Metabolic Disorders
Metabolic disorders can affect numerous body tissues, including bony structures. Primary metabolic diseases and their implications for the therapist are discussed in Chapter 11. Metabolic disorders primarily affecting the skeletal system are the focus of this chapter.
The skeleton is a metabolically active organ that undergoes continuous remodeling throughout life with an annual turnover of cortical and trabecular bone of about 10% of the adult skeleton.104 This remodeling is necessary both to maintain the structural integrity of the skeleton and to serve a metabolic function as a storehouse of calcium and phosphorus. These dual functions can come into conflict under conditions of changing mechanical forces or metabolic and nutritional stress.96
Clinical disorders in which bone resorption is increased are common and include Paget’s disease of bone, osteoporosis, and the bone changes secondary to cancer such as occur in myeloma and metastases from breast or prostate cancer. Clinical disorders of reduced bone resorption are less common and have a genetic basis (e.g., osteopetrosis).
Metabolic bone disease is typically manifested by diffuse loss of bone density and bone strength, but increased bone density and decreased bone strength can occur such as with Paget’s disease. Significant disability, marked by bone pain, postural deformity, and fracture, can occur secondary to these bony changes. The commonly observed accentuated thoracic spine kyphosis in clients with vertebral collapse secondary to osteoporosis can compromise cardiopulmonary function, affecting the person’s ability to participate in a rehabilitation program.
The most serious, potentially life-threatening and costly complication of metabolic bone disease is fracture (see the section on Fractures in Chapter 27). Osteoporosis alone is estimated to be responsible for more than 1.5 million fractures annually (700,000 vertebral fractures, 300,000 hip fractures, 250,000 wrist fractures, and 250,000 other fractures); 1 in 3 women older than 50 years of age experience a fracture in their lifetime.79 The monetary cost of these fractures is estimated at $10 billion to $15 billion annually in the United States. This estimate does not include the indirect costs of lost wages or productivity of either the individual or the caregiver.76
Therapists have an important role in the primary prevention of disability secondary to the complications associated with metabolic bone disease. Client education regarding posture, body mechanics, and proper exercise is an important component of any prevention program. Physical therapy intervention is also a vital part of the rehabilitation of clients disabled by the resultant pain or postural deformities that can accompany these diseases. Therapists also treat many clients who have experienced traumatic injury precipitated by the presence of metabolic bone disease or conversely, people who are experiencing the metabolic consequences of trauma.
The regulation of all metabolic functions requires proper fluid and electrolyte balance, which in turn relies on a finely tuned integration between the endocrine system and the nervous system. The effects of metabolic disorders on muscle and muscle performance are discussed more completely in Chapter 5. Finally, therapists treat many people with a primary diagnosis of low back, knee, or shoulder conditions with osteoporosis as a secondary diagnosis. The presence of such a secondary diagnosis may influence the therapist’s choice of evaluation and intervention. A thorough understanding of this group of diseases will enable the therapist to treat clients safely and effectively.
OSTEOPOROSIS
Definition and Overview
Osteoporosis, which means porous bones, is a combination of decreased bone mass and microdamage to the bone structure that results in a susceptibility to fracture. Osteoporosis can be classified as primary or secondary, depending on the underlying etiology. Primary osteoporosis, the most common, can occur in both genders at all ages but often follows menopause in women, and occurs later in life in men. Primary osteoporosis has two subtypes: postmenopausal/estrogen-deficient (type 1) and age-related/senile (type 2).118 Osteoporosis associated with medications, other conditions, or diseases is referred to as secondary osteoporosis (Box 24-1).
Incidence
Osteoporosis is by far the most common metabolic bone disease, affecting approximately 10 million people living in the United States. An additional 34 million Americans already have low bone mass (osteopenia) that places them at increased risk of osteoporosis. With the aging of America, osteoporosis is expected to increase in prevalence.77
The disease is much more common in women, especially postmenopausal women who are estrogen deficient. However, osteoporosis in men represents a major public health problem, which until recently has received little recognition. Approximately 2 million men are affected by osteoporosis (usually age-related, type 2), and another 12 million are at risk as a result of low bone mass. According to the National Osteoporosis Foundation (NOF), this condition in men is underdiagnosed, undertreated, and underreported.78
One in every 2 women older than 50 years of age will experience fragility fractures secondary to osteoporosis. One in 4 men will experience an osteoporosis-related fragility fracture during their lifetime, usually later (because of greater bone mass than women) at approximately age 70 years. When affected, men have a higher morbidity and mortality than that of women (30% versus 9%).7,131 This is because men are older at the time they sustain a fracture and are more likely to have comorbid disease, malnutrition, and complicated hospitalizations.133
Etiologic Factors
The cause of primary osteoporosis is unknown, but many contributory factors exist such as mild but prolonged negative calcium balance, declining gonadal and adrenal function, relative or progressive estrogen deficiency, or sedentary lifestyle. Secondary osteoporosis may be caused by prolonged therapy with corticosteroids, heparin, anticonvulsants, and other medications; alcoholism, malnutrition, malabsorption, or lactose intolerance; endocrine disorders; or other conditions or diseases (see Box 24-1).
Risk Factors
Between the ages of 25 and 35 years, bone mass peaks, and the rate of bone resorption begins to exceed the rate of bone formation. This physiologic mismatch can progress to a point at which osteopenia (Box 24-2) may be noted radiographically and a diagnosis of osteoporosis made. Box 24-1 lists the many risk factors and conditions or diseases associated with osteoporosis. Chronic diseases, or medications used to treat these diseases, may have side effects that can damage bone or interfere with bone formation, leading to osteoporosis.
A difference is certainly evident between risk factors for osteoporosis and risk factors for fractures (see Box 27-17). Estrogen status, heredity, and ethnicity are important risk factors for osteoporosis in women; alcoholism, cigarette smoking, steroid therapy, and hypogonadism or androgen withdrawal therapy for prostate cancer are the most common factors for men,152 although in 50% of cases, a cause is unknown.131 Age is a risk factor for both men and women; the 10-year fracture risk at age 50 years quadruples by age 80 years; although women are affected earlier in the decades compared to men, the associated mortality and morbidity is much greater for men.1
Hormonal Status.: Postmenopausal women are at higher risk to develop the disease. Diagnosis of primary postmenopausal osteoporosis occurs with increasing frequency from age 51 to 75 years. The increased risk of osteoporosis related to menopause is due primarily to the decreased production of estrogen. Decreased intestinal calcium absorption, increased bone resorption to compensate for low calcium levels, and impaired osteoblastic activity have all been associated with estrogen deficiency.
Women lose bone at the usual rate of 1% per year after peak bone density has been achieved; however, bone loss accelerates to a varying degree (depending on such factors as calcium intake and absorption, hormonal balance, and activity level) for about 5 to 8 years after menopause, increasing the risk of fracture. Researchers report a wide range from 2% to 11% loss for the 10 years after menopause, slowing after that to about 0.5% to 1% of bone mass per year.17,84,101
Men experience a gradual slowing of testosterone production with age, and below-normal testosterone levels have been associated with loss of bone mineral mass. The outward signs of this phenomenon (e.g., loss of muscle strength, fatigue, and a decreased interest in sex) are often attributed to aging without considering the accompanying osteoporosis that ensues. Other risk factors include androgen-deprivation treatment for prostate cancer and hypogonadism associated with erectile dysfunction.10 Low serum testosterone levels have also been documented in men treated with corticosteroids for chronic inflammatory conditions such as sarcoidosis.2
Heredity/Genetics.: Peak bone mass is partly genetically determined as evidenced by the varying prevalence of osteoporosis among different ethnic groups, but other variables also exist such as differences in bone geometry and rate of bone loss. Individual differences among people of the same ethnic background can occur, possibly in part because of inheriting specific genes that affect bone mass and turnover.108
Eight genes have been linked to bone density with a strong connection to the receptor for parathyroid hormone (PTHR1). This hormone plays a crucial role in determining the level of calcium in the blood. Genetic differences in the levels of receptors for PTH in the blood may determine bone density. The results of some research suggest that gender specificity as to how these genes are expressed plays a key role (i.e., men and women may inherit these genes differently).26
Body build is related to bone fragility. Thin women have less cortical bone and are therefore at greater risk of fractures. Obesity, by increasing the mechanical strain on bone, may result in increased peak bone mass, reducing bone fragility. In addition, obesity increases the amount of biologically available estrogen, protecting against fracture.17 Women with a family history of osteoporosis are at high risk of developing osteoporotic fractures.
Ethnicity.: Ethnicity is also considered a risk factor because bone mass correlates positively with skin pigmentation. Whites have the least amount of bone mass, whereas blacks have the greatest amount. Men have wider long bones than women, and blacks have wider long bones than whites. Therefore black men are at lowest risk of developing osteoporosis.114 Because white women generally have lower peak bone mass, the complications of the bone loss associated with aging can affect them earlier in life.
Limited information available suggests that Native American women have lower bone densities than white non-Hispanic women, and Mexican-American women have bone densities intermediate between those of white non-Hispanic women and black women.76 On the other hand, skin pigmentation also correlates with vitamin D synthesis, with more heavily pigmented people requiring greater amounts of sunshine to synthesize vitamin D needed for bone strength. Early reports indicate that osteoporosis may be more prevalent in Hispanic, Native American, Asian, and black women than once thought.
Physical Inactivity.: Inactivity and immobilization have been associated with decreased bone formation. The prolonged inactivity results in reduced gravitational and muscular forces acting on the skeletal system. The decreased mechanical stress on bony structures alters bone physiology, resulting in decreased bone mass.
Disuse osteopenia, which can be caused by immobilization (e.g., cast, bed rest, long-term care, or neurologic impairment), results from a change in cellular function or an increase in the recruitment of osteoclasts. The difference in the quality of bone produced is in the decreased number of trabeculae and in increased osteoclastic performance. Residents in nursing home or long-term care facilities have a fivefold to tenfold greater fracture risk than community dwellers.28
Tobacco.: Cigarette smoking is associated with a reduction of bone mass and is a well-known risk factor for spinal and hip fractures. The effects of smokeless tobacco and secondhand smoke have not been evaluated as risk factors but may be important considerations. Some evidence suggests that cigarette smoke may affect bone progenitor cells by directly contributing to the development of osteoporosis.62,129
Alcohol.: More than two “units” of alcohol per day is a risk factor for osteoporosis.52 A unit is defined as one 12 oz beer, one 5 oz glass of wine, or 1.5 oz of hard liquor. Excessive alcohol intake alters osteoblast gene expression and matrix synthesis, thereby reducing the number of effective cells.149 Chronic alcohol use can impair intestinal absorption and increase renal excretion of calcium, contribute to poorer overall nutrition, and increase the likelihood of a fall.
In men, consuming large amounts of alcohol is a major independent risk factor for hip fractures46; combined alcohol and corticosteroid use has a significant impact on the development of osteoporosis.54,133 (See the sections on Tobacco and Depressants in Chapter 2.)
Medications.: Long-term use of medications, including corticosteroids, has been associated with the presence of osteoporosis. People with a wide range of medical conditions (e.g., asthma, chronic obstructive pulmonary disease [COPD], organ transplantation, renal impairment, arthritis, systemic lupus erythematosus [SLE], Crohn’s disease or regional enteritis, sarcoidosis, and others) may be taking corticosteroids.
Most bone loss occurs during the first 6 months of systemic corticosteroid therapy. Loss of 10% or 15% of spinal trabecular bone mass is possible; after that, bone loss averages 1% to 2% annually. Corticosteroids impair osteoblastic activity and the maturation of preosteoblastic cells to osteoblasts, increase osteoclastic activity, and impair vitamin D–dependent intestinal calcium absorption, which can result in secondary hyperparathyroidism. The hyperparathyroidism increases bone resorption and decreases renal resorption of calcium, thereby increasing the amount of calcium excretion.
Other medications have also been implicated (see Box 24-1). Selective serotonin reuptake inhibitors (SSRIs) are not on this list, but a recent study has shown that people over the age of 50 years who regularly took SSRIs had double the fracture rate as people not taking such medications.100 More research is needed to identify if depression itself is the source of endocrine changes that can damage bone.42
Depression.: Depression is now recognized as a risk factor for osteoporosis.112,153 Individuals with a history of a major depressive disorder are more likely to have lower bone densities and higher levels of cortisol than people without depression, regardless of physical activity levels.70 Researchers do not know how depression might cause a loss of bone mineral density (BMD), but increases in the stress hormone cortisol could account for some of the loss. As mentioned, scientists are looking for an explanation of the relationship between hormones and brain chemistry that could be involved in both depression and bone loss.
Depression may be a risk factor in other ways. A National Institute of Mental Health (NIMH) study of bone loss in premenopausal women with and without major depression showed that those who were depressed had unfavorable levels of cytokines that specifically affect bone mass. Cytokines act as chemical messengers between the immune, nervous, and endocrine systems. The connection between bone loss and depression has led to a new discipline referred to as the “neuropsychoendocrinology” of bone regulation.15,68 Poor nutrition as a result of loss of appetite may be another depression-related risk factor for osteoporosis.
Diet and Nutrition.: Different sources of dietary protein may have different effects on bone metabolism. Older women age 65 and older with a high dietary ratio of animal-to-vegetable protein intake have more rapid femoral neck bone loss and a greater risk of hip fracture than do those with a low ratio.
Animal foods provide predominantly acid precursors, whereas protein in vegetable foods is accompanied by base precursors not found in animal foods. Imbalance between dietary acid and base precursors leads to a chronic net dietary acid load that may have adverse consequences on bone.113,115 Excess acid is excreted in the urine, but with aging, the kidneys are less capable of excreting acid.
Bone density is decreased in anorexic and bulimic women and is possibly the result of estrogen deficiency, low intake of nutrients, low body weight, early onset and long duration of amenorrhea, low calcium intake, reduced physical activity, and hypercortisolism. This type of reduced bone density is associated with a significantly increased risk of fracture even at a young age.36 A new term, female athlete triad, has been coined to describe the combination of disordered eating, amenorrhea, and osteoporosis, which is a situation that often goes unrecognized and untreated.109
A significant number of people with osteoporosis also have celiac disease, a gastrointestinal (GI) disorder that impairs the absorption of calcium, various nutrients, and vitamin D needed for maintaining healthy bones. Identification and effective dietary therapy for celiac disease can lead to improved absorption of vital nutrients and potentially reverse the decline in BMD. Celiac disease increases the long-term risk of fracture, especially hip fracture.45,65
New information about the role of cholesterol in bone health has come to light. High cholesterol associated with a high-fat diet may decrease overall bone production through the process of oxidation. Oxidative stress modulates differentiation of vascular and bone cells oppositely so that a parallel buildup and loss of calcification occur in vascular calcification and osteoporosis, respectively.73 Preliminary studies in animals suggest that an atherogenic diet inhibits bone formation by blocking differentiation of osteoblast progenitor cells.92
Pathogenesis
Bone is composed of a meshwork of collagen fibers inlaid with calcium and phosphate. These minerals are mixed with water to form a hard, cementlike substance called hydroxyapatite. Sodium, magnesium, and potassium are also present in smaller amounts. The human skeleton is composed of two main bone types: trabecular and cortical.
Bone Composition.: Trabecular, also referred to as spongy or cancellous, bone is the center meshwork of bones. Trabecular mass constitutes approximately 20% of the adult skeleton and is sensitive to influences relating to metabolic and hormonal implications. The outer substance of bone mass is the cortical (compact) bone, found primarily in the shafts of the long bones and comprising roughly 80% of skeletal mass. The concentric orientation of lamellae deposited around a central nutrient canal in cortical bone forms very dense tissue. Of the total volume of cortical bone, 80% to 90% is calcified (versus 15% to 25% of trabecular bone volume that is calcified). The remaining volume is occupied by bone marrow, fat, and blood vessels.117
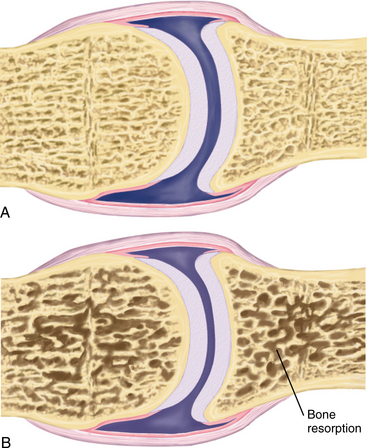
Because bone turnover is a surface event and trabecular bone has a high surface area, bones rich in trabeculae (e.g., vertebrae, metaphyseal segments of long bones, and the calcaneus) are more active, metabolically exhibiting a greater rate of turnover and resorption with loss of structural integrity during the aging process (Fig. 24-1). The ultimate strength of a specific bone appears to be related to the percentage of trabecular versus cortical bone, the structural integrity, and the trabecular interconnections of the bone.150
Bone Remodeling.: Bone renews itself in a constant process called remodeling, a repeating cycle of osteoclasts breaking down the existing bone bit by bit (resorption) and osteoblasts laying down new cells (formation) for mineralization. The purpose of remodeling is to maintain the strength and integrity of the skeleton by replacing fatigue-damaged older bone with new bone and to act as a source of vital minerals (e.g., calcium) necessary for the maintenance of mineral homeostasis. Calcium’s importance extends far beyond the bone. It circulates throughout the body to regulate heart rate, muscle contractions, blood pressure, and other systemic functions. When blood levels of calcium drop below the level necessary to carry out these functions, it is replenished from bone.
BMD increases during growth and development, and the peak bone mass is achieved in the third decade of life. This is the bone bank for the rest of a person’s life.12 An individual with a high peak bone mass who starts to lose bone later in life and loses it slowly is unlikely to sustain osteoporotic fractures, whereas someone with suboptimal peak bone mass who starts to lose bone earlier in life or loses bone rapidly is more likely to develop osteoporosis with fractures.
Osteoporosis develops when new bone formation falls behind resorption, possibly related to impaired new bone formation due to declining osteoblast function (Fig. 24-2). The onset of bone loss is likely to be genetically predetermined, and the subsequent rate of bone loss may also be influenced by genetic factors.
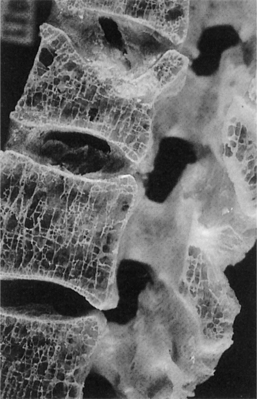
Figure 24-2 Osteoporosis of the lumbar vertebra with generalized loss of bone. The vertical plates have become perforated and the number of horizontal cross-braces are decreased markedly in proportion to the vertical plates. (From McPherson RA: Henry’s clinical diagnosis and management by laboratory methods, ed 21, Philadelphia, 2006, WB Saunders.)
Bone loss increases at the time of menopause as a result of the marked reduction in the circulating concentrations of estradiol and progesterone (Fig. 24-3).33 Other causes of age-related bone loss have been discussed previously in this chapter. Each one of those contributing factors has its own pathologic process. Only the general pathogenic concepts are discussed here.
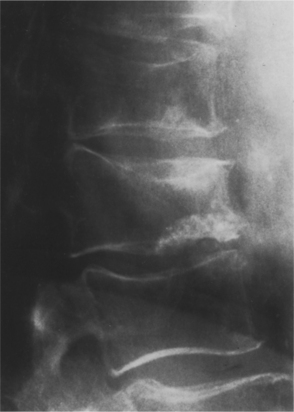
Figure 24-3 Postmenopausal osteoporosis in a 59-year-old woman. Lateral view of the thoracolumbar spine sows protrusion of disc material through a weakened end plate (Schmorl’s node). Note also the biconcave appearance of several vertebral bodies. (From Grainger: RG: Grainger & Allison’s diagnostic radiology: a textbook of medical imaging, ed 4, Philadelphia, 2001, Churchill Livingstone.)
Small defects in formation remaining at the completion of a normal remodeling cycle accumulate and also contribute to age-related losses in bone mass. Thus the bone that experiences the greatest number of remodeling cycles is at the highest risk for age-related losses in mass.117 New information about metabolic bone diseases has revealed that maintenance of adult skeletal mass is controlled not only by changes in the production of osteoclasts and osteoblasts but also by altering the duration of their respective lifespans through regulated apoptosis (programmed cell death). During bone remodeling, disruption of the rate of supply of new osteoblasts and osteoclasts and the timing of this supply by apoptosis may be an important mechanism behind the deranged bone turnover found in most metabolic disorders of the adult skeleton.148
Defects in a protein called alphaV beta3 integrin appear to play a role in the development of osteoporosis. The effects of this protein can be reversed by enhancing another protein, macrophage colony-stimulating factor (M-CSF). The interaction between M-CSF and alphaV beta3 integrin is under investigation. These proteins may have a role in bone cell differentiation and the signaling pathway that causes a change in osteoclast and osteoblast balance.137,154
Bone Demineralization.: Bone demineralization, which can lead to osteopenia, takes place when a deficit in hormonal levels, inadequate physical activity (mechanical load), or poor nutrition occurs. Bone strength is a function of skeletal load—that is, bone responds to alterations in mechanical forces. Although the exact physio- logic mechanism causing bone to sense and respond to alterations in mechanical loads is unclear, adaptations appear to be site specific.
Mechanical stimuli may be the only type of stimuli capable of inducing modeling in mature bone; young bone responds more favorably to mechanical loading than old bone, again emphasizing the importance of physical activity and exercise in the adolescent and young adult.146 Physical activity transmits mechanical loads to the skeleton through gravitational forces and muscular pull at sites of attachment. In the absence of mechanical forces (space flight or prolonged bed rest), urinary calcium excretion increases and bone density decreases.117 BMD changes induced by loading are not maintained long-term, which is why regular site-specific and weight-bearing exercises must be done routinely to prevent osteoporosis and reduce bone fracture risk.146
Subsequently, hormonal levels, physical activity, and nutrition are the key factors that facilitate bone growth because they regulate the osteoblastic and osteoclastic remodeling cycles, initiate a natural cycle of microscopic bone damage and subsequent repair, and foster solid bone architecture.16
Gender Differences.: Studies of normal age-related changes in human bone have been investigated by using a combination of different techniques. Men show an age-related compensatory increase in bone size (cross- sectional area of the vertebral bodies), which cannot be found in women. Women have a higher tendency, after age 50 years, to demonstrate disconnection of the trabecular network that can be positively affected by physiologic loading such as occurs during exercise.74
Clinical Manifestations
Loss of height, postural changes, back pain, and fracture are the most common presenting features of osteoporosis. Postural changes may include lax abdominal musculature, protuberant abdomen, forward head, kyphosis, dowager’s hump, loss of lumbar lordosis, posterior pelvic tilt, knee hyperextension, shoulder internal rotation, scapular forward rotation, palms facing backward, and other deviations in alignment observed when assessed by a physical therapist. Marked thoracic spine kyphosis and loss of overall body height are common findings, especially after a vertebral compression fracture. Similarly, bone loss in the mandible can contribute to changes in facial appearance.
Muscular pain and spasm can occur in the lower back paravertebral muscles, as can burning pain in the midthoracic region lateral to the spine because of excess stretch placed on the rhomboid muscles from the compensatory forward rotation of the scapulae. Trigger points secondary to kyphoscoliosis, rhomboid imbalance, and paravertebral muscle spasm are common. Similar muscle imbalance and muscular symptoms are observed or reported in the lower quadrant with involvement of the lumbosacral and sacroiliac joints and surrounding musculature.
Fractures.: The vertebral bodies, hip, ribs, radius, and femur are the most common fracture sites (in that order) although any bone in the body can be affected. Fractures are often “silent” compression fractures of vertebral bodies, sacral insufficiency fractures, or complete fractures of the spine or femoral neck. Recently, metatarsal insufficiency fracture in both men and women has been brought to the forefront as a previously unrecognized early sign of osteoporosis.139
Vertebral compression fractures (VCFs) are the most common osteoporosis-related spinal fractures presenting with clinical symptoms of back pain, posture change, loss of height, functional impairment, disability, and diminished quality of life (Fig. 24-4). These can occur without injury or fall when the bone becomes so porous or weak and it begins to compress (see the section on Fractures in Chapter 27). By age 90 years, the force required to produce failure of the L3 vertebrae is approximately one-fourth the compressive failure force at age 30 years.55
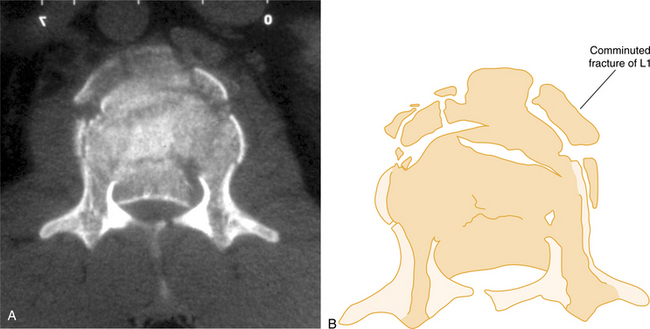
Figure 24-4 Computed tomography of unstable comminuted vertebral compression (burst) fracture of the lumbar vertebra (L1). (From Marx JA: Rosen’s emergency medicine: concepts and clinical practice, ed 6, St Louis, 2006, Mosby.)
The prevalence of vertebral fractures increases steadily with age, ranging between 20% for 50-year-old postmenopausal women and 64% for older women. The majority of vertebral fractures are not connected with severe trauma, and only one in three is diagnosed clinically. Almost 20% of women will experience another fracture within 1 year after a vertebral fracture.38
Pain associated with VCFs is usually severe and localized to the site of fracture, typically midthoracic, lower thoracic, and lumbar spine. Tenderness to palpation over the fracture is common in both symptomatic and otherwise asymptomatic cases. Pain may radiate to the abdomen or flanks and is aggravated by prolonged sitting or standing, bending, or performing a Valsalva maneuver. Side lying with hips and knees flexed may alleviate the pain. Generalized bone pain is more suggestive of meta- static carcinoma or osteomalacia. Neurologic symptoms may not occur immediately but rather develop insidiously over days to months.83
MEDICAL MANAGEMENT
Since no cure is available for osteoporosis, prevention and more effectively, early intervention is essential for everyone (men, women, young, and old) but especially for those at risk (Box 24-3). By minimizing modifiable risk factors, people at high risk for developing osteoporosis may be able to achieve higher peak bone mass in the hope of delaying or preventing the onset of osteoporosis.118 Because peak adult bone density depends on factors during growth and development, preventing osteoporosis in the aging adult begins by providing necessary dietary calcium intake during bone development and calcification in childhood and adolescence (Table 24-1).
Table 24-1
| Age Group | Minimum Daily Requirements (Elemental Calcium) | Calcium Administration |
| Birth to 6 months | 400 mg | Calcium supplements come in several preparations (e.g., carbonate, citrate, gluconate, lactate, phosphate). Calcium carbonate should be taken with meals that do not contain calcium-rich foods. Recommended daily requirements refer to elemental or actual calcium. Check the label for the %Daily Value, and note how many tablets or capsules are required to obtain this amount. |
| 6 months to 1 year | 600 mg | The body can absorb 500 mg of calcium at a time. Spread calcium intake (food or supplements) over the course of a day. Separate calcium supplements away from foods high in calcium or vitamins with calcium added. |
| Children ages 2 to 8 years | 800 mg | Beware of foods containing wheat bran or oxalic acid (e.g. chocolate, cauliflower, rhubarb, beet greens, brussel sprouts) that interfere with calcium absorption. Take calcium supplements away from these foods. |
| Children and adolescents ages 9 to 17 years* | 1300 mg | Calcium can interfere with the effectiveness of a variety of tetracycline antibiotics and quinolones such as Cipro, Floxin, Maxaquin, Noroxin, and Penetrex. Avoid consuming calcium (food or supplements) within 2 to 4 hrs of taking these drugs. |
| Adolescents and young adults (11 to 24 years) | 1200-1500 mg; the NOF recommends 1000 mg for this age group | Calcium, especially in antacids, may interfere with certain calcium channel blockers and β-blockers and thyroid medication (thyroxine). Check with prescribing physician about how to take calcium. |
| Women, age 25 to 50 years | 1000-1200 mg | Calcium can interfere with bisphosphonate absorption. If taking bisphosphonates, delay consuming calcium (food or supplements) at least 30 minutes. |
| Pregnant or lactating females; women >50 years of age | 1200 to1500 mg | Calcium just before bedtime can aid with reducing muscle cramps and improving sleep. |
| Postmenopausal women on estrogen therapy | 1000 mg (some report 1200 mg) | Spend at least 10 min daily in the sun (longer if using sunscreen) to obtain vitamin D necessary for calcium absorption and bone formation. |
| Postmenopausal women not on estrogen therapy | 1500 mg | For every ounce over 4 oz of animal protein, an additional 100 mg of calcium is required to stay even. |
| Men up to age 65 years* | 1200 mg | Take calcium supplements with a meal rather than on an empty stomach, unless the foods contain significant amounts of calcium. |
| Men >65 years† | 1500 mg | Extra-strength antacid made with calcium carbonate in tablet form (e.g., Tums) is used by some people to obtain calcium. Beware of this antacid; decreased stomach acid alters food digestion and absorption. For the individual with reduced stomach acid and especially the older adult, this may not be a good choice. |
| All women and men over age 65 | 1500 mg |
Anyone receiving medications for osteoporosis prevention should also be taking at least 1200 mg calcium and 400 IU/day vitamin D (800 IU/day for men over 65 years) each day in addition to a weight-bearing exercise program that is site specific; calcium is best absorbed when taken several times a day in amounts of 500 mg or less.
NOF, National Osteoporosis Foundation.
*This category is included by the Institute of Medicine.76
†Some sources recommend over age 55 years.
From National Women’s Health Information Center, S Department of Health and Human Services, 2000; Swan KG et al. Osteoporosis in men: a serious but under-recognized problem, J Musculoskel Med 18(6):310-316, 2001.
Information about the effect of maternal diet during pregnancy and its role in the early life-prevention of osteoporosis is being gathered through longitudinal studies.51 Other studies to assess the influence of maternal milk and soft drink consumption on children’s consumption show that parents play a powerful role in children’s eating behavior. They influence children’s developing preferences and eating behaviors by making some foods available rather than others, and by acting as models of eating behavior.32,110
Serum screening for celiac disease in anyone with osteopenia or osteoporosis is advised because dietary changes can restore normal absorption of dietary nutrients in this group, including calcium and vitamin D, contributing to a reversal in the decline of BMD associated with osteoporosis.130
Calcium.
Peak bone mass is attained around age 30 years, at which time the pattern of bone building and bone loss is reversed with more calcium loss than deposit. Regular exercise and physical activity, combined with adequate calcium, is considered both a prophylactic and treatment measure for osteoporosis from childhood through the adult years. The risk of fractures can be reduced by 50% if vitamin and nutrient requirements are met in the first 2 to 3 decades of life.44
The NOF, in conjunction with the Centers for Disease Control and Prevention (CDC), has initiated a multiyear campaign to promote optimal health in girls age 9 to 12 years. The goal is to educate and encourage young girls to establish lifelong healthy habits, especially increased calcium consumption and physical activity. Healthy People 2010 has eight goals (available for review)43 for children as young as age 2 years through the adult years.
Low-fat dairy and other calcium-and magnesium-rich foods (e.g., broccoli or kale, sardines or salmon with the bones, fresh or dried apricots, figs, turnip greens, oranges or calcium-enriched orange juice, or tofu) and calcium supplements are the primary means of achieving an adequate calcium intake.
Vitamin D.
Vitamin D helps the body absorb, synthesize, and transport calcium within the body, therefore necessitating adequate sunshine each day. Vitamin D requirements vary by geographic location and age. In northern areas (above the 42-degree latitude parallel, that is, in the United States, imagine a line stretching from the California-Oregon border to Boston), the sunlight in winter angles too obliquely to produce the ultraviolet light needed for adequate vitamin D production. Additional vitamin D (food or supplementation) is recommended; many calcium supplements contain vitamin D.
Diet.
Dietary considerations, such as reducing animal sources of protein, increasing vegetable sources,113,115 and increasing whole soy foods,111 may result in reduced risk for both cardiovascular disease and osteoporosis. Soybeans and foods made from soy are high in phytoestrogens, which are plant chemicals classified as isoflavones that display estrogen-like activity because of their structural similarity to human estrogens. These substances exhibit high affinity binding for the estrogen receptor and may have a beneficial influence in the prevention of osteoporosis, cardiovascular disease, and endocrine-controlled cancer (e.g., breast, prostate). This claim is not supported by all research results and remains highly controversial.127
According to some reports, the lipid-lowering oral statins (HMG-CoA reductase inhibitors, see Table 12-5) increase BMD by enhancing bone morphogenetic protein-2 (BMP-2), a powerful stimulator of osteoblast differentiation and osteoblastic expression, thereby promoting mineralization while reducing the risk of fractures. Contradictory data suggest that further testing is necessary before more specific recommendations can be made.49,132
Baseline BMD tests performed at the time of menopause (cessation of menstrual flow) or even earlier (during the perimenopausal phase) can provide a baseline assessment for future reference. Falls prevention (as a means of fracture prevention) is a separate component of osteoporosis management but not necessarily osteoporosis prevention (see the section on Falls Preventions in Chapter 27).
SCREENING.
Whereas osteoporosis was once called a “silent disease” because it was not recognized until a fracture signaled its presence, the widespread availability of technology to measure bone density has made it possible to identify people at risk for osteoporosis before fractures are imminent. Currently, no accurate measure of overall bone strength and standards of routine screening assessment have been established.
The NOF’s guide for physicians suggests a bone density test for every woman age 65 years and older.80 Additionally, the NOF has recommended testing for postmenopausal women under age 65 years who have sustained a bone fracture in adulthood, have a family history of osteoporosis, are thin, or smoke. Optimal intervals between tests have not been established. The measurements given here apply to white, adult women; it is not clear how to apply this diagnostic criterion to men and children and across ethnic groups. Because of the difficulty in accurate measurement and standardization between instruments and sites, controversy exists among experts regarding the continued use of this diagnostic criterion.76
Until evidence supports the cost-effectiveness of routine screening or the efficacy of early initiation of preventive drugs, an individualized approach has been recommended.76,95 However, even given this guideline, appropriate screening is not taking place. According to at least one study of Medicare recipients, bone density testing was the lowest for women in the highest fracture risk group based on age. This may occur because physicians do not realize the benefit of screening and intervention in fracture prevention for older adults (aged 80 years and older).82
Screening is particularly important for men at risk because fragility fractures are more likely to lead to fatal consequences than in women. Without a diagnosis of osteoporosis, treatment interventions that can prevent fractures are not initiated.2
DIAGNOSIS.
Careful assessment of the osteoporotic person is essential in developing a comprehensive plan that reduces fracture risk and improves quality of life. Assessment of the individual with osteoporosis includes history and physical examination, laboratory testing, and imaging studies. Information gathered during this assessment assists clinicians in targeting strategies to prevent fractures. Diagnosis and intervention are based on bone density and risk assessment120; in the case of secondary osteoporosis, the specific underlying cause must be determined through the diagnostic process before intervention can be initiated.
History.
The medical history should contain the personal and family history of fractures, lifestyle, intake of substances such as vitamin D, calcium, corticosteroids, and other medications. Clinicians should be aware of problems with vitamin D measurement, including seasonal variation, variability among laboratories, and the desirable therapeutic range. The physical examination can reveal relevant information such as height loss and risk of falls.60
Bone Mineral Density Testing.
BMD is often used as a proxy measure and accounts for approximately 70% of bone strength.76 A BMD test is the simplest way to assess for osteoporosis. Without this test, most people are unaware they have osteoporosis until a fracture occurs, although some fractures can be painless or the pain may be mistaken for arthritis, delaying diagnosis.
BMD is a measurement of the mineral content of bone in gram/square centimeter (g/cm2) for the area of the body that has been scanned. Researchers have evaluated the bone densities of thousands of women and used the peak bone density of women in their thirties as a reference point for average bone mass. Osteoporosis is now defined in terms of standard deviations from the average peak bone mass, also called a T-score. Sometimes a Z-score is also reported, which is the number of standard deviations by which the BMD differs from the mean value for the woman’s age and ethnicity.
Multiple imaging modalities can be used to evaluate BMD and diagnose osteoporosis, including dual energy x-ray absorptiometry (previously DEXA, now DXA), which measures spine, hip, or total body density; peripheral DXA (pDXA), which measures wrist, heel, or finger density only; quantitative ultrasound (QUS), in which sound waves are used to measure calcaneal, tibial, or patellar density; computed tomography (CT); or radiographic absorptiometry (RA), which is a radiograph of the hand. These methods are not interchangeable and do not provide equivalent information.
DXA is the preferred procedure because it measures bone density at the hip and spine where bone loss occurs more rapidly (Table 24-2). DXA has the highest sensitivity and specificity but sensitivity may not be adequate. The World Health Organization (WHO) recommends combining BMD to predict fracture risk with clinical risk factors.120 DXA best predicts fracture risk in adults without previous fracture. BMD testing is most efficient in women over 65 years of age but is also helpful for men and women with risk factors. Serial BMD tests can identify individuals losing bone mass, but clinicians should be aware of what constitutes a significant change.60
Table 24-2
World Health Organization (WHO) Classification for Bone Mineral Density
| T-Score | Significance |
| −1.0 or higher | Normal, low risk for fracture |
| −1.0 to 2.5* | Osteopenia (low bone mass) |
| −2.5 or lower† | Osteoporosis |
*Half of fragility fractures occur in this group.
†The National Osteoporosis Foundation (NOF) suggests that anyone with a T-score of −2.0 or less or −1.5 or less with at least 1 risk factor should be treated to reduce fracture risk.
From World Health Organization Study Group: Assessment of fracture risk and its application to screening for postmenopausal osteoporosis, Tech Rep Series 843, Geneva, 1994, WHO.
X-rays.
Fractures are usually diagnosed by radiograph examination (x-ray) that demonstrates the fracture (Fig. 24-5) and also reveals the osteopenia leading to the diagnosis of osteoporosis. Once osteopenia is noted, other causes of metabolic bone disease must be ruled out, including hyperthyroidism, hyperparathyroidism, osteomalacia, testicular failure, malignancies, and so on. Diagnostic criteria for men with this condition are at present based on those for women, although the validity of this approach is under discussion.131 Histologically, a thinning of cortical bone and a reduction in the number and size of the trabeculae of cancellous bone occurs (see Fig. 24-1). Thirty percent or greater bone density loss must occur before such abnormalities can be noted on an x-ray film.
Laboratory Testing.
Laboratory testing can detect other risk factors and can provide clues to etiology. Selection of laboratory tests should be individualized because there is no consensus regarding which tests are optimal. Biochemical markers of bone turnover (e.g., alkaline phosphatase, osteocalcin, urinary hydroxyproline, urinary deoxypyridinoline, urinary N-telopeptide, and others) reflect the rate of bone remodeling and may be helpful in assessing for fracture risk and for effectiveness of the treatment regimen but are not widely used at present.60
TREATMENT.
Osteoporosis is not curable, but intervention can stop the progression of bone loss and prevent further morbidity. Secondary osteoporosis intervention begins with treatment of the underlying cause. Management of primary and secondary osteoporosis in the adult should include lifestyle measures to reduce bone loss and prevent falls such as a high calcium intake, smoking cessation, reducing alcohol intake, and physical activity and exercise. Besides eliminating factors, such as chronic alcohol abuse and cigarette smoking, a key to prevention is developing as great a peak bone mass as possible through adequate calcium intake and regular exercise. Calcium is required for bone mineralization and also suppresses bone turnover, slowing the remodeling process.
Reports suggest that premenopausal women need more than 1000 mg of calcium daily, and postmenopausal women need more than 1500 mg daily (see Table 24-1). The average calcium intake of postmenopausal women is 400 to 500 mg/day, well below the recommended levels. Vitamin D (800 units/day) is advocated to assist in maximal absorption of dietary calcium.
Medications.
Pharmacotherapy to reduce fracture risk in women with reduced BMD is initiated when the T-score is below-2 in the absence of risk factors or below-1.5 if other risk factors are present.16,80 A T-score is not the only measure of fracture risk. A 70-year-old woman with a T-score of-2 is at greater immediate risk of fracture than a woman 50 years of age with the same T-score because of other variables common in the older age population that can increase fracture risk (e.g., poor eyesight, reduced muscle mass, balance problems, or medications).
Paying attention to individuals who are osteopenic (low bone mass but without the microarchitectural bone changes seen in osteoporosis) is important because there are increasing numbers of people who are osteopenic and who do experience bone fracture.120 Despite the availability and favorable safety profile of bisphosphonates, many people are not receiving therapy, including men who have low BMD or who are at risk of fracture from osteoporosis.8,10
Postmenopausal women need estrogen replacement with selective estrogen receptor modulators (SERMs), which maximize the beneficial effect of estrogen on bone and minimize or antagonize the deleterious effects on the breast and endometrium, or antiresorptive agents, such as bisphosphonate, to help prevent bone loss. Although calcitonin is not approved for prevention, it is approved for treatment because it acts directly on osteoclasts, suppressing activity.
Bisphosphonates are a family of drugs that inhibit bone resorption and actually reverse bone loss. These drugs are currently being used in various bone conditions involving increased levels of bone resorption such as osteoporosis from a variety of clinical causes, glucocorticoid-induced bone loss, hypercalcemia of cancer, and Paget’s disease of bone. The use of bisphosphonates in reducing bone pain associated with cancer is described in Chapter 26.
Researchers are evaluating two long-term effects of various treatment interventions: (1) the effect of each agent on BMD and (2) the effect of each agent on fracture development. For women in a state of estrogen deficiency, exercise or estrogen supplementation alone does not prevent bone loss, but exercise combined with estrogen causes a synergistic effect.86
Criticism of studies using exercise alone includes the nonspecificity of the exercise to the targeted area being measured. For example, walking and lifting weights are not enough of a stimulus for the forearm. Further research is needed to clarify the role of specific loading exercises in exercise prescription for postmenopausal women.
Estrogen supplementation has been found to protect against hip fracture only when used for more than 7 years,31 and once discontinued, bone loss resumes at a level similar to that in women who have not received such therapy.14 For this reason, the labeling of estrogens has been modified to state that they may be used to “manage” osteoporosis. The lack of efficacy of calcitonin to prevent bone loss during the first 5 years after menopause and the lack of prospective fracture reduction data for estrogen have resulted in these labeling restrictions. SERMs and bisphosphonates have been shown to increase BMD and decrease fracture risk.72 For men the addition of testosterone therapy for those individuals with normal gonadal function is under investigation.141
Raloxifene (Evista) is the only SERM approved by the Food and Drug Administration (FDA) at the time of this writing for the treatment and prevention of osteoporosis. It is the first compound with selective estrogen agonist activity in bone but with estrogen antagonist activity or no activity in reproductive tissues and breast.
Other second-generation SERMs, such as lasofoxifene (Oporia), remain under investigation. Raloxifene reduces the risk of positive estrogen-receptor breast cancer, decreases total cholesterol and low-density lipoprotein (LDL) cholesterol, and increases high-density lipoprotein (HDL) cholesterol but does not increase the risk of endometrial cancer or cause spotting and bleeding. After 3 years of treatment, raloxifene reduces the risk of first vertebral fracture by 55% with sustained efficacy in the continued reduction of fracture reduction in the fourth year. Raloxifene treatment does not change the physiologic structure of bone quality and does not cause fibrosis, osteomalacia, or other toxic effects.38
All resorptive agents currently approved for treatment of osteoporosis (e.g., bisphosphonates, SERMs, or nasal calcitonin) decrease bone resorption but cannot induce new bone formation. Consequently, their effects are limited. The use of agents that stimulate new bone formation, such as recombinant human PTH (rhPTH [teriparatide]), is under investigation as a single agent and in combination with antiresorptive agents.35,102
Early results of a new once-a-year pharmacologic treatment with zoledronic acid (Zometa) have been reported from the HORIZON Pivotal Fracture trials involving multiple investigators from several countries. Postmenopausal women with osteoporosis treated with a single infusion of intravenous zoledronic acid had a 70% reduced risk of vertebral fracture during a 3-year period and a 41% reduced risk of hip fracture. The advantage of this drug is the increased compliance rate because of the convenience of a once yearly infusion as opposed to a weekly or monthly dose regimen. It should be noted that higher rates of serious atrial fibrillation occurred in the zoledronic acid group.5
Combined administration of hormone replacement therapy (HRT) and bisphosphonates appears to have a more pronounced effect in the appendicular skeleton than the axial skeleton with greater increases of bone mass at the lumbar spine and hip than for either of the therapies alone.41
Fracture prevention has been shown for SERMS only at the spine, whereas bisphosphonates have been shown to reduce fracture rate by 50% at all sites. The benefit of other agents, such as calcitonin, PTH, and fluoride in preventing fracture, remains under investigation.50,102
The use of an ultralow-dose estrogen patch has been shown to improve bone density in animal and human trials. The skin patch contains a very small amount of estrogen with no progestin and has been used to increase bone density without harmful side effects in small groups of women. There is some evidence to support the protective effects of ultralow-dose estrogen therapy depends on a woman’s endogenous estrogen levels before treatment. More research is needed before this becomes a viable treatment option.29,47
A recent development has been the discovery of osteoprotegerin (OPG), a protein belonging to the tumor necrosis factor (TNF) receptor family. OPG occurs naturally in the body and plays a key role in the physiologic regulation of osteoclastic bone resorption. Animal studies have shown that OPG increases bone mass by reducing osteoclast production. Limited data suggest that OPG and PTH work together synergistically to improve bone mass and strength beyond what either can accomplish alone. The discovery of OPG has opened a new era in bone research by increasing the molecular knowledge available and providing new therapeutic targets in bone disease.56,63
Exercise.
Exactly how much benefit can be gained from given levels or types of exercise remains unclear, but the consensus is that regular exercise has a positive effect on bone mass levels. Weight-bearing exercises (against gravity), such as walking and jogging, stress the skeletal system and are associated with greater changes in bone remodeling and result in larger bone mass, although the effects on bone mass differ in premenopausal versus postmenopausal women. For example, walking alone has not been shown to be effective in arresting bone loss in postmenopausal women.13
The minimal or most effective frequency, intensity, and duration of exercise have not been determined. Optimal overload at specific skeletal sites during various activities remains to be determined by future studies. The NOF recommends a preventive program of 45 to 60 minutes of weight-bearing exercise 4 times/week.79
Exercises combined with bracing or other rehabilitative measures aimed at reducing the anterior translation of the cervicothoracic spine and thoracic hyperkyphosis may be beneficial in reducing the risk or occurrence of osteoporotic fractures.55
Vibration.
Whole body vibration, also known as oscillating plate therapy, is a new method of treatment being tested as a tool for the prevention of osteoporosis and bone fractures. Vibrating plates that the size and shape of a bathroom scale have shown effectiveness in increasing BMD (and balance). Vibratory exercise may be more effective than walking to improve both these measures. Researchers continue to investigate the physiologic mechanisms involved and to identify the most appropriate parameters to use that are both safe and effective.11,37,48
Surgery.
Because of the challenges of reconstruction of osteoporotic bone, open surgical management is reserved only for those rare cases that involve neurologic deficits or an unstable spine. New minimally invasive procedures for management of acute vertebral fractures, vertebroplasty, and kyphoplasty, which involve the injection of bone cement into the fractured vertebra, are discussed in Chapter 27.
PROGNOSIS.
Osteoporosis, once thought to be a natural part of aging among women, is no longer considered age-or gender-dependent. Although osteoporosis is one of the greatest deterrents to women’s health and accounts for significant morbidity and mortality, it is largely preventable because of the remarkable progress in the scientific understanding of its causes, diagnosis, and treatment. Even so, at the present time, 24% of women over the age of 50 years die from complications (e.g., pneumonia, infections, and fracture emboli) in the first year after an osteoporotic-associated hip fracture.79
Medical treatment for osteoporosis has been shown to decrease the incidence of vertebral fractures by 40% to 60% after just 1 year of treatment. The occurrence of a single vertebral fracture substantially increases the likelihood of future fractures and progressive kyphotic deformity.76 Even so, only 50% of women with vertebral compression fractures diagnosed incidentally with chest radiographs are started on any pharmacologic treatment.57
Vertebral fractures are accompanied by increased mortality with the relative risk of death after such a fracture being almost nine times higher than the person without osteoporosis-associated vertebral fracture.38 Low bone density at the hip is a strong and independent predictor of all cause and cardiovascular mortality in older men age 65 years and older.142
Adverse side effects of long-term use of bisphosphonates have been reported that affect the GI system and include esophagitis, heartburn, abdominal pain, and diarrhea. Intravenous bisphosphonates can cause transient acute phase reactions with fever, myalgia, and flu-like symptoms. Osteonecrosis of the jaw has also been raised as a possible concern, most often with intravenous administration of bisphosphonates used to manage hypercalcemia associated with metastatic disease.102
Adherence to osteoporosis medications is relatively poor with up to 30% of individuals suspending their treatment within 6 to 12 months of initiating therapy. Poor adherence is usually attributed to drug-induced adverse effects and results in increased risk of fracture and hospitalization.91 Some women may discontinue pharmacologic treatment early because their BMD test results do not show osteoporosis (or they think that is the interpretation of the test results).140
OSTEOMALACIA
Definition
In contrast to osteoporosis, which results in a loss of bone mass and brittle bones, osteomalacia is a progressive disease in which lack of mineralization of new bone matrix results in a softening of bone without loss of the present bone matrix. Osteomalacia is a generalized bone condition in which insufficient mineralization of the bone matrix results from calcium, vitamin D, and/or phosphate deficiency. The disease is sometimes referred to as the adult form of rickets, but the absence of epiphyseal plates in adults precludes the epiphyseal plate changes seen in rickets.
Etiologic Factors
The two primary causes of osteomalacia are insufficient intestinal calcium absorption and increased renal phos phorus losses. The insufficient calcium absorption could occur because of either a lack of calcium or a resistance to the action of vitamin D. The increased renal phosphorus losses can occur associated with renal osteodystrophy (see the section on Chronic Renal Failure in Chapter 18 and Fig. 18-7) and renal tubular insufficiency. In addition, the long-term use of antacids and the presence of long-standing hyperparathyroidism can lead to phosphate deficiencies contributing to the development of osteomalacia. Antacids containing aluminum hydroxide bind with dietary forms of phosphate to prevent GI absorption.
The dietary deficiency type of osteomalacia has been eradicated in the United States for the most part by the widespread supplementation of dairy products with vitamin D. However, osteomalacia does occur in the malnourished aging adult who may not receive adequate nutrition or enough exposure to sunlight.107
Incidence and Risk Factors
Osteomalacia is essentially a histologic diagnosis, so little information on its overall prevalence in the adult population is available. Housebound and institutionalized individuals are especially at risk, thus the prevalence of osteomalacia is expected to increase with the aging of the American population.24 Diseases of the small intestine, cholestatic disorders of the liver, biliary obstruction, and chronic pancreatic insufficiency increase the risk of developing osteomalacia. These conditions adversely affect the absorption of calcium and the action of vitamin D. The incidence of osteomalacia is greater in the world’s colder regions. This phenomenon is related to decreased exposure to sunlight, affecting vitamin D levels.
Osteomalacia is seen with greater frequency in cultures where the population has increased skin pigmentation and the diet is deficient in vitamin D (e.g., northern China, Japan, and northern India).24 Osteomalacia is common in the aged adult because of calcium-and vitamin D-deficient diets and decreased sunlight exposure. This situation is worsened by the intestinal malabsorption problems associated with aging or the presence of SLE that usually requires avoidance of sunlight to prevent ultraviolet-induced flare-ups.108
Long-term use of commonly prescribed medications also increases the risk of developing osteomalacia. Anticonvulsant medications, such as phenobarbital and phenytoin, accelerate breakdown of the active forms of vitamin D by inducing hepatic hydroxylases. As mentioned, antacids can cause phosphate deficiency. Box 24-6 summarizes the risk factors associated with osteomalacia.
Pathogenesis
Although the bone matrix remains intact (i.e., the bone structure remains unchanged) in osteomalacia, a generalized decrease in calcification of the matrix (calcification results in bone) and an increase in uncalcified matrix occur, leaving instead osteoid (bone that has not matured or calcified). Thus too little calcified bone occurs; failure of calcium salts to be deposited promptly in newly formed organic bone matrix (osteoid) results in osteomalacia.107
Demineralization results in an exaggeration of the osteoid seams seen radiographically adjacent to the relatively sparse areas of calcified bone (Fig. 24-6). As the osteoid accumulates, bone strength declines. These exaggerated seams occur because of the excessive time lag between collagen deposition and the appearance of the calcium salt. Areas of abundant osteoid appear radiographically as radiolucent stripes. These so-called pseudofractures, known as Looser’s zones, occur most commonly on the concave side of long bones, the ischial and pubic rami, and the ribs and scapula (Fig. 24-7). These pseudofractures develop from the healing of multiple microstress fractures in the moderately severe form of osteomalacia sometimes referred to as Milkman’s syndrome.107
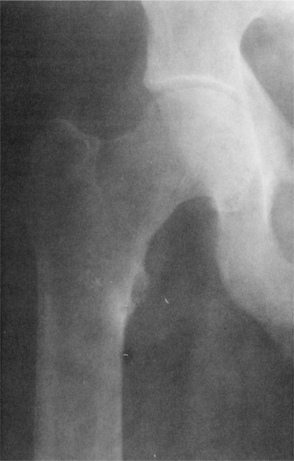
Figure 24-6 Osteomalacia of the femur. Note the loss of the sharp interface between cortical bone and cancellous bone caused by demineralization of the cortex. (From Richardson JK, Iglarsh ZA: Clinical orthopaedic physical therapy, Philadelphia, 1994, WB Saunders.)
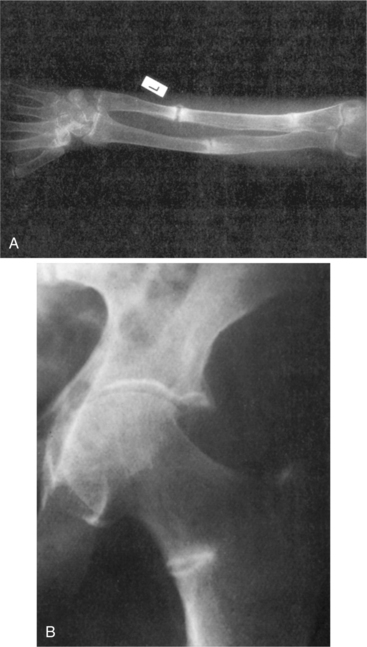
Figure 24-7 Osteomalacia. A, Forearm and B, femoral neck. Looser zones are seen as translucent zones with sclerotic margins. Usual sites include the medial femoral neck, pubic rami, lateral borders of the scapulae, and ribs. Complete fractures can extend through Looser’s zones; these will heal with appropriate treatment. (From Bullough P: Orthopaedic pathology, ed 3, London, 1997, Mosby-Wolfe.)
Clinical Manifestations
The diagnosis of osteomalacia is difficult and often delayed because many people present initially with diffuse, generalized aching and fatigue in the presence of anorexia and weight loss. Proximal myopathy and sensory polyneuropathy may also be present, resulting in a confusing clinical presentation. Bone pain and periarticular tenderness can occur in the spine, ribs, pelvis, and proximal extremities.
The combination of muscle weakness and softening of bone contributes to postural deformities, including increased thoracic kyphosis, a heart-shaped pelvis, and marked bowing of the femurs and tibias. The muscular weakness (proximal myopathy) may lead to a waddling gait and difficulties with transitional movements such as rising from sitting to standing, climbing stairs, or moving into and out of bed. Occasionally, the hypocalcemia associated with osteomalacia leads to latent tetany, with paresthesias of the hands and around the mouth, muscle cramps, and positive Chvostek and Trousseau signs (see Figs. 5-8 and 5-9).24
MEDICAL MANAGEMENT
Osteomalacia may present with a variety of clinical and radiographic manifestations mimicking other musculoskeletal disorders (e.g., fibromyalgia or polymyalgia rheumatica).98 For this reason, numerous methods are used to diagnose osteomalacia, including radiographs, bone scan, bone biopsy, and a laboratory workup (blood tests and urinalysis). Serum for levels of calcium, albumin, phosphate, alkaline phosphatase, and PTH are also obtained. Urine is collected to assess calcium and phosphate excretion rates.
Radiographically, osteomalacia, like osteoporosis, may present as osteopenia. A bone biopsy may be done at the site of osteopenia to evaluate the bone matrix. Besides osteopenia, radiolucent bands in the bone cortex (Looser’s zones) may be revealed radiographically (see Fig. 24-7).
Although not occurring nearly as often as in osteoporosis, acute fracture may be what leads to the diagnosis of osteomalacia. The radius, femur, vertebral bodies, ribs, and pubic ramus are common sites of fracture.
TREATMENT.
The treatment of osteomalacia depends on the cause. If inadequate nutrition is the problem, strengthening the dietary regimen with calcium and vitamin D is necessary. This step may be sufficient to improve the calcification of the organic matrix and thereby result in healing of the pseudofractures and strengthening the bones in general.
If osteomalacia is a result of intestinal malabsorption, treatment is directed to correct the primary disease. Phosphate supplementation can be prescribed in the presence of renal phosphate wasting. If used, vitamin D must be given to enhance calcium absorption impaired by the phosphate.
PAGET’S DISEASE
Definition
Paget’s disease (also known as osteitis deformans) is the second most common metabolic bone disease after osteoporosis. A progressive disorder of the adult skeletal system, this disease is characterized by abnormal bone remodeling with increased bone resorption and excessive, unorganized new bone formation caused by activated osteoclasts. Eventually, the normal bone marrow is replaced by vascular and fibrous tissue. Although Paget’s disease is a state of high bone turnover, the excess bone that is formed lacks the structural stability of normal bone (enlarged bone but weakened), leading to complications such as deformity, fracture, arthritis, and pain. The disease may involve one or more sites.
Incidence and Prevalence
Paget’s disease is a common disease of the aging adult population, rarely presenting before age 35 years with increasing prevalence among adults over age 50 years. Approximately 3% to 4% of the population over age 50 years and 10% of those over age 70 years may be affected (up to 3 million people in the United States).3
Although still unclear, both prevalence and severity of Paget’s disease seem to be declining. Current prevalence is only approximately 50% of that in 1983.18 Men and women are both affected, although a slight increased prevalence is evident among men. The disease is often familial and has an unusual geographic distribution. Populations of the British Isles and countries where migration from Britain occurred (the United States, Australia, New Zealand, and Canada) have a greater incidence. The disease is almost nonexistent in Asia and in the native African and South American populations.
Etiologic Factors
The cause of Paget’s disease is unknown. Sir James Paget, who described the disease more than a century ago, thought it was likely a bone infection. Paget’s disease is often inherited in an autosomal dominant pattern, but the genetic basis of the condition is not well understood.20,64
Family studies have localized gene susceptibility to Paget’s disease at three different chromosomal regions, although these defects do not appear to account for the majority of cases. The results of a number of studies suggest that variations in the protein p62 affect the function of osteoclasts and that the production of p62 is directed by the sequestosome 1 gene. Genomic studies of human sequestosome 1 gene containing a Paget’s disease mutation are ongoing. Because of the late age of onset of this disease, finding large families with a number of generations with the disease has been difficult and has hindered progress in research.
Environmental factors, specifically slow viruses that take years to progress to a point where symptoms become evident, may play a role in the development of Paget’s disease58; the hereditary factor may be the reason family members are susceptible to the suspected virus.116 Exactly how the viruses affect osteoclasts remains unknown. Some bacteria can lyse bone, and others produce osteolytic materials or activate osteoclasts, but the mechanisms by which this happens is obscure.23
Paget’s disease has become less prevalent and people are presenting even later and with less severe disease than before, thus environmental factors may be an important etiologic factor in this disease. People born recently and presenting with Paget’s disease have substantially less severe bone disease.18
Pathogenesis
Traditionally, Paget’s disease has been considered a disorder of the osteoclasts (bone-resorbing cells) rather than the osteoblasts (bone-forming cells). However, scientists now understand that osteoblasts are major regulators of osteoclast development and function, and a tightly coupled pathway of communication and collaboration is evident between these two groups of cells. The exact mechanism by which these two cell types contribute to the formation of Paget’s disease is under investigation.
An initial osteoclastic, resorptive stage where abnormal osteoclasts proliferate unrestrained is evident. The bone resorption is so rapid that osteoblastic activity cannot keep up and fibrous tissue replaces bone. Radiographically, the resultant lytic areas are sharply defined and flame-or wedge-shaped. The initial resorption is followed by abnormal regeneration called the osteoblastic sclerotic phase.
In the sclerotic phase, the normal cancellous architecture is replaced by coarse, thickened struts of trabecular bone, and the cortical bone is irregularly thickened, rough, and pitted. The abnormal arrangement of the lamellar bone, separated by so-called cement lines, gives the bone the look of a mosaic. Although heavily calcified, the bone is now enlarged but weakened with a chaotic woven pattern, rather than the well-organized lamellar structure seen in normal bone.
Involvement of the vertebral bodies presents with a picture-frame appearance radiographically as the cortical shell and endplates become greatly exaggerated in comparison to the coarse, cancellous bone portion of the vertebral body (Fig. 24-8). The final stage of the disease is characterized by little cellular activity.
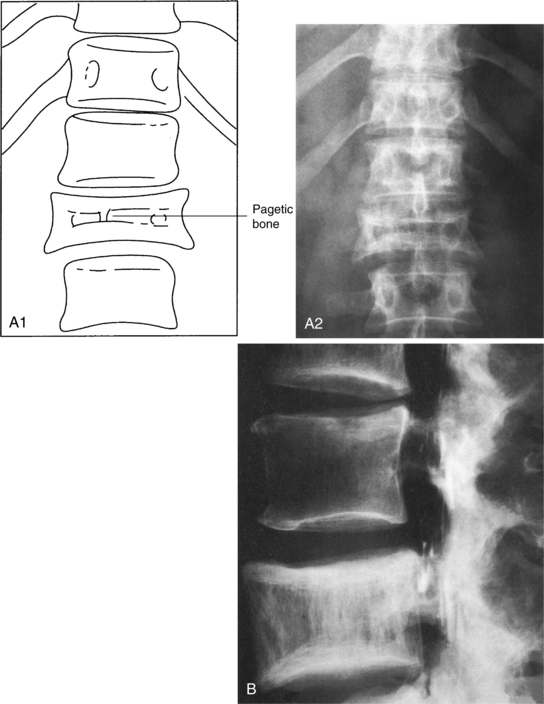
Figure 24-8 A, Clinical radiograph of the spine shows a solitary focus of pagetic bone. A loss of height of the vertebra (compression), some increases in the width of the vertebra, and a typical “picture-frame” appearance are evident. B, Radiograph of the lumbar spine in a 66-year-old male shows enlargement of L4 with coarsening of the trabecular pattern and a margin sclerosis, the so-called picture-framing characteristic of Paget’s disease. (From Bullough P: Orthopaedic pathology, ed 3, London, 1997, Mosby-Wolfe; B, Courtesy Dr. Alex Norman.)
Clinical Manifestations
Paget’s disease begins insidiously and progresses slowly; in mild cases, a person may have a few symptoms or may be symptom free over a very long period, eventually presenting with bone pain and skeletal deformities. When Paget’s disease is active in several bones, overactive osteoclasts can release enough calcium in the blood to cause hypercalcemia with fatigue, weakness, loss of appetite, abdominal pain, and/or constipation.3
Musculoskeletal.: The progressive deossification that weakens the bony structure primarily affects the axial skeleton. The lesions occur at multiple sites, particularly the skull, spine, pelvis, femurs, and tibias. Pathologic fractures can occur in any bone (Fig. 24-9), especially in the proximal femurs, pelvis, and lumbar spine.
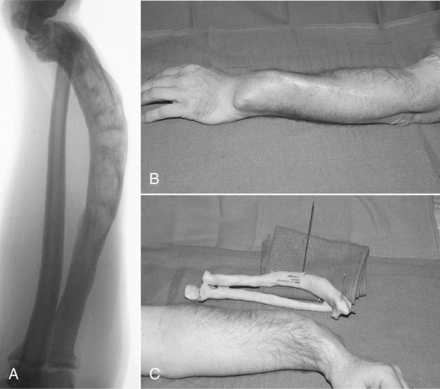
Figure 24-9 Paget’s disease. A complex malunion of the radius in a 62-year-old man after several fractures. A, The presenting lateral radiograph of the severe deformity. B, The clinical appearance. C, A bone model made from computed tomographic scans and used for preoperative planning for correction of the deformity. (From Browner BD: Skeletal trauma: basic science, management, and reconstruction, ed 3, Philadelphia, 2003, WB Saunders.)
Affected bones change in shape, size, and direction, causing bone pain, arthritis, deformities, and fractures (Box 24-7). The most common presenting symptom is pain, which may be of headache, radicular, osteoarthritic, muscular, or other skeletal origin. Direct pain from periosteal irritation of involved bones is deep and boring, worse at night, and reduced but not eliminated with activity. In some people, this direct pain may be referred to nearby muscles and joints.144
Clients may also experience fatigue, lightheadedness, and general stiffness. New onset of pain may be related to pathologic fracture of the vertebral bodies, pelvis, or long bones. Damage to the cartilage of joints adjacent to the affected bone and distortion of the normal joint alignment from bony changes may lead to osteoarthritis. This is often more disabling than the Paget’s disease itself and will not respond to treatment of the underlying bone disease.33
Clinical findings also include postural deformities such as increased thoracic kyphosis and bowing of the femurs and tibias (Fig. 24-10). Bony softening of the femoral neck can cause coxa vara (reduced angle of the femoral neck) and may result in a waddling gait. These changes may produce increased local mechanical stresses resulting in pain.
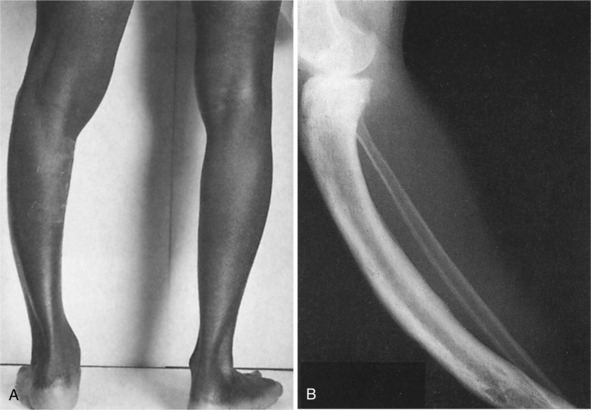
Figure 24-10 A, Bowing of the leg is often seen in Paget’s disease. B, Radiograph of a person with Paget’s disease affecting the tibia (but not the fibula). Overgrowth has resulted in an increase in length of the tibia, associated with bowing. Irregularity of both the periosteal and endosteal surfaces is shown. (From Bullough P: Orthopaedic pathology, ed 3, London, 1997, Mosby-Wolfe.)
Neurologic.: Paget’s disease of the skull and spinal column can produce neurologic complications as a result of either direct impingement (myelopathy) or ischemia related to a pagetic steal syndrome (hypervascular pagetic bone “steals” blood from the neural tissue).144
Typical neurologic deficits may include eighth cranial nerve involvement (most common) with hearing loss related to involvement of the ossicles or bony foraminal encroachment. Headaches may occur if the skull is involved, and the forehead may enlarge as the amount of bone in the skull expands and the foramen for the cranial nerves gets smaller (Fig. 24-11). Affected individuals also report tinnitus, vertigo, and hearing loss.
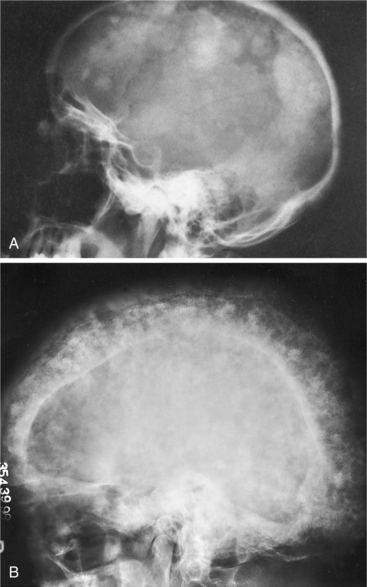
Figure 24-11 Clinical radiographs of the skull in later stages of Paget’s disease. A, Marked patchy sclerosis appears in the bone, the organized architecture is lost, and the bone becomes extremely thick, on occasion several times thicker than normal. B, Advanced involvement of the skull with marked thickening of the entire vault, areas of osteolysis, and patchy new bone formation resulting in a “cotton-wool” appearance called osteoporosis circumscripta cranii. This person experienced progressive hearing loss. (A from Bullough P: Orthopaedic pathology, ed 3, London, 1997, Mosby-Wolfe; B from Goldman L: Cecil textbook of medicine, ed 22, Philadelphia, 2004, WB Saunders.)
Other nerve or spinal cord compression syndromes may occur as enlarged pagetic bones put pressure on various nerve structures. Findings may include myelopathy, spinal stenosis, radiculoneuropathy, cauda equina syndrome, peripheral nerve entrapment, carpal and tarsal tunnel syndromes, and any of the effects of cranial nerve compression.144 Thickening of the skull causes compression of the auditory nerves most often; other cranial nerves are only rarely involved in Paget’s disease.
Cardiovascular.: Other findings may include mental deterioration (Paget’s disease causes reduced blood flow to the brain) and cardiovascular disease (rare). The cardiovascular disease is due to vasodilation of blood vessels in the bones and skin and subcutaneous tissues overlying the affected bones. When one-third to one-half of the skeleton is involved, an increase in cardiac output may be severe enough to cause heart failure. This is the most common cause of death in people with advanced Paget’s disease.144
MEDICAL MANAGEMENT
Because Paget’s disease progresses slowly and the severity of the disease varies considerably among individuals, it may be many years before a diagnosis is made or the person may be misdiagnosed with arthritis or other disorders. In fact, the diagnosis is often made incidentally on the basis of radiographs or laboratory tests done for other reasons.
Researchers are trying to develop a screening test that could be used to detect those at risk of Paget’s disease at an early age. At the present time, siblings and children of someone with Paget’s disease may have a standard alkaline phosphatase blood test every 2 or 3 years. Alkaline phosphatase is an enzyme that is produced by bone cells and over-produced by pagetic bone. Alkaline phosphatase levels can be used to diagnose Paget’s disease and to monitor response to therapy (see Table 40-5). If the alkaline phosphatase level is above normal, other tests, such as the bone-specific alkaline phosphatase test, bone scan, or radiographic examination, can be performed.
If the disease is advanced, the diagnosis is made based on the characteristic bone deformities and radiologic bony changes. Bone scans are positive only if the disease is active and marked by rapid bone turnover but provide information that helps determine the extent and activity of the condition. Bone scans must be confirmed by radiographic examination as other conditions are also accompanied by a metabolically active lesion. A bone biopsy may occasionally be done to make a differential diagnosis ruling out hyperparathyroidism, bone metastasis, multiple myeloma, and fibrous dysplasia.
TREATMENT.
The goal of treatment is to normalize Paget’s disease activity for a prolonged period. Because of the osteoclast involvement, inhibitors of bone resorption (nitrogen-containing bisphosphonates such as oral alendronate or risedronate) are the first line of treatment. These pharmacologic agents (see Box 24-3) decrease bone turnover through the inhibition of osteoclastic activity, improve bone density, and reduce fracture incidence.
These effects last after stopping administration of the agent, thus providing clinical and biochemical and long-lasting remissions in the majority of people.46 Treatment response is assessed by the extent of reduction of biochemical markers of bone turnover, especially plasma total alkaline phosphatase. The goal of treatment is to induce full remission (i.e., normal levels of alkaline phosphatase) and prevent complications of the disease.88 Some experts in the field are recommending treatment of asymptomatic people who have active disease at sites where complications are likely to develop, although no data are available at this time to prove that complications can be prevented by pharmacologic intervention.138
Individuals under long-term oral bisphosphonate use are treated with caution because of the potential side effects, especially recently reported osteonecrosis of the jaw (ONJ).75 Well-controlled, prospective clinical trials on the effect of oral bisphosphonates on bone are needed to determine which individuals may be at risk for such complications.99,145 At the present time, it looks like ONJ may occur in individuals receiving intravenous bisphosphonates for an underlying malignant condition. Further investigation is ongoing.75
Management of Paget’s disease depends on the degree of pain and extent of the pathologic changes. Nonsteroidal and other antiinflammatory agents are used to control the pain. Surgical intervention may be indicated for reasons other than fracture repair. Interventions include occipital craniectomy to relieve basilar and nerve compression, tibial osteotomy if the deformity is severe, and joint replacement(s) if severe degenerative joint disease is present (Fig. 24-12).
Figure 24-12 Extensive Paget’s disease of the acetabulum and femur in an 82-year-old man. A, Protrusion deformity and varus femoral neck are seen. B, After total hip replacement. Acetabulum required autogenous bone graft from femoral head. Cementless acetabular component appears bone ingrown 5 years after surgery. (From Canale ST: Campbell’s operative orthopaedics, ed 10, St Louis, 2003, Mosby.)
PROGNOSIS.
The course of Paget’s disease varies widely from individual to individual. It may be completely stable and asymptomatic, or at the other end of the continuum, rapid progression can occur. The outlook is generally good, particularly if treatment is given before major changes have occurred. Biochemical remission with bisphosphonates is achievable in a majority of individuals.126
Although rarely fatal by itself, Paget’s disease is accompanied by osteogenic sarcoma in less than 1% of all Paget’s disease cases; this figure represents an increase in risk that is several thousand-fold higher than the general population.39,40 There may be a link between the putative gene for Paget’s disease and the tumor suppressor gene for osteosarcoma on chromosome 18q.4
Peak incidence of the associated sarcoma occurs in the seventh and eighth decades. Tumors tend to be high grade and found in areas commonly affected by Paget’s disease, especially in the pelvis, femur, humerus, and skull. Metastases has often occurred at the time of discovery.
Prognosis for individuals with Paget sarcoma is poor and is unrelated to the site or stage of presentation.22 Little progress has occurred in the treatment of Paget’s sarcoma over the years despite improvement in the treatment of standard osteosarcoma.67 See Chapter 26 for an overview of osteogenic sarcoma.
References
1. Adler, RA. Epidemiology and pathophysiology of osteoporosis in men. Curr Osteoporos Rep. 2006;4(3):110–115.
2. Adler, RA. The need for increasing awareness of osteoporosis in men. Clin Cornerstone. 2006;8(suppl 3):7–13.
3. American Academy of Orthopaedic Surgeons (AAOS) Paget’s disease of bone. Available on-line at http://orthoinfo.aaos.org/ Accessed July 27, 2007
4. ME Betancourt-Albrecht A man with pain in his bones. Available on-line at www.medscape.com/viewarticle/445158 Accessed July 27, 2007
5. Black, DM, Delmas, PD, Eastell, R, et al. Once-yearly zoledronic acid for treatment of postmenopausal osteoporosis. N Engl J Med. 2007;356(18):1809–1822.
6. Block, JE, et al. Determinants of bone density among athletes engaged in weight-bearing and non-weight-bearing activity. J Appl Physiol. 1989;67:1100–1105.
7. Bonnick, SL. Osteoporosis in men and women. Clin Cornerstone. 2006;8(1):28–39.
8. Boonen, S. The diagnosis and treatment of male osteoporosis: defining, assessing, and preventing skeletal fragility in men. Eur J Intern Med. 2007;18(1):6–17.
9. Bravo, G, et al. Impact of a 12-month exercise program on the physical and psychological health of osteopenic women. JAGS. 1996;44:756–762.
10. Brown, SA. Drug insight: the use of bisphosphonates for the prevention and treatment of osteoporosis in men. Nat Clin Pract Urol. 2007;4(6):310–320.
11. Cardinale, M. Vibration exercise makes your muscles and bones stronger: fact or fiction? J Br Menopause Soc. 2006;12(1):12–18.
12. Cavalieri, RJ. Nursing presence in osteoporosis research. AJN. 2001;98(6):60–63.
13. Cavanaugh, DJ, Cann, CE. Brisk walking does not stop bone loss in postmenopausal women. Bone. 1988;9:201–204.
14. Christiansen, C, Christiansen, MS, Transbol, I. Bone mass in postmenopausal women after withdrawal of oestrogen/gestagen replacement therapy. Lancet. 1998;1:459–461.
15. Cizza, G. Depression: a major, unrecognized risk factor for osteoporosis. Trends Endocrinol Metab. 2001;12(5):198–203.
16. Croarkin, E. Osteopenia: implications for physical therapists managing patients of all ages. PT Mag. 2001;9(2):80–89.
17. Cummings, S, et al. Epidemiology of osteoporosis and osteoporotic fractures. Epidemiol Rev. 1985;7:178–208.
18. Cundy, T. Is the prevalence of Paget’s disease of bone decreasing? J Bone Miner Res. 2006;21(suppl 2):9–13.
19. Cussler, EC. Exercise frequency and calcium intake predict 4-year bone changes in postmenopausal women. Osteoporosis Int. 2005;16(12):2129–2141.
20. Daroszewska, A. Genetics of Paget’s disease of bone. Clin Sci (Lond). 2005;109(3):257–263.
21. Devereux, K. Effects of a water-based program on women 65 years and over: a randomized controlled trial. Aust J Physiother. 2005;51(2):102–108.
22. Deyrup, AT. Sarcomas arising in Paget disease of bone: a clinicopathologic analysis of 70 cases. Arch Pathol Lab Med. 2007;131(6):942–946.
23. Dickinson, CJ. Mouth bacteria as the cause of Paget’s disease of bone. Med Hypotheses. 1999;52(3):209–212.
24. Dieppe, P, Tobias, J. Bone and joint aging. In Tallis RC, Fillit HM, Brocklehurst JC, eds.: Geriatric medicine and gerontology, ed 5, Edinburgh: Churchill Livingstone, 1998.
25. Drinkwater, BL. Weight-bearing exercise and bone mass. In: Kraft GH, Matkovic V, eds. Physical medicine and rehabilitation clinics of North America. Philadelphia: WB Saunders, 1995.
26. Duncan, EL, et al. Suggestive linkage of the parathyroid receptor type 1 to osteoporosis. J Bone Miner Res. 1999;14(12):1993–1999.
27. Dunn, CJ, Goa, KL. Risedronate: a review of its pharmacological properties and clinical use in resorptive bone disease. Drugs. 2001;61(5):685–712.
28. Elliott, ME. Osteoporosis assessment strategies for male nursing home residents. Maturitas. 2004;48(3):225–233.
29. Ettinger, B. Effects of ultralow-dose transdermal estradiol on bone mineral density: a randomized clinical trial. Obstet Gynecol. 2004;104(3):443–451.
30. Fahy, UM, et al. Osteoporotic fractures in young amenorrheic women. Acta Obstet Gynecol Scand. 1994;73:417–419.
31. Felson, DT, Zhang, Y, Hannan, MT, et al. The effect of postmenopausal estrogen therapy on bone density in elderly women. N Engl J Med. 1993;329(16):1141–1146.
32. Fisher, J, et al. Maternal milk consumption predicts the tradeoff between milk and soft drinks in young girls’ diets. J Nutr. 2001;131(2):246–250.
33. Francis, RM. Metabolic bone disease. In Tallis RC, Fillit HM, Brocklehurst JC, eds.: Geriatric medicine and gerontology, ed 5, Edinburgh: Churchill Livingstone, 1998.
34. Frost, HM. Suggested fundamentals concepts in skeletal physiology. Calcif Tissue Int. 1993;52:1–4.
35. K Gray Current concepts in osteoporosis management. Presented at the American Association of Clinical Endocrinologists Tenth Annual Meeting and Clinical Congress, San Antonio, TX, May 2-6, 2001
36. Grinspoon, S, et al. Severity of osteopenia in estrogen-deficient women with anorexia and hypothalamic amenorrhea. J Clin Endocrinol Metab. 1999;84(6):2049–2055.
37. Gusi, N. Low-frequency vibratory exercise reduces the risk of bone fracture more than walking: a randomized controlled trial. BMC Musculoskeletal Disord. 2006;307:92.
38. Haczynski, J, Jakimiuk, A. Vertebral fractures: a hidden problem of osteoporosis. Med Sci Monit. 2001;7(5):1108–1117.
39. Hansen, MF. Osteosarcoma in Paget’s disease of bone. J Bone Miner Re. 2006;21(suppl 2):58–63.
40. Hansen, MF, Nellissery, MJ, Bhatia, P. Common mechanisms of osteosarcoma and Paget’s disease. J Bone Miner Res. 1999;14(suppl 2):39–44.
41. Harris, ST. The effect of combined risedronate and hormone replacement therapies on bone mineral density in postmenopausal women. J Clin Endocrin Metab. 2001;86(5):1890–1897.
42. Harvard Women’s Health Watch. Exploring the depression-bone connection. Harvard Womens Health Watch. 2007;14(10):1–3.
43. Healthy People 2010 Objectives-osteoporosis, 2007. Available on-line at http://www.health.gov/healthypeople/ Accessed July 30, 2007
44. Heany, RP. Nutrition and bone mass. In: Kraft GH, Matkovic V, eds. Physical medicine and rehabilitation clinics of North America. Philadelphia: WB Saunders, 1995.
45. Hernandez, L. Extraintestinal manifestations of celiac disease. Curr Gastroenterol Rep. 2006;8(5):383–389.
46. Hoidrup, S. Alcohol intake, beverage preference, and risk of hip fracture in men and women (Copenhagen Centre for Prospective Population Studies). Am J Epidemiol. 1999;149:993–1001.
47. Huang, AJ, Ettinger, B, Vittinghoff, E, et al. Endogenous estrogen levels and the effects of ultra low-dose transdermal estradiol therapy on bone turnover and bone density in postmenopausal women. J Bone Miner Res. 2007;22(11):1791–1797.
48. Iwamoto, J. Effect of whole-body vibration exercise on lumbar bone mineral density, bone turnover, and chronic back pain in post-menopausal osteoporotic women treated with alendronate. Aging Clin Exp Res. 2005;17(2):157–163.
49. Jadhav, SB. Statins and osteoporosis: new role for old drugs. J Pharm Pharmacol. 2006;58(1):3–18.
50. Javaid, MK, Cooper, C. How to prevent fractures in the individual with osteoporosis. Best Pract Res Clin Rheumatol. 2001;15(3):497–515.
51. Jones, G, Riley, MD, Dwyer, T. Maternal diet during pregnancy is associated with bone mineral density in children: a longitudinal study. Eur J Clin Nutr. 2000;54(10):749–756.
52. Kanis, JA. Alcohol intake as a risk factor for fracture. Osteoporosis Int. 2004;16:737–742.
53. Kaplan, FS. Surgical management of Paget’s disease. J Bone Miner Res. 1999;14(suppl 2):34–38.
54. Kelepouris, NI. Severe osteoporosis in men. Ann Intern Med. 1995;123:452–460.
55. Keller, TS. Prediction of osteoporotic spinal deformity. Spine. 2003;28(5):455–462.
56. Kostenuik, PJ, Shalhoub, V. Osteoprotegerin: a physiologic and pharmacologic inhibitor of bone resorption. Curr Pharm Des. 2001;7(8):613–635.
57. Kroth, PJ. Undertreatment of osteoporosis in women, based on detection of vertebral compression fractures on chest radiography. Am J Geriatr Pharmacother. 2004;2(2):112–118.
58. Kurihara, N. Experimental models of Paget’s disease. J Bone Miner Res. 2006;21(suppl 2):55–57.
59. Lane, JM, Nydick, M. Osteoporosis: current modes of prevention and treatment. J Am Acad Orthop Surg. 1999;7(1):19–31.
60. Lata, PF. Patient assessment in the diagnosis, prevention, and treatment of osteoporosis. Nutr Clin Pract. 2007;22(3):261–275.
61. Layne, JE, Nelson, ME. The effects of progressive resistance training on bone density: a review. Med Sci Sports Exerc. 1999;31(1):25–30.
62. Liu, XD, et al. Cigarette smoke inhibits osteogenic differentiation and proliferation of human osteoprogenitor cells in monolayer and three-dimensional collagen gel culture. J Lab Clin Med. 2001;137(3):208–219.
63. Lories, RJ, Luyten, FP. Osteoprotegerin and osteoprotegerin-ligand balance: a new paradigm in bone metabolism providing new therapeutic targets. Clin Rheumatol. 2001;20(1):3–9.
64. Lucas, GJ. Contribution of genetic factors to the pathogenesis of Paget’s disease of bone and related disorders. J Bone Miner Res. 2006;21(suppl 2):31–37.
65. Ludvigsson, JF. Coeliac disease and the risk of fractures-a general population-based cohort study. Aliment Pharmacol Ther. 2007;25(3):273–285.
66. Maciaszek, J. Effect of Tai Chi on body balance: randomized controlled trial in men with osteopenia or osteoporosis. Am J Chin Med. 2007;35(1):1–9.
67. Mankin, HJ. Paget’s sarcoma: a historical outcome review. Clin Orthop Relat Res. 2005;438:97–102.
68. Marques-Deak, A. Brain-immune interactions and disease susceptibility. Mol Psychiatry. 2005;10(3):239–250.
69. Meeks, S Walk tall: a program for the prevention and treatment of osteoporosis. Gainesville, FL: Triad; 1999. Available on-line at http://www.triadpublishing.com or by calling (800) 525-6902.
70. Michelson, D. Bone mineral density in women with depression. N Engl J Med. 1996;335(16):1176–1181.
71. Millar, AL, Hunter, D. Osteopenia: Its relation to menstrual disorders in female athletes. JOSPT. 1990;11(8):351–354.
72. Miller, PD. New possibilities for diagnosis and treatment of osteoporosis. Int J Fertil Womens Med. 2001;46(4):215–221.
73. Mody, N, et al. Oxidative stress modulates osteoblastic differentiation of vascular and bone cells. Free Radic Biol Med. 2001;31(4):509–519.
74. Mosekilde, L. Age-related changes in bone mass, structure, and strength-effects of loading. Z Rheumatol. 2000;59(suppl 1):1–9.
75. Murad, OM. Bisphosphonates and osteonecrosis of the jaw: a retrospective study. Endocr Pract. 2007;13(3):232–238.
76. National Institutes for Health (NIH) Consensus Statement Osteoporosis, prevention, diagnosis, and therapy Bethesda, MD, March 27-29, 2000
77. National Osteoporosis Foundation (NOF) About osteoporosis (2007). Available on-line at http://www.nof.org/ Accessed June 6, 2007
78. National Osteoporosis Foundation (NOF) Men and osteoporosis (2007). Available on-line at http://www.nof.org/ Accessed June 6, 2007
79. National Osteoporosis Foundation (NOF) Osteoporosis. Washington DC: NOF; 2001. Available on-line at http://www.nog.org
80. National Osteoporosis Foundation (NOF). Physician’s guide to prevention and treatment of osteoporosis. Washington DC: NOF, 1998.
81. Nelson, M, Wernick, S. Strong women, strong bones: everything you need to prevent, treat, and beat osteoporosis. New York: Putnam, 2000.
82. Neuner, JM. Bone density testing in older women and its association with patient age. J Am Geriatr Soc. 2006;54(3):485–489.
83. Nguyen, H-V. Osteoporotic vertebral burst fractures with neurologic compromise. J Spinal Disorders. 2003;16(1):10–19.
84. Nordin, C, et al. The relative contributions of age and years since menopause to postmenopausal bone loss. J Clin Endocrinol Metab. 1990;70:83–88.
85. North American Menopause Society. Available on-line at http://www.menopause.org or by calling (440) 442-7550
86. Notelovitz, M, et al. Estrogen therapy and variable-resistance weight training increase bone mineral in surgically menopausal women. J Bone Min Res. 1991;6:583–590.
87. Notelevitz, M, Tonnessen, D Stand tall: every woman’s guide to the prevention and treatment of osteoporosis. Gainesville, FL: Triad; 1999. Available by calling (800) 525-6902 (www.triadpublishing.com).
88. Orcel, P. Paget’s disease and its therapeutic management. Presse Med. 2005;34(8):612–616.
89. Orwall, ES. The influence of exercise on osteoporosis and skeletal health. In: Goldberg L, Elliot DL, eds. Exercise for prevention and treatment of illness. Philadelphia: FA Davis, 1994.
90. Osteoporosis functional disability questionnaire. J Gerontology Med Sci. 1995;50A(2):M91–98.
91. Papaioannou, A. Patient adherence to osteoporosis medications: problems, consequences and management strategies. Drugs Aging. 2007;24(1):37–55.
92. Parhami, F, et al. Atherogenic high-fat diet reduces bone mineralization in mice. J Bone Miner Res. 2001;16(1):182–188.
93. Prior, JC. Spinal bone loss and ovulatory disturbances. N Engl J Med. 1990;323:1221–1227.
94. Qin, L. Regular Tai Chi Chuan exercise may retard bone loss in postmenopausal women: a case-control study. Arch Phys Med Rehabil. 2002;83(10):1355–1359.
95. Raisz, LG. Clinical practice: screening for osteoporosis. N Eng J Med. 2005;353(2):164–171.
96. Raisz, LG. Physiology and pathophysiology of bone remodeling. Clin Chem. 1999;45(10):1885.
97. Randell, AG, Bhalerao, N, Nguygen, TV, et al. Quality of life in osteoporosis: reliability, consistency, and validity of the Osteoporosis Assessment Questionnaire. J Rheumatol. 1998;25:1171–1179.
98. Reginato, AJ, et al. Musculoskeletal manifestations of osteomalacia: report of 26 cases and literature review. Semin Arthritis Rheum. 1999;28(5):287–304.
99. Reid, IR. Emerging issues with bisphosphonates. Rheum Dis Clin North Am. 2006;32(4):691–702.
100. Richards, JB. Effect of selective serotonin reuptake inhibitors on the risk of fracture. Arch Intern Med. 2007;167(2):188–194.
101. Riggs, BL, Melton, LJ. Evidence for two distinct syndromes of involutional osteoporosis. Am J Med. 1992;75:899–901.
102. Rubenstein, B. Update on osteoporosis treatment: new and better approaches. J Musculoskel Med. 2007;24(6):264–268.
103. Rubin, CT, Lanyon, CE. Regulation of bone mass by mechanical strain magnitude. Calcif Tissue Int. 1985;37:411–417.
104. Russell, G, et al. Clinical disorders of bone resorption. Novartis Found Symp. 2001;232:251–267.
105. Rutherford, OM. Is there a role for exercise in the prevention of osteoporotic fractures? Br J Sports Med. 1999;33(6):378–386.
106. Salkeld, G, et al. Quality of life related to fear of falling and hip fracture in older women: a time trade off study. BMJ. 2000;320(7243):1206.
107. Salter, RB. Textbook of disorders and injuries of the musculoskeletal system, ed 3. Baltimore: Williams and Wilkins, 1999.
108. Sammaritano, LR. Osteoporosis in patients with rheumatic diseases: prevention and management. J Musculoskel Med. 2001;18(5):240–249.
109. Sanborn, CF, et al. Disordered eating and the female athlete triad. Clin Sports Med. 2000;19(2):199–213.
110. Savage, JS. Parental influence on eating behaviors: conception to adolescence. J Law Med Ethics. 2007;35(1):22–34.
111. Schreiber, MD, et al. NAMS Fellowship findings: dietary inclusion of whole soy foods results in significant reductions in clinical risk factors for osteoporosis and cardiovascular disease in normal postmenopausal women. Menopause. 2001;8(5):384–392.
112. Schweiger, U, et al. Lumbar bone mineral density in patients with major depression: evidence of increased bone loss at follow-up. Am J Psychiatry. 2000;157(1):118–120.
113. Sebastian, A, et al. Dietary ratio of animal to vegetable protein and rate of bone loss and risk of fracture in postmenopausal women. Am J Clin Nutr. 2001;74(3):411–412.
114. Seeman, E. The structural basis of bone fragility in men. Bone. 1999;25(1):143–147.
115. Sellmeyer, DE, et al. A high ratio of dietary animal to vegetable protein increases the rate of bone loss and the risk of fracture in postmenopausal women (Study of Osteoporotic Fractures Research Group). Am J Clin Nutr. 2001;73(1):118–122.
116. Shaw, J. The genetic analysis of Paget’s disease in a large pedigree. Newsletter Paget Found. 2001;23(1):3.
117. Shaw, JM, Witzke, KA, Exercise for skeletal health and osteoporosis prevention. American College of Sports Medicine: Resource manual for guidelines for exercise testing and prescription. ed 4. Philadelphia: Lippincott Williams and Wilkins; 2001.
118. Shen, M, Kim, Y. Osteoporotic vertebral compression fractures: a review of current surgical management techniques. Am J Ortho. 2007;36(5):241–248.
119. Shipp, KM. Questions and answers about exercise and Paget’s disease) update). Newsletter Paget Found. 2005;27(1):10.
120. Silverman, SL. Selecting patients for osteoporosis therapy: a new approach. J Musculoskel Med. 2007;24(5):207–211.
121. Sinaki, M. Exercise and physical therapy. In: Riggs BL, Melton JB, eds. Osteoporosis: etiology, diagnosis, and management. New York: Raven Press, 1988.
122. Sinaki, M. Musculoskeletal challenges of osteoporosis. Aging. 1998;10(3):249–262.
123. Sinaki, M, McPhee, MC, Hodgson, SF. Relationship between bone mineral density of spine and strength of back extensors in healthy postmenopausal women. Mayo Clin Proc. 1986;61:116–122.
124. Sinaki, M, Mikkelsen, BA. Postmenopausal spinal osteoporosis: flexion vs extension exercises. Arch Phys Med Rehabil. 1988;69:277–280.
125. Sinaki, M, et al. Effect of gender, age, and anthropometry on axial and appendicular muscle strength. Am J Phys Med Rehabil. 2001;80(5):330–338.
126. Siris, ES. Medical management of Paget’s disease of bone: indications for treatment and review of current therapies. J Bone Miner Res. 2006;21(suppl 2):94–98.
127. Sirtori, CR. Risks and benefits of soy phytoestrogens in cardiovascular diseases, cancer, climacteric symptoms and osteoporosis. Drug Saf. 2001;4(9):665–682.
128. Skerry, TM. Mechanical loading and bone: what sort of exercise is beneficial to the skeleton? Bone. 1997;20:179–181.
129. Spangler, JG, Quandt, S, Bell, RA. Smokeless tobacco and osteoporosis: a new relationship? Med Hypotheses. 2001;56(5):553–557.
130. Stenson, WF. Increased prevalence of celiac disease and need for routine screening among patients with osteoporosis. Arch Intern Med. 2005;165(4):393–399.
131. Summers, GD. Osteoporosis in men. Radiography. 2001;7(2):119–123.
132. Svejda, P. Statins and osteoporosis. Vnitr Lek. 2006;52(12):1190–1193.
133. Swan, KG, et al. Osteoporosis in men: a serious but under-recognized problem. J Musculoskel Med. 2001;18(6):310–316.
134. Swezey, RL. Exercise for osteoporosis: is walking enough? Spine. 1996;21:2809–2813.
135. Swezey, RL. Preventing osteoporotic fractures: the role of exercise, posture, and safety. J Musculoskel Med. 1997;14(4):9–28.
136. Taafe, DR, et al. Differential effects of swimming versus weight-bearing activity on bone mineral status of eumenorrheic athletes. J Bone Miner Res. 1995;10:586–593.
137. Teitelbaum, SL. Osteoclasts: what do they do and how do they do it? Am J Pathol. 2007;170(2):427–435.
138. Tiegs, RD. Paget’s disease of bone: indications for treatment and goals of therapy. Clin Ther. 1997;19(6):1309–1329.
139. Tomaczak, RL, VanCourt, R. Metatarsal insufficiency fractures in previously undiagnosed osteoporosis patients. J Foot Ankle Surg. 2000;39(3):174–183.
140. Tosteson, AN. Early discontinuation of treatment for osteoporosis. Am J Med. 2003;115(3):209–216.
141. Tracz, MJ. Testosterone use in men and its effects on bone health. A systematic review and meta-analysis of randomized placebo-controlled trials. J Clin Endocrinol Metab. 2006;91(6):2011–2016.
142. Trivedi, DP, Khaw, KT. Bone mineral density at the hip predicts mortality in elderly men. Osteopor Int. 2001;12(4):259–265.
143. Vargo, MM. Osteoporosis: strategies for prevention and treatment. J Musculoskel Med. 1995;12(5):19–30.
144. Wallach, S. Identifying and controlling Paget’s disease. J Musculoskel Med. 1997;14(6):66–82.
145. Wang, HL. Effect of long-term oral bisphosphonates on implant wound healing: literature review and case report. J Periodontol. 2007;78(3):373–376.
146. Warden, SJ. Does exercise during growth influence osteoporotic fracture risk later in life? J Musculoskel Neuronal Interact. 2005;5(4):344–346.
147. Wayne, PM. The effects of Tai Chi on bone mineral density in postmenopausal women: a systematic review. Arch Phys Med Rehabil. 2007;88(5):673–680.
148. Weinstein, RS, Manolagas, SC. Apoptosis and osteoporosis. Am J Med. 2000;108(2):153–164.
149. Wezeman, FH, et al. Chronic alcohol consumption during male rate adolescence impairs skeletal development through effects on osteoblast gene expression, bone mineral density, and bone strength. Alcohol Clin Exp Res. 1999;23(9):1534–1542.
150. Wirganowicz, PZ, Lane, JM. Postmenopausal osteoporosis: an update for the clinician. J Musculoskel Med. 1996;13(10):29–38.
151. Woods, EN. Building better bones: managing and preventing osteoporosis. PT Magazine. 1996;4(5):40–42.
152. Wright, VJ. Osteoporosis in men. J Am Acad Orthop Surg. 2006;14:347–353.
153. Yirmiya, R. Depression induces bone loss through stimulation of the sympathetic nervous system. Proc Natl Acad Sci. 2007;103(45):16876–16881.
154. Zhao, H. Critical role of beta3 integrin in experimental postmenopausal osteoporosis. J Bone Miner Res. 2005;20(12):2116–2123.