Collecting and Managing Data
Data collection is one of the most exciting parts of research. After all the planning, writing, and negotiating, you are ready for the real part of research—the action part. There is a sense of euphoria and excitement, an eagerness to start the study. However, before you leap into data collection, spend some time carefully planning this adventure. It may save you difficulties later on as you implement the final steps of the research process. Consider problems you might encounter while collecting data, and develop strategies for addressing them. You must make careful plans for managing data as you collect it. This chapter is divided into three sections to assist you in planning data collection, collecting data, and managing data for quantitative studies. Data collection strategies for qualitative studies are described in Chapter 23.
PLANNING DATA COLLECTION
A data collection plan details how you will implement your study. The plan for collecting data is specific to the study being conducted and requires that you consider some of the more commonplace elements of research. You will need to map out the procedures you will use to collect data, anticipate the time and cost of data collection, develop data collection forms that ease data entry, and prepare a codebook that will help you to identify data to be entered in a database. This extensive planning increases the accuracy of the data collected and the validity of the study findings. The strength of the findings from several studies increases the quality of the research evidence that is available for use in practice (Craig & Smith, 2007; Melnyk & Fineout-Overholt, 2005).
Planning Data Collection Procedures
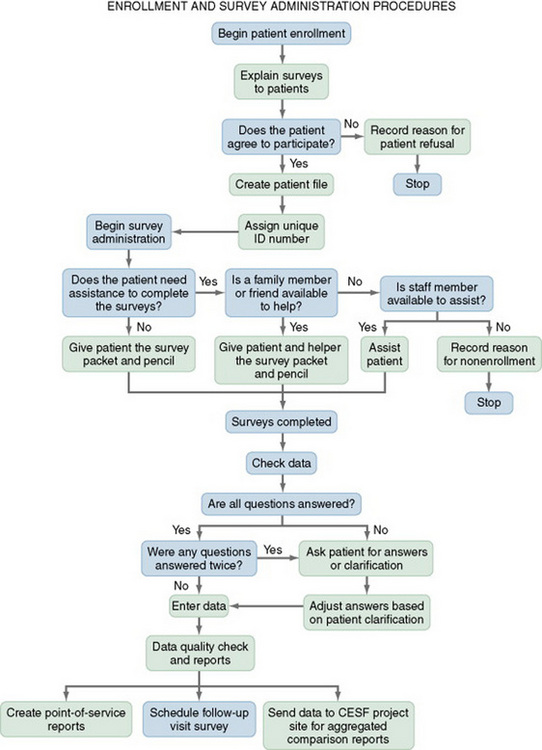
To plan the process of data collection, you must determine step by step how and in what sequence data will be collected from a single subject. The timing of this process also must be established. For example, how much time will be required to identify potential subjects, explain the study, and obtain consent? How much time is needed for activities such as completing questionnaires or obtaining physiological measures? Next, envision the overall activities that will be occurring during data collection. At what point are subjects assigned to groups? When and how will you implement the study treatment? Will data be collected from more than one subject at a time, or is it necessary to focus attention on one subject at a time? How many subjects per day can be accessed for data given the study design and the setting? It might be helpful to conduct a trial run or even a pilot study by collecting data from three to five subjects to determine the strengths and weaknesses of the data collection plan. You will need a minimum of five subjects if you plan to conduct a pilot study. You might develop a data collection tree or flow diagram to illustrate the process for collecting data in your study. An example is shown in Figure 17-1.
Decision Points
Decision points that occur during data collection must be identified and all options considered. Decisions might include whether potential subjects meet the sampling criteria, whether a subject understands the information needed to give informed consent, what group the subject will be assigned to, whether the subject comprehends instructions related to providing data, and whether the subject has provided all the data needed. Your data collection tree should indicate each point at which a decision is made.
Consistency
Consistency in data collection across subjects is critical. If more than one person is collecting the data, consistency among data collectors (interrater reliability) is also necessary (see Chapter 15). Identify situations in your study that might interfere with consistency, and develop a plan that will maximize consistency. The specific days and hours of data collection may influence the consistency of the data collected and thus must be carefully considered. For example, the energy level and state of mind of subjects from whom data are gathered in the morning may differ from that of subjects from whom data are gathered in the evening. Visitors are more likely to be present at certain times of day and may interfere with data collection or influence subject responses. Patient care routines vary with the time of day. In some studies, the care recently received or the care currently being provided may alter the data you gather. The subjects you approach on Saturday to participate in the study may differ from the subjects you approach on weekday mornings. Subjects seeking care on Saturday may have a full-time job, whereas those seeking care on weekday mornings may be either unemployed or too ill to work.
You and your research team also must decide who will collect the data. Will the researcher collect all the data, or will data collectors be employed for this purpose? Can data collectors be nurses working in the area? Researchers have experienced difficulties in studies in which they expected nurses providing patient care to also be data collectors. Patient care takes priority over data collection, which may lead to missing data or missed subjects.
If you decide to use data collectors, they must be informed about the research project, familiarized with the instruments to be used, and provided equivalent training in the data collection process. In addition to training, data collectors need written guidelines or protocols that indicate which instruments to use, the order in which to introduce the instruments, how to administer the instruments, and a time frame for the data collection process (Gift, Creasia, & Parker, 1991).
After training, data collectors must be evaluated to determine their consistency in the data collection process. Washington and Moss (1988) suggested that a minimum of 10 subjects must be rated with the complete instrument before interrater reliability can be adequately assessed. The data collectors’ interrater reliability is usually assessed intermittently throughout data collection to ensure consistency. Data collectors also must be encouraged to identify and record any problems or variations in the environment that affect the data collection process.
Determine how you will reliably and competently deliver the study treatment. Often researchers develop detailed protocols to guide them in delivering the treatment or intervention, and they train data collectors in this process. Stein, Sargent, and Rafaels (2007) stressed the importance of achieving intervention fidelity in a study, which involves training an individual called an interventionalist to deliver the intervention protocol. To achieve intervention fidelity, the delivery of the intervention must include the core components of adherence and competence. Adherence is the most basic and exists when the interventionalist reliably or consistently implements the behaviors of the intervention protocol. Competence is more complex and focuses on the interventionalist’s skill and expertise in delivering the study intervention. For more details on intervention protocol development and implementation, refer to Chapters 11 and 13.
Time Factors
Researchers often underestimate the time required to collect data for a study, which sometimes takes two to three times longer than anticipated. It is helpful to write out a time plan for the data collection period. Conduct a pilot study to refine the data collection process, and determine the time required to collect data from a subject.
Events during the data collection period sometimes are not under the researcher’s control. For example, a sudden heavy staff workload may make data collection temporarily difficult or impossible, or the number of potential subjects might be reduced for a period. In some situations, researchers must obtain permission from each subject’s physician before they are permitted to collect data on that subject. Activities required to meet this stipulation—such as contacting physicians, explaining the study, and obtaining permission—require extensive time. In some cases, potential subjects are lost before the researcher can obtain the mandatory permission, thus extending the time required to obtain the necessary number of subjects.
Cost Factors
Cost is another consideration when planning a study. Measurement tools—such as Holter monitors, spirometers, infrared thermometers, pulse oximeters, or Glucometers—used in physiological studies may need to be rented, purchased, or obtained from the company manufacturing the equipment. You may need to pay a fee for questionnaires or scales and for analyzing the data. Data collection forms must be typed and duplicated. In some cases, printing costs for materials that are to be distributed during data collection must be factored in, such as teaching materials, questionnaires, or scales. In some studies, postage is an additional expense. There may be costs involved in coding the data for entry into the computer and for conducting data analyses. Consultation with a statistician early in the development of a research project and during data analysis must also be budgeted. You may need to hire a secretary to type the final report, research presentations, or a manuscript for publication.
In addition to these direct costs, there are also indirect costs. The researcher’s time is a cost and costs for travel to and from the study site and for meals eaten out while working on the study must be taken into account. You also must estimate the expense of presenting the research project at conferences, and include that cost in the budget. To prevent unexpected expenses from delaying the study, examine all costs in an organized manner during the planning phase of the study. A budget is best developed early in the planning process and revised as plans are modified (see Chapter 28 for a sample budget). Seeking funding for at least part of the study costs can facilitate the conduct of a study (see Chapter 29 on funding for research).
Neophyte researchers have difficulty making reasonable estimates of time and costs related to a study. We advise validating the time and cost estimates with an experienced researcher. If the cost and time factors are prohibitive, simplify your study so that fewer variables are measured, fewer instruments are used. Make the design less complex, and use fewer data collectors. These are serious modifications, however, so you and your team should thoroughly examine the consequences before making such revisions. If time or cost estimates go beyond expectations, you can revise the time schedules and budget with a new projection for completing the study.
Developing Data Collection Forms
Before data collection begins, you may need to develop or modify forms on which to record data. These forms can be used to record demographic data, information from the patient record, observations, or values from physiological measures. The demographic variables commonly collected in nursing studies include age, gender, race, education, income or socioeconomic status, employment status, diagnosis, and marital status. You also might need to collect other data that may be either extraneous or confounding variables such as the subject’s physician, stage of illness, length of illness or hospitalization, complications, date of data collection, time of day and day of week of data collection, and any untoward events that occur during the data collection period. In some cases, the length of time required of individual subjects for data collection may be a confounding variable and must be recorded. If it is necessary to contact the subject at a later time, you will need to obtain the subject’s address and telephone number, but only with that person’s awareness and permission. Names and phone numbers of family members may also be useful if subjects are likely to move or be difficult to contact. Consider the importance of each piece of datum and the amount of the subject’s time required to collect it. If the data can be obtained from patient records or any other written sources, you do not need to ask the subject to provide this information; just make sure that the institutional review board (IRB) has authorized your team to collect these data in the study setting. You also need to protect the participant’s private health information that is regulated by the Health Insurance Portability and Accountability Act (HIPAA) (available online at www.hhs.gov/ocr/hipaa).
Data collection forms must be designed so that the data are easily recorded and entered into the computer. Decide whether data will be collected in raw form or coded at the time of collection. Coding is the process of transforming data into numerical symbols that can be entered easily into the computer. For example, variables such as gender, ethnicity, and diagnoses can be categorized and given numerical labels. For gender, the male category could be identified by a 1 and the female category by a 2. For the ethnicity variable, the African-American category could be represented by the number 1, Caucasian by a 2, Hispanic by a 3, and Other by a 4.
The coding categories developed for a study must be not only mutually exclusive but also exhaustive, which means that the value for a specific variable fits into only one category, and each observation must fit into a category. For example, the income ranges would not be mutually exclusive or exhaustive if they were categorized in the following way on a demographic questionnaire:
Income Range (Please check the range that most accurately reflects your income.)
These categories are not exclusive because they overlap and a subject with a $35,000 income could mark category 1, or category 2, or both. The categories are not exhaustive because a subject may have an income of either $25,000 or $39,500, yet the questionnaire does not contain categories that include each of these incomes. For many items, a code for “Other” should be included for unexpected classifications of variables such as marital status, ethnicity, or diagnosis. The following income ranges are both exclusive and exhaustive and would be appropriate for collecting demographic data from subjects:
Income Range (Please check the range that most accurately reflects your income.)
Data collection forms offer a number of response styles. The person completing the form (subject or data collector) might be asked to check a blank space before or after the words male or female, to circle the word male or female, or to write a 1 or a 2 in a blank space before or after the word selected. If codes are used, the meaning of the codes should be indicated on the collection forms so that the individual completing the form will understand them.
Placement of the data on the forms is important, because careful placement makes it easier for users to complete the form and to locate responses for computer entry. Placement of blanks on the left side of the page seems to be most efficient for data entry, but this layout may prove problematic when subjects are completing the forms. The least effective arrangement is when the data are positioned irregularly on the form, because the risk of data being missed during data entry is high. Subjects’ names should not be on the data collection forms; only the subject’s identification number should appear. The researcher will usually keep a master list of subjects and associated coding numbers, which is stored in a separate location to ensure the subjects’ privacy. Often this master list of subjects and codes is kept with the subject consent forms.
Figure 17-2 provides a sample data collection form. It includes four items that could be problematic in terms of coding, data analysis, or both. The blank used to enter Surgical Procedure Performed would lead to problems when it is time to enter the data into a computerized data set. Because multiple surgical procedures could have been performed, developing codes for the various surgical procedures would be difficult and time consuming. In addition, different words might be used to record the same surgical procedure. It may be necessary to tally the surgical procedures manually. Unless this degree of specification of procedures is important to the study, an alternative would be to develop larger categories of procedures before data collection and place the categories on the data collection form. A category of “Other” might be useful for less commonly performed surgical procedures. This method would require the data collector to make a judgment regarding which category was appropriate for a particular surgical procedure. Another option would be to write in the category code number for a particular surgical procedure after the data collection form is completed but before data entry. Similar problems occur with the items Narcotics Ordered after Surgery and Narcotic Administration. Unless these data are to be used in statistical analyses, it might be better to manually categorize this information for descriptive purposes. If these items are needed for planned statistical procedures, use care to develop appropriate codes. In this study, the researcher might be interested in determining differences in the amount of narcotics administered in a given period in relation to weight and height. Recording the treatment groups on the data collection form may be problematic because the information could influence the data recorded by the data collectors.
Using Electronic Devices for Data Collection
Electronic devices can be used to collect a variety of scale, questionnaire, or physiological data. However, the use of these devices for research may require considerable preparation. You may need to purchase, rent, or borrow the equipment. You also may need to make arrangements with the data collection site or to place measurement scales on special forms.
Scantron Sheets
Scantron sheets are forms that allow subjects to respond to test questions or scale items by using a pencil to bubble in responses. These responses can be entered directly into the computer by optic scanner (Dennis, 1994) and stored into a database for analysis. This practice speeds up the process of entering data and reduces errors related to data entry. However, subjects not familiar with Scantron sheets may be reluctant to use them, and some inaccuracies in data may occur because of subject error. These forms have been used commonly for administering multiple-choice tests to nursing students. Scantrons are best used when subjects cannot be accessed by e-mail or there is no computer for direct data entry and data must be collected using paper-and-pencil forms.
Teleform
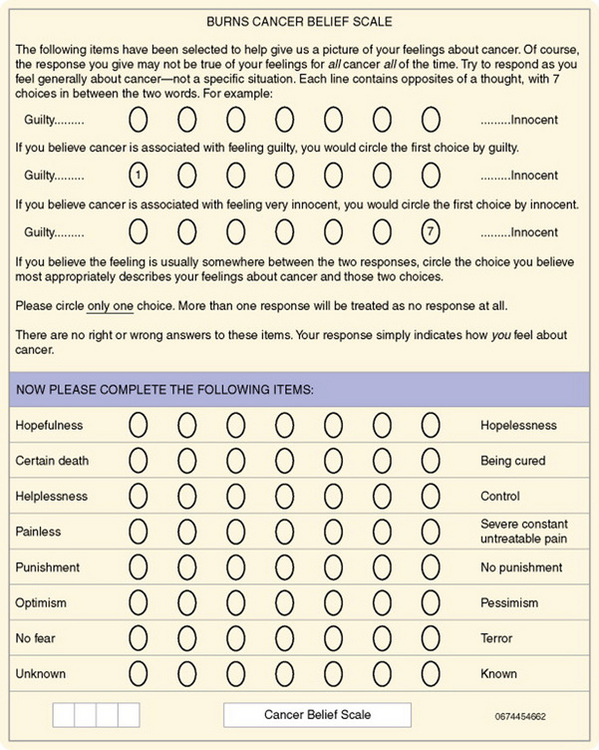
Teleform is a computer software package developed by Cardiff (see www.cardiff.com) that enables researchers to design a form specific to a scale or questionnaire to be used for data collection. Cardiff software has unique features that allow users to develop point-and-click automated forms that can be distributed electronically. Additional features include data accuracy verification, selective data extraction and analysis, digital record signature support, auditing and tracking, print merge applications, and flexible export interfaces (www.cardiff.com/products/teleform/index.html). Training is available at the Cardiff website or in person at locations around the United States. Figure 17-3 shows a Teleform version of the Burns Cancer Belief Scale, which allows data to be scanned and stored in a database. Or this form could be developed and sent to subjects electronically, and the data are collected online and automatically entered into a database. Universities and schools of nursing are purchasing this software, as are some researchers who can purchase it with grant monies. The costs of acquiring the hardware and software are considerably less than the costs of entering data manually.
Im et al. (2007) conducted a multiethnic survey in the United States of the gender and ethnic differences in the cancer pain experience. These researchers administered their questionnaire over the Internet and through a paper-and-pencil format based on the desires of the subjects. The following excerpt describes the data collection procedure for this study.
Im et al. (2007) maximized their sample size and obtained a more representative sample by giving participants an option to complete their questionnaire on the Internet or via the pen-and-pencil format. The researchers then took steps to ensure that the data collected by the two formats were comparable by testing for significant differences and finding none. The time to complete the Internet and pen-and-pencil questionnaires did not vary. Im et al. (2007) also ensured that the rights of the subjects were protected and an ethical study was conducted.
Computerized Data Collection
With the advent of microcomputers, data collectors can code data directly into a microcomputer at the data collection site. If a computer is used for data collection, a program must be written for entering, cleaning, and storing data. A microcomputer enables users to collect large amounts of data with few errors, which can be readily analyzed with a variety of statistical software packages.
Personal Digital Assistants
Personal digital assistants (PDAs) are small hand-held computers that allow the researcher to enter data directly into the computer from observations as they occur or to download data from a larger computer for easy access. Bernhardt et al. (2001) used PDAs to collect survey data and found that participants preferred the PDA to paper-and-pencil surveys. Health care providers are loading programs on their PDAs that facilitate accurate assessment, diagnosis, and pharmacological and nonpharmacological management of patients with a variety of health needs. In addition, PDAs are being used to store patient data from office computers in a form that is easily transportable. Thus, they have easy access to information about patients who call during off hours. These small computers can also be used for research purposes. PDA software is currently available that may help nurse practitioners in busy offices to collect data for research. Multiple nurse practitioners involved in a research project could forward data electronically from PDAs to a central research site for analysis. Care would need to be taken to protect the confidentiality of the data during transmission. Also, PDAs can be misplaced or stolen, thus threatening confidentiality. Researchers need to protect the information on their PDAs with a security code to ensure that no one but themselves can access their PDAs.
Bioinstruments
Advancements in technology have made it possible to interface bioinstruments with computers for data collection. The advantages of using computers for the acquisition and storage of physiological data from bioinstruments are numerous. Harrison (1989) summarized them as follows:
1. Increased accuracy and reliability are achieved by reducing errors that may occur when manually recording or transcribing physiologic data from patient monitors or other clinical instruments.
2. Linking microcomputers with biomedical instruments (e.g., cardiac, respiratory, blood pressure, or oxygen saturation monitors) permits more frequent acquisition and storage of larger amounts of data (e.g., once or more per second) than is practical with manual recording procedures.
3. Once established, computerized data acquisition systems save researcher time during both the data collection and analysis phases of research.
4. Even though the initial cost of equipment may be high, over the long run computerized data collection systems are less expensive, more efficient, and more reliable than hiring and training multiple human data collectors. (Harrison, 1989, p. 131)
There are some concerns with the use of computerized bioinstruments, but physiological data are usually best gathered and stored directly into computer databases to ensure accurate, complete data collection. Researchers must make every effort to deal with any problems. The microcomputer and the equipment required to interface it with the bioinstruments take up space in an already crowded clinical setting; when possible, existing equipment should be used to collect data. Purchasing the equipment, setting it up, and installing the software can be time consuming and expensive at the start of the research project. Thus, initial studies will usually require external funding. Another concern is that the nurse researcher will focus on the machine and neglect observing and interacting with the patient. The most serious disadvantage is the possibility of measurement error that can occur with equipment malfunctions and software errors, although regular maintenance and reliability checks of the equipment and software will reduce this problem.
Savian, Paratz, and Davies (2006) conducted a single-blind randomized, crossover study with 14 mechanically ventilated intensive care unit (ICU) patients. The purpose of the study was to determine the effectiveness of
manual hyperinflation (MHI) and ventilator hyperinflation (VHI) on respiratory mechanics (static compliance [Cst]), oxygenation (arterial oxygen tension [PaO2]/fraction of inspired oxygen [FIO2] ratio), and secretion removal (wet weight of sputum and peak expiratory flow rate [PEFR]) at different levels of PEEP [positive end-expiratory pressure]… a secondary aim was to investigate the hemodynamics (heart rate [HR], mean arterial pressure [MAP] and metabolic response (carbon dioxide output [VCO2]) during MHI and VHI. (Savian et al., 2006, p. 335)
The computerized bioinstruments that were used to collected and record data in the Savian et al. (2006) study are detailed in the following excerpt.
Savian et al.’s (2006) use of computerized bioinstruments enabled them to collect repeated measures on several physiological variables in an accurate and precise way. The data were collected by sensors and stored in the computer to reduce error and ease data analysis.
Developing a Codebook for Data Definitions
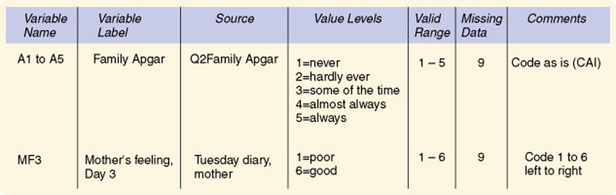
A codebook identifies and defines each variable in your study and includes an abbreviated variable name (often limited to six to eight characters), a descriptive variable label, and the range of possible numerical values for every variable entered in a computer file. Some codebooks also identify the source of each datum, thus linking your codebook with your data collection forms and scales. The codebook keeps you in control and provides a safety net for when you access the data later. Some computer programs, such as SPSS for Windows, allow you to print out your data definitions after setting up a database. Figure 17-4 is an example of data definitions from SPSS for Windows. Another example of coding is presented in Figure 17-5.
Developing a logical method of abbreviating variable names can be challenging. For example, you might use a quality-of-life (QOL) questionnaire in your study. It will be necessary for you to develop an abbreviated variable name for each item in the questionnaire. For example, the fourth item on a QOL questionnaire might be given the abbreviated variable name QOL4. A question asking the last time a home health nurse visited might be abbreviated HHN Lstvisit. Although abbreviated variable names usually seem logical at the time the name is created, it is easy to confuse or forget these names unless they are clearly documented.
We advise that you develop your codebook before initiating data collection. This practice encourages you to identify places in your forms that might prove to be a problem during data entry because of lack of clarity. Also, you may find that a single question contains not one but five variables. For example, an item might ask whether the subject received support from her or his mother, father, sister, brother, or other relatives and ask the subject to circle the number that represents those who provided support. You might think that you could code mother as 1, father as 2, sister as 3, brother as 4, and other as 5. However, because the individual can circle more than one, each relative must be coded separately. Thus, mother is one variable and would be coded 1 if circled and 0 if not circled. The father would be coded similarly as a second variable, and so on. Identifying these items before data collection may allow you to restructure the item on the questionnaire or data collection form to simplify computer entry.
Give the codebook with its data definitions to the individual or individuals who will enter your data into the computer before initiating data collection. In addition, provide the following information:
1. Copies of all scales, questionnaires, and data collection forms to be used in the study.
2. Information on the location of every variable on scales, questionnaires, or data collection forms.
3. Information on the statistical package to be used for analysis of the data.
4. Identification of the analyses to be conducted to describe the sample and to address the research purpose and the objectives, questions, or hypotheses.
5. Identification of a statistician to consult with about data analysis.
6. Determination of the database in which the data will be entered.
7. Information related to receiving the data, for example, whether you will deliver the data in batches or wait until all the data have been gathered before delivering it.
8. Estimation of the number of subjects to be included in the study.
9. Plan for documenting refusal rate, sample size, and attrition during the study.
10. Plan of the dates for data collection initiation and completion and for data entry.
With this information, the assistant can develop the database in preparation for receiving the data. It will take an average of 16 hours of concentrated work to prepare the database. Approximate dates for completion of the data entry, analyses, or both must be negotiated before beginning data collection. If you have a deadline for completing or presenting your results, such as an upcoming conference, you should share this information with those performing data entry and analysis.
COLLECTING DATA
Data collection is the process of selecting subjects and gathering data from these subjects. The actual steps of collecting the data are specific to each study and depend on the research design and measurement methods. Data may be collected on subjects by observing, testing, measuring, questioning, recording, or any combination of these methods. The researcher is actively involved in this process either by collecting data or by supervising data collectors. You will apply people-management and problem-solving skills constantly as data collection tasks are implemented, kinks in the research plan are resolved, and support systems are used.
Data Collection Tasks
In both quantitative and qualitative research, the investigator performs four tasks during the process of data collection. These tasks are interrelated and occur concurrently rather than in sequence. The tasks are selecting subjects, collecting data in a consistent way, maintaining research controls as indicated in the study design, and solving problems that threaten to disrupt the study. Selecting subjects is discussed in Chapter 14. Collecting data may involve administering Internet or paper-and-pencil scales; asking subjects to complete data collection forms in person or online; or recording data from observations, patients’ records, or health care equipment (Chapter 16 focuses on measurement strategies). Data collection tasks for qualitative studies are discussed in more detail in Chapter 23.
Maintaining Controls
Maintaining consistency and controls during subject selection and data collection protects the integrity or validity of the study. Research controls were built into the design to minimize the influence of intervening forces on the study findings. Maintenance of these controls is essential. They are not natural in a field setting, and letting them slip is easy. In some cases, these controls slip without the researcher realizing it. In addition to maintaining the controls identified in the plan, you must continually watch for previously unidentified extraneous variables that might have an impact on the data being collected. These variables are often specific to a study and tend to become apparent during the data collection period. The extraneous variables identified during data collection must be considered during data analysis and interpretation. These variables also must be noted in the research report to allow future researchers to control them.
Problem Solving
Problems can be perceived either as a frustration or as a challenge. The fact that the problem occurred is not as important as successfully resolving it. Therefore, the final and perhaps most important task of the data collection period may be problem resolution. Little has been written about the problems encountered by nurse researchers. Research reports often read as though everything went smoothly. The implication is that if you are a good researcher, you will have no problems, which is not true. Research journals generally do not provide enough space for the researcher to describe the problems encountered, and inexperienced researchers may get a false impression. A more realistic picture can be obtained through personal discussions with researchers about the process of data collection. Some of the common problems experienced by researchers are discussed in the following section.
Data Collection Problems
Murphy’s law (if anything can go wrong, it will, and at the worst possible time) seems to prevail at times in research, just as in other dimensions of life. For example, data collection frequently requires more time than was anticipated, and collecting the data is often more difficult than was expected. Sometimes changes must be made in the way the data are collected, in the specific data collected, or in the timing of data collection. People react to the study in unpredicted ways. Institutional changes may force modifications in the research plan, or unusual or unexpected events may occur. You must be as consistent as possible during the data collection process, but you must also be flexible in dealing with unforeseen problems. Sometimes, sticking with the original plan at all costs is a mistake. Skills in finding ways to resolve problems that will protect the integrity of the study can be critical.
In preparation for data collection, possible problems must be anticipated, and solutions for these problems must be explored. The following discussion describes some of the common problems and concerns and presents possible solutions. Problems that tend to occur with some regularity in studies have been categorized as people problems, researcher problems, institutional problems, and event problems.
People Problems
Nurses cannot place a subject in a laboratory test tube, instill one drop of the independent variable, and then measure the effect. Nursing studies are conducted by examining subjects as they interact with their environments. When research involves people, nothing is completely predictable. People, in their complexity and wholeness, have an impact on all aspects of nursing studies. Researchers, potential subjects, family members of subjects, health professionals, institutional staff, and others (“innocent bystanders”) interact within the study situation. You will need to closely observe and evaluate these interactions to determine their impact on your study.
Problems Selecting a Sample: The first step in initiating data collection—selecting a sample—may be the beginning of people problems. You may find that few available people fit your sample criteria or that many of those you approach refuse to participate in the study even though the request seems reasonable. Appropriate subjects, who were numerous a month earlier, seem to have disappeared. Institutional procedures may change, which might make many potential subjects ineligible for participation in the study. You may have to reevaluate the sample criteria or seek additional sources for potential subjects. In research institutions that care for the indigent, patients tend to be reluctant to participate in research. This lack of participation might arise because these patients are frequently exposed to studies, feel manipulated, or misunderstand the research. Patients may feel that they are being used or are afraid that they will be harmed.
Subject Attrition or Mortality: After you have selected a sample, certain problems might cause subject attrition (a loss of subjects from the study). For example, some subjects may agree to participate but then fail to follow through. Some may not complete needed forms and questionnaires or may fill them out incorrectly. To reduce these problems, someone from the research team can supervise the subjects while they complete essential documents. Some subjects may not return for a second interview or may not be home for a scheduled visit. Although you have invested time to collect data from these subjects, their data may have to be excluded from analysis because of incompleteness.
Sometimes subjects must be dropped from the study because of changes in health status. For example, the patient may be transferred out of intensive care where the study is being conducted; the patient’s condition may worsen, so he or she may no longer meet sample criteria; or the patient may die. Clinic patients may be transferred to another clinic or be discharged from the service. In the community, subjects may choose to discontinue services, or the limits of third-party reimbursement may force the health care provider to discontinue the services you are studying.
Subject attrition occurs, to some extent, in all studies. One way to deal with this problem is to anticipate the attrition rate and increase the planned number of subjects to ensure that a minimally desired number will complete the study. If subject attrition is higher than expected, consider continuing the data collection process for a longer period of time to achieve an adequate sample size. Sometimes a study might have to be completed with a smaller than expected sample size. If so, the effect of a smaller sample on the power of planned statistical analyses must be considered, because the smaller sample may not be adequate to test the hypotheses and result in a type II error.
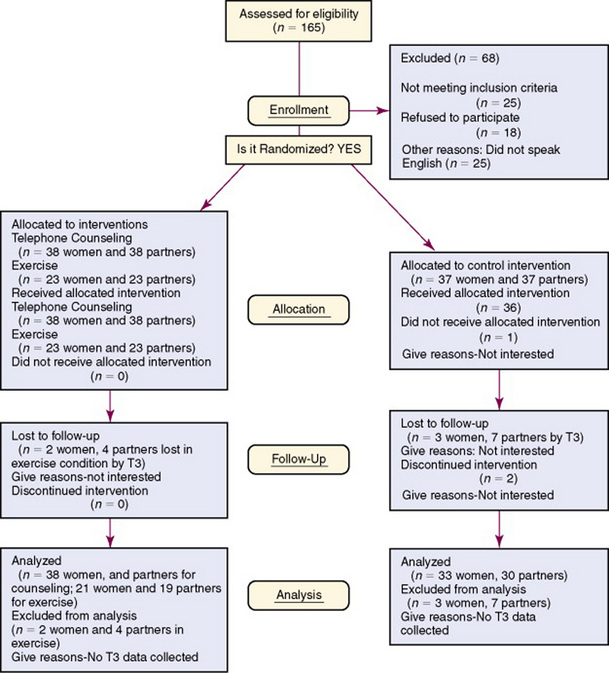
An increasing number of researchers are providing important information about subjects’ acceptance to participate in a study and attrition during the study to determine if the sample is representative of the study target population. Some published studies include a flow diagram that indicates the number of subjects meeting sample criteria, the potential subjects refusing to participate and their rationale, and the sample size for the study. If data are collected over time (repeated measures) or the study intervention is implemented over time, subjects will often drop out of a study, and it is important to document the attrition of these subjects. Badger, Segrin, Dorros, Meek, and Lopez (2007) provided a flow diagram that documented the participants’ selection, refusal rate, assignment to groups, and attrition over the weeks of data collection (Figure 17-6). The flow diagram clearly identifies important aspects of the sampling process and the rationale for the attrition of study subjects. This information enabled these researchers to determine the representativeness of their sample and any potential for bias.
Subject as an Object: The quality of interactions between the researcher and subjects during the study is a critical dimension for maintaining subject participation. When researchers are under pressure to complete a study, people can be treated as objects rather than as subjects. In addition to being unethical, such treatment alters interactions, diminishes the subject’s satisfaction, and increases subject mortality. Subjects are scarce resources and must be treated with care. The researcher’s treatment of the subject as an object can lead to similar treatment by other health care providers and result in poor quality of care. In this case, participation in the study becomes detrimental to the subject.
External Influences on Subject Responses: People interacting with the subject, the researcher, or both can have an important impact on the data collection process. Family members may not agree to the subject’s participation in the study or may not understand the study process. These people will, in most cases, influence the subject’s decisions related to the study. Researchers benefit from taking the time to explain the study and to seek the cooperation of family members. Family cooperation is essential when the potential subject is critically ill and unable to give informed consent.
Family members or other patients may also influence the subject’s responses to scales or interview questions. In some cases, subjects may ask family members, friends, or other patients to complete study forms for them. The subject may discuss questions on the forms with whomever happens to be in the room, and therefore the questionnaire may not record the subjects’ real feelings. If interviews are conducted while other persons are in the room, the subject’s responses may depend on his or her need to meet the expectations of other persons. Sometimes, a family member may answer questions addressed verbally to the patient. Thus, the setting in which a questionnaire is completed or an interview is conducted may determine the extent to which the answers are a true reflection of the subject’s feelings. If the privacy afforded by the setting varies from one subject to another, the subjects’ responses may also vary.
Usually, the most desirable setting for an interview is a private area away from distractions. If it is not possible to arrange for such a setting, the researcher can be present at the time the questionnaire is completed to decrease the influence of others. If the questionnaire is to be completed later or taken home and returned at a later time, the probability of influence by others increases and the return of questionnaires greatly decreases. The impact of this problem on the integrity of the study depends on the nature of the questionnaire.
Passive Resistance: Health professionals and institutional staff working with the subject may affect the data collection process. Some professionals verbalize strong support for the study and yet passively interfere with data collection. For example, nurses providing care may fail to follow the guidelines agreed on for providing the specific care activities being studied, or information needed for the study may be left off patient records. The researcher may not be informed when a potential subject has been admitted, and a physician who has agreed that his or her patients can be study subjects may decide as each patient is admitted that this one is not quite right for the study. In addition, the physician might become unusually unavailable to the researcher.
Nonprofessional staff may not realize the impact of the data collection process on their work patterns until the process begins. The data collection process may violate their beliefs about how care should be provided (and has been provided for the past 20 years). If ignored, their resistance can completely undo a carefully designed study. For example, research on skin care may disrupt a nursing aide’s bathing routine, so he or she may continue the normal routine regardless of the study protocol and thereby invalidate the study findings.
Because of the potential impact of these problems, the researcher must maintain open communication and nurture positive relationships with other professionals and staff during data collection. Problems that you and your team recognize early and deal with promptly will have fewer serious consequences than will those that you ignore. However, not all problems can be resolved. Sometimes you may need to seek creative ways to work around an individual or to counter the harmful consequences of passive resistance.
Researcher Problems
Some problems are a consequence of the researcher’s interaction with the study situation or lack of skill in data collection techniques. These problems are often difficult to identify because of the researcher’s personal involvement. However, their effect on the study can be serious.
Researcher Interactions: The researcher can become so involved in interactions with people involved in the study that data collection on the subject is not completed. Researcher interactions can also interfere with data collection in interview situations. If you are collecting data while you are surrounded by professionals with whom you interact socially and professionally, it is sometimes difficult to focus completely on the study situation. This lack of attention usually leads to loss of data.
Lack of Skill in Data Collection Techniques: The researcher’s skill in using a particular data collection technique can affect the quality of the data collected. A researcher who is unskilled at the beginning of data collection might practice the data collection techniques with the assistance of an experienced researcher. A pilot study to test data collection techniques can be helpful. If data collectors are being used, they also need opportunities to practice data collection techniques before the study is initiated. Sometimes a skill is developed during the course of study; if this is the case, as one’s skill increases, the data being collected may change and thereby confound the study findings and threaten the validity of the study. If more than one data collector is used, changes in skill can occur more frequently than if the researcher is the data collector. The skills of data collectors must be evaluated during the study to detect any changes in their data collection techniques.
Researcher Role Conflict: Professional nurses conducting clinical research often experience a conflict between the researcher role and the clinician role during data collection. As a researcher, one is observing and recording events. In some cases, the researcher’s involvement in the event, such as providing physical or emotional care to a patient during an interview, could alter the event and thus bias the results. It would be difficult to generalize the findings to other situations in which the researcher was not present to intervene. However, the needs of patients must take precedence over the needs of the study. The dilemma is to determine when the needs of patients are great enough to warrant researcher intervention.
Some patient situations are life threatening—such as respiratory distress and changes in cardiac function— and require immediate action by anyone present. Other patient needs are simple, can be addressed by any nurse available, and are not likely to alter the results of the study. Examples of these interventions include giving the patient a bedpan, informing the nurse of the patient’s need for pain medication, or helping the patient to open food containers. These situations seldom cause a dilemma.
Solutions to other situations are not as easy. Suppose, for example, that your study involves examining the emotional responses of patients’ family members during and immediately after the patients’ operations. Your study includes an experimental group that receives one 30-minute family support session before and during the patients’ operations and a control group that receives no support session. Both sets of families are being monitored for 1 week after the surgeries to measure their levels of anxiety and coping strategies. You are currently collecting data on the control group. The data consist of demographic information and scales measuring anxiety and coping. One of the family members is in great distress. After completing the demographic information, she verbally expresses her fears and the lack of support she has received from the nursing staff. Two other subjects from different families hear the expressed distress and concur; they move closer to the conversation and look to you for information and support. In this situation, a supportive response from you is likely to modify the results of the study because these responses are part of the treatment to be provided to the experimental group only. This interaction is likely to narrow the difference between the two groups and thus decrease the possibility that the results will show a significant difference between the two groups. How should you respond? Are you obligated to provide support? To some extent, almost any response will be supportive. One alternative is to provide the needed support and not include these family members in the control group. Another alternative is to recruit the help of a nonprofessional to collect the data from the control group. However, recognize that most people will provide some degree of support in the described situation, even though their skills in supportive techniques may vary.
Other dilemmas include witnessing unethical behavior that interferes with patient care or witnessing subjects’ unethical or illegal behavior (Field & Morse, 1985). Try to anticipate these dilemmas before data collection whenever possible. Pilot studies can help you to identify dilemmas likely to occur in a study, and you can build strategies into the design to minimize or avoid them. However, some dilemmas cannot be anticipated, and you must respond to these problems spontaneously. There is no prescribed way to handle difficult dilemmas; each case must be dealt with individually. We recommend discussing unethical and illegal behavior with colleagues, members of ethics committees, or legal advisors. After you have resolved the dilemma, it is wise to reexamine the situation for its effect on study results and consider options in case the situation arises again.
Maintaining Perspective: Data collection includes both joys and frustrations. Researchers must be able to maintain some degree of objectivity during the process and yet not take themselves too seriously (Marshall & Rossman, 2006). A sense of humor is invaluable. You must be able to experience the emotions and then become the rational problem solver. Management skills and mental health are invaluable to a lifetime researcher.
Institutional Problems
Institutions are in a constant state of change. They will not stop changing for the period of a study, and these changes often affect data collection. A nurse who has been most helpful in your study may be promoted or transferred. The unit on which your study is conducted may be reorganized, moved, or closed during the study. An area used for subjects’ interviews may be transformed into an office or a storeroom or may be torn down. Patient record forms may be revised, omitting data that you and your team collected. The record room personnel may be reorganizing their files and be temporarily unable to provide needed charts.
These problems are, for the most part, completely outside of the researcher’s control. Keep an ear to the internal communication network of the institution for advanced warning of impending changes. Contacts within the institution’s administrative decision-making system could warn you about the impact of proposed changes on an ongoing study. However, in many cases, data collection strategies might have to be modified to meet the newly emerging situation. Again, flexibility while maintaining the integrity of the study may be the key to successful data collection. Byers (1995) suggested that in the future the home setting may be more desirable than institutions as a data collection site and that the response rate in this setting is better than that in institutions. The disadvantage is that home visits are time intensive and the subject may not be home at the agreed appointment time.
Event Problems
Unpredictable events can be a source of frustration during a study. Research tools ordered from a testing company may be lost in the mail. The duplicating machine may break down just before 500 data collection forms are to be copied, or a machine to be used in data collection may break down and require 6 weeks for repair. A computer ordered for data collection may not arrive when promised, a tape recorder may jam in the middle of an interview, or after an interview the data collector may discover that he or she had not pushed the record button and there is no recording of the interview. Data collection forms may be misplaced, misfiled, or lost.
Local, national, or world events and nature can also influence subjects’ responses to a study. For example, one of our graduate students was examining patients’ attitudes toward renal dialysis. She planned to collect data for 6 months. Three months into the data collection process, three patients died as a result of a dialysis machine malfunction in the city where the study was being conducted. The event made national headlines. Obviously, this event could be expected to modify subjects’ responses. In attempting to deal with the impact of the event on the study, the graduate student could have modified the study and continued collecting data to examine the impact of news such as this on attitudes. However, the emotional climate of the clinics participating in the study was not conducive to this option. She chose to wait 3 months before collecting additional data and examined the data before and after the event for statistically significant differences in responses. Because she could not find any differences, she could justify using all the data for analysis.
Other less dramatic events can also have an impact on data collection. If data collection for the entire sample is planned for a single time, a snowstorm or a flood may require that the researcher cancel the meeting or clinic. Weather may decrease attendance far below that expected at a support group or series of teaching sessions. A bus strike can disrupt transportation systems to such an extent that subjects can no longer get to the data collection site. A new health agency may open in the city, which may decrease demand for the care activities being studied. Conversely, an external event can also increase attendance at clinics to such an extent that existing resources are stretched and data collection is no longer possible. These events are also outside the researcher’s control and are impossible to anticipate. In most cases, however, restructuring the data collection period can salvage the study. To do so, it is necessary to examine all possible alternatives for collecting the study data. In some cases, data collection can simply be rescheduled; in other situations, the changes needed may be more complex.
SERENDIPITY
Serendipity is the accidental discovery of something useful or valuable. During the data collection phase of studies, researchers often become aware of elements or relationships that they had not previously identified. These aspects may be closely related to the study being conducted or have little connection with it. They come from increased awareness and close observation of the study situation. Because the researcher is focused on close observation, other elements in the situation can come into clearer focus and take on new meaning. Like the open context situation discussed in Chapter 4, the researcher’s perspective shifts, and new gestalts are formed.
Serendipitous findings are important to the development of new insights in nursing theory. They can be important for understanding the totality of the phenomenon being examined. Additionally, they lead to areas of research that generate new knowledge. Therefore, it is essential to capture these insights as they occur. These events must be carefully recorded, even if their impact or meaning is not understood at the time. Sometimes, when these notes are reexamined at a later time, patterns begin to emerge.
Serendipitous findings can also lead the researcher astray. Sometimes researchers forget the original plan and redirect their attention to the newly discovered dimensions. Although modifying data collection to include data related to the new discovery may be valid, there has not been time to carefully plan a study related to the new findings. Examination of the new data should only be an offshoot of the initial study. Data collected as a result of serendipitous findings can guide future studies and must be included in presentations and publications related to the study. Although the meaning of the discovery may not be understood, sharing the information may lead to insights by researchers studying related phenomena.
HAVING ACCESS TO SUPPORT SYSTEMS
The researcher must have access to individuals or groups who can provide support and consultation during the data collection period. Support systems themselves have been the subject of much study in recent years. In some cases, they can be the source of both stress and support. However, current theorists propose that to be classified as support, the individual or group must enhance the ego strength of the individual. Three dimensions of support have been identified: (1) physical assistance, (2) the provision of money or other concrete needs such as equipment or information, and (3) emotional support. These types of support can usually be obtained from academic committees; from institutions serving as research settings; and from colleagues, friends, and family.
Support of Academic Committees
Although thesis and dissertation committees are basically seen as stern keepers of the sanctity of the research process, they also serve as support systems for neophyte researchers. In fact, committee members must be selected from faculty who are willing and able to provide the needed support. Experienced researchers among faculty are usually more knowledgeable about the types of support needed. Because they are directly involved in research, they tend to be sensitive to the needs of neophyte researchers.
Institutional Support
A support system within the institution where the study is being conducted is also important. Support might come from people serving on the institutional research committee or from nurses working on the unit where the study is to be conducted. These people often have knowledge of how the institution functions, and their closeness to the study can increase their understanding of the problems experienced by the researcher and subjects. Do not overlook their ability to provide useful suggestions and assistance. Your ability to resolve some of the problems encountered during data collection may depend on having someone within the power structure of the institution who can intervene.
Personal Support
In addition to professional support, it is helpful to have at least one significant other with whom one can share the joys, frustrations, and current problems of data collection. A significant other can often serve as a mirror to allow you to see the situation clearly and perhaps more objectively. Through personal support, the researcher can share and release feelings and distance himself or herself from the data collection situation. Alternatives for resolving the problem can then be discussed. Data collection is a demanding, but rewarding, time that increases the confidence and expertise of the neophyte researcher.
MANAGING DATA
When data collection begins, you will have to handle large quantities of data. To avoid a state of total confusion, make careful plans before data collection begins. Plans are needed to keep all data from a single subject together until analysis is begun. Write the subject code number on each form, and check the forms for each subject to ensure that they are all present. Researchers have been known to sort their data by form, such as putting all the scales of one kind together, only to realize afterward that they had failed to code the forms with subject identification numbers first. They then had no idea which scale belonged to which subject, and valuable data were lost.
Allot space needs for storing forms. Purchase file folders, and design a labeling method to allow easy access to data; color coding is often useful. For example, if you are using multiple forms, the subject’s demographic sheet could be one color, with different colors for the visual analogue scale; the pain questionnaire; the physiological data sheet with blood pressure, pulse, and respiration readings; and the interview notes. Use envelopes to hold small pieces of paper or note cards that might fall out of a file folder. Plan to code data and enter them into the computer as soon as possible after data collection to reduce the loss or disorganization of data. If data are collected on a computer, make sure the data are backed up so that they are not lost if the computer fails.
Preparing Data for Computer Entry
Data must be carefully checked and problems corrected before you initiate the data entry phase. The data entry process should be essentially automatic and require no decisions regarding the data. Such simplicity in data entry will reduce the number of data entry errors and markedly decrease the time required for entry. It is not sufficient to establish general rules for those entering data such as “in this case always do X.” This action still requires the data enterer to recognize a problem, refer to a general rule, and correct the data before entering them. Anything that alters the rhythm of data entry increases errors. Carefully examine each datum to search for the following problems and resolve them before data entry:
1. Missing data. Provide the data if possible or determine the impact of the missing data on your analysis. In some cases, the subject must be excluded from at least some of the analyses, so you must determine what data are essential.
2. Items in which the subject provided two responses when only one was requested. For example, if the item asked the subject to mark the most important in a list of 10 items and the subject selected 2, you must decide how to resolve this problem; do not leave the decision to an assistant who is entering the data. On the form, indicate how the datum is to be coded.
3. Items in which the subject has marked a response between two options. This problem commonly occurs with Likert-type scales, particularly those using forced choice options. Given four options, the subject places a mark on the line between response 2 and response 3. On the form, indicate how the datum is to be coded (missing data is an option).
4. Items that ask the subject to write in some information such as occupation or diagnosis. Such items are a data enterer’s nightmare. Develop a list of codes for entering such data. Rather than leaving it up to the assistant to determine which code matches the subject’s written response, the researcher should enter this code before turning the data over for entry. After the data have been checked and needed codes written in, it is prudent to make a copy rather than turning over the only set of your data to an assistant.
Data Entry Period
If you are entering your own data, develop a rhythm to your entry. Avoid distractions while entering data, and limit your data entry periods to 2 hours at a time to reduce errors. Back up the database after each data entry period, and store it on a flash stick or CD-ROM. It is possible for the computer to crash and lose all of your data. If an assistant is entering your data, make yourself as available as possible to respond to questions and address problems. After entry, the data should be checked for accuracy. Data checking is discussed in Chapter 18 in the Preparation of the Data for Analysis section.
Storage and Retrieval of Data
In this time of flash sticks and CD-ROM burners, it is relatively easy to store data. Decide how long you wish to store the data. The original data forms must be stored, as well as the database. There are several reasons to store data. The data can be used for secondary analyses. For example, individuals who are participating in a research program related to a particular research focus may pool data from various studies for access by all members of the group. The data are available to document the validity of your analyses and the published results of your study. Because of nationally publicized incidents of scientific misconduct where researchers invented data from which multiple publications were developed, you are wise to preserve documentation that your data were obtained as you claim. Issues that have been raised include how long data should be stored, the need for institutional policy regarding data storage, and whether graduate students who conduct a study should leave a copy of their data at the university. Thomas (1992) surveyed 153 researchers to determine their responses to these questions. She found that the length of data storage varied greatly, with 29% storing their data 5 years, 31% storing it 10 years, and 21% storing it forever. Most researchers stored their data in their office (84%), and a few used a central location (12%) or a laboratory (4%). The forms of data storage devices preferred were disk (54%), tape (47%), and paper/raw data (32%). Some researchers preferred more than one storage device for their data. The majority of the researchers (86%) indicated that their institutions did not have a policy for data storage, and most graduate students (74%) did not leave a copy of their data at the university.
SUMMARY
• To plan the process of data collection, the researcher must determine step by step how and in what sequence data will be collected and the timing of the process.
• Decision points that occur during data collection must be identified and all options considered.
• The researcher must decide who will collect the data.
• If data collectors are used, they must be informed about the research project, introduced to the instruments, and provided equivalent training in the data collection process. After training, data collectors must be evaluated to determine their consistency.
• Consistency in data collection across subjects is critical, and so is consistency among data collectors if more than one data collector is used.
• The researcher must develop data collection forms so that data can be recorded and entered into the computer more easily.
• A research treatment or intervention must be implemented in a reliable and competent way to promote intervention fidelity in a study. The person implementing the study intervention (an interventionalist) must be trained to ensure adherence and competence in the intervention protocol.
• Data collection involves four tasks: selecting subjects, collecting data in a consistent way, maintaining research controls, and solving problems that threaten to disrupt the study.
• Some of the problems researchers encounter during data collection include problems in selection of a sample, subject mortality, treatment of the subject as an object, external influences on subject responses, passive resistance, researcher interactions, lack of skill in data collection techniques, researcher role conflicts, and maintaining perspective.
• A successive study requires support that is often obtained from academic committees; health care agencies; and personnel, family members, and friends.
• Data collected during a study must be accurately entered in a computer and safely stored.
References
Badger, T., Segrin, C., Dorros, S., Meek, P., Lopez, A.M. Depression and anxiety in women with breast cancer and their partners. Nursing Research. 2007;56(1):44–53.
Bernhardt, J.M., Strecher, V.J., Bishop, K.R., Potts, P., Madison, E.M., Thorp, J. Handheld computer-assisted self-interviews: User comfort level and preferences. American Journal of Health Behavior. 2001;25(6):557–563.
Byers, V.L. Overview of the data collection process. Journal of Neuroscience Nursing. 1995;27(3):188–193.
Craig, J.V., Smyth, R.L. The evidence-base practice manual for nurses, (2nd ed.). Edinburgh, Scotland: Churchill Livingstone, 2007.
Dennis, K.E. Managing questionnaire data through optical scanning technology. Nursing Research. 1994;43(6):376–378.
Field, P.A., Morse, J.M. Nursing research: The application of qualitative approaches. Rockville: MD: Aspen, 1985;65–90.
Gift, A.G., Creasia, J., Parker, B. Utilizing research assistants and maintaining research integrity. Research in Nursing & Health. 1991;14(3):229–233.
Harrison, L.L. Interfacing bioinstruments with computers for data collection in nursing research. Research in Nursing & Health. 1989;12(2):129–133.
Im, E., Chee, W., Guevara, E., Liu, Y., Lim, H., Tsai, H. Cancer pain management: Decision support computer program, 2006. [Unpublished manuscript].
Im, E., Chee, W., Guevara, E., Liu, Y., Lim, H., Tsai, H. Gender and ethnic differences in cancer pain experience: A multiethnic survey in the United States. Nursing Research. 2007;56(5):296–306.
Marshall, C., Rossman, G.B. Designing qualitative research, (4th ed.). Thousand Oaks: CA: Sage, 2006.
Melnyk, B.M., Fineout-Overholt, E. Evidence-based practice in nursing & healthcare: A guide to best practice. Philadelphia: Lippincott Williams & Wilkins, 2005.
Savian, C., Paratz, J., Davies, A. Comparison of the effectiveness of manual and ventilator hyperinflation at different levels of positive end-expiratory pressure in artificially ventilated and intubated intensive care patients. Heart & Lung. 2006;35(5):334–341.
Stein, K.F., Sargent, J.T., Rafaels, N. Intervention research: Establishing fidelity of the independent variable in nursing clinical trials. Nursing Research. 2007;56(1):54–62.
Thomas, S.P. Storage of research data: Why, how, where? Nursing Research. 1992;41(5):309–311.
Washington, C.C., Moss, M. Methodology corner: Pragmatic aspects of establishing interrater reliability in research. Nursing Research. 1988;37(3):190–191.