1 Abd-Allah S.A., et al. Helium-oxygen therapy for pediatric acute severe asthma requiring mechanical ventilation. Pediatr Crit Care Med. 2003;4:353-357.
2 Aboab J., et al. Effect of inspired oxygen on alveolar decruitment in acute respiratory distress syndrome. Intensive Care Med. 2006;32:1979-1986.
3 Acosta B., et al. Hemodynamic effects of noninvasive bilevel positive airway pressure on patients with chronic congestive heart failure with systolic dysfunction. Chest. 2000;118:1004.
4 Acute Respiratory Distress Network. Ventilation with lower tidal volumes as compared with conventional tidal volumes for acute lung injury and the acute respiratory distress syndrome. N Engl J Med. 2000;342:1301-1308.
5 Agus M.S., Alexander J.L., Mantell P.A. Continuous noninvasive end-tidal CO2 monitoring in pediatric in patients with diabetes ketoacidosis. Pediatr Diabetes. 2006;7:196-200.
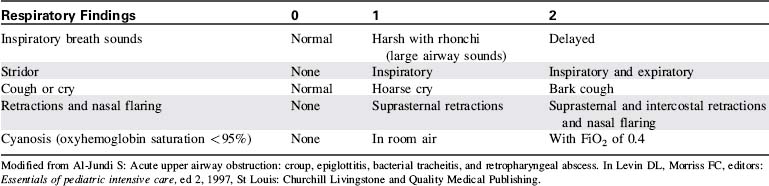
6 Al-Sundi S. Acute upper airway obstruction: croup, epiglottitis, bacterial tracheitis, and retropharyngeal abrasions. In: Levin D.L., Morriss F.C., editors. Essentials of pediatric intensive care. New York: Churchill Livingstone, 1997.
7 Amato M.B., et al. Effect of a protective-ventilation strategy on mortality in the acute respiratory distress syndrome. N Engl J Med. 1998;338:347.
8 American Academy of Pediatrics Committee on Fetus and Newborn. Postnatal steroids to treat or prevent chronic lung disease in preterm infant. Pediatrics. 109(330), 2002.
9 Anas N. Respiratory failure. In: Levin D.L., Morriss F.C., editors. Essentials of pediatric intensive care. New York: Churchill Livingstone, 1997.
10 Arnold J.H., et al. Prospective, randomized comparison of high-frequency oscillatory ventilation and conventional mechanical ventilation in pediatric respiratory failure. Crit Care Med. 1994;22:1530-1539.
11 Ashbaugh D.G., et al. Acute respiratory distress in adults. Lancet. 1967;12:7511.
12 Bancalari E. Mechanical ventilation. In: Rudolph C.D., Rudolph A.M., editors. Rudolph's pediatrics. New York: McGraw Hill, 2003.
13 Baraldi E., Filippone M. Chronic lung disease after premature birth. N Engl J Med. 2007;357:1946-1955.
14 Bartlett J.G. Aspiration pneumonia. Clin Notes Respir Disease. 1980;18:3.
15 Berg M.D., Schexnayder S.M., Chameides L., et al. Pediatric basic life support. 2010 American heart association guidelines for cardiopulmonary resuscitation and emergency cardiovascular care. Circulation. 122(18 Suppl. 3), 2010. S862-875
16 Berg M.D. Pediatric in-hospital cardiac arrest and therapeutic hypothermia: where we are and where we are going. Pediatr Crit Care Med. 2009;10:601-602.
17 Berkenbosch J.W., Tobias J. Transcutaneous carbon dioxide monitoring during high frequency oscillatory ventilation in infants and children. Crit Care Med. 2002;30:1024-1027.
18 Bernard G.R., et al. The American-European consensus conference on ARDS definitions, mechanisms, relevant outcomes and clinical trial coordination. Am J Respir Crit Care Med. 1994;149:818.
19 Bernard S.A., et al. Treatment of comatose survivors of out-of-hospital cardiac arrest with induced hypothermia. N Engl J Med. 2002;346:557.
20 Bernet V.B., et al. Predictive factors for the success of noninvasive mask ventilation in infants and children with acute respiratory failure. Pediatr Crit Care Med. 2005;6:660.
21 Bhende M.S. End-tidal carbon dioxide monitoring in pediatrics—clinical applications. J Postgraduate Med. 2001;47:215-218.
22 Biagas K. Drowning and near drowning: submersion injuries. In Nichols D.G., editor: Rogers' textbook of pediatric intensive care, ed 4, Philadelphia: Lippincott Williams and Wilkins, 2008.
23 Bindslev L., Hedenstierna G., Santesson J., et al. Ventilation-perfusion distribution during inhalation anaesthesia. Effects of spontaneous breathing, mechanical ventilation and positive end-expiratory pressure. Acta Anaesthesiol Scand. 1981;25(4):360-371.
24 Bjornson C.L., Johnson D.W. Croup. Lancet. 2008;371:329.
25 Bledsoe G.H., Schexnayder S.M. Pediatric rapid sequence intubation. Pediatr Emerg Care. 2004;20:339.
26 Boogaard R., et al. Recombinant human deoxyribonuclease for the treatment of acute asthma in children. Thorax. 2008;63:141-146.
27 Boyce J.A.K. Auto positive end-expiratory pressure calculation. In: Verger J.T., Lebet R.M., editors. AACN procedure manual for pediatric acute and critical care. St Louis: Saunders, 2008.
28 Branson R.D. Secretion management in the mechanically ventilated patient. Respir Care. 2007;52:1328-1342.
29 Briassoulis G.C., et al. Air leaks from the respiratory tract in mechanically ventilated children with severe respiratory disease. Pediatr Pulmonol. 2000;29:127-134.
30 Brochard L., Pluskwa R., Lemaire F. Improved efficacy of spontaneous breathing with inspiratory pressure support. Am Rev Respir Dis. 1987;136:411.
31 Bronicki R., Anas N. Cardiopulmonary interaction. Pediatr Crit Care Med. 2009;10:313-322.
32 Bryan M., et al. Pulmonary function studies during the first year of life in infants recovering from the respiratory distress syndrome. Pediatrics. 1973;52:169.
33 Buford A.E., et al. Drowning and near-drowning in children and adolescents. Pediatr Emerg Care. 2005;21:610.
34 Cahn M.D., et al. Long-term follow up of Greenfield inferior cava filter placement in children. J Vascular Surg. 2001;34:820-825.
35 Carlson J., et al. Extubation force: tape versus endotracheal tube holders. Ann Emerg Med. 2007;50:686-691.
36 Carroll C.L., Schramm C.M. Noninvasive positive pressure ventilation for the treatment of status asthmaticus in children. Ann Allergy Asthma Immunol. 2006;96:454-459.
37 Carroll C.L., Zucker A.R. The increased cost of complications in children with status asthmaticus. Pediatr Pulmonol. 2007;42:914-919.
38 Carter E.R., Webb C.R., Moffitt D.R. Evaluation of heliox in children hospitalized with acute severe asthma. A randomized crossover trial. Chest. 1996;109:1256-1261.
39 Casati A., et al. Transcutaneous monitoring of partial pressure of carbon dioxide in the elderly patient: a prospective, clinical comparison with end-tidal monitoring. J Clin Anesth. 2006;18:436-440.
40 Caviedes I., et al. Effect of intrinsic positive end-expiratory pressure on respiratory compliance. Crit Care Med. 1986;14:947.
41 Celebi S., Hacimustafaoglu S., Demirkaya M. Septic pulmonary emboli in a child. Indian Pediatr. 2008;45:415-417.
42 Centers for Disease Control and Prevention, National Center for Injury Prevention and Control: Web-based Injury Statistics Query and Reporting System (WISQARS) [online]. 2008 Available at: http://www.cdc.gov/ncipc/wisqars Accessed July 28, 2008
43 Chameides L.C., Samson R.A., Schexnayder S.M., Hazinski M.F., editors. Pediatric advanced life support manual. Dallas: American Heart Association, 2011.
44 Chatte G., et al. Prone position in mechanically ventilated patients with severe acute respiratory failure. Am J Respir Crit Care Med. 1997;155:473.
45 Cherry J.D. Croup. N England Med. 2008;358:384.
46 Choi S.J. Comparison of desaturation and resaturation response times between transmission and reflectance pulse oximeters. Acta Anaesthesiol Scand. 2010;54:212-217.
47 Ciarallo L., Brousseau D., Reinert S. Higher-dose intravenous magnesium therapy for children with moderate to severe acute asthma. Arch Pediatr Adolesc Med. 2000;154:979-983.
48 Ciarallo L., Sauer A.H., Shannon M.W. Intravenous magnesium therapy for moderate to severe pediatric asthma: results of a randomized, placebo-controlled trial. J Pediatr. 1996;129:809-814.
49 Coalson J.J. Stucture of the respiratory system. In Fuhrman B.P., Zimmerman J., editors: Pediatric critical care, ed 3, Philadelphia: Mosby-Elsevier, 2006.
50 Collaborative European Multicenter Study Group. Surfactant replacement therapy for severe neonatal respiratory distress syndrome: an international randomized clinical trial. Pediatrics. 1988;82:683-691.
51 Colvin J., et al. Outcomes of congenital diaphragmatic hernia: a population-based study in Western Australia. Pediatrics. 2005;116:e356-e363.
52 Craven D., et al. Iprtropium bromide plus nebulized albuterol for the treatment of hospitalized children with acute asthma. J Pediatr. 2001;138:51-58.
53 Curley M.A.Q., et al. Effect of prone positioning on clinical outcomes in children with acute lung injury. J Am Med Assoc. 2005;294:229.
54 Curley M.A.Q., Thompson J.E., Arnold J.H. The effects of early and repeated prone positioning in pediatric patients with acute lung injury. Chest. 2000;118:156.
55 D'Ávila R.S., et al. Early administration of two intravenous bolus of aminophylline added to the standard treatment of children with acute asthma. Respir Med. 2008;102:156-161.
56 Davison C., et al. Efficacy of interventions for bronchiolitis in critically ill infants: a systematic review and meta analysis. Pediatr Crit Care Med. 2004;5:482.
57 Datin-Dorriere V. Prenatal prognosis in isolated congenital diaphragmatic hernia. Am J Obstet Gynecol. 2008;198(1):80-81.
58 Daya H., et al. Pediatric vocal fold paralysis: a long-term retrospective study. Arch Otolaryngol Head Neck Surg. 2000;126:21-25.
59 deCaen A., Duff J., Coovadia A.H., et al Airway Management. In Nichols DG, editor-in-chief: Roger's textbook of pediatric critical care. Philadelphia. ed 4, Lippincott; Williams and Wilkins:2008
60 Deep A., De Munter C., Desai A. Negative pressure ventilation in pediatric critical care setting. Indian J Pediatr. 2007;74:483.
61 DeJonge M.H., White M. A comparison of two methods or oral endotracheal tube stabilization in neonatal patients. J Perinatol. 1998;18:463-465.
62 deKlerk A. Humidified high-flow nasal cannula: is it the new and improved CPAP? Adv Neonatal Care. 8(98), 2008.
63 Denne S.C. Energy Expenditure in infants with pulmonary insufficiency: is there evidence for increased energy needs? J Nutr. 2001;131:935S.
64 Dobyns E.L. Assessment and monitoring of respiratory function. In: Fuhrman B.P., Zimmerman J., editors. Pediatric critical care. Philadelphia: Mosby, 2006.
65 Doshi J., Krawiec M.E. Clinical manifestations of airway malacia in young children. J Allergy Clin Immunol. 2007;120:1276.
66 Drover D.W. Gastric versus postpyloric feeding. Gastrointest Endosc Clin N Am. 2007;17:765-775.
67 Duffett M., et al. Surfactant therapy for acute respiratory failure in children: a systematic review and meta-analysis. Critical Care. 11(R66), 2007. Available at: http://pedsccm.org/view.php?id=498 Accessed June 26, 2011
68 Dullenkopf A., et al. Performance of a novel pressure release valve for cuff pressure control in pediatric tracheal tubes. Pediatr Anesth. 2006;16:19-24.
69 Duruisseau O., et al. Endoscopic rehabilitation of vocal cord paralysis with a silicone elastomer suspension implant. Otolaryngol Head Neck Surg. 2004;131:241-247.
70 Ehrlich P.F. Coran Eventration of the Diaphragm. In Kleigman R.M., editor: Nelson textbook of pediatrics, ed 18, Philadelphia: Elsevier/Saunders, 2007.
71 Essouri S., et al. Noninvasive positive pressure ventilation: five years of experience in a pediatric intensive care unit. Pediatr Crit Care Med. 2006;7:329.
72 Farias J.A., et al. An evaluation of extubation failure predictors in mechanically ventilated infants and children. Intensive Care Med. 2002;28:752.
73 Farias J.A., et al. A comparison of two methods to perform a breathing trial before extubation in pediatric intensive care patients. Intensive Care Med. 2001;27:1649.
74 Ferrara T.B., et al. Routine use of Dexamethasone for the prevention of postextubation respiratory distress. J Perinatol. 1989;9:287.
75 Fewell JE, Abendschein DR, Carlson J, et al. Mechanism of decreased right and left ventricular end-diastolic volumes during continuous positive-pressure ventilation in dogs. Circ Res. 1980;47:467-472.
76 Fiastro J.F., Habib M.P., Quan S.E. Pressure support compensation for inspiratory work due to endotracheal tubes and demand continuous positive airway pressure. Chest. 1988;93:499.
77 Flori H.R., et al. Pediatric acute lung injury: prospective evaluation of risk factors associated with mortality. Am J Respir Crit Care Med. 2005;171:995-1001.
78 Foglia E., Meier M.D., Elward A. Ventilator-associated pneumonia in neonatal and pediatric intensive care unit patients. Clin Microbiol Rev. 2007;20:409-425.
79 Fontán J.P., Haddad G.G. Respiratory Pathophysiology. In: Behrman R.E., Kliegman R.M., Jenson H.B., editors. Nelson textbook of pediatrics. Philadelphia: Saunders, 2004.
80 Fontán J.P., Lister G. The acutely ill infant and child. In: Rudolph C.D., et al, editors. Rudolph's pediatrics. New York: McGraw-Hill, 2003.
81 Frank B.S., Lewis R.J. Experience with intubated patients does not effect the accidental extubation rate in pediatric intensive care units and intensive care nurseries. Pediatr Pulmonol.. 1997;23:424-428.
82 Frankel L. Respiratory distress and failure. In: Berhman R.E., Kliegman R.M., Jenson H.B., editors. Nelson textbook of pediatrics. Philadelphia: Saunders, 2004.
83 Gal T.G. Pulmonary function testing. In: Miller R.D., editor. Miller's Anesthesia. Philadelphia: Elsevier, 2005.
84 Gardner A., et al. Best practice in stabilization of oral endotracheal tubes: a systemic review. Aust Crit Care. 2005;18:160-165.
85 Gawchik S.M., et al. The safety and efficacy of nebulized levalbuterol compared with racemic albuterol and placebo, in treatment of asthma in pediatric patients. J Allergy Clin Immunol. 1999;103:615-621.
86 Geelhoed G.C. Sixteen years of croup in a western Australian teaching hospital: effects of routine steroid treatment. Ann Emerg Med. 1996;28:621.
87 Glover M.L., Machado C., Totapally B.R. Magnesium sulfate administered via continuous infusion in pediatric patients with refractory wheezing. J Crit Care. 2002;17:255-258.
88 Goggi N., Macarthur C., Parkin P.C. Randomized trial of the addition of ipratropium bromide to albuterol and corticosteroid therapy in children hospitalized because of an acute asthma exacerbation. Arch Pediatr Adolesc Med. 2001;155:1329-1334.
89 Gold W.M. Pulmonary Function Testing. In: Mason R.J., editor. Murray & Nadel's texbook of respiratory medicine. Philadelphia: Elsevier, 2005.
90 Gorelick M.H., Baker M.D. Epiglottitis in children, 1979 through 1992. Effects of haemophilus influenzae type b immunization. Arch Pediatr Adolesc Med. 148(47), 1994.
91 Grant M.J.C., Curley M.A.Q. Pulmonary critical care problems. In: Curley M.A.Q., Maloney-Harmon P.A., editors. Critical care nursing of infants and children. Philadelphia: WB Saunders, 2001.
92 Grant M.J.C., Webster H.F. Pulmonary system. In Slota M.C., editor: Core curriculum for pediatric critical care nursing, ed 2, St Louis: Saunders, 2006.
93 Grillo H.C., et al. Management of congenital tracheal stenosis by means of slide tracheoplasty or resection and reconstruction, with long term follow-up of growth after slide tracheoplasty. J Thorac Cardiovasc Surg. 2002;112:145.
94 Gupta V.K., Cheifetz I. Heliox administration in the pediatric intensive care unit: an evidence-based review. Pediatr Crit Care Med. 2005;6:204-211.
95 Guyton A.C., Jones C.E., Coleman T.G. Mean circulatory pressure, mean systemic pressure, and mean pulmonary pressure and their effects on venous return. In: Guyton A.C., Jones C.E., Coleman T.G., editors. Circulatory physiology: cardiac output and its regulation. ed 2. Philadelphia: WB Saunders; 1973:205-221.
96 Guyton D.C., Barlow M.R., Besselievre T.R. Influence of airway pressure on minimum occlusive endotracheal tube cuff pressure. Crit Care Med. 1997;25:91-94.
97 Hales R.L. High frequency oscillatory ventilation. In: Verger J.T., Lebet R.M., editors. AACN procedure manual for pediatric acute and critical care. Philadelphia: WB Saunders, 2008.
98 Happ M.B., et al. Use of the quasi-experimental sequential cohort design in the study of patient-nurse effectiveness with assisted communication. Contemp Clin Trials. 2008;29:801-808.
99 Hazinski T.A. Tools for Diagnosis and Management of Respiratory Disorders. In Rudolph C.D., et al, editors: Rudolph's pediatrics, ed 21, New York: McGraw-Hill, 2003.
100 Jul 18Henderson-Smart D.J., et al. Elective high frequency oscillatory ventilation versus conventional ventilation for acute pulmonary dysfunction in preterm infants. Cochrane Database Syst Rev. 3(CD000104), 2007.
101 Henrickson K.J. Cost-effective use of rapid diagnostic techniques in the treatment and prevention of viral respiratory infections. Pediatr Ann. 2005;34:24.
102 Herting E., et al. Surfactant improves oxygenation in infants and children with pneumonia and acute respiratory distress syndrome. ACTA Paediatr. 2002;91:1174.
103 Heulitt M.J. Physiology of the respiratory system. In Fuhrman B.P., Zimmerman J., editors: Pediatric critical care, ed 3, Philadelphia: Mosby-Elsevier, 2006.
104 Heyland D.K., et al. Optimizing the benefits and minimizing the risks of enteral nutrition in the critically ill: role of small bowel feeding. J Parenter Enteral Nutr. 2002;28:60.
105 Hickling K.G., Henderson S.J., Jackson R. Low mortality associated with low volume pressure limited ventilation with permissive hypercapnia in severe adult respiratory distress syndrome. Intensive Care Med. 1990;16:372.
106 Hypothermia after Cardiac Arrest Study Group. Mild therapeutic hypothermia to improve the neurological outcome after cardiac arrest. N Engl J Med. 246(549), 2002.
107 Idris A.H., et al. Recommended guidelines for uniform reporting of data from drowning the “Utstein Style”. Circulation. 2003;108:2565.
108 Isben L.M., Koch T. Submersion and asphyxia injury. Crit Care Med. 2002;30(11 Suppl.):S402-S408.
109 Jaber S., et al. Volume-guaranteed pressure-support ventilation facing acute changes in ventilatory demand. Intensive Care Med. 2005;31:1181.
110 Jardine D.S., Crone R.K. Specific diseases of the respiratory system: upper airway. In: Fuhrman B.P., Zimmerman J., editors. Pediatric critical care. Philadelphia: Mosby, 2006.
111 Jarog D.L. Endotracheal tube: taping. In: Verger J.T., Lebet R.M., editors. AACN procedure manual for pediatric acute and critical care. St Louis: Saunders Elsevier, 2008.
112 Jin-Hee K., et al. Elongation of the trachea during neck extension in children: implications of the safety of endotracheal tubes. Anesth Analg. 2005;101:974-977.
113 Jobe A.H., Bancalari E. Bronchopulmonary Dysplasia (electronic version). Am J Respir Crit Care Med. 2001;163:1723. Available at: http://ajrccm.atsjournals.org/cgi/content/full/163/7/1723?ijkey=9885160ff371fb7a8a8b47bf80f44a06e051598c&keytype2=tf_ipsecsha Accessed June 26, 2011
114 Johansson F., et al. Effects of albuterol enantiomers on in vitro bronchial reactivity. Clin Rev Allergy Immunol. 1999;14:57-64.
115 Art. No.: CD000437Joshi V.H., Bhuta T. Rescue high frequency jet ventilation versus conventional ventilation for severe pulmonary dysfunction in preterm infants. Cochrane Database of Systematic Reviews. 1, 2006. doi: 10.1002/14651858.CD000437.pub2
116 Kane-Gill S.L., et al. Multicenter treatment and outcome evaluation of aspiration syndromes in critically ill patients. Ann Pharmacother. 2007;41:549-555.
117 Karakoc F., et al. Late diagnosis of foreign body aspiration in children with chronic respiratory symptoms. Int J Pediatr Otorhinolaryngol. 2007;71:241-246. Epub 2006 Nov 27
118 Kelley P.J., Arney T.D. Use of magnesium sulfate for pediatric patients with acute asthma exacerbations. J Infus Nurs. 2005;28:329-336.
119 Kellner J.D., Ohlsson A., Gadomski A.M., Wang E.E.L. Bronchodilators for bronchiolitis. Cochrane Database Syst Rev. 2(CD001266), 2000.
120 Kim I.K., et al. Helium/oxygen-driven albuterol nebulization in the treatment of children with moderate to severe asthma exacerbations: a randomized, controlled trial. Pediatrics. 2005;116:1127-1133.
121 Kirk V.G., Batuyong E.D., Bohn S.G. Transcutaneous carbon dioxide monitoring and capnography during pediatric polysomnography. Sleep. 2006;29:1601-1608.
122 Kissoon N., Rimensberger P.C., Bohn D. Ventilation strategies and adjunctive therapy in severe lung disease. Pediatr Clin N Am. 2008;55:709.
122a Kleinman ME, Chameides L, Schexnayder SM, et al. Part 14: pediatric advanced life support: 2010 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation. 2010;122(18 Suppl 3):S876-908.
123 Kline A.M. Arterial Puncture: perform. In: Verger J.T., Lebet R.M., editors. AACN procedure manual for pediatric acute and critical care. St Louis: Saunders-Elsevier, 2008.
124 Kregenow D.A., et al. Hypercapnic acidosis and mortality in acute lung injury. Crit Care Med. 2006;34:1.
125 Krishnan J.A., Brower R.G. High-frequency ventilation for acute lung injury and ARDS. Chest. 2000;118:795.
126 Lacroix J., et al. Transfusion strategies for patients in pediatric intensive care units. New Engl J Med. 2007;356:1609-1619.
127 Lee C.C., et al. Outcome of vocal cord paralysis in infants. Acta Paediatr Taiwan. 2004;45:278-281.
128 Lee L.K., Mao C., Thompson K.M. Demographic factors and their association with outcomes in pediatric submersion injury. Acadc Emerg Med. 2006;13:308.
129 Levin D. Morphological analysis of the pulmonary vascular bed in congenital left-sided diaphragmatic hernia. J Pediatr. 1978;92:805-809.
130 Levin D.L., et al. A prospective, randomized controlled blinded study of three bronchodilators in infants with respiratory syncytial virus bronchiolitis on mechanical ventilation. Pediatr Crit Care Med. 2008;9:598-604.
131 Levin D.L. Near-drowning. Crit Care Med. 1980;8:590-595.
132 Levin D.L., et al. Persistent pulmonary hypertension of the newborn. J Pediatr. 1976;89:626-630.
133 Levin D.L., Tribuzio W., Green-Wrzesinkit T. Empiric antibiotics are justified for infants with respiratory syncytial virus lower respiratory tract infection presenting with respiratory failure. A prospective slides and evidence review. Pediatr Crit Care Med. 2010;11:289-294.
134 Little L.A., Koenig J.C., Newth C.J.L. Factors affecting accidental extubations in neonatal and pediatric intensive care patients. Crit Care Med. 1990;18:163.
135 Lloyd T.C. Effect of inspiration on inferior vena caval blood flow in dogs. J Appl Phyisol. 1983;55:1701-1708.
136 Lodato R.F. Oxygen toxicity. Crit Care Clin. 1990;6:749.
137 Loh L.E., Chan Y.H., Chan I. Noninvasive ventilation in children: a review. J Pediatr (Rio J). 2007;83:S91.
138 Loughead J.L., et al. Reducing accidental extubation in neonates. Jt Comm J Qual Patient Saf. 2008;34:164-170.
139 Luten R.C., et al. Length-based endotracheal tube and emergency equipment in pediatrics. Ann Emerg Med. 1992;21:900.
140 Macintyre N.R. Principles of mechanical ventilation. In: Mason R.J., editor. Murray and Nadel's texbook of respiratory medicine. Philadelphia: Elsevier, 2005.
141 Maclntyre N.R. Pressure support ventilation: effects on ventilatory reflexes and ventilatory-muscle workloads. Respir Care. 1987;32:447.
142 Magnus V.S., Turkington L. Communication interaction in ICU—patient and staff experiences and perceptions. Intensive Crit Care Nursing. 2006;22:167-180.
143 Mallick M.S., et al. Late presentation of tracheobronchial foreign body aspiration in children. Pediatrics. 2005;51:145-148.
144 Mancebo J., et al. A multicenter trial of prolonged prone ventilation in severe acute respiratory distress syndrome. Am J Respir Crit Care Med. 2006;173:1233.
145 Mannix R., Bachur R. Status asthmaticus in children. Curr Opin Pediatr. 2007;19:281-287.
146 Marcin J.P., et al. Nurse staffing and unplanned extubation in the pediatric intensive care unit. Pediatr Crit Care Med. 2005;6:254-257.
147 Marini J.J. Mechanical ventilation. Curr Pulmonol. 1988;9:164.
148 Markovitz B.P., Randolph A.G., Khemani R.G. Corticosteroids for the prevention and treatment of post-extubation stridor in neonates, children and adults. Cochrane Database Sys Rev. 2(CD001000), 2008.
149 Marraro G.A. Innovative practices of ventilatory support with pediatric patients. Pediatr Crit Care Med. 2003;4:8.
150 Martins R.H.G., et al. Endoscopic findings in children with stridor. Braz J Otorhinolaryngol. 2006;72:649.
151 McClave S.A., et al. Poor validity of residual volumes as a marker for risk of aspiration in critically ill patients. Crit Care Med. 2005;33:324.
152 McNamara V.M., Crabbe D.C.G. Traceomalacia. Paediatr Respir Rev. 2004;5:147.
153 Meert K.L., Daphtary K.M., Metheny N.A. Gastric vs small bowel feeding in critically ill children receiving mechanical ventilation. Chest. 2004;126:872-878.
154 Meissner H.C. Selected populations at increased risk from respiratory syncytial virus infection. Pediatr Infect Dis J. 2003;22:S40.
155 Mercier J., Dauger S., Durand Javouey E. Acute respiratory distress syndrome. In: Fuhrman B.P., Zimmerman J., editors. Pediatric critical care. Philadelphia: Mosby-Elsevier, 2006.
156 Meyer R.J., Theodorou A.A., Berg R.A. Childhood drowning. Pediatr Rev. 2006;27:163.
157 Mhanna M.J., et al. The “air leak” test around the endotracheal tube, as predictor of postextubation stridor, is age dependent in children. Crit Care Med. 2002;30:2639-2643.
158 Michelow I.C., et al. Epidemiology and clinical characteristics of community-acquired pneumonia in hospitalized children. Pediatrics. 2004;113:701.
159 Michiels J.J., et al. Noninvasive exclusion and diagnosis of pulmonary embolism by sequential use of the rapid ELISA d-dimer assay, clinical score and spiral CT. Int Angiol. 2003;22:1-14.
160 Mitchell R.B., Kelly J. Adenotonsillectomy for obstructive sleep apnea in obese children. Otolaryngol Head Neck Surg. 2004;131:104-108.
161 Mitra A., Bassler D., Ducharme F.M. Intravenous aminophylline for acute severe asthma in children over 2 years of age using inhaled bronchodilators. Cochrane Database Syst Rev. 4(CD001276), 2001.
162 Miyamoto R.C., et al. Bilateral congenital vocal cord paralysis: a 16 year institutional review. Otolaryngol Head Neck Surg. 2005;133:241-245.
163 Möller J.C., et al. Treatment with bovine surfactant in severe acute respiratory distress syndrome in children: a randomized multicenter study. Intensive Care Med. 2003;29:437.
164 Moore M., Little P. Humidified air inhalation for treating croup. Cochrane Database Syst Rev. 3(CD002870), 2006.
165 Morris L.G., Zoumalan R.A., Roccaforte J.D., et al. Monitoring tracheal tube cuff pressures in the intensive care unit: a comparison of digital palpation and manometry. An Otol Rhinol Laryngol. 2007;116:639-642.
166 Morrow B., Futter M., Argent A. Effect of endotracheal suction on lung dynamics in mechanically-ventilated paediatric patients. Aust J Physiother. 2006;52:121-126.
167 Myers T.R. Use of heliox in children. Respir Care. 2006;51:6.
168 National Heart, Lung, and Blood Institute Acute Respiratory Distress Syndrome (ARDS) Clinical Trials Network, et al. Comparison of two fluid-management strategies in acute lung injury. N Engl J Med. 2006;354:2564-2575.
169 Nawab U.S., et al. Heliox attenuates lung inflammation and structural alterations in acute lung injury. Pediatr Pulmonol. 2005;40:524-532.
170 Nelson H.S., et al. Improved bronchodilation with levalbuterol compared with racemic albuterol in patients with asthma. J Allergy Clin Immunol. 1998;102:943-952.
171 Newth C.J., et al. The use of cuffed versus uncuffed endotracheal tubes in pediatric intensive care. J Pediatr. 2004;144:333.
172 New C.J., et al. Eunice Shriver Kennedy National Institute of Child Health and Human Development Collaborative Pediatric Critical Care Research Network. Weaning and extubation readiness in pediatric patients. Pediatr Crit Care Med. 2009;10:1-11.
173 Noda A.K., et al. Beneficial effect of bilevel positive airway pressure on left ventricular function in ambulatory patients with idiopathic dilated cardiomyopathy and central sleep apnea-hypopnea: a preliminary study. Chest. 2007;131:1694.
174 Northway W., Rosan R., Porter D. Pulmonary disease following respiratory therapy of hyaline membrane disease. N Engl J Med. 1967;276:357.
175 Olshaker J.S. Submersion. Emerg Med Clin North Am. 2004;22:357-367.
176 Openshaw P.J.M. Potential therapeutic implications of new insights into respiratory syncytial virus disease. Respir Res. 2002;3:S15.
177 Paintal H.S., Kuschner W.G. Aspiration syndromes: 10 clinical pearls every physician should know. Int J Clin Pract. 2007;61:846-852.
178 Panitch H.B. Viral respiratory infection in children with technology dependence and neuromuscular disorders. Pediatr Infect Dis J. 2004;23:S222.
179 Parakininkas D., Rice T.B. Pneumonitis and Interstitial Lung Disease. In: Fuhrman B.P., Zimmerman J.J., editors. Pediatric critical care. Philadelphia: Mosby, 2006.
180 Paz-Priel I., et al. Thromboembolic events in children and young adults with pediatric sarcoma. J Clin Oncol. 2007;25:1519-1524.
181 Pelosi P., et al. Effects of the prone position on respiratory mechanics and gas exchange during acute lung injury. Am J Respir Crit Care Med. 1998;157:387.
182 Perket E.A. Lung Growth in Infancy and Childhood. In Rudolph C.D., et al, editors: Rudolph's pediatrics, ed 21, New York: McGraw-Hill, 2003.
183 Peter J.V., Chacko B., Moran J.L. Comparison of closed endotracheal suction versus open endotracheal suction in the development of ventilator-associated pneumonia in intensive care units: an evaluation using meta-analytic techniques. Indian J Sci. 2007;61:201-211.
184 Peters J., Kindred M.K., Robotham J.L. Transient analysis of cardiopulmonary interactions. I. Diastolic events. J Appl Physiol. 1988;64:1506-1517.
185 Peters J., Kindred M.K., Robotham J.L. Transient analysis of cardiopulmonary interaction. II. Systolic events. J Appl Physiol. 1988;64:1518-1526.
186 Petty T.L. A historical perspective of mechanical ventilation. Crit Care Clin. 1990;6:489.
187 Phipps L.M., et al. Prospective assessment of guidelines for determining appropriate depth of endotracheal tube placement in children. Pediatr Crit Care Med. 2005;6:519.
188 Popernack M.L., Thomas N.J., Lucking S.E. Decreasing unplanned extubations: utilization of the Penn State Children's Hospital sedation algorithm. Pediatr Crit Care Med. 2004;5:58-62.
189 Prakash O., Meij S. Cardiopulmonary response to inspiratory pressure support during spontaneous ventilation vs conventional ventilation. Chest. 1985;88:403.
190 Priestly M.A., Helfaer M.A. Approaches in the management of acute respiratory failure in children. Curr Opin Pediatr. 2004;16:293.
191 Putensen C., et al. The impact of spontaneous breathing during mechanical ventilation. Curr Opin Crit Care. 2006;12:13.
192 Raffini L., et al. A prospective observational study of IVC filters in pediatric patients. Pediatr Blood Cancer. 2008;51:517-520.
193 Rajpurkar M., et al. Pulmonary embolism—experience at a single children's hospital. Thromb Res. 2007;119:699-703.
194 Ralston M., et al. PALS provider manual and student CD. Dallas: American Heart Association, 2006.
195 Randolph A.G., et al. Cumulative fluid intake minus output is not associated with ventilator weaning duration or extubation outcomes in children. Pediatr Crit Care Med. 2005;6:642.
196 Randolph A.G., et al. Effect of mechanical ventilator weaning protocols on respiratory outcomes in infants and children: a randomized controlled trial. J Am Med Assoc. 2002;288:2561.
197 Ream R.S., et al. Association of nursing workload and unplanned extubations in a pediatric intensive care unit. Pediatr Crit Care Med. 2007;8:366-371.
198 Reid C., Chan L., Tweeddale M. The who, where, and what of rapid sequence intubation: prospective observational study of emergency RSI outside the operating theatre. Emer Med J. 2004;21:296.
199 Rich J.M., et al. The critical airway, rescue ventilation and the combitube: part 1. AANA J. 2004;72:17.
200 Roosevelt G. Infectious upper airway obstruction. In: Berhman R.E., Kliegman R.M., Jenson H.B., editors. Nelson textbook of pediatrics. Philadelphia: Saunders, 2004.
201 Roosevelt G.E. Acute Inflammatory upper airway obstruction (croup, epiglottitis, laryngitis, and bacterial tracheitis). In: Kleigman R.M., et al, editors. Nelson textbook of pediatrics. Philadelphia: Saunders, 2007.
202 Rosenberg A.A. The Neonate. In: Gabbe S.G., et al, editors. Obstetrics: normal and problem pregnancies. Philadelphia: Churchill Livingston, 2007.
203 Rothe C.F. Mean circulatory filling pressure: its meaning and measurement. J Appl Physiol. 1993;74:499.
204 Rotta A.T., Steinhorn D.M. Conventional mechanical ventilation in pediatrics. J Pediatr. 2007;83:S100-S108.
205 Royall J.A., Levin D.L. Adult respiratory distress syndrome in pediatric patients, I: clinical aspects, pathophysiology, and mechanisms of lung injury. J Pediatr. 1988;112:169.
206 Rubens D., Schenkman K.A., Martin L.D. Noninvasive monitoring in children. In Fuhrman B.P., Zimmerman J., editors: Pediatric critical care, ed 3, Philadelphia: Mosby-Elsevier, 2006.
207 Russell K., et al. Glucocorticoids for croup. Cochrane Database of Syst Rev. 1(CD001955), 2004.
208 Santamore W.P., Gray L. Significant left ventricular contributions to right ventricular systolic function. Chest. 1995;107:1134-1145.
209 Saragin M.J., et al. Rapid sequence intubation for pediatric emergency airway management. Pediatr Emerg Care. 2002;18:417.
210 Sarnaik A.P., et al. Pressure-controlled ventilation in children with status asthmaticus. Pediatr Crit Care Med. 2004;5:133-138.
211 Sarrell E.M., et al. Nebulized 3% hypertonic saline solution treatment in ambulatory children with viral bronchiolitis decreases symptoms. Chest. 2002;223:2015.
212 Scharf S.M., Ingram R.H.Jr. Influence of abdominal pressure and sympathetic vasoconstriction of the cardiovascular response to positive end-expiratory pressure. Am Rev Respir Dis. 1977;116:661-670.
213 Schuerer D.J.E., et al. Extracorporeal membrane oxygenation current clinical practice, coding and reimbursement. Chest. 2008;134:179.
214 Sectish T.C., Prober C.G. Pneumonia. In: Kliegman R.M., Behrman R.I., Stanton B.F., editors. Nelson textbook of pediatrics. Philadelphia: Elsevier, 2008. Available at: http:www.mdconsult.com/das/book/body/100677694-7/730226910/1608/939.html Accessed July 29
215 Sersar S.I., et al. Inhaled foreign bodies: presentation, management and value of history and plain chest radiography in delayed presentation. Head Neck Surg. 2006;134:92-99.
216 Severinghaus J.W. History of measuring O2 and CO2 responses. Adv Exp Med Biol. 2008;605:3-8.
217 Shah P.S., Ohlsson A., Shah J.P. Continuous negative extrathoracic pressure or continuous positive airway pressure for acute hypoxemic respiratory failure in children. Cochrane Database Syst Rev. 1(DC003699), 2008.
218 Shah R.K., Roberson D.W., Jones D.T. Epiglottitis in the Hemophilus influenzae type B vaccine era: changing trends. Laryngoscope. 2004;114:557.
219 Shah S., Sharieff G.Q. Pediatric respiratory infections. Emerg Med Clin North Am. 2007;25:961-979.
220 Shapiro B.A., Cane R.D., Harrison R.A. Positive end-expiratory pressure therapy in adults with special reference to acute lung injury: a review of the literature and suggested clinical correlations. Crit Care Med. 1984;12:127.
221 Shekerdemian L.S., et al. Cardiorespiratory responses to negative pressure ventilation after tetralogy of Fallot repair: a hemodynamic tool for patients with a low-output state. J Am Coll Cardiol. 1999;33:549-555.
222 Siempos I.I., Vardakas K.Z., Falagas M.E. Closed tracheal suction systems for prevention of ventilator associated pneumonia. Br J Anaesth. 2008:299-306.
223 Siempos I.I., et al. Impact of passive humidification on clinical outcomes of mechanically ventilated patients: a meta-analysis of randomized controlled trials. Crit Care Med. 2007;35:2843-2851.
224 Silvestri J.M., Weese-Mayer D.E. Apnea and SIDS. In: Rudolph C.D., et al, editors. Rudolph's pediatrics. ed 21. New York: McGraw-Hill; 2003:1934-1937.
225 Simoes E.A. Environmental and demographic risk factors for respiratory syncytial virus lower respiratory tract disease. J Pediatr. 2003;143(5 Suppl.):S118.
226 Singer M., et al. Oxygen toxicity in man: a prospective study in patients after open heart surgery. N Engl J Med. 1970;283:1473.
227 Smith M.M., et al. Flexible fiber-optic laryngoscopy in the first hours after extubation for the evaluation of laryngeal lesions due to intubation in the pediatric intensive care unit. J Pediatr Otorhinolaryngol. 2007;71:1423-1428.
228 Stahlman M.T. Clinical description of bronchopulmonary dysplasia. J Pediatr. 1979;8:829.
229 Staworn D., et al. Brain death in pediatric intensive care unit patients: incidence, primary diagnosis, and the clinical occurrence of Turner's triad. Crit Care Med. 1994;22:1301-1305.
230 Steinhorn R.H., Green T.P. Use of extracorporeal membrane oxygenation in the treatment of respiratory syncytial virus bronchiolitis: the national experience, 1983 to 1988. J Pediatr. 1990;116:338-342.
231 Stensballe L.G., et al. Atopic disposition, wheezing and subsequent respiratory syncytial virus hospitalization in Danish children younger than 18 months: a nested case-control study [electronic version]. Pediatrics. 2006;118:1360.
232 Subirana M., Solà I., Benito S. Closed tracheal suction systems versus open tracheal suction systems for mechanically ventilated adult patients. Cochrane Database Syst Rev. 4, 2007. Art. No.: CD004581. Doi: 10.1002/14651858.CD004581.pub2
233 Sur J.P., Garg R.K., Jolly N.J. Rheolytic percutaneous thrombectomy for acute pulmonary embolism in a pediatric patient. Catheter Cardiovasc Interv. 2007;70:450-453.
234 Suter P.M., Fairley B., Isenberg M.D. Optimum end-expiratory airway pressure in patients with acute pulmonary failure. N Engl J Med. 1975;292:284.
235 Swaniker F., et al. Extracorporeal life support outcome for 128 pediatric patients with respiratory failure. J Pediatric Surgery. 2000;35:197.
236 Takata M., Robotham J.L. Ventricular external constraint by the lung and pericardium during positive end-expiratory pressure. Am Rev Respir Dis. 1991;143:872-875.
237 Teague W.G. Noninvasive positive pressure ventilation: current status in paediatric patients. Paediatr Respir Rev. 2005;6:52-60.
238 Teboul J.L., et al. Estimating cardiac filling pressure in mechanically ventilated patients with hyperinflation. Crit Care Med. 2000;28:3631-3636.
239 Telford K., et al. Outcome after neonatal continuous negative-pressure ventilation: follow-up assessment. Lancet. 2006;367:1080.
240 The Red Book Online: Summaries of Infectious Diseases, Staphylococcal Infections. 2011. http://aapredbook.aappublications.org/cgi/content/full/2006/1/3.121 Accessed on June 30
241 Thierbach A.R., Pieipho T., Maybauer M. The easytube for airway management in emergencies. Prehosp Emerg Care. 2005;9:445.
242 Thill P.J., et al. Noninvasive positive-pressure ventilation in children with lower airway obstruction. Pediatr Crit Care Med. 2004;5:337-342.
243 Thompson A.E. Pediatric airway management. In: Fuhrmans B.P., Zimmerman J., editors. Pediatric critical care. Philadelphia: Mosby, 2006.
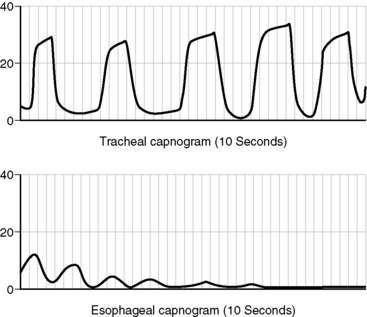
244 Thompson J., Jaffe M. Capnographic waveforms in the mechanically ventilated patient. Respir Care. 2005;50:100-109.
245 Tingay D.G., et al. Effects of open endotracheal suction on lung volume in infants receiving HFOV. Intensive Care Med. 2007;33:689-693.
246 Tobias J.D., Meyer D.J. Noninvasive monitoring of carbon dioxide during respiratory failure in toddlers and infants: end-tidal versus transcutaneous carbon dioxide. Anesth Analg. 1997;85:55-58.
247 Todisco T., et al. Treatment of acute exacerbations of chronic respiratory failure: integrated use of negative pressure ventilation and noninvasive positive pressure ventilation. Chest. 2004;125:2217.
248 Tokar B., Ozkan R., Ilhan H. Tracheobronchial foreign bodies in children: importance of accurate history and plain chest radiography in delayed presentation. Clin Radiol. 2004;59:609-615.
249 Tooley W.H. Epidemiology of bronchopulmonary dysplasia. J Pediatr. 1979;85:851.
250 Truong M.T., et al. Pediatric vocal fold paralysis after cardiac surgery: rate of recovery and sequelae. Otolaryngol Head Neck Surg. 2007;137:780-784.
251 Truwit J.D., Marini J.D. Evaluation of thoracic mechanics in the ventilated patient, I: primary measurements. J Crit Care. 1988;3:133.
252 Tsugawa J., et al. Development of acquired tracheal stenosis in premature infants due to prolonged endotracheal intubation: etiologic considerations and surgical management. Pediatr Surg Int. 2006;22:887-890.
253 Turner D.A., Arnold J.H. Insights in pediatric ventilation: timing of intubation, ventilatory strategies, and weaning. Curr Opin Crit Care. 2007;13:57.
254 Turner G.R., Levin D.L. Improvement of neurological status after pediatric near-drowning accidents. Crit Care Med. 1985;13:1080.
255 Tyler D.C. Positive end-expiratory pressure: a review. Crit Care Clin. 1983;11:300.
256 Ukleja A., Sanchex-Fermin M. Gastric versus post-pyloric feeding: relationship to tolerance, pneumonia risk, and successful delivery of nutrition. Curr Gastroenterol Rep. 2007;9:309-316.
257 Vaughan D.J., Brogan T.V. Ventilation/perfusion mismatch. In Fuhrman B.P., Zimmerman J., editors: Pediatric critical care, ed 3, Philadelphia: Mosby-Elsevier, 2006.
258 Venkataraman S.T., Khan N., Brown A. Validation of predictors of extubation success and failure in mechanically ventilated infants and children. Crit Care Med. 2000;28:2991.
259 Venkataraman S.T. Mechanical ventilation and respiratory care. In: Fuhrman B.P., Zimmerman J.J., editors. Pediatric critical care. ed 4. Philadelphia: Mosby Elsevier; 2011:657-688.
260 Venkataraman S.T. Mechanical ventilation and respiratory care. In: Fuhrman B.P., Zimmerman J.J., editors. Pediatric critical care. ed 4. Philadelphia: Mosby Elsevier; 2011:657-688.
261 Villar J., et al. A high positive end expiratory pressure, low tidal volume ventilatory strategy improves outcome in persistent acute respiratory distress syndrome: a randomized, controlled trial. Crit Care Med. 2006;34:1311.
262 Villeneuve E.J., Zed P.J. Nebulized magnesium sulfate in the management of acute exacerbations of asthma. Ann Pharmacother. 2006;40:1118-1124.
263 Ware L.B., et al. Pathogenetic and prognostic significance of altered coagulation and fibrinolysis in acute lung injury/acute respiratory distress syndrome. Crit Care Med. 2007;35:1821.
264 Ware L.B., Matthay M.A. The acute respiratory distress syndrome. N Engl J Med. 2000;342:1334.
265 Ware L.B., Matthay M.A. Clinical practice. Acute pulmonary edema. N Engl J Med. 2005;353(26):2788-2796.
266 Weber J.E., et al. A randomized comparison of helium-oxygen mixture (heliox) and racemic epinephrine for the treatment of moderate to severe croup. Pediatrics. 107(E96), 2001. Available at: http://www.pediatrics.org/cgi/content/full/107/6/e96 Accessed April 8, 2008
267 Weber T.R., Keller M.S., Fiore A. Aortic supspension (aortopexy) for severe tracheomalacia in infants and children. Amer J Surg. 2002;184:573.
268 Webster H.F. Weaning from mechanical ventilation. In: Verger J.T., Lebet R.M., editors. AACN procedure manual for pediatric acute and critical care. St Louis: Saunders-Elsevier, 2008.
269 Weiss M., et al. Tracheal tube displacement in children during head-neck movement—a radiological assessment. Br J Anaesth. 2006;96:486-491.
270 Welliver R.C. Review of epidemiology and clinical risk factors for severe respiratory syncytial virus (RSV) infection. J Pediatr. 2003;143(5 Suppl.):S112.
271 West J.B.: Gas transport by the blood. . Pulmonary pathophysiology: the essentials. ed 7, Baltimore; Lippincott Williams & Wilkins:2008
272 West J.B. Challenges in teaching the mechanics of breathing to medical and graduate students. Adv Physiol Educ. 2008;32:177-184.
273 Westley C.R., Cotton E.K., Brooks J.G. Nebulized racemic epinephrine by IPPB for the treatment of croup. Am J Dis Child. 1978;132:484.
274 Wheeler D.S., Dauplaise D.J., Giuliano J.S. An infant with fever and stridor. Pediatr Emerg Care. 2008;24:46.
275 Wheeler D.S., et al. Theophylline versus terbutaline in treating critically ill children with status asthmaticus: a prospective, randomized, controlled trial. Pediatr Crit Care Med. 2005;6:204-211.
276 Willson D.F., et al. Effect of exogenous surfactant (calfactant) in pediatric acute lung injury: a randomized controlled trial. J Am Med Assoc. 2005;293:470.
277 Wilson J., et al. Noninvasive monitoring of carbon dioxide in infants and children with congenital heart disease: end-tidal versus transcutaneous techniques. J Intensive Care Med. 2005;20:291-295.
278 Wratney A.T., et al. The endotracheal tube air leak test does not predict extubation outcome in critically ill pediatric patients. Pediatr Crit Care Med. 2008;9:490-496.
279 Wright M.L., Romano M.J. Ventilator-associated pneumonia in children. Sem Pediatr Infectious Dis. 2006;17:58-64.
280 Writing Group for the Christopher Study Investigators. Effectiveness of managing suspected pulmonary embolism using and algorithm combining clinical probability, d-dimer testing and computed tomography. J Am Med Assoc. 2006;295:172-197.
281 Wunsch J., Mapstone J. High-frequency ventilation versus conventional ventilation for treatment of acute lung injury and acute respiratory distress syndrome. Cochrane Database Syst Rev. 1(CD004085), 2004.
282 Youngquist S., Gausche-Hill M., Burbulys D. Alternative airway devices for use in children requiring prehospital airway management. Pediatr Emerg Care. 2007;23:250.
283 Yuksel H., et al. A pediatric case of pyomyositis presenting with septic pulmonary emboli. Joint Bone Spine. 2007;74:491-494.
284 Zelicof-Paul A., et al. Controversies in rapid sequence intubation in children. Curr Opin Pediatr. 2005;17:355.
285 Zuckerbraun N.S., Saladino R.A. Pediatric drowning: current management strategies for immediate care. Clin Pediatr Emerg Med. 6, 2005. Available at: http://www.mdconsult.com/das/article/body/101018360-3/jorg=journal&source=&sp=15573844&sid=731277684/N/473050/1.html?issn=1522-8401 Accessed July 28, 2008
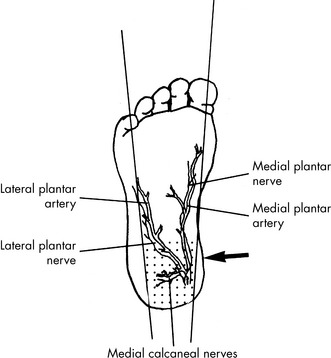
 matching.
matching.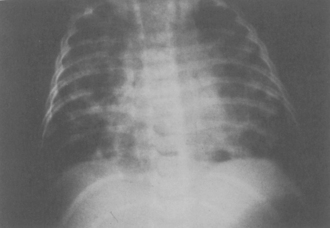
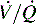 mismatching. Intubation and mechanical ventilation may be necessary.
mismatching. Intubation and mechanical ventilation may be necessary.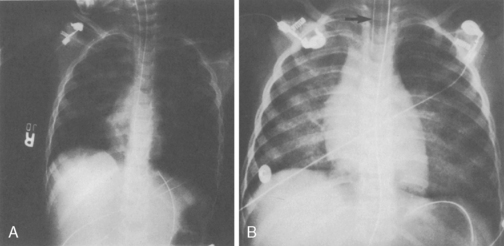
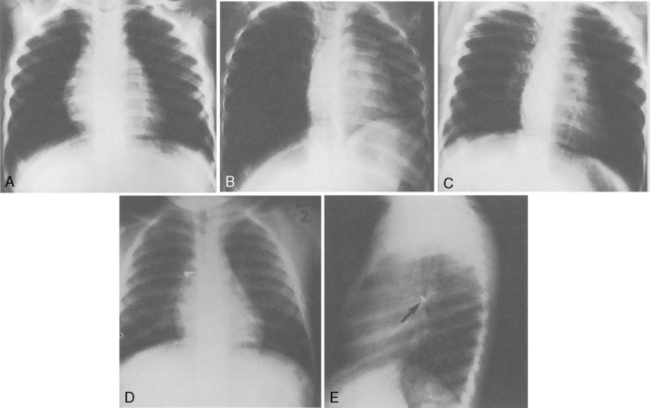
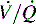 ) abnormalities. The duration and extent of the obstruction will determine the severity of signs and symptoms. In the acute phase, or the first 48 hours of this process, cardiac output is the main determinate in distribution of
) abnormalities. The duration and extent of the obstruction will determine the severity of signs and symptoms. In the acute phase, or the first 48 hours of this process, cardiac output is the main determinate in distribution of 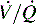 abnormalities, rather than redistribution of pulmonary blood flow to nonoccluded areas or increasing or redistributing ventilation to areas maintaining perfusion.
abnormalities, rather than redistribution of pulmonary blood flow to nonoccluded areas or increasing or redistributing ventilation to areas maintaining perfusion.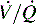 mismatch and acute changes in pulmonary function.
mismatch and acute changes in pulmonary function.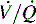 ratio increases.
ratio increases.
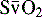 ) is a reflection of O2 delivery. This mixed venous O2 saturation reflects the balance between O2 delivery and O2 utilization. The superior vena caval O2 saturation (ScvO2) is often used as a surrogate for the mixed venous O2 saturation (see
) is a reflection of O2 delivery. This mixed venous O2 saturation reflects the balance between O2 delivery and O2 utilization. The superior vena caval O2 saturation (ScvO2) is often used as a surrogate for the mixed venous O2 saturation (see 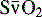 is possible using a pulmonary artery catheter with fiberoptic light and sensor, but pulmonary artery catheters are rarely used in children. Generally venous O2 saturation samples are obtained from a central venous catheter, typically from the SVC. Pediatric fiberoptic catheters allow for continuous monitoring of central venous saturation and can facilitate rapid detection of a fall in ScvO2, which can be associated with a compromise in O2 delivery (i.e., either pulmonary function or cardiac output) or an increase in oxygen consumption.
is possible using a pulmonary artery catheter with fiberoptic light and sensor, but pulmonary artery catheters are rarely used in children. Generally venous O2 saturation samples are obtained from a central venous catheter, typically from the SVC. Pediatric fiberoptic catheters allow for continuous monitoring of central venous saturation and can facilitate rapid detection of a fall in ScvO2, which can be associated with a compromise in O2 delivery (i.e., either pulmonary function or cardiac output) or an increase in oxygen consumption.