Atherosclerosis.: Because of the hyperglycemia and increased fat metabolism associated with type 1 DM, atherosclerosis begins earlier and is more extensive among people with diabetes than in the general population. Atherosclerotic changes in large blood vessels, caused by lipid accumulation and thickening of vessel walls, result in decreased vessel lumen size, compromised blood flow, and ischemia to adjacent tissues. As a consequence, people with diabetes have a much higher risk of myocardial infarction, stroke, and limb amputation.
Atherosclerosis and the accompanying large-vessel changes result in cardiovascular and cerebrovascular changes, skin and nail changes, poor tissue perfusion, decreased or absent pedal pulses, and impaired wound healing. Atherosclerosis combined with peripheral neuropathy and the subsequent foot deformities increases the risk for ulceration of skin and underlying tissues and limb amputation.
Individuals with undiagnosed type 2 DM are at significantly higher risk for CAD, stroke, and peripheral vascular disease than the population without diabetes. Screening of the type 2 at-risk population is essential in the prevention and treatment of diabetes-related complications. In addition, all individuals with diabetes should be aware of the strong and consistent data regarding the risks of smoking and the exacerbation of atherosclerosis-related diabetic complications.
Clients and families should be consistently and continuously counseled and encouraged in smoking cessation. The combination of smoking and diabetes dramatically increases the risks related to atherosclerotic vessel disease, impaired wound healing, and the associated morbidity and mortality rates.5
Cardiovascular Complications.: CVD is the leading cause of mortality and morbidity in diabetes and accounts for approximately two-thirds of all deaths among the diabetic population.41 People with diabetes have 1.5-to 4-fold increased risk of having CAD, stroke, and myocardial infarction.41 Although diabetes has long been recognized as a potent and prevalent risk factor of ischemic heart disease caused by coronary atherosclerosis, only recently has diabetes become associated with left ventricular dysfunction independent of hypertension and CAD. This is a disease of a cardiac muscle itself and is called diabetic cardiomyopathy.71
Left ventricular diastolic and systolic dysfunction, left ventricular hypertrophy, and alterations in the coronary microcirculation have all been observed in diabetic cardiomyopathy and are not fully explained by the cellular effects of hyperglycemia alone. The most important mechanisms of diabetic cardiomyopathy are metabolic disturbances, myocardial fibrosis, small vessel disease, cardiac autonomic neuropathy, and insulin resistance.71 Because of the presence of autonomic neuropathy, people with diabetes may have what is called “silent ischemia” or silent heart attack. They do not experience typical pain because of the damage to nerves that occurs in diabetes.
The cardiovascular and renal systems are intricately connected and affected by diabetes. Low blood flow to the kidney causes a release of renin, which in turn triggers a cascade of events as angiotensin is converted to angiotensin I then to angiotensin II, resulting in large increases in blood pressure. The risk of myocardial infarction and stroke increases as well.
Retinopathy and Nephropathy.: Diabetic retinopathy is a highly specific vascular complication in persons with both type 1 and type 2 DM and its prevalence is correlated closely with duration and control of high blood glucose levels. After 20 years with DM, nearly all individuals with type 1 DM and more than 60% of type 2 DM have some degree of retinopathy.
Diabetic retinopathy poses a serious threat to vision. Underlying microvascular occlusion of the retina resulting in progressive areas of retinal ischemia and tissue death causes diabetic retinopathy. Studies have established that intensive management of blood glucose level control to consistent near-normal levels can prevent and delay the progression of diabetic retinopathy.55
Diabetes is now the leading cause of end-stage renal disease (ESRD), which is kidney failure requiring dialysis or transplantation, in the United States and Europe.171 Hardening and thickening of the glomerular basement membrane, which result in eventual destruction of critical renal filtration structures, cause diabetic nephropathy. The presence of small amounts of albumin in the urine is the earliest clinical evidence of nephropathy. The eventual destruction of the filtering ability of the kidney causes chronic renal failure and the need for permanent dialysis or renal transplantation.
Renal destruction, as with retinopathy, can be slowed significantly with early detection and monitoring, tight glucose control, early treatment of hypertension (particularly with angiotensin-converting enzyme [ACE] inhibitors), careful monitoring of dietary protein, and strong encouragement of cessation of smoking.55,60,102 Hypertension is managed with ACE inhibitors initially and if blood pressure is not less than 130/85 mm Hg, a β-blocker may be added. However, combining a β-blocker with a diuretic can blunt awareness and symptoms of low glucose, so this combination usually is not recommended.
Infection.: Chronic, poorly controlled diabetes mellitus can lead to a variety of blood vessel and tissue changes that result in impaired wound healing and markedly increased risk for infections. Impaired vision and peripheral neuropathy contribute to the decreased ability of the person with diabetes to feel or see breaks in skin integrity and developing wounds. Vascular disease contributes to tissue hypoxia, which further decreases healing ability.
In addition, once pathogens are inside the body, they multiply rapidly because the increased glucose content in body fluids and tissues fosters bacterial growth. Because the blood supply to tissues is already compromised, white blood cells are not mobilized to the affected areas efficiently or adequately. Diabetes results in higher incidences of skin, urinary tract, vaginal, and other types of tissue infections.111
Musculoskeletal Problems.: Musculoskeletal complications are common, often involving the hands, shoulders, spine, and feet. Carpal tunnel syndrome, Dupuytren’s contracture, trigger finger, and adhesive capsulitis occur four times more often in people with diabetes compared with those who do not have diabetes.30,32 Available data show that more than 30% of people with type 1 or type 2 DM have some kind of hand or shoulder disease. More people with type 1 DM have musculoskeletal disorders than those with type 2 DM and the degree of stiffness is greater with this type of diabetes. The exact mechanism by which the specific metabolic abnormalities of diabetes are linked to rheumatic manifestations remains unclear.31
Although these disorders are not life-threatening, they can add significant functional impairment to a person’s life. See also the discussion of orthopedic problems that can develop secondary to sensory and motor neuropathy in the section on Sensory, Motor, and Autonomic Neuropathy in this chapter.
UPPER EXTREMITY.: In the hand, the syndrome of limited joint mobility (SLJM or LJM) and the stiff hand syndrome are unique to diabetes. SLJM is characterized by painless stiffness and limitation of the finger joints (Fig. 11-10). Flexion contractures typically progress to result in loss of dexterity and grip strength. The SLJM is an underdiagnosed complication of diabetes, largely because this type of loss of hand range of motion is considered a common normal sign of aging.89 The severity of this syndrome in diabetes is correlated with the duration of disease, duration and quantity of insulin therapy, and smoking. Joint contractures also may develop in larger joints, such as the elbows, shoulders, knees, and spine.
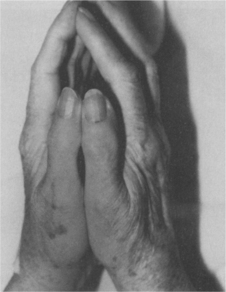
Figure 11-10 The prayer sign. The individual is unable to press the palms flat against each other, a diagnostic sign for the syndrome of limited joint mobility in diabetic persons. Other conditions also may result in loss of extension with a positive prayer sign. (From Kaye T: Watching for and managing musculoskeletal problems in diabetes, J Musculoskel Med 11:25-37, 1994.)
The stiff hand syndrome often is confused with or included in SLJM, but it has a distinct pathogenesis and clinical presentation. The stiff hand syndrome occurs uniquely with diabetes and is seen more frequently with type 1 DM and poor blood glucose control. Paresthesias, which eventually become painful, are accompanied by subcutaneous tissue changes such as stiffness and hardness. Vascular insufficiency may be the underlying cause or may be secondary to neuropathy, nodular tenosynovitis, and osteoarthritis.
Dupuytren’s contracture is characterized by the formation of a flexion contracture, palmar nodules, and thickening band or cord of palmar fascia (Fig. 11-11), usually involving the third and fourth digits in the population with diabetes (rather than the fourth and fifth digits in the population without diabetes). Pain and decreased range of motion are the primary presentation. Painless nodules develop in the distal palmar crease, often in line with the ring finger, which slowly mature into a longitudinal cord that is readily distinguishable from a tendon. The skin overlying the nodules is usually puckered.
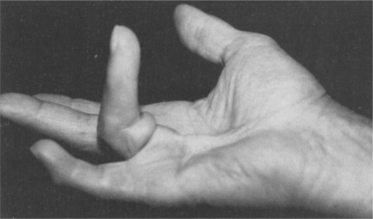
Figure 11-11 Dupuytren’s contracture. Painless nodules develop in the distal palmar crease, often in line with the ring finger, that slowly mature into a longitudinal cord that is readily distinguishable from a tendon. The skin overlying the nodules is usually puckered. The contracture may be symptomatic (painful), but with or without pain it results in impaired hand function. (From Kaye T: Watching for and managing musculoskeletal problems in diabetes, J Musculoskel Med 11:25-37, 1994.)
In some cases, regression of symptoms does occur without intervention, although the underlying mechanism for this phenomenon remains unknown. Surgical excision has not been shown to be a reliable cure for the disease and is not recommended unless there is a contracture that is bothersome. It has been reported that if the disease recurs after surgical excision, the rate of progression may be faster.204
Flexor tenosynovitis (also called chronic stenosing tenosynovitis) is another rheumatologic condition seen more commonly in persons with diabetes. Tenosynovitis is caused by accumulation of fibrous tissue in the tendon sheath and can cause aching, nodularity along the flexor tendons, and contracture. Locking of the digit, called trigger finger, can occur in flexion or extension and may be associated with crepitus or pain. In the population with diabetes, tenosynovitis is found predominantly in women and affects the thumb, middle, and ring fingers most often.
Diabetes is the systemic disease most often seen in connection with peripheral neuropathy of the hand, including CTS. The clinical presentation of CTS is the same for the person with diabetes as for the person without diabetes, although in diabetes CTS can be either a neuropathic process or an entrapment problem. Both neuropathy and compression within the carpal tunnel may exist together.
Adhesive capsulitis (also known as periarthritis or frozen shoulder) is characterized by diffuse shoulder pain and loss of motion in all directions, often with a positive painful arc test and limited joint accessory motions. The pattern is slightly different from that of typical adhesive capsulitis, in which regional tightness in the anteroinferior joint capsule primarily compromises external rotation, followed by loss of abduction and less often, internal rotation and flexion.
The pattern in diabetes is one of significant global tightness with external and internal rotation equally limited in the dominant shoulder, followed by limitations in abduction and hyperextension. External rotation and hyperextension are most limited in the nondominant shoulder, followed by internal rotation and abduction. The pathogenesis of the capsular thickening and adherence to the humeral head remains unknown. The long head of the biceps tendon may become glued down in its tendon sheath on the anterior humeral head.239
Adhesive capsulitis may be accompanied by vasomotor instability of the hand previously referred to as reflex sympathetic dystrophy (RSD) but now classified as the complex regional pain syndrome (CRPS). This condition is characterized by severe pain, swelling, and trophic skin changes of the hand (e.g., thinning and shininess of the skin with loss of wrinkling, sometimes with increased hair growth).
Skin changes in diabetic hand arthropathy, in addition to changes caused by CRPS, may occur in association with adhesive capsulitis. Other skin changes associated with diabetes include scleroderma diabeticorum, an asymptomatic thickening of the skin that may lead to a peau d’orange appearance, which usually involves the posterior neck, upper back, and shoulders.27
Skin and subcutaneous tissue atrophy and tendon flexion contractures develop. The natural history of this condition ranges from spontaneous remission to perma- nent loss of function. (See the section on Complex Regional Pain Syndrome in Chapter 39.)
Tendinopathy with thickening of the plantar fascia and Achilles tendon and tendo-Achilles tightening occurs as glucose deposits in tendons and ligaments result in loss of flexibility and rigid foot. In the diabetic population, loss of Achilles tendon flexibility, especially when combined with a flatfoot, increases pressure under the foot, adding to the compressive forces that contribute to ulcer formation.82
SPINE.: Diffuse idiopathic skeletal hyperostosis (DISH; also known as ankylosing hyperostosis or Forestier disease) is a condition of the spine seen most often in people with type 2 DM, although it can occur in a person who does not have diabetes. In DISH, osteophytes develop into bony spurs, typically right-sided syndesmophytes that may join to form bridges (Fig. 11-12). The thoracic spine most commonly is involved. In contrast to ankylosing spondylitis, the sacroiliac joints are spared, and vertebral body osteoporosis is absent. Calcaneal and olecranon spurs may develop, and new bone may form around hips, knees, and wrists.
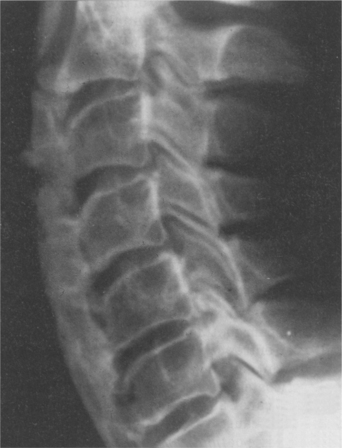
Figure 11-12 Diffuse idiopathic skeletal hyperostosis (DISH), or ankylosing hyperostosis, associated with type 2 diabetes mellitus (DM). DISH can occur with other conditions such as ankylosing spondylitis. Although the dense anterior bony bridging of the cervical vertebrae is pictured on this lateral roentgenogram, the thoracic spine most commonly is involved in diabetes. This type of DISH can be distinguished from ankylosing spondylitis by the preservation of sacroiliac joints, a site of typical involvement in ankylosing spondylitis. (From Kaye T: Watching for and managing musculoskeletal problems in diabetes, J Musculoskel Med 11:25-37, 1994.)
People with DISH may be asymptomatic or they may experience back pain and stiffness without limitations in range of motion. Dysphagia may develop if extensive cervical spine involvement occurs. The pathogenesis of DISH is unknown, and apparently no correlation exists between the degree of diabetic control and the extent of hyperostosis.
OSTEOPOROSIS.: Generalized osteoporosis usually develops within the first 5 years after the onset of DM and is more severe in persons with type 1 DM. It is hypothesized that bone matrix formation may be inadequate in the absence of normal circulating insulin levels. Results of bone density studies in persons with type 2 DM are conflicting, with some studies demonstrating decreased bone density and others indicating increased bone density. People with type 2 DM have decreased circulating insulin levels because of beta-cell exhaustion, and others are hyperinsulinemic because of insulin resistance.
As in any case of osteoporosis, regardless of the underlying cause, this condition places the person at greater risk for fractures. With the additional loss of sensation associated with diabetes, minor trauma easily produces injury. Microfractures can occur in already weakened bone and cartilage and may remain unrecognized because of the lack of pain appreciation. A vicious circle is started, leading to further damage.
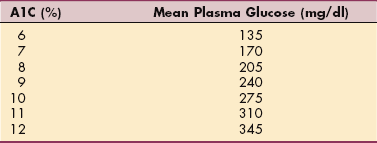
Sensory, Motor, and Autonomic Neuropathy.: Sensory, motor, and autonomic neuropathy associated with DM is a common phenomenon with known risk factors (e.g., duration of diabetes, current glycated hemoglobin value [HbA1c, also written as HbA1c], BMI, smoking, hypertension, and high triglycerides). The presence of CVD doubles the risk of neuropathy.242
Neuropathy may affect the CNS, peripheral nervous system, or autonomic nervous system (see Box 39-5). The most common form of diabetic neuropathy is a sensory polyneuropathy, usually affecting the hands and feet and causing symptoms that range from mild tingling, burning, numbness, or pain to a complete loss of sensation (usually feet) and foot drop. See further discussion of diabetic neuropathy in Chapter 39.
SENSORY NEUROPATHY.: Many people with diabetes suffer from diabetic peripheral neuropathic pain (DPNP) associated with nerve damage. Spontaneous pain, allodynia (painful response to benign stimuli), hyperalgesia, and other unpleasant symptoms are common with DPNP. Neuropathic pain often progressively increases in intensity throughout the day and is worse at night, significantly impairing sleep. Some individuals experience painful neuropathy called insulin neuritis syndrome at the beginning of therapy for diabetes; the feet are affected more often than the hands and it is usually self-limiting.256
The loss of sensation in diabetic neuropathy predisposes joints to repeated trauma and progressive joint destruction. Chronic progressive degeneration of the stress-bearing portion of a joint associated with loss of proprioceptive sensation in the joint produces a condition called Charcot’s disease, Charcot’s arthropathy, neuroarthropathy, or neuropathic arthropathy. Diabetes is the most common cause of neuropathic joints.
Several stages of neuropathic arthropathy (Charcot’s foot) occur involving bone destruction and absorption leading to dislocation and an unstable joint. Bone fragments and debris are deposited in the affected joint. Subluxation of the tarsal and metatarsal joints commonly results in a rocker-bottom foot deformity (see Fig. 23-9) and a redistribution of pressure on the plantar surface of the foot with progressive ulceration. An acute neuropathic joint is swollen, warm, and edematous, but pain may be minimal because of the underlying altered sensation.
Left untreated, neuropathic changes can progress to complete destruction of the joint. The presence of autonomic neuropathy may hasten this process as the blood vessels are unable to respond appropriately (e.g., vasoconstrict) to even minor trauma. Prolonged and unregulated hyperemia in the foot may lead to excessive bone resorption resulting in decreased bone mineral density, further increasing the risk of bone and joint destruction.100
Joints with less movement transmit abnormal forces through the foot to injure already damaged joints. This is especially true during walking, when large forces are placed on the midtarsal and tarsometatarsal joints. Obesity further increases these forces, and in the presence of any preexisting gait abnormalities or deformities, both create additional stress that compounds the condition.
Assessment of the underlying problem is important in planning the appropriate treatment intervention. For example, improving circulation may be a goal with macrovascular or peripheral vascular disease, whereas foot care and orthoses are more appropriate treatments for microvascular-caused neuropathy. The underlying neurologic disorder should be treated but this has no effect on the existing arthropathy. Reduction of weight bearing, joint immobilization, and joint protection are important conservative treatment tools. Surgical fusion can be performed if all else fails, but joint replacement is contraindicated in this condition.36,212
MOTOR NEUROPATHY.: Motor neuropathy is more common with long-standing disease and produces weakness and atrophy; bilateral but asymmetric proximal muscle weakness is called diabetic amyotrophy. Diabetic amyotrophy leads to bony deformities (e.g., claw toes, severe flatfoot with valgus of the midfoot, or collapse of the longitudinal arch) that contribute to biomechanical changes in foot function resulting in abnormal patterns of loading. Pain and erythema of the forefoot may constitute forefoot osteolysis, which is sometimes considered another form of neuropathy distinguished from cellulitis or osteomyelitis by laboratory values (leukocyte count) and roentgenographic appearance.
AUTONOMIC NEUROPATHY.: Autonomic neuropathy is sometimes referred to as diabetic autonomic neuropathy (DAN) and affects nerves that innervate heart, lung, stomach, intestines, bladder, and reproductive organs. It may manifest itself through the loss of control of blood pressure, blood glucose levels, temperature, regulation of sweating (skin becomes dry and cracked with buildup of callus), and blood flow in the limbs. Skin changes such as these can create more openings for bacteria to enter. The combination of all three types of neuropathy can ultimately lead to gangrene and possible amputation, largely preventable with proper care (see Special Implications for the Therapist: Diabetes and Foot Care in this section).
Cardiovascular autonomic neuropathy (CAN) is manifested by the lack of heart rate variability in response to deep breathing and exercise, exercise intolerance, persistent sinus tachycardia, bradycardia, and postural hypotension. Stress testing should be considered before starting an exercise program, especially in the older adult.8 CAN may also result in reduced perception of ischemic pain, making a person with diabetes unaware of having a heart attack. This may delay appropriate medical treatment and lead to death.255
DAN may lead to hypoglycemia without awareness because of loss of the warning signs of hypoglycemia such as sweating and palpitations. Being unaware of hypoglycemia and unresponsive to it are troublesome metabolic complications because they impair the person’s ability to manage the disease and may result in death. Other forms of autonomic neuropathy include gastroparesis (decreased gastrointestinal motility accompanied by diarrhea and fecal incontinence), constipation, urinary tract infections (nerve damage can prevent the bladder from emptying completely, allowing bacteria to grow in the bladder and kidneys), urinary incontinence, and sexual (erectile) dysfunction.
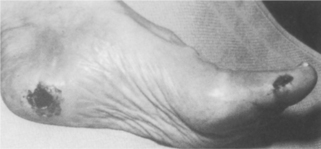
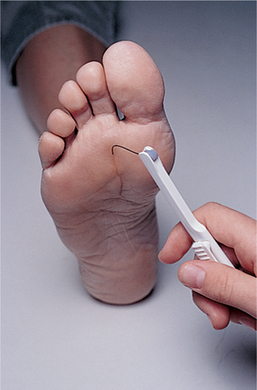
ULCERATION.: Sensory neuropathy, occurring as a result of improper glucose metabolism and diminished vascular perfusion to nerve tissues, places the diabetic person at risk for the development of ulcers. Diabetic foot ulcers are caused primarily by repetitive stress on the insensitive skin with increased pressure and/or horizontal (shear) stress. Body weight and activity level increase the force that the foot must transmit, and this also may increase pressure and shear force, especially in the presence of an underlying bony prominence or foot imbalance. In addition, previously healed ulcers leave scars that transmit force to underlying tissues in a more concentrated manner and hold the fat pad locally so that it cannot function physiologically. As a result, it cannot transmit shear forces, and it becomes damaged easily.
The loss of autonomic nerve function eliminates the production of sweat, leaving the skin dry and inelastic. Changes in pressure and gait, fat atrophy, and muscle weakness are mechanical factors that, along with sensory neuropathy, influence the development of plantar skin abnormalities, especially ulceration.21,229 Diabetes-induced changes in the skin are likely to contribute to ulceration because the collagen and keratin (a protein that is the principal constituent of epidermis, hair, and nails) may be glycosylated (saturated with glucose) with increased cross-linking, which makes the skin stiff. Keratin builds up in response to the increased pressure, covering the openings of unhealed ulcers, and cannot be removed as readily as normal keratin.
The areas most commonly affected by foot ulcers are the plantar areas of the metatarsal heads, the toes, and the plantar area of the hallux (Fig. 11-13). In the Charcot foot, the incidence of ulceration beneath the talus and navicular bones becomes more common because of the rigid rocker-bottom deformity.