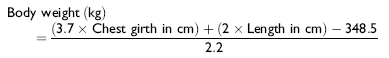Chapter 9 Alterations in Body Weight or Size
Slowed growth and below-normal weight gain usually happen at the same time, although occasionally they develop separately. By definition a decrease in growth and weight gain is limited to the growing animal. Similar pathogenic mechanisms cause weight loss or an emaciated condition in an adult patient. This arbitrary age division allows the clinician to consider possible causes that are more or less common for a given age-group.
Potential growth and weight gain are genetically determined. They differ according to species, breed, and sex, and marked differences in potential growth exist within a breed. The potential for growth in ruminants is greater in the offspring of multiparous females than in those from primiparous females. The normal or minimum growth and weight gain rates for common breeds of the various large animal species are outlined in the section on assessment of growth and weight gains.
MECHANISMS OF DECREASED GROWTH AND DECREASED WEIGHT GAIN
Major pathogenic mechanisms that result in decreased growth and decreased weight gain include the following:
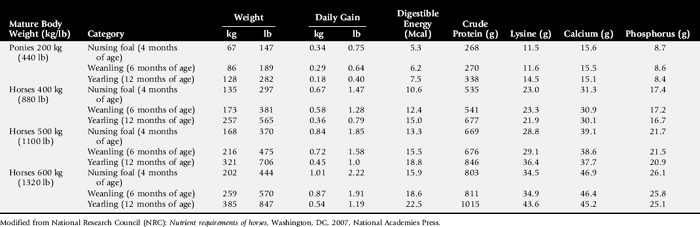
Inadequate intake of one or more essential nutrients is an important cause of decreased growth. In many cases growing animals are not provided with a sufficient volume of feed to meet their nutrient requirements. Young animals rely on a highly digestible diet that provides energy and essential nutrients for growth. Even animals fed an appropriate volume of a poor-quality milk replacer could suffer from poor growth. Milk replacers formulated with sources of protein, fat, vitamins, and minerals that have limited nutrient digestibility may induce a state of energy, protein, vitamin, or mineral malnutrition. For some young animals, poor-quality forage is the only feed available. Weaned foals and ruminants rely on forages and cereal grains to provide essential nutrients. Hay that has been harvested at a late stage of growth usually has a lower nutrient digestibility compared with young forages. Diets low in digestible energy or protein or both reduce total daily intake in ruminants (Table 9-1) because of the increased turnover time (T½) in the gastrointestinal tract and subsequent decreased throughput. This compounds the problems caused by an inadequate intake of digestible nutrients. The digestibility of forages is even lower for horses than for ruminants.
Table 9-1 Maximum Dry Matter Intake (DMI) Related to Forage Quality for Cattle
| Forage Quality | Maximum DMI/Day (% Body Weight) | Maximum DMI for 500 kg/Cow/ Day (kg) |
|---|---|---|
| Poor | 1–1.5 | 5–7.5 |
| Oat straw | ||
| Corn stover | ||
| Average | 2 | 10 |
| Meadow grass hay | ||
| Excellent | 2.5 | 12.5 |
| Alfalfa hay (25% crude fiber) | ||
| Corn silage |
From Maas J: Relationship between nutrition and reproduction in beef cattle, Vet Clin North Am, 3:634, 1987.
Protein-calorie malnutrition (PCM) is the most common clinical cause of decreased growth and decreased weight gain in young animals. It is characterized by smaller size and lower weight than the normal minimums for age, breed, and sex. Inadequate intake of digestible energy and protein (or essential fatty acids in the neonate primarily adapted to a milk diet) results in inadequate levels of amino acids, fats, and carbohydrates for normal metabolism and growth. Diets that lack any of the other essential nutrients (fatty acids, vitamins, macrominerals, or trace minerals) can also decrease growth. Deficiencies of calcium, phosphorus, and magnesium result in improper skeletal formation. Deficiencies in other macrominerals (e.g., sodium, chloride, potassium), trace minerals (e.g., copper, zinc, manganese, cobalt, iron), and vitamins (e.g., A, D, E, thiamin) cause biochemical dysfunctions that lead to inefficient metabolism and growth. Large animal patients that grow slowly as a result of inadequate diets often have normal or increased appetites until they are terminally ill. Physical findings and clinicopathologic data from animals with PCM often are within the normal range until the disease process is well advanced.
Infections or inflammatory processes are important causes of decreased growth and decreased weight gain in young horses and ruminants. The decrease in growth can be of short duration followed by recovery and compensatory gain (cryptosporidiosis) or can persist (chronic bronchopneumonia). Infections or inflammatory processes can also result in nutrient malabsorption (chronic salmonellosis, acute rotavirus diarrhea), anorexia (pharyngeal abscesses), increased nitrogen turnover, and direct protein losses (gastrointestinal disease). Energy or protein requirements may be increased as a result of infection and inflammation.
Parasitism often affects young horses and ruminants and results in decreased growth and decreased weight gain by increasing nutrient requirements, increasing nutrient losses, and/or decreasing nutrient absorption. The animal’s metabolic rate and nutrient requirements may also increase as a result of inflammatory and immune reactions that arise secondary to parasitism.
Genetic diseases (α-mannosidosis, dwarfism) result in decreased growth through generalized errors in the genetic code or interference with strategic reactions in one or more metabolic pathways. Congenital cardiac malformations (tetralogy of Fallot, interventricular septal defect) create physiologic inefficiencies that require energy beyond the body’s ability to supply it. Congenital renal disease (agenesis, dysplasia, hypoplasia, polycystic kidney disease) affects homeostatic mechanisms that regulate electrolyte and acid-base balance, results in the production of uremic toxins, and often results in partial anorexia and PCM. Digestive tract malformations including cleft palate, megaesophagus, and brachygnathism can reduce nutrient ingestion and impair growth.
Toxicities, although rare in growing animals, result in decreased weight gain by interfering with metabolic pathways (e.g., ammonia toxicity, zinc-induced copper deficiency with abnormal skeletal development in foals), by causing loss of body reserves (e.g., thiamin deficiency in horses, bone marrow hypoplasia and associated bleeding diatheses in ruminants associated with bracken fern toxicity), by inducing anorexia, or by a combination of mechanisms. The pathogenic mechanisms of many toxins are not yet known.
Environmental factors including extreme heat or cold or high humidity result in decreased growth and decreased weight gain. Extremely cold conditions increase an animal’s daily energy requirements. During extremely hot weather feed intake often decreases, which may contribute to decreased growth. Often, environmental conditions influence the development of disease, resulting in a subsequent increase in nutrient requirements in a growing animal (e.g., calves with PCM housed in poorly ventilated or overly humid conditions become much more susceptible to infectious pneumonia).
In many cases a combination of these diverse factors may influence the growth and weight gain of young animals. A period of increased growth rate and weight gain, called compensatory gain, often occurs after a period of restricted growth. In growing foals, compensatory gain should be closely monitored to prevent excessively rapid growth and abnormal skeletal development.
Boxes 9-1 and 9-2 list many of the possible causes of decreased growth and decreased weight gain in horses and ruminants, respectively.
Box 9-1 Causes of Decreased Growth and Decreased Weight Gain in Horses
Box 9-2 Causes of Decreased Growth and Decreased Weight Gain in Ruminants
Approach to the Diagnosis and Management of Decreased Growth and Decreased Weight Gain in Horses
Table 9-21 Body Condition Scoring System for Horses
| Score | Description |
|---|---|
| 1 | Poor. Animal is extremely emaciated. Spinous processes, ribs, tailhead, tuber coxae, and tuber ischii project prominently. Bone structure of the withers, shoulders, and neck is noticeable. No fatty tissue can be felt. |
| 2 | Very thin. Animal is emaciated. There is a slight fat covering over the base of the spinous processes; the transverse processes of the lumbar vertebrae feel rounded. Spinous processes, ribs, tailhead, tuber coxae, and tuber ischii are prominent. Bone structure of the withers, shoulders, and neck is faintly discernible. |
| 3 | Thin. Fat buildup is present about halfway on the spinous processes; the transverse processes cannot be felt. There is a slight fat cover over the ribs. Spinous processes and ribs are easily discernible. Tailhead is prominent, but individual vertebrae cannot be visually identified. Tuber coxae appear rounded but are easily discernible; tuber ischii are not distinguishable. Bone structure of the withers, shoulders, and neck is accentuated. |
| 4 | Moderately thin. Negative crease can be seen along the back. Faint outline of ribs is discernible. Tailhead prominence depends on conformation; fat can be felt around tailhead. Tuber coxae are not discernible. Withers, shoulders, and neck are not obviously thin. |
| 5 | Moderate. Back is level. Ribs cannot be visually distinguished but can be felt easily. Fat around tailhead is somewhat spongy. Withers appear rounded over spinous processes, and shoulders and neck blend smoothly into the body. |
| 6 | Moderately fleshy. Slight crease may be seen down the back. Fat over ribs is spongy, and fat around tailhead is soft. Fat is beginning to be deposited along withers, behind shoulders, and along neck. |
| 7 | Fleshy. Crease may be seen down the back. Individual ribs can be felt, but there is noticeable filling of fat between ribs. Fat around tailhead is soft. Fat is deposited along withers, behind shoulders, and along neck. |
| 8 | Fat. Crease is seen down the back. Ribs are difficult to feel. Fat around tailhead is very soft. Areas along withers and behind shoulders are filled with fat, and neck is noticeably thickened. Fat is deposited along inner thighs. |
| 9 | Extremely fat. Obvious crease is seen down the back. Patchy fat appears over ribs. Bulging fat is seen around tailhead along withers, behind shoulders, and along neck. Fat along inner thighs may cause thighs to rub together. Flank is filled with fat. |
From Henneke GD, Potter GD, Kreider JL, Yeates BF: Relationship between condition score, physical measurements and body fat percentage in mares, Equine Vet J, 15:371, 1983.
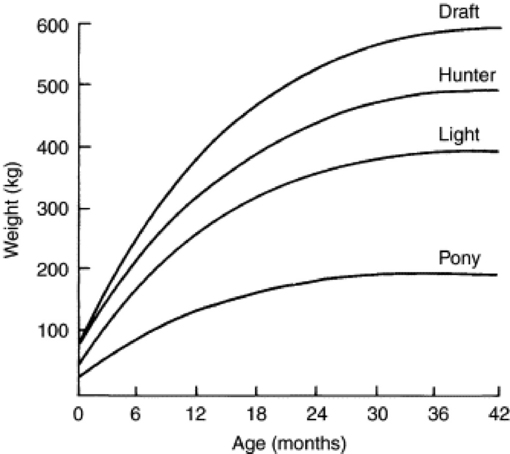
Fig. 9-1 Estimated weight gain of horses of various mature body weights.
Modified from National Research Council [NRC]: Nutrient requirements of horses, Washington, DC, 1978, National Academy of Sciences, NRC.
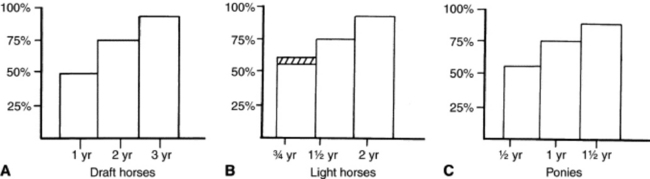
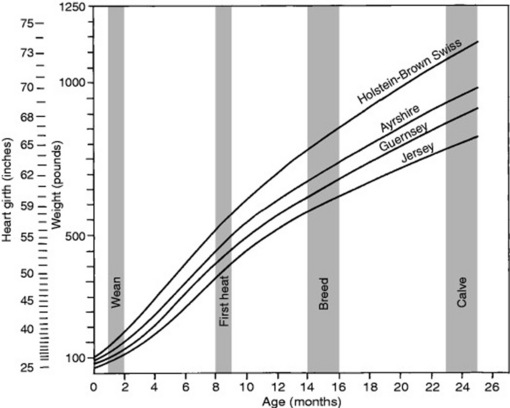
Fig. 9-2 A to C, Body weight as a percentage of mature body weight for horses at a given age.
A modified from Crampton WW: J Agric Hortic 26:172, 1923; B modified from Lewis LD: Feeding and care of the horse, Philadelphia, 1982, Lea & Febiger; C modified from Hintz HF: Factors affecting the growth rate of horses, Horse Short Course Proceedings, Texas A&M Animal Agriculture Conference, 1979, College Station, Texas.
What is the consistency of the feces? Refer to Chapter 20 for the diagnosis and management of neonatal diarrhea; refer to Chapter 7 if the foal is older and has evidence of diarrhea. Is there evidence of sand in the manure? Perform a fecal egg count. If the foal has a positive fecal egg count, follow the parasite control program recommendations in Chapter 49. If a negative fecal egg count is reported but parasitic infestation is still suspected, repeat the test in 2 to 3 weeks or follow the deworming protocols in Chapter 49. Evaluate the feces occult blood. If the foal has a positive fecal occult blood test, review the medical management for melena in Chapter 7.
If the foal is consuming a milk replacer, review the guaranteed analysis for the nutrient content of the product. Review the mixing instructions with the owner or farm manager. Develop a feeding program appropriate for the foal’s age.
The most accurate way to determine the nutrient content of forage or pasture is with an analysis. Forage sampling instructions and forage analysis companies are listed in Boxes 9-3 and 9-4. University Extension services often provide a forage analysis service. If the client does not purchase a large volume of hay, or if analysis cannot be performed, forage tables from the Nutrient Requirement Council reference books (www.nap.edu) or nutrient tables from the Equi-Analytical Laboratories forage laboratory database (www.equi-analytical.com) can be referenced to estimate the concentration of different nutrients in common forages and supplemental feeds. Use the daily nutrient requirement table (see Table 9-3) to recommend the type and amount of forage the foal should consume based on the nutrient content of the forage.
Box 9-3 Forage and Large Animal Feed Sampling Instructions
SAMPLING PASTURE
SAMPLING HAY USING A CORE HAY SAMPLER
SAMPLING HAY BY HAND
SAMPLING GRAIN OR PELLETED FEED
The guaranteed analysis on the feed tag label provides the nutrient content of certain ingredients. Contact commercial feed companies for the energy content of their product. Make recommendations about the appropriate use of commercial equine feeds, grain, or grain mixes for young growing foals based on the clinical health of the foal.
If the cause of the decreased growth and/or poor weight gain has not been determined, additional diagnostic tests should be performed. Possible tests include, but are not limited to ultrasound, radiographs, serum or whole blood trace mineral analysis, and carbohydrate absorption tests (oral D-glucose, D-xylose).
Approach to the Diagnosis and Management of Decreased Growth and Decreased Weight Gain in Ruminants
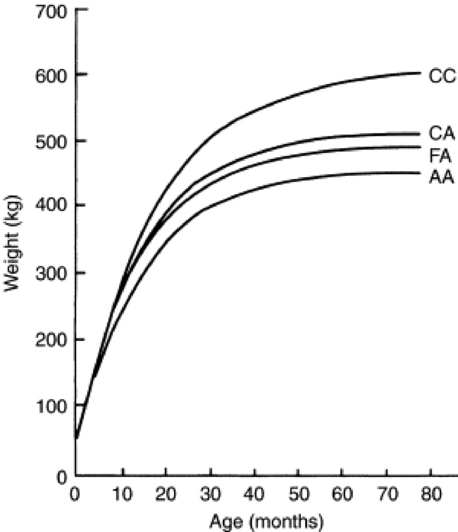
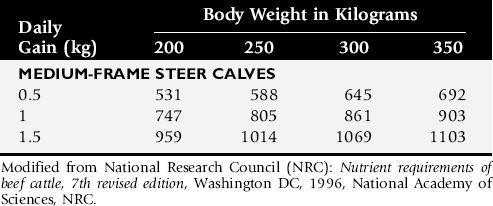
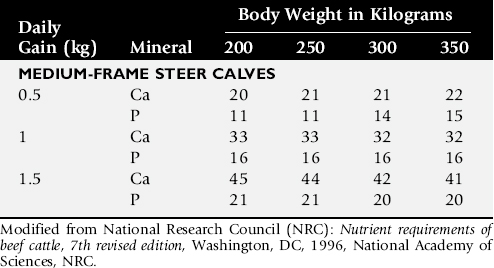
Fig. 9-4 Estimated growth curves for beef cows of various breeds. AA, Angus; CC, Charolais; CA, Charolais × Angus; FA, Holstein × Angus.
Modified from Nadarajah K, Marlowe TJ, Notter DR: Growth patterns of Angus, Charolais, Charolais X Angus and Holstein X Angus cows from birth to maturity, J Anim Sci 59:957, 1984.
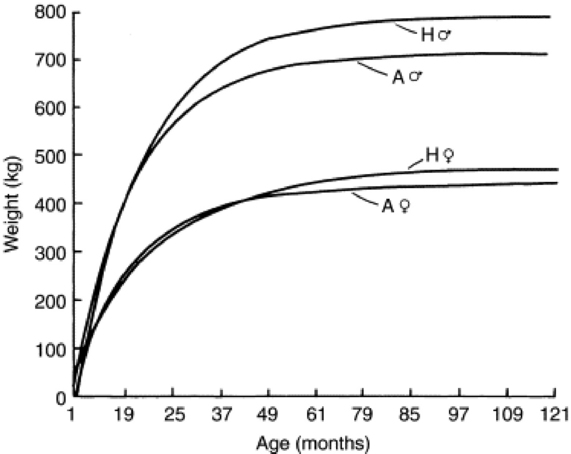
Fig. 9-5 Mean growth curves of Angus (A), and Hereford (H), males ( ) and females (
) and females ( ).
).
Modified from Brown JE, Brown CJ, Butts WT: A discussion of the genetic aspects of weight, mature weight and rate of maturing in Hereford and Angus cattle, J Anim Sci 34:525, 1972.
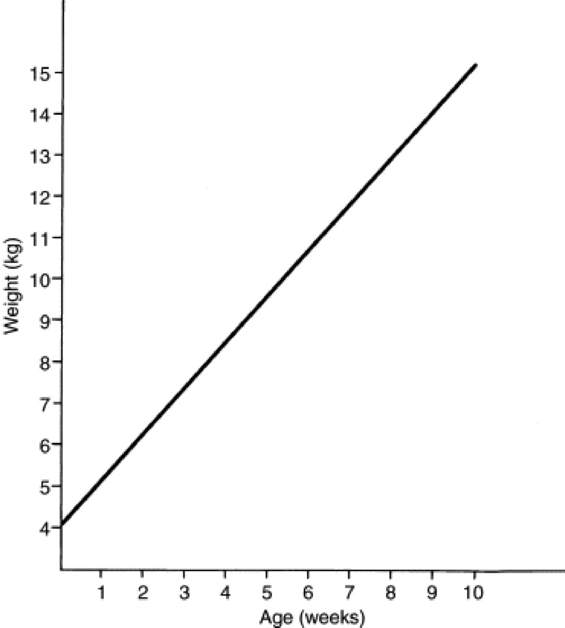
Fig. 9-6 Growth curve for young goats.
Modified from Morand-Fehr P, Hervieu J, Bas P, Sauvant D: Proc Third Int Conf Goat Prod Dis 3:96, 1982.
Perform flotation, sedimentation, and Baermann’s procedures to detect patent parasitic infestation. Perform a fecal occult blood test; if the result is positive or if there is evidence of diarrhea, see the section on melena or diarrhea in Chapter 7. If diarrhea is noted in neonatal calves, refer to Chapter 20 for diagnostic and therapeutic management.
Hypophosphatemia may be the result of dietary deficiency or Brassica, feeding, or it may be associated with copper deficiency. Measure serum (plasma) copper if a herd problem exists in a copper-deficient region (or a region with excess molybdenum or sulfate or both). Copper (serum or plasma) concentrations below 0.5 μg/mL (ppm) indicate deficiency. Liver copper levels are even more indicative of status. Serum glucose may be increased with stress or decreased or normal near death.
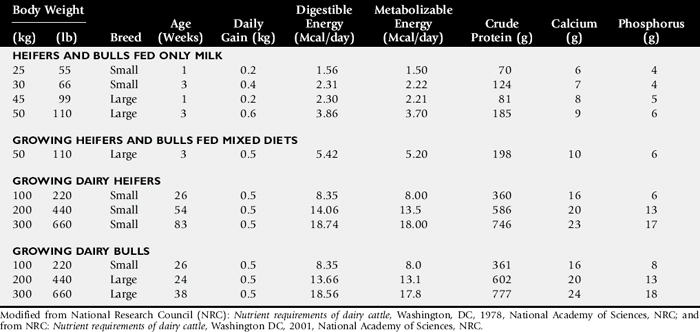
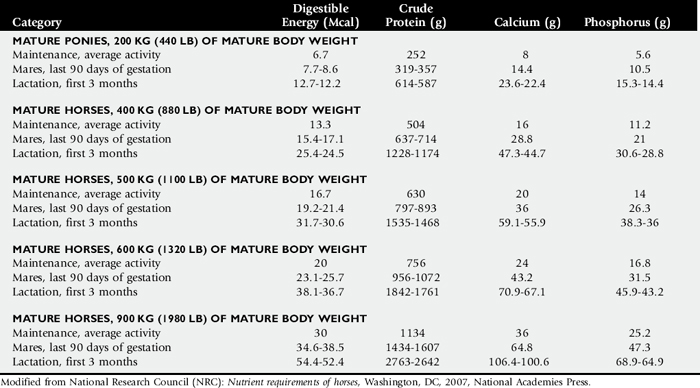
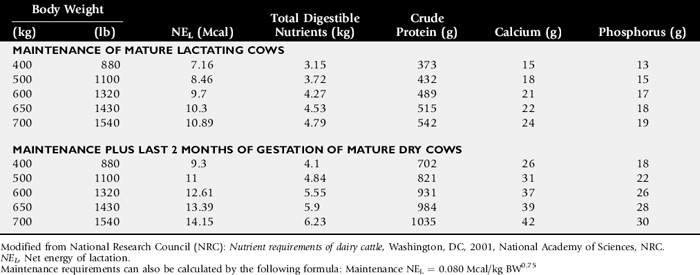
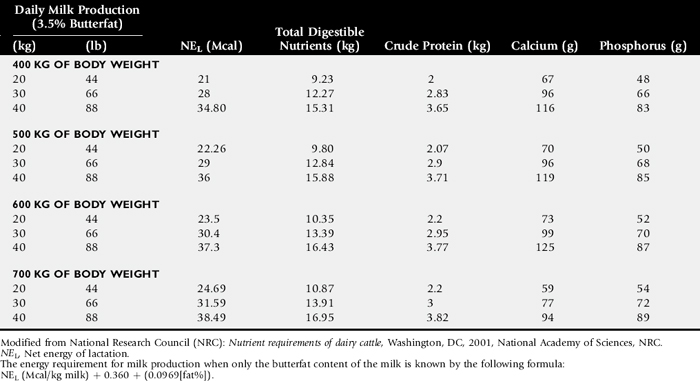
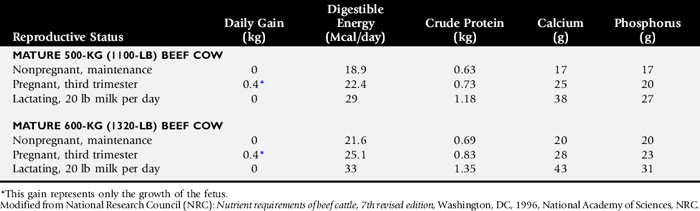
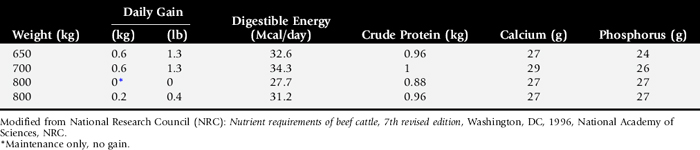
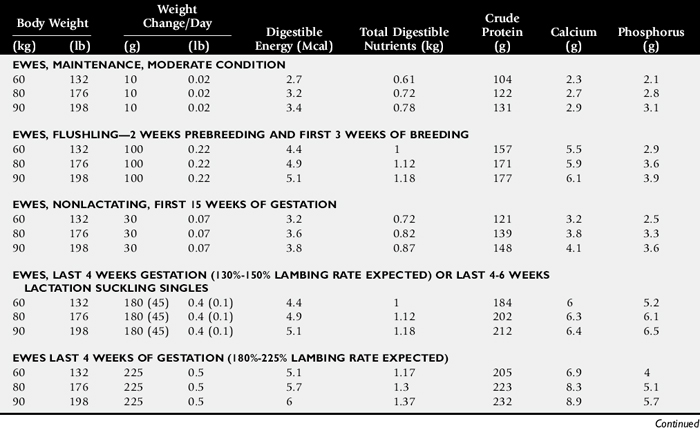
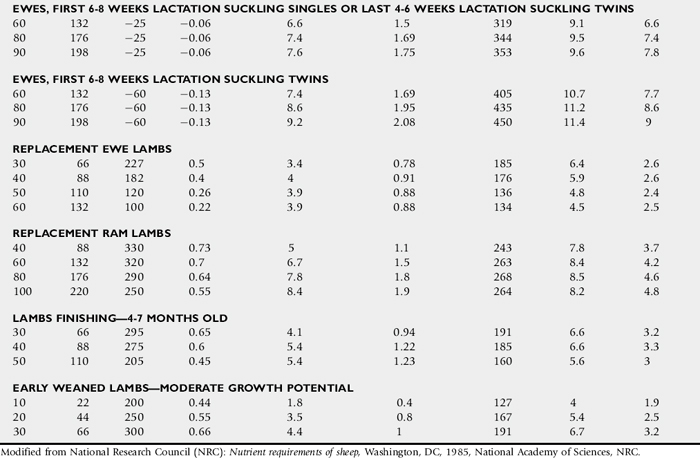
Compare nutrient intake with the requirements for maintenance and growth of the various ruminant species (Tables 9-4 through 9-10). If the neonate is being fed a milk diet, evaluate the quality of the product and ensure that the animal’s intake meets the dietary requirements (see Tables 9-4 and 9-5). Ensure that the milk replacer is mixed properly. If the ruminant is consuming a grain mix or forage, ensure that the quantity and the quality of the feed are adequate to allow sufficient intake in developed ruminants (see Table 9-1). Forage sampling instructions are listed in Box 9-3. If anorexia is present, look for more specific signs of a primary disease process. If the diet supplies adequate nutrients for maintenance and growth, consider decreased growth and decreased weight gain to be caused by a primary disease condition.
Table 9-4 Daily Energy and Protein Requirements for 50-kg Calves on a Milk Diet
| Digestible Energy Requirements | Digestible Protein Requirements | |
|---|---|---|
| Maintenance | 45–55 kcal/kg body weight | 0.5 g/kg body weight |
| Gain | 300 kcal/100 g gain in body weight* | 22 g/100 g of weight gain† |
| 0.5 kg daily gain | 1500 kcal | |
| 1 kg daily gain | 3000 kcal |
* A 50-kg calf gaining 0.75 kg/day would have a daily energy requirement of 5000 kcal of digestible energy (2750 kcal maintenance + 2250 kcal/0.75 kg of gain).
† A 50-kg calf gaining 0.75 kg/day would have a daily protein requirement of 190 g of digestible protein (25 g of maintenance + 165 g of gain).
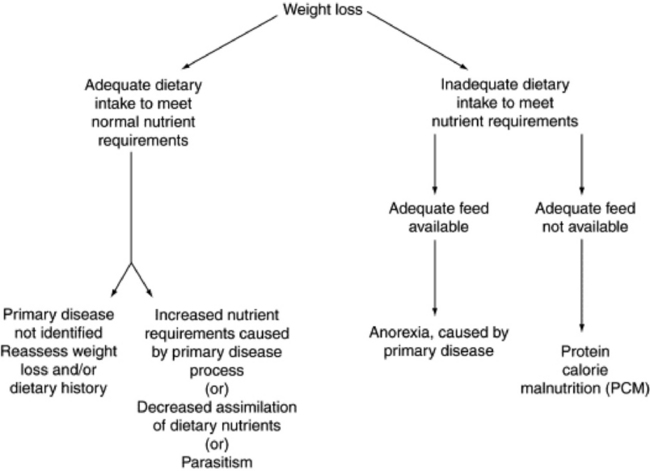
WEIGHT LOSS
The clinical problem of weight loss suggests that an individual large animal patient or a herd has lost weight over a known period of time. It may also suggest that the patient has reached a subnormal adult weight and size (see section on decreased growth and decreased weight gain, earlier). Late pregnancy, early lactation, and intense exercise are normal physiologic conditions commonly accompanied by mild to moderate weight loss. Late pregnancy can be associated with decreased body condition without actual weight loss, because weight is gained with the conceptus. During pregnancy and lactation the loss of body condition may be mild, resulting in a low normal BCS, or the loss may be severe and can threaten the health of both the dam and the neonate.
Weight loss in adult animals is most commonly associated with one or more of the following circumstances (other causes are listed in Boxes 9-5 and 9-6):
Anorexia usually occurs secondary to a primary disease. Increased nutrient requirements are associated with normal physiologic conditions (e.g., pregnancy, lactation, exercise, cold weather) and with pathologic processes (e.g., sepsis, trauma, parasitism, burns). Mild to severe PCM is often associated with inadequate feed quality (see Table 9-1) or quantity but can also be caused by increased energy requirements resulting from adverse environmental conditions. Weight loss can also occur with a deficiency of essential micronutrients such as copper, cobalt (vitamin B12), or vitamin A. Parasitism should always be on the differential list in an animal that has lost weight.
Mechanisms of Weight Loss
Anorexia is the loss of appetite or lack of desire for food; it may be complete or partial. It is a primary mechanism for weight loss of short or intermediate duration. Weight loss results from decreased nutrient intake. When partial anorexia occurs over a long period, the weight loss may be subtle and go unrecognized. Acute, complete anorexia results in more dramatic weight loss.
In domestic species, anorexia is usually associated with a primary disease condition and is regulated by cytokines, including interleukin (IL)-1 and tumor necrosis factor alpha (TNF-α), released during an inflammatory response. Resolution of the primary disease process usually results in a return to voluntary food consumption. Anorexia must be differentiated from dysphagia by observation. The distinction between the conditions that cause anorexia and those that control hunger and satiety is not clear; however, many diseases that cause anorexia also result in dehydration, electrolyte imbalances, and/or acid-base disorders.
In addition to causing anorexia, many disease processes cause an increase in the nutrient requirements for basal metabolism. Nutrient requirements for maintenance, growth, pregnancy, lactation, and exercise have been well defined for many large animal species. Nutrient requirements in disease have not been adequately evaluated in large animals, and most information is currently extrapolated from humans, laboratory animals, and small animal species. In human patients, published estimates indicate that requirements for energy and protein increase approximately 10% after elective surgery, 20% with fractures, 30% to 60% with severe infection or sepsis, 40% with peritonitis, and 50% to 110% with major burns.7,8 In humans, the resting energy expenditure is estimated to increase by 14% for each degree Celsius increase in body temperature.9 Extrapolation of these data directly to equine and ruminant patients is probably not possible; however, the figures do indicate the degree of change in nutrient requirements as a result of disease. The stress of many disease processes results in an increase in serum cortisol and in glucagon. The decreased insulin/glucagon ratio alters the production of glucose and results in hyperglycemia because of enhanced hepatic gluconeogenesis. An increase in sympathetic activity appears to regulate fat oxidation, the increased release of fatty acids from cellular lipid stores, and the development of hypertriglyceridemia in many patients with sepsis. Protein degradation and a negative nitrogen balance are also hallmarks of the acute response to infection. Weight loss resulting from protein and lipid catabolism is often observed in large animal patients with sepsis, owing to altered metabolic activity and nutrient requirements.
In conditions such as burns, peritonitis, pleuritis, colitis, and granulomatous bowel disease, nutrients (particularly proteins) are lost. In many disease conditions, concurrent anorexia and increased nutrient requirements greatly increase the risk of PCM and weight loss. Certain conditions, such as Johne’s disease in ruminants and granulomatous enteritis in horses, are also associated with a malabsorption or malassimilation syndrome. In these types of diseases, nutrients are not efficiently digested and absorbed; anorexia may be absent, and dietary intake may appear normal, but weight loss still occurs.
PCM continues to be a persistent problem in domestic animals. Inadequate ingestion of energy and protein obviously results in weight loss, but PCM and associated weight loss can occur through several other mechanisms. The most direct cause is that the animal receives an inadequate volume of feed to meet their dietary requirements. This can occur as frank underfeeding of all animals or as a consequence of inadequate feeding facilities that create competition among animals for available feed. The latter circumstance occurs most dramatically when animals of varying ages are mixed; the younger animals with the highest requirements are often pushed away by older, dominant individuals.
The quality of the diet, particularly dietary forages such as hay and pasture, is an important factor in the development of PCM and total nutrient intake. Table 9-1 lists guidelines for estimating the maximum daily intake by cattle. It is evident that as forage quality (digestibility) decreases, maximum daily intake decreases because poor-quality feed must remain in the rumen for an increased period of time before it is sufficiently digested to allow passage through the reticuloomasal orifice. Maximum dry matter intake (DMI) as a percentage of body weight is somewhat higher in small ruminants than in cattle. However, the energy requirement per kilogram of body weight is higher in small ruminants than in cattle. Similar estimates for maximum DMI in horses related to forage quality are not available. Horses do not have a pregastric fermentation organ (rumen) and can ingest slightly more of the same quality forage than cattle. Low-quality forages are often the cause of PCM, even when an unlimited quantity is available. The best way to determine the nutrient content of forage, grain, and pelleted feeds is to have the feed analyzed by a forage laboratory. Feed analysis instructions are provided in Box 9-3. Feed tag labels or forage databases can be referenced if a forage analysis is not performed.
Environmental factors can have a major influence on nutrient requirements and can increase the subsequent risk for PCM and weight loss. The most important environmental factor is the ambient temperature. Nutrient requirements for maintenance change with a decreasing ambient temperature as follows:
 Adult horses:, Estimated increase in digestible energy (DE) requirements by 2.5% for every degree Celsius below the lower critical temperature (LCT). The LCT for adult horses ranges from 5° C to −15° C, depending on the horse’s adaptation to the environment. In cold temperatures, when the hair coat is wet, the maintenance DE requirement may be increased by as much as 50%.10
Adult horses:, Estimated increase in digestible energy (DE) requirements by 2.5% for every degree Celsius below the lower critical temperature (LCT). The LCT for adult horses ranges from 5° C to −15° C, depending on the horse’s adaptation to the environment. In cold temperatures, when the hair coat is wet, the maintenance DE requirement may be increased by as much as 50%.10 Beef cattle:, 1% increase in maintenance energy requirements (total digestible nutrients [TDN], net energy of maintenance [NEm], digestible energy [DE], and metabolizable energy [ME]) for each 1° C drop below 20° C (68° F).
Beef cattle:, 1% increase in maintenance energy requirements (total digestible nutrients [TDN], net energy of maintenance [NEm], digestible energy [DE], and metabolizable energy [ME]) for each 1° C drop below 20° C (68° F). Dairy cattle (lactating):, 25% increase in energy requirements (TDN, net energy of lactation [NEL]) as ambient temperature drops from 20° C (68° F) to −10° C (14° F).
Dairy cattle (lactating):, 25% increase in energy requirements (TDN, net energy of lactation [NEL]) as ambient temperature drops from 20° C (68° F) to −10° C (14° F). Sheep with 10-cm wool:, 1% increase in energy requirements (TDN, ME, DE) for each 1° C drop below lower critical temperature (approximately −10° C).
Sheep with 10-cm wool:, 1% increase in energy requirements (TDN, ME, DE) for each 1° C drop below lower critical temperature (approximately −10° C).There are also additive effects of wind and rain that increase energy requirements in large animal species. As nutrient requirements increase, the dietary intake must also increase to prevent weight loss associated with PCM. Horses in inclement weather may not be able to consume enough forage to meet their increased energy requirements, and for these animals, dietary fat and limited grain supplementation may be required.
Deficiencies of micronutrients (trace minerals, B vitamins) often result in inefficiencies in basic biochemical pathways. These inefficiencies, if marked, can be associated with weight loss. Genetic errors in metabolism can cause similar disturbances, but these usually manifest as decreased growth and even death in young animals.
Parasitism is a common cause of weight loss in adult domestic animals. The mechanisms by which parasite infestation can result in weight loss include a loss of body fluid and tissues resulting in increased nutrient requirements, competition for nutrients in the gastrointestinal tract, malassimilation and malabsorption, inflammation resulting in increased nutrient requirements, micronutrient deficiencies, and organ or vascular damage from migrating parasite larvae. Anorexia may also develop in the advanced stages of severe parasitism.
Approach to the Diagnosis and Management of Weight Loss in Adult Horses
Use the flow sheet in Fig. 9-7 to aid in decision making.
If a weight tape will not fit around the girth of the horse, or if the horse has a BCS of 1 to 3, a body weight estimate is made using measurements of the length and girth of the horse.11-12 Length is measured from the tuber ischium to the point of the shoulder, and girth is measured at the withers, behind the elbows, at the end of expiration.
The weight of miniature horses should be obtained using a small animal clinic scale. If a scale is not available to weigh a miniature horse, then the following equation can be used to estimate the weight of the miniature horse.13
What is the consistency of the feces? If the horse has evidence of diarrhea, review the section on diarrhea in Chapter 7. How long are the fibers in the feces? Perform a glove test. Is there evidence of sand in the feces? Horses can have a significant volume of sand in the large intestine while having negative fecal sand test results. Perform a fecal egg count. An enzyme-linked immunosorbent assay (ELISA) may be useful in diagnosing a tapeworm infection. Follow the parasite control program in Chapter 49 if the horse has evidence of fecal parasites or if a parasite infection is suspected despite a negative fecal egg count. Perform a fecal occult blood test; if the result is positive, see the section on melena in Chapter 7.
The most accurate way to determine the nutrient content of forage or pasture is with an analysis. See Box 9-3 for instructions on how to sample feeds for analysis. Contact a university extension service for forage analysis. If the client does not purchase a large volume of hay, or if analysis cannot be performed, forage tables from the Nutrient Requirement Council reference books (www.nap.edu), or nutrient tables from the Equi-Analytical Laboratories forage laboratory database (www.equi-analytical.com) can be used to estimate the concentration of different nutrients in common forages and supplemental feeds.
If the weight loss is not caused by inadequate or poor-quality feed, additional diagnostic tests should be performed. Possible tests include but are not limited to ultrasound, gastric endoscopy, radiographs, serum or whole blood trace mineral analysis, serum insulin concentration, carbohydrate absorption tests (oral D-glucose, D-xylose [see Chapter 32]), and appropriate organ biopsy.
Approach to the Diagnosis and Management of Weight Loss in Adult Ruminants
Question the caretaker closely about any clinical signs that might indicate a primary disease (e.g., diarrhea, coughing, dysphagia, polyuria, depression, agalactia). Note if body condition is less than desired. Quantitate the weight loss or BCS if possible. Acute weight loss of 5% to 10% is quite significant. Carefully note the production level of the animal or herd (e.g., pregnancy [single, twins, triplets], lactation [level of milk production]). Evaluate the parasite control program. Does the herd have a history of chronic or recurrent disease (Johne’s disease, bovine virus diarrhea, ruminal lactic acidosis, laminitis, mastitis, pneumonia)?
Obtain an accurate, dietary history, particularly when signs of a primary disease are absent. Inspect all forages, concentrates, and feed additives for quality, signs of spoilage, abnormal color or odor, and quantity on hand. Be sure the feeding system allows for adequate consumption by all animals and that competition for feedstuffs does not occur. Check to see if the feeding program was changed before the onset of observed weight loss or loss of body condition. The history should include the weight, of each feedstuff and supplement fed and consumed per day. The maximum DMI can be estimated according to feed quality for cattle (see Table 9-1). Determine or estimate the nutrient analysis of the feedstuffs being fed. Examine the environment for possible toxic plants or substances.
Examine patients carefully for signs of concurrent disease (e.g., diarrhea, decreased ruminal motility, pyrexia, dysphagia, abnormal dentition, melena, icterus, mastitis, metritis, dyspnea, tachycardia). Is the patient hungry? Weigh the patient (or use a heart-girth measurement) and note the BCS (see Tables 9-18 through 9-20). Observe the patient for signs of muscle wasting and the presence or absence of subcutaneous fat. Test the milk with nitroprusside powder (a positive reaction indicates an acetoacetate concentrate above 5 mg/dL and is diagnostic of ketonlactia and ketonemia), or measure the ketone concentration in the urine. Measure the ruminal pH (pH above 7 is indicative of anorexia). Examine the skin for evidence of lice or keds.
Table 9-19 Body Conditioning Scoring System for Dairy Cattle
| Score | Description |
|---|---|
| 1 | Individual spinous processes have limited flesh covering and are prominent; the ends are sharp to the touch, and together the processes form a definite overhanging shelf effect to the loin region. Individual vertebrae of the chine, loin, and rump regions are prominent and distinct. Hooks and pin bones are sharp with negligible flesh covering, and severe depressions between hooks and pin bones are noted. The area below the tailhead and between the pin bones is severely depressed, causing the bone structure of the area to appear extremely sharp. (Body fat = 3.77%) |
| 2 | Individual spinous processes are visually discernible but not prominent. The ends of processes are sharp to the touch, although they have greater flesh covering, and the processes do not have a distinct overhanging shelf effect. Individual vertebrae of chine, loin, and rump regions are not visually distinct but are readily distinguishable by palpation. Hooks and pin bones are prominent, but the depression between them is less severe. The area below the tailhead and between the pin bones is depressed, but the bone structure is not devoid of flesh covering. (Body fat = 11.3%) |
| 3 | Spinous processes are discernible by applying slight pressure. Together the processes appear smooth, and the overhanging shelf effect is not noticeable. Vertebrae of the chine, loin, and rump regions appear as rounded ridges, and hooks and pin bones are rounded and smooth. The area between the pin bones and around the tailhead appears smooth, with no sign of fat deposition. (Body fat = 18.84%) |
| 4 | Individual spinous processes can be distinguished only by firm palpation, and together the processes appear flat or rounded with no overhanging shelf effect. The ridge formed by the vertebral column of the chine region is rounded and smooth, but loin and rump regions appear flat. Hooks are rounded, and the span between the hooks is flat. The area around the tailhead and pin bones is rounded, with evidence of subcutaneous fat deposition. (Body fat = 26.38%) |
| 5 | Bone structure of the vertebral column, spinous processes, hooks, and pin bones is not visually apparent, and evidence of subcutaneous fat deposition is prominent. The tailhead appears to be buried in fatty tissue. (Body fat = 33.9%) |
From Wildman EE et al: J Dairy Sci, 65:495–501, 1982; and modified from National Research Council (NRC): Nutrient requirements of dairy cattle, Washington, DC, 2001, National Academy of Sciences, NRC.
Table 9-20 Body Condition Scoring System for Sheep
| Score | Description |
|---|---|
| 0 | Animal is extremely emaciated and at the point of death. No muscular or fatty tissue can be detected between the skin and the bone. |
| 1 | The spinous processes are prominent and sharp. The transverse processes are also sharp; the fingers pass easily under the ends, and it is possible to feel between each process. The eye muscle areas are shallow with no fat cover. |
| 2 | The spinous processes still feel prominent but also smooth, and individual processes can be felt only as fine corrugations. The transverse processes are smooth and rounded, and the fingers can be passed under the ends with a little pressure. The eye muscle areas are of moderate depth but have little fat cover. |
| 3 | The spinous processes are detected only as small elevations; they are smooth and rounded, and individual bones can be felt only with pressure. The transverse processes are smooth and well covered, and firm pressure is required to feel over the ends. The eye muscle areas are full and have a moderate degree of fat cover. |
| 4 | With pressure the spinous processes can just be detected as a hard line between the fat-covered muscle areas. The ends of the transverse processes cannot be felt. The eye muscle areas are full and have a thick covering of fat. |
| 5 | The spinous processes cannot be detected even with firm pressure, and there is a depression between the layers of fat where the spinous processes would normally be felt. The transverse processes cannot be detected. The eye muscle areas are very full and have a very thick fat cover. Large deposits of fat may be seen over the rump and tail. |
From Russel A: Body condition scoring of sheep, In Pract, 6:91, 1984.
Perform flotation, sedimentation, and Baermann’s procedures to detect patent parasitic infestations. If the feces test positive for occult blood or are very dark, see the section on melena in Chapter 7. If there is evidence of or apparent diarrhea, see the section on diarrhea in Chapter 7.
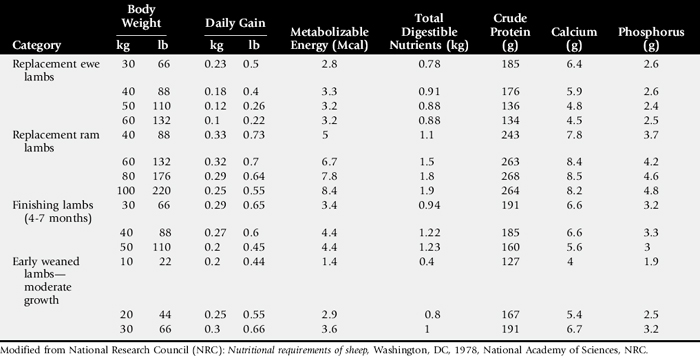
Is the feed quantity and quality adequate to allow sufficient intake of nutrients (see Table 9-1)? Compare the nutrient intake from the diet with the requirements for the appropriate species (Tables 9-12 through 9-17). Consider any important environmental and management factors in the daily nutrient requirements. Review nutrient analysis profiles on the feeds and total mixed ration (TMR) ingredients. Is the TMR mixed properly? Are grain rations mixed properly? Is too much grain being fed to the animal or herd? Analyze the pasture, forage, or concentrate rations (see Box 9-3). Make appropriate recommendations to optimize the ration. Determine if the patient or patients have a normal appetite. Is anorexia present? If the dietary history and analysis indicate that adequate nutrients have been steadily consumed, the search for a primary cause for the weight loss should be resumed.
OBESITY
Mechanisms of Obesity
Obesity is a common problem in domestic species. The prevalence of obesity is increasing in the domestic horse population, and obesity is a significant problem in many ruminant species raised as companion animals. Obese patients (especially ruminants) are at particular risk for reproductive failure or metabolic disease late in pregnancy or during lactation. The risk of obstructive urolithiasis increases in overweight goats and sheep. In horses, obesity may be related to a variety of diseases including equine metabolic syndrome, laminitis, and colic associated with strangulating lipomas. Obese horses and ponies that are rapidly losing weight or that are anorexic are particularly susceptible to hyperlipidemia and hyperlipemia.
The mechanism of obesity is invariably a prolonged intake of total dietary energy above that needed for maintenance and either production or exercise. Obesity occurs most commonly in stabled horses fed high-energy feeds such as grain or high-fat supplemental feeds and in horses and ponies on lush pasture. Even horses that are fed a forage diet but have limited access to exercise may gain weight if they consume an excess amount of energy for their body size. Purebred or pet sheep and goats (particularly wethers) tend to be overfed. In dairy cattle, obesity occurs when cattle are fed well above requirements for maintenance and milk production. Poor reproductive performance is often associated with the initiation of obesity and is also a common sequela to obesity. Feeding for high milk production for lengthy periods in production groups predisposes infertile cows to become fat cows. Dry cows with access to high-energy diets are also predisposed to fat cow syndrome. The systemic complications associated with fat cow syndrome (fatty liver), and its diagnosis and treatment, are described in Chapter 33.
Diagnosis of Obesity in Horses and Ruminants
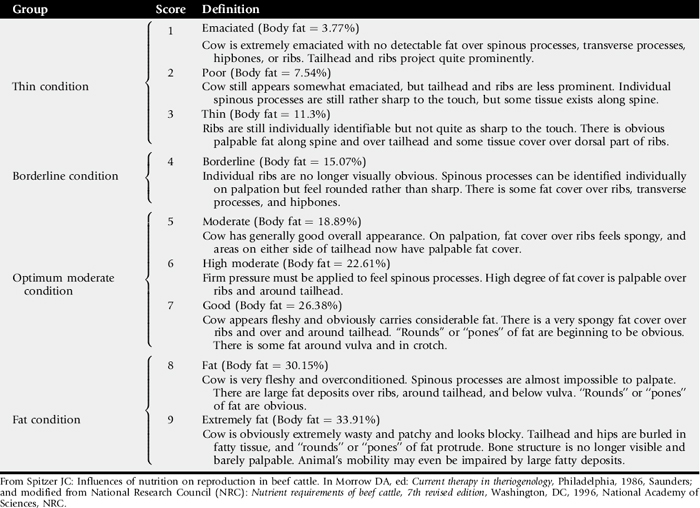
Diagnosis is obvious; it is made by physical examination. Overweight and obese animals have an elevated BCS (7 to 9/9 for horses; 8 to 9/9 for beef cattle; 4 to 5/5 for dairy cattle, sheep, and goats). Palpation of the back, gluteal area, and ribs should be included in the physical examination of sheep and camelids with long wool or fiber and of horses with a long winter hair coat so that a BCS can be accurately assigned. In some animals, external signs of fat deposition may be subtle. In these cases, ultrasonography can be used to identify the extent of deposition of intraabdominal fat in horses and of back fat in cattle. Clinical descriptions of obesity provided by the body condition scoring system are straightforward (Tables 9-18 to 9-21) and require minimal interpretation.
In some animals, obesity can be mistaken for the normal physiologic condition of pregnancy. In other cases, obesity could be mistaken for a distended abdomen from acute pathologic disease conditions including uroabdomen and peritonitis. A full physical examination should always be performed on a large animal patient before a weight loss ration is fed.
Treatment of Obesity
Overweight and obese horses and ruminants should lose weight to extend life span and to improve production efficiency. In many cases a reduction in energy intake can be achieved by simply eliminating excess calories from grain or supplemental feeds. Animals that fail to lose weight after 1 to 2 months after a reduction in calories require a more aggressive weight loss program, which may include reformulation of their ration and the gradual implementation of an exercise program. Voluntary activity in animals that are turned out in pasture is rarely high enough to promote weight loss. Obese laminitic horses present a difficult challenge because exercise may not be practical, and weight loss must rely solely on dietary energy restriction.
Horses and companion ruminants that require a managed weight loss program should have the energy content of their diet evaluated by the veterinarian. The actual energy intake should be compared against the energy requirement for the animal’s current lifestage (see Tables 9-11 to 9-17). The animal’s current calorie intake should be reduced by 10% to 20% at the start of the weight loss program. Dietary protein should not be restricted. Supplemental feeds and treats should be reduced or eliminated from the diet, and only high-quality forage should be fed.
Feeding straw should be avoided because gastrointestinal complications including impaction colic could develop. A vitamin and mineral supplement should be fed during a weight loss program to ensure that the diet meets nutrient requirements. To prevent boredom, small amounts of forage should be fed frequently. When horses are being fed, hay can be placed in a double hay net or hay bag, or a restricted feeder (The Grazer Hay Feeding Machine) can be used to decrease the rate of intake. The target weight loss goal is between 0.5% and 2% of the original body weight each week. A weight tape should be used to assess the horse’s weight if a scale is not available. A BCS should be assigned to the animal at the start of the weight loss program. The body weight and BCS should be assessed every month to track changes in the animal’s weight.
Owner compliance is essential during a weight loss program for horses and companion large animals. Owners should be encouraged to keep a weight loss journal and to include digital pictures to assess the animal’s BCS during the program. Excessive, rapid weight loss should be avoided. Weight loss programs should be approached with caution in animals that are either pregnant or lactating. Once the animal has achieved the ideal body weight and BCS, it should be placed on a maintenance ration and exercise program that will ensure that the ideal weight is maintained.
In a production management setting the ration should be evaluated and revised to reduce calories while maintaining sufficient energy intake to maximize reproduction or milk production. Care should be taken to ensure the animals consume an adequate concentration of vitamins and minerals to meet their requirements when they are fed an energy-restricted ration.
PICA
Pica (geophagia) is defined as a depraved or abnormal appetite. It is usually associated with animals that chew or eat wood (fences, trees, buildings), dirt, bones, or other inanimate objects not usually considered feedstuffs. The mechanism or mechanisms of pica are not yet understood. Pica has been associated with PCM, parasitism, obesity, and deficiencies of phosphorus, salt, protein (kwashiorkor), and micronutrients. Diagnosis is by observation or history or both. The main emphasis must be placed on identification and resolution of the primary problem. Pica must be differentiated from abnormal behavior associated with central nervous system diseases, bovine ketosis, and equine behavioral abnormalities associated with boredom.
1 Budzynski M, et al. Growth in halfbred horses. Roczniki Nauk Roln B. 1961;93:21.
2 Cunningham K, Fowler S. A study of growth in quarter horses. La Agric Exp Stn Bull. 1961:546.
3 Hintz HF. A review of recent studies on the growth of horses. Calif Vet. 1979:17. March
4 Jordan RM. Growth pattern in ponies. Proc Fifth Equine Nutr Physiol Symp. 1977;5:63.
5 Reed KF, Dunn NK. Growth of the Arabian horse. Proc Fifth Equine Nutr Physiol Symp. 1977;5:99.
6 National Research Council. Nutrient requirements of horses, ed 6 revised, Washington, DC: National Academies Press; 2007:54-68. 294–303
7 Kinney JM. The application of indirect calorimetry to clinical studies. In: Kinney JM, editor. Assessment of energy metabolism in health and disease. Columbus, Ohio: Ross Laboratories, 1980.
8 Long CL, Schaffel N, Geiger JW, et al. Metabolic response to injury and illness: estimation of energy and protein needs from indirect calorimetry and nitrogen balance. JPEN J Parenter Enteral Nutr. 1979;3:452.
9 Trujillo EB, Chertow GM, Jacobs DO. Metabolic assessment. In: Rombeau JL, Rolandelli RH, editors. Clinical nutrition: parenteral nutrition. ed 3. Philadelphia: Saunders; 2001:80.
10 National Research Council:. Nutrient requirements of horses, ed 6 revised, Washington, DC: National Academies Press; 2007:3.
11 Carroll CL, Huntington PJ. Body condition scoring and weight estimation of horses. Equine Vet J. 1988;20:41.
12 Reavell DG. Measuring and estimating the weight of horses with tapes, formulae and by visual assessment. Equine Vet Educ. 1999:188. December
13 Kentucky Equine Research: Feeding the miniature horse, Kentucky Equine Research Equine Review,
14 Russel A. Nutrition of the pregnant ewe. Vet Rec,(In Pract, 7:23, suppl). 1985;116:29.