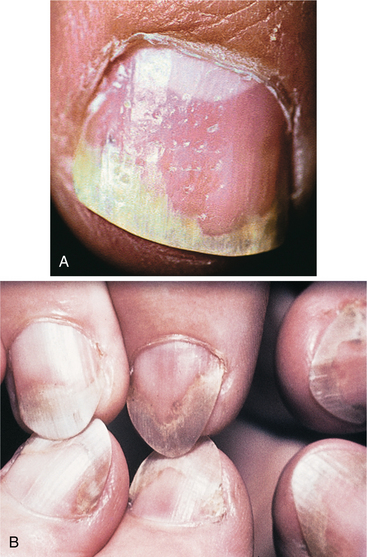
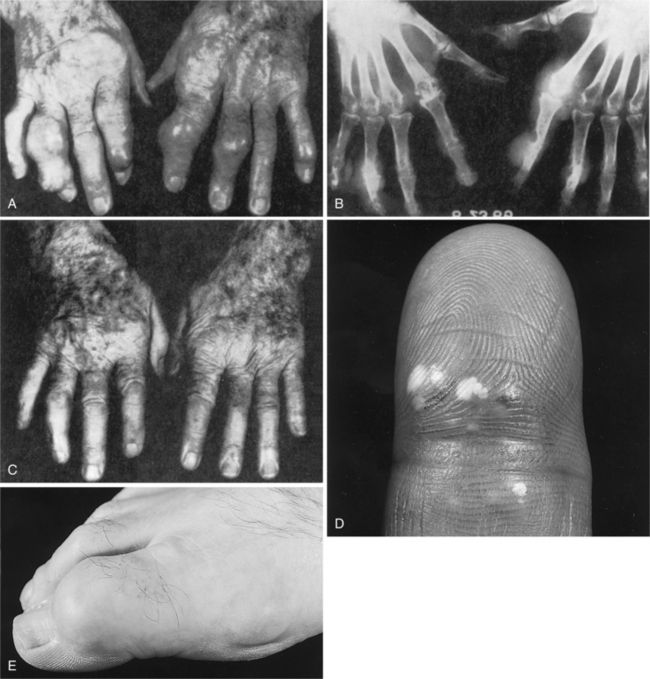
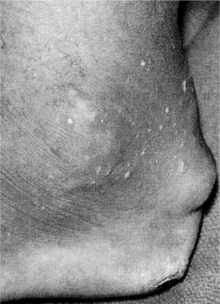
Rheumatoid Arthritis
PREFERRED PRACTICE PATTERNS
4A:
Primary Prevention/Risk Reduction for Skeletal Demineralization (osteopenia, osteoporosis)
4B:
Impaired Posture (cervical involvement)
4C:
Impaired Muscle Performance
4D:
Impaired Joint Mobility, Motor Function, Muscle Performance, and Range of Motion Associated with Connective Tissue Dysfunction
4H:
Impaired Joint Mobility, Motor Function, Muscle Performance, and Range of Motion Associated with Joint Arthroplasty
4I:
Impaired Joint Mobility, Motor Function, Muscle Performance, and Range of Motion Associated with Bony or Soft Tissue Surgery (tenosynovectomy, tendon reconstruction)
5H:
Impaired Motor Function, Peripheral Nerve Integrity, and Sensory Integrity Associated with Nonprogressive Disorders of the Spinal Cord (cervical spine)
6B:
Impaired Aerobic Capacity/Endurance Associated with Deconditioning
Medical Screening of Joint Pain
Therapists see many people with joint pain. Most of these people have joint pain secondary to degenerative OA as opposed to rheumatic joint disease, but the therapist must be aware of the symptoms and signs associated with RA. The therapist can ask a series of questions to help identify the cause of joint pain.156 An abbreviated version of this list can include the following:
• Are you stiff in the morning? If yes, how long does this last?
• Does your stiffness increase after sitting?
• Where do you think the pain is coming from?
• If joints, which ones are involved?
• Do you ever notice any joint swelling or other changes?
• Does anyone else in your family have RA or other kinds of arthritis?
• Does aspirin or ibuprofen help you feel better?
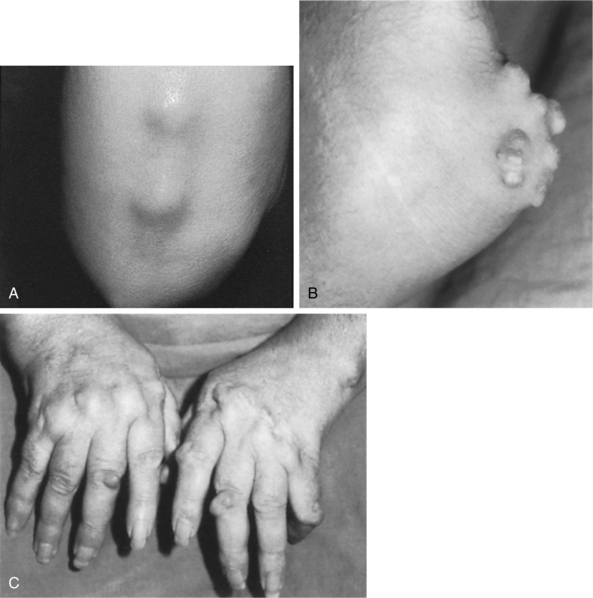
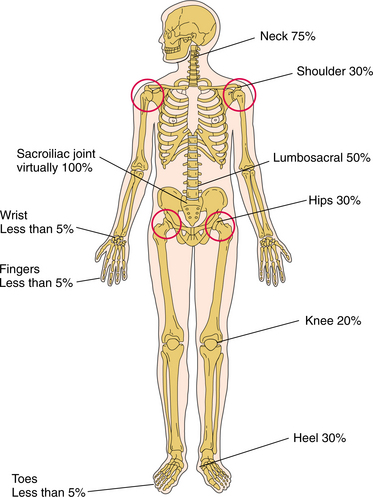
Being aware of the clinical signs and symptoms of RA will help the therapist make an early referral. The distribution of joint involvement is an important clue. RA usually affects the small joints of the feet and hands symmetrically; generalized pain (“I hurt all over”) is not characteristic of RA.
Quick, aggressive medical treatment is necessary to minimize joint destruction. Unexplained joint pain for 1 month or more, especially accompanied by systemic complaints, skin rash, or extensor nodules, no matter how mild, should raise concern on the therapist’s part. Cervical pain with reports of urinary retention or incontinence warrants immediate medical evaluation. Also, insidious onset of polyarthritis or joint pain within 6 weeks of taking a medication should raise suspicion regarding the nature of the pain complaints. Any of these red flags suggests the need for a medical referral if a physician has not recently evaluated the affected individual.
Anyone with known RA should be instructed in recognizing the signs and symptoms of progression of disease (e.g., increased duration of morning stiffness, increased number of tender and painful joints, increased intensity of joint pain, increased fatigue, increased gait disturbances, worsening of visible deformities) so that the intervention plan can be modified to meet the individual’s needs.
When distinguishing articular pain from periarticular involvement, remember that true arthritis produces pain and limitation during both active and passive range of motion, while limitation from tendinitis is much worse during active than during passive motion. Inflammatory joint involvement typically produces warmth, erythema, and tenderness. Frequently, there is bogginess related to underlying synovitis or effusion. These indicators are not present with joint pain of a mechanical cause.
Patient/Client Education
Helping individuals affected by RA understand the disease, disease process, treatment, possible outcomes, and role of exercise and self-care is a major part of the therapist’s task. Self-management includes learning pacing, joint protection, and energy conservation; monitoring symptoms; and maintaining or progressing an exercise program.
Each person must be taught ways to minimize trauma to inflamed joints by unloading joints and reducing mechanical joint stresses. This can be done through modification of activities such as using assistive devices to open doors and jars, and avoiding excessive weight bearing on inflamed joints by reducing movements such as bending and stooping. Energy conservation (see Box 9-8) should become a way of life for anyone with acute or subacute disease. The systemic nature of this disease produces global fatigue; the demand for energy to move joints may increase if biomechanics are altered.223
The need for frequent rest breaks, change in level of activity, and change in positions throughout the day should be taught and their use encouraged, but they should be balanced by the need to avoid muscle wasting and weakness from immobilization. Range of motion, stretching, and isometric exercises must be taught, monitored, and reinforced for as long as the therapist follows the individual, always teaching the client how to modify the program during periods of active inflammation.
Cervical Spine Involvement
Cervical collars may be used for comfort but do not protect against progressive subluxation or neurologic compromise. Rigid cervical collars can partially limit anterior atlantoaxial subluxation, but they also prevent reduction of the deformity in extension.238,239 Rigid orthoses are also poorly tolerated in these individuals because of skin sensitivity and temporomandibular joint involvement. Anyone with cervical spine involvement should be taught to avoid cervical flexion. The therapist can focus on isometric strengthening of neck muscles and overall postural training.
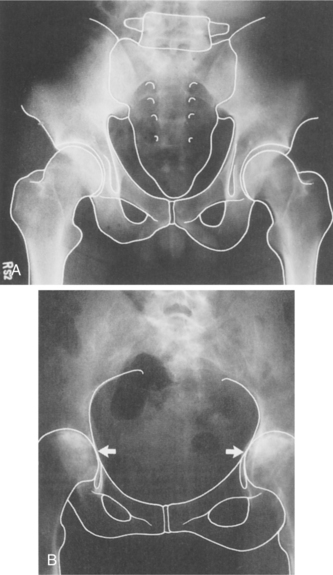
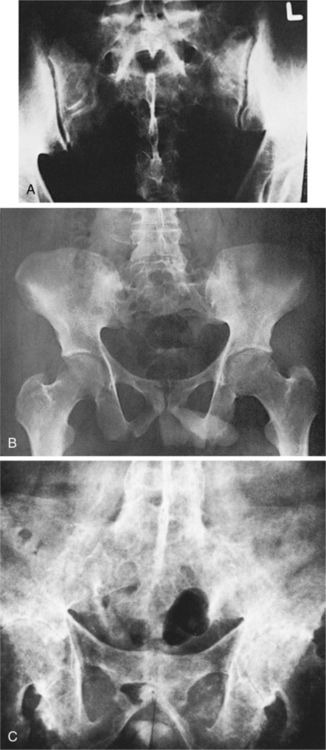
In the case of conservative intervention with nonsurgical management, the therapist must observe for (and teach the client to observe for) gradual deterioration in function that may indicate the development of subtle myelopathy (see Clinical Manifestations in this section). Radiographic evaluation is important in observing for impending neurologic compromise.420
Rehabilitation
RA is a chronic, progressive disease requiring an interdisciplinary team approach that is individual to the client and comprehensive, with long-range planning that extends beyond the initial acute phase. The role of the physical therapist as an integral part of the management of RA has been well established,59,287,370 with a renewed focus on outcome-based intervention.
The American College of Rheumatology has developed criteria for classification of functional status for individuals with RA that may help guide the therapist in designing and monitoring the results of an appropriate plan of care (Box 27-11).
Box 27-11 ACR CRITERIA FOR CLASSIFICATION OF FUNCTIONAL STATUS IN RHEUMATOID ARTHRITIS
Class 1: Completely able to perform usual activities of daily living (self-care, vocational, avocational)
Class 2: Able to perform usual self-care and vocational activities, but limited in avocational activities
Class 3: Able to perform usual self-care activities, but limited in vocational and avocational activities
Class 4: Limited in ability to perform usual self-care, vocational, and avocational activities
ACR, American College of Rheumatology.
The Ottawa Panel has identified nine goals in the rehabilitation of individuals with RA, including decreasing pain, effusion (swelling), and stiffness; correcting or preventing joint deformity; increasing motion and muscle force (decreasing weakness); improving mobility and walking; increasing physical fitness; reducing fatigue; and increasing functional status.383
Before initiating any rehabilitation program for this group of individuals, a thorough limb and joint examination must be done to provide an objective way to assess and document disease activity and progression or, conversely, remission and improved function. Numerous resources are available to assist the clinician in carrying out a thorough clinical assessment of the individual with RA, including a helpful joint-by-joint guide (describing where and how to palpate).347,431,459,460 Physical therapy examination should also include observation of functional performance, limitations, impairments, and a systems review as outlined in the Guide to Physical Therapist Practice.17
Complete bed rest is rarely indicated and is saved for those with severe, uncontrolled inflammation. For many people, a rest period of up to 2 hours during the day is important for dealing with general body fatigue and protection of involved joints. Splints can be applied to rest involved joints, prevent excessive movement, and reduce mechanical stresses. Crutches, canes, or walkers can be used to reduce weight-bearing stresses and enhance balance.
Adaptations may be necessary (e.g., platform crutches) because of upper extremity involvement. A home program of self-management will include instruction in proper body mechanics, positioning, joint protection, and energy conservation (see Box 9-8). Adaptive equipment designed to make tasks easier may include large key handle attachments allowing the person to use the whole palm to turn a key, spring-open scissors with big loops for anyone with hand involvement, jar openers and electric can openers, clip-on bottle openers, and ergonomic kitchen utensils with large handles and ergonomically angled handles.318
Instability at any joint, particularly at the atlantoaxial segment, requires caution on the therapist’s part. Such a joint may present with a marked reduction in range of motion, such as the shoulder or neck feeling stuck or caught with a certain movement. A history of periods of significant loss of range of motion alternating with full range of motion suggests joint hypermobility. Restoration of mobility is an important goal, but choosing techniques that are gentle or applying traction while stretching is necessary.
The extraarticular problems may affect the rehabilitation program. For example, if fatigue is present, the therapist may have to allow periods of rest during the treatment session. During periods of symptom exacerbation there is a fine line between overextending the client and maximizing activity. There are times when active exercise may have to be curtailed, but passive stretching remains important to prevent contractures.
Splenomegaly may account for tenderness on palpation and fullness or increased resistance of the left upper abdominal quadrant. Deep soft tissue techniques are contraindicated in this area. Percussion techniques may help the therapist delineate the caudal boundaries of the spleen.
Remission
Whether or not remission from RA was possible was a point of discussion and debate in 1981 when preliminary criteria for remission were first proposed.403 These criteria have been modified over the years to reflect current treatment, trends, and outcomes. For example, the complete absence of tender or swollen joints is the most important sign of remission.380
Changes must last for more than 2 months, but a definitive time line has not been established. Today even some people with long-standing disease may achieve remission, although this is not possible for everyone. After remission, researchers hope to be able to effect a curative approach.
Postoperative Care
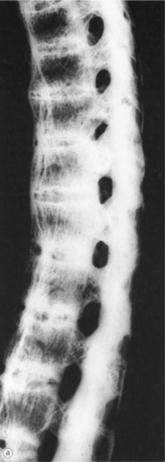
Surgical treatment of RA is often complicated by the client’s generalized debilitated condition. People with RA tend to have poor skin condition, poorly healing wounds, and osteopenic bone. Generalized bone loss occurs early in the course of RA and correlates with disease activity. This condition is further affected by the use of corticosteroids.
Poor nutritional status has been associated with higher complication rates following surgery, including infection. Following Standard Precautions with adequate handwashing is important. Likewise, promoting respiration with good breathing techniques is an important component of the therapist’s postoperative intervention.
Anyone with RA should be taught early on that, if surgery is ever indicated, a program of isometric exercises and range of motion before surgery is advised. Review dislocation precautions and restrictions prior to the surgical procedure. After arthroplasty, correction of deformity and relief of pain are typical, but recurrence of deformity can occur even with appropriate rehabilitation. Many clients are still very satisfied with the improved cosmesis, reduced pain, and improved function. Maximum benefit from arthroplasty may not occur for up to 1 year after surgery.77
The postoperative rehabilitation regimen must be tailored to the specific needs of each individual. The surgeon’s intraoperative assessment of the quality of tissues, component stability, and any associated repairs is critical to the rehabilitation protocol selected. Specific motion limitations vary depending on intraoperative repairs made, complications, and adverse events (e.g., infection, wound dehiscence, dislocation, implant fracture or failure, nerve damage).
Exercise and Rheumatoid Arthritis
A group of experts from the University of Ottawa reviewed comparative controlled trials and compiled evidence to suggest and support the conclusion that therapeutic exercises, including specific functional strengthening and whole-body functional strengthening, are a beneficial intervention for individuals with RA. The benefit may vary depending on the stage of disease (acute, subacute, inactive) but includes reduced pain, improved overall function, and decreased number of sick leaves.384
There is a need for more research to investigate the impact and effects of exercise on individuals with RA. Whereas rest has often been the treatment of choice, a balance must be attained between rest required during acute flare-ups and activity necessary to prevent the deconditioning effects of prolonged rest, immobilization, and inactivity. Education as to the efficacy of exercise and its proper use in self-management of RA has been shown effective in reducing stiffness and improving function in as little as 4 hours of a community-based physical therapy intervention delivered over a period of 6 weeks.39
To date, there is no evidence that active exercise beneficially affects the inflammatory processes associated with adult RA, but it has been shown that a short-term intensive exercise program in active RA is more effective in improving muscle strength than a conservative exercise program and does not have deleterious effects on disease activity.516
Individual studies of long-term intensive exercise have not shown an increase in joint swelling or pain with such a regimen. In fact, those who exercise rigorously at least twice a week for an hour show more improvement in physical abilities such a stair climbing and reduced psychologic distress compared to those who received standard care.100
The beneficial effect of exercise in lowering cytokine levels and increasing antiinflammatory compounds in plasma has been demonstrated in children with juvenile RA.522 Other effects of exercise on the immune system are discussed in the section on Exercise Immunology in Chapter 7.
GENERAL CONCEPTS
Exercises to prevent contractures, improve strength and flexibility, and enhance cardiorespiratory or aerobic conditioning are important components of the rehabilitation program.176,255,370 Joint pain leads to a reflex inhibition of muscle surrounding the joints, causing disuse atrophy of these muscles. Use of corticosteroids may lead to an additional decrease in strength and function.370
The feet are often overlooked in people with RA, but foot involvement occurs frequently, can impair gait, and can prevent safe participation in an exercise program. Foot involvement resembles that of the hand, with one important difference being that alterations in biomechanics may cause excessive stress to proximal lower extremity joints and to the spine areas forced to compensate for the altered gait.
Careful assessment of the feet may reveal uneven or pathologic weight-bearing patterns. Gait analysis and assessment of shoe wear can provide additional significant information regarding altered biomechanics. Providing assistive devices or orthotics before initiating an exercise program may be essential.431,459,460
Avoid overloading and overtraining. For the individual with active (acute) disease, adequate sleep is essential. Encourage 8 to 10 hours of rest each night. Range-of-motion exercises should begin with low repetitions several times throughout the day. Isometric exercises with short holds (4 to 6 seconds) have been suggested, once again using low repetitions (start with one or two and build up gradually to four to six).223
Range-of-motion exercises can be increased up to 8 to 10 repetitions in subacute cases, with the addition of dynamic strengthening exercises. Stable, quiescent, or inactive disease makes it possible to add an aerobic component such as walking, aquatics, or biking for at least 15 minutes each day three times a week. Range of motion and strengthening can be continued and monitored.223
EXERCISE PRESCRIPTION
A helpful guide in establishing the level of acceptable exercise intensity is as follows: acute pain during exercise indicates a need to modify the program; if joint pain persists for more than 1 hour after exercise is completed, the exercise was probably excessive.144 Recent research indicates that it is not necessary to work out as vigorously as once thought to derive benefits from exercise.
Engaging in moderate-level exercise for 30 min/day 4 or 5 days/wk appears to substantially increase physical fitness, even for older adults.484 Exercise spaced out over the course of the day can help loosen up stiff, achy joints while still providing cardiovascular benefits.343,390
The Arthritis Foundation provides a 24-page booklet (Exercise and Your Arthritis) describing the benefits and types of exercises, how to start, and how to keep going. This publication educates people about the possible effects of not exercising (e.g., increased joint stiffness, muscle weakness, muscle atrophy, increased risk of fracture and deformity). The foundation also provides valuable exercise tips for before, during, and after exercise.25
Currently the National Arthritis Foundation offers the largest standardized exercise program to individuals with arthritis through two community-based programs: an on-land exercise program called People with Arthritis Can Exercise (PACE) and an aquatic exercise program called the Arthritis Foundation Aquatic Program (AFAP).
The PACE program involves 72 exercises to improve motion, strength, endurance, balance, coordination, posture, and body mechanics. Other recommendations and guidelines for including conditioning exercise in a comprehensive management program for RA are available.15,26,345
STRENGTH TRAINING
Dynamic strengthening (gradually increasing resistance) through the full available range of motion helps stabilize joints, reducing erosive wear and tear on the structures. Regular, dynamic strength training combined with endurance-type physical activities improves muscle strength and physical function but not bone mineral density (BMD) in adults with early and long-standing RA, without causing detrimental effects on disease activity.168,169
Low-load resistive muscle training has been shown to increase functional capacity and is a clinically safe form of exercise in mild to moderate RA.255 Other studies report that moderate-or high-intensity strength training programs have better training effects on muscle strength in RA. It is essential to maintain the training routine to obtain long-term benefits.167
Strengthening in some cases of RA can be difficult, because exercise may lead to increased joint swelling and subsequent joint destruction. Before initiating a strengthening program, the physical therapist must be sure that pain is controlled, range of motion is optimized, and contractures are minimized.370 Paying attention to biomechanics, deformities, and muscle imbalances is important. Exercising misaligned joints without properly distributing the load can place too much pressure on vulnerable joints.452
AEROBIC EXERCISE
Aerobic exercise in this population is necessary to help reduce weight and improve cardiovascular fitness without increasing pain.330 Aerobic exercises are safe to perform during the subacute and inactive stages of RA. Aerobic capacity can be estimated using a single-stage submaximal treadmill test.344 Training programs begin at 50% (and work towards 80%) of maximal oxygen uptake based on the baseline test results. Without baseline testing, the therapist can rely upon (and teach the client to use) the Borg scale for rate of perceived exertion (RPE; see Table 12-13). Heart rate monitors are also helpful in enabling clients to track their cardiovascular responses.223
Screening for unknown coronary artery disease is recommended before initiating resistance or aerobic exercise. RA can also affect the bony structures of the rib cage and cause a decrease in chest expansion. The usual precautions for cardiopulmonary screening still apply based on the individual’s age, risk factors, and health history (especially heart health history).
People with RA may have normal pulmonary function tests but reduced respiratory muscle strength and endurance with reduced aerobic capacity compared to adults without RA.82 Assessment of respiration and intervention to improve breathing patterns are important components of the rehabilitation program.
AQUATIC THERAPY
Aquatic therapy may be beneficial for conditioning, strengthening, and flexibility while reducing mechanical stress on the joints. Water exercise provides the means by which people with RA can reach needed training levels in a comfortable environment. The AFAP program consists of 72 exercises similar to the PACE program that can be done in water with decreased joint loading, a reduced effect of gravity, increased buoyancy, and increased circulation.
Modalities and Rheumatoid Arthritis
Various modalities provide temporary pain relief and may be used in effectively and safely controlling symptoms of the acute inflammatory phase of RA. Information on the rationale for use and effectiveness of the various physical modalities is available.64,335,346
Although cold may be more suitable in acute inflammation, people with RA usually prefer heat. Superficial heat (e.g., paraffin baths, moist hot packs, hydrotherapy or aquatic therapy) is recommended, whereas prolonged or deep heat is contraindicated, since it may increase intraarticular temperature, leading to increased collagenase activity, possibly contributing to joint destruction.194
Electrotherapeutic modalities and thermotherapy physical agents are often used as part of a rehabilitation program mainly for pain relief, to control inflammation, and to reduce joint stiffness.
The Ottawa Panel recommends the use of low-level laser therapy, therapeutic ultrasound, thermotherapy, electrical stimulation, and transcutaneous electrical nerve stimulation for the management of RA. This recommendation is based on the analysis of systematic and literature reviews. Specifics of studies reviewed and a summary of the findings have been published in our own journal (Physical Therapy).383
The Ottawa Panel reported on available systematic reviews on the efficacy of ultrasound in the management of RA and noted that these are limited and do not offer conclusive evidence-based support for the use of ultrasound alone or combined with exercise.
Medications
Because of the long-term nature of RA, intervention is usually an ongoing process. Aspirin and NSAIDs are potentially ulcerogenic, and prolonged use of corticosteroids can lead to osteoporosis.
The analgesics and slow-acting antirheumatic drugs such as gold and penicillamine can impair renal function. Periodic screening of each of the body systems is imperative when working with this population; anyone taking DMARDs but still having joint pain and swelling should be reevaluated by the rheumatologist.
Numerous other side effects can occur with any of the pharmacologic agents used in the management of RA. The therapist should be aware of the potential side effects with any of the medications each client is taking.
For the individual with Felty’s syndrome (RA in combination with low white blood cell count or leukopenia and an enlarged spleen), there is an increased risk of infection, even when treatment with DMARDs raises the white blood count. Careful handwashing and Standard Precautions are always warranted, but especially in the case of this syndrome.