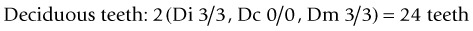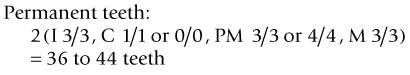CHAPTER 5 Dental anatomy
Introduction
Equine dental nomenclature
Adult mammals have four types of teeth, termed incisors, canines, premolars (PM) and molars (M), in a rostrocaudal order.1 Teeth embedded in the incisive (premaxillary) bone are by definition termed incisors. The most rostral teeth in the maxillary bone are the canines. In horses, the main three premolars have evolved to become more complex and morphologically identical to the molars (i.e., molarization of premolars) to facilitate grinding of foodstuffs. Consequently, in horses, premolars 2–4 (Triadan 06–08) and the three molars (Triadan 09–11) can be collectively termed cheek teeth. Each type of tooth has certain morphological characteristics and specific functions. Incisor teeth are specialized for the prehension and cutting of food, and the canine teeth are for defence and offence (for capture of prey in carnivores). Equine cheek teeth function as grinders for mastication. The occlusal or masticatory surface is the area of tooth in contact with the opposing teeth; the term coronal refers to the crown. The anatomical crown is that part of the tooth covered by enamel and in brachydont (short crowned) teeth, such as in humans, is usually the same as the clinical (erupted) crown, i.e., the erupted part of the tooth. However, in equine teeth (hypsodont – long crowned), especially young teeth, most of the crown is unerupted and is termed unerupted or reserve crown, and a smaller proportion (circa 10–15% in young adult horses) of crown is clinical crown. The term occlusal (‘coronal’ is a much less satisfactory term for hypsodont teeth) is used when referring to direction towards the occlusal surface. The reserve crown can be subdivided into alveolar crown (i.e., that part lying in the alveolus) and the gingival crown, i.e., that part which has erupted from the alveolus, but which is still lying sub-gingivally.2
Apical refers to the area of tooth furthest away from the occlusal surface, i.e., the area where the roots later develop and is the opposite of occlusal. Lingual refers to the medial aspect (area closest to the tongue) of all the lower teeth, whilst palatal refers to the same aspect of the upper cheek teeth. Buccal (aspect closest to cheeks) refers to the lateral aspect of both upper and lower (cheek) teeth, whilst labial refers to the rostral and rostrolateral aspect of teeth (incisors and canines only in horses) close to lips. The terms interproximal or interdental refer to the area of teeth that face the adjoining teeth in the same arcade (incisors) or row (cheek teeth). The terms mesial and distal, which refer, respectively, to the surfaces of teeth that face towards and away from an imaginary line between the central incisors, are satisfactory for equine incisors – that form a true arch. However, these terms are unsatisfactory for the equine cheek teeth, because they do not form part of a continuous dental arch as they are separated from the incisors by the ‘interdental space (‘physiological diastema’, ‘bars of mouth’). The term cheek teeth row is a more appropriate term to describe the straight rows of six cheek teeth.
Equine dental evolution
The evolution of equine dentition is comprehensively covered in Chapter 1, and the functional consequences of this dental evolution are discussed in Chapter 6; nevertheless, some salient anatomical aspects of the differentiation of brachydont into hypsodont teeth are briefly discussed here. Following ingestion of their coarse forage diet, the necessary grinding down of this foodstuff to a small particle size (the average length of fibers in equine feces is just 3.7 mm)3 to allow more efficient endogenous and microbial digestion, causes a high degree of wear on their cheek teeth. However, unlike ruminants that can later regurgitate their food to allow further mastication, horses have only one opportunity to effectively grind their foodstuffs.
Brachydont teeth (permanent dentition) fully erupt prior to maturity and are normally long and hard enough to survive for the life of the individual because they are not subjected to the prolonged and high levels of dietary abrasive forces that herbivore teeth must contend with. In contrast, hypsodont teeth slowly erupt over most of the horse’s life at a rate of 2–3 mm/year,4,5 which is similar to the rate of attrition (wear) on the occlusal surface of the tooth, provided that the horse is on a grass (or some alternative fibrous diet, e.g., hay or silage) rather than being fed high levels of concentrate food. The latter type of diet reduces the rate of occlusal wear and also restricts the range of lateral chewing actions6 and thus dental overgrowths can occur. Both brachydont and hypsodont teeth have a limited growth period (although this period is very prolonged in the latter group) and thus are termed anelodont teeth. A further evolutionary progression to cope with highly abrasive diets, as has occurred in some rodents such as rabbits (and some extinct equid lineages), is the presence of teeth that continually grow throughout all of the animal’s life, that are termed elodont teeth.
Many brachydont teeth have a distinct neck between the crown and root, a feature that could not be present in permanent hypsodont teeth that have a prolonged eruption period. At eruption, hypsodont teeth have no true roots and in this text the term root specifically refers to the apical area which is enamel free.7,8 The formation of roots in equine teeth permits further dental growth for some year or so after these teeth erupt, in addition to the very prolonged eruption of these teeth for most of the horse’s life. The terms apical or periapical are much more appropriate to describe this area of equine teeth that, for example, commonly develop apical infections of the mandibular 07s and 08s (2nd and 3rd cheek teeth) even prior to the development of any roots. About 25 % of equine mandibular cheek teeth still have no root development even 12 months following eruption.9
Because of the marked physiological wear (attrition) on the surface of hypsodont teeth, exposure on the occlusal surface of enamel ridges, and also of dentin and cement (cementum) is inevitable and leads to the presence of alternate layers of these three calcified dental tissues on the occlusal surface. This is in contrast to the sole presence of enamel on the occlusal surface of brachydont teeth. The presence of infolding of the peripheral enamel, and also of enamel cup-like infoldings (infundibula) in the upper cheek teeth and all incisors also increases the amount and irregularity of exposed enamel ridges on the occlusal surface. This peripheral enamel infolding is greater in mandibular cheek teeth to compensate for the lack of enamel infundibulae that are present in the maxillary cheek teeth.10 This feature confers additional advantages to hypsodont teeth, as the different calcified tissues wear at different rates (enamel slowest, dentin and cementum fastest) and therefore a permanently irregular occlusal surface that is advantageous in the grinding of coarse fibrous foodstuffs is created by a self-sharpening mechanism.
Embryology of teeth
Dental development (dentogenesis) involves several sequential processes, including epithelial-mesenchymal interaction, growth, remodeling and calcification of tissues until a tooth is fully developed.11,12,13 During dental development, the tooth germ undergoes a series of distinct, consecutive events termed the initiating, morphogenetic and cytodifferentiative phases. These phases occur in all types of mammalian dentition;14 however, their timing and termination vary, i.e., compared to brachydont teeth, hypsodont teeth have a delayed termination of the morphogenetic and cytodifferentiative stages (at their apical region), whilst in elodont teeth (such as in some rodents), these stages continue throughout all of the animal’s life. Tooth formation begins by the development of a horseshoe-shaped, epithelial thickening along the lateral margin of the fetal oral cavity. This epithelial thickening (termed the primary epithelial band) invaginates into the underlying mesenchymal tissue to form two distinct ridges, the vestibular lamina, and (caudal to it) the dental lamina. The dental lamina produces a series of epithelial swellings called tooth buds along its buccal margin. This stage is known as the bud stage of tooth development (Fig. 5.1). At this stage, a mesenchymal cell proliferation develops beneath the hollow ectodermal tooth buds and invaginates into these tooth buds, which then develops into inverted cap-shaped structures called the enamel organs. This is termed the cap stage of dental development (Fig. 5.1).
All deciduous teeth and the permanent molars develop from the enamel organ of the dental laminae. However, permanent incisors, permanent canines, and permanent premolars are formed from separate enamel organs that are derived from lingual (medial) extensions of the dental laminae of the deciduous teeth (Fig. 5.1). Consequently, the deciduous incisors are normally displaced labially (towards the lips) by the erupting permanent incisors.
After formation of the enamel organ, the mesenchymal cells continue to proliferate within the concave aspect of the enamel organ, and are then termed the dental papilla, a structure that is later responsible for dentin and pulp formation. These cells now also extend peripherally, as a structure termed the dental sac (follicle), which surrounds and protects the enamel organ and dental papilla until tooth eruption occurs (Fig. 5.2).1,15 The enamel organ, dental papilla and dental sac are together termed the tooth germ, with each germ responsible for an individual tooth.
The enamel organ proliferates further, and in brachydont teeth now assumes a concave, bell-like shape, which is termed the bell stage of dental development. At this stage, the concavity of the enamel organ increases, while the mesenchymal cells of the dental papilla invaginate further into its hollow aspect (Fig. 5.1). Additionally, in some equine teeth (all incisors and maxillary cheek teeth), invaginations of enamel epithelium that will later become infundibula develop from the convex aspect of the ‘bell’ into the papilla (one per incisor and two per upper cheek teeth). Equine cheek teeth have multiple cusps (raised occlusal areas) that arise from protrusions on the convex aspect of the bell. The enamel organ in equine incisors and in all brachydont teeth is circular on transverse section; however, the enamel organ of equine cheek teeth (which are rectangular to square on transverse section) develops peripheral infoldings16 that later produce the infolded peripheral enamel.
Most cytodifferentiative events in the tooth germs occur during the transitional period between the cap and bell stages. The ectodermal cells lining the concave aspect of the enamel organ become the internal enamel epithelium, and the cells lining the convex aspect of the enamel organ form the external enamel epithelium.12 Between them lies a third layer containing star-shaped cells with large intracellular spaces, termed the stellate reticulum (Fig. 5.1), which has nutritive and mechanical functions in enamel development. The cells of the internal dental epithelium develop into tall columnar cells with large, proximally located nuclei. This development induces alterations at the molecular level in the underlying dental papilla whose uppermost cells now rapidly enlarge, becoming odontoblasts. The first layer of dentin is now laid down along the basal membrane, which then disintegrates. These events reciprocally induce the overlying internal enamel epithelial cells to differentiate into ameloblasts which now begin to produce enamel.17
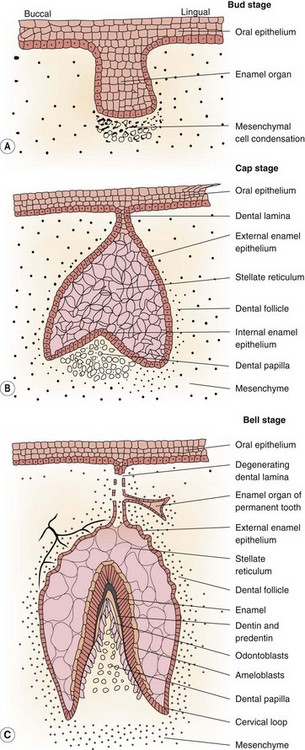
Fig. 5.1 The three early stages of development of a brachydont or hypsodont tooth.
(Reproduced from Kilic72 with permission.)
The ameloblasts initially deposit a structureless enamel layer and then migrate away from the enamel-dentinal interface, and form a projection termed Tome’s process at their distal aspect. Secretions from the proximal aspect of Tome’s process form interprismatic enamel, and secretions from the surface of Tome’s process form the enamel prisms. The development of enamel and dentin (and later, also of cement) occurs in two consecutive phases, the secretion of extra cellular matrix of mucopolysaccharides and organic fibers, which is then followed by its mineralization.18,19
Odontoblasts, like ameloblasts and cementoblasts (that produce cement), are end cells, meaning that they cannot further differentiate into other cell types. During dentin deposition, the basal aspects of odontoblasts gradually become thinner and form long fine cytoplasmic extensions termed odontoblast processes, that remain within the dental tubule, whilst the odontoblast cell body gradually moves centrally on the peripheral of the pulp horn, remaining on the surface of the developing dentin.13
In multi-cusped teeth (such as equine cheek teeth) mineralization begins independently at each cusp tip (Figs 5.2 & 5.3) and then merges, as calcification progresses down towards the amelodentinal (enamel-dentin) junction.1 As dentin and enamel deposition continues, odontoblasts and ameloblasts move in opposite directions and thus avoid becoming entrapped in their own secretions. Radiography has shown the calcification of equine deciduous cheek teeth buds (three in each quadrant) to be underway by the 120th day of fetal life and to be completed by 240 days.20 The deciduous 06 (PM2) germs are largest, indicating that they develop first. Calcification of the first permanent tooth bud (09s) begins about 6 months later.20
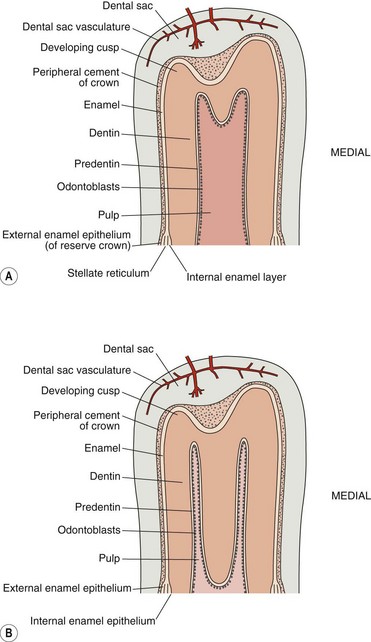
Fig. 5.2 Two stages of the development of a multicusped hypsodont tooth without an infundibulum (i.e., a lower cheek tooth) showing the presence of coronal cement and enamel that are covered by the dental sac. The large common pulp chamber (A) later develops separate pulp horns (B) due to deposition of dentin by the odontoblasts within the common pulp chamber.
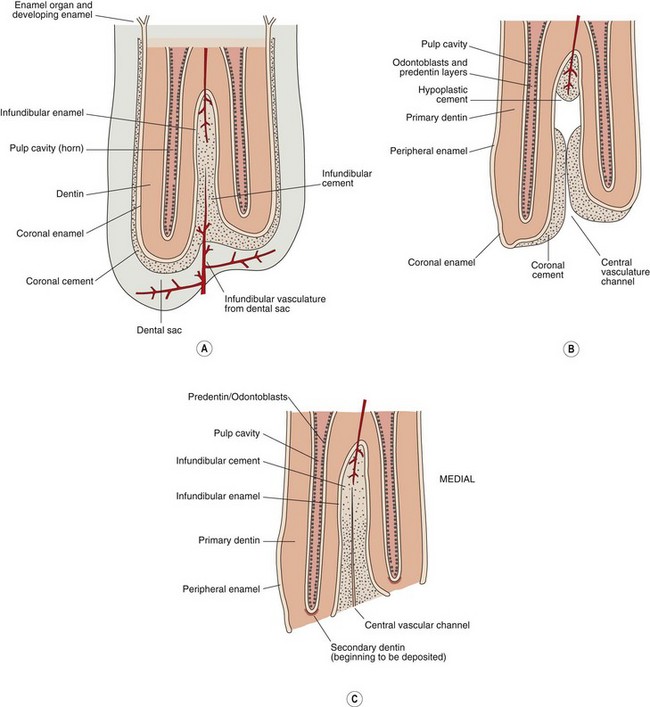
Fig. 5.3 The crown and occlusal surface of a multicusped hypsodont tooth with an infundibulum (i.e., an upper cheek tooth) (a) immediately prior to eruption (b) immediately following eruption, showing loss of the dental sac over the occlusal surface and (c) following wear of the primary occlusal surface to expose the secondary occlusal surface which is the permanent occlusal surface in hypsodont teeth. Note the additional apical blood supply to the infundibulum.
In brachydont teeth, vascularization begins at the periphery of the tooth germs at the early cap stage, and blood vessels then grow into the dental sac and dental papilla.13 Until this stage, the enamel epithelium is supplied by small mesenchymal capillaries. Once dentinal and enamel mineralization begins, the connection between the enamel epithelium and the dental papilla is completely lost. The developing enamel is now solely nourished by the vasculature of the surrounding dental sac (Figs 5.3 & 5.4).
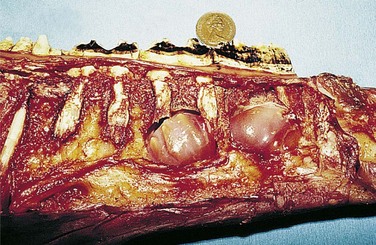
Fig. 5.4 Dissected hemimandible of a yearling Thoroughbred showing the tooth germs of the permanent 06 and 07 mandibular cheek teeth developing beneath and causing resorption of their overlying temporary counterparts. Some calcified developing cusps can be seen on the developing teeth through the semi-transparent dental sac. Note the resorption of the apices of the overlying deciduous teeth by the developing permanent teeth.
After crown formation is completed in brachydont teeth, the external and internal enamel epithelial cells at the cervical region proliferate down over the dental papilla as a double layer of cells that (at this site) is termed Hertwig’s epithelial root sheath (Fig. 5.2). This epithelium induces the underlying mesenchymal cells to differentiate into odontoblasts, which produce dentin.13 With the progressive distal disintegration of Hertwig’s epithelial root sheath, the dental sac cells come into direct contact with dentin. Interaction between these two tissues now induces the cells of the dental sac (mesenchymal cells) to convert into cementoblasts and then to lay down cement (cementum).11,15 In equine teeth cement deposition occurs over the entire crown, including over the future occlusal surface just prior to eruption16 (Fig. 5.3). When the equine tooth has reached its full length, the epithelial root sheath disintegrates, and no further enamel can be formed.
In the infundibula (two in all upper cheek teeth and one in all incisors), cement deposition proceeds by cementoblasts, that are nourished by vasculature from the dental sac (Fig. 5.3) and also via openings in the apical aspects of the infundibula.21 Immediately after eruption (or following loss of the overlying deciduous tooth), the soft tissue of the dental sac is quickly destroyed by mastication and consequently infundibular cement no longer has any occlusal blood supply (Fig. 5.3). The cement at the occlusal aspect of the infundibulum can now be regarded as an inert or ‘dead’ tissue, but cement deposition can continue more apically, possibly for some years if an apical infundibular blood supply is present. Because of the frequent absence of complete filling of more central areas of the cheek teeth infundibula, the term central infundibular cemental hypoplasia has been advocated for this feature22 as discussed further in the cementum section.
Dental structures
Enamel
Enamel is the hardest and most dense substance in the body. Due to its high (96–98 %) mineral content, it is almost translucent, and gains its color from that of the underlying dentin. Being ectodermal in origin, much of its limited organic component is composed of the keratin family of proteins, in contrast to the largely collagenous proteins of dentin and cement (i.e., connective tissue – reflecting their mesodermal origin). In equine teeth, enamel (except on the occlusal surface) is usually covered by dull, chalk-like peripheral cement. However, at the rostral aspect of the incisors, and more occlusally on the cheek teeth, this peripheral cement is usually worn away, thus exposing the shiny underlying enamel. The deciduous incisors often have little overlying cementum and thus appear whiter and shinier than their permanent successors. Enamel, with its high mineral content and absence of cellular inclusions (unlike dentin or cement) can be regarded as almost an inert or ‘dead’ tissue. Therefore, as the ameloblasts die off once the tooth is fully formed, enamel has no ability to repair itself. Enamel is almost fully composed of impure hydroxyapatite crystals (Fig. 5.5) which are larger than the hydroxyapatite crystals of dentin, cement, or bone. Enamel hydroxyapatite crystals are arranged both into structured prisms which may be contained in a prism sheath, and also into less structured, interprismatic enamel. Different species, different teeth within a species and even different areas of teeth in an individual can have differently shaped enamel prisms or different arrangements of prismatic and interprismatic enamel, which form the basis for enamel classification in equidae.23

Fig. 5.5 Scanning electron micrograph of Equine Type-1 enamel. This shows parallel rows of enamel prisms (P) lying on flat plates of interprismatic enamel (IP). The enamel crystals within the enamel prisms are oriented parallel to the long axes of the prisms while the enamel crystals of the interprismatic enamel plates are oriented at right angles to the prisms (×3860).
Equine enamel is composed of two main types termed Equine Types-1 and -2 enamel, with smaller amounts of a third enamel, i.e., Equine Type-3 enamel sometimes detected.23 Equine Type-1 enamel is present on the medial aspect of the enamel folds, i.e., at the amelodentinal junction. It is composed of prisms that are rounded or oval on cross section and lie in parallel rows between flat plates of dense interprismatic enamel (Figs 5.5 & 5.6). Equine Type-2 enamel is present on the periphery of the enamel layer, i.e., at the amelocemental (enamel to cement) junction, and is composed solely of enamel prisms ranging from horseshoe to keyhole in shape (Fig. 5.7) with no interprismatic enamel present. Equine Type-3 enamel is composed of prisms completely surrounded by large quantities of interprismatic enamel in a honeycomb-like structure and is inconsistently present as a thin layer at both the amelodentinal and amelocemental junctions (Fig. 5.7).
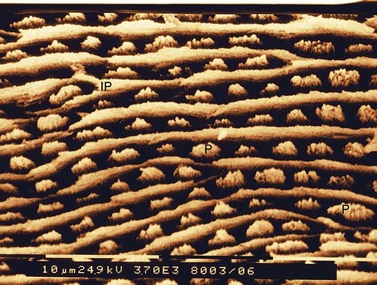
Fig. 5.6 Scanning electron micrograph of Equine Type-1 enamel showing interprismatic plates (IP) alternating with rows of prisms (P). Note the convergence and branching of some of the interprismatic enamel plates (×3700).
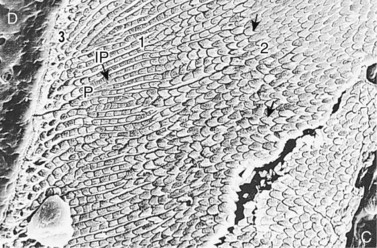
Fig. 5.7 Scanning electron micrograph of a section of an equine tooth showing dentin (D) enamel and cement (C). A thin layer of Equine Type-3 enamel (3) is visible on the left at the amelodentinal junction. Adjacent to this area is a wider layer of Equine Type-1 enamel (1) showing interprismatic enamel (IP) that is contiguous with Type-3 enamel and enamel prisms (P). To the right is a wider layer of Equine Type-2 enamel (2) that in this area has horseshoe-shaped prisms (↑) (×482).
(Reproduced from Kilic et al23 courtesy of the Editor of Equine Veterinary Journal.)
The distributions of Equine Type-1 and -2 enamels vary throughout the teeth, with Equine Type-2 enamel increasing in thickness in the peripheral enamel folds (ridges) and decreasing where these folds invaginate towards the center of the tooth (Figs 5.8 & 5.9). Almost all enamel folds contain both Type-1 and Type-2 enamel; however, increased amounts of Equine Type-1 enamel are present in the upper cheek teeth. Similar quantities of Equine Type-1 and-2 enamel occur in the lower cheek teeth, whereas incisor enamel is composed almost solely of Equine Type-2 enamel. Equine Type-1 prisms are oriented at angles of approximately 45° to both the amelodentinal junction and the occlusal surface, but bundles of Equine Type-2 enamel prisms are oriented at a very wide variety of oblique angles.23
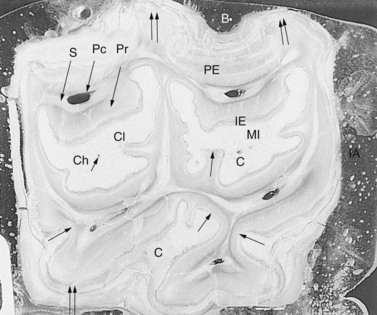
Fig. 5.8 Transverse subocclusal section of a methyl methacrylate embedded upper 09 of an 18-year-old horse. The rostral (mesial) infundibulum (MI) and the caudal (distal) infundibulum (CI) are surrounded by infundibular enamel (IE), and the infundibular cement has a central channel (Ch). Five pulp cavities (Pc) are present and are surrounded by areas of secondary dentin (S) that in turn are surrounded by primary dentin (Pr). Both the peripheral enamel (PE) and infundibular enamel (IE) are thicker at the palatal (Pa) and buccal (B) aspects than at the interdental aspects (IA). Additionally, the enamel is thicker in ridges (↑↑) than in invaginations (↑) (×4).
(Reproduced from Kilic72 with permission.)
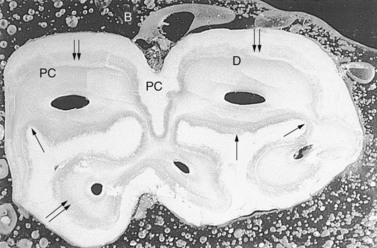
Fig. 5.9 Transverse section 2 cm beneath the occlusal surface of a methyl methacrylate embedded lower fourth cheek tooth of an 8-year-old horse. The enamel (peripheral only) is thickest (↑↑) in regions that are parallel to the long axis of the mandible and thinnest (↑) in invaginations of enamel. One peripheral infolding is apparent on the buccal (B) aspect while two deeper infoldings are present on the lingual (L) aspect. PC, peripheral cementum; D, dentin (×4).
(Reproduced from Kilic70 with permission.)
Although enamel is the hardest substance in the mammalian body, it is brittle. The closely packed prisms of Equine Type-1 enamel form a composite structure including dense interprismatic plates that confer very strong wear resistance. However, these often-parallel rows of enamel prisms and interprismatic enamel are susceptible to cracking along prismatic and interprismatic lines. One adaptive process to prevent such cracks, which is particularly noticeable in Equine Type-2 enamel, is the presence of enamel decussation (which means interweaving, with changes of direction of bundles of enamel prisms that run in three-dimensions) (Fig. 5.10). In contrast, Equine Type-1 enamel has no decussation. Equine incisors are smaller and flatter than cheek teeth, have less support from adjacent teeth and yet undergo great mechanical stresses during prehension that could readily cause enamel cracks. Therefore, it is not surprising that they are largely composed of Equine Type-2 enamel prisms. Cheek teeth primarily have a grinding function, and so the presence of enamel that confers high wear resistance is more essential, and this requirement is fulfilled by the high proportion of Equine Type-1 enamel present in cheek teeth.23 Close examination of cheek teeth enamel sometimes shows the presence of fine transverse fissures (micro fractures) through the peripheral enamel,24 which does not appear to be clinically significant, as the progression of these cracks through the remaining part of the tooth often appears to be prevented by the adjacent cementum and dentin, but some fissure fractures can lead to pulpar exposure and thus to apical infection.25,26 Donkeys have similar enamel types and distribution of enamel types to horses except that their maxillary cheek teeth have similar proportions of Equine Type-1 and Type-2 enamels.27
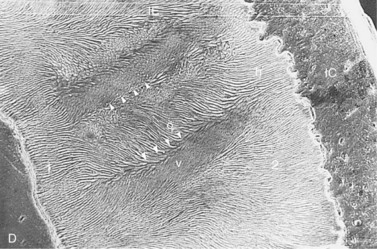
Fig. 5.10 Scanning electron micrograph of a section of an equine incisor tooth showing dentin (D) infundibular enamel (IE) and infundibular cement (IC). A thin layer of Equine Type-1 enamel is present on the left (1). The bulk of the enamel is Equine Type-2 (2) and this is oriented at a wide variety of angles including horizontal (h) obliquely (o) and vertically (v) relative to the occlusal surface. The bands of enamel oriented obliquely and vertically form alternating bands that are oriented perpendicular to the amelodentinal and amelocemental junctions with the changes in direction of the enamel bands are demarcated by grooves (∇∇∇∇) (×131).
(Reproduced from Kilic et al23 courtesy of the Editor of Equine Veterinary Journal.)
In equine cheek teeth, both peripheral and infundibular enamel are about three times thicker in areas where they are parallel to the long axis of the maxillae or mandible, than where perpendicular to this axis, i.e., are invaginated into the tooth.23 It appears that enamel may have evolved to become thinner or thicker in certain regions of the tooth in response to the level of localized masticatory forces. However, enamel thickness remains constant throughout the length of the tooth, therefore, as the animal ages the enamel thickness remains constant at the different sites in the transverse plane.
Dentin
The bulk of the tooth is composed of dentin, a cream colored, calcified tissue composed of approximately 70 % minerals (mainly hydroxyapatite crystals) and 30 % organic components (including collagen fibers and mucopolysaccharides) and water. The latter content is obvious in dried equine teeth specimens where the dentin (and also cement) develop artefactual cracks following loss of their water content. The mechanical properties of dentin, including its tensile strength and compressibility, are highly influenced by the arrangements and relationships of its matrix collagen fibers (Fig. 5.11), other organic components, water content and its calcified components, with the heterogeneity of its structure contributing to its overall strength.28 Electron microscopic examination of equine dentin shows that it contains both calcified fibers and calcospherites. In equine teeth, the presence of dentin (and also cement) interspersed between the hard but brittle enamel layers forms an elegant laminated structure (a biological ‘safety glass’) allowing the two softer calcified tissues (dentin and cementum) to act as ‘crack stoppers’ for the enamel28 as well as creating an irregular occlusal surface, due to the differential wear between the hard enamel and the softer cementum and dentin.
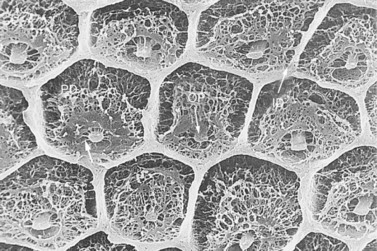
Fig. 5.11 Scanning electron micrograph of partially decalcified dentin. The hexagonal shaped intertubular dentin (ID) has a compact appearance. A network of collagenous fibers is apparent in the fully decalcified intratubular dentin (formerly termed peritubular dentin) (PD) that are attached to the odontoblast processes (OP) (×2020).
(Reproduced from Kilic72 with permission.)
Dentin can be divided into three main types: primary dentin; secondary dentin that can be subdivided into regular and irregular secondary dentin;27,29,30 and tertiary dentin that forms in response to local insults that in turn can be subdivided into reactionary tertiary dentin if formed by pre-existing odontoblasts or reparative tertiary dentin if formed by previously undifferentiated mesenchymal cells.31,32 Even in a morphological resting phase, odontoblasts remain capable of synthesizing dentin throughout their lives if appropriately stimulated.13,33 Similarly undifferentiated connective tissue cells of the pulp can also be stimulated to differentiate into odontoblasts. In equine teeth, odontoblasts (Fig. 5.12) synthesize regular secondary dentin and also irregular secondary dentin on the periphery of the pulp horn throughout most of the life of the tooth, which gradually reduces the size of the pulp cavity and thus of the pulp and eventually fully occludes the pulp horn (Fig. 5.13). In equids, irregular secondary dentin is a physiological dentin that is laid down last, subocclusally in the centre of the pulp cavity and, along with regular secondary dentin, it prevents pulpal exposure with normal wear (attrition). The physiological nature of irregular secondary dentin has been shown in horse and donkey teeth, when irregular secondary dentin was present sub-occlusally in every normal cheek tooth examined histologically (Fig. 5.13).27,32 A recent study examining cheek teeth from 17 skulls (age range 4–30) showed the median depth of occlusal secondary dentin in mandibular and maxillary cheek teeth to be 10.8 and 9.0 mm, respectively, and does not appear to increase in thickness with age.34 These values are similar to the mean occlusal secondary dentin depth determined (by CAT examinations) in donkeys of 14.6 and 13.4 mm in mandibular and maxillary cheek teeth, respectively.10 However, the donkey study showed a trend towards thicker secondary dentin in older donkeys.10
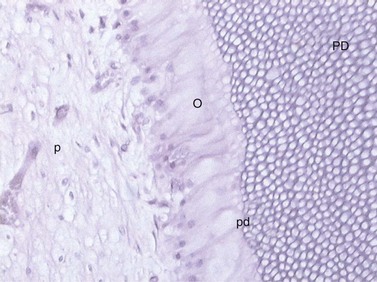
Fig. 5.12 Odontoblast cells (O) withdrawing toward the center of the pulp (p) laying down a secretory matrix i.e., a paler staining layer of predentin (pd) adjacent to the primary dentin (PD).
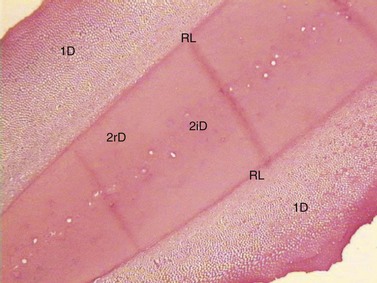
Fig. 5.13 Decalcified transverse histological section through a mandibular CT pulp horn that has become completely filled with dentin. Primary equine dentin (1D) is peripheral to regular secondary dentin (2rD) and irregular secondary dentin (2iD) with a resting line (RL) present between them (H and E).
This process has great practical significance because the occlusal surface of equine teeth would otherwise develop exposure of the pulp horns and vital pulp, due to normal attrition on the occlusal aspect and normal eruption. Following insults to teeth, such as traumatic injury, dental caries, or excessive attrition, primary dentin can respond by developing sclerosis of the primary dentinal tubules to prevent microorganisms or their molecular products gaining access to the pulp, a defensive feature that is additional to the deposition of tertiary dentin.
As noted, the cream color of dentin largely contributes to the color of brachydont teeth. Because equine primary dentin contains very high levels of heavily mineralized intratubular dentin, it too has an almost translucent appearance, similar to enamel. In contrast, the less mineralized regular secondary dentin (produced at the site of the former pulp cavity) has a dull opaque appearance. Secondary dentin also absorbs pigments from foods such as grass (but little from grains), which give it a dark brown color that is obvious in the so-called ‘dental star’ of incisors35 or in the brown linear areas of secondary dentin that develop on the occlusal surface of cheek teeth that are in wear (Fig. 5.14).
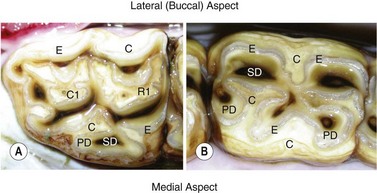
Fig. 5.14 A Triadan 11 maxillary cheek tooth (Triadan 211) showing 7 pulp horns and a Triadan 08 mandibular cheek tooth with (the standard) 5 pulp horns. The cheek tooth pulp numbering system is presented in Fig. 5.20. C, cementum; E, enamel; PD, primary dentin; SD, secondary dentin.
Dentin is composed of several distinct structures, including dentinal tubules, which are its characteristic histological feature, intratubular dentin (which lines the tubule walls), intertubular dentin (which lies between the tubules) and odontoblast processes. Dentinal tubules extend from the pulp cavity across the width of the tooth to the enamel at the amelodentinal junction. The odontoblasts reside in the predentin at the periphery of the pulp cavity, but their odontoblast processes extend through the dental tubules (Figs 5.11, 5.12 & 5.15) as far as the enamel, sometimes subdividing into two or three tubules and displaying a sharp curvature just before reaching the amelodentinal junction. There is a debate on whether the odontoblast processes reach as far as the amelodentinal junction in other species, but in the horse it appears that the odontoblast processes do reach this far.36 Because there is an intimate association between the pulp and dentin that act as a single functional unit, the term pulpodentinal complex is appropriately used for these two tissues.30 Because its tubules contain odontoblast processes, dentin is considered to be a sensitive living tissue and thus mechanical interference with dentin, e.g., reducing larger overgrowths that contain dentin, can damage sensitive odontoblast processes and can thus potentially cause pain.37
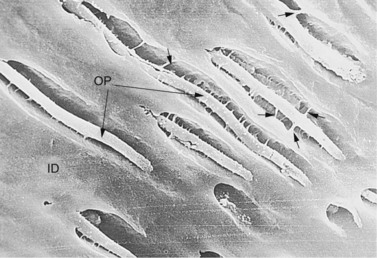
Fig. 5.15 Scanning electron micrograph of an untreated dentinal section showing a longitudinal profile of dentinal tubules containing odontoblast processes (OP) that are attached to the intertubular dentin (ID) by calcified fibrils (↑) (×1010).
(Reproduced from Kilic et al36 courtesy of the Editor of Equine Veterinary Journal.)
In brachydont species, odontoblast processes or their surrounding fluid can convey pain signals from insulted (e.g., by excessive heat or cold, trauma, infection) dentin to the pulp, by incompletely understood mechanisms. In horses, where exposed dentin constitutes a major part of the occlusal surface, it is most unlikely that such a pain-producing mechanism exist on the normal occlusal surface. It is interesting that on the occlusal surface of normal equine teeth, apparently intact, odontoblast-like processes are visible protruding from the dentinal tubules of primary and regular secondary dentin (Fig. 5.16), even though this area is constantly exposed to oral microbial and biochemical insults38 and pulpar infection is rare. A possible explanation for their apparently undamaged morphology is that they have become calcified. Some studies have suggested that these structures are not odontoblast processes, but are in fact collagen fibrils, termed laminae limitantes, that are the un-mineralized inner layer of intratubular dentin.39 However, even if microorganisms could enter patent dentinal tubules on the occlusal surface, they may not reach the pulp cavity because the dentinal tubules are sealed by a smear layer of ground dental tissue and additionally, retrograde flow of fluid from the pulp through the dentinal tubules to the occlusal surface40 may also prevent descent of microorganisms down these tubules. Irregular (reparative) secondary dentin is less organized than primary dentin and contains no odontoblast processes as its dentinal tubules are fully obliterated. This type of dentin can fully seal off the pulp from the oral environment.

Fig. 5.16 Scanning electron micrograph of the occlusal surface of an equine cheek tooth showing regular secondary dentin that has been etched to remove the organic pellicle. Almost all the dentinal tubules contain protruding odontoblast processes (OP) which may be calcified and many are hollow (×1010).
(Reproduced from Kilic72 with permission.)
Intratubular dentin (Fig. 5.11) has a higher mineral content than intertubular dentin and therefore has a higher resistance to wear. A transitional region exists between equine primary and secondary dentin where intratubular dentin is absent, and is sometimes very distinct histologically.32 Because regular secondary dentin contains no (dense) intratubular dentin, it is more susceptible to attrition than primary dentin. Likewise, the dentin near the amelodentinal junction contains the lowest amounts of intratubular dentin and would theoretically be expected to wear faster; however, it is protected from excessive wear by the adjacent enamel.
Pulp
The histology of equine teeth pulp has been poorly evaluated to date, with most information derived from studies on brachydont teeth pulp. Pulp is a soft tissue within the dental pulp cavities that contains a connective tissue skeleton, including fibroblasts, thick collagen fibers and a network of fine reticulin fibers, connective tissue cells (that, as previously noted, can differentiate into odontoblasts if appropriately stimulated), extensive vasculature (to allow active continuous secondary dentin deposition), lymphatics, and nerves (sensory and vasoregulatory). In mature teeth, pulp is contiguous with the periodontal connective tissue at the apical foramen. A thin layer of predentin (that becomes thinner in older brachydont teeth) lies between the formed dentin and pulp periphery that contains the odontoblasts (Fig. 5.12) whose cytoplasmic processes extend into the dentinal tubules.
At eruption, equine permanent teeth possess a large common pulp (Fig. 5.17) that is contiguous with the primordial pulp that surrounds the developing apices (Figs. 5.18 & 5.19). At the apex of these young teeth, a thin layer of enamel surrounds this pulp. Later, following deposition of apical dentin and cement, roots are developing well in all equine cheek teeth by circa 2 years after eruption. One study suggested that separate pulp canals may not develop in mandibular cheek tooth until 5–6 years following eruption,9 but more extensive studies of both mandibular and maxillary CT have shown separate pulp horn development 1 year following cheek teeth eruption.32 The distinct anatomical features of equine cheek teeth pulps have significant implications for endodontic therapy. The 07s to the 10s all contain 5 pulp horns but the fully-developed 06s and mandibular 11s usually contain 6 and the maxillary 11s have 7 pulp horns.32,41 Whilst the original pulp horn classification of Dacre32,41,42 was a major step forward, its use of differing pulp numberings for upper and lower CT has caused some confusion. Consequently, a modified pulp classification10 is used in this text (Fig. 5.20). Equine incisors are similar to brachydont incisors in having just a single pulp.
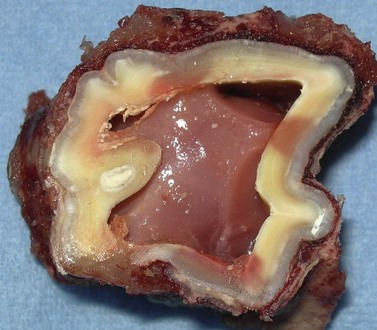
Fig. 5.17 Transverse section of a young equine maxillary cheek tooth sectioned just above the apex showing the common pulp chamber that later will become divided into separate pulp horns. A thick vascular periodontal membrane surrounds a thin layer (not apparent in some areas) of cementum that overlies the almost translucent layer of enamel that, in turn, overlies a layer of thicker cream-colored dentin.
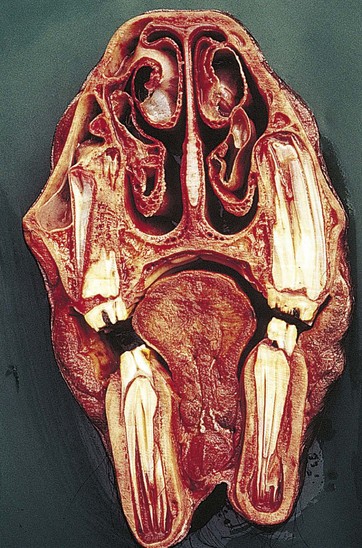
Fig. 5.18 Transverse section of the skull of a 3.5-year-old horse at the level of the 207 (second right maxillary cheek tooth) and between 107 and 108 on the left with part of 108 lying within the rostral maxillary sinus. Note the remnants of the deciduous teeth (‘caps’), the angulation of the occlusal surfaces, and the anisognathia. There are wide common pulp chambers of the apices of the permanent teeth and absence of roots. The infundibula of the temporary maxillary teeth have cemental hypoplasia.
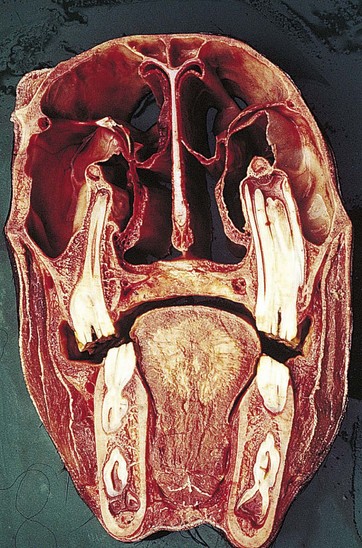
Fig. 5.19 Transverse section of the skull of a 3.5-year-old horse at the level of the 110 and 210 (with overlying infra-orbital canals) that lie at the junction of the rostral and caudal maxillary sinuses. Due to their curvature, parts of the mandibular 10s and 11s are shown. The mandibular canal lies on the ventromedial aspect of the mandible. The wide common pulp cavity of 210 has pulp horns that extend to within 1 cm of the occlusal surface.
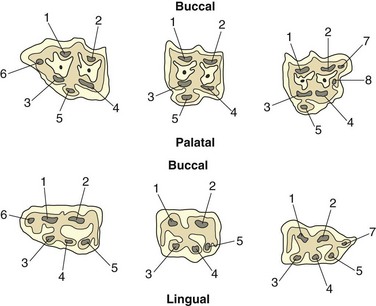
Fig. 5.20 A revised cheek teeth pulp numbering system (maxillary cheek teeth on top row and mandibular cheek teeth on bottom row) as described by du Toit et al.10
Unlike brachydont teeth, hypsodont teeth need to continue to lay down secondary dentin over a prolonged period (most of their life) in order to prevent occlusal pulp exposure. Consequently, in order to supply the metabolically active odontoblasts, the apical foramina through which the tooth vasculature passes into the pulp must remain relatively dilated (‘open’) for a prolonged period, although progressive reduction in foramen size does occur with age.43 The apical foramina also become displaced more occlusally by continued cement deposition at the apical aspect of the teeth with age. Kirkland et al found constricted (‘closed’) apical foramina in equine mandibular cheek by 5–8 years after their eruption, with development of two apical foramina in the rostral root.9 This is in contrast to the apical foramina of brachydont teeth which become more rapidly and extensively constricted (‘closed’) by deposition of secondary dentin within the pulp canal1 and also by cement deposition externally.
A practical result of these features is that pulp exposure in mature brachydont teeth causes pulpitis due to pH changes, irritation from molecules in saliva and foodstuffs, and an inevitable bacterial infection. The resultant pulpar inflammation within the totally rigid confines of the pulp chamber compresses the limited pulpar vasculature, usually leading to pulpar ischemia and necrosis, resulting in the death of the tooth. However, in hypsodont teeth, especially when young, the dilated apices and good blood supply often allows the pulp to withstand such inflammation by maintaining its blood supply. Local macrophages within the pulp, along with extravasated white blood cells and their molecules can then control pulpar infection. Additionally, the odontoblasts laying down secondary dentin can also lay down reactionary tertiary dentin in response to infection of the overlying dentin or following traumatic pulp exposure. In the absence of sufficient local odontoblasts as noted, adjacent undifferentiated connective tissue cells or fibroblasts in the pulp can transform into odontoblasts and lay down tertiary reparative dentin to seal off the exposed pulp from the healthy more apically situated pulp.
In addition to progressive, complete occlusion of the pulp horn beneath their occlusal aspect with secondary dentin, a continuous, but slower deposition of secondary dentin over all of the pulp horn walls causes the overall pulp size to reduce with age, as the surrounding dentin becomes thicker. A practical consequence of this is that the cheek teeth in younger (e.g. <7–8 year old) horses contain a high proportion of hard but brittle enamel with minimal secondary dentin, and thus are somewhat shell-like. These teeth are readily rasped but may fracture if cut with shears (whose use is no longer advocated – as mechanical burrs are much safer). In contrast, the teeth of older horses contain large amounts of secondary dentin, which makes them more solid and less likely to shatter when cut, but more difficult to rasp (float) than young teeth. With age, the pulp of brachydont teeth loses much of its vasculature, fibroblasts, and odontoblasts, while its collagen content increases. This process should be delayed in equine teeth due to the prolonged, higher metabolic requirements of their pulp to allow prolonged secondary dentin deposition (Fig. 5.12).
Cement
Cement (cementum) is a white or cream-colored, calcified dental tissue with mechanical characteristics and a histological appearance similar to bone. It contains circa 65% inorganic (again mainly impure hydroxyapatite crystals) and 35% organic and water components. Similar to dentin, its high organic and water content give it flexibility. The organic component of cement is comprised mainly of extensive collagen fibers, which include small intrinsic fibrils (produced by cementoblasts) and larger extrinsic fibers (produced by fibroblasts of the periodontal membrane), some of which form tight bundles termed Sharpey’s fibers (median 2.5 microns in diameter in horses) that cross the periodontal space to become anchored in the alveolar bone3 (Fig. 5.21), thus indirectly attaching the cement and alveolar bone. Cementum may be variously classified by its cellular content, i.e., cellular or acellular; its anatomical location, i.e., peripheral or infundibular; or coronal or root. A recent study examining cell proliferation within the equine periodontium demonstrated that a dynamic process of cell proliferation and migration is involved in the periodontal ligament remodeling associated with continued eruption.44 The recent successful culture of equine periodontal fibroblasts and equine dental cementoblasts will result in better elucidation of the process of continued eruption.45
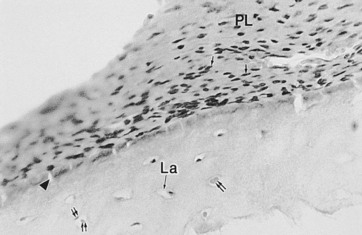
Fig. 5.21 Light microscopy of the periphery of an upper cheek tooth showing the periodontal ligament (PL) containing fibroblast-like cells (↑). The adjacent peripheral cement contains lacunae (La) of the cementoblasts (↑↑). Projections of the periodontal ligament into the cementum (arrowhead) probably represent Sharpey’s fibers (×1000).
(Reproduced from Kilic et al22 courtesy of the Editor of Equine Veterinary Journal.)
Under polarized light, undecalcified (ground) transverse sections of equine CT show two distinct regions. Adjacent to the peripheral amelocemental junction, the crystalloid nature of the cementum has irregular orientation of its hydroxyapatite crystal similar to those of maxillary CT infundibular cement. More peripherally, the crystal orientation changes to a more regular pattern, with the crystals having a similar concentric orientation. ‘Peripheral lines’ may be observed in decalcified transverse sections in this more peripheral zone. These two zones of regular and irregular peripheral cementum are most obvious in sections of older teeth near the occlusal surface.
Like dentin, cement (of the subgingival area and a few millimeters more occlusally, i.e., of reserve crown and roots) is a living tissue with its cells (cementoblasts) nourished by the vasculature of the periodontal ligament.2 Peripheral cement and the adjacent periodontal membrane can be considered as a single functional unit29 (as are dentin and pulp). After eruption onto the clinical crown, cementoblasts lose their blood supply from the periodontium and, in general, cement on the clinical (erupted) crown can be regarded as an inert tissue. However, recent work has shown active vasculature extending from the gingival margin beneath the surface of cementum on the clinical crown.2,27 Cement is the most adaptable of the calcified dental tissues and can be quickly deposited (within the alveolus or subgingivally) in response to insults such as infection or trauma,46 as commonly observed in some teeth with chronic apical infections.25,26 As noted earlier, hypsodont teeth have cementum covering all of the crown at eruption (including the occlusal surface) but the latter is soon worn away after eruption. Cement also fills the infundibula, usually incompletely at eruption.21
In hypsodont teeth, cement deposition continues throughout the life of the tooth, both around the roots (root cement) and also on the reserve crown (coronal cement) (Fig. 5.22). The latter allows new Sharpey’s fibers (Fig. 5.23) to be laid down, a process required to allow the necessary prolonged eruption of hypsodont teeth and also to allow additional cement deposition to mechanically contribute to the clinical crown. However, further cement cannot be deposited in the more occlusal aspect of the infundibulum that has lost its blood supply following tooth eruption, or more apically in the infundibulum unless some apical vasculature remains;21 likewise on the cement of the clinical crown once it moves a few millimetres away from the gingival vasculature.10 The main functions of cement are to provide anchorage for fibers of the periodontal ligament that support (with some flexibility) the tooth in the alveolus, and to protect the underlying dentin at the dental apex, and these two features of cement are present in both brachydont and hypsodont teeth. However, in hypsodont teeth, cement has major additional roles by contributing significantly to the bulk and thus mechanical strength of the clinical crown (especially in the lower cheek teeth), protecting the coronal enamel from cracking and helping form the protruding enamel ridges on the occlusal surface.2,43 The mandibular cheek teeth have a greater amount of peripheral cementum than maxillary cheek teeth.10
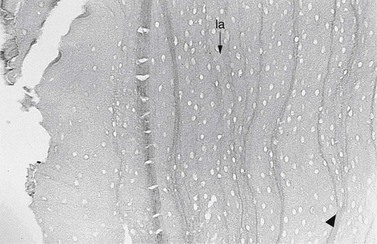
Fig. 5.22 Light micrograph of the peripheral cement of the deep reserve crown (adjacent to the apex) of a recently erupted cheek tooth. This contains wavy incremental lines (arrowhead) between successive depositions of cement that have occurred even at this early stage of tooth growth. Cementoblast lacunae (la) are present at all levels of the cement (×44).
(Reproduced from Kilic72 with permission.)
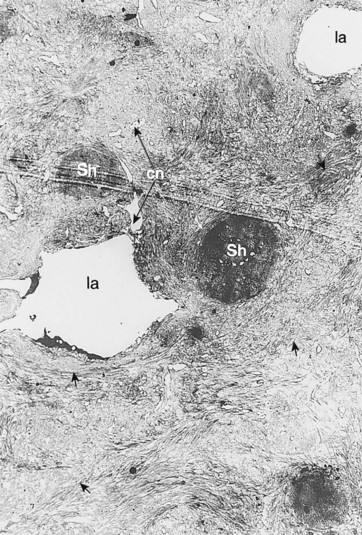
Fig. 5.23 Transmission electron micrograph of peripheral cement of a cheek tooth. This shows irregularly shaped lacunae (la) and their canaliculae (cn) but the cementoblasts have been lost during sample preparation. The dense Sharpey’s fibers (Sh) have been transversely sectioned. The intrinsic fibrils of the cement (↑) are also apparent (×2150).
(Reproduced from Kilic et al,22 courtesy of the Editor of Equine Veterinary Journal.)
To provide additional cement on the clinical crown, there is a large increase in cement deposition, once the tooth exits from the rigid spatial restrictions of the alveolus2 (Fig. 5.24). In some aged horses, the dental remnants exposed at the occlusal surface may eventually be composed only of roots (dentin and cement) with surrounding heavy cemental deposits. As this dental remnant contains no enamel, it becomes smooth on its occlusal surface (smooth mouth) and wears away quickly (Fig. 5.25).
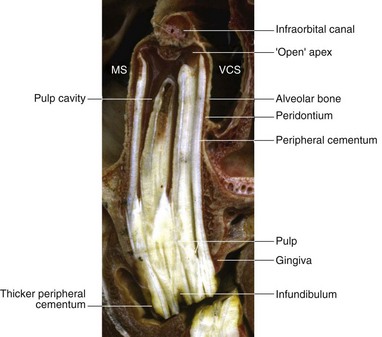
Fig. 5.24 Longitudinal section of a young maxillary cheek tooth lying in the maxillary sinus (MS) and ventral conchal sinus (VCS). Note the very extensive common pulp chamber and large pulp horns and thus a limited amount of (secondary) dentin present, which is characteristic of young equine teeth. Localized (clinically insignificant) central cemental caries is present in the transected infundibulum. The alveolar bone and periodontal membrane can be identified adjacent to the cement at the periphery of the tooth. Note the increase in thickness of the peripheral cement (of the ‘gingival reserve crown’) immediately following eruption of the tooth from the alveolus.
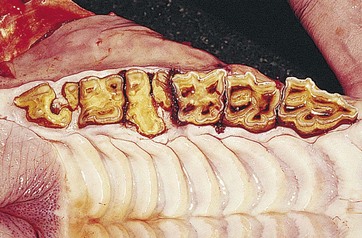
Fig. 5.25 Occlusal view of a maxillary cheek teeth row of an aged horse. Just the roots (with the rostral roots separated) remain of the 109 and 111 that have heavy peripheral cement deposits. These remnants contain little enamel (‘smooth mouth’) and consequently are ineffective at grinding forage and will soon fully wear out. The infundibula of some of the remaining teeth have fully worn out, and diastemata are present between some teeth.
A thin layer of peripheral cement covers the incisors and canine teeth, but much greater amounts overlie the cheek teeth, where its thickness varies greatly, largely depending on the degree of infolding of peripheral enamel. Peripheral cement is thickest in deeply infolded areas, especially in the two deep infoldings on the medial aspect of the lower cheek teeth (Fig. 5.9). At these sites, especially towards the tooth apex, this thick peripheral cement can be fully enclosed by these deep enamel folds, and these areas of cement can resemble infundibula.
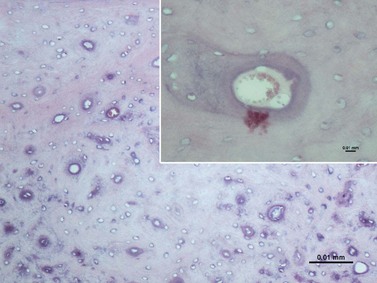
As noted, the infundibula (in all incisors and the upper cheek teeth) are usually incompletely filled by (infundibular) cement. Kilic et al22 found that in addition to the 24 % of (upper) cheek teeth that had gross caries (mineralized dental tissue dissolution) of their infundibular cement (Fig. 5.26), a further 65 % of horses had one or more small central vascular channels in this cement.22,38 These channels extended from the occlusal surface to a variable depth and contained smaller lateral channels extending as far as the infundibular enamel. This type of cement hypoplasia was termed central infundibular cemental hypoplasia. In addition, some infundibula had linear areas of cement hypoplasia at the enamel junction termed junctional cemental hypoplasia.22 As this latter cemental hypoplasia was commonly found in incisor infundibula, that show little evidence of caries (albeit they are much shallower infundibula than those of cheek teeth), consequently junctional cemental hypoplasia is not believed to be clinically significant.22 A more recent study showed infundibular cemental hypoplasia present in 22 % of 786 maxillary cheek teeth infundibula, with discoloration of an often porous-appearing infundibular cement present in 72 % of infundibula and true cemental caries present in 8 % of infundibula (Figs 5.27 & 5.28). The most marked cemental hypoplasia was present in the apical third of the infundibula.21 As only 15 % of the infundibula were completely filled with normal cementum, the hypoplasia (Figs 5.27–5.29) and discoloration so commonly observed in this study further supports the theory that cemental hypoplasia and discoloration could almost be considered as normal anatomical variations. Viable cementocytes and an active local blood supply were found in the more apical aspects of infundibula in one study (Figs 5.28 & 5.29).21
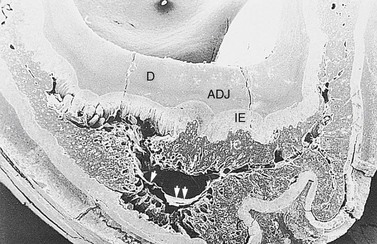
Fig. 5.26 Scanning electron micrograph of a mid tooth transverse section of the infundibular area of a maxillary cheek tooth of a 14-year-old horse. This contains dentin (D), an amelodentinal junction (ADJ), infundibular enamel (IE), and infundibular cement (IC). The infundibular cement shows extensive hypoplasia and areas of porous cementum with the large central defect partially lined by shrunken organic tissue (arrowheads). The infundibular enamel is exposed in several areas (×10.6).
(Reproduced from Kilic et al,31 courtesy of the Editor of Equine Veterinary Journal.)
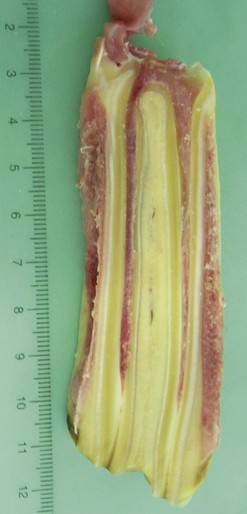
Fig. 5.27 Longitudinal section of a young maxillary cheek tooth circa 10 cm long with an infundibulum of 90 % of that length. Note the central area of discolored darker staining infundibular cementum that represents the site of the central vascular channel. Two pulp horns derived from the common pulp chamber are partially visible except at their occlusal aspects. Enamel extends to the full extent of the immature apex indicating absence of root development. Note the extensive peridontal attachments peripherally.

Fig. 5.28 Longitudinal section of a recently erupted maxillary cheek tooth with both infundibula partly exposed. The outlines of both infundibula are curved and irregular with a dilation of one infundibulum near its apex that additionally contains some vascular cementum with a blood supply from the apical area. This infundibulum also has a central cemental defect running through almost all of the infundibulum with more marked dark food-staining present 2–3 cm subocclusally. Note that one pulp horn contains viable pulp circa 5 mm sub-occlusally.
The occlusal surface
At eruption, the crown, including the occlusal surface of equine teeth, is fully covered by coronal cement, which in turn covers a thin layer of coronal enamel. With normal occlusal wear, the coronal cement and coronal enamel are very soon worn away thus exposing the secondary occlusal surface of these teeth, which is the permanent occlusal surface of hypsodont teeth (Fig. 5.3). The normal wear process, i.e., attrition on the occlusal surface of hypsodont teeth is a complex phenomenon depending on many factors, including the type of diet, e.g., in the winter outdoor horses may be forced to graze lower and thus ingest more soil-covered roots and leaves, or even eat the roots of plants such as nettles (M. Booth 1996, personal communications), thus greatly increasing the amount of silicates that are ingested. When grazing is scarce, they may also eat coarser food, including bushes, such as gorse. The duration of eating also varies according to the season from up to 13 hours/day in summer to 16.5 hours/day in winter in outdoor horses, in some environments (M Booth 1996, personal communications). While eating hay, horses and ponies have 58–66 chews a minute, with 4200 chews /Kg of dry matter,47 whilst at grass they have 100–105 chewing movements per minute.48 Dental attrition also depends on the force and the direction of the chewing action, the sizes, shapes, and angles of the opposing occlusal surfaces, and the relationship of opposing cusps and crest patterns to the occlusal motion. Horses eating roughage exhibit a more lateral masticatory action compared with horses eating a concentrate/pelleted diet, which have a more vertical crushing stroke.49,50 Consequently, painful oral disorders can cause changes in the direction and forces of mastication and thus affect the wear patterns of cheek teeth, as further discussed in Chapters 6 and 9.
The occlusal surfaces of equine teeth are covered by an organic pellicle38 containing microorganisms, small food particles, and a smear layer of finely ground dental particles formed by masticatory actions. The underlying enamel contains differing wear patterns, including polished areas, small local fractures, pit striations, and depressions. Most large striations are at right angles to the long axis of the cheek teeth rows (i.e., in the buccolingual/palatal plane) and appear to be caused by the normal side-to-side chewing motion of the cheek teeth when grinding down small ingested phytoliths (calcified plant particles). Scanning electron microscopy of such deep grooves shows that prismatic enamel is more deeply worn than interprismatic enamel, confirming that the former structures are softer. Additionally, some shorter striations are present on the occlusal surface of equine teeth perpendicular to the buccolingual/palatal plane (at right angles to the normal chewing direction), and it is suggested that these striations are caused by ingested phytoliths during the crushing phase of chewing.38
The softer dentin on the occlusal surface wears faster than the surrounding enamel, and therefore the dentinal surface becomes depressed, relative to the adjacent enamel. The depth of these depressions is directly related to the area of occlusal dentin, with larger exposed areas more deeply recessed.38 In contrast, smaller areas of occlusal dentin are better protected from wear by the adjacent enamel folds and so undergo less wear and have shallower depressions on their surface. Therefore, the orientation and invaginations of the enamel folds (peripheral and infundibular) play an important role in dividing the occlusal surface of dentin into smaller areas and thus protecting it from excessive attrition. The lower cheek teeth have three very deep infoldings of enamel, two on the medial (lingual) aspect and one on the buccal aspect. The upper cheek teeth have less marked peripheral enamel infoldings; however, they contain two enamel-containing infundibula, which further subdivides and compartmentalize their occlusal dentin, thus protecting it from excessive wear centrally in the teeth.38 The ratio of peripheral enamel length to tooth perimeter on sub-occlusal transverse sections is greater in maxillary cheek teeth with infundibular enamel (1.87) compared to mandibular cheek teeth (1.48) despite the greater peripheral enamel infolding present in mandibular cheek teeth.10 There is usually less infolding of peripheral enamel folds more apically in teeth, and therefore some older teeth may show excessive dentinal wear at such unprotected areas (Fig. 5.30). Similarly, if infundibula (in upper cheek teeth) are absent or are short and wear away prematurely, excessive local dentinal wear also occurs centrally in such teeth (Fig. 5.31). A study of normal infundibula has shown lengths (depths) from 89 mm (in a 4-year-old horse) to 2 mm (in a 30-year-old horse), with infundibular length being a mean of 82 % of the total dental crown length.21
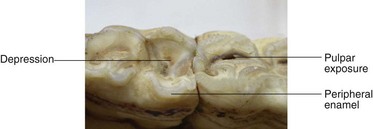
Fig. 5.30 Occlusal aspect of two aged mandibular cheek teeth with the tooth on the left showing limited peripheral cement infolding of its caudal aspect – with consequent excessive wear of the adjacent dentin causing a depression in this area of the occlusal surface (‘cupping’ ‘senile excavation’). The adjacent cheek tooth has normal enamel infolding but has occlusal exposure of one of its pulps.
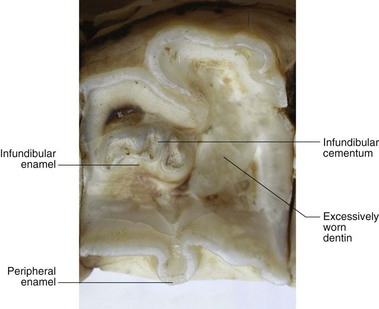
Fig. 5.31 Occlusal surface of a maxillary cheek tooth that is missing one of its infundibula with resultant excessive wear causing a deep depression (‘cupping’ ‘senile excavation’) in the occlusal surface at this site. Such ‘cupping’ may predispose to sharp overgowths on the lateral and medial aspects of upper and lower cheek teeth in older horses. Unusually the remaining infundibulum appears to consist of two separate smaller infundibula.
Gross Anatomy of Equine Teeth
Incisors
The deciduous 01s (central incisors), 02s (middle-intermediate) and 03s (corner) incisors erupt within a few days of birth, 4–6 weeks, and 6–9 months of age, respectively.51 Deciduous incisors are whiter and contain wider and shallower infundibula than their permanent successors, which erupt on their lingual aspect. As noted, the eruption of both deciduous and permanent teeth can be used to estimate the age of horses up to 5 years old with a reasonable degree of accuracy52,53 (see Ch. 7).
The dental formula of deciduous and permanent teeth in horses
depending on the presence and number of canine teeth or 1st premolar (wolf teeth).54
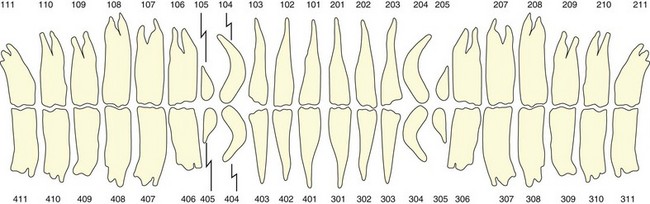
The Triadan System of dental nomenclature utilizes three digits to identify each tooth. The first digit refers to the quadrant, with 1 for upper right, 2 for upper left, 3 for lower left, and 4 for lower right (Fig. 5.32).55 The deciduous teeth are similarly identified using the prefix 5–8 for the four quadrants.
Adult horses also have 12 incisors in total, six in each arcade. The upper incisor teeth are embedded in the premaxillary (incisive) bone, and the lower incisors in the rostral mandible, with the reserve crowns and apices of incisors converging towards each other. Incisor teeth are curved convexly on their labial aspect (concavely on their lingual aspect) and taper in uniformly from the occlusal surface toward the apex (unlike equine deciduous incisors, and all brachydont incisors that have a distinct neck). Therefore with age, spaces eventually develop between equine permanent incisors, but the development of these spaces is delayed by the medial (mesial) pressure of the 03s on the remaining incisors. The fully developed incisor arcade in a young adult horse has an almost semicircular appearance, which gradually becomes shallower with age, due to alteration of teeth shape caused by progressive wear.56 The occlusal angle of incisors also changes from almost vertical apposition in the young horse (Fig. 5.33) to an increasing angle of incidence with age.
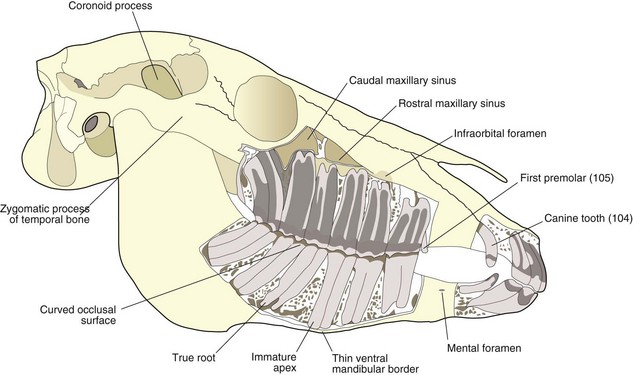
Fig. 5.33 Diagram of a skull of a 5-year-old horse demonstrating ventral deviation of the mandible associated with eruption of 408. Note the shape and apposition of the incisors of this young horse. The angulation of the rostral and caudal cheek teeth and the curvature of the sixth teeth maintain tight apposition of all six cheek teeth at the occlusal surface. The TMJ is high (approximately 15 cm higher in an adult Thoroughbred) above the level of the cheek teeth occlusal surface. Note the small coronoid process and the large area of muscle attachment of the mandible.
Equine incisors also develop certain wear-related macroscopic features that have been traditionally (if not very accurately) utilized for estimating age52–54 as discussed in detail in Chapter 7. The infundibulum present in all incisors is termed the incisal cup (‘cup’). This funnel-like enamel structure is oval in shape and circa 10 mm deep when the tooth first erupts. However, variations in its depth may cause the infundibulum to wear away more rapidly or slower than ‘normal’ and thus make aging difficult. The incisor infundibulum is usually incompletely filled with cement and consequently later becomes filled with food material and appears dark.52 When the infundibular cavity is worn away, it leaves behind a small ring of the remaining apical aspect of the infundibular enamel, located on the lingual aspect of the tooth, which is called the enamel spot (enamel ring or mark).52 Due to the slower wear of enamel as compared to dentin, the enamel spot becomes elevated above the occlusal surface. The dental star represents exposure of secondary (regular and irregular) dentin on the occlusal surface of incisor teeth that was deposited in the former pulp cavity. It appears sequentially in the 01s, 02s and 03s (see Ch. 7). This secondary dentin initially appears as a dark yellow (due to food staining), transverse line on the labial aspect of the infundibulum. With further tooth wear, it gradually becomes oval in shape and moves toward the centre of the occlusal surface.
Galvayne’s groove is a longitudinal groove that appears on the labial aspect of the permanent upper 03s (corner incisors), and is traditionally stated to first appear at about 10 years of age, reaching halfway down the tooth by 15 years of age and extending to the occlusal surface by 20 years of age. However, recent critical studies, as reviewed in Chapter 7, have shown much variation in the time that these features develop. Another, variable anatomical feature is the development of a ‘hook’ (a colloquial term for a localized dental overgrowth) on the caudolabial aspect of the occlusal surface of 103 and 203 after circa 6 years of age, due to incomplete occlusal contact between the upper and lower 03s. It is often termed a ‘7 year notch or hook’ because it was traditionally (but erroneously) believed to always appear at 7 years of age.7,56 Variations in incisor teeth appearance can also be due to individual and breed variation, differences in diets, environmental conditions, eruption times, mineralization rates, depth of enamel infundibulum, amount of infundibular cement and the presence of certain stereotypic behaviors, such as crib-biting and wind sucking.57,58 The occlusal surface of individual incisors is elliptical in recently erupted incisors, but, with wear, they successively become round, triangular, and then oval in shape. These changes are more apparent in the lower 01s and 02s than in the lower 03s.53,54
Canine teeth
The deciduous canine teeth (Triadan 504, 604, 704 and 804) are vestigial spicule-like structures, 0.5–1.0 cm long, that do not erupt above gum level. The lower deciduous canine is situated caudal to the 03s (corner incisor).51 Male horses normally have four permanent canine teeth, two maxillary (104, 204) and two mandibular (304, 404), that erupt between 4 and 6 years of age in the interdental space (physiological diastema).54 They are often stated to be simple teeth, but while they have no enamel infolding, their clinical crown is covered in peripheral cementum, and some degree of prolonged eruption can occur. Canine teeth have a pointed occlusal surface, are convex on their buccal border, and slightly concave on their medial (lingual and buccal) aspect, with a slight caudal facing curvature. The lower canines are more rostrally positioned than the upper, and thus there is no occlusal contact between them. This is alleged to be a reason why canine teeth (especially the lowers) are prone to develop calculus. Canine teeth are usually absent or rudimentary in female horses, with a reported prevalence of 7.8–28 % in horses57 and 17.3–30 % in donkeys.59 Canines do not continually erupt like cheek teeth, and thus long reserve crowns can be present in older horses. In the young adult Thoroughbred, canine teeth are 5–7 cm long with most present as unerupted crown. In some horses, just 10–20 % of the crown is erupted and consequently, due to the great length and size of the reserve crown and roots, extraction of these teeth is a major undertaking. Canine teeth have a wide pulp cavity that in young adult horses may extend to within 5 mm of the occlusal surface; consequently, the reduction (grinding down) of canines in horses (usually for non-scientifically validated reasons) risks causing pulpar exposure.
1st premolar (‘wolf tooth’)
One or both of the upper 05s (1st premolar), and less commonly, the lower 05s, can be present as the small, vestigial ‘wolf teeth’. These should normally lie immediately in front of the 06s. They are simple brachydont teeth whose clinical crown can vary from 1 to 2 cm in length. Their roots can vary from being non-existent (with loose attachments of the teeth to the gingiva), to being > 30 mm in length. These teeth are sometimes rostrally or rostrolaterally displaced and also may be angulated (i.e., not vertically aligned in relation to the hard palate). The (permanent) 05s usually erupt at 6–12 months of age, and they do not have a deciduous precursor. Wolf teeth have a reported prevalence of 24.4 % in females and 14.9 % in males;60 and of 13 %20 to 31.9 %61 in horses of both sexes. This wide range of prevalence is likely due to loss of some wolf teeth at circa 2.5 years of age, when the adjacent deciduous 06s are shed (J. Easley, personal communications).
Cheek teeth
The 12 temporary premolars (506–508, 606–608, 706–708 and 806–808) are erupted at birth or do so within a week or so. These deciduous teeth are replaced by the larger, permanent premolars at circa 2.5, 3, and 4 years of age for the 06s, 07s and 08s, respectively. In contrast to brachydont teeth and to equine incisors (where the deciduous teeth are much smaller than the permanent teeth) the transverse (cross sectional) area of equine deciduous cheek teeth can be similar to those of adult teeth,43 and thus a retained remnant of a deciduous cheek tooth remnant (‘cap’) can be difficult to identify from the underlying permanent tooth. The three deciduous cheek teeth in each row have a distinct neck between the crown and roots, unlike their permanent successors.62 Latterly, these deciduous cheek teeth erupt into the oral cavity due to traction by their periodontal ligaments and pressure from the underlying permanent tooth. They are simultaneously resorbed at their apices by immunologically-mediated mechanisms until eventually just a thin ‘cap’ of the temporary tooth remains lying on the occlusal aspect of the permanent cheek tooth (Fig. 5.18).
An adult equine mouth normally contains 24 cheek teeth (06s–11s, i.e., 2nd – 4th premolars and 1st – 3rd molars), forming four rows of six teeth that are accommodated in the maxillary and mandibular bones. The molars erupt at approximately 1, 2, and 3.5 years of age, respectively. On transverse section, equine cheek teeth are rectangular shaped, except the first (06) and last (11), which are somewhat triangular shaped (Figs 5.14, 5.34). The maxillary cheek teeth are about 50 % wider and so are squarer in comparison with the mandibular cheek teeth, which are narrower and more rectangular in outline. The long axes of all cheek teeth are relatively vertical, except the first cheek tooth (06) whose clinical crown tilts caudally (reserve crown tilts rostrally) and the last cheek tooth (11) and to a lesser and variable extent the (10), whose clinical crowns tilt rostrally (reserve crowns tilt caudally; Fig. 5.33). The purpose of these angulations is to compress all six cheek teeth together at the occlusal surface to prevent the development of diastemata. The buccal aspects of the upper cheek teeth have two prominent vertical (longitudinal) ridges (cingula, styles) rostrally and a less prominent caudal ridge with two deep grooves between them, except the 06s which can have 3–4 small grooves and ridges. These ridges can decrease with age; they vary in size between individual horses and different breeds. Dental overgrowths (enamel ‘points’) can often be sharp and prominent on the lateral and medial aspects of these occlusal ridges (especially on larger ridges such as 10s and 11s), even in horses on a permanent forage diet. In contrast, horses with less prominent vertical ridges appear to be less likely to develop enamel overgrowths on their maxillary cheek teeth. The palatal aspect of the upper, and both lingual and buccal aspects of the mandibular cheek teeth contain much less distinct vertical grooves and ridges.
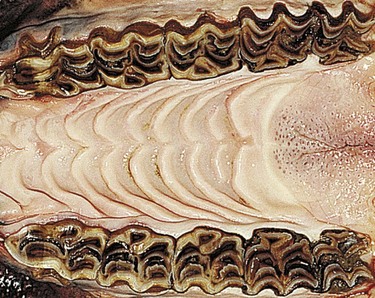
Fig. 5.34 Occlusal view of the five maxillary cheek teeth of a 3-year-old horse. Note the absence of spaces between these teeth, the pronounced vertical ridges on the buccal aspects of these teeth, and the triangular shape of the 06s (as will be the case for the 11s when they erupt).
In younger horses, the permanent cheek teeth possess long crowns, most of which is un-erupted reserve crown, that are embedded in deep alveoli (Figs 5.18, 5.19, 5.35). In Thoroughbreds, the 06 cheek tooth is the shortest (circa 5–6 cm maximum length), with the remaining cheek teeth being up to 9 cm long at eruption. Dental eruption proceeds throughout the life of equine teeth, and normally the eruption rate corresponds with tooth wear (attrition) and has been calculated as 2–3 mm per year;4,5 therefore a 75-mm long permanent 08 cheek tooth that comes into wear at 4 years of age should be fully worn by 30 years of age.4
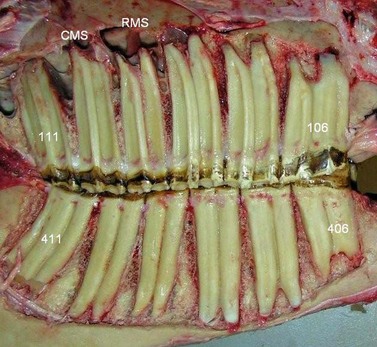
Fig. 5.35 Skull of young horse (circa 7 years) with the lateral maxillary and mandibular walls removed to expose the cheek teeth. Note the long reserve crowns – with the mandibular 408 tooth almost touching the ventral cortex of the mandible. The rostral maxillary sinus (RMS) and caudal maxillary sinus (CMS) overlie the apical aspects of 408–411. Note the angulation of the reserve crowns that keeps the occlusal surface of all 6 teeth tightly compressed together. Root development is present in all teeth.
Image courtesy of Istvan Gere.
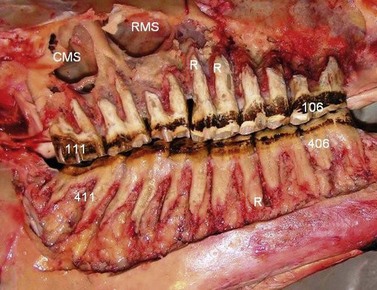
Fig. 5.36 Skull of an aged horse with the lateral maxillary and mandibular walls removed to expose the cheek teeth. Minimal reserve crowns now remain, with most teeth just having alveolar attachment to their (cemental) roots (R) that are very elongated in all teeth. The mandibular teeth in particular still have good angulation of their remnants, that is compressing their occlusal aspects together. There is a ‘step-like’ overgrowth of the caudal three mandibular cheek teeth. Note the larger rostral maxillary sinus (RMS) and caudal maxillary sinus (CMS) in this older skull as compared to the skull in Fig. 5.35.
(Image courtesy of Istvan Gere.)
Once their roots have clearly developed (circa 2 years following eruption), the upper cheek teeth usually have three roots, two small lateral roots and a large, flat medial root, but occasionally the medial root will form two separate roots giving a total of four roots.63 The lower cheek teeth (except the 11s, which have three roots) usually have two equally sized roots, one rostral and one caudal, that tend to become longer than those of their maxillary counterparts.
The alveoli of the first two upper cheek teeth (06s and 07s) and often the rostral aspect of the 08, and usually all of this tooth in older horses62 are embedded in the maxillary bone (Fig. 5.18). The caudal aspect of the 08 and the 09 alveoli usually lie in the rostral maxillary sinus, and the alveoli of the 10s and 11s usually lie in the caudal maxillary sinus (Fig. 5.19). However, there can be much variation in this finding, with the rostral aspect of the rostral maxillary sinus varying in position from overlying the 07s to the 09s, and the shell-like, bony transverse maxillary septum (separating the rostral and caudal maxillary sinuses) varying in position from the caudal aspect of the 08s to the caudal aspect of the 09s.64,65 In young horses, the alveoli of the large cheek teeth reserve crowns occupy much of these maxillary sinuses, but with age and subsequent eruption of their reserve crowns and remodelling and retraction of their alveoli, the sinus cavities increase in volume, as their floor lowers due to cheek teeth eruption (Fig. 5.36). In younger horses, the infraorbital canal lies directly over the apices of the caudal maxillary cheek teeth, and is often curved dorsally at this site, whereas in the older horse a thin plate of bone (that divides the sinuses sagittally) attaches the alveoli to the infraorbital canal.
Additionally, the cheek teeth migrate rostrally in the sinuses as they erupt.5 For example, the apex of the curved upper 11s (caudal maxillary cheek tooth) drifts rostrally from its site beneath the orbit in the young adult, to become sited rostral to the orbit in the aged horse. The intimate relation between the caudal cheek teeth and sinuses can allow periapical infections of the caudal cheek teeth to cause maxillary sinus empyema, as discussed in Chapters 13 (imaging) and 14 (ancillary techniques). The rostral maxillary teeth are embedded in maxillary bones: for example, in young Thoroughbreds, the apices of the 08s often lie 2–3 cm rostral to the rostral aspect of the facial crest, the 07s lie 2–3 cm rostral to this site, and the apices of the 06s lie a further 2–3 cm rostrally. In many young horses, eruption cysts occur at these sites during dental eruption, but due to the presence of the overlying levator nasolabialis and levator labii superioris muscles, these swellings may not be detected.
The six maxillary and six mandibular cheek teeth form slightly curved rows (more pronounced in the maxillary cheek teeth), with their concavity facing buccally (laterally) and lingually (medially), respectively.52 This convex curvature of the lateral aspect of the maxillary cheek teeth can be marked in some horses and a practical consequence is that it renders effective dental rasping impossible, unless a selection of angulated rasps are available. A common feature of all ungulates (mammals with hooves), including horses, is the presence of an interdental space (‘bars of mouth’) between the incisors and the premolars, that is likely due to the evolutionary increase in face length (dolicephalic) to allow these long-legged animals to more comfortably graze off the ground66 whilst being able to view potential predators. Its presence, however, necessitates the clinical crown of the rostral cheek tooth (06) facing caudally to help compress the occlusal aspect of each row of cheek teeth, as noted earlier (Fig. 5.33). In contrast, the complete arch of teeth of omnivores and many carnivores needs to be compressed in just a single direction with the rostrally (mesially) facing caudal tooth (‘wisdom tooth’) to promote compression of teeth.66
Continued eruption of the angulated rostral and caudal cheek teeth usually maintains the tight occlusal contact between the six cheek teeth until late in life in normal horses, despite the fact that equine teeth slightly taper in towards their apex and so, with age, would otherwise develop spaces between the teeth (interdentally–interproximally) that is termed diastema(ta) (Fig. 5.25).67,68 Many very old horses (>20 years) do develop diastemata between their incisors, which is usually of little consequence in these teeth, unlike the situation with cheek teeth, where diastemata can cause food to accumulate between the teeth and in the adjacent periodontal space, possibly leading to severe dental disease,65 and this situation is even more pronounced where cheek teeth diastemata are widespread and cause severe clinical disease.69
The occlusal surfaces of the rows of cheek teeth are not level in the longitudinal plane as occurs in some other species, but instead the surfaces of the caudal 2–3 cheek teeth curve dorsally to a variable degree in the caudal direction that is termed the Curve of Spee 6 (Fig. 5.33). This curvature is often marked in Arabian-type horse breeds that often have a similar curvature on their (dished) facial bones, but can also be marked in other breeds of horses, even in larger draught horses with convex faces (i.e., ‘rams head’ or ‘Roman nose’). Some horses also have a marked upward curvature of the rostral aspect of their cheek teeth rows, with the lower 06 becoming quite tall (dominant) and little clinical crown present on the upper 06s. This anatomical configuration is likely be a normal anatomical variation in such horses, that causes no clinical problems, whereas misguided attempts to create a standard appearance of equine teeth certainly can have deleterious clinical consequences. If both a Curve of Spee and a rostral curvature are present, this gives the lower cheek teeth row occlusal surface a concave appearance in the rostrocaudal plane, i.e., raised at the 06s and the 11s.
In normal horses, the distance between the maxillary cheek teeth rows is approximately 23 % wider than the distance between the mandibular rows,70 a feature which is termed anisognathia. Anisognathia is even more marked (27 % difference between upper and lower CT) in donkeys.10 This is in contrast to many brachydont arcades, such as human upper and lower dental arcades, that are equally spaced (isognathic). As noted, the maxillary cheek teeth are also wider than their lower counterparts. Consequently, when the equine mouth is closed, approximately one-third of the occlusal surface of the upper cheek teeth is in contact with about half of the lower cheek teeth’s occlusal surface. Additionally, the occlusal surfaces of the cheek teeth are not level in the transverse (bucco-lingual) plane as is usually the case in brachydont species, but are angled between 15 and 35 degrees [angled from dorsal on their lingual (buccal) aspect to ventral on their buccal aspect] (Figs 5.18 & 5.19). The maxillary cheek teeth have a lower angulation than the mandibular cheek teeth, varying from circa 19.2 degrees at the 06s and decreasing to 9.2 degrees at the 11s. In contrast, the mandibular cheek teeth occlusal angulation increases from 15.3 degrees on the 06s to up to 31.5 degrees on the 11s.71 Cheek teeth angulation is also influenced by masticatory activity.50 For example, on a normal forage diet where horses have a wide range of lateral masticatory movement, the angle is believed to remain within the normal range. In contrast, on a diet high in concentrates, e.g., processed grains, or with an intercurrent painful dental disorder that causes pain on mastication, the masticatory action will be more vertical and lead to a higher degree of occlusal surface angulation (e.g., >45 degrees in the caudal mandibular cheek teeth), which is termed shear mouth if severe.
The terminology concerning the irregularities present on the occlusal surface of the cheek teeth can be confusing. A cusp is a pronounced elevation on the occlusal surface of a cheek tooth and is an area with thicker enamel. A ridge (or style) is a linear elevation on the surface (peripheral or occlusal) of a tooth, and on the occlusal surface may be formed by interconnecting cusps. Horses usually have about 12 such ridges running transversely across the occlusal surface of their cheek teeth that are commonly termed transverse ridges, two on the occlusal surface of each tooth, except the first and last, which can contain one to three ridges. These ridges can be quite tall, especially over the caudal cheek teeth in younger horses of certain breeds. Diet and age may also influence their size. These normal anatomical structures that increase the masticatory surface (and efficiency) of cheek teeth should not be confused with narrower acquired transverse overgrowths (usually just a lesser number) often termed ‘exaggerated or accentuated transverse ridges’ due, for example, to being opposite a wide diastema or some other area of reduced contact with their occlusal counterpart.
Because equine cusps contain sharp ridges of exposed occlusal enamel adjacent to hollows (craters) of dentin (and cementum at some sites) they are classified as lophs and thus the cusp pattern of equine teeth is termed lophodont. A fossa is a rounded depression, and a fissure is a linear depression between cusps or ridges.1,71 The latter physiological fissures should be distinguished from pathological fissure fractures (cracks) in cheek teeth24 that can lead to pulpar exposure.25,26 The opposing ridges and (physiological) fissures of the upper and lower equine cheek teeth interdigitate when the mouth is shut. Other variations in cusp number, size and distribution are used for paleontological research and for taxonomic classification of different species.71
Nerve supply of teeth
Because of its great importance in human dentistry, the innervation of teeth has been well studied in brachydont teeth. Pulpar nerves enter through the apical foramen and include sensory nerves derived from the trigeminal (5th cranial) nerve, which are most extensive in the coronal (occlusal) region of the pulp where they form the plexus of Raschkow,13 and sympathetic fibers from the cervical ganglion that supply the vascular smooth muscles to regulate blood flow in the pulp.72,73 The latter are also believed to control the differentiation and function of odontoblasts, including their circadian rhythm of activity.30
The type and duration of pain caused by stimulation of dentin are different from those of pulp. In brachydont teeth, dentin responds to various stimuli, including excessive heat and cold, and to therapeutic procedures, such as drilling, with a sharp pain which stops when these stimuli cease. In contrast, stimulation of the pulpal nerves produces dull pain (sometimes a throbbing pain synchronous with the heartbeat caused by the effect of arterial pulsations within the inflamed pulp) which continues for some time after the stimulus is removed.30 Nerves are also present in the pulp of hypsodont teeth, although the role of sensory nerves is unclear, as these teeth have dentin, including open dentinal tubules, and odontoblast processes constantly exposed on the occlusal surface,38 a situation that would cause marked pain in brachydont teeth. Following significant dental overgrowth reductions, some horses do not masticate properly for days to weeks, and this is without any evidence, attributed to temporomandibular joint (TMJ) pain caused by prolonged opening of the mouth with a speculum during the dental procedures. However, the recent work showing exposed, apparently viable dentinal processes following dental rasping37 makes it more likely that in fact pain from damaged sensitive dentin, or in some cases from actual pulp exposure, is the cause of such post-treatment pain.74
Blood supply of teeth
In brachydont teeth, the blood vessels enter pulp through the apical foramen and form an extensive capillary network, particularly in the coronal region of the pulp.30 These capillaries drain into an extensive venous network that has a more tortuous course than the arterioles and also exits via the apical foramen.30 Due to difficulties in microscopically distinguishing lymphatics from vascular capillaries it remains unclear if lymph vessels are actually present in pulp.30 However, other authors believe that pulp tissues (like all other connective tissues), contain lymph vessels that in human beings drain into the submandibular and deep cervical lymph nodes. As previously noted, the good blood supply and wide apical foramina of even adult equine teeth can allow them to retain a blood supply following pulpar exposure and then allow them to seal off the exposed pulp with tertiary dentin. Although not involved in dental blood supply, the greater palatine artery can be iatrogenically damaged during dental procedures and awareness of its site and size is necessary. This artery runs around the periphery of the hard palate and is not an end-artery as it adjoins its counterpart rostrally and thus receives a blood supply from both internal maxillary arteries. It can be damaged if a dental elevator slips medially when extracting wolf teeth, especially if they are medially displaced. It can also be damaged when orally extracting maxillary cheek teeth – which usually have short erupted crowns on their medial (palatal) aspect – if the extraction forceps is placed too high on the gingiva above these teeth.
Supporting bones and muscles of prehension and mastication
Alveolar bone
Alveolar bone is very flexible and constantly remodels to accommodate the changing shape and size of the dental structures it contains. Alveolar bone can be divided into two main parts: a thin layer of compact (radiodense) bone (the ‘cortex’ of alveolus) that lines the alveolus proper, in which Sharpey’s fibers insert, that is radiographically termed the lamina dura (lamina dura denta). This area is radiographically detectable (but not on computed tomography) as a thin radiodense line in brachydont teeth but due to irregularities of the periphery of some normal equine cheek teeth, this feature is not always obvious on lateral radiographs of equine teeth (Fig. 5.37). Secondly, the main alveolar bone surrounding the lamina dura denta cannot be morphologically differentiated from the main bone of the mandible or maxilla in adult brachydont teeth.1 However, recent studies have shown that in horses, the alveolar bone beneath the lamina dura remains spongy and porous throughout life – similar to the alveolar bone of developing children’s teeth – probably a reflection of its constant remodeling as the equine teeth constantly erupt.2 This presents an area of anatomical weakness, which may explain why sequestration of the alveolar cortical bone can occur following oral extraction of cheek teeth. The most prominent aspect of the alveolar bone beneath the gingival margin (occlusally) is termed the alveolar crest.
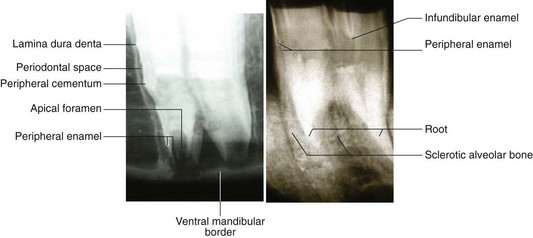
Fig. 5.37 Radiographs of the apical aspects of a young mandibular (left) and an inverted aged maxillary cheek tooth (right). Note the wide periodontal space of the younger tooth that merges with soft tissue of the apical area and into the large apical foramen (of such younger teeth) and on into wide pulp cavities. The peripheral enamel folds reach to the apex of this tooth i.e., it has no enamel-free apical area or true roots. In contrast, the older tooth (right) has long true roots composed of cementum with no radiographically obvious apical foramen or pulp horns. The peripheral (and infundibular enamel) folds are positioned high above the alveolus on this old tooth due to prolonged root cementum deposition.
Mandible
The mandible, the largest bone of the equine face, is composed of two component hemimandibles that fuse together at the symphysis at 2–3 months of age.51 The mandible articulates with the squamous temporal bone at the TMJ and contains the alveoli of the mandibular incisors; canines; wolf teeth (if present) and lower cheek teeth. The ventral border of the horizontal ramus of the mandible is wide and rounded in the young horse because of the length and size of the reserve crowns of the cheek teeth it contains (Figs 5.18 & 5.19) and, conversely, becomes thinner and sharper in older horses as the cheek teeth erupt – a feature used to age horses in some Eastern countries. Some breeds, especially those that are descendants of the Arabian horse (which in turn are descendants of Equus cracoviensis – Type IV horse), have shallow mandibles and maxillae and commensurately short reserve crowns, whereas most other breeds, e.g., derived from E. muniensis (Types I or Mountain Pony) or E. mosbachensis (Type III – Forest or Marshland Horse) such as the North European Draught and native British pony types (such as Exmoor ponies) have deep alveoli and long reserve crowns.75,76
It has been proposed that crosses between these two types of horses can develop pronounced ventral swellings under the developing apices of the 2nd and 3rd cheek teeth,75 due to an imbalance between mandibular depth and tooth length. These mandibular eruption cysts (‘osseous tubercles’) (Fig. 5.33) usually occur at 3–5 years of age and exceptionally, unless they become infected (usually by blood borne mechanisms, i.e., anachoresis), they usually regress over the following 1–2 years.43 Other authors suggest that some breeds of horses are predisposed to retention of deciduous cheek teeth remnants (‘caps’), which causes these mandibular swellings,67 but clinical studies have not verified this.77
The mental nerve (branch of Cranial Nerve V) enters the mandibular foramen on the medial aspect of the vertical ramus, level with the occlusal surface of the cheek teeth. The mental nerve can be locally anesthetized at the mandibular foramen to facilitate painful dental procedures (e.g., oral extraction of a mandibular tooth) in the standing horse. The nerve then continues rostroventrally in the mandibular canal until it reaches the ventral aspect of the horizontal ramus, where it then continues rostrally within the mandible below the apices of the cheek teeth. However, in recently erupted teeth whose apices reach the ventral border of the mandible, the nerve usually lies on the medial aspect of the developing tooth (Fig. 5.18). The main part of the mental nerve emerges through the mental foramen on the rostrolateral aspect of the horizontal ramus, approximately halfway between the lower 06 and the incisors, while a smaller branch continues rostrally in a smaller canal along with the vasculature of the lower incisors. The nerve supply to the lower incisors and lower canine tooth can be anesthetized within the rostral aspect of the mandibular canal.
Immediately caudal to the alveoli of the lower 11s (6th cheek teeth), the mandible becomes a very thin sheet of bone. This flattened, thin area progressively increases in size with eruption of the caudal mandibular cheek teeth and subsequent contraction of their alveoli. More caudally, at the angle of the jaw, this thin plate of bone expands medially and laterally into two thick bony protrusions that are roughened to allow muscle attachment (Fig. 5.33). These protrusions reduce in size towards the dorsal border of the vertical ramus. These normal roughened mandibular areas may be radiologically confused with pathological mandibular changes.
TMJ and muscles of mastication
In contrast to carnivores that have a vertical power stroke, horses also have a transverse power stroke in a lingual (medial) direction that is termed a lingual power stroke,78 as described in detail in Chapter 6 (physiology) and consequently, their masseter and medial pterygoideus muscles are their most highly developed masticatory muscles and are innervated (like most muscles of mastication) by the mandibular branch of the 5th cranial nerve. The facial (7th) nerve just innervates the superficial facial muscles (i.e., muscles of expression). The powerful masseter muscle originates along the full length of the facial crest and zygomatic arch and has wide insertions along the caudo-lateral aspect of the mandible, with its deeper fibers running ventrocaudally and its more superficial fibers running almost vertically. Its elevated rostral border is caudal to the site where the facial artery, facial vein and parotid duct cross the ventral border of the mandible and ascend vertically. In horses, the TMJ lies circa 15 cm above the level of the cheek teeth occlusal surface, and thus the movement arm of the masseter is longer. The powerful pterygoideus medialis and lateralis muscles lie on the medial aspect of the mandible, and have similar attachments and orientation to the masseter, and can move the jaw sideways almost continually with a strong power stroke.5 In some horses the pterygoideus muscles are larger than the more obvious masseters. The relatively small digastricus muscle, which attaches from the occipital bone to the caudal aspect of the mandible, functions to open the mouth – a gravity-assisted process that takes little mechanical effort, hence the small muscle size. Horses can generate massive occlusal pressure (up to 875N during the power stroke) during mastication, that is highest between the caudal cheek teeth, i.e., closest to the fulcrum (TMJ).79 Further details on masticatory function and on the TMJ are presented in Chapter 6 (physiology) and Chapter 23 (TMJ).
The articular extremities of the mandible are composed of the condyle that lies caudally and the coronoid process, rostrally. The latter is poorly developed in the horse (Fig. 5.33) because it has smaller temporalis muscles (which close the jaw) compared to carnivores, where the power stroke of the jaws is vertical (to catch and crush prey), consequently both the temporalis muscle and coronoid process are larger in carnivores. Between the articular surfaces of the mandible and the squamous temporal bone lies an articular disc that divides the TMJ cavity in two. The joint capsule is tight and reinforced by an indistinct lateral ligament, and an elastic posterior ligament.51
Although it allows just limited opening of the jaws, the equine TMJ has a wide range of lateral movements to permit the cheek teeth to effectively grind coarse foodstuffs, utilizing a side to side movement that is combined with a slight rostrocaudal movement of the TMJ, with one side gliding rostrally and the other caudally. This rostrocaudal movement can vary greatly between horses and can be demonstrated in some sedated horses by gentle pushing and pulling of the mandible rostrocaudally relative to the TMJ. More clinically significantly, this rostrocaudal mandibular movement can be demonstrated by closing the mouth and then elevating the head – which causes caudal movement of the mandible relative to the maxilla. Lowering the head causes the opposite (rostral) mandibular movement. This maneuver can cause a horse with mild overjet to have normal occlusion on lowering the head. Horses with large focal dental overgrowths may have restriction of their rostrocaudal mandibular movement – but due to individual variation between horses this parameter is difficult to quantify.
Maxillary bones
The upper jaws are largely formed by the paired maxillary bones that contain the alveoli of the upper cheek teeth, wolf teeth and canine teeth (if present). The relationship of the cheek teeth to the maxillary bones and maxillary sinuses has been discussed earlier. As noted, the rostral maxillary area in younger horses may become focally swollen because of the presence of the underlying eruption cysts of the 06s–08s. The overlying bone may become thin and distended, with a temporary and even focal loss of bone over the developing apices occurring, but as noted these features are usually masked by the overlying muscles. Some 3–4-year-old equids (mainly ponies) develop marked bilateral firm swellings of the rostral maxillary bones during eruption of these teeth, giving their face a ‘box-like’ appearance. These are the equivalent of the mandibular eruption cysts (‘osseous tubercles’, ‘3 or 4-year-old bumps’) of this same age group.
The facial crest is a lateral protrusion of the maxilla that continues caudally as the zygomatic process and then joins the zygomatic parts of the malar and temporal bones to form the zygomatic arch (Fig. 5.33). After giving off a small branch that runs rostrally to innervate the maxillary incisor teeth, the infraorbital nerve (a sensory branch of cranial nerve V) emerges through the infraorbital foramen, circa 5 cm dorsal to the rostral aspect of the facial crest. Its point of exit is covered by the pencil-like levator labii superiorus muscle, that can be dorsally displaced to allow local anesthesia of this nerve within the canal to anesthetise the upper 06 (possibly the 07), wolf teeth, canines, and incisors.
The dorsal and caudal borders of the maxillary bone are attached to the nasal and lacrimal bones respectively, whilst rostrally, the maxillary bone is attached to the premaxilla (incisive bone). The thicker ventral border of the maxillary bones contains the alveoli. The individual cheek teeth alveoli are fully separated by transverse, inter-alveolar bony septa. As noted, the equine maxillary sinuses are uniquely divided into rostral and caudal compartments by a thin, transversely angulated bony septum that can vary greatly in position. The medial aspect of each maxillary bone forms a horizontal bony shelf (the palatine process) that joins mid-line with its opposite counterpart to form the supporting bone of most of the hard palate; the remainder of the hard palate is supported by similar flat bony extensions, caudally by the palatine bone, and rostrally by the premaxilla (incisive bone).
Premaxillary (incisive) bone
The paired premaxillary (incisive) bones form the rostral aspect of the upper jaw. Their thick rostral aspects contain the alveoli of the incisors, whilst their thinner caudal aspects form the rostral aspect of the hard palate. The almost transverse suture line between the premaxillae and maxillae is an anatomically weak site that is a common site of fractures, especially in young horses. The canine teeth (if present) lie on the maxillary side of this suture.
Oral mucosa
The mucosa of the gingiva and hard palate is a specialized masticatory mucosa. It can be keratinized, orthokeratinized or parakeratinized and has deep interdigitating rete pegs extending into the underlying vascular, subcutaneous connective tissue that limit its mobility. Most of the gingiva is firmly attached to the supporting bone, with a slightly more mobile (usually non-keratinized) area, termed the free (marginal) gingiva, which is the prominent area close to the tooth. Between the free gingiva and the tooth lies a depression termed the gingival sulcus, which is lined by non-keratinized epithelium. In the deepest area of the gingival sulcus lies the junctional epithelium, which is attached to the peripheral cementum of the tooth, with the periodontal ligament lying directly below this layer. In the horse, with its prolonged dental eruption, this area is constantly remodeling and reforming new periodontal ligaments and new gingival-dental attachments. In other species, interdental papillae of gingiva are present between teeth to prevent food trapping and subsequent periodontal disease, but as noted, most equine teeth are tightly compressed at the occlusual surface and so have no interproximal spaces occlusally.
The salivary glands
The (paired) main equine salivary glands are the parotid, mandibular, and sublingual glands whose ducts drain directly into the mouth. Minor salivary glandular tissue is also present in the lips, tongue, palate, and buccal regions. The largest salivary gland is the parotid, which is circa 20–25 cm long, 2–3 cm thick, and weighs circa 200g in Thoroughbreds, producing up to 50 ml of saliva/min. This salivary gland lies behind the horizontal ramus of the mandible, ventral to the base of the ear, rostral to the wing of atlas, extending ventrally just caudal to the mandible as far as the tendon of origin of the sternomandibularis muscle and the external maxillary vein (Fig. 5.38).5,51 The lateral aspect of the parotid salivary gland is usually level with the masseter muscle, except for a small flat area of this salivary gland which can protrude above the surface of the masseter muscle at the level of the lateral canthus of the eye, and which often overlies some of the parotid lymph nodes at this site. In some apparently normal horses, the parotid salivary gland may swell and protrude 1–3 cm above the masseter muscle level when they are turned out to grass, in the ill-defined condition termed idiopathic parotitis (colloquially termed ‘grass glands’).
The jugular vein is often embedded in the parotid salivary gland and the medial aspect of this gland covers the stylohyoid bone, carotid artery, facial nerve, guttural pouch and the origins of the brachiocephalicus and sternocephalicus muscles – the latter separating the parotid and mandibular salivary glands.5 The dorsal aspect of the parotid salivary gland contains lymphatic tissue within its substance or lying beneath it, which can become focally distended following purulent infections (especially strangles) of that region. The parotid duct originates from an amalgamation of 3–4 large ducts that converge on the rostroventral aspect of the parotid gland and this large duct then initially runs on the medial aspect of the pterygoideus muscles (and mandible), and then crosses beneath the ventral aspect of the mandible, just caudal to the facial artery and vein. The parotid duct then moves dorsally on the lateral aspect of the mandible, moving rostral to the accompanying vasculature, and it perforates the cheek at the level of the upper 08s35 (Fig. 5.38). At its caudal sites, the parotid duct is vulnerable to injury during cheek teeth repulsion, whilst it can be damaged more rostrally by cheek teeth extraction by the lateral buccotomy technique.80
The smaller, (20–25 cm long and 2–3 cm wide, circa 50 g weight in adult medium-sized horses) mandibular salivary gland lies deep to the mandible and parotid salivary glands and so is not palpable. It curves around beneath the parotid salivary gland and mandible, extending from the base of the atlas as far ventrally as the basihyoid bone. Its duct arises on its concave aspect and travels almost the full length of the oral cavity in the sublingual fold, running beside the sublingual salivary glands rostrally, and enters into the oral cavity on the lateral aspect of the sublingual caruncle.5 The long, thin sublingual salivary glands lie superficially in the floor of the mouth beneath the sublingual fold of the oral mucosa. They lie between the tongue and mandible, extending from the mandibular symphysis to the level of the lower 09 and then drain through multiple small ducts into the oral cavity.51
1 Berkovitz BKB, Moxham B. Development of dentition: early stages of tooth development. In: Osborn JW, ed. Dental Anatomy and Embryology. Oxford: Blackwell Scientific; 1981:166–174.
2 Mitchell SR, Kempson SA, Dixon PM. Structure of peripheral cementum of normal equine cheek teeth. Journal of Veterinary Dentistry. 2003;20:199–208.
3 Butler P. Dentition in function. In: Osborn JW, ed. Dental anatomy and embryology. Oxford: Blackwell Scientific; 1991:345.
4 Baker GJ. Oral examination and diagnosis: management of oral diseases. In: Harvey CE, ed. Veterinary dentistry. Philadelphia: WB Saunders; 1985:217–228.
5 Dyce KM, Sack WO, Wensing CJG. Textbook of veterinary anatomy. Philadelphia: WB Saunders; 1987. pp 473–477
6 Easley J, Equine dental development and anatomy. In-Depth Dentistry Seminar. Proceedings of the American Association of Equine Practitioners, 1996:1–10.
7 DeLahunta A, Habel RE. Applied veterinary anatomy. Philadelphia: WB Saunders; 1986. pp 4–16
8 Miles AEW, Grigson C. Colyer’s variations and diseases of the teeth of animals, revised edition. Cambridge: Cambridge University Press; 1990. pp 2–15, 482
9 Kirkland KD, Baker GJ, Marretta SM, et al. Effect of ageing on the endodontic system reserve crown and roots of equine mandibular cheek teeth. American Journal of Veterinary Research. 1996;57:31–38.
10 du Toit N, Kemspon SA, Dixon PM. Donkey dental anatomy. Part 1: Gross and computed axial tomography examinations. The Veterinary Journal. 2008;176:338–344.
11 Warshawsky H. The teeth. In: Weiss L, ed. Histology. 5th edn. New York: Macmillan Press; 1983:609–655.
12 Fortelius M. Ungulate cheek teeth: developmental functional and evolutionary interrelations. Acta Zoologica Fennica. 1985;180:1–76.
13 Ten Cate AR. Development of the tooth and its supporting tissues; hard tissue formation and its destruction; dentinogenesis. In: Ten Cate AR, ed. Oral histology. 4th edn. St Louis: CV Mosby; 1994:58–80. 111–119, 147–168
14 Kollar EJ, Lumsden AGS. Tooth morphology: the role of innervation during induction and pattern formation. The Journal of Biological Buccale. 1979;7:49–60.
15 Brescia NJ. Development and growth of the teeth. In: Sicker H, ed. Orban’s Oral Histology and Embryology. 6th edn. London: CV Mosby; 1966:18–37.
16 Latshaw WK. Face mouth and pharynx. In: Latshaw WK, ed. Veterinary developmental anatomy – a clinically oriented approach. Toronto: BC Decker, 1987.
17 Ferguson M. The dentition throughout life. In: Elderton RJ, ed. The Dentition and Dental Care, vol 3. Oxford: Heinemann; 1990:1–18.
18 Suga SJ. Comparative histology of progressive mineralisation patterns of developing incisor enamel of rodents. The Journal of Dental Research. 1979;58:1025–1026.
19 Eisenmann DR. Amelogenesis; enamel structure. In: Ten Cate AR, ed. Oral histology. 4th edn. St Louis: Ed CV Mosby; 1994:218–256.
20 Baker GJ. A study of dental disease in the horse, PhD Thesis. Glasgow University; 1979. pp 3–96
21 Fitzgibbons C, du Toit N, Dixon PM. Anatomical studies of equine cheek teeth infundibulae. Equine Veterinary Journal. 2010;42:37–43.
22 Kilic S, Dixon PM, Kempson SA. A light and ultrastructural examination of calcified dental tissues of horses: 4 Cement and the amelo-cemental junction. Equine Veterinary Journal. 1997;29:213–219.
23 Kilic S, Dixon PM, Kempson SA. A light and ultrastructural examination of calcified dental tissues of horses: 2 Ultrastructural enamel findings. Equine Veterinary Journal. 1997;29:198–205.
24 Simhofer H, Griss R, Zetner K. The use of oral endoscopy for detection of cheek teeth abnormalities in 300 horses. The Veterinary Journal. 2008;178:396–404.
25 Dacre IT, Kempson SA, Dixon PM. Pathological studies of cheek teeth apical infections in the horse.4: Aetiopathological findings in 41 apically infected mandibular cheek teeth. The Veterinary Journal. 2008;178:341–351.
26 Dacre IT, Kempson SA, Dixon PM. Pathological studies of cheek teeth apical infections in the horse 5: Aetiopathological findings in 57 apically infected maxillary cheek teeth and histological and ultrastructural findings. The Veterinary Journal. 2008;178:352–363.
27 du Toit N, Kemspon SA. Dixon PM Donkey dental anatomy Part 1: Histological and scanning electron microscopy examinations. The Veterinary Journal. 2008;176:345–353.
28 Shellis P. Dental tissue. In: Osborn JW, ed. Dental anatomy and embryology. Oxford: Blackwell Scientific; 1981:193–209.
29 Stanley HR, White CL, McCray L. The rate of tertiary reparitive dentine formation in the human tooth. Oral Surgery, Oral Medicine and Oral Pathology. 1966;21:180–189.
30 Nanci A. The pulp-dentin complex. In: Nanci A, ed. Ten Cate’s Oral Histology; Development Structure and Function. 7th edn. Elsevier: Mosby; 2007:191–238.
31 Magloire H, Joffre A, Bleicher F. An in vitro model of human dental pulp repair. Journal of Dental Research. 1996;75:1971–1978.
32 Dacre IT, Kempson SA, Dixon PM. Pathological studies of cheek teeth apical infections in the horse 1: Normal endodontic anatomy and dentinal structure of equine cheek teeth. The Veterinary Journal. 2008;178:311–320.
33 Fawcett DW. A textbook of histology. Philadelphia: WB Saunders; 1987. pp 603–618
34 White C, Dixon PM. A study of the thickness of cheek teeth subocclusal secondary dentine in horses of different ages. Equine Veterinary Journal. 2010. in press
35 Muylle S, Simoens P, Lauwers H. A study of the ultrastructure and staining characteristics of the ‘dental star’ of equine incisors. Equine Veterinary Journal. 2002;34:230–234.
36 Kilic S, Dixon PM, Kempson SA. A light and ultrastructural examination of calcified dental tissues of horses: 3 Dentine. Equine Veterinary Journal. 1997;29:206–212.
37 Kempson SA, Davidson M, Dacre IT. The effects of three types of rasps on the occlusal surface of equine cheek teeth: a scanning electron microscopic study. Journal of Veterinary Dentistry. 2003;20:19–27.
38 Kilic S, Dixon PM, Kempson SAX. A light and ultrastructural examination of calcified dental tissues of horses: 1 The occlusal surface and enamel thickness. Equine Veterinary Journal. 1997;29:206–212.
39 Muylle S, Simoens P, Lauwers H. Tubular contents of equine dentin: a scanning electron microscopic study. Journal of Veterinary Medicine Series A. 2000;47:321–330.
40 Holland GR. Morphological features of dentin and pulp related to dentin sensitivity. Archives of Oral Biology. 1994;39:3–11. supplement
41 Dacre I. A pathological study of equine dental disorders PhD Thesis. Edinburgh: University of Edinburgh; 2005.
42 Dacre IT. Equine dental pathology. In: Baker GJ, Easley J. Equine Dentistry. 2nd edn. Philadelphia: Saunders; 2005:91–110.
43 Dixon PM, Copeland AN. The radiological appearance of mandibular cheek teeth in ponies of different ages. Equine Veterinary Education. 1993;5:317–323.
44 Warhonowicz M, Staszyk C, Rohn K, Gasse H. The equine periodontium as a continuously remodeling system: morphometrical analysis of cell proliferation. Archive of Oral Biology. 2006;51:1141–1149.
45 Staszyk C, Gasse H. Primary culture of fibroblasts and cementoblasts of the equine periodontium. Research Veterinary Science. 2007;82:150–157.
46 Jones SJ. Human tissue: cement. In: Osborn JW, ed. Dental anatomy and embryology. Oxford: Blackwell Scientific; 1981:193–209.
47 Capper SR. The effects of feed types on ingestive behaviour in different horse types, BSc Thesis. Edinburgh: University of Edinburgh; 1992.
48 Myers JS. The effects of body size grass height and time of day on the foraging behaviour of horses, MSc Thesis. Aberystwyth: Aberystwyth University; 1994. p. 56
49 Leue G. Zahne. In: Dobberstein J, Pallaske G, Stunzi H. Handbuch der speziellen pathologischen Anatomie der Haustiere. 3rd edn. Berlin: Verlag Paul Parey; 1941:131–132.
50 Bonin SJ, Clayton HM, Lanovaz JL, Johnston T. Comparison of mandibular motion in horses chewing hay and pellets. Equine Veterinary Journal. 2007;39:258–262.
51 Sisson S, Grossman JD. Splanchnology. In The anatomy of domestic animals, 4th edn, Philadelphia: WB Saunders; 1953:406–407.
52 Walmsley JP. Some observations on the value of ageing 5–7 year-old horses by examination of their incisor teeth. Equine Veterinary Education. 1993;5:195–298.
53 Richardson JD, Lane JG, Waldron KR. Is dentition an accurate indication of the age of a horse? Veterinary Record. 1994;137:88–90.
54 St Clair LE, Teeth, 5th edn. Getty R, ed., Sisson and Grossman’s the Anatomy of the Domestic Animals, WB Saunders, 1975;Vol 1;460–470.
55 Floyd MR. The modified Triadan system nomenclature for veterinary dentistry. Journal of Veterinary Dentistry. 1991;8:18–20.
56 Goody PC. Horse anatomy. London: JA Allen; 1983. pp 38–41
57 Eisenmenger E, Zetner K. Veterinary dentistry. Philadelphia: Lea, Febiger; 1985. pp 55–57, 153–157
58 Mueller POE. Equine dental disorders: cause diagnosis and treatment. The Compendium of Continuing Education. 1991;13:1451–1460.
59 du Toit N. An anatomical pathological and clinical study of donkey cheek teeth, PhD thesis. Edinburgh: University of Edinburgh; 2008. p. 416
60 Colyer JF. Variations and diseases of teeth of horses. Transactions of the Odontological Society of Great Britain New Series. 1906;38:47–74.
61 Wafa NSY. A study of dental disease in the horse, MVM Thesis. Dublin: University College; 1988. pp 1–203
62 Huidekoper RS. Age of domestic animals. Philadelphia and London: FA Davis; 1891. pp 33–35
63 Bradley OC. The topographical anatomy of the head and neck of the horse. Edinburgh: W Green; 1923. pp 85–92
64 Perkins J. Quantitative and qualitative anatomy of the equine maxillary sinuses with particular reference to the nasomaxillary aperture, MScThesis. Edinburgh: University of Edinburgh; 2001.
65 Tremaine WH, Dixon PM. Equine sinonasal disorders: a long term study of 277 cases Part I. Historical clinical and ancillary diagnostic findings. Equine Veterinary Journal. 2001;33:274–282.
66 Osborn H. Cement. In: Sicker H, ed. Orban’s Oral histology and embryology. 6th edn. London: CV Mosby; 1966:155–212.
67 Dixon PM. Dental extraction in horses: Indications and preoperative evaluation. Compendium of Continuing Education for the Equine Practitioner. 1997;19:366–375.
68 Dixon PM, Tremaine WH, McGorum BC. Equine dental disease – a long term study of 400 cases part 2: disorders of development eruption and variations in position of the cheek teeth. Equine Veterinary Journal. 1999;31:519–528.
69 du Toit N, Burden FA, Dixon PM. Post mortem survey in 349 donkeys from an aged population 2005–2006. Part 1: prevalence of specific dental disorders. Equine Veterinary Journal. 2008;40:204–208.
70 Taylor AC. An investigation of mandibular width and related dental disorders in the equine oral cavity, thesis. Coventry: Coventry University; 2008.
71 Kilic S. A light and electron microscopic study of the calcified dental tissues in normal horses. PhD Thesis. Edinburgh: University of Edinburgh; 1995.
72 Hayward AF. Human tissue: Pulp. In: Osborn JW, ed. Dental anatomy and embryology. Oxford: Blackwell Scientific; 1981:187–190.
73 Torneck CD. Dentine-pulp complex. In: Ten Cate AR, ed. Oral Histology. 4th edn. St Louis: Mosby; 1994:169–213.
74 Dixon PM. The gross histological and ultrastructural anatomy of equine teeth and their relationship to disease. Proceedings of the 49th Annual Convention of the American Association of Equine Practitioners. 2002;48:421–424.
75 Speed JG. Horses and their teeth. The Journal of the Royal Army Veterinary Corps. 1951;22:136–141.
76 Copeland AN. 1990, unpublished observations
77 Dixon PM, Tremaine WH, McGorum BC. Equine dental disease – a long term study of 400 cases: Part 4 – Apical infections of the cheek teeth. Equine Veterinary Journal. 2000;32:182–194.
78 Tremaine WH. Dental care in horses. Practice Journal of Veterinary Postgraduate Clinical Study. 1997;19(1):86–199.
79 Staszyk C, Lehmann F, Bienert A. Measurement of masticatory forces in the horse. Pferdeheilkunde. 2006;22:12–16.
80 Dixon PM, Gerard M. Oral cavity and salivary glands. In: Auer G, Stick JA. Equine surgery. 3rd edn. St Louis: Elsevier; 2008:321–350.