Chapter 16 Inflammatory arthritis
KEY POINTS
 Initial detailed assessment is important in identifying where therapies can be most efficacious – opportunities for subsequent review allow timely response, especially in flares.
Initial detailed assessment is important in identifying where therapies can be most efficacious – opportunities for subsequent review allow timely response, especially in flares. Early therapy interventions minimise inflammation, pain, joint damage, deconditioning, and functional limitation.
Early therapy interventions minimise inflammation, pain, joint damage, deconditioning, and functional limitation. Self-management programmes that emphasize self efficacy and behaviour change improve health outcomes in RA.
Self-management programmes that emphasize self efficacy and behaviour change improve health outcomes in RA. Relate any exercise to improving performance of individually relevant functional tasks; individualise range, resistance, eccentric, concentric muscle work, length of hold of contraction
Relate any exercise to improving performance of individually relevant functional tasks; individualise range, resistance, eccentric, concentric muscle work, length of hold of contraction Aim to improve exercise tolerance as well as increasing range of movement/muscle strength and joint stability
Aim to improve exercise tolerance as well as increasing range of movement/muscle strength and joint stability Reassure new exercisers that joint/muscle aches may last up to 30–60 minutes after exercise – this is normal!
Reassure new exercisers that joint/muscle aches may last up to 30–60 minutes after exercise – this is normal!INTRODUCTION
Inflammatory arthritis is a category of rheumatic conditions involving inflammation of the synovial joints and includes specific forms such as rheumatoid arthritis, psoriatic arthritis, systemic lupus erythematosus, ankylosing spondylitis, reactive arthritis and juvenile idiopathic arthritis, among others. This chapter will focus on rheumatoid arthritis (RA), as a common presentation of inflammatory arthritis.
RA is a systemic, autoimmune disease characterized by symmetrical involvement of the peripheral joints, especially the small joints of the hands and feet, wrists, elbows, shoulders, knees and cervical spine. Symptoms include joint pain, stiffness and generalized fatigue, with exacerbations and remissions. Its aetiology is unknown and there is no cure. Despite significant recent improvements in medical management, RA remains a chronic condition resulting in mild to severe limitations in mobility and participation in everyday activities. RA has an impact across the spectrum of International Classification of Functioning, Disability and Health (ICF: World Health Organization, 2002) (see Ch. 4). In terms of body structure and function, joint mobility, muscle function, hand strength and dexterity are frequently impaired. Activity limitations, such as difficulty walking or handling objects, may subsequently restrict participation in self care, household work, employment, social relationships and leisure. The available resources and way a person responds to their illness and the challenges it presents (personal characteristics) influences perceived health, as will the environment, such as accessibility of the physical or built environment or support provided by institutional policies. For example, work disability begins early in RA, affecting over one-third, depending on disease severity, age of onset and job demands (Burton et al 2006). RA has a substantive physical, psychosocial, and economic impact. Rehabilitation services are important in maintaining, restoring and improving patient function as well as enhancing quality of life.
CHARACTERISTICS OF RHEUMATOID ARTHRITIS
PREVALENCE AND INCIDENCE
The prevalence of RA is remarkably consistent at 0.5–1% of the population in Western nations and women are affected twice as much as men (Kremers & Gabriel 2004, Symmons et al 2002). There is a genetic susceptibility, supported by a higher prevalence in North American aboriginal populations (up to 7%) and lower rates in China, Japan and rural Africa (Ferucci et al 2004). Incidence varies but is typically close to 40 per 100,000 (Symmons 2002), although declining over the past few decades, possibly due to increased oral contraceptive use, dietary influences and cohort effects (Symmons 2002). However, a recent cohort study suggested rising incidence of RA in women after four decades of decline (Gabriel et al 2008). Mortality studies show RA decreases life expectancy, possibly due to its associated co-morbidities, including cardiovascular, respiratory and gastrointestinal problems (Kremers & Gabriel 2004). Co-morbidities may be due to chronic illness effects (e.g. de-conditioning leading to cardiovascular problems) or treatments (e.g. non-steroidal anti-inflammatory drugs leading to gastrointestinal problems). The increased mortality associated with RA has been relatively stable over the past few decades. Recent changes in medical management will take several years to effect mortality rates (Kremers & Gabriel 2004).
PATHOLOGY AND IMMUNOLOGY
RA is an autoimmune disease with abnormal antibody and T-cell responses to an auto-antigen (Haynes 2004). The result is a wide-spread inflammatory process in the synovial cells lining joint capsules and other body tissues, manifesting as a range of extra-articular features. The normal joint has a thin synovium lining the joint capsule. These cells produce synovial fluid which lubricates and provides nutrition to the articular cartilage. In early RA, lymphocytes infiltrate the joint capsule, proliferation of the synovial lining occurs, resulting in increased synovial fluid production. This presents as swollen, warm, red and painful joints. Prolonged periods of inflammation stress the surrounding ligaments and tendons causing laxity and subsequent joint instability. Therefore, intervention focuses on reducing the inflammatory response. In more advanced stages pannus forms: the synovium proliferates with fibroblasts, macrophages, T cells, and blood vessels. The pannus invades and erodes articular cartilage, eventually exposing the bone. Bone resorption and remodelling may occur in end-stage disease when cartilage destruction may be unavoidable. Intervention for end-stage disease is typically joint replacement.
Synovitis (inflammation of synovium) is not limited to joint capsules. The long tendons of the hand pass through synovial sheaths and this tenosynovitis may restrict tendon gliding, causing stiffness and limited finger mobility. Other tendon sheaths may be similarly affected. RA is said to be ‘active’ or ‘exacerbated’ when joints are inflamed and laboratory indicators of disease activity, such as erythrocyte sedimentation rate (ESR) are elevated. RA is said to be ‘in remission’ or ‘controlled’ when inflammation is minimal.
Extra-articular features of RA include a range of inflammatory processes: cutaneous changes such as vasculitis (inflammation of the small blood vessels) and rheumatoid nodules (fibrosis nodes in subcutaneous tissue, commonly near the elbow); inflammation of tissues in the eye (scleritis, uveitis); cardiac disease (myocarditis, pericarditis and effusions); lung and pleural disease; kidney disease and peripheral neuropathies (Haynes 2004). Generally speaking, extra-articular manifestations suggest more severe disease. Therapeutic recommendations and interventions need to accommodate systemic symptoms and impairments as well as joint disease.
DIAGNOSIS, DIFFERENTIAL DIAGNOSIS AND SPECIAL TESTS
Diagnosis results from a careful history together with physical examination, radiological and serological tests. The American College of Rheumatology (ACR) established criteria for the diagnosis of RA (Arnett et al 1988), listed in Table 16.1. Diagnosis is confirmed if a patient has at least four of the seven criteria. The first four must have been present for at least 6 weeks. Rheumatoid factor (RF) is important for both diagnosis and prognosis of RA (Shin et al 2005).
Table 16.1 Revised criteria for classification of RA (Arnett et al 1988)
| CRITERION | DEFINITION |
|---|---|
| 1. Morning stiffness | Morning stiffness in and around the joints, lasting at least 1 hour before maximal improvement |
| 2. Arthritis of 3 or more joint areas | At least 3 joint areas simultaneously have had soft tissue swelling or fluid (not bony overgrowth alone) observed by a physician. The 14 possible areas are right or left PIP, MCP, wrist, elbow, knee, ankle and MTP joints. |
| 3. Arthritis of the hand joints | At least 1 area swollen (as defined above) in a wrist, MCP or PIP joint |
| 4. Symmetrical arthritis | Simultaneous involvement of the same joint areas (as defined in 2) on both sides of the body (bilateral involvement of PIPs, MCPs or MTPs is acceptable without absolute symmetry) |
| 5. Rheumatoid nodules | Subcutaneous nodules, over bony prominences or extensor surfaces or in juxta-articular regions, observed by a physician |
| 6. Serum rheumatoid factor | Demonstration of abnormal amounts of serum rheumatoid factor by any method for which the results have been positive in < 5% of normal control subjects |
| 7. Radiographic changes | Radiographic changes typical of rheumatoid arthritis on postero-anterior hand and wrist radiographs, which must include erosions or unequivocal bony decalcification localized in or most marked adjacent to the involved joints (osteoarthritis changes alone do not qualify) |
Key: MCP, metacarpophalangeal; MTP, metatarsophalangeal; PIP, proximal interphalangeal
Other tests aid monitoring disease activity. Most commonly ESR and C-reactive protein (CRP) blood tests assess levels of inflammation and are known as ‘inflammatory markers’. CRP is a better indicator of the acute phase response in the first 24 hours, but a more expensive test. (See Chapter 3 for details). Raised markers often indicate a ‘flare’ of RA, but the possibility of infection causing this elevation should always be considered.
Therapists should be aware of a patient’s haemoglobin (Hb) because, not only may RA patients present with ‘anaemia of chronic disease’, but as inflammatory markers rise, Hb may fall and vice versa. Consequently, a patient with low Hb, may not be as able to actively participate with therapy; appearing pale and possibly reporting overwhelming fatigue.
MONITORING DISEASE ACTIVITY, PROGRESSION AND PROGNOSIS
Predicting prognosis is challenging, but outlook is now more positive with recent significant pharmacological advances and the advent of anti-TNF therapies (see Ch. 15). A sero-positive rheumatoid factor (RF), at, or soon after diagnosis, indicates a worse prognosis in terms of long term erosive joint disease (Shin et al 2005). Auto-antibodies against cyclic citrullinated peptide (CCP) may be more specific than RF, not only for predicting prognosis (Kastbom et al 2004), but also diagnosing RA (Nishimura et al 2007). El Miedany et al (2008) suggest longer duration of early morning stiffness (EMS), greater percentage change in health assessment questionnaires (HAQ) and anti-CCP positivity are all predictors of persistent arthritis. Ongoing disease activity, both clinically and serologically, has been linked to increasing morbidity, loss of function and mortality. Therapists should be aware of anticipated prognosis and pro-actively target therapies accordingly.
Felson et al (1993) published a core set of disease activity measures for use in RA clinical trials. These include articular indices (joint counts), the patient’s assessment of their pain and physical function and both the patient and clinician’s global assessment of disease activity, as well as results of one acute inflammatory marker. Therapists should be aware of these valid and reliable measures, but may use them in isolation and/or combination with other functional measures of therapy progress. Twenty percent, 50% and 70% response criteria (known as ACR 20, 50 and 70) have been defined to identify improvement in RA (Felson et al 1995), which therapists should understand. The Disease Activity Score, calculated on 28 specific joints (DAS-28), is a standard approach to monitoring RA progress and drug response (Prevoo et al 1995).
RADIOGRAPHIC FEATURES
Conventional plain film x-rays remain the most frequently used method of evaluating disease progression in RA and assisting in diagnosis (Cimmino et al 2004). Bony erosions caused by RA may be visible from a few months following symptom onset. They are found at lateral joint margins, typically first seen in the small joints of the hands and feet (Fig. 16.1). Joint spaces may also be narrowed as the thickness of hyaline cartilage of synovial joints is reduced. Osteopaenia and peri-articular osteoporosis may be seen around these small joints on x-ray, but are more accurately quantified on DEXA scans, used to quantify the osteoporosis presenting in many RA patients (Tourinho et al 2005) particularly in early inflammatory arthritis (Murphy et al 2008).
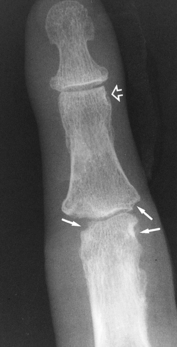
Figure 16.1 Illustrating erosions on the radial and ulnar aspects of the proximal interphalangeal joint as seen on radiology.
With permission from: Theodorou DJ et al 2003 Ch. 71 Imaging of rheumatoid arthritis. In: Hochberg MC et al (eds) Rheumatology 3rd edn, Elsevier, London. Fig. 71.1 p801.
Synovial joint subluxation may also be seen on x-ray. On examination, the hands can present with deformities associated with RA: the swan-neck, boutonnière and ulnar-deviation affecting the inter-phalangeal (IP) and metacarpo-phalangeal (MCP) joints and Z deformity affecting the thumbs. Any level of the cervical spine can also be affected. The consequences of subluxation of the atlanto-axial and atlanto-occipital joints are the most serious (compression of the spinal cord, nerve roots or cervical artery). Lateral views with the cervical spine in flexion or extension help assess this as well as views of the odontoid peg from an anterior to posterior view through the mouth.
Magnetic resonance imaging (MRI) aids evaluating synovitis, tenosynovitis and bursitis in the hands and feet of newly diagnosed patients (Boutry et al 2003). MRI may be used more in future for monitoring purposes (Cimmino et al 2004) as it is more sensitive, detecting both soft tissue and bony changes (Uetani 2007).
More recent research has focussed on using ultrasound scanning (USS) imaging in RA (Filippucci et al 2007), particularly to detect sub-clinical synovitis or bony erosions not evident with conventional radiology.
CLINICAL EVALUATION
Initial assessment and ongoing evaluation will be undertaken by many multidisciplinary team members. Although overlap is inevitable, therapists have a unique line of inquiry to establish a baseline level of function, particularly related to activities of daily living, as well as identifying current clinical problems. Ay et al (2008) reported that RA patients experience most functional challenges with gripping, hygiene and grooming, running errands and shopping. For those in acute flare up, only a subjective history may be possible, perhaps with part of an objective examination. Therapists work collaboratively with patients to establish functional goals and devise plans to achieve them.
SUBJECTIVE ASSESSMENT
History of presenting condition
The patient’s main problems must be identified from the outset. Some information may be provided on referral documentation. Therapists may have access to in-patient or on-line records, but a careful and logical history will elicit:
Past medical history
A relevant past medical history is needed, as certain treatments may be contraindicated or alter a patient’s capacity for therapy. Key questions include:
Ask the patient if there is anything they believe to be important to assess that has not yet been discussed.
Drug history
This provides clues to disease severity and helps determine optimal timing for therapy. Relevant aspects to note are:
The timing of medication may also be relevant, especially for patients who have recently started or stopped disease-modifying anti-rheumatic drugs (DMARD’s), biologics or received intra-articular injections. Knowing the timescale for drugs to reach full efficacy is important when planning therapy. See Chapter 15 for further details.
It will be important for therapists to be aware of allergic reactions, including latex (for Theraband™ purposes or if gloves are required).
Social and functional history
This provides an understanding of the patient’s roles and responsibilities, typical daily activities and any difficulties encountered in these as a result of symptoms or joint impairment. Understanding the activities the patient needs and wants to do helps establish functional goals. Occupational performance areas should be assessed (Law et al 2005):
The context in which these are performed is also important to query, such as where the patient lives and works, household members, sociocultural expectations, community resources and transportation. This helps establish priorities for intervention relevant to the patient.
When problems are noted in self-care, productivity or leisure, probe for contributing factors, including pain, physical factors (strength, endurance, mobility), or environmental factors (physical barriers, lack of proper equipment for a particular task). This determines which observational assessments are necessary. Discussing social roles provides an opportunity to inquire about psychological status, such as depression resulting from withdrawal from valued activities or roles (Katz & Yelin 1994), cultural beliefs and expectations, intimate relationships or coping issues. Although not feasible to assess every area in an initial evaluation, one should be alert to those most relevant to the individual, their circumstances and stage of illness. Some may be ready to act while others may still be adjusting to the diagnosis or a change in functional status and wondering how to cope. People with more established disease may come to therapy for specific and well-delineated purposes, as they are already experienced in managing their illness.
Asking the patient to describe their usual levels of mobility (including mobility aids and if mobility level has recently changed) helps the therapist plan objective assessment safely. Ask about estimated walking distance, what limits this, e.g. pain or shortness of breath, whether steps or stairs have to be negotiated (with or without banisters/stair lifts, etc.) and corroborate ability in objective examination. Subjective evaluation may include self-report measures of health and functional status (discussed later, and in Chs 4 & 5).
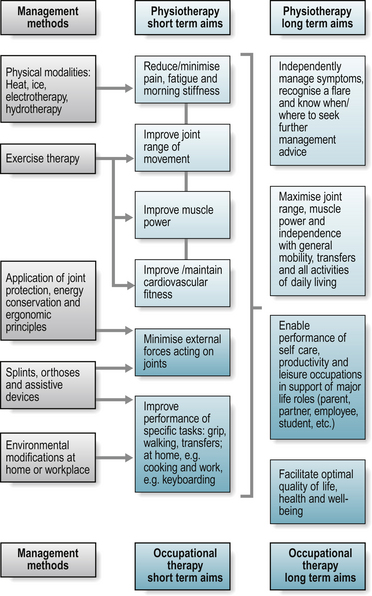
These two case examples illustrate how subjective findings will guide organizing the objective examination. The presenting issues and functional priorities differ for each patient. When time or patient tolerance is limited, focus on a few key issues for the objective examination and begin priority interventions to establish the therapeutic relationship. When necessary, evaluation and intervention planning can evolve over subsequent visits. See the clinical pathway in Fig. 16.2 for a comparison of functional goals, assessment and intervention plan for Ms Jones and Mrs Edwards.
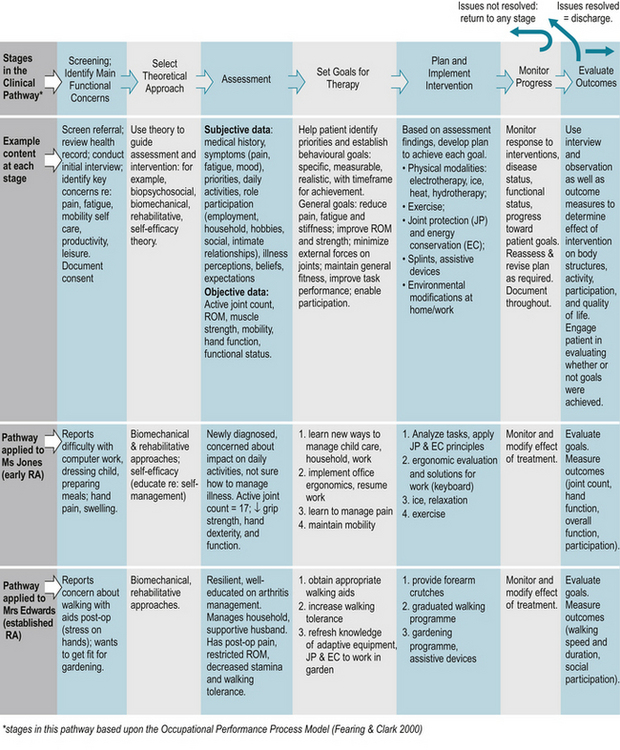
Figure 16.2 Clinical pathway for physiotherapy and occupational therapy for individuals with rheumatoid arthritis.
CASE STUDY 16.1 NEWLY DIAGNOSED PATIENT-SUBJECTIVE HISTORY
Ms Sarah Jones, aged 38, a part-time computer programmer and mother of two young children, consults her general practitioner. She complains of a 6 month history of wrist and hand pain, increasing weakness in her grip and general fatigue. Initially, she thought her symptoms related to a house move, but on questioning, she admits things have not improved over time. She reports general body stiffness experienced in the mornings, recently lasting over an hour, and episodes of swelling in the small joints of her fingers. This has made it difficult for her to work and she is concerned as she has had to take some time off. She describes how she has ‘trouble dressing my two-year-old’ and ‘peeling vegetables’. She has had some relief taking Ibuprofen medication.
CASE STUDY 16.2 PATIENT WITH MORE ESTABLISHED DISEASE-SUBJECTIVE HISTORY
Mrs Edwards, aged 62, has had RA for 22 years. She is referred to out-patient therapy following a right total knee arthroplasty 10 days previously. Medically, her RA is managed with combination therapy (methotrexate and sulfasalazine). Over the years, she has learned to ‘listen to her body’ and recognizes how to pace activities to manage fatigue. As a result of increasing knee pain and decreasing mobility prior to surgery, she was unable to maintain her exercise routine, and reports declining general strength and stamina. She is a little concerned about the stress placed on her hand and wrist while using a walking stick/cane as she recovers from surgery, because several joints are subluxed. Mrs Edwards is a retired office manager and lives in a small, one-level house with her husband. Both are avid gardeners.
OBJECTIVE EXAMINATION
If possible objective examination should follow the subjective history, but if a patient is acutely unwell, this may not be feasible. A logical approach ensures nothing of great importance should be missed if performed over several sessions. Whilst the focus will usually be the musculoskeletal system, a full respiratory or neurological assessment may be required with some patients (see Table 16.2).
Table 16.2 Components of the objective examination
| COMPONENT | DESCRIPTION | ADDITIONAL NOTE |
|---|---|---|
| General observation | Transfers, amount of assistance required, quality of movement, sitting/standing postures, eye contact | Observe patient’s response to proposed therapy, willingness to actively participate |
| UL joint observations | Swelling and/or erythema – especially MCP, IP and wrist joints | Severity of hand signs and symptoms may not correlate with poorer hand function |
| LL joint observations | Foot posture whilst weight-bearing; medial arch flattening, tendo-achilles angle, subluxation of MTP joints, tread on footwear | Document regularly used assistive, orthotic devices |
| Palpation | Small joints of hands especially but any symptomatic joints; active or inactive synovitis, tenderness | DAS-28 joint scoring adopted internationally (Prevoo et al 1995) |
| Range of movement | For UL, LL and spinal joints – measure actively and passively. Note reason for limitation of range, muscle length and neurodynamics as appropriate. | Should be linked with a functional goal Reliability of goniometry disputed: standard errors between 15 and 30° |
| Measure with manual or electronic goniometry or “eye-ball” technique | ||
| Muscle strength | Note hand dominance – power grip and key grip most functional | Jamar dynamometry and pinch/key grip dynamometry (Mathiowetz et al 1984) |
| Individual and key muscle groups should be assessed depending on functional challenges | Oxford scale method (Medical Research Council, 1976) | |
| UL – wrist extensors and rotator cuff muscles | ||
| LL – quadricep and gluteal muscle groups | ||
| Joint stability | Some agonist muscles may be a lot stronger than antagonist exacerbating instability at a joint | Key joints: wrists, MCP and IP joints in hands, knees, MTP joints and cervical spine |
| Mobility | Gait patterns +/− mobility aids and support required | Note “quality of gait” including stride length, cadence, heel strike, and distance the patient can (or cannot) walk |
| Steps/stairs | ||
| Note ability and safety | ||
| Transfers | Sit to stand, lie to sit +/− assistive devices | Make as functional and replicate home circumstances as much as possible |
| Note ability and safety | ||
| Exercise tolerance | Borg scale (Borg 1985) is a speedy and pragmatic method where patients state their “rate of perceived exertion” (RPE) | Most patients have reduced exercise tolerance |
Key: UL, upper limbs; LL, lower limbs; MTP, metatarsophalangeal, MCP, metacarpophalangeal; IP, interphalangeal
Observation
Welsing et al (2001) identified that functional capacity is most reduced with higher disease activity in early RA and with joint damage in late RA. Very often initial impressions can be deceptive; someone with apparently established and significant hand deformities may have accommodated and adapted over time and have good residual hand function. However, the opposite can also be true (see Fig. 16.3a-d).
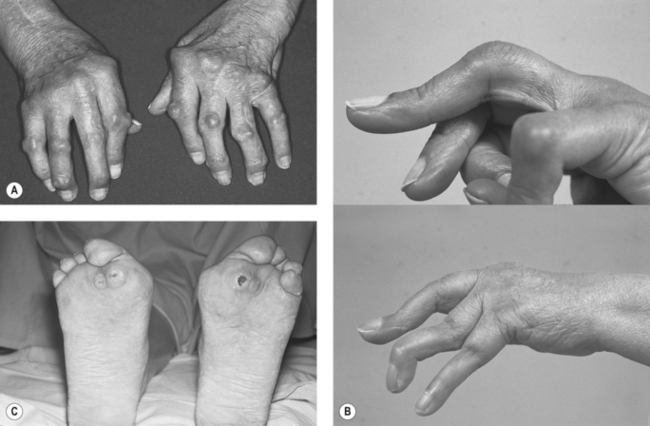
Figure 16.3 Clinical features that may be observed in advanced RA in the hand and foot in RA. (A) Ulnar deviation and rheumatoid nodules, (B) Swan-neck and Boutonniere deformities, (C) RA- forefoot deformity with callosity under MT head.
With permission from: (A) Matteson EL Ch.69 Extra-articular features of rheumatoid arthritis and systemic involvement, Fig 69.4 p782, (B) Gordon DA and Hastings DE 2003 Ch. 68 Clinical features of rheumatoid arthritis Fig 68.7 p771, (C) Gordon DA and Hastings DE 2003 Ch. 68 Clinical features of rheumatoid arthritis Fig 68.15 p776. In: Hochberg MC et al (eds) Rheumatology 3rd edn, Elsevier, London.
Palpation
The DAS-28 joint scoring method identifies pain and swelling on joint palpation and is part of a disease activity score (DAS), including patient and clinician opinion, questionnaires and ESR levels as part of disease and drug monitoring (Prevoo et al 1995). Knowing which joints are most affected helps therapists to plan relevant and targeted treatment.
Range of movement
Caution is needed when assessing passive range, especially in the cervical spine. In most instances this is not advisable and could elicit symptoms of cervical artery insufficiency. Assessing joint range can be very time consuming if all spinal, UL and LL joints are included. The therapist may direct range of movement (ROM) assessment to specific joints impacting on previously identified key functional difficulties.
Joint stability/muscle strength
RA often affects joints bilaterally, but a symmetrical distribution of either joint range or muscle power should not be presumed. Power grip can be reliably measured using a Jamar dynamometer (Mathiowetz et al 1984) but may not always be comfortable for patients with RA (see Fig. 16.4). Key grip however, is rarely uncomfortable to assess (e.g. using B&L Engineering Pinch Gauge) and other dynamometers are valid and reliable tools to use (e.g. MIE digital grip measures (MIE Medical Research, Leeds UK); Grippit digital measure, AB Detektor, Sweden).

Figure 16.4 Jamar dynamometer.
Reproduced, with permission, from the ‘Benefits Now’ website-http://www.benefitsnowshop.co.uk/
Some units may have more sophisticated electronic methods of measuring muscle strength (e.g. isokinetic machines), both isometrically as well as isotonically within a stated range of motion. Whilst affording more accuracy for research, it is not essential for patients in standard clinical settings. Using isokinetic machines may, however, be contraindicated in patients with active inflammatory joint disease.
Functional mobility and transfers
The ‘get up and go’ test quantifies mobility, especially in more elderly patients (Wall et al 1998, Podsiadlo & Richardson 1991). Alternatively, a quick timed ‘10 metre walk test’ needs only a measured walk way and stop watch for equipment. Slippers and soft-soled slip-on shoes with minimal support should be discouraged and ‘trainer’ type footwear, possibly with velcro fastening, promoted for both safety and joint protection reasons.
ASSESSMENT OF OCCUPATIONAL PERFORMANCE (FUNCTIONAL ASSESSMENT)
The initial interview identifies priority occupational performance issues and guides selecting additional evaluations to determine underlying performance components or environmental conditions contributing to the problem (Backman & Medcalf 2000). In addition to ROM, strength, and mobility, evaluation may include:
Useful measures are summarized in Table 16.3 and outcome measures for baseline and follow up assessments are discussed in chapters 4 and 5. During formal evaluation, particular attention is given to assessing hand function because RA typically affects these joints and the hands are involved in almost all daily activities. Early hand involvement is one of the strongest predictors of poor outcomes in RA (Schumacher et al 2004). Early detection and intervention through hand therapy may be one way to improve outcomes. Questionnaires measuring overall function may not be sensitive enough to capture specific problems with hand use in everyday activity (O’Connor et al 1999). Detailed observations of joint effusions, synovial proliferation, ligamentous integrity and deformity also contribute to a comprehensive hand assessment. Combining subjective and objective measures gives an overall picture of functional status.
Table 16.3 Selected functional tests/measures of activity and participation
| TEST | DESCRIPTION | REFERENCE |
|---|---|---|
| Performance-based Tests of hand function | ||
| Arthritis Hand Function Test (AHFT) | 11-item test of hand strength, dexterity, and functional tasks for adults with RA, osteoarthritis, and scleroderma | Backman et al 1991; Poole et al 2000 |
| Sequential Occupational Dexterity Assessment (SODA) | Measures ability to do 12 functional tasks, with difficulty scored by both occupational therapist and client | van Lankveld et al 1996 |
| Self-report assessments of hand and upper limb function | ||
| Cochin Rheumatoid Hand Disability Scale | Evaluates level of difficulty performing 18 functional tasks | Duruoz et al 1996 |
| Disabilities of the Arm, Shoulder and Hand (DASH) Questionnaire | 30-items evaluate level of pain and difficulty with functional tasks; optional modules for sports, work, and musicians; for any diagnosis | Beaton et al 2001 (includes entire questionnaire) |
| Michigan Hand Outcomes Questionnaire (MHQ) | Scores symptoms, function, and aesthetics for pre- and postoperative evaluations | Chung et al 1998 (includes entire questionnaire) |
| Overall health and functional status | ||
| Arthritis Impact Measurement Scales 2 (AIMS-2) | Self-report of mobility, physical, household and social activities, ADL, pain, depression & anxiety | Meenan et al 1992 |
| Canadian Occupational Performance Measure (COPM) | Semi-structured interview, rating performance and satisfaction with performance in self care, productivity, leisure on 1–10 scale | Law et al 2005 |
| Evaluation of Daily Activities Questionnaire (EDAQ) | Self-report or interview rating performance of 102 ADL with and without assistive devices/ adapted methods | Nordenskiold et al 1998 (UK version in development) |
| Health Assessment Questionnaire Disability Index (HAQ) and HAQ-II | HAQ: Self-report of difficulties performing 20 ADL tasks (in 8 categories). HAQ-II: 10 item version. | Fries et al 1980 Wolfe 2005 |
| Medical Outcomes Study Short Form-36 (SF-36) | Health survey with 8 subscales: physical function, social function, mental health, pain, physical role limitations, emotional role limitations, energy/fatigue, and general health | Ware & Kosinski, 2001 |
| Impact of fatigue on function | ||
| Multi-dimensional Assessment of Fatigue (MAF) | 16-item questionnaire addressing fatigue severity, frequency, distress and impact on ability to perform 11 activities | Belza 1995 |
| Parenting disability | ||
| Parenting Disability Index | Self-report of difficulty caring for children ≤ 5 yrs, 6–12 yrs, and/or > 12 yrs | Katz et al 2003 |
| Participation | ||
| Keele assessment of participation | Self-report, 11-item questionnaire; measures whether or not one is engaging in activities when and as they would like | Wilkie et al 2005 |
| Social role participation questionnaire | Self-report questionnaire rating salience and satisfaction with 10 domains of social participation | Gignac et al 2008 |
| Work disability | ||
| Ergonomic Assessment Tool for Arthritis | Customized self-assessment and semi-structured interview re: work organization, job demands and environment | Backman et al 2008 |
| Work Limitations Questionnaire | Self-report questionnaire rating difficulty performing 25 specific job demands | Lerner et al 2001 |
| Work Instability Scale | Self-report, 23-item questionnaire to identify risk for problems believed to lead to work disability | Gilworth et al 2003 |
The ICF (World Health Organization, 2002) distinguishes between activity limitations and participation restrictions. To capture these changes, specific measures of activity and participation are being developed or refined, potentially as part of a core set of outcome measures (Uhlig et al 2007). Staying abreast of emerging tools will improve comprehensive and valid functional assessment.
IMPORTANT FIRST TREATMENTS - EARLY RECOGNITION OF SIGNS AND SYMPTOMS
Because RA is episodic, recognising when a patient is entering a ‘flare up’ phase is very important. Patients should be aware of a sudden increase in joint swelling with associated pain, increasing EMS as well as over-whelming fatigue. Experienced patients will recognise signs and symptoms and appropriately self-manage, but the newly diagnosed may need guidance. Therapists are ideally positioned to help the patient to identify tell-tale signs, and manage the situation to best advantage. For example, for acute joint effusions (red, warm, swollen and painful joints), local treatments like ice and resting splints are indicated. To alleviate more systemic pain and fatigue, balancing rest and activity will help significantly. Some gentle daily activity (pottering in the home, gentle stretching) is important to prevent soft tissue shortening, but during the acute few days of a flare many patients will benefit from additional chair or bed rest. The balance is specific to each individual and dependant upon which joints are involved. Ensuring family members and significant others understand this principle is essential and therapists can often play an important role acting as an advocate for the individual.
Flare ups can be induced by stress and significant life events (Curtis et al 2005, Straub et al 2005) but the aetiology may also be unexplained. Early recognition and prompt management can minimise effects. Appropriate medication, as well as using ice (with hot joints, unless Raynaud’s phenomenon is an issue) or heat in the form of hot packs, warm showers or baths when muscular spasms are painful, can all ease symptoms.
IDENTIFICATION OF RED FLAGS
REHABILITATION INTERVENTIONS FOR RA
Guidelines for pharmacological and non-pharmacological management of RA have been documented in both the United States (American College of Rheumatology 2002) and Britain (Luqmani et al 2006, NICE 2009). Special attention has been given to early RA management to minimize disability (Hennell & Luqmani 2008), and to rehabilitation interventions (Hammond 2004). There is some evidence to show RA symptoms are relieved by thermotherapy, laser therapy, acupuncture, splints and assistive devices; and that pain and function can improve with patient education, joint protection behaviours, exercise therapy, hand exercises, and cognitive-behavioural therapy. However since Hammond’s review in 2004 few treatment studies have been published and well-designed rehabilitation trials are still required to identify which treatments work best for patients at different disease stages.
Figure 16.2 introduced a clinical pathway from assessment to outcome evaluation, providing an overview of occupational therapy and physical therapy management. Specific interventions are summarized in turn below.
PHYSIOTHERAPY EVIDENCE-BASED TREATMENT PROGRAMMES
The Association of Rheumatology Health Professionals has published standards of care documenting physical therapy competencies in rheumatology (2006), based on work by Moncur (1988). Treatment programmes must be individualised and devised in agreement and collaboration with the patient (Heller & Shadick 2007). The purpose and intended outcome for each stage of therapy should be discussed to clarify realistic expectations. As a rule the earlier a referral is made to therapists, the more successful interventions will be, especially if joint protection principles are adopted early on in joint disease. However for some individuals the shock of diagnosis may mean time is required to contemplate, before any actions can be positively adopted. Therapists should include promotion of long term health in their treatment strategies (see Ch. 6) and facilitate the patient to remain in work or education. In the context of inter-professional practice, physiotherapy and occupational therapy colleagues work collaboratively with patients to achieve shared therapy objectives. Increasingly, therapy will be delivered in community settings and the emphasis will continue to be on empowering the individual to manage their own symptoms.
Exercise therapy
Exercise therapy, in its many possible settings, is the mainstay of physiotherapy treatment (Hurley et al 2002, Lineker et al 2006). Exercise is prescribed to: improve joint range and muscle power, maximise joint stability, increase exercise tolerance, reduce morning stiffness/muscle spasm and thereby associated pain. The overall aim is to optimise function in ADL. Physiological benefits are well known, but Hurley et al (2003) also documented many psychosocial benefits of exercising in osteoarthritis, much of which is applicable in RA. Lee et al (2006) evaluated exercising three times a week for at least 20 minutes for more than 6 months and found that exercisers with RA experienced significantly less fatigue and disability compared with non-exercisers. Patients report that pain remains a significant barrier to their exercising (Bajwa & Rogers 2007) so unless this is addressed and an end purpose understood, exercise may seem irrelevant at best, painful at worst and therefore rarely undertaken independently.
Key features of exercising with RA:
Within the United Kingdom, ‘Arthritis Care’ and ‘Young Arthritis Care’ organisations encourage individuals to exercise in groups. This is popular and helps individuals remain motivated. Similarly, ‘People with Arthritis Can Exercise’ (PACE) programmes operate in the USA and “JointWorks” and “Waterworks” are offered by the Arthritis Society in Canada. These group initiatives are cost effective and for many peer support is invaluable. For others, exercising 1:1 with a therapist or alone is preferred (Bajwa & Rogers 2007) and therapists should select or produce audio-visual material to support this.
Tai Chi has become increasingly popular among people with arthritis. Whilst evidence is limited, Han et al (2004) report no detrimental affects in RA patients. A higher level of participation and generally greater enjoyment was also documented, compared with a traditional range of movement exercise class.
Previously, concern has been raised about exercise potentially increasing joint pain and exacerbating inflammatory disease, especially during a flare up. However, many studies reassure therapists and patients alike that appropriately prescribed exercises are significantly beneficial (Bearne et al 2002, Kennedy 2006, American Physical Therapy Association Ottawa Panel 2004a, Panel 2004, Van den Ende et al 2000, 2006).
The frequency of exercise required remains generally poorly documented and evaluated. Many recommend daily practice of simple stretching exercises to maintain or improve joint range for all large joints e.g. shoulder, hip and knee, and strength exercises most days (ARC 2005).
For all, early exercise therapy should be encouraged. Prophylactic exercises to prevent contractures, muscle atrophy and general deconditioning help the individual to maximise quality of life long term. Patients should be reassured it is never too late to start and regular exercise can be beneficial for everyone.
Research has demonstrated that hand exercises improve hand function over a six month period (O’Brien et al 2006). A combination of daily stretching and strengthening exercises improved hand function as measured by the Arthritis Impact Measurement Scales upper limb function subscale. Further research into this is ongoing.
Hydrotherapy
Hydrotherapy uses the properties of water, buoyancy, turbulence and drag during exercise to achieve an end functional goal. Hydrotherapy remains popular, despite efficacy having been challenged in some studies (Verhagen et al 1997, 2004). However, benefit has been demonstrated (Hall et al 1996) with patients reporting continued pain relief and reduced morning stiffness by diminishing muscle spasm. Some consider this an expensive resource (Epps et al 2005) not available to all. Consequently, therapists should be clear about their rationale for prescribing hydrotherapy as opposed to land-based exercise. However, it may be cost-effective if sufficient numbers utilise the resource throughout the day.
Cryotherapy
For many patients cryotherapy can be a useful adjunct to treatment of hot swollen joints (Robinson et al 2006) before or after exercise. For most patients a simple ice pack can be replicated at home by using ‘frozen peas’ or equivalent to ease symptoms temporarily (Hirvonen et al 2006). Information relating to optimal dosage and frequency remains illusive. Contraindications should be checked prior to treatment, e.g. a significant number of RA patients have Raynaud’s phenomenon.
Thermotherapy
Some patients experiencing muscle spasm or morning stiffness report benefits from applying heat and many report a morning shower minimises morning stiffness. Simple hot packs can be used, heated up in microwaves at home. Evidence for thermotherapy remains limited, but no detrimental effects have been reported (Welch et al 2002). Wax therapy, previously popular especially for hands, is less favoured today as being too passive a treatment. Benefit is only maintained if followed by hand exercises (Dellhag et al 1992). Therapists should be cautious of applying heat to joints with active synovitis. Some evidence indicates that metalloproteinases are activated with increasing temperature which could accelerate degeneration of hyaline cartilage (Yasura et al 2006). However, the clinical relevance of this is yet to be established in RA patients.
Electrotherapy
Many electrotherapy modalities have historically been used, but studies have been criticised for lack of methodological rigour as well as low sample sizes. The American Physical Therapy Association Ottawa Panel has published practice guidelines (2004b) for electrotherapy in RA. This panel recommends the use of therapeutic ultrasound, low level laser, transcutaneous electrical nerve stimulation (TENS), electrical stimulation and thermotherapy.
Berliner & Piegsa (1997) studied the physiological effects of continuous therapeutic ultrasound in a water bath on skin microcirculation and temperature in RA patients. Comparing healthy participants with patients with high and low disease activity, they concluded (based on a single treatment intervention) that microcirculation increased significantly in healthy participants, but least in those with high inflammatory activity.
A meta-analysis of low level (non-thermal) laser therapy concluded this has small short term effects on pain relief, range of movement and reduction in morning stiffness (Brosseau et al 2005). This systematic review highlights that future research must investigate dosage, wavelength, site of application and treatment duration.
Transcutaneous electrical nerve stimulation (TENS) is used for pain relief. It can be independently applied by the patient and used with many peripheral and spinal joints at home. Brosseau et al (2003) systematically reviewed effects of TENS in the rheumatoid hand. TENS significantly reduced resting pain and joint tenderness, and improved grip strength. However there were conflicting results between the differing modalities of TENS (‘conventional’ and ‘acupuncture-like’ TENS). Further research is needed, with increased power, to draw more meaningful conclusions.
Acupuncture
Acupuncture has been reported to have anti-inflammatory properties (Zijlstra et al 2003) and is increasingly used for symptomatic treatment of RA. As with many other modalities, data are not compelling and there are often methodological limitations in trials, but its popularity with patients suggests an anecdotal analgesic benefit. Casimiro et al (2005) report benefit from electro-acupuncture in knee pain in RA, but conclusions are limited due to few trials and small sample sizes. Results to date suggest no effect on inflammatory markers, swollen or tender joint counts or analgesic intake. Figure 16.5 summarises the management of a patient with RA from a physiotherapy and occupational therapy perspective.
OCCUPATIONAL THERAPY INTERVENTIONS
Occupational therapy interventions are directed at resolving functional limitations identified by the patient while managing symptoms like pain and fatigue. It is important to stay involved and connected with family, friends, and community, because withdrawal from everyday activities, including housework, leisure and social activities, is more closely linked to poor psychological outcomes than difficulty with ADL (Katz & Alfieri 1997). Patients may grieve the loss of participation in valued roles or experience mood changes, such as depression or anxiety, as they negotiate living with chronic illness. Engaging in meaningful activities is critically important to maintaining a sense of identity and positive self-worth. To have the greatest impact on health and well-being, it is highly recommended that occupational therapists take time to evaluate the occupations of greatest value to the client and focus on improving participation in these.
Comprehensive occupational therapy programmes have demonstrated sustained improvements in function (Helewa et al 1991, Steultjens et al 2002). Typical programmes involve: patient education (including joint protection, energy conservation, psychosocial support and cognitive-behavioural approaches to improve coping); splinting; assistive devices and environmental modifications; and activity adaptation.
PATIENT EDUCATION: SUPPORTING SELF-MANAGEMENT
Arthritis self-management programmes improve health outcomes for people with RA (Lorig & Holman 2006, Riemsma et al 2003). Successful programmes are based on enhancing self efficacy, i.e. the patient’s confidence that they can adopt behaviours to control disease symptoms and enhance function. Facilitating new habits through counselling on joint protection, energy conservation and cognitive-behavioural approaches are the cornerstone of occupational therapy patient education, which focuses on practical strategies to engage in life despite pain, fatigue, depression, and physical challenges.
Joint protection principles are based upon biomechanical and ergonomic guidelines and aim to reduce pain and local inflammation during task performance, preserve the integrity of vulnerable joint structures and improve function (Backman et al 2004). Energy conservation principles involve planning and pacing activities, to balance energy and fatigue in support of valued activities (Backman et al 2004). To be effective, general principles of joint protection and energy conservation need to be demonstrated and applied to the patient’s specific roles and activities (see Table 16.4). Compared to standard care, comprehensive joint protection education has been shown to reduce pain and morning stiffness, and improve ADL performance (Hammond et al 1999; Hammond & Freeman 2001). See chapters 1 and 6 for further descriptions of techniques and evidence.
Table 16.4 Joint-protection and energy-conservation principles and sample techniques
| PRINCIPLE | SAMPLE TECHNIQUES OR APPLICATION |
|---|---|
| Respect your pain | Reduce time and/or effort spent on an activity if pain occurs and lasts for more than 2 hours after the activity has been discontinued. Avoid nonessential activities that aggravate your pain |
| Balance rest and work | Take short breaks during your work. For example, take a 5-minute rest at the end of an hour of work. Intersperse more active tasks with more passive or quiet work |
| Reduce the amount of effort needed to do the job | Use assistive devices such as a jar opener or lever taps. Slide pots across the counter instead of lifting. Use a trolley to transport heavy items. Use a raised toilet seat and seat cushion to reduce stress on hips, knees, and hands. Use frozen vegetables to minimize peeling and chopping |
| Avoid staying in one position for prolonged periods of time | Change position frequently to avoid joint stiffness and muscle fatigue. For example, take a 30-second range of motion break after 10–20 minutes of typing or holding a tool; after standing for 20 minutes perch on a stool for the next 20 minutes; walk to the mailroom after 20–30 minutes sitting at your desk |
| Avoid activities that cannot be stopped immediately if you experience pain or discomfort | Plan ahead. Be realistic about your abilities so you don’t walk or drive too far, or leave all your shopping and errands to a single trip |
| Reduce unnecessary stress on your joints while sleeping | Use a firm mattress for support. Sleep on your back with a pillow to support the curve in your neck. If you prefer to lay on your side, place a pillow between your knees and lay on the least painful side |
| Maintain muscle strength and joint range of motion | Do your prescribed exercises regularly. Strong muscles will help support your joints. Regular exercise will reduce fatigue |
| Use a well-planned work space | Organize your work space so that work surfaces and materials are at a convenient height for you, to ensure good posture. Place frequently used items within close reach. Reduce clutter by getting rid of unnecessary items, or storing less frequently used items away from the immediate work space |
Note. From the Mary Pack Arthritis Program, Vancouver Coastal Health, Vancouver, BC Canada. Used with permission.
The psychosocial impact of RA cannot be underestimated: symptoms may significantly impact daily routines, and depression often accompanies RA. For example, morning stiffness can delay self care routines and present a challenge getting to work on time, or frustrate a mother needing to care for a young child waking early or during the night. Fatigue may limit participation in family events or recreational activities if all one’s energy is expended in obligatory paid and unpaid work activities. Ongoing disruption to daily routines creates distress and coping difficulties for many patients. There may be added worries regarding employment, financial issues, or family obligations.
One-to-one and group educational programmes (including joint protection and energy conservation mentioned above) help patients develop self-management skills and identify reasonable ways to plan ahead and adjust routines where possible to minimize pain and fatigue while doing daily activities. Cognitive-behavioural approaches and counselling assist patients to recognize ineffective thought processes, feelings and behaviours, then set goals and implement practical strategies to cope with the emotional consequences of RA (Backman 2006). For example, stress management and relaxation techniques might focus on changing attitudes and responses to challenging situations, rhythmic breathing and guided imagery. Because of differing priorities among patients and the episodic nature of RA, it is wise to offer a range of strategies and encourage patients to select those most applicable to their unique situation.
Involving family members in education and counselling sessions may be welcomed, as social support is important to living with RA. In many ways, RA affects the entire family, and spouses, parents or children may have questions of their own. Sometimes, problem-solving involves coordinating the efforts of several members of the health care team, for example, coordinating medications, ROM exercises, and joint protection techniques to optimize performance, or engaging the services of a vocational counsellor, social worker or psychologist to resolve complex employment, family, or emotional issues.
Splinting
Splints support and protect joints, minimize pain, and enhance function. They may provide localized rest to reduce pain and inflammation (e.g. a hand resting splint, see Fig. 16.6a); enhance joint stability to improve function (e.g. a wrist splint to improve ability to grasp, lift and carry items, see Fig. 16.6b); align joints in a stable anatomical plane to minimize deformity and stretching of the joint capsule and ligaments (e.g. a silver ring splint to position a swan-neck deformity of the finger into a slightly flexed posture, see Fig. 16.6c); or facilitate recovery from surgery (e.g. dynamic flexion or extension splints following arthroplasty or tendon repairs to encourage early finger motion in a controlled range (Backman et al 2004).
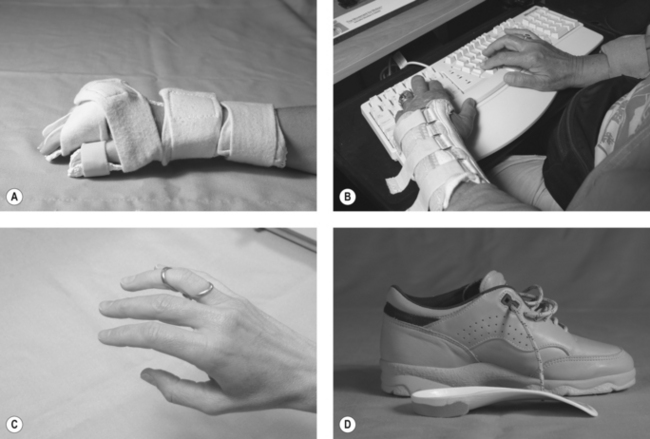
Figure 16.6 Splints used in RA. (A) hand resting splint; (B) wrist splint; (C) anti-swan neck silver ring splint; (D) foot orthosis and supportive shoe.
A more comprehensive discussion of splinting is presented in chapter 12. With respect to RA, evidence suggests that wrist splints improve hand strength (Haskett et al 2004, Pagnotta et al 1998) and reduce pain (Haskett et al 2004, Veehof et al 2008), and after a break-in period, do not compromise dexterity (Haskett et al 2004). During flares, resting splints may be recommended for the wrists and hands, knees, or elbow, at night use or during daytime rests. A systematic review concluded there is little evidence that resting splints improve function, yet patients prefer using them rather than going without, suggesting they alleviate pain (Egan et al 2001).
For prolonged MCP joint effusion, MCP protection splints may be indicated. These restrict flexion and ulnar deviation at the MCP joints with the intent to prevent or minimize subluxation. One study, using x-rays to evaluate 27 hands with RA, showed that while worn the splints corrected the alignment of all MCP joints except the index finger (Rennie 1996), although long-term benefits have not been evaluated.
Foot orthoses in combination with supportive shoes (Fig. 16.6d) support the transverse and longitudinal arches of the foot, alleviate metatarsalgia, and enable people with RA to walk comfortably (Chalmers et al 2000, Hawke et al 2008) (see also Ch. 13).
Assistive devices and environmental modifications
Dozens of assistive devices are available to accommodate physical limitations or promote adherence to joint protection principles. Common devices include jar openers (to accommodate for weak grasp), long-handle shoe horns, sock aids, or dressing sticks (to accommodate limited motion), and raised toilet seats (to reduce stress on hips, knees, and hands, because the higher seat means less need to push up). Once only available through therapy departments and medical supply catalogues, the move toward universal accessibility means many helpful devices can be found in department, hardware, office supply and other shops. For example, cook shops have an array of utensils with large grips, making grasp easier for all, not just people with RA. They also have lightweight dishes and small appliances for easy use at counter height. Ergonomic office equipment, appropriately selected, facilitates improved posture to minimize pain and fatigue.
Not all assistive devices will ‘fit’ all patients, and careful assessment and patient education may be necessary to prevent inappropriate suggestions. The long-handled reacher that accommodates decreased range of motion may present a mechanical disadvantage in some situations, putting greater stress on the hands and wrists, counteracting the intended benefit. Devices that fail to resolve the functional problem are not used. However, in one follow-up study, as many as 91% of kitchen devices reduced pain and were still in use by women with RA 6 to 12 months after they were first provided (Nordenskiold 1994). Not all patients are ready to consider multiple assistive devices and environmental modifications at once, and some devices reinforce beliefs about ‘being disabled’. Limit recommendations to the most important problems first, and build on successes as they occur.
Home modifications may enhance independence and functional capacity. Physical environment modifications can be divided into three categories (Mann et al 1999): re-arranging the living environment, such as moving dishes to lower shelves; adding to the environment, such as the placement of hooks and storage bins to keep frequently used supplies within easy reach; and structurally modifying the environment, such as installing a walk-in shower or ramps in place of stairs. Such modifications help people compensate for physical limitations arising from RA. In a small qualitative study, women described physical modifications, such as stair lifts and grab bars, accessing services such as home delivery of groceries, and involving friends and family to assist with tasks like home maintenance to successfully adapt to living with RA (Moss 1997). Similar accommodations, individualized to the patient’s chosen activities, delay or prevent disability at home and work.
Activity adaptation at home and work
For many patients, what matters most is actively engaging in parenting, household work, employment, socializing and similar roles. Mothers with arthritis describe learning to adjust expectations and set priorities, and recommend strategies such as omitting non-essential tasks, delegating to others, and ‘figuring out’ how to adapt tasks to match their physical ability and energy (Backman et al 2007). Work place transitions are also common (Gignac et al 2008), with patients identifying fluctuations in productivity, job changes, and job loss. Assessing workplace ergonomics (Backman et al 2008a, Backman 2008b) and adapting the workplace to accommodate arthritis symptoms may support productivity. Specific education on job accommodation legislation, vocational rehabilitation resources, and problem-solving in the workplace may reduce work disability (Lacaille et al 2007, 2008).
REFERRAL TO SPECIALIST SERVICES
Ideally patients with RA should have access to a diverse multi-disciplinary team (Hennell & Luqmani 2008). Within a secondary care environment this may include specialist nurses, podiatrists, social welfare staff, dieticians, orthotists and others. In the primary care setting staff involved include general practitioners with a ‘specialist interest’ in rheumatology and specialist community nurses. Employers may need to be advised of their employee’s health challenges, if the patient is willing to disclose their arthritis. Therapists are often the key staff who undertake workplace assessments with their patients and may involve a Disability Employment Advisor. Within a school or college environment, therapists may assess young people and liaise closely with parents and dedicated ‘special needs’ staff.
CONCLUSION
Proactive therapists will enjoy working with patients who have diverse and challenging problems arising from inflammatory arthritis. Therapists have an integral role within multi-disciplinary teams and their early targeted intervention maximises functional outcome. Evidence is increasing to support therapy interventions and further studies are essential to advance the practice of physical and occupational therapy and improve the health and well-being of people living with arthritis.
 What evidence is there for the effectiveness of splinting the MCP joints in terms of preventing deformity, reducing pain, or improving hand function?
What evidence is there for the effectiveness of splinting the MCP joints in terms of preventing deformity, reducing pain, or improving hand function?Arthritis and Musculoskeletal Alliance (ARMA). Standards of Care for people with infl ammatory arthritis. http://www.arma.uk.net.
Association of Rheumatology Health Professionals (ARHP), 2006. Standards of practice: Physical therapy competencies in rheumatology http://www.rheumatology.org/arhp/practice/standards.
Musculoskeletal Services Framework Document – Department of Health UK. http://www.dh.gov.uk/en/Publicationsandstatistics/Publications/PublicationsPolicyAndGuidance/DH_4138413
Arthritis Research Campaign (UK. http://www.arc.org.uk.
To obtain the exercise leaflet - “Keep moving”.
http://ww.arc.org.uk/arthinfo/patpubs/6282/6282.asp.
Hydrotherapy Association of Chartered Physiotherapists - UK guidelines.
http://www.csp.org.uk/director/groupandnetworks/ciogs/skillsgroups/hydrotherapy.cfm.
Joint zone – a study of rheumatology – UK. http://www.jointzone.org.uk/.
National Rheumatoid Arthritis Society (NRAS) – UK. http://www.rheumatoid.org.uk.
NHS Evidence (UK): Musculoskeletal Specialist Library. http://www.library.nhs.uk/musculoskeletal.
British Health Professionals in Rheumatology (BHPR). http://www.rheumatology.org.uk/bhpr.
The Health Talk Online Website personal experiences of health and illness – Rheumatoid Arthritis. http://www.healthtalkonline.org.
The Wright Stuff Arthritis Supplies. www.arthritissupplies.com.
Able Data, hundreds of assistive devices and technologies. www.abledata.com.
Arthritis Foundation (US), includes community-based self-management, exercise programs and resources for living well. http://www.arthritis.org/resources.php.
Arthritis Society (Canada), includes Arthritis Self-Management
Program information and a storefront for patients to purchase educational materials and assistive devices. http://www.arthritis.ca.
Psychometric Laboratory for Health Sciences – references to useful scales & outcome measures. http://home2.btconnect.com/Psylab_at_Leeds/scales.htm.
The DAS-28 for disease activity monitoring. http://www.dasscore.nl.
References and further reading
American College of Rheumatology. Guidelines for the management of rheumatoid arthritis 2002 update. Arthritis Rheum.. 2002;46(2):328-346.
American Physical Therapy Association Ottawa Panel. Evidence-based clinical practice guidelines for therapeutic exercises in the management of rheumatoid arthritis in adults. Phys. Ther.. 2004;84:934-972.
American Physical Therapy Association Ottawa Panel. Evidence-based clinical practice guidelines for electrotherapy and thermotherapy interventions in the management of rheumatoid arthritis in adults. Phys. Ther.. 2004;84(11):1016-1143.
ARC, 2005. Keep Moving Education Leaflet. Chesterfield, UK. Online. Available: http://www.arc.org.uk (accessed 9.2.09.).
Arnett F.C., Edworthy S.M., Bloch D.A., et al. The American Rheumatism association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum.. 1988;31(3):315-324.
Association of Rheumatology Health Professionals, 2006. Standards of practice: Physical therapy competencies in rheumatology. Online. Available: http://www.rheumatology.org/arhp/practice/standards 17 July 2007. (accessed 9.2.09.).
Ay S., Tur B.S., Kucukdeveci A. Evaluation of disability in patients with degenerative and inflammatory arthritis. Int. J. Rehabil. Res.. 2008;31(2):159-163.
Backman C.L. Psychosocial aspects in the management of arthritis pain. Arthritis Res. Ther.. 2006;28:221. (doi:10.1186/ar2083)
Backman C., Mackie H., Harris J. Arthritis Hand Function Test: Development of a standardized assessment tool. Occup. Ther. J. Res.. 1991;11:245-256.
Backman C., Medcalf N. Identifying components and environmental conditions contributing to occupational performance issues. In: Fearing V.J., Clark J., editors. Individuals in Context: A Practical Guide to Client Centred Practice. Thorofare, NJ: Slack Inc; 2000:55-67.
Backman C.L., Fairleigh A., Kuchta G. Occupational therapy. In: St Clair E.W., Pisetsky D.S., Haynes B.F., editors. Rheumatoid Arthritis. Philadelphia: Lippincott Williams & Wilkins; 2004:431-439.
Backman C.L., Del Fabro Smith L., Smith S., et al. The experiences of mothers living with inflammatory arthritis. Arthritis Rheum.. 2007;57(1):381-388.
Backman C.L., Village J., Lacaille D. The ergonomic assessment tool for arthritis (EATA): development and pilot testing. Arthritis Rheum.. 2008;59(10):1495-1503.
Backman, C., 2008b. The Ergonomic Assessment Tool for Arthritis (Ergotool). http://ergotool.arthritisresearch.ca (accessed 14.1.09.).
Bajwa H.A., Rogers L.Q. Physical activity barriers and program preferences among indigent internal medicine patients with arthritis. Rehabil. Nurs.. 2007;32(1):31-34. 40
Bearne L.M., Scott D.L., Hurley M.V. Exercise can reverse quadriceps sensorimotor dysfunction that is associated with rheumatoid arthritis without exacerbating disease activity. Rheumatology. 2002;41(2):157-166.
Beaton D.E., Katz J.N., Fossel A.H., et al. Measuring the whole or the parts? Validity, reliability, and responsiveness of the disabilities of the arm, shoulder and hand outcome measure in different regions of the upper extremity. J. Hand Ther.. 2001;14(2):128-146.
Belza B. Comparison of self-reported fatigue in rheumatoid arthritis and controls. J. Rheumatol.. 1995;22(4):639-643.
Berliner M., Piegsa M. Effects of therapeutic ultrasound in a water bath on skin microcirculation and skin temperature in rheumatoid arthritis. Eur. J. Phys. Med. Rehabil.. 1997;7(2):46-49.
Bibbo C., Goldberg J.W. Infectious and healing complications after elective orthopaedic foot and ankle surgery during tumor necrosis factor-alpha inhibition therapy. Foot Ankle Int.. 2004;25(5):331-335.
Boardman N.D.3rd, Cofield R.H., Bengtson K.A., et al. Rehabilitation after total shoulder arthroplasty. J. Arthroplas.. 2001;16(4):483-486.
Borg G.E. An Introduction to Borg’s RPE scale. New York: Ithaca; 1985.
Boutry N., Lardé A., Lapègue F., et al. Magnetic resonance imaging appearance of the hands and feet in patients with early rheumatoid arthritis. J. Rheumatol.. 2003;30(4):671-679.
Brosseau, L., Welch, V. & Wells, G.A., et al., 2005. Low level laser therapy (Classes I, II and III) for treating rheumatoid arthritis. Cochrane Database of Systematic Reviews (4) Art. No.: CD002049. DOI: 10.1002/14651858.CD002049.pub2.
Brosseau, L., Yonge, K.A. & Welch, V., et al., 2003. Transcutaneous electrical nerve stimulation (TENS) for the treatment of rheumatoid arthritis in the hand. Cochrane Database of Systematic Reviews (2) Art No. CD004377. DOI: 10.1002/14651858.CD004377.
Burton W., Morrison A., Maclean R., et al. Systematic review of studies of productivity loss due to rheumatoid arthritis. Occup. Med.. 2006;56(1):18-27.
Casimiro, L., Barnsley, L. & Brosseau, L., et al., 2005. Acupuncture and electroacupuncture for the treatment of rheumatoid arthritis. Cochrane Database of Systematic Reviews (4) Art No. CD003788. DOI: 10.1002/14651858.CD003788. pub2.
Chalmers A.C., Busby C., Goyert J., et al. Metatarsalgia and rheumatoid arthritis: A randomized, single blind, sequential trial comparing 2 types of foot orthoses and supportive shoes. J. Rheumatol.. 2000;27:1643-1647.
Chung K.C., Pillsbury M.S., Walters M.R., et al. Reliability and validity testing of the Michigan Hand Outcomes Questionnaire. J. Hand Surg. [Am]. 1998;23:575-587.
Cimmino M.A., Parodi M., Silvestri E., et al. Correlation between radiographic, echographic and MRI changes and rheumatoid arthritis progression. Reumatismo. 2004;56(1):28-40.
Curtis R., Groarke A., Coughlan R., et al. Psychological stress as a predictor of psychological adjustment and health status in patients with rheumatoid arthritis. Patient Edu. Counsel.. 2005;59(2):192-198.
Dellhag B., Wollersjo I., Bjelle A. Effect of active hand exercise and wax bath treatment in rheumatoid arthritis patients. Arthritis Care Res.. 1992;5:87-92.
Dreyer S.J., Boden S.D. Natural history of rheumatoid arthritis of the cervical spine. Clin. Orthop. Relat. Res.. 1999;366:98-106.
Duruoz M.T., Poiraudeau S., Fermanian J., et al. Development and validation of a rheumatoid hand functional disability scale that assesses functional handicap. J. Rheumatol.. 1996;23(7):1167-1172.
Egan, M., Brosseau, L. & Farmer, M., et al., 2001. Splints and Orthosis for treating rheumatoid arthritis. Cochrane Database of Systematic Reviews (4) Art No. CD004018. DOI: 10.1002/14651858.CD004018.
El Miedany Y., Youssef S., Mehanna A.N., et al. Development of a scoring system for assessment of outcome of early undifferentiated inflammatory synovitis. Joint, Bone, Spine. 2008;75(2):155-162.
Epps H., Ginnelly L., Utley M., et al. Is hydrotherapy cost-effective? A randomised controlled trial of combined hydrotherapy programmes compared with physiotherapy land techniques in children with juvenile idiopathic arthritis. Health Technol. Ass.. 2005;9(39):1-59. iii-iv, ix-x
Fearing V.G., Clark J. Individuals in context: A practical guide to client centred practice. Thorofare, NJ: Slack Inc; 2000.
Felson D.T., Anderson J.J., Boers M., et al. The American College of Rheumatology preliminary core set of disease activity measures for rheumatoid arthritis clinical trials. Arthritis Rheum.. 1993;36:729-740.
Felson D.T., Anderson J.J., Boers M., et al. The American College of Rheumatology preliminary definition of improvement in rheumatoid arthritis. Arthritis Rheum.. 1995;38:727-735.
Ferucci E.D., Templin D.W., Lanier A.P. Rheumatoid arthritis in American Indians and native Alaskans: A review of the literature. Semin. Arthritis Rheum.. 2004;34:662-667.
Filippucci E., Iagnocco A., Meenagh G., et al. Ultrasound imaging for the rheumatologist VII. Clin. Exp. Rheumatol.. 2007;25(1):5-10.
Fries J.F., Spitz P.W., Kraines R.G., et al. Measurement of patient outcome in arthritis. Arthritis Rheum.. 1980;23:137-145.
Gabriel S., Crowson C.S., Kremers H.M., et al. The rising incidence of rheumatoid arthritis. Arthritis Rheum.. 2008;58(conference supplement):773. [abstract]
Gignac M.A.M., Cao X., Lacaille D., et al. Arthritis-related work transitions: A prospective analysis of reported productivity losses, work changes and leaving the labor force. Arthritis Rheum.. 2008;59(12):1805-1813.
Gilworth G., Chamberlain A., Harvey A., et al. Development of a work instability scale for rheumatoid arthritis. Arthritis Rheum.. 2003;49(3):349-354.
Hall J., Skevington S.M., Maddison P.J., et al. A randomised and controlled trial of hydrotherapy in rheumatoid arthritis. Arthritis Care Res.. 1996;9:206-215.
Hammond A., Lincoln N., Sutcliffe L. A crossover trial evaluating an educational-behavioural joint protection programme for people with rheumatoid arthritis. Patient Edu. Counsel.. 1999;37:19-32.
Hammond A., Freeman K. One-year outcomes of a randomized controlled trial of an educational-behavioural joint protection programme for people with rheumatoid arthritis. Rheumatology. 2001;40:1044-1051.
Hammond A. Rehabilitation in rheumatoid arthritis: a critical review. Musculoskeletal Care. 2004;2(3):135-151.
Han, A., Judd, M. & Welch, V., et al., 2004. Tai chi for treating rheumatoid arthritis. Cochrane Database of Systematic Reviews (3) Art No. Art No. CD004849. DOI: 10.1002/14651858.CD004849
Haskett S., Backman C., Porter B., et al. A crossover trial of commercial versus custom-made wrist splints in the management of inflammatory polyarthritis. Arthritis Rheum.. 2004;51:792-799.
Hawke, F., Burns, J. & Radford, J.A., et al., 2008. Custom-made foot orthoses for the treatment of foot pain. Cochrane Database of Systematic Reviews (3) Art No. CD006801. DOI: 10.1002/14651858.CD006801. pub2.
Haynes B.F. Pathology. In: St Clair E.W., Pisetsky D.S., Haynes B.F., editors. Rheumatoid Arthritis. Philadelphia: Lippincott Williams & Wilkins; 2004:118-139.
Helewa A., Goldsmith C.H., Lee P., et al. Effects of occupational therapy home service on patients with rheumatoid arthritis. Lancet. 1991;337:1453-1456.
Heller J.E., Shadick N.A. Outcomes in rheumatoid arthritis: incorporating the patient perspective. Curr. Opin. Rheumatol.. 2007;19(2):101-105.
Hennell S., Luqmani R. Developing multidisciplinary guidelines for the management of early rheumatoid arthritis. Musculoskeletal Care. 2008;6(2):97-107.
Hirvonen H.E., Mikkelsson M.K., Kautiainen H., et al. Effectiveness of different cryotherapies on pain and disease activity in active rheumatoid arthritis. A randomised single blinded controlled trial. Clin. Exp. Rheumatol.. 2006;24(3):295-301.
Hurley M.V., Mitchell H.L., Walsh N. In osteoarthritis, the psychosocial benefits of exercise are as important as physiological improvements. Exerc. Sport Sci. Rev.. 2003;31(3):138-143.
Hurley M.V., Dziedzic K., Bearne L., et al. The clinical and cost-effectiveness of physiotherapy management of elderly people with common rheumatological conditions. London: The Chartered Society of Physiotherapy; 2002.
Kastbom A., Strandberg G., Lindroos A., et al. Anti-CCP antibody predicts the disease course during 3 years in early rheumatoid arthritis. Ann. Rheum. Dis.. 2004;63:1085-1089.
Katz P.P., Alfieri W.S. Satisfaction with abilities and well-being: Development and validation of a questionnaire for use among persons with rheumatoid arthritis. Arthritis Care Res.. 1997;10(1):89-98.
Katz P.P., Pasch L.A., Wong B. Development of an instrument to measure disability in parenting activity among women with rheumatoid arthritis. Arthritis Rheum.. 2003;48:935-943.
Katz P.P., Yelin E.H. Life activities of persons with rheumatoid arthritis with and without depressive symptoms. Arthritis Care Res.. 1994;7(2):69-77.
Kennedy N. Exercise therapy for patients with rheumatoid arthritis: safety of intensive programmes and effects upon bone mineral density and disease activity: a literature review. Phys. Ther. Rev.. 2006;11(4):263-268.
Kremers H.M., Gabriel S.E. Epidemiology. In: St Clair E.W., Pisetsky D.S., Haynes B.F., editors. Rheumatoid Arthritis. Philadelphia: Lippincott Williams & Wilkins; 2004:1-10.
Lacaille D., White M.A., Backman C.L., et al. Problems faced at work due to inflammatory arthritis: new insights gained from understanding patients’ perspective. Arthritis Rheum.. 2007;57(7):1269-1279.
Lacaille D., White M.A., Rogers P.A., et al. Employment and arthritis: making it work – A proof of concept study. Arthritis Rheum.. 2008;59(11):1647-1655.
Law M., Baptiste S., Carswell A., et al. Canadian Occupational Performance Measure, fourth ed. Ottawa, ON: CAOT Publications; 2005.
Lerner D., Amick B.C., Rogers W.H., et al. The work limitations questionnaire. Med. Care. 2001;39:72-85.
Lee E.O., Kim J.I., Davis A.H., et al. Effects of regular exercise on pain, fatigue and disability in patients with rheumatoid arthritis. Fam. Commun. Health. 2006;29(4):320-327.
Lineker S.C., Hurley L., Wilkins A., et al. Investigating care provided by physical therapists treating people with rheumatoid arthritis: pilot study. Physiother. Can.. 2006;58(1):53-60.
Lorig K.R., Holman H. Self-management education: history, definition, outcomes, and mechanisms. Ann. Behav. Med.. 2006;26:1-7.
Luqmani R., Hennell S., Estrach C., et al. British Society for Rheumatology and British Health Professionals in Rheumatology guideline for the management of rheumatoid arthritis (the first 2 years). Rheumatology. 2006;45:1167-1169.
Mann W.C., Tomita M., Hurren D., et al. Changes in health, functional and psychosocial status and coping strategies of home-based older persons with arthritis over three years. Occup. Ther. J. Res.. 1999;19:126-146.
Mathiowetz V., Weber K., Volland G., et al. Reliability and validity of grip and pinch strength evaluations. J. Hand Surg. (Am). 1984;9:222-226.
Medical Research Council. Aids to the Investigation of Peripheral Nerve Injuries. London, UK: HMSO; 1976.
Meenan R.F., Mason J.H., Anderson J.J., et al. AIMS2: the content and properties of a revised and expanded Arthritis Impact Measurement Scales Health Status Questionnaire. Arthritis Rheum.. 1992;35(1):1-10.
Moncur C. Discipline-specific standards of care: Physical therapy competencies in Physical Therapy Management of Arthritis. New York: Churchill Livingstone, 1988. p 29-41
Moss P. Negotiating spaces in home environments: older women living with arthritis. Soc. Sci. Med.. 1997;45:23-33.
Murphy E., Bresnihan B., Fitzgerald O. Measurement of periarticular bone mineral density in the hands of patients with early inflammatory arthritis using dual energy x-ray absorpitometry. Clin. Rheumatol.. 2008;27(6):763-766.
NICE. (2009). Rheumatoid Arthritis: National clinical guideline for management and treatment in adults http://www.nice.org.uk/nicemedia/pdf/CG79FullGuideline.pdf(accessed 13.9.09)
Nishimura K., Sugiyama D., Kogata Y., et al. Meta-analysis: diagnostic accuracy of anti-cyclic citrullinated peptide antibody and rheumatoid factor for rheumatoid arthritis. Ann. Intern. Med.. 2007;146(11):797-808.
Nordenskiold U. Evaluation of assistive devices after a course in joint protection. Int. J. Technol. Assess. Health Care. 1994;10:293-304.
Nordenskiold U., Grimby G., Dahlin-Ivanoff S. Questionnaire to evaluate the effects of assistive devices and altered working methods in women with rheumatoid arthritis. Clin. Rheumatol.. 1998;17:6-16.
O’Brien A.V., Jones P., Mullis R., et al. Conservative hand therapy treatments in rheumatoid arthritis. Rheumatology. 2006;45:577-583.
O’Connor D., Kortman B., Smith A., et al. Correlation between objective and subjective measures of hand function in patients with rheumatoid arthritis. J. Hand Ther.. 1999;12:323-329.
Pagnotta A., Baron M., Korner-Bitensky N. The effect of a static wrist orthosis on hand function in individuals with rheumatoid arthritis. J. Rheumatol.. 1998;25:879-885.
Podsiadlo D., Richardson S. The timed “Up & Go”: a test of basic functional mobility for frail elderly persons. J. Am. Geriat. Soc.. 1991;39(2):142-148.
Poole J.L., Gallegos M., O’Linc S. Reliability and validity of the Arthritis Hand Function Test in adults with systemic sclerosis (scleroderma). Arthritis Care Res.. 2000;13(1):69-73.
Prevoo M.L., van’t Hof M.A., Kuper H.H., et al. Modified disease activity scores that include twenty-eight joint counts. Development and validation in a prospective longitudinal study of patients with rheumatoid arthritis. Arthritis Rheum.. 1995;38(1):44-48.
Rennie R.J. Evaluation of the effectiveness of a metacarpophalangeal ulnar deviation orthosis. J. Hand Ther.. 1996;9:371-377.
Riemsma, R.P., Kirwan, J.R. & Taal, E., et al., 2003. Patient education for adults with rheumatoid arthritis. Cochrane Database of Systematic Reviews (2) Art No. CD003688. DOI: 10.1002/14651858.CD003688.
Ronkainen A., Niskanen M., Auvinen A. Cervical spine surgery in patients with rheumatoid arthritis: longterm mortality determinants. J. Rheumatol.. 2006;33(3):517-522.
Shin Y., Choi J., Nahm D., et al. Rheumatoid factor is a marker of disease activity in Korean rheumatoid arthritis. Yonsei Med. J.. 2005;46(4):464-470.
Straub R.H., Dhabhar F.S., Bijlsma J.W.J., et al. How psychological stress via hormones and nerve fibers may exacerbate rheumatoid arthritis. Arthritis Rheum.. 2005;52(1):16-26.
Schumacher HR., Habre W., Meador R., Hsia E.C. Predictive factors in early arthritis: long-term follow-up. Semin. Arthritis Rheum.. 2004;33(4):264-272.
Steultjens E.M.J., Dekker J., Bouter L.M., et al. Occupational therapy for rheumatoid arthritis: a systematic review. Arthritis Rheum.. 2002;47:672-685.
Symmons D., Turner G., Webb R., et al. The prevalence of rheumatoid arthritis in the United Kingdom: New estimates for a new century. Rheumatology. 2002;41:793-800.
Symmons D.P.M. Epidemiology of rheumatoid arthritis: Determinants of onset, persistence and outcome. Best Prac. Res. Clin. Rheumatol.. 2002;16(5):707-722.
Tourinho T.F., Stein A., Castro J.A., et al. Rheumatoid arthritis: evidence for bone loss in premenopausal women. J. Rheumatol.. 2005;32(6):1020-1025.
Uetani M. Imaging approach for the evaluation of the bone and joint destruction in rheumatoid arthritis. Clin. Calcium. 2007;17(4):453-462.
Uhlig T., Lillemo S., Moe R.H., et al. Reliability of the ICF Core Set for rheumatoid arthritis. Ann. Rheum. Dis.. 2007;66:1078-1084.
Van den Ende C.H., Breedveld F.C., le Cessie S., et al. Effect of intensive exercise on patients with active rheumatoid arthritis: a randomised clinical trial. Ann. Rheum. Dis.. 2000;59:615-621.
Van den Ende, C.H., Vliet Vlieland, T.P.M., Munneke, M., et al., 2006. Dynamic exercise therapy for treating rheumatoid arthritis: a systematic review. Coch. Lib. (4).
van Lankveld W., van’t Pad Bosch P., Bakker J., et al. Sequential occupational dexterity assessment (SODA): a new test to measure hand disability. J. Hand Ther.. 1996;9(1):27-32.
Veehof M.M., Taal E., Heijnsdijk-Rouwenhorst L.M., et al. Efficacy of wrist working splints in patients with rheumatoid arthritis: A randomized controlled study. Arthritis Rheum.. 2008;59(12):1698-1704.
Verhagen AP, de Vet H.C.W., de Bie R.A., et al. Taking baths: the efficacy of balneotherapy in patients with arthritis. A systematic review. J. Rheumatol.. 1997;24:1964-1971.
Verhagen, A.P., Bierma-Zeinstra, S.M.A. & Boers, M., et al., 2004. Balneotherapy for rheumatoid arthritis. Cochrane Database of Systematic Reviews (1) Art No. CD000518. DOI: 10.1002/14651858. CD000518.
Wall J.C., Bell C., Campbell S., et al. The expanded timed get up and go test. Gait Post.. 1998;7:187.
Ware J.E., Kosinski M. The SF-36® Physical and Mental Health Summary Scales: A Manual for users of Version 1, second ed. Lincoln, RI: Quality Metric; 2001.
Welch, V., Brosseau, L. & Casimiro, L., et al., 2002. Thermotherapy for treating rheumatoid arthritis. Cochrane Database of Systematic Reviews (2) Art No. CD002826. DOI: 10.1002/14651858.CD002826.
Welsing P.M., van Gestel A.M., Swinkels H.L., et al. The relationship between disease activity, joint destruction and functional capacity over the course of rheumatoid arthritis. Arthritis Rheum.. 2001;44:2009-2017.
Wilkie R., Peat G., Thomas E., et al. The Keele assessment of participation: A new instrument to measure participation restriction in population studies. Combined qualitative and quantitative examination of its psychometric properties. Qual. Life Res.. 2005;14:1889-1899.
Wolfe F. Why the HAQ-II can be an effective substitute for the HAQ. Clin. Exp. Rheumatol.. 2005;23(Suppl 39):S29-S30.
World Health Organization. International Classification of Functioning, Disability and Health. Geneva: World Health Organization, 2002.
Yasura K., Nakagawa Y., Kobayashi M., et al. Mechanical and biochemical effect of monopolar radiofrequency energy on human articular cartilage: an in vitro study. Am. J. Sports Med.. 2006;34(8):1322-1327.
Zijlstra F.J., van den Berg-de-Lange I., Huygen F.J., et al. Anti-inflammatory actions of acupuncture. Mediators Inflam.. 2003;12(2):59-69.
Robinson V.A., Brosseau L., Casimiro L., et al. Thermotherapy for treating rheumatoid arthritis. Coch. Lib.. (4):2006.