Acyanotic defects: left-to-right shunts
A common feature of left-to-right shunt defects is increased pulmonary blood flow. The magnitude of the shunt and the resulting volume and pressure of the increased pulmonary blood flow is affected by pulmonary vascular resistance (PVR). PVR, in turn, is affected by a variety of factors including alveolar oxygenation, and by the maturation of the pulmonary vasculature, the response of the pulmonary vasculature to increased blood flow, and the anatomic features (e.g., size, location) of the defect.628
When a left-to-right shunt is present, the increased pulmonary blood flow is the basis for most resulting symptoms, particularly tachypnea. Tachypnea is frequently associated with mild respiratory distress. Greater respiratory effort further increases metabolic demand. Because a significant volume of blood recirculates through the pulmonary vascular bed, venous return to the left heart is increased and combined ventricular output is increased. Heart failure in these patients is referred to as “high-output” failure rather than “low-output” failure or shock. When heart failure develops, ventricular end-diastolic pressures increase, leading to increased systemic and pulmonary venous pressures. Hepatomegaly develops because the compliant hepatic veins dilate to accommodate the increase in circulating blood volume and pressure.30 Hypertrophy of the ventricular myocardium will develop if the increased volume load persists.628
The increase in work of breathing can substantially increase oxygen consumption and caloric requirements, and so may result in failure to thrive. The tachypneic infant will likely not feed well, compounding nutritional compromise. Heart failure also results in increased catecholamine secretion modulated by neural and hormonal mechanisms (“fight or flight” response). As a result, the infant/child with high-output heart failure is also tachycardiac and diaphoretic. To maintain physical growth, the patient with high-output failure must increase caloric intake.30 For further information, see Congestive Heart Failure and Altered Nutrition in the Common Clinical Conditions section of this chapter.
Patent Ductus Arteriosus (PDA)
Pearls
• The maturity of the pulmonary vascular bed, the anatomic features of the ductus arteriosus and the relative difference between pulmonary and systemic vascular resistances affect the degree of left-to-right shunt.
• The gestational age of the infant determines the response of the ductus arteriosus to oxygen and prostaglandins.
Etiology
A patent ductus arteriosus (PDA) is persistence of the fetal structure, the ductus arteriosus, after birth (Fig. 8-30). The ductus arteriosus is derived from the left sixth embryologic aortic arch and connects the main pulmonary trunk (at the origin of the left pulmonary artery) to the descending aorta just below the left subclavian artery. When a right aortic arch is present, the ductus typically connects the right pulmonary artery with the right aortic arch. The ductus varies in length and is as large as or larger in diameter than the descending aorta in the fetus.449,628
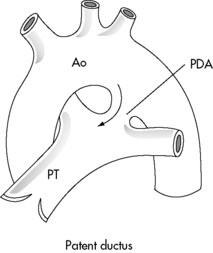
Fig. 8-30 Patent ductus arteriosus (PDA). The PDA extends from the bifurcation of the pulmonary trunk (PT) to the aorta (Ao), joining the aorta just beyond the origin of the left subclavian artery.
(From Perloff M: Clinical recognition of congenital heart disease, ed 5, Philadelphia, 2003, Saunders. Fig. 20-1.)
During fetal life the ductus arteriosus diverts blood away from the (high-resistance) pulmonary circulation to the descending aorta and toward the low-resistance placental circulation. The ductus carries 55% to 60% of the combined ventricular output.628
The intimal layer of the ductus arteriosus is thicker and more mucoid than the intimal layer of the pulmonary artery and the aorta. The smooth muscle in the medial layer of the ductus is arranged in both leftward and rightward patterns with an increased amount of hyaluronic acid; this facilitates constriction and closure of the ductus after birth. By comparison, the medial layer of the wall of the pulmonary artery and aorta consists of circumferential layers of muscle fibers.
The precise mechanisms of ductal closure are not fully understood. Functional closure occurs in two phases. In the first phase, immediately after birth, contraction and cellular migration of the medial smooth muscle produce shortening and thickening of this layer and protrusion of the media into the ductal lumen with thickening of the intimal layer. These thickened cushions or mounds result in functional closure of the ductus within about 12 hours of birth. Infolding of the endothelium; disruption and fragmentation of the elastic fibers; and cellular proliferation, hemorrhage, and necrosis in the subintimal layers result in replacement of the muscle fibers with fibrotic connective tissue to form the ligamentum arteriosum within about 2 to 3 weeks after birth.628
The increase in the partial pressure of oxygen (PaO2) that normally occurs after birth stimulates constriction of the ductus arteriosus. However in preterm infants the ductus is not constricted at even high levels of oxygen tension. Prostaglandins play an important role in ductal patency or constriction. Prostaglandins dilate the ductus, and a normal fall in endogenous prostaglandins after birth contributes to constriction of the ductus. Administered prostaglandin inhibitors (e.g., indomethacin) can cause ductal constriction.449,628 Other vasoactive substances (bradykinin, acetylcholine, and endogenous catecholamines) also contribute to ductal constriction and closure.
The PDA accounts for about 5% to 10% of all congenital heart defects in full-term infants.686 The incidence is much higher in premature infants (8 of 1000 premature infants have a PDA), particularly those with pulmonary disease and varies inversely with birthweight. Hemodynamically significant PDA is noted in almost 80% of infants with a birthweight of less than 1000 g, because ductal constrictive response to oxygen is related to gestational age.628
Ductus closure may be delayed if the neonate's arterial oxygen tension does not rise normally after birth. For example, the incidence of PDA is about 30% higher at high altitudes (i.e., 4500-5000 m [about 14,700-16,000 feet] above sea level), and neonates with cyanotic congenital heart disease characteristically demonstrate delayed closure of the ductus. A PDA also may be present as part of the rubella syndrome, and it may be seen occasionally in otherwise healthy, normal full-term infants.
A functionally closed ductus may reopen if the arterial oxygen tension falls, such as occurs with asphyxia, meconium aspiration, or pneumonia. It may also reopen in response to administered prostaglandin (e.g., for infants with cyanotic congenital heart disease or lesions with ductal-dependent systemic perfusion).628
Pathophysiology
With a PDA, several factors impact the magnitude of the left-to-right shunt: the diameter and length of the ductus, the pressure difference between the aorta and pulmonary artery, and the relative resistances in the systemic and pulmonary circulations.628 With the first breaths after birth, alveolar oxygenation improves and pulmonary vascular resistance begins to fall; as a result, the pulmonary to systemic flow through the ductus decreases dramatically.449 Once the umbilical cord is clamped, the systemic vascular resistance becomes slightly greater than the pulmonary vascular resistance, so any flow through the ductus initially becomes bidirectional and then ultimately reverses so it flows from the aorta into the pulmonary circulation.
As noted, the diameter and length of the ductus influences the magnitude of the left-to-right shunt. If the ductus is long and narrow the small diameter and the length add to the resistance to flow. By comparison, a short, wide ductus will offer little resistance to blood flow and allow a large shunt that will increase pulmonary blood flow. In addition, a short, wide ductus allows the blood to flow from the aorta into the pulmonary artery under relatively high pressure. The magnitude and pressure of the pulmonary blood flow through a PDA is likely to increase as pulmonary vascular resistance continues to fall over the first 2 months of life.628
There are several compensatory mechanisms, including the Frank-Starling law, adrenergic response, and myocardial hypertrophy that all help maintain cardiac output and systemic perfusion despite high-output heart failure. However, these mechanisms may not be as well developed or expressed in the neonate and particularly the premature neonate as they are in the older infant or child.628 Even at term the newborn's myocardial structure has higher water content and fewer contractile elements than the myocardium of adults.30 Neonatal myocardium—especially the myocardium of the preterm neonate—has a more modest response to stretch, so there is limited ability to increase stroke volume in response to volume administration and increased ventricular end-diastolic pressure. All result in decreased ability to compensate for increased ventricular end-diastolic volume and pressure. In addition, sympathetic nervous innervation of the left ventricle is not complete until term or just after birth.
Myocardial perfusion is also affected by a large PDA. Higher left ventricular diastolic pressure, faster heart rates with shorter diastolic filling time, and lower aortic pressures from ductal flow (runoff) all can compromise coronary artery blood flow. Physiologic anemia also affects myocardial function. Lower hemoglobin concentrations in newborns (even lower in premature newborns) and fetal hemoglobin affect oxygen delivery to the myocardial tissue.628 In addition to decreased myocardial perfusion, the decrease in systemic blood flow may compromise other organ systems, with resulting renal insufficiency, necrotizing enterocolitis, and intraventricular hemorrhage.30
Increased pulmonary blood flow through the PDA is associated with a variety of pulmonary complications, including increased pulmonary interstitial water, increased work of breathing, and decreased diaphragm blood flow. Reduced diaphragm blood flow probably substantially reduces the effectiveness of ventilation. Pulmonary edema may be observed in neonates with only a moderate ductal shunt because capillary permeability is higher in neonates than in older infants.628
The high pressure, high volume pulmonary blood flow that can result from a PDA (particularly a short, wide PDA) creates a risk of pulmonary vascular disease. The systemic and pulmonary arterial pressures can become equal. This high pulmonary vascular pressure can prevent the normal postnatal regression of medial smooth muscle in the pulmonary arteries. If the ductus remains open, true pulmonary vascular disease may develop with increased medial smooth muscle, intimal damage with cellular proliferation and hyalinization, and ultimately thrombosis and fibrosis of the small pulmonary arteries.628
With some congenital heart defects, flow through the ductus arteriosus is required for pulmonary or systemic blood flow; these defects are called ductal-dependent defects. Defects such as pulmonary atresia have ductal-dependent pulmonary blood flow. This means that blood flow from the aorta to the pulmonary artery provides the only source of pulmonary blood flow. When the ductus begins to constrict after birth, the newborn develops profound hypoxemia. Defects such as hypoplastic left heart syndrome have ductal-dependent systemic blood flow. With such defects, blood flow from the ductus into the aorta supports systemic blood flow. When the ductus begins to constrict after birth, systemic perfusion is compromised and shock develops.
Clinical Signs and Symptoms
Clinical features vary depending on the volume and pressure of the left-to-right shunt and the age of the patient. In the preterm infant with no lung disease a systolic murmur may be heard at 24 hours of age. The widespread use of surfactant has reduced severe respiratory disease, so the symptoms of a PDA appear earlier than when severe disease is present. As lung disease resolves and oxygenation improves, vasodilation causes pulmonary vascular resistance to fall, increasing the left-to-right shunt.30 As the magnitude of the left-to-right shunt increases, the systolic murmur becomes louder and extends into early diastole. A middiastolic flow rumble may be heard at the apex.
If the shunt is small, a murmur may be the only sign of a PDA.30,628,686 The classic continuous so-called “machinery murmur” heard in an older infant or child with a moderate PDA is not initially heard in the premature or term infant. With an increasing shunt the murmur is louder in all affected patients and may be accompanied by a suprasternal thrill. The pulmonic component of the second heart sound may be increased, especially in the infant with pulmonary disease. The precordium will be active, and the peripheral pulses bounding (because aortic flow runs off into the pulmonary artery).
The preterm, term infant, and child with a significant left-to-right shunt is dyspneic, tachypneic, and tachycardic. (For further information regarding auscultatory findings, see Table 8-25.)
Table 8-25 Clinical, Radiographic and Electrocardiographic Characteristics of Acyanotic Congenital Heart Defects
If the neonate is receiving mechanical ventilator support for lung disease, increased support requirements (including increased inspiratory pressure or increased supplementary oxygen requirements) may indicate the development of a shunt through the PDA. The murmur may only be heard between breaths or when the neonate is briefly removed from ventilator support (e.g., for suctioning).628
Evidence of left ventricular hypertrophy often is noted on clinical examination and by electrocardiographic criteria in the older infant and child (Table 8-26). The echocardiogram usually documents evidence of a large shunt, and an increase in the ratio of left atrial size/aortic size. The chest radiograph may be normal in asymptomatic patients with a small shunt, but cardiomegaly and increased pulmonary vascular markings are generally identified if a large shunt and congestive heart failure are present. Pulmonary interstitial edema also may be apparent. The main pulmonary artery may be prominent (for further information, see, also, Table 8-25).
If the clinical presentation is typical and if no additional abnormality is suspected, the diagnosis is made on the basis of clinical examination, chest radiograph, and echocardiogram. Cardiac catheterization is commonly used as an intervention to close the duct, using one of several devices. If the clinical presentation is atypical or if the presence of other cardiac anomalies is suspected, a cardiac catheterization may be performed. The catheterization will reveal an increase in oxygen saturation in the pulmonary artery. Right ventricular and pulmonary artery pressures will be elevated if pulmonary hypertension is present. Aortic contrast injection will demonstrate the shunt into the pulmonary artery.
If pulmonary hypertension develops, the patent ductus arteriosus murmur may decrease in intensity or be absent. The pulmonary component of the second heart sound will be increased.
If pulmonary vascular resistance is approximately equal to systemic vascular resistance, the child may develop bidirectional shunting through the PDA. This causes arterial oxygen desaturation; the child may demonstrate cyanosis, particularly of the lower extremities and when the infant cries.
If the neonate has ductal-dependent pulmonary blood flow (i.e., cyanotic congenital heart disease with pulmonary blood flow occurring only through the ductus arteriosus), profound hypoxemia and cyanosis will develop when the ductus begins to constrict. The hypoxemia is not relieved with oxygen administration (refer to Hypoxemia in the second section of this chapter and Cyanotic Defects later in this section of the chapter).
If the neonate has ductal dependent systemic blood flow, signs of poor systemic perfusion will develop when the ductus begins to constrict. Without treatment, these signs will progress to circulatory collapse (see section, Obstructive Lesions, Single Functioning Ventricle).
Management
Treatment of a PDA is aimed at closing the ductus. In the asymptomatic older child and adult with a PDA presenting only with a murmur, the need for PDA closure remains controversial.399,628 Before echocardiography was available, the natural history of the PDA included some spontaneous closure, but also included high infant mortality, and risk of bacterial endocarditis, heart failure, and pulmonary vascular disease with a mortality rate of 60% before 60 years of age. However, the widespread use of echocardiography enables early identification and evaluation of the ductal shunt.
With current therapy and closure at the time of diagnosis the complications of the ductal shunt are eliminated.449 The PDA may be closed pharmacologically, it may be closed surgically, and it can be closed using a device in older infants and young children. The timing and method of ductal closure in the premature infant remain open to debate. Before the ductus is closed in the preterm infant, medical management includes the maintenance of adequate hemoglobin concentration and hematocrit, normal serum electrolytes, adequate blood glucose, and nutritional support. To provide peripheral oxygenation and improve cardiac output, the hematocrit should be maintained greater than 45%. Management of congestive heart failure includes careful management of intravascular volume, diuretic administration, fluid restriction, and possible sodium restriction. Digitalis is not administered in the preterm infant because complications are high and therapeutic effect is minimal.628 For further information, see Common Clinical Conditions, Congestive Heart Failure.
Pharmacologic Closure
In the premature neonate, the use of a prostaglandin synthetase inhibitor, indomethacin, to promote ductal closure has until recently replaced surgical intervention as the first-line therapy for the management of symptomatic PDA in this patient population.628 Indomethacin therapy is most effective when it is administered before 10 days of age. The initial dose is 0.2 mg/kg either intravenously or by nasogastric tube. The intravenous dose is preferred because of the unpredictable absorption of the nasogastric dose. Subsequent doses depend on the age of the neonate at the time of the initial dose. For neonates less than 48 hours of age at initial dose, the next two doses are 0.1 mg/kg; if the neonate is 2 to 7 days of age at initial dose, the next two doses are 0.2 mg/kg; and if the neonate is greater than 7 days of age at initial dose, the next two doses are 0.25 mg/kg. The three doses are given 12 to 24 hours apart. If the urine output decreases, the number of doses may be reduced or the time between doses increased. Renal side effects are more severe if fluid intake is restricted before indomethacin therapy.628 If the symptoms of the left-to-right shunt remain, and renal and platelet function remain within normal limits (or adequate), additional doses of indomethacin may be administered (in some cases, consideration is given to repeating the course).
Indomethacin should not be administered to neonates with decreased renal function (serum creatinine greater than 1.6 mg/dL or blood urea nitrogen greater than 20 mg/dL), overt bleeding, shock, necrotizing enterocolitis, or echocardiographic evidence of myocardial ischemia.628 In recent series, indomethacin therapy failed in more than one fourth of the premature infants who received it, with either the ductus failing to constrict or reopening following initial closure.173,184,778 The risk factors associated with indomethacin failure included extreme prematurity (all failures occurred in neonates less than 27 weeks' gestation) and lack of exposure to antenatal betamethasone.
Indomethacin has been administered prophylactically during the first 24 hours of life to prevent symptomatic deterioration from a ductal shunt in extremely premature neonates (birthweight less than 1000 g). Although this practice often prevents the cardiorespiratory deterioration associated with the development of a large ductal shunt, it has not been demonstrated to reduce morbidity or mortality from the PDA. The current practice is to initiate therapy immediately on diagnosis of a PDA, usually before 72 h of age.628 Indomethacin is not effective beyond the neonatal period.30
Ibuprofen has been used as an alternative to indomethacin. Early studies indicated that ibuprofen had less negative effect on renal function and urine output. Ibuprofen also has less effect on cerebral vasculature and cerebral blood flow, but does not reduce risk of intraventricular hemorrhage. A recent trial of ibuprofen prophylaxis was ended because an increased incidence of pulmonary hypertension was detected in study patients.628
If medical therapy has failed to close the PDA, surgical intervention should be performed 1 to 2 days after failed medical therapy.
Device Closure
For infants and children, the goal of treatment is to interrupt the left-to-right shunt. If the ductus in a term infant is abnormal, the failure to constrict is related to a structural abnormality.628 Heart failure may be seen in infants less than 6 months of age but rarely develops later. Closure of the PDA in the catheterization laboratory may be delayed until the child is large enough to have a device placed in the ductus. But if congestive heart failure or failure to thrive is present, surgical intervention is indicated.449
At the present time the majority of PDAs in children and adults are closed during cardiac catheterization. The exceptions are the premature infant and when the infant's PDA is very large and short.545 The most common devices used are Gianturco embolization coils (more than one may be used), each consisting of a stainless steel wire with Dacron fibers twisted in the wire, and Amplatzer Vascular Plug, a plug constructed of nitinol wire mesh. The Dacron in a Gianturco embolization coil adds thrombogenicity to the wire, but Dacron is not included in the Amplatzer Vascular plug.628
The coil is placed through a catheter that is advanced retrograde through the aorta to the site of the junction of the ductus and the aorta. The catheter delivering the coil crosses the ductus and enters the pulmonary artery. The coil is advanced beyond the tip of the catheter, and one loop is placed in the pulmonary artery. The catheter is then pulled back through the narrowest part of the ductus to the ampulla at the attachment of the ductus to the aorta. The rest of the coil is then advanced out of the catheter.
If a left-to-right shunt is detected after the first coil is placed, one or more additional coils may be deployed. The greatest risk of this procedure is coil migration (4%) into the pulmonary arteries. More than half of these embolized coils were successfully retrieved; the rest remained in place with no adverse effects.545,628 The greatest risk of coil migration occurs when a patent ductus arteriosus is greater than 4 mm in diameter.
The Amplatzer Vascular Plug (AVP) is used for moderate to large ducts. The AVP is delivered in an antegrade fashion through a central venous catheter that is threaded through the right ventricle and then into the pulmonary artery and through the ductus to the junction of the aorta. The device is positioned in the aorta and pulled back into the aortic ampulla. The rest of the body of the plug is deployed in the body of the ductus with a small part of the plug at the pulmonary artery end of the ductus. The plug is not recommended for infants less than or equal to 5 kg.
With current use of the coil and the plug, the PDA is closed successfully in greater than 97% of the procedures with the overall risk of inadvertent embolizations at less than 1%.545,580,628,910 After therapeutic catheter intervention, antiplatelet prophylaxis is usually prescribed for 3 to 6 months. Antibiotic prophylaxis for subacute bacterial endocarditis is recommended for 12 months or longer if a persistent shunt remains.399
If the neonate has a ductal-dependent cyanotic congenital heart defect, prostaglandin E1 will be administered to maintain ductal patency. For further information the reader is referred to Hypoxemia in the second section of this chapter and to Cyanotic Defects later in this section of the chapter.
Surgical Intervention
Early surgical ligation of a PDA in premature neonates was shown to reduce the duration of mechanical ventilatory support and the length of the hospital stay.628 Therefore prompt surgical PDA ligation is recommended if pharmacologic ductal closure fails.
Closed-heart surgery may be performed thorascopically or through a left, lateral thoracotomy. Although there is risk of recannulation, ligation (tying of the ductus) is favored over division (cutting and oversewing) for the smallest premature neonates (less than 1000 g) because it minimizes the dissection required and shortens the time required for lung retraction and anesthesia. Most surgeons clip the ductus to minimize the need for dissection and the risk of vocal cord dysfunction associated with dissection.628,757 For all others the ductus is divided and oversewn. If the ductus is calcified, hypertensive, and fragile (generally this occurs only in elderly patients) the procedure is performed with cardiopulmonary bypass on standby.
Morbidity and mortality for surgical elimination of the ductus are typically extremely low (less than 1% mortality). The neonatal myocardium is very sensitive to afterload, and the change in afterload with ligation of the ductus arteriosus will affect the myocardial performance index and cardiac output of the premature neonate. To maintain cardiac output, inotropic support, usually with dopamine, is required for approximately 24 hour postoperatively.656
The highest surgical risk is observed in children and adults with PDA who have developed pulmonary hypertension and Eisenmenger syndrome (a left-to-right shunt becomes a right-to-left shunt as the result of the development of pulmonary hypertension). These patients require careful evaluation and diagnostic studies preoperatively.275 PDAs have been successfully closed using devices in adult patients with pulmonary hypertension.
Postoperative complications of PDA surgical repair (see Table 8-27)851-853 include those of a thoracotomy (bleeding, atelectasis, hemothorax, and pneumothorax). In addition, phrenic or recurrent laryngeal nerve injury may occur.851-853 Vocal cord paralysis may be associated with recurrent laryngeal nerve injury; this complication is relatively uncommon and is observed almost exclusively in very small neonates.
Advanced concepts regarding the care of the child with a PDA are presented in Box 8-22.
Box 8-22 Advanced Concepts: Patent Ductus Arteriosus
• Heart failure from a left-to-right shunt is “high output” failure.
• Patent ductus arteriosus appears earlier in the preterm infant when surfactant is used to prevent pulmonary disease.
• The preterm infant has less compensatory mechanisms to handle the increased volume load from the left-to-right shunt.
• The symptoms of a PDA in a neonate during mechanical ventilation for lung disease may include requirement for increased pressure and/or oxygen support.
• Abrupt closure of the PDA with surgical ligation in the preterm infant will decrease myocardial performance and require inotropic support.
Aortopulmonary Window (Aortopulmonary Septal Defect)
Etiology
An aortopulmonary window is a defect between the ascending aorta and the pulmonary artery. With this defect, there must be two distinct semilunar valves.445
Septation of the fetal truncus arteriosus is influenced by neural crest cells. However, embryologic studies demonstrate that the neural crest cells do not affect the development of an aortopulmonary window. Studies also demonstrate that the aortopulmonary window is not associated with DiGeorge syndrome or any other chromosomal deletions in 22Q11.30
Aortopulmonary window accounts for 0.2% to 0.6% of all congenital heart defects. Nearly half of these patients have associated anomalies; the most common include interrupted aortic arch, anomalous origin of the coronary arteries, and anomalous origin of right pulmonary artery. Although these associated anomalies are found in the same area in the heart, they appear to be embryologically unrelated.628
Pathophysiology
The physiologic effects of an aortopulmonary window are similar to the left-to-right shunt of a large patent ductus arteriosus (PDA) or a ventricular septal defect. Once the pulmonary vascular resistance falls in the first days to weeks of life, blood is shunted from the aorta across the defect into the pulmonary circulation. These defects are discrete, most commonly large and unrestrictive, and are positioned between the semilunar valves and the bifurcation of the pulmonary artery as illustrated in Fig. 8-31.
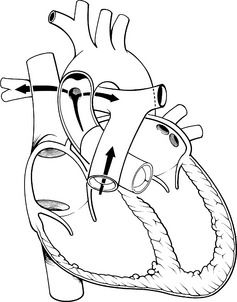
Fig. 8-31 Aortopulmonary window. The defect between the aorta and pulmonary artery produces a left-to-right shunt from the aorta into the pulmonary circulation. Shunting of blood occurs during both systole and diastole. If the defect is large, the volume of the shunt will be large and under high pressure.
These defects typically produce early congestive heart failure. Without repair, pulmonary vascular disease often develops, perhaps as early as 1 year of age.445 This defect does not close spontaneously. Prompt surgical repair is indicated when the diagnosis is made.30,686
Clinical Signs and Symptoms
Clinical signs and symptoms of an aortopulmonary window vary with the size of the defect and the associated anomalies. A very small defect may not produce signs or symptoms other than a systolic ejection murmur.
The defect characteristically produces a loud, systolic murmur at the upper left sternal boarder or a machinery murmur, similar to that produced by a patent ductus arteriosus. Additionally, there may be a mid-diastolic murmur at the apex caused by the flow of the large volume of pulmonary venous return across the mitral valve. The second sound may have a louder pulmonary component from pulmonary hypertension. An ejection click may also be heard in the pulmonary area.445,628 A right ventricular impulse may be felt along the left sternal boarder, and a thrill may be present at the upper left sternal border. Pulses may be bounding, or if the defect is associated with an interrupted aortic arch, femoral pulses may be markedly decreased when the patent ductus arteriosus closes.
Signs of congestive heart failure often are present, and left ventricular hypertrophy may be apparent on the clinical examination, the chest radiograph, and the ECG. Signs of right ventricular hypertrophy will be present if pulmonary vascular disease develops.
Echocardiography can accurately diagnose an aortopulmonary window and associated anomalies. The defect can also be identified by a fetal echocardiogram.628 Cardiomegaly and increased pulmonary vascular markings are typically apparent on chest radiograph, unless the defect is very small.
Cardiac catheterization is usually not necessary but will document elevated pulmonary pressures, usually equal to systemic pressures. Contrast injection into the aorta will reveal the shunt into the pulmonary artery. Small defects have been successfully closed with a device during catheterization.445,628
Management
Closure of the defect is indicated in virtually all cases. Surgical closure is usually planned as soon as the diagnosis is made. If signs and symptoms of heart failure are severe, preoperative treatment of congestive heart failure (e.g., diuretics) is helpful to reduce pulmonary congestion. Newborns with an aortopulmonary septal defect and interrupted aortic arch may require prostaglandin E1 to maintain ductal patency to support systemic blood flow preoperatively.
Recommended surgical treatment of an aortopulmonary window requires a median sternotomy with hypothermic cardiopulmonary bypass. Best outcomes are achieved with patch closure through an aortic incision under direct visualization.275 Postoperative complications include bleeding, low cardiac output, pulmonary hypertension, and congestive heart failure.156
Atrial Septal Defect (ASD)
Pearls
• Atrial septal defects are present in 1 in 1000 live births.
• Most atrial septal defect in infants close spontaneously.
• Secundum defects may be closed by a septal occluder device.
• Some atrial septal defects require surgical closure. These include sinus venosus atrial septal defect, coronary sinus atrial septal defects, and primum atrial septal defects.
Etiology
An atrial septal defect is any opening in the atrial septum other than a competent foramen ovale.708 Atrial septal defects result from improper septal formation early in fetal cardiac development. Most occur sporadically. However, some occur with genetic syndromes, most commonly Holt-Oram syndrome, Down syndrome, and Noonan syndrome. The incidence of atrial septal defects is 1 per 1000 live births,458 but ASDs are found in about 30% to 50% of children with other forms of congenital heart disease.686
ASDs are classified into three major types, according to their location relative to the fossa ovalis (Fig. 8-32 illustrates the three types of ASDs: the ostium secundum, ostium primum, and sinus venosus).
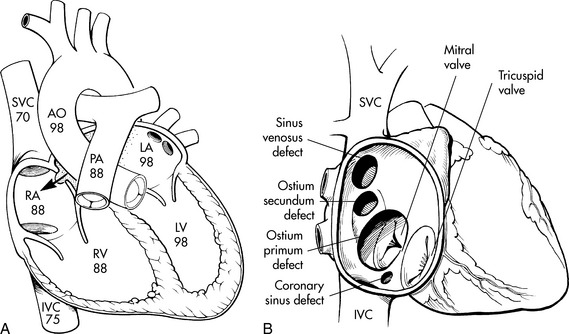
Fig. 8-32 Atrial septal defect (ASD). A, Typical oxyhemoglobin saturations observed in the cardiac chambers and great vessels when an ASD is present. B, Common types of ASD, and their relationship to the superior vena cava (SVC), the inferior vena cava (IVC) and the mitral and tricuspid valves.
(From Kambam J: Patent ductus arteriosus. In Kambam J, editors: Cardiac anesthesia for infants and children. St Louis, 1994, Mosby.)
The ostium secundum is the most common type of ASD, accounting for 50% to 70% of all ASDs. It is located in the region of the fossa ovalis (the foramen ovale). Historically, a patent foramen ovale has not been included in this category unless atrial dilation occurs from increased volume, and the flap closing the foramen ovale becomes incompetent, allowing a left-to-right or a right-to left shunt.708 Although functional closure of the fetal foramen ovale may occur at birth, anatomic closure of the foramen ovale does not occur in 25% to 30% of the population. This failure is thought to be responsible for paradoxic emboli (from a right-to-left shunt across the foramen ovale) associated with cryptogenic stroke and decompression illness.169 Migraine headaches have also been linked to patent foramen ovale.
The ostium primum defect accounts for about 30% of ASDs. This defect is located anterior to the fossa ovalis where the atrial septum originates from the endocardial cushion. It usually is associated with a defect in ventricular septal tissue as well as with anomalies of one or both atrioventricular valves. (See section, Atrioventricular Septal Defects/Endocardial Cushion Defects.)
The sinus venosus ASD is located posterior and superior to the fossa ovalis, typically near the junction of the superior vena cava and the right atrium. This defect can also occur at the junction of the inferior vena cava and right atrium. It often is associated with partial anomalous pulmonary venous return (PAPVR). With PAPVR, some of the pulmonary veins empty directly into the right atrium or superior vena cava instead of into the left atrium.
Anomalies of the atrioventricular valves may be associated with ASDs. As noted, ostium primum ASDs are often associated with anomalies of one or both atrioventricular valves. Mitral valve prolapse is present in about 20% of patients with either a secundum or sinus venosus ASD.458,686
Less common forms of ASDs include Chiari network, characterized by multiple fenestrations in the atrial septum that allow a left-to-right shunt (from left-to-right atrium), and a coronary sinus septal defect at the expected site of the coronary sinus as an “unroofed” coronary sinus. When the coronary sinus is unroofed, coronary sinus (i.e., venous) blood flows into the left atrium (a form of right-to-left shunt). Thus, this defect results in bidirectional shunting of blood at the atrial level, with resulting hypoxemia and cyanosis. This unusual form of ASD typically is associated with persistent left superior vena cava.458
A common atrium is the most severe form of ASD. The septum primum, septum secundum, and the atrioventricular canal septum are all absent. A common atrium is usually associated with heterotaxy syndrome458 (for further information, see section, Essential Anatomy and Physiology, Fetal Development of the Heart and Great Vessels).
A patent foramen ovale may serve as a necessary channel for shunting of blood between the right and left atria. This is most commonly observed in patients with obstruction to right atrial flow (tricuspid atresia), obstruction to right ventricular flow (pulmonary atresia), transposition of the great arteries, or obstruction of left ventricular flow (hypoplastic left heart syndrome).362
Pathophysiology
The resistance (and compliance) of the ventricles and the ventricular outflow tracts determine the magnitude and direction of shunt across an ASD.458,708 At birth, the right ventricle is thicker and less compliant than the left ventricle. Pulmonary vascular resistance begins to fall with the first few breaths following birth, but the right ventricle takes weeks to remodel and for resistance to decrease.30 Therefore, a significant shunt does not typically develop for the first weeks of life.
This defect usually is tolerated well and often produces no symptoms during childhood. An occasional neonate with ASD will develop symptoms of heart failure. Such newborns typically have extracardiac anomalies and developmental delay but do not have hemodynamic changes that are different from newborns without heart failure.708 Heart failure may also develop in neonates with ASD and additional cardiac defects, such as ventricular septal defect, patent ductus arteriosus, coarctation of the aorta, or those with myocardial dysfunction, an anatomically small left ventricle and systemic hypertension.458
If an ASD remains unrepaired to adulthood, significant right atrial and right ventricular dilation and hypertrophy can develop, producing atrial arrhythmias (e.g., atrial fibrillation), congestive heart failure, and possible paradoxic emboli. The risk of pulmonary vascular disease is low in the child with an uncomplicated ASD. However, pulmonary vascular disease is reported in 5% to 10% of all adults with unrepaired ASD, although it usually does not occur before the patient is 20 to 30 years of age.458 This complication should decrease in frequency because most ASDs greater than 7 to 8 mm in children more than 6 years of age are now closed when they are discovered. The risk of bacterial endocarditis in association with an uncomplicated ASD is virtually zero.708
When the child has cyanotic congenital heart disease, an ASD may provide an important shunt that allows mixing of oxygenated and venous blood within the atria. In this case the size of the ASD can influence the degree of mixing as well as the arterial oxygen saturation.
Clinical Signs and Symptoms
Most children with secundum or sinus venosus ASD are asymptomatic. (See Atrioventricular Septal Defects for information about ostium primum atrial septal defects.) The characteristic heart murmur associated with an ASD is a soft systolic ejection murmur heard over the left second intercostal space and an early to mid-diastolic murmur at the lower left sternal border. This systolic murmur results from increased blood flow across an otherwise normal pulmonary valve; this causes a “relative stenosis” because when a larger than normal volume passes through a normal (not larger than normal) valve orifice, turbulence results. The diastolic murmur results from increased flow from the right atrium across the tricuspid valve into the right ventricle.
The first heart sound is louder at the left lower sternal boarder, and the second heart sound is heard best at the left upper sternal border. The pathognomonic auscultatory finding of an ASD is the presence of fixed splitting of the second heart sound. This split does not vary with respirations and it results from prolonged right ventricular ejection, caused by the increased blood flow into the right atrium, right ventricle and the pulmonary circulation (see Table 8-25). If pulmonary hypertension develops, the split narrows and the pulmonary component of the second heart sound increases.
Right ventricular hypertrophy may produce a sternal lift on clinical examination and right ventricular hypertrophy may be apparent on the ECG (see Table 8-26). A two-dimensional echocardiogram will demonstrate the position as well as the size of the atrial septal defect. It will also demonstrate the effects of the left-to-right shunt, including right ventricular and right atrial enlargement as well as a dilated pulmonary artery. Color-flow Doppler studies will enhance the evaluation of the shunt.
In adolescents and children who are overweight, the transthoracic echo may not provide clear visualization, and a transesophageal echocardiogram is needed. Echocardiographic studies may be augmented by “bubble” studies. During this study agitated normal saline is injected into a peripheral vein, and contrast bubbles are visualized in the left atrium.458,686,708
The chest radiograph may be normal, or it may demonstrate cardiomegaly and increased pulmonary vascular markings. The main pulmonary artery is often prominent on the chest radiograph (see Table 8-25).
Cardiac catheterization is necessary only when device closure is planned, if other defects are present, or if pulmonary vascular disease has developed. During cardiac catheterization of the patient with pulmonary vascular disease, pulmonary pressures are measured directly and effects of pulmonary vasodilators, including medications, nitric oxide, and oxygen on pulmonary vascular resistance are evaluated. These studies help to determine the risks associated with surgical or device closure of the ASD.708,892
Cyanosis may be present if the coronary sinus is unroofed. Patients with this form of ASD are at risk for development of systemic consequences of chronic hypoxemia, including cerebral thromboembolic complications and brain abscess formation (see Hypoxemia).
Management
Management of an atrial septal defect is determined by the type of defect and presence of symptoms.
Nonsurgical Treatment
Most children with secundum ASDs are asymptomatic. Because of the frequency of echocardiograms, ASDs have been identified in as many as 24% of newborns. The average age at diagnosis is 5 months. Most infants with ASDs (estimated as greater than 90%) have spontaneous closure of this defect by 1 year. Defects 8 mm or larger in diameter are the defects that commonly cause a significant left-to-right shunt and most likely will not close spontaneously. These defects should be surgically closed.351,458,708
Device closure has also been successfully performed in infants, even premature newborns. Placement involves hybrid techniques with hepatic and periatrial access.235,537
Spontaneous ASD closure can occur in infants with congestive heart failure.351,708 Children diagnosed with secundum ASD after infancy usually have large defects. These children are followed until they are 4 to 6 years old before elective closure of the secundum ASD. Spontaneous closure of the smaller defects occurs in 34% of children, with 77% showing regression in the size of the defect. Unfortunately, some ASDs can become larger; typically this occurs in children who were diagnosed at an older age, and they require device or surgical closure.
The primary reason to close an atrial septal defect is to prevent pulmonary vascular disease458 and adult complications such as atrial fibrillation and congestive heart failure. The presence and severity of functional limitations increases with age, with 5% to 10% of patients with ASDs developing pulmonary vascular disease if the defect is not closed. Although pulmonary vascular disease may develop in childhood, it usually does not develop before 20 years of age.708
Catheter Intervention
Transcatheter closure of ASDs has been performed with increasing success during the past several years. Criteria for device placement include evidence of right ventricular overload and appropriate septal anatomy. The secundum ASD must have a surrounding rim that is sufficient to anchor the device, and the stretch diameter must not exceed that specific to the device.
Several devices have been evaluated and the Amplatzer Septal Occluder (ASO) was the first to receive FDA approval in 2001.708 (See Evolve Fig. 8-3 for illustrations of Amplatzer Occluders in the Chapter 8 Supplement on the Evolve Website.) The ASO is a double-disc device of nitinol mesh. The two discs are linked to each other with a central waist. Dacron fabric is incorporated into each disc and the waist. The ASO device is the most effective for closing large defects; stretch diameter must not be larger than 36 to 38 mm.892 The Helex septal occluder is currently being evaluated to close small or moderate defects with a stretch diameter less than 20 mm (see illustration of Helex septal occluder in Evolve Fig. 8-4 in the Chapter 8 Supplement on the Evolve Website).422,515,910
Closure devices are placed in the catheterization laboratory with either transthoracic or transesophageal echocardiographic guidance. Complete closure rates have been satisfactory, with the ASO achieving 98% to 100% closure with few complications. Complications include device malposition, device embolization, thrombus formation, infection, and erosion of adjacent structures. Transient atrioventricular heart block has also been reported.27,708 These devices become endothelialized in a few weeks. Aspirin is prescribed for 3 to 6 months after placement to minimize the risk of thrombus formation. Both the Amplatzer Septal Occluder and the Helex septal occluder can safely be used in the magnetic resonance imaging (MRI) scanner. The artifact created by the occluder is small enough to allow anatomic and functional MRI even in the immediate vicinity of the device.545
Although device closure has been attempted in sinus venosus ASDs, surgical closure is preferred. Because these defects are almost always associated with anomalous pulmonary venous return, the surgical procedure involves patch placement to close the defect and direct the pulmonary venous return to the left atrium.
Coronary sinus atrial septal defects also require surgical closure. Surgical repair depends on the anatomy.275,458
Surgical Intervention
Surgical closure of the ASD most frequently is completed through a median sternotomy incision on cardiopulmonary bypass. To reduce visible scarring, other surgical approaches can be used. The most frequent alternative approach is a “mini-sternotomy” through an incision on the lower half of the sternum.458 Other approaches include a limited right lateral thoracotomy with the incision starting below the scapula and ending at the mid axial line.11,800 Thorascopic approaches have also been used.
A secundum ASD is closed directly with sutures (primary closure) or with autologous pericardium or prosthetic patch material (patch closure). Mortality is virtually zero, and hospital stay is normally less than 3 days. Complications include bleeding, arrhythmias, and post-pericardiotomy syndrome.458
Surgical repair of a sinus venosus defect is more complicated, especially if anomalous pulmonary veins are present. An intracardiac patch may be required as a baffle or the superior vena cava may be translocated to direct pulmonary venous return to the left atrium. If the defect is an inferior sinus venous defect, the procedure becomes more complex to direct hepatic venous return or anomalous pulmonary venous return to the appropriate atrium.
Repair of a coronary sinus ASD is tailored to the anatomy. If a left superior vena cava (SVC) is present, it may be ligated if a bridging vein to the right superior vena cava exists, or flow from the left SVC will be baffled (directed under a patch) to the right atrium. Postoperative mortality is less than 4%.
Complications following repair of a sinus venosus or coronary sinus ASD include arrhythmias, such as heart block, and postoperative obstruction to venous return. If cyanosis is noted in the postoperative period, reevaluation of pulmonary or systemic drainage is required.
Long term followup from surgical closure of ASDs in children with low pulmonary artery pressures shows excellent results (Table 8-28),851-853 with a 95% survival rate compared with a matched population survival rate of 98%.708 Surgical closure usually alleviates symptoms, and residual defects are rare. Occasional patients demonstrate persistent arrhythmias or develop late arrhythmias. Cardiomegaly as evident on a preoperative chest radiograph or ECG may be evident for months or years postoperatively.458
Advanced concepts regarding atrial septal defects are listed in Box 8-23.
Box 8-23 Advanced Concepts: Atrial Septal Defect
• The direction of blood flow across an atrial septal defect is determined by the resistance or compliance of the ventricles.
• A significant shunt from an atrial septal defect usually does not develop in the newborn because it takes the right ventricle weeks to months to remodel and resistance to decrease.
• Because the defect is well tolerated and a significant number of secundum atrial septal defects close spontaneously, closure can wait until the child is 4 to 6 years of age.
• The primary reason to close an atrial septal defect is to prevent pulmonary vascular disease and other complications (e.g., atrial arrhythmias). Such complications usually do not develop until the second decade of life or later.
Ventricular Septal Defect (VSD)
Etiology
A ventricular septal defect is the most common congenital heart defect, accounting for 15% to 20% of all congenital heart defects.686 It occurs as frequently as 2.5 in 1000 live births, although only about one in five or fewer of the defects require closure, either during catheterization or surgery.457
A VSD results when the interventricular septum fails to close after the first 7 weeks of fetal life. The reasons for this failure are not clear and are felt to be multifactorial. Although these defects are the most common present in many genetic and chromosomal syndromes, including trisomy 13, 18, and 21, Holt-Orem and Cornelia de Lange syndromes, the vast majority of VSDs are not associated with any defects.457,597,686 They are, however, more common in premature and low-birthweight neonates.
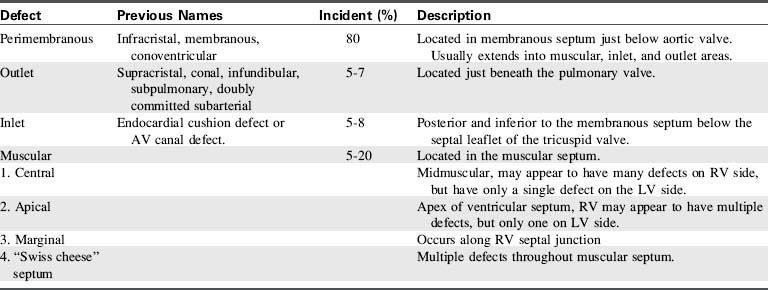
VSDs can occur as a single defect, as multiple defects, in association with another defect, or as a component of more complex congenital heart disease. Physiologic consequences range from trivial to severe. VSDs are classified by location in the ventricular septum.597
Although many terms have been used to classify VSD location, the current terms for the four major locations are: perimembranous, outlet, inlet, and muscular. A summary of the four classifications is presented in Table 8-29, and locations are illustrated in Fig. 8-33.
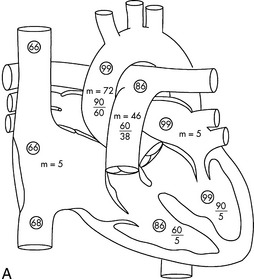
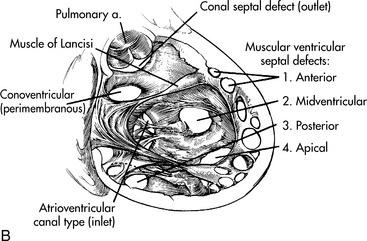
Fig. 8-33 Ventricular septal defect (VSD). A, Typical intracardiac pressures (systolic, diastolic and mean [m]) and oxyhemoglobin saturations (in circles). Note step-up in oxyhemoglobin saturation from the right atrium (66%) to the right ventricle (86%) indicating left-to-right shunt at the ventricular level. Right ventricular pressure (60/5) is elevated compared with the normal right ventricular pressure, but is less than the left ventricular pressure (90/5), indicating that the VSD in this illustration is restrictive. The pulmonary artery pressure is elevated as well. (From Nichols DG. Critical heart disease in infants and children, ed 2, St Louis, 2006, Mosby (Fig. 24-8). B, Classification of ventricular septal defects (VSDs) and potential locations: atrioventricular canal (inlet) type; muscular VSDs (anterior [1], midventricular [2], posterior [3], and apical [4]); conoventricular septal defect, which includes perimembranous and malalignment conoventricular septal defects; and conal septal (outlet) defects. The orifice of the pulmonary artery (pulmonary a.) is indicated.
(From Sellke F: Sabiston and spencer's surgery of the chest, ed 8, Philadelphia, 2009, Saunders; Fig. 117-4.)
Perimembranous VSDs, also called membranous conoventricular and infracristal defects, are the most common type of VSD, accounting for 70% to 80% of all VSDs.597,686 The membranous septum is a small area just below the aortic valve. Defects in this area include the muscular tissue adjacent to the membranous septum. The membranous septum overlaps a small segment of the right atrium, and defects occasionally occur in this area.
Outlet (conal) VSDs account for 5% to 7% of all VSDs with several-fold higher incidence in Far Eastern countries. Outlet VSDs have also been called supracristal, conal, subpulmonary, or subarterial VSDs.457,686 The outlet defect is located in the conal septum just below the pulmonary valve. Because of its location, a cusp of the aortic valve can prolapse into this defect, causing aortic valve insufficiency.
The third type of ventricular septal defect, an inlet defect, occurs in 5% to 8% of all VSDs. It is located posterior and inferior to the membranous septum below the septal leaflet of the tricuspid valve. This form of a VSD may also be referred to as an endocardial cushion defect or AV canal defect, but unlike the atrioventricular septal defects, it usually does not involve either of the atrioventricular valves.597
The fourth type of VSD is the muscular defect, located in the muscular septum. Muscular VSDs constitute 5% to 20% of all VSDs. The defect may be single when viewed from the left side but appears to be multiple when viewed from the right ventricle because the shunted blood flows around trabeculations (criss-crossing muscular and fibrous tissue strands) that form the right ventricular walls. Muscular defects are found in the apex, in the central or mid-muscular region, or in the anterior or marginal region of the septum. Multiple defects involving all components of the ventricular septum are usually referred to as a “Swiss cheese” septum.
Many (30% to 40%) membranous and muscular VSDs may close spontaneously. Spontaneous closure occurs more frequently in small defects and during the first 6 months of life. Defects do not enlarge with age, so they may become relatively smaller as the infant grows. Inlet and outlet defects do not close spontaneously.
Pathophysiology
The primary variable that determines the hemodynamic consequences of a VSD is the size of the defect. Small to medium defects restrict the volume and the pressure of the left-to-right shunt. However, large defects (approaching the size of the aortic orifice) do not restrict flow, so the magnitude of the shunt is determined by the difference between pulmonary and systemic vascular resistances.597
Immediately after birth, pulmonary vascular resistance normally falls rapidly as the small, muscular pulmonary arteries change from vessels with small lumens and thick medial muscle walls to vessels with larger lumens and thinner walls.30 When pulmonary vascular resistance falls normally, right ventricular pressure decreases and approaches adult levels by 7 to 10 days of life.597
The normal postnatal decline in pulmonary vascular resistance may be delayed if a large VSD is present, and this will, in turn, delay development of symptoms of the shunt. The increase in pulmonary blood flow resulting from a VSD will increase left atrial and pulmonary venous pressure and slow the normal decline in pulmonary vascular resistance, so it may take weeks to fall. When pulmonary vascular resistance eventually falls, the left-to-right shunt through the VSD increases. Increased pulmonary blood flow causes increased pulmonary venous return to the left side of the heart with resultant left atrial and ventricle enlargement and development of congestive heart failure, usually at 2 to 8 weeks of age.597 If signs of congestive heart failure develop at this age, blood flow to the pulmonary arteries is typically at least twice the systemic blood flow or more.457
When the VSD is large, the volume of the shunt into the pulmonary circulation is large. Left ventricular hypertrophy develops rapidly in response to the volume load resulting from increased pulmonary venous return. In addition, when the VSD is large, the pressures in the right ventricle and the pulmonary circulation increase, and right ventricular hypertrophy and failure and pulmonary hypertension can develop.
Increased pulmonary blood flow can cause changes in the pulmonary vessels; these changes are more likely to develop and can develop more rapidly when the increased pulmonary blood flow is under high pressure. Medial hypertrophy and intimal proliferation can eventually cause pulmonary vascular obstructive disease.597 Such pulmonary vascular obstructive disease may be permanent as early as 12 months of age.
Once pulmonary vascular resistance approaches systemic vascular resistance, the shunt flow through the VSD decreases. If the shunt flow reverses as the result of development of pulmonary hypertension, this is called Eisenmenger syndrome. If Eisenmenger syndrome develops, the defect is irreparable, because acute right heart failure is likely to develop if the defect is closed.457 For this reason, children with moderate to large VSDs are followed closely with surgical intervention planned before the development of pulmonary hypertension.686 For further information, see Common Clinical Conditions, Pulmonary Hypertension.
Several factors may cause the newborn with a VSD to develop signs of congestive heart failure within the first few days of life rather than at several weeks of age. In a rare form of membranous defect, the left ventricle ejects directly to the right atrium. This defect produces a significant shunt and substantial increase in pulmonary blood flow. Other factors that promote early signs of congestive failure include additional cardiac anomalies (particularly any defect that increases resistance to left ventricular or systemic blood flow), respiratory infection, anemia, other congenital anomalies, and prematurity. Left heart abnormalities, including aortic stenosis, coarctation of the aorta, and interrupted aortic arch, can contribute to left ventricular failure and pulmonary edema, and they also magnify the difference between pulmonary and systemic resistance and increase the magnitude of the left-to-right shunt. Mitral valve stenosis or any pulmonary venous obstruction will substantially increase pulmonary venous congestion and pulmonary hypertension.597
Other factors may affect the pulmonary or systemic blood flow in patients with a VSD. In a membranous defect the great arteries may be malaligned, either to the right or left and the malalignment may obstruct right or left ventricular outflow. Such malalignment may affect the direction, volume, and pressure of the shunt flow. Tetralogy of Fallot and double outlet right ventricle with a subaortic VSD and pulmonary stenosis are examples of malaligned VSD associated with obstruction to pulmonary blood flow and a right-to-left shunt through the VSD (see section, Tetralogy of Fallot, Double Outlet Right Ventricle).457
In an outlet defect the right coronary cusp of the aortic valve may prolapse into the defect, because the defect in the ventricular septum results in inadequate support for the aortic root. In addition, a venturi effect on the valve leaflets can contribute to development of aortic insuffiency.361,597 Once aortic insufficiency develops it is likely to increase with age.597 The right or noncoronary cusp of the aortic valve may also prolapse into a membranous defect; this may reduce flow through the defect but obstruct the right ventricular outflow tract.686
The risk of bacterial endocarditis in infants and young children with VSD is low, rarely occurring before the age of 2 years.597
Clinical Signs and Symptoms
Moderate and large VSDs typically produce signs and symptoms of heart failure as early as 2 weeks of age. The infant will develop signs of pulmonary venous congestion, with tachypnea and respiratory rates as high as 80 to 100 breaths per minute with retractions. Systemic venous congestion produces hepatomegaly and systemic edema.457 These infants have difficulty feeding; as symptoms of congestive heart failure worsen, the infant takes a progressively longer time to take less formula, and fails to gain weight. Adrenergic stimulation produces tachycardia, peripheral vasoconstriction, decreased urine output, and diaphoresis.
Infants with a moderate to large VSD and congestive heart failure frequently present with respiratory infection. The respiratory infection makes it difficult to determine if the respiratory distress is caused by the left-to-right shunt or the infection.597
The murmur of a moderate VSD is harsh and holosystolic, and is usually associated with a thrill. When pulmonary blood flow is twice systemic blood flow, a third heart sound may be heard, with a mid-diastolic rumble. This sound is best heard at the apex and results from increased flow of pulmonary venous return across the mitral valve. The second heart sound is split with the pulmonary component at normal intensity.457,597,686
With large shunts, increased precordial activity is apparent over both the right ventricular (parasternal) and left ventricular (apical) areas. The high volume of pulmonary blood flow and pulmonary venous return increase left ventricular volume, resulting in a hyperdynamic precordium. The systolic murmur is S1 coincident and is heard best along the left sternal border; it may end before the second heart sound. The second heart sound is narrowly split and has a loud pulmonary component. Some patients with VSDs have an early diastolic murmur indicating mild pulmonary or aortic valve insufficiency. Either condition indicates increased pathology: the pulmonary valve insufficiency indicates increased pulmonary artery pressure and the aortic valve insufficiency indicates prolapse of the aortic valve cusp. There is also a third heart sound, and a diastolic rumble can be heard at the apex.457,597
Infants with infundibular pulmonary stenosis may develop cyanosis from a right-to-left shunt pathology similar to tetralogy of Fallot. Cyanosis may also be seen in the older child with a large VSD and pulmonary hypertension (i.e., pulmonary vascular disease with Eisenmenger syndrome—see section, Common Clinical Conditions, Pulmonary Hypertension).
The chest x-ray and electrocardiogram also vary with the size of the ventricular septal defect (see Tables 8-25 and 8-26). If the VSD is small the ECG and chest x-ray may be normal.686 With a moderate VSD there is increased pulmonary venous return to the left ventricle, resulting in left ventricular hypertrophy (LVH) on the ECG. Cardiomegaly, increased pulmonary vascular markings, and an enlarged main pulmonary artery segment will be visible on the chest radiograph.597 In young infants with large VSDs, right ventricular hypertrophy may not be as marked on the ECG as in older infants who have pulmonary hypertension. When the right ventricular pressure equals the left ventricular pressure, biventricular hypertrophy is apparent on ECG and the left atrial enlargement may produce a biphasic P wave.
Cardiomegaly will be present on chest radiograph and results from enlargement of both ventricles and the left atrium. Increased pulmonary vascular markings (increased pulmonary blood flow) will also be present.597,686
Two-dimensional echocardiography accurately identifies the defect anatomy and any associated anomalies, including overriding atrioventricular valves, prolapse of the aortic valve cusp, aortic valve regurgitation, and ventricular outflow tract obstruction. Color-flow Doppler studies provide information regarding restriction of flow through the defect and allow assessment of pulmonary and right ventricular pressures. Measurement of left atrial and ventricular diameters provides information about shunt volume.
Transesophageal echocardiography is used preoperatively to further define the defect. It may also be used intraoperatively to evaluate closure of the defect(s).457,597
Because echocardiography provides precise details of the anatomy and allows assessment of pulmonary and ventricular pressures and shunts, cardiac catheterization is rarely necessary unless device closure is anticipated. Cardiac catheterization is used to evaluate pulmonary vascular disease (see section, Common Clinical Conditions, Pulmonary Hypertension).
Management
Medical Management
Because most VSDs are small, the infant who is asymptomatic (i.e., without signs of congestive heart failure or pulmonary hypertension) by 6 months of age can be followed conservatively. A large number of these defects close spontaneously; 75% to 80% close within the first 2 years. Children with small defects should be followed every 3 years to monitor for aortic valve prolapse or regurgitation.457,597,686,759 Prophylaxis for bacterial endocarditis is recommended for VSDs associated with complex heart disease (see Bacterial Endocarditis).
Infants with moderate and large defects and early (i.e., within the first few weeks of life) signs and symptoms of congestive heart failure and failure to thrive are treated medically with nutritional supplementation. A high-calorie formula is generally needed to maximize caloric intake while limiting fluid intake (see section, Common Clinical Conditions, Nutritional Compromise). Medical treatment usually includes diuretics and medications to reduce systemic afterload (see section, Common Clinical Conditions, Congestive Heart Failure).597 Infants who do not respond to medical therapy, especially those with signs and symptoms of pulmonary hypertension, require device (during catheterization) or surgical closure of the defect.481,597
A large number of infants with moderate defects and early signs and symptoms of congestive heart failure improve with medical management. In these infants the defect either decreases in size or becomes relatively smaller as the infant grows. When the defect becomes relatively smaller, it restricts the volume and pressure of the left-to-right shunt, which in turn results in decreased signs and symptoms of heart failure and decreased risk of pulmonary vascular disease.481
VSD closure has been recommended to prevent bacterial endocarditis, decrease the risk of aortic valve regurgitation, and prevent ventricular outflow obstruction and pulmonary vascular disease. However, as many as 94% of patients with small to moderate VSDs who were medically managed are in New York Heart Association Class I at 15 years after their diagnosis. In addition, both surgical repair and device closure have been associated with late rhythm disturbances, aortic valve regurgitation, and bacterial endocarditis.67,481,597 The medical and surgical team must always monitor outcomes and weigh the potential risks and benefits of medical management versus defect closure.
Device Closure
Experience with percutaneous device closure of muscular and membranous VSDs is more limited than device closure of patent ductus arteriosus or atrial septal defects. The initial experience with device closure of VSDs included mid-muscular defects, postoperative residual defects, VSDs that were difficult to reach surgically, or fenestrated defects.484 Devices can be placed percutaneously, intraoperatively, and periventricularly through an open sternotomy,27,40,389,545 and they have evolved rapidly over the past several years.
The Amplatzer septal occluder is currently the most frequently used ventricular closure device, with FDA approval since December 2001. The Amplatzer device is a double-disc device of nitinol mesh. The two discs are linked to each other with a central waist. Dacron fabric is incorporated into each disc and the waist. Modifications of this device, still in investigational use, include the Amplatzer muscular VSD occluder and the Amplatzer membranous VSD occluder. The Amplatzer membranous VSD occluder, the newest member and the first with an asymmetric design, was first used clinically in 2002. The Amplatzer family of devices is illustrated in Evolve Fig. 8-3 in the Chapter 8 Supplement on the Evolve Website.
Percutaneous transcatheter VSD closure is more difficult than that of atrial septal defects. Multicenter studies report successful closure in 95.3% of cases using several types of devices. Postcatheter complications include vascular complications, hemolysis, infection, device embolization, arrhythmias, valve regurgitation, and residual shunt. Mortality averages 0.2%. The most significant complicating arrhythmia is complete atrioventricular block, occurring in 2.8% of patients at various intervals after device placement.139,285 Aspirin is usually given for 3-6 months after device implantation until the device becomes endothelialized.27
Surgical Closure
Palliative surgery using a pulmonary artery band is no longer used, even for small infants with significant left-to-right shunts. Indications for early surgery include uncontrolled congestive heart failure with failure to thrive. Surgical repair is successfully performed within the first few months of life in infants as small as 2 kg. Additional indications for closure include large defects even without symptoms, defects with elevated pulmonary pressures, defects producing a pulmonary-to-systemic shunt ratio greater than 2:1, and defects with associated aortic cusp prolapse. Defects associated with elevated pulmonary pressure and signs of pulmonary hypertension are repaired as soon as signs of pulmonary hypertension are noted or before 2 years of age to prevent pulmonary vascular disease.457,597
VSDs are closed surgically through a median sternotomy on hypothermic, cardiopulmonary bypass. Muscular, perimembranous, and inlet defects are closed from the right atrium through the tricuspid valve (i.e., without a ventriculotomy). Subpulmonary defects are closed through the pulmonary valve (so the incision is made in the pulmonary artery). Outlet and perimembranous defects that involve prolapse of the aortic valve cusps require careful attention to support of the aortic valve cusp and avoiding injury to the conduction system.275,457 Multiple defects, especially near the ventricular apex, may require a small apical ventriculotomy or device closure. The device may be placed intraoperatively.457
A variety of materials are used to close VSDs, including autologous pericardium, woven Dacron, or homograft material, and selection is determined by surgeon preference.275 Intraoperative echocardiography and color flow Doppler are used to determine residual shunts or additional defects before the patient is removed from cardiopulmonary bypass or before the patient leaves the operating room.
Postoperative complications include congestive heart failure and arrhythmias. Right bundle branch block is usually present postoperatively if surgery is performed in patients less than 6 months of age. If closure of an apical ventricular septal defect required a ventriculotomy, postoperative myocardial dysfunction is likely.457 Postoperative complications are likely in infants with respiratory infections before surgical repair.597
Long-term survival of patients with VSD is good. Spontaneous closure of small defects is almost 80%. Ninety-four percent of symptomatic infants with moderate defects who respond to medical treatment and do not require surgical or catheter intervention continue to do well 15 years after initial diagnosis. Most infants with large, symptomatic VSDs who require surgery within the first year have excellent results with normal growth, development, and activity. Limited longitudinal studies show that pulmonary hypertension is rare; sinus node dysfunction is also rare; but progressive aortic valve insufficiency is more common. Children repaired after 2 years of age may demonstrate increased postoperative pulmonary resistance that may be progressive.597,759
In the discharge mortality data (Table 8-30) from the STS Congenital Heart Surgery Data Summary,851-853 mortality for surgical closure of VSD is highest in neonates. Infant discharge mortality is twice that of the child, but is just slightly greater than 1%. Therefore it appears that repairing symptomatic infants in the first year of life is the best management of large symptomatic ventricular septal defects.
Advanced concepts for the nurse caring for the infant or child with VSD are provided in Box 8-24.
Box 8-24 Advanced Concepts: Ventricle Septal Defect (VSD)
• The primary determinant of a left-to-right shunt and the pulmonary artery pressure is the size of the defect; once it is approximately half the size of the aortic outflow tract, it allows a relatively unrestricted shunt. Location of the defect also influences shunt volume and pressure.
• If the defect is large, the relative difference between pulmonary and systemic outflow tract resistance (including any subvalvular or valvular stenosis or pulmonary artery or aortic narrowing and resistances in the vascular bed) determines the volume of a left-to-right shunt.
• The presence of a loud murmur and a thrill does not indicate a significant left- to-right shunt.
Atrioventricular Septal Defect (AVSD)/AV Canal Defect
Pearls
• Atrioventricular septal defects are a group of abnormalities with a common finding—absence of atrioventricular septum. There are two forms of AVSDs—partial and complete.
 With complete AVSD, the mitral and tricuspid valve share some tissue—there is a single atrioventricular valve annulus.
With complete AVSD, the mitral and tricuspid valve share some tissue—there is a single atrioventricular valve annulus.• AVSDs do NOT close spontaneously. All require surgical repair.
Etiology
Atrioventricular septal defects (AVSD) are a group of abnormalities of the atrioventricular septum and atrioventricular valves.150 The one common finding in this group of abnormalities is the absence of the atrioventricular septum; therefore the term atrioventricular septal defect is used for this group of defects.30 AVSDs are mainly derived from faulty fetal development of the atrioventricular septum and the embryologic endocardial cushion tissue; thus, they are also called endocardial cushion defects.
Atrioventricular septal defects occur in 4% to 5% of all congenital heart defects. The estimated incidence is 0.19 in 1000 live births. An AVSD is the most common defect identified on fetal echocardiography,150,686 and the most common congenital heart defect in children with Down syndrome (see section, Essential Anatomy and Physiology, Etiologies of CHD: Noninherited and Genetic Factors). Patients with Down syndrome and AVSD rarely have associated cardiac defects. Children with AVSD but no Down syndrome may also have heterotaxy syndrome with asplenia and polysplenia.575 A common atrium is associated with Ellis-van Creveld syndrome.150
The AVSDs are divided into two forms: partial and complete (Fig. 8-34). In the partial form there are two distinct atrioventricular valve annuli: the tricuspid valve (with possible cleft) and a cleft mitral valve. In the complete form of AVSD there is a common atrioventricular valve annulus.
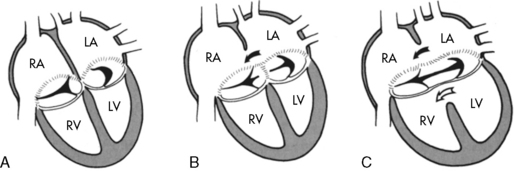
Fig. 8-34 Diagram of the atrioventricular (AV) valve(s) and cardiac septa in partial and complete atrioventricular septal defects (AVSDs). A, Normal AV valve anatomy with no septal defect. B, Partial AVSD with an ostium primum atrial septal defect (ASD) (solid arrow showing left to right atrial shunt) and clefts in the mitral and tricuspid valves. C, Complete AVSD. An ostium primum ASD (solid arrow indicates resulting atrial shunt) and an inlet ventricular septal defect (open arrow indicates ventricular shunt) are present. There is a common AV valve with large anterior and posterior bridging leaflets. LA, left atrium; LV, left ventricle; RA, right atrium; RV, right ventricle.
(From Park MK. Pediatric cardiology for practitioners, ed 5, Philadelphia, 2008, Elsevier, Fig. 12-15.)
A typical partial AVSD includes a primum atrial septal defect (ASD) with a cleft in the anterior leaflet of the mitral valve. The tricuspid valve may also have a cleft in the septal leaflet.
The complete form of AVSD includes a persistent fetal common atrioventricular canal. It is composed of a primum atrial septal defect that is contiguous with an inlet ventricular septal defect (VSD). When the common atrioventricular valve is open, the valve and the septal defects form a “canal” in the center of the heart, hence the name atrioventricular canal defect.150,575,686
In both partial and complete forms of AVSD there is downward displacement of the left atrioventricular valve so that both valves are on the same plane. This creates a deficiency in the length of the ventricular septum from the inlet portion to the apex and increases the length from the apex to the aortic valve. The result is an elongated and narrowed left ventricular outflow tract, commonly called a “gooseneck” deformity as illustrated in Fig. 8-35.
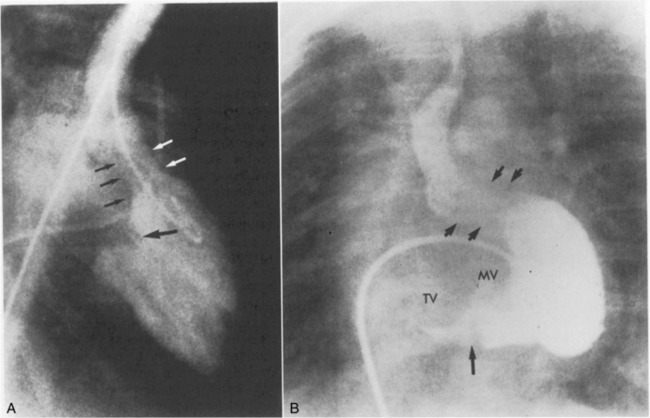
Fig. 8-35 Left ventricular angiograms depicting characteristic features of atrioventricular septal defects (AVSDs). A, The injection of contrast into the left ventricle creates a silhouette of the large cleft in the mitral valve (large black arrow). During systole, contrast material can be seen flowing back into the left atrium through the insufficient mitral valve. The downward displacement of the mitral valve creates a characteristic elongation of the left ventricular outflow tract (the “gooseneck” deformity), which is outlined by five small (black and white) arrows. B, This complete atrioventricular canal defect can be identified, because injection of contrast material into the left ventricle outlines both the mitral and tricuspid valve orifices (TV and MV); these valve rings are not separate and complete. A small indentation in the contrast material (single large arrow) is created by some ventricular septal tissue, but the contrast material reveals the ventricular level shunt. The elongation (“gooseneck” deformity) of the left ventricular outflow tract (four small arrows) is apparent.
There is wide variability among AVSDs with many variations of the atrial and ventricular septal defects and of atrioventricular valve anomalies. These variations provided the basis for complex terminology used in previous classifications. The current trend is to use the generic term AVSD and then describe the anatomy and shunting present.150,575
Pathophysiology
The physiology seen in this group of defects may be similar to that observed with an atrial septal defect (ASD), ventricular septal defect (VSD), or both, with the possibility of atrioventricular valve regurgitation (refer to Atrial Septal Defect and Ventricular Septal Defect, above). Every possible variation is seen with anatomic differences in both the size of the defect(s) and/or the degree of atrioventricular valve incompetence.575
Partial AVSD
In the partial form of AVSD, the primum ASD is usually large. A tongue of connective tissue (it is different from endocardial cushion tissue) separates the tricuspid valve annulus from the mitral valve annulus. This tissue displaces the right and left atrioventricular valves toward the ventricular septum where they may be firmly attached.686 A small, restrictive inlet ventricular septal defect may also be present; it is typically referred to as an intermediate or transitional AVSD.275
Patients with primum ASDs usually demonstrate symptoms of left-to-right shunts earlier that those with secundum ASDs. The cleft in the anterior leaflet of the mitral valve may allow for left ventricle to right atrial shunting; the degree of shunting depends on the competence of the valve. Infants with significant mitral regurgitation may develop early (i.e., within the first weeks of life) symptoms of congestive heart failure. Patients with partial AVSDs and competent atrioventricular valves may not be symptomatic until adulthood.150
Complete AVSD
A complete AVSD consists of a large septal defect with both interatrial and interventricular components and a single valve orifice that connects the atrial and ventricular chambers (see Fig. 8-34). This single atrioventricular valve usually has five leaflets: two on the right, one on the left, plus an anterior and a posterior leaflet that bridge the ventricular septum. Although the valve is very abnormal, it may be competent. The resulting pathophysiology is that of a large ventricular septal defect: Increased pulmonary blood flow and signs of congestive heart failure typically develop in the first months of life. The risk of pulmonary vascular disease is high. Left-to-right shunting and resultant symptoms and risk of pulmonary vascular disease are enhanced if the common atrioventricular valve is regurgitant.
In patients with Down syndrome, pulmonary vascular resistance may remain elevated during the first weeks of life. In these patients, development of a significant left-to-right shunt and signs of congestive heart failure may be delayed until the infant is several months old.575
The relationships of the leaflets to the crest of the septum and the insertion of the chordae that attach the valve leaflets to the ventricles affect the magnitude of the resulting shunt and influence the surgical repair. If the leaflets override the septum or the chordae insert through the defect into the opposite ventricle (straddle), blood flow from the atrium may be directed proportionately more to one ventricle than the other. Decreased blood flow to one ventricle may cause that ventricle to become hypoplastic, creating an “unbalanced” defect with either right or left ventricular dominance. Marked ventricular hypoplasia may prevent a two-ventricular repair. Ventricular hypoplasia is uncommon in patients with Down syndrome and AVSDs.575,686
When an AVSD is present, the location of the conduction system is abnormal. The septal defect results in posterior displacement of the atrioventricular (AV) node, so it is closer to the coronary sinus. In addition, the bundle of His is displaced inferiorly so it courses along the lower rim of the ventricular septal defect. On the electrocardiogram the posterior displacement of the AV node produces a prolonged PR interval (first-degree AV block), and “superior” QRS axis (left axis deviation). The abnormal location of the conduction system must be considered during the surgical correction.150,275
Children with unrepaired AVSDs require antibiotic prophylaxis against subacute bacterial endocarditis (SBE), although SBE is rare during in infancy. Postoperatively SBE prophylaxis is typically required for patients with valvular insufficiency, because that will cause turbulence near the septal defect patch (refer to Bacterial Endocarditis later in this section of the chapter).686
Clinical Signs and Symptoms
Partial AVSD
Children with a partial AVSD are usually asymptomatic. When this defect is associated with moderate or severe mitral valve regurgitation, symptoms of excessive pulmonary blood flow and congestive heart failure may develop during early infancy. These children are tachypneic, tire easily, and have recurrent respiratory infections and poor weight gain.150,686
The left-to-right shunt and resulting symptoms will be more severe in the presence of associated left heart abnormalities, such as subaortic stenosis, left atrioventricular valve disease, or aortic arch abnormalities.575 Symptoms may also be more severe if a common atrium is present.
The typical murmur of a partial AVSD is a systolic ejection murmur heard best at the upper left sternal border, radiating to the lung fields. The second heart sound is widely split and fixed during inspiration (because ejection of the increased volume of blood flow through the pulmonary valve takes longer than ejection of blood flow through the aortic valve). If mitral valve regurgitation is present, the murmur is holosystolic. A mid-diastolic murmur may be present at the apex if the left-to-right shunt is large.
Many children with AVSD have only left-to-right shunting of blood unless or until pulmonary vascular disease develops. However, the child may be mildly cyanotic if a common atrium allows mixing of systemic and pulmonary venous blood.150,686
On electrocardiogram about half of patients with AVSD demonstrate left axis deviation and a prolonged PR interval (first-degree AV block). Many patients demonstrate right ventricular hypertrophy with increased right ventricular volume load.686
The primary imaging technique for a partial AVSD is two-dimensional echocardiography. The defect in the lower atrial septum is clearly visualized, as is the cleft in the mitral valve. As with all forms of AVSD the deficiency of the inlet portion of the ventricular septum, the inferior displacement of the atrioventricular valves, and the attachment of the AV valves to the ventricular septum are visible. In a partial AVSD the two separate atrioventricular valve orifices are equidistant from the cardiac apex. Color-flow Doppler demonstrates the left-to right shunt and enables estimation of the right ventricular systolic pressure.150
Cardiac catheterization is rarely necessary but if performed, angiography will demonstrate the characteristic “gooseneck” deformity. Chest radiography may demonstrate cardiomegaly and increased pulmonary vascular markings. Right atrial enlargement may result from the jet of blood flow through the cleft mitral valve.150
Complete AVSD
In the complete form of AVSD, excessive pulmonary blood flow results in tachypnea, frequent respiratory infections, and failure to thrive within 1 to 2 months after birth (see section, Congestive Heart Failure). If the infant does not develop early symptoms, persistence of increased pulmonary resistance or early onset of pulmonary vascular obstructive disease should be suspected.
Atrioventricular valve regurgitation increases the symptoms of congestive heart failure and complicates management preoperatively and postoperatively. Patients with AV valve regurgitation are at increased risk for reoperation.
Preoperatively, the murmur resembles that of a large ventricular septal defect with a holosystolic murmur heard best at the lower left sternal boarder; it is usually accompanied by a thrill. There is also a holosystolic apical murmur from left AV valve regurgitation that may radiate toward the left axilla. The second heart sound is narrowly split with a louder pulmonary component. The precordium is usually hyperactive, and the liver is enlarged.686
Characteristic left axis deviation with a prolonged PR interval is present on electrocardiogram (see Table 8-26). Right ventricular hypertrophy is always evident, and most patients also have left ventricular hypertrophy.150
As noted, two-dimensional echocardiography is the primary diagnostic tool for identifying complete AVSDs. It provides complete imaging of all components and allows evaluation of the severity of abnormalities. Additional defects, including secundum atrial septal defects and additional ventricular septal defects, can be visualized and important information about the size and position of the atrioventricular valves, the anatomy of the leaflets, and the locations of the attachment of the papillary muscles in the left ventricle and the septal wall can be demonstrated with this technology. The relative size of both ventricles is also assessed.150,686 This information is essential for the surgical repair.
The chest radiograph of a child with a complete AVSD demonstrates cardiomegaly with a prominent pulmonary artery and increased pulmonary vascular markings. Cardiac catheterization is rarely needed to define the anatomy, but the characteristic “gooseneck” deformity will be demonstrated on the left ventricular angiogram. In an older child cardiac catheterization is necessary to evaluate pulmonary vascular resistance and rule out pulmonary vascular obstructive disease before complete repair.150 For further information, please refer to section, Common Clinical Conditions, Pulmonary Hypertension.
Frequently children with Down syndrome and AVSD have higher pulmonary vascular resistance than systemic vascular resistance, and their pulmonary vascular resistance is higher than that of children with AVSD without Down syndrome. Although some histologic studies show no difference in lung tissue between children with and without Down syndrome, other studies have demonstrated that children with Down syndrome have relative pulmonary parenchymal hypoplasia.150 However, when 100% oxygen is administered to children with Down syndrome and AVSD in the cardiac catheterization lab, pulmonary vascular resistance decreases and the difference between the pulmonary and systemic resistances is eliminated or reversed. This supports the belief that the cause of the high pulmonary vascular resistance in children with AVSD and Down syndrome is related to chronic nasopharyngeal obstruction, relative hypoventilation, and alveolar hypoxia. In addition, sleep apnea seen in children with Down syndrome produces hypoventilation, hypercarbia, and increased pulmonary vascular resistance.150 Hemodynamic studies of patients with AVSD and Down syndrome have documented a fixed and elevated pulmonary vascular resistance in 11% of patients less than 1 year of age.150
Management
The management of the child with AVSD is determined by the child's symptoms, anatomy, and presence of pulmonary vascular disease. Surgical intervention is ultimately required because the defects do not close spontaneously and are not amenable to catheter closure.
Repair of Partial AVSD
Because the partial AVSD rarely causes congestive heart failure, management consists of surgical closure between 6 and 12 months of age. If the diagnosis is not made until the infant is older, surgical repair is performed when the diagnosis is made.575
If significant mitral valve regurgitation is present, repair should not be delayed. Regurgitant flow thickens the leaflets around the cleft. Although some surgeons feel that this is beneficial to provide more secure sutures, most feel that the distortion and foreshortening of the valve leaflets prevent the surgical achievement of a competent valve.145
Repair of an AVSD is preformed through a median sternotomy incision and uses hypothermic cardiopulmonary bypass. The defect is approached through the right atrium. If there is a small, restrictive inlet ventricular septal defect, this defect will be closed first. Careful placement of sutures in this area is necessary to avoid surgically induced atrioventricular block.275 The cleft in the mitral valve is then sutured to prevent regurgitation and the primum ASD is closed with autologous pericardium. Synthetic patch material is avoided because it can contribute to hemolysis if there is persistent mitral valve regurgitation postoperatively. At the time of surgery, the surgeon verifies that the mitral valve is competent. Surgical results are excellent with an operative mortality of 2%.575
The most frequent postoperative complication is arrhythmia. Supraventricular tachycardia is the most common rhythm disturbance, occurring in about 16% of children following correction of AVSD. Complete heart block is seen in 3%.575
Reoperation may be needed in 10% to 15% of patients after repair of AVSD. The most common indication for reoperation is mitral valve regurgitation, although some patients may require reoperation for subaortic stenosis. The goal for reoperation of the mitral valve is to repair the valve, but some require replacement. Surgery may be delayed with little added risk if valve replacement seems likely.575,686
Repair of Complete AVSD
For the child with complete atrioventricular septal defect preoperative management includes medical therapy for congestive heart failure using diuretics and afterload reduction (see section, Common Clinical Conditions, Congestive Heart Failure). Medical management also includes nutritional support to minimize failure to thrive (see section, Common Clinical Conditions, Altered Nutrition and Potential Gastrointestinal Complications).
To prevent permanent development of pulmonary vascular obstructive disease and to treat congestive heart failure, surgical repair for uncomplicated complete AVSDs is typically performed in early infancy (at about 2 to 4 months).575
The goals of surgical repair of an infant with a balanced, complete AVSD include complete closure of the atrial and ventricular septal defects and effective use of the available atrioventricular septal valve tissue to create two competent atrioventricular valves. In an uncomplicated complete AVSD this may be accomplished in infants in the neonatal period, including those weighing as little as 3 kg.45,492 The critical component of this repair is the repair of the valve, particularly the left atrioventricular or mitral valve. Dysfunction of the right-sided atrioventricular (tricuspid) valve is much better tolerated than dysfunction of the left atrioventricular valve.275,575
A pulmonary artery banding is a palliative procedure performed through a left thoracotomy, without cardiopulmonary bypass. A “band” of synthetic material is placed around the pulmonary artery in an attempt to reduce pulmonary blood flow, decrease the severity of congestive heart failure, and reduce the risk of pulmonary vascular disease. This band on the pulmonary artery may cause distortion of the pulmonary valve or artery.153
Only in extreme cases—including complex anatomy, sepsis or respiratory illnesses such as respiratory syncytial virus or pneumonia—is palliative surgery with placement of pulmonary artery band considered.145,150,153 Some centers report successful outcomes with pulmonary artery banding for infants less than 1 month of age and less than 5 kg who exhibit significant congestive heart failure unresponsive to medical treatment. Successful banding has also been reported for infants with significant associated anomalies. However, the combined reported mortality rate (for banding and later debanding with complete repair) is as high as 15%.150,686
For complete correction of AVSD the defect is approached through a median sternotomy incision and repaired during hypothermic, cardiopulmonary bypass. The repair is performed through an incision in the right atrium. Based on the preference of the surgeon, either a single patch or double patch technique is used to close the atrial septal defect and ventricular septal defect and reconstruct the atrioventricular valve (Fig. 8-36).41,145,150,275,575,623
Fig. 8-36 Surgical correction of atrioventricular septal defect. A patch is used to close the atrial septal defect (pericardium is usually used). When a complete AVSD is present (as shown), a second woven patch (usually Dacron) can be used to close the ventricular septal defect. The patches are placed to divide common atrioventricular valve tissue, particularly the bridging anterior leaflet. Clefts in the mitral valve are sutured to prevent significant postoperative mitral insufficiency while avoiding the creation of mitral stenosis.
(From Bender HW, et al: Repair of atrioventricular canal malformation in the first year of life. J Thorac and Cardiovasc Surg 84:518, 1982.)
In the single patch technique repair of AVSD, autologous pericardium is used to close the defects. In the double patch procedure, a Dacron patch is used to close the VSD and autologous pericardium is used to close the ASD. Usually the cleft in the left atrioventricular valve is sutured to form a bi-leaflet valve. Chordal attachments of the atrioventricular valve must be identified preoperatively with a surface echocardiogram or in the operating room with a transesophageal echocardiogram. The surgeon visually confirms the anatomy and makes the appropriate adjustments in patch placement. The abnormal location of the conduction system makes it critically important that the surgeon place the sutures to avoid the conduction tissue. Direct injury to the conduction tissue or edema around the sutures can cause postoperative heart block, including complete atrioventricular block.275
Postoperative complications following repair of complete AVSDs include persistent mitral valve regurgitation, low cardiac output, pulmonary hypertension, elevated left atrial pressures, and arrhythmias such as heart block and junctional atrial tachycardia. Because the atrioventricular valve has been reconstructed and risk of persistent AV valve regurgitation remains, volume loading to increase cardiac output should be avoided if at all possible for the first 24 postoperative hours. Volume loading may put tension on the site of valve sutures and can increase AV valve regurgitation. Atrial filling pressures should be maintained at lower values with inotropic support used to increase cardiac output, as needed. If either a residual ventricular septal defect or significant AV valve regurgitation is thought to be present, aggressive afterload reduction is needed.153
Postoperative mortality is related to pulmonary hypertension. It is important to recognize any anatomic causes of pulmonary hypertension, such as severe left atrioventricular valve stenosis or regurgitation, or a residual ventricular septal defect.153 The trend toward earlier age of repair has decreased the risk of pulmonary hypertension. When correction is performed at less than 3 months of age, fixed elevated pulmonary vascular resistance is rare and outcomes are good. If surgical repair occurs at 6 months of age or older, pulmonary vascular resistance may be elevated and postoperative pulmonary arterial hypertensive crises may be observed postoperatively.575 (For additional information, please refer to Common Clinical Conditions, Low Cardiac Output and Common Clinical Conditions, Pulmonary Hypertension, as well as the third section of this chapter, Postoperative Care and Anticoagulation.)
Postoperative arrhythmias may be problematic following repair of complete AVSD. As noted, the conduction tissue is in an abnormal position at the rim of the septal defect. Sinoatrial dysfunction may be present preoperatively, and complete heart block may be present postoperatively. When dysfunction of the sinoatrial node is present, cardiac output may be improved with atrial pacing. The preferred treatment of complete heart block is AV sequential pacing. Atrioventricular valve regurgitation may increase if ventricular (ventricular sensing, pacing inbited, or VVI) pacing is provided, rather than atrioventricular pacing.153
The mortality rate following repair of AVSD averages 3% to 10%. The surgical mortality is the same for patients with and without Down syndrome. Increased mortality risk for complete repair of an AVSD is associated with very young age at operation, severity of preoperative atrioventricular valve regurgitation, hypoplasia of the left ventricle, increased or fixed pulmonary vascular resistance, and severity of preoperative symptoms.150,686
If additional defects are present they can increase the operative risk. Patients with complex forms of atrioventricular septal defects include unbalanced AVSD (surgical risk is highest for right ventricular dominant defect; this means left ventricular hypoplasia is present) and when the defect is present in combination with tetralogy of Fallot, transposition of the great arteries, and double outlet ventricles. Please refer to those associated defects discussed in the following sections.
Surgical repair of AVSDs is one of the great success stories in the past several decades of congenital cardiac surgery.150 In the STS Congenital Heart Surgery Data Summary, partial AVSD repairs in children have a postoperative mortality of 0%, and infant repairs of complete AVSD repairs has a mortality of 2% (Table 8-31).851-853 Long-term survival is excellent. Reoperation is required for 2.5% of patients, most often indicated for progressive left atrioventricular valve regurgitation or for relief of left ventricular outflow tract obstruction.150 Endocarditis remains a long-term postoperative risk for most patients, so antibiotic prophylaxis is recommended for periods of increased risk of bacteremia (see section, Bacterial Endocarditis).
Care of the infant or child with AVSD is both challenging and rewarding. Advanced concepts regarding atrioventricular septal defects are listed in Box 8-25.
Box 8-25 Advanced Concepts: Atrioventricular Septal Defect (AVSD)
• Atrioventricular septal defects are the most common defects in patients with Down syndrome.
• The degree of atrioventricular valve regurgitation determines the onset of symptoms in both forms of AVSDs.
• A delay in the onset of symptoms past 2 months may indicate persistent pulmonary hypertension.
• Surgical repair of complete AVSD is performed before 6 months of age to prevent pulmonary vascular obstructive disease.
• Abnormal development of the conduction system is an important risk factor for postoperative arrhythmias, especially complete heart block.