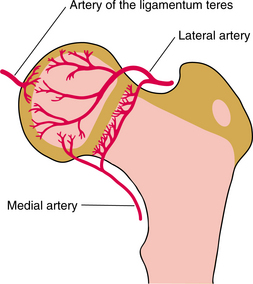
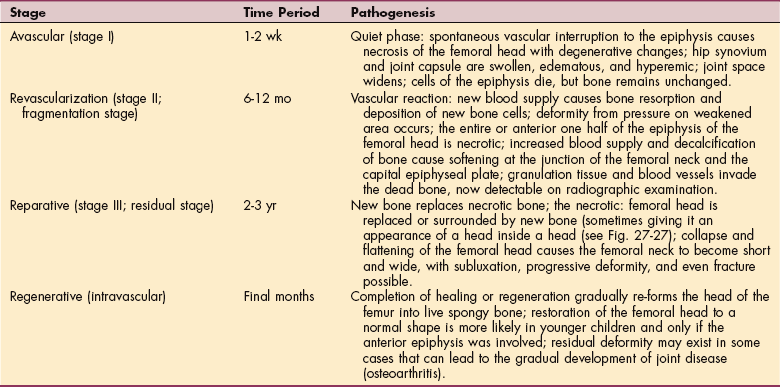
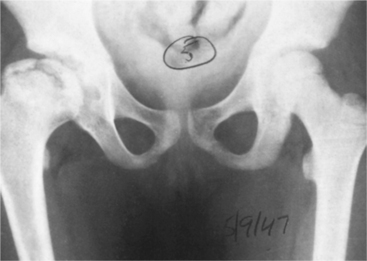
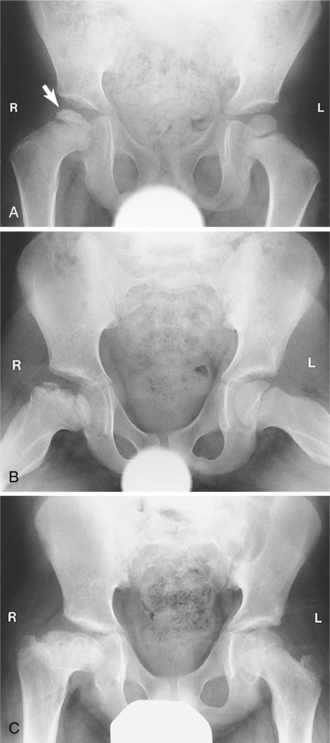
1. Adams, MA. What is intervertebral disc degeneration, and what causes it? Spine. 2006;31(18):2151–2161.
2. Afleck, G. Everyday life with osteoarthritis or rheumatoid arthritis: independent effects of disease and gender on daily pain, mood, and coping. Pain. 1999;83:601–609.
3. Ahuja, A, Brent, L. Revisting the spondyloarthropathies: a new era of treatment. J Musculoskelet Med. 2006;23(9):654–666.
4. Ala-Kokko, L. Genetic risk factors for lumbar disc disease. Ann Med. 2002;34:42–47.
5. Alarcon, GS. Unclassified or undifferentiated connective tissue disease. Baillieres Best Pract Res Clin Rheumatol. 2000;14(1):125–137.
6. Alexanderson, H. Benefits of intensive resistance training in patients with chronic polymyositis or dermatomyositis. Arthritis Rheum. 2007;57(5):768–777.
7. Alexanderson, H. Exercise: an important component of treatment in the idiopathic inflammatory myopathies. Curr Rheumatol Rep. 2005;7:115–124.
8. Alexanderson, H. Safety of a home exercise programme in patients with polymyositis and dermatomyositis: a pilot study. Rheumatology. 1999;38:608–611.
9. Alexanderson, H. The safety of a resistive home exercise program in patients with recent onset active polymyositis or dermatomyositis. Scand J Rheumatol. 2000;29:295–301.
10. Alpay, M, Cassem, EH. Mood disorders in rheumatic disease: evaluation and management. J Musculoskelet Med. 1999;16(11):643–650.
11. Altman, DA, Titus, L, Hair, GA, et al. Molecular biology and spinal disorders. Spine. 1999;24(7):723–730.
12. Altman, R. Development of criteria for the classification and reporting of osteoarthritis: classification of osteoarthritis of the knee, Diagnostic and Therapeutic Criteria Committee of the American Rheumatism Association. Arthritis Rheum. 1986;29:1039–1049.
13. Ambrosio, AM, Sahota, JS, Khan, Y, et al. A novel amorphous calcium phosphate polymer ceramic for bone repair: I. Synthesis and characterization. J Biomed Mater Res. 2001;58(3):295–301.
14. American College of Rheumatology (ACR) Seventieth annual meeting, Washington, DC, November 10-15, 2006. Presentations by Tom WJ Huizinga, MD, chairman of rheumatology, Leiden University Medical Center, Leiden, Netherlands; Steven B Abramson, MD, director of rheumatology, New York University and the Hospital for Joint Diseases, New York; Iain B McInnes, FRCP, PhD, professor of experimental medicine and rheumatology, University of Glasgow, Scotland; Cornelia F Allaart, MD, rheumatologist, Leiden University Medical Center, Netherlands
15. American College of Sports Medicine (ACSM). Guidelines for exercise testing and prescription, ed 7. Philadelphia: Lippincott Williams & Wilkins, 2005.
16. American Geriatrics Society, British Geriatrics Society, American Academy of Orthopaedic Surgeons Panel on Falls Prevention, Guidance for prevention of falls in older persons. Ann Long Term Care 2001;9:42–47. Available on-line at http://www.healthinaging.org/public_education/falls_index.php Accessed July 17, 2007.
17. American Physical Therapy Association (APTA). Guide to physical therapist practice, rev ed 2. Alexandria, VA: The Association, 2003.
18. American Physical Therapy Association (APTA). What you need to know about balance and falls: a physical therapist’s perspective. Alexandria, VA: The Association, 1999. [[Brochure.]].
19. Amin, S. Cigarette smoking and the risk for cartilage loss and knee pain in men with knee osteoarthritis. Ann Rheum Dis. 2007;66(1):18–22.
20. Anaya, JM. Genetic basis of Sjögren’s syndrome: how strong is the evidence? Clin Dev Immunol. 2006;13(2-4):209–222.
21. Andersson, GBJ. Intervertebral disc: clinical aspects. In: Buckwalter JA, Goldberg VM, Woo SL-Y, eds. Musculoskeletal soft tissue aging: impact on mobility. Rosemont, IL: American Academy of Orthopaedic Surgeons; 1993:331–347.
22. Arnett, FC, Chakraborty, R. Ankylosing spondylitis: the dissection of a complex genetic disease. Arthritis Rheum. 1997;40(10):1746–1748.
23. Arthritis Foundation (AF) Alternative treatments for arthritis: an A to Z guide. Marietta, GA: Longstreet Press; 2007. Available on-line at www.arthritis.org Accessed May 30, 2008.
24. Arthritis Foundation (AF) Arthritis self-help course. Atlanta: The Foundation; 2001. Available on-line at www.arthritis.org Accessed May 30, 2008.
25. Arthritis Foundation (AF) Exercise and your arthritis. Atlanta: The Foundation; 2007. Available on-line at http://ww2.arthritis.org/afstore/ProductSearchResults.asp Accessed July 13, 2007.
26. Arthritis Foundation (AF) Water exercise. Atlanta: The Foundation; 2007. Available on-line at http://www.arthritis.org/exercise-water.php Accessed July 13, 2007.
27. Azuma, Y, Ito, M, Harada, Y, et al. Low-intensity pulsed ultrasound accelerates rat femoral fracture healing by acting on the various cellular reactions in the fracture callus. J Bone Miner Res. 2001;16(4):671–680.
28. Baer, AN. Myotoxicity associated with lipid-lowering drugs. Curr Opin Rheumatol. 2007;19(1):67–73.
29. Balboni, TA. Heterotropic ossification: pathophysiology, clinical features, and the role of radiotherapy for prophylaxis. Int J Radiat Oncol Biol Phys. 2006;65(5):1289–1299.
30. Bandura, A. Self-efficacy: toward a unifying theory of behavioral change. Psychol Rev. 1977;84:191–215.
31. Barak, Y, et al. Characteristics of elderly people with a history of falls: a dynamic approach. Phys Ther. 2006;86(11):1501–1510.
32. Baron, JA, Karagas, M, Barrett, J, et al. Basic epidemiology of fractures of the upper and lower limb among Americans over 65 years of age. Epidemiology. 1996;7(6):612–618.
33. Bath Ankylosing Spondylitis Functional Index (BASFI) BASFI test. Available on-line at http://www.basdai.com/BASFI.php/. Accessed July 10, 2007
34. Battié, M.C. Lumbar disc degeneration: epidemiology and genetic influences. Spine. 2004;29:2679–2690.
35. Battié, M.C. 1995 Volvo Award in clinical sciences: determinants of lumbar disc degeneration: a study relating lifetime exposures and magnetic resonance imaging findings in identical twins. Spine. 1995;20:2601–2612.
36. Beauchet, O. Dual-task related gait changes in transitionally frail older adults: the type of the walking-associated cognitive task matters. Gerontology. 2005;51:48–52.
37. Beissel, MD. Acetabular labrum tears: a late complication of Legg-Calvé-Perthes disease. Orthop Pract. 2005;17(1):16–21.
38. Bekkering, WP, ten Cate, R, van Suijlekom-Smit, LW, et al. The relationship between impairments in joint function and disabilities in independent function in children with systemic juvenile idiopathic arthritis. J Rheumatol. 2001;28(5):1099–1105.
39. Bell, MJ, Lineker, SC, Wilkins, AL, et al. A randomized controlled trial to evaluate the efficacy of community based physical therapy in the treatment of people with rheumatoid arthritis. J Rheumatol. 1998;25(2):231–237.
40. Bergstrom, B. Morphology of the vestibular nerve: II. The number of myelinated vestibular nerve fibers in man at various ages. Acta Otolaryngol. 1973;76:173–179.
41. Bhattacharyya, T. Rate and risk factors for acute inpatient mortality after orthopaedic surgery. J Bone Joint Surg Am. 2002;84-A(4):562–572.
42. Bigos, S. Acute low back problems in adults. Rockville, MD: Agency for Healthcare Research and Quality, Public Health Service, U.S. Department of Health and Human Services, 1994. [Clinical practice guideline no. 14].
43. Birks, YF. Randomised controlled trial of hip protectors for the prevention of second hip fractures. Age Ageing. 2003;32(4):442–444.
44. Boden, S. Rheumatoid arthritis of the cervical spine. Spine. 1994;19:2275–2280.
45. Boden, SD. Abnormal magnetic-resonance scans of the lumbar spine in asymptomatic subjects: a prospective investigation. J Bone Joint Surg Am. 1990;72:403–408.
46. Bonnick, SL. Osteoporosis in men and women. Clin Cornerstone. 2006;8(1):28–39.
47. Boonen, A. The burden of ankylosing spondylitis. J Rheumatol Suppl. 2006;78:4–11.
48. Borchers, AT. Juvenile idiopathic arthritis. Autoimmun Rev. 2006;5(4):279–298.
49. Borenstein, D. Inflammatory arthritides of the spine: surgical versus nonsurgical treatment. Clin Orthop Relat Res. 2006;443:208–221.
50. Boulgarides, LK, et al. Use of clinical and impairment based tests to predict falls by community-dwelling older adults. Phys Ther. 2003;83(4):328–339.
51. Braun, J. First update of the international ASAS consensus statement for the use of anti-TNF agents in patients with ankylosing spondylitis. Ann Rheum Dis. 2006;65(3):316–320.
52. Bridges, SL. Spotting aggressive RA early: the physical examination, testing, and imaging. J Musculoskelet Med. 2006:S10–S14. [Suppl].
53. Brody, DM. Running injuries. Clin Symp. 1980;32(4):1–36.
54. Brody, DM. Running injuries: prevention and management. Clin Symp. 1987;39(3):1–36.
55. Brontzman, B. Clinical orthopaedic rehabilitation, ed 2. St Louis: Mosby, 2003.
56. Brophy, S, Calin, A. Ankylosing spondylitis: interaction between genes, joints, age at onset, and disease expression. J Rheumatol. 2001;28(10):2283–2288.
57. Brothers, A. Basic clinical management of muscle strains and tears. J Musculoskelet Med. 2003;20(6):303–307.
58. Buckwalter, JA. Spine update-aging and degeneration of the human intervertebral disc. Spine. 1995;20(11):1307–1314.
59. Buljina, AI, Taljanovic, MS, Avdic, DM, et al. Physical and exercise therapy for treatment of the rheumatoid hand. Arthritis Rheum. 2001;45(4):392–397.
60. Bullough, PG. Orthopaedic pathology, ed 3. London: Mosby-Wolfe, 1997.
61. Burr, DB, Milgrom, C. Musculoskeletal fatigue and stress fractures. Boca Raton, FL: CRC Press, 2000.
62. Calin, A. A new approach to defining functional ability in ankylosing spondylitis: the development of the Bath Ankylosing Spondylitis Functional Index. J Rheumatol. 1994;21(12):2281–2285.
63. Calin, A, Brophy, S, Blake, D. Impact of sex on inheritance of ankylosing spondylitis: a cohort study. Lancet. 1999;354(9191):1687–1690.
64. Cameron, MH. Physical agents in rehabilitation: from research to practice. Philadelphia: Saunders, 1999.
65. Campbell, SK. Physical therapy for children, ed 3. Philadelphia: Saunders, 2005.
66. Cannella, AC. Use of combination therapy for RA. J Musculoskelet Med. 2003;20(4):162–172.
67. Cantini, F, Salvarani, C, Olivieri, I, et al. Inflamed shoulder structures in polymyalgia rheumatica with normal erythrocyte sedimentation rate. Arthritis Rheum. 2001;44(5):1155–1159.
68. Capezuti, E, Evans, L, Strumpf, N, et al. Physical restraint use and falls in nursing home residents. J Am Geriatr Soc. 1996;44(6):727–728.
69. Carmona, L. Increased risk of tuberculosis in patients with rheumatoid arthritis. J Rheumatol. 2003;30:1436–1439.
70. Carragee, EJ. Clinical outcomes after lumbar discectomy for sciatica: the effects of fragment type and anular competence. J Bone Joint Surg. 2003;85-A(1):102–108.
71. Casavant, AM. Heterotopic ossification about the elbow: a therapist’s guide to evaluation and management. J Hand Ther. 2006;19(2):255–266.
72. Centers for Disease Control and Prevention (CDC). Arthritis prevalence and activity limitations-United States, 1990. MMWR Morb Mortal Wkly Rep. 1994;43:433–448.
73. Centers for Disease Control and Prevention (CDC) National Center for Health Statistics,: 2007. Available on-line at http://www.cdc.gov/nchs/ Accessed May 30, 2008
74. Centers for Disease Control and Prevention (CDC). Prevalence of disabilities and associated health conditions among adults-United States, 1999. MMWR Morb Mortal Wkly Rep. 2001;50:120–125.
75. Chao, ST. Treatment of heterotopic ossification. Orthopedics. 2007;30(6):457–466.
76. Charrois, O. Rapid chondrolysis after arthroscopic external meniscectomy. Rev Chir Orthop Reparatrice Appar Mot. 1998;84:88–92.
77. Chen, AL. Rheumatoid arthritis of the shoulder. J Am Acad Orthop Surg. 2003;11(1):12–24.
78. Chikanza, I, Fernandes, L. Novel strategies for the treatment of osteoarthritis. Expert Opin Investig Drugs. 2000;9(7):1499–1510.
79. Christopher-Stine, L. Statin myopathy: an update. Curr Opin Rheumatol. 2006;18(6):647–653.
80. Chung, KC, Spilson, SV. The frequency and epidemiology of hand and forearm fractures in the United States. J Hand Surg Am. 2001;26(5):908–915.
81. Chung, YL. Creatine supplements in patients with idiopathic inflammatory myopathies who are clinically weak after conventional pharmacologic treatment: six-month, double-blind, randomized, placebo-controlled trial. Arthritis Rheum. 2007;57(4):694–702.
82. Cimen, B, Deviren, SD, Yorgancloglu, ZR. Pulmonary function tests, aerobic capacity, respiratory muscle strength and endurance of patients with rheumatoid arthritis. Clin Rheumatol. 2001;20(3):168–173.
83. Claeys, V. Sjögren’s syndrome: a review of the literature and a case report. Rev Belge Med Dent. 2006;61(3):161–172.
84. Clarke, MJ. Long-term incidence of subaxial cervical spine instability following cervicalarthrodesis surgery in patients with rheumatoid arthritis. Surg Neurol. 2006;66(2):136–140.
85. Clarkson, PM. Exertional rhabdomyolysis and acute renal failure in marathon runners. Sports Med. 2007;37(4-5):361–363.
86. Clegg, DO. Glucosamine, chondroitin sulfate, and the two in combination for painful knee osteoarthritis. N Engl J Med. 2006;354(8):795–808.
87. Cohen, M. Anterior cruciate ligament reconstruction after 10 to 15 years: association between meniscectomy and osteoarthrosis. Arthroscopy. 2007;23(6):629–634.
88. Cooney, LM. Hip fracture outcomes. Arch Intern Med. 1997;157:485–486.
89. Cooper, C. Fracture risk in patients with ankylosing spondylitis: a population based study. J Rheumatol. 1994;21:1877–1882.
90. Crock, HV. Internal disc disruption: a challenge to disc prolapse fifty years on. Spine. 1986;11:650–653.
91. Cross, J, et al. Public health and aging: nonfatal fall-related traumatic brain injury among older adults. MMWR Morbid Mortal Wkly Rep. 2003;52(13):276–278.
92. Csizy, M, Buckley, RE, Rennell, C. Benign calcaneal bone cyst and pathologic fracture-surgical treatment with injectable calcium-phosphate bone cement (Norian). Foot Ankle Int. 2001;22(6):507–510.
93. Dagfinrud, H. The Cochrane review of physiotherapy interventions for ankylosing spondylitis. J Rheumatol. 2005;32(10):1899–1906.
94. Dagfinrud, H, Hagen, K. Physiotherapy interventions for ankylosing spondylitis (Cochrane review). Cochrane Database Syst Rev. 2001;4:CD002822.
95. Daubney, ME. Lower-extremity muscle force and balance performance in adults aged 65 years and older. Phys Ther. 1999;79(12):1177–1185.
96. Davidson, MH. Safety of aggressive lipid management. J Am Coll Cardiol. 2007;49(17):1753–1762.
97. De Kleer, IM. Autologous stem cell transplantation for refractory juvenile idiopathic arthritis: analysis of clinical effects, mortality, and transplant related morbidity. Ann Rheum Dis. 2004;63(10):1318–1326.
98. Deane, K. Managing comorbidities in RA. J Musculoskelet Med. 2006:S24–S31. [Suppl].
99. DeFranco, MJ. Radial nerve injuries associated with humeral fractures. J Hand Surg. 2006;31A(4):655–663.
100. deJong, Z. Is a long-term high-intensity exercise program effective and safe in patients with rheumatoid arthritis? Results of a randomized controlled trial. Arthritis Rheum. 2003;48(9):2415–2424.
101. Deyle, GD. Physical therapy treatment effectiveness for osteoarthritis of the knee: a randomized comparison of supervised clinical exercise and manual therapy procedures versus a home exercise program. Phys Ther. 2005;85(12):1301–1317.
102. Deyle, GD, Henderson, NE, Matekel, RL, et al. Effectiveness of manual physical therapy and exercise in osteoarthritis of the knee: a randomized, controlled trial. Ann Intern Med. 2000;132(3):173–181.
103. Dieppe, P. Osteoarthritis: time to shift the paradigm. BMJ. 1999;318:1299–1300.
104. Distler, J. Evidence-based practice: review of clinical evidence on the efficacy of glucosamine and chondroitin in the treatment of osteoarthritis. J Am Acad Nurse Pract. 2006;18(10):487–493.
105. Dite, W, Temple, VA. A clinical test of stepping and change of direction to identify multiple falling older adults. Arch Phys Med Rehabil. 2002;83(11):1566–1571.
106. Divine, JG. Viscosupplementation for knee osteoarthritis. Clin Orthop Relat Res. 2007;455:113–122.
107. Doran, MR. Frequency of infection in patients with rheumatoid arthritis compared to controls: a population-based study. Arthritis Rheum. 2002;46:2287–2293.
108. Dreyer, SJ, Boden, SD. Natural history of rheumatoid arthritis of the cervical spine. Clin Orthop. 1999;366:98–106.
109. Dunkin, MA. Juvenile rheumatoid arthritis. Arthritis Today. 2000:32–33.
110. Elliott, JM, Grainger, AJ, Grigorian, MA, et al. Rheumatoid arthritis: a guide to imaging studies. J Musculoskelet Med. 1999;16(9):507–514.
111. Ensrud, KE. Prevalent vertebral deformities predict mortality and hospitalization in older women with low bone mass, Fracture Intervention Trial Research Group. J Am Geriatr Soc. 2000;48:241–249.
112. Ernst, E. Complementary or alternative therapies for osteoarthritis. Nat Clin Pract Rheumatol. 2006;2(2):74–80.
113. Esselman, PC. Burn rehabilitation: state of the science. Am J Phys Med Rehabil. 2006;85(4):383–413.
114. Ettinger, WH, Burns, R, Messier, SP, et al. A randomized trial comparing aerobic exercise and resistance exercise with a health education program in older adults with knee osteoarthritis. JAMA. 1997;277:25–31.
115. Falkenbach, A. Disability motivates patients with ankylosing spondylitis for more frequent physical exercise. Arch Phys Med Rehabil. 2003;84(3):382–383.
116. Feldman, M. Rheumatoid arthritis. Cell. 1996;85(3):307–310.
117. Feldtkeller, E. Prevalence and annual incidence of vertebral fractures in patients with ankylosing spondylitis. Rheumatol Int. 2006;26(3):234–239.
118. Felson, DR. An update on the pathogenesis and epidemiology of osteoarthritis. Radiol Clin North Am. 2004;42:1–9.
119. Felson, DT, Lawrence, RC, Dieppe, RA, et al. Osteoarthritis: new insights: I. The disease and its risk factors. Ann Intern Med. 2000;133(8):635–646.
120. Felson, DT, Lawrence, RC, Hochberg, MC, et al. Osteoarthritis: new insights: II. Treatment approaches. Ann Intern Med. 2000;133(9):726–737.
121. Fernandez-de-Las-Penas, C. One-year follow-up of two exercise interventions for the management of patients with ankylosing spondylitis: a randomized controlled trial. Am J Phys Med Rehabil. 2006;85(7):559–567.
122. Fernandez-de-Las-Penas, C. Two exercise interventions for the management of patients with ankylosing spondylitis: a randomized controlled trial. Arch Phys Med Rehab. 2005;84(6):407–419.
123. Fiatarone, MA. High-intensity strength training in nonagenarians: effects on skeletal muscle. JAMA. 1990;263(22):3029–3034.
124. Fisher, N, Exercise improves juvenile arthritis. Univ Buffalo Res Q. 2000;10(1). Available on-line at http://www.research.buffalo.edu/quarterly/vol10/num01/f2.shtml Accessed July 12, 2007.
125. Fitzgerald, GK. Role of physical therapy in management of knee osteoarthritis. Curr Opin Rheumatol. 2007;16(2):143–147.
126. Focht, BC. Exercise, self-efficacy, and mobility performance in overweight and obese older adults with knee osteoarthritis. Arthritis Rheum. 2005;53(5):659–665.
127. Focht, BC. The unique and transient impact of acute exercise on pain perception in older, overweight, or obese adults with knee osteoarthritis. Ann Behav Med. 2002;24(3):201–210.
128. Foldes, K, Balint, P, Gaal, M, et al. Nocturnal pain correlates with effusion in diseased hips. J Rheumatol. 1992;19:1756–1758.
129. Foley, A. Does hydrotherapy improve strength and physical function in patients with osteoarthritis-a randomized controlled trial comparing a gym based and a hydrotherapy based strengthening programme. Ann Rheum Dis. 2003;62(12):1162–1167.
130. Fox, PC. Autoimmune diseases and Sjögren’s syndrome: an autoimmune exocrinopathy. Ann N Y Acad Sci. 2007;1098:15–21.
131. Fransen, M, Crosbie, J, Edmons, J, et al. Physical therapy is effective for patients with osteoarthritis of the knee: a randomized controlled clinical trial. J Rheumatol. 2001;28:156–164.
132. Franzen, C, Salzberger, B, Fatkenheuer, G. Avascular necrosis of both femoral heads in an HIV-infected patient receiving protease inhibitors. Eur J Med Res. 2001;6(2):83–84.
133. Frech, TM. The utility of nutraceuticals in the treatment of osteoarthritis. Curr Rheumatol Rep. 2007;9(1):25–30.
134. Fujiwara, K. Cervical lesions related to the systemic progression in rheumatoid arthritis. Spine. 1998;23(19):2052–2056.
135. Fye, KH. Recognizing the extra-articular manifestations of RA. J Musculoskelet Med. 2002;19(8):307–316.
136. Gannotti, ME. Can exercise influence low bone mineral density in children with juvenile rheumatoid arthritis? Pediatr Phys Ther. 2007;19(2):128–139.
137. Garabrant, DH, Dumas, C. Epidemiology of organic solvents and connective tissue disease. Arthritis Res. 2000;2(1):5–15.
138. Garcia-Carrasco, M. Pathophysiology of Sjögren’s syndrome. Arch Med Res. 2006;37(8):921–932.
139. Gardam, M. Rheumatoid arthritis and tuberculosis: time to take notice (editorial). J Rheumatol. 2003;30:1397–1399.
140. Gautschi, OP. Osteoinductive effect of cerebrospinal fluid from brain-injured patients. J Neurotrauma. 2007;24(1):154–162.
141. Gelber, AC, Hochberg, MC, Mead, LA, et al. Joint injury in young adults and risk for subsequent knee and hip osteoarthritis. Ann Intern Med. 2000;133:321–328.
142. Genant, HK, Li, J, Wu, CY, et al. Vertebral fractures in osteoporosis: a new method for clinical assessment. J Clin Densitom. 2000;3(3):281–290.
143. Gennari, L. Osteoporosis in men. Endocrinol Metab Clin North Am. 2007;36(2):399–419.
144. Gerber, L. Exercise and arthritis. Bull Rheum. 1990;39:1–9.
145. Gerwin, RD. A review of myofascial pain and fibromyalgia-factors that promote their persistence. Acupunct Med. 2005;23(3):121–134.
146. Gerwin, RD. An expansion of Simons’ integrated hypothesis of trigger point formation. Curr Pain Headache Rep. 2004;8(6):468–475.
147. Gholve, PA. Osgood Schlatter syndrome. Curr Opin Pediatr. 2007;19(1):44–50.
148. Ghose, RR. Vitamin D deficiency and muscle weakness in the elderly. N Z Med J. 2005;118(1219):U1582.
149. Glueck, CJ, Freiberg, RA, Crawford, A, et al. Secondhand smoke, hypofibrinolysis, and Legg-Perthes disease. Clin Orthop. 1998;352:159–167.
150. Gold, DT. Group treatment improves trunk strength and psychological status in older women with vertebral fractures: results of a randomized, clinical trial. J Am Geriatr Soc. 2004;52(9):1471–1478.
151. Gold, L. Levofloxacin-induced tendon rupture: a case report and review of the literature. J Am Board Fam Pract. 2003;16(5):458–460.
152. Goldberg, SH. Pharmacologic therapy for osteoarthritis. Am J Orthop. 2002;31(12):673–680.
153. Goldman, AB, Freiberger, RH. Localized infectious and neuropathic disease. Semin Roentgenol. 1979;14:19–32.
154. Goldstein, SA. Tissue engineering solutions for traumatic bone loss. J Am Acad Orthop Surg. 2006;14(10):S152–S156.
155. Gonzalez-Gay, MA. Genetic epidemiology: giant cell arteritis and polymyalgia rheumatica. Arthritis Res. 2001;3(3):154–157.
156. Goodman, CC, Snyder, TE. Differential diagnosis for physical therapists: screening for referral, ed 4. Philadelphia: Saunders, 2007.
157. Green, MJ, Deodhar, AA. Bone changes in early rheumatoid arthritis. Baillieres Best Pract Res Clin Rheumatol. 2001;15(1):105–123.
158. Greenspan, A. Evaluating suspected stress fracture. J Musculoskelet Med. 1999;16(3):144.
159. Greenspan, SL, Myers, ER, Maitland, LA, et al. Fall severity and BMD as risk factors for hip fracture in ambulatory elderly. JAMA. 1994;271:128–133.
160. Gryfe, CI, Amies, A, Ashley, MJ. A longitudinal study of falls in an elderly population: I. Incidence and morbidity. Age Ageing. 1977;6:201–210.
161. Grzegorzewski, A. The role of the acetabulum in Perthes disease. J Pediatr Orthop. 2006;26(3):316–321.
162. Guccione, AA. Geriatric physical therapy, ed 2. St Louis: Mosby, 2000.
163. Guccione, AA, Fagerson, TL, Anderson, JJ. Regaining functional independence in the acute care setting following hip fracture. Phys Ther. 1996;76(8):818–826.
164. Günther, KP. Prevalence of generalised osteoarthritis in patients with advanced hip and knee osteoarthritis: the Ulm Osteoarthritis Study. Ann Rheum Dis. 1998;57:717–723.
165. Gür, H. Muscle mass, isokinetic torque, and functional capacity in women with osteoarthritis of the knee. Arch Phys Med Rehabil. 2003;84:1534–1541.
166. Guthrie, B. Review of medications used in juvenile rheumatoid arthritis. Pediatr Emerg Care. 2007;23(1):38–46.
167. Häkkinen, A. Effectiveness and safety of strength training in rheumatoid arthritis. Curr Opin Rheumatol. 2004;16(2):132–137.
168. Häkkinen, A. Effects of concurrent strength and endurance training in women with early or longstanding rheumatoid arthritis: comparison with healthy subjects. Arthritis Rheum. 2003;49(6):789–797.
169. Häkkinen, A, Sokka, T, Kotaniemi, A, et al. A randomized-two year study of the effects of dynamic strength training on muscle strength, disease activity, functional capacity, and bone mineral density in early rheumatic arthritis. Arthritis Rheum. 2001;44(3):515–522.
170. Hall, CM, Brody, LT. Therapeutic exercise: moving towards function, ed 2. Philadelphia: Lippincott Williams & Wilkins, 1998.
171. Hall, T, Zusman, M, Elvey, R. Adverse mechanical tension in the nervous system? Analysis of straight leg raise. Man Ther. 1998;3(3):140–146.
172. Hannallah, D, Kange, J. Use of growth factors for bone repair. J Musculoskelet Med. 2003;20(8):365–372.
173. Hansen, BP. Postarthroscopic glenohumeral chondrolysis. Am J Sports Med. 2007;35(10):1628–1634.
174. Hanten, WP, Olson, SL, Butts, NL, et al. Effectiveness of a home program of ischemic pressure followed by sustained stretch for treatment of myofascial trigger points. Phys Ther. 2000;80(10):997–1003.
175. Harada, ND. Patterns of rehabilitation utilization after hip fracture in acute hospitals and skilled nursing facilities. Med Care. 2000;38(11):1119–1130.
176. Harkcom, TM, Lampman, RM, Banwell, BF, et al. Therapeutic value of graded aerobic exercise training in rheumatoid arthritis. Arthritis Rheum. 1985;28:32–39.
177. Harris, E. Rheumatoid arthritis: pathophysiology and implications for therapy. N Engl J Med. 1990;322:1277–1289.
178. Harris ED, ed. Kelley’s textbook of rheumatology, ed 7, Philadelphia: Saunders, 2005.
179. Harrison, AL. The influence of pathology, pain, balance, and self-efficacy on function in women with osteoarthritis of the knee. Phys Ther. 2004;84(9):822–831.
180. Hartman, CA, Manos, TM, Winter, C, et al. Effects of T’ai Chi training on function and quality of life indicators in older adults with osteoarthritis. J Am Geriatr Soc. 2000;48(12):1553–1559.
181. Hausdorff, JM, et al. Gait variability and fall risk in community-living older adults: a 1-year prospective study. Arch Phys Med Rehabil. 2001;82(8):1050–1056.
182. Hazes, JMW, Dijkmans, BAC, Vandenbroucke, JP, et al. Pregnancy and the risk of developing rheumatoid arthritis. Arthritis Rheum. 1990;33:1770–1775.
183. Headley, B. Myofascial pain syndrome. Fibromyalgia Netw. 2007;76:2–6.
184. Headley, BJ. For stabilization, timing is everything. Adv Phys Ther PT Assist. 2005;16(21):54.
185. Healthy people 2010: arthritis objectives 2001. Available on-line at www.health.gov/healthypeople/ Accessed May 30, 2008
186. Heikkila, S, Viitanen, JV, Kautiainen, H, et al. Sensitivity to change of mobility tests: effect of short term intensive physiotherapy and exercise on spinal, hip, and shoulder measurements in spondyloarthropathy. J Rheumatol. 2000;27(5):1251–1256.
187. Heinonen, M. Post-operative degree of mobilization at two weeks predicts one-year mortality after hip fracture. Aging Clin Exp Res. 2004;16(6):476–480.
188. Henroitin, Y, Labasse, A, Zheng, SX, et al. Strontium ranelate increases cartilage matrix formation. J Bone Miner Res. 2001;16(2):299–308.
189. Herkowitz, HN. Spine update: degenerative lumbar spondylolisthesis. Spine. 1995;20:1084–1090.
190. Herrero-Beaumont, G. Glucosamine sulfate in the treatment of knee osteoarthritis symptoms: a randomized, double-blind, placebo-controlled study using acetaminophen as a side comparator. Arthritis Rheum. 2007;56(2):555–567.
191. Herring, JA. Legg-Calvé-Perthes disease: Part II: Prospective multicenter study of the effect of treatment on outcome. J Bone Joint Surg Am. 2004;86-A(10):2121–2134.
192. Hicks, JE. Rehabilitating patients with idiopathic inflammatory myopathy. J Musculoskelet Med. 1995;12(4):41–54.
193. Hicks, JE. Role of rehabilitation in the management of myopathies. Curr Opin Rehabil. 1998;10(6):548–555.
194. Hicks, JE, Gerber, L. Rehabilitation of the patient with arthritis and connective tissue disease. In: Delisa J, ed. Rehabilitation medicine. ed 2. Philadelphia: Lippincott; 1993:1047–1081.
195. Hicks, JE, Miller, F, Plotz, P, et al. Isometric exercise increases strength and does not produce sustained creatine phosphokinase increases in a patient with polymyositis. J Rheumatol. 1993;20:1399–1402.
196. Hinman, RS. Aquatic physical therapy for hip and knee osteoarthritis: results of a single-blind randomized controlled trial. Phys Ther. 2007;87(1):32–43.
197. Hitchon, PW. Fractures of the thoracolumbar spine complicating ankylosing spondylitis. J Neurosurg. 2002;97:218–222.
198. Hoenig, H, Rubenstein, LV, Sloane, R, et al. What is the role of timing in the surgical and rehabilitative care of community-dwelling older persons with acute hip fracture? Arch Intern Med. 1997;157:513–520.
199. Hoffman, RW, Greidinger, EL. Mixed connective tissue disease. Curr Opin Rheumatol. 2000;12(5):386–390.
200. Hong, CZ, Simons, DG. Pathophysiologic and electrophysiologic mechanisms of myofascial trigger points. Arch Phys Med Rehabil. 1998;79:863–872.
201. Hoover, RT. Avascular necrosis of the femoral head and hemiarthroplasty. Phys Ther Case Rep. 2000;3(1):25–27.
202. Hoppenfeld, S, Murthy, VL. Treatment and rehabilitation of fractures. Philadelphia: Lippincott Williams & Wilkins, 2000.
203. Hornbrook, MC, et al. Preventing falls among community-dwelling older persons: results from a randomized trial. Gerontologist. 1994;34(1):16–23.
204. Horowitz, S, Evinson, B, Borer, A, et al. Mycoplasma fermentans in rheumatoid arthritis and other inflammatory arthritides. J Rheumatol. 2000;27(12):2747–2753.
205. Hotchkiss, A, et al. Convergent and predictive validity of three scales related to falls in the elderly. Am J Occup Ther. 2004;58(1):100–103.
206. Hsu, S. A new approach to managing oral manifestations of Sjögren’s syndrome and skin manifestations of lupus. J Biochem Mol Biol. 2006;39(3):229–239.
207. Hudson, JI, Kenzora, JE, Hebel, JR, et al. Eight-year outcome associated with clinical options in the management of femoral neck fractures. Clin Orthop. 1998;348:59–66.
208. Hughes, MA, Cooperman, J, Peterson, C, et al. Partial weight-bearing in the older person. Top Geriatr Rehabil. 1996;11(3):1–8.
209. Hund, E. Critical illness polyneuropathy. Curr Opin Neurol. 2001;14(5):649–653.
210. Hunder, GG, Kaye, RL, Lane, NE. Osteoporosis, fibromyalgia, Lyme disease, polymyalgia, lupus, and gout. J Musculoskelet Med. 1999;16(1):12–16. [31-32].
211. Hunt, JL. Heterotopic ossification revisited: a 2-year experience. J Burn Care Res. 2006;27(4):535–540.
212. Hunt, SA. Arthroscopic management of osteoarthritis of the knee. J Am Acad Orthop Surg. 2002;10:356–363.
213. Hunter, DJ. Knee alignment does not predict incident osteoarthritis: the Framingham osteoarthritis study. Arthritis Rheum. 2007;56(4):1212–1218.
214. Hunter, DJ. Patella malalignment, pain and patellofemoral progression: the Health ABC Study. Osteoarthritis Cartilage. 2007;15(10):1120–1127.
215. Hunter, DJ, Felson, DT. Osteoarthritis: clinical review. BMJ. 2006;332:638–642.
216. Hutton, JT. Preventing falls: a defensive approach. New York: Prometheus Books, 2000.
217. Hyrich, KL, Inman, RD. Infectious agents in chronic rheumatic diseases. Curr Opin Rheumatol. 2001;13(4):300–304.
218. Iaria, J, Houseknecht, H, Craik, R. Effects of hip fracture on impairment and function in older persons. Phys Ther. 2001;81(5):A17. [(abstract)].
219. Ince, G. Effects of a multimodal exercise program for people with ankylosing spondylitis. Phys Ther. 2006;86(7):924–935.
220. Inman, RD. Mechanisms of disease: infection and spondyloarthritis. Nat Clin Pract Rheumatol. 2006;2(3):163–169.
221. Intrator, O. Benefits of home health care after inpatient rehabilitation for hip fracture. Arch Phys Med Rehabil. 1998;79(10):1195–1199.
222. Ismail, AA. Prevalent vertebral deformity predicts incident hip though not distal forearm fracture: results from the European Prospective Osteoporosis Study. Osteoporosis Int. 2001;12:85–90.
223. Iversen, MD. Physical therapy for management of RA. J Musculoskelet Med. 2002;19(9):352–362.
224. James, J, Steijn-Myagkaya, GL. Death of osteocytes: electron microscopy after in vitro ischemia. J Bone Joint Surg Br. 1986;68:620–624.
225. Janowsky, EC, Kupper, LL, Hulka, BS. Meta-analysis of the relation between silicone breast implants and the risk of connective-tissue diseases. N Engl J Med. 2000;342:781–790.
226. Järvinen, M. The effect of early mobilization and immobilization on the healing process following muscle injuries. Sports Med. 1993;15:78–89.
227. Järvinen, TAH. Muscle injuries: biology and treatment. Am J Sports Med. 2005;33(5):745–764.
228. Jensen, MC. Magnetic resonance imaging of the lumbar spine in people without back pain. N Engl J Med. 1994;331:69–73.
229. Johnson, EO. Hypothalamic-pituitary-adrenal axis function in Sjögren’s syndrome: mechanisms of neuroendocrine and immune system homeostasis. Ann N Y Acad Sci. 2006;1088:41–51.
230. Johnson, EO, Skopouli, FN, Moutsopoulos, HM. Neuroendocrine manifestations in Sjögren’s syndrome. Rheum Dis Clin North Am. 2000;26(4):927–949.
231. Jonssou, B, Stromquist, B. Symptoms and signs in degeneration of the lumbar spine. J Bone Joint Surg Br. 1993;75:381–385.
232. Joseph, B. Natural evolution of Perthes disease: a study of 610 children under 12 years of age at disease onset. J Pediatr Orthop. 2003;23:590–600.
233. Min, JH, Jang, JS, Lee, SH. Comparison of anterior-and posterior-approach instrumented lumbar interbody fusion for spondylolisthesis. J Neurosurg Spine. 2007;7(1):21–26.
234. Kalichman, L, et al. The association between patellar alignment and patellofemoral joint osteoarthritis features-an MRI study. Rheumatology (Oxford). 2007;9(2):R26.
235. Kalichman, L. The association between patellar alignment on magnetic resonance imaging and radiographic manifestations of knee osteoarthritis. Arthritis Res Ther. 2007;9(2):R26.
236. Kannus, P. Prevention of hip fracture with hip protectors. Age Ageing. 2006;35(suppl 2):ii51–ii54.
237. Kapural, L. Novel intradiscal biacuplasty (IDB) for the treatment of lumbar discogenic pain. Pain Pract. 2007;7(2):130–134.
238. Kauppi, M, Anttila, P. A stiff collar can restrict atlantoaxial instability in rheumatoid cervical spine in selected cases. Ann Rheum Dis. 1995;54:305–307.
239. Kauppi, M, Anttila, P. A stiff collar for the treatment of rheumatoid atlantoaxial subluxation. Br J Rheumatol. 1996;35:771–774.
240. Kauppila, LI. MR aortography and serum cholesterol levels in patients with long-term nonspecific lower back pain. Spine. 2004;29(19):2147–2152.
241. Keating, RM, Vyas, A. Reactive arthritis following Clostridium difficile colitis. West J Med. 1995;162(1):61–63.
242. Kellgren, J, Lawrence, J. Radiologic assessment of osteoarthritis. Ann Rheum Dis. 1957;16:494–501.
243. Kellgren, JH. Observations of referred pain arising from muscle. Clin Sci. 1938;3:175.
244. Kelly, AKW. Managing stress fractures in athletes. J Musculoskelet Med. 2005;22(9):463–472.
245. Kerrigan, DC. Effectiveness of a lateral-wedge insole on knee varus torque in patients with knee osteoarthritis. Arch Phys Med Rehabil. 2002;83(7):889–893.
246. Kiebzak, GM. Undertreatment of osteoporosis in men with hip fracture. Arch Intern Med. 2002;162(19):2217–2222.
247. Kiebzak, GM. Vitamin D status of patients admitted to a hospital rehabilitation unit: relationship to function and progress. Am J Phys Med Rehabil. 2007;86(6):435–445.
247a. Kim, DH. Factors influencing segmental range of motion after lumbar total disc replacement using the ProDisc II prosthesis. J Neurosurg Spine. 2007;7(2):131–138.
248. Kim, DH. Rheumatoid arthritis in the cervical spine. J Am Acad Orthop Surg. 2005;13(7):463–474.
249. Kim, M. Transverse sacral fractures: case series and literature review. Can J Orthop Surg. 2001;44:359–363.
250. Kirk-Sanchez, NJ, Roach, KE. Factors predicting mobility at discharge following rehabilitation for femur fracture. Phys Ther. 2001;81(5):A16. [(abstract)].
251. Kirkaldy-Willis, WH, Hill, RJ. A more precise diagnosis for low back pain. Spine. 1979;4:102–109.
252. Kirschner, RE, Karmacharya, J, Ong, G, et al. Synthetic hybrid grafts for craniofacial reconstruction: sustained gene delivery using a calcium phosphate bone mineral substitute. Ann Plast Surg. 2001;46(5):538–545.
253. Knop, C, Lange, U, Bastian, L, et al. Biomechanical compression tests with a new implant for thoracolumbar vertebral body replacement. Eur Spine J. 2001;10(1):30–37.
254. Koch, AE. Targeting cytokines and growth factors in RA. J Musculoskelet Med. 2005;22(3):130–136.
255. Komatireddy, GR, Leitch, RW, Cella, K, et al. Efficacy of low load resistive training in patients with rheumatoid arthritis functional class II and III. J Rheumatol. 1997;24(8):1531–1539.
256. Konig, A. Insufficiency fractures of the ileum and sacrum. Z Rheumatol. 2000;59(5):343–347.
257. Konttinen, YT. Immunohistology of Sjögren’s syndrome. Autoimmun Rev. 2006;6(1):16–20.
258. Kostopoulos, D, Rizopoulos, K, Kurman, RJ, et al. The manual of trigger point and myofascial therapy. Thorofare, NJ: Slack, 2001.
259. Koval, KJ, Zuckerman, JD. Hip fractures: a practical guide to management. New York: Springer-Verlag, 2000.
260. Kraemer, J. Natural course and prognosis of intervertebral disc diseases. Spine. 1995;20:635–639.
261. Krebs, DE, et al. Is base of support greater in unsteady gait? Phys Ther. 2002;82(2):138–147.
262. Kristmundsdottir, F. Delayed skeletal maturation in Perthes’ disease. Acta Orthop Scand. 1987;58(3):277–279.
263. Kruithof, E. Identification of synovial biomarkers of response to experimental treatment in early-phase clinical trials in spondyloarthritis. Arthritis Rheum. 2006;54(6):1733–1735.
264. Kujala, UM. Hamstring injuries: current trends in treatment and prevention. Sports Med. 1997;23:397–404.
265. Kurowski, P. The relationship of degeneration of the intervertebral disc to mechanical loading on lumbar vertebrae. Spine. 1986;11:726–731.
266. Lacomis, D. Causes of neuromuscular weakness in the intensive care unit: a study of 92 patients. Muscle Nerve. 1998;21(5):610–617.
267. Lacomis, D. Critical illness myopathy. Curr Rheumatol Rep. 2002;4(5):403–408.
268. Laing, TJ, Schottenfeld, D, Lacey, JV, et al. Potential risk factors for undifferentiated connective tissue disease among women: implanted medical devices. Am J Epidemiol. 2001;154(7):610–617.
269. Laurencin, CT, Attawia, MA, Lu, LQ, et al. Poly/hydroxyapatite delivery of BMP-2-producing cells: a regional gene therapy approach to bone regeneration. Biomaterials. 2001;22(11):1271–1277.
270. Lavernia, CJ, Sierra, RJ, Grieco, FR. Osteonecrosis of the femoral head. J Am Acad Orthop Surg. 1999;7(4):250–261.
271. Lawley, J, Thompkins, P. Metaphors in mind. Highgate, London: Developing Company Press, 2000.
272. Lawlor, DA, et al. Association between falls in elderly women and chronic disease and drug use: cross sectional study. BMJ. 2003;327(7417):712–717.
273. LeBoff, MS, Kohlmeier, L, Hurwitz, S, et al. Occult vitamin D deficiency in postmenopausal US women with acute hip fracture. JAMA. 1999;281(16):1505–1511.
274. Lee, S. Bone age delay patterns in Legg-Calvé-Perthes disease. J Pediatr Orthop. 2007;27(2):198–203.
275. Leech, JA. Relationship of lung function to severity of osteoporosis in women. Am Rev Respir Dis. 1990;141:951–956.
276. Leoux, JL. Sacral insufficiency fractures presenting as acute low-back pain-biomechanical aspects. Spine. 1993;18(12):2502–2506.
277. Lephart, SM. Proprioception and neuromuscular control in joint stability. Champaign, IL: Human Kinetics, 2000.
278. Levine, WN. Chondrolysis following arthroscopic thermal capsulorrhaphy to treat shoulder instability. J Bone Joint Surg Am. 2005;87:616–621.
279. Lewis, C. Discovering best treatments for hip fractures. Adv Phys Ther PT Assist. 2002;13(11):6–7.
280. Lewis, C. PTs should be hip to orthopedic rehab. Adv Phys Ther PT Assist. 2003;14:6, 49.
281. Lewis, C. Shoulder fracture. Adv Phys Ther PT Assist. 2003;14(18):6–7.
282. Lewis, R. Ankylosing spondylitis: early diagnosis and management. J Musculoskelet Med. 2003;20(4):184–198.
283. Lewit, D, Simons, DG. Myofascial pain: relief by post-isometric relaxation. Arch Phys Med Rehabil. 1984;65:452–456.
284. Lie, H. Mobilizing or stabilizing exercise in degenerative disk disease in the lumbar region? Tidsskr Nor Laegeforen. 1999;119(14):2052–2053.
285. Lin, DH. Comparison of proprioceptive functions between computerized proprioception facilitation exercise and closed kinetic chain exercise in patients with knee osteoarthritis. Clin Rheumatol. 2007;26(4):520–528.
286. Lindsay, R. Risk of new vertebral fracture in the year following a fracture. JAMA. 2001;285:320–323.
287. Lineker, SC, Bell, MJ, Wilkins, AL, et al. Improvements following short term home based physical therapy are maintained at one year in people with moderate to severe rheumatoid arthritis. J Rheumatol. 2001;28(1):165–168.
288. Lo, TC. Heterotopic bone formation after hip surgery: prevention with single-dose postoperative hip irradiation. Radiology. 1988;168:851–854.
289. Long, T. Handbook of pediatric physical therapy. Philadelphia: Lippincott Williams & Wilkins, 2001.
290. Lord, SR. A physiological profile approach to falls risk assessment and prevention. Phys Ther. 2003;83(3):237–252.
291. Lorig, KR, Mazonson, PD, Holman, HR. Evidence suggesting that health education for self-management in patients with chronic arthritis has sustained health benefits while reducing health care costs. Arthritis Rheum. 1993;36:439–446.
292. Lotke, PA, Battish, R, Nelson, CL. Treatment of osteonecrosis of the knee. Instr Course Lect. 2001;50:483–488.
293. Lovell, DJ. Update on treatment of arthritis in children: new treatments, new goals. Bull N Y U Hosp Jt Dis. 2006;64(1-2):72–76.
294. Lowe, JC, Honeyman-Lowe, G. Ultrasound treatment of trigger points: differences in technique for myofascial pain syndrome and fibromyalgia. Myalgia ’99. 1999;2:12–15. [Available from the Fibromyalgia Research Foundation, PO Box 396, Tulsa, OK 74101-0396].
295. Lozada, CJ. Glucosamine in osteoarthritis: questions remain. Cleve Clin J Med. 2007;74(1):65–71.
296. Lundberg, IE. Technology insight: tools for research, diagnosis and clinical assessment of treatment in idiopathic inflammatory myopathies. Nat Clin Pract Rheumatol. 2007;3(5):282–290.
297. Luu, HH. Distinct roles of bone morphogenetic proteins in osteogenic differentiation of mesenchymal stem cells. J Orthop Res. 2007;25(5):665–677.
298. MacDonald, CW, Whitman, JM, Cleland, JA, et al. Clinical outcomes following manual physical therapy and exercise for hip osteoarthritis: a case series. J Orthop Sports Phys Ther. 2006;36(8):588–599.
299. Maddison, PJ. Mixed connective tissue disease: overlap syndromes. Baillieres Best Pract Res Clin Rheumatol. 2000;14(1):111–124.
300. Magaziner, J, Lydick, E, Hawkes, W, et al. Excess mortality attributable to hip fracture in white women aged 70 years and older. Am J Public Health. 1997;87(10):1630–1636.
301. Majlesi, J. High-power pain threshold ultrasound technique in the treatment of active myofascial trigger points: a randomized, double-blind, case-control study. Arch Phys Med Rehabil. 2004;85(5):833–836.
302. Maki, BE, et al. Fear of falling and postural performance in the elderly. J Gerontol. 1991;46:M123–M131.
303. Malm, C. Immunological changes in human skeletal muscle and blood after eccentric exercise and multiple biopsies. J Physiol. 2000;529:243–262.
304. Mandelbaum, B. Etiology and pathophysiology of osteoarthritis. Orthopedics. 2005;28(suppl 2):S207–S214.
305. Mangione, KK. The effects of high-intensity and low-intensity cycle ergometry in older adults with knee osteoarthritis. J Gerontol A Biol Sci Med Sci. 1999;54(4):M184–M190.
306. Mannion, AF. A randomized clinical trial of three active therapies for chronic low back pain. Spine. 1999;24:2435–2448.
307. Mansi, IA. Rhabdomyolysis in response to weight-loss herbal medicine. Am J Med Sci. 2004;327(6):356–357.
308. March, LM. Economics of osteoarthritis: a global perspective. Baillieres Clin Rheumatol. 1997;11:817–834.
309. Marinelli, WA. Neuromuscular disorders in the intensive care unit. Crit Care Clin. 2002;18(4):915–929.
310. Martin, TM, Smith, JR, Rosenbaum, JT. Anterior uveitis: current concepts of pathogenesis and interactions with the spondyloarthropathies. Curr Opin Rheumatol. 2002;14:337–341.
311. Martinez-Taboada, VM, Blanco, R, Rodriquez-Valverde, V. Polymyalgia rheumatica with normal erythrocyte sedimentation rate: clinical aspects. Clin Exp Rheumatol. 2000;18(4 suppl 20):S34–S37.
312. Masuko Hongo, K, Kato, T. The association between autoimmune thyroid diseases and rheumatic diseases: a review. Nippon Rinsho. 1999;57(8):1873–1877.
313. Matsumoto, T. Cyclic mechanical stretch stress increases the growth rate and collagen synthesis of nucleus pulposus in vitro. Spine. 1999;24:315–319.
314. Mattila, VM, Niva, M, Kiuru, M, et al. Risk factors for bone stress injuries: a follow-up study of 102,515 person-years. Med Sci Sports Exerc. 2007;39(7):1061–1066.
315. Mauer, AC, et al. Bilateral total knee arthroplasty increases the propensity to trip on an obstacle. Clin Orthop Relat Res. 2005;433:160–165.
316. Mavragani, CP. The management of Sjögren’s syndrome. Nat Clin Pract Rheumatol. 2006;2(5):252–261.
317. Maxey, L, Magnusson, J. Rehabilitation for the postsurgical orthopedic patient, ed 2. St Louis: Mosby, 2006.
318. McDonough, A, Saidoff, D. Clinical pathways in therapeutic intervention: upper extremity. St Louis: Mosby, 1997.
319. McGuire, KJ. The 2004 Marshall Urist Award: delays until surgery after hip fracture increases mortality. Clin Orthop Relat Res. 2004;428:294–301.
320. McKenzie, R. The lumbar spine: mechanical diagnosis and therapy. Waikanae, New Zealand: Spinal Publications, New Zealand, 1981.
321. McLain, RF. Determining treatment of lumbar disk protrusion and disk extrusion. J Musculoskelet Med. 2005;22(1):21–28.
322. McNair, PJ, Marshall, RN, Maguire, K. Swelling of the knee joint: effects of exercise on quadriceps muscle strength. Arch Phys Med Rehabil. 1996;77(9):896–899.
323. Mecagni, C, Smith, JP, Roberts, KE, et al. Balance and ankle range of motion in community-dwelling women aged 64 to 87 years: a correlational study. Phys Ther. 2000;80(10):1004–1011.
324. Medical Multimedia Group (MMG) A patient’s guide to lumbar artificial disc replacement, 2007. Available on-line at http://www.eorthopod.com. Accessed July 11, 2007
325. Medical Multimedia Group (MMG) A patient’s guide to lumbar degenerative disc disease, 2002. Available on-line at http://www.eorthopod.com. Accessed July 11, 2007
326. Medical Multimedia Group (MMG) A patient’s guide to Osgood-Schlatter lesion of the knee. Available on-line at http://www.eorthopod.com. Accessed July 10, 2007
327. Medical Multimedia Group (MMG). A patient’s guide to Perthes disease of the hip. Missoula, MT: MMG, 2005.
328. Meisel, HJ. Clinical experience in cell-based therapeutics: disc chondrocyte transplantation: a treatment for degenerated or damaged intervertebral disc. Biomol Eng. 2007;24(1):5–21.
329. Melaragno, PG, Cheney, RA, Bennett, GL. Lower leg compartment syndromes: when to suspect and how to diagnose. J Musculoskelet Med. 1996;13(11):14–27.
330. Melton-Rogers, S, Hunter, G, Walter, J, et al. Cardiorespiratory responses of patients with rheumatoid arthritis during bicycle riding and running in water. Phys Ther. 1996;76(10):1058–1065.
331. Mense, S, Simons, DG, Russell, IJ. Muscle pain: understanding its nature, diagnosis, and treatment. Philadelphia: Lippincott Williams & Wilkins, 2000.
332. Messier, SP. Exercise and dietary weight loss in overweight and obese older adults with knee osteoarthritis: the Arthritis, Diet, and Activity Promotion Trial. Arthritis Rheum. 2004;50(5):1501–1510.
333. Messier, SP, Royer, TD, Craven, TE, et al. Long-term exercise and its effect on balance in older, osteoarthritic adults: results from the Fitness, Arthritis, and Seniors Trial (FAST). J Am Geriatr Soc. 2000;48(2):131–138.
334. Messinger-Rapport, BJ. Prevention for the older woman: mobility: a practical guide to managing osteoarthritis and falls. Geriatrics. 2003;58(7):22–29.
335. Michlovitz, SL. Thermal agents in rehabilitation, ed 3. Philadelphia: FA Davis, 1996.
336. Mikuls, TR. Coffee, tea, and caffeine consumption and risk of rheumatoid arthritis: results from the Iowa Women’s Health Study. Arthritis Rheum. 2002;46(1):83–91.
337. Miller, D. Endothelial dysfunction and decreased vascular responsiveness in the anterior cruciate ligament-deficient model of osteoarthritis. J Appl Physiol. 2007;102(3):1161–1169.
338. Miller, GD. Intensive weight loss program improves physical function in older obese adults with knee osteoarthritis. Obesity. 2006;14(7):1219–1230.
339. Miller, JA. Lumbar disc degeneration: correlation with age, sex, and spine level in 600 autopsy specimens. Spine. 1988;13:173–178.
340. Min, BW, Koo, KH, Song, HR, et al. Subcapital fractures associated with extensive osteonecrosis of the femoral head. Clin Orthop. 2001;390:227–231.
341. Minden, K. What are the costs of childhood-onset rheumatic disease? Best Pract Res Clin Rheumatol. 2006;20(2):223–240.
342. Minor, MA. Exercise in the treatment of osteoarthritis. Rheum Dis Clin North Am. 1999;25(2):397–415.
343. Minor, MA. Exercise proves a vital tool against rheumatoid arthritis. Arthritis Today. 1997:36–37.
344. Minor, MA, Johnson, JC. Reliability and validity of a submaximal treadmill test to estimate aerobic capacity in women with rheumatic disease. J Rheumatol. 1996;23(9):1517–1523.
345. Minor, MA, Lane, NE. Recreational exercise in arthritis. Rheum Dis Clin North Am. 1996;22(3):563–577.
346. Minor, MA, Sanford, MK. The role of physical therapy and physical modalities in pain management. Rheum Dis Clin North Am. 1999;25(1):233–248.
347. Moder, KG. A working guide to joint examination in rheumatoid arthritis. J Musculoskelet Med. 1995;12(11):17–34.
348. Mohler, R. Painless hand mass in an elderly man. J Musculoskelet Med. 2001;18(3):153–156.
349. Monauni, T, Zenti, MG, Cretti, A, et al. Effects of glucosamine infusion on insulin secretion and insulin action in humans. Diabetes. 2000;49(6):926–935.
350. Monu, JU. Gout: a clinical and radiologic review. Radiol Clin North Am. 2004;42(1):169–184.
351. Moorthy, LN. Juvenile idiopathic arthritis: making the diagnosis. J Musculoskelet Med. 2004;21(11):581–588.
352. Morbidity Mortality Weekly Report (MMWR). Fatalities and injuries from falls among older adults, 2005. MMWR Morbid Mortal Wkly Rep. 2006;55(45):1221–1224.
353. Morbidity Mortality Weekly Report (MMWR). Visual impairment and eye care among older adults, 2005. MMWR Morbid Mortal Wkly Rep. 2006;55(49):1321–1325.
354. Morehead, K. Early diagnosis of RA: the therapeutic implications. J Musculoskelet Med. 2005;22(11):599–606.
355. Moreland, LW. Etanercept therapy in rheumatoid arthritis: a randomized, controlled trial. Ann Intern Med. 1999;130(6):478–486.
356. Morse, CG. The incidence and natural history of osteonecrosis in HIV-infected adults. Clin Infect Dis. 2007;44(5):739–748.
357. Munin, MC, Hockenberry, PS, Flynn, PG, et al, Rehabilitation. Callaghan, JJ, Rosenberg, AG, Rubash, HE, eds. The adult hip, II. Philadelphia: Lippincott-Raven, 1998.
358. Mustafa, K, Khan, MA. Recognizing and managing reactive arthritis. J Musculoskelet Med. 1996;13(6):28–41.
359. Myer, GD. Specialized neuromuscular training to improve neuromuscular function and biomechanics in a patient with quiescent juvenile rheumatoid arthritis. Phys Ther. 2005;85(8):791–802.
360. Nannapaneni, R. Surgical outcome in rheumatoid Ranawat class IIIb myelopathy. Neurosurgery. 2005;56(4):706–715.
361. National Ankylosing Spondylitis Society (NASS) The Bath Indices, 2004. Available on-line at http://www.mc.uky.edu/rheumatology/education/nass-bath-indices.pdf Accessed July 10, 2007
362. National Center for Health Statistics. 1990 and 1991 National hospital discharge survey. Rosemont, IL: American Academy of Orthopedic Surgeons Department of Research and Scientific Affairs, 1994.
363. National Center for Injury Prevention and Control (NCIPC) Preventing falls among older adults. Available on-line at http://www.cdc.gov/ncipc/duip/preventadultfalls.htm Accessed December 26, 2006. Lots of great patient education materials, checklists, posters, prevention strategies
364. National Institutes of Health (NIH). Consensus statement: osteoporosis prevention, diagnosis, and therapy. Bethesda, MD: The Institutes, 2000.
365. National Osteoporosis Foundation Important disease facts, 2001. Available on-line at www.nof.org/other/statistics.html Accessed May 30, 2008
366. Nativ, A. Stress fractures and bone health in track and field athletes. J Sci Med Sport. 2000;3(3):268–279.
367. NeuroCom Assessment Protocols. NeuroCom International, Clackamas, O.R., (800) 767-6744. Available on-line at www.onbalance.com Accessed May 30, 2008. (Reference to this product does not represent endorsement.)
368. Neva, MH. Mortality associated with cervical spine disorders: a population-based study of 1,666 patients with rheumatoid arthritis who died in Finland in 1989. Rheumatology (Oxford). 2001;40:123–127.
369. Nevitt, MC. Risk factors for a first-incident radiographic vertebral fracture in women greater than or equal to 65 years of age: the study of osteoporotic fractures. J Bone Miner Res. 2005;20(1):131–140.
370. Newcomer, K, Jurrison, ML. Rheumatoid arthritis: the role of physical therapy. J Musculoskelet Med. 1994;11:14–26.
371. Newton RA, Fall Prevention Project, Temple University Falls and Fall Prevention, 1999. Available on-line at www.temple.edu/older_adult Accessed July 17, 2007
372. Nicola, PJ. The risk of congestive heart failure in rheumatoid arthritis: a population based study over 46 years. Arthritis Rheum. 2005;52:412–420.
373. Niva, MH. Bone stress injuries causing exercise-induced knee pain. Am J Sports Med. 2006;34(1):78–84.
374. Noonan, TJ. Thermal effects on skeletal muscle tensile behavior. Am J Sports Med. 1993;21:517–522.
375. Norwalk, MP, Prendergast, JM, Bayles, CM, et al. A randomized trial of exercise programs among older individuals living in two long-term care facilities: the FallsFREE program. J Am Geriatr Soc. 2001;49(7):859–865.
376. Nyberg, L, Gustafson, Y, Berggren, D, et al. Falls leading to femoral neck fractures in lucid older people. J Am Geriatr Soc. 1996;44(2):156–160.
377. Oda, T, Fujiwara, K, Yonenobu, K, et al. Natural course of cervical spine lesions in rheumatoid arthritis. Spine. 1995;20:1128–1135.
378. O’Dell, JR, Blakely, KW, Mallek, JA, et al. Treatment of early seropositive rheumatoid arthritis: a two-year, double-blind comparison of minocycline and hydroxychloroquine. Arthritis Rheum. 2001;44(10):2235–2241.
379. O’Halloran, PD. Factors affecting adherence to use of hip protectors amongst residents of nursing homes-a correlation study. Int J Nurs Stud. 2007;44(5):672–686.
380. Olsen, NJ. Initial diagnosis and early management of RA. J Musculoskelet Med. 2005;22(10 suppl):S6–S11.
381. Ostensen, M. Sex, reproduction, and gynecological disease in young adults with a history of juvenile chronic arthritis. J Rheumatol. 2000;27:1783–1787.
382. O’Sullivan, SB. Physical rehabilitation, ed 5. Philadelphia: FA Davis, 2006.
383. Ottawa Panel evidence-based clinical practice guidelines for electrotherapy and thermotherapy interventions in the management of rheumatoid arthritis in adults. Phys Ther. 2004;84(11):1016–1043.
384. Ottawa Panel evidence-based clinical practice guidelines for therapeutic exercises in the management of rheumatoid arthritis in adults. Phys Ther. 2004;84(10):934–972.
385. Paap, E. Physiologic response of the six-minute walk test in children with juvenile idiopathic arthritis. Arthritis Rheum. 2005;53(3):351–356.
386. Paget, SA. Modern-day RA management: resetting the inflammatory thermostat. J Musculoskelet Med. 2006:S3. [Suppl].
387. Paimela, L. Progression of cervical spine changes in patients with early rheumatoid arthritis. J Rheumatol. 1997;24(7):1280–1284.
388. Parker, MJ, Gillespie, LD, Gillespie, WJ. Hip protectors for preventing hip fractures in the elderly. Cochrane Database Syst Rev. 2000;4:CD001255.
389. Parkkari, J, Kannus, P, Palvanen, M, et al. Majority of hip fractures occur as a result of a fall and impact on the greater trochanter of the femur: a prospective controlled hip fracture study with 206 consecutive patients. Calcif Tissue Int. 1999;65(3):183–187.
390. Pate, RR, Pratt, M, Blair, SN, et al. Physical activity and public health: a recommendation from the Centers for Disease Control and Prevention and the American College of Sports Medicine. JAMA. 1995;273(5):402–407.
391. Paterno, MV. Early rehabilitation following surgical fixation of a femoral shaft fracture. Phys Ther. 2006;86(4):558–572.
392. Paulozzi, LJ, et al. Recent trends in mortality from unintentional injury in the U.S. J Safety Res. 2006;37(3):277–283.
393. Pavlica, P, Barozzi, L, Salarani, C, et al. Magnetic resonance imaging in the diagnosis of PMR. Clin Exp Rheumatol. 2000;18(4 suppl 20):S38–S39.
394. Pawl, RP. Chronic neck syndromes: an update. Compr Ther. 1999;25(5):278–282.
395. Penninx, BW, Messier, SP, Rejeski, J, et al. Physical exercise and the prevention of disability in activities of daily living in older persons with osteoarthritis. Arch Intern Med. 2001;161(19):2309–2316.
396. Pépin, JL. Sleep apnoea syndrome secondary to rheumatoid arthritis. Thorax. 1995;50:692–694.
397. Peri, P. Stress fractures. Best Pract Res Clin Rheumatol. 2003;17(6):1043–1061.
398. Petrofsky, J, Lee, S, Bweir, S. Gait characteristics in people with type 2 diabetes mellitus. Eur J Appl Physiol. 2005;93(5-6):640–647.
399. Petrofsky, J, Lee, S, Macnider, M, et al. Autonomic, endothelial function and the analysis of gait in patients with type 1 and type 2 diabetes. Acta Diabetol. 2005;42(1):7–15.
400. Petty, DH. Glenohumeral chondrolysis after shoulder arthroscopy. Am J Sports Med. 2004;32:509–515.
401. Phelan, JD. Genomic progress in pediatric arthritis: recent work and future goals. Curr Opin Rheumatol. 2006;18(5):482–489.
402. Pill, SG. Managing and preventing overuse injuries in young athletes. J Musculoskelet Med. 2003;20(9):434–442.
403. Pinals, RS. Preliminary criteria for clinical remission in rheumatoid arthritis. Arthritis Rheum. 1981;24:1308–1315.
404. Podichetty, VK. Managing vertebral compression fractures in older patients. J Musculoskelet Med. 2004;21(7):372–378.
405. Pollintine, P. Neural arch load-bearing in old and degenerated spines. J Biomech. 2004;37:197–204.
406. Pongratz, DF. Soft tissue pain syndromes: clinical diagnosis and pathogenesis. Binghamton, NY: Haworth Press, 2006.
407. Pope, JE, Stevens, A, Howson, W, et al. The development of rheumatoid arthritis after recombinant hepatitis B vaccination. J Rheumatol. 1998;25:1687–1693.
408. Pope, RM. Rheumatoid arthritis: pathogenesis and early recognition. Am J Med. 1996;100(suppl 2A):3–9.
409. Praemer, A. Musculoskeletal conditions in the United States, ed 2. Rosemont, IL: American Academy of Orthopaedic Surgeons, 1999.
410. Prescott, K. Ankylosing spondylitis: the difficulty and importance of early diagnosis. J Musculoskelet Med. 2007;24(4):163–173.
410a. Price, CT. The lateral pillar classification for Legg-Calvé-Perthes disease. J Pediatr Orthop. 2007;27(5):592–593.
411. Puett, DW, Griffen, MR. Published trials of non-medicinal and non-invasive therapies for hip and knee osteoarthritis. Ann Intern Med. 1994;121:133–140.
412. Racette, SB. Creatine supplementation and athletic performance. J Orthop Sports Phys Ther. 2003;33(10):615–621.
413. Rang, M. Management of Legg-Calvé-Perthes disease varies with severity. J Musculoskelet Med. 1996;13(4):10–11.
414. Rathjen, KE. Stainless steel flexible intramedullary fixation of unstable shaft fractures. J Pediatr Orthop. 2007;27(4):432–441.
415. Ravelli, A. Juvenile idiopathic arthritis. Lancet. 2007;369(9563):767–778.
416. Reddy, R. Avascular necrosis and protease inhibitors. J Natl Med Assoc. 2005;97(11):1543–1546.
416a. Reichenbach, S. Meta-analysis: chondroitin for osteoarthritis of the knee or hip. Ann Intern Med. 2007;146(8):580–590.
417. Reginster, JY. Current role of glucosamine in the treatment of osteoarthritis. Rheumatology (Oxford). 2007;46(5):731–735.
418. Reginster, JY, Deroisy, R, Rovati, LC, et al. Long-term effects of glucosamine sulfate on osteoarthritis progression: a randomized, placebo-controlled clinical trial. Lancet. 2001;357(9252):247–248.
419. Reginster, JY, Gillot, VV, Bruyere, O, et al. Evidence of nutraceutical effectiveness in the treatment of osteoarthritis. Curr Rheumatol Rep. 2000;2(6):472–477.
420. Reiter, MF, Boden, SD. Inflammatory disorders of the cervical spine. Spine. 1998;23(24):2755–2766.
421. Robbins, L. Clinical care in the rheumatic diseases. Atlanta: American College of Rheumatology, 2001.
422. Robin, DW, Hasan, SS, Edeki, T, et al. Increased baseline sway contributes to increased losses of balance in older people following triazolam. J Am Geriatr Soc. 1996;44(3):300–304.
423. Robinson, CP, Yamachika, S, Bounous, DI, et al. A novel NOD-derived murine model of primary Sjögren’s syndrome. Arthritis Rheum. 1998;41:150–156.
424. Robinson, E. Weakness after critical illness-just deconditioning? Or something more? Acute Care Perspect. 2006;15(3):7–9.
425. Roos, EM. Joint injury causes knee osteoarthritis in young adults. Curr Opin Rheumatol. 2005;17(2):195–200.
426. Roos, EM. Positive effects of moderate exercise on glycosaminoglycan content in knee cartilage: a four-month, randomized, controlled trial in patients at risk of osteoarthritis. Arthritis Rheum. 2005;52(11):3507–3514.
427. Rosenthal, MD. Diagnosis of medial knee pain: atypical stress fracture about the knee joint. J Orthop Sports Phys Ther. 2006;36(7):526–534.
428. Russek, LN. Examination and treatment of a patient with hypermobility syndrome. Phys Ther. 2000;80(4):386–398.
429. Safran, MR. Warm-up and muscular injury prevention. Am J Sports Med. 1989;16:123–129.
430. Salter, RB. Textbook of disorders and injuries of the musculoskeletal system, ed 3. Baltimore: Williams & Wilkins, 1999.
431. Saltzman, CL, Vogelgesang, S. Clinical evaluation of the rheumatoid foot. J Musculoskelet Med. 1997;14(8):27–42.
432. Salvarani, C, Cantini, F, Olivieri, I. Distal musculoskeletal manifestations in polymyalgia rheumatica. Clin Exp Rheumatol. 2000;18(4 suppl 20):S51–S52.
433. Salvarani, C, Gabriel, SE, O’Fallon, WM, et al. Epidemiology of polymyalgia rheumatic in Olmsted County, Minnesota, 1970-1991. Arthritis Rheum. 1995;38:369–373.
434. Sambrook, PN. Genetic influences on cervical and lumbar disc degeneration: a magnetic resonance imaging study in twins. Arthritis Rheum. 1999;42:366–372.
435. Sanchez-Sotelo, J, Munuera, L, Madero, R. Treatment of fractures of the distal radius with a remodelable bone cement: a prospective, randomized study using Norian SRS. J Bone Joint Surg Br. 2000;82(6):856–863.
436. Santos, H, Brophy, S, Calin, A. Exercise in ankylosing spondylitis: how much is optimum? J Rheumatol. 1998;25:2156–2160.
437. Santos, H, Brophy, S, Calin, A. Exercise in ankylosing spondylitis: how much is optimum? J Rheumatol. 1998;25(11):2156–2160.
438. Sashika, H, Matsuba, Y, Watanabe, Y. Home program of physical therapy: effect on disabilities of patients with total hip arthroplasty. Arch Phys Med Rehabil. 1996;77(3):273–277.
439. Sattin, RW. Falls among older persons: a public health perspective. Annu Rev Public Health. 1992;13:489–508.
440. Saunders, SR. Physical therapy management of hand fractures. Phys Ther. 1989;69(12):1065–1076.
441. Sawka, AM. Do hip protectors decrease the risk of hip fracture in institutional and community-dwelling elderly? A systematic review and meta-analysis of randomized controlled trials. Osteoporos Int. 2005;16(12):1461–1474.
442. Sawka, AM. Hip protectors decrease hip fracture risk in elderly nursing home residents: a Bayesian meta-analysis. J Clin Epidemiol. 2007;60(4):336–344.
443. Scher, JU. Managing osteoarthritis: NSAIDs past, present, and future. J Musculoskelet Med. 2006;23(11):779–787.
444. Schnitzer T Advances in osteoarthritis research: investigating subchondral bone as etiologic agent and therapeutic target. Medscape, 2004. Available on-line at www.medscape.com/viewprogram/2892_pnt Accessed July 17, 2007
445. Schreiber, DH. Statin-induced rhabdomyolysis. J Emerg Med. 2006;31(2):177–180.
446. Scribner, AN, Troia-Cancio, PV, Cox, BA, et al. Osteonecrosis in HIV: a case-control study. J Acquir Immune Defic Syndr Hum Retrovirol. 2000;25(1):19–25.
447. Seeger, JD. Achilles tendon rupture and its association with fluoroquinolone antibiotics and other potential risk factors in a managed care population. Pharmacoepidemiol Drug Saf. 2006;15(11):784–792.
448. Serhal, D, Calabrese, LH. Diagnosing and managing rheumatic disorders in HIV-infected persons. J Musculoskelet Med. 2000;17(10):606–620.
449. Shakoor, N. Osteoarthritis: 20 clinical pearls. J Musculoskelet Med. 2003;20(4):173–182.
450. Shankar, K. Exercise prescription. Philadelphia: Hanley & Belfus, 1999.
451. Shankman, G. Fundamental orthopedic management for the physical therapist assistant. St Louis: Mosby, 1997.
452. Sharma, L. Quadriceps strength and osteoarthritis progression in malaligned and lax knees. Ann Intern Med. 2003;138(8):613–619.
453. Shen, M, Kim, Y. Osteoporotic vertebral compression fractures: a review of current surgical management techniques. Am J Orthop. 2007;36(5):241–248.
454. Sherrington, C, Lord, S. Home exercises to improve strength and walking velocity after hip fracture. Arch Phys Med Rehabil. 1997;78(2):208–212.
455. Sherrington, C, Lord, S. Increased prevalence of fall risk factors in older people following hip fracture. Gerontology. 1998;44(6):340–344.
456. Shih, CH, Du, YK, Lin, YH, et al. Muscular recovery around the hip joint after total hip arthroplasty. Clin Orthop. 1994;302:115–120.
457. Shumway-Cook, A. Incidence of and risk factors for falls following hip fracture in community-dwelling older adults. Phys Ther. 2005;85(7):648–655.
458. Shumway-Cook, A, Gruber, W, Baldwin, M, et al. The effect of multidimensional exercises on balance, mobility, and fall risk in community-dwelling older adults. Phys Ther. 1997;77(1):46–57.
459. Siegel, LB, Gall, EP. The systematic approach to the physical examination in RA: I. J Musculoskelet Med. 1999;16(6):329–335.
460. Siegel, LB, Gall, EP. The systematic approach to the physical examination in RA: II. J Musculoskelet Med. 1999;16(7):392–400.
461. Sieper, J. Ankylosing spondylitis: an overview. Ann Rheumatol Dis. 2002;61(suppl 3):iii8–iii18.
462. Simons, DG, Travell, JG, Simons, LS. ed 2. Myofascial pain and dysfunction. Baltimore: Williams & Wilkins; 1999;1.
463. Singh-Grewal, D. Pilot study of fitness training and exercise testing in polyarticular childhood arthritis. Arthritis Rheum. 2006;55(3):364–372.
464. Skopouli, FN, Kassan, SS, Moutsopoulos, HM. Sjögren’s syndrome: causes, detection, and management. J Musculoskelet Med. 2000;17(1):33–42.
465. Skovgaard, D, Feldt-Rasmussen, B, Nimb, L, et al. Bilateral Achilles tendon rupture in kidney-transplanted individuals. Dan Med Bull. 1997;159:57–58.
466. Slemenda, C. Quadriceps weakness and osteoarthritis of the knee. Ann Intern Med. 1997;27:97–104.
467. Slemenda, C. Reduced quadriceps strength relative to body weight: a risk factor for knee osteoarthritis in women? Arthritis Rheum. 1998;41:1951–1959.
468. Soubrier, M. Polymyalgia rheumatica: diagnosis and treatment. Joint Bone Spine. 2006;73(6):599–605.
469. Spackey, J. Diagnosis: gout. Orthopedics. 2007;30(5):405–408.
470. Spector, TD, Roman, E, Silman, AJ. The pill, parity and rheumatoid arthritis. Arthritis Rheum. 1990;33:782–789.
471. Spirduso, W. Physical dimensions of aging. Champaign, IL: Human Kinetics, 1995.
472. Spondylitis Association of America. Facts about ankylosing spondylitis. The Association: Sherman Oaks, CA, 2007:777–8189. (800) Available on-line at www.spondylitis.org Accessed July 19, 2007.
473. Srbely, JZ. Randomized controlled study of the antinociceptive effect of ultrasound on trigger point sensitivity: novel applications in myofascial therapy? Clin Rehabil. 2007;21(5):411–417.
474. Stahl, CE. No pain, no gain-exercise-induced rhabdomyolysis associated with the performance herbal supplement ephedra. Med Sci Monit. 2006;12(9):CS81–CS84.
475. Steinberg, ME, Larcom, PG, Strafford, B, et al. Core decompression with bone grafting for osteonecrosis of the femoral head. Clin Orthop. 2001;386:71–78.
476. Stevens, DB, Tao, SS, Glueck, CJ. Recurrent Legg-Calvé-Perthes disease: case report and long-term followup. Clin Orthop. 2001;385:124–129.
477. Stevens, JA. Fatalities and injuries from falls among older adults-United States. MMWR Morbid Mortal Wkly Rep. 2005;55(45):1221–1224.
478. Stitik, TP. Nonpharmacologic management of knee and hip osteoarthritis. J Musculoskelet Med. 2005;22(2):61–70.
479. Strömbeck, B. The role of exercise in rehabilitation of patients with systemic lupus erythematosus and patients with primary Sjögren’s. Curr Opin Rheumatol. 2007;19(2):197–203.
480. Subbaro, JV. Heterotopic ossification: diagnosis and management, current concepts and controversies. J Spinal Cord Med. 1999;22:273–283.
481. Sullivan, DH, Wall, PT, Bariola, JR, et al. Progressive resistance muscle strength training of hospitalized frail elderly. Am J Phys Med Rehabil. 2001;80:503–509.
482. Sullivan, KE. The role of bone marrow transplantation in pediatric rheumatic diseases. J Rheumatol. 2000;58:49–52.
483. Summers, B. Low back pain on passive straight leg raising: the anterior theca as a source of pain. Spine. 2005;30(3):L342–L345.
484. Suomi, R. Effects of arthritis exercise programs on functional fitness and perceived activities of daily living measures in older adults with arthritis. Arch Phys Med Rehabil. 2003;84:1589–1594.
485. Symmons, DPM, Jones, MA, Scott, DL, et al. Long-term mortality outcome in patients with rheumatoid arthritis: early presenters continue to do well. J Rheumatol. 1998;25:1072–2077.
486. Takikawa, S, Matsui, N, Kokubu, T, et al. Low-intensity pulsed ultrasound initiates bone healing in rat nonunion fracture model. J Ultrasound Med. 2001;20(3):197–205.
487. Takken, T. Aquatic fitness training for children with juvenile idiopathic arthritis. Rheumatology (Oxford). 2003;42(11):1408–1414.
488. Takken, T. Physical activity and health related physical fitness in children with juvenile idiopathic arthritis. Ann Rheum Dis. 2003;62(9):885–889.
489. Tan, V, Dormans, JP. A persistent lesion following forearm fracture. J Musculoskelet Med. 1999;16(7):429–430.
490. Tanaka, J. Outcomes of hip fracture surgery in patients aged > 90 years. Orthopedics. 2003;26(1):55–58.
491. Tanaka, M. A pathologic study of discs in the elderly: separation between cartilaginous endplate and the vertebral body. Spine. 1993;18:1456–1462.
492. Tecklin, JS. Pediatric physical therapy, ed 3. Philadelphia: Lippincott Williams & Wilkins, 1999.
493. Terrono, AL, Horner, G. Carpometacarpal pain: is it osteoarthritis? J Musculoskelet Med. 2000;17:744–753.
494. Thelander, U. Straight leg raising test versus radiologic size, shape, and position of lumbar disc hernias. Spine. 1992;17:395–399.
495. Theodosakis, J. The arthritis cure. New York: St Martin’s Press, 2004.
496. Theodosakis, J, Adderly, B, Fox, B, et al. Maximizing the arthritis cure: a step by step program to faster stronger healing during any stage of the cure. New York: St Martin’s Press, 1999.
497. Thigpen, MT, Light, KE, Creel, GL, et al. Turning difficulty characteristics of adults aged 65 years or older. Phys Ther. 2000;80(12):1174–1187.
498. Thomas, E. National study of cause-specific mortality in rheumatoid arthritis, juvenile chronic arthritis, and other rheumatic conditions: a 20 year followup study. J Rheumatol. 2003;30:958–965.
499. Tinetti, ME. Clinical practice: preventing falls in elderly persons. N Engl J Med. 2003;348(1):42–49.
500. Tinetti, ME. Instability and falling in elderly patients. Semin Neurol. 1989;9:39–45.
501. Tinetti, ME. Risk factor abatement strategy for fall prevention. J Am Geriatr Soc. 1993;41:315–320.
502. Tinetti, ME, et al. Falls efficacy as a measure of fear of falling. J Gerontol. 1990;45:239–243.
503. Tinetti, ME, et al. Prevention of falls among the elderly. N Engl J Med. 1989;320:1055–1059.
504. Tinetti, ME, Baker, D, McAvay, G. A multifactorial intervention to reduce the risk of falling among elderly people living in the community. N Engl J Med. 1994;331(13):821–827.
505. Tinetti, ME, Liu, WL, Ginter, SF. Mechanical restraint use and fall-related injuries among residents of skilled nursing facilities. Ann Intern Med. 1992;116:369–374.
506. Tinetti, ME, Richman, D, Powell, L. Falls efficacy as a measure of fear of falling. J Gerontol Psychol Sci. 1990;45(6):P239–P243.
507. Tough, EA. Variability of criteria used to diagnose myofascial trigger point pain syndrome-evidence from a review of the literature. Clin J Pain. 2007;23(3):278–286.
508. Travell, J, Rinzler, SH. The myofascial genesis of pain. Postgrad Med. 1952;11:425–431.
509. Travell, JG, Simons, DG. Myofascial pain and dysfunction. Baltimore: Williams & Wilkins; 1992;2.
510. Tropiano, P. Lumbar total disc replacement. J Bone Joint Surg. 2005;87-A(3):490–497.
511. Uhrin, Z, Kuzis, S, Ward, MM. Exercise and changes in health status in patients with ankylosing spondylitis. Arch Intern Med. 2000;160(19):2969–2975.
512. Unwin, B. Polymyalgia rheumatic and giant cell arteritis. Am Fam Phys. 74(9), 2006. [1547-1154].
513. Urquhart, N, Stanish, WD. Drugs for knee osteoarthritis: what primary care physicians need to know. J Musculoskelet Med. 2007;24(4):178–187.
514. van Baar, ME, Assendelft, WJJ, Dekker, J, et al. Effectiveness of exercise therapy in patients with osteoarthritis of the hip or knee: a systematic review of randomized clinical trials. Arthritis Rheum. 1999;42:1361–1369.
515. Vanden Bossche, L. Heterotopic ossification: a review. J Rehabil Med. 2005;37(3):129–136.
516. van den Ende, CH, Breedveld, FC, le Cessie, S, et al. Effect of intensive exercise on patients with active rheumatoid arthritis: a randomized clinical trial. Ann Rheum Dis. 2000;59(8):615–621.
517. Van der Esch, M. Joint proprioception, muscle strength, and functional ability in patients with osteoarthritis of the knee. Arthritis Rheum. 2007;57(5):787–793.
518. van der Linden, PD. Increased risk of Achilles’ tendon rupture with quinolone antibacterial use especially in elderly patients taking oral corticosteroids. Arch Intern Med. 2003;163(15):1801–1807.
519. van der Linden, S. Physiotherapy in ankylosing spondylitis: what is the evidence? Clin Exper Rheum. 2002;6(suppl 28):S60–S64.
520. Van Dillen, LR. Symmetry of timing of hip and lumbopelvic rotation motion in two different subgroups of people with low back pain. Arch Phys Med Rehabil. 2007;88(3):351–360.
521. van Tulder, MW. Conservative treatment of acute and chronic nonspecific low back pain: a systematic review of randomized controlled trial of the most common interventions. Spine. 1997;22:2128–2156.
522. Velazquez V, Fisher NM, Venkatraman JT, et al The effect of a lower extremity resistance exercise rehabilitation program on TNF-alpha and TNF receptors in juvenile arthritis. Presentation at the Annual Scientific Meeting of the Association of Rheumatology Health Professionals and the American College of Rheumatology, Boston, November 16, 1999
523. Venning, G. Recent developments in vitamin D deficiency and muscle weakness among elderly people. BMJ. 2005;330(7490):524–526.
524. Vestergaard, P, Rejnmark, L, Mosekilde, L. Hip fracture prevention: cost-effective strategies. Pharmacoeconomics. 2001;19(5 pt 1):449–468.
525. Vestibular Autorotation Test (VAT). Western Systems Research: Pasadena, CA, 2001:578–7363. (626) Available on-line at www.4wsr.com Accessed May 30, 2008. (Reference to this tool does not imply endorsement.).
526. Videman, T. Magnetic resonance imaging findings and their relationships in the thoracic and lumbar spine: insights into the etiopathogenesis of spinal degeneration. Spine. 1995;20:928–935.
527. Videman, T, Levälahti, E, Battié, M.C. The effects of anthropometrics, lifting strength, and physical activities in disc degeneration. Spine. 2007;32(13):1406–1413.
528. Vinik, AI, Mehrabyan, A. Diabetic neuropathies. Med Clin North Am. 2004;88(4):947–999.
529. Vivino, FB, Sjögren’s syndrome: a disease in evolution. Medscape Rheum. 2004;5(2). Available on-line at www.medscape.com/viewarticle/493493 Accessed July 11, 2007.
530. Vlad, SC. Glucosamine for pain in osteoarthritis: why do trial results differ? Arthritis Rheum. 2007;56(7):2267–2277.
531. Vogt, TM, Ross, PD, Palermo, L, et al. Tool identifies women at risk for vertebral fracture. Mayo Clin Proc. 2000;75:888–896.
532. Wallace, CA. Current management of juvenile idiopathic arthritis. Best Pract Res Clin Rheumatol. 2006;20(2):279–300.
533. Wang, TJ. Effects of aquatic exercise on flexibility, strength, and aerobic fitness in adults with osteoarthritis of the hip or knee. J Adv Nurs. 2007;57(2):141–152.
534. Warden, SJ, Bennell, KL, McMeeken, JM, et al. Acceleration of fresh fracture repair using the sonic accelerated fracture healing system (SAFHS): a review. Calcif Tissue Int. 2000;66(2):157–163.
535. Warden, SJ, Favaloro, JM, Bennell, KL, et al. Low-intensity pulsed ultrasound stimulates a bone-forming response in UMR-106 cells. Biochem Biophys Res Commun. 2001;286(3):443–450.
536. Weinblatt, ME. A trial of etanercept, a recombinant tumor necrosis factor receptor: Fc fusion protein, in patients with rheumatoid arthritis receiving methotrexate. N Engl J Med. 1999;340(4):253–259.
537. Welsing Paco, MJ. Is the disease course of rheumatoid arthritis becoming milder? Time trends since 1985 in an inception cohort of early RA. Arthritis Rheum. 2005;52(9):2616–2624.
538. Wetzel, ET. The role of repeated end-range/pain response assessment in the management of symptomatic lumbar discs. Spine J. 2003;3(2):146–154.
539. Whipple, RH, Wolfson, LI, Amerman, P. The relationship of knee and ankle weakness to falls in nursing home residents: an isokinetic study. J Am Geriatr Soc. 1987;35:13–20.
540. White, JH. Imaging of sacral fractures. Clin Radiol. 2003;58(12):914–921.
541. Whitney, SL. Management of the elderly person with vestibular dysfunction. In Herdman SJ, ed.: Vestibular rehabilitation, ed 3, Philadelphia: FA Davis, 2007.
542. Will, R, Kennedy, G, Elswood, J, et al. Ankylosing spondylitis and the shoulder: commonly involved but infrequently disabling. J Rheumatol. 2000;27(1):177–182.
543. Wlodarski, KH. Bone histogenesis mediated by nonosteogenic cells. Clin Orthop. 1991;272:8–15.
544. Wolfe, F. Increase in cardiovascular and cerebrovascular disease prevalence in rheumatoid arthritis. J Rheumatol. 2003;30:36–40.
545. Yacyshyn, EA, Cohen, MD. Ankylosing spondylitis: early detection and management. J Musculoskelet Med. 1999;16(12):703–711.
546. Yang, YX. Long-term proton pump inhibitor therapy and risk of hip fracture. JAMA. 2006;296(24):2947–2953.
547. Yarbrough, R. Chondrolysis: an update. J Pediatr Orthop. 2005;25(5):702–704.
548. Yazici, Y. Bright future for RA therapies. J Musculoskelet Med. 2006:32–35. [Suppl].
549. Young, JWR. Fracture of the pelvis: current concepts of classification. AJR Am J Roentgenol. 1990;155:1169–1175.
550. Zeni, AI, Street, CC, Dempsey, RL, et al. Stress injury to the bone among women athletes. Phys Med Rehabil Clin North Am. 2000;11(4):929–947.