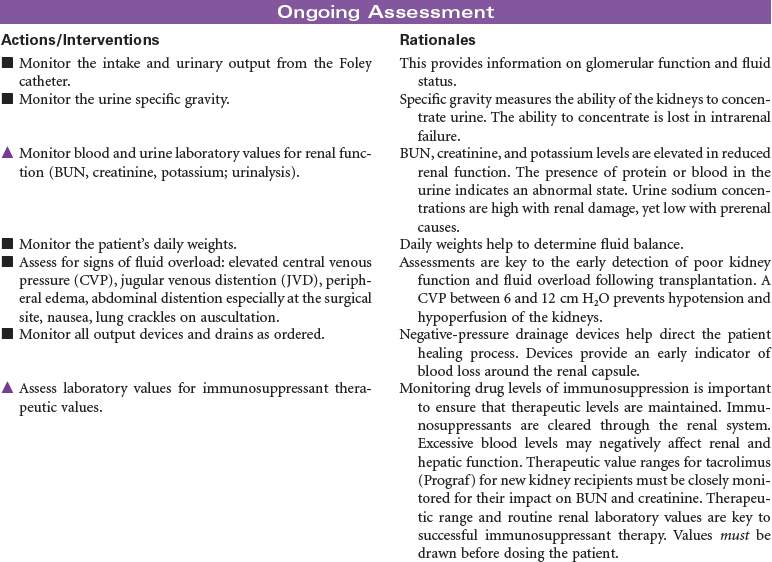
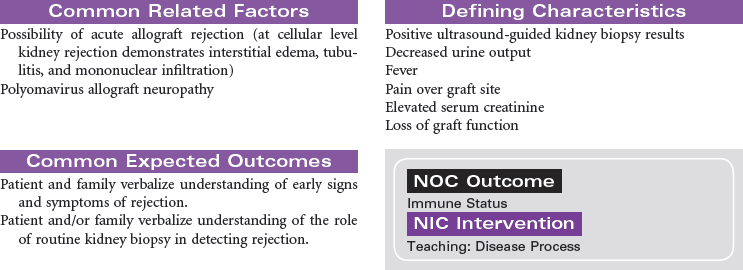
Kidney Transplantation
Kidney transplant is viewed as the superior treatment for end-stage renal disease (ESRD). Receiving a kidney transplant entails an anterior surgical incision that may or may not remove the patient’s own kidney, depending on the disease process that contributed to the development of ESRD. Most individuals will keep their native organ, and the new kidney will be placed in the abdomen with the donor ureter connected to the recipient’s bladder. In the case of abscessed polycystic kidney disease, the affected native kidney will be removed to minimize the risk for abscess and infection development in the recipient.
Living Kidney Donors
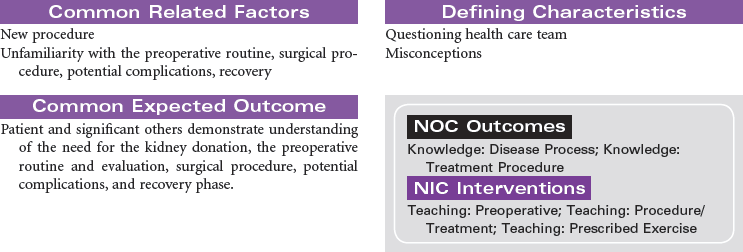
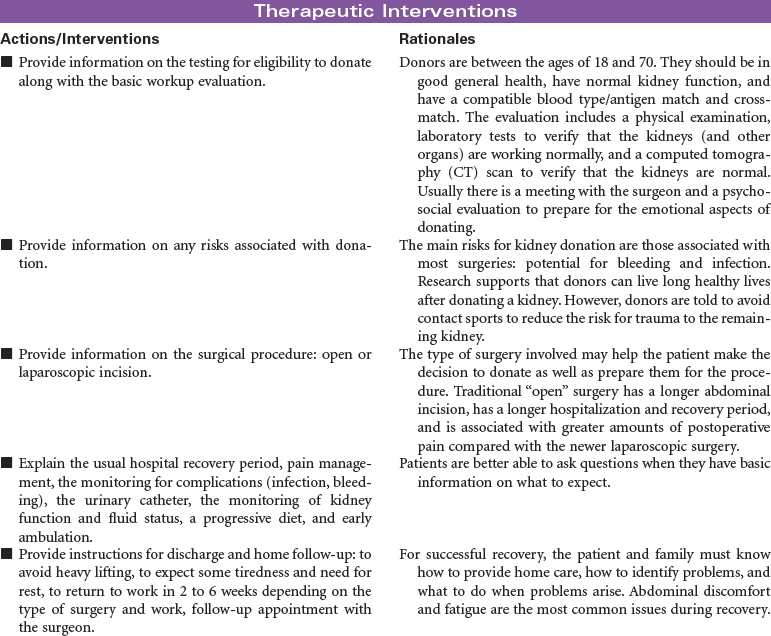
The most frequently performed transplant procedure, kidney transplantation, also accounts for the largest majority of living donors and is associated with the highest patient and graft survival rates. Education and information are key to maximizing outcomes, and they minimize stress for living kidney donors. Living kidney donors experience a thorough evaluation by a special living-donor advocacy team, including: assessment of physical well-being; review of family history; review of past medical history of diabetes, hypertension, heart disease, and weight control; and a psychiatric evaluation by a designated psychiatrist who has a specialty in living organ donation. The living-donor team is composed of a separate group of health care professionals from the recipient transplant team and includes a social worker, living donor transplant coordinator, living donor advocate, psychiatrist, nephrologist, and surgeon.
Living kidney donors are healthy patients who experience a major abdominal surgery that will be painful, with a recovery expected to last 6 to 8 weeks. Care providers must be ready to manage the juxtaposition of this situation. Most hospitalized patients undergoing major surgery are very uncomfortable before a procedure with hope of improving their condition; in contrast, living organ donors are healthy people who will potentially feel much worse after their health care experience. This can be difficult to cope with for family and donors alike.
Sickle Cell Disease
Vasoocclusive Crisis; Sickle Cell Pain Crisis
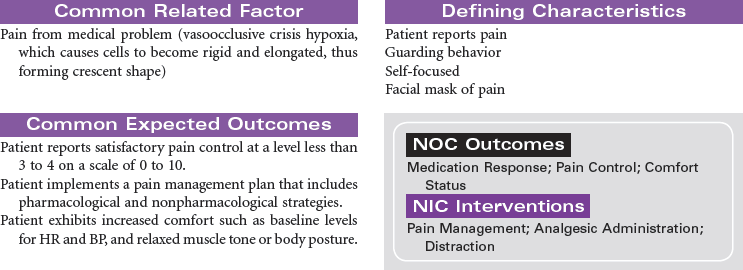
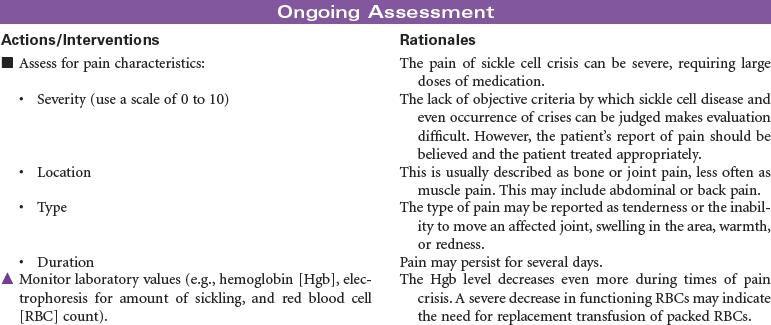
Sickle cell disease is a severe genetic hemolytic anemia caused by mutations in the HBB gene resulting in a defective hemoglobin molecule (HbS). This disease is found in Africans, African Americans, and people from Mediterranean countries. The formation of sickle cells is increased by low oxygen partial pressure. Factors associated with sickling include hypoxia, dehydration, infection, acidosis, cold exposure, and exertion. This chronic disease can cause impaired renal, pulmonary, nervous system, and spleen function; increased susceptibility to infection; and ultimately decreased life span. Sickle cell pain crisis is defined as pain of sufficient severity to require medical attention and hospitalization. The severe pain, usually in the extremities, is caused by the occlusion of small blood vessels by sickle-shaped red blood cells. Research continues in identifying effective antisickling agents and possible gene therapy to correct this defect. Hematopoetic stem cell transplantation can cure sickle cell disease. These are difficult procedures currently reserved for children with minimal organ damage from the disease. This care plan focuses on the physical and emotional aspects of sickle cell disease.
Systemic Lupus Erythematosus
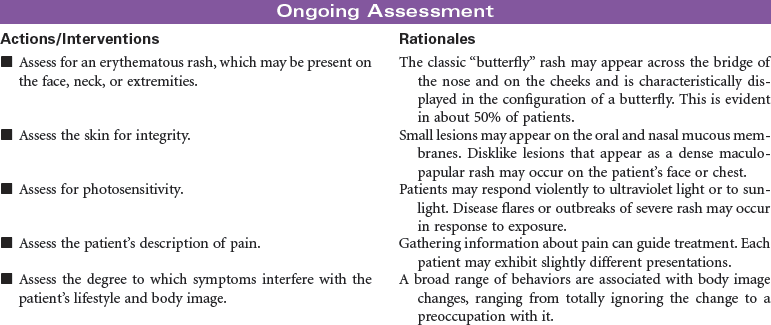
Systemic lupus erythematosus (SLE) is a chronic, autoimmune disease that causes a systemic inflammatory response in various parts of the body. The cause of SLE is unknown, but genetics and hormonal and environmental factors are involved. Under normal circumstances the body’s immune system produces antibodies against invading disease antigens to protect itself. In individuals with SLE the body loses its ability to discriminate between antigens and its own cells and tissues. It produces antibodies against itself, called autoantibodies, and these antibodies react with the antigens and result in the development of immune complexes. Immune complexes proliferate in the tissues of the patient with SLE and result in inflammation, tissue damage, and pain. Mild disease can affect joints and skin. More severe disease can affect kidneys, heart, lung, blood vessels, central nervous system, joints, and skin.
There are three type of lupus. The discoid type is limited to the skin and only rarely involves other organs. Systemic lupus is more common and usually more severe than discoid; it can affect any organ system in the body. With systemic lupus there may be periods of remission and flares. The third type of lupus is drug induced. The drugs most commonly implicated in precipitating this condition are hydralazine, procainamide, isoniazid, d-penicillamine, and some antiseizure drugs. Symptoms usually do not present until after months or years of continued administration. The symptoms are usually abolished when the drugs are discontinued.
Women are affected by SLE nine times more often than men, most commonly between 10 and 50 years of age. It is also more common among African Americans and in Asians. That the symptoms occur more frequently in women, especially before menstrual periods and during pregnancy, may indicate that hormonal factors influence development and progression of the disease. For some individuals, the disease remains mild and affects only a few organ systems; for others, the disease can cause life-threatening complications that can result in death. This care plan addresses the nursing management of patients with systemic lupus in an ambulatory setting.
Hematopoietic Stem Cell Collection
Bone Marrow Transplantation; Bone Marrow Donor; Apheresis; Peripheral Blood Stem Cell Donor; Autologous; Allogenic; Syngeneic
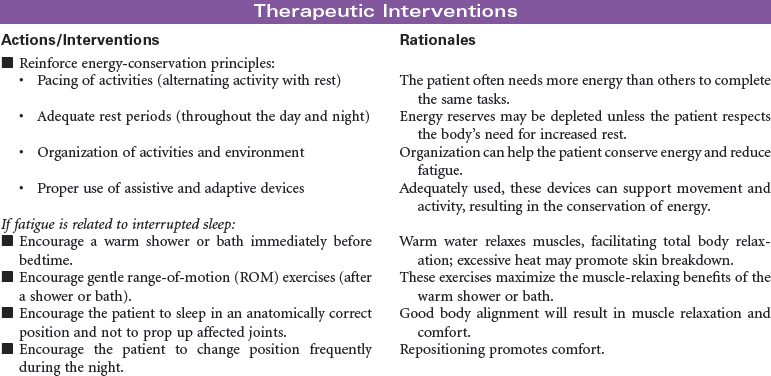
Hematopoietic stem cells (HSCs) normally reside in the bone marrow and can be collected from this compartment via multiple needle aspirations from the posterior iliac crest. When collected from the bone marrow, all blood components are collected (e.g., red blood cells [RBCs], platelets, plasma), not just the stem cell component. HSCs can be moved or mobilized out of the bone marrow into the bloodstream or peripheral circulation. The mobilization of HSCs into the bloodstream occurs after the administration of certain types of chemotherapy and/or colony-stimulating factors (CSFs) such as granulocyte colony-stimulating factor (G-CSF) (Neupogen) or granulocyte-macrophage colony-stimulating factor (GM-CSF) (Leukine). Mozobil is a new CXCR4 chemokine receptor antagonist that is available to use in combination with G-CSF in patients identified as poor mobilizers. HSCs can then be collected via a procedure known as apheresis. During apheresis, needles are inserted into the arm veins (or a central venous catheter), and blood is passed through an apheresis machine, which separates the blood components. Only the HSCs are removed and the rest of the blood components and volume are returned to the patient. This procedure occurs in the outpatient setting (usually in a blood bank or apheresis center), takes from 2 to 4 hours, and may be repeated for several days until the desired number of HSCs is collected. Most transplants performed today rely almost exclusively on peripheral blood stem cells (PBSCs) rather than bone marrow because PBSCs are easier to collect and are associated with more rapid hematopoietic recovery, which results in shortened hospitalizations and decreased costs.
Hemophilia
Clotting Abnormality; Factor VIII Deficiency; Factor IX Deficiency; Hemophilia A; Hemophilia B
Hemophilia is an inherited disorder of the clotting mechanism caused by diminished or absent factors necessary to the formation of prothrombin activator (the catalyst to clot formation). Hemophilia is a sex-linked recessive disorder transmitted by females and occurring almost exclusively in males. Classic hemophilia (type A) is caused by the lack of factor VIII; it is the most common and usually most severe type of hemophilia. Type B (Christmas disease) is caused by the lack of factor IX. Symptom severity is directly proportional to the plasma levels of available clotting factors; depending on these levels, the disease is classified as mild (5% to 30% of normal clotting factor), moderate (1% to 5% of normal clotting factor), or severe (<1% of normal clotting factor). Patients with close to normal factor levels may experience only frequent bruising and slightly prolonged bleeding times. Bleeding can be acute, delayed, or prolonged oozing over time. Common sites of bleeding include the head, which can be life threatening; joints, resulting in hemarthroses, arthroses, and destruction; or muscle tissue, causing ecchymosis, hematoma, or compartment syndrome. Bleeding in the abdomen can present with blood in the stool, or the patient may have excruciating pain from hematomas in the abdominal or bowel wall. This care plan addresses the more moderate symptoms of hemophilia. Most patients can lead normal lives if they manage their disorder appropriately. Comprehensive care is best provided through designated treatment centers.
 = Independent
= Independent  = Collaborative
= Collaborative