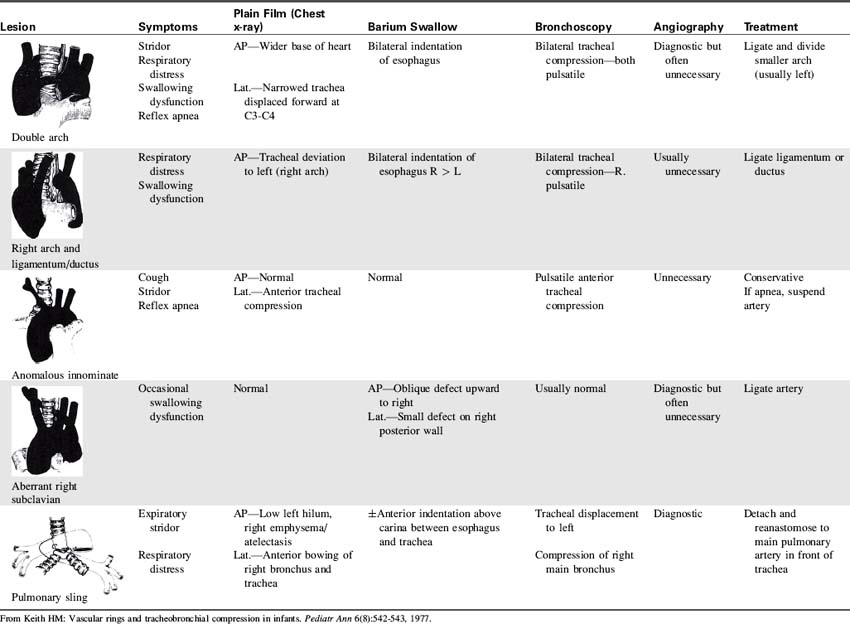
From Korkola SJ, Tchervenkov CI, Mavroudis C. Infective endocarditis. In Mavroudis C, Backer CL, editors. Pediatric cardiac surgery, ed 3, Philadelphia, 2003, Mosby/Elsevier, p. 769.
Clinical Signs and Symptoms
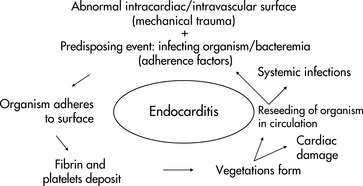
The onset of endocarditis can be acute and fulminant, potentially resulting in death within weeks, or slow and insidious with low-grade fever, malaise, and night sweats recurring over months. A high index of suspicion for endocarditis is needed when caring for a patient with a cardiac lesion. Findings are often subtle and early detection and treatment are imperative to limit endocardial damage and reduce mortality and morbidity. Any indication that a patient may have endocarditis warrants investigation: obtain blood cultures promptly.
Some of the symptoms of endocarditis can mimic those seen with active rheumatic fever but positive blood cultures will distinguish between the two. Acute rheumatic fever and endocarditis do not happen simultaneously.289
If endocarditis is present on the left side of the heart, systemic emboli often produce extremity ischemia with pain and compromise of perfusion, renal dysfunction, arthralgia, or focal neurologic signs; specific signs are determined by the location of the embolus. Additional signs of endocarditis include the development of new or changing murmurs, fever, and subtle signs of illness, including malaise, headache, or arthralgia. The list of possible signs and symptoms is extensive (Table 8-40).104
Table 8-40 Signs and Symptoms of Endocarditis
| Sign/Symptom |
Incidence in Endocarditis Population (%) |
| Fever* |
95-100 |
| Onset of murmur |
80-85 |
| New or changing murmur |
10-40 |
| Chills |
42-75 |
| Sweating |
25 |
| Anorexia and weight loss |
25-55 |
| Malaise |
25-40 |
| Dyspnea |
20-40 |
| Cough |
25 |
| Stroke |
13-20 |
| Headache |
15-40 |
| Nausea, vomiting |
15-20 |
| Myalgia/arthralgia |
15-30 |
| Chest pain |
8-35 |
| Abdominal pain |
5-15 |
| Back pain |
7-10 |
| Confusion |
10-20 |
| Neurological abnormalities |
30-40 |
| Embolic event |
20-40 |
| Splenomegaly |
15-50 |
| Clubbing |
10-20 |
| Peripheral manifestations* |
|
| Osler's nodes (painful lesions on finger and toes) |
7-10 |
| Splinter hemorrhages (seen in nail beds) |
5-15 |
| Petechiae |
10-40 |
| Janeway lesions (nontender lesions on palms/soles) |
6-10 |
| Retinal lesions/Roth spots (retinal hemorrhage with pale center) |
4-10 |
| Evidence of vascular phenomena (emboli, infarcts, hemorrhage, aneurysm)* |
Variable depending on location |
From Braunwald E, Zipes DP, Libby P. Heart disease, ed 6, Philadelphia, 2001, Saunders, p. 1730, lexi.com/web/news.isp?id=100025.
The presence of endocarditis is suggested by a positive blood culture. Blood cultures are positive in 85% to 95% of cases of infective endocarditis.248 If a child has received antibiotics before the identification of the infecting organism, special techniques may allow isolation of the organism from subsequent blood cultures. Repeat blood cultures (up to three sets in 24 h) are often necessary to isolate an organism, and a small number of children with endocarditis never have a positive blood culture.
Additional nonspecific laboratory tests suggestive of endocarditis include positive acute phase reactants. The erythrocyte sedimentation rate is typically elevated, as is the C-reactive protein. Some laboratories are able to perform complex antigen or antibody studies or isolate peripheral reticuloendothelial cells; these studies are not used widely.
Although the ECG may not be helpful in the diagnosis of endocarditis (other than possible onset of new conduction disturbances), the echocardiogram often enables visualization of vegetations and is considered an essential tool in diagnosis. A negative echocardiogram, however, will not rule out the presence of endocarditis. The chest radiograph may show evidence of congestive heart failure and/or changes indicative of pulmonary embolic lesions or abscess.
Diagnosis often is based on the Duke Criteria for the Diagnosis of Infective Endocarditis.248,532 Major criteria include532:
• positive blood culture for microorganisms typical for endocarditis (two positive cultures typically required, drawn more than 12 h apart although single positive culture for Coxiella burnetii or antiphase 1 IgG antibody titer greater than 1:800 acceptable)
• evidence of endocardial involvement
• echocardiographic evidence of infectious endocarditis (oscillating intracardiac mass, abscess or new partial dehiscence of prosthetic valve)
• new valvular regurgitation.
Minor criteria include532:
• predisposing heart condition or drug use
• fever (temperature above 38° C)
• vascular phenomena
• immunologic phenomena
• microbiologic evidence.
Diagnosis is positive if a patient has two major criteria (at least two positive blood cultures and evidence of endocardial involvement typically determined by transthoracic or ?transesophageal echocardiographic evidence), one major plus three minor criteria, or five minor criteria.248,532
Prevention and Management
Prevention
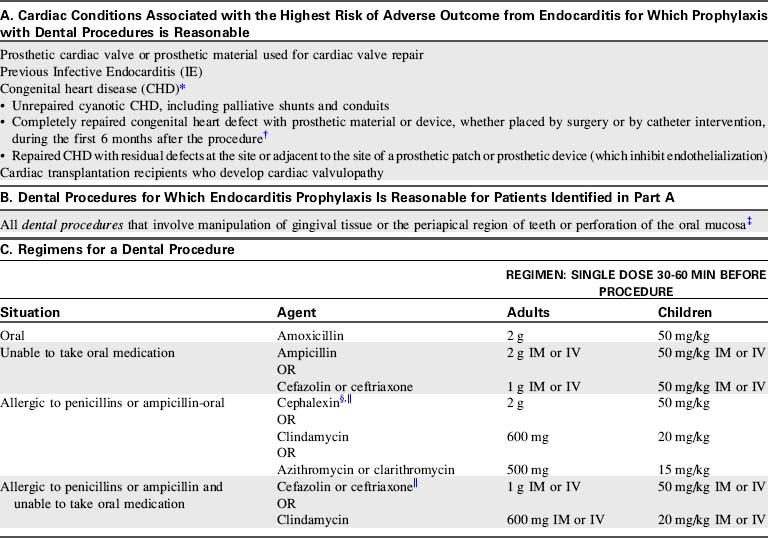
Prevention rather than need to treat endocarditis is the goal. However, several aspects of endocarditis prophylaxis remain controversial. For many years the American Heart Association (AHA) has published extensive recommendations regarding antibiotic prophylaxis for the prevention of endocarditis under a variety of circumstances; these recommendations have been revised over the years based on review of published evidence. These guidelines address the administration of specified antibiotics before an event that is likely to cause entry of causative organisms into the circulation of an individual with risk factors. It is always prudent to obtain and follow the most current guidelines published by the AHA.951
In 2007 significant revisions were made in the AHA Guidelines for Endocarditis Prophylaxis because of the absence of high-level evidence that the prophylaxis decreases costs, reduces the risk for allergic reactions (including anaphylaxis) by reducing exposure to antibiotics, and reduces antibiotic overuse that can promote development of resistant organisms.
The 2007 Guidelines noted:
The major changes in the updated recommendations include the following: (1) The Committee concluded that only an extremely small number of cases of infective endocarditis might be prevented by antibiotic prophylaxis for dental procedures even if such prophylactic therapy were 100% effective. (2) Infective endocarditis prophylaxis for dental procedures is reasonable only for patients with underlying cardiac conditions associated with the highest risk of adverse outcome from infective endocarditis. (3) For patients with these underlying cardiac conditions, prophylaxis is reasonable for all dental procedures that involve manipulation of gingival tissue or the periapical region of teeth or perforation of the oral mucosa. (4) Prophylaxis is not recommended based solely on an increased lifetime risk of acquisition of infective endocarditis. (5) Administration of antibiotics solely to prevent endocarditis is not recommended for patients who undergo a genitourinary or gastrointestinal tract procedure. These changes are intended to define more clearly when infective endocarditis prophylaxis is or is not recommended and to provide more uniform and consistent global recommendations.”951
Table 8-41 highlights the AHA guidelines for prevention of infective endocarditis, published in October 2007.951
The 2007 AHA Endocarditis Prevention guidelines produced some controversy. Concerns have been expressed regarding confusion over changes, medical and dental legal considerations, and no recommended prophylaxis for some lesions that were previously considered at higher risk for endocarditis, such as ventricular septal defect, patent ductus arteriosus, and aortic stenosis. Apprehension is natural following major guidelines changes because antibiotic prophylaxis was recommended in past guidelines for more types of congenital heart defects, and was recommended preoperatively and postoperatively (without a 6-month limitation) and in some cases was recommended even if postoperatively the child had no residual defect.
Currently, in many cases the recommendation for prophylaxis is based on the presence of residua.951 Because the heart is a dynamic organ in dynamic individuals the absence of residua at one moment may not be so later. Also, residua may be difficult to rule out when diagnostic techniques may be hindered by lack of cooperation in young children. Practitioners are advised to use judgment to make appropriate decisions in each individual case.
Nonpharmacologic endocarditis prevention measures are those which educate patients and parents and can reduce events that cause bacteremia (Box 8-46). These precautions should always be followed and included in published and online information for patients and parents. It is the duty of nurses and the entire healthcare team to teach patients and parents about these precautions, which include appropriate antibiotic treatment of infections, good dental hygiene and care, timing of dental care to avoid dental procedures close to the time of cardiac interventions, good skin care, and education about risky behaviors and risk factors.
Box 8-46 Nonpharmacologic Endocarditis Prevention Measures
• Provide patient/family with a current AHA Prevention of Bacterial Endocarditis wallet card, which is available in English and Spanish at www.americanheart.org.
• Teach the child and family that they should seek appropriate antibiotic treatment for infections.
• Encourage good dental hygiene and regular dental care.
• Elective dental treatment should be performed at least 1-2 months before or 4-6 months after cardiac surgery to try to avoid a potentially significant bacteremic episode in temporal proximity to cardiac surgery.818
• Counsel patients and families regarding potentially greater risks of complications from intravenous drug abuse, piercings, and tattooing for individuals with many types of cardiac disease.706
• Encourage good skin care—cleaning wounds, avoiding nail biting, and refraining from manipulation of acne lesions. Treatment of chronic acne should be discussed with a physician.706
• Educate patients and families about:

Risks and pathophysiology

Their responsibility to be advocates for the child and to inform care providers of risks

Signs of endocarditis with emphasis on fever and the need to inform physicians of any symptoms that might warrant obtaining blood cultures
Treatment
Effective treatment of endocarditis requires several weeks of intravenous antibiotic therapy. Delay of effective antibiotic therapy is associated with greater incidence of complications. The choice of antibiotics is determined by the organism and its sensitivity to antibiotics, as well as the site(s) of associated infection. During therapy the serum peak bactericidal titer will be monitored to evaluate the effectiveness of the antibiotic regimen and the likelihood of cure.
If a prosthetic valve is infected, 6 weeks of intravenous antibiotic therapy will be required. If cardiovascular deterioration, systemic embolic events, obstruction, regurgitation, or staphylococcal endocarditis with heart failure are present with prosthetic valve endocarditis, valve replacement will be required under urgent conditions.499
Surgery may be necessary to remove infected tissue and restore better hemodynamics. For endocarditis without a prosthetic valve, surgical indications may include refractory congestive heart failure, uncontrolled infection, repeated serious embolic episodes, fungal or other difficult-to-treat endocarditis, the presence of prosthetic material, progressive renal failure, severe valve dysfunction, the presence of an echocardiographically identifiable intracardiac vegetation (although this is controversial), and invasion of cardiac tissue leading to conduction disturbances, abscesses, or cardiac fistulae.495,499 Surgery may be indicated after the acute endocarditis is resolved if there are significant hemodynamic sequelae amenable to repair. Before surgical intervention the child's neurologic status must be monitored closely to detect any evidence of cerebral emboli.
Postoperative care includes all typical interventions after cardiac surgery, with emphasis on care likely to be required in light of the valve or tissue involved. Additional considerations are hemodynamic consequences associated with sepsis (see Chapter 6), treatment of other affected organs, anticoagulation implications if a mechanical valve is used and the patient has experienced hemorrhagic complications, continued antibiotic therapy, and observation for recurrent infection.499
Throughout therapy, support of cardiovascular function is required. Valvular endocarditis often results in congestive heart failure, so limitation of fluid intake and diuretic therapy are frequently required (see Common Clinical Problems, Congestive Heart Failure).
Treatment of lesions of other organs must occur simultaneous with treatment of the endocarditis. Outcomes have improved with the advent of interventional radiology and catheterization techniques such as device occlusion of vessels that have become hemorrhagic as the result of erosion from infection.
Myocarditis
Gail L. Stendahl
Etiology
Myocarditis is an inflammatory process that involves the cardiac muscle; most result from viral infection (Box 8-47). Each decade there has been a shift in viral demographics of myocarditis. In the 1970s and 1980s, coxsackievirus was most common, but in the 1990s and 2000s viral causes included adenovirus and a broader range of enteroviruses. Most recently, parvovirus B19 has become a commonly identified pathogen in patients with suspected myocarditis.873
Box 8-47 Viral Causes of Myocarditis
From Towbin, JA. Myocarditis. In Allen HD, Shaddy RE, Driscoll DJ, Feltes TF, editors. Moss and Adams' heart disease in infants, children, and adolescents, including the fetus and young adult, ed 7. Philadelphia, 2008, Lippincott Williams & Wilkins, p. 1208, Table 58-1.
| Enterovirus |
Varicella |
| Coxsackie A |
Mumps |
| Coxsackie B |
Measles |
| Echovirus |
Rabies |
| Poliovirus |
Hepatitis B, C |
| Adenovirus |
Rubella |
| Parvovirus B19 |
Rubeola |
| Cytomegalovirus |
Respiratory syncytial virus |
| Herpesvirus |
Human immunodeficiency virus |
| Influenza A |
Epstein-Barr virus |
Nonviral causes of myocarditis include infectious agents such as rickettsiae, fungi, bacteria, protozoa, and other parasites (Box 8-48). Noninfectious etiologies include drug toxicity, as with some antimicrobial medications, along with hypersensitivity, autoimmune, collagen-vascular diseases, or other disorders such as Kawasaki disease and sarcoidosis (Box 8-49).873
Box 8-48 Nonviral Causes of Myocarditis
From Towbin, JA. Myocarditis. In Allen HD, Shaddy RE, Driscoll DJ, Feltes TF, eds. Moss and Adams' heart disease in infants, children, and adolescents, including the fetus and young adult, ed 7. Philadelphia, 2008, Lippincott Williams & Wilkins, p. 1208, Table 58-2.
| Rickettsial |
Protozoal |
Fungi and Yeasts |
| Rickettsia rickettsii |
Trypanosoma cruzi |
Actinomycosis |
| Rickettsia tsutsugamushi |
Toxoplasmosis |
Coccidioidomycosis |
| |
Amebiasis |
Histoplasmosis |
| |
|
Candida |
| Bacterial |
Other Parasites |
| Meningococcus |
Toxocara canis |
| Klebsiella |
Schistosomiasis |
| Leptospira |
Heterophyiasis |
| Mycoplasma |
Cysticercosis |
| Salmonella |
Echinococcus |
| Clostridia |
Visceral larva migrans |
| Tuberculosis |
Trichinosis |
| Brucella |
|
| Legionella pneumophila |
|
| Streptococcus |
|
| Smallpox |
|
Box 8-49 Causes Of Myocarditis: Noninfectious Etiologic Agents
From Towbin, JA. Myocarditis. In Allen HD, Shaddy RE, Driscoll DJ, Feltes TF, eds. Moss and Adams' heart disease in infants, children, and adolescents, including the fetus and young adult, ed 7. Philadelphia, Lippincott Williams & Wilkins, 2008, p. 1208, Table 58-3.
| Toxic |
Hypersensitivity/Autoimmune |
| Scorpion |
Rheumatoid arthritis |
| Diphtheria |
Rheumatic fever |
| |
Ulcerative colitis |
| |
Systemic lupus erythematosus |
| |
Mixed connective tissue disease |
| |
Scleroderma |
| |
Whipple disease |
| Drugs |
Other |
| Sulfonamides |
Sarcoidosis |
| Phenylbutazone |
Kawasaki disease |
| Cyclophosphamide |
Cornstarch |
| Neomercazole |
|
| Acetazolamide |
|
| Amphotericin B |
|
| Indomethacin |
|
| Tetracycline |
|
| Isoniazid |
|
| Methyldopa |
|
| Phenytoin |
|
| Penicillin |
|
In most cases, myocarditis remains idiopathic.873 The incidence of myocarditis is difficult to determine because of the varied presentation; patients may be asymptomatic or may develop cardiogenic shock and die. Estimated prevalence frequently ranges from 1 to 10 per 100,000 persons. Viral RNA has been detected in myocardial tissue in pediatric patients with sudden death.179 Infants and young children may be more prone to the development of myocarditis because of a higher overall rate of enteroviral and adenoviral infections.528
Pathophysiology
Three related mechanisms result in myocardial injury from infectious agents, including invasion of the myocardial cells, production of a myocardial toxin, and immune-mediated myocardial damage.528 The process can be divided into three phases: infection and proliferation, autoimmunity, and progression to dilated cardiomyopathy. The immune-mediated changes led by T-lymphocytes and macrophages are the predominant mechanisms that result in myocardial injury. The increased effect of T cells, activation of cytokines, and synthesis of nitric oxide destroys the myocytes, causing ventricular remodeling and progression to dilated cardiomyopathy.528
Several changes result in the pathophysiologic response in patients with myocarditis. First, the sympathetic nervous system may preserve blood pressure and systemic blood flow through vasoconstriction. This adrenergic response is associated with tachycardia and an increase in ventricular afterload. Congestive heart failure develops with disease progression. The increase in ventricular end-diastolic volume and pressure results in increased left atrial pressure, which, in turn, increases pulmonary venous pressure and leads to pulmonary edema.
Eventually, all cardiac chambers dilate, particularly the left ventricle. This dilation, in addition to causing poor ventricular function, creates worsening pulmonary edema and symptoms of congestive heart failure. The ventricular dilation may also cause mitral regurgitation, further increasing left atrial volume and pressure.
During the healing stages of myocarditis, fibroblasts replace normal cells, resulting in scar formation. Reduced elasticity and ventricular performance can produce persistent heart failure. In addition, ventricular arrhythmias commonly accompany fibrosis.528
There are subtypes of myocarditis based on presenting symptoms, clinical course, outcome, and histology.528 Fulminant myocarditis is characterized by a distinct, sudden onset of cardiac failure, severe left ventricular dysfunction, and cardiogenic shock. Acute myocarditis represents the largest group of patients. The onset of symptoms is often indistinct, and patients seem to develop a more gradual deterioration in ventricular function. Chronic myocarditis is characterized by an unclear onset of congestive heart failure with a slow progressive deterioration in ventricular function.528
Clinical Signs and Symptoms
The clinical presentation depends on the age of the child. Nonspecific flulike illness or episodes of gastroenteritis may precede symptoms of congestive heart failure. Newborns and infants present with poor appetite, fever, irritability or listlessness, pallor, and diaphoresis. Older children and adolescents commonly have a recent history of viral disease before presentation.873
Typically the child has fever, tachycardia disproportionate to the degree of fever present, arrhythmias, and signs of congestive heart failure, including a gallop rhythm, tachypnea, and signs of systemic and pulmonary edema (see section, Common Clinical Conditions, Congestive Heart Failure). The parents may note lethargy, and the child may complain of chest pain, weakness, myalgia, or constant fatigue. A history of asthma or congenital heart disease can skew the clinical picture.885
If significant myocardial dysfunction is present the child may have signs of poor systemic perfusion or shock (see Shock in Chapter 6). A systolic tricuspid or mitral murmur may be noted that is consistent with the development of atrioventricular valve insufficiency resulting from progressive ventricular dilation. Often, a third heart sound (gallop rhythm) develops as the result of rapid filling of a noncompliant, poorly contractile left ventricle.885 Pulsus alternans may result from decreased ventricular contractility, and a pericardial or pleural friction rub also may be present.
The initial evaluation of a child with suspected myocarditis includes evaluation of the chest radiograph, electrocardiogram (ECG), and cardiac biomarkers. An echocardiogram should be performed to evaluate ventricular function and the degree of any mitral regurgitation.870
The chest radiograph may reveal cardiomegaly, pulmonary venous engorgement, and pulmonary edema. It may be difficult to distinguish any increase in the size of the cardiac silhouette produced by a pericardial effusion from the cardiomegaly associated with a poorly functioning myocardium. Pleural effusions also may be noted.
Blood tests (e.g., complete blood count, erythrocyte sedimentation rate, C-reactive protein, and other chemistry profiles) usually are not helpful to confirm the diagnosis of myocarditis. Cardiac biomarkers showing myocardial injury often are elevated, including creatinine phosphokinase myocardial band (CPK-MB) fraction, Troponin I and/or Troponin T. These can be followed serially to assess ongoing inflammation and injury.528
The B-Type natriuretic peptide (BNP) blood test is a measurement of cardiac hormone produced by the ventricular myocardium in response to volume expansion and pressure overload. It is a hormonal marker of ventricular dysfunction and a surrogate quantifiable marker of the degree of congestive heart failure; BNP can also be useful when monitored serially. Viral serologic tests and polymerase chain reaction (PCR) assays of blood, stool, urine, and nasopharyngeal specimens can be adjunctive methods in diagnosing myocarditis; however, they are often negative.873
The echocardiogram is required to rule out the presence of structural heart disease, and it will enable evaluation of heart size, ventricular contractility, and atrioventricular (AV) valve function. The echocardiogram also will confirm the presence of any significant pericardial effusion.
Cardiac magnetic resonance imaging (MRI) is an emerging field that is capable of showing delayed enhancement of the affected myocardium. The cardiac MRI can show nodular and patchy areas of inflammation, often seen first in the lateral and inferior wall of the heart; these can be used later to guide biopsy.
A cardiac catheterization may be performed if there is any question about the presence of structural heart disease, pulmonary hypertension, coronary artery anatomy, or severe ventricular dysfunction with AV valve disease. The cardiac catheterization typically shows depressed cardiac index, elevated left ventricular end-diastolic pressure, and elevated mean atrial pressure. Angiography shows decreased left ventricular function with or without mitral regurgitation although the utility of angiography to evaluate ventricular and valve function has recently decreased because the quality of available noninvasive imaging is extremely high.
During catheterization an endomyocardial biopsy (EMB) may be obtained to allow histologic grading of the myocarditis and possible explanation of the causative organism. Right ventricle EMB using the Dallas Criteria for classification continues to be the “gold standard” for the diagnosis of myocarditis.528 Improvement in diagnostic accuracy can be achieved by using immunohistochemical markers for lymphocytes and applying polymerase chain reaction (PCR) techniques to detect the presence of virus in a biopsy specimen.189 This is often extremely valuable in the establishment of the cause and in some cases to guide therapy.
Management
Treatment of the child with myocarditis includes managing the underlying infection or disease (if identified), maximization of ventricular function, and cardiovascular support (optimize heart rate, preload and contractility, and reduce afterload). Because the symptomatic child with myocarditis is at risk for development of serious arrhythmias and sudden cardiac arrest (sudden death), admission to the critical care unit and continuous electrocardiographic monitoring and observation are required.
If an infectious agent is found the child may require isolation or treatment with antimicrobial agents. The physician may recommend that the child be maintained on bedrest to reduce cardiac output requirements. Fever should be treated with antipyretics, because fever will increase oxygen consumption and myocardial work.
Assisted ventilation along with continuous positive airway pressure relieves the excessive work of breathing, improves pulmonary edema and oxygenation, and may have a beneficial effect in reducing left ventricular afterload. In the setting of low cardiac output, severe ventricular dysfunction, and absence of cardiac reserve, endotracheal intubation should be carefully planned with contingency plans ready to enact if the child decompensates. Risk of decompensation is high during intubation because the sedation needed may lead to hypotension, cardiovascular collapse, and even cardiac arrest.528
Patients with fulminant myocarditis and cardiogenic shock require aggressive intervention to support circulation. Treatment of shock requires maintenance of adequate intravascular volume, correction of electrolyte or acid-base imbalances and use of inotropic and vasodilator therapy. Intravenous inotropic agents should be used judiciously and may be more helpful for short-term stabilization than for longer-term support. When low cardiac output and elevated systemic vascular resistance are present, milrinone therapy should be considered. The major advantages of milrinone are that it does not increase heart rate and it improves relaxation of the heart.528
Mechanical circulatory support with extracorporeal membrane oxygenation (ECMO) or ventricular assist devices may be life-saving in patients with fulminant myocarditis when inotropic agents are ineffective in maintaining adequate cardiac output (see Chapter 7). The use of ventricular assist devices versus transplantation in acute and fulminant myocarditis as a bridge to recovery remains controversial; assist devices are thought to be underused as a therapy.528
Transplantation may be the only option for children with significant cardiac failure resulting from myocarditis. Preferably, children are not listed for transplant until they have progressed to the chronic phase of the disease, because recovery is possible even in the most severe cases.64
Treatment of arrhythmias requires administration of antiarrhythmic drugs, although these medications should be used with caution because many of them also depress myocardial contractility. If antiarrhythmic therapy is prescribed the nurse must assess the child carefully for signs of decreased systemic perfusion and notify a physician or other on-call provider immediately if these develop. If arrhythmias remain unresponsive to pharmacologic therapy, pacing wires may be inserted to allow overdrive pacing (see section, Common Clinical Conditions, Arrhythmias).
If a significant pericardial effusion is present the nurse must monitor for signs of cardiac tamponade, including a rise in central venous pressure, poor systemic perfusion, progressive unremitting tachycardia, and pulsus paradoxus. Pericardiocentesis may be required to decompress the pericardium (see Evolve Fig. 19-2 in the Pediatric Trauma information in the Chapter 19 Supplement on the Evolve Website).
Patients with mild-to-moderate cardiac failure should be treated with conventional pharmacologic agents, including diuretics (e.g., furosemide, spironolactone), angiotensin-converting enzyme inhibitors or angiotensin receptor blockers, and possibly digoxin.528 Digoxin, if employed, should be used judiciously in low dose and without a loading dose, to reduce risk of toxicity. Anticoagulation with aspirin, warfarin, or intravenous heparin may be used to prevent thrombus formation when the left ventricle is dilated and function is severely depressed.
After stabilization, beta-adrenergic antagonists, such as carvedilol, may be introduced cautiously and the dose slowly titrated up over time. Beta blockers are effective in improving systolic function in adults with heart failure, although data remain limited regarding effectiveness in children.793 Beta-blockers are currently being used in pediatrics in hopes to reduce mortality and improve left ventricular ejection fraction in chronic heart failure.528
Use of corticosteroids in the treatment of myocarditis is controversial, because steroids may suppress the child's immune response, and result in progression of the initial infectious process. Occasionally, if myocarditis produces severe complications, including arrhythmias unresponsive to medical management, corticosteroids may be administered in an attempt to reduce myocardial inflammation. Overall survival in children has not differed in patients who received steroids in addition to conventional therapy versus those who received conventional therapy alone.252
Immunosuppression for the treatment of myocarditis remains controversial.873 Immunosuppressive agents that are administered early in the course of myocarditis increase viral replication, damage heart cells, and increase mortality. If immunosuppression is used it should be initiated only during the autoimmune phase of the disease process. Treatment with intravenous immunoglobulin (IVIG) can alter the immune response and may improve outcomes by reducing ongoing myocardial cell damage.189 The IVIG may also neutralize the antibodies that form against the causative virus and lead to ongoing damage via an autoimmune mechanism.
Monitoring the critically ill child with myocarditis in the critical care unit can be very challenging. The nurse must be able to recognize early clinical signs of deterioration. The most common assessment techniques used for children at risk for cardiovascular compromise or collapse include invasive hemodynamic monitoring, frequent careful clinical evaluation by experienced providers, and laboratory assessment of end-organ function, blood gases, and serum lactate, along with routine continuous noninvasive measures of vital signs, pulse oximetry and the use of near infrared spectroscopy (NIRS) to detect trends in oxygen delivery or consumption.
These critically ill patients are often treated with a myriad of agents to manage afterload, prevent arrhythmias, and support oxygenation, ventilation and nutrition and to optimize cardiac output. When the child is unresponsive to these management strategies, the timely use of ECMO and/or ventricular assist devices are additional modalities available as supportive therapy for myocarditis (see Chapter 7).
Potential novel therapies for future treatment of myocarditis include vaccinations, virus specific drugs, and protease inhibitors. Vaccination has been used successfully to prevent diseases.873 The efficacy of vaccines has led to the suggestion that a broadly specific enteroviral vaccine would be beneficial for reducing the incidence of myocarditis. The use of antiviral medications requires further investigation. In addition, because viruses release proteases that can lead to myocardial damage, protease inhibitors may be reasonable agents to consider for clinical investigation. Continued improvements in alternatives for myocardial support along with ongoing research into genetic markers that may identify patients at risk hold promise for the future.
Throughout the management of the child with myocarditis, the patient and the family will require emotional support and clear, concise, and consistent information. If the child is admitted with signs of cardiopulmonary compromise that require critical care support and monitoring, the possibility of mortality is real and heightens the importance of effective communication with the family.
The parents should be allowed to remain with the child as often as is feasible in hopes of reducing their anxiety and that of the child. It is only through careful coordination of care, a shared understanding of the pathophysiology and clinical issues, and thoughtful open communication between healthcare providers and the family that we can maximize the opportunities for these children to have the best possible outcome.
Cardiomyopathy
Gail L. Stendahl
Etiology
Cardiomyopathy is defined as a disease of the myocardium with cardiac dysfunction. It is usually associated with mechanical and/or electrical problems, and the patient may exhibit ventricular hypertrophy or dilation. Cardiomyopathies can be confined to the heart or may be related to a systemic disorder, often leading to death or a progressive heart failure-related disability.570
The term primary cardiomyopathy is used to indicate myocardial disease unrelated to congenital heart disease, pulmonary or systemic hypertension, or coronary artery or valvular heart disease. Cardiomyopathy also may develop as a secondary complication of systemic disease, viral infection, or exposure to chemicals or drugs. Some forms of congenital heart disease produce secondary ventricular dysfunction with effects similar to cardiomyopathy. For example, the child with transposition may develop severe right ventricular dysfunction after intraatrial (atrial—not arterial) correction. In most patients, however, the cause of cardiomyopathy is unknown.
Cardiomyopathy is uncommon, accounting for only 1% of all pediatric cardiac disease.524 Risk factors for the development of pediatric cardiomyopathy include the infant or adolescent age group, gender (3:2 male predominance), African-American race, and, most importantly, the presence of an affected family member. More than 50% of pediatric cardiomyopathy cases develop before 1 year of age. This early incidence may represent a genetic component. The incidence peaks again in adolescence. Lower socioeconomic status is associated with higher incidence of cardiomyopathy. These data suggest that genetic and environmental factors are interrelated and contribute to the risk of cardiomyopathy. The presence of a family member with cardiomyopathy increases the incidence for all first-degree relatives: 9% to 20% of all cardiomyopathies are inherited.212
Pathophysiology
Three forms of cardiomyopathy are commonly identified: (1) dilated cardiomyopathy, (2) hypertrophic cardiomyopathy, and (3) restrictive cardiomyopathy. A discussion of each of these follows. All forms of cardiomyopathy are associated with ventricular dysfunction, decreased ventricular ejection fraction, and an increase in myocardial mass.
Dilated Cardiomyopathy
Dilated cardiomyopathy (DCM) is the most common form of the cardiomyopathies; it accounts for at least half of all cases, with a population incidence of 0.58 per 100,000 children.189 Most cases of DCM are idiopathic in origin. Severe infections and overwhelming systemic inflammation may cause severe systolic dysfunction that may look like DCM. Myocarditis is a relatively common cause of DCM, accounting for 2% to 15% (diagnosed by endomyocardial biopsy) of DCM cases; the percentage is slightly higher in children less than 2 years of age.
There are multiple patterns of inheritance in familial DCM. Neuromuscular diseases may result from genetic mutations and may be autosomal or related to abnormalities of mitochondrial DNA (maternal inheritance). Duchenne's and Becker's muscular dystrophy (both X-linked disorders located in the dystrophin gene locus) have cardiomyopathy as a prominent feature. Barth syndrome, another X-linked disorder, is caused by a genetic mutation for the tafazzin gene. It is characterized by both cardiac and skeletal involvement and can present with DCM, with or without ventricular noncompaction.524
Regardless of the etiology, depressed cardiac function is the common feature in all forms of DCM. Some patients with cardiomyopathy, particularly those with restrictive or hypertrophic variants, may present initially with predominantly diastolic dysfunction. Systolic function may initially be preserved; however, the systolic function can deteriorate over time. Furthermore, the various forms of cardiomyopathy can have overlapping phenotypes (e.g., dilated cardiomyopathy with restrictive physiology).
Initially, cardiac output is often maintained despite decreased contractility by increased end-systolic function and end-diastolic volume creating increased wall tension. This increased tension stimulates myocyte hypertrophy and normalizes cardiac output. Over time, these compensatory changes result in unfavorable remodeling and progressive ventricular dilation with alteration in the normal ventricular geometry. As the function declines, the dilation and wall tension continue to increase, causing pooling of blood in the heart chambers. At this point, the elevated wall tension decreases myocardial efficiency and increases myocardial oxygen consumption. Eventually, these compensatory mechanisms are not sufficient to maintain adequate cardiac output. Additional stress, such as a febrile illness or exercise, may exacerbate symptoms.
Diminished cardiac output results in hypoperfusion of organs and may cause end-organ damage. Decreased renal blood flow activates the renin-angiotensin system to help maintain perfusion pressure by promoting fluid retention and vasoconstriction, and potentiating catecholamines. The sympathetic nervous system is stimulated, producing an increase in heart rate and contractility. At the expense of increased afterload, vasoconstriction maintains perfusion to vital organs. In short, the compensatory physiologic changes caused by the failing heart produce additional myocardial strain, ventricular dilation, and dysfunction.
The failing ventricle continues to dilate; the stretch on the cardiac myocytes distorts the conduction system, there is increased myocardial work and decreased cardiac output, and the patient is susceptible to arrhythmias, end organ dysfunction, and ischemia. Current medical therapies largely serve to neutralize this counterproductive physiology.
Microscopic examination of the cells shows hypertrophy of the myocytes. The progression of symptoms is dependent on the acuity of the decompensation.524
Hypertrophic Cardiomyopathy
Hypertrophic cardiomyopathy (HCM) is the second most common form of cardiomyopathy524; it accounts for 42% of childhood cardiomyopathy with an incidence of 0.47/100,000 children.189 HCM is one of the most common inherited cardiac disorders, with a prevalence in young adults of 1 in 500. Many children with this form of cardiomyopathy have first-degree relatives with similar heart disease, and sudden death is frequently reported in family members.
As is the case with dilated cardiomyopathy, metabolic and mitochondrial abnormalities may cause HCM.212 In addition, Wolff-Parkinson-White syndrome has been associated with HCM. Although this disease generally appears in the second or third decade of life, occasionally symptoms develop during childhood and may be progressive, with significant morbidity and mortality.524
The characteristic feature of HCM is the progressive and asymmetric thickening of the myocardium, especially in the area of the ventricular septum. The thickened myocardium invades the ventricular cavity, dramatically decreasing the cavity size. Left ventricular outflow tract obstruction is often present and may be associated with systolic anterior motion (SAM) of the mitral valve with secondary mitral insufficiency.
Intracellular calcium is known to regulate contractile function and relaxation. As the cardiac myocytes begin to hypertrophy, there is an increase in the release of intracellular calcium during diastole, which leads to diastolic dysfunction. Histologic examination reveals myocyte hypertrophy, myofiber disarray, and patchy fibrosis.872
Hypertrophic cardiomyopathy usually produces a decrease in ventricular compliance and ejection fraction, and it can result in the development of mitral regurgitation, arrhythmias, congestive heart failure, and/or low cardiac output.
Restrictive Cardiomyopathy
Restrictive cardiomyopathy is the least common form of cardiomyopathy; among children it is quite rare, accounting for fewer than 3% of pediatric cardiomyopathy cases.189 Restrictive cardiomyopathy presents with normal or near-normal systolic function but impaired ventricular filling resulting from increased stiffness and reduced diastolic volume of either or both ventricles.189 Primary types of restrictive cardiomyopathy include endomyocardial fibrosis, hypereosinophilic syndrome, familial, and most commonly idiopathic cardiomyopathy. Secondary types are related to multisystem disorders, including infiltrative and storage disease.
Pediatric cases are generally idiopathic or related to cardiotoxicity or endocardial fibroelastosis (EFE), with up to one third occurring in a familial pattern. Differentiation from many of the secondary causes relies on morphologic criteria. Tissue analysis is often pursued given the dismal prognosis of the disease and the desire to exclude any potentially treatable disorder.189
The characteristic feature of restrictive cardiomyopathy is a marked increase in the stiffness of the myocardium or endomyocardium, resulting in a reduced ventricular size. The consequence is impaired ventricular filling, leading to elevated diastolic pressures and marked atrial dilation. Pulmonary hypertension secondary to left atrial hypertension is present from the early stages of this disease.
If endomyocardial fibrosis is present, it may be noted as a distinct disease or in association with bacterial endocarditis or eosinophilic leukemia. As endocardial or myocardial lesions progress, ventricular expansion during diastole is restricted so ventricular end-diastolic pressure increases and stoke volume declines.231 Eventually the ventricular lumen may be obstructed by fibrotic tissue and thrombus formation. These ventricular thrombi can embolize to the pulmonary or systemic circulations.
Clinical Signs and Symptoms
Children with cardiomyopathy may be asymptomatic or show signs of severe congestive heart failure (CHF). The rate of symptom progression varies and depends on the nature of the disease and the body's ability to increase cardiac output to meet the demands of the body. As ventricular dysfunction worsens, the child shows symptoms of left- and/or right-heart failure.
The findings of CHF vary with the age of the patient. Infants usually present with feeding intolerance. Symptoms include tachypnea, increased respiratory effort with retractions, diaphoresis during feeding, prolonged periods of irritability, and failure to thrive. Children and adolescents initially present with exercise intolerance or dyspnea with exertion, fatigue, orthopnea, tachypnea, dyspnea, and edema. Sympathetic nervous system redistribution of blood flow away from skin, gut, and kidney can lead to gastrointestinal symptoms, including abdominal pain after meals, nausea, vomiting, and anorexia. Additional symptoms may include new onset palpitations and syncope. Occasionally, patients are asymptomatic except when arrhythmias are present; sudden cardiac arrest (sudden death) may be the first sign of the disease.
Vital signs reveal an elevated heart rate (for age) with decreased heart rate variability. Tachycardia initially compensates for the decreased stroke volume. Pulses may be weak with a narrow pulse pressure, as systemic vascular resistance increases and cardiac output decreases. The blood pressure is low to normal and may be maintained until just before cardiovascular collapse. The respiratory rate is elevated. Cyanosis is uncommon but pallor or mottling may be present.212 The patient may also be febrile as the result of an acute infectious process that may be exacerbating symptoms that otherwise would be subtle.671
Cardiac examination may reveal cardiomegaly, increased precordial impulse or lateral displacement of the apical impulse. The heart sounds may be distant or muffled. Although murmurs are not always present, a systolic murmur often is noted along the left sternal border. The murmur may be caused by progressive left ventricular outflow tract obstruction (in the child with hypertrophic cardiomyopathy) or by mitral regurgitation. An extra heart sound, secondary to elevated ventricular filling pressure, may be auscultated as a gallop rhythm. Jugular venous distension may be present, but is difficult to appreciate in young children.671
Abdominal examination can reveal hepatomegaly and ascites. Nonspecific generalized abdominal pain also may be present, resulting from mesenteric ischemia. Dependent edema is common and can be seen in the eyelids and the scrotum of infants or in the legs of children and adolescents.212 Vasoconstriction can cause extremities to be cool and poorly perfused. Capillary refill time is increased.
The chest radiograph classically reveals cardiomegaly, typically from left atrial and left ventricular enlargement. The left atrial enlargement may elevate the left main stem bronchus and cause airway obstruction or left lung atelectasis. In younger children, lung fields often appear hyperexpanded with flattening of the diaphragm. Elevated pulmonary venous pressure leads to pulmonary congestion. Pleural effusions may also be present.212
The echocardiogram demonstrates ventricular dilation, disproportionate ventricular septal thickening, or possible obstruction of the left and/or right ventricular outflow tracts. Echocardiographic findings show globally decreased contractility without regional wall motion abnormalities. Color and pulse-wave Doppler can determine the presence and degree of valve regurgitation, assess cardiac output by aortic flow velocities, determine the degree of diastolic dysfunction by atrioventricular (AV) valve inflow pattern, and estimate pulmonary artery and right ventricular pressures. Echocardiography is beneficial for monitoring patients longitudinally as well as for detecting changes in ventricular function, thrombus formation, wall stress, pulmonary vascular disease, and valvular regurgitation.212
The electrocardiogram (ECG) is usually abnormal, although no specific criteria are diagnostic for cardiomyopathy. Usually, evidence of left (and possible right) ventricular hypertrophy, arrhythmias and ST segment changes consistent with myocardial injury may be noted (see section, Common Clinical Conditions, Arrhythmias, ST-Segment Deviation), T-wave inversion, abnormal Q waves, and diminished R waves also may be present on lateral precordial leads. Evidence of atrial hypertrophy may be present. A Holter monitor allows for determination of chronotropic (heart rate) variability, identification of ST segment changes, and assessment of T-wave alternans.
Acute myocardial injury or inflammation can be detected with serum biomarkers, such as cardiac troponins I and T. These biomarkers are intracellular proteins that are sensitive and specific for acute myocyte injury. Elevation of troponin should alert clinicians to look for ischemia, myocarditis, or an acute inflammatory process as the cause of the cardiomyopathy. Patients with infectious causes for cardiomyopathy may have an elevated white blood cell count with a lymphocytosis, and trends in inflammatory markers (e.g., C-reactive protein [CRP] and erythrocyte sedimentation rate [ESR]) should be monitored. Providers must closely monitor end-organ function. Blood and nasopharyngeal cultures for viruses are usually not diagnostic.
In patients presenting with CHF, progression of heart failure can be monitored with B-type natriuretic peptide levels. These hormones are secreted from the ventricles in response to dilation and wall stress. Monitoring of BNP levels may be useful in managing cardiomyopathy in children. Depending on the age and presentation of the cardiomyopathy, a metabolic evaluation with blood and urine analysis should be considered. In addition, molecular testing is available to screen for genetic defects that led to cardiomyopathy.212
Cardiac catheterization is performed to evaluate left ventricular filling, pulmonary capillary wedge pressure, and pulmonary artery and central venous pressures; these measurements and evaluation can help guide therapy. Endomyocardial biopsy remains the gold standard to exclude myocarditis and may be useful in the diagnosis of metabolic abnormalities, mitochondrial defects, and infiltrative disease. Biopsy results are often nonspecific, showing myocyte hypertrophy and fibrosis. Polymerase chain reaction (PCR) analysis and electron microscopy may provide a more definitive diagnosis that can affect prognosis and treatment.212
Cardiac magnetic resonance imaging (MRI) is a useful tool for the measurement of myocardial mass and thickness, particularly when echocardiography is inadequate. Three-dimensional reconstruction of data enables quantification of the disorder. Recent work using delayed enhancement after gadolinium administration has demonstrated the capacity to identify areas of myocardial fibrosis, a finding with prognostic importance.189
Management
Management of a child diagnosed with cardiomyopathy should begin by ruling out treatable causes, minimizing the risks of complications from the cardiomyopathy and providing supportive care for the heart failure symptoms.212 The clinical picture of a child with cardiomyopathy includes low cardiac output, fluid retention and peripheral vasoconstriction. Therapy is aimed at increasing cardiac output, enhancing tissue oxygen delivery, and sustaining vital organ function. If a treatable metabolic abnormality (e.g., carnitine deficiency) is present, appropriate treatment should be started without delay. Confirmatory diagnostic testing from urine and plasma sampling can still be performed with concurrent treatment of the presumed deficiency.
The short-term management of patients with heart failure consists of supportive care. Acute symptoms of CHF may be improved with administration of inotropes, such as dopamine or epinephrine, but these strategies are rarely useful as longer-term therapies. At low doses, dopamine increases renal blood flow. At higher doses, it increases cardiac output but can also increase peripheral vascular resistance and cause arrhythmias.671 Therefore, inotropes should be used judiciously and may serve as a bridge to alternative therapies such as mechanical support, biventricular pacing, or cardiac transplantation. Incremental titration should be used as the clinical situation dictates. Milrinone, an inodilator (inotrope and vasodilator), may decrease left ventricular work by promoting relaxation and increasing ventricular compliance.
Diuresis is essential to control signs and symptoms of CHF. Intravenous diuretic therapy is used during acute decompensation and can be transitioned to oral therapy once the systemic or venous congestion improves. It is critical to monitor electrolytes during initial or escalation of diuretic therapy because electrolyte imbalances can lead to life-threatening arrhythmias.524
When the acute decompensation has been controlled, patients are often converted to an oral regimen of afterload reduction with angiotensin-converting enzyme (ACE) inhibitors such as enalapril or captopril, diuretic therapy with spironolactone, and a loop diuretic such as furosemide or hydrochlorothiazide, and a beta-receptor antagonist, usually carvedilol or metoprolol.524
Beta-adrenergic agents may be effective in the treatment of both dilated and hypertrophic cardiomyopathy in adults, but their utility in pediatrics remains controversial and unproven. Approximately one third of symptomatic adults improve after administration of a beta-blocker. Although the predominant mechanism by which improvement occurs is unclear, it is thought that carvedilol reduces myocardial oxygen requirements and may decrease apoptosis (programmed cell death).671 Carvedilol does not usually produce immediate improvement in the short term and has not been shown to reduce the risk of sudden fatal arrhythmias among patients with cardiomyopathy.
Calcium channel blockers, such as amlodipine, have also been effective in the treatment of hypertrophic cardiomyopathy, although experience in children is limited. Sudden death is a risk factor for children receiving calcium channel blockers, so they require close monitoring with initiation of this therapy.
Digoxin should be used with caution in acutely ill children. Use of digoxin in patients with inflamed myocardium may promote ventricular arrhythmias. Digoxin is not recommended for children with hypertrophic cardiomyopathy and adequate ventricular systolic function because digoxin may worsen the left ventricular outflow tract obstruction. Digoxin, however, may be beneficial in low doses for symptomatic patients with dilated cardiomyopathy.671
If the child has developed intraventricular or intraatrial thrombi, anticoagulant therapy will be initiated. Careful monitoring of systemic perfusion and pulmonary function is required to detect evidence of systemic or pulmonary emboli. Thrombus formation can occur when there is stasis of blood in enlarged chambers of the heart. If a clot is detected, it should be aggressively treated with heparin and eventually switched to warfarin or Lovenox. Patients with global ventricular dilation and dysfunction may benefit from low dose aspirin as an antiplatelet agent or prophylactic anticoagulation with Coumadin even in the absence of identified thrombus formation.212
Prevention or treatment of secondary arrhythmias (particularly malignant ones) can be challenging because antiarrhythmic drugs often produce myocardial depression. Despite this, many patients with chronic arrhythmias benefit from antiarrhythmic therapy. Depending on the nature of the arrhythmia, treatment may include beta-blockade, amiodarone, or class 1 agents such as lidocaine or mexiletine. Antiarrhythmic drugs and/or radiofrequency ablation are used when the diagnosis of tachycardia-induced cardiomyopathy is established.671 The therapy of choice depends on the nature of the tachycardia, but establishing sinus rhythm with rate control is imperative and usually leads to marked improvement in ventricular function. Many effective antiarrhythmics, such as procainamide, have negative inotropic effects, may not produce rapid cardioversion, and should be used with caution in this group. Ultimately, these patients should often be considered for ablative therapy as a potential cure (see section, Common Clinical Conditions, Arrhythmias).
Cardiac resynchronization therapy has been used in adult patients with left bundle branch block and decreased ventricular function. This therapy has been shown to be helpful in improving symptoms and decreasing hospitalizations. Experience with this therapy is limited in pediatric patients, although early reports suggest that this is a promising therapy for selected patients. Further studies are needed in the pediatric population.671
Children that are not responsive to medical management for CHF may benefit from surgical palliative therapies before heart transplantation. Surgical palliation with bridging techniques, such as ventricular assist devices, intraaortic balloon pumps, and extracorporeal membrane oxygenation, have allowed some patients who normally would not have survived to live long enough to receive a heart transplant (see Chapter 7).212
Cardiac transplantation should be considered if short-term survival is unlikely or when severe symptoms are unresponsive to conventional therapy. Patients with heart failure symptoms who cannot be weaned from intravenous inotropic support fall into this category. In addition, patients managed on diuretics, angiotensin-converting enzyme inhibitors, and beta-blockers who have persistent New York Heart Association class III or IV heart failure symptoms, ongoing failure to thrive, or severely depressed ventricular function after 6 months of appropriate therapy should be considered candidates for cardiac transplantation (see Chapter 17).679
The natural history of cardiomyopathy is affected by the cause (primary versus secondary) and any coexisting risk factors. Primary cardiomyopathies are usually progressive, despite aggressive anticongestive medical therapy, whereas secondary causes may or may not be progressive. Although many children with secondary cardiomyopathy recover ventricular function, children with marked myocardial injury in early childhood should be followed closely.212
The prognosis of children with cardiomyopathy is dependent on the cause of the disease and the age at presentation. Traditionally, approximately one third of children with cardiomyopathy were expected to recover completely, another one third were expected to improve but with some residual dysfunction, and the remaining one third were predicted to die or require transplantation. However, these results have improved in recent years. Sudden death may occur at any stage regardless of ventricular function and may be precipitated by comorbidities, such as progressive ventricular dysfunction, CHF, arrhythmias, conduction disturbances, and/or thromboembolic events.212
Children with ventricular dysfunction benefit from reducing modifiable cardiovascular risk factors, such as hypertension, diabetes, hypercholesterolemia, and obesity. Secondary prevention strategies can limit the ongoing myocardial injury after the initial insult. Children should be encouraged to eat a well-balanced diet and participate in a regular physical activity or a cardiac rehabilitation program whenever possible. In addition, routine screening for some high-risk groups is recommended.212
Cardiac Tumors
Gwen Paxson Fosse
Etiology
The incidence of primary cardiac tumors is reported to be 0.027% to 0.32% in children.199,576 Primary myocardial, intracardiac, or epicardial masses/tumors are rare but do occur in infants and children, and are most commonly benign. However, they may produce hemodynamic compromise, hemolysis, or embolic phenomena, so they must be identified and treated. The incidence of benign primary cardiac tumors in children is increasing, but these tumors are also more frequently detected and diagnosed, particularly with echocardiographic techniques.574 Primary cardiac malignant tumors represent less than 10% of the primary cardiac tumors in children.199,576
Pathophysiology
Cardiac tumors in pediatrics may cause no significant pathophysiology, but they may have very significant effects depending on the type, number, location, and size of the tumor(s). Right heart tumors can cause right heart failure and left heart tumors can cause or mimic mitral or aortic valve dysfunction. Tumors can result in cardiac inflow and/or outflow obstruction, they may compress vessels and chambers, interfere with valve function, cause coronary artery changes with potential ischemia, infiltrate the myocardium, and cause arrhythmias, embolization, and sudden death.199,574 There are both benign and malignant cardiac tumors in children (described in the following).
Benign Masses and Tumors
Benign masses and tumors include rhabdomyomas, fibromas, myxomas, teratomas, hemangiomas, angiomas, hamartomas, thrombi, and other uncommon tumors. These are presented briefly in the following.
Rhabdomyoma
The rhabdomyoma is the most common cardiac tumor seen in children, and is typically diagnosed in infants less than 1 year of age. These benign tumors, composed of cardiac myocytes, often (in 78% to 90% of patients) occur as a result of tuberous sclerosis (an autosomal dominant disorder involving tumors of many organ systems and developmental delay) and may be diagnosed prenatally.574 The rhabdomyoma is usually multiple and is located within the walls of the ventricles, particularly within the septum. Cardiovascular effects occur when the tumor obstructs a coronary artery or a valve or the outflow tract of a ventricle. Arrhythmias also may develop.
These tumors can be removed successfully, resulting in resolution of all symptoms (including all arrhythmias). In more than half of the cases these tumors spontaneously regress or completely disappear.574
Fibroma
A fibroma is the second most common cardiac tumor seen during childhood. It, too, is benign, and is diagnosed most frequently during the first year of life. This tumor is firmer than a rhabdomyoma, and is usually a solitary tumor that compresses surrounding structures as it grows. These tumors appear to invade the myocardium and can cause inflow and/or outflow tract obstruction, distortion and changes to atrioventricular valve apparatus, and coronary artery changes.574 Arrhythmias are reported in approximately one third of affected patients, and sudden death may occur.
In approximately half of involved patients the fibroma cannot be excised completely, although it can be debulked. Long-term survival after complete excision is excellent, but must be more guarded after partial resection.
Myxomas
Myxomas are common in adults but rare in the pediatric population. They most often are found in the left atrium but can occur in other chambers. There is typically a stalk that connects the myxoma, which allows it to be mobile, so embolization can occur. Some myxomas are a familial form and they can be associated with Carney complex, an autosomal dominant disorder.574
Teratomas
Teratomas are usually single and intrapericardial but in rare cases have been reported to involve cardiac chambers. They are composed of embryonic germinal layers and may be diagnosed in the fetus. They are often large and cystic, causing compression of vessels and chambers and pericardial effusion. Surgical removal is typically possible without recurrence. On rare occasions teratomas may be malignant.574
Hemangiomas/angiomas
Hemangiomas/angiomas consist of large blood vessels and vascular channels involving the myocardium. They are usually single. If symptoms develop, surgical removal may be warranted. Many hemangiomas/angiomas have been successfully treated with steroids or interferon resulting in regression.574
Hamartomas
Hamartomas typically involve the left ventricle and present early with ventricular tachycardia necessitating electrophysiologic studies and surgery with inspection and excision of the tumor (typically very small) and cryoablation of the margins. The outcome using this approach is reported to be good.199
Thrombi
Thrombi have increased in incidence and there is also increased detection through the use of improved imaging techniques including echocardiography. The growth in the population of critically ill infants and children has resulted in conditions amenable to the formation of thromboemboli. The use of indwelling catheters also has added to this phenomenon. Survival of children with single ventricle physiology, Fontan operations, cardiomyopathies, and those who have had Kawasaki disease contribute to the population at risk for thrombi.574 With an intracardiac mass in the presence of an indwelling catheter, a blood culture can be obtained from the site to determine if the mass is infected or is an endocarditis vegetation.401
Other Masses
Other tumors including lipomas, papillary tumors, accessory endocardial cushion tissue, and fibroelastoma can be associated with pathophysiology similar to that mentioned for other tumors.576
Malignant Cardiac Tumors
Malignant cardiac tumors are extremely rare in children.574 The most common malignant types found in pediatric patients are rhabdomyosarcomas, angiosarcomas, fibrosarcomas, lymphosarcoma, giant cell sarcoma, fibromyxosarcoma, neurogenic sarcoma, leiomyosarcomas, and undifferentiated sarcoma.199,576 They are associated with right heart chamber involvement, local invasion of the heart, and hemorrhagic pericardial effusions.199 Stroke and myocardial infarction have been reported complications of pediatric cardiac tumors.636 Metastases to lung, brain, and liver are not unusual.
Without treatment survival is typically less than 1 year. With surgery and chemotherapy reported survival has improved.199
Secondary Cardiac Tumors
Secondary cardiac tumors, aggressive local tumors, or metastatic malignancies are more common than primary cardiac neoplasms in children and can cause the same pathophysiology as that seen with primary malignant tumors. Lymphomas, leukemias, neuroblastomas, and extracardiac sarcomas are associated with secondary cardiac involvement in children.199,576 Orthopnea and abnormal echocardiography should raise the suspicion of cardiac infiltration, which might not otherwise be appreciated promptly enough for meaningful intervention.786 Renal and hepatic malignancies, including Wilms tumors, may have direct extension of tumor into the inferior vena cava and right atrium requiring chemotherapy with possible surgery.199,576 Life-threatening embolic phenomena have been reported with extracardiac malignancies resulting in myocardial infarction and pulmonary emboli.28,423
Clinical Signs and Symptoms
The size, location and number of tumors have a major effect on presenting symptoms. Some tumors are small and asymptomatic and may remain undetected. The tumor location may be indicative of tumor type and composition. Diagnosis may be made in the fetus, neonate, child, and adolescent.
Signs and symptoms may be related to myocardial dysfunction and obstruction of blood flow. Clinical features may resemble congenital heart defects with congestive heart failure and include low cardiac output, murmurs, cyanosis, and arrhythmias. Additional findings in the child and adolescent may include fever, malaise, weight loss, arthralgias, myalgias, emboli, exercise intolerance, chest pain, and syncope.199,574
Electrocardiography will certainly reveal arrhythmias, but also may be suggestive of chamber enlargement and myocardial ischemia.574 Ventricular preexcitation or supraventricular tachycardia can be presenting symptoms in infants with some cardiac tumors.607
The chest radiograph may show the contours of a mass as well as areas of calcification if they are present in the tumor. Echocardiography can be invaluable in evaluating the hemodynamics and deciphering details about the tumor to aid in diagnosis of the tumor type. Other imaging modalities, including magnetic resonance imaging, computed tomography, and cardiac catheterization, can provide additional information.199,574
Management
Diagnosis of the type of tumor and knowledge of the specific characteristics can be paramount in determining the management approach. Medical management with serial surveillance may be the only recommendation.
Management of thrombi can be very challenging. For a thrombus mass, antibiotic therapy may be employed if infection is present. Assessment for a hypercoagulable state or collagen vascular disease may be helpful in determining etiology and management of a thrombus. Thrombolysis may be warranted and is most effective with freshly clotted material, but the risk of emboli must be considered. Intracardiac thrombi can pose a risk of emboli or obstruction to flow, in which case surgical resection may be recommended. Thrombi in a child with underlying cardiac anomalies, foreign surfaces, stagnant areas of flow, or abnormal flow through a baffle or tunnel may warrant surgical intervention.401 Decision-making must involve consideration of the risk/benefit ratio for both thrombolysis and surgical intervention.199,574
In cases of hemodynamic compromise with the presenting symptoms of a tumor, medical interventions to stabilize the child must be the first priority. In newborns this may involve the use of prostaglandin E1 (PGE1) to maintain ductal patency (initial dose: 0.05-0.1 mcg/kg per minute, refer to Box 8-33).576
Surgical resection can be successful and life-saving for tumors such as myxomas, teratomas, and others that are causing embolic phenomena, hemodynamic compromise, or arrhythmias. Surgical management may or may not require cardiopulmonary bypass. The approach depends on the location of the tumor(s), but if at all possible, incisions are made in the atria or great vessels, avoiding ventriculotomy. Ideally, complete resection occurs, but in some cases the involvement of critical structures may prevent total resection.
If complete resection is not possible the child will require serial evaluation to detect changes in the tumor. In rare cases cardiac transplantation has been performed for massive fibromas. Radiofrequency or other ablation techniques may be employed successfully to treat tumor-associated arrhythmias.199,574
Malignant tumors, both primary and secondary, raise issues of both a medical and surgical nature. The cardiology, cardiovascular surgery, and hematology/oncology teams must work closely together to determine the treatment plan. Surgical cavectomy has been reported with success for children with Wilms tumor and intravascular thrombus into the right atrium.733 Individualized treatment plans are necessary to address the specific medical needs in children with primary cardiac malignant tumors to avoid potential complications.636
Outcomes for those with benign tumors are usually quite good. Those with underlying congenital heart disease, systemic conditions, or malignancy have varied outcomes depending on the nature of their associated disorders.
Kawasaki Disease (Mucocutaneous Lymph Node Syndrome)
Caroline A. Rich
Etiology and Epidemiology
Kawasaki disease (KD) is an immune-mediated, multisystem vasculitis of infancy and early childhood with self-limited clinical course and unknown etiology. It was formerly called mucocutaneous lymph node syndrome.
Most cases of KD occur in children between 1 and 8 years of age with 85% of children affected under the age of 5 years. The disease is more common in the Japanese-American population. The male to female ratio is 1.5:1.684,838
Pathophysiology and Clinical Signs and Symptoms
Kawasaki disease is characterized by systemic inflammation manifested by fever for at least 5 days (usually greater than 39° C and remittent) and four of five clinical criteria (Box 8-50).684,838
Box 8-50 Clinical Criteria for Kawasaki Disease684,838
• Bilateral bulbar conjunctival infection without exudate
• Oral mucous membrane changes, including erythema of mouth and pharynx, strawberry tongue, and red cracked lips
• Erythema and edema of palms of hands and soles of feet, and periungual desquamation (after acute phase of illness)
• Polymorphous rash
• Cervical lymphadenopathy (at least one lymph node >1.5 cm in diameter)
Supporting Laboratory Findings in Kawasaki Disease
• Albumin less than 3 g/dL
• Anemia for age
• Elevated alanine aminotransferase
• Platelet count greater than 450,000/mm3 after 7 days
• White blood cell count greater than 15,000/mm3
• Elevated C-Reactive Protein
• Erythrocyte sedimentation rate greater than 40 mm/h
• Sterile pyuria
Other associated features include irritability, abdominal pain, diarrhea, vomiting, elevated platelet count (by second week of illness), C-reactive protein and erythrocyte sedimentation rate, hyponatremia, and leukocytosis with bandemia (high percentage of band forms). Other findings include urethritis with sterile pyuria, anterior uveitis, mild hepatic dysfunction, arthralgia, aseptic meningitis, pericardial effusion, gallbladder hydrops, and myocarditis manifested by congestive heart failure (CHF).
There is no diagnostic test available to confirm KD. The diagnosis is established by fulfillment of the clinical criteria and exclusion of other possible illnesses, such as measles, parvovirus B19 infection, adenovirus, enterovirus, scarlet fever, drug reactions (Stevens-Johnson syndrome), staphylococcal scalded skin syndrome, toxic shock syndrome, and juvenile rheumatoid arthritis.684,838
Children with fever and two or three of the typical clinical criteria for 5 days or more, with three or more supporting laboratory findings are said to have incomplete KD. These children should have an echocardiogram and receive treatment. If there are less than three supporting laboratory findings, an echocardiogram is suggested; treatment is warranted if the echocardiogram is positive for coronary artery aneurysms or dilation.
The major complication of KD, especially in infants younger than 1 year of age, is coronary artery aneurysms. These aneurysms occur in 20% to 25% of untreated children with KD, but develop in only 4% of those who receive adequate therapy. Other cardiac sequelae may include myocarditis, decreased myocardial contractility, coronary arteritis without aneurysms, mild mitral valvular regurgitation, and pericardial effusion.684,838
Clinical findings at presentation that have been associated with an increased incidence of coronary artery aneurysms include treatment delay beyond 10 days; age less than 1 year or greater than 8 years; male gender; fever greater than 14 days; or fever persisting after IVIG administration.684,838
Coronary artery aneurysms have been visualized with echocardiogram as soon as a few days after onset of illness, but more typically occur between 1 and 4 weeks after onset of illness; later than 6 weeks is uncommon. Giant coronary artery aneurysms (greater than 8 mm in diameter) are likely to be associated with long-term complications, such as myocardial infarction.
Acute myocardial infarction (AMI) may develop during the acute phase of illness. This diagnosis can be difficult to make in children because it is so rare. The infarction often occurs at night during sleep or rest and is accompanied by pallor, inconsolable crying, abdominal pain, and vomiting.
Management
Management of KD during the acute phase is directed at decreasing inflammation of the myocardium and coronary artery wall. Once the acute phase has passed, therapy is directed at prevention of coronary artery thrombosis.
Intravenous immune globulin (IVIG) and aspirin initiated within 10 days of the onset of the fever substantially decreases progression to coronary artery dilation and aneurysms.20 The recommended IVIG dose is 2 g/kg as a single dose given over 10 to 12 hours. Aspirin is administered in doses of 80 to 100 mg/kg per day in four divided doses during the acute phase. The dose of aspirin is decreased to 3 mg/kg per day, 48 hours after the resolution of fever. Aspirin is discontinued if no coronary artery abnormalities have been detected by 6 to 8 weeks after the onset of illness.684,838
An echocardiogram should be obtained early in the acute phase of illness and 6 to 8 weeks after onset. Children diagnosed with KD should be assessed during the first 2 months for arrhythmias, CHF, and valvular regurgitation.
Approximately 10% of patients with KD fail to defervesce with initial IVIG treatment. Failure to respond is usually defined as persistent or recrudescent fever more than 36 h after completion of initial IVIG infusion. Retreatment with IVIG (dose: 2 g/kg) is recommended.918 Although corticosteroids are the treatment of choice in other forms of vasculitis, their use has been limited in children with KD.805
Early detection of KD and prompt initiation of therapy with IVIG and aspirin have reduced mortality to less than 0.1% to 0.2% and the prevalence of coronary artery aneurysm to approximately 5%. The principal cause of death is AMI resulting from coronary artery occlusion attributable to thrombosis or progressive stenosis.684,838
Measles and varicella immunizations should be deferred 11 months after IVIG administration.
Echocardiography
Description
Sonography has evolved to become one of the most versatile modalities for diagnosing and guiding treatment in critically ill children. It consists of both cardiac (echocardiography) and noncardiac (head, lung, abdominal, and vascular) ultrasound. Echocardiographic examination provides extensive noninvasive anatomic and hemodynamic information in real time. This painless procedure involves the transmission of high-frequency, pulsed-sound waves from piezoelectric crystals located within the ultrasound transducer. The pulse-sound waves are transmitted into the chest and then are reflected back, received, and recorded by the same transducer.
All tissues in the body impede or absorb high-frequency sound waves in a different way, so that the sound waves reflected back to the receiver are of differing strengths. The distance of a reflecting surface from the transducer is calculated on the basis of the time it takes the energy to reach the structure and return to the transducer. This information then determines the location of dots representing that structure on a display screen. This process is repeated approximately 1000 times/s (1000 Hz) to create an image.259,312,675,808,819
Echocardiography can be a rapid, noninvasive, objective tool in the assessment of ventricular function and preload during resuscitation of a critically ill child. Echocardiography can also be used to evaluate the dimensions of the cardiac chambers and great vessels, the location and motion of cardiac valves, and the size and location of septal and other defects. In addition, bedside echocardiography can be used to differentiate fine ventricular fibrillation from true asystole during resuscitation.105,106,710 Box 8-51 summarizes some of the common indications for echocardiography in critically ill children.
Box 8-51 Common Indications for Echocardiography in Critically Ill Children
Assessment of hemodynamics
Assess myocardial contractility and regional wall abnormalities
Assess volume status
Assess noninvasive cardiac output
Estimate filling pressures and predict volume responsiveness
Assess right heart function
Detect pericardial effusion/tamponade
Detect pulmonary embolus
Diagnose diastolic ventricular dysfunction
Resuscitation
Differentiate fine ventricular fibrillation from true asystole
Assess cardiac standstill for prognostication purposes
Perform focused evaluation in resuscitation (FEER) to look for potentially reversible causes of PEA or asystole, such as pericardial effusion/tamponade, hypovolemia, pulmonary embolism, or tension pneumothorax
Other Diagnostic Uses
Congenital heart disease
Primary or secondary cardiomyopathy
Primary pulmonary hypertension
Cerebrovascular infarct (embolic)
Rhythm and conduction abnormalities
Cardiac and pericardiac tumors
Arteriovenous malformations
Rheumatic heart disease
Kawasaki disease
Chest pain
Syncope
Suspected endocarditis
Deep vein thrombosis
Procedural Guidance in PCCU
Balloon atrial septostomy
Pericardiocentesis
Line displacement or dislodgement
The ability to assess myocardial function, volume status, and cardiac output noninvasively is very useful in the management of complicated or mixed shock states.166,716,779 Although the culture is changing, echocardiography still remains a forte of cardiologists in the majority of tertiary care centers in United States.
Three forms of echocardiography currently are used, namely M-mode (or motion-mode) echocardiography, 2D echocardiography, and 3D echocardiography.259,675 M-mode (or motion-mode) echocardiography uses a single crystal to obtain an image of the heart at one point. The only axis that is evaluated is depth, so only structures located directly under the transducer are visualized. Spatial orientation of objects can only be achieved by moving or angling the transducer. M-mode is now used primarily for evaluation of left ventricular dimensions and function; it has largely been replaced with 2D echo imaging. In rare cases, 2D-directed M-mode may be useful for assessing certain structures, such as native and prosthetic valve leaflets.
Two-dimensional or cross-sectional echocardiography uses many crystals to pass a planar beam of ultrasound through the heart to obtain an image of a cardiac plane (Fig. 8-65, A). The ultrasound beam is swept rapidly in an arc, creating multiple M-mode images, which are then aligned to create a cross-sectional image of the heart. Because this form of echocardiography can provide a spatially correct image of the heart and great vessels, it is more useful for the determination of anatomic relationships of structures within the heart. The three basic elements of any 2D echocardiogram include imaging sweeps for anatomic delineation, Doppler interrogation for blood flow velocity and direction, and measurement of systolic and diastolic function.
Three-dimensional echocardiography is one of the significant advances in this field because it provides realistic visualization of cardiac valves and congenital abnormalities as well as accurate assessment of cardiac chamber volumes (Fig. 8-65, B). Real-time 3D echocardiography allows fast acquisition of pyramidal datasets during a single breath-hold without the need for off-line reconstruction, thus eliminating motion artifacts known to have adversely affected the earlier multiplane acquisition and reconstruction methodology.715,801
Color Doppler echocardiography yields color-coded Doppler signals overlaid on an echocardiographic image of the heart. With such Doppler studies, blood flow of differing speeds (e.g., shunting blood) is identified by different colors. Directionality of blood flow is determined by whether the frequency shift is higher (i.e., the flow is toward the probe) or lower (the flow is away from the probe). Various color maps are used to encode this information, but a commonly used map displays blood flow toward the transducer in red and flow away in blue (see Evolve Fig. 8-7 in the Chapter 8 Supplement on the Evolve Website). The use of color enables rapid and obvious visualization of small or multiple shunts or small amounts of valvular regurgitation and may enable identification of small vascular pathways (e.g., sources of pulmonary blood flow in patients with pulmonary atresia) that are not detected by standard echocardiography. In addition, Doppler derived calculations provide quantitative and semiquantitative estimation of valvular regurgitation, intracardiac and extracardiac shunts, and myocardial motion.324
Transesophageal echocardiography (TEE) is another modality of echocardiography, most frequently used in the operating room or during the perioperative period, where the acoustic windows are limited by dressings over the incision (Fig. 8-66).690 The transesophageal approach does not compromise the operative field and images are not obscured by chest tubes and dressings as with transthoracic imaging. Occasionally the image quality with transesophageal echocardiography can be compromised by adjacent nasogastric tubes.
TEE offers high-quality imaging of patients who otherwise pose a challenge to the echocardiographer. Structures that are particularly well suited to TEE examination are those located closest to the esophagus, including the atrial septum, pulmonary veins, superior vena cava, AV valves, left ventricular outflow tract, aortic valve, and coronary artery origins. With the advancement in technology, transesophageal probes are available for patients as small as 2 to 2.5 kg.
Procedure
Echocardiography can be performed at the bedside or in the echocardiography laboratory. It does not require any specific preparation. The infant or child is placed in a reclining or semireclining position at the start of the procedure and it may be necessary to turn the patient occasionally during the procedure.
A single, flat-tipped transducer that is approximately 6 inches in length is used to obtain the surface echocardiogram; this transducer sends and receives the sound waves. To minimize artifact and maximize sound transmission, small amounts of electrocardiographic gel or paste are applied to the child's chest, where the transducer is placed. During the procedure the infant or child merely feels the touch of the transducer and gel. TEE is obtained through a transducer inserted into the esophagus.
Because the images produced during the echocardiogram will be blurred by motion artifact, it is very important that the infant and/or child be motionless during the procedure. Usually the infant and/or child will be quieted by the use of a pacifier or feeding with a bottle. Parent presence, the use of pacifiers or favorite objects, a quiet environment, and the therapeutic use of music or videos can all be provided in a hospital environment. If all attempts at holding and quieting fail, the physician may elect to prescribe a mild sedative for the infant. Oral chloral hydrate provides deep sedation usually lasting 60 to 120 min, which should be adequate for a complete study in a child with complex congenital heart disease.542,644 Intravenous midazolam or narcotics may be preferable in some situations, particularly in critical care settings. Institutional guidelines for procedural sedation should be followed, and qualified medical personnel not involved in performing the echocardiography should be responsible for monitoring the child's cardiorespiratory status.19
Potential Complications
Although a relatively safe procedure, it has been our experience that a child with marginal hemodynamic status has the potential to decompensate during echocardiography. Therefore, the bedside nurse should monitor the hemodynamic status of the child very closely throughout the procedure. Procedural sedation may produce complications. Because managing procedural sedation is the responsibility of pediatric cardiovascular nurses, the bedside nurse should be well versed with these complications. Adequate care must be taken to keep the critically ill infant or child warm and comfortable during the procedure.
A transesophageal echocardiogram (TEE) may produce complications such as oropharyngeal and esophageal injury and compression of airways and vascular structures. Contraindications to TEE include unrepaired tracheoesophageal fistula, significant esophageal stricture, active gastrointestinal bleeding, and an uncontrolled airway in a patient with significant cardiorespiratory compromise.690
Nursing Interventions
There is no specific nursing care required before, during, or after echocardiography. It is important that the nurse observe the patient carefully throughout the procedure, and ensure that the patient is kept warm. Unstable children must be monitored closely. The study will progress more quickly if the nurse is able to help keep the infant or child quiet, content, and motionless. Often this requires creativity and a lot of patience. The nurse should keep a bottle (unless the infant can have nothing by mouth) and pacifier nearby during echocardiography of the infant and several toys available during echocardiography of the child. Children often enjoy watching their heart “on TV.”
Nuclear magnetic resonance imaging (NMRI or MRI)
Description
Clinical magnetic resonance imaging (MRI) uses the magnetic properties of hydrogen and its interaction with both a large external magnetic field and radio waves to produce highly detailed images of the human body. In its early days, MRI was known as NMR, or nuclear magnetic resonance. Although the name has changed, the basic principles are the same. MR images are generated by analysis of signals produced by hydrogen nuclei of molecules in varying tissues as the spins of the nuclei are aligned in a strong external magnetic field and then perturbed by radiofrequency pulses. In an MRI machine, a radio frequency transmitter is briefly turned on, producing an electromagnetic field. The photons of this field have just the right energy, known as the resonance frequency, to flip the spin of the aligned protons in the body. As the intensity and duration of application of the field increase, more aligned spins are affected. After the field is turned off, the protons decay to the original spin-down state and the difference in energy between the two states is released as a photon. It is these photons that produce the electromagnetic signal that the scanner detects. An image can be constructed because the protons in different tissues return to their equilibrium state at different rates, a difference that can be detected.
MRI is used to image every part of the body, and is particularly useful for tissues with many hydrogen nuclei and little density contrast, such as the brain, muscle (including myocardium), connective tissue, and most tumors. MRI often provides more sensitive and specific imaging information about pediatric CNS abnormalities than does ultrasound or computed tomography technology. MRI also has redefined the role of invasive procedures such as myelography, ventriculography, cisternography, and angiography. Box 8-53 summarizes some of the common indications of MRI in critically ill children.24,52,57-59,635
Box 8-53 Common Indications for Magnetic Resonance Imaging in Critically Ill Children
Acute trauma
Nonaccidental trauma
Hypoxic ischemic injury
Meningitis or meningoencephalitis
Intracranial or spinal fluid collections and/or Infections
Intractable seizure activity
Evaluation of spinal column and spinal neuraxis
Unexplained and intractable headaches
Unexplained hydrocephalus
Investigation of orbital pathology
Toxic metabolic injury
Vascular disease and hemorrhage
Muscular and vascular anomalies in any body part
Postinfectious encephalomyelitis
All personnel and monitoring and support equipment must be safe for the magnet. The American Society for Testing and Material (ASTM) International developed the following terminology for labeling of implanted devices21,796:
• MR safe: An item that poses no known hazards in any MR environment
• MR conditional: An item that has been demonstrated to pose no known hazards in a specified MR imaging environment with specified conditions of use
• MR unsafe: An item that is known to pose hazards in all MR environments
Procedure
The MRI scanner is normally located well away from critical care units. The patient is positioned on the moveable examination table. The patient lies inside a large, cylinder-shaped magnet. Radio waves 10,000 to 30,000 times stronger than the magnetic field of the earth are then sent through the body. These radio waves and the magnet affect the body's atoms, forcing the nuclei into a different position. As they move back into place, they send out radio waves of their own. The scanner picks up these signals and a computer turns them into a picture. These pictures are based on the location and strength of the incoming signals. If a contrast material is used during the examination, it is injected into the intravenous (IV) catheter after an initial series of scans. Additional series of images are taken during or after the injection.
Straps and bolsters may be used to keep the child still and maintain the correct position during imaging. An audiovisual system within the scanner greatly reduces the need for sedation in children greater than 3 years of age.
Potential Complications
There are no known complications to MRI scanning. The principal drawback of MR imaging is the long acquisition time, which frequently necessitates procedural sedation for children less than 8 years old and for some who are older. As noted, the use of audiovisual systems may distract or calm the child and reduce need for sedation.
Another drawback of MRI is the incompatibility of ferromagnetic materials within the imaging suite. In critically ill ventilator-dependent children, plastic and aluminium MR-compatible monitors and ventilators are available, but children with metallic bullet fragments or implants, including cardiac pacemakers and neurostimulators, may not be able to undergo MRI.
Although the scanning itself causes no pain, there may be some discomfort for some children from having to remain still for several minutes to a few hours. The MRI machine produces loud thumping and humming noises; therefore earplugs are typically provided during the scan. The MRI scan is particularly stressful for children with claustrophobia and/or chronic pain.
Because the child must remain in a closed chamber with limited access during the procedure, this procedure has the potential for short-term complications such as inadvertent extubation, hemodynamic collapse, contrast infiltration at the injection site, anaphylactoid reactions from the gadolinium contrast agents, and/or long-term complications from gadolinium contrast as nephrogenic systemic fibrosis (NSF). MR contrast agents are generally safer than iodinated contrast agents in patients who are unstable or have impaired renal function or a prior history of anaphylactoid reaction to iodinated contrast agents.
Nursing Interventions
Before entering the MRI suite, the bedside nurse should remove any MRI-incompatible metal jewelry or clothing accessories. A detailed list of MR-compatible medical devices is available on the web site maintained by Frank Shellock at http://www.mrisafety.com. In addition to magnet issues, metallic devices can also create artifacts and distortions in the surrounding tissues.
Given the long acquisition time of the procedure, all devices and catheters must be secured before the start of the procedure. Careful cardiorespiratory monitoring is required throughout the procedure, often performed via a camera in the scanner and a screen in the observation area. For children who cannot hold still for the examination, procedural sedation may be needed. If the patient is in the critical care unit, the bedside nurse should accompany the child throughout transportation to and from and during the procedure in the MRI suite. (The nurse will observe the child in the observation room during the actual procedure.)
To continue sedation or vasopressor infusions during the MRI, the bedside nurse should attach and flush long tubing extensions before the transport to allow continuation of all infusions during the MRI. Because most infusion pumps are incompatible with the MRI, they must remain outside of the MRI suite. The long tubing allows use of these infusion pumps; the tubing stretches from the patient through a hole in the wall of the MRI suite to the pumps. If the patient is intubated, both the bedside nurse and the respiratory therapist are responsible for ensuring that the endotracheal tube is well secured and remains in place throughout the procedure.
Although intravenous contrast is generally administered by the radiology technicians or a physician in the radiology suite, the bedside nurse is responsible for maintaining the intravenous catheter and monitoring the injection site. Infiltration of the contrast material can result in a painful tissue injury around the site.
Cardiac Catheterization and Intervention
Jo Ann Nieves and Ruby L. Whalen
Pearls
• Pediatric cardiac catheterization procedures are primarily for interventional purposes.
• Cardiac catheterization interventional procedures alter cardiac hemodynamics. Each patient must be monitored for potential complications, including decreased systemic perfusion, arrhythmias, bleeding, tamponade, and compromised limb perfusion.
• Interventional procedures are typically performed under general anesthesia.
• Anemia may develop from blood loss during the procedure. A hematocrit less than 40% is undesirable in the child with single ventricle physiology or cyanotic heart disease and will lead to inadequate oxygen carrying capacity and delivery. Transfusion therapy may be required.
• Increasingly complex interventions: About 70% of cardiac catheterizations are now interventional, and many delay or eliminate the need for surgical intervention. Patient acuity is often high.
• When patients have short length of stay (admitted and discharged within 24 hours) the teaching provided by the bedside nurse is crucial.
• Care is anticipatory; to anticipate complications the bedside nurse requires knowledge of the child's diagnosis, intervention, and change in anatomy/and physiology caused by the catheterization.
Cardiac catheterization involves insertion of a radiopaque catheter through an artery and/or vein into the heart. This procedure is performed under fluoroscopy so the location and movement of the catheter can be visualized. Throughout catheterization, pressure measurements are made and oxygen saturations are recorded to provide complete hemodynamic and anatomic data about blood flow patterns (including shunts) within the heart. Structures such as cardiac chambers, valves, or great vessels can be visualized through injection of a radiopaque contrast agent. Rapid sequential radiographs, called angiograms, are made to record the flow of contrast through the heart. References that provide excellent reviews of the technique of cardiac catheterization and analysis of the data include Grifka336 and Lock545.
Assessment of the pulmonary vascular bed is required in patients with elevated pulmonary vascular resistance to determine operability or before transplant. Response to vasodilator therapy will be evaluated, including response to oxygen and medications such as nitric oxide, prostacyclin, and sildenafil. Fontan and Glenn procedures require low pulmonary vascular resistance. Pulmonary hypertensive precautions in care are required to avoid exacerbation of the pulmonary vascular hypertension. (See section, Common Clinical Conditions, Pulmonary Hypertension, and Specific Diseases, Single Functioning Ventricle.)
Since the 1980s advances in echocardiography and magnetic resonance imaging have progressively improved the precision of noninvasive diagnosis of congenital heart defects, decreasing the need for diagnostic cardiac catheterization.336 Diagnostic cardiac catheterization is still required when complete diagnosis or hemodynamic information is not possible with noninvasive methods. Diagnostic catheterization may also take place when the patient's clinical signs and symptoms are not consistent with the diagnosis, or when the clinical course is not progressing as expected.336
The assessment of pulmonary vascular resistance is one of the most common indications for diagnostic catheterization,336 particularly in those patients who will undergo creation of a complete systemic venous to pulmonary arterial connection.253 Selected patients undergo MRI study and, therefore, no longer require routine preoperative diagnostic cardiac catheterization prior to bidirectional Glenn procedure.118
Therapeutic interventions have rapidly evolved as the primary reason for cardiac catheterization. The interventional cardiologist uses devices to dilate stenotic structures, close defects, and create vascular connections in patients with congenital heart disease of all ages, from the unborn fetus to the adult. In many cases patients no longer require surgical intervention, and in selected cases surgery is delayed. Many procedures take place as a day procedure or require overnight admission, have a relatively painless and rapid recovery, and are safe and effective.660 Many lesions previously classified as inoperable can be managed at cardiac catheterization.545 In selected patients a hybrid procedure will combine the simultaneous skills of the surgeon with the interventional cardiology team to optimize patient management.
Critically ill patients requiring cardiac catheterization postoperatively may be moved to the catheterization suite on mechanical cardiopulmonary support circuits for diagnostic catheterization and catheter intervention. Electrophysiologic studies are used to identify and eliminate intracardiac conduction pathways and sources of arrhythmias.
The most common percutaneous interventional procedures in the adult patient with congenital heart disease include lesion closures by devices or coils, balloon valvuloplasty, balloon angioplasty with or without stent placement to treat native, recurrent or prosthetic conduit lesions, and arrhythmia assessment or therapies. Adult interventions are detailed in Inglessis and Landzberg399 and Holzer and Cheatham.388
Procedure
Cardiac catheterization is performed in a catheterization laboratory where appropriate catheters, devices, anesthesia, and radiographic equipment are located. The infant or child usually receives nothing by mouth before the catheterization to minimize the possibility of vomiting and aspiration during the procedure. Oral sedation is prescribed before the catheterization in patients presenting for outpatient, elective catheterization, and additional sedation is administered as needed during the procedure. General anesthesia is often used for interventional procedures.664 Vascular access is typically placed in patients with cyanotic heart disease and/or polycythemia before the catheterization to ensure adequate hydration and sedation.336
The patient's right femoral artery and vein often are used for the procedure; the umbilical vessels may be used in the neonate. In the older child the femoral artery and vein or vessels in the antecubital fossa or axilla, jugular, or subclavian veins may be used for the catheterization. Percutaneous puncture, usually via the Seldinger technique, is performed most often to gain access to the artery or vein. Transhepatic access may be required if femoral access is not possible. Surgical access via a cutdown in the neck may be used as access for interventional procedures treating the aortic valve in newborns or the descending aorta to optimize access during hybrid intervention.663
Right heart catheterization is accomplished through insertion of a catheter into a vein; for purposes of simplicity, the remaining description assumes normally related atria, ventricles, and great vessels. The catheter is passed into the superior or inferior vena cava and into the right atrium and then the right ventricle and pulmonary artery. Pressure measurements and oxygen saturation analyses are made in each location to detect the presence and location of abnormal chamber hypertension or intracardiac shunts.
The left heart often can be entered by passage of the catheter from the right atrium through a probe-patent foramen ovale to the left atrium. If the cardiologist is not able to enter the left atrium from the right atrium, a catheter can be inserted into a systemic artery, retrograde into the aorta and then into the left ventricle, or the atrial septum can be punctured to allow access into the left heart.
When all necessary pressure and oxygen saturation measurements have been recorded, angiograms are performed. Angiograms depict any shunting that occurs as the result of congenital heart defects or valvular dysfunction, and document areas of narrowing or dilation in blood flow pathways.
Associated procedures may be accomplished during cardiac catheterization. As noted, defects may be created or closed, and valves and vessels may be dilated. Specific information regarding therapeutic catheterization is comprehensively covered by Nykanen and Zahn,663 Lock,545 and Holzer and Cheatham.388,389
Conduction system mapping can be performed along with therapeutic management of refractory arrhythmias (comprehensive information in Saul772). Transvenous intracardiac pacing wires also may be inserted during catheterization and fluoroscopy
Sedation and analgesia are continued during the procedure with a variety of agents, including morphine, fentanyl, ketamine, midazolam, and propofol (see Chapter 5).336,664 Agents that produce systemic vasodilation, such as propofol, are avoided in patients with right-to-left shunts, because they enhance the right-to-left shunt and can worsen hypoxemia.336
Medication may be administered during the catheterization to allow detailed assessment of the patient's hemodynamic response. Continual monitoring of all hemodynamic and respiratory responses occurs throughout the procedure, observing for variations in heart rate, oxygen saturation, blood pressure, and ventilatory status.
At the end of the catheterization procedure the catheters are removed and pressure is applied to the puncture site. Products that apply pressure over the access sites may be secured in place with specific removal guidelines (i.e., they are removed several hours later). The infant or child is returned to the unit for close observation. If a catheter was passed through a surgical systemic to pulmonary artery shunt, anticoagulation therapy may be ordered to minimize the risk of thrombus within the shunt (see Postoperative Care, Anticoagulation).
If the child is normally ambulatory, the physician typically requests that he or she remain in bed for several hours after the catheterization. After percutaneous cardiac device placement a chest radiograph or echocardiogram is typically obtained to evaluate device position before discharge home.664 Nursing care is reviewed in the following sections.
If cardiac catheterization is performed electively on a stable child the mortality rate is low, reported at 0.2%,143 with 2.4% serious complications. If, however, the procedure is performed on an emergency basis on a critically ill patient, the morbidity and mortality will be higher. The risks of catheterization also are increased for infants and children with elevated pulmonary vascular resistance, arrhythmias, or hypoxemia, and for those children with tetralogy of Fallot or other cyanotic defect who have a history of hypercyanotic episodes (see section, Common Clinical Conditions, Hypoxemia).
Therapeutic Cardiac Catheterization
Cardiac catheterization interventions in congenital heart disease are often guided by transesophageal echocardiogram (TEE), intravascular ultrasound (IVUS) or intracardiac echocardiogram (ICE). Hybrid procedures combine the simultaneous skills of the surgeon with the interventional cardiology team. Interventions can be completed intraoperatively, including implantation of stents in the pulmonary or systemic vasculature under direct vision or cardioscopic imaging.663 Surgeons can assist planned catheter interventions by providing alternative open surgical vessel access for interventions via the carotid artery or direct access to the aortic valve or descending aorta.663 Improved outcomes are reported with early (less than 24 hours) cardiac catheterization and catheter based intervention in children who cannot be separated from cardiopulmonary bypass at the end of a surgical procedure.663,965
Occlusion of Lesions
Atrial septal defect (ASD) transcatheter device closure began in 1975.781 Secundum ASDs are most frequently closed by the Amplatzer septal occluder device, approved by the FDA in 2001 (see Evolve Figs. 8-3 through 8-5 in the Chapter 8 Supplement on the Evolve Website). The Amplatzer is a double-disk of nitinol wire mesh and fibers, available up to 38 mm in diameter.389 The Helex (up to a 35 mm device) has a long nitinol wire with polytetrafluoroethylene fabric that allows tissue in-growth to seal the defect.649 ASD occluders have also included the Sideris, CardioSEAL, and STARFlex devices.389
Rhythm disturbances are common within the first 24 hours after ASD device closure,374 but typically resolve quickly.389 Patients with ASDs are at increased risk for paradoxic emboli if they develop deep vein thrombosis.26 Device embolization is rare, but can occur into the pulmonary, left heart, or systemic circulation.664 Adults with large ASD with deficient rims are at highest risk for embolization.389
Adults with large unrepaired ASDs can develop atrial arrhythmias and heart failure. They may have left ventricular diastolic dysfunction, with development of elevated left atrial pressure, pulmonary edema, and ventilator dependence after closure of the ASD, when the “pop off” flow across the atrial septum is sealed.256 Closure of large ASDs may precipitate headaches in patients with a previous history.
Ventricular septal defects (VSDs) in the muscular septum may be closed by percutaneous placement of the FDA approved muscular VSD Amplatzer occluder device, available up to 18 mm (see Evolve Fig. 8-3 in the Chapter 8 Supplement on the Evolve Website).139,389 A transcatheter approach or periventricular (midsternal chest incision with the heart beating, no cardiopulmonary bypass) closure may be used.389 Complications include potential embolization, arrhythmias (most frequently heart block), and blood loss requiring transfusion.664 The transcatheter device closure of perimembranous VSD is under investigation with potential concerns including development of heart block in 1%579 to 5%,389 within days to months after the procedure. New or increased valve regurgitation (aortic or tricuspid) has been reported in up to 9%, with most improving or resolving by 6 months after the procedure.390
Vascular structure occlusions include sealing of aortopulmonary collateral vessels, pulmonary arteriovenous fistulae producing cyanosis, residual shunts, Blalock-Taussig shunts, venous malformations and coronary AV fistulae.545 Devices used to occlude these vessels include Gianturco embolization coils and stainless steel coils with synthetic Dacron fibers.389 The Gianturco-Grifka vascular occlusion device consists of a nylon bag filled with long stainless steel wires. It may be used to occlude vascular tubular structures.389
Closure of a patent ductus arteriosus (PDA) can use coils or the Amplatzer duct occluder device made of nitinol wire mesh (FDA approved). The Nit-Occlud PDA occlusion system (nitinol wire device wound into a helix-type loop) is also available.389 Complications can include dislodgement and migration of coils into the systemic or pulmonary circulations.664
Fontan fenestrations may be closed with septal closure devices in patients with transcutaneous oxygen saturations of 90% or less after Fontan completion.389 The Amplatzer septal occluder is the most common device used after balloon test occlusion of the fenestration to assure favorable hemodynamics.389
Implanted devices can embolize. When monitoring for device embolization assess for signs of altered perfusion status or development of arrhythmias or ECG changes. Auscultation may reveal a change in the cardiac murmur. Changes in peripheral perfusion can occur with migration into the aorta or branches. Some embolizations may be completely asymptomatic. CXR and echo are monitored for status of device location before the patient's discharge home. Dislodged devices may be retrieved with cardiac catheterization or surgery.
Structure Enlargement
Obstructive congenital heart lesions can be treated with percutaneous cardiac catheterization procedures that use balloon angioplasty and cutting balloons and may include placement of endovascular stents.389 Balloon angioplasty is performed to stretch a vessel to its maximum diameter.545 The angioplasty results in stretching or vessel tear of the intima, media, and rarely the adventitia.389,664 The vessel will then heal in an open position.545 Because vessels recoil to a smaller size after angioplasty, a stent may be placed to increase the diameter to a larger size than would result from an angioplasty alone.545 Stent implantation is most commonly used to enlarge branch pulmonary artery stenosis, for treatment of coarctation (see Fig. 8-67),389 and to enlarge narrowed modified Blalock-Taussig or Sano Gore-Tex grafts.725
All stents used for congenital cardiovascular lesions in the United States are used in an “off-label” basis, because they were not designed and have not been approved for the specific intervention.389 Covered stents are used to treat ruptured vessels or aortic aneurysms.389 Stent implantation in infants is safe with re-dilations scheduled to keep pace with somatic growth.827 Hybrid procedures may be completed with placement of stents planned in the operating suite, along with the cardiac surgical procedure.389
Balloon angioplasty of all types of pulmonary arteries is a widely accepted procedure,389 and is one of the most common interventions performed in the developed world.26 Repeated, staged interventions may be needed to treat distal pulmonary artery stenosis389 or long segment stenosis (also called hypoplastic pulmonary arteries),545 particularly to decrease RV strain before replacement of the pulmonary valve or in the presence of RV dysfunction.388
Vessels treated by angioplasty or stenting may develop aneurysm or dissection, or they may rupture.664 Patients with diseases involving arteriopathies, such as William syndrome, are at increased risk of vessel rupture with dilation. The pulmonary vessels are the most likely to rupture after angioplasty, with presenting signs including acute hemoptysis, pulmonary hemorrhage, hemothorax, and pleural or pericardial effusions with tamponade.664 Progression of bleeding must be monitored and ventilation supported. Pulmonary edema may develop because of increased blood flow after angioplasties, particularly in patients with multiple peripheral pulmonary artery stenoses (as in William or Alagille syndrome).664 Surgical intervention may be required.
All bioprosthetic conduits placed for surgical palliation of tetralogy of Fallot, truncus arteriosus, or other complex repairs develop obstruction because of calcification, shrinkage, or compression,545 and they do not grow as the child grows. Right ventricle to pulmonary artery conduit stenosis can be treated with angioplasty and stents to delay reoperations for conduit change.545
In newborns to adults, valvar pulmonic stenosis requiring intervention is treated by percutaneous balloon dilation as the standard therapeutic intervention.389 For the first several days after balloon valvuloplasty for treatment of critical pulmonary valve stenosis of the newborn, decreased right ventricular compliance and residual elevation of right ventricular pressure may initially prevent sufficient antegrade flow across the valve, with resultant cyanosis and continued requirement for prostaglandin E1 infusion. Once right ventricular compliance improves, pulmonary blood flow and arterial oxygen saturation rise days later.664 Restenosis of the pulmonary valve may occur in 25% to 50% of neonates.545 Beyond the neonatal period, a single valvotomy will likely produce sufficient pulmonary blood flow for decades, with restenosis rare.
Atretic pulmonary valves may be perforated with radiofrequency energy during cardiac catheterization, then balloon dilation is used to increase forward blood flow.26 An estimated 50% of these infants require infusion of prostaglandin E1 to provide additional pulmonary blood flow for several hours or days after cardiac catheterization. If severe hypoxemia and cyanosis persist, the infant may require future surgical aortopulmonary shunt or ductal stenting.389 The major risk of radiofrequency valve perforation is perforation of the heart with resultant pericardial effusion and potential tamponade.26 In patients with pulmonary atresia/intact ventricular septum, radiofrequency valve perforation can facilitate pulmonary artery growth and provide the potential for eventual biventricular repair.
Aortic valve stenosis treated by balloon valvotomy increases valve diameter and decreases the gradient across the valve. It is important to note that neonates with critical aortic obstruction may have a falsely low transvalvular gradient at the beginning of the procedure because cardiac output is low.545
An aortic valvotomy may produce aortic regurgitation if excessive dilation or torn leaflets result. Severe aortic regurgitation produces symptoms including low diastolic pressure, tachycardia, diastolic murmur, signs of coronary ischemia, hypotension, and poor systemic perfusion. Surgical intervention may be required.
Critically ill neonates may continue to require hemodynamic support after successful aortic valvotomy because of pre-existing poor left ventricular function. Aortic valve balloon valvuloplasty is usually considered palliative versus curative,389 although as many as two thirds of patients are free from surgery in the long term.276 Percutaneous placement of a stent can relieve tunnel subaortic stenosis.545 In the neonate with critical aortic stenosis, surgical access to the carotid artery permits safe and effective access to the valve for intervention.
Mitral valve balloon valvotomy may be performed for congenital or acquired (rheumatic) stenosis. In most patients with mitral stenosis caused by rheumatic heart disease, mitral balloon valvotomy is the initial intervention of choice.545 These patients require careful assessment and care, particularly if they have pulmonary hypertension before intervention.664 Mitral regurgitation may be present postprocedure and require surgical intervention.
Aortic coarctation balloon angioplasty is less common in small children and infants because of the high risk of recoarctation (near 66%).389 The goal of the procedure is to reduce the gradient to less than 10 mm Hg, or effect a 90% relief of the obstruction.389 Adults and school-aged children with primary coarctation or recoarctation are often treated with placement of an aortic stent (Fig. 8-67).388,545 After angioplasty, vessel dissection is uncommon, but if it occurs it can be treated by placement of a covered stent. An aneurysm may be seen early and late after coarctation dilation.389 Persistent, severe chest pain can indicate extension of the aortic tear; therefore this should be reported to a physician immediately.664 Hypertension is present before intervention because of the aortic coarctation, and severe elevations must be controlled after the treatment to avoid the risk of vessel rupture.664
An unrestrictive atrial shunt is required for patients with simple transposition of the great arteries and other complex lesions to improve mixing of systemic and pulmonary venous blood and optimize arterial oxygen saturation. In hypoplastic left heart syndrome (HLHS), adequate cardiac output requires an adequate interatrial communication. The balloon atrial septostomy is a life-saving procedure for infants with simple transposition of the great arteries and acidosis related to a restrictive atrial shunt.389 Percutaneous access through umbilical or femoral vein is typical with echocardiographic guidance in the PCCU. A deflated balloon is passed through the venous system, into the right atrium, across the atrial septum. The balloon is inflated with dilute contrast and pulled rapidly across the atrial opening, into the right atrium, to force a perforation in the septum. After adequate septostomy, the atrial pressures should immediately equalize, and hemodynamic status should improve.389 Above the age of 1 month, the atrial septum may be too thick or tough for adequate septostomy requiring additional use of a blade atrial septostomy or cutting balloon atrial septostomy.389
A highly restrictive atrial shunt or intact atrial septum in HLHS has been managed by emergent balloon atrial septostomy in the effort to stabilize hemodynamics before first-stage palliation or cardiac transplantation.908 Fetal intervention has been performed for this condition with reports of improving hospital survival after first-stage palliation.908 Placement of an intraatrial stent may be required to maintain nonrestrictive atrial communication,525 and cutting balloons or radiofrequency perforation may be used.389,691 Before catheterization, infants with HLHS and severely restrictive atrial shunt or intact atrial septum are typically gravely ill, with severe cyanosis and pulmonary hypertension.685 After atrial septostomy or stent implantation, the interatrial communication can progressively restenose.691 Despite atrial septostomy, mortality in patients born with severely restrictive atrial shunt or intact atrial septum remains high.323
After an atrial switch procedure (Mustard or Senning operation for transposition of the great arteries) residual anatomic lesions can emerge.388 Systemic or pulmonary venous baffle obstruction can be treated by percutaneous catheter dilation and stent implantation. Leaks within the atrial baffles may be sealed with a septal device occluder.388 Electrophysiology therapy may be required for recurrent arrhythmias.
Pulmonary vein stenosis has a poor long-term outcome, despite surgical and therapeutic cardiac catheterization intervention.232 Complications frequently develop after placement of pulmonary vein stents, and include stent embolization into the systemic circulation.389
Fontan fenestrations can be created with percutaneous catheterization by perforation of the extracardiac conduit and a covered stent may be inserted to maintain fenestration patency. Such fenestrations can result in improved cardiac output, pleural effusions, and ascites.615 A Fontan procedure can be completed during cardiac catheterization with percutaneous placement of a large covered stent extending from the inferior vena cava into the superior vena cava and pulmonary arteries.292
Significant pulmonary insufficiency leads to RV dilation with subsequent risk for RV dysfunction and arrhythmias. Such insufficiency may develop in patients after tetralogy of Fallot repair,483 and some patients ultimately require reoperation for insertion of a valved right ventricle to pulmonary artery conduit. Surgically placed valved conduits have limited life spans.550 Since 2000, successful percutaneous valve replacement has been accomplished within a stent placed in the pulmonary position to treat both pulmonary stenosis and regurgitation.91 A bovine jugular venous valve is used within a platinum stent that can be expanded to a maximum of 22 mm in diameter (Fig. 8-68, A).389,598 Potential complications can include homograft rupture, device migration, coronary compression, and later stent fracture.550 Surgical intervention for valved conduit replacement is delayed,550 but evaluation and long-term outcome analysis continue. Percutaneous aortic valve implantations and mitral valve repairs have begun in adults, with increasing success.260,389
Coils may be used to close PDAs (Fig. 8-69). Stents may be used to keep the PDA open in patients awaiting cardiac transplant for HLHS,94 and as an alternative to initial palliative surgical shunt, to maintain pulmonary blood flow in ductal-dependent pulmonary circulation (Fig. 8-70).18,93,614 Stents may also be placed to widen a narrowed Gore-Tex shunt. Potential complications include progressive narrowing of the stent with cyanosis requiring re-intervention, stent migration, intravascular hemolysis, and worsening of pulmonary artery stenosis.18,614
Other Cardiac Catheterization Procedures
Endomyocardial biopsy by cardiac catheterization remains the mainstay in the diagnosis of rejection both early and late after heart transplantation.545 The biopsies can produce complications; myocardial perforation is the most common (see the section on Endomyocardial Biopsy).
Electrophysiology studies are performed to identify cardiac rhythm abnormalities and definitively treat them by elimination of abnormal atrial or ventricular arrhythmia pathways. The procedures are similar to cardiac catheterization with various potential access sites at the antecubital veins, jugular, and femoral vessels. Radiofrequency energy applies heat to burn lesions and interrupt arrhythmia pathways. Cryoablation applies extreme cold at the catheters tip to ultimately burst the cells responsible for arrhythmias.772 These procedures are reviewed in detail by Saul.772
General anesthesia is often used to ensure that the patient remains motionless during critical points in the procedure, and to keep the patient comfortable during a potentially long procedure.772 Radiofrequency energy can cause AV block in children775 and coronary artery injury.85 Cryoablation is safer,772 having minimal effect on the adjacent coronary arteries814 and a lower incidence of thrombus formation.465 Additional potential complications include vascular injury, embolic injury, valvular regurgitation, and minor skin irritations or burns at electrode skin sites.503 Mortality is low for ablation procedures, reported at 0.097%,503 but up to 0.89% in patients with structural heart disease.774
Hybrid Procedures
Hybrid procedures combine the interventional cardiac catheterization procedure with a surgical procedure. Hybrid procedures include periventricular closure of the ventricular septal defect,40 as well as intraoperative placement of stents in the pulmonary arteries or the aorta.
Newborns with hypoplastic left heart syndrome (HLHS) may undergo a hybrid first-stage intervention to extend the waiting time for cardiac transplant, allow time for left ventricular growth,12 and minimize the newborn's exposure to cardiopulmonary bypass, cardioplegic arrest, and circulatory arrest.534 The stage I hybrid procedure for HLHS includes surgical bilateral pulmonary artery banding with cardiac catheterization placement of a stent within the ductus arteriosus (Fig. 8-71), possibly followed by enlargement of the interatrial communication via septostomy.130,340,783 Postcatheterization hemodynamics in stage I hybrid HLHS patients can include early low cardiac output, elevated systemic vascular resistance, acidosis and high serum lactate, and elevated pulmonary blood flow,534 requiring close monitoring and management.
The atrial defect can become progressively restrictive even after atrial stent implantation,691 indicated by increasing cyanosis, rising left atrial pressure, and respiratory distress. Preductal coarctation or arch hypoplasia may compromise pulmonary blood flow.340 Percutaneous intravascular devices to control pulmonary blood flow are under development.94 Long-term results for stage I hybrid palliation for HLHS and Fontan completion with a covered stent are unknown.
Fetal Intervention
Fetal in utero interventional valvuloplasty is performed at select programs for cases of critical aortic valvar obstruction, with the aim of preventing fetal progression to hypoplastic left heart syndrome.791,884 Intact or highly restrictive interatrial communication,908 and pulmonary atresia with intact ventricular septum689 have also been treated by fetal in utero dilation. Fetal aortic valve intervention has led to biventricular repairs.545 Long-term outcomes must be analyzed.
Clinical Trials
The development of transcatheter devices to treat congenital heart lesions have rapidly progressed over the past decade. Septal occluder devices, intravascular stents, and transcatheter valves have become more readily available. This has led to the expansion of clinical research trials into the domain of interventional congenital cardiology. Clinical trials exist for ASD and VSD closure devices, stent procedures, and transcatheter pulmonary valves. When caring for a child who is enrolled in a clinical trial, the nurse must be aware of the responsibilities of each member of the research team. Adherence to the trial's protocol is imperative if access to devices is to continue and grow. Byers126 reported that 75% of families enrolled in clinical research trials thought that the treatment provided was the standard of care.
Potential Complications of Cardiac Catheterization
Potential complications of any cardiac catheterization include arrhythmias, particularly atrial or ventricular irritability (and resultant supraventricular or ventricular arrhythmias), or the development of atrioventricular (AV) block. Bleeding, cardiac perforation, tamponade, cerebrovascular accident, stroke, pulmonary hemorrhage, pulmonary edema, contrast agent reactions, brachial plexus injury, skin pressure lesions, or hypothermia may also occur. Arterial or venous vascular injury may occur as well.336
Interventional catheterization often involves anticoagulation and insertion of large sheaths, and may require long procedure times. This can increase the risk of bleeding from the access site, hematoma formation, vessel spasm, and thrombus. Frequent assessment of the extremity is vital for early detection and treatment to avoid limb ischemia and permanent complications.
Hypoventilation may result because of the effects of sedation or prostaglandin infusions, or in patients with Down syndrome because of the risk of airway obstruction.336 Emboli into the systemic or venous circulations can be caused by air, thrombus, or broken catheters or wires.336 Patients with infundibular pulmonic stenosis (including tetralogy of Fallot) may experience hypercyanotic episodes during or after the catheterization despite careful maintenance of hydration and sedation and careful catheter manipulation (see section, Common Clinical Conditions, Cyanotic Heart Disease).336 Air or thrombus embolization to cerebral vessels is a known complication. This risk increases in the cyanotic or iron-deficient patient, or if multiple catheter exchanges occurred during the catheterization.660,663 Devices placed during interventional procedures may embolize within the heart or distal vascular sites. Aortic stents can migrate distally. Although complications are rare, the nurse should exercise a high index of suspicion with focus on anticipation of potential problems.336,660,663
Nursing Management
Any patient who returns from the cardiac catheterization procedure with hemodynamic instability or new, sudden arrhythmias after a device placement must be assessed for suspected device displacement. A device placed to occlude an atrial septal defect can migrate into the mitral valve or aorta. A device placed to close a patent ductus arteriosus can migrate into the pulmonary arteries or the aorta, particularly if the patient has pulmonary hypertension. Initial efforts focus on maintaining adequate oxygenation, ventilation, and hemodynamic stability. An immediate echocardiogram can assist in evaluating the position of the device within the heart. A chest radiograph also provides a general idea of device location, but interpretation may be complicated by unusual anatomy.
Patients with elevated pulmonary vascular resistance may develop acute elevations in pulmonary resistance with right heart failure. If a pulmonary hypertension event occurs after cardiac catheterization, management focuses on optimizing airway, ventilation, and oxygenation; treatment of pain and anxiety; optimizing carbon dioxide tension, and correction of hypothermia (see section, Common Clinical Conditions, Pulmonary Hypertension, Management).
Alternative routes of vascular access (ARVA) have grown with the increasing complexity of patients and interventional procedures. Alternative access routes involve, but are not limited to, access for catheterization via carotid, brachial, hepatic, and open chest/direct cardiac puncture.222 Two common ARVA sites are carotid and hepatic access. Carotid access is often used emergently to treat critical aortic stenosis, or for PDA stent placement. Hepatic venous access is used in the setting of femoral vein occlusion. The nurse must be aware of the access route used for the procedure and assess the patient accordingly.
When caring for a child who is part of a clinical research study, the bedside nurse must ensure that the family understands they are part of a clinical trial. Family support and education are paramount. Compliance with the treatment protocol, prompt notification of adverse events to the clinical research team, and reporting of conflicts of interest are the responsibility of the nursing team.
The past decade has witnessed significant changes in cardiac catheterization therapy. Although the volume of diagnostic procedures has decreased, an increase in interventional treatment of congenital heart disease has been associated with higher acuity and complexity of cases.660 Improved noninvasive diagnostic methods have brought about earlier diagnosis and treatment. This, in turn, has had a significant effect on required postcatheterization nursing care.
Anesthesia is used for many interventional cases, patients are younger, undergoing corrective treatment in the cath lab, and with short LOS as most are day procedures with patient admitted day of the procedure and discharged within 24 h. In addition, more patients undergoing cardiac catheterization are unstable, with problems such as early postoperative low cardiac output or pulmonary hypertension,336 and they require critical care.
Most cardiac catheterizations are performed percutaneously, and therefore do not include an incision that requires sutures. However, pressure is applied to the vascular access site, and the nurse should monitor for evidence of bleeding or hematoma formation and for evidence of obstruction of the venous or arterial catheterization site.
Patients may return to the nursing unit with only intravenous fluids and oxygen for recovery. However, interventional catheterizations create significant changes in physiology. This necessitates need for nursing care centered on a high index of suspicion for potential complications.
General nursing management of the patient before and after cardiac catheterization is summarized in Box 8-54. Intervention-specific care after interventional cardiac catheterization is summarized in Box 8-55. Finally, advanced concepts related to care of the child undergoing cardiac catheterization are summarized in Box 8-56.
Box 8-54 Nursing Care for the Child Undergoing Cardiac Catheterization
Precardiac Catheterization
Potential Problems
Anxiety (patient/family) and knowledge deficit related to patient's health status, anticipated catheterization
Expected Patient Outcomes
• Patient/family demonstrates comprehension of preparation for procedure, catheterization itself, and postcatheterization care
• Patient/family's anxiety does not interfere with appropriate activity
Nursing Interventions
• Orient child/family to nursing care unit, policies, personnel, catheterization lab (as age appropriate)
• Orient child/family to preparation for catheterization, including tests, medications (including holding anticoagulants, premedication, and effects and planned sedation or anesthesia), need for nothing by mouth before catheterization

Instruct patient (as appropriate to age) and family regarding procedure itself

Discuss postcatheterization care with patient (if appropriate to age) and family

If child is greater than 2 years old, toys or puppets may be used to demonstrate experiences the child will remember
• During and after catheterization procedure, provide support and simple explanations of catheterization results; orient patient to time and place frequently while patient is recovering from sedation. Involve child life therapist.
• Provide written information on procedure and child's condition
• Provide online teaching web sites to reinforce teaching
Postcardiac Catheterization
General Care for All Patients
• Continuous cardiorespiratory monitoring, including monitoring of oxyhemoglobin saturation by pulse oximetry
• Assessment and support of airway, oxygenation, and ventilation as needed
• Frequent assessment of perfusion of affected extremity
• Exposure of pressure dressing, monitoring for bleeding, hematoma
• Bedrest/immobilization
• Administration of intravenous fluids
• Frequent neurologic checks
• Strict recording of fluid intake and output
• Discharge planning and teaching
Initial Evaluation at Admission after Catheterization (“Quick look” assessment)
• Evaluate general appearance, color, perfusion
• Evaluate airway, oxygenation, ventilation including color, oxyhemoglobin saturation, respiratory effort
• Evaluate systemic perfusion: heart rate and rhythm, four extremity pulses, blood pressure, skin color, skin temperature, capillary refill
• Check catheterization site(s)
• Evaluate and monitor temperature (core and peripheral)
• Evaluate hydration
• Evaluate neurologic function, responsiveness, and movement
• Check all infusion sites
1 Potential Arrhythmias, Decreased Cardiac Output
• Related to injury to conduction system, physiology stress of catheterization, underlying cardiac disease, reaction to sedation/anesthesia or contrast agent, hypothermia. Arrhythmias may be more common in patients with single ventricle anatomy, or severe/critical aortic stenosis.336
• Common arrhythmias include: tachycardia/supraventricular tachycardia (SVT), bradycardia, heart block (particularly with AV discordance, d-transposition of the great arteries [d-TGA], and tetralogy of Fallot [TOF]),336 or severe right ventricular outflow tract obstruction. Ventricular fibrillation may develop in patients with aortic stenosis or pulmonary atresia with intact ventricular septum.664
Expected Patient Outcomes
Stable cardiac output as measured by adequate BP, good peripheral perfusion, regular/appropriate cardiac rate and rhythm, intimal bleeding from catheterization site, adequate urine output (average, approximately 1-2 mL/kg per hour), absence of systemic or pulmonary venous congestion, normal body temperature
Nursing Interventions
• Obtain baseline ECG on arrival to unit and continuously (or per unit routine). If arrhythmia develops:

Monitor for changes in ECG, ECG axis, or ST-segment changes, palpitations, chest pain, and shortness of breath

Note effect on systemic perfusion (note if BP drops with aberrant beat)

Note any precipitating or alleviating factors, and response to medications

Notify provider

Administer oxygen and support perfusion as needed

SVT/tachycardia: Anticipate adenosine or cardioversion

Bradycardia/heart block

Ensure adequate oxygenation and ventilation

Determine if it is a first-, second-, or third-degree heart block.

Anticipate the administration of medications, and initiate pacemaker therapy.
• Assess heart rate: Ascertain if it is adequate for clinical condition (see Table 8-12 earlier in this chapter and pages inside front cover); notify physician and on-call provider if heart rate is excessive or insufficient
• Assess vital signs, including body temperature
• Assess apical and peripheral pulses for strength, regular rate, and rhythm. If any irregularities or pulse discrepancies exist, notify physician, assess perfusion, and obtain rhythm strip. Be prepared to provide CPR.
• Assess central and peripheral perfusion and blood pressure every l5 min initially, then every 1-2 h as appropriate or per unit routine. Notify on-call provider of evidence of poor perfusion or hypotension and treat
• Assess level of consciousness
2 Potential Bleeding/Cardiac Tamponade
Related to vessel rupture or cardiac perforation, embolization of an implanted device and subsequent heart or vessel damage. When caring for a child who has undergone placement of a device such as ASD/VSD/transcatheter valve implant/endovascular stent, device migration and internal bleeding must be considered if the patient develops signs of hemodynamic compromise or decreased perfusion.
Expected Patient Outcomes
Stable cardiac output as measured by adequate blood pressure, normal heart rate and rhythm, normal peripheral pulses and perfusion, normal hemoglobin/hematocrit, absence of signs of cardiac tamponade, absence of hemoptysis, or hematuria (consider retroperitoneal bleed)
Nursing Interventions
• Monitor hemoglobin and hematocrit as ordered
• Closely monitor systemic perfusion and blood pressure for evidence of low cardiac output—tachycardia; cool, clammy extremities; decreased urine output; change in behavior or responsiveness; cyanosis, mottling, or pallor; evidence of pulmonary edema; decreasing blood pressure. Notify physician/on-call provider immediately if they develop.
• Monitor for signs of cardiac tamponade—pallor, tachycardia, decreased blood pressure or decreased pulse pressure, decreased heart sounds, restlessness, cool extremities, tachypnea, neck vein distension, hypotension not responsive to fluid administration. Notify physician/on-call provider immediately if they occur and be prepared for emergency measures if necessary.
Note: Signs of tamponade may be virtually identical to the signs of shock. Pulsus paradoxus may be impossible to appreciate if child is tachypneic or hypotensive.
3 Potential Alteration in Body Temperature
Related to cool temperatures in catheterization suite and flushing of catheters with cold fluids during the procedure.664
Expected Outcomes
Patient will be kept warm during and after cardiac catheterization, and core and peripheral temperature will be normal.
Nursing Interventions
• Assess temperature with vital signs. Keep patient warm but avoid hyperthermia.
• Monitor for signs of hypothermia, including, bradycardia/hypotension, hemodynamic compromise because of vasoconstriction, lethargy/CNS depression, prolonged recovery from sedation or anesthesia, peripheral cyanosis, metabolic acidosis
4 Potential Site Complications: Bleeding/Compromised Perfusion to Extremity
• Related to anticoagulation, arterial access, larger sheath size, especially for device implantation such as aortic/pulmonary stents, device closures, or transcatheter valve implantation.
• Treatment of coarctation requires a larger sheath in the aorta/femoral artery.
• Bleeding from catheterization site may cause hematomas, weakened pulse, or venous/arterial congestion. Arterial compromise can lead to long-term issues with growth of the affected extremity and significant ischemia; may ultimately require amputation of extremity if allowed to progress. Vigilant monitoring and prompt treatment are important.
• The incidence of arterial spasm and vessel thrombosis increases with smaller children (<5 kg) larger sheaths, and longer vessel access times.
Expected Patient Outcomes
Perfusion of catheterized extremity remains adequate as indicated by: warmth, brisk capillary refill, pink color, strong pulses, appropriate movement and sensation (use opposite limb for comparison)
Nursing Interventions
• Monitor catheterization site and dressing for evidence of bleeding. If bleeding is excessive and does not stop with application of pressure, notify physician/on-call provider immediately.
• If bleeding occurs, apply pressure one to two fingers cephalad to (above) catheterization site, remove the dressing to inspect the site while maintaining pressure, hold pressure until hemostasis is achieved, while assessing peripheral pulses frequently during this time to ensure adequate perfusion to extremity. If pressure device is present (i.e., Safeguard), inflate device to apply pressure while palpating distal pulse to the extremity.
Note: Many institutions have protocols for management of pressure devices.
• Maintain bedrest for 4 to 8 hours as ordered. Head of bed can be elevated 30-45 degrees as per institution protocol.
• Monitor for arterial compromise: Often results in cool, pale, mottled extremity. If any of these symptoms occur, notify physician/on-call provider immediately.

Monitor pulses of extremity distal to catheterization site; notify physician immediately of any decrease in pulses (if spasm or thrombus occurs in artery, distal artery can rapidly become thrombosed, and ischemia of extremity will result)

Assess color and warmth of extremity

Application of heat to contralateral extremity may help maintain circulation to catheterized extremity (by producing reflex vasodilation), but heat should never be applied to involved extremity because it merely increases O
2 consumption of already compromised tissue

If thrombus develops in artery, surgical removal may be required

Anticipate anticoagulation therapy such as Heparin infusion, Lovenox injections. If administered early may prevent thrombus formation. Streptokinase or tPA may be used if pulse does not return.
336
Attempt to prevent flexion of catheterized extremity at catheterization site for 6 hours or as ordered

Maintain bed rest for 6 to 12 hours after catheterization (or as ordered)

Administer pain medication as ordered (and needed); monitor patient's response and systemic perfusion

Monitor for evidence of excessive edema or bleeding at catheterization site; notify physician if bleeding is not stopped by application of pressure

Apply ice to catheterization site as needed and ordered
• Monitor for venous compromise: Often results in signs of venous congestion, such as edema, extremity discoloration (extremity becomes dusky), increase in size of extremity.
Note: When venous cutdown is performed, vein used for the catheterization is often tied off at end of procedure, especially in small infants. In this case, extremity distal to catheterization site is likely to become edematous and slightly cyanotic as venous blood is trapped in extremity; collateral veins will quickly provide venous drainage, but initial discomfort should be expected.
• Monitor pulses of extremity distal to catheterization site
• If edema is present, elevate extremity to facilitate venous return; notify physician immediately if edema causes decrease in arterial pulses (indicates compromise of arterial circulation)
• Monitor for evidence of bleeding at catheterization site and notify physician if it is not relieved by pressure
• Maintain bed rest for 4 to 6 hours after catheterization (as ordered)
• Monitor for evidence of arteriovenous fistula: AV fistulas are rare but can develop after cardiac catheterization, particularly in older children. Smaller AV fistulas are usually asymptomatic, but larger communications may cause symptoms and may require treatment.
 Monitor for pain, development of large hematoma/bruise, presence of visible veins on surface of leg, leg swelling, palpation of a bruit, or auscultation of shunt at the catheterization site
Monitor for pain, development of large hematoma/bruise, presence of visible veins on surface of leg, leg swelling, palpation of a bruit, or auscultation of shunt at the catheterization site
 Monitor perfusion to extremity
Monitor perfusion to extremity
 Provide pain management
Provide pain management
 Anticipate further treatment involving coil embolization or surgical ligation
Anticipate further treatment involving coil embolization or surgical ligation
5 Potential Respiratory Compromise
Related to sedation or general anesthesia, precatheterization respiratory compromise or upper airway obstruction (e.g., Down syndrome, velocardiofacial syndrome), postextubation upper airway edema, or pulmonary edema associated with reperfusion syndrome (e.g., with tetralogy of Fallot, pulmonary atresia, and other diagnoses after pulmonary artery angioplasty or stent implantation to increase pulmonary blood flow)
Expected Patient Outcomes
Adequate respiratory function as demonstrated by appropriate rate and effort, equal and adequate lung aeration bilaterally, appropriate oxyhemoglobin saturation and heart rate for patient
Nursing Interventions
• Monitor respiratory rate (see pages inside front cover) and effort. Notify physician/on-call provider if rate or effort is insufficient or excessive; respiratory support (oxygen therapy, ventilation) may be necessary
• Monitor for airway obstruction: Snoring, nasal flaring, retractions, gasping for breath, use of accessory muscles. Reposition head (head tilt/jaw thrust), consider need for oxygen
• Monitor for laryngospasm: Agitation, stridor, hypoxia, diminished/absence of breath sounds. Provide positive-pressure ventilation via bag/mask and notify physician/on-call provider immediately
• Monitor oxyhemoglobin saturation: Notify physician/on-call provider of inadequate/inappropriate oxyhemoglobin saturation. Consider assessments compared to the baseline status prior to catheterization procedure and potential presence of single ventricle circulation.
• Auscultate lungs: Notify physician/on-call provider of inadequate aeration, ventilation
• Monitor CXR for atelectasis, symptoms of pulmonary congestion
• If reperfusion syndrome is suspected, anticipate administration of oxygen, diuretics, aggressive pulmonary toilet, or mechanical ventilation
6 Potential Dehydration/ Potential Compromise in Renal Function
Related to inadequate fluid intake before and during procedure, diuresis, or nephrotoxicity caused by contrast agents. Patients with cyanotic heart disease are at increased risk for development of hemoconcentration if dehydrated.
Expected Patient Outcome
Patient will demonstrate adequate urine output (1-2 mL/kg per hour)
Nursing Interventions
• Monitor urine output; notify physician if urine output is inadequate despite sufficient fluid intake
Note: A small child may rapidly become dehydrated when kept NPO for hours while awaiting catheterization; ensure adequate parenteral and/or oral fluid intake before, during, and after catheterization.
• Typically, generous fluid administration is provided (if tolerated) to facilitate urinary excretion of contrast agent. If patient is oliguric despite sufficient intake, restriction of fluid intake may be needed to prevent overload.
• If the patient has a persistent right to left intracardiac shunt, no air can be allowed into any IV system due to the risk for cerebral or systemic embolization.
• Assess fluid balance; notify physician or on-call provider of imbalance
• Test urine for hematuria; notify physician if blood is present and monitor urine output closely
6 Potential Allergic Reaction
Related to contrast agent, catheters, medications, equipment. Most occur during or in the early postcatheterization period. Latex allergies should be considered in some patients. Many pediatric catheterization suites now use low osmolality/nonionic agents, yet contrast agents can still result in pulmonary compromise/edema.
Expected Patient Outcomes
Patient will remain free of symptoms of allergic reaction
Nursing Interventions
Monitor for signs of allergic reaction: changes in skin color, rash, hives, itching, low-grade fever, diaphoresis, respiratory difficulty, vasodilation, and hypotension. If allergic reaction is suspected, notify physician/on-call provider immediately, administer oxygen if necessary, anticipate administration of antihistamines, treat specific symptoms (i.e., support airway, oxygenation, ventilation, perfusion)
7 Potential Neurologic Compromise
Related to embolization of thrombus, air or device, cyanotic heart disease, and polycythemia. Potential for neurologic event increases with multiple catheter exchanges, use of large sheaths, polycythemia with iron deficiency anemia, and procedures involving left heart, head and neck vessels, aorta, and right-to-left intracardiac shunts.
Expected Patient Outcomes
Patient will respond in age-appropriate and developmentally appropriate manner
Nursing Interventions
• Document baseline neurologic status
• Immediately report failure to progress to baseline movement, speech, activity
• Frequently assess neurologic function: Notify physician/on-call provider if changes. Note that increased somnolence may indicate neurologic compromise.
• Monitor for seizure activity; notify physician/on-call provider immediately if present
8 Potential Knowledge Deficit/Comfort/Anxiety
Many cardiac catheterization patients are admitted and discharged within 24 h. Extensive teaching is necessary in a short time period. Postprocedure patients are often awake but with enforced limited mobility, which can be challenging for the nurse, patient, and family. Pain may be present.
Expected Patient Outcome
Patient/family possesses adequate information to comply with postcatheterization care regimen and general health maintenance
Nursing Interventions
• Assess level of pain and provide medication as ordered
• Offer child services where available
• Assess family's understanding of diagnosis, procedure and necessary care. Provide additional teaching if necessary, consult with support services such as social work where applicable
• Provide child/family with appropriate instruction regarding wound care, physician and other follow-up appointments and emergency telephone numbers, signs of infection, activity restrictions (if any), and medications
• Discuss implications of catheterization results with patient/family to obtain their perceptions of physician's recommendations and clarify any misconceptions they may have
• Review appropriate diet with child/family as indicated
9 Potential for Infection Caused by Invasive Procedure, Wound
Expected Patient Outcome
Patient will remain free of symptoms of infection, including fever or temperature instability, leukocytosis, erythema or drainage at catheterization site, evidence of endocarditis or pericarditis
Nursing Interventions
• Monitor catheterization site for edema, erythema, heat, or discharge; notify physician if present
• Monitor patient's temperature; blood cultures are usually recommended if fever higher than 101.3° F (38.5° C)
• Inform family that a low-grade fever is not uncommon 24 to 48 hours after cardiac catheterization. Notify physician if fever persists or temperature is higher than 100.4° F (or accepted parameters per institution).
• Monitor white blood cell (WBC) count and platelet count if infection is suspected
• Monitor for evidence of endocarditis (high fever, appearance of new heart murmur, hematuria) and pericarditis (cardiac friction rub, loss of heart tones, ECG changes)
Box 8-55 Intervention-Specific Nursing Management Following Cardiac Catheterization*