Chapter 24 The Endocrine System
The endocrine system consists of a highly integrated and widely distributed group of organs that orchestrate a state of metabolic equilibrium, or homeostasis, among the various organs of the body. Signaling by extracellular secreted molecules can be classified into three types—autocrine, paracrine, or endocrine—on the basis of the distance over which the signal acts. In endocrine signaling, the secreted molecules, which are frequently called hormones, act on target cells that are distant from their site of synthesis. An endocrine hormone is frequently carried by the blood from its site of release to its target. In response, the target tissue often secretes factors that down-regulate the activity of the gland that produces the stimulating hormone, a process known as feedback inhibition.
Several processes can disturb the normal activity of the endocrine system, including impaired synthesis or release of hormones, abnormal interactions between hormones and their target tissues, and abnormal responses of target organs. Endocrine diseases can be generally classified as (1) diseases of underproduction or overproduction of hormones and their resulting biochemical and clinical consequences and (2) diseases associated with the development of mass lesions. Such lesions might be nonfunctional, or they might be associated with overproduction or underproduction of hormones. The study of endocrine diseases requires integration of morphologic findings with biochemical measurements of the levels of hormones, their regulators, and other metabolites.
PITUITARY GLAND
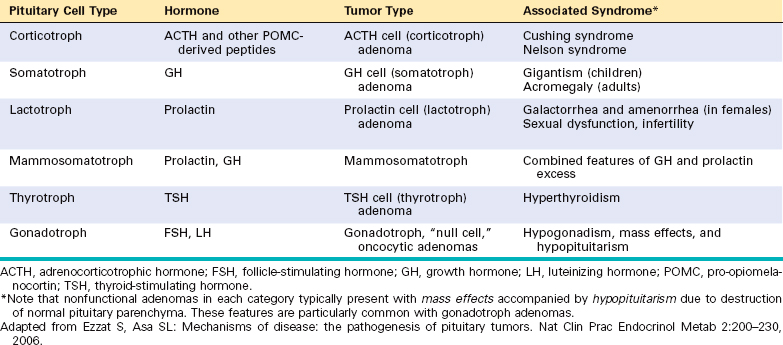
The pituitary gland is composed of two morphologically and functionally distinct components: the anterior lobe (adenohypophysis) and the posterior lobe (neurohypophysis). The anterior pituitary constitutes about 80% of the gland. The production of most pituitary hormones is controlled predominantly by positive-acting releasing factors from the hypothalamus (Fig. 24-1), which are carried to the anterior pituitary by a portal vascular system. Prolactin is the major exception; its primary hypothalamic control is inhibitory, through the action of dopamine. Pituitary growth hormone also differs in that it receives both stimulatory and inhibitory influences via the hypothalamus. In routine histologic sections of the anterior pituitary, a colorful array of cells is present that contain eosinophilic cytoplasm (acidophil), basophilic cytoplasm (basophil), or poorly staining cytoplasm (chromophobe) cells (Fig. 24-2). Specific antibodies against the pituitary hormones identify five cell types:
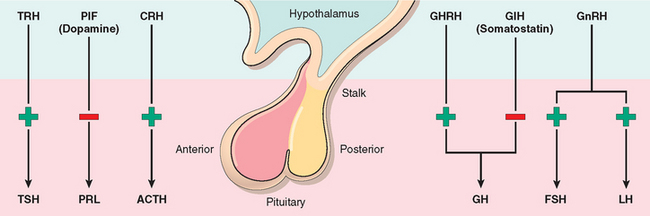
FIGURE 24-1 Hormones released by the anterior pituitary. The adenohypophysis (anterior pituitary) releases five hormones that are in turn under the control of various stimulatory and inhibitory hypothalamic releasing factors. TSH, thyroid-stimulating hormone (thyrotropin); PRL, prolactin; ACTH, adrenocorticotropic hormone (corticotropin); GH, growth hormone (somatotropin); FSH, follicle-stimulating hormone; LH, luteinizing hormone. The stimulatory releasing factors are TRH (thyrotropin-releasing hormone), CRH (corticotropin-releasing hormone), GHRH (growth hormone–releasing hormone), GnRH (gonadotropin-releasing hormone). The inhibitory hypothalamic influences comprise PIF (prolactin inhibitory factor or dopamine) and growth hormone inhibitory factor (GIH or somatostatin).
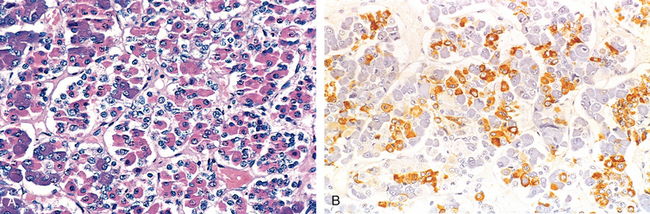
FIGURE 24-2 A, Photomicrograph of normal pituitary. The gland is populated by several distinct cell populations containing a variety of stimulating (tropic) hormones. Each of the hormones has different staining characteristics, resulting in a mixture of cell types in routine histologic preparations. B, Immunostain for human growth hormone.
The posterior pituitary consists of modified glial cells (termed pituicytes) and axonal processes extending from the hypothalamus through the pituitary stalk to the posterior lobe (axon terminals). The two peptide hormones secreted from the posterior pituitary—oxytocin and antidiuretic hormone (ADH, also called vasopressin)—are actually synthesized in the hypothalamus and are stored within the axon terminals residing in the posterior pituitary. In response to appropriate stimuli, the pre-formed hormones are released directly into the systemic circulation through the venous channels of the pituitary. For example, dilatation of the cervix in pregnancy results in massive oxytocin release, leading to contraction of the uterine smooth muscle, facilitating parturition (uterine labor). Similarly, oxytocin released upon nipple stimulation in the postnatal period acts on the smooth muscles surrounding the lactiferous ducts of the mammary glands and facilitates lactation. Synthetic oxytocin can be administered during pregnancy for artificial induction of labor. The most important function of ADH is to conserve water by restricting diuresis during periods of dehydration and hypovolemia. Decreased blood pressure, sensed by baroreceptors (pressure-sensing receptors) in the cardiac atria and carotids, stimulates ADH release. An increase in plasma osmotic pressure detected by osmoreceptors also tiggers ADH secretion. In contrast, states of hypervolemia and increased atrial distention result in inhibition of ADH secretion.
Clinical Manifestations of Pituitary Disease
The manifestations of pituitary disorders are as follows:
Diseases of the posterior pituitary often come to clinical attention because of increased or decreased secretion of ADH.
Pituitary Adenomas and Hyperpituitarism
The most common cause of hyperpituitarism is an adenoma arising in the anterior lobe. Pituitary adenomas are classified on the basis of hormone(s) produced by the neoplastic cells, which are detected by immunohistochemical stains (Table 24-1). Some pituitary adenomas can secrete two hormones (GH and prolactin being the most common combination), and rarely, pituitary adenomas are plurihormonal. Pituitary adenomas can be functional (i.e., associated with hormone excess and clinical manifestations thereof) or nonfunctioning (i.e., immunohistochemical and/or ultrastructural demonstration of hormone production at the tissue level, without clinical symptoms of hormone excess). Less common causes of hyperpituitarism include pituitary carcinomas and some hypothalamic disorders. Large pituitary adenomas, and particularly nonfunctioning ones, may cause hypopituitarism as they encroach on and destroy adjacent anterior pituitary parenchyma.
Pituitary adenomas are usually found in adults, with a peak incidence from 35 to 60 years of age. They are designated, somewhat arbitrarily, microadenomas if they are less than 1 cm in diameter and macroadenomas if they exceed 1 cm in diameter. Silent and hormone-negative adenomas are likely to come to clinical attention at a later stage than those associated with endocrine abnormalities and are therefore more likely to be macroadenomas. A meta-analysis of autopsy studies estimates the population prevalence of pituitary adenomas to be about 14%, although the vast majority of these lesions are incidentally diagnosed microadenomas (“pituitary incidentaloma”).1
With recent advances in molecular techniques, substantial insight has been gained into the genetic abnormalities associated with pituitary adenomas2 (Table 24-2):
TABLE 24-2 Genetic Alterations in Pituitary Tumors
| Gene | Mechanism of Alteration | Pituitary Tumor Subtype |
|---|---|---|
| GAIN OF FUNCTION | ||
| Gsα | Activating mutation | GH adenomas |
| Protein kinase A (PKA)* | Germline inactivating mutations of PRKARIA (Carney complex), a negative regulator of PKA | GH and prolactin adenomas |
| Cyclin D1 | Overexpression | Aggressive adenomas |
| HRAS | Activating mutation | Pituitary carcinomas |
| LOSS OF FUNCTION | ||
| Menin* | Germline inactivating mutations of MEN1 (multiple endocrine neoplasia, type 1) | GH, prolactin, and ACTH adenomas |
| CDKN1B (p27/KIP1)* | Germline inactivating mutations of CDKN1B (“MEN-1-like” syndrome) | ACTH adenomas |
| Aryl hydrocarbon receptor interacting protein (AIP)* | Germline mutations of AIP (pituitary adenoma predisposition [PAP] syndrome) | GH adenomas |
| Retinoblastoma (RB) protein | Methylation of RB gene promoter | Aggressive adenomas |
ACTH, adrenocorticotrophic hormone; GH, growth hormone.
* Genetic alterations associated with familial predisposition to pituitary adenomas.
Adapted from Boikos SA, Stratakis CA: Molecular genetics of the cAMP-dependent protein kinase pathway and of sporadic pituitary tumorigenesis. Hum Mol Genet 16:R80–R87, 2007.
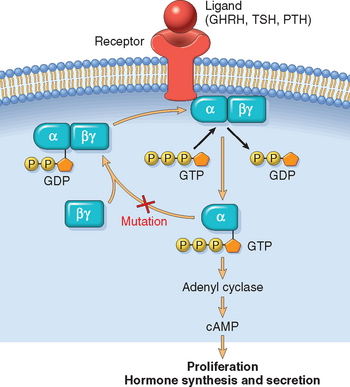
FIGURE 24-3 G-protein signaling in endocrine neoplasia. Mutations that lead to G-protein hyperactivity are seen in a variety of endocrine neoplasms, including pituitary, thyroid, and parathyroid adenomas. G proteins play a critical role in signal transduction, transmitting signals from cell surface receptors (GHRH, TSH, or PTH receptor) to intracellular effectors (e.g., adenyl cyclase), which then generate second messengers (cAMP, cyclic adenosine monophosphate). GDP, guanosine diphosphate; GTP, guanosine triphosphate; Pi, inorganic phosphate. See Figure 24-1 for other abbreviations.
Morphology. The typical pituitary adenoma is a soft, well-circumscribed lesion that may be confined to the sella turcica. Larger lesions typically extend superiorly through the diaphragm sella into the suprasellar region, where they often compress the optic chiasm and adjacent structures, such as some of the cranial nerves (Fig. 24-4). As these adenomas expand, they frequently erode the sella turcica and anterior clinoid processes. In as many as 30% of cases, the adenomas are not grossly encapsulated and infiltrate neighboring tissues such as the cavernous and sphenoid sinuses, dura, and on occasion, the brain itself. Such lesions are termed invasive adenomas. Not unexpectedly, macroadenomas tend to be invasive more frequently than smaller tumors. Foci of hemorrhage and necrosis are also more common in these larger adenomas.
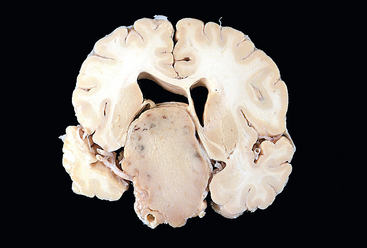
FIGURE 24-4 Pituitary adenoma. This massive, nonfunctional adenoma has grown far beyond the confines of the sella turcica and has distorted the overlying brain. Nonfunctional adenomas tend to be larger at the time of diagnosis than those that secrete a hormone.
Histologically, typical pituitary adenomas are composed of relatively uniform, polygonal cells arrayed in sheets or cords. Supporting connective tissue, or reticulin, is sparse, accounting for the soft, gelatinous consistency of many of these lesions. Mitotic activity is usually sparse. The cytoplasm of the constituent cells may be acidophilic, basophilic, or chromophobic, depending on the type and amount of secretory product within the cells, but it is generally uniform throughout the tumor. This cellular monomorphism and the absence of a significant reticulin network distinguish pituitary adenomas from non-neoplastic anterior pituitary parenchyma (Fig. 24-5). The biologic behavior of the adenoma cannot always be reliably predicted from its histologic appearance. A subset of pituitary adenomas demonstrates brisk mitotic activity and staining of greater than 3% of the nuclei with the proliferation marker Ki-67; these tumors typically also demonstrate extensive nuclear p53 immunoreactivity in the neoplastic cells, a feature that correlates with the presence of p53 mutations. It is recommended that adenomas with this profile be classified as atypical adenomas, since these tumors have a higher propensity for aggressive behavior, including invasion and recurrence.
Clinical Course.
The signs and symptoms of pituitary adenomas include endocrine abnormalities and mass effects. The effects of excessive secretion of anterior pituitary hormones are mentioned below, when the specific types of pituitary adenoma are described. Local mass effects may be encountered in any type of pituitary tumor and have been discussed previously under clinical manifestations of pituitary disease. Briefly, these include radiographic abnormalities of the sella turcica, visual field abnormalities, signs and symptoms of elevated intracranial pressure, and occasionally hypopituitarism. Acute hemorrhage into an adenoma is sometimes associated with pituitary apoplexy, as was noted above.
With this general introduction to pituitary adenomas, we proceed to a discussion of the individual types of tumors.
PROLACTINOMAS
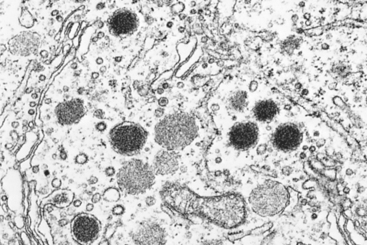
Prolactinomas (lactotroph adenomas) are the most frequent type of hyperfunctioning pituitary adenoma, accounting for about 30% of all clinically recognized cases. These lesions range from small microadenomas to large, expansile tumors associated with substantial mass effect. Microscopically, the overwhelming majority of prolactinomas are composed of weakly acidophilic or chromophobic cells (sparsely granulated prolactinoma); rare prolactinomas are strongly acidophilic (densely granulated prolactinoma) (Fig. 24-6). Prolactin can be demonstrated within the secretory granules in the cytoplasm of the cells using immunohistochemical stains. Prolactinomas have a propensity to undergo dystrophic calcification, ranging from isolated psammoma bodies to extensive calcification of virtually the entire tumor mass (“pituitary stone”). Prolactin secretion by functioning adenomas is usually efficient (even microadenomas secrete sufficient prolactin to cause hyperprolactinemia) and proportional, in that serum prolactin concentrations tend to correlate with the size of the adenoma.
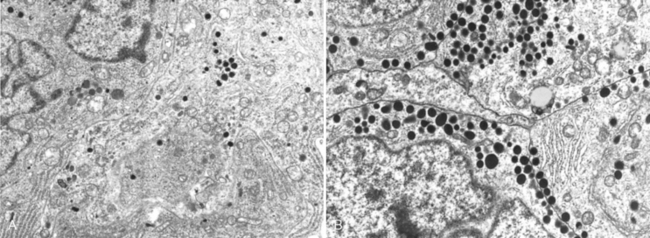
FIGURE 24-6 Ultrastructural features of prolactinomas. A, Electron micrograph of a sparsely granulated prolactinoma. The tumor cells contain abundant granular endoplasmic reticulum (indicative of active protein synthesis) and small numbers of electron-dense secretory granules. B, Electron micrograph of densely granulated growth hormone–secreting adenoma. The tumor cells are filled with numerous large, electron-dense secretory granules.
(Courtesy of Dr. Eva Horvath, St. Michael’s Hospital, Toronto, ON, Canada.)
Increased serum levels of prolactin, or prolactinemia, cause amenorrhea, galactorrhea, loss of libido, and infertility. The diagnosis of an adenoma is made more readily in women than in men, especially between the ages of 20 and 40 years, presumably because of the sensitivity of menses to disruption by hyperprolactinemia. Prolactinoma underlies almost a quarter of cases of amenorrhea. In contrast, in men and older women, the hormonal manifestations may be subtle, allowing the tumors to reach considerable size (macroadenomas) before being detected clinically.
Hyperprolactinemia may result from causes other than prolactin-secreting pituitary adenomas. Physiologic hyperprolactinemia occurs in pregnancy; serum prolactin levels increase throughout pregnancy, reaching a peak at delivery. Prolactin levels are also elevated by nipple stimulation, as occurs during suckling in lactating women, and as a response to many types of stress. Pathologic hyperprolactinemia can also result from lactotroph hyperplasia, such as when there is interference with normal dopamine inhibition of prolactin secretion. This may occur as a result of damage to the dopaminergic neurons of the hypothalamus, damage to the pituitary stalk (e.g., due to head trauma), or drugs that block dopamine receptors on lactotroph cells. Any mass in the suprasellar compartment may disturb the normal inhibitory influence of the hypothalamus on prolactin secretion, resulting in hyperprolactinemia. Therefore, a mild elevation in serum prolactin in a person with a pituitary adenoma does not necessarily indicate a prolactin-secreting tumor. Other causes of hyperprolactinemia include several classes of drugs (such as dopamine antagonists), estrogens, renal failure, and hypothyroidism. Prolactinomas are treated by surgery or, more commonly, with bromocriptine, a dopamine receptor agonist that causes the lesions to diminish in size.
GROWTH HORMONE CELL (SOMATOTROPH) ADENOMAS
GH-secreting tumors are the second most common type of functioning pituitary adenoma. Somatotroph cell adenomas may be quite large by the time they come to clinical attention because the manifestations of excessive GH may be subtle. Histologically, pure GH cell–containing adenomas are also classified into two subtypes: densely granulated and sparsely granulated. The densely granulated adenomas are composed of cells that are monomorphic and acidophilic in routine sections, retain strong cytoplasmic GH reactivity on immunohistochemistry, and demonstrate cytokeratin staining in a perinuclear distribution. In contrast, the sparsely granulated variants are composed of chromophobe cells with considerable nuclear and cytologic pleomorphism and focal, weak staining for GH. Bihormonal mammosomatotroph adenomas that express both GH and prolactin are being increasingly recognized with the availability of better immunohistochemical reagents; morphologically, most bihormonal adenomas resemble the densely granulated pure somatotroph adenomas.
Persistently elevated levels of GH stimulate the hepatic secretion of insulin-like growth factor 1 (IGF-1 or somatomedin C), which causes many of the clinical manifestations. If a somatotrophic adenoma appears in children before the epiphyses have closed, the elevated levels of GH (and IGF-1) result in gigantism. This is characterized by a generalized increase in body size with disproportionately long arms and legs. If the increased levels of GH are present after closure of the epiphyses, patients develop acromegaly. In this condition, growth is most conspicuous in skin and soft tissues; viscera (thyroid, heart, liver, and adrenals); and bones of the face, hands, and feet. Bone density may be increased (hyperostosis) in both the spine and the hips. Enlargement of the jaw results in protrusion (prognathism), with broadening of the lower face. The hands and feet are enlarged with broad, sausage-like fingers. In most instances gigantism is also accompanied by evidence of acromegaly. These changes develop for decades before being recognized, hence the opportunity for the adenomas to reach substantial size. GH excess is also correlated with a variety of other disturbances, including gonadal dysfunction, diabetes mellitus, generalized muscle weakness, hypertension, arthritis, congestive heart failure, and an increased risk of gastrointestinal cancers.
The diagnosis of pituitary GH excess relies on documentation of elevated serum GH and IGF-1 levels. In addition, failure to suppress GH production in response to an oral load of glucose is one of the most sensitive tests for acromegaly. The underlying pituitary adenoma can be either removed surgically or treated via pharmacologic means. The latter includes somatostatin analogs (recall that somatostatin has an inhibitory effect on pituitary GH secretion) or the use of GH receptor antagonists, which prevent hormone binding to target organs such as the liver. When effective control of high GH levels is achieved, the characteristic tissue overgrowth and related symptoms gradually recede, and the metabolic abnormalities improve.
ACTH CELL (CORTICOTROPH) ADENOMAS
Corticotroph adenomas are usually small microadenomas at the time of diagnosis. These tumors are most often basophilic (densely granulated) and occasionally chromophobic (sparsely granulated). Both variants stain positively with periodic acid–Schiff (PAS) because of the presence of carbohydrate in POMC, the ACTH precursor molecule; in addition, they demonstrate variable immunoreactivity for POMC and its derivatives, including ACTH and β-endorphin.
Excess production of ACTH by the corticotroph adenoma leads to adrenal hypersecretion of cortisol and the development of hypercortisolism (also known as Cushing syndrome). This syndrome is discussed in more detail later with the diseases of the adrenal gland. It can be caused by a wide variety of conditions in addition to ACTH-producing pituitary tumors. When the hypercortisolism is due to excessive production of ACTH by the pituitary, the process is designated Cushing disease. Large destructive adenomas can develop in patients after surgical removal of the adrenal glands for treatment of Cushing syndrome. This condition, known as Nelson syndrome, occurs most often because of a loss of the inhibitory effect of adrenal corticosteroids on a preexisting corticotroph microadenoma. Because the adrenals are absent in persons with this disorder, hypercortisolism does not develop. In contrast, patients present with mass effects of the pituitary tumor. In addition, there can be hyperpigmentation because of the stimulatory effect of other products of the ACTH precursor molecule on melanocytes.
OTHER ANTERIOR PITUITARY ADENOMAS
Pituitary adenomas may elaborate more than one hormone. For example, prolactin may be demonstrable by immunolabeling of somatotroph adenomas. In other cases, unusual plurihormonal adenomas are capable of secreting multiple hormones; these tumors are usually aggressive. A few comments are made about several of the less frequent functioning tumors.
Gonadotroph (LH-producing and FSH-producing) adenomas can be difficult to recognize because they secrete hormones inefficiently and variably, and the secretory products usually do not cause a recognizable clinical syndrome (non-functioning adenomas, see below). Gonadotroph adenomas are most frequently found in middle-aged men and women when they become large enough to cause neurologic symptoms, such as impaired vision, headaches, diplopia, or pituitary apoplexy. Pituitary hormone deficiencies can also be found, most commonly impaired secretion of LH. This causes decreased energy and libido in men (due to reduced testosterone) and amenorrhea in premenopausal women. Thus, gonadotroph adenomas are paradoxically associated with secondary gonadal hypofunction. The neoplastic cells usually demonstrate immunoreactivity for the common gonadotropin α-subunit and the specific β-FSH and β-LH subunits; FSH is usually the predominant secreted hormone.
Thyrotroph (TSH-producing) adenomas are rare, accounting for approximately 1% of all pituitary adenomas. Thyrotroph adenomas are a rare cause of hyperthyroidism.
Nonfunctioning pituitary adenomas are a heterogeneous group that constitutes approximately 25% to 30% of all pituitary tumors. Their lineage can be established by immunohistochemical staining for hormones or by biochemical demonstration of cell type-specific transcription factors. In the past, many such tumors have been called silent variants or null-cell adenomas. Not surprisingly, the typical presentation of nonfunctioning adenomas is mass effects. These lesions may also compromise the residual anterior pituitary sufficiently to cause hypopituitarism. This may occur as a result of gradual enlargement of the adenoma or after abrupt enlargement of the tumor because of acute hemorrhage (pituitary apoplexy).
Pituitary carcinomas are quite rare, accounting for less than 1% of pituitary tumors. The demonstration of craniospinal or systemic metastases is a sine qua non of a pituitary carcinoma. The majority of pituitary carcinomas are functional neoplasms, with prolactin and ACTH being the most common secreted products. Metastases usually appear late in the course, following multiple local recurrences.
Hypopituitarism
Hypopituitarism refers to decreased secretion of pituitary hormones, which can result from diseases of the hypothalamus or of the pituitary. Hypofunction of the anterior pituitary occurs when approximately 75% of the parenchyma is lost or absent. This may be congenital or the result of a variety of acquired abnormalities that are intrinsic to the pituitary. Hypopituitarism accompanied by evidence of posterior pituitary dysfunction in the form of diabetes insipidus (see below) is almost always of hypothalamic origin. Most cases of hypofunction arise from destructive processes directly involving the anterior pituitary, although other mechanisms have been identified.
The clinical manifestations of anterior pituitary hypofunction can be protean, and depend on the specific hormone(s) that are lacking. Children can develop growth failure (pituitary dwarfism) due to growth hormone deficiency. Gonadotropin (LH and FSH) deficiency leads to amenorrhea and infertility in women and decreased libido, impotence, and loss of pubic and axillary hair in men. TSH and ACTH deficiencies result in symptoms of hypothyroidism and hypoadrenalism, respectively, and are discussed later in the chapter. Prolactin deficiency results in failure of postpartum lactation. The anterior pituitary is also a rich source of MSH, synthesized from the same precursor molecule that produces ACTH; therefore, one of the manifestations of hypopituitarism includes pallor due to a loss of stimulatory effects of MSH on melanocytes.
Posterior Pituitary Syndromes
The clinically relevant posterior pituitary syndromes involve ADH and include diabetes insipidus and secretion of inappropriately high levels of ADH.
Hypothalamic Suprasellar Tumors
Neoplasms in this location may induce hypofunction or hyperfunction of the anterior pituitary, diabetes insipidus, or combinations of these manifestations. The most commonly implicated lesions are gliomas (sometimes arising in the chiasm; see Chapter 28) and craniopharyngiomas. The craniopharyngioma is thought to be derived from vestigial remnants of Rathke pouch. These slow-growing tumors account for 1% to 5% of intracranial tumors; a small minority of these lesions arise within the sella, but most are suprasellar, with or without intrasellar extension. A bimodal age distribution is observed, with one peak in childhood (5 to 15 years) and a second peak in adults 65 years or older. Patients usually come to attention because of headaches and visual disturbances, while children might present with growth retardation due to pituitary hypofunction and GH deficiency. Abnormalities of the WNT signaling pathway, including activating β-catenin mutations, have been reported in craniopharyngiomas.
Morphology. Craniopharyngiomas average 3 to 4 cm in diameter; they may be encapsulated and solid, but more commonly they are cystic and sometimes multiloculated. In their strategic location, they often encroach on the optic chiasm or cranial nerves, and not infrequently they bulge into the floor of the third ventricle and base of the brain. Two distinct histologic variants are recognized: adamantinomatous craniopharyngioma (most often observed in children) and papillary craniopharyngioma (most often observed in adults). The adamantinomatous type frequently contains radiologically demonstrable calcifications; the papillary variant calcifies only rarely.
Adamantinomatous craniopharyngioma consists of nests or cords of stratified squamous epithelium embedded in a spongy “reticulum” that becomes more prominent in the internal layers. “Palisading” of the squamous epithelium is frequently observed at the periphery. Compact, lamellar keratin formation (“wet keratin”) is a diagnostic feature of this tumor (Fig. 24-7). As was mentioned above, dystrophic calcification is a frequent finding. Additional features include cyst formation, fibrosis, and chronic inflammatory reaction. The cysts of adamantinomatous craniopharyngiomas often contain a cholesterol-rich, thick brownish-yellow fluid that has been compared to “machine oil.” These tumors extend fingerlets of epithelium into adjacent brain, where they elicit a brisk glial reaction.
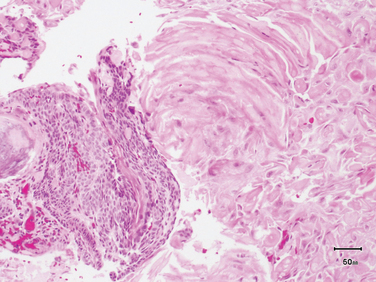
FIGURE 24-7 Adamantinomatous craniopharyngioma, demonstrating characteristic compact, lamellar “wet” keratin (right half of photomicrograph) and cords of squamous epithelium with peripheral palisading on the left.
(Courtesy of Dr. Charles Eberhart, Department of Pathology, Johns Hopkins University, Baltimore, MD.)
Papillary craniopharyngiomas contain both solid sheets and papillae lined by well-differentiated squamous epithelium. These tumors usually lack keratin, calcification, and cysts. The squamous cells of the solid sections of the tumor lack the peripheral palisading and do not typically generate a spongy reticulum in the internal layers.
Patients with craniopharyngiomas, especially those < 5 cm in diameter, have an excellent recurrencefree and overall survival. Larger lesions are more invasive but this does not impact on the prognosis. Malignant transformation of craniopharyngiomas into squamous carcinomas is exceptionally rare and usually occurs after irradiation.
THYROID GLAND
The thyroid gland consists of two bulky lateral lobes connected by a relatively thin isthmus, usually located below and anterior to the larynx. The thyroid is divided by thin fibrous septae into lobules composed of about 20 to 40 evenly dispersed follicles, lined by a cuboidal to low columnar epithelium, and filled with PAS-positive thyroglobulin. In response to hypothalamic factors, TSH (thyrotropin) is released by thyrotrophs in the anterior pituitary into the circulation. The binding of TSH to its receptor on the thyroid follicular epithelium results in conformational change and activation of the receptor, allowing it to associate with a Gs protein (Fig. 24-8). Activation of the G protein eventually results in an increase in intracellular cAMP levels, which stimulates thyroid growth, and hormone synthesis and release via cAMP-dependent protein kinases. The dissociation of thyroid hormone synthesis and release from the controlled influence of TSH-signaling pathways results in so-called thyroid autonomy and hyperfunction (see below).
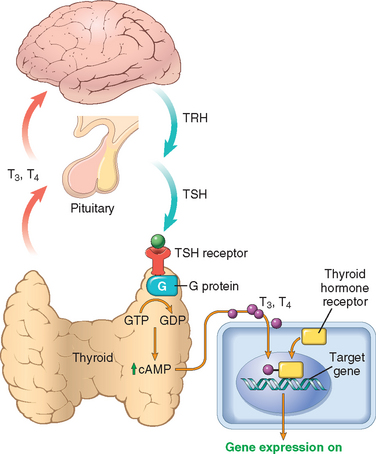
FIGURE 24-8 Homeostasis in the hypothalamus-pituitary-thyroid axis and mechanism of action of thyroid hormones. Secretion of thyroid hormones (T3 and T4) is controlled by trophic factors secreted by both the hypothalamus and the anterior pituitary. Decreased levels of T3 and T4 stimulate the release of thyrotropin-releasing hormone (TRH) from the hypothalamus and thyroid-stimulating hormone (TSH) from the anterior pituitary, causing T3 and T4 levels to rise. Elevated T3 and T4 levels, in turn, feed back to suppress the secretion of both TRH and TSH. TSH binds to the TSH receptor on the thyroid follicular epithelium, which causes activation of G proteins, and cAMP-mediated synthesis and release of thyroid hormones (T3 and T4). In the periphery, T3 and T4 interact with the thyroid hormone receptor (TR) to form a hormone-receptor complex that translocates to the nucleus and binds to so-called thyroid response elements (TREs) on target genes to initiate transcription.
Thyroid follicular epithelial cells convert thyroglobulin into thyroxine (T4) and lesser amounts of triiodothyronine (T3). T4 and T3 are released into the systemic circulation, where most of these peptides are reversibly bound to circulating plasma proteins, such as thyroxine-binding globulin and transthyretin, for transport to peripheral tissues. The binding proteins serve to maintain the serum unbound (“free”) T3 and T4 concentrations within narrow limits yet ensure that the hormones are readily available to the tissues. In the periphery, the majority of free T4 is deiodinated to T3; the latter binds to thyroid hormone nuclear receptors in target cells with tenfold greater affinity than does T4 and has proportionately greater activity. The interaction of thyroid hormone with its nuclear thyroid hormone receptor (TR) results in the formation of a multiprotein hormone-receptor complex that binds to thyroid hormone response elements (TREs) in target genes, regulating their transcription (see Fig. 24-8). Thyroid hormone has diverse cellular effects, including up-regulation of carbohydrate and lipid catabolism and stimulation of protein synthesis in a wide range of cells. The net result of these processes is an increase in the basal metabolic rate. One of the most important functions of thyroid hormone is its critical role in brain development in the fetus and neonate (see below).
The function of the thyroid gland can be inhibited by a variety of chemical agents, collectively referred to as goitrogens. Because they suppress T3 and T4 synthesis, the level of TSH increases, and subsequent hyperplastic enlargement of the gland (goiter) follows. The antithyroid agent propylthiouracil inhibits the oxidation of iodide and thus blocks production of the thyroid hormones; parenthetically, propylthiouracil also inhibits the peripheral deiodination of circulating T4 into T3, thus ameliorating symptoms of thyroid hormone excess (see below). Iodide, when given to individuals with thyroid hyperfunction, also blocks the release of thyroid hormones but through different mechanisms. Iodides in large doses inhibit proteolysis of thyroglobulin. Thus, thyroid hormone is synthesized and incorporated within increasing amounts of colloid, but it is not released into the blood.
The thyroid gland follicles also contain a population of parafollicular cells, or C cells, which synthesize and secrete the hormone calcitonin. This hormone promotes the absorption of calcium by the skeletal system and inhibits the resorption of bone by osteoclasts.
Diseases of the thyroid include conditions associated with excessive release of thyroid hormones (hyperthyroidism), those associated with thyroid hormone deficiency (hypothyroidism), and mass lesions of the thyroid. We first consider the clinical consequences of disturbed thyroid function, then focus on the disorders that generate these problems.
Hyperthyroidism
Thyrotoxicosis is a hypermetabolic state caused by elevated circulating levels of free T3 and T4. Because it is caused most commonly by hyperfunction of the thyroid gland, it is often referred to as hyperthyroidism. However, in certain conditions the oversupply is related to either excessive release of preformed thyroid hormone (e.g., in thyroiditis) or to an extra-thyroidal source, rather than hyperfunction of the gland (Table 24-3). Thus, strictly speaking, hyperthyroidism is only one (albeit the most common) cause of thyrotoxicosis. The terms primary and secondary hyperthyroidism are sometimes used to designate hyperthyroidism arising from an intrinsic thyroid abnormality and that arising from processes outside of the thyroid, such as a TSH-secreting pituitary tumor. With this caveat, we will follow the common practice of using the terms thyrotoxicosis and hyperthyroidism interchangeably. The three most common causes of thyrotoxicosis are also associated with hyperfunction of the gland and include the following:
TABLE 24-3 Disorders Associated with Thyrotoxicosis
| ASSOCIATED WITH HYPERTHYROIDISM |
| Primary |
| Secondary |
| TSH-secreting pituitary adenoma (rare)* |
| NOT ASSOCIATED WITH HYPERTHYROIDISM |
* Associated with increased thyroid-stimulating hormone (TSH); all other causes of thyrotoxicosis associated with decreased TSH.
Clinical Course.
The clinical manifestations of hyperthyroidism are protean and include changes referable to the hypermetabolic state induced by excess thyroid hormone and to overactivity of the sympathetic nervous system (i.e., an increase in the β-adrenergic “tone”).
Excessive levels of thyroid hormone result in an increase in the basal metabolic rate. The skin of thyrotoxic patients tends to be soft, warm, and flushed because of increased blood flow and peripheral vasodilation to increase heat loss. Heat intolerance is common. Sweating is increased because of higher levels of calorigenesis. Increased basal metabolic rate also results in characteristic weight loss despite increased appetite.
Cardiac manifestations are among the earliest and most consistent features of hyperthyroidism. Individuals with hyperthyroidism can have an increase in cardiac output, due to both increased cardiac contractility and increased peripheral oxygen requirements. Tachycardia, palpitations, and cardiomegaly are common. Arrhythmias, particularly atrial fibrillation, occur frequently and are more common in older patients. Congestive heart failure may develop, particularly in elderly patients with preexisting cardiac disease. Myocardial changes, such as foci of lymphocytic and eosinophilic infiltration, mild fibrosis in the interstitium, fatty changes in myofibers, and an increase in size and number of mitochondria, have been described. Some individuals with thyrotoxicosis develop reversible left ventricular dysfunction and “low-output” heart failure, so-called thyrotoxic or hyperthyroid cardiomyopathy.
In the neuromuscular system, overactivity of the sympathetic nervous system produces tremor, hyperactivity, emotional lability, anxiety, inability to concentrate, and insomnia. Proximal muscle weakness and decreased muscle mass are common (thyroid myopathy).
Ocular changes often call attention to hyperthyroidism. A wide, staring gaze and lid lag are present because of sympathetic overstimulation of the levator palpebrae superioris (Fig. 24-9). However, true thyroid ophthalmopathy associated with proptosis is seen only in Graves disease (see below).
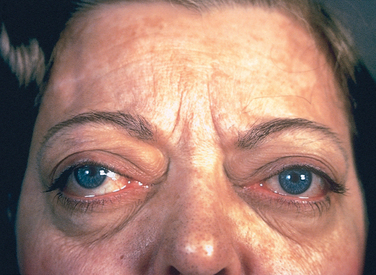
FIGURE 24-9 A person with hyperthyroidism. A wide-eyed, staring gaze, caused by overactivity of the sympathetic nervous system, is one of the features of this disorder. In Graves disease, one of the most important causes of hyperthyroidism, accumulation of loose connective tissue behind the eyeballs, also adds to the protuberant appearance of the eyes.
In the gastrointestinal system, sympathetic hyperstimulation of the gut results in hypermotility, malabsorption, and diarrhea.
The skeletal system is also affected. Thyroid hormone stimulates bone resorption, increasing porosity of cortical bone and reducing the volume of trabecular bone. The net effect is osteoporosis and an increased risk of fractures in patients with chronic hyperthyroidism.
Other findings include atrophy of skeletal muscle, with fatty infiltration and focal interstitial lymphocytic infiltrates; minimal liver enlargement due to fatty changes in the hepatocytes; and generalized lymphoid hyperplasia and lymphadenopathy in patients with Graves disease.
Thyroid storm is used to designate the abrupt onset of severe hyperthyroidism. This condition occurs most commonly in patients with underlying Graves disease and probably results from an acute elevation in catecholamine levels, as might be encountered during infection, surgery, cessation of antithyroid medication, or any form of stress. Patients are often febrile and present with tachycardia out of proportion to the fever. Thyroid storm is a medical emergency: A significant number of untreated patients die of cardiac arrhythmias.
Apathetic hyperthyroidism refers to thyrotoxicosis occurring in the elderly, in whom advanced age and various co-morbidities may blunt the typical features of thyroid hormone excess seen in younger patients. The diagnosis of thyrotoxicosis in these individuals is often made during laboratory work-up for unexplained weight loss or worsening cardiovascular disease.
A diagnosis of hyperthyroidism is made using both clinical and laboratory findings. The measurement of serum TSH concentration using sensitive TSH assays provides the most useful single screening test for hyperthyroidism, since its levels are decreased even at the earliest stages, when the disease may still be subclinical. A low TSH value is usually confirmed with measurement of free T4, which is expectedly increased. In an occasional patient, hyperthyroidism results predominantly from increased circulating levels of T3 (“T3 toxicosis”). In these cases, free T4 levels may be decreased, and direct measurement of serum T3 may be useful. In rare cases of pituitary-associated (secondary) hyperthyroidism, TSH levels are either normal or raised. Determining TSH levels after the injection of thyrotropin-releasing hormone (TRH stimulation test) is used in the evaluation of cases of suspected hyperthyroidism with equivocal changes in the baseline serum TSH level. A normal rise in TSH after administration of TRH excludes secondary hyperthyroidism. Once the diagnosis of thyrotoxicosis has been confirmed by a combination of TSH assays and free thyroid hormone levels, measurement of radioactive iodine uptake by the thyroid gland may be valuable in determining the etiology. For example, there may be diffusely increased uptake in the whole gland (Graves’ disease), increased uptake in a solitary nodule (toxic adenoma), or decreased uptake (thyroiditis).
The therapeutic options for hyperthyroidism include multiple medications, each of which has a different mechanism of action. Typically, these include a β-blocker to control symptoms induced by increased adrenergic tone, a thionamide to block new hormone synthesis, an iodine solution to block the release of thyroid hormone, and agents that inhibit peripheral conversion of T4 to T3. Radioiodine, which is incorporated into thyroid tissues, resulting in ablation of thyroid function over a period of 6 to 18 weeks, may also be used.
Hypothyroidism
Hypothyroidism is caused by any structural or functional derangement that interferes with the production of adequate levels of thyroid hormone. Hypothyroidism is a fairly common disorder, and by some estimates the population prevalence of overt hypothyroidism is 0.3%, while subclinical hypothyroidism can be found in greater than 4%.7 The prevalence of hypothyroidism increases with age, and it is nearly tenfold more common in women than in men. It can result from a defect anywhere in the hypothalamic-pituitary-thyroid axis. As in the case of hyperthyroidism, this disorder is divided into primary and secondary categories, depending on whether the hypothyroidism arises from an intrinsic abnormality in the thyroid itself, or occurs as a result of pituitary and hypothalamic disease (Table 24-4). Primary hypothyroidism accounts for the vast majority of cases of hypothyroidism, and can be accompanied by an enlargement in the size of the thyroid gland (goiter). Primary hypothyroidism can be congenital, acquired, or autoimmune.
TABLE 24-4 Causes of Hypothyroidism
| PRIMARY |
| SECONDARY (CENTRAL) |
* Associated with enlargement of thyroid (“goitrous hypothyroidism”). Hashimoto thyroiditis and postablative hypothyroidism account for the majority of cases of hypothyroidism in developed countries. FOXE1, forkhead box E1; PAX8, paired box 8; THRB, thyroid hormone receptor β.
Worldwide, congenital hypothyroidism is most often the result of endemic iodine deficiency in the diet (see below). Other less common forms of congenital hypothyroidism include inborn errors of thyroid metabolism (dyshormonogenetic goiter), wherein any one of the multiple steps leading to thyroid hormone synthesis may be deficient, such as (1) iodide transport into thyrocytes, (2) iodide “organification” (binding of iodide to tyrosine residues of the storage protein, thyroglobulin), and (3) iodotyrosine coupling to form hormonally active T3 and T4. Mutations in the thyroid peroxidase (TPO) gene are the most common cause of dyshormonogenetic goiter. Pendred syndrome, characterized by hypothyroidism and sensorineural deafness, is caused by mutations in the SLC26A4 gene, whose product, pendrin, is an anion transporter expressed on the apical surface of thyrocytes and in the inner ear.8 In rare instances there may be complete absence of thyroid parenchyma (thyroid agenesis), or the gland may be greatly reduced in size (thyroid hypoplasia). Germline mutations in transcription factors that are expressed in the developing thyroid and regulate follicular differentiation, such as thyroid transcription factor-2 (TTF-2), also known as FOXE1, and paired box-8 (PAX-8), have been reported in individuals with thyroid agenesis. These patients typically present with a constellation of extra-thyroidal malformations. Inactivating germline mutations of the TSH receptor (TSHR) is a rare genetic cause of isolated hypothyroidism (note that activating somatic mutations of TSHR are found in autonomous thyroid nodules, see below). Thyroid hormone resistance syndrome is a rare autosomal-dominant disorder caused by inherited mutations in the thyroid hormone receptor, which abolish the ability of the receptor to bind thyroid hormones. Patients demonstrate a generalized resistance to thyroid hormone, despite high circulating levels of T3 and T4. Since the pituitary is also resistant to feedback from thyroid hormones, TSH levels tend to be high as well.
Acquired hypothyroidism can be caused by surgical or radiation-induced ablation of thyroid parenchyma. A large resection of the gland (total thyroidectomy) for the treatment of hyperthyroidism of a primary neoplasm can lead to hypothyroidism. The gland may also be ablated by radiation, whether in the form of radioiodine administered for the treatment of hyperthyroidism, or exogenous irradiation, such as external radiation therapy to the neck. Drugs given intentionally to decrease thyroid secretion (e.g., methimazole and propylthiouracil) can cause acquired hypothyroidism, as can agents used to treat nonthyroid conditions (e.g., lithium, p-aminosalicylic acid).
Autoimmune hypothyroidism is the most common cause of hypothyroidism in iodine-sufficient areas of the world. The vast majority of cases of autoimmune hypothyroidism are due to Hashimoto thyroiditis. Circulating autoantibodies, including anti-microsomal, anti-thyroid peroxidase, and anti-thyroglobulin antibodies, are found in this disorder, and the thyroid is typically enlarged (goitrous). Autoimmune hypothyroidism can occur in isolation or in conjunction with autoimmune polyendocrine syndrome (APS), types 1 and 2 (see discussion in “Adrenal Glands”).
Secondary (or central) hypothyroidism is caused by deficiency of TSH, and far more uncommonly, that of TRH. Any of the causes of hypopituitarism (for example, pituitary tumor, postpartum pituitary necrosis, trauma, and nonpituitary tumors), or of hypothalamic damage from tumors, trauma, radiation therapy, or infiltrative diseases can cause central hypothyroidism.
Classic clinical manifestations of hypothyroidism include cretinism and myxedema.
CRETINISM
Cretinism refers to hypothyroidism that develops in infancy or early childhood. The term cretin was derived from the French chrétien, meaning “Christian” or “Christlike,” and was applied to these unfortunates because they were considered to be so mentally retarded as to be incapable of sinning. In the past this disorder occurred fairly commonly in areas of the world where dietary iodine deficiency is endemic, such as the Himalayas, inland China, Africa, and other mountainous areas. It has become much less frequent in recent years, as a result of the widespread supplementation of foods with iodine. On rare occasions, cretinism may also result from inborn errors in metabolism that interfere with the biosynthesis of normal levels of thyroid hormone (dyshormonogenetic goiter, see above).
Clinical features of cretinism include impaired development of the skeletal system and central nervous system, manifested by severe mental retardation, short stature, coarse facial features, a protruding tongue, and umbilical hernia. The severity of the mental impairment in cretinism seems to be related to the time at which thyroid deficiency occurs in utero. Normally, maternal hormones, including T3 and T4, cross the placenta and are critical to fetal brain development. If there is maternal thyroid deficiency before the development of the fetal thyroid gland, mental retardation is severe. In contrast, reduction in maternal thyroid hormones later in pregnancy, after the fetal thyroid has developed, allows normal brain development.
MYXEDEMA
The term myxedema is applied to hypothyroidism developing in the older child or adult. Myxedema, or Gull disease, was first linked with thyroid dysfunction in 1873 by Sir William Gull in an article addressing the development of a “cretinoid state” in adults.9 The clinical manifestations vary with the age of onset of the deficiency. The older child shows signs and symptoms intermediate between those of the cretin and those of the adult with hypothyroidism. In the adult the condition appears insidiously and may take years to reach the level of clinical suspicion.
Clinical features of myxedema are characterized by a slowing of physical and mental activity. The initial symptoms include generalized fatigue, apathy, and mental sluggishness, which may mimic depression in the early stages of the disease. Speech and intellectual functions become slowed. Patients with myxedema are listless, cold intolerant, and frequently overweight. Decreased sympathetic activity results in constipation and decreased sweating. The skin of these patients is cool and pale because of decreased blood flow. Reduced cardiac output probably contributes to shortness of breath and decreased exercise capacity, two frequent complaints in individuals with hypothyroidism. Thyroid hormones regulate the transcription of several sarcolemmal genes, such as calcium ATPases, whose encoded products are critical in maintaining efficient cardiac output. In addition, hypothyroidism promotes an atherogenic profile—an increase in total cholesterol and low-density lipoprotein (LDL) levels—probably contributing toward the adverse cardiovascular mortality rates in this disease. Histologically, there is an accumulation of matrix substances, such as glycosaminoglycans and hyaluronic acid, in skin, subcutaneous tissue, and a number of visceral sites. This results in non-pitting edema, a broadening and coarsening of facial features, enlargement of the tongue, and deepening of the voice.
Laboratory evaluation plays a vital role in the diagnosis of suspected hypothyroidism because of the nonspecific nature of symptoms. Patients with unexplained increase in body weight or hypercholesterolemia should be assessed for potential hypothyroidism. Measurement of the serum TSH level is the most sensitive screening test for this disorder. The TSH level is increased in primary hypothyroidism as a result of a loss of feedback inhibition of TRH and TSH production by the hypothalamus and pituitary, respectively. The TSH level is not increased in persons with hypothyroidism due to primary hypothalamic or pituitary disease. T4 levels are decreased in individuals with hypothyroidism of any origin.
Thyroiditis
Thyroiditis, or inflammation of the thyroid gland, encompasses a diverse group of disorders characterized by some form of thyroid inflammation. These diseases include conditions that result in acute illness with severe thyroid pain (e.g., infectious thyroiditis, subacute granulomatous thyroiditis) and disorders in which there is relatively little inflammation and the illness is manifested primarily by thyroid dysfunction—subacute lymphocytic thyroiditis and fibrous (Reidel) thyroiditis.
Infectious thyroiditis may be either acute or chronic. Acute infections can reach the thyroid by hematogenous spread or through direct seeding of the gland, such as through a fistula from the piriform sinus adjacent to the larynx. Other infections of the thyroid, including mycobacterial, fungal, and Pneumocystis infections, are more chronic and frequently occur in immunocompromised patients. Whatever the cause, the inflammatory involvement may cause sudden onset of neck pain and tenderness in the area of the gland and is accompanied by fever, chills, and other signs of infection. Infectious thyroiditis can be self-limited or can be controlled with appropriate therapy. Thyroid function is usually not significantly affected, and there are few residual effects except for possible small foci of scarring. This section focuses on the more common and clinically significant types of thyroiditis: (1) Hashimoto thyroiditis, (2) granulomatous (de Quervain) thyroiditis, and (3) subacute lymphocytic thyroiditis.
HASHIMOTO THYROIDITIS
Hashimoto thyroiditis is the most common cause of hypothyroidism in areas of the world where iodine levels are sufficient. The name Hashimoto thyroiditis is derived from the 1912 report by Hashimoto describing patients with goiter and intense lymphocytic infiltration of the thyroid (struma lymphomatosa).10 Hashimoto thyroiditis and Graves disease (see below) are the two most common immunologically mediated disorders of the thyroid. Hashimoto thyroiditis is characterized by gradual thyroid failure because of autoimmune destruction of the thyroid gland. This disorder is most prevalent between 45 and 65 years of age and is more common in women than in men, with a female predominance of 10 : 1 to 20 : 1. Although it is primarily a disease of older women, it can occur in children and is a major cause of nonendemic goiter in the pediatric population.
Akin to other autoimmune diseases, Hashimoto thyroiditis has a strong genetic component. This is supported by the concordance of disease in as many as 40% of monozygotic twins, as well as the presence of circulating antithyroid antibodies in approximately 50% of asymptomatic siblings of Hashimoto patients. Increased susceptibility to Hashimoto thyroiditis is associated with polymorphisms in multiple immune regulation–associated genes, the most significant of which is the linkage to cytotoxic T lymphocyte–associated antigen-4 (CTLA4) polymorphisms.11 CTLA4 is a negative regulator of T-cell responses, and not surprisingly, polymorphisms of the CTLA4 gene that result in reduced protein level or function are associated with a predisposition to autoimmune disease. Another recently described genetic determinant of susceptibility to Hashimoto thyroiditis is a functional polymorphism in protein tyrosine phosphatase-22 (PTPN22) gene that encodes a lymphoid tyrosine phosphatase, which is also thought to inhibit T-cell function.12 Susceptibility to other autoimmune diseases, such as type 1 diabetes (see below), has been associated with polymorphisms in both CTLA4 and PTPN22.
Pathogenesis.
Hashimoto thyroiditis is caused by a breakdown in self-tolerance to thyroid auto-antigens. This is exemplified by the presence of circulating autoantibodies against thyroglobulin and thyroid peroxidase in the vast majority of Hashimoto patients. The inciting events leading to breakdown in self-tolerance in Hashimoto patients have not been fully elucidated, but possibilities include abnormalities of regulatory T cells (Tregs)13, or exposure of normally sequestered thyroid antigens (Chapter 6). Induction of thyroid autoimmunity is accompanied by a progressive depletion of thyrocytes by apoptosis and replacement of the thyroid parenchyma by mononuclear cell infiltration and fibrosis. Multiple immunologic mechanisms may contribute to thyroid cell death, including (Fig. 24-10):
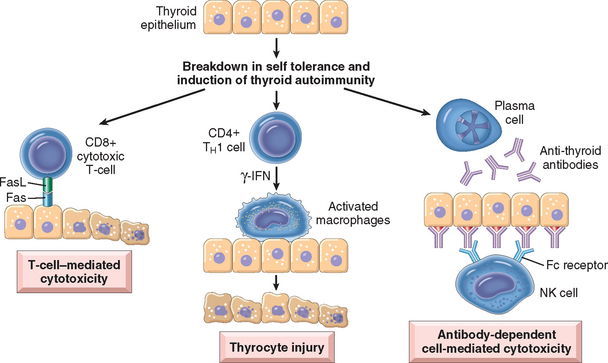
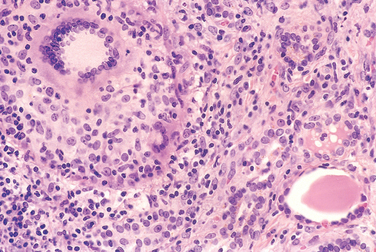
FIGURE 24-10 Pathogenesis of Hashimoto thyroiditis. Breakdown of peripheral tolerance to thyroid auto-antigens, results in progressive autoimmune destruction of thyrocytes by infiltrating cytotoxic T cells, locally released cytokines, or by antibody-dependent cytotoxicity.
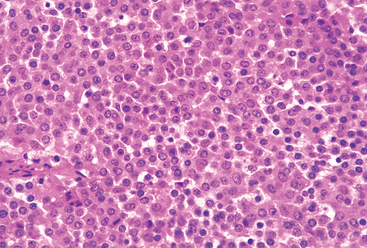
Morphology. The thyroid is often diffusely enlarged, although more localized enlargement may be seen in some cases. The capsule is intact, and the gland is well demarcated from adjacent structures. The cut surface is pale, yellowtan, firm, and somewhat nodular. Microscopic examination reveals extensive infiltration of the parenchyma by a mononuclear inflammatory infiltrate containing small lymphocytes, plasma cells, and well-developed germinal centers (Fig. 24-11). The thyroid follicles are atrophic and are lined in many areas by epithelial cells distinguished by the presence of abundant eosinophilic, granular cytoplasm, termed Hürthle cells. This is a metaplastic response of the normally low cuboidal follicular epithelium to ongoing injury. In fine-needle aspiration biopsy samples, the presence of Hürthle cells in conjunction with a heterogeneous population of lymphocytes is characteristic of Hashimoto thyroiditis. In “classic” Hashimoto thyroiditis, interstitial connective tissue is increased and may be abundant. A fibrous variant is characterized by severe thyroid follicular atrophy and dense “keloid-like” fibrosis, broad bands of acellular collagen encompassing residual thyroid tissue. Unlike Reidel thyroiditis (see below), the fibrosis does not extend beyond the capsule of the gland. The remnant thyroid parenchyma demonstrates features of chronic lymphocytic thyroiditis.
Clinical Course.
Hashimoto thyroiditis most often comes to clinical attention as painless enlargement of the thyroid, usually associated with some degree of hypothyroidism, in a middle-aged woman. The enlargement of the gland is usually symmetric and diffuse, but in some cases it may be sufficiently localized to raise a suspicion of neoplasm. In the usual clinical course, hypothyroidism develops gradually. In some cases, however, it may be preceded by transient thyrotoxicosis caused by disruption of thyroid follicles, with secondary release of thyroid hormones (“hashitoxicosis”). During this phase, free T4 and T3 levels are elevated, TSH is diminished, and radioactive iodine uptake is decreased. As hypothyroidism supervenes, T4 and T3 levels fall, accompanied by a compensatory increase in TSH. Individuals with Hashimoto thyroiditis are at increased risk for developing other autoimmune diseases, both endocrine (type 1 diabetes, autoimmune adrenalitis) and nonendocrine (systemic lupus erythematosus, myasthenia gravis, and Sjögren syndrome; see Chapter 6). They are also at increased risk for the development of B-cell non-Hodgkin lymphomas, especially marginal zone lymphomas of mucosa-associated lymphoid tissues (MALT lymphomas; see Chapter 13). The relationship between Hashimoto disease and thyroid epithelial cancers remains controversial, with some morphologic and molecular studies suggesting a predisposition to papillary carcinomas.
SUBACUTE (GRANULOMATOUS) THYROIDITIS
Subacute thyroiditis, which is also referred to as granulomatous thyroiditis or De Quervain thyroiditis, occurs much less frequently than does Hashimoto disease. The disorder is most common between the ages of 40 and 50 and, like other forms of thyroiditis, affects women considerably more often than men (4 : 1).
Pathogenesis.
Subacute thyroiditis is believed to be triggered by a viral infection. The majority of patients have a history of an upper respiratory infection just before the onset of thyroiditis. The disease has a seasonal incidence, with occurrences peaking in the summer, and clusters of cases have been reported in association with coxsackievirus, mumps, measles, adenovirus, and other viral illnesses. Although the pathogenesis of the disease is unclear, one model suggests that it results from a viral infection that leads to exposure to a viral or thyroid antigen, which is released secondary to virus-induced host tissue damage. This antigen stimulates cytotoxic T lymphocytes, which then damage thyroid follicular cells. In contrast to autoimmune thyroid disease, the immune response is virus-initiated and not self-perpetuating, so the process is limited.
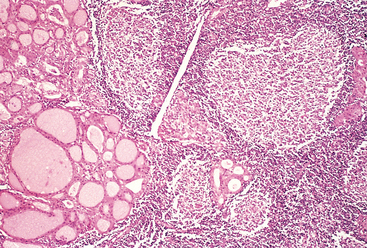
Morphology. The gland may be unilaterally or bilaterally enlarged and firm, with an intact capsule. It may be slightly adherent to surrounding structures. On cut section, the involved areas are firm and yellow-white and stand out from the more rubbery, normal brown thyroid substance. Histologically, the changes are patchy and depend on the stage of the disease. Early in the active inflammatory phase, scattered follicles may be entirely disrupted and replaced by neutrophils forming microabscesses. Later, the more characteristic features appear in the form of aggregates of lymphocytes, activated macrophages, and plasma cells about collapsed and damaged thyroid follicles. Multinucleate giant cells enclose naked pools or fragments of colloid (Fig. 24-12), hence the designation granulomatous thyroiditis. In later stages of the disease a chronic inflammatory infiltrate and fibrosis may replace the foci of injury. Different histologic stages are sometimes found in the same gland, suggesting waves of destruction over a period of time.
Clinical Course.
Granulomatous (de Quervain) thyroiditis is the most common cause of thyroid pain. There is a variable enlargement of the thyroid. The thyroid inflammation and hyperthyroidism are transient, usually diminishing in 2 to 6 weeks, even if the patient is not treated. Nearly all patients have high serum T4 and T3 levels and low serum TSH levels during this phase. However, unlike in hyperthyroid states such as Graves disease, radioactive iodine uptake is diminished. After recovery, generally in 6 to 8 weeks, normal thyroid function returns.
SUBACUTE LYMPHOCYTIC (PAINLESS) THYROIDITIS
Subacute lymphocytic thyroiditis, which is also referred to as painless thyroiditis, usually comes to clinical attention because of mild hyperthyroidism, goitrous enlargement of the gland, or both. Although it can occur at any age, it is most often seen in middle-aged adults and is more common in women. A disease process resembling painless thyroiditis can occur during the postpartum period in up to 5% of women (postpartum thyroiditis). Painless and postpartum thyroiditides are variants of Hashimoto thyroiditis, since the majority of patients have circulating anti-thyroid peroxidase antibodies or a family history of other autoimmune disorders. As many as a third of cases can evolve into overt hypothyroidism over time, and the thyroid histology is also reminiscent of Hashimoto thyroiditis (see below).
Morphology. Except for possible mild symmetric enlargement, the thyroid appears normal on gross inspection. The most specific histologic features consist of lymphocytic infiltration with hyperplastic germinal centers within the thyroid parenchyma and patchy disruption and collapse of thyroid follicles. Unlike in frank Hashimoto thyroiditis, however, fibrosis and Hürthle cell metaplasia are not prominent features.
Clinical Course.
Individuals with painless thyroiditis can present with a painless goiter, transient overt hyperthyroidism, or both. Some patients transition from a hyperthyroid state to hypothyroidism before recovery. The vast majority (∼80%) of individuals with postpartum thyroiditis are euthyroid by 1 year. Postpartum thyroiditis can resemble Graves disease, the incidence of which is also increased in the setting of pregnancy. Infiltrative ophthalmopathy and other manifestations of Graves disease (see below) are, however, not present in the former. As stated, as many as a third of affected individuals eventually progress to overt hypothyroidism over a 10-year period.
Other, less common forms of thyroiditis include Riedel thyroiditis, a rare disorder of unknown etiology characterized by extensive fibrosis involving the thyroid and contiguous neck structures. The presence of a hard and fixed thyroid mass clinically simulates a thyroid carcinoma. It may be associated with idiopathic fibrosis in other sites in the body, such as the retroperitoneum. The presence of circulating anti-thyroid antibodies in most patients suggests an autoimmune etiology.
Graves Disease
Graves reported in 1835 his observations of a disease characterized by “violent and long continued palpitations in females” associated with enlargement of the thyroid gland.14 Graves disease is the most common cause of endogenous hyperthyroidism. It is characterized by a triad of clinical findings:
Graves disease has a peak incidence between 20 and 40 years of age. Women are affected as much as 10 times more frequently than men. This disorder is said to be present in 1.5% to 2.0% of women in the United States. As previously mentioned, Graves disease (hyperthyroidism) and Hashimoto thyroiditis (hypothyroidism) span two extremes of autoimmune thyroid disorders, and not surprisingly share many underlying tenets. For example, akin to Hashimoto thyroiditis, genetic factors are also important in the etiology of Graves disease, with a concordance rate in monozygotic twins of 30% to 40%, as compared with less than 5% among dizygotic individuals. Like Hashimoto thyroiditis, genetic susceptibility for Graves disease has been linked to polymorphisms in immune-function genes like CTLA4 and PTPN22 and the HLA-DR3 allele.15
Pathogenesis.
Graves disease is characterized by a breakdown in self-tolerance to thyroid auto-antigens, most importantly the TSH receptor. The result is the production of multiple autoantibodies, including:
The key role of anti–TSH receptor antibodies in the pathogenesis of hyperthyroidism is supported by animal models of Graves disease. Immunization of mice with the TSH receptor results in generation of antibodies that cause thyroid stimulation, thyroid enlargement with lymphocytic infiltration, elevated thyroxine levels, and, in a subset of mice, ocular signs reminiscent of Graves ophthalmopathy (see below).
Autoimmunity also plays a role in the development of the infiltrative ophthalmopathy that is characteristic of Graves disease. In Graves ophthalmopathy, the volume of the retro-orbital connective tissues and extraocular muscles is increased for several reasons, including (1) marked infiltration of the retro-orbital space by mononuclear cells, predominantly T cells; (2) inflammatory edema and swelling of extraocular muscles; (3) accumulation of extracellular matrix components, specifically hydrophilic glycosaminoglycans such as hyaluronic acid and chondroitin sulfate; and (4) increased numbers of adipocytes (fatty infiltration). These changes displace the eyeball forward and can interfere with the function of the extraocular muscles. Recent evidence suggests that orbital preadipocyte fibroblasts express the TSH receptor and thus become targets of an autoimmune attack. T cells reactive against these fibroblasts secrete cytokines, which stimulate fibroblast proliferation and synthesis of extracellular matrix proteins (glycosaminoglycans) and increase surface TSH receptor expression, perpetuating the autoimmune response. The result is progressive infiltration of the retro-orbital space and ophthalmopathy.
Morphology. The thyroid gland is usually symmetrically enlarged because of diffuse hypertrophy and hyperplasia of thyroid follicular epithelial cells (Fig. 24-13A). Increases in weight to over 80 gm are not uncommon. On cut section, the parenchyma has a soft, meaty appearance resembling normal muscle. Histologically, the follicular epithelial cells in untreated cases are tall and more crowded than usual. This crowding often results in the formation of small papillae, which project into the follicular lumen and encroach on the colloid, sometimes filling the follicles (Fig. 24-13B). Such papillae lack fibrovascular cores, in contrast to those of papillary carcinoma (see below). The colloid within the follicular lumen is pale, with scalloped margins. Lymphoid infiltrates, consisting predominantly of T cells, with fewer B cells and mature plasma cells, are present throughout the interstitium; germinal centers are common.
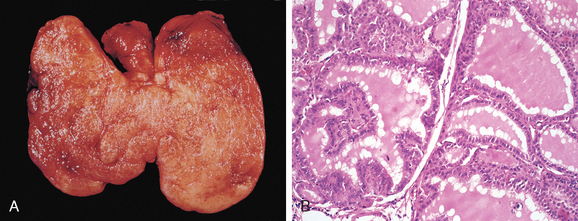
FIGURE 24-13 Graves disease. A, There is diffuse symmetric enlargement of the gland and a beefy deep red parenchyma. Compare with gross photograph of multinodular goiter in Figure 24-15. B, Diffusely hyperplastic thyroid in a case of Graves’ disease. The follicles are lined by tall, columnar epithelium. The crowded, enlarged epithelial cells project into the lumens of the follicles. These cells actively resorb the colloid in the centers of the follicles, resulting in the scalloped appearance of the edges of the colloid.
(A, Reproduced with permission from Lloyd RV et al. (eds): Atlas of Nontumor Pathology: Endocrine Diseases. Washington, DC, American Registry of Pathology, 2002.)
Preoperative therapy alters the morphology of the thyroid in Graves disease. Preoperative administration of iodine causes involution of the epithelium and the accumulation of colloid by blocking thyroglobulin secretion. Treatment with the antithyroid drug propylthiouracil exaggerates the epithelial hypertrophy and hyperplasia by stimulating TSH secretion. Thus, in pretreated patients it is impossible from histologic examination of surgical specimens to evaluate the functional activity of the gland.
Changes in extra-thyroidal tissue include generalized lymphoid hyperplasia. The heart may be hypertrophied, and ischemic changes may be present, particularly in patients with preexisting coronary artery disease. In patients with ophthalmopathy, the tissues of the orbit are edematous because of the presence of hydrophilic mucopolysaccharides. In addition, there is infiltration by lymphocytes and fibrosis. Orbital muscles are edematous initially but may undergo fibrosis late in the course of the disease. The dermopathy, if present, is characterized by thickening of the dermis due to deposition of glycosaminoglycans and lymphocyte infiltration.
Clinical Course.
The clinical findings in Graves disease include changes referable to thyrotoxicosis as well as those associated uniquely with Graves disease, diffuse hyperplasia of the thyroid, ophthalmopathy, and dermopathy. The degree of thyrotoxicosis varies from case to case and is sometimes less conspicuous than other manifestations of the disease. Diffuse enlargement of the thyroid is present in all cases. The thyroid enlargement may be accompanied by increased flow of blood through the hyperactive gland, often producing an audible “bruit.” Sympathetic overactivity produces a characteristic wide, staring gaze and lid lag. The ophthalmopathy of Graves disease results in abnormal protrusion of the eyeball (exophthalmos). The extraocular muscles are often weak. The exophthalmos may persist or progress despite successful treatment of the thyrotoxicosis, sometimes resulting in corneal injury. The infiltrative dermopathy, or pretibial myxedema, is most common in the skin overlying the shins, where it presents as scaly thickening and induration. However, it is present only in a minority of patients. Sometimes individuals spontaneously develop thyroid hypofunction. Patients are at increased risk for other autoimmune diseases, such as systemic lupus erythematosus, pernicious anemia, type 1 diabetes, and Addison disease.
Laboratory findings in Graves disease include elevated free T4 and T3 levels and depressed TSH levels. Because of ongoing stimulation of the thyroid follicles by thyroid-stimulating immunoglobulins, radioactive iodine uptake is increased, and radioiodine scans show a diffuse uptake of iodine.
Graves disease is treated with β-blockers, which address symptoms related to the increased β-adrenergic tone (e.g., tachycardia, palpitations, tremulousness, and anxiety), and by measures aimed at decreasing thyroid hormone synthesis, such as the administration of thionamides (e.g., propylthiouracil), radioiodine ablation, and surgical intervention.
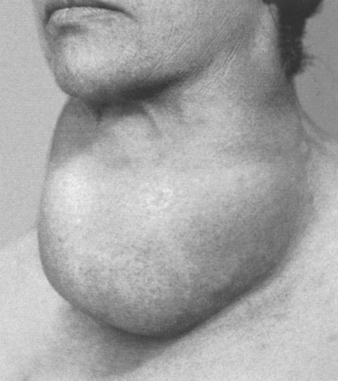
Diffuse and Multinodular Goiters
Enlargement of the thyroid, or goiter, is the most common manifestation of thyroid disease. Diffuse and multinodular goiters reflect impaired synthesis of thyroid hormone, which is most often caused by dietary iodine deficiency. Impairment of thyroid hormone synthesis leads to a compensatory rise in the serum TSH level, which, in turn, causes hypertrophy and hyperplasia of thyroid follicular cells and, ultimately, gross enlargement of the thyroid gland. The compensatory increase in functional mass of the gland is able to overcome the hormone deficiency, ensuring a euthyroid metabolic state in most individuals. If the underlying disorder is sufficiently severe (e.g., a congenital biosynthetic defect or endemic iodine deficiency, see below), the compensatory responses may be inadequate to overcome the impairment in hormone synthesis, resulting in goitrous hypothyroidism. The degree of thyroid enlargement is proportional to the level and duration of thyroid hormone deficiency.
DIFFUSE NONTOXIC (SIMPLE) GOITER
Diffuse nontoxic (simple) goiter causes enlargement of the entire gland without producing nodularity. Because the enlarged follicles are filled with colloid, the term colloid goiter has been applied to this condition. This disorder occurs in both an endemic and a sporadic distribution.
Endemic goiter occurs in geographic areas where the soil, water, and food supply contain low levels of iodine. The term endemic is used when goiters are present in more than 10% of the population in a given region. Such conditions are particularly common in mountainous areas of the world, including the Andes and Himalayas, where iodine deficiency is widespread. The lack of iodine leads to decreased synthesis of thyroid hormone and a compensatory increase in TSH, leading to follicular cell hypertrophy and hyperplasia and goitrous enlargement. With increasing dietary iodine supplementation, the frequency and severity of endemic goiter have declined significantly, although as many as 200 million people worldwide continue to be at risk for severe iodine deficiency.
Variations in the prevalence of endemic goiter in regions with similar levels of iodine deficiency point to the existence of other causative influences, particularly dietary substances, referred to as goitrogens. The ingestion of substances that interfere with thyroid hormone synthesis at some level, such as vegetables belonging to the Brassicaceae (Cruciferae) family (e.g., cabbage, cauliflower, Brussels sprouts, turnips, and cassava), has been documented to be goitrogenic. Native populations subsisting on cassava root are particularly at risk. Cassava contains a thiocyanate that inhibits iodide transport within the thyroid, worsening any possible concurrent iodine deficiency.
Sporadic goiter occurs less frequently than does endemic goiter. There is a striking female preponderance and a peak incidence at puberty or in young adult life. Sporadic goiter can be caused by several conditions, including the ingestion of substances that interfere with thyroid hormone synthesis. In other instances, goiter may result from hereditary enzymatic defects that interfere with thyroid hormone synthesis, all transmitted as autosomal-recessive conditions (dyshormonogenetic goiter; see above). In most cases, however, the cause of sporadic goiter is not apparent.
Morphology. Two phases can be identified in the evolution of diffuse nontoxic goiter: the hyperplastic phase and the phase of colloid involution. In the hyperplastic phase, the thyroid gland is diffusely and symmetrically enlarged, although the increase is usually modest, and the gland rarely exceeds 100 to 150 gm. The follicles are lined by crowded columnar cells, which may pile up and form projections similar to those seen in Graves disease. The accumulation is not uniform throughout the gland, and some follicles are hugely distended, whereas others remain small. If dietary iodine subsequently increases or if the demand for thyroid hormone decreases, the stimulated follicular epithelium involutes to form an enlarged, colloid-rich gland (colloid goiter). In these cases the cut surface of the thyroid is usually brown, somewhat glassy, and translucent. Histologically the follicular epithelium is flattened and cuboidal, and colloid is abundant during periods of involution.
Clinical Course.
As stated above, the vast majority of persons with simple goiters are clinically euthyroid. Therefore, the clinical manifestations are primarily related to mass effects from the enlarged thyroid gland (Fig. 24-14). Although serum T3 and T4 levels are normal, the serum TSH is usually elevated or at the upper range of normal, as is expected in marginally euthyroid individuals. In children, dyshormonogenetic goiter, caused by a congenital biosynthetic defect, may induce cretinism.
MULTINODULAR GOITER
With time, recurrent episodes of hyperplasia and involution combine to produce a more irregular enlargement of the thyroid, termed multinodular goiter. Virtually all long-standing simple goiters convert into multinodular goiters. Multinodular goiters produce the most extreme thyroid enlargements and are more frequently mistaken for neoplastic involvement than any other form of thyroid disease. Because they derive from simple goiter, they occur in both sporadic and endemic forms, having the same female-to-male distribution and presumably the same origins but affecting older individuals because they are late complications.
It is believed that multinodular goiters arise because of variations among follicular cells in their response to external stimuli, such as trophic hormones. If some cells in a follicle have a growth advantage, perhaps because of intrinsic genetic abnormalities similar to those that give rise to adenomas, those cells can give rise to clones of proliferating cells. This may result in the formation of a nodule whose continued growth is autonomous, without the external stimulus. Consistent with this model, both polyclonal and monoclonal nodules coexist within the same multinodular goiter, the latter presumably having arisen because of the acquisition of a genetic abnormality favoring growth. Not surprisingly, mutations affecting proteins of the TSH-signaling pathway that lead to constitutive activation of this pathway have been identified in a subset of autonomous thyroid nodules (TSH-signaling pathway mutations and their implications are discussed under “Adenomas”). The uneven follicular hyperplasia, generation of new follicles, and uneven accumulation of colloid produce physical stress that leads to rupture of follicles and vessels followed by hemorrhages, scarring, and sometimes calcifications. With scarring, nodularity appears, which may be accentuated by the preexisting stromal framework of the gland.
Morphology. Multinodular goiters are multilobulated, asymmetrically enlarged glands that can reach weights of more than 2000 gm. The pattern of enlargement is quite unpredictable and may involve one lobe far more than the other, producing lateral pressure on midline structures, such as the trachea and esophagus. In other instances the goiter grows behind the sternum and clavicles to produce the so-called intrathoracic or plunging goiter. Occasionally, most of it is hidden behind the trachea and esophagus; in other instances one nodule may so stand out as to impart the clinical appearance of a solitary nodule. On cut section, irregular nodules containing variable amounts of brown, gelatinous colloid are present (Fig. 24-15A). Older lesions have areas of hemorrhage, fibrosis, calcification, and cystic change. The microscopic appearance includes colloid-rich follicles lined by flattened, inactive epithelium and areas of follicular hyperplasia, accompanied by the degenerative changes noted previously. In contrast to follicular neoplasms, a prominent capsule between the hyperplastic nodules and residual compressed thyroid parenchyma is not present (Fig. 24-15B).
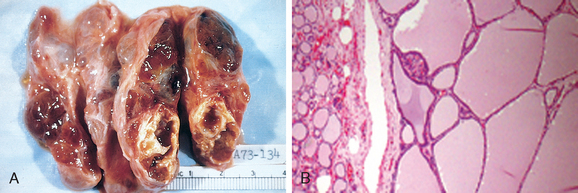
FIGURE 24-15 Multinodular goiter. A, Gross morphology demonstrating a coarsely nodular gland, containing areas of fibrosis and cystic change. B, Photomicrograph of a hyperplastic nodule, with compressed residual thyroid parenchyma on the periphery. Note absence of a prominent capsule, a distinguishing feature from follicular neoplasms.
(B, Courtesy of Dr. William Westra, Department of Pathology, Johns Hopkins University, Baltimore, MD.)
Clinical Course.
The dominant clinical features of multinodular goiter are those caused by the mass effects of the enlarged gland. In addition to the obvious cosmetic effects of a large neck mass, goiters may cause airway obstruction, dysphagia, and compression of large vessels in the neck and upper thorax (superior vena cava syndrome). Most patients are euthyroid or have subclinical hyperthyroidism (identified only by reduced TSH levels), but in a substantial minority of patients an autonomous nodule may develop within a long-standing goiter and produce hyperthyroidism (toxic multinodular goiter). This condition, known as Plummer syndrome, is not accompanied by the infiltrative ophthalmopathy and dermopathy of Graves disease. It is estimated that clinically apparent autonomous nodules can develop in approximately 10% of multinodular goiters over a 10-year follow-up. The incidence of malignancy in long-standing multinodular goiters is low (<5%) but not zero, and concern for malignancy arises in goiters that demonstrate sudden changes in size or symptoms (e.g., hoarseness). Dominant nodules in a multinodular goiter can present as a “solitary thyroid nodule” (see below), mimicking a thyroid neoplasm. A radioiodine scan demonstrates uneven iodine uptake (including the occasional “hot” autonomous nodule) consistent with the diffuse parenchymal involvement, and an admixture of hyperplastic and involuting nodules in multinodular goiter. A fine-needle aspiration biopsy is helpful and can often, albeit not always, facilitate the distinction of follicular hyperplasia from a follicular neoplasm (see below).16
Neoplasms of the Thyroid
The solitary thyroid nodule is a palpably discrete swelling within an otherwise apparently normal thyroid gland. The estimated incidence of solitary palpable nodules in the adult population of the United States varies between 1% and 10%, although it is significantly higher in endemic goitrous regions. Single nodules are about four times more common in women than in men. The incidence of thyroid nodules increases throughout life.
From a clinical standpoint, the possibility of neoplastic disease is of major concern in persons who present with thyroid nodules. Fortunately, the overwhelming majority of solitary nodules of the thyroid prove to be localized, non-neoplastic conditions (e.g., a dominant nodule in multinodular goiter, simple cysts, or foci of thyroiditis) or benign neoplasms such as follicular adenomas. In fact, benign neoplasms outnumber thyroid carcinomas by a ratio of nearly 10 : 1. While under 1% of solitary thyroid nodules are malignant, this still represents about 15,000 new cases of thyroid carcinoma per year in the United States. Fortunately, most of these cancers are indolent, permitting a 90% survival at 20 years. Several clinical criteria provide clues to the nature of a given thyroid nodule:
Such general trends and statistics, however, are of little significance in the evaluation of a given patient, in whom the timely recognition of a malignancy can be lifesaving. Ultimately, it is the morphologic evaluation of a given thyroid nodule, by fine-needle aspiration and histologic study of surgically resected thyroid parenchyma, that provides the most definitive information about its nature. In the following sections we consider the major thyroid neoplasms, including adenoma and carcinoma in its various forms.
ADENOMAS
Adenomas of the thyroid are typically discrete, solitary masses, derived from follicular epithelium, and hence they are also known as follicular adenomas. Clinically, follicular adenomas can be difficult to distinguish from dominant nodules of follicular hyperplasia or from the less common follicular carcinomas. In general, follicular adenomas are not forerunners to carcinomas; nevertheless, shared genetic alterations support the possibility that at least of subset of follicular carcinomas arise in preexisting adenomas (see below). Although the vast majority of adenomas are nonfunctional, a small proportion produces thyroid hormones and causes clinically apparent thyrotoxicosis. Hormone production in functional adenomas (“toxic adenomas”) is independent of TSH stimulation and represents another example of thyroid autonomy, analogous to toxic multinodular goiters. Not surprisingly, both of these benign processes share altered genetic pathways.
Pathogenesis.
A minority (<20%) of nonfunctioning follicular adenomas have mutations of RAS or phosphotidylinositol-3-kinase subunit (PIK3CA),17 or bear a PAX8-PPARG fusion gene,18 all of which are genetic alterations shared with follicular carcinomas. These are discussed in further detail under “Carcinomas” (see below).
Somatic mutations of the TSH receptor signaling pathway have been found in toxic adenomas, as well as in toxic multinodular goiter. Gain-of-function mutations in one of two components of this signaling system—most often TSHR itself or the α-subunit of Gs (GNAS)—allow follicular cells to secrete thyroid hormone independent of TSH stimulation (“thyroid autonomy”). This causes symptoms of hyperthyroidism and produces a “hot” thyroid nodule on imaging. Overall, mutations in the TSH receptor signaling pathway seem to be present in slightly over half of toxic thyroid nodules. Notably, TSHR and GNAS mutations are rare in follicular carcinomas; thus toxic adenomas and toxic multinodular goiter do not seem to be forerunners of malignancy.
Morphology. The typical thyroid adenoma is a solitary, spherical, encapsulated lesion that is well demarcated from the surrounding thyroid parenchyma (Fig. 24-16A). Follicular adenomas average about 3 cm in diameter, but some are much larger (≥10 cm in diameter). In freshly resected specimens the adenoma bulges from the cut surface and compresses the adjacent thyroid. The color ranges from gray-white to red-brown, depending on the cellularity of the adenoma and its colloid content. The neoplastic cells are demarcated from the adjacent parenchyma by a well-defined, intact capsule. These features are important in making the distinction from multinodular goiters, which contain multiple nodules on their cut surface even though the patient may present clinically with a solitary dominant nodule. Areas of hemorrhage, fibrosis, calcification, and cystic change, similar to those encountered in multinodular goiters, are common in follicular adenomas, particularly within larger lesions.
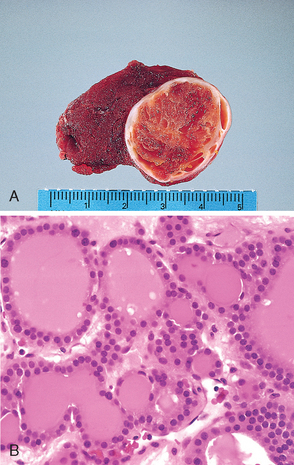
FIGURE 24-16 Follicular adenoma of the thyroid. A, A solitary, well-circumscribed nodule is seen. B, The photomicrograph shows well-differentiated follicles resembling normal thyroid parenchyma.
Microscopically, the constituent cells often form uniform-appearing follicles that contain colloid (Fig. 24-16B). The follicular growth pattern within the adenoma is usually quite distinct from the adjacent non-neoplastic thyroid. The epithelial cells composing the follicular adenoma reveal little variation in cell and nuclear morphology, and mitotic figures are rare. Extensive mitotic activity, necrosis, or high cellularity warrants careful examination of the capsule to exclude a follicular carcinoma, as well as of the nuclear features to exclude a follicular variant of papillary carcinoma (see below). Occasionally the neoplastic cells acquire brightly eosinophilic granular cytoplasm (oxyphil or Hürthle cell change) (Fig. 24-17); the clinical presentation and behavior of a follicular adenoma with oxyphilia (Hürthle cell adenoma) is no different from that of a conventional adenoma. The hallmark of all follicular adenomas is the presence of an intact, well-formed capsule encircling the tumor. Careful evaluation of the integrity of the capsule is therefore critical in distinguishing follicular adenomas from follicular carcinomas, which demonstrate capsular and/or vascular invasion (see below).
Clinical Features.
Many follicular adenomas present as a unilateral painless mass, often discovered during a routine physical examination. Larger masses may produce local symptoms, such as difficulty in swallowing. Nonfunctioning adenomas take up less radioactive iodine than does normal thyroid parenchyma. On radionuclide scanning, therefore, nonfunctioning adenomas usually appear as cold nodules relative to the adjacent thyroid tissue. As many as 10% of cold nodules eventually prove to be malignant on histologic analysis. By contrast, malignancy is rare in hot nodules (toxic adenomas). Other techniques used in the preoperative evaluation of suspected adenomas are ultrasonography and fine-needle aspiration biopsy. Because of the need for evaluating capsular integrity, the definitive diagnosis of adenomas can be made only after careful histologic examination of the resected specimen. Suspected adenomas of the thyroid are therefore removed surgically to exclude malignancy. Follicular adenomas have an excellent prognosis and do not recur or metastasize.
CARCINOMAS
Carcinomas of the thyroid are relatively uncommon in the United States, accounting for about 1.5% of all cancers. A female predominance has been noted among patients who develop thyroid carcinoma in the early and middle adult years. In contrast, cases presenting in childhood and late adult life are distributed equally among males and females. Most thyroid carcinomas (except medullary carcinomas) are derived from the thyroid follicular epithelium, and of these, the vast majority are well-differentiated lesions. The major subtypes of thyroid carcinoma and their relative frequencies include the following:
Because of the unique clinical and biologic features associated with each variant of thyroid carcinoma, these subtypes are described separately. We begin with a discussion of the molecular pathogenesis of all thyroid cancers.
Pathogenesis
Genetic Factors.
Distinct genetic events are involved in the pathogenesis of the four major histologic variants of thyroid cancer. As stated, medullary carcinomas do not arise from the follicular epithelium. Genetic alterations in the three follicular cell–derived malignancies are clustered along two oncogenic pathways—the mitogen-activated protein (MAP) kinase pathway and the phosphatidylinositol-3-kinase (PI-3K)/AKT pathway (Fig. 24-18). In normal cells, these pathways are transiently activated by binding of soluble growth factor ligands to the extracellular domain of receptor tyrosine kinases, which results in autophosphorylation of the cytoplasmic domain of the receptor, permitting intracellular signal transduction. In thyroid carcinomas, as with many solid cancers (see Chapter 7), gain-of-function mutations along components of these pathways lead to constitutive activation even in the absence of ligand and thus promote carcinogenesis.17
Papillary Carcinomas.
Activation of the MAP kinase pathway is a feature of most papillary carcinomas and can occur by one of two major mechanisms. The first mechanism involves rearrangements of RET or NTRK1 (neurotrophic tyrosine kinase receptor 1), both of which encode transmembrane receptor tyrosine kinases, and the second mechanism involves activating point mutations in BRAF, whose product is an intermediate signaling component in the MAP kinase pathway (see Fig. 24-18). The RET gene is located on chromosome 10q11, and the receptor tyrosine kinase it encodes is normally not expressed in thyroid follicular cells. In papillary cancers, either a paracentric inversion of chromosome 10 or a reciprocal translocation between chromosomes 10 and 17 places the tyrosine kinase domain of RET under the transcriptional control of genes that are constitutively expressed in the thyroid epithelium. The novel fusion proteins that are so formed are known as RET/PTC (RET/papillary thyroid carcinoma) and are present in approximately 20% to 40% of papillary thyroid cancers. There are more than 15 fusion partners of RET, and two—designated as RET/PTC1 and RET/PTC2—are most commonly observed in sporadic papillary cancers. The frequency of RET/PTC rearrangements is significantly higher in papillary cancers arising in the backdrop of radiation exposure. Irrespective of frequency, the presence of a RET/PTC fusion protein leads to constitutive expression of the tyrosine kinase in thyroid follicular cells, with resultant activation of the MAP kinase pathway. Similarly, paracentric inversions or translocations of NTRK1 on chromosome 1q21 are present in 5% to 10% of papillary thyroid cancers, and the resultant fusion proteins are constitutively expressed in thyroid cells, leading to activation of MAP kinase and other oncogenic signaling pathways. One third to one half of papillary thyroid carcinomas harbor a gain-of-function mutation in the BRAF gene, which is most commonly a valine-to-glutamate change on codon 600 (BRAFV600E).19 The presence of BRAF mutations in papillary carcinomas correlates with adverse prognostic factors like metastatic disease and extra-thyroidal extension. The histologic variants of papillary carcinoma demonstrate some unique characteristics vis-vis the frequency or nature of BRAF mutation (see below). Since chromosomal rearrangements of the RET or NTRK1 genes and mutations of BRAF have redundant effects on the thyroid epithelium (both mechanisms result in activation of the MAP kinase signaling pathway), papillary thyroid carcinomas demonstrate either one or the other molecular abnormality, but not both. RET/PTC rearrangements and BRAF point mutations are not observed in follicular adenomas or carcinomas.
Follicular Carcinomas.
Approximately one third to one half of follicular thyroid carcinomas harbor mutations in the PI-3K/AKT signaling pathway, resulting in constitutive activation of this oncogenic pathway. This subset of cases includes tumors with gain-of-function point mutations of RAS and PIK3CA, tumors with amplification of PIK3CA, and those with loss-of-function mutations of PTEN, a tumor suppressor gene and negative regulator of this pathway (see Fig. 24-18). The genetic alterations activating the PI-3K/AKT pathway are almost always mutually exclusive in follicular carcinomas, in line with their functional equivalence. The progressive increase in the prevalence of RAS and PIK3CA mutations from benign follicular adenomas to follicular carcinomas to anaplastic carcinomas (see below) suggests a shared histogenesis and molecular evolution among these follicular cell–derived tumors. A unique (2;3)(q13;p25) translocation has been described in one third to one half of follicular carcinomas. This translocation creates a fusion gene composed of portions of PAX8, a paired homeobox gene that is important in thyroid development, and the peroxisome proliferator-activated receptor gene (PPARG), whose gene product is a nuclear hormone receptor implicated in terminal differentiation of cells. Fewer than 10% of follicular adenomas harbor PAX8-PPARG fusion genes, and these have not been documented thus far in other thyroid neoplasms.
Anaplastic (Undifferentiated) Carcinomas.
These highly aggressive and lethal tumors can arise de novo, or more commonly, by “dedifferentiation” of a well-differentiated papillary or follicular carcinoma. Molecular alterations present in anaplastic carcinomas include those also seen in well-differentiated carcinomas (e.g., RAS or PIK3CA mutations), albeit at a significantly higher rate, suggesting that the presence of these mutations might predispose existing thyroid neoplasms to transform.20 Other genetic “hits,” such as inactivation of p53 or activating mutations of β-catenin, are essentially restricted to anaplastic carcinomas, and might also relate to their aggressive behavior.
Medullary Thyroid Carcinomas.
Familial medullary thyroid carcinomas occur in multiple endocrine neoplasia type 2 (MEN-2, see below) and are associated with germline RET proto-oncogene mutations that lead to constitutive activation of the receptor. RET mutations are also seen in approximately one half of nonfamilial (sporadic) medullary thyroid cancers. Chromosomal rearrangements involving RET, such as the RET/PTC translocations reported in papillary cancers, are not seen in medullary carcinomas.
Environmental Factors.
The major risk factor predisposing to thyroid cancer is exposure to ionizing radiation, particularly during the first 2 decades of life. In keeping with this, there was a marked increase in the incidence of papillary carcinomas among children exposed to ionizing radiation after the Chernobyl nuclear disaster in 1986.21 Deficiency of dietary iodine (and by extension, an association with goiter) is linked with a higher frequency of follicular carcinomas.
Papillary Carcinoma
Papillary carcinomas are the most common form of thyroid cancer, accounting for nearly 85% of primary thyroid malignancies in the United States. They occur throughout life but most often between the ages of 25 and 50, and account for the majority of thyroid carcinomas associated with previous exposure to ionizing radiation. The incidence of papillary carcinoma has increased markedly in the last 30 years, partly because of the recognition of follicular variants (see below) that were misdiagnosed in the past.
Morphology. Papillary carcinomas are solitary or multifocal lesions. Some tumors may be well circumscribed and even encapsulated; others may infiltrate the adjacent parenchyma with ill-defined margins. The lesions may contain areas of fibrosis and calcification and are often cystic. The cut surface sometimes reveals papillary foci that may point to the diagnosis. The microscopic hallmarks of papillary neoplasms include the following (Fig. 24-19):
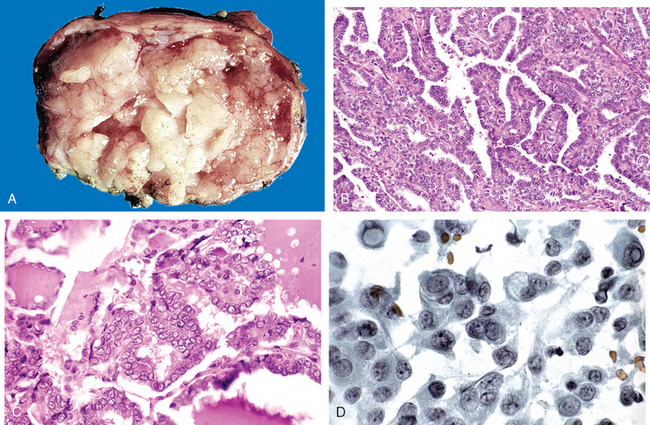
FIGURE 24-19 Papillary carcinoma of the thyroid. A, The macroscopic appearance of a papillary carcinoma with grossly discernible papillary structures. This particular example contains well-formed papillae (B), lined by cells with characteristic empty-appearing nuclei, sometimes called “Orphan Annie eye” nuclei (C). D, Cells obtained by fine-needle aspiration of a papillary carcinoma. Characteristic intranuclear inclusions are visible in some of the aspirated cells.
There are over a dozen histologic variants of papillary carcinoma that can mimic other lesions of the thyroid or harbor distinct prognostic implications22; their discussion is beyond the scope of this book. The most common variant, and the one most liable to misdiagnosis, is the follicular variant, which has the characteristic nuclei of papillary carcinoma but has an almost totally follicular architecture. The incidence of this variant has sharply increased in recent years with greater recognition of its existence among pathologists. The genetic alterations in the follicular variant demonstrate several distinctions from conventional papillary carcinomas, including a lower frequency of RET/PTC rearrangements and a significantly higher frequency of RAS mutations.23 Further, the follicular variant often harbors a distinct mutation in BRAF, which results in a lower degree of BRAF kinase activation than the mutation present in conventional papillary carcinomas.24 The follicular variant is more frequently encapsulated and has a lower incidence of lymph node metastases and extra-thyroidal extension than conventional papillary carcinomas. Recent studies suggest that encapsulated follicular variant carcinomas have a favorable prognosis and can be managed with conservative surgical excision, while the more infiltrative tumors associated with metastases should be treated more aggressively.
A tall-cell variant is marked by tall columnar cells with intensely eosinophilic cytoplasm lining the papillary structures. These tumors tend to occur in older individuals and have higher frequencies of vascular invasion, extra-thyroidal extension, and cervical and distant metastases than conventional papillary thyroid carcinoma. Tall-cell variant papillary carcinomas harbor BRAF mutations in most (55% to 100%) cases, and often have a RET/PTC translocation. The occurrence of these together may be responsible for the aggressive behavior of this variant.
An unusual diffuse sclerosing variant of papillary carcinoma occurs in younger individuals, including children. The tumor demonstrates a prominent papillary growth pattern, intermixed with solid areas containing nests of squamous metaplasia. As the name suggests, there is extensive, diffuse fibrosis throughout the thyroid gland, often associated with a prominent lymphocytic infiltrate, simulating Hashimoto thyroiditis. Lymph node metastases are present in almost all cases. The diffuse sclerosing variant carcinomas lack BRAF mutations, but RET/PTC translocations are found in approximately half the cases.
In passing, we should mention the papillary microcarcinoma, which is defined as an otherwise conventional papillary carcinoma less than 1 cm in size, and usually confined to the thyroid gland. These lesions are most often observed as an incidental finding upon surgery. These might well be precursors of the usual papillary cancers.
Clinical Course.
Most conventional papillary carcinomas present as asymptomatic thyroid nodules, but the first manifestation may be a mass in a cervical lymph node. Interestingly, the presence of isolated cervical nodal metastases does not seem to have a significant influence on the generally good prognosis of these lesions. The carcinoma, which is usually a single nodule, moves freely during swallowing and is not distinguishable from a benign nodule. Hoarseness, dysphagia, cough, or dyspnea suggests advanced disease. In a minority of patients, hematogenous metastases are present at the time of diagnosis, most commonly in the lung.
A variety of diagnostic tests have been used to help separate benign from malignant thyroid nodules, including radionuclide scanning and fine-needle aspiration. Papillary carcinomas are cold masses on scintiscans. Improvements in cytologic analysis have made fine-needle aspiration cytology a reliable test for distinguishing between benign and malignant nodules. The nuclear features are often nicely demonstrable in aspirated specimens.
Papillary thyroid cancers have an excellent prognosis, with a 10-year survival rate in excess of 95%. Between 5% and 20% of patients have local or regional recurrences, and 10% to 15% have distant metastases. The prognosis of someone with papillary thyroid cancers is dependent on several factors including age (in general, the prognosis is less favorable among patients older than 40 years), the presence of extra-thyroidal extension, and presence of distant metastases (stage).
Follicular Carcinoma
Follicular carcinomas account for 5% to 15% of primary thyroid cancers. They are more common in women (3 : 1) and present at an older age than do papillary carcinomas, with a peak incidence between 40 and 60 years of age. Follicular carcinoma is more frequent in areas with dietary iodine deficiency (25% to 40% of thyroid cancers), while its incidence has either decreased or remained stable in iodine-sufficient areas of the world.
Morphology. Follicular carcinomas are single nodules that may be well circumscribed or widely infiltrative (Fig. 24-20A). Sharply demarcated lesions may be exceedingly difficult to distinguish from follicular adenomas by gross examination. Larger lesions may penetrate the capsule and infiltrate well beyond the thyroid capsule into the adjacent neck. They are gray to tan to pink on cut section and, on occasion, are somewhat translucent due to the presence of large, colloid-filled follicles. Degenerative changes, such as central fibrosis and foci of calcification, are sometimes present.
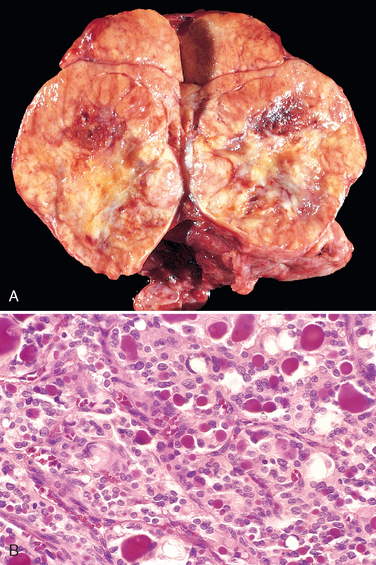
FIGURE 24-20 Follicular carcinoma. A, Cut surface of a follicular carcinoma with substantial replacement of the lobe of the thyroid. The tumor has a lighttan appearance and contains small foci of hemorrhage. B, A few of the glandular lumens contain recognizable colloid.
Microscopically, most follicular carcinomas are composed of fairly uniform cells forming small follicles containing colloid, quite reminiscent of normal thyroid (Fig. 24-20B). In other cases follicular differentiation may be less apparent, and there may be nests or sheets of cells without colloid. Occasional tumors are dominated by cells with abundant granular, eosinophilic cytoplasm (Hürthle cell or oncocytic variant of follicular carcinoma). Whatever the pattern, the nuclei lack the features typical of papillary carcinoma, and psammoma bodies are not present. It is important to note the absence of these details, because some papillary carcinomas appear almost entirely follicular. Follicular lesions in which the nuclear features are typical of papillary carcinomas should be treated as papillary cancers. While nuclear features are helpful in distinguishing papillary from follicular neoplasms, they are of little value in distinguishing follicular adenomas from minimally invasive follicular carcinomas. This distinction requires extensive histologic sampling of the tumor-capsule-thyroid interface to exclude capsular and/or vascular invasion (Fig. 24-21). The criterion for vascular invasion is applicable only to capsular vessels and vascular spaces beyond the capsule; the presence of tumor plugs within intra-tumoral blood vessels has little prognostic significance. Unlike in papillary cancers, lymphatic spread is uncommon in follicular cancers.
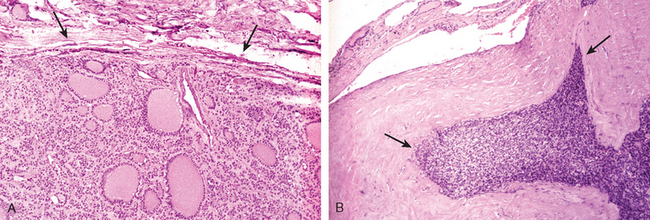
FIGURE 24-21 Capsular integrity in follicular neoplasms. In adenomas (A), a fibrous capsule, usually thin but occasionally more prominent, circumferentially surrounds the neoplastic follicles and no capsular invasion is seen (arrows); compressed normal thyroid parenchyma is usually present external to the capsule (top of the panel). In contrast, follicular carcinomas demonstrate capsular invasion (B, arrows) that may be minimal, as in this case, or widespread. The presence of vascular invasion is another feature of follicular carcinomas.
In contrast to minimally invasive follicular cancers, the diagnosis of carcinoma is obvious in widely invasive follicular carcinomas, which infiltrate the thyroid parenchyma and extra-thyroidal soft tissues. Histologically, these cancers tend to have a greater proportion of solid or trabecular growth pattern, less evidence of follicular differentiation, and increased mitotic activity.
Clinical Course.
Follicular carcinomas present as slowly enlarging painless nodules. Most frequently they are cold nodules on scintigrams, although rare, better differentiated lesions may be hyperfunctional, take up radioactive iodine and appear warm on scintiscan. Follicular carcinomas have little propensity for invading lymphatics; therefore, regional lymph nodes are rarely involved, but vascular (hematogenous) dissemination is common, with metastases to bone, lungs, liver, and elsewhere. The prognosis is largely dependent on the extent of invasion and stage at presentation. Widely invasive follicular carcinomas not infrequently present with systemic metastases, and as many as half the patients succumb to their disease within 10 years. This is in stark contrast to minimally invasive follicular carcinoma, with which the 10-year survival rate is greater than 90%. Most follicular carcinomas are treated with total thyroidectomy followed by the administration of radioactive iodine, the rationale being that metastases are likely to take up the radioactive element, which can be used to identify and ablate such lesions. In addition, because any residual follicular carcinoma may respond to TSH stimulation, patients are usually treated with thyroid hormone after surgery to suppress endogenous TSH. Serum thyroglobulin levels are used for monitoring tumor recurrence, since this product should be barely detectable in a patient who is free of disease.
Anaplastic (Undifferentiated) Carcinoma
Anaplastic carcinomas are undifferentiated tumors of the thyroid follicular epithelium, accounting for less than 5% of thyroid tumors. They are aggressive, with a mortality rate approaching 100%. Patients with anaplastic carcinoma are older than those with other types of thyroid cancer, with a mean age of 65 years. Approximately a quarter of patients with anaplastic thyroid carcinomas have a past history of a well-differentiated thyroid carcinoma, and another quarter harbor a concurrent well-differentiated tumor in the resected specimen.
Morphology. Microscopically, these neoplasms are composed of highly anaplastic cells, with variable morphology, including: (1) large, pleomorphic giant cells, including occasional osteoclast-like multinucleate giant cells; (2) spindle cells with a sarcomatous appearance; and (3) mixed spindle and giant cells. Foci of papillary or follicular differentiation may be present in some tumors, suggesting an origin from a better differentiated carcinoma. The neoplastic cells express epithelial markers like cytokeratin, but are usually negative for markers of thyroid differentiation, like thyroglobulin.
Clinical Course.
Anaplastic carcinomas usually present as a rapidly enlarging bulky neck mass. In most cases, the disease has already spread beyond the thyroid capsule into adjacent neck structures or has metastasized to the lungs at the time of presentation. Symptoms related to compression and invasion, such as dyspnea, dysphagia, hoarseness, and cough, are common. There are no effective therapies, and the disease is almost uniformly fatal. Although metastases to distant sites are common, in most cases death occurs in less than 1 year as a result of aggressive growth and compromise of vital structures in the neck.
Medullary Carcinoma
Medullary carcinomas of the thyroid are neuroendocrine neoplasms derived from the parafollicular cells, or C cells, of the thyroid, and account for approximately 5% of thyroid neoplasms. Medullary carcinomas, similar to normal C cells, secrete calcitonin, the measurement of which plays an important role in the diagnosis and postoperative follow-up of patients. In some instances the tumor cells elaborate other polypeptide hormones, such as serotonin, ACTH, and vasoactive intestinal peptide (VIP). About 70% of tumors arise sporadically. The remainder occurs in the setting of MEN syndrome 2A or 2B or as familial tumors without an associated MEN syndrome (familial medullary thyroid carcinoma, or FMTC; see “Multiple Endocrine Neoplasia Syndromes”). Recall that activating point mutations in the RET protooncogene play an important role in the development of both familial and sporadic medullary carcinomas. Cases associated with MEN types 2A or 2B occur in younger patients, and may even arise during the first decade of life. In contrast, sporadic as well as familial medullary carcinomas are lesions of adulthood, with a peak incidence in the 40s and 50s.
Morphology. Sporadic medullary thyroid carcinomas present as a solitary nodule (Fig. 24-22A). In contrast, bilaterality and multicentricity are common in familial cases. Larger lesions often contain areas of necrosis and hemorrhage and may extend through the capsule of the thyroid. The tumor tissue is firm, pale gray to tan, and infiltrative. There may be foci of hemorrhage and necrosis in the larger lesions.
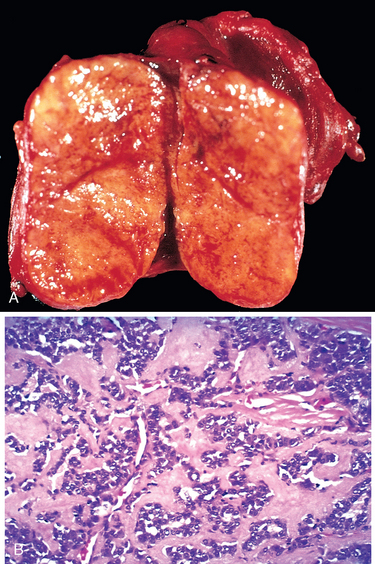
FIGURE 24-22 Medullary carcinoma of thyroid. A, These tumors typically show a solid pattern of growth and do not have connective tissue capsules. B, Histology demonstrates abundant deposition of amyloid, visible here as homogeneous extracellular material, derived from calcitonin molecules secreted by the neoplastic cells.
(A, Courtesy of Dr. Joseph Corson, Brigham and Women’s Hospital, Boston, MA.)
Microscopically, medullary carcinomas are composed of polygonal to spindle-shaped cells, which may form nests, trabeculae, and even follicles. Small, more anaplastic cells are present in some tumors and may be the predominant cell type. Acellular amyloid deposits, derived from altered calcitonin polypeptides, are present in the adjacent stroma in many cases (Fig. 24-22B). Calcitonin is readily demonstrable within the cytoplasm of the tumor cells as well as in the stromal amyloid by immunohistochemical methods. As with all neuroendocrine tumors, electron microscopy reveals variable numbers of membrane-bound electron-dense granules within the cytoplasm of the neoplastic cells (Fig. 24-23). One of the peculiar features of familial medullary cancers is the presence of multicentric C-cell hyperplasia in the surrounding thyroid parenchyma, a feature that is usually absent in sporadic lesions. While the precise criteria for defining C-cell hyperplasia are not established, the presence of multiple prominent clusters of C cells scattered throughout the parenchyma should raise the specter of a familial tumor, even if that history is not explicitly present. Foci of C-cell hyperplasia are believed to represent the precursor lesions from which medullary carcinomas arise.
Clinical Course.
Sporadic cases of medullary carcinoma come to medical attention most often as a mass in the neck, sometimes associated with local effects such as dysphagia or hoarseness. In some instances, the initial manifestations are those of a paraneoplastic syndrome, caused by the secretion of a peptide hormone (e.g., diarrhea due to the secretion of VIP, or Cushing syndrome due to ACTH). Notably, hypocalcemia is not a prominent feature, despite the presence of raised calcitonin levels. In addition to circulating calcitonin, secretion of carcinoembryonic antigen by the neoplastic cells is a useful biomarker, especially for presurgical assessment of tumor load and in calcitonin-negative tumors. Patients with familial syndromes may come to attention because of symptoms localized to the thyroid or as a result of endocrine neoplasms in other organs (e.g., adrenal or parathyroid glands). Medullary carcinomas arising in the context of MEN-2B are generally more aggressive and metastasize more frequently than those occurring in patients with sporadic tumors, MEN-2A, or FMTC. As will be discussed later, all asymptomatic MEN-2 kindred carrying germline RET mutations are offered prophylactic thyroidectomy as early as possible to protect against the inevitable development of medullary carcinomas, the major risk factor for poor outcome in these families. Sometimes the only histologic finding in the resected thyroid of asymptomatic carriers is the presence of C-cell hyperplasia or small (<1 cm) “micromedullary” carcinomas. Several promising small-molecule inhibitors of RET tyrosine kinase have recently been developed, and are being tested in individuals with medullary carcinomas.25
Congenital Anomalies
Thyroglossal duct or cyst is the most common clinically significant congenital anomaly of the thyroid. A persistent sinus tract may remain as a vestigial remnant of the tubular development of the thyroid gland. Parts of this tube may be obliterated, leaving small segments to form cysts. These occur at any age and might not become evident until adult life. Mucinous, clear secretions may collect within these cysts to form either spherical masses or fusiform swellings, rarely over 2 to 3 cm in diameter. These are present in the midline of the neck anterior to the trachea. Segments of the duct and cysts that occur high in the neck are lined by stratified squamous epithelium, which is essentially identical with that covering the posterior portion of the tongue in the region of the foramen cecum. The anomalies that occur in the lower neck more proximal to the thyroid gland are lined by epithelium resembling the thyroidal acinar epithelium. Characteristically, subjacent to the lining epithelium, there is an intense lymphocytic infiltrate. Superimposed infection may convert these lesions into abscess cavities, and rarely, they give rise to cancers.
PARATHYROID GLANDS
The four parathyroid glands are composed mainly of chief cells. The chief cells vary from light to dark pink with hematoxylin and eosin stains, depending on their glycogen content. They are polygonal, 12 to 20 μm in diameter, and have central, round, uniform nuclei. In addition, they have secretory granules containing parathyroid hormone (PTH). Sometimes these cells have a water-clear appearance due to lakes of glycogen. Oxyphil cells and transitional oxyphils are found throughout the normal parathyroid, either singly or in small clusters. They are slightly larger than the chief cells, have acidophilic cytoplasm, and are tightly packed with mitochondria. Glycogen granules are also present in these cells, but secretory granules are sparse or absent. In early infancy and childhood, the parathyroid glands are composed almost entirely of solid sheets of chief cells. The amount of stromal fat increases up to age 25, reaching a maximum of approximately 30% of the gland, and then plateaus.
The activity of the parathyroid glands is controlled by the level of free (ionized) calcium in the bloodstream rather than by trophic hormones secreted by the hypothalamus and pituitary. Normally, decreased levels of free calcium stimulate the synthesis and secretion of PTH. The metabolic functions of PTH in regulating serum calcium levels can be summarized as follows:
The net result of these activities is an increase in the level of free calcium, which, in turn, inhibits further PTH secretion in a classic feedback loop.
Hypercalcemia is one of a number of changes induced by elevated levels of PTH. As was discussed in Chapter 7, hypercalcemia is also a relatively common complication of malignancy, occurring both with solid tumors, such as lung, breast, head and neck, and renal cancers, and with hematologic malignancies, notably multiple myeloma. In fact, malignancy is the most common cause of clinically apparent hypercalcemia, while primary hyperparathyroidism (see below) is a more common cause of asymptomatic elevated blood calcium (incidental hypercalcemia). The prognosis of individuals with malignancy-associated hypercalcemia is generally poor, because it more frequently occurs with advanced cancers. Hypercalcemia of malignancy is due to increased bone resorption and subsequent release of calcium. Bone resorption can occur in tumors that have not metastasized to the bone, as well as in tumors with osteolytic metastases. This is because many solid cancers secrete PTH-related protein (PTHrP) (Chapter 7), which promotes the expression of receptor activator of nuclear factor κB ligand (RANKL) on osteoblasts. RANKL is an osteoclast differentiation factor, which binds to its receptor, RANK, on the surface of osteoclast progenitor cells, and promotes their differentiation into mature osteoclasts, capable of bone resorption. Calcium homeostasis in bones is maintained by the secretion of osteoprotegerin, a decoy receptor of RANKL, capable of “siphoning” off excess ligand, and thereby preventing unbalanced bone resorption (Chapter 26). PTHrP inhibits osteoprotegerin secretion by osteoblastic cells, thereby altering the RANKL/osteoprotegerin ratio in a direction favoring osteoclastogenesis.26 Thus, administration of soluble osteoprotegerin and monoclonal antibodies against RANKL have both emerged as promising therapeutic strategies in cancer patients with hypercalcemia of malignancy.
Similar to the other endocrine organs, abnormalities of the parathyroid glands include both hyperfunction and hypofunction. Tumors of the parathyroid glands, in contrast to thyroid tumors, usually come to attention because of excessive secretion of PTH rather than mass effects.
Hyperparathyroidism
Hyperparathyroidism occurs in two major forms—primary and secondary—and, less commonly, tertiary. The first condition represents an autonomous, spontaneous overproduction of PTH; the latter two conditions typically occur as secondary phenomena in individuals with chronic renal insufficiency.
PRIMARY HYPERPARATHYROIDISM
Primary hyperparathyroidism is one of the most common endocrine disorders, and it is an important cause of hypercalcemia. The frequency of the various parathyroid lesions underlying the hyperfunction is as follows:
Primary hyperparathyroidism is usually a disease of adults and is more common in women than in men by a ratio of nearly 4 : 1. The annual incidence is now estimated to be about 25 cases per 100,000 in the United States and Europe; as many as 80% of patients with this condition are identified in the outpatient setting, when hypercalcemia is discovered incidentally on a serum electrolyte panel. Most cases occur in the 50s or later in life.
The most common cause of primary hyperparathyroidism is a solitary parathyroid adenoma arising in the sporadic (nonfamilial) setting (Fig. 24-24). Although familial syndromes are a distant second, they have provided a unique insight into the pathogenesis of primary hyperparathyroidism. The genetic syndromes associated with familial primary hyperparathyroidism include the following:
FIGURE 24-24 Parathyroid adenomas are almost always solitary lesions. Technetium-99m-sestamibi radionuclide scan demonstrates an area of increased uptake corresponding to the left inferior parathyroid gland (arrow). This person had a parathyroid adenoma. Preoperative scintigraphy is useful in localizing and distinguishing adenomas from parathyroid hyperplasia, where more than one gland would demonstrate increased uptake.
Most, if not all, sporadic parathyroid adenomas are monoclonal, suggesting that they are true neoplasms. Sporadic parathyroid hyperplasia is also monoclonal in many instances, particularly when associated with a persistent stimulus for parathyroid growth (refractory secondary or tertiary parathyroidism; see below). Among the sporadic adenomas there are two molecular defects that have an established role in pathogenesis:
Morphology. The morphologic changes seen in primary hyperparathyroidism include those in the parathyroid glands as well as those in other organs affected by elevated levels of PTH and calcium. Parathyroid adenomas are almost always solitary and, similar to the normal parathyroid glands, may lie in close proximity to the thyroid gland or in an ectopic site (e.g., the mediastinum). The typical parathyroid adenoma averages 0.5 to 5.0 gm; is a well-circumscribed, soft, tan to reddish-brown nodule; and is invested by a delicate capsule. In contrast to primary hyperplasia, the glands outside the adenoma are usually normal in size or somewhat shrunken because of feedback inhibition by elevated levels of serum calcium. Microscopically, parathyroid adenomas are mostly composed of fairly uniform, polygonal chief cells with small, centrally placed nuclei (Fig. 24-25). At least a few nests of larger oxyphil cells are present as well; uncommonly, entire adenomas may be composed of this cell type (oxyphil adenomas). These may resemble Hürthle cell tumors in the thyroid. A rim of compressed, non-neoplastic parathyroid tissue, generally separated by a fibrous capsule, is often visible at the edge of the adenoma. Mitotic figures are rare, but it is not uncommon to find bizarre and pleomorphic nuclei even within adenomas (so-called endocrine atypia); this is not a criterion for malignancy. In contrast to the normal parathyroid parenchyma, adipose tissue is inconspicuous.
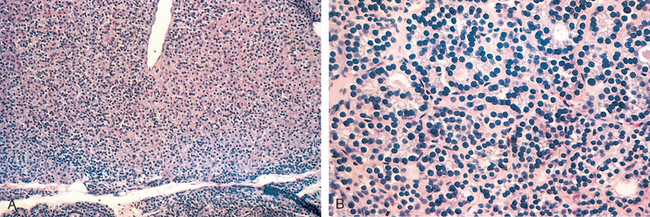
FIGURE 24-25 Parathyroid adenoma. A, Solitary chief cell parathyroid adenoma (low-power photomicrograph) revealing clear delineation from the residual gland below. B, High-power detail of a chief cell parathyroid adenoma. There is some slight variation in nuclear size but no anaplasia and some slight tendency to follicular formation.
Primary hyperplasia may occur sporadically or as a component of MEN syndrome. Although classically all four glands are involved, there is frequently asymmetry with apparent sparing of one or two glands, making the distinction between hyperplasia and adenoma difficult. The combined weight of all glands rarely exceeds 1.0 gm and is often less. Microscopically, the most common pattern seen is that of chief cell hyperplasia, which may involve the glands in a diffuse or multinodular pattern. Less commonly, the constituent cells contain abundant water-clear cells (“water-clear cell hyperplasia”). In many instances there are islands of oxyphils, and poorly developed, delicate fibrous strands may envelop the nodules. As in the case of adenomas, stromal fat is inconspicuous within the foci of hyperplasia.
Parathyroid carcinomas may be circumscribed lesions that are difficult to distinguish from adenomas, or they may be clearly invasive neoplasms. These tumors enlarge one parathyroid gland and consist of gray-white, irregular masses that sometimes exceed 10 gm in weight. The cells are usually uniform and resemble normal parathyroid cells. They are arrayed in nodular or trabecular patterns with a dense, fibrous capsule enclosing the mass. There is general agreement that a diagnosis of carcinoma based on cytologic detail is unreliable, and invasion of surrounding tissues and metastasis are the only reliable criteria. Local recurrence occurs in one third of cases, and more distant dissemination occurs in another third.
Morphologic changes in other organs deserving special mention include skeletal and renal lesions. Skeletal changes include increased numbers of osteoclasts, which erode bone matrix and mobilize calcium salts, particularly in the metaphyses of long tubular bones. Bone resorption is accompanied by increased osteoblastic activity and the formation of new bony trabeculae. In many cases the resultant bone contains widely spaced, delicate trabeculae reminiscent of those seen in osteoporosis. In more severe cases the cortex is grossly thinned, and the marrow contains increased amounts of fibrous tissue accompanied by foci of hemorrhage and cyst formation (osteitis fibrosa cystica) (Chapter 26). Aggregates of osteoclasts, reactive giant cells, and hemorrhagic debris occasionally form masses that may be mistaken for neoplasms (brown tumors of hyperparathyroidism). PTH-induced hypercalcemia favors formation of urinary tract stones (nephrolithiasis) as well as calcification of the renal interstitium and tubules (nephrocalcinosis). Metastatic calcification secondary to hypercalcemia may also be seen in other sites, including the stomach, lungs, myocardium, and blood vessels.
Clinical Course.
Primary hyperparathyroidism may be: (1) asymptomatic and identified after a routine chemistry profile, or (2) associated with the classic clinical manifestations of primary hyperparathyroidism.
Asymptomatic Hyperparathyroidism.
Because serum calcium levels are routinely assessed in the work-up of most patients who need blood tests, clinically silent hyperparathyroidism is often detected early. Hence, many of the classic manifestations, particularly those referable to bone and renal disease, are now seen infrequently in clinical practice. Primary hyperparathyroidism is the most common cause of asymptomatic hypercalcemia and indeed the most common manifestation of primary hyperparathyroidism is hypercalcemia. Among many other less common causes of hypercalcemia (Table 24-5), malignancy stands out as the most frequent cause of clinically apparent hypercalcemia in adults and must be excluded by appropriate clinical and laboratory investigations in patients with suspected hyperparathyroidism. In individuals with primary hyperparathyroidism, serum PTH levels are inappropriately elevated for the level of serum calcium, whereas PTH levels are low to undetectable in hypercalcemia caused by of nonparathyroid diseases (see Table 24-5). In persons with hypercalcemia caused by secretion of PTHrP by certain nonparathyroid tumors, radioimmunoassays specific for PTH and PTHrP can distinguish between the two molecules. Other laboratory alterations referable to PTH excess include hypophosphatemia and increased urinary excretion of both calcium and phosphate. Secondary renal disease may lead to phosphate retention with normalization of serum phosphates.
TABLE 24-5 Causes of Hypercalcemia
| Raised [PTH] | Decreased [PTH] |
|---|---|
| Hyperparathyroidism | Hypercalcemia of malignancy* |
| Primary (adenoma > hyperplasia)* | Vitamin D toxicity |
| Secondary† | Immobilization |
| Tertiary† | Thiazide diuretics |
| Familial hypocalciuric hypercalcemia | Granulomatous disease (sarcoidosis) |
[PTH], parathyroid hormone concentration.
* Primary hyperparathyroidism is the most common cause of hyper-calcemia overall. Malignancy is the most common cause of symptomatic hypercalcemia. Primary hyperparathyroidism and malignancy account for nearly 90% of cases of hypercalcemia.
† Secondary and teriary hyperparathyroidism are most commonly associated with progressive renal failure.
Symptomatic Primary Hyperparathyroidism.
The signs and symptoms of hyperparathyroidism reflect the combined effects of increased PTH secretion and hypercalcemia. Primary hyperparathyroidism is associated with “painful bones, renal stones, abdominal groans, and psychic moans.” The constellation of symptoms includes:
The abnormalities most directly related to hyperparathyroidism are nephrolithiasis and bone disease, whereas those attributable to hypercalcemia include fatigue, weakness, and constipation. The pathogenesis of many of the other manifestations of the disorder remains poorly understood.
SECONDARY HYPERPARATHYROIDISM
Secondary hyperparathyroidism is caused by any condition that gives rise to chronic hypocalcemia, which in turn leads to compensatory overactivity of the parathyroid glands. Renal failure is by far the most common cause of secondary hyperparathyroidism, although several other diseases, including inadequate dietary intake of calcium, steatorrhea, and vitamin D deficiency, may also cause this disorder. The mechanisms by which chronic renal failure induces secondary hyperparathyroidism are complex and not fully understood. Chronic renal insufficiency is associated with decreased phosphate excretion, which in turn results in hyperphosphatemia. The elevated serum phosphate levels directly depress serum calcium levels and thereby stimulate parathyroid gland activity. In addition, loss of renal substance reduces the availability of α-1-hydroxylase necessary for the synthesis of the active form of vitamin D, which in turn reduces intestinal absorption of calcium (Chapter 9). Because vitamin D has suppressive effects on parathyroid growth and PTH secretion, its relative deficiency compounds the hyperparathyroidism in renal failure.
Morphology. The parathyroid glands in secondary hyperparathyroidism are hyperplastic. As in primary hyperparathyroidism, the degree of glandular enlargement is not necessarily symmetric. Microscopically, the hyperplastic glands contain an increased number of chief cells, or cells with more abundant, clear cytoplasm (so-called water-clear cells) in a diffuse or multinodular distribution. Fat cells are decreased in number. Bone changes similar to those seen in primary hyperparathyroidism may also be present. Metastatic calcification may be seen in many tissues, including lungs, heart, stomach, and blood vessels.
Clinical Course.
The clinical features of secondary hyperparathyroidism are usually dominated by symptoms of chronic renal failure. Bone abnormalities (renal osteodystrophy) and other changes associated with PTH excess are, in general, less severe than are those seen in primary hyperparathyroidism. The vascular calcification associated with secondary hyperparathyroidism may occasionally result in significant ischemic damage to skin and other organs, a process sometimes referred to as calciphylaxis. Patients with secondary hyperparathyroidism often respond to dietary vitamin D supplementation, as well as phosphate binders to decrease the prevailing hyperphosphatemia.
In a minority of patients, parathyroid activity may become autonomous and excessive, with resultant hypercalcemia, a process that is sometimes termed tertiary hyperparathyroidism. Parathyroidectomy may be necessary to control the hyperparathyroidism in such patients.