Writing Research Proposals
With a background in the quantitative, qualitative, and outcomes research methodologies, you are ready to propose a study. A research proposal is a written plan that identifies the major elements of a study, such as the research problem, purpose, and framework, and outlines the methods and procedures to conduct the study. A proposal is a formal way to communicate ideas about a proposed study and to seek approval for conducting the study and funding. To seek approval for the conduct or funding of a study, the researcher submits a research proposal to a selected group for review and, in many situations, verbally defends that proposal. Receiving approval to conduct research has become more complicated because of the increasing complexity of nursing studies, the difficulty involved in recruiting subjects, and rising concerns over legal and ethical issues. In many large hospitals, both the lawyer and the institutional research review committee will evaluate the research proposals. The expanded number of health care studies being conducted has led to conflict among investigators over who has the right to recruit potential research subjects. This chapter focuses on writing a research proposal and seeking approval to conduct a study. Chapter 29 presents the process of seeking funding for research.
WRITING A RESEARCH PROPOSAL
A well-written proposal communicates a significant, carefully planned research project, demonstrates the qualifications of the researcher, and generates support for the project. Conducting research requires precision and rigorous attention to detail. Thus, reviewers often judge a researcher’s ability to conduct a study by the quality of the proposal. A quality study proposal is clear, concise, and complete (Dexter, 2000; Tornquist, 1999). Writing a quality proposal involves (1) developing ideas logically, (2) determining the depth or detail of the proposal’s content, (3) identifying critical points in the proposal, and (4) developing an aesthetically appealing copy.
Developing Ideas Logically
The ideas in a research proposal must logically build on each other to justify or defend a study, just as a lawyer would logically organize information in the defense of a client. The researcher builds a case to justify why a problem should be studied and proposes the appropriate methodology for conducting the study. Each step in the research proposal builds on the problem statement to give a clear picture of the study and its merit (Brink, 1993; Turabian, Booth, Colomb, & Williams, 2007). Universities, medical centers, federal funding agencies, and grant writing consultants have developed websites to help you and other researchers write successful proposals for quantitative, qualitative, and outcomes research; these sites include www.research.umich.edu/proposals/proposals.html,www.ninr.nih.gov/Training, and www.mayo.edu/research/crtp/resproposal.html. You can use a search engine of your choice, such as Google, and search for proposal guidelines, proposal writing tips, and courses on proposal development for different universities, medical centers, and government agencies. In addition, a variety of publications have been developed to help individuals improve their scientific writing skills (American Psychological Association [APA], 2001; Dexter, 2000; Pyrczak & Bruce, 2005; Turabian et al., 2007; University of Chicago Press Staff, 2003).
Determining the Depth of a Proposal
The depth of a proposal is determined by guidelines developed by schools of nursing, funding agencies, and institutions where research is conducted. Guidelines provide specific directions for the development of a proposal and should be followed explicitly. Omission or misinterpretation of a guideline is frequently the basis for rejection. In addition to following the guidelines, you need to determine the amount of information necessary to describe each step of your study clearly. The content in a proposal needs to be detailed enough to inform the reader yet concise enough to be interesting and easily reviewed. Often, the guidelines stipulate a page limit, which will determine the depth of the proposal. The relevant content of a research proposal is discussed later in this chapter.
Identifying Critical Points
The key or critical points in a proposal must be evident, even to a hasty reader. You might highlight your critical points with bold or italicized type. Sometimes researchers create headings to emphasize critical content, or they may organize the content into tables or graphs. It is critical in a proposal to detail the background and significance of the research problem, your purpose, your methodology/research design or qualitative method, and your research production plans (data collection and analysis plan, personnel, schedule, and budget) (Tornquist, 1999; Turabian et al., 2007).
Developing an Aesthetically Appealing Copy
An aesthetically appealing copy is typed without spelling, punctuation, or grammatical errors. Even a proposal with excellent content that is poorly typed or formatted will probably not receive the full attention or respect of the reviewer (Dexter, 2000). The format used in typing the proposal should follow the guidelines developed by the reviewer. If no particular format is requested, researchers commonly follow the APA (2001) format. An appealing copy is legible (the print is dark enough to be read) and neatly organized in a folder so that the reviewer can examine it easily.
CONTENT OF A RESEARCH PROPOSAL
The content of a proposal is written with the interest and expertise of the reviewers in mind. Proposals are typically reviewed by faculty, clinical agency members, and representatives of funding institutions. The content of a proposal varies with the reviewer, the guidelines developed for the review, and the type of study (quantitative or qualitative) proposed. This section addresses the content of (1) a student proposal for both quantitative and qualitative studies, (2) condensed research proposals, and (3) preproposals.
Content of a Student Proposal
Student researchers develop proposals to communicate their research projects to the faculty and members of university and agency research review committees. Student proposals are written to satisfy requirements for a degree and are usually developed according to guidelines outlined by the faculty. The faculty member who will be assisting with the research project (the chair of the student’s thesis or dissertation committee) generally reviews these guidelines with the student. Each faculty member has a unique way of interpreting and emphasizing aspects of the guidelines. In addition, a student needs to evaluate the faculty member’s background regarding a research topic of interest and determine whether a productive working relationship can be developed. Faculty members who are actively involved in their own research have extensive knowledge and expertise that can be helpful to a novice researcher. Both the student and the faculty member benefit when a student becomes involved in an aspect of the faculty’s research. This collaborative relationship can lead to the development of essential knowledge for providing evidenced-based nursing practice (Craig & Smyth, 2002; Melnyk & Fineout-Overholt, 2005).
The content of a student proposal usually requires greater detail than does a proposal developed for an agency or funding organization. The proposal is often the first three or four chapters of the student’s thesis or dissertation, and the proposed study is discussed in the future tense—that is, what the student will do in conducting the research. A student research proposal usually includes a title page, with the title of the proposal, the name of the investigator, and the date, and a table of contents that precede the proposal content.
Content of a Quantitative Research Proposal
A quantitative research proposal usually includes the following chapters or sections: (1) introduction, (2) review of relevant literature, (3) framework, and (4) methods and procedures. Some graduate schools require an in-depth development of these sections, whereas others require a condensed version of the same content. Another approach is that proposals for theses and dissertations be written in a format that can be transformed into a publication. Table 28-1 outlines the content often covered in the chapters of a quantitative research proposal.
Introduction: The introductory chapter identifies the research topic and problem and discusses their significance and background. The significance of the problem addresses its importance in nursing practice and the expected generalizability of the findings. A problem’s magnitude is partly determined by the interest of nurses, other health care professionals, policy makers, and health care consumers at the local, state, national, or international level. You can document this interest with sources from the literature. The background describes how the problem was identified and historically links the problem to the nursing practice. Your background information might also include one or two major studies conducted to resolve the problem, some key theoretical ideas related to the problem, and possible solutions to the problem. The background and significance will form the basis for your problem statement, which identifies what is not known and the need for further research. Follow your problem statement with a succinct statement of the research purpose or the goal of the study (Kerlinger & Lee, 2000; Peat, Mellis, Williams, & Xuan, 2002).
Review of Relevant Literature: The review of relevant literature provides an overview of the essential information that will guide you as you develop your study and includes relevant theoretical and empirical literature. The theoretical literature provides a background for defining and interrelating relevant study concepts, whereas the empirical literature includes a summary and critical appraisal of previous studies. Here you will discuss the recommendations made by other researchers, such as changing or expanding a study, in relation to the proposed study. The depth of the literature review varies; it might include only recent studies and theorists’ works, or it might be extensive and include a description and critical appraisal of a number of past and current studies and an in-depth discussion of theorists’ works. The literature review might be presented in a narrative format or in a pinch table that summarizes relevant studies (see Chapter 6) (Pinch, 1995). The literature review demonstrates that you have a command of the current empirical and theoretical knowledge regarding the proposed problem (Tornquist, 1999).
This chapter concludes with a summary. The summary includes a synthesis of the theoretical literature and findings from previous research that describe the current knowledge of a problem (Pinch, 1995). Gaps in the knowledge base are also identified, with a description of how the proposed study is expected to contribute to the nursing knowledge needed for evidence-based practice.
Framework: A framework provides the basis for generating and refining the research problem and purpose and linking them to the relevant theoretical knowledge in nursing or related fields. The framework includes concepts and relationships among concepts or propositions, which are sometimes represented in a model or a map. The concepts to be studied are conceptually defined and linked to the study variables. If you use another theorist’s or researcher’s model from a journal article or book, letters documenting permission to use this model from the publisher and the theorist or researcher must be included in your proposal appendices.
In some studies, research objectives, questions, or hypotheses are developed to direct the study. The objectives, questions, or hypotheses evolve from the research purpose and study framework, in particular the proposition(s), and identify the study variables. The variables are conceptually defined to show the link to the framework, and they are operationally defined to describe the procedures for manipulating or measuring the study variables. You also will need to define any relevant terms and to identify assumptions that provide a basis for your study.
Methods and Procedures: The researcher describes the design or general strategy for conducting the study, sometimes including a diagram of the design. Designs for descriptive and correlational studies are flexible and can be unique to the study being conducted. Because of this uniqueness, the descriptions need to include the design’s strengths and weaknesses. Presenting a design for quasi-experimental and experimental studies involves (1) describing how the research situation will be structured, (2) detailing the treatment to be implemented, (3) explaining how the effect of the treatment will be measured, (4) specifying the variables to be controlled and the methods for controlling them, (5) identifying uncontrolled extraneous variables and determining their impact on the findings, (6) describing the methods for assigning subjects to the treatment and control groups, and (7) exploring the strengths and weaknesses of a design. The design needs to account for all the objectives, questions, or hypotheses identified in the proposal. If a pilot study is planned, the design should include the procedure for conducting the pilot and for incorporating the results into the proposed study.
Your proposal should identify the target population to which your study findings will be generalized and the accessible population from which the sample will be selected. Outline the criteria you will use to select a study participant or subject, and present the rationale for these criteria. For example, a participant might be selected according to the following criteria: female age 18 to 60 years, hospitalized, and 1 day post–abdominal surgery. The rationale for these criteria might be that the researcher wants to examine the effects of selected pain management interventions on adult females who have recently experienced hospitalization and abdominal surgery. The sampling method and the approximate sample size are discussed in terms of their adequacy and limitations in investigating the research purpose. Usually, a power analysis is conducted to determine an adequate sample size to identify significant relationships and differences in studies.
A proposal includes a description of the proposed study setting, which frequently includes the name of the agency and the structure of the units or sites where the study is to be conducted. The specific setting is often identified in the proposal but not in the final research report. The agency you select should have the potential to generate the type and size of sample required for the study. Thus, your proposal might include the number of individuals who meet the sample criteria and are cared for by the agency in a given period of time. In addition, the structure and activities in the agency need to be able to accommodate the proposed design of the study. If you are not affiliated with this agency, then it would be helpful if you had a letter of support for your study from the agency.
Ethical considerations in a proposal include the rights of the subjects and the rights of the agency where the study is to be conducted. Describe how you plan to protect subjects’ rights as well as the risks and potential benefits of your study. Also address the steps you will take to reduce any risks that the study might present. Many agencies require a written consent form, and that form is often included in the appendices of the proposal. With the implementation of the Health Insurance Portability and Accountability Act (HIPAA), health care agencies and providers must have a signed authorization form from patients to release their health information for research (U.S. Department of Health and Human Services [DHHS], 2004) (see Chapter 9 for details). You must also address the risks and potential benefits of the study for the institution (Fullwood, Granger, Bride, & Taylor, 1999). If your study places the agency at risk, outline the steps you will take to reduce or eliminate these risks. It is also necessary for you to state that the proposal will be reviewed by the thesis or dissertation committee, university human rights review committee, and agency research review committee.
Describe the methods you will use to measure study variables, including each instrument’s reliability, validity, methods of scoring, and level of measurement. A plan for examining the reliability and validity of the instruments in the present study needs to be addressed. If an instrument has no reported reliability and validity, you may need to conduct a pilot study to examine these qualities. If the intent of the proposed study is to develop an instrument, describe the process of instrument development. If physiological instruments are used, address the instrument’s accuracy, precision, sensitivity, selectivity, and error rate. A copy of the interview questions, questionnaires, scales, physiological instruments, or other tools to be used in the study is usually included in the proposal appendices. You must obtain permission from the authors to use copyrighted instruments, and letters documenting that permission has been obtained must be included in the proposal appendices.
The data collection plan clarifies what data are to be collected and the process for collecting the data. In this plan you will identify the data collectors, describe the data collection procedures, and present a schedule for data collection activities. If more than one person will be involved in data collection, it is important to describe methods used to train your data collectors to ensure consistency. The method of recording data is often described, and sample data recording sheets are placed in the proposal appendices. Also discuss any special equipment you will use or develop to collect data for the study, and address data security, including the methods of data storage.
The plan for data analysis identifies the analysis techniques that the researcher will use to summarize the demographic data and answer the research objectives, questions, or hypotheses. The analysis section is best organized by the study objectives, questions, or hypotheses. The analysis techniques identified need to be appropriate for the type of data collected. For example, if an associative hypothesis is developed, correlational analysis is planned. If a researcher plans to determine differences among groups, the analysis techniques might include a t-test or analysis of variance (ANOVA) (Corty, 2007; Kerlinger & Lee, 2000). A level of significance (α = 0.05, 0.01, or 0.001) is also identified. Often, a researcher projects the type of results that will be generated from data analysis. Dummy tables, graphs, and charts can then be developed to present these results and are included in the proposal appendices. The investigator might also project the possible findings for a study. For example, the researcher might consider what support or nonsupport of a proposed hypothesis would mean in light of the study framework and previous research findings. Projecting a study’s findings encourages others to logically examine the findings when the study is complete.
The methods and procedures chapter of a proposal usually concludes with a discussion of the study’s limitations and a plan for communication of the findings. Both methodological and theoretical limitations are addressed. Methodological limitations might include areas of weakness in the design, sampling method, sample size, measurement tools, data collection procedures, or data analysis techniques, whereas theoretical limitations set boundaries for the generalization of study findings. For example, the accuracy with which the conceptual definitions and relational statements in a theory reflect reality has a direct impact on the generalization of study findings. Theory that has withstood frequent testing through research provides a stronger framework for the interpretation and generalization of findings. A plan is included for communicating the research through presentations and publications to audiences of nurses, other health professionals, policy makers, and health care consumers.
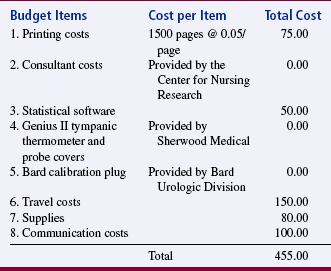
Frequently, a budget and timetable are included in the proposal appendices. The budget projects the expenses for the study, which might include the cost for data collection tools and procedures; special equipment; consultants for data analysis; computer time; travel related to data collection and analysis; typing; copying; and developing, presenting, and publishing the final report. Study budgets requesting external funding for researchers’ time include investigators’ salaries and secretarial costs. You will need a timetable to direct the steps of your research project and increase the chance that you will complete the project on schedule. A timetable identifies the tasks to be done, who will accomplish these tasks, and when these tasks will be completed. An example proposal for a quantitative study is presented at the end of this chapter.
Content of a Qualitative Research Proposal
A qualitative research proposal might include content similar to that of a quantitative proposal, but the guidelines are unique for the development of knowledge and theories using a variety of qualitative research methods. A qualitative proposal usually includes chapters with the following foci: (1) introduction, (2) research paradigm and general method, (3) applied method of inquiry, and (4) current knowledge, limitations, and plans for communication of the study findings (Boyd & Munhall, 2001; Marshall & Rossman, 2006; Patton, 2002; Sandelowski, Davis, & Harris, 1989). We propose the guidelines in Table 28-2 to assist you in developing a qualitative research proposal.
Introduction: The introduction usually provides a general background for the proposed study by identifying the phenomenon to be investigated and linking this phenomenon to nursing knowledge. The general aim or purpose of the study is identified and indicates the type of qualitative study to be conducted. The study aim is often followed by research questions that direct the investigation (Boyd & Munhall, 2001; Parse, Coyne, & Smith, 1985). For example, an aim for a qualitative study might be to “describe the lived experience of coping with the chronic illness of type 1 diabetes.” The research questions would focus on real-world problems and dilemmas, such as the following: How do people cope with a new diagnosis of type 1 diabetes? What is it like to live with the chronic illness of diabetes for 1 year, 2 years, 5 years, and 10 years? What type of support exists for a person with type 1 diabetes? What does it mean to have your diabetes under control? What is the impact of the chronic illness diabetes on the individual and family?
The introduction also includes the evolution of the study and its significance to nursing practice, patients, the health care system, and health policy. The discussion of the evolution of the study often includes how the problem developed (historical context), who or what is affected by the problem, and the researcher’s experience with the problem (experiential context) (Boyd & Munhall, 2001). Whenever possible, the significance and evolution of the study aim needs to be documented from the literature. Marshall and Rossman (2006) identified the following questions to assess the significance of a study: (1) Who has an interest in this domain of inquiry? (2) What do we already know about the topic? (3) What has not been answered adequately in previous research and practice? and (4) How will this research add to knowledge, practice, and policy in this area? The introduction section concludes with an overview of the remaining sections that are covered in the proposal.
Research Paradigm and General Methods for the Proposed Study: This section introduces you to the conceptual foundation for the qualitative research method (phenomenological, ethnographic, grounded theory, historical, philosophical inquiry, or critical social theory) selected for the proposed study. The researcher provides a rationale for the qualitative method selected and discusses its ability to generate the knowledge needed in nursing. Qualitative research methods are supported by different paradigms, such as naturalistic, postpositivism, feminism, and postmodernism (Miller, 1997). The investigator introduces the research paradigm and provides the essential elements of philosophy and assumptions of the specific type of qualitative study to be conducted. The philosophy varies for the different types of qualitative research and guides the conduct of the study. For example, a proposal for a phenomenological study might identify the study aim, “to describe the experience of women recovering from substance abuse,” discuss the naturalistic paradigm as a basis of understanding for the study, and describe the works of Giorgi (1985) and Frankl (1984) as the philosophical basis for conducting the study (Wright, 2003). Assumptions about the nature of the knowledge and the reality that underlie the type of qualitative research to be conducted are also identified. The assumptions and philosophy provide a theoretical perspective for the study that influences the focus of the study, data collection and analysis, and articulation of the findings.
Method of Inquiry: Applied: Developing and implementing the methodology of qualitative research require a certain expertise that some believe can only be obtained through a mentorship relationship with an experienced qualitative researcher. The role of the researcher and the intricate techniques of data collection and analysis are thought to be best communicated through a one-to-one relationship. Thus, planning the methods of a qualitative study requires knowledge of relevant sources that describe the different qualitative research techniques and procedures (Boyd & Munhall, 2001; Chenitz & Swanson, 1986; Leininger, 1985; Marshall & Rossman, 2006; Miles & Huberman, 1994; Munhall, 2001; Parse et al., 1985; Patton, 2002), in addition to requiring interaction with a qualitative researcher. The proposal needs to reflect the researcher’s credentials for conducting the particular type of qualitative study proposed.
Identifying the methods for conducting a qualitative study is a difficult task because the design of the study is still emerging. Unlike quantitative research, in which the design is a fixed blueprint for a study, the design in qualitative research emerges or evolves as the study is conducted. Thus, you must document the logic and appropriateness of the qualitative method and develop a tentative plan for conducting your study. Because this plan is tentative, reserve the right to modify or change the plan as needed during the conduct of the study (Sandelowski et al., 1989). However, the design or plan must be consistent with the study aim and research questions, be well conceived, and address prior criticism, as appropriate (Cohen, Knafl, & Dzurec, 1993). The tentative plan describes the process for selecting a site and population. The site will allow you entry and will include the participants necessary to answer the research aim and questions. For the research question “How do individuals cope with a new diagnosis of type 1 diabetes?” the participants might be identified in hospitals, clinics, practitioners’ offices, home care organizations, or rehabilitation facilities, and data collection might be conducted in the participants’ homes.
The researcher must gain entry into the setting, develop a rapport with the participants that will facilitate the detailed data collection process, and protect the rights of these participants (Marshall & Rossman, 2006; Sandelowski et al., 1989; U.S. DHHS, 2004). Address the following questions in describing the researcher’s role: (1) What is the best setting for the study? (2) How will I ease my entry into the research site? (3) How will I gain access to the participants? (4) What actions will I take to encourage the participants to cooperate? (5) What precautions will I take to protect the rights of the participants and to prevent the setting and the participants from being harmed? You need to describe the process you followed to obtain informed consent and the actions you will use to decrease study risks. The sensitive nature of some qualitative studies increases the risk for participants, which makes ethical concerns and decisions a major focus of the study (Munhall, 2001; Patton, 2002).
The primary data collection techniques used in qualitative research are observation and in-depth interviewing. Observations can range from highly detailed, structured notations of behavior to ambiguous descriptions of behavior or events. The interview can also range from structured, closed-ended questions to unstructured, open-ended questions (Marshall & Rossman, 2006; Munhall, 2001). Address the following questions when describing the proposed data collection process: (1) Who will collect data and provide any training required for the data collectors? (2) What data will be collected? For example, will the data be field notes from memory, audiotapes of interviews, transcripts of conversations, videotapes of events, or examination of existing documents? (3) What techniques or procedures will my research team use to collect the data? For example, if interviews are to be conducted, will I include a list of the proposed questions in the appendix? In historical research, data are collected through an exhaustive review of published and unpublished literature. (4) Where are the identified sources of data located? (5) How much time will I need to collect data? (6) How will my data be recorded and stored?
The methods section also needs to address how you will document the research process. For example, you might keep progress notes during the course of the study. These notes can document the day-to-day activities, methodological events, decision-making procedures, and personal notes about the informants. This information becomes part of the audit trail that you can provide to ensure the quality of the study (Miles & Huberman, 1994; Munhall, 2001; Patton, 2002).
The methods section of the proposal also includes the analysis techniques and the steps for conducting these techniques. In qualitative research, data collection and analysis often occur simultaneously. The data are in the form of notes, tapes, and other material obtained from observation, interviews, and completing questionnaires. Through qualitative analysis techniques, these data are structured and reduced to determine meaning (Miles & Huberman, 1994; Patton, 2002). Determine what strategies you will apply to ensure the credibility, fittingness, and auditability of the findings (Beck, 1993). These qualitative terms relate to the concepts of reliability and validity used in quantitative research and are addressed inChapters 23 and 26.
Current Knowledge Base, Limitations, and Plans for Communication of the Study: This section of the proposal summarizes and documents all relevant literature that was reviewed for the study. In some qualitative studies, the researcher reviews the literature before conducting the study. For example, ethnographic research requires a literature review to provide a background for conducting the study, as in quantitative research. Historical research involves a literature review to develop research questions, as does philosophical inquiry to generate philosophical questions. The literature review needs to provide a basis for the aims of the study and clarify how this study will expand nursing knowledge (Boyd & Munhall, 2001; Marshall & Rossman, 2006).
In phenomenological research, grounded theory research, and critical social theory, the literature review is usually conducted toward the end of the research project. The findings from a phenomenological study are compared and combined with findings from the literature to determine current knowledge of the phenomenon. In grounded theory research, the literature is used to explain, support, and extend the theory generated in the study. Study findings obtained through critical social theory are examined in light of the existing literature to determine the current knowledge of a social situation (see Chapter 4).
As the researcher, you will need to describe how the literature reviewed has influenced your proposed research methods. Biases and previous experience with the research problem need to be addressed, as does their potential impact on the proposed study. Often, anticipated findings, hypotheses, and hunches are identified before the study is conducted, followed by a discussion of the procedures that might be used to remain open to new information. You will also need to address the limitations of the study in the context of limitations of similar studies.
Conclude your proposal by describing how you plan to communicate your findings to a variety of audiences through presentations and publications. Often, a realistic budget and timetable are provided in the appendix. A qualitative study budget is similar to a quantitative study budget and includes costs for data collection tools and procedures; consultants for data analysis; travel related to data collection and analysis; typing and copying related to data collection and analysis; and developing, presenting, and publishing the final report. However, the greatest expenditure in qualitative research is usually the researcher’s time. Develop a timetable to project how long the study will take; often 2 years or more is designated for data collection and analysis (Marshall & Rossman, 2006; Munhall, 2001; Patton, 2002). You can use your budget and timetable to make decisions regarding funding inasmuch as funding is essential for many qualitative studies.
Some excellent websites have been developed to assist novice researchers in identifying an idea for qualitative study and developing a qualitative research proposal and reports (for example, see www.nova.edu/ssss/QR/qualres.html). The Office of Behavior and Social Sciences Research within the National Institutes of Health (NIH) has developed a website to assist researchers in developing qualitative and quantitative research proposals for funding (see http://grants.nih.gov/grants/writing_application.htm). You can use these websites and other publications to promote the quality of your qualitative research proposal. The quality of a proposal is based on the potential scientific contribution of the research to nursing knowledge; the research paradigm guiding the study; the research methods; and the knowledge, skills, and resources available to the investigators (Cohen et al., 1993; Marshall & Rossman, 2006; Munhall, 2001; Patton, 2002).
Content of a Condensed Proposal
The content of proposals developed for review by clinical agencies and funding institutions is usually a condensed version of the student proposal. However, even though these proposals are condensed, the logical links between components of the study need to be clearly demonstrated. A condensed proposal often includes a statement of the problem and purpose; previous research that has been conducted in the area (usually limited to no more than three to five studies); the framework, variables, design, sample, ethical considerations, and plans for data collection and analysis; and plans for dissemination of findings.
A proposal submitted to a clinical agency needs to identify the specific setting clearly, such as the emergency department or intensive care unit, as well as the projected time span for the study. Members of clinical agencies are particularly interested in the data collection process and involvement of institutional personnel in the study. The researcher will need to identify any expected disruptions in institutional functioning, with plans for preventing these disruptions when possible. The researcher must recognize that anything that slows down or disrupts employee functioning costs the agency money and can interfere with the quality of patient care. By showing that you are aware of these concerns and have proposed ways to minimize their effects, you increase the probability of obtaining approval to conduct your study.
Various companies, corporations, and organizations provide funding for research projects. A proposal developed for these types of funding institutions frequently includes a brief description of the study, the significance of the study to the institution, a timetable, and a budget. Most of these proposals are brief and might contain a one-page summary sheet or abstract at the beginning of the proposal that summarizes the steps of the study. The salient points of the study are included on this page in simple, easy-to-read, nontechnical terminology. Some proposal reviewers for funding institutions are laypersons with no background in research or nursing. An inability to understand the terminology might put the reviewer on the defensive or create a negative reaction, which could lead to disapproval of the study. When a funding institution is examining multiple studies, the summary sheet is often the basis for final decisions. The summary should be concise, informative, and designed to sell the study.
In proposals for both clinical and funding agencies, investigators need to document their research background and supply curriculum vitae if requested. The research review committee for approval of funding will be interested in previous research, research publications, and clinical expertise, especially if a clinical study is proposed. If you are a graduate student, the committee may request the names of the university committee members and verification that your proposal has been approved by the student’s thesis or dissertation committee and the university human subjects review committee.
Content of a Preproposal
Sometimes a researcher will send a preproposal or query letter rather than a proposal to a funding institution. A preproposal is a short document of four to five pages plus appendices that explores the funding possibilities for a research project. The parts of the preproposal are logically ordered as follows: “(1) letter of transmittal, (2) proposal for research, (3) personnel, (4) facilities, and (5) budget” (Malasanos, 1976, p. 223). The proposal provides a brief overview of the proposed project, including the research problem, purpose, methodology (brief description), and, most important, a statement of the significance of the work to knowledge in general and the funding institution in particular. By developing a preproposal, researchers are able to determine the agencies interested in funding their study and limit submission of their proposals to only institutions that indicate an interest.
SEEKING APPROVAL FOR A STUDY
Initially, only graduate students developing theses or dissertations and researchers seeking grant money were subject to proposal reviews. However, as a consequence of stricter rules related to the protection of human subjects, most nursing studies are reviewed by at least one research committee. Seeking approval to conduct a study is an action that should be based on knowledge and guided by purpose. Obtaining approval for a study from a research review committee requires understanding the approval process, writing a research proposal for review, and, in many cases, verbally defending the proposal. Little has been written to guide the researcher who is going through the labyrinth of approval mechanisms. This section provides a background for obtaining approval to conduct a study.
Clinical agencies and health care corporations review studies for the following reasons: (1) to evaluate the quality of the study, (2) to ensure that adequate measures are being taken to protect human subjects, and (3) to evaluate the impact of the study on the reviewing institution (Fullwood et al., 1999). What does the researcher hope to result from this institutional review? Most hope to receive approval to collect data at the reviewing institution and to obtain support for the proposed study.
Approval Process
An initial step in seeking approval is to determine exactly what committees in which agencies must grant approval before the study can be conducted. You need to take the initiative to determine the formal approval process rather than assume that you will be told if a formal review system exists. Information on the formal research review system might be obtained from administrative personnel, special projects or grant officers, chairs of institutional review boards (IRBs) in clinical agencies, clinicians who have previously conducted research, and university faculty who are involved in research.
Graduate students usually require approval from their thesis or dissertation committee, the university human subjects review committee, and the agency IRB where the data are to be collected. University faculty conducting research seek approval through the latter two committees. Nurses conducting research in an agency where they are employed must seek approval only at that agency. If the researcher seeks outside funding, additional review committees are involved. Not all studies require full review by the IRB (see Chapter 9 for the types of studies that qualify for exempt or expedited review). However, the IRB, not the researcher, determines the type of review that the study requires for conduct in that agency.
When multiple committees must review a study, sometimes the respective committees agree that the review for the protection of human subjects will be done by only one of the committees, with the findings of that committee generally being accepted by the other committees. For example, if the university human subjects review committee examined and approved a proposal for the protection of human subjects, funding agencies usually recognize that review as sufficient. Reviews in other committees then focus on approval to conduct the study within the institution or decisions to provide study funding.
As part of the approval process, the researcher must determine the agency’s policy regarding (1) the use of the clinical facility’s name in reporting findings, (2) the presentation and publication of the study, and (3) the authorship of publications. The facility’s name is used when presenting or publishing a study only with prior written administrative approval. The researcher may feel freer to report findings that could be interpreted negatively in terms of the institution if the agency is not identified. Some institutions have rules that limit what is presented or published in a study, where it is presented or published, and who is the presenter or author. Before conducting a study, researchers, especially employees of health care agencies, must clarify the rules and regulations of the agency regarding authorship, presentations, and publications. In some cases, recognition of these rules must be included in the proposal if it is to be approved.
Preparing Proposals for Review Committees
The initial proposals for theses and dissertations are often developed as part of a formal class. The faculty members teaching the class provide students with specific proposal guidelines approved by the graduate faculty and assist them in developing their initial proposals. If students elect to conduct a thesis, they ask an appropriate faculty member to serve as chair. With the chair’s assistance, the student identifies committee members with expertise in the focus of the proposed study or in conducting research who can work effectively together to refine the final proposal. The number of committee members varies for theses (usually a chair and two members) and dissertations (often a chair and four members) and with the university requirement. This proposal requires approval by the thesis or dissertation committee and the university human rights review committee or IRB.
Conducting research in a clinical agency requires approval by the agency’s IRB. This committee has the responsibility to (1) provide researchers with copies of institutional policies and requirements, (2) screen proposals for conducting research in the agency, and (3) assist the researcher with the IRB process (Vessey & Campos, 1992). The approval process policy and proposal guidelines are usually available from the chair of the IRB; and the guidelines should be followed carefully, particularly page limitations. Some committees refuse to review proposals that exceed these limitations. Reviewers on these committees are usually evaluating proposals in addition to other full-time responsibilities, and their time is limited.
Investigators also need to be familiar with the IRB’s process for screening proposals. Most agency IRBs screen proposals for (1) scientific merit, (2) protection of human rights, (3) congruence of the study with the agency’s research agenda, and (4) impact of the study on patient care (Vessey & Campos, 1992). Researchers need to develop their proposal with these ideas in mind. They also need to determine whether the committee requires specific forms to be completed and submitted with the research proposal. Other important information can be gathered by addressing the following questions: (1) How often does the committee meet? (2) How long before the next meeting? (3) What materials should be submitted before the meeting? (4) When should these materials be submitted? (5) How many copies of the proposal are required? (6) What period of time is usually involved in committee review?
Social and Political Factors
Social and political factors play an important role in obtaining approval to conduct a study. You will need to treat the review process with as much care as development of the study. The dynamics of the relationships among committee members is important to assess. This detail is especially important in the selection of a thesis or dissertation committee to ensure that the members are willing to work together productively. Thorough assessment of the social and political situation in which the study will be reviewed and implemented may be crucial to success of the study.
Clinical agency IRBs may include nurse clinicians who have never conducted research, nurse researchers, and researchers in other disciplines. The reactions of each of these groups to a study could be very different. Sometimes committees are made up primarily of physicians, which is frequently the case in health science centers. Physicians are often not oriented to nursing research methods. The lack of control in nursing studies concerns them, and some believe that the topics of these studies are not important. Sometimes they do not see the nurse researcher as credible because of educational differences, lack of previous experience in research, and few published studies. However, not all physicians view nursing research negatively. Many are strong supporters of nursing research, helpful in suggesting changes in design to strengthen the study, and eager to facilitate access to subjects.
The researcher needs to anticipate potential responses of committee members, prepare the proposal to elicit a favorable response, and consider means of minimizing negative responses. It is wise to meet with the chair of the agency IRB early in the development of a proposal. This meeting could facilitate proposal development, rapport between the researcher and agency personnel, and approval of the research proposal.
In addition to the formal committee approval mechanisms, you will need the tacit approval of the administrative personnel and staff who will be affected in some way by the study. Obtaining informal approval and support often depends on the way in which a person is approached. Demonstrate interest in the institution and the personnel, as well as interest in the research project. The relationships formed with agency personnel should be equal, sharing ones, because these people can often provide ideas and strategies for conducting the study that you may not have considered. The support of agency personnel during data collection can also make the difference between a successful and an unsuccessful study (Fullwood et al., 1999).
Conducting nursing research can benefit the institution, as well as to the researcher. Clinicians have an opportunity to see nursing research in action, which can influence their thinking and clinical practice if the relationship with the researcher is positive. These clinicians may be having their first close contact with a researcher, and interpretation of the researcher’s role and the aspects of the study may be necessary. In addition, clinicians tend to be more oriented in the present than researchers are, and they need to see the immediate impact that the study findings can have on nursing practice in their institution. Interactions with researchers might help clinicians see the importance of research in providing evidence-based practice and encourage them to become involved in study activities in the future (Craig & Smyth, 2002). Conducting research and providing evidence-based practice are essential if a hospital is to achieve and maintain magnet status. The award of magnet status from the American Nursing Credentialing Center (ANCC) is prestigious to an institution and validates the excellence in evidence-based nursing care provided by the facility.
Verbal Defense of a Proposal
Graduate students writing theses or dissertations are frequently required to defend their proposal verbally to their university committee members, which is called a thesis or dissertation proposal defense. Most clinical agencies require researchers to meet with the IRB to defend their proposals. In a verbal defense, reviewers can evaluate the researcher as a person, the researcher’s knowledge and understanding of the content of the proposal, and his or her ability to reason and provide logical explanations related to the study. And as a researcher, these face-to-face meetings give you opportunity to persuade reluctant committee members to approve your study.
Appearance is important in a personal presentation because it can give an impression of competence or incompetence. These presentations are business-like, with logical and rational interactions, so one should dress in a business-like manner. The committee might perceive individuals who are casually dressed as not valuing the review process.
Nonverbal behaviors are important during the meeting as well, so appearing calm, in control, and confident projects a positive image. Plan and rehearse your presentation to reduce anxiety. Obtain information on the personalities of committee members, their relationships with each other, the vested interests of each member, and their areas of expertise, as this can increase your confidence and provide a sense of control. It is important to arrive at the meeting early, assess the environment for the meeting, and carefully select a seat. Because you are the presenter, all members of the committee need to be able to see you. However, selecting a seat on one side of a table with all the committee members on the other side could make you feel uncomfortable and simulate an interrogation rather than a scholarly interaction. Sitting at the side of a table rather than at the head might be a strategic move to elicit support.
The verbal defense usually begins with a brief presentation of the study. Your presentation needs to be carefully planned, timed, and rehearsed. Salient points should be highlighted, which you can accomplish with the use of audiovisuals. After the presentation the reviewers will ask questions, so be prepared to defend or justify the methods and procedures used in your study. Sometimes it is beneficial to practice responding to questions related to the study with a friend; this rehearsal will help you to determine the best way to defend your ideas without appearing defensive. When the meeting ends, thank the members of the committee for their time. If the committee did not make a decision regarding the study during the meeting, ask when the decision will be made.
Revising a Proposal
Reviewers sometimes suggest changes in a proposal; however, some of these changes may benefit the institution, but not the study. Remain receptive to the suggestions, explore with the committee the impact of the changes on the proposed study, and try to resolve the conflict. If the conflict cannot be resolved, you might need to find another setting.
Many times reviewers make valuable suggestions that might improve the quality of a study or facilitate the data collection process. Revision of the proposal is often based on these suggestions before the study is implemented. Sometimes a study requires revision while it is being conducted because of problems with data collection tools or subjects’ participation. However, if clinical agency personnel or representatives of funding institutions have approved a proposal, the researcher needs to examine the situation seriously before making major changes in the study.
Before revising a proposal, address three questions: (1) What needs to be changed? (2) Why is the change necessary? (3) How will the change affect implementation of the study and the study findings? Students need to seek advice from the faculty before revising their studies. Sometimes it is beneficial for seasoned researchers to discuss their proposed study changes with other researchers or agency personnel for suggestions and additional viewpoints.
If a revision is necessary, revise your proposal and discuss the change with members of the IRB in the agency where the study is being conducted. The committee members might indicate that the investigators can proceed with the study or that they will have to seek approval for the revised proposal. If a study is funded, the study changes must be discussed with the representatives of the funding agency. The funding agency has the power to approve or disapprove the changes. However, realistic changes that are clearly described and backed with a rationale will probably be approved.
SAMPLE QUANTITATIVE RESEARCH PROPOSAL
The following proposal of a quantitative study will help you to prepare your first research proposal. The proposal was developed by a thesis student to conduct a quasi-experimental study (Ulbrich, 1995). The content of this proposal is brief and does not include the detail normally presented in a thesis or dissertation. The example provides ideas regarding the content areas that would be covered in developing a proposal for a quantitative study.
SUMMARY
• This chapter focuses on writing a research proposal and seeking approval to conduct a study.
• A research proposal is a written plan that identifies the major elements of a study, such as the problem, purpose, and framework, and outlines the methods and procedures to conduct a study.
• Writing a quality proposal involves (1) developing the ideas logically, (2) determining the depth or detail of the proposal content, (3) identifying the critical points in the proposal, and (4) developing an aesthetically appealing copy.
• A quantitative research proposal usually has four chapters or sections: (1) introduction, (2) review of relevant literature, (3) framework, and (4) methods and procedures.
• A qualitative research proposal generally includes the following chapters or sections: (1) introduction; (2) research paradigm and general method; (3) applied method of inquiry; and (4) current knowledge, limitations, and plans to communicate the study.
• Most clinical agencies and funding institutions require a condensed proposal, which usually includes a problem, a purpose, previous research conducted in the area, a framework, variables, design, sample, ethical considerations, plan for data collection and analysis, and plan for dissemination of findings.
• Sometimes a researcher will send a preproposal or query letter to a funding institution rather than a proposal; and the parts of the preproposal are logically ordered as follows: (1) letter of transmittal, (2) proposal for research, (3) personnel, (4) facilities, and (5) budget.
• Seeking approval for the conduct or funding of a study is a process that involves submission of a proposal to a selected group for review and, in many situations, verbally defending that proposal.
• Research proposals are reviewed to (1) evaluate the quality of the study, (2) ensure that adequate measures are being taken to protect human subjects, and (3) evaluate the impact of conducting the study on the reviewing institution.
• Proposals sometimes require revision before or during the implementation of a study; and if a change is necessary, the researcher needs to discuss the change with the members of the university and clinical agency IRBs and the funding agency.
• The chapter concludes with an example of a brief quantitative research proposal of a quasi-experimental study (Ulbrich, 1995).
REFERENCES
Alexander, D., Kelly, B. Cost effectiveness of tympanic thermometry in the pediatric office setting. Clinical Pediatrics. 1991;30(4 Suppl):57–59.
Alexander, D., Kelly, B. Responses of children, parents, and nurses to tympanic thermometry in the pediatric office. Clinical Pediatrics. 1991;30(4 Suppl):53–56.
American Psychological Association. Publication manual of the American Psychological Association, (5th ed.). Washington, DC: Author; 2001.
Bates, B. A guide to physical examination, (3rd ed.). Philadelphia: Lippincott; 1983.
Beck, C.T. Qualitative research: The evaluation of its credibility, fittingness, and auditability. Western Journal of Nursing Research. 1993;15(2):263–266.
Bland, J.M., Altman, D.G. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet. 1986;1(8476):307–310.
Boyd, C.O., Munhall, P.L. Qualitative research proposal and report. In: Munhall P.L., ed. Nursing research: A qualitative perspective. 3rd ed. Sudbury, MA: Jones & Bartlett; 2001:613–638.
Brink, H. Academic nurse leaders’ interpretation of concepts and priorities related to the examination of scientific short papers, dissertations and theses: Part 1. Curationis. 1993;16(3):62–67.
Bruce, J.L., Grove, S.K. Fever: Pathology and treatment. Critical Care Nurse. 1991;12(1):40–49.
Burns, N., Grove, S.K. The practice of nursing research: Conduct, critique, and utilization, (5th ed.). Philadelphia: Saunders; 2005.
Chenitz, W.C., Swanson, J.M. From practice to grounded theory: Qualitative research in nursing. Menlo Park, CA: Addison-Wesley, 1986.
Cohen, M.Z., Knafl, K., Dzurec, L.C. Grant writing for qualitative research. Image: Journal of Nursing Scholarship. 1993;25(2):151–156.
Corty, E.W. Using and interpreting statistics: A practical text for the health, behavioral, and social sciences. St. Louis: Mosby, 2007.
Craig, J.V., Smyth, R.L. The evidence-base practice manual for nurses. Edinburgh: Churchill Livingstone, 2002.
Dexter, P. Tips for scholarly writing in nursing. Journal of Professional Nursing. 2000;16(1):6–12.
Doyle, F., Zehner, W.J., Terndrup, T.E. The effect of ambient temperature on tympanic and oral temperatures. American Journal of Emergency Medicine. 1992;10(4):285–289.
Egan, E.C., Snyder, M., Burns, K.R. Intervention studies in nursing: Is the effect due to the independent variable? Nursing Outlook. 1992;40(4):187–190.
Erickson, R.S., Meyer, L.T. Accuracy of infrared ear thermometry and other temperature methods in adults. American Journal of Critical Care. 1994;3(1):40–54.
Erickson, R.S., Woo, T.M. Accuracy of ear thermometry and traditional temperature methods in young children. Heart & Lung. 1994;23(3):181–195.
Erickson, R.S., Yount, S.T. Comparison of tympanic and oral temperatures in surgical patients. Nursing Research. 1991;40(2):90–93.
Fraden, J. Noncontact temperature measurements in medicine. In: Wise D.L., ed. Bioinstrumentation and biosensors. New York: Marcel Dekker; 1991:511–549.
Frankl, V.E. Man’s search for meaning. New York: Simon & Schuster, 1984.
Fullwood, J., Granger, B.B., Bride, W., Taylor, M.C. Heart center nursing research: A team effort. Progress in Cardiovascular Nursing. 1999;14(1):25–29.
Giorgi, A. Phenomenology and psychological research. Pittsburgh: Duquesne University Press, 1985.
Guthrie, K.A., Keunke, N.E. Tympanic-based core temperature measurement in relation to thermometer and technique. Portland: Unpublished master’s thesis, Oregon Health Sciences University, 1992.
Holtzclaw, B.J. The febrile response in critical care: State of the science. Heart & Lung. 1992;21(5):482–501.
Kerlinger, F.N., Lee, H.B. Foundations of behavioral research, (4th ed.). Fort Worth, TX: Harcourt College Publishers, 2000.
Leininger, M.M. Qualitative research methods in nursing. Orlando: Grune & Stratton, 1985.
Livornese, L.L., Dias, S., Samuel, C., Romanowski, B., Taylor, S., May, P., et al. Hospital acquired infection with vancomycin-resistant Enterococcus faecium transmitted by electronic thermometers. Annals of Internal Medicine. 1992;117(2):112–116.
Malasanos, L.J. What is the preproposal? What are its component parts? Is it an effective instrument in assessing funding potential of research ideas? Nursing Research. 1976;25(3):223–224.
Marshall, C., Rossman, G.B. Designing qualitative research, (4th ed.). Thousand Oaks, CA: Sage, 2006.
Melnyk, B.M., Fineout-Overholt, E. Evidence-based practice in nursing & healthcare: A guide to best practice. Philadelphia: Lippincott Williams & Wilkins, 2005.
Miles, M.B., Huberman, A.M. Qualitative data analysis, 2nd ed. Thousand Oaks, CA: Sage, 1994.
Milewski, A., Ferguson, K.L., Terndrup, T.E. Comparison of pulmonary artery, rectal, and tympanic membrane temperatures in adult intensive care patients. Clinical Pediatrics. 1991;30(4 Suppl):13–16.
Miller, S. Multiple paradigms for nursing. In: Thorne S.E., Hayes V.E., eds. Nursing praxis: Knowledge and action. Thousand Oaks, CA: Sage; 1997:140–156.
Munhall, P.L. Nursing research: A qualitative perspective, (3rd ed.). Sudbury, MA: Jones & Bartlett; 2001.
Munro, B.H. Statistical methods for health care research, (3rd ed.). Philadelphia: Lippincott; 1997.
Nierman, D.M. Core temperature measurement in the intensive care unit. Critical Care Medicine. 1991;19(6):818–823.
Parse, R.R., Coyne, A.B., Smith, M.J. Nursing research: Qualitative methods. Bowie, MD: Brady, 1985.
Patton, M.Q. Qualitative research and evaluation methods, (3rd ed.). Thousand Oaks, CA: Sage; 2002.
Peat, J.K., Mellis, C., Williams, K., Xuan, W. Health science research: A handbook of quantitative methods. Thousand Oaks, CA: Sage, 2002.
Pinch, W.J. Synthesis: Implementing a complex process. Nurse Educator. 1995;20(1):34–40.
Pransky, S.M. The impact of technique and conditions of the tympanic membrane upon infrared tympanic thermometry. Clinical Pediatrics. 1991;30(4 Suppl):50–51.
Pyrczak, F., Bruce, R.R. Writing empirical research reports: A basic guide for students of the social and behavioral sciences, (5th ed.). Glendale, CA: Pyrczak; 2005.
Sandelowski, M., Davis, D.H., Harris, B.G. Artful design: Writing the proposal for research in the naturalist paradigm. Research in Nursing & Health. 1989;12(2):77–84.
Schmitz, T., Bair, N., Falk, M., Levine, C. A comparison of five methods of temperature measurement in febrile intensive care patients. American Journal of Critical Care. 1995;4(4):286–292.
Schooley, J.F. Thermometry. Boca Raton, FL: CRC Press, 1986.
Summers, S. Axillary, tympanic, and esophageal measurement: Descriptive comparisons in postanesthesia patients. Journal of Post Anesthesia Nursing. 1991;6(6):420–425.
Terndrup, T.E. An appraisal of temperature assessment by infrared emission detection tympanic thermometry. Annals of Emergency Medicine. 1992;21(12):1483–1492.
Tornquist, E.M. From proposal to publication: An informal guide to writing about nursing research, (2nd ed.). Menlo Park, CA: Addison-Wesley; 1999.
Turabian, K.L., Booth, W.C., Colomb, G.G., Williams, J.M. A manual for writers of research papers, theses, dissertations, seventh edition: Chicago style for students and researchers. Chicago: University of Chicago Press, 2007.
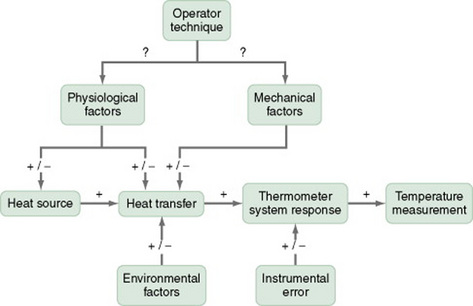
Ulbrich, S.L. The effect of operator technique on tympanic membrane thermometry. University of Texas at Arlington: Unpublished master’s thesis, 1995.
University of Chicago Press Staff. The Chicago manual of style, (15th ed.). Chicago: University of Chicago Press; 2003.
U.S. Department of Health and Human Services Information for covered entities and researchers on authorizations for research uses or disclosures of protected health information. HIPAA Privacy Rule: Information for Researchers, 2004, July 1. Retrieved October 25, 2007, from http://www.privacyruleandresearch.nih.gov/authorization.asp.
Vessey, J.A., Campos, R.G. Commentary: The role of nursing research committee. Nursing Research. 1992;41(4):247–249.
Wolff, S.M., The febrile patient. Wyngaarden, J.B, Smith, L.H., eds. Cecil textbook of medicine, (18th ed.), vol. 2. Philadelphia: Saunders, 1988;1524–1525.
Wright, V.L. A phenomenological exploration of spirituality among African American women recovering from substance abuse. Archives of Psychiatric Nursing. 2003;17(4):173–185.
Zehner, W.J., Terndrup, T.E. The impact of moderate temperature variance on the relationship between oral, rectal, and tympanic membrane temperatures. Clinical Pediatrics. 1991;30(4 Suppl):61–64.