Soft Tissue, Joint, and Bone Disorders
People presenting with muscle, joint, and bone disorders make up a significant percentage of the therapist’s practice. These conditions are primarily manifested by pain, deformity, and loss of mobility and function. Many of the people seen by therapists have these conditions secondary to trauma or repetitive overuse; these conditions are generally self-contained or local in terms of involved tissues.
Therapists may treat dysfunction in other body regions to reduce the mechanical stresses on the involved joint, but the disorder itself (i.e., degenerative joint disease, bursitis, tendinitis) does not spread to other body regions. This is in contrast to rheumatic disease, a systemic disorder, which can be manifested not only by local joint or muscle pain and dysfunction but also by additional complaints associated with other body systems.
Although this book is primarily a compilation of diseases and conditions of all systems, this chapter contains both orthopedic and systemic conditions that affect the bones, joint, or muscles that may not fall into any other category. Because the focus of this text is not orthopedics, many orthopedic conditions have not been included. For the most part, those conditions with a more generalized effect or accompanied by a systemic component are included here. The concepts presented in Chapter 22 are especially important to the discussion of this chapter and should be reviewed or read along with this chapter.
This chapter is divided into three distinct anatomic areas (soft tissue, joint, bone) with conditions and diseases placed in the area most notably affected. Frequently there is overlap, and one condition affecting more than one area is found in a single section. As always, the reader is encouraged to keep a broad perspective whenever studying an isolated condition or anatomic area.
SOFT TISSUE
Soft tissue injuries, such as strains and sprains, lacerations, tendon ruptures, muscle injuries, myofascial compartment syndromes, dislocations, and subluxations, are described briefly in this section. For a more detailed description of these conditions, the reader is referred to the Guidelines for Exercise Testing and Prescription by the American College of Sports Medicine (ACSM).15
Strains refer to stretching or tearing of the musculotendinous unit; they may be partial or full tears. The musculotendinous junction is a region of highly folded basement membranes between the end of the muscle fiber and the tendon. These involutions maximize surface area for force transmission but contain a transition zone where the compliant muscle fibers become relatively noncompliant tendon, placing this junction at increased risk for injury.57 A helpful mnemonic device to recall strain versus sprain is that the t in strain can be matched to the t in musculotendinous. A sprain is an injury of the ligamentous structures around a joint caused by abnormal or excessive joint motion.
Strains and sprains can be classified as mild, moderate, or severe (complete) tears or as injuries of first, second, or third degree depending on the severity of tissue damage. Stretching or minor tearing of a few fibers without loss of integrity is classified as first degree (mild), with only minor swelling and discomfort accompanied by no or only minimal loss of strength and restriction of movement.227
Second-degree (moderate) strain refers to partial tearing of tissue with clear loss in function (ability to contract). Pain, moderate disability, point tenderness, swelling, localized hemorrhaging, and slightly to moderately abnormal motion are typical.
A third-degree (severe) strain or sprain refers to complete loss of structural or biomechanical integrity extending across the entire cross section of the muscle and usually requires surgical repair. An alternate classification scheme uses three grades of injury (I, II, III). Common sites for this type of injury include the ankle, knee, and fingers.
The tendon is most vulnerable to injury (tendinitis, tendon rupture) when it is tense or the attached muscle is maximally contracted or stressed and tension is applied quickly or obliquely. Tendinitis and spontaneous tendon ruptures have been reported to occur as a potential side effect of antibiotic treatment, especially with the use of fluoroquinolone antibiotics (e.g., drugs ending in “floxacin,” such as ofloxacin, norfloxacin, levofloxacin).
Reports suggest that fluoroquinolone-associated tendon disorders are more common in people over 60 years of age, especially those who are also taking oral corticosteroids.518 Tendon injuries occur at a higher rate in kidney transplant recipients, possibly caused by medications. In such cases, care should be taken to avoid overloading tendons, since dramatic ruptures following even small trauma have been reported.465
Injured muscle is also at increased risk for complete rupture if the muscle is subjected to high tensile force. The clinical manifestations of soft tissue injuries are local pain, edema, increased local tissue temperature, ecchymosis, hypermobility or instability, and loss of function. Muscle contusion (bruising with intact skin) is common in contact sports and incites an inflammatory response, sometimes involving hematoma formation.
Inflammatory reaction from injured soft tissue may lead to structural adaptation of tissue, scarring, weakness, and inflexibilities that can cause structural deficits or functional adaptations. If, after an injury, the therapist notes quick onset of joint effusion and the joint feels hot to the touch and movement is extremely painful and limited, the joint needs to be examined by a physician to rule out hemarthrosis.
Myofascial compartment syndromes develop when increased interstitial pressure within a closed myofascial compartment compromises the functions of the nerves, muscles, and vessels within the compartment. Compartment syndromes may be acute or chronic and are most likely to occur within the “envelopes” of the lower leg, forearm, thigh, and foot where the fascia cannot give or expand.
Many clinical conditions predispose to the development of compartment syndromes, including fractures, severe contusions, crush injuries, excessive skeletal traction, and reperfusion injuries and trauma. Other risk factors may include burns, circumferential wraps or restrictive dressings, or a cast or other unyielding immobilizer. Ischemia and irreversible muscle loss can occur, resulting in functional disability (and even potential loss of limb) if the condition is left untreated.
The earliest clinical symptom of impending compartment ischemia is pain out of proportion to that expected from the injury. The pain is described as deep, throbbing pressure. There may be sensory deficit or paresthesia within the region distal to the area of involvement. In severe compartment syndromes, objective signs are visible, such as a swollen extremity with smooth, shiny, or red skin. The extremity is tense on palpation, and passive stretch increases the pain.329 Prompt surgical decompression is the standard intervention.
Injury to the growth cartilage can occur in skeletally immature children and adolescents. The three areas of growing cartilage in a skeletally immature individual include the physis (growth plate), articular cartilage of joint surfaces, and major bone-tendon attachments (apophyses). These sites account for a large number of sports injuries in young athletes, including osteochondritis dissecans (articular surface) and Osgood-Schlatter disease (apophysis); both conditions are discussed later in this chapter.
The terms subluxation and dislocation relate to joint integrity. Subluxation is partial disruption of the anatomic relationship within a joint. Mobile joints are at risk of subluxation. These include the glenohumeral, acromioclavicular, sacroiliac, and atlantoaxial joints. Once the joint condition has stabilized, rehabilitation should address local muscle imbalances and adjacent joint hypomobility, which could increase mechanical stresses at the joint.
Dislocation implies complete loss of joint integrity with loss of anatomic relationships. Often significant ligamentous damage occurs with this type of injury. Dislocations most often occur at the glenohumeral joint. Congenital dislocations are most frequently seen at the hip joints (see the section on Developmental Dysplasia in Chapter 23).
Joint dislocation can also be a late manifestation of chronic disease, such as rheumatoid arthritis (RA), paralysis, and neuromuscular disease. In the presence of a joint dislocation, the integrity of nerve and vascular tissue must be assessed. If compromise is suspected, timely reduction is essential to prevent serious complications.
Heterotopic Ossification
Heterotopic ossification (HO) is defined as bone formation in nonosseous tissues (usually muscles and other soft tissue areas). It is considered a benign condition of abnormal bone formation in soft tissue that occurs most commonly after trauma such as fractures, surgical procedures (especially total hip replacements), spinal cord and traumatic brain injuries, burns, and amputations. HO is the most common complication of total hip arthroplasty.75 Classification of HO is based on the anatomic location and effect on functional motion (Box 27-1).
There is an increased incidence of HO among military personnel with blast injuries. The extreme force destroys bone, muscles, and tendons, resulting in amputation. Bone growth associated with HO in the residual limb does not follow a predictable pattern, and bone may grow into long spikes or develop more like cobwebs.
In addition to acquired forms of HO, there are forms due to hereditary causes such as fibrodysplasia ossificans progressiva, progressive osseous heteroplasia, and Albright’s hereditary osteodystrophy. These conditions are extremely rare but do provide helpful information on the pathophysiology of the condition.515
Heterotopic ossification and myositis ossificans (MO) are terms often used interchangeably. Both conditions represent the deposition of mature lamellar bone and share radiographic and histologic characteristics, but the locations in which they occur are different. HO develops in nonosseous tissues, while MO forms in bruised, damaged, or inflamed muscle.71
HO in people with spinal cord injuries is often referred to as neurogenic heterotopic ossification (NHO). NHO appears to be related more to the degree of completeness of spinal cord injury than the level involved; individuals with complete transverse spinal cord injuries are more likely to develop HO compared to those with incomplete spinal cord injuries.71
Risk Factors
Risk factors for HO include a serious traumatic injury, previous history of HO, hypertrophic osteoarthritis, ankylosing spondylitis (AS), and diffuse idiopathic skeletal hyperostosis (DISH). Men seem to be at higher risk for HO than women. Other risk factors may include Paget’s disease, RA, posttraumatic arthritis, neural axis and thermal injuries, and osteonecrosis.71,75
Surgery-related factors may contribute to the formation of HO. Individuals who have undergone multiple surgical interventions over a short period of time are at increased risk of HO. This may be attributed to the extensive damage to soft tissues, presence of disseminated bone dust, or formation of hematoma. Length of time in surgery has also been implicated.71
HO occurs in 1% to 3% of the burn population. It appears to be related more to the degree of thermal injury than to the location of the burn. Individuals with third-degree burns affecting more than 20% of the total body surface are at greatest risk for the development of HO. Systemic physiologic factors in conjunction with local factors are the likely underlying etiology.113,211
Etiology and Pathogenesis
The cause of HO remains unknown. Direct trauma is the most common cause of heterotopic bone formation in the elbow. It appears that there is a link between the severity of injury and the amount of ectopic bone formation that develops. Someone who sustains a massive traumatic injury is very likely to develop HO; HO is five times more likely if there is both fracture and dislocation of the elbow.71
It is most likely that pluripotent mesenchymal (stem) cells that could differentiate into cartilage, bone, or tendon/ligament become osteoblasts instead. Differentiation begins early after surgery and peaks at 32 hours, possibly induced by a bone-inducing substances such as bone morphogenetic protein (BMP). The stimulus and mechanism that causes this to happen in soft tissues after trauma has not been determined. There may be local factors such as mechanical stress (e.g., articular disruption, muscle damage) and/or systemic factors.297
Individuals with traumatic brain injury are predisposed to HO, most likely due to osteoinductive factors released at the site of the brain injury, although little is known about this process.140 In the case of bone fracture or reaming of the bone during joint replacements, bone marrow, which is capable of forming bone, may spread into well-vascularized muscle tissue. Bone marrow combined with growth factors from traumatized tissues may set off a series of steps leading to bone development and HO.29,75
Histologically, in the acute phase, the inflammatory process results in edema and degeneration of muscle tissue. After a few weeks, the inflamed tissue is replaced with cartilage and bone, and the bone undergoes intensive turnover. Histologically, this process cannot be distinguished from the formation of bone callus in fractures.480
There are histologic differences between normal bone and the ectopic (displaced) bone formed in HO. In normal bone, the periosteal layer covering the external surface of the bone has an inner vascular cambium layer surrounded by an outer fibrosus layer. In HO, the ectopic bone is not enveloped by periosteum. Instead there are three zones: the center is made up of dense cells and is surrounded by a layer of osteoid. The outermost layer consists of highly organized bone, although ectopic bone has twice the number of osteoclasts compared with normal bone and a higher number of osteoblasts as well.543
Clinical Manifestations
HO may be asymptomatic and without pain, but pain and loss of motion are the most common presenting symptoms, often within 2 weeks of the precipitating trauma, surgery, burn, or neurologic insult. Swelling, warmth, erythema, and tenderness mimic a low-grade infection or, in the case of surgery, the normal postoperative inflammation that is often present. The hallmark sign of HO is a progressive loss of joint motion at a time when posttraumatic inflammation should be resolving.
As the ectopic ossification advances, the acute symptoms described may subside, but motion continues to decrease, even with intervention such as dynamic and/or static progressive splinting. Over the next 3 to 6 months, the HO matures and the individual develops a rigid or abrupt end feel with pain at the end range of motion. Delayed nerve palsy is common when the elbow is affected.71
Areas of calcification and bone spurs may progress to ankylosis. Sites affected most often include the hip, elbow, knee, shoulder, and temporomandibular joints. The elbow is the most common site of HO in burn patients; of the 1% to 3% of burn patients affected, the elbow is involved more than 90% of the time.211 Typically, a bridge of ectopic bone forms across the posterolateral aspect of the elbow, possibly filling in the olecranon fossa.
Pressure from the bone formation can result in pressure ulcers and interfere with skin grafts. Loss of motion can have serious consequences for daily function, especially for those individuals who are already neurologically compromised.
Different classification schemes are used depending on the site affected. Most grade the condition based on a scale from 0 to 3 or 0 to 4. Grade 0 is no islands of bone visible on x-ray. The final grade is bony ankylosis, with progressive involvement between the lowest and highest grade (e.g., bone spurs, periarticular bone formation).
MEDICAL MANAGEMENT
Measures can be taken to prevent HO, such as radiation treatment and pharmaceuticals (e.g., nonsteroidal antiinflammatory drugs [NSAIDs], diphosphonates). Diphosphonates inhibit osteoid cells from calcifying, thus preventing HO. The effect lasts only as long as the drug is taken. Gastrointestinal disturbance and osteomalacia are adverse side effects of this treatment, making it less than optimal.
NSAIDs (indomethacin) are effective in reducing the frequency and magnitude of ectopic bone formation in some areas (e.g., hip). Used during the first 3 weeks postoperatively, indomethacin inhibits precursor (undifferentiated) cells from developing into osteoblasts.
Low-dose external beam radiation is another effective preventive measure. Fractionated radiation of the pluripotent mesenchymal cells has been shown to be effective in preventing HO from developing when delivered within 72 hours after surgery.288
It can be used alone or in combination with NSAIDs. Prevention is recommended for individuals at high risk of ectopic ossification, including those with neurologic injury, burns, past history of HO, and/or a previous history of other conditions previously mentioned.
The best prevention for HO is to avoid soft tissue trauma, especially among high-risk individuals undergoing surgery of any kind. Complete wound lavage and the removal of all bone debris and reamings may help prevent HO.75
DIAGNOSIS.
Radiographic evidence with mineralization may be observed 4 to 6 weeks after the trauma (sometimes as early as 2 weeks after the incident event). X-rays show both the location, extent, and maturity of pathologic bone. HO must be differentiated from metastatic calcification, most often associated with hypercalcemia, and from dystrophic calcifications in tumors. History and radiographic examination usually provide the tools needed to diagnose this condition. Ultrasound may prove useful in diagnosing HO around the hip or elbow.
A computed tomographic (CT) scan may be best to show the exact location and involvement of the articular surfaces. Laboratory tests to measure the level of serum alkaline phosphatase are used by some, but they are not consistently accurate.
TREATMENT AND PROGNOSIS.
Radiation applied to the damaged limb site within a few days after the injury may respond but there is always the risk of impaired healing for those with bone fractures. Surgical resection is delayed until the bone matures and develops a distinct fibrous capsule in order to minimize trauma to the tissues and reduce the risk of recurrence and may only be done in cases where activities of daily living (ADLs) are compromised by loss of motion.211
Indication for surgery may not be just the presence of HO but rather the severity of functional restriction when loss of motion prevents the individual from using the affected extremity. A comprehensive rehabilitation program is needed to maximize motion, restore function, and reduce the risk of developing ankylosis. Once surgical removal is done, radiation and NSAIDs are continued to prevent recurrence.
Connective Tissue Disease
Sometimes people have features of more than one rheumatic disease. This has been called the overlap syndrome or mixed connective tissue disease (MCTD). This category includes people who have overlapping features of systemic lupus erythematosus (SLE), scleroderma, or polymyositis. The incidence of this disease is unknown, but adults, particularly women, are predominantly affected.
Initially MCTD was considered a distinct entity defined by a specific autoantibody to ribonuclear protein (RNP). In the late 1980s this concept of MCTD was considered flawed, since with time, in many of the affected people, the manifestations evolve to one predominant disease, and since many people with autoantibodies to RNP have clearcut SLE. Therefore the designation overlap connective tissue disease (OCTD) became the preferred name for the disorder in people having features of different rheumatic diseases.
There is also a condition called undifferentiated connective tissue syndrome in which the systemic rheumatic diseases present have several properties shared to a variable extent by RA, SLE, polymyositis, dermatomyositis, and Sjögren’s syndrome, which makes a specific diagnosis for a recognizable connective tissue disease difficult.5
More advanced technology has brought about immunogenetic and serologic studies that demonstrate once again that MCTD is quite distinctive from other disorders, especially SLE and systemic sclerosis. There is now good evidence that the clinical and serologic features of MCTD are not just a haphazard association but represent a distinctive subset of connective tissue disease in which specific autoimmune response is relevant to clinical expression and to understanding the underlying pathogenesis.299
Etiologic and Risk Factors and Pathogenesis
The cause of connective tissue disease is unknown, but hypotheses implicating modified self-antigens or infectious agents in the pathogenesis of MCTD have been advanced.199 Persons with this condition often have hypergammaglobulinemia and test positive for rheumatoid factor, suggesting an immune injury.
There is also a high titer of antibody to RNP (anti-RNP), but as previously mentioned this feature is also present in SLE. The cause for the formation and maintenance of the high titer of anti-RNP antibody is unclear. There is no direct evidence that these antibodies induce the characteristic involvement of the various organ systems.
There has been considerable controversy over the possible connection between silicone breast implants (and other silicone-containing devices, such as shunts and catheters) and the risk of connective tissue diseases. To date, there has been no convincing evidence of an association between breast implants in general, or silicone gel–filled breast implants specifically, and any of the individual connective tissue diseases or other autoimmune or rheumatic conditions.
From a health perspective, breast implants appear to have a minimal effect on the number of women in whom connective tissue diseases develop; the elimination of implants would not be likely to reduce the incidence of connective tissue diseases.225,268 See also the section on Infections with Prostheses and Implants in Chapter 25.
Likewise, efforts to prove an association between organic solvents and connective tissue disease have not been consistently replicated; which solvents convey risk remains unknown.137
Clinical Manifestations
OCTD/MCTD combines features of SLE (rash, Raynaud’s phenomenon, arthritis, arthralgias), scleroderma (swollen hands, esophageal hypomotility, pulmonary interstitial disease), polymyositis (inflammatory myositis), and, in most people, polyarthralgias. Seventy-five percent have RA. Proximal muscle weakness with or without tenderness is common.
Pulmonary, cardiac, and renal involvement, as well as such findings as Sjögren’s syndrome, Hashimoto’s thyroiditis, fever, lymphadenopathy, splenomegaly, hepatomegaly, intestinal involvement, and persistent hoarseness, may occur. Neurologic abnormalities, including organic mental syndrome, aseptic meningitis, seizures, multiple peripheral neuropathies, and cerebral infarction or hemorrhage, occur in about 10% of people affected by this disorder. A trigeminal sensory neuropathy appears to be seen much more frequently in MCTD/OCTD than in other rheumatic diseases.
MEDICAL MANAGEMENT
DIAGNOSIS, TREATMENT, AND PROGNOSIS.
The diagnosis is considered when additional overlapping features are present in persons appearing to have SLE, scleroderma, polymyositis, RA, juvenile idiopathic arthritis, Sjögren’s syndrome, vasculitis, idiopathic thrombocytopenic purpura, or lymphoma. High titers of serum antibodies to U1-RNP are a characteristic serologic finding seen much more often with OCTD/MCTD than with any other rheumatic disease.
General medical management and drug therapy are similar to the approach used in SLE. Most persons are responsive to immunosuppression with corticosteroids, especially if administered early in the course of the disease. Mild disease often is controlled by salicylates, other NSAIDs, antimalarials, or very low doses of corticosteroids. High doses of steroids may be used in combination with cytotoxic drugs when the disease is progressive and widespread.
The overall mortality has been reported as 13%, with the mean disease duration varying from 6 to 12 years. Individuals who respond well to steroid therapy have a good prognosis. Pulmonary and cardiac complications (e.g., pulmonary hypertension) are the most common cause of death in MCTD.178 Sustained remissions for several years in some people receiving little or no maintenance corticosteroid therapy have been observed.
Polymyalgia Rheumatica
Polymyalgia rheumatica (PMR, literally “pain in many muscles”) is a disorder marked by diffuse pain and stiffness that primarily affects the shoulder and pelvic girdle musculature. This condition is significant in that diagnosis is difficult and often delayed; severe disability can occur unless proper intervention is initiated. PMR may be the first manifestation of a condition called giant cell arteritis, an endocrine disorder, malignancy, or an infection.468
The initial symptoms associated with PMR are often subtle and of gradual onset, resulting in a delay in the person’s seeking care. The complaints also may be localized to one shoulder, leading to an initial diagnosis of bursitis. As the disease progresses, carrying out ADLs becomes increasingly difficult. Bed mobility and sit-to-stand transfers are among the functional activities affected.
Finally, a significant number (15% to 20%) of those with PMR also develop giant cell arteritis, a condition characterized by inflammation in the arteries of the head and neck (see further discussion in Chapter 12). The risk related to the arteritis is blindness secondary to obstruction of the ciliary and ophthalmic arteries from inflammation-associated swelling.
Incidence and Risk Factors
Female gender, age, and race are the three primary risk factors associated with PMR. Women are affected twice as often as men, and the disease is rare before the age of 50 years; most cases occur after age 70 years. White women are more commonly affected than are women of other ethnicity. PMR is a relatively common condition, with incidence estimated at 1 in 200 (one half as common as RA).433
Etiologic Factors and Pathogenesis
The cause of PMR is unknown, but experts suspect that genetic factors, infection, or an autoimmune malfunction may play a role. There is a genetic predisposition for PMR; human leukocyte antigen (HLA) DR4 (see Table 40-20) has been identified. Besides associations with HLA, tumor necrosis factor (TNF) appears to influence susceptibility to both PMR and giant cell arteritis. Additional studies are under way to clarify the genetic influence on susceptibility to these conditions.155
Despite complaints of pain and stiffness in the muscles, PMR is not associated with any histologic abnormalities. Serum creatinine kinase levels, electromyograms, and muscle biopsy results are negative in this population. Rather, the aching and stiffness typical of this condition are caused by joint inflammation.
More specifically, magnetic resonance imaging (MRI) studies have shown that subacromial and subdeltoid bursitis of the shoulders, iliopectineal bursitis, and hip synovitis are the predominant and most frequently observed lesions in active PMR. The inflammation of the bursae associated with glenohumeral synovitis, bicipital tenosynovitis, and hip synovitis may explain the diffuse discomfort and morning stiffness.393
Clinical Manifestations
PMR may begin gradually, taking days or weeks for symptoms to become fully evident, but more often it develops suddenly, and the person wakes up one morning feeling stiff and sore for no apparent reason. Getting out of bed in the morning can be the biggest challenge for individuals with PMR before initiating drug therapy.
Even though the initial muscle pain and stiffness may occur unilaterally, the symptoms are often bilateral and symmetric, affecting the neck, sternoclavicular joints, shoulders, hips, low back, and buttocks. Painful stiffness lasts more than 1 hour in the morning upon arising and is a hallmark feature of this disorder. Flulike symptoms such as fever, malaise, and weight loss are not uncommon.
Peripheral manifestations (e.g., wrists or metacarpophalangeal joints) are present in about one half of all cases of PMR and include joint synovitis, diffuse swelling of the distal extremities with or without pitting edema, tenosynovitis, and carpal tunnel syndrome.432 Many people are misdiagnosed with fibromyalgia, myositis, tendonitis, thyroid problems, or depression and spend months searching for answers and help before the correct diagnosis is made.
Despite the complaints of difficulties with bed mobility, sit-to-stand maneuvers, and accomplishing ADLs such as combing the hair or brushing the teeth, muscle weakness is not the problem. Pain and stiffness are the primary issues. Local tenderness of the involved muscles is noted with palpation. In addition, fever, malaise, unexplained weight loss, and depression may occur.
For those individuals with concomitant giant cell arteritis, additional symptoms of headache, jaw pain, scalp tenderness, fever, fatigue, weight loss, anemia, or blurred or double vision can occur.
MEDICAL MANAGEMENT
Since there are no definitive tests to identify PMR, the diagnosis is often based on the presence of a constellation of findings and the person’s rapid response to a trial of prednisone. Besides the complaints noted under Clinical Manifestations, the person may be anemic and present with an elevated erythrocyte sedimentation rate (ESR; measure of viscosity); lowered hemoglobin and elevated platelet count (indicators of inflammation); and elevated C-reactive protein (indicator of current disease activity).
The current diagnostic criteria include as a requirement an ESR higher than 30 or 40 mm/hr. However, several reports have indicated that a large number of people with PMR (7% to 22%) have a normal or slightly increased ESR at the time of diagnosis, supporting the notion that an increased ESR should not be an absolute requirement for the diagnosis of PMR. This subset is characterized by younger age, less marked predominance of females, lower frequency of constitutional symptoms (e.g., weight loss, fever), and a longer diagnostic delay.311
The lack of rheumatoid factor, the presence of ANAs, and the lack of histologic changes in the muscles contribute to the diagnosis by excluding other conditions. MRI or ultrasonography of the joint or joints may facilitate diagnosis in anyone with typical proximal symptoms of PMR who also has normal ESR values.67
TREATMENT AND PROGNOSIS.
Untreated, PMR can result in significant disability. It is imperative that the individual be checked for giant cell arteritis, a frequently concurrent condition that can cause irreversible blindness.512
Treatment is with corticosteroids (e.g., prednisone); the response is dramatic. In fact, if dramatic improvement is not noted within 1 week of starting the prednisone, the diagnosis of PMR is questioned and the person must be reevaluated.
Most people require a maintenance dosage of prednisone for 6 months to 2 years that is gradually tapered to the lowest effective dose required to control symptoms. Treatment may take up to 5 years or longer before complete clinical remission occurs.512 Methotrexate may be used for individuals who develop a dependency on corticosteroids.468
PMR is not life-threatening but it can limit daily activities, decrease restful sleep with nighttime awakenings and difficulty turning in bed, and decrease a sense of well-being and quality of life. With proper treatment, the prognosis is good, as the disease is self-limiting in many people with resolution within a period of 1½ to 2 years; however, recurrence can be as high as 30% in people who received treatment for 1 to 2 years. Those individuals with temporal arteritis are at increased risk for stroke or blindness.
Rhabdomyolysis
Rhabdomyolysis is the rapid breakdown of skeletal muscle tissue due to mechanical, physical, or chemical traumatic injury (Box 27-2). The principal result is a large release of the creatine phosphokinase (CPK) enzymes and other cell by-products into the blood system. Accumulation of muscle breakdown products can lead to acute renal failure.
Etiology and Risk Factors
Of particular note is the potential for muscle pain from statins (cholesterol-lowering medications) and rhabdomyolysis from high-dose statins.96 Less than 5% of the adult population who take statins develop this problem. However, with more than 15 million Americans taking these drugs, the prevalence is on the rise.445
Underlying neuromuscular diseases may become clinically apparent during statin therapy and may predispose to myotoxicity.28,79 Rhabdomyolysis also has been reported in performance athletes taking herbal supplements containing ephedra; there are similar reports of rhabdomyolysis in individuals using weight-loss herbal supplements.307,474
Strenuous exercise, including marathon running, biking, and exercises such as push-ups, sit-ups, or pull-ups can result in damage to skeletal muscle cells, a process known as exertional rhabdomyolysis.85
Pathogenesis and Clinical Manifestations
The individual may report muscle pain (myalgia) and weakness ranging from mild to severe.
The exact mechanism for statin-induced myopathy remains unknown. There may be a drug influence on deoxyribonucleic acid (DNA), an enzyme deficiency, or autoimmune reaction triggered by the drug.79 The effect of the process is well known; specifically, when muscle proteins are released into the blood, one of these proteins (myoglobin) can precipitate in the kidneys and spill into the urine. The client may report a change in color of the urine, most often tea colored or the color of cola soft drinks.
The therapist is most likely to see this with military recruits or marathon runners who have been exercising in hot and humid weather, or who have taken analgesics, had a viral or bacterial infection, and/or have a preexisting condition.85 Acute excessive consumption of alcohol exacerbated by a hot environment and dehydration can also predispose individuals competing in athletic events to exercise-induced rhabdomyolysis.
Massive skeletal muscle necrosis can also occur, further complicating the situation with reduced plasma volumes leading to shock and reduced blood flow to the kidneys resulting in acute renal failure. As the injured muscle leaks potassium, hyperkalemia may cause fatal disruptions in heart rhythm.
MEDICAL MANAGEMENT
DIAGNOSIS, TREATMENT, AND PROGNOSIS.
The diagnosis is typically made by history and clinical presentation and confirmed by laboratory studies when an abnormal renal function and elevated CPK are observed. To distinguish the causes, a careful medication history is considered useful. Often the diagnosis is suspected when a urine dipstick test is positive for blood, but no cells are seen on microscopic analysis. This suggests myoglobinuria, and usually prompts a measurement of the serum CPK, which confirms the diagnosis.
Treatment is directed toward rehydration and correction of electrolyte imbalances by administering intravenous fluids, and in the case of renal failure, dialysis may be necessary. In most cases of rhabdomyolysis, especially in the case of exertional rhabdomyolysis, damage to skeletal muscle cells resolves without consequence. Clinically significant rhabdomyolysis is uncommon but, when present, can be life-threatening.445
Myopathy
Myopathy is a term used to describe nonspecific muscle weakness secondary to an identifiable disease or condition. The term myositis is also used to describe idiopathic inflammatory myopathies.
Many metabolic and hormonal diseases and autoimmune diseases can cause muscle weakness. Myopathies are usually classified as either hereditary or acquired (Box 27-3; see also Table 39-3) Myopathy associated with polymyositis or dermatomyositis is discussed in Chapter 10. Myopathy (myositis) associated with infectious causes is mentioned briefly in Chapter 25. Information about other sources of muscle pain not discussed in this chapter and their neurophysiologic bases is also available.331
Etiologic Factors and Pathogenesis
The idiopathic inflammatory myopathies are thought to be immune-mediated processes that are triggered by environmental factors in genetically susceptible individuals. The pathogenesis of acquired myopathies and their course are highly variable and depend on the underlying cause. For example, in thyrotoxicosis, the high metabolic rate reduces the muscle stores of nutrients, whereas in hypothyroidism, the entire metabolism, including the energy-generating metabolism of muscles, is slowed down. Myopathy associated with RA is caused by the rheumatic joint disease.
Expression of proinflammatory cytokines such as interleukin-1 (IL-1) on endothelial cells and expression of major histocompatibility complex (MHC) class I antigens on muscle fibers have been associated with muscle weakness in individuals with active and chronic disease.7
Diabetes is associated with myopathy of three origins: vascular, neurogenic, and metabolic. Diabetes affects the small blood vessels and is associated with chronic hypoperfusion of muscles with blood. Diabetes also affects the peripheral nerves and causes neurogenic muscle atrophy and weakness. The disturbances of carbohydrate and lipid metabolism caused by insulin deficiency or insulin resistance adversely affect muscle function.
Acquired myopathy can also occur as part of a paraneoplastic syndrome (see discussion in Chapter 9). Tumors may produce muscle weakness with or without inflammation. Human immunodeficiency virus (HIV)–associated myopathies are less common now with improved medical intervention but may still be encountered by the therapist.
A new disorder called critical illness myopathy (CIM) has also been introduced. Disorders associated with prolonged stays in intensive care units (ICUs; e.g., acute respiratory illness, septic inflammatory response syndrome, acute respiratory distress syndrome) often result in excessive and prolonged weakness. CIM is a nonnecrotizing myopathy accompanied by fiber atrophy, fatty degeneration of muscle fibers, and fibrosis. As improvements in medical technology and medical management of individuals with severe illness continue to improve, the incidence of CIM is expected to rise.424
Use of systemic corticosteroids combined with prolonged exposure to neuromuscular blocking (paralytic) agents during the treatment of various critical illnesses in the ICU may be the key risk factor for this type of acute myopathy. Septic inflammatory response syndrome may be another risk factor.424
Clinical Manifestations
Myopathy is characterized by progressive proximal muscle weakness with varying degrees of pain and tenderness. Distal involvement is possible but is more common in myositis. During the early stages of disease, the muscles may be acutely inflamed and painful to move and touch. Muscle weakness and easy fatigability eventually compromise aerobic capacity and affect the person’s ability to work, socialize, and complete ADLs.192 Other symptoms of systemic illness may be present, including fever, fatigue, morning stiffness, and anorexia.
MEDICAL MANAGEMENT
The management of myopathy is determined by the underlying cause. Muscle biopsy, electromyography (EMG), and laboratory findings (measurement of muscle enzymes) are essential to ensure diagnostic accuracy, especially in the case of idiopathy myopathy. EMG can allow differentiation between myopathy and neuropathy and can localize the site of the neuropathic condition. The typical laboratory profile reveals mild to marked elevations in muscle enzymes, including creatine kinase and aldolase.
Some imaging techniques such as MRI and magnetic resonance spectroscopy of muscles can assess changes in local inflammatory activity. Changes in protein and gene expression patterns in repeated biopsy specimens provide molecular information that may lead to a more precise disease classification scheme and improved treatment, but these are research tools at this time.296
TREATMENT AND PROGNOSIS.
Inflammatory myopathies may respond to pharmacologic treatment, especially corticosteroids but also immunosuppressives and antimalarial agents. Oral creatine supplements combined with exercise have proven effective for improving muscle function without adverse effects in adults with inflammatory myopathies.81
Effective therapy for noninflammatory myopathies remains lacking; antiinflammatory agents do not appear to be helpful in these cases. Presently there is no known pharmacologic treatment or prevention for CIM. Medical management to minimize the risks is suggested.424
Prognosis is variable, with some people responding well to medical therapy and rehabilitation and others continuing to decline. Long-standing disability is not uncommon despite aggressive immunosuppressive treatment; the reasons for the persisting disability remain unknown.7 Additionally, corticosteroid-related complications can have a significant impact.
Factors associated with poor survival include onset after age 45 years, delayed diagnosis and intervention, severe weakness and pharyngeal dysphagia, malignancy, myocardial involvement, and interstitial lung disease. CIM is reversible, but there is often considerable morbidity (e.g., persistent pain and weakness, HO with frozen joints).267 ICU-acquired myopathy prolongs hospitalization because of the need for extensive rehabilitation. Even with rehabilitation, many affected individuals remain heavily dependent upon others for personal care and ADLs.309
Myofascial Pain Syndrome
Myofascial pain syndrome (MPS) is an overuse or muscle stress syndrome marked by the presence of myofascial trigger points (TrP) within a taut band of muscle. These hyperirritable foci located in skeletal muscle or its fascial components were first described in 1952.508 On palpation of these points, a characteristic pattern of local and referred pain is provoked. Referred pain may be provoked at quite a distance from the points of local tenderness.
TrPs may be either active (those that cause pain at rest or with activity of the involved muscle) or latent (not painful but causing movement restriction and weakness of the involved muscle with increased muscle tension and muscle shortening present). A latent TrP may become active in the presence of an acute, sudden overload of the muscle or a more chronic strain. A satellite TrP may develop in the same or other muscles within the referred pain pattern of the primary TrP or in synergistic muscles.
TrPs are separate and distinct from the tender points associated with fibromyalgia (see description of fibromyalgia tender points in Chapter 7). The terms should not be used interchangeably, and the term fibromyalgia TrPs is a misnomer.
TrPs appear to be a peripheral muscle phenomenon, whereas the widespread pain of fibromyalgia syndrome is a combination of both peripheral and central nervous system factors (e.g., abnormal pain processing). Although it is possible to have both TrPs and tender points in the same person, usually the individual presents with a distinct clinical presentation of predominantly either MPS or fibromyalgia syndrome. The widespread tender points of fibromyalgia have a very different underlying physiology compared with the TrPs of MPS.
Etiologic and Risk Factors
The cause of myofascial pain dysfunction is thought to be related to a sudden overload or overstretching of a muscle, direct impact trauma, postural faults, psychologic stress, or chronic repetitive or sustained muscle activity.462 TrPs can be the source of pain in other conditions such as tension headache.
People involved in occupations or recreation marked by repetitive or sustained activities or postures are at increased risk of developing this condition. Structural abnormalities or postural or mechanical stress could also place a chronic strain on certain muscle groups. Structural abnormalities that can predispose an individual to TrPs include significant leg length discrepancy, small hemipelvis, short upper arms in relation to torso height, and a foot with a relatively long second metatarsal compared to the first metatarsal.462
Other predisposing factors that can trigger activation of TrPs include overwork fatigue, chronic infection, impaired sleep, psychologic stress, and nerve entrapment (i.e., radiculopathy).462,508 TrPs can be activated indirectly by other existing TrPs, visceral disease (e.g., myocardial infarction, peptic ulcer, renal colic, gallbladder disease), arthritic joints, joint dysfunctions, and emotional stress.462
Pathogenesis
Previously, the TrP concept was viewed as a syndrome of unknown etiopathogenesis, but this has been replaced by the notion that TrPs are the result of an established neuromuscular disease characterized by muscle tension and its sequelae.406
According to one highly respected researcher in the area of TrP research, myofascial TrPs are most likely triggered by the performance of unaccustomed eccentric muscle activity. Eccentric movement of a muscle requires it to contract while being lengthened at the same time (e.g., action of the quadriceps muscle walking downhill). A buildup of acetylcholine causes the muscle to remain tense and even activate nearby muscles in the referral zone to activate latent triggers, making them satellite myofascial TrPs.146
Electrophysiologic and histopathologic evidence now makes it clear that dysfunctional motor endplates of skeletal muscle fibers are at the heart of the pathophysiology that characterizes myofascial TrPs. Spontaneous EMG activity in the TrP is greater than EMG activity in a noninvolved area of the same muscle, with a specific electrical discharge characteristic of a TrP identified. It appears that the electrical signal originates from the motor endplate rather than from the muscle spindle.200
The myofascial TrP mechanism is also closely related to spinal cord integration. The pathogenesis of TrPs is probably related to an integrative mechanism in the spinal cord in response to sensitized sensory nerve fibers (nociceptors) associated with dysfunctional endplates. When the input from nociceptors in an original receptive field persists (pain from an active TrP), central sensitization in the spinal cord may develop and the receptive field corresponding to the original dorsal horn neuron may be expanded (referred pain). Through this mechanism, a new (satellite) TrP may develop in the referred zone of the original TrP.200
Muscles protecting themselves against perceived trauma from overuse or repetitive contraction do not go into a protective spasm as was once thought but rather shorten and “shut off” as a means of guarding and self-protection. Energy requirements are reduced through this mechanism, and the body compensates by finding other muscles to do the task. It is hypothesized that the nervous system forgets to “turn the muscle back on” as identified by surface EMG studies.183
It has been hypothesized that there are two different types of TrPs: central and attachment. Pathophysiologic differences between central TrPs located in the muscle belly or endplate region and those located in a region of the muscle attachment where the taut band attaches to the tendon or where the tendon attaches to the bone (attachment TrPs) have yet to be adequately explored. When the pathologic mechanisms for TrPs have been fully determined, more specific intervention can be developed as well.462
The pathophysiology of the taut band (a localized contracture within the muscle) is now much clearer as well, although much of this remains a hypothesis that requires further validation. Intracellular calcium in certain muscle fibers may be excessively released in response to trauma or abnormal stress. The abnormally increased calcium may cause uncontrolled shortening activity and increased metabolism.
The muscle fiber shortening also impairs local circulation, which causes a loss of oxygen and nutrient supply to the region. This completes a vicious cycle; thus an energy crisis occurs, and taut bands form. It appears that these taut bands are necessary precursors for the development of TrPs based on the fact that taut bands frequently exist in pain-free individuals.200
Clinical Manifestations
A taut, palpable myofascial band that is exquisitely tender on palpation with a characteristic and reproducible referred pain pattern (with sustained palpable pressure) is the hallmark of myofascial pain dysfunction. This clinical manifestation has also been described as a ropelike, nodular, or crepitant (crackling or grating) area within a muscle; there may be fibrotic tissue resembling a small pea present that exhibits a highly localized, exquisitely tender spot. Once present, the TrPs are self-sustained and self-perpetuating hyperirritable foci.
Mechanical stimulation of the TrP, either by dry needling or by manual palpation, frequently results in a local twitch response (a brief contraction of the palpable mass in response to a brisk rolling or snapping palpation of the band, a visible indication of an active TrP). The same stimulus also produces a jump sign characterized by vocalization or withdrawal (person jumps away from the examiner in response to pressure exerted on the TrP). These two signs are usually present, observable, and reproducible before effective intervention eliminates the TrP and the signs. Additionally, the examiner may palpate a distinct nodule in the center of the taut band that is tender, sometimes exquisitely tender.
Pain referral patterns associated with TrPs are documented by several authors.258,462,509 Besides the pain, myofascial pain dysfunction is manifested by a reduced range of motion of joints under the control of the involved muscle and muscle weakness. The affected individual may be aware of numbness or paresthesia rather than pain, but there are no neurologic abnormalities, and the hypesthesia does not follow a radicular distribution.394
Systemic signs and symptoms are absent (unlike in fibromyalgia syndrome with its multiple presentation of various systemic manifestations), although a mild autonomic nervous system response to pain may result in nausea, diaphoresis, or change in blood pressure when TrPs are palpated. Related proprioceptive disturbances caused by TrPs may also include imbalance, dizziness, tinnitus, and distorted weight perception of lifted objects.462
Disturbances of motor function caused by TrPs include spasm of other muscles, weakness of the involved muscle function, loss of coordination by the involved muscle, and decreased work tolerance of the involved muscle. The weakness and loss of work tolerance are often misinterpreted as an indication for strengthening exercise, but if this is attempted without inactivating the responsible TrPs, the exercise is likely to encourage and further ingrain muscle substitution and further deconditioning of the involved muscle.462
MEDICAL MANAGEMENT
Because myofascial pain dysfunction is more of a clinical entity, the diagnosis is first made by clinical examination. Several diagnostic tests can help substantiate objectively the presence of characteristic TrP phenomena, including surface EMG, needle EMG, and ultrasound.
There is limited consensus on the diagnostic criteria for TrPs associated with MPS. A literature review of criteria used to diagnose TrPs found no less than 19 different diagnostic criteria. The four most commonly applied criteria were a tender spot in a taut band of skeletal muscle, patient pain recognition, predicted pain referral pattern, and local twitch response.507
Simons et al462 state that the criteria have changed in response to clinical observation and evidence from diagnostic reliability studies. According to this source, criteria no longer considered essential for diagnosis are local twitch response (least reliable diagnostic test) and predicted pain pattern, which is considered nonspecific. Simons et al have made further changes to the diagnostic criteria by adding nodule in a taut band and painful limitation to motion.462 However, these modifications were made on the basis of clinical experience and have not been tested experimentally for reliability, sensitivity, and specificity.507
Clearly in the absence of diagnostic laboratory tests or molecular markers, there is a need to define and standardize the way in which diagnosis of TrPs and MPS is made. At the present time, there is not a single set of criteria that is universally accepted.507
TREATMENT.
Many techniques aimed at desensitizing TrPs have been employed, such as injections using dry needling, saline, or local anesthetics (performed by a physician or in some states by a qualified physical therapist); application of ice in the direction of prescribed patterns; laser; ultrasound; and sustained (ischemic) manual pressure to the TrP. High-power, pain-threshold, static ultrasound technique has been shown to resolve acute TrPs more rapidly than conventional methods of using ultrasound for this condition.301,473 All of these techniques should be accompanied by sustained stretch of the involved muscle to desensitize the band.462,509
Ischemic compression is described as one that applies a steady pressure using the thumbs or four fingers on one or both hands inward toward the center and then is slowly released. Pressure application varies and may start from a few pounds and increase up to 10 lb, lasting from 30 to 45 seconds. On release, the skin blanches and then shows reactive hyperemia. The person should breathe deeply and slowly as pressure is progressively increased.
Non–ozone depleting vapocoolant spray (a topical skin refrigerant) may be used with the stretching procedures to facilitate pain relief and return of function. It is hypothesized that elongation of the muscle to its full normal length is the underlying mechanism that relieves pain caused by myofascial TrPs. Muscle lengthening utilizing postisometric relaxation may also be a successful technique.283
Additional rehabilitation to restore muscle strength and proprioception is required (Box 27-4). The physical therapist may employ a variety of additional modalities in the treatment of TrPs, such as low-voltage electrical stimulation, ultrasound, moist heat, or laser. Some experts recommend nutritional counseling with supplementation of vitamins B1, B6, and B12; folic acid; and vitamin C.
PROGNOSIS.
Whereas fibromyalgia syndrome is a chronic, long-term neuroendocrine condition requiring years of management, MPS is a local and/or regional condition of the soft tissue structures that responds well to local intervention as described.
Active TrPs can revert spontaneously to a latent state under the right conditions (i.e., adequate rest, absence of perpetuating factors) (Box 27-5).462 Chronic myalgia may not improve until the underlying precipitating or perpetuating factors are removed or properly managed.145 And even with proper clearing of the TrPs, the muscle activation process might not return to normal until specific training is done to facilitate that process. Recovery of proper motor control strategies is not automatic but depends on restoring normal motor plans.184 Biofeedback may help with this process; more research is needed to identify specific ways to accomplish this.
JOINT
Overview
Chondrolysis is a process of progressive cartilage degeneration resulting in narrowing of the joint space and loss of motion. It is seen most often as a complication of slipped capital femoral epiphysis (SCFE) but can occur in association with infection, trauma, and prolonged immobilization for any reason. Trauma can also include orthopedic procedures such as arthroscopic meniscectomy, shoulder arthroscopy, anterior cruciate ligament reconstruction, and thermal capsulorrhaphy.76,173,278,400
The hip is the most likely location for chondrolysis to occur, but cases have been reported affecting the knee, shoulder, and ankle. Spontaneous chondrolysis without known risk factors occurs occasionally, most commonly in adolescent girls. In fact chondrolysis occurs five times more often in females than in males; adolescence is the most common period of onset.547
Etiology and Pathogenesis
The etiology is unknown; many theories have been proposed, including nutritional abnormalities, mechanical injury, ischemia, abnormal chondrocyte metabolism, ischemia, and abnormal intracapsular pressure. There may be some evidence to support an autoimmune mechanism responsible for the cartilage destruction.
Various studies have implicated IL-1, which has chondrolytic action by stimulating the release of inflammatory mediators, enhancing the breakdown of cartilage proteoglycans. Clearly, some disruption of the cartilage extracellular matrix occurs leading to chondrolysis, but the key to the process has not been discovered.
Clinical Manifestations
Regardless of the underlying cause of this condition, the affected individual presents with progressive joint stiffness with progressive loss of motion and pain. Chondrolysis of the hip causes anterior hip and/or groin pain accompanied by an antalgic gait. Soft tissue contracture can result in an apparent leg length discrepancy and pelvic obliquity with muscle atrophy. Painful ankylosis may develop in some individuals, while others experience an improvement in pain and range of motion.547
MEDICAL MANAGEMENT
DIAGNOSIS, TREATMENT, AND PROGNOSIS.
Imaging studies are used to make the diagnosis. Plain radiographs are the first choice, but in difficult cases, the definitive diagnosis may be made on the basis of scintigraphy and/or MRI.
Treatment is with nonsteroidal antiinflammatory medications to control synovial inflammation. Protected weight bearing and maintaining joint motion are important components of the treatment plan. Surgery may be indicated (e.g., capsulectomy, tendon release of the adductor and iliopsoas), but the best course of operative treatment is unknown.547
Osteoarthritis
Osteoarthritis (OA), or degenerative joint disease, is a slowly evolving articular disease that appears to originate in the cartilage and affects the underlying bone, soft tissues, and synovial fluid.
OA is divided into two classifications: primary and secondary. Primary OA is a disorder of unknown cause, and the cascade of joint degeneration events associated with it is thought to be related to a defect in the articular cartilage. Secondary OA has a known cause, which may be trauma, infection, hemarthrosis, osteonecrosis, or some other condition.
OA is present worldwide as a heterogeneous group of conditions that lead to slow, progressive degeneration of joint structures with defective integrity of articular cartilage in addition to related changes in the underlying bone at the joint margins. OA can lead to loss of mobility, chronic pain, deformity, and loss of function.
Incidence
OA is the single most common joint disease, with an estimated prevalence of 60% in men and 70% in women later in life after the age of 65 years, affecting an estimated 40 million people in the United States.
In fact, it is the most common musculoskeletal disorder worldwide affecting the hands and large weight-bearing joints such as the hip and knee and causing disability.308 And the overall prevalence is expected to increase dramatically over the next 20 years as the population ages.164
Before age 50 years, the prevalence of OA in most joints is higher in men than in women, but this changes after age 65 years. In the United States OA is second only to ischemic heart disease as a cause of work disability in men older than 50 years.106
In the United States, about 6% of adults over 30 years of age have OA of the knee and 3% have OA of the hip; incidence rises with increasing age. Both incidence and prevalence are expected to rise in the coming decades as a result of the aging of America combined with more extreme sports and activities (see the following section on Etiologic and Risk Factors). OA is the most common indication for total joint replacements.
Etiologic and Risk Factors
The etiology of OA is multifactorial, including many components of biomechanics and biochemistry. Evidence is growing for the role of systemic factors such as genetics, nutrition and weight control, estrogen use, bone density, and local biomechanical factors (e.g., muscle weakness, obesity, joint laxity).120
Serious injury and an inherited predisposition account for one half of all cases of OA in the hands and hips and knees.119,141,425 Smokers with knee OA sustain greater cartilage loss and have more severe knee pain than those who do not smoke, suggesting a role for tobacco in cartilage degeneration.19
There is low or no additional risk of OA from regular, moderate running, but sports that involve high-intensity, acute, direct joint impact from contact with other players do carry an increased risk of OA, especially when repetitive joint impact and twisting are combined. Football players, soccer players, hockey players, and baseball pitchers are especially at increased risk. Anterior cruciate ligament injury may predispose athletes to knee OA, especially when accompanied by meniscectomy.87,337
Although the theory is as yet unproven, some experts warn that extreme sports such as snow boarding, mountain biking, and aggressive in-line skating with the increased incidence of repeated impact or injury may be risk factors for OA developing earlier in life. Much of the OA in men is attributable to occupational activities, particularly kneeling or squatting, along with heavy lifting and repetitive use of heavy machinery.119,120
Generalized ligamentous laxity appears to be a predisposing factor; this may be related to the presence of estrogen receptors on the ligaments. Postmenopausal women appear to be at increased risk.493 Some women have a condition called hypermobility joint syndrome or hypermobility syndrome with loose, unstable joints resulting from a dominant inherited connective tissue disorder.
Hypermobility syndrome is characterized by excessive laxity of multiple joints, a condition that is separate from the generalized hypermobility associated with disorders such as Ehlers-Danlos syndrome, RA, SLE, or Marfan’s syndrome. Hypermobility syndrome appears to be a systemic collagen abnormality with a decreased ratio of type I to type III collagen (see Table 6-2).428
Women with this syndrome may develop OA earlier than the norm. Muscle weakness in anyone can also cause joint changes leading to OA, such as occurs with prolonged immobilization, polymyositis, multiple sclerosis, or any of the myopathies listed in Box 27-3.
There have been some studies that show a link between patellar alignment and patellofemoral OA manifested by a loss of cartilage thickness and knee pain and disability.214,234 Other studies suggest that malalignment is not a risk factor for OA but rather a marker of disease severity and its progression.213 Additional studies will be needed to establish the normal and abnormal ranges of patellar alignment indices and their relationship to patello-femoral OA.235
Pathogenesis
The pathophysiologic events associated with OA are beginning to be understood more definitively. It is quite clear now that OA is a disorder of the whole synovial joint organ, not just “wear and tear” on the cartilage. In fact, it may be that damage to the articular cartilage is the by-product of a disease process that is centered in subchondral bone in particular. Emphasis is now on the joint as a whole rather than just the cartilage.
In recent years, the view of OA has shifted to that of both a local and systemic condition in which inflammation plays an important part in determining the symptoms and disease progression.118,304 The former wear-and-tear concept already mentioned has been replaced by the idea that OA is an active disease process with joint tissue destruction and aberrant repair as a result of alterations in cellular function.
Although joint cartilage is the final target of the pathologic processes, the underlying subchondral bone may be the primary etiologic agent. Treatment focused on modifying changes in the bone may alter the pathologic processes observed in the adjacent cartilage. The use of new, potent bone antiresorptive agents in clinical use will help test this hypothesis.444
Tissue changes in OA are the result of active joint remodeling processes involving an imbalance between catabolic and anabolic repair activity. People with OA may have a general tendency toward increased bone metabolic activity, especially in response to biomechanical or other stimuli such as occurs with obesity and injury.
As OA develops, loss of cartilage, hypertrophic changes in neighboring bone and joint capsule, mild synovial inflammation, and degenerative changes in the menisci, ligaments, and tendons all contribute to pain and loss of joint function, resulting in joint failure.449
In more recent years, it has been discovered that essential inflammatory cytokines such as IL-1β and TNF-α initiate this cycle of catabolic and degradative events in the cartilage, mediated by metalloproteinases, enzymes that degrade cartilage extracellular matrix as part of the normal turnover in all tissues. These enzymes have been shown to be up-regulated after joint injury.
The role of inflammation in the pathophysiology and progression of early OA is supported further by the observation that C-reactive protein levels are raised in women with early knee OA and higher levels predict those whose disease will progress. The synovium from OA joints stains positive for IL-1β and TNF-α. Nitric oxide, which exerts proinflammatory effects, is released during inflammation. In experimental OA, nitric oxide induces chondrocyte apoptosis, thus contributing to cartilage degradation. Hence, unregulated nitric oxide production in humans plays a part in the pathophysiology of the disease.78
Articular cartilage has an important role in joint physiology by providing a smooth, relatively friction-free surface between the bony ends making up the joint. In addition, the cartilage attenuates the mechanical load transmitted through the joint. With progressive loss of cartilage, inflammation develops, with resultant bony overgrowth, ligament laxity, and progressive muscle weakness and atrophy accompanied by joint pain.
Once the cartilage begins to break down, excessive mechanical stress begins to fall on other joint structures. Eventually, fissuring and eburnation of the cartilage (thinning and loss of the articular cartilage resulting in exposure of the subchondral bone, which becomes denser with the surface becoming worn and polished) can occur.
The joint space narrows as the cartilage thins, and sclerosis of the subchondral bone occurs as new bone is formed in response to the now excessive mechanical load. New bone also forms at the joint margins (osteophytes) (Fig. 27-1) with the end result being mechanical joint failure and varying degrees of loss of joint function.
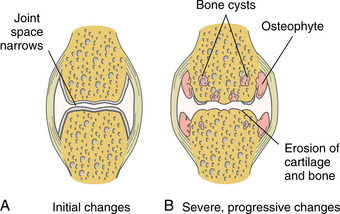
Figure 27-1 A, Early degenerative changes associated with osteoarthritis include joint space narrowing and articular cartilage erosion. B, Late degenerative changes associated with osteoarthritis include osteophyte formation and articular cartilage fissuring and eburnation.
Immobilization is another factor that can result in articular cartilage degeneration. Secondary to the lack of vascular supply, articular cartilage depends on repetitive mechanical loading and unloading for the nutritional elements to reach the chondrocytes and the cellular waste products to return to the synovial fluid and eventually to the bloodstream. This nutritional mechanism of articular cartilage is interrupted by immobilization. If the nutritional cycle is interrupted long enough, structural changes will occur.
Clinical Manifestations
The most common symptoms of OA include bony enlargement, limited range of motion, crepitus on motion, tenderness on pressure, joint effusion, malalignment, and joint deformity. Inflammation is a prominent sign that plays a role in symptom generation. Soft tissue inflammation and edema are observed during acute exacerbations.118 The most commonly involved joints associated with this disorder are the weight-bearing joints, especially the hip and knee but also the shoulder, lumbar and cervical spine and the first carpometacarpal and metatarsophalangeal joints.
The onset of symptoms related to OA can occur insidiously or suddenly. Only a portion of people who have radiographic evidence of OA have associated pain. For most people, however, the pain complaints progress slowly and gradually. Since the cartilage is not innervated, pain is not perceived until the bone or other structures surrounding the joint are involved. The predominate cause of joint pain is attributed to a breakdown in the mechanics of movement rather than inflammation. The pain is often described as a deep ache that is worse with activity and better after rest; pain can occur at rest and at night with advanced disease.128,231
Pain with activity is most likely due to enthesopathy and mechanical factors, whereas pain at rest may be caused by synovial inflammation. Night pain is a poor prognostic indicator and may occur as a result of intraosseous hypertension, which stretches periosteal pain neurons.152
Stiffness of relatively short duration (less than 30 minutes) can occur after periods of inactivity, including sitting and sleeping. Morning stiffness, referred to as the gel phenomenon or “joint gelling,” usually only lasts 5 to 10 minutes after awakening. Movement and activity dissipate this stiffness until the individual sits or rests for a long period of time. This differs from RA, in which the morning stiffness or gelling can last until noon or even midafternoon.
Swelling, if present, is mild and localized to the joint. Loss of flexibility is usually associated with significant disease and can occur secondary to soft tissue contractures, intraarticular loose bodies, large osteophytes, and loss of joint surface congruity.
Crepitus (audible crackling or grating sensation produced when roughened articular or extraarticular surfaces rub together during movement) may be noted on physical examination, and enlarged joint surfaces, including osteophytes, may be palpable. Although many people have physical and radiographic findings of OA, they may not have symptoms, whereas others with minimal changes observed develop significant symptoms. The reasons for this remain unknown.
For many women, OA typically develops within a few years of menopause and is often associated with mild inflammation for the first year or two that a particular joint is involved. The joints may intermittently be warm and tender. The disease is strikingly symmetric, although the degree of involvement may vary somewhat.
OA of the hands affecting the distal interphalangeal and proximal interphalangeal joints occurs most often in this group of women. The gradual loss of joint motion can assume major significance, with the person finding it difficult to grasp small objects.
After 1 or 2 years of inflammation, the joints enlarge with osteophyte (spur) formation, referred to as Heberden’s nodes (affecting the distal interphalangeal joints) and Bouchard’s nodes (affecting the proximal interphalangeal joints) (Fig. 27-2) and become unsightly. Pain may also be noted with loss of joint articular cartilage. Lateral deformities of the joints are common, with stretching of the collateral ligaments and bone resorption. This leads to overlapping of the fingers and considerable loss of functional ability.
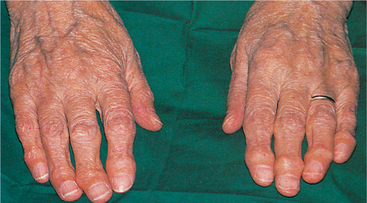
Figure 27-2 Typical hand deformities in osteoarthritis. Heberden’s nodes are seen on the distal interphalangeal joints, and Bouchard’s nodes are at the proximal interphalangeal joints. (From Forbes CD, Jackson WF: Color atlas and text of clinical medicine, ed 3, London, 2003, Mosby.)
Some individuals experience OA of the carpometacarpal joint. With advanced disease, individuals with carpometacarpal involvement may develop joint subluxation as the metacarpal flexes and adducts, leaving the metacarpal base prominent. Axial loading (e.g., pinching) and rotation characteristically reproduce symptoms and cause crepitus. An excellent review of carpometacarpal OA is available.493
MEDICAL MANAGEMENT
Arthritis (including OA and rheumatic conditions) is the leading cause of disability in the United States, affecting a total of over 43 million people in the United States, with an estimated prevalence of nearly 60 million by the year 2020.72,74 The Arthritis Foundation, Centers for Disease Control and Prevention (CDC), and Healthy People 2010 are working together to implement the National Arthritis Action Plan to promote progress toward reaching arthritis-related national objectives for 2010.185
Arthritis research is providing a growing body of knowledge about prevention as well as slowing the disease’s progression and new, more effective combinations of drug and behavioral interventions. Education is a cornerstone of prevention and management for this condition. A healthy lifestyle helps prevent OA, and exercise can lessen disability if OA has developed. Moderate exercise has been shown to improve the knee cartilage glycosaminoglycan content in individuals at high risk of developing OA.426 Strengthening the quadriceps muscle and maintaining an appropriate body weight for height reduce risk of OA at the knee by 30%.119,120
Sports officials and athletes need to work with athletic trainers, exercise physiologists, and physical therapists to evaluate and modify rules, equipment, and playing sur- faces while providing adequate training to help reduce injuries. Early diagnosis and intervention with complete rehabilitation of joint injuries can decrease the risk of subsequent OA.119,120 In the future, biomarkers found in joint fluid, blood, or urine that indicate changes in bone or cartilage may help identify people at risk for OA, allowing for prevention of disease progression and early intervention.
High intakes of vitamin C are associated with lower rates of OA on radiograph examination and less knee pain from OA. High levels of vitamin D protect against new and progressive OA.119,120
DIAGNOSIS.
OA is diagnosed by correlation of history, physical examination, radiologic findings (Figs. 27-3 and 27-4), and laboratory tests, which rule out rheumatic disease. Box 27-6 lists radiographic changes associated with OA. The history of location of symptoms, symptom duration, functional limitations, trauma, medical comorbidities, and family history helps guide the physician in making the diagnosis.
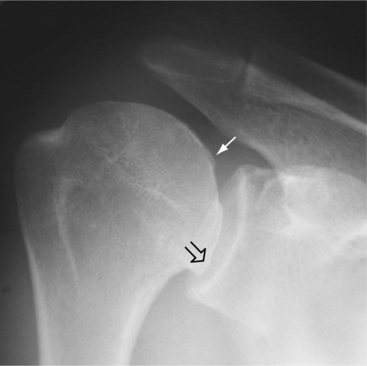
Figure 27-3 Osteoarthritis of the shoulder. There is osteophytic lipping (open arrow) from the humeral head, including new bone formation deep to the cartilage (closed arrow). (From Harris ED: Kelley’s textbook of rheumatology, ed 7, Philadelphia, 2005, Saunders.)
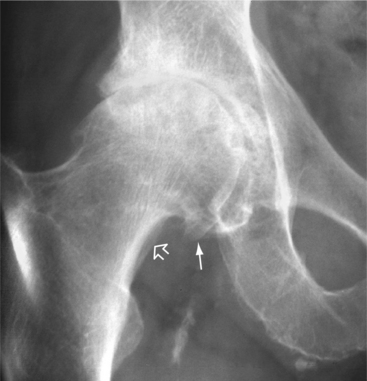
Figure 27-4 Osteoarthritis of the hip. The anteroposterior view of the hip shows complete cartilage space loss superiorly. There is osteophytic lipping from the femoral head, especially medially (arrow), and buttressing bone (open arrow) is present along the femoral neck. (From Harris ED: Kelley’s textbook of rheumatology, ed 7, Philadelphia, 2005, Saunders.)
The American College of Rheumatology’s guidelines for the diagnosis of knee OA include knee pain with radiographic changes of osteophyte formation and at least one of the following: age more than 50 years old, morning stiffness lasting less than 30 minutes, or crepitus on motion.12
Other symptoms diagnostic of OA include a locking or a “giving way” sensation in the knees, swelling, and exacerbation of symptoms with inactivity or overactivity. Walking on uneven ground and climbing stairs also aggravate knee and/or hip OA.513
The physician also relies on findings from the physical examination, such as joint line or bony tenderness, joint effusion (not always present), quadriceps muscle atrophy, varus or valgus deformity (knee), and any abnormalities such as Heberden’s nodes, a classic osteoarthritic change observed in the distal interphalangeal joints of the hands (see Fig. 27-2).
OA is classified based on clinical information and radiologic evidence. The radiographic classification used most often is the 0 to 4 grading system proposed by Kellgren and Lawrence (Box 27-7).242 Grade 4 changes include large osteophytes, severe joint space narrowing, bony sclerosis, and bone exposure (Fig. 27-5).
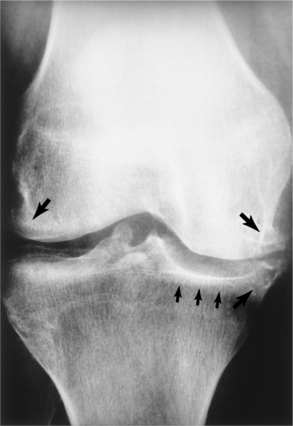
Figure 27-5 Osteoarthritis of the knee. Proliferative marginal osteophytes (larger arrows), narrowing of the medial weight-bearing joint space, and eburnation (exposure of the subchondral bone, surface becomes smooth and polished as it wears down) (smaller arrows). (From Noble J: Textbook of primary care medicine, ed 3, St Louis, 2001, Mosby.)
MRI is becoming increasingly helpful in determining OA pathology because of its ability to show the condition of cartilage and the surrounding soft tissues. Laboratory evaluation may include ESR and rheumatoid factor, but generally these tests are not needed.513
A goal of current research is to develop laboratory tests (i.e., serum, synovial, or urine biomarkers) that would help identify people who are predisposed to OA, detect the disease in its earliest stages, and assess the response to therapy.
TREATMENT.
OA is managed on an individual basis, and treatment consists of a combination of nonsurgical and surgical options. Treatment is modified based on response and should begin with conservative care, including education, weight loss, exercise, orthotics and/or braces, medications, and complementary approaches.
The combination of modest weight loss and moderate exercise provides better overall improvements in function, pain, and mobility in older adults who are overweight or obese with knee OA.332 Greater improvements in function have been observed in older obese adults with the most weight loss.338 Surgery is avoided and considered only when debilitating pain and major limitation of functions interfere with walking and daily activities or impair ability to sleep or work.215
Pharmacotherapy.
For many years, analgesics and NSAIDs have been the first-line medical treatment approach for OA. Newly discovered information about the pathophysiology of OA is paving the way for researchers to design medical therapy that targets specific sites of pathophysiologic pathways involved in the pathogenesis of OA. Medical attention has shifted from easing the pain of OA to slowing the disorder’s progression and actually preventing it.
Strategies under consideration at this time include cyclooxygenase-2 (COX-2) inhibitors (new agents or “super aspirins” that act against inflammation with much less effect on the gastrointestinal system), nitric oxide synthesis inhibitors and antioxidants, chondrocyte and bone growth promoters, metalloproteinase and cytokine inhibitors, and gene therapy.78
Research on cytokines, growth factors, and signaling pathways that was started in the 1990s is now producing new concepts for disease-modifying OA drugs (DMOADs), much like the disease-modifying drugs developed and now available for RA.443,513
Antiresorptive drugs aimed at altering the increased metabolic states of the subchondral bone may have an effect in altering damage done to the overlying cartilage. This approach is based on the hypothesis that the underlying subchondral bone, either indirectly through biomechanical effects or directly via release of cytokines, is responsible for driving the release of degradative enzymes and, ultimately, the destruction of overlying cartilage.
A fairly recent nondrug, nonsurgical treatment known as viscosupplementation has been developed. This intervention involves direct injections into the knee of substances derived from sodium hyaluronate (hylan G-F 20 [Synvisc], Hyalgan), a principal component of natural synovial fluid. These injections help restore some of the viscosity and elasticity of the diseased joint fluid and offer pain relief for 6 to 12 months. Viscosupplementation is used when standard conservative treatments for knee OA (e.g., medications, physical therapy, behavioral therapy) have been inadequate or ineffective.
Education.
The CDC has launched a major public health initiative, called the National Arthritis Action Plan, to identify and change behaviors that may cause OA, calling for a significant change in the way this disease is treated. Medical interventions involving expensive medications, joint injections, and surgery are now suggested for use in the 10% to 30% of cases where OA will progress to severe joint damage.
Multimodal treatment should include client education and self-management, Two excellent resources for consumer education and self-management of OA are available: The Arthritis Cure495 and Maximizing the Arthritis Cure.496 More attention to psychosocial problems (e.g., isolation, depression) that may influence the person’s perception of pain, and to exercise is recommended. The value of a well-designed exercise program including training for strength and endurance also has been recognized.103,119,120,514 Aquatic therapy is especially helpful as a form of moist heat and gravity-eliminated resistive exercise.
Behavioral interventions directed toward enhancing self-management are important, including prevention (see previous discussion), diet and weight control, and low-impact exercise. Complementary approaches to OA (e.g., chondroitin sulfate and glucosamine supplements, acupuncture, yoga, tai chi180) are also gaining in popularity with many people.
Complementary or Alternative Therapy.
Glucosamine and chondroitin sulfate are components of cartilage taken with the hope of decreasing pain and improving function while halting the progression of disease by stopping an enzyme that is believed to break down cartilage. Glucosamine is derived from the shells of lobster, shrimp, and crabs; chondroitin sulfate is derived from cow cartilage. These are available as a nutritional supplement, sometimes referred to as a nutraceutical.
Long-term use of these agents may provide combined structure-modifying and symptom-modifying effects, making these potentially disease-modifying agents for OA. Most people take 1500 mg of glucosamine and 1200 mg of chondroitin daily; anyone weighing more than 200 lb should increase the dosage to 2000 mg of glucosamine and 1600 mg of chondroitin sulfate.190,418,419,495
Investigators in various trials report a range of results with this treatment, making conclusions difficult. Differences in studies are too great to make comparisons or metaanalysis conclusive. Different glucosamine preparations add to the complexity of comparative studies.417,530 The safety of this product used over a long period of time has also been questioned.295
An extensive review of four metaanalyses as well as the recently published findings of the Glucosamine/Chondroitin Arthritis Intervention Trial (GAIT)86 on the efficacy of glucosamine sulfate, glucosamine hydrochloride, and chondroitin sulfate for the treatment of OA has been published.104 Results of previous studies were inconclusive because of poor research design. The GAIT study used rigorous research design to elicit cause and effect and found that glucosamine sulfate and chondroitin sulfate were not effective in reducing knee pain; when combined together, these two were more effective for individuals with moderate to severe pain.104
In another metaanalysis, 20 trials were reviewed to compare the effects of chondroitin with either placebo or no treatment in adults with hip or knee OA. The evidence did not support the use of chondroitin for OA and found no clinically relevant benefit for individuals with advanced OA. According to researchers, the symptomatic benefit of chondroitin is minimal or nonexistent.416a
Results of long-term studies in these two areas (behavioral interventions, complementary medicine) are expected to be published in the next few years. Preliminary results of clinical trials are available but limited.133 Best available evidence suggests that acupuncture, several herbal preparations (e.g., devil’s claw root, white willow bark), and capsaicin cream are beneficial. Evidence is weak or contradictory for homeopathy, magnet therapy, tai chi, leech therapy, music therapy, yoga, imagery, and therapeutic touch. Many other treatments have not been scientifically tested.112
The Arthritis Foundation has published an excellent resource on alternative therapies23; some experts advise people not to think of these therapies as “alternative” but rather as synergistic measures that can be integrated into a total care plan. An area of medicine called complementary and integrative medicine (CIM) has developed as more medical doctors have started incorporating these treatment methods into their overall management plans.
Surgery.
Surgical intervention is considered when pain and loss of function are severe. Arthroscopic management, including lavage and debridement, abrasion arthroplasty, subchondral penetration procedures such as drilling and microfracture, and laser/thermal chondroplasty, may benefit some individuals, potentially delaying reconstructive procedures (e.g., osteotomy, joint arthrodesis or fusion, total joint replacement). Each of these procedures is under investigation for efficacy and long-term results.212
To help joint replacements last longer, intense research is focusing on more wear-and corrosion-resistant materials as well as investigating how the tissue around the replacement responds. Replacement of damaged cartilage is also under investigation using one of three types of cartilage: one’s own cartilage, donor cartilage, and cartilage produced by tissue engineering of progenitor cells (see the section on Tissue Engineering in Chapter 21). Medications that stimulate human cartilage matrix formation without stimulating the chondroresorption processes are also under investigation.188
PROGNOSIS.
OA is a major contributor to functional impairment and reduced independence in older adults (more than 65 years). It is a chronic condition with unpredictable symptoms that often cause fluctuations in pain and function.2
Mobility disability, defined as needing help walking or climbing stairs, is common for those with hip and/or knee OA. The social burden in terms of personal suffering and use of health resources is expected to increase with the increasing prevalence of obesity and the aging of the American population.215
Although there is no known cure for OA, by following the guidelines for lifestyle changes, pain management, and self-management incorporating exercise and weight loss, affected individuals can substantially decrease the pain and dysfunction associated with OA.
Degenerative Intervertebral Disk Disease
The degenerative joint process described in the previous section applies to any synovial joint, including the facet joints of the spinal column. Degenerative joint changes are not limited to synovial joints, however, and in the spine (particularly the low lumbar segments) they commonly occur at the intervertebral disk articulations as well.262
Currently, there is no consensus on what “disk degeneration” actually is or how it should be defined or distinguished from the physiologic processes of growth, aging, healing, and adaptive remodeling. The following working definition has been proposed as a starting point: “The process of disk degeneration is an aberrant, cell-mediated response to progressive structural failure.”1
Lumbar disk degeneration begins early in life. Severe macroscopic changes are visible from the age of 30 onward. Atherosclerosis is present by middle age or even earlier for some people in the abdominal aorta.339 It is estimated that half of all Americans over the age of 40 are affected by degenerative disk disease (DDD).
Disk degeneration follows a predictable pattern. First, the nucleus in the center of the disk begins to lose its ability to absorb water. The disk becomes dehydrated. Then the nucleus becomes thick and fibrous, so that it looks much the same as the annulus. As a result, the nucleus isn’t able to absorb shock as well. Routine stress and strain begin to take a toll on the structures of the spine. Tears form around the annulus. The disk weakens. It starts to collapse, and the bones of the spine compress.325 This process will be discussed in greater detail later in the Pathogenesis section.
Incidence
Lumbar DDD is a common musculoskeletal disease estimated to affect up to 5% of all adults. Although this disease and resulting condition can occur before age 20, it tends to peak in the fourth and fifth decades of life and declines after that. It has been reported that men suffer from sciatica due to disk herniation about 1.5 to 3 times more often than women, but it is not clear if this is a true difference in prevalence of DDD or can be attributed to anatomic and mechanical factors that contribute to nerve root compression.4
Risk Factors
The greatest risk factor for disk degeneration is genetic inheritance, which accounts for approximately 50% to 70% of the variability in disk degeneration between identical twins. Individual genes associated with disk degeneration have been identified (e.g., aggrecan, collagen type IX, matrix metalloproteinase-3 [MMP-3], vitamin D receptor).1,34,434
Twin and family studies have shown that the risk of developing a lumbar disk herniation is approximately five times greater in people who have a positive family history. Mutations that alter collagen I structure (the major component of the outer annulus) may contribute to annular tears and disk ruptures. Sequence variations in the genes for collagen IX may also be linked with DDD.4
Age and body weight appear to be two other significant risk factors for DDD. There has been a prevailing view that DDD occurs as a result of excessive forces, particularly from the cumulative effects of repeated loading from occupational physical demands, such as manual material handling. Results of research to confirm or refute this hypothesis have been mixed; some studies find a correlation between disk degeneration and physical demands, while other do not.35,527 It appears that actual time spent lifting and carrying loads is less important than body weight or mass.527
Results of a recent study of the Finnish Twin Cohort suggest that routine (daily) physical loading of the spine may actually have a training (rather than detrimental) effect. Occupational lifting or repeated loading of the spine from physical activity may in fact benefit the disks527; cyclic mechanical stress may increase the growth rate and collagen fibers of the nucleus pulposus.313
Height has been suggested but not consistently found to be a risk factor in all studies; the link between DDD and obesity and smoking also remains controversial. The role of psychosocial factors has also been investigated, and they have been found to have a positive link as risk factors for disk herniation, which can lead to further DDD. Atherosclerosis is another potential risk factor. Obstruction in the abdominal aorta, lumbar, and middle arteries can lead to ischemia in the lumbar spine, resulting in hypoxia and tissue dysfunction with eventual disk degeneration.240
Etiology and Pathogenesis
The intervertebral disk undergoes marked changes with age, although genetic inheritance, inadequate metabolite transport, and loading history combined with age contribute significantly to structural failure that can occur during ADLs.1
The most significant alterations occur in the nucleus pulposus. The number of cells and the concentration of proteoglycans and water decrease. In addition, there is fragmentation of proteoglycans, which contributes to water loss. As the nucleus breaks down, the fibrocartilaginous inner annulus expands. Fissures and clefts may form within the disk, and the height of the disk decreases. This loss of disk height can contribute to the age-related condition of spinal stenosis.21
A number of events can contribute to the three stages of age-related disk degeneration (Box 27-8).251 The most important event appears to be the decreased cellular function and concentration. The progressive decline in arterial supply to the periphery of the disk and the impairment of nutrient delivery across the cartilaginous endplate contribute to the reduced nutritional supply to the cells, affecting cellular function.
In addition, the impaired cartilaginous endplate diffusion results in reduced cellular waste product removal and an increased lactic acid concentration. The resultant decreased pH level compromises cellular metabolism and biosynthesis and can lead to cell death. The reduced biosynthesis can adversely affect the biomechanical properties of the matrix over an extended period of time.
Besides the internal events affecting the general health of the intervertebral disk, repetitive external mechanical loading on the structure can lead to fatigue failure of the matrix. When enough structural breakdown occurs, what once were normal mechanical loads acting on a normal disk are now excessive loads on a compromised disk. At this point the degenerative process is accelerated.58
Collapse of the inner annulus into the nucleus is a common feature in the disks of older adults, with the anterior annulus being affected more than the posterior (Fig. 27-6). This could be caused by nucleus decompression following endplate fracture. In many older disks, the cartilage endplate becomes detached from the underlying bone, presumably because the high internal pressure that presses it against the bone in young disks has been lost.1,90,491
Figure 27-6 Lumbar intervertebral disks (midsagittal section; anterior on the left). A, Young disk in a male, 35 years old. B, Mature disk in a male, 47 years old. C, Disrupted disk in a male, 31 years old. Note the endplate and inward collapse of the inner anulus. D, Severely disrupted disk in male, 31 years old. Note the collapse of disk height. E, Disk induced to prolapse in cadaver male, 40 years old. Some nucleus pulposus has herniated through a radial fissure in the posterior anulus (right). (From Adams MA: The biomechanics of back pain, Edinburgh, 2002, Churchill Livingstone.)
The results of the Videman et al study527 go against the traditional view that physical loading and repetitive spinal movements are bad for the back. The authors suggest that lifelong loading from body weight is more detrimental than work-related lifting and loading. Loading from daily physical activity may actually help offset the effects of aging. Forces across the lumbar spine vary with body weight and lifting strength; this may explain why some disks degenerate more than others. The authors also suggest that smaller disks hold up better because it is easier for nutrients to reach each cell.
Associated with intervertebral disk degeneration are spinal stenosis and degenerative spondylolisthesis, two common conditions in the older adult (more than 65 years). As the intervertebral disk loses height, the annulus may bulge circumferentially and the ligamentum flavum can buckle. Both encroach on the spinal canal, subarticular (facet) recesses, and lateral intervertebral foramina. With a loss of disk height, compressive force increases on the neural arch,405 causing OA of the facet (apophyseal) joints and osteophytes around the margins of the vertebral bodies (Fig. 27-7).526 In addition, concurrent osteophyte formation on the vertebral bodies or articular processes may occur, compounding the stenosis.
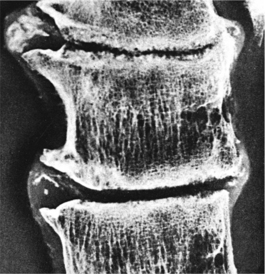
Figure 27-7 Radiograph of a lumbar spine (anterior on left) with severe disk narrowing, vertebral osteophytes, sclerosis of the endplates, and selective loss of horizontal trabeculae from the vertebral body. (From Adams MA: The biomechanics of back pain, Edinburgh, 2002, Churchill Livingstone.)
Degenerative spondylolisthesis is marked by anterior slippage of one vertebra over another with an intact posterior neural arch. The L4L5 spinal segment is the most common site for this to occur (Fig. 27-8). Lytic spondylolisthesis is marked by anterior slippage of one vertebra over another with a defective posterior neural arch (Fig. 27-9). The L5S1 spinal segment is the most common site for lytic spondylolisthesis to occur. The loss of disk height associated with degeneration allows for a buckling of the annulus and ligamentum flavum, slackening them some what. This allows the vertebrae to migrate anteriorly in response to the shear forces inherent to the lumbar lordosis.189
Figure 27-8 Degenerative spondylolisthesis at L4-L5. (From DeRosa C, Porterfield JA: Lumbar spine and pelvis. In Richardson JK, Iglarsh ZA, eds: Clinical orthopaedic physical therapy, Philadelphia, 1994, Saunders, p 144.)
Figure 27-9 Spondylolysis or posterior arch defect, which can lead to a lytic spondylolisthesis. The Scottie dog with a collar, which is visible on the radiograph (posterior oblique view), is outlined. (From Magee D: Orthopedic physical assessment, ed 2, Philadelphia, 1992, Saunders.)
In contrast to the L5-S1 segment, the facet joint orientation at the L4-L5 segment tends to be more in the sagittal plane, so there is no structural bar to anterior slippage. The facet joint orientation at L5-S1 tends to be more in the frontal plane, making anterior migration of L5 difficult unless there is a structural defect in the posterior neural arch. Stenosis can be caused by the displacement of one vertebra over another as well as by the concurrent buckling of the annulus and ligamentum flavum.
Clinical Manifestations
Disk degeneration is most likely to affect the lower lumbar spine but can affect the upper lumbar spine and cervical spine as well. Low back pain is often the first symptom, but DDD is asymptomatic in up to one third of all affected individuals.45,228
DDD is one of the most common causes of low back pain with radiculopathy. The intervertebral disk changes include alterations in volume, shape, structure, and composition. Although there is not a 100% correlation between the presence of DDD and pain complaints, the structural alterations will decrease motion and alter the mechanical properties of the spine.58
Most people with DDD have a gradual onset of increasingly severe midline lower back pain. At first, symptoms only last a few days. This type of back pain is often intermittent but recurring over the years. Each time it occurs, the pain may seem worse than the time before. Eventually the pain may spread into the buttocks or thighs, and it may take longer to subside.325
When pain is present, it tends to get worse after heavy physical activity or after a prolonged time in one position. The back may also begin to feel stiff. Resting the back eases pain. Aching of the buttock(s) and posterior thigh(s) with ambulation is common.
The symptoms of DDD should not be confused with disk herniation, which may occur as part of the process of disk degeneration but is not a constant feature and is a separate entity from DDD. Clinical presentation of disk protrusion and herniation can vary widely but can occur as a result of tears of the annulus fibrosus, causing acute back or neck pain or radicular pain if there is herniation of the nucleus pulposus.
In the lumbar spine, radiculopathy causing sciatic pain and restricted straight leg raise may occur as the sheath of the nerve root is compressed483 or as a result of inflammatory irritation from chemicals released by the damaged disk.
Centralization of radiating pain is characteristic of sciatica from disk protrusion or herniation. Specifically, leg pain routinely reduces or “retreats” to the lumbar midline before the disappearance of back pain. This distinctive pain pattern was first described by McKenzie.320
Other signs of disk herniation include ankle dorsiflexion weakness; great toe extensor weakness; impaired ankle reflexes; loss of light touch sensation in the medial, dorsal, and lateral aspects of the foot; and positive ipsilateral or crossed straight leg raise test. These six neurologic tests allow detection of most clinically significant nerve root compromises resulting from L4-L5 or L5-S1 disk herniations.42,171
Gait abnormality, muscle weakness, sensory changes such as numbness and tingling, and bowel or bladder dysfunction can occur as a result of myelopathy associated with either disk herniation or degeneration. In the case of cervical disk involvement, difficulty swallowing, hand numbness, Lhermitte’s sign, and hoarseness or voice difficulties may occur.
MRI observations of disk degeneration and herniation are poorly correlated with clinical signs and symptoms. For example, a large, extruded disk may be clinically tolerable if the spinal canal is large and the spinal nerve roots are not compressed. On the other hand, a focal, contained subligamentous herniation may produce severe symptoms if it occurs in the foramen adjacent to the dorsal root ganglion of the affected nerve.321
In the case of disk degeneration associated with stenosis and spondylolisthesis, the person can be asymptomatic, even when moderate to severe changes are observed on imaging studies. Conversely, early or mild changes can be accompanied by severe pain and neurologic symptoms.
MEDICAL MANAGEMENT
Diagnosis requires a thorough history, physical examination, and imaging protocol; provocative diskography (with dye injected into the suspected disk or disks) will reveal annular tears that cause pain and offer a definitive diagnosis, especially when used in conjunction with other imaging studies. The dye seen on x-ray can give information about the health of the disk(s). This test may be done when the surgeon is considering surgery, since it can help determine which disk is causing the symptoms.
The x-rays provide a measure of disk space; narrowing of the disk spaces is suggestive of the degenerative process. The presence of osteophytes affecting facet joints and any spondylolysis or spondylolisthesis can also be visualized on x-rays.
MRIs visualize the soft tissues of the body and are helpful for showing if the disks are able to absorb water and any radial fissures present inside the disk. They can also show if there are problems in other soft tissues, such as the spinal nerves.
TREATMENT.
Conservative care, including NSAIDs, mild analgesics for short-term pain control, physical therapy, lifestyle changes (e.g., smoking cessation, weight loss, fitness program), and aerobic conditioning is the first line of treatment for painful DDD.321 The goal is to reduce painful symptoms and enable the person to get back to normal activities as soon as possible. Bed rest is no longer the standard of care.
Surgery.
Surgery to remove the disk or disk fragments (diskectomy), laminectomy, and/or spinal fusion may be considered if conservative care has been unsuccessful and/or neurologic symptoms persist. The timing of surgery can be important to avoid permanent nerve damage and footdrop.
Long-term studies show that in the absence of neurologic compromise, the outcomes for conservative care and surgical intervention are the same 10 years after the initial incident, suggesting that surgery can be postponed indefinitely. The exceptions are when the individual is at risk for neurologic damage and when conservative care does not alter the pain, thereby compromising function and quality of life.
For individuals with chronic, nonradicular, diskogenic low back pain, intradiskal electrothermal therapy (IDET) has been used with success in 60% to 80% of the cases. Thermal energy directed at the disk material through a catheter uses heat to induce tissue shrinkage. IDET curbs the release of irritating substances and destroys the surrounding pain receptors and offers a less invasive alternative to surgery for the treatment of chronic and severe DDD.538
Other minimally invasive treatments include epidural injections and transdiskal radiofrequency therapy (called intradiskal biacuplasty); both have been used with varying degrees of success.237 The use of one to three epidural steroid injections for DDD has gained in popularity for individuals whose symptoms continue to limit function. A steroids combined with a long-lasting numbing agent is injected into the space around the lumbar nerve roots.
For some individuals, artificial disk replacement (ADR) is an alternative to spinal fusion. After removing what is left of the damaged or worn-out disk, the ADR device is inserted in the space between two lumbar vertebrae. The goal is to replace the diseased disk while keeping the normal spinal motion. ADR devices are not designed at this time for the treatment of herniated disks but for one or two levels of DDD. Cervical arthroplasty is also available now, with primary indications for the treatment of radiculopathy and myelopathy at one or two levels.
Artificial disk surgery is relatively new in the United States but has been used in Europe for many years. In the United States, the first lumbar artificial disk surgery was done in clinical trials in October 2001. The U.S. Food and Drug Administration (FDA) approved the use of the lumbar ADR implants in October 2004.324
The advantages of this treatment over spinal fusion are maintenance of spinal movement, disk height, and neural foramina, thus simulating a more normal spinal alignment, angulation, and mechanics. Immobilization is avoided with ADR, and early return of function is possible. There is the additional advantage of preventing adjacent-segment deterioration that can occur after spinal fusion.
There are also risks and disadvantages (e.g., infection; ossification; neurologic impairment; implant failure; and fracture, migration, or subsidence or sinking down into the bone).233 Although short-term and medium-term results are favorable, long-term results are not yet available.247a,510
Gene Therapy.
Researchers are investigating the use of gene therapy to slow, prevent, or reverse the biochemical changes associated with disk degeneration. Alternatively, if the correct gene could be identified, it might be possible to manage an annular tear by direct repair or regeneration.11 In other areas of study, autologous chondrocyte transplantation may become a future treatment for DDD. Animal studies have shown that chondrocytes removed from damaged cartilaginous tissues maintain a capacity to proliferate and reproduce tissue similar to normal intervertebral disk material.328
PROGNOSIS.
Disk structural failure is irreversible, always progresses by physical and biologic mechanisms, and is closely associated with mechanical dysfunction and pain.1 The potential for recovery varies based on the size of the protrusion, the size of the canal, the person’s age and activity level, the extent of disk disruption, and similar parameters related to spondylolisthesis and stenosis.494 Laminectomy and/or diskectomy removes the mechanical cause of pain, but the inflammatory process may cause persisting pain for a few more days or, in some cases, indefinitely as a result of fibrosis impinging on the nerve tissue.
For those with disk herniation, the type of disk herniation can predict the final outcome. Individuals with a bulging disk and normal annulus have the best result. Pain from sciatica is relieved after diskectomy. Individuals with bulging disk and large annular defects have a high rate of reoperation.70
Some, but not all, individuals with spondylolisthesis and stenosis may experience pain relief and improved function after surgery (fusion). In the older adult, recovery can be very slow; rehabilitation is a key feature in this process.
Rheumatic Diseases
Rheumatic disorders are systemic diseases encompassing over 100 different diseases divided into 10 classification categories. The pathogenesis and progression of these disorders can affect any and all body systems. The onset of joint pain and loss of function may be accompanied by fever, rash, diarrhea, scleritis, or neuritis symptoms that are not typically associated with joint or muscle conditions normally brought on by repetitive overuse or trauma.
Rheumatic disorders are also often marked by periods of exacerbation and remission. During a period of exacerbation the therapist will often need to modify the treatment approach considerably. In addition, aggressive medical intervention (i.e., medications) may need to be initiated to prevent or minimize the tissue destruction that can occur with these disorders. Many of the rheumatic conditions are chronic and progressive, requiring long-term rehabilitation and ongoing adjustment of functional goals.
Therapists must be able to differentiate between degenerative joint disease (OA) and rheumatic joint conditions (Table 27-1). If there is any suspicion of the presence of a rheumatic disorder, immediate referral to a physician is warranted. When someone with RA presents with systemic symptoms or if existing complaints worsen, communication with a physician is advised. An understanding of the diseases discussed in this chapter will assist the therapist regarding this clinical decision-making process.
Table 27-1
Osteoarthritis and Rheumatoid Arthritis
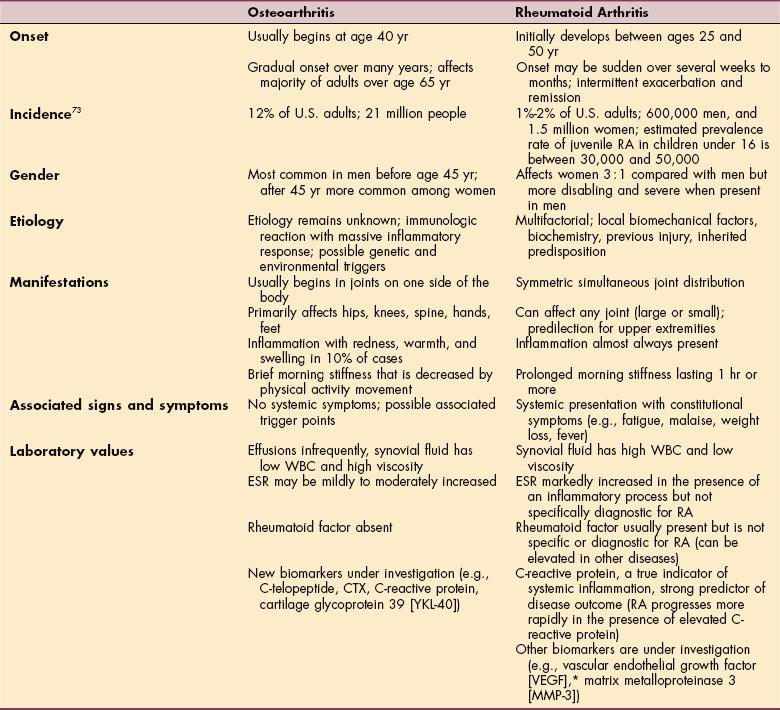
RA, Rheumatoid arthritis; WBC, white blood cell count; ESR, erythrocyte sedimentation rate.
*VEGF is an angiogenetic cytokine; its presence supports the theory that expansion of the synovial vasculature is important for the development of joint destruction in RA.
Rheumatoid Arthritis
Overview.: RA is a chronic systemic inflammatory disease presenting with a wide range of articular and extraarticular findings. Chronic polyarthritis, which perpetuates a gradual destruction of joint tissues, can result in severe deformity and disability. Systems that may be involved include the cardiovascular, pulmonary, and gastrointestinal systems. Eye lesions, infection, and osteoporosis are other potential extraarticular manifestations.
RA is a major subclassification within the category of diffuse connective tissue diseases that also includes juvenile arthritis, SLE, progressive systemic sclerosis (scleroderma), polymyositis, and dermatomyositis.162
Incidence and Risk Factors.: RA has a worldwide distribution and affects all races. Approximately 1% to 2% of the U.S. adult population (2.1 million people) has RA, which is the second most prevalent form of arthritis after OA.
Age and female gender are the two primary risk factors associated with RA. Although the onset of the disorder can occur at any age, the peak onset is usually between 20 and 50 years; with the aging of America, the prevalence of RA is expected to rise. Women are affected two to three times more frequently than men; although it is less common, children can also develop the disorder (see the section on Juvenile Idiopathic Arthritis in this chapter).
Pregnancy and oral contraceptives appear to influence the incidence and severity of the disease. The incidence of RA in women who have borne a child is lower, and oral contraceptives diminish the incidence of severe arthritis. A nulliparous woman who does not use oral contraceptives has a fourfold increased risk of developing RA.72,182,470
Prophylactic administration of recombinant hepatitis B vaccine may trigger the development of RA in previously healthy people. Vaccine-related arthritis may be linked to genetic factors, such as the presence of MHC class II molecules.407 An association between autoimmune thyroid diseases and rheumatic diseases has been established, although its precise mechanism is unclear. For example, RA often occurs in association with Graves’ disease and Hashimoto’s thyroiditis. In these individuals there is a significant presence of antithyroid autoantibodies.312
Drinking decaffeinated coffee (four or more cups per day) may be an independent risk factor among older women, especially in the presence of seropositive disease. The mechanism for this to occur remains unknown.336
Etiologic Factors.: Little is known about the exact causes of RA, except that joint inflammation is a consequence of massive infiltration of immune cells, especially T lymphocytes, into the synovial fluid. Genetic predisposition and environmental triggers, such as a bacteria (e.g., Mycoplasma fermentans), are both considered possible etiologic factors in the stimulation of T cells.116,204
Pathogenesis.: RA is considered an autoimmune disease, with inflammation and destruction targeted at the joint capsule (articular) and elsewhere throughout the body (extraarticular). Approximately 80% of people with RA are rheumatoid factor positive.177
Rheumatoid factors are autoantibodies that react with immunoglobulin antibodies found in the blood. Rheumatoid factor has also been found in the synovial fluid and synovial membranes of those with the disease. It is hypothesized that the interaction between rheumatoid factor and the immunoglobulin triggers events that initiate an inflammatory reaction.
RA begins attacking the joint in the synovium. The normal synovial membrane consists of loose connective tissue that contains blood vessels and is covered by a layer of synovial lining consisting of macrophages and synoviocytes and is only minimally infiltrated by lymphocytes. In RA, the cells of the synovial lining multiply, there is an influx of leukocytes from the peripheral circulation, and the synovium becomes edematous. The synovial lining thickens, resulting in the clinical synovitis seen so often.
These changes can result in the development of thickened synovium, a destructive vascular granulation tissue called pannus. The inflammatory cells found within the pannus are destructive, preventing the synovium from performing its two primary functions: lubricating the joint and providing nutrients to the avascular articular cartilage. As this tissue proliferates, encroaching on the joint space at the margins where the hyaline cartilage and synovial lining do not adequately cover the bone, it dissolves collagen, cartilage, subchondral bone, and other periarticular tissues in its path (Fig. 27-10).
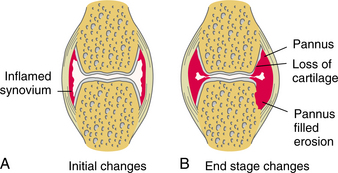
Figure 27-10 A, Early synovial changes associated with rheumatoid arthritis. B, Late joint changes associated with rheumatoid arthritis, including pannus formation and articular cartilage eburnation.
Although the cause of RA remains unknown, recent advances in molecular techniques have allowed for identification of distinct cell subtypes, surface markers, and products that may initiate and propagate the inflammatory and destructive components of the disease. Two cytokines, TNF-α and IL-1, appear to play a major role in the pathophysiologic process of RA.66
The dominant cytokine, TNF, stimulates production of other cytokines from the interleukin family, thus prompting a massive inflammatory response. As the attracted leukocytes, monocytes, and lymphocytes phagocytose the immune complexes, TNF stimulates the secretion of matrix metalloproteinases (protein-degrading enzymes that lyse the cartilage and destroy the joint), leading to articular cartilage destruction and synovial hyperplasia with local tenderness, swelling, and intense joint pain. TNF also inhibits bone formation and induces bone resorption.
Fortunately, a new member of the tumor necrosis family has been identified: osteoprotegerin (OPG). This protein plays a key role in the physiologic regulation of osteoclastic bone resorption, counteracting the destruction and bone degradation caused by the cytokine-induced inflammatory process. OPG might represent an effective therapeutic option for diseases such as RA associated with excessive osteoclastic activity.
The result of the synovial changes that occur in RA can be irreversible joint instability, joint deformity, or ankylosis (adhesions and fibrous or bony fusion of the joint). Joint destruction eventually leads to laxity of the tendons and ligaments, which contributes to the altered biomechanics and deformities frequently observed.110 The wide range of extraarticular problems is also probably a result of local inflammatory injury induced by the immune complexes traveling through the circulatory system.
Clinical Manifestations.: RA is a systemic disease typically manifested by articular and extraarticular complaints (Box 27-9; see also the section on Rheumatoid Arthritis in Chapter 5). The symptoms usually begin insidiously and progress slowly as the disease process moves from cartilage degradation to ligamentous laxity and, finally, synovial expansion with erosion. Complaints of fatigue, weight loss, weakness, and general, diffuse musculoskeletal pain are often the initial presentation. Deconditioning and depression are common complications of this disease.
The course of RA can vary considerably from mild to severely disabling and is difficult to predict, but it appears that adults with RA today have less severe symptoms and less functional disability than even a decade ago. This positive trend and more favorable course of disease may be attributed to earlier diagnosis with a shorter duration of symptoms at the time of diagnosis and more aggressive use of drug therapy.537
Joint.: The musculoskeletal symptoms gradually localize to specific joints. Multiple joints are usually involved, with symmetric, bilateral presentation. The most frequently involved joints are the wrist, knee, and joints of the fingers, hands, and feet, although RA can affect any joint, including the temporomandibular joints. The metacarpophalangeal and proximal interphalangeal joints of the hand are involved early. The involved joints can be edematous, warm, painful, and stiff. After periods of rest (e.g., prolonged sitting, sleeping) intense joint pain and stiffness may last 30 minutes to several hours as activity is initiated.
As the disease progresses, joint deformity can occur, including subluxation. Deformities in the fingers are common, including ulnar deviation, swan-neck deformity, and boutonnière deformity. The ulnar deviation occurs as the extensor tendons slip to the ulnar aspect of the metacarpal head. Hyperextension of the proximal interphalangeal joint and partial flexion of the distal interphalangeal joint make up the swan-neck deformity (Fig. 27-11). The boutonnière deformity is marked by flexion of the proximal interphalangeal joint and hyperextension of the distal interphalangeal joint (Fig. 27-12).
Soft Tissue.: Soft tissue manifestations of RA can include synovitis, bursitis, tendinitis, fasciitis, neuritis, and vasculitis. These problems are often overlooked but can be very debilitating. Soft tissue imbalance combined with joint involvement can result in significant deformity, especially in the hands and feet.
Spine.: Early involvement of the spinal column is common and typically limited to the cervical spine, with deep, aching neck pain radiating into the occipital, retroorbital, or temporal areas reported in 40% to 88% of persons.44,248,420 Neck movement precipitates or aggravates neck pain; facial and ear pain and occipital head- aches occur frequently with active disease from irritation of the C2 nerve root supply to the spinal trigeminal tract, greater auricular nerve, or greater occipital nerve.248
There is a potential for atlantoaxial subluxation (usually anterior) and brainstem or spinal cord compression. The upper cervical spine is affected most commonly because the occiput-C1 and the C1-C2 articulations are purely synovial and are thus primary targets for rheumatoid involvement. In addition, because the C1 and C2 facets are oriented in the axial plane, there is no bony interlocking to prevent subluxation in the face of ligamentous destruction.420
The natural history of cervical instability in people with RA is variable, and only some develop neurologic deficits.108 Symptoms of C1-C2 subluxation include a sensation of the head falling forward with neck flexion, loss of consciousness or syncope, dysphagia, vertigo, seizures, hemiplegia, dysarthria, nystagmus, and peripheral paresthesias and loss of dexterity of the hands.49 Urinary retention and later incontinence are symptoms of more severe involvement. Sleep apnea may be caused by brainstem compression associated with atlantoaxial impaction.396
There may be a positive Lhermitte sign with shocklike sensations of the torso or extremities with neck flexion. Atlantoaxial instability may result in vertebrobasilar insufficiency with visual disturbances, loss of equilibrium, vertigo, tinnitus, and dysphagia. These symptoms can also be caused by mechanical compression of the cervicomedullary junction or brainstem.420 Asymmetrical destruction of the lateral atlantoaxial joints may result in a clinical presentation of head tilt down and to one side. When the neck is flexed, the spinous process of the axis may be prominent.
Pain associated with RA in the subaxial segments of the cervical spine is located in the lateral aspects of the neck and clavicles (C3-C4) and over the shoulders (C5-C6). Neurologic symptoms include burning paresthesias and numbness, which may be attributed to carpal tunnel syndrome, delaying the diagnosis of cervical myelopathy.