CHEMOTHERAPY111
Systemic chemotherapy plays a major role in the management of the 60% of malignancies that are not curable by regional modalities. As with radiation therapy, chemotherapy acts by interfering with cellular function and division. Chemotherapy may be used to cure cancer, to palliate or stabilize disease as preliminary therapy before bone marrow transplantation, or as adjuvant therapy.
In contrast to most cells in the body, tumor cells undergo frequent cell division, leading to an accumulation of cells that are cytologically and histologically defective. Cellular processes needed to support this increased cell division, such as DNA synthesis, DNA repair, DNA replication, and RNA transcription, are themselves accelerated. The principal goal of chemotherapy is to destroy malignant cells with the least harm to normal cells or the host. However, most chemotherapeutic agents are nonspecific and therefore affect both malignant and normal cells.144
Researchers first used these unique characteristics of tumor cells as targets for antitumor drugs in the mid1940s. Goodman and Gillman successfully reduced tumor size in adults with non-Hodgkin’s lymphoma with mustine, a drug that disrupts the normal structure of DNA.94 This discovery led to the development of many new drugs, commonly referred to as chemotherapeutic drugs, which specifically target those processes needed to support mitotic activity and cell division. Although such drugs have been successful in treating a wide variety of cancers, they are unable to distinguish cancerous from noncancerous cells (i.e., they lack specificity) often attacking normal, as well as cancerous, cells.
Characteristics and Categories of Chemotherapeutic Drugs
Chemotherapeutic drugs are systemic drugs, meaning that they travel throughout the body rather than remain confined to a specific area. They are able to reach cells in the primary tumor and cancerous cells that may have escaped from the primary tumor. Many chemotherapeutic agents are systemic and nonspecific, which means they can reach and exert their toxic effects on noncancerous cells as well.
Normal cells most at risk for damage by chemotherapeutic agents are those that normally have high mitotic rates such as hepatic cells, cells that make up epithelial layers, bone marrow cells, and hair cells. However, virtually every organ in the body can be affected by these drugs; for this reason, chemotherapy is often accompanied by multisystem problems and disease.
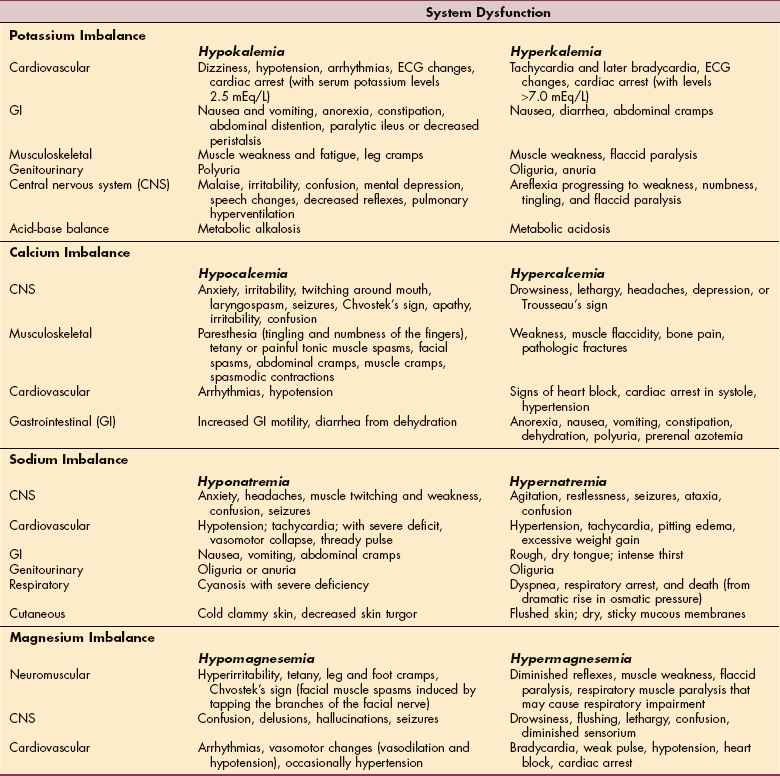
Four broad categories of systemic chemotherapeutic agents are generally recognized, each of which interferes in some manner with compounds or processes that contribute to cell growth (Table 5-8). Specifically, alkylating agents insert themselves into DNA strands, disrupting the normal structure of the strand, preventing the successful replication of an exact copy of that DNA strand, creating a break in the DNA strand. These compounds are mutagenic and potentially carcinogenic.
Table 5-8
Major Toxicities Commonly Associated with Cancer Chemotherapeutic Agents
*Myelosuppression: bone marrow suppression resulting in anemia, leukopenia leading to infection, and thrombocytopenia.
Drugs known as antimetabolites are structurally similar to the purine and pyrimidine bases that form the backbone of each DNA strand. These drugs act either by being incorporated into the DNA strand, leading to the synthesis of a defective DNA strand, or by inhibiting enzymes necessary for DNA and RNA replication, as well as protein synthesis.
Several chemotherapeutic agents are antibiotics. These compounds are incorporated into the DNA strand, preventing the synthesis of DNA and RNA. These compounds also lead to the formation of free radicals, which can damage DNA and cell membranes. Finally, a variety of plant alkaloids are effective in treating cancers, because they interfere with the formation of the mitotic spindle, subcellular structures that transfer genetic material from the mother cell to the two daughter cells.
Although systemic chemotherapy drugs remain the mainstay of chemotherapy, drugs targeting critical biochemical pathways unique to tumor cells have become available. These so-called targeted therapies include imatinib mesylate (Gleevec) and trastuzumab (Herceptin). Gleevec specifically attacks the Philadelphia chromosome, which is a chromosome translocation found in chronic myelogenous leukemia.
The anti-HER2 antibody trastuzumab (Herceptin) is used to treat some women with breast cancer. About 30% of individuals with breast cancer have increased expression of the human epidermal growth factor receptor (HER2), resulting in aggressive tumors and poor prognosis. Herceptin specifically targets these receptors and in doing so, increases the likelihood of tumor regression in select individuals. These targeted therapies do not randomly attack rapidly dividing cells, so they generally cause fewer and less severe side effects than do chemotherapy drugs.77
Adverse Effects of Chemotherapy
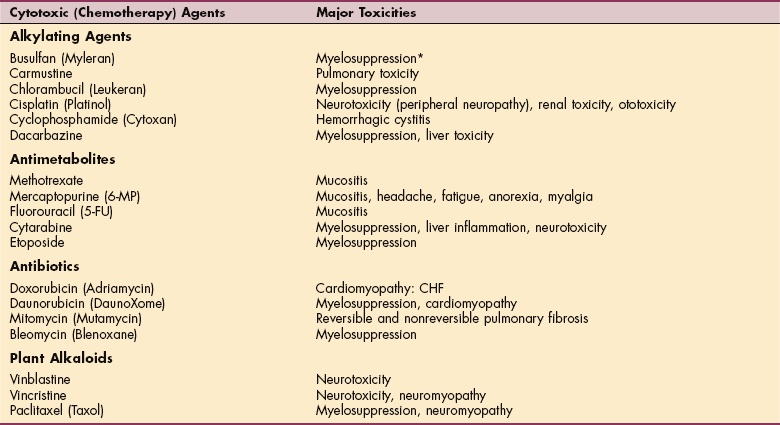
Many chemotherapy agents have unique, dose-limiting toxicities. Chemotherapy drugs are used in combination for their specific actions on cells and care is taken not to use agents with significant overlapping toxicities. The short-and long-term toxicities of commonly used chemotherapy drugs are outlined in Table 5-8 (see also Table 9-7).
Most chemotherapeutic agents have the propensity to cause nausea and vomiting with the administration of the drug, and mucositis, diarrhea, myelosuppression, and alopecia often occur after treatment. Many cause sterility and are toxic to a fetus.132
Cognitive deficits referred to as chemotherapy related cognitive dysfunction (CRCD) can have a dramatic effect on a person’s quality of life. These deficits can be subtle or dramatic, transient or permanent, or stable or progressive.69
Alopecia
Alopecia (hair loss) is the most noticeable cutaneous side effect of chemotherapy and often the most distressing because it has a profound social and psychologic impact on the individual. Actively growing hair or anagen hair is the most rapidly proliferating cell population in the human body and therefore very susceptible to the effects of systemic chemotherapeutic agents. Chemotherapy also renders the hair shaft more fragile and thus prone to significant damage with minimal trauma. As a result, hair breaks, falls out, and is not replaced.
Alopecia is more likely to occur when high-dose chemotherapy is used to treat the cancer (not all chemotherapy regimens have this effect) and typically occurs within 2 to 3 weeks after the onset of chemotherapy. Hair loss is not restricted to the scalp, but because this area has the greatest amount of anagen hair, the greatest losses occur here. Hair loss is temporary, with regrowth typically occurring 4 to 6 weeks after termination of treatment. Full hair restoration may require 1 to 2 years to complete and may be accompanied by changes in hair color, texture, and type.83
Gastrointestinal Toxicity
Over 75% of clients receiving combination chemotherapy experience nausea and vomiting, which typically occurs 1 to 2 hours after the administration of the agent, with the effects peaking at 4 to 10 hours after administration and lasting approximately 12 to 24 hours. Potential medical complications of chemotherapy-induced emesis include dehydration, electrolyte and acid-base disturbances, and anorexia with accompanying weight loss.12 A variety of antiemetic drugs are currently available, and often these drugs are given in combination.117 However, these drugs are not without their own side effects.
Other agents, such as high-dose cisplatin, are known to cause delayed nausea and vomiting, with symptoms occurring 1 to 5 days after drug administration. Some people suffer nausea and vomiting before drug administration apparently in anticipation of becoming sick. Acute nausea and vomiting is usually the most severe, whereas the course of delayed nausea and vomiting can be prolonged, leading to dehydration and poor nutrition.
The mechanisms responsible for nausea and vomiting are varied and not completely understood, and different drugs may cause nausea by utilizing different pathways. One mechanism, which has been described in animals for high-dose cisplatin, proposes that the drug causes the release of serotonin in the small intestine (perhaps because of mucosal damage), which then triggers afferent vagal nerves to the vomiting center of the brain.56 Research in this area has led to the development of antiemetics that block specific receptors. These agents may alleviate delayed nausea and vomiting.
Chemotherapy drugs cause the most damage to rapidly dividing cells. Although this is the means by which eradication of tumor cells occurs, these cytotoxins also affect cells that normally divide quickly, such as cells of the oral cavity and GI tract. Damage to the cells lining the GI tract result in the diarrhea so often seen after treatment. The cytotoxic effects of many chemotherapy drugs cause an inflammation of the mucosal epithelium in the GI tract. The resulting mucositis occurs when these injured cells are unable to replace themselves. This effect can occur anywhere in the GI tract and presents differently depending on where the damage occurs.73 Like nausea and vomiting, mucositis and diarrhea lead to dehydration and malnutrition.
Myelosuppression
Because many chemotherapeutic agents are not specific for cancer cells, they adversely affect normal cells, causing a number of serious side effects. Myelosuppression (inhibition of bone marrow production of blood cells and platelets resulting in anemia, leukopenia, and thrombocytopenia) is the most common and dose-limiting adverse effect of chemotherapy and can be the most lethal.101
Chemotherapeutic agents destroy the proliferating progenitor cells found in bone marrow that are destined to develop into mature blood cells. In doing so, these drugs limit the ability of the body to replace blood cells as they die or lose their effectiveness. Individuals receiving chemotherapy agents often develop some form of cytopenia, including anemia, leukocytopenia, and thrombocytopenia, 10 to 14 days after starting a course of chemotherapy. Withdrawal of the drug usually allows the progenitor cell populations to rebound and levels of blood cells to increase. High-dose chemotherapy may prolong the time needed to repopulate the bone marrow, leaving the person cytopenic for extended periods of time.
Prolonged or severe myelosuppression frequently delays scheduled treatments and increases the risk of serious infections, bleeding, or reduced endurance. With increased dosages or more frequent administration of drugs, more profound and prolonged myelosuppression may occur. However, the use of prophylactic antibiotics and colony-stimulating factors (granulocyte colony-stimulating factor [G-CSF] and/or granulocyte-macrophage colony–stimulating factor [GM-CSF]) has helped dramatically in the prevention of infection and control of myelosuppression.
Erythropoietin is often administered to increase RBC synthesis and reduce anemia; interleukin II is administered to reduce thrombocytopenia. Administration of blood products can also help alleviate adverse effects. Infection is the most serious adverse effect of myelosuppression (neutropenia) and is associated with significant morbidity and mortality.147 Individuals who develop neutropenic infections are treated with antibiotics and various colony-stimulating factors to stimulate the proliferation and differentiation of hematopoietic progenitor cells, ultimately increasing the numbers of mature WBCs.
Fatigue
It has been estimated that between 70% and 100% of all individuals with cancer will experience cancer-related fatigue (CRF).66 Symptoms of CRF include persistent sense of tiredness that is not relieved by rest, shortness of breath, decreased ability to focus or concentrate, and decreased ability to perform daily tasks.125 Although most people will experience fatigue during treatment (chemotherapy, postsurgery, or postradiation), upwards of 35% still experience fatigue 24 months after completing therapy.116
Fatigue often peaks within a few days for individuals receiving cyclic chemotherapy then declines until the next treatment cycle. Fatigue significantly reduces quality of life. It is generally agreed that fatigue has multiple cancer-related or treatment-induced causes that can be described as being either physiologic or psychologic. Physiologic causes of fatigue include underlying disease; cancer treatment; anemia; infection; accompanying pulmonary, hepatic, cardiac, and renal disorders; sleep disorders; poorly controlled pain; and malnutrition. Psychologic causes of fatigue include anxiety disorders, depressive disorders, and cognitive losses that include decreased attention span and concentration.137
Because CRF is multifactorial, multidimensional interventions involving both physical and psychologic components are required to successfully treat it. Such an approach has proved to be successful.174
Cardiotoxicity
All categories of chemotherapeutic agents can cause cardiac disease, but the anthracyclines are the most damaging to the heart. The anthracycline class of drugs, particularly doxorubicin (Adriamycin) and daunorubicin (DaunoXome), is well known for its increase risk of cardiomyopathy leading to irreversible CHF.191
Trastuzumab has been associated with a 2% risk of developing significant cardiac dysfunction, which increases to approximately 16% when this drug is used in combination with anthracyclines or cyclophosphamide (Cytoxan).154
Chemotherapy drugs known as antimetabolites (see Table 5-8), including the widely used agent 5-fluorouracil (5-FU), can produce ischemia and sequelae (e.g., heart attack) if not treated. Other nonchemotherapy agents used to treat cancer such as interleukin-2 or Gleevec are also linked to cardiotoxicity.
High-dose regimens and the total dose per course increase the likelihood of developing cardiac disease. Cessation of the drug will often decrease symptoms.191 Target therapies are also known to cause cardiotoxicity.
CHF and left ventricular failure with decreased endurance and other associated symptoms can occur early (within 6 to 12 months after initial treatment) or as late as 5 to 20 years after treatment. The risk for developing a cardiomyopathy increases as cumulative doses exceed 550 mg/m2. This factor often limits the amount of these drugs that can be given even if the tumor is responding to the drug. Currently, this can be at least partially avoided by adjusting the dose, using liposomal-based agents, or using a cardioprotective drug such as dexrazoxane.
Alkylating agents and antimetabolites are known to cause heart failure months to years after treatment, especially if the person taking these drugs also received radiation or an anthracycline. Acute cardiotoxicity is a greater adverse effect of these drugs, causing ischemic syndrome ranging from angina pectoris to myocardial infarctions.
The earliest signs of drug-induced cardiomyopathy include tachycardia and inability to return to baseline heart rate after exertion.158 There has also been a relationship noted between the use of 5-FU and the development of myocardial ischemia.171
Chemotherapeutic agents have proved very effective in reducing the mortality and morbidity from many forms of cancer. With chemotherapy increasing life expectancy, many people who have a history of cancer are just as much at risk for cardiac disease as they are from recurrent cancer.152
Pulmonary Toxicity
Pulmonary toxicity from chemotherapy is relatively uncommon but potentially fatal. Bleomycin, BCNU (carmustine), and mitomycin have the highest incidences of pulmonary toxicity, compared with other agents, causing a diffuse pneumonitis. For those clients with previous unrelated pulmonary disease, the development of pulmonary pneumonitis or fibrosis may be life-threatening.
Renal Toxicity
Many medications have renal toxicities, including cisplatin, methotrexate, and mitomycin. A careful evaluation of renal function must be performed before each course of chemotherapy. While the drugs themselves may be renal toxic, these agents also contribute to the occurrence of tumor lysis syndrome.
Tumor lysis syndrome occurs when cytotoxic drugs destroy malignant cells, releasing large amounts of metabolites and ions into the bloodstream. The kidneys are unable to tolerate the sudden load, leading to hyperkalemia, hyperuricemia, hypocalcemia, and uremia. This can be life-threatening, leading to cardiac dysrhythmias and renal failure. Clients who have renal insufficiency before treatment, large tumors, or rapidly dividing tumors that are sensitive to chemotherapy are at highest risk for this syndrome.
Hemorrhagic cystitis can occur in clients receiving cyclophosphamide or ifosfamide. The metabolite created from these agents (acrolein) irritates the bladder, causing bleeding. Mesna (2-mercaptoethanesulfonate) can be administered with the drug to detoxify this metabolite and reduce the risk of hemorrhagic cystitis.
Hepatic Toxicity
Acute or chronic hepatotoxicity is possible because many of the chemotherapeutic agents are metabolized in the liver. Drugs requiring dose modification for hepatitis include daunorubicin, doxorubicin, methotrexate, VP-16, vincristine, and vinblastine. Venoocclusive disease of the liver is seen in half of the bone marrow transplant clients, secondary to preconditioning chemotherapy, causing significant mortality.45
Neuropathies
Many chemotherapeutic agents adversely affect the nervous system either peripherally or systemically. The vinca-alkaloids, cisplatin, and the taxanes are particularly damaging to the peripheral nervous system, damaging the peripheral neurons and leading to a variety of defects (e.g., myalgia, distal symmetric sensory loss, motor weakness, foot drop, numbness, tingling, and muscle atrophy).
Symptoms can develop within hours after an infusion or may not appear for several weeks to months after treatment has stopped. Peripheral neuropathies tend to be directly related to the cumulative dose administered and the dose intensity. The appearance of a peripheral neuropathy often limits the total dose given and forces the discontinuation of a specific drug.
Signs and symptoms of a peripheral neuropathy tend to resolve with removal of the drug. Other factors that may influence the development of neuropathies include patient age, type, dose and frequency of chemotherapy, preexisting nerve condition, and nutritional status. Preexisting neuropathies and previous chemotherapy treatment are thought to make individuals more vulnerable to peripheral neuropathies.176
Central nervous neuropathies have also been described for chemotherapeutic agents capable of crossing the blood-brain barrier such as cisplatin, methotrexate, and cytarabine.176 Symptoms can appear quickly after receiving drugs and may include headaches, strokes, seizures, and aseptic encephalopathy.
Affected individuals may complain of noticeable short-term memory loss or difficulty concentrating classified as a mild cognitive impairment (MCI) but often referred to as “chemo brain” or “brain fog.” Chemotherapy-induced MCI occurs in up to 40% of patients.159,179 Many people using computers on the job report delays in their work output as they have to relearn and retrain themselves to do tasks that were automatic before cancer treatment.67
Symptoms are transient but may take several years to disappear.104 Of increasing concern is the observation that chemotherapy-induced CNS defects may take many months to years to develop. For example, 26% of long-term survivors of methotrexate treatment presented with neurologic complications, including both motor and cognitive deficits 68 months after treatment.18
Adverse Effects of Stem-Cell Transplantation
The use of high-dose chemotherapy with stem-cell transplantation (SCT) has increased as a treatment for malignant diseases such as leukemia and lymphomas. The individual may receive reinfusion of his or her own stem cells (autologous or autoSCT) or infusion of stem cells from a healthy donor (alloSCT).
SCT is preceded by an intensive course of high-dose chemotherapy and/or total body irradiation to destroy any residual malignant cells in the body. The process is called the conditioning regimen. The risk of infection is great during this phase before the “rescue” when tumor-free stem cells are introduced into the body.
Diminished functional capacity, poorer quality of life, and increased symptoms are typical immediately after the ablative regimen. Side effects of this treatment are those typical of transplantation, chemotherapy, and/or radiotherapy and include nausea, vomiting, anorexia (appetite and weight loss), mucositis, hemorrhagic cystitis, fluid and electrolyte imbalances, dermatologic reactions, renal failure, venoocclusive disease, and graft-versus-host disease.
SPECIFIC DISORDERS AFFECTING MULTIPLE SYSTEMS
Vasculitis is a term that applies to a diverse group of diseases characterized by inflammation in blood vessel walls. The primary forms of vasculitis encountered in a therapy practice include giant cell (temporal) arteritis, Takayasu’s arteritis, Behçet’s disease, polyarteritis nodosa, Wegener’s granulomatosis, and Kawasaki disease. These are discussed in greater detail in Chapter 12. Because the pathogenesis of most forms of vasculitis remains poorly understood and cases of vasculitis show great variability, it may not be possible to apply a specific disease label to such cases. Such instances of vasculitis may be diagnosed as systemic vasculitis.
Blood vessels of different sizes in various parts of the body may be affected by vasculitis, causing a wide spectrum of clinical manifestations. The inflammation often causes narrowing or occlusion of the vessel lumen and produces ischemia of the tissues that are supplied by the involved vessels. The inflammation may weaken the vessel wall, resulting in aneurysm or rupture. Large vessel disease often produces limb claudication, aortic dilation, and bruits. Vasculitis of the medium vessels causes cutaneous nodules (Fig. 5-6), gangrene of the digits, mononeuritis multiplex, and microaneurysms. Palpable purpura, glomerulonephritis, and alveolar hemorrhage can be seen in affected small vessels.
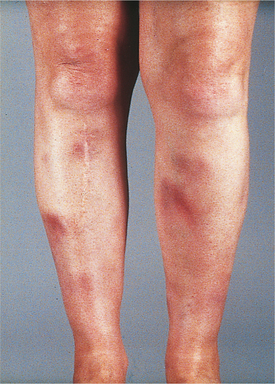
Figure 5-6 Nodular vasculitis caused by inflammation of the medium blood vessels. (From du Vivier A: Atlas of clinical dermatology, ed 2, London, 2002, Gower.) Gower
Most clients with vasculitis will exhibit constitutional symptoms such as fever, arthralgias, arthritis, weight loss, and malaise.161 Vasculitis may occur as a primary disease (as described previously); as a secondary manifestation of other illnesses such as RA, infection, malignancy, or serum sickness; or as a drug-induced illness.
Rheumatoid Arthritis
RA is best known as a progressive, autoimmune disease affecting the synovial tissue and joints. Yet RA has many extraarticular manifestations involving bone, muscle, eyes, lung, heart, and the skin. The most frequent skin manifestation is the rheumatoid nodule. These are most commonly found subcutaneously on extensor surfaces, such as the forearm, but have been noted on the heart, lung, sacrum, larynx, and leptomeninges.
RA involvement of the lungs and heart is multiple and varied, depending on the portion of the organ affected. Other extraarticular conditions that can occur with RA include vasculitis, anemia, and osteopenia/osteoporosis. Rheumatoid vasculitis has become much less frequent over the last decade, probably because of disease-modifying agents, yet it remains the most feared complication of RA, with considerable morbidity and mortality. Vasculitis is more common in men and usually develops in persons with the most significant active disease (deforming arthritis and high rheumatoid factor titers). (See the section on Collagen Vascular Disease in Chapter 12.)
Clinical features of systemic rheumatoid vasculitis are diverse because any size blood vessel may be involved anywhere in the body. The most common findings are cutaneous lesions such as nail-edge infarctions (e.g., splinter hemorrhages; see Fig. 27-14), purpura (see Fig. 5-1), and skin ulcers (e.g., pyoderma gangrenosum). Skin ulcers usually develop suddenly as deep, punched-out lesions at sites that are unusual for venous ulceration, such as the dorsum of the foot or the upper calf.
Neurologic manifestations of RA vasculitis present most commonly as either a mild distal sensory neuropathy (paresthesia or numbness) or as a severe sensorimotor neuropathy such as mononeuritis multiplex. These clients exhibit sensory symptoms with muscle weakness (e.g., foot drop). These may be the only extraarticular manifestations of RA.
Vasculitis may involve visceral organs such as the heart (causing a myocardial infarction) and lungs (leading to pulmonary hypertension). Infarction of the intestine may occur, requiring bowel resection.
Systemic manifestations of rheumatoid vasculitis may include unexplained weight loss, anorexia, and malaise. Malaise may be related to the release of cytokines (substances released by lymphocytes with various immunologic functions) and may be accompanied by fatigue, low-grade fever, and night sweats. Individuals with severe RA who experience any of these symptoms should be referred to the physician for further evaluation. Clients with multiple manifestations of vasculitis have a poor prognosis and require aggressive treatment.
Anemia secondary to RA is usually mild with normocytic/hypochromic features. Over three-fourths of people with RA and anemia have anemia of chronic disease, which is typically proportional to the disease severity and resolves as RA is brought under control. If iron deficiency is noted in an RA client, sources of bleeding must be explored such as GI bleeding from therapy with NSAIDs. Vitamin B12 and folate deficiency are also common in this population and should also be addressed. The therapist should follow special precautions related to anemia until the disease is under control (see Special Implications for the Therapist: Anemia in Chapter 14).
Osteopenia and osteoporosis may result from postmenopausal bone loss, treatment with glucocorticoids, or general immobility, but it may also be an inherent part of RA. Because most clients with RA may have all these risk factors for bone loss, they should be aggressively treated to reduce bone loss. With long-standing disease, osteoporosis may become generalized and can lead to fractures after minimal stress, particularly the fibula.
Systemic Lupus Erythematosus
Lupus erythematosus is an autoimmune disease that appears in two forms: discoid lupus erythematosus (DLE), which affects only the skin, and systemic lupus erythematosus (SLE), which affects both multiple organ systems and the skin and can be fatal. SLE most commonly causes rashes of the skin, polyarthritis, and myalgias. The most serious complications affect the heart, kidneys, and CNS. Like RA, SLE is characterized by recurring remissions and exacerbations, although complete remission is rare.
SLE affects women eight times as often as men, increasing to 15 times as often during childbearing years. (For further discussion of DLE see Chapter 10; see Chapter 7 for discussion of SLE.)
Systemic Sclerosis
Systemic sclerosis, also known as progressive systemic sclerosis (PSS) or scleroderma, is a generalized connective tissue disorder of unknown etiology characterized by thickening and fibrosis of the skin. It may also affect internal organs, namely the heart, lungs, GI tract, and kidneys.
Although there are many subgroups termed scleroderma, it is often categorized into two main subgroups: diffuse cutaneous scleroderma (skin involvement of the trunk, face, and proximal and distal extremities) and limited cutaneous scleroderma (involvement of the skin of the face and neck but distal to the elbow and knee). Limited cutaneous scleroderma was previously known as the CREST syndrome (Calcinosis, Raynaud’s phenomenon, Esophageal dysmotility, Sclerodactyly, and Telangiectasia). These subgroups help clinically prognosticate since individuals with diffuse cutaneous scleroderma are more likely to develop organ involvement earlier in the progression of the disease.
There is significant variability of symptoms and organ involvement between clients. It affects women more than men, especially between ages 30 and 50 years. Scleroderma has no current significant disease-modifying treatments and the morbidity and mortality are high. Approximately 30% of people with PSS die within 5 years of onset. (See Chapter 10 for discussion of this condition.)
Tuberculosis
Tuberculosis (TB) is an acute or chronic infection caused by Mycobacterium tuberculosis. Although the primary infection site is the lung, mycobacteria commonly exist in other parts of the body; this is referred to as extrapulmonary tuberculosis. The extrapulmonary sites may include the renal system, skeletal system (osteomyelitis; vertebral TB is known as Pott’s disease), GI tract, meninges (tuberculous meningitis), and genitals. Extrapulmonary tuberculosis occurs with increased frequency in people with HIV infection (see Chapter 15 for more on pulmonary tuberculosis; see Chapter 25 for more on tuberculous spondylitis [Pott’s disease]).
Sarcoidosis
Sarcoidosis is a multisystem disorder characterized by the formation of noncaseating granulomas, which are inflammatory cells (e.g., mononuclear inflammatory cells or macrophages) usually surrounded by a rim of lymphocytes. These granulomas may develop in any organ but often are noted in multiple organs at once, including the lungs, lymph nodes, liver, bones, or eyes (see Box 15-10) and may be accompanied by skin lesions (see Fig. 15-21). Presenting symptoms of sarcoidosis can often be confused with other inflammatory or infectious processes, making the diagnosis difficult. In the United States, sarcoidosis occurs predominantly among blacks and affects twice as many women as men.
Sarcoidosis is often referred to as either acute or chronic. Acute sarcoidosis is abrupt in onset, frequently involving the eyes and skin. Bell’s palsy may also be seen. Acute sarcoidosis is transient, often with a good prognosis and complete resolution of symptoms. Chronic sarcoidosis is typically more insidious in onset and occurring in older individuals. Fibrosis formation is prevalent, involving the heart, lung, kidneys, and bone. There is significant morbidity and mortality associated with chronic sarcoidosis, with recurrence of the disease despite treatment (see Chapter 15 for a complete discussion of this condition).
Multiple Organ Dysfunction Syndrome
Care of critically ill people has progressed significantly during the last 50 years. Substantial advances have been made in the care of shock, acute renal failure, acute brain injury, and acute respiratory failure, with more people surviving these conditions.
However, despite these advances, progressive deterioration of organ function may occur in people who are critically ill or injured. People often die of complications of disease, rather than from the disease itself. Multiple organ dysfunction syndrome (MODS) is often the final complication of a critical illness; it is one of the most common causes of death in the ICU.63
Definition and Etiologic and Risk Factors
MODS, also called multiple organ failure syndrome (MOFS), is the progressive failure (more than 24 hours) of two or more organ systems after a severe illness or injury. Although sepsis and septic shock are the most common causes,192 infection is not required for its development. MODS also can be triggered by ARDS, severe inflammatory processes (e.g., pancreatitis), other types of shock, and traumatic injury (e.g., burns or surgery). MODS carries a high mortality rate that increases with each organ that fails.
Systemic inflammatory response syndrome (SIRS) characterizes the clinical manifestations of hypermetabolism (e.g., increased temperature, heart rate, and respirations) present in many clients with MODS. Because it is a response to tissue insult or injury, SIRS is present in many individuals admitted to an ICU.
After an initial insult or injury, other factors can increase the risk of developing MODS/SIRS, including inadequate or delayed resuscitation, age over 65 years, alcoholism, diabetes, surgical complications (e.g., infection, hematoma formation), bowel infarction, or the previous existence of organ dysfunction (e.g., renal insufficiency).
Pathogenesis
Although MODS may be a final common pathway in critical illnesses, actual causes and cellular changes leading to MODS are not completely understood. Most likely multiple mechanisms and factors are responsible or contribute to the development of MODS. In response to illness or traumatic injury, the neuroendocrine system activates stress hormones (e.g., cortisol, epinephrine, norepinephrine, or endorphins) to be released into the circulation, whereas the sympathetic nervous system is stimulated to compensate for complications such as fluid loss and hypotension.
Because of the initial insult, proinflammatory cytokines (e.g., interferons and tumor necrosis factor) and enzymes are released with the overall effect of massive uncontrolled systemic immune and inflammatory responses. This hyperinflammation and hypercoagulation perpetuates edema formation, cardiovascular instability, endothelial damage, and clotting abnormalities.
At the same time, initial oxygen consumption demand increases because the oxygen requirements at the cellular level increase. Flow and oxygen consumption are mismatched because of a decrease in oxygen delivery to the cells caused by maldistribution of blood flow, myocardial depression, and a hypermetabolic state. The end result is abnormal cellular respiration and function (tissue hypoxia with cellular acidosis and death), resulting in the multiple organ dysfunction characteristic of MODS.127
Clinical Manifestations
A clinical pattern in the development of MODS has been well established. After the precipitating event, low-grade fever, tachycardia, dyspnea, SIRS, and altered mental status develop. The lung is the first organ to fail, resulting in ARDS (see Chapter 15).
Between 7 and 10 days, the hypermetabolic state intensifies, GI bacteremia is common, and signs of liver and kidney failure develop. During days 14 to 21, renal and liver failures progress to a severe status and the GI and immune systems fail, with eventual cardiovascular collapse. Ischemia and inflammation are responsible for the CNS manifestations. Protein metabolism is also affected, and amino acids derived from skeletal muscle, connective tissue, and intestinal viscera become an important energy source. The result is a significant loss of lean body mass.
MEDICAL MANAGEMENT
Prevention and early detection and supportive therapy are essential for MODS, as no specific medical treatment exists for this condition. A way to halt the process, once it has begun, has not yet been discovered. Pharmacologic treatment may include antibiotics to treat infection, inotropic agents (e.g., dopamine or dobutamine) to counteract myocardial depression, and supplemental oxygen and ventilation to keep oxygen saturation levels at or above 90%.
Fluid replacement and nutritional support are also provided. The recent development of monoclonal antibodies to modulate or inhibit the immune and inflammatory responses may lead to more specific pharmacologic treatment.97
MODS is the major cause of death (usually occurring between days 21 and 28) after septic, traumatic, and burn injuries. If the affected individual’s condition has not improved by the end of the third week, survival is unlikely. The mortality rate of MODS is 60% to 90% and approaches 100% if three or more organs are involved, sepsis is present, and the individual is older than 65 years.
FLUID AND ELECTROLYTE IMBALANCES
Observing clinical manifestations of fluid or electrolyte imbalances may be an important aspect of client care, especially in the acute care and home health care settings. Identifying clients at risk for such imbalances is the first step toward early detection.
The causes of fluid and electrolyte imbalance are many and varied and include disease processes, injury, medications, medical treatment, dietary restrictions, and imbalance of fluid intake with fluid output.169 The most common causes of fluid and electrolyte imbalances in a therapy practice include burns, surgery, diabetes mellitus, malignancy, alcoholism, and the various factors affecting the aging adult population (Box 5-7).
This is a brief presentation of the normal homeostatic processes of fluid and electrolyte balance. The interactions of these systems and how they maintain fluid and electrolyte balance and acid-base regulation are beyond the scope of this text. For a more in-depth study of these concepts, the reader is referred to the latest edition of Guyton AC, Hall JE: Textbook of medical physiology, Philadelphia, WB Saunders.
Aging and Fluid and Electrolyte Balance
The volume and distribution of body fluids composed of water, electrolytes, and nonelectrolytes vary with age, gender, body weight, and amount of adipose tissue. Throughout life, a slow decline occurs in lean body or fat-free mass with a corresponding decline in the volume of body fluids. Only 45% to 50% of the body weight of aging adults is water compared with 55% to 60% in younger adults. This decrease represents a net loss of muscle mass and a reduced ratio of lean body weight to total body weight and places older people at greater risk for water-deficit states.
There are also changes in the kidney that further potentiate the risk for fluid and electrolyte disturbances. With increasing age, there is a decrease in renal mass and glomerular filtration rate (GFR). This in turn may lead to the inability of the aging kidney to excrete free water in the face of fluid excess, causing hyponatremia.
Yet hypernatremia can also be problematic in the aging adult secondary to a defect in the ability of the kidney to concentrate urine combined with a decreased thirst despite dehydration, often seen with age. Although these changes are seen in normal aging, factors that depress the sensorium in the frail and sick elderly (stroke and medications) further complicate hypernatremia by suppressing the natural compensatory mechanism for fluid intake.
Infection, dementia, neurologic disorders, and other systemic illnesses can decrease the release of arginine vasopressin (AVP), further placing older adults at high risk for dehydration.25 Renin and aldosterone decrease with age accompanied by a blunted response to aldosterone. These changes can lead to hyperkalemia, particularly if other factors are present such as the use of potassium-sparing diuretics.
Fluid Imbalances
Approximately 45% to 60% of the adult human body is composed of water, which contains the electrolytes that are essential to human life (see the section on Electrolytes in this chapter). This life-sustaining fluid is found within various body compartments, including the intracellular (within cells), interstitial (space between cells), intravascular (within blood vessels), and transcellular compartments.
Fluid in the transcellular compartment is present in the body but is separated from body tissues by a layer of epithelial cells. This fluid includes digestive juices, water, and solutes in the renal tubules and bladder, intraocular fluid, joint-space fluid, and cerebrospinal fluid. The fluid in the interstitial and intravascular compartments comprises approximately one-third the total body fluid, called the extracellular fluid (ECF). Fluid found inside the cells accounts for the remaining two-thirds of total body fluid, called the intracellular fluid (ICF).
The cell membrane is water permeable with equal concentrations of dissolved particles on each side of the membrane maintaining equal volumes of ECF and ICF and preventing passive shifts of water. Passive shifts occur only if an inequality occurs on either side of the membrane in the concentration of solutes that cannot permeate the membrane. For example, water will move from one compartment to another if there is a change in sodium ion concentration.
The following five types of fluid imbalances may occur:
A simpler approach to this subject is to view fluid shifts in terms of intravascular or extravascular movement. Movement from the vascular space to the extravascular areas and vice versa takes place easily and is the first mechanism of extracellular movement.
Increased intravascular fluid results in CHF and increased pulse and respiration. Decreased intravascular fluid results in decreased blood pressure and increased pulse and respirations. However, increased extravascular fluid may cause edema, ascites, or pleural effusion. Decreased extravascular fluid results in decreased skin turgor and fatigue. The material in this section is presented on the bases of three broad categories: fluid deficit, fluid excess, and fluid shift (see Chapter 13).
Etiologic Factors and Pathogenesis
Maintaining constant internal conditions (homeostasis) requires the proper balance between the volume and distribution of ECF and ICF to provide nutrition to the cells, allow excretion of waste products, and promote production of energy and other cell functions. Maintenance of this balance depends on the differences in the concentrations of ICF and ECF fluids, the permeability of the membranes, and the effect of the electrolytes in the fluids.
A fluid imbalance occurs when either the ICF or ECF gains or loses body fluids or electrolytes, causing a fluid deficit or a fluid excess. Sodium is the major ion that influences water retention and water loss. A deficit of body fluids occurs with either an excessive loss of body water or an inadequate compensatory intake. The result is an insufficient fluid volume to meet the needs of the cells. It is manifested by dehydration (Box 5-8); hypovolemia, such as blood or plasma loss; or both. Severe fluid volume deficit can cause vascular collapse and shock.
An excess of water occurs when an overabundance of water is in the interstitial fluid spaces or body cavities (edema) or within the blood vessels (hypervolemia). A fluid shift occurs when vascular fluid moves to interstitial or intracellular spaces or interstitial or intracellular fluid moves to vascular fluid space.
Fluid that shifts into the interstitial space (i.e., fluid not in the vascular compartment) and remains there is referred to as third-space fluid. Third-space fluid is commonly seen in a therapy practice as a result of altered capillary permeability secondary to tissue injury or inflammation, but the most common cause is liver disease. Decreased serum protein (albumin) associated with liver disease and/or states of malnutrition results in third-space fluid.
Other areas called potential spaces can fill with fluid in the presence of inflammation or fluid imbalances. Examples of potential spaces include the peritoneal cavity fluid (e.g., ascites) and the pleural cavity (e.g., pleural effusion).
Clinical Manifestations
Fluid volume deficit (FVD) is most often accompanied by symptoms related to a decrease in cardiac output such as decreased blood pressure, increased pulse, and orthostatic hypotension. FVD can occur from loss of blood (whether obvious hemorrhage or occult GI bleeding), loss of plasma (burns or peritonitis), or loss of body fluids (diarrhea, vomiting, diaphoresis, or lack of fluid intake), resulting in dehydration.
Hypernatremia occurs if the body fluid loss is a loss of body water without solute components (e.g., diabetes insipidus). Most often, however, body fluid losses contain both body water and its solute components. The affected individual experiences symptoms of thirst, weakness, dizziness, decreased urine output, weight loss, and altered levels of consciousness (i.e., confusion). Significant decreases in systolic blood pressure (less than 70 mm Hg) result in symptoms of shock and require immediate medical treatment and possibly life-sustaining emergency management.
Fluid volume excess (FVE) is primarily characterized by weight gain and edema of the extremities. With intravascular FVE, other clinical manifestations include dyspnea, engorged neck veins, and a bounding pulse. In the early stages, if the fluid is in the third space (interstitial fluid between cells), the person may not exhibit any of these symptoms.
Fluid shift from the vascular to the extravascular (interstitial) spaces (e.g., burns or peritonitis) is manifested by signs and symptoms similar to fluid volume deficit and shock, including skin pallor, cool extremities, weak and rapid pulse, hypotension, oliguria, and decreased levels of consciousness. When the fluid returns to the blood vessels, the clinical manifestations are similar to those of fluid overload such as bounding pulse and engorgement of peripheral and jugular veins.
MEDICAL MANAGEMENT
The ECF is the only fluid compartment that can be readily monitored; clinically, the status of ICF is inferred from analysis of plasma and the condition of the person. A fluid balance record is kept on any individual who is susceptible or already experiencing a disturbance in the balance of body fluids. In addition, medical evaluation of clinical signs and laboratory tests are helpful in the assessment of a person’s hydration status. Laboratory tests may include serum osmolality, sodium, hematocrit, and BUN measurements (see the section on Laboratory Values in Chapter 40).
Serum osmolality measures the concentration of particles in the plasma portion of the blood. Osmolality increases with dehydration and decreases with overhydration. Serum sodium is an index of water deficit or excess; an elevated level of sodium in the blood (hypernatremia) would indicate that the loss of water from the body has exceeded the loss of sodium such as occurs in the administration of osmotic diuretics, uncontrolled diabetes insipidus, and extensive burns. Hematocrit increases with dehydration and decreases with excess fluid. BUN serves as an index of kidney excretory function; BUN increases with dehydration and decreases with overhydration (see Table 40-2).
Treatment is directed to the underlying cause; in the case of FVD, the aim is to improve hydration status. This may be accomplished through replacement of fluids and/or electrolytes by oral, nasogastric, or IV means.
Electrolyte Imbalances
Electrolytes are chemical substances that separate into electrically charged particles, called ions, in solution. The electrolytes that consist of positively charged ions, or cations, are sodium (Na+), potassium (K+), calcium (Ca2+), and magnesium (Mg2+). Those that consist of negatively charged ions, or anions, are chloride (Cl+); bicarbonate (HCO3−); and phosphate (PO43−).
Concentration gradients of sodium and potassium across the cell membrane produce the membrane potential and provide the means by which electrochemical impulses are transmitted in nerve and muscle fibers. Sodium affects the osmolality of blood and therefore influences blood volume and pressure and the retention or loss of interstitial fluid. Sodium imbalance affects the osmolality of the ECF and is often associated with fluid volume imbalances.
Adequate potassium is necessary to maintain function of sodium-potassium membrane pumps, which are essential for the normal muscle contraction-relaxation sequence. Imbalances in potassium affect muscular activities, notably those of the heart, intestines, and respiratory tract, and neural stimulation of the skeletal muscles.
Calcium influences the permeability of cell membranes and thereby regulates neuromuscular activity. Calcium plays a role in the electrical excitation of cardiac cells and in the mechanical contraction of the myocardial and vascular smooth muscle cells. An imbalance in calcium concentrations affects skeletal muscle, bones, kidneys, and the GI tract. Conditions that can cause movement of calcium from the bones into the ECF (e.g., bone tumors, multiple fractures, or osteoporosis) can cause hypercalcemia. (Other causes of hypocalcemia or hypercalcemia are listed in Table 5-9).
Table 5-9
Causes of Electrolyte Imbalances



NSAIDs, Nonsteroidal antiinflammatory drugs; ACE, angiotensin-converting enzyme; GI, gastrointestinal; IV, intravenous; SIADH, syndrome of inappropriate antidiuretic hormone; ADH, antidiuretic hormone; CVA, cerebrovascular accident; PTH, parathyroid hormone; MEN, multiple endocrine neoplasia; AT N, acute tubular necrosis.
Modified from Horne M, Bond E: Fluid, electrolyte, and acid-base imbalances. In Lewis S, Heitkemper M, Dirksen S, eds: Medical-surgical nursing: assessment and management of clinical problems, ed 5, St Louis, 2000, Mosby.
Magnesium, an important intracellular enzyme activator, exerts physiologic effects on the nervous system that resemble the effects of calcium. Magnesium plays a role in maintaining the correct level of electrical excitability in the nerves and muscle cells by acting directly on the myoneural junction. Magnesium depresses acetylcholine release at synaptic junctions; when concentrations of Mg2+ in the plasma are altered, then associated changes also occur in the concentration of Ca2+. Magnesium imbalances affect parathyroid hormone (PTH) function and if severe, can change or impair end-organ response to PTH.
Neuromuscular irritability results from hypomagnesemia (e.g., poor diet, chronic alcohol abuse, diuretic use, or prolonged diarrhea), and magnesium excess (rare but occurs with renal failure or the overuse of magnesium-containing antacids) causes neuromuscular depression affecting the musculoskeletal and cardiac systems.123
Etiologic and Risk Factors
An electrolyte imbalance exists when the serum concentration of an electrolyte is either too high or too low. Stability of the electrolyte balance depends on adequate intake of water and the electrolytes and on homeostatic mechanisms within the body that regulate the absorption, distribution, and excretion of water and its dissolved particles.
Bodily fluid loss associated with weight loss, excessive perspiration, or chronic vomiting and diarrhea are the most common causes of electrolyte imbalance. Many other conditions can interfere with these processes and result in an imbalance (see Table 5-9).
Ischemia is accompanied by electrolyte disturbances, particularly the release of potassium, calcium, and magnesium from cells when cellular death ensues (see the section on Cell Injury in Chapter 6). Myocardial cells deprived of necessary oxygen and nutrients lose contractility, thereby diminishing the pumping ability of the heart. Diuretics also can produce mild-to-severe electrolyte imbalance. These factors explain the careful observation of specific electrolyte levels in the cardiac client.
Clinical Manifestations
In a therapy practice, paresthesias, muscle weakness, muscle wasting, muscle tetany, and bone pain are the most likely symptoms first observed with electrolyte imbalances (Table 5-10). (See the Clinical Manifestations section under Common Causes of Fluid and Electrolyte Imbalances in this chapter.)
MEDICAL MANAGEMENT
Potassium, calcium, sodium, and chloride can be measured in plasma. Intracellular levels of electrolytes cannot be measured; therefore all values for electrolytes are expressed as serum values. Serum values for electrolytes are given as milliequivalents per liter (mEq/L) or milligrams per deciliter (mg/dl) (see Table 40-2). As with fluid imbalances, the underlying cause of electrolyte imbalances must be determined and corrected. Electrolyte supplementation, when needed, can be given orally or intravenously.
Common Causes of Fluid and Electrolyte Imbalances
The exact mechanisms of fluid and electrolyte imbalances are outside the scope of this text. A brief description of the common causes and overall clinical picture encountered in a therapy practice is included here. Burns, surgery, and trauma may result in a fluid volume shift from the vascular spaces to the interstitial spaces. Tissue injury causes the release of histamine and bradykinin, which increases capillary permeability, allowing fluid, protein, and other solutes to shift into the interstitial spaces.
In the case of burns, the fluid shifts out of the vessels into the injured tissue spaces, as well as into the normal (unburned) tissue. This causes severe swelling of these tissues and a significant loss of fluid volume from the vascular space, which results in hypovolemia. Severe hypovolemia can result in shock, vascular collapse, and death. In the case of major tissue damage, potassium is also released from the damaged tissue cells and can enter the vascular fluids, causing hyperkalemia.
In an attempt to treat shock, large quantities of fluid are administered intravenously to maintain blood pressure, cardiac output, and renal function. After 24 to 72 hours, capillary permeability is usually restored and fluid begins to leave the tissue spaces and shift back into the vascular space. If renal function is not adequate, the accumulation of fluid used for treatment and fluid returning from the tissue spaces into the vascular space can cause fluid volume overload. Fluid overload can then cause CHF or pulmonary edema.
Diabetes mellitus (type 1) may result in a condition called diabetic ketoacidosis, which is caused by an overproduction of ketones and the accompanying metabolic acidosis that occurs (see Chapter 11). As the pH of the blood decreases (acidosis), the accumulating hydrogen moves from the ECF to the ICF. Movement of hydrogen into the cells promotes the movement of potassium out of the cells and into the ECF. As the potassium enters the vascular space, the plasma potassium levels increase. However, since significant diuresis is also occurring, the accumulated potassium is quickly excreted in the urine. As a result, severe potassium losses occur (hypokalemia), which unless treated immediately, cause life-threatening cardiac dysrhythmias.
Tumors often produce peptides that can affect fluid and electrolyte balance. These peptides cause neurologic, hormonal, dermatologic, and hematologic symptoms or syndromes often referred to as paraneoplastic syndromes (see Chapter 9). The peptides released by tumors are not regulated by normal suppression feedback loops; consequently, the ectopic hormone continues to be released by the tumor, often causing serious electrolyte imbalances. An ectopic hormone arises at or is produced at an abnormal site or in a tissue where it is not normally found. One example of this phenomenon is the ectopic production of antidiuretic hormone (ADH) by lung carcinomas, resulting in hyponatremia.
A more local effect of malignancy occurs when metastases to the skeletal system produce hypercalcemia from the osteolysis of bone. The treatment of malignancies also can create fluid and electrolyte imbalances such as occurs with hormonal treatment for breast cancer (e.g., tamoxifen can cause hypercalcemia). Hyponatremia and hypokalemia may also result from nausea and vomiting caused by chemotherapy. Certain chemotherapeutic drugs (e.g., vincristine and cyclophosphamide) are associated with the syndrome of inappropriate antidiuretic hormone (SIADH), causing hyponatremia.
Hyponatremia is also the most common electrolyte imbalance affecting hospitalized patients. Causes of hyponatremia in this population group include sodium loss from diuretics, vomiting, or wound draining and water gain if the person receives too much of a hypotonic IV fluid.
Alcohol withdrawal and eating disorders are also associated with physiologic changes that can include electrolyte imbalances. See discussion of each individual condition in Chapter 3.
Clinical Manifestations
The effects of a fluid or electrolyte imbalance are not isolated to a particular organ or system (Box 5-9). Symptoms most commonly observed by the therapist may include skin changes, neuromuscular irritability (muscle fatigue, twitching, cramping, or tetany), CNS involvement, edema, and changes in vital signs, especially tachycardia and postural (orthostatic) hypotension (see Box 12-11).
Skin changes include changes in skin turgor and alterations in skin temperature. In a healthy individual, pinched skin will immediately fall back to its normal position when released, a measure of skin turgor. In a person with FVD, such as dehydration, the skin flattens more slowly after the pinch is released and may even remain elevated for several seconds, referred to as tenting of tissue (Fig. 5-7). Tissue turgor can vary with age, nutritional state, race, and complexion and must be accompanied by other signs of FVD to be considered meaningful.
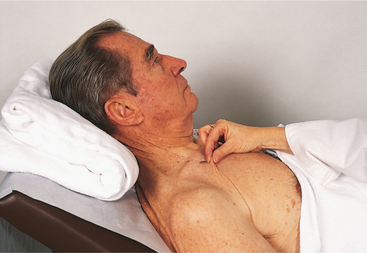
Figure 5-7 Testing skin turgor (normal resiliency of a pinched fold of skin). Turgor is measured by the time it takes for the skin and underlying tissue to return to its original contour after being pinched up. If the skin remains elevated (i.e., tented) for more than 3 seconds, turgor is decreased. Normal turgor is indicated by a return to baseline contour within 3 seconds when the skin is mobile and elastic. Turgor decreases with age as the skin loses elasticity; testing turgor of some older persons on the forearm (the standard site for testing) is less valid because of decreased skin elasticity in this area. (From Jarvis C: Physical examination and health assessment, ed 4, Philadelphia, 2004, WB Saunders.)
Skin turgor may be more difficult to assess in older adults because of reduced skin elasticity compared with that of younger clients. Skin temperature may become warm and flushed as a result of vasodilation (e.g., in metabolic acidosis) or pale and cool because of peripheral vasoconstriction compensating for hypovolemia.
Neuromuscular irritability can occur as a result of imbalances in calcium, magnesium, potassium, and sodium. (See Chapter 24 for discussion of osteoporosis associated with calcium loss.) Specific signs of neuromuscular involvement associated with these imbalances occur because of increased neural excitability, specifically increased acetylcholine action at the nerve ending, resulting in lowering of the threshold of the muscle membrane.
Tetany (continuous muscle spasm) is the most characteristic manifestation of hypocalcemia. The affected person may report a sensation of tingling around the mouth (circumoral paresthesia) and in the hands and feet, and spasms of the muscles of the extremities and face. Less overt signs (latent tetany) can be elicited through Trousseau’s sign (Fig. 5-8); Chvostek’s sign (Fig. 5-9); and changes in deep tendon reflexes (DTRs) (Table 5-11). Many other factors can produce abnormalities in DTRs requiring the therapist to evaluate altered DTRs in light of other clinical signs and client history.
Table 5-11
Changes in Deep Tendon Reflexes Associated With Fluid/Electrolyte Imbalance
| Increased (Hyperactive) | Decreased (Hypoactive) |
| Hypocalcemia | Hypercalcemia |
| Hypomagnesemia | Hypermagnesemia |
| Hypernatremia | Hyponatremia |
| Hyperkalemia* | Hyperkalemia |
| Alkalosis | Acidosis |
*Generally hyperkalemia is accompanied by decreased or absent deep tendon reflexes (DTRs); some sources do report hyperactive DTRs with hyperkalemia. In the clinical situation, DTRs are never used to determine a potassium imbalance. They are a warning sign for the therapist to assess the client further and report all pertinent findings.
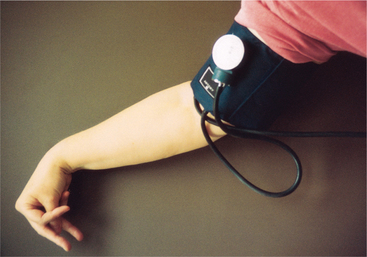
Figure 5-8 Carpopedal attitude of the hand, a form of latent tetany associated with hypocalcemia, is called Trousseau’s sign. This can be tested for by inflating a blood pressure cuff on the upper arm to a level between diastolic and systolic blood pressure and maintaining this inflation for 3 minutes. A positive test results in the carpal spasm shown here. (From Ignatavicius DD, Workman ML: Medical-surgical nursing: critical thinking for collaborative care, ed 5, Philadelphia, 2006, WB Saunders.)

Figure 5-9 To check for Chvostek’s sign, tap the facial nerve above the mandibular angle, adjacent to the ear lobe. A facial muscle spasm that causes the person’s eye and upper lip to twitch, as shown, confirms tetany. (From Ignatavicius DD, Workman ML: Medical-surgical nursing: critical thinking for collaborative care, ed 5, Philadelphia, 2006, WB Saunders.)
Nervous system involvement may occur in the peripheral system (hyperkalemia) or the CNS (hypocalcemia, hypercalcemia, hyponatremia, and hypernatremia). CNS manifestations of hypocalcemia may include seizures, irritability, depression, memory impairment, delusions, and hallucinations. In chronic hypocalcemia, the skin may be dry and scaling, the nails become brittle, and the hair is dry and falls out easily.
Signs and symptoms of hyponatremia occur when a drop in the serum sodium level pulls water into cells. When this happens, the client may experience headaches, confusion, lethargy, muscle weakness, and nausea. These symptoms are easily mistaken for complications from anesthesia or analgesia.
Hypokalemia seen in a therapy practice can be associated with diuretic therapy; excessive sweating, vomiting, or diarrhea; diabetic acidosis; trauma; or burns. It is accompanied by muscular weakness that can progress to flaccid quadriparesis. The weakness is initially most prominent in the legs, especially the quadriceps; it extends to the arms, with involvement of the respiratory muscles soon after.123 Severe hypokalemia can cause paralysis, respiratory failure, cardiac arrhythmias, and hypotension. Finally, a condition called rhabdomyolysis (disintegration of striated muscle fibers with excretion of myoglobin in the urine) can occur with potassium or phosphorus depletion.
Edema, defined as an excessive accumulation of interstitial fluid (fluid that bathes the cells), may be either localized or generalized. Generalized edema may be characterized by shortness of breath, ankle swelling, nocturia, and orthopnea. Other manifestations of generalized edema may include decreased urinary output; weight gain; labored, shallow, and increased respiratory rate; distended neck veins at 45-degree elevation of the head; changes in blood pressure; and abnormal laboratory findings (e.g., electrolytes, serum creatinine, BUN, and hemoglobin).
Pulmonary edema results from excessive shifting of fluid from the vascular space into the pulmonary interstitium and air spaces. When edema forms secondary to fluid retention, the clinical picture is usually one of pitting edema (Fig. 5-10).
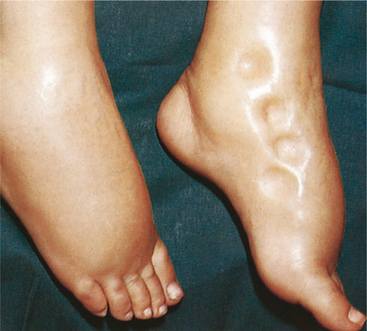
Figure 5-10 Severe, dependent, pitting edema occurs with some systemic diseases, such as congestive heart failure and hepatic cirrhosis. Note the finger-shaped depressions that do not refill after pressure has been exerted by the examiner. (From Thidodeau GA, Patton KT: The human body in health and disease, ed 4, St. Louis, 2005, Mosby.)
Vital sign changes, including pulse, respirations, and blood pressure, may signal early development of fluid volume changes. Decreased blood pressure and tachycardia are usually the first signs of the decreased vascular volume associated with FVD as the heart pumps faster to compensate for the decreased plasma volume. Irregular pulse rates and dysrhythmias may also be associated with magnesium, potassium, or calcium imbalances.
Orthostatic hypotension is another sign of volume depletion (hypovolemia). Moving from a supine to standing position causes an abrupt drop in venous return, which is normally compensated for by sympathetically mediated cardiovascular adjustments. For example, in the healthy individual, increased peripheral resistance and increased heart rate maintain cardiac output. Blood pressure is unaffected or characterized by a small decrease in systolic pressure, and the diastolic pressure may actually rise a few millimeters (mm) of mercury.
In contrast, for the person with FVD, systolic pressure may fall 20 mm Hg or more, accompanied by an increase in the pulse rate greater than 15 beats/minute.85 The decreased volume results in compensatory increases in pulse rate as the heart attempts to increase output in the face of decreased stroke volume.
As fluid volume depletion worsens, blood pressure becomes low in all positions due to loss of compensatory mechanisms and autonomic insufficiency. Conditions such as diabetes, associated with autonomic neuropathy, can also produce orthostatic blood pressure and pulse changes (see the section on Orthostatic Hypotension in Chapter 12).
ACID-BASE IMBALANCES
Overview
Normal function of body cells depends on regulation of hydrogen ion concentration (H+) so that H+ levels remain within very narrow limits. Acid-base imbalances occur when these limits are exceeded and are recognized clinically as abnormalities of serum pH (i.e., the measure of acidity or alkalinity of blood). Normal serum pH is 7.35 to 7.45. Cell function is seriously impaired when pH falls to 7.2 or lower or rises to 7.55 or higher (see the section on Laboratory Values in Chapter 40).
Three physiologic systems act interdependently to maintain normal serum pH: immediate buffering of excess acid or base by the blood buffer systems, excretion of acid by the lungs (occurs within hours), and excretion of acid or reclamation of base by the kidneys (occurs within days). The four general classes of acid-base imbalance are respiratory acidosis, respiratory alkalosis, metabolic acidosis, and metabolic alkalosis. Table 5-13 summarizes these four imbalances (see Table 40-3).
Table 5-13
Overview of Acid-Base Imbalances
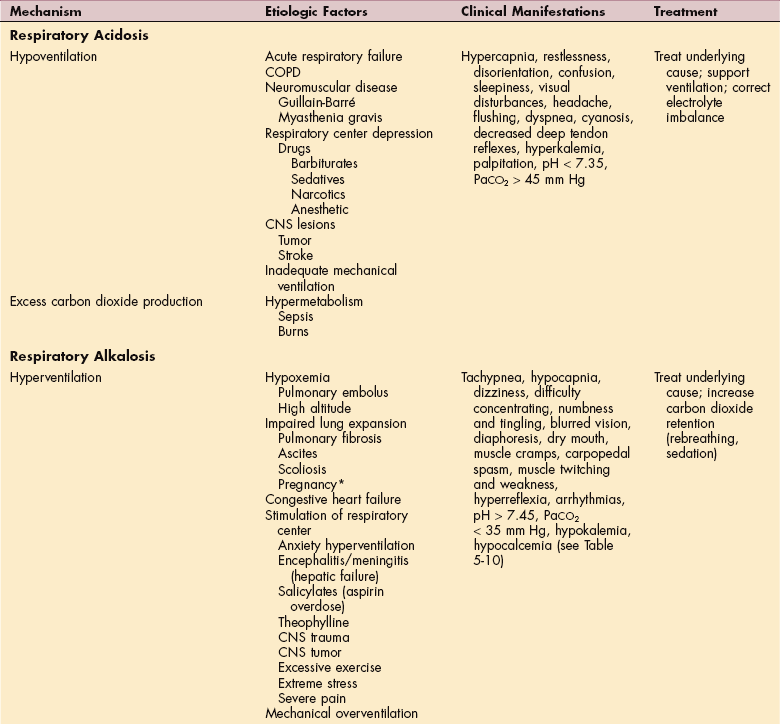


COPD, Chronic obstructive pulmonary disease; CNS, central nervous system; ARDS, adult respiratory distress syndrome; PaCO2, partial pressure of carbon dioxide; NaCO3, sodium bicarbonate; HCO3−, bicarbonate ion.
*In the third trimester of pregnancy, the hormone progesterone also stimulates respiration.
Acidosis refers to any pathologic process causing a relative excess of acid in the body. This can occur as a result of accumulation of acid or depletion of the alkaline reserve (bicarbonate content, HCO3−) in the blood and body tissues.
Acidemia refers to excess acid in the blood and does not necessarily confirm an underlying pathologic process. The same distinction may be made between the terms alkalosis and alkalemia; alkalosis indicates a primary condition resulting in excess base in the body. Although efforts have been made to standardize acid-base terminology, these terms are often used interchangeably.
Incidence
The incidence of acid-base imbalances in hospital settings is high. Acid-base imbalances are often related to respiratory and/or metabolic problems typical of the critically ill or injured individual. Some people have more than one acid-base imbalance at the same time.
Clinical Manifestations
A guide to the clinical presentation of acid-base imbalances is shown in Table 5-13. Besides the major distinguishing characteristics of acid-base imbalance described in this chapter, potassium excess (hyperkalemia) is associated with both respiratory and metabolic acidosis, and neuromuscular hyperexcitability is associated with both respiratory and metabolic alkalosis.35
MEDICAL MANAGEMENT
Pulse oximetry is used most often to measure oxygen saturation, yet it does not provide needed information regarding the effectiveness of ventilation or the pH of the blood. A more comprehensive procedure is the arterial blood gas (ABG) test (see Table 40-18).
This measurement is important in the diagnosis and treatment of ventilation, oxygen transport, and acid-base problems. The test measures the amount of dissolved oxygen and carbon dioxide in arterial blood and indicates acid-bases status by measurement of the arterial blood pH. The pH is inversely proportional to the hydrogen ion concentration (H+) in the blood. Therefore, as the hydrogen ion concentration (H+) increases (acidosis), the pH decreases; as the hydrogen ion concentration (H+) decreases (alkalosis), the pH increases.
The PCO2 is a measure of the partial pressure of carbon dioxide in the blood. PCO2 is termed the respiratory component in acid-base measurement because the carbon dioxide level is primarily controlled by the lungs. As the carbon dioxide level increases, the pH decreases (respiratory acidosis); as the carbon dioxide level decreases, the pH increases (respiratory alkalosis).
TREATMENT.
Treatment in acid-base imbalances is directed toward the underlying cause and correction of any coexisting electrolyte imbalance. For example, respiratory infections contributing to ventilatory failure (respiratory acidosis) are managed with appropriate antibiotic therapy, pulmonary hygiene, oxygen support, and possibly, continuous mechanical ventilation. Use of pharmaceutical agents that depress the respiratory control center is minimized. Dialysis may be indicated in renal failure (metabolic acidosis) or overdose of toxins.
Respiratory Acidosis
Respiratory acidosis is nearly always due to hypoventilation and subsequent retention of carbon dioxide (CO2). In a therapy setting, respiratory acidosis is most commonly observed in the population with COPD or asthma, depressed CNS (drugs, infection, or brain injury), or whenever the diaphragm is impaired (e.g., Guillain-Barré syndrome, myasthenia gravis, or chest wall deformities); secondary to burns; and as a result of lesions of the CNS (e.g., tumor, stroke, or muscular dystrophy).
The respiratory system has an important role in maintaining acid-base equilibrium. In response to an increase in the hydrogen ion concentration in body fluids, the respiratory rate increases, causing more CO2 to be released from the lung.
Anything that impairs this CO2 exhalation causes the CO2 to accumulate in the blood, where it unites with water to form carbonic acid (H2CO3), decreasing the blood pH. In addition, the kidneys begin to excrete more acid and retain more bicarbonate to further correct the acid imbalance.
Respiratory acidosis can be acute because of a sudden failure in ventilation, or chronic, as with long-term pulmonary disease (e.g., COPD). In the acute episode, the blood buffer systems cannot compensate to restore the acid-base balance because normal blood circulation and tissue perfusion are impaired. The lungs may not be functioning properly and the kidneys require more time to compensate than the acute condition permits.
Chronic respiratory acidosis results from gradual and irreversible loss of ventilatory function. Although there is increased retention of CO2, the kidneys have time to compensate by retaining bicarbonate and thereby maintaining a pH within tolerable limits. However, if even a minor respiratory infection develops, the person is subjected to a rapidly developing state of acute acidosis because the lungs remove only a limited amount of carbon dioxide.
Clinical Manifestations
Acute respiratory acidosis produces CNS disturbances. Effects range from restlessness, confusion, and apprehension to somnolence (sleepiness), with a fine or flapping tremor (see the section on Asterixis in Chapter 17) or coma. The person may report headaches and shortness of breath with retraction and use of accessory muscles. On examination, DTRs may be depressed. This disorder may also cause cardiovascular abnormalities such as tachycardia, hypertension, atrial and ventricular arrhythmias, and in severe acidosis, hypotension with vasodilation.
Respiratory Alkalosis
Respiratory alkalosis, the opposite of respiratory acidosis, occurs as a result of a loss of acid without compensation and most commonly when the lungs excrete excessive amounts of carbon dioxide (hyperventilation).
Conditions associated with respiratory alkalosis fall into the following two categories:
1. Pulmonary, caused by hypoxemia in early stage pulmonary problems (e.g., pulmonary edema, pulmonary embolism, pneumonia, and acute asthma) and by overuse of a mechanical ventilator
2. Nonpulmonary, which includes anxiety, hysteria, pain, fever, high environmental temperature, pregnancy, drug toxicities (salicylates, theophylline), CNS disease (brainstem tumors, infection), and hyperthyroidism (see Table 5-13).
Hyperventilation and the subsequent respiratory alkalosis is a common finding in ICU patients.
Clinical Manifestations
The cardinal sign of respiratory alkalosis is deep, rapid breathing, possibly exceeding 40 breaths/minute (much like the Kussmaul’s respirations that characterize diabetic acidosis) (see Table 5-15). Such hyperventilation usually leads to CNS and neuromuscular disturbances such as dizziness or light-headedness (caused by below-normal CO2 levels that decrease cerebral blood flow); inability to concentrate; tingling and numbness of the extremities and around the mouth; blurred vision; diaphoresis; dry mouth; muscle cramps; carpopedal (wrist and foot) spasms; twitching (possibly progressing to tetany); and muscle weakness. Severe respiratory alkalosis may cause cardiac arrhythmias, seizures, and syncope.
Metabolic Acidosis
Metabolic acidosis is an accumulation of acids or a deficit of bases in the blood. This type of acidosis can occur with an acid gain (e.g., ketones with diabetic ketoacidosis, lactic acid with hypoxia, toxins such as ethylene glycol, and renal failure) or bicarbonate loss (e.g., diarrhea). Specific etiologic factors are listed in Table 5-13.
Ketoacidosis occurs when insufficient insulin for the proper use of glucose results in increased breakdown of fat. This accelerated fat breakdown produces ketones and other acids. Although the body attempts to neutralize these increased acids, the plasma bicarbonate (HCO3−) is depleted.
In the case of renal failure, the failing kidney cannot rid the body of excess acids and cannot produce the necessary bicarbonate to buffer the acid load that is accumulating in the body. Lactic acidosis occurs as excess lactic acid is produced during strenuous exercise or when oxygen is insufficient (hypoxemia). Severe diarrhea depletes the body of highly alkaline intestinal and pancreatic secretions.123
Clinical Manifestations
The symptoms of metabolic acidosis can include muscular twitching, weakness, malaise, nausea, vomiting, diarrhea, and headache (see Table 5-13). If the acidosis is severe, myocardial depression and hypotension can occur. Compensatory hyperventilation may occur as a result of stimulation of the hypothalamus as the body attempts to rid itself of excess CO2. As the acid level goes up, these symptoms progress to stupor, unconsciousness, coma, and death. The breath may have a fruity odor in the presence of acetone associated with ketoacidosis.
Metabolic Alkalosis
Metabolic alkalosis occurs when either an abnormal loss of acid or excess accumulation of bicarbonate occurs. Postoperative loss of acids through vomiting or gastric suctioning may also result in metabolic alkalosis. In the outpatient setting, diarrhea, excessive use of laxatives, diuretics, antacids, and milk (milk alkali syndrome) may also lead to metabolic alkalosis. Other causes are listed in Table 5-13.
Clinical Manifestations
Signs and symptoms occur as the body attempts to correct the acid-base imbalance, primarily through hypoventilation. Respirations are shallow and slow as the lungs attempt to compensate by building up carbonic acid stores. Clinical manifestations may be mild at first, with muscle weakness, irritability, confusion, and muscle twitching (see Table 5-13). If untreated, the condition progresses and the person may become comatose, with possible seizures, cardiac arrhythmias, and respiratory paralysis.
Aging and Acid-Base Regulation
The normal aging process results in decreased ventilatory capacity and loss of alveolar surface area for gas exchange; thus older adults are prone to respiratory acidosis caused by hypoventilation and to respiratory alkalosis caused by hypoxemia and subsequent hyperventilation. Older adults are often taking multiple medications for hypertension or cardiovascular disease that may contribute to hypokalemia and metabolic alkalosis. Respiratory compensation in these conditions can be compromised because of the structural and functional changes mentioned.
Aldosterone is less effective in older adults, as is ammonia buffering. These changes limit renal compensation for respiratory imbalances and place the individual at higher risk for metabolic imbalance.138,169
Older adults who are unable to excrete an acid load may develop a chronic metabolic acidosis. While the bicarbonate level and pH of the blood remain normal, mild metabolic acidosis may contribute to muscle wasting and bone loss.
References
1. American College of Sports Medicine (ACSM). ACSM guidelines for exercise testing and prescription, ed 6. Baltimore: Lippincott Williams and Wilkins, 2000.
2. Adamsen, L, Midtgaard, J, Andersen, C, et al. Transforming the nature of fatigue through exercise: qualitative findings from a multidimensional exercise programme in cancer patients undergoing chemotherapy. Eur J Cancer Care. 2004;13(4):362–370.
3. Adamsen, L, Midtgaard, J, Rorth, M, et al. Feasibility, physical capacity, and health benefits of a multidimensional exercise program for cancer patients undergoing chemotherapy. Support Care Cancer. 2003;11(11):707–716.
4. Aft, RL, Lewis, JS, Zhang, F, et al. Enhancing targeted radiotherapy by copper(II) diacetyl-bis(N4-methylthiosemicarbazone) using 2-deoxy-D-glucose. Cancer Res. 2003;63(17):5496–5504.
5. American Diabetes Association. Clinical practice recommendations, 2000. Diabetes Care. 2000;23(Suppl 1):S1–S116.
6. American Physical Therapy Association. Guide to physical therapist practice, ed 2. Alexandria, VA: The Association, 2003.
7. Anscher, MS, Chen, L, Rabbani, Z, et al. Recent progress in defining mechanisms and potential targets for prevention of normal tissue injury after radiation therapy. Int J Radiat Oncol Biol Phys. 2005;62(1):255–259.
8. Arvary, D. Anabolic-androgenic steroids as a gateway to opioid dependence. N Engl J Med. 2000;342(20):1532.
9. Augustine, E. Oncology section of the APTA position statement-physical therapy: management of lymphedema in patients with a history of cancer. Rehabil Oncol. 2000;18(1):9–12.
10. Bahrke, MS, et al. Risk factors associated with anabolic-androgenic steroid use among adolescents. Sports Med. 2000;29(6):397–405.
11. Bajrovic, A, Rades, D, Rades, D, et al. Is there a life-long risk of brachial plexopathy after radiotherapy of supraclavicular lymph nodes in breast cancer patients? Radiother Oncol. 2004;71(3):297–301.
12. Baker, PD, Morzorati, SL, Ellett, ML. The pathophysiology of chemotherapy-induced nausea and vomiting. Gastroenterol Nurs. 2005;28(6):469–480.
13. Bayry, J, Misra, N, Latry, V, et al. Mechanisms of action of intravenous immunoglobulin in autoimmune and inflammatory diseases. Transfus Clin Biol. 2003;10(3):165–169.
14. Bhasin, S, Calof, OM, Storer, TW, et al. Drug insight: testosterone and selective androgen receptor modulators as anabolic therapies for chronic illness and aging. Nat Clin Pract Endocrinol Metab. 2006;2(3):146–159.
15. Bhasin, S, Storer, TW, Berman, N, et al. The effects of supraphysiologic doses of testosterone on muscle size and strength in normal men. N Engl J Med. 1996;335(1):1–7.
16. Bhasin, S, Woodhouse, L, Casaburi, R, et al. Older men are as responsive as young men to the anabolic effects of graded doses of testosterone on the skeletal muscle. J Clin Endocrinol Metab. 2005;90(2):678–688.
17. Bijlsma, JWJ, Saag, KG, Buttgereit, F, et al. Developments in Glucocorticoid Therapy. Rheum Dis Clin North Am. 2005;31(1):1–17.
18. Blay, JY, Conroy, T, Chevreau, C, et al. High-dose methotrexate for the treatment of primary cerebral lymphomas: analysis of survival and late neurologic toxicity in a retrospective series. J Clin Oncol. 1998;16(3):864–871.
19. Bombardier, C, Laine, L, Reicin, A, et al. Comparison of upper gastrointestinal toxicity of rofecoxib and naproxen in patients with rheumatoid arthritis. N Engl J Med. 2000;343:1520–1528.
20. Boockvar, K, Fishman, E, Kyriacou, CK, et al. Adverse events due to discontinuations in drug use and dose changes in patients transferred between acute and long-term care facilities. Arch Internal Medicine. 2004;164(5):545–550.
20a. Braith, RW, et al. Resistance exercise prevents glucocorticoid-induced myopathy in heart transplant recipients. Med Sci Sports Exerc. 1998;30(4):483–489.
21. Bresalier, RS, Sanlder, RS, Quan, H, et al. Cardiovascular events associated with rofecoxib in a colorectal adenoma chemoprevention trial. N Engl J Med. 2005;352(11):1092–1102.
22. Bushbacher, L. Rehabilitation of patients with peripheral neuropathies. In Braddom R, ed.: Physical medicine and rehabilitation, ed 2, Philadelphia: WB Saunders, 2000.
23. Cheng, HF, Harris, RC. Cyclooxygenases, the kidney, and hypertension. Hypertension. 2004;43(3):525–530.
24. Cheng, HF, Harris, RC. Renal effects of non-steroidal anti-inflammatory drugs and selective cyclooxygenase-2 inhibitors. Curr Pharm Des. 2005;11(14):1795–1804.
25. Choudhury, D, Rau, DSC, Levi, M. Effect of aging on renal function and disease. In Brenner BM, ed.: Brenner & Rector’s The Kidney, ed 7, Philadelphia: Saunders, 2004.
26. Chyka, P. How many deaths occur annually from adverse drug reactions in the United States? Am J Med. 2000;109:122–130.
27. Ciccone, C. Introduction: pharmacology. Phys Ther. 1995;75:342–351.
28. Cina, JL, Gandhi, TK, Churchill, W, et al. How many hospital pharmacy medication dispensing errors go undetected? Jt Comm J Qual Patient Saf. 2006;32(2):73–80.
29. Cohen, J. Avoiding adverse reactions: effective lower-dose drug therapies for older patients. Geriatrics. 2000;55:54–56. [59-60, 63-64].
30. Cohen, R, Moelleken, B. Disorders due to physical agents. In Tierney L, McPhee S, Papadakis M, eds.: Current medical diagnosis and treatment, ed 39, New York: Lange Medical Books/McGraw-Hill, 2000.
31. Cotran, R, Kumar, V, Collins, T. Acute and chronic inflammation. In Cotran R, Kumar V, Collins T, eds.: Robbins pathologic basis of disease, ed 6, Philadelphia: WB Saunders, 1999.
32. Cross, NE, Glantz, MJ. Neurologic complications of radiation therapy. Neurol Clin. 2003;21(1):249–277.
33. Crossen, JR, Garwood, D, Glatstein, E, et al. Neurobehavioral sequelae of cranial irradiation in adults: a review of radiation-induced encephalopathy. J Clin Oncol. 1994;12:627–642.
34. Dantas, F, Rampes, H. Do homeopathic medicines provoke adverse effects? A systematic review. Br Homeopath J. 2000;89:S35–S38. [(Suppl)].
35. Dean, E. Monitoring systems in the intensive care unit. In Frownfelter D, Dean E, eds.: Principles and practice of cardiopulmonary physical therapy, ed 3, St Louis: Mosby, 1996.
36. Dean, E. Oxygen transport deficits in systemic disease and implications for physical therapy. Phys Ther. 1997;77(2):187–202.
37. Dean, E, et al. Cardiovascular/cardiopulmonary physical therapy sinks or swims in the twenty-first century: addressing the health care issues of our time. Phys Ther. 2000;80(12):1275–1278.
38. DeAngelis, LM, Delattre, JY, Posner, JB. Radiation-induced dementia in patients cured of brain metastases. Neurology. 1989;39(6):789–796.
39. Decramer, M, de Bock, V, Dom, R. Functional and histologic picture of steroid-induced myopathy in chronic obstructive pulmonary disease. Am J Respir Crit Care. 1996;153:1958–1964.
40. De Jonghe, B, Sharshar, T, Lefaucheur, JP, et al. Paresis acquired in the intensive care unit. JAMA. 2002;288(22):2859–2867.
41. Delanian, S, Balla-Mekias, S, Lefaix, JL. Striking regression of chronic radiotherapy damage in a clinical trial of combined pentoxifylline and tocopherol. J Clin Oncol. 1999;17:3283–3290.
42. Demling, RH, DeSanti, L. Oxandrolone, an anabolic steroid, significantly increases the rate of weight gain in the recovery phase after major burns. J Trauma. 1997;43(1):47–51.
43. Demling, RH, DeSanti, L. Closure of the “non-healing wound” corresponds with correction of weight loss using the anabolic agent oxandrolone. Ostomy Wound Manage. 1998;44(10):58–68.
44. Demling, RH, Orgill, DP. The anticatabolic and wound healing effects of the testosterone analog oxandrolone after severe burn injury. J Crit Care. 2000;15(1):12–17.
45. Diehl, KM, Chang, AE. Acute abdomen, bowel obstruction, and fistula. In Abeloff MD, Armitage JO, Niederhuber JE, Kastan MB, McKenna WG, eds.: Clinical oncology, ed 3, Philadelphia: Churchill Livingstone, 2004.
46. Dimeo, FC. Effects of aerobic exercise on the physical performance and incidence of treatment-related complications after high-dose chemotherapy. Blood. 1997;90:3390–3394.
47. Dimeo, FC. Effects of physical activity on the fatigue and psychological status of cancer patients during chemotherapy. Cancer. 1999;85(10):2273–2277.
48. Dimeo, FC, Rumberger, BG, Keul, J. Aerobic exercise as therapy for cancer fatigue. Med Sci Sports Exerc. 1998;30:475–478.
49. Dimeo, FC, Stieglitz, D, Novelli-Fischer, U, et al. Effects of physical activity on the fatigue and psychological status of cancer patients during chemotherapy. Cancer. 1999;85(10):2273–2277.
50. Dirksen, S. Integumentary system. In Lewis S, Heitkemper M, Dirksen S, eds.: Medical-surgical nursing: assessment and management of clinical problems, ed 5, St Louis: Mosby, 2000.
51. Dorr, W, Hendry, JH. Consequential late effects in normal tissues. Radiother Oncol. 2001;61:223–231.
52. Drouin, J, Pfalzer, LA. Moderate intensity aerobic exercise for the person with cancer. In: Wruble E, ed. Acute care perspectives. Robbinsville, NJ: Acute Care Section-APTA, 2001.
53. Eason, JM, Dodd, SL, Powers, SK. Use of anabolic steroids to attenuate the effects of glucocorticoids on the rat diaphragm. Phys Ther. 2003;83(1):29–36.
54. Efficacy and safety of corticosteroids for persistent acute respiratory distress syndrome; The national heart, lung, and blood institute acute respiratory distress syndrome (ARDS) clinical trials network. N Eng J Med. 2006;354(16):1671–1684.
55. Eifel, PJ. Chemoradiation for carcinoma of the cervix: advances and opportunities. Radiat Res. 2000;154(3):229–236.
56. Endo, T, Minami, M, Monama, Y, et al. Emesis-related biochemical and histopathological changes induced by cisplatin in the ferret. J Toxicol Sci. 1990;15:235–244.
57. Fathers, E, Thrush, D, Huson, SM, et al. Radiation-induced brachial plexopathy inwomen treated for carcinoma of the breast. Clin Rehabil. 2002;16(2):160–165.
58. Feldmeier, J. Hyperbaric oxygen 2003: indications and results. The hyperbaric oxygen therapy committee report. Undersea and Hyperbaric Medical Society. 2003.
59. Felsenthal, G, Lehman, J, Stein, B. Principles of geriatric rehabilitation. In Braddom R, ed.: Physical medicine and rehabilitation, ed 2, Philadelphia: WB Saunders, 2000.
60. Ferrando, AA, Sheffield-Moore, M, Wolf, SE, et al. Testosterone administration in severe burns ameliorates muscle catabolism. Crit Care Med. 2001;29(10):1936–1942.
61. Ferrando, AA, Sheffield-Moore, M, Yeckel, CW, et al. Testosterone administration to older men improves muscle function, molecular and physiological mechanisms. Am J Physiol Endocrinol Metab. 2002;282(3):E601–E607.
62. Fine, N, Scallion, L. Hodgkin’s disease. In: Miaskowski C, Buchsel P, eds. Oncology nursing: assessment and clinical care. St Louis: Mosby, 1999.
63. Fink, MP, Delude, RL. Unifying theme to explain the pathogenesis of multiple organ dysfunction at the cellular level. Crit Care Clin. 2005;21(2):177–196.
64. Finn, S, Scherer, R. Seizure after exercise in the heat: recognizing life-threatening hyponatremia. Phys Sports Med. 2000;28:61. [62, 65-67].
65. Fitzgerald, P. Endocrinology. In McPhee L, McPhee S, Papadakis M, eds.: Current medical diagnosis and treatment, ed 39, Tierney: Lange Medical Books/McGraw-Hill New York, 2000.
66. Franklin, DJ, Packer, L. Cancer related fatigue. Arch Phys Med Rehab. 2006;87(3):91–93.
67. Freedman, TG. Prescriptions for health providers from cancer patients. Cancer Nurs. 2003;26(4):323–330.
68. Freml, JM, Smith, KM. Medical management in the elderly: do you know the risks? Orthopedics. 2005;28(9):915–917.
69. Galantino, ML, Henderson, A, Michaels, J. Cognitive challenges for women undergoing adjuvant chemotherapy for treatment for breast cancer: the role of rehabilitation oncology. Rehab Oncol. 2005;23(1):7–10.
70. Gandhi, TK, Bartel, SB, Shulman, LN, et al. Medication safety in the ambulatory chemotherapy setting. Cancer. 2005;104(11):2289–2291.
71. Gandhi, TK, Weingart, SN, Borus, J, et al. Adverse drug events in ambulatory care. N Engl J Med. 2003;348(16):1587–1589.
72. Gerber, L.H. Cancer rehabilitation: the bridge to cross between organ and organism. In L’Esprit Rehabilitation Centers Symposium: Cancer rehabilitation: the multidisciplinary integration of traditional and “whole person” care, Montreal, January 21-23, 2000, The Symposium.
73. Gibson, RJ, Keefe, DMK. Cancer chemotherapy-induced diarrhea and constipation: mechanisms of damage and prevention strategies. Support Care Cancer. 2007;15(5):483–490.
74. Glantz, MJ, Burger, PC, Friedman, AH, et al. Treatment of radiation-induced nervous system injury with heparin and warfarin. Neurology. 1994;44:2020–2027.
75. Goldstat, R, Briganti, E, Tran, J, et al. Transdermal testosterone therapy improves well-being, mood, and sexual function in premenopausal women. Menopause. 2003;10(5):390–398.
76. Goulding, MR. Inappropriate medication prescribing for elderly ambulatory care patients. Arch Intern Med. 2004;164:305–312.
77. Green, MR. Targeting target therapies. N Engl J Med. 2004;350(21):2191–2193.
78. Gudas, S. Shoulder rehabilitation post surgery and irradiation: brachial plexopathy. In L’Esprit Rehabilitation Centers Symposium: Cancer rehabilitation: the multidisciplinary integration of traditional and “whole person” care Montreal, Canada, January 21-23, 2000, The Symposium.
79. Guyton, A, Hall, J. Textbook of medical physiology, ed 10. Philadelphia: WB Saunders, 2000.
80. Halloran, PF. Immunosuppressive drugs for kidney transplantation. N Engl J Med. 2004;351(26):2715–2729.
81. Hartgens, F, Kuipers, H. Effects of androgenic-anabolic steroids in athletes. Sports Med. 2004;34(8):513–554.
82. Heidenreich, PA, Hancock, SL, Lee, BK, et al. Asymptomatic cardiac disease following mediastinal irradiation. J Am Coll Cardiol. 2003;42(4):743–749.
83. Hesketh, PJ, Batchelor, D, Golant, M, et al. Chemotherapy-induced alopecia: psychosocial impact and therapeutic approaches. Support Care Cancer. 2004;12(8):543–549.
84. Hooper, L, Brown, TJ, Elliott, R, et al. The effectiveness of five strategies for the prevention of gastrointestinal toxicity induced by non-steroidal anti-inflammatory drugs: systematic review. BMJ. 2004;329:948.
85. Horne, M, Bond, E. Fluid, Electrolyte and acid base imbalances. In Lewis S, Heitkemper M, Dirksen S, eds.: Medical-surgical nursing; assessment and management of clinical problems, ed 5, St Louis: Mosby, 2000.
86. Hosseinimehr, SJ, Mahmoudzadeh, A, Akhlagpour, S. Captopril protects mice bone marrow cells against genotoxicity induced by gamma irradiation. Cell Biochem Funct. 2006.
87. Hull, MC, Morris, CG, Pepine, CJ, et al. Valvular dysfunction and carotid, subclavian, and coronary artery disease in survivors of Hodgkin Lymphoma treated with radiation therapy. JAMA. 2003;290(21):2831–2837.
88. Hurst, JW, Radiation-induced heart disease. Medscape Cardiology. 2004;8(1). Available on-line at http://www.medscape.com/viewarticle/465851, Accessed on September 9, 2006.
89. Hymes, SR, Strom, EA, Fife, C. Radiation dermatitis: Clinical presentation, pathophysiology, and treatment 2006. J Am Acad Dermatol. 2006;54(1):28–46.
90. Iskander, J.K., Miller, E.R. & Pless, R.P. et al, Vaccine safety post-marketing surveillance: the vaccine adverse event reporting system (VAERS). Continuing education activity sponsored by the Centers for disease control and prevention (CDC), July 2006. Available on-line at http://www.cdc.gov/nip/vacsafe/VAERS/CME-post-mktg-surv.pdf, Accessed on September 6, 2006.
91. Israel, E, Banerjee, TR, Fitzmaurice, GM, et al. Effects of inhaled Glucocorticoids on bone density in premenopausal women. N Engl J Med. 2001;345:941–947.
92. Jeter, MD, Janne, PA, Brooks, S. Gemcitabine-induced radiation recall. Int J Radiat Oncol Biol Phys. 2002;53(2):394–400.
93. Johansson, S, et al. Brachial plexopathy after postoperative radiotherapy of breast cancer patients-a long term follow-up. Acta Oncol. 2000;39(3):373–382.
94. Jones, GB. From mustard gas to medicines. Chemical Heritage. 15(2), 1998.
95. Jordan, KM, Arden, NK, Doherty, M, et al. EULAR Recommendations 2003: an evidence based approach to the management of knee osteoarthritis: Report of a task force of the standing committee for international clinical studies including therapeutic trials (ESCISIT). Ann Rheum Dis. 2003;62:1145–1155.
96. Kang, SK. Radiation recall reaction after antimicrobial therapy. N Engl J Med. 2006;354(6):622.
97. Kim, PK, Deutschman, CS. Inflammatory responses and mediators. Surg Clin North Am. 2000;80:885–894.
98. Kompoliti, K, Horn, SS. Drug-Induced and Iatrogenic Neurological Disorders. In Goetz CG, ed.: Textbook of clinical neurology, ed 2, Philadelphia: Saunders, 2003.
99. Lewis, S. Inflammation and infection. In Lewis S, Heitkemper M, Dirksen S, eds.: Medical-surgical nursing: assessment and management of clinical problems, ed 5, St Louis: Mosby, 2000.
100. Liu, CY, Yim, BT, Wozniak, AJ. Anticoagulation therapy for radiation-induced myelopathy. 2001;35(2):188–191.
101. Lowenthal, RM, Eaton, K. Toxicity of chemotherapy. Hematol Oncol Clin North Am. 1996;10:967–990.
102. Ludin, A, Macklis, RM. Radiotherapy for pediatric genitourinary tumors. Its role and long-term consequences. Urol Clin North Am. 2000;27(3):553–562.
103. Machtay, M. Pulmonary complications of anticancer treatment. In Abeloff MD, Armitage JO, Niederhuber JE, et al, eds.: Clinical oncology, ed 3, New York: Churchill-Livingstone, 2004.
104. Matsuda, T, Takayama, T, Tashiro, M, et al. Mild cognitive impairment after adjuvant chemotherapy in breast cancer patients-evaluation of appropriate research design and methodology to measure symptoms. Breast Cancer. 2005;12(4):279–287.
105. McArdle, W, Katch, F, Katch, V. Exercise physiology: energy, nutrition and human performance, ed 4. Philadelphia: Lippincott Williams and Wilkins, 2000.
105a. Meek, AG. Breast radiotherapy and lymphedema. Cancer. 1998;83:2788–2797.
106. Midgley, SJ, et al. Risk behaviors for HIV and hepatitis infection among anabolic-androgenic steroid users. AIDS Care. 2000;12(2):163–170.
107. MM Wolfe. Rofecoxib, Merck, and the FDA. N Engl J Med. 2004;351(27):2877.
108. Mock, V, et al. Effects of exercise on fatigue, physical functioning, and emotional distress during radiation therapy for breast cancer. Oncology Nurs Forum. 1997;24(6):991–1000.
109. Moeller, BJ, Batinic-Haberle, I, Spasojevic, I, et al. A manganese porphyrin superoxide dismutase mimetic enhances tumor radioresponsiveness. Int J Radiat Oncol Biol Phys. 2005;63(2):545–552.
110. Moncur, C, Williams, HG. Rheumatoid arthritis: status of drug therapies. Phys Ther. 1995;75:61–75.
111. Morris, G.S. Chemotherapy-induced cardiotoxicity. Adapted from presentation at Combined Sections, February 2, 2006.
112. Mukand, JA, Cai, C, Zielinski, A, et al. The effects of dehydration on rehabilitation outcomes of elderly orthopedic patients. Arch Phys Med Rehabil. 2003;84(1):58–61.
113. Mukherjee, D, Nissen, SE, Topol, EJ. 2001 Risk of cardiovascular events associated with selective COX-2 inhibitors. JAMA. 2001;286:945–949.
114. National Academy of Science Research CouncilHealth risks from exposure to low levels of ionizing radiation: BEIR VII-Phase 2, 2005. Available on-line at http://www.books.nap.edu/catalog/11340.html Accessed September 5, 2006.
115. National Cancer Institute. Radiation therapy and you. Bethesda, MD: U.S. Department of Health and Human Services, 1997.
116. National Comprehensive Cancer Network (NCCN) Side effects of cancer treatment. Available on-line at http://www.nccn.org Accessed September 5, 2006.
117. Navari, RM, Province, PS. Emerging drugs for chemotherapy-induced emesis. Expert Opin Emerg Drugs. 2006;11(1):137–151.
118. Nguyen, NP, Antoine, JE. Radiation enteritis. In Feldman M, Friedman LA, Sleisenger MH, eds.: Sleisenger & Fordtran’s gastrointestinal and liver disease: pathophysiology, diagnosis, management, ed 7, Philadelphia: WB Saunders, 2002.
119. Nicol, N, Ruszkowski, A. Integumentary problems. In Lewis S, Heitkemper M, Dirksen S, eds.: Medical-surgical nursing: assessment and management of clinical problems, ed 5, St Louis: Mosby, 2000.
120. Nicholas, J, Lennard, T. Joint and soft tissue injection techniques. In Braddom R, ed.: Physical medicine and rehabilitation, ed 2, Philadelphia: WB Saunders, 2000.
121. Nseir, S, Di Pompeo, C, Soubrier, S, et al. Impact of ventilator-associated pneumonia on outcome in patients with COPD. Chest. 2005;128:1650–1656.
122. Oeffinger, KC, Hudson, MM. Long-term complications following childhood and adolescent cancer: foundations for providing risk-based health care for survivors. A Cancer J Clin. 2004;54(4):208–236.
123. Okuda, T, Kiyoshi, K, Papadakis, M. Fluid and electrolyte disorders. In Tierney L, McPhee S, Papadakis M, eds.: Current medical diagnosis and treatment, ed 39, New York: Lange Medical Books/McGraw-Hill, 2000.
124. Okunieff, P, Augustine, E, Hicks, JE. Pentoxifylline in the treatment of radiation-induced fibrosis. J Clin Oncol. 2004;22(11):2207–2213.
125. Packel, L, Claghorn, KVB, Dekerlegand, J. Cancer-related fatigue and deconditioning: A program evaluation. Rehab Oncol. 2006;24(2):3–8.
126. Palmer, BF, Henrich, WL. Toxic nephropathy. In Brenner BM, ed.: Brenner & Rector’s the kidney, ed 7, Philadelphia: WB Saunders, 2004.
127. Papathanasoglou, E, et al. Does programmed cell death (apoptosis) play a role in the development of multiple organ dysfunction in critically ill patients: a review and theoretical framework. Crit Care Med. 2000;28:537–542.
128. Parkinson, AB, Evans, NA. Anabolic androgenic steroids: a survey of 500 users. Med Sci Sports Exerc. 2006;38(4):644–651.
129. Pennisi, P, Trombetti, A, Rizzoli, R. Glucocorticoid-induced osteoporosis and its treatment. Clin Orthop Relat Res. 2006;443:39–47.
130. Percy, L, Fang, M. Geropharmacology for the rheumatologist. Rheum Dis Clin North Am. 2000;26:1–21.
131. Perera, GK, Child, FJ, Heaton, N, et al. Skin lesions in adult liver transplant recipients: a study of 100 consecutive patients. Br J Dermatol. 2006;154(5):868–872.
132. Perry, MC, Anderson, CM, Donehower, RC. Chemotherapy. In Abeloff MD, Armitage JO, Niederhuber JE, Kastan MB, McKenna WG, eds.: Clinical oncology, ed 3, Philadelphia: Churchill Livingstone, 2004.
133. Petersson, A, Garle, M, Holmgren, P, et al. Toxicological findings and manner of death in autopsied users of anabolic androgenic steroids. Drug Alcohol Depend. 2006;81(3):241–249.
134. Ponticelli, C, Campise, MR. Neurological complications in kidney transplant recipients. J Nephrol. 2005;18(5):521–528.
135. Pope, JE, Anderson, JJ, Felson, DT. A meta-analysis of the effects of nonsteroidal anti-inflammatory drugs on blood pressure. Arch Intern Med. 1993;153(4):477–484.
136. Porock, D, Nikoletti, S, Cameron, F. The relationship between factors that impair wound healing and the severity of acute radiation skin and mucosal toxicities in head and neck cancer. Cancer Nursing. 2004;27(1):71–78.
137. Portenoy, RK, Itri, LM. Cancer-related fatigue: guidelines for evaluation and management. Oncologist. 1999;4(1):1–10.
138. Priebe, H. The aged cardiovascular risk patient. Br J Anaesth. 2000;85:763–778.
139. Pritchard, J, Anand, P, Broome, J, et al. Double-blind randomized phase II study of hyperbaric oxygen in patients with radiation-induced brachial plexopathy. Radiother Oncol. 2001;58(3):279–286.
140. Recchia, F, Saggio, G, Amiconi, G, et al. Gonadotropin-releasing hormone analogues added to adjuvant chemotherapy protect ovarian function and improve clinical outcomes in young women with early breast carcinoma. Cancer. 2006;106(3):514–523.
141. Recommendations for the prevention and treatment of glucocorticoid-induced osteoporosis. 2001 update. American College of Rheumatology Ad Hoc Committee on Glucocorticoid-Induced Osteoporosis. Arthritis Rheum. 2001;44(7):1496–1503.
142. Reents, S. Sport and exercise pharmacology. Champain, IL: Human Kinetics, 2000.
143. Cidlowski, JA. Antiinflammatory action of glucocorticoids-new mechanisms for old drugs. N Engl J Med. 2005;353:1711–1723.
144. Rieger, PT, Escalante, CP. Complications of cancer treatment. In: Boyer K, et al, eds. Primary care oncology. Philadelphia: WB Saunders, 1999.
145. Robbins, ME, Diz, DI. Pathogenic role of the renin-angiotensin system in modulating radiation-induced late effects. Int J Radiat Oncol Biol Phys. 2006;64(1):6–12.
146. Robinson-Bostom, L, Pan, TD, McDonald, CJ. Alopecia and cutaneous complications. In Abeloff MD, Armitage JO, Niederhuber JE, Kastan MB, McKenna WG, eds.: Clinical oncology, ed 3, New York: Churchill-Livingstone, 2004.
147. Rolston, KV. Expanding the options for risk based therapies in febrile neutropenia. Diagn Microbial Infect Dis. 1998;25:411–416.
148. Sader, MA, Griffiths, KA, McCredie, RJ, et al. Androgenic anabolic steroids and arterial structure and function in male bodybuilders. J Am Coll Cardiol. 2001;37(1):224–230.
149. Sambrook, P, Lane, NE. Corticosteroid osteoporosis. Best Pract Res Clin Rheumatol. 2001;15:401–413.
150. Schäcke, H, Schottelius, A, Döcke, WD. Dissociation of transactivation from transrepression by a selective glucocorticoid receptor agonist leads to separation of therapeutic effects from side effects. Proc Natl Acad Sci USA. 2004;101:227–232.
151. Schindl, A, Schindl, M, Pernerstorfer-Schon, H, et al. Low intensity laser irradiation in the treatment of recalcitrant radiation ulcers in patients with breast cancer-long-term results of 3 cases. Photodermatol Photoimmunol Photomed. 2000;16:34–37.
152. Schultz, PN, Beck, ML, Stava, C, et al. Health profiles in 5836 long-term cancer survivors. Int J Cancer. 2003;104(4):488–495.
153. Segar, ML, et al. The effect of aerobic exercise on self-esteem and depressive and anxiety symptoms among breast cancer survivors. Oncol Nurs Forum. 1998;25(1):107–113.
154. Seidman, A, Hudis, C, Pierri, MK, et al. Cardiac dysfunction in the trastuzumab clinical trials experience. J Clin Oncol. 2002;20(5):1215–1221.
155. Shaddick, N. The natural history of exercise-induced anaphylaxis: survey results from a 10-year follow-up study. J Allergy Clin Immunol. 1999;104:123–127.
156. Singh, G, Triadafilopoulus, G. Epidemiology of NSAID-induced GI complications. J Rheumatol. 1999;26(suppl 26):18–24.
157. Solomon, SD, McMurray, JJV, Pfeffer, MA, et al. Cardiovascular risk associated with Celecoxib in a clinical trial for colorectal adenoma prevention. N Engl J Med. 2005;352(11):1071–1080.
158. Speyer, JL, Ewer, MS, Freedberg, RS. Cardiac effects of cancer therapy. In Abeloff MD, Armitage JO, Niederhuber JE, et al, eds.: Clinical oncology, ed 3, Philadelphia: Churchill Livingstone, 2004.
159. Staat, K, Segatore, M. The phenomenon of chemo brain. Clin J Oncol Nurs. 2005;9(6):713–721.
160. Stilger, VG, Yesalis, CE. Anabolic-androgenic steroid use among high school football players. J Community Health. 1999;24(2):131–145.
161. Stone, JH. The classification and epidemiology of systemic vasculitis. In Harris ED, Budd RC, Genovese MC, et al, eds.: Kelly’s textbook of rheumatology, ed 7, Philadelphia: Elsevier, 2005.
162. Sturmi, J, Diorio, D. Anabolic agents. Clin Sports Med. 1998;17:261–281.
163. Sullivan, ML, Martinez, CM, Gallagher, EJ. Atrial fibrillation and anabolic steroids. J Emerg Med. 1999;17(5):851–857.
164. Sullivan, P, Markos, P. Ambulation: a framework of practice applied to a functional outcome. In Guccione A, ed.: Geriatric physical therapy, ed 2, St Louis: Mosby, 2000.
165. Suter, PM. Lung inflammation in ARDS-friend or foe. N Engl J Med. 2006;354(16):1739–1742.
166. Taylor, CW, McGale, P, Darby, SC. Cardiac risks of breast-cancer radiotherapy: a contemporary view. Clin Oncol. 2006;18(3):236–245.
167. Thiblin, I, Lindquist, O, Rajs, J. Cause and manner of death among users of anabolic androgenic steroids. J Forensic Sci. 2000;45(1):16–23.
168. Thiessen, B, DeAngelis, LM. Hydrocephalus in radiation leukoencephalopathy: results of ventriculoperitoneal shunting. Arch Neurol. 1998;55(5):705–710.
169. Thompson, L. Physiological changes associated with aging. In Guccione A, ed.: Geriatric physical therapy, ed 2, St Louis: Mosby, 2000.
170. Tikka, T, Usenius, T, Tenehunen, M, et al. Tetracycline derivatives and ceftriaxone, a cephalosporin antibiotic, protect neurons against apoptosis induced by ionizing radiation. J Neurochem. 2001;78:1409–1414.
171. Tsibiribi, P, Descotes, J, Lombard-Bohas, C, et al. Cardiotoxicity of 5-fluorouracil in 1350 patients with no prior history of heart disease. Bull Cancer. 2006;93(3):E27–E30.
172. Van den Belt-Dusebout, AW, Nuver, J, de Wit, R, et al. Long-term risk of cardiovascular disease in a 5-year survivors of testicular cancer. J Clin Oncol. 2006;24(3):467–475.
173. van der Eerden, BC, Karperien, M, Wit, JM. Systemic and local regulation of the growth plate. Endocr Rev. 2003;24:782–801.
174. van Weert, E, Hoekstra-Weebers, J, Renée Otter, R, et al. Cancer related fatigue: predictors and effects of rehabilitation. The Oncologist. 2006;11(2):184–196.
175. Varricchio, C, eds. ed 7. A cancer source book for nurses. Atlanta: American Cancer Society; 1997.
176. Verstappen, CC, Heimans, JJ, Hoekman, K, et al. Neurotoxic complications of chemotherapy in patients with cancer. Drugs. 2003;63:1549–1563.
177. Warren, JL, Bacon, WE, Harris, T, et al. The burden and outcomes associated with dehydration among US elderly. Am J Public Health. 1994;84(8):1265–1269.
178. Warren, JL, Harris, T, Phillips, C. Dehydration in older adults. JAMA. 1996;275(12):912.
179. Wefel, JS, Lenzi, R, Theriault, RL, et al. The cognitive sequelae of standard-dose adjuvant chemotherapy in women with breast carcinoma. Cancer. 2004;101(9):2143–2145.
180. Werner-Wasik, M. Treatment-related esophagitis. Semin Oncol. 2005;32(2 Suppl 3):S60–S66.
181. Werner-Wasik, M, Langer, C, Movsas, B. Amifostine in chemoradiation therapy for non-small lung cancer: review of experience and design of a phase II trial assessing subcutaneous and intravenous bolus administration. Semin Oncol. 2005;32(2 Suppl 3):S105–S108.
182. Werner-Wasik, M, Yu, X, Marks, LB, et al. Normal-tissue toxicities of thoracic radiation therapy: esophagus, lung, and spinal cord as organs at risk. Hematol Oncol Clin North Am. 2004;18(1):131–160.
183. Wilson, B, Shannon, M, Stang, C. Nursing drug guide. Upper Saddle River, NJ: Prentice-Hall, 2001.
184. Winningham, ML. The role of exercise in cancer therapy. In: Watson R, Eisinger M, eds. Exercise and disease. Boca Raton, FL: CRC Press, 1992.
185. Wolfe, MM, Lichtenstein, DR, Singh, G. Gastrointestinal toxicity of nonsteroidal antiinflammatory drugs. N Engl J Med. 1999;340(24):1888–1899.
186. Wollina, U, Liebold, K, Konrad, H. Treatment of chronic radiation ulcers with recombinant platelet-derived growth factor and a hydrophilic copolymer membrane. J Eur Acad Dermatol Venereol. 2001;15:455–457.
187. Workman, M, Visovsky, C. Cancer pathophysiology. In: Miaskowski C, Buchsel P, eds. Oncology nursing: assessment and clinical care. St Louis: Mosby, 1999.
188. Worthington, K. Guarding against radiation exposure. Am J Nurs. 2000;100(5):104.
189. Yagami, T, Ueda, K, Hata, S, et al. S-2474, a novel nonsteroidal anti-inflammatory drug, rescues cortical neurons from human group IIA secretory phospholipase A(2)-induced apoptosis. Neuropharmacology. 2005;49(2):174–184.
190. Yarnold, J. Double-blind randomised phase II study of hyperbaric oxygen in patients with radiation-induced brachial plexopathy. Radiother Oncol. 2005;77(3):327.
191. Yeh, ETH, Ton, AT, Lenihan, DJ, et al. Cardiovascular complications of cancer therapy. Diagnosis, pathogenesis and management. Circulation. 2004;109(25):3122–3131.
192. Young, L. Sepsis syndrome. In Mandell G, Bennett J, Dolin R, eds.: Principles and practice of infectious diseases, ed 5, Philadelphia: Churchill Livingstone, 2000.