Injury, Inflammation, and Healing
Pathology is defined as the structural and functional changes in the body caused by disease or trauma. Understanding the normal structure and function of the tissues is required before the discussion of pathology. The organization of the material presented in this chapter parallels the processes underlying pathology—that is, cell injury and the factors causing this injury, inflammation as a secondary response to cell injury, and tissue healing, the third step of the process toward homeostasis.
The role of nerve-immune interactions in regulation and tissue healing is just beginning to be revealed and will continue to enhance our understanding of and intervention in injury, inflammation, and recovery. A new area of science called psychoneuroimmunology (see description in Chapters 1 and 7) describes the influences of the nervous system on immune and inflammatory responses and how these contribute to the healing and repair process. For example, it is now clear that mast cells, T cells, neutrophils, and monocytes can directly alter tissue physiology through the release of mediators and cytokines. In addition, aging, age-related changes, and various other factors can influence homeostasis and the recovery process and are the major focus of the next two sections.
CELLULAR AGING
Various components of cells (e.g., mitochondria, ribosomes, or cell membrane) are subject to changes associated with aging. Mitochondrial deoxyribonucleic acid (DNA) is considered a prime target for age-related changes. DNA has to replicate and maintain itself to preserve the primary genetic message. This takes place through division, which can result in alterations of the genetic code by anything that can damage DNA (e.g., physical, chemical, or biologic factors; spontaneous mutations of genes; exposure to radiation). Anything that can alter the information content of the cell can cause changes in function and affect the ability of the cell to maintain homeostasis.
The ability of a cell to resist microorganisms or to recover from injury or inflammation is dependent in part on the underlying state or health of the cells. Age-related changes at the cellular level are present but remain difficult to measure or quantify; researchers are working toward finding satisfactory biomarkers of aging at the cellular level. Age-associated deterioration in cells leads to tissue or organ deficiencies and ultimately to the expression of aging or disease. The most well-described age-associated change in the subcellular structure (lysosomes) of postmitotic cells, especially neurons and cardiac myocytes, is the presence of a component called lipofuscin, an aging-pigment granule that is found in high concentrations in old cells. The explanation for the increase of lipofuscin with age and the effects of these intracellular deposits on function remains under investigation. It is suspected that pressure from this pigmented lipid on the cell nucleus may interfere with cellular function.124,142
Theories of Cellular Aging
The aging process is often associated with impaired wound healing, but the cellular and molecular mechanisms implicated are not completely understood.37,78 More than 300 theories exist to explain the aging phenomenon from a cellular level. Many of these theories originate from the study of changes that accumulate with time. In organs composed of cells that cannot regenerate, such as those of the heart and brain, the wear-and-tear theory may account for the decline in function of these organs. Other factors may also play a role, such as the influence of genetics suggested by the genetic hypothesis that aging is a genetically predetermined process.
The free radical theory of aging is the most popular and widely tested and is based on the chemical nature and wide presence of free radicals causing DNA damage and cellular oxidative stress as it relates to the aging process (see the Chemical Factors section under Mechanisms of Cell Injury in this chapter).
The discovery of the telomeres, the structure at the end of chromosomes, has added the telomere aging clock theory for the molecular mechanisms that lead to senescence. This theory suggests that the telomere acts as a molecular clock signaling the onset of cell senescence. Normal human cells will not divide forever but eventually enter a viable nondividing state (senescence). The progressive accumulation of senescent cells contributes to but does not exclusively cause the aging process. Cell senescence acts as an anticancer mechanism to control the potential for cellular proliferation71,146 (see further discussion in Chapter 9). Because of the close association between telomere dysfunction and malignancy, both pathologists and clinicians expect this molecule to be a useful malignancy marker.
Pathologic changes associated with aging vary from individual to individual but usually consist of reduced functional reserve caused by atrophy of tissues or organs. Resistance to infection declines with age, and pathologic processes such as atherosclerosis result in increased cardiovascular and cerebrovascular injuries or death.
Studies have shown that considerable potential exists for improving aerobic capacity by training. This observation has cellular implications. For example, mitochondria of cardiac and skeletal78 muscle cells improve function under appropriate training conditions. If changes in diet and exercise or treatment with hormones or compounds, such as antioxidants (see Fig. 6-2), are able to modify damage by reactive oxygen species and the body can reestablish cellular norms, then this information has great implications for the various cellular and molecular theories on aging and our approach to the aging process.
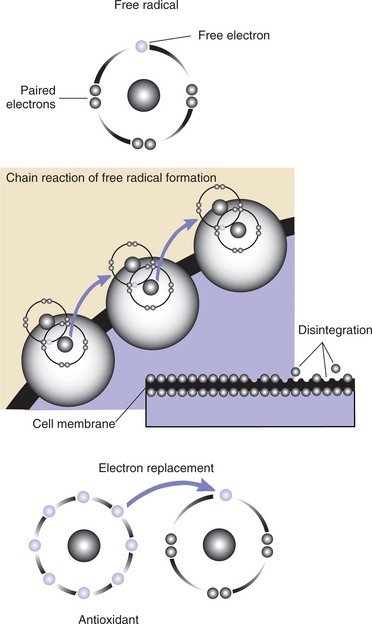
Figure 6-2 The oxidative process and formation of free radicals. Normal metabolic processes and a variety of other extrinsic factors, such as pollution, poor nutrition, and exposure to toxic chemicals, can result in the formation of free radicals when normal oxygen atoms lose one of their four paired electrons. The resulting unstable atom attempts to replace the missing electron by “stealing” an electron from a healthy cell, creating another unstable atom (free radical) and setting off a chain reaction referred to as oxidation. Oxidation as a by-product of metabolism damages cell membranes, leading to intrinsic cellular damage, a part of the normal aging process. Free radical damage (oxidation) is believed to alter the way cells encode genetic information in the DNA and may contribute to a variety of diseases and disorders. Antioxidant molecules freely give up an electron to stabilize the oxygen atom without becoming unstable and without initiating a chain reaction.
CELL INJURY
Understanding cell injury, inflammation, and tissue healing serves as a solid foundation for the other topics presented in this text. We begin by acknowledging that the structural and functional changes produced by pathology start with injury to the cells that make up the tissues. Mild injury produced by stressors leads to sublethal alterations of the affected cells, whereas moderate or severe injury leads to lethal alterations. After cell injury, the body reacts by initiating the process of inflammation. The amount, type, and severity of the inflammatory reaction are dependent on the amount, type, and severity of the injury. As part of the healing process, the inflammatory process is responsible for the removal of the injurious agent, removal of cellular debris, and the initiation of the healing process. The healing process occurs to allow restoration of structure and function whenever possible.
To achieve complete restoration of function, regeneration of the damaged tissue must occur. Often, regeneration of the tissue is not possible, and the body must settle for tissue repair by nonfunctional, connective tissue (fibrosis or scar tissue). This connective tissue helps maintain structural integrity but has none of the functional properties of the original cells and tissues.
Mechanisms of Cell (Tissue) Injury
Cells may be damaged by a variety of mechanisms. The most important mechanisms are listed in Box 6-1. Each of these mechanisms leads to either a reversible (sublethal) or irreversible (lethal) injury. Whether the injury is reversible is dependent on the cell’s ability to withstand the derangement of homeostatic mechanisms and its adaptability (i.e., ability to return to a state of homeostasis). Reversing the injury and achieving homeostasis are determined by a combination of factors including the mechanism of injury, length of time the injury is present without intervention, and the severity of the injury.
Ischemia
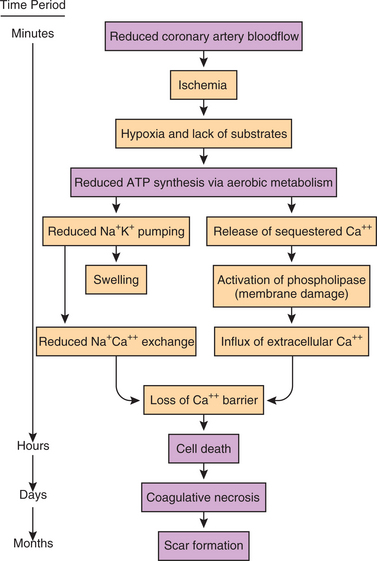
At the tissue or organ level, ischemia is defined as blood flow below the minimum necessary to maintain cell homeostasis and metabolic function. This can be due to a reduction in flow or an increase in metabolism of the tissue beyond the capability of the arterial vascular system. Insufficient blood flow results in a critical reduction in oxygen delivery to the tissue that is partial (hypoxia) or total (anoxia), a decreased delivery of nutrients, and decreased removal of waste products from the tissue. The lack of oxygen leads to loss of aerobic metabolism. The resulting reduction in adenosine triphosphate (ATP) synthesis leads to accumulation of ions and fluid intracellularly. The cells swell and their function is compromised. This concept is discussed further in the section on Reversible Cell Injury in this chapter.
Hypoxia or anoxia may occur under many circumstances, including obstruction of the respiratory tree (e.g., suffocation secondary to drowning), inadequate transport of oxygen across the respiratory surfaces of the lung (e.g., pneumonia), inadequate transport of oxygen in the blood (e.g., anemia), or an inability of the cell to use oxygen for cellular respiration (e.g., chemical poisoning).26
Ischemia is usually the result of arterial lumen obstruction and narrowing caused by atherosclerosis and/or an intravascular clot called a thrombus. Ischemia, resulting in myocardial infarction (MI) and stroke (lack of blood flow to the heart or brain, respectively), can cause death of tissue (necrosis) and accounts for two of the three leading causes of mortality in industrialized nations.
Infectious Agents
Infectious agents, such as bacteria, viruses, mycoplasmas, fungi, rickettsiae, protozoa, prions, and helminths (see Chapter 8), may also cause cell injury or death. Bacterial and viral agents are responsible for the vast majority of infections. Bacterial infections cause cell injury primarily by invading tissue and releasing exotoxins and endotoxins that can cause cell lysis and degradation of extracellular matrix and aid in the spread of the infection. Injury can also result from the inflammatory/immunologic reactions induced by bacteria in the host. For example, exotoxins may be released by clostridial organisms that cause gas gangrene, tetanus, and botulism.
Clostridium tetani, for example, releases an exotoxin that is preferentially absorbed by the alpha motor neurons and delivered into the central nervous system (CNS). Once inside the CNS, the exotoxin crosses the synapse of the anterior horn cell and interferes with release of inhibitory neurotransmitters. This disruption of homeostasis eventually causes the activation of motor neurons that in turn cause involuntary muscular contractions (tetanus).130
When microorganisms or their toxins are present in the blood, a condition called sepsis can occur. Endotoxins released from gram-negative bacteria induce the synthesis of cytokines (extracts of normal leukocytes such as tumor necrosis factor [TNF] and interleukins [ILs]) that are responsible for many of the systemic manifestations of sepsis (see Box 6-5).
In sepsis, endothelial cell damage, loss of plasma volume, and maldistribution of blood flow result in hypovolemia. Cardiovascular collapse may ensue and lead to a condition called septic shock. The detection of an infectious agent initiates an inflammatory reaction designed to contain and inactivate the pathogen, but the magnitude of this defensive response by the host may also cause cellular or tissue destruction in the infected area.
Viruses kill cells by one of two mechanisms (Fig. 6-1) and are the consequence of complete redirection of the cell’s biosynthesis towards viral replication. The first is a direct cytopathic effect usually found with ribonucleic (RNA) viruses. These viruses kill from within by disturbing various cellular processes or by disrupting the integrity of the nucleus and/or plasma membrane.
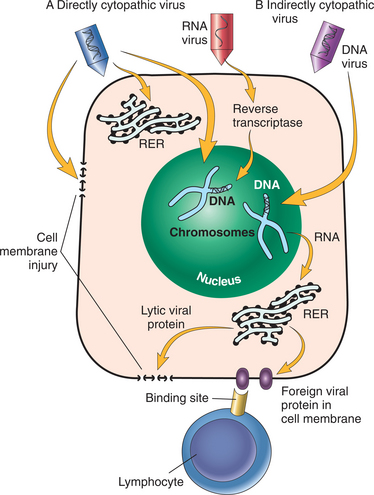
Figure 6-1 Mechanisms of cell destruction by viruses. A, Direct cytopathic effect: RNA virus inserts itself in a receptor on the cell membrane and is brought into the cell. The RNA virus is altered into DNA by reverse transcriptase. The DNA within the nucleus of the cell forms various types of RNA that allows for protein synthesis in the rough endoplasmic reticulum (RER). The protein formed inserts itself into the cell membrane, forming a channel that allows ions and extracellular fluid to enter, leading to cell lysis (directly killing the cell). B, Indirect cytopathic effect mediated by immune mechanisms: DNA virus inserts itself in a receptor on the cell membrane and is brought into the cell. The DNA virus within the nucleus of the cell forms various types of RNA that allow for protein synthesis in the RER. This foreign viral protein inserts into the cell membrane and becomes a neoantigen. This neoantigen will be recognized by the T lymphocytes that will react to and kill (indirectly) the infected cell. (From Damjanov I: Pathology for the health-related professions, ed 3, Philadelphia, 2006, WB Saunders.)
Virally encoded proteins become inserted into the plasma membrane of the host cell (forming a channel) and alter the permeability of the cell membrane to ions. The resulting loss of the ionic barrier leads to cell swelling and death. DNA type viruses also kill cells through an indirect cytopathic effect by integrating themselves into the cellular genome. These viruses encode the production of foreign proteins, which are exposed on the cell surface and recognized by the body’s immune cells.
Immunocompetent cells, such as the T lymphocyte, recognize these virally encoded proteins inserted into the plasma membrane of host cells and attack and destroy the infected cell. When the immune system is compromised or if the number of invading microorganisms overwhelms the immune system, disease (and the symptoms of illness) occurs.
Immune Reactions
Although the immune system normally functions in defense against foreign antigens, sometimes the system becomes overzealous in its activity, leading to hypersensitivities ranging from a mild allergy to life-threatening anaphylactic reactions or autoimmune disorders (attacking oneself). The mechanisms by which the immune system can lead to cell injury or death include antibody attachment, complement activation, and activation of the inflammatory cells (e.g., neutrophils, macrophages, T and B lymphocytes, mast cells, and basophils).
Cell injury and disease can be caused by the immune system in numerous ways. For example, allergies are caused by the presence of high numbers of a specific antibody-E (IgE) on the surface of specialized cells (mast cells and basophils, which release histamine), resulting in mild, moderate, or severe allergic reactions. Examples of mild reactions include the runny nose and watery eyes caused by a mild allergic response.
Moderate reactions include severe hypoxia caused by asthmatic bronchoconstriction. Severe reactions can result in a potentially life-threatening circulatory collapse seen in anaphylaxis (a whole-body allergic reaction). The presence of what would normally be considered optimal ratios of antigen to antibody in the circulation may lead to damage of filtration in the kidney because of excess deposition of antigen-antibody complexes in the glomeruli.
Cross-reactivity between foreign and host antigens is another immune mechanism that can compromise the body. For example, cross-reaction between streptococcal and myocardial antigens can occur in rheumatic fever and result in injury of cardiac valves. Alternately, the chronic persistence of a foreign antigen by a foreign body or microorganism that cannot be cleared by the body may lead to a specific type of chronic inflammatory reaction called a granuloma (e.g., tuberculosis). Finally, sensitization to endogenous antigens can lead to type 1 diabetes mellitus caused by destruction of islet cells by T lymphocytes sensitized by islet antigens released during an antecedent viral infection.
Genetic Factors
Genetic alterations lead to cellular injury or death by three primary means: (1) alterations in the structure or number of chromosomes that induce multiple abnormalities, (2) single mutations of genes that cause changes in the amount or functions of proteins, and (3) multiple gene mutations that interact with environmental factors to cause multifactorial disorders. These genetic alterations can be severe enough to cause fetal death in utero, resulting in spontaneous abortion. Some may cause congenital malformations, whereas others do not manifest pathologic alterations until midlife such as Huntington’s chorea. Down syndrome is an example of an alteration in the number of chromosomes that results in multiple abnormalities. This condition, caused by the abnormal presence of a third chromosome in the twenty-first pair, includes cardiac malformations, increased susceptibility to severe infections, cognitive and developmental delays, and increased risk of leukemia and Alzheimer’s dementia.
Sickle cell anemia, low-density lipoprotein (LDL) receptor deficiency, and α-antitrypsin deficiency are examples of single gene mutations. In the case of α-antitrypsin deficiency, the deficiency in a protease inhibitor causes enhanced degradation of elastic tissue surrounding the alveoli of the lungs, which in turn leads to emphysema. Examples of multiple gene mutations that can cause disease include hypertension and type 2 diabetes mellitus. In type 2 diabetes mellitus, obesity and other environmental factors induce the expression of the diabetic genetic trait.
Nutritional Factors
Imbalances in essential nutrients can lead to cell injury or cell death. For example, deficiencies of essential amino acids interfere with protein synthesis. Synthesis of proteins is required to replace cell proteins lost through normal catabolism, through growth, and in preparation for cell replication. Cell replication is essential for the healing processes after cell injury and the replacement of cells lost through normal turnover.
The consequence of protein malnutrition is a condition called kwashiorkor; marasmus, another form of malnutrition, is a consequence of generalized dietary deficiency. These two diseases are still leading causes of death in impoverished countries. In many industrialized countries, excessive nutrient intake leads to obesity and its many complications.
Nutritional imbalance can also occur as a result of abnormal levels of either vitamins or minerals. These nutrients function as cofactors for biosynthetic reactions or are essential components of proteins or membranes; their deficiency usually affects selected cells or tissues. For example, a deficiency of iron leads to anemia, and the presence of excessive amounts of iron in the tissues can cause damage by the formation of free radicals.
Vitamin C (ascorbic acid) deficiency can be associated with a wide range of connective tissue symptoms (Box 6-2). Frank deficiencies of ascorbate are uncommon in the United States, although certain population groups may be at increased risk for deficient intake sometimes referred to as biochemical scurvy (Box 6-3).
Physical Factors
Trauma and physical agents can lead to cell injury and/or death. Blunt trauma caused by motor vehicle accidents is a leading killer in the United States. Massive brain contusions, injury to internal organs and soft tissues, and blood loss may lead to immediate mortality. Survivors may succumb to infections and multiple organ failure. Repair of injuries to soft tissue, skeletal and muscular systems, and internal organs often requires prolonged periods of rehabilitation. Penetrating trauma inflicted by a variety of weapons can result in multiple complications.
Extremes of physical agents, such as temperature, radiation, and electricity, may damage cells. Generalized increases in body temperature (hyperthermia) or reduction in body temperature (hypothermia) can lead to cell injury; high or low tissue temperatures can cause tissue injury or death. With increased temperature, the resulting morbidity and mortality are dependent on the severity of the burn and the total surface area that was burned. Markedly reduced temperatures may induce the freezing of tissue (frostbite). Ice crystals in cellular tissue rupture the cell membrane, which leads to cell death.
Irradiation for the treatment of cancer can cause injury of susceptible normal cells. Ionizing radiation causes radiolysis of water and the production of hydroxyl radicals (−OH). These radicals will lead to membrane damage and breakdown of structural and enzymatic proteins that result in cell death. Often, arterioles that supply oxygenated blood are damaged by ionizing radiation, resulting in inadequate nutritional supply leading to ischemia and death of the irradiated tissues. Irradiation also causes damage to nucleic acids and may result in gene mutations, possibly leading to neoplasia years later.
Mechanical Factors
The physical stress theory may help explain mechanical factors influencing tissue adaptation and injury. The physical stress theory proposes that changes in the relative level of physical stress cause a predictable adaptive response in all biologic tissue. Typical tissue response to physical stress includes decreased stress tolerance (e.g., atrophy), maintenance, increased stress tolerance (e.g., hypertrophy), injury, and death.98
Failure of a tissue occurs when the applied load exceeds the failure tolerance of the tissue. Soft tissues are influenced by the history of recent physical stresses, so that the accumulation of individual stresses can cause injury. Characteristics of the load, such as rate, compression, and forces (e.g., torsion, shear), along with the properties of the affected tissue determine the type and extent of tissue damage. The time elapsed since injury and the extent of tissue damage determine the inflammatory response.8,90
With repetitive and/or forceful tasks, the initiating stimuli for inflammatory responses include repeated overstretch, compression, friction, and anoxia. These insults lead to mechanical injury of cellular membranes and intracellular structures and a localized release of proteins such as collagen, fibronectin, and cytokines.8
A single high load or stress from a traumatic fall, car accident, or other traumatic event can cause significant injury. Bones can fracture from one episode of high-magnitude force, while workers lifting heavy boxes repeatedly can incur a slow degradation of the tissue tolerance. Decreasing tissue tolerance may explain why there are no active acute inflammatory indicators in tendons associated with tendinitis. Instead, antiinflammatory mediators and fibrotic proliferation are observed, suggesting the acute inflammatory phase has resolved. Tennis elbow or golfer’s elbow is recognized in many cases as a noninflammatory condition after an inflammatory episode.8 In fact, research is ongoing to find ways to reinitiate the inflammatory cascade and promote healing in an otherwise degenerative process.35 Low loads sustained over a long period of time, such as workers who remain in a fixed, flexed posture for prolonged periods of time, can also result in tissue injury because of decreased tissue tolerance.98
Some soft tissues, such as ligaments, can rupture with a single high-magnitude force but can also fail from repeated bouts of moderate-magnitude stress. Likewise, as mentioned, bone can fracture from high-magnitude force but can also develop stress fractures or stress reactions from repeated episodes of moderate-magnitude force.70 Altering mechanical stress (either increasing or decreasing forces) can be used to benefit individuals under varying circumstances. For example, reducing mechanical stress by offloading or pressure reduction is a concept used for healing ulcers and preventing their recurrence.
Controlled increase in physical stress is the underlying principle of progressive resistive exercise used to cause muscle fibers to hypertrophy and thereby able to withstand and generate greater force. Higher than normal levels of physical stress can promote remodeling in bone. Musculoskeletal tissues subjected to higher than normal levels of stress become more tolerant to subsequent physical stresses and are more resistant to injury.98
Chemical Factors
Toxic substances cause chemical injury. These substances can be divided into two categories: those that can injure cells directly and those that require metabolic transformation into the toxic agent. Examples of chemicals that injure cells directly are heavy metals, such as mercury, that bind to and disrupt critical membrane proteins and a number of toxins and drugs, such as alkylating agents, used in chemotherapy.
Alkylating agents, such as nitrogen mustards, induce cross linking of DNA and inactivation of other essential cellular constituents. Carbon tetrachloride and acetaminophen are examples of inert substances that must be metabolized to reactive intermediates to cause cell injury. Taken in large amounts, most medications can be toxic, and many are even lethal. Suicide by drug overdose is a common example of drug-induced chemical toxicity.
Free Radical Formation.: An important mechanism of cell injury and disease is the production of reactive oxygen species sometimes referred to as the formation of free radicals. Free radicals are an integral part of metabolism and are formed continuously in the body. They can exert positive effects (e.g., on the immune system) or negative effects (e.g., lipid, protein, or DNA oxidation). A variety of normal and pathologic reactions can lead to the activation of oxygen by the sequential addition or subtraction, respectively, of one electron at a time (Fig. 6-2).
For example, the body’s natural process of using oxygen and food to produce energy can create free radicals as a by-product of these functions. These unpaired electrons are reactive and commonly bind to oxygen for stabilization. The oxygen then binds to hydrogen for stabilization. This series of reactions generated by normal cellular metabolism results in a phenomenon referred to as oxygen toxicity and yields superoxide (O2−), hydrogen peroxide (H2O2), and hydroxyl radical (−OH). These forms of reactive oxygen are referred to as oxygen radicals, which are toxic to cells.
The cellular enzymes always scavenging the body to protect cells from this type of injury normally inactivate these radicals and convert the radical back to usable oxygen. Some unstable oxygen molecules (i.e., free radicals) enable the body to fight inflammation, kill bacteria, and help regulate the autonomic nervous system.
However, if produced in excess amounts (a situation referred to as oxidative stress), these radicals can become the mechanism of cell injury and subsequent cell death. Free radicals have been considered central to the damaging effects that can lead to degenerative conditions such as heart disease, cerebrovascular disease, diabetes mellitus, cataracts, Parkinson’s disease, premature aging, and cancer. In fact, research has shown that oxidative stresses caused by reactive oxygen species are factors in over 90% of lifestyle-related diseases.96
Reactive oxygen species or free radical formation occur as a result of many events such as prolonged exercise; exposure to high levels of oxygen, irradiation, ultraviolet or fluorescent light, pollutants, tobacco smoke, and pesticides (airborne or in food); drug overdose; heat stress; and the reperfusion injury that is induced by the restoration of normal blood flow after a period of ischemia such as occurs during organ transplantation or after MI.
Free radical toxicity may also be the underlying cause of degeneration of neurons located in the substantia nigra leading to the loss of dopamine necessary for the normal control of movements that produces the abnormal movements seen in Parkinson’s disease.24
Antioxidants.: Oxygen is the most common form of free radical in the human body, but the utilization of oxygen as a life-supporting mechanism means oxidative stress is an inescapable part of the human biologic system. The simultaneous presence of antioxidants is an adaptive response to help the body ward off the potentially harmful effects of oxygen and its derivatives, including free radicals.154
Antioxidants neutralize the extra free radicals and keep them from taking electrons from other molecules, resulting in cellular and DNA damage. A variety of enzymatic and nonenzymatic defense mechanisms are present within cells to perform the function of antioxidants detoxifying reactive oxygen species and protecting the cells from this type of injury. These are called endogenous antioxidants. Researchers are finding a variety of uses for natural antioxidants in combating the effects of aging and disease.
There are also exogenous antioxidants that can be taken from outside the body through our diet. Vitamin C, E, and beta-carotene are three important exogenous antioxidants. Over 200 antioxidants have been identified through food or plant substances. For example, vitamin E effectively scavenges several types of free radicals and other reactive species in lipid membranes and other lipid concentrations, making it a potentially effective antioxidant (able to neutralize the free radical before damage occurs) in preventing LDL cholesterol from adhering to the walls of arteries. In the case of the prostate, lycopene, the compound that makes tomatoes red, is a potent antioxidant potentially effective in promoting prostate health.51,79
Adequate folate intake has been shown to reduce the risk of breast cancer associated with alcohol consumption by providing bioactive compounds to counteract the formation of oxidative compounds.50,155
Multiple trials are ongoing to investigate oxidation and its effect on cellular injury, aging, and disease (e.g., cancer, heart disease, and cataracts) and the use of antioxidants found naturally in food and plants to combat oxidative stress, thereby preventing or possibly modifying diseases at the cellular level. Animal and human studies have confirmed that regular, moderate physical activity and exercise strengthen the antioxidant defense system, whereas intense or prolonged, strenuous exercise (especially in a person who has a sedentary lifestyle) constitutes an oxidative stress.40,116,119
Nitric Oxide.: The nitric oxide (NO) molecule is composed of one nitrogen atom and one oxygen atom. It is present in all mammals including humans and is one of the few gaseous signaling molecules known. NO should not be confused with nitrous oxide (N2O), a general anesthetic, or with nitrogen dioxide (NO2), which is a poisonous air pollutant.
The nitric oxide molecule is a free radical, which is relevant to understanding its high reactivity. NO is recognized as an important modulator of an enormous number of physiologic responses. Reduced NO bioavailability that is a result of oxidative stress seems to be the common molecular disorder causing many pathologic effects within the body.
NO assists in long-term memory. It also influences neuronal transmission by increasing the permeability of nerve endings, making acetylcholine transfer across the synapses easier. NO alters the ability of the gastrointestinal mucosa to resist injury induced by toxins, thereby influencing the immune system. NO inhibits virally induced cytokine and chemokine production, possibly combating the common cold.120 It also stimulates collagen synthesis for wound healing, modulates fracture healing, and is useful in the treatment of tendinopathy.107,108
NO is an antilipid that provides a nonstick coating to the lining of blood vessels, much like Teflon. These two effects have helped explain how NO might prevent heart attacks and strokes and why nitroglycerin works—nitroglycerin is converted to NO inside vascular tissue, where it relaxes smooth muscle in arteries and causes blood vessels to dilate. It also controls platelet function by preventing platelets from clumping together, thus preventing the formation of blood clots.
Researchers are studying the effect of NO on free radicals that cannot be stabilized or removed. Studies show that NO appears to play a role in exercise-induced dilation of blood vessels supplying cardiac and skeletal muscle. Exercise training enhances NO-mediated vasodilation. The exact mechanism is not clear yet, but a growing number of studies suggest that exercise training, perhaps via increased capacity for NO formation, retards atherosclerosis.85 There is also accumulating evidence that NO is involved in skeletal muscle glucose uptake during exercise.86
Exercise and Free Radicals.: Physical activity and exercise can have positive or negative effects on oxidative stress depending on training load, training specificity, and basal level of training. Oxidative stress seems to be involved in muscular fatigue and overtraining.43 Excessive exercise has been shown to induce DNA damage in peripheral leukocytes. Exhaustion of the leukocyte reactive oxygen species (ROS) may reduce the body’s ability to combat microbial invasions (i.e., infections) before the system has been restored.103 On the other hand, moderate stress in the form of regular exercise training may have protective effects against exercise-induced DNA damage.
Evidence is emerging to support a role for improved NO bioavailability with exercise training.128 Up-regulation of endogenous antioxidant defense systems and complex regulation of repair systems are seen in response to training and exercise. Up-regulation of antioxidants and modulation of the repair response may be mechanisms by which exercise can influence our health in a positive way.41
Regular, long-term aerobic exercise has been shown to reduce migraine pain severity, frequency, and duration, possibly a result of increased NO production.102
Psychosocial Factors
Psychosocial factors can have an impact on tissue adaptation, especially as related to tissue injury.89 Psychosocial factors (e.g., fear, tension, or anxiety) may influence individual threshold values for tissue adaptation and injury. Many studies have investigated the role of mechanical and psychosocial factors in the onset of musculoskeletal (and other regional) acute and chronic pain.121 For example, people who are only occasionally or never satisfied in their work settings or who describe their work as “monotonous” have a higher risk of injury than those who are satisfied or completely satisfied with support from supervisors and colleagues.11,57,58,100
Reversible Cell Injury
Alteration in a cell’s functional environment, either acute or chronic, produces a stress to the cell’s ability to attain or maintain homeostasis. The extent to which the cell is able to alter mechanisms and regain homeostasis in the altered environment is considered an adaptation by the cells or tissues. When the cell is unable to adapt, injury can occur. A sublethal or reversible injury occurs if the stress is sufficiently small in magnitude or short enough in duration that the cell is able to recover homeostasis after removal of the stress.
Cells react to injurious stimuli by changing their steady state to continue to function in a hazardous environment. Reversible (sublethal) injury caused by any of the mechanisms of cell injury listed in Box 6-1 is a transient impairment in the cell’s normal structure or function. Normal cell structure and function can return after removal of the stressor or injurious stimulus (Fig. 6-3).
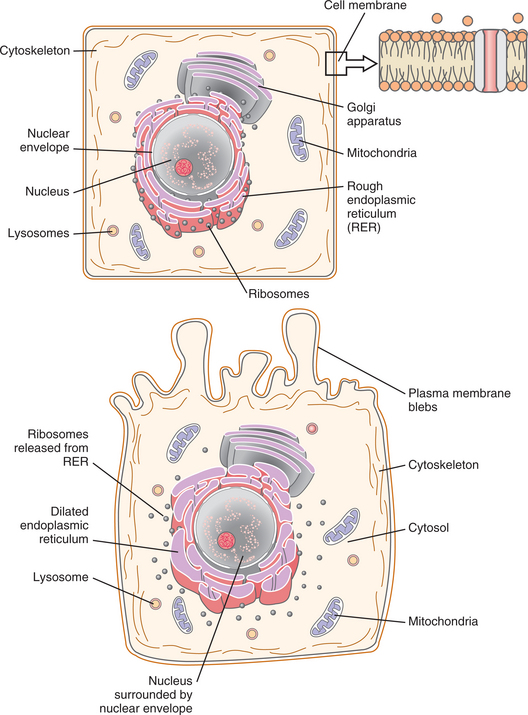
Figure 6-3 A, A normal cell with its organelles. B, Reversible cell injury with cellular swelling, accumulation of fluid in endoplasmic reticulum, and the release of ribosomes and formation of membrane blebs. (Courtesy SH Tepper, PhD, PT, Winchester, VA, Shenandoah University.)
Acute reversible injury causes an impairment of ion homeostasis within the cell and leads to increased intracellular levels of sodium and calcium. An influx of interstitial fluid into the cell accompanies these ionic shifts and causes increased cell volume (swelling). Swelling occurs within the cytosol (liquid medium of the cytoplasm) and within organelles such as mitochondria and the endoplasmic reticulum. Swollen mitochondria generate less energy. Thus, instead of oxidative ATP production, the cell reverts to less efficient anaerobic glycolysis, which results in excessive production of lactic acid. The pH of the cell becomes acidic, which slows down the cell metabolism, resulting in further cellular damage. The injured cell forms plasma membrane blebs that can seal off and detach from the cell surface. In severely injured cells, ribosomes detach from the rough endoplasmic reticulum (RER), and a decrease in the number of polysomes occurs. These changes lead to reduced protein synthesis by the affected cells and the cycle of damage can continue.
However, if the cell nucleus remains undamaged and the energy source is restored or the toxic injury is neutralized, the cell is able to recover and pump the ions and excess fluid back out. The swelling disappears, and the cell is returned to the original steady state, constituting a reversible cell injury.
Cellular Adaptations in Chronic Cell Injury
When a sublethal stress remains present over a period of time, stable alterations (adaptations) take place within the affected cells, tissues, and organs. Adaptation enables the cells to function in an altered environment and thereby avoid injury. Characteristics of cell adaptation, such as change in size, number, or function, increase the cell’s ability to survive. In many but not all cases, these changes benefit the function of the parent organ or structure within which the cell resides. These changes are potentially reversible; common cellular adaptations include atrophy, hypertrophy, hyperplasia, metaplasia, and dysplasia (Fig. 6-4).
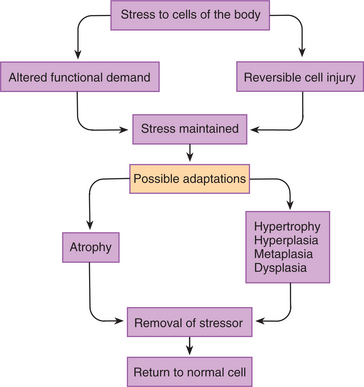
Figure 6-4 Cellular adaptations and reversible cell injury in response to stress. When the body is under persistent stress leading to either reversible cell injury or altered functional demand, the tissues adapt. Adaptations could include atrophy, hypertrophy, hyperplasia, metaplasia, or dysplasia. All of these changes are reversible with removal of the stressor. (Courtesy SH Tepper, PhD, PT, Winchester, VA, Shenandoah University.)
Atrophy is a reduction in cell and organ size. Atrophy can occur with vascular insufficiency, reduction in hormone levels, malnutrition, immobilization, pain limiting function, and chronic inflammation. Atrophy, involving the entire body, may occur with aging and is referred to as physiologic atrophy. Bone loss, thymus involution, muscle wasting, and brain cell loss are examples of either tissue or organ atrophy associated with aging. Pathologic atrophy occurs as a result of some of the mechanisms of cell injury, such as ischemia, inadequate nutrition, or physical factors, listed in Box 6-1. For example, ischemia of the viscera results in atrophied organs; cancer or malnutrition can result in cachexia, a general wasting of the body; and spinal cord injury results in atrophy of the affected muscles.
Hypertrophy is an increase in the size of the cell and organ. Hypertrophy can occur when increased functional demands are placed on the cells, tissue, or organs and with increased hormonal input (e.g., exercise stress can induce skeletal muscle hypertrophy). Pure hypertrophy only occurs in the heart and striated muscles because these organs consist of cells that cannot divide. Hypertrophy of the heart is a common pathologic finding that occurs as an adaptation of heart muscle to an increased workload. Specifically, hypertrophy of the left ventricle is a typical complication of hypertension. Increased blood pressure requires that the heart produce more force to eject the blood. The additional force is produced by hypertrophy of muscle fibers in the left ventricle.
Hyperplasia is an increase in the number of cells leading to increased organ size. Tissues can divide, and in the presence of excessive functional demands, these tissues increase in cell number. Pure hyperplasia typically occurs because of hormonal stimulation (e.g., prolonged estrogen exposure causes the endometrium of the uterus to become thick) or chronic stimulation (e.g., persistent pressure on the skin induces hyperplasia and the formation of a callus). Some hyperplasia has no discernible cause and may represent early neoplasia. Hypertrophy and hyperplasia often occur together such as in the case of prostate enlargement and obstruction of the urethra and bladder. The result is an increase in size and number of smooth muscle cells in the wall of the urinary bladder.
Metaplasia is a change in cell morphology and function resulting from the conversion of one adult cell type into another. For example, in smokers, portions of the respiratory tract change from ciliated pseudostratified columnar epithelium into stratified squamous epithelium, leading to a thickening of the respiratory epithelium and loss of the functional clearance of mucus and debris along the respiratory tree.
Dysplasia is an increase in cell numbers that is accompanied by altered cell morphology and loss of histologic organization. Considered to be a preneoplastic alteration, dysplasia can be found in areas that are chronically injured and undergoing hyperplasia or metaplasia.
Intracellular Accumulations or Storage
Intracellular accumulations are increases in the storage of lipids, proteins, carbohydrates, or pigments within the cell that occur as a result of an overload of various metabolites or exogenous material. These accumulations can also be caused by metabolic disturbances altering cell function. For example, when the liver is sublethally injured, lipid (triglyceride) accumulates within the hepatocyte. This lipid accumulation occurs when a reduction in protein synthesis occurs as a result of disaggregation of the ribosomes from the rough endoplasmic reticulum as previously discussed. Hepatocytes normally produce our endogenous lipoproteins.
With sublethal damage to hepatocytes (e.g., alcohol abuse), a lack of protein shell formation occurs so that lipoproteins cannot be packaged and transported to the plasma. As a result, lipids remain within the hepatocyte, causing the characteristic “fatty liver” found in alcoholics.
Irreversible Cell Injury
If the injurious or stressful stimulus is of sufficient magnitude or duration or if the cell is unable to adapt, the cell will be irreversibly injured. Irreversible cell injury is synonymous with cell death. Hallmarks of lethally injured cells include alterations in the cell nucleus, mitochondria, and lysosomes and the rupture of the cell membrane.
Damage to the nucleus can present in three forms: pyknosis, karyorrhexis, and karyolysis. Nuclei undergo clumping or pyknosis, which is a degeneration of the cell as the nucleus shrinks in size and the chromatin condenses to a solid mass. The pyknotic nuclei can fragment, a process termed karyorrhexis, or it can undergo dissolution (karyolysis).
Mitochondria lose their membrane potential and become unable to synthesize ATP, leaving the cell without the necessary energy production for cell function. Morphologically, irreversibly injured mitochondria appear swollen, contain large lipid-protein aggregates called flocculent densities, and may also contain dense crystalline deposits of calcium (Fig. 6-5).
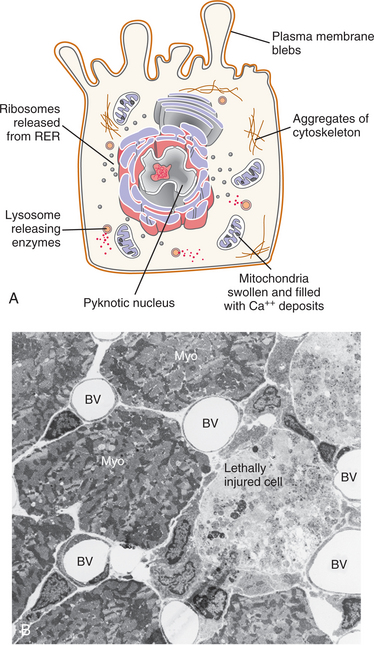
Figure 6-5 Irreversible cell injury: ultrastructural alterations in an irreversibly killed cell. A, Mitochondria are nonfunctional and filled with flocculent densities. Lysosomes are releasing their digestive enzymes. The nucleus is condensing upon itself (pyknosis). Membrane breakdown allows intracellular enzymes to be released into the interstitial area. B, Electron micrograph of lethally injured cardiomyocytes next to healthy viable cardiomyocytes (Myo). Note lethally injured cells to the right of the Myo are swollen, mitochondria are filled with flocculent densities, there is a loss of myofilaments, and mononuclear phagocytic cells are beginning to remove these dead cells. BV, Blood vessel. Original magnification ×1500. (A, Courtesy SH Tepper, PhD, PT, Winchester, VA, Shenandoah University; B, From Tepper SH, Anderson PA, Mergner WJ: Recovery of the heart following focal injury induced by dietary restriction of potassium, Path Res Prac 186(2):265-285, 1990.)
After cell death, lysosomes release their digestive enzymes within the cytoplasm of the cell, initiating enzymatic degradation of all cellular constituents, a process that may be aided by enzymes released from inflammatory cells. The active process of degradation of dead cells is called necrosis. Enzymes help dissolve the dead tissue, making it easier for phagocytic cells to remove the dead tissue in preparation for healing by repair (laying down of a collagenous tissue scar) or regeneration (regrowth of parenchymal tissue). Dead cells release their contents into the extracellular fluid, eventually making their way into the circulation, where they can be measured as clinically useful signs of cell injury. For example, levels of aspartate aminotransferase (AST), creatine kinase (CK), and lactate dehydrogenase (LDH) are typically elevated in the serum of people with myocardial infarct or viral hepatitis (see Tables 40-5, 40-15, and 40-16).
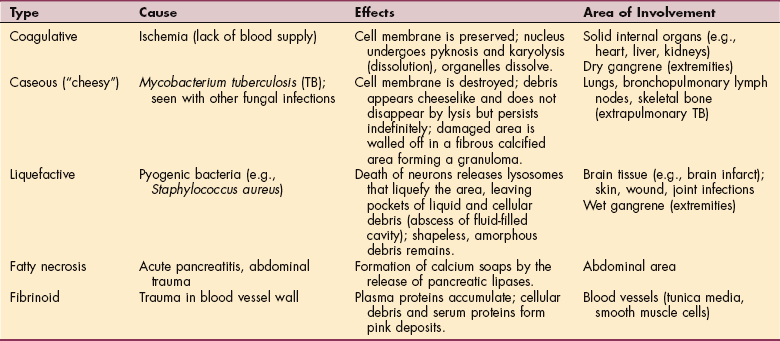
Types of Necrosis
Dead tissue becomes morphologically distinguishable from healthy tissue only after the process of necrosis begins with the dissolution of irreversibly injured cells within living tissue. Removal of this dead tissue is essential for healing to take place. Histologically, several different types of necrosis are recognized (Table 6-1) with some additional subcategories.
For example, gangrene caused by bacterial infection and associated with tissue ischemia (peripheral vascular disease) may form coagulative necrosis (dry gangrene) or liquefactive necrosis (wet gangrene). The fermentation reactions caused by certain bacterial pathogens may cause the formation of gas bubbles in the infected tissue. In muscle necrosis, one of the causative agents is Clostridium perfringens. The term used to describe this condition is clostridial myonecrosis or gas gangrene (see Chapter 8).
Pathologic Tissue Calcification
Calcification is the deposition of calcium salts, primarily calcium phosphate, in body tissues. Normally, 99% of all calcium is deposited in the teeth and bone matrix to ensure stability and strength; the remaining 1% is dissolved in body fluids, such as blood, or within skeletal muscle. Two types of pathologic calcification are evident. The first type is dystrophic calcification, the deposition of calcium salts in an area of damaged tissue. Classic examples of dystrophic calcification include tuberculosis and atherosclerosis. With tuberculosis, calcification occurs in the granulomas (accumulations of macrophages and connective tissue) that may be found in lymph nodes or in the lung parenchyma and may be seen on radiograph. In the case of atherosclerosis, vessels damaged by the deposition of cholesterol may become calcified. Calcifications within the vessel wall lead to a reduction in elasticity of the vessel.
Dystrophic calcification can occur in soft tissues such as the tendons. Calcifying tendinitis occurs in up to 20% of the adult population but is mostly asymptomatic. Women between the ages of 30 and 60 years old are affected most often but men can be affected, too. The shoulder is the primary site involved; calcific tendinitis is bilateral in approximately 25% to 30% of all cases. The etiopathology remains unknown but hydroxyapatite crystals are the main component of the calcifications within the tendons. Iontophoresis remains a common treatment for this condition along with antiinflammatory medications. Extracorporeal shockwave therapy has been used; surgery is a rare treatment approach.81
The second type of calcification is metastatic calcification. This type occurs with hypercalcemia (increased blood calcium levels) in living tissue. The normal absorption of calcium is facilitated by parathyroid hormone and vitamin D. When there are increased levels of parathyroid hormone in the blood (e.g., hyperparathyroidism), an increased accumulation of calcium in the pulmonary alveoli, renal tubules, thyroid gland, gastric mucosa, and arterial walls interferes with normal organ function.
TISSUE HEALING
The process of tissue healing begins soon after tissue injury or death and occurs either by regeneration (regrowth of original tissue) or by repair (formation of a connective tissue scar). The inflammatory cells that are recruited from the blood circulation begin the healing process by breaking down and removing the necrotic tissue. This is accomplished primarily by phagocytes that secrete degradative enzymes and also phagocytose the cellular debris, connective tissue fragments, and plasma proteins present in the dead tissue.
The healing process is complex and influenced by many components such as fibronectin, proteoglycans and elastin, collagen, and parenchymal and endothelial cells. In addition, there is a wide range of factors that affect tissue healing and must be taken into account during recovery and rehabilitation. Both the components and the factors that affect tissue healing are presented in this section, followed by a discussion of the multiphasic process of tissue healing and recovery.
Components of Tissue Healing
Fibronectin has numerous functions in wound healing, the most important of which are the formation of scaffold, the provision of tensile strength, and the ability to “glue” other substances and cells together. It is one of the earliest proteins to provide the structural support that stabilizes the healing tissue. Plasma proteins that leak from inflamed vessels are the first source of fibronectin for the healing tissue. Plasma-derived fibronectin binds to and stabilizes fibrin, a protein that makes up the blood clots that are present in the injured tissue.
Fibronectin binds together several types of proteins present in the extracellular matrix and can also bind to debris, such as DNA material derived from necrotic cells, thereby acting as an opsonin (molecule that acts as a binding enhancer to facilitate phagocytosis) during the breakdown of necrotic tissue. Fibronectin is also responsible for attracting fibroblasts and macrophages by chemotaxis to the healing tissue. The stimulated fibroblasts, in turn, secrete more fibronectin. Fibronectin binds to proteoglycans and collagens and this binding further stabilizes the healing tissue.
The importance of fibronectin can be seen as researchers seek to explain the lack of a functional healing response in the anterior cruciate ligament (ACL) after injury.154 Studies focusing on the signaling pathways and on binding to fibronectin for specific tissues, such as the ACL, may yield improved prevention and intervention strategies in the future.91,138,153
Proteoglycans and Elastin
Proteoglycans, proteins containing carbohydrate chains and sugars, are secreted in abundance by fibroblasts early during the tissue repair reaction. Proteoglycans bind to fibronectin and to collagen and help stabilize the tissue that is undergoing repair. Proteoglycans also retain water and aid in the hydration of the tissue being repaired. Once the tissue is healed, proteoglycans contribute to the organization and stability of collagen and create an electrical charge that gives basement membranes the property of functioning like molecular sieves. Fibroblasts also synthesize and secrete elastin, a protein that becomes cross-linked to form fibrils or long sheets that provide tissues with elasticity.
Collagen
Collagen is the most important protein to provide structural support and tensile strength for almost all tissues and organs of the body. The different types of collagen give stability to healing tissue; the word collagen is derived from the Greek and means glue producer. Collagen is a fibrous protein molecule consisting of three chains of amino acid coiled around each other in a triple helix (Fig. 6-7). Improved technology has made it possible to identify collagen types and measure protein turnover. It is the most abundant protein in the body; at least 27 collagen types have been identified.
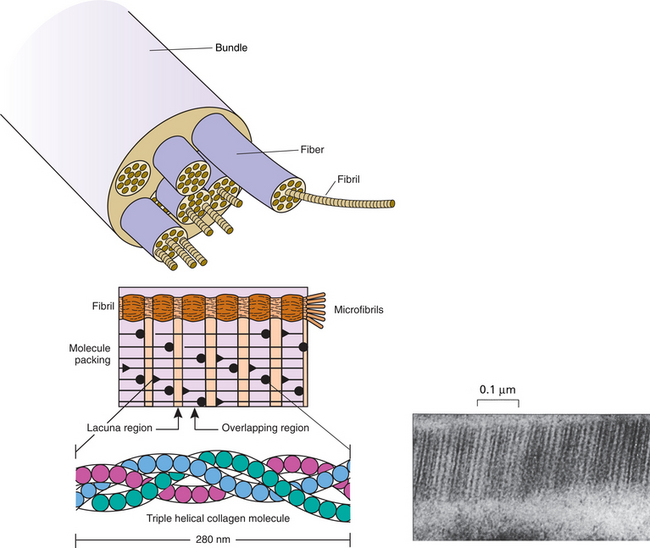
Figure 6-7 Structure of collagen. A, The collagen fiber is composed of fibrils, each of which is composed of microfibrils. B, The molecule itself consists of three polypeptide chains called alpha chains that wrap around each other in a triple helix. The helix is made possible because each third amino acid in the polypeptide chain is glycine. The molecules are quarter-staggered one to another, which ensures that no weak points occur across the fibril to prevent overload and slippage. C, Visualized by transmission electron microscopy, the individual collagen fibrils are seen to have two orders of banding. The larger bands result from the gaps between the individual molecules of collagen, which then overlap the adjacent molecules to form a strong bond. (From Bullough PG: Bullough and Vigorita’s orthopaedic pathology, ed 3, St Louis, 1997, Mosby.)
We know that exercise is a potent stimulus for protein synthesis in skeletal muscle. Collagen in the extracellular matrix of muscle and tendon is also sensitive to mechanical stimuli. Collagen does not appear to be nutritionally sensitive, which may contribute to the loss of muscle during aging. It is possible that the tissue is unable to respond adequately to increased availability of nutrients.150
Organization of Collagen.: Each collagen type has a specialized function (Table 6-2). The amino acid makeup of the collagen molecule and the manner in which the molecules are assembled together vary for each one of the collagen types. The differences in organization and composition account for the structural properties of each collagen type. For example, collagen organized in unidirectional or parallel bundles contributes to the strength of tendons. Collagen is the principal extracellular component of normal tendon.
Table 6-2
| Type | Location |
| Type I | Predominant structural collagen of the body; constitutes 80% to 85% of dermal collagen; prominent in mature scars, tendon, bone, and dentin; joints |
| Type II | Predominant component of hyaline cartilage (e.g., outer ear, end of nose, joint); not present in skin; found in nucleus pulposus external annulus |
| Type III | Prominent in vascular and visceral structures (e.g., blood vessels, gastrointestinal tract, liver, uterus) but absent in bone and tendon, constitutes 15% to 20% of dermal collagen, abundant in embryonic tissues, first collagen deposited in wound healing (granulation tissue) |
| Type IV | Found in basement membranes (base of epithelial, endothelial, and mesenchymal cells found in developing fetus), glomeruli of kidney nephron |
| Type V | Present in most tissues but never as a major component; prominent in fetal membrane, cornea, heart valve; minor component of skin; synovial membranes |
| Type VI | Prevalent in most connective tissues |
| Type VII | May be involved in matrix and bone disorders, anchoring filaments of lymphatic vessels and at dermal-epidermal junctions |
| Type VIII | Secreted by rapidly proliferating cells; found in basement membranes; may provide a molecular bridge between different types of matrix molecules |
| Type IX | Minor component in hyaline cartilage; vitreous humor (fluid of the eye) |
| Type X | Only formed in the epiphyseal growth plate cartilage; may have a role in angiogenesis; may be involved in matrix and bone disorders |
| Type XI | Hyaline cartilage |
| Type XII | Embryonic skin and tendon, periodontal ligament |
| Type XIII | Endothelial cells |
| Type XIV | Fetal skin and tendons; similar to type I |
| Types XV-XXVII | Identified but not clearly understood |
Collagen in random arrangement provides flexibility of the skin and rigidity of bone. When organized at right angles, collagen allows transmission of light in the cornea and vitreous. Collagen laid down in a tubular fashion contributes to the elasticity of the blood vessels.
Some collagen molecules are assembled into progressively thicker and stronger filamentous structures, allowing the molecules to become cross-linked. These cross-links impart tensile strength to collagen fibers and prevent slippage of molecules past one another when under tension. The structural stability of the extracellular matrix is primarily a consequence of collagen and the extent of cross-linking.23
Types of Collagen.: Type I collagen, the most common form, is assembled as a thick bundle that is structurally very strong and can be found in all body tissues, where it forms bundles together with other collagen types. Type I collagen is the main component of mature scars and is also predominant in strong tissues such as tendons and bones.
Type II collagen is assembled into thin supporting filaments and is the predominant collagen type found in cartilaginous tissue. Type II fibers of the external annulus have a half-life of about 3 months. This allows maintenance of the nutritive exchanges between degenerative external annulus and any healthy remaining tissue, possibly delaying or avoiding further degeneration.48
Type III collagen is assembled into thin filaments that make tissues strong but supple and elastic. It contains interchain disulfide bonds or bridges not found in type I or II and is the collagen type first deposited in wound healing (i.e., fresh scars). This type of highly soluble collagen accounts in part for the plasticity of skin and blood vessels. Overexposure to the sun speeds up the breakdown of collagen and elastin, two proteins that give skin its strength and resilience, thus contributing to the development of skin wrinkling.
Type III collagen is more prevalent in newborns; with each passing decade, collagen-producing cells make less soluble collagen and progressively convert to synthesizing an insoluble, more stable type I collagen. The changing ratio of collagen types I and III throughout the body is so reliable that chronologic age can be determined by analyzing the collagen type III content of a skin sample.7
During the initial stages of tissue repair, fibroblasts secrete large amounts of type III collagen, which provides support for the developing capillaries. Within a few days after the tissue injury, type III collagen is degraded by enzymes secreted by fibroblasts and other cells and is replaced by newly synthesized type I collagen. Type I collagen enhances wound tensile strength and is the main component of the scar tissue that remains after repair is completed. Type IV collagen is not assembled into fibers. Together with other proteins, it forms the basement membrane to which epithelial, endothelial, and certain mesenchymal cells are anchored.
Mutations in the genes for collagen cause a wide spectrum of diseases of bone, cartilage, and blood vessels, including osteogenesis imperfecta, a variety of chondrodysplasias, Alport syndrome, the Ehlers-Danlos syndrome, and more rarely, some forms of osteoporosis, osteoarthritis, and familial aneurysms. Scientists are finding that aberrant collagen cross-linking and increased collagen synthesis are present in some malignancies,12 whereas the presence of free radical scavengers inhibits the rate of collagen formation.93
When either collagen or elastin becomes resorbed, elements are released into blood and concentrate in urine. Determining the presence of these components in tissues and body fluids provides important markers in the clinical investigation of various diseases.133 Methods to quantify the number of collagen cross-links in tissue are also being further developed at this time.112,131,147
Differences in collagen fibril diameter have been demonstrated in people with unidirectional (anterior) shoulder instability versus multidirectional instability. Smaller collagen diameters in the multidirectional instability group suggest the possibility of an underlying connective tissue abnormality.127
Factors That Affect Tissue Healing
Many variables regulate or affect the healing process and either facilitate, inhibit, or delay wound healing (Box 6-4). Since local blood supply is vital to the delivery of the materials necessary for wound healing, factors that impede local circulation or depletion of the necessary materials could delay rehabilitation. Certain tissues (e.g., tendons, ligaments, cartilage, disk) have a decreased blood supply, thus the healing process may require additional time.
Growth Factors
The cells involved in the tissue repair response produce proteins called growth factors that regulate a number of cellular reactions involved in healing. Growth factors regulate cell proliferation, differentiation, and migration; biosynthesis and degradation of proteins; and angiogenesis. Through all of these varying functions, growth factors integrate the inflammatory events with the reparative processes. When these complex mechanisms are disturbed, the result can be delayed healing and an inferior scar (hypotrophic) or elevated levels of growth factor, resulting in hypertrophic scarring such as occurs after a burn injury or in the formation of keloids.117,144
Growth factors act by binding to receptors on the plasma membranes of specific cells and have a stimulatory or inhibitory effect on these cells. This binding initiates a process of transmembrane signaling that results in the phosphorylation of proteins (the process of attaching a phosphate group to the protein). These steps lead to the activation of gene expression and DNA synthesis in the cell.
The signals that turn on proliferation of normal cells and cause tissue healing are also responsible for turning on proliferation of cancer cells. With continued growth of neoplastic cells, a neoplasm or tumor may occur. The significant difference between the healing process and cancer is that the growth of the cancer cells goes on unchecked. These analogies have led to the designation of cancers as wounds that do not heal.
Platelets, endothelial cells, fibroblasts, macrophages, and cytokines are important sources of growth factors. Two important growth factors are platelet-derived growth factor (PDGF), which activates fibroblasts and macrophages, and fibroblast growth factor (FGF), which stimulates endothelial cells to form new blood vessels. An example of a growth factor that inhibits cell growth and inactivates macrophages is transforming growth factor–β (TGF–β).
Several growth factors (e.g., recombinant human PDGF-BB and granulocyte colony-stimulating factor) are being tested clinically to establish if these can boost the healing process in people who have deficiencies in wound healing (e.g., diabetic lower extremity ulcers).36,99
PDGF-BB has been approved by the FDA for the treatment of neuropathic ulcers when there is adequate blood supply.110 Wound dressings of the future may include several growth factors, each with a specific function. The application of topically active growth factors to chronic ulcers remains in the experimental phase.31,149 Efforts to improve methods of delivering growth factors are also under investigation.110
Finally, it should be mentioned that cytokines, such as IL-1, IL-2, IL-15, and TNF, can also regulate some aspects of the healing response. Some ILs have been identified as T-cell growth factors with proinflammatory properties or the transforming growth factor associated with hypertrophic scarring. Further studies are necessary to clarify the mechanism of cytokine release in normal postoperative wounds before therapeutic use can be developed.67
Nutrition
Nutrition is an important factor influencing healing. Adequate nutritional intake is necessary to support the active metabolism of cells involved in repair. Trauma, including surgery, infections, or large draining wounds, often increases the systemic rate of protein catabolism (loss). This has adverse effects on the synthesis of proteins required for healing. Inadequate intake of specific nutritional factors can specifically affect collagen production and remodeling. Examples are vitamin C deficiency, which causes defective collagen molecules to form, and deficiency of zinc.
Zinc is essential for the activity of enzymes that degrade collagen and of enzymes that are responsible ultimately for the induction of protein synthesis. Zinc deficiency therefore impairs healing. People with cancer often manifest delayed healing because of poor nutritional status often associated with the cancer process or the medical treatment (e.g., chemotherapy); particularly notable is the poor healing in tissues that have been subjected to radiation therapy. For an excellent source of information related to nutrition and healing in the therapist’s practice, see Nutrition Applied to Injury Rehabilitation and Sports Medicine.16
Other Factors
Other factors that influence healing include vascular supply, presence of infection, immune reaction, client’s age, and the presence of other medical conditions referred to as comorbidities. Healing is often adversely affected in people who smoke, who are immunosuppressed, or who have other compromising medical conditions. For example, incontinence, peripheral vascular disease, confusion associated with dementia or Alzheimer’s disease, or other neurologic impairment can contribute to delayed wound healing.
Diseases associated with decreased oxygen (tissue) perfusion (e.g., anemia, congestive heart failure, chronic obstructive pulmonary disease [COPD], or diabetes mellitus) can also delay healing. Diabetes mellitus is associated with poor healing; one of the causes appears to be impaired function of phagocytic cells and another is a defect in granulation tissue formation.27
Medications can directly affect healing, especially the prolonged use of corticosteroids, or undergoing chemotherapy or radiation treatment. Anyone taking prednisone or other corticosteroids may be at risk, as steroids are well known to impair the healing process by inhibiting the inflammatory response necessary for tissue regeneration or repair.
An adequate vascular supply is critical to provide oxygen and nutrients to support healing. Vascular insufficiency, particularly in the lower limbs, is an important cause of slow-healing or nonhealing wounds. When blood return is not normal, a buildup of fluid can occur, reducing the body’s ability to supply nutrients and oxygen to the wound site.
Infection interferes with healing by inciting a severe and prolonged inflammatory reaction that can increase tissue damage. Certain microorganisms can also release toxins that directly cause tissue necrosis and lysis. Foreign bodies may retard healing by inducing a chronic inflammatory reaction, by interfering with closure of a tissue defect, and by providing a site protected from leukocytes and antibiotics where bacteria can multiply.
It may be necessary to offload weight-bearing surfaces to relieve pressure on the wound and surrounding area. Immobility, lack of desire to exercise or follow a plan of care, and refusal to change dietary or other lifestyle behaviors contributing to poor wound healing must also be considered.
Healing may be delayed or inhibited for individuals who are in a constant state of survival or sympathetic nervous system (SNS) stimulation. When the SNS is locked in a hyperactive mode, exaggerated responses to relatively minor stimuli cause the body to work against itself for healing and recovery. Concepts discussed in Chapter 3 can be applied with these individuals to “reset” the system and facilitate forward movement in the healing process.
PHASES OF HEALING
Acute wounds caused by trauma or surgery usually heal according to a well-defined process that has the following four phases that overlap each other and can take months to years to complete6:
Hemostasis and Degeneration
When tissue injury occurs, hemostasis is the first step. Hemostasis occurs immediately after an acute injury as the body tries to stop the bleeding by initiating coagulation. Blood fills the gap, and the coagulation cascade commences immediately, clumping platelets together to form a loose clot. Platelets release chemical messengers, including growth factors that summon inflammatory cells to the wounded tissue. Growth factors stimulate proliferation and migration of epithelial cells, fibroblasts, and vascular endothelial cells. Growth factors also regulate the differentiation of cells such as expression of extracellular matrix proteins.6
The inflammatory process described in detail in the next section begins right away, bringing fluid to the area to dilute harmful substances and support infection-fighting and scavenger cells (neutrophils and macrophages). Some sources describe this first phase as degeneration and inflammation.
The degeneration phase is characterized by the formation of a hematoma, necrosis of dead cells, and as mentioned, the start of the inflammatory cell response. After the removal of the dead tissue, the healing process undertakes the repair of the tissue defect that remains. Tissue repair begins within 24 hours of the injury with the migration of fibroblasts from the margins of the viable tissue into the defect caused by the injury. The fibroblasts proliferate and synthesize and secrete proteins such as fibronectin, various proteoglycans and elastin, and several types of collagen. The function of these proteins is to reconstitute the extracellular matrix and provide a scaffolding-like framework for the developing endothelial and parenchymal cells.
It is at this point that proliferation and migration occur as epidermal skin cells in the top layer move down the sides of the wound to help fill in the gap. Fibroblasts move in from the dermis, and new blood vessels form to create granulation tissue, which later becomes scar tissue. The next phase of remodeling eventually progresses into the final maturation phase as the regenerated tissue reorganizes into healthy scar tissue.
But we have just jumped ahead to tell you the “rest of the story” by discussing proliferation and migration before describing the inflammatory process. Since the phases of tissues healing overlap, it is difficult to describe the process from start to finish without interrupting the discussion.
Inflammation
Inflammation serves a vital role in the healing process. Inflammation has both protective and curative features. Every step serves a specific purpose and is necessary as the body responds to tissue injury or damage. The ultimate goal of the inflammatory process is to replace injured tissue with healthy regenerated tissue, a fibrous scar, or both.8
The inflammatory phase begins once the blood clot forms. Vasodilation and increased capillary permeability activate the movement of various cells, such as polymorphonuclear leukocytes and macrophages, to the wound site. These cells destroy bacteria; release proteases, such as elastase and collagenase; and secrete additional growth factors.
Growth factors, cytokines, and chemokines are the key molecular bioregulators of the inflammatory phase of tissue healing. The functions of these three bioregulators overlap considerably. About 5 days after injury, fibroblasts, epithelial cells, and vascular endothelial cells move into the wound to form granulation tissue. This newly developing tissue is not strong, so there is a higher risk of wound dehiscence during this time.6
In contrast to cell injury, which occurs at the level of single cells, inflammation is the coordinated reaction of body tissues to cell injury and cell death that involves vascular, humoral, neurologic, and cellular responses. Regardless of the type of cell injury or death, the inflammatory response follows a basically similar pattern. As a result of all of these factors, inflammation occurs only in living organisms.
The functions of the inflammatory reaction are to inactivate the injurious agent, to break down and remove the dead cells, and to initiate the healing of tissue. The key components of the inflammatory reaction are as follows:
• Connective or interstitial tissue cells (fibroblasts, mast cells, and resident macrophages)
• Chemical mediators derived from inflammatory cells or plasma cells
• Specific extracellular matrix constituents, primarily collagen and basement membranes
Basement membranes are thin, sheetlike structures deposited by endothelial (cells that line the heart, blood vessels, lymph vessels, and serous body cavities) and epithelial cells (cells that cover the body and viscera) but are also found surrounding nerve and muscle cells. They provide mechanical support for resident cells and function as a scaffold for accurate regeneration of preexisting structures of tissue. Basement membrane tissue also serves as a semipermeable filtration barrier for macromolecules in organs, such as the kidney and the placenta, and act as regulators of cell attachment, migration, and differentiation. The major constituents are collagen type IV and proteoglycans.
Inflammation of sudden onset and short duration is referred to as acute inflammation, whereas inflammation that does not resolve but persists over time is called chronic inflammation. Although inflammation has been linked with many other conditions (e.g., Alzheimer’s, cardiovascular disease, cancer, diabetes, insulin resistance syndrome, and obesity), the focus of this chapter is inflammation and the musculoskeletal system.
Acute Inflammation
Normally, inflammation has a protective role and is generally beneficial to the body. However, inflammation, whether in the acute or chronic stage (and with all of its components), can be detrimental, causing damage and even death to adjacent healthy tissue.
In the acute stage, the inflammatory stimulus acts on blood cells and plasma constituents. Chemical mediators are produced that alter vascular tone and permeability. These mediators also cause the accumulation of plasma proteins, fluid (edema), and blood cells in the injured site (Fig. 6-8).
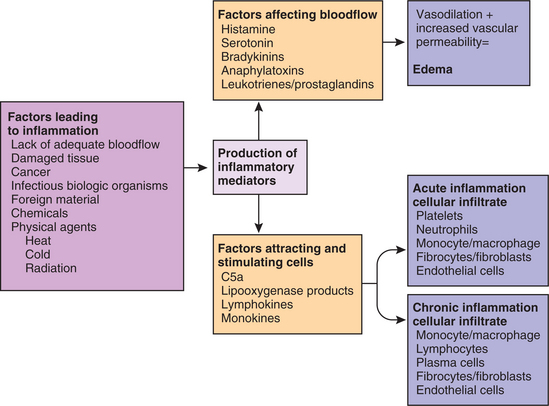
Figure 6-8 Contributing factors and components of inflammation. Note the vascular alterations associated with factors affecting blood flow (vasoactive mediators) leading to edema and the factors attracting and stimulating cellular alterations (chemotactic factors) resulting in acute (and sometimes) chronic inflammation. (Courtesy SH Tepper, PhD, PT, Winchester, VA, Shenandoah University.)
The clinical manifestations of this inflammatory reaction are redness, swelling, increased temperature, pain, and decreased function of the affected site (Table 6-3). Arteriolar constriction followed by vasodilation gives rise to the redness and heat. The exudation and leukocyte infiltration give rise to the swelling. Pain and loss of function occur as a result of the increased pressure from the edema on the peripheral nerves.8
Table 6-3
Four Cardinal Signs/Symptoms of Inflammation
| Sign | Precipitating Events |
| Erythema | Vasodilation and increased blood flow |
| Heat | Vasodilation and increased blood flow |
| Edema | Fluid and cells leaking from local blood vessels into the extravascular spaces |
| Pain | Direct trauma; chemical mediation by bradykinins, histamines, serotonin; internal pressure secondary to edema; swelling of the nerve endings |
Accompanying clinical findings include increased muscle tone or spasm and loss of motion or function. Cyriax describes two components of passive movement testing that also suggest acute inflammation: a spasm end feel and pain reported before resistance is noted by the practitioner as the limb is moved passively.25 If movement testing suggestive of acute inflammation persists, inflammation can become chronic with proliferation of blood vessels and connective tissue components.
In the normal, healthy individual, symptoms may be more intense because the body is vital and capable of healing quickly. Conversely, immunocompromised individuals and especially older adults with multiple comorbidities often require much longer time to heal. The symptoms may be less in intensity, but healing and repair are often delayed and chronic inflammation may occur.
There are three primary outcomes of acute inflammation: (1) complete resolution with restoration of normal tissue structure, (2) healing with scar formation, and (3) chronic fibrosis. Complete resolution usually occurs after mild trauma and minimal tissue damage. Healing with scar formation occurs after substantial tissue destruction in tissues with little capacity for regeneration or after prolonged edema. The soft tissue structures of the musculoskeletal system are often characterized by this result.
Chronic Inflammation
As described, acute inflammation follows injury. Once the injurious agent is removed, acute inflammation subsides. If little necrosis is present and replacement of lost parenchymal cells is possible, restitution of normal structure and function of the tissue occurs. In the presence of extensive necrosis or if regeneration of parenchymal cells is not possible (e.g., heart, CNS, or peripheral nervous system cells), the inflammatory reaction can become chronic. Chronic inflammation also develops if the underlying cause is not addressed and the injurious agent persists for a prolonged period. Repeated episodes of acute inflammation in the same tissue over time or low-grade, persistent immune reactions can also result in a chronic inflammatory response (Fig. 6-9).
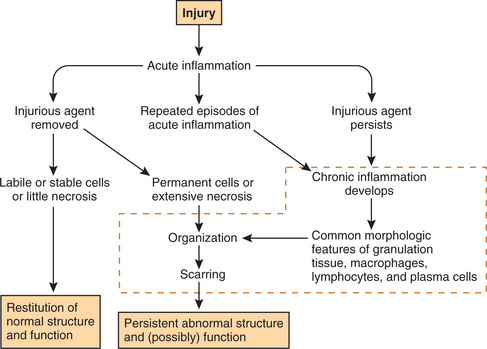
Figure 6-9 Overview after tissue injury: acute inflammation, chronic inflammation, and the likely healing process. (Courtesy SH Tepper, PhD, PT, Winchester, VA, Shenandoah University.)
The hallmark of chronic inflammation in a tissue is the accumulation of macrophages, lymphocytes, and plasma cells (see Fig. 6-8). The macrophage accumulation is the result of chemotaxis of monocytes (precursors to macrophages) to the area of injury. Macrophages modulate lymphocyte functions and promote growth of endothelial cells and fibroblasts by the release of growth factors. Eosinophils may also be present, particularly if allergic reactions or parasite invasions are involved.
Granulation tissue made up of proliferating endothelial cells and fibroblasts is also seen in areas of chronic inflammation. Granulation tissue can be seen in well-healing, open wounds. Inspection of the wound site reveals red “beefy” tissue with pinpoint red dots (new capillaries) and a granular surface composed of newly formed collagen.
Certain diseases cause the formation of a specific type of chronic inflammation called a granuloma. The granuloma is a microscopic (less than 2 mm in diameter) aggregate of macrophages often surrounded by lymphocytes. Most of the macrophages are flattened “epithelioid” in appearance and some may fuse together, giving rise to large cells with multiple nuclei (Langerhans and foreign body giant cells).
The presence of granulomatous inflammation is clinically important because it aids in the diagnosis of the injurious stimulus. Tuberculosis, a disease caused by Mycobacterium tuberculosis, classically causes granulomas or tubercles in this condition with a central focus of caseous necrosis. The presence of a foreign body (e.g., a suture) is another common cause of granulomatous inflammation.
Chronic inflammation can contribute to the healing of injured tissue but usually without a full return of function. The proliferation of endothelial cells reconstitutes the vasculature in the injured tissue, whereas proliferation of fibroblasts and the production of collagens and proteoglycans (polymers that form the gel between collagen fibrils) reconstitute the extracellular matrix. Together, these constituents make up the granulation tissue and lead to the formation of a connective tissue scar. This process is regulated by growth factors derived from macrophages, platelets, and plasma.
Components of the Inflammatory Reaction
Vascular Alterations.: Acute inflammation can last from a few minutes (e.g., redness and swelling from scratching your skin) to a few days (e.g., after an open cut on the finger), during which time a series of vascular events occurs. After an injury that disrupts the integrity of a vessel wall, the small arteries supplying blood to the area undergo vasoconstriction. This is mediated by a neural reflex and results in a slowing down of blood flow to the affected area.
At the same time, the blood flowing into the surrounding tissue exerts pressure on the damaged vessels, compressing them from outside. The slowdown of the blood flow promotes aggregation of platelets, which leads to the formation of a blood clot, resulting in a reduction in the amount of blood loss.
In the case of an injury that does not disrupt the integrity of the blood vessel wall but does cause tissue injury, the temporary neurally mediated constriction of arterioles is followed by a more sustained and overriding vasodilation, resulting in an increased blood flow to the affected area. The increased blood volume raises hydrostatic pressure, and an increased loss of protein-poor fluid occurs from the vasculature into the injured tissue.
At this stage, clinical manifestations include redness (erythema) and warmth of the injured area due to the increased blood flow. The leakage of protein-poor fluid (transudate) from the vasculature into the interstitial spaces is called transudation and causes the affected area to appear swollen (Fig. 6-10).
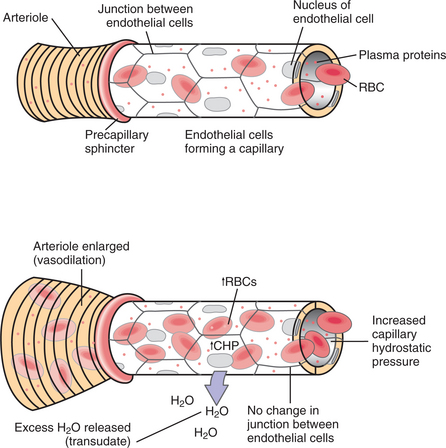
Figure 6-10 Normal capillary (A) reveals endothelial cells connected by tight junctions limiting flow of plasma proteins into the interstitium. With mild injury (B) vasodilation results in increased capillary hydrostatic pressure pushing more water into the interstitial area (transudate). (Courtesy SH Tepper, PhD, PT, Winchester, VA, Shenandoah University.)
Transudation, the passage of fluid through a membrane or tissue surface, occurs as a result of a difference in hydrostatic pressure, primarily in conditions in which there is protein loss and low protein content (e.g., left ventricular failure, cirrhosis, nephrosis). Typically, transudate is thin and watery, containing few blood vessels or other large proteins. The terms transudate, exudate, effusion, and edema are often used interchangeably, although each of these has its own clinical significance. When fluid transudates or leaks from blood vessels and accumulates inside an anatomic space, such as the pleural, pericardial, or peritoneal cavities or the joint space, these accumulations are called effusions.
Effusion is a more general term referring to the escape of a fluid and can either be a transudate or an exudate. Exudates occur when an increase in capillary permeability allows proteinaceous fluid and/or cells to leak out primarily through openings created between adjacent endothelial cells in the capillaries or venules (Fig. 6-11).
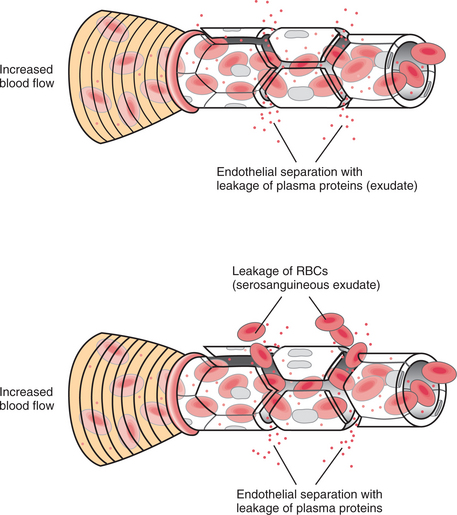
Figure 6-11 A, With a more severe inflammatory response, endothelial cells separate, causing leakage of plasma proteins into the interstitium (exudate). This accentuates the edema. B, With damage to the endothelial cells, the separation between the damaged cells may allow even erythrocytes to extravasate (serosanguineous exudate). (Courtesy SH Tepper, PhD, PT, Winchester, VA, Shenandoah University.)
Exudate contains much more protein than transudate. Exudate may also contain inflammatory phagocytic cells that occur in response to necrotic tissue and/or an infection. Protein-rich fibrinous (stringy) material found within some blisters or pus are sometimes identified as exudates. Various types of exudate are evident, depending on the stage of inflammation and its cause (Table 6-4).
Table 6-4
| Type | Appearance | Significance |
| Hemorrhagic; sanguineous | Bright red or bloody; presence of RBCs | Small amounts expected after surgery or trauma. Large amounts may indicate hemorrhage. Sudden large amounts of dark red blood may indicate a draining hematoma. |
| Serosanguineous | Blood-tinged yellow or pink; presence of RBCs | Expected for 48 to 72 hours after injury or trauma to the microvasculature. A sudden increase may precede wound dehiscence (rupture or separation). |
| Serous | Thin, clear yellow, or straw-colored; contains albumin and immunoglobulins | Occurs in the early stages of most inflammations; common with blisters, joint effusion with rheumatoid arthritis, viral infections (e.g., skin vesicles caused by herpesvirus); expected for up to 1 week after trauma or surgery. A sudden increase may indicate a draining seroma (pocket of serum within tissue or organ). |
| Purulent | Viscous, cloudy, pus; cellular debris from necrotic cells and dying neutrophils (PMNs) | Usually caused by pus-forming bacteria (streptococci, staphylococci) and indicates infection. May drain suddenly from an abscess (boil). |
| Catarrhal | Thin, clear mucus | Seen with inflammatory process within mucous membranes (e.g., upper respiratory infection). |
| Fibrinous | Thin, usually clear; may be yellow or pink, tinged, or cloudy | Occurs with severe inflammation or bacterial infections (e.g., strep throat, pneumonia); does not resolve easily; can cause fibrous scarring and restriction (e.g., constrictive pericarditis). |
RBCs, Red blood cells; PMNs, polymorphonuclear neutrophils.
Modified from Black J: Wound healing. In Black JM, Matassarin-Jacobs E, eds: Luckmann and Sorensen’s medical-surgical nursing, ed 5, Philadelphia, 2000, WB Saunders.
Removal of the fluid for analysis is required when differentiating between transudates and exudates and helps establish a specific diagnosis. Sometimes exudates are described by visual appearance (e.g., serosanguineous exudate, a fluid containing erythrocytes or red blood cells [RBCs]).
The time of onset of the vascular reaction to injury varies. Mild injuries may induce an increase in vascular permeability that occurs very soon after injury and resolves in a few minutes. In this case, the anatomic site responsible for the leak is the capillary/venule.88 The leak occurs because endothelial cells lining the lumen of the capillary/venules actively contract and open up their intercellular junctions. This increase in vascular permeability allows proteins to shift from the plasma into the interstitium, causing a greater attraction and retention of fluid in this area.
The increase in vascular permeability caused by the injury may be delayed for some time and may persist for days such as the delayed reaction seen in tissue injury caused by ultraviolet light (sunburn) or irradiation (radiation therapy); typically, the vascular leak begins a few hours after exposure to the sun. In severely injured tissues (e.g., trauma or extensive burns), all vascular structures may be directly injured and become leaky instantly.
Leukocyte Accumulations.: An important consequence of the exudation of protein and fluid from the vasculature is the engorgement of vessels with blood cells. This causes a slowing or cessation of blood flow in the affected vessels, a phenomenon called stasis. During stasis, the leukocytes (white blood cells [WBCs]) accumulate and adhere to the endothelial cells of blood vessel walls at the site of injury in a process called margination. Inflammatory mediators cause an increased expression of specific glycoproteins called adhesion molecules on the surface membrane of leukocytes and endothelial cells. These adhesion glycoproteins, by adhering to each other, function as receptors and counterreceptors. The adhesion glycoproteins are the glue that binds the leukocytes to each other and to the endothelium of venules and capillaries.
The binding of leukocytes to receptors on endothelial cells of venules is the first step in the migration of leukocytes from the vasculature to the interstitial tissues. This process initiates the circulation of leukocytes through the extravascular space in normal conditions and the infiltration of leukocytes into the site of inflammation.
In the next stage, the leukocytes actively migrate out of the vessels, passing through the vascular walls without damaging the blood vessels and entering the interstitial space in a process called diapedesis, or oozing (Fig. 6-12). The continued migration of leukocytes in interstitial space is directed by a chemical trail created by a concentration gradient of one of many possible attractants. The attractants are called chemotactic agents, and the process of locomotion is called chemotaxis. In other words, leukocytes are attracted to and accumulate at the site of an inflammatory reaction in response to a chemical stimulus.
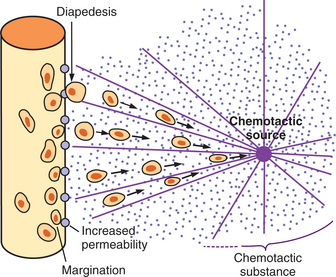
Figure 6-12 Many different chemical substances in the tissues cause both neutrophils and macrophages to move through the capillary pores in a process called diapedesis and toward the area of tissue damage by chemotaxis. Chemotaxis depends on the concentration gradient of the chemotactic substance. The concentration is greatest near the source, which directs the unidirectional movement of the white blood cells. Chemotaxis is effective up to 100 μm away from an inflamed tissue. Since almost no tissue area is more than 50 μm away from a capillary, the chemotactic signal can easily move vast numbers of WBCs from the capillaries into the inflamed area. (From Guyton AC, Hall JE: Textbook of medical physiology, ed 11, Philadelphia, 2006, WB Saunders.)
The presence of leukocyte accumulations in tissue or fluid specimens is diagnostic of an inflammatory process. The predominant cell type found in a specimen identifies the type of inflammation and/or its duration and original stimulus (see Table 40-11). Typically, during acute inflammation, neutrophils predominate (neutrophilia). Neutrophils inhibit bacterial growth by releasing lactoferrin, a protein that binds with iron, thus preventing microorganisms from using iron for growth and development. Neutrophils also demonstrate direct cytotoxic activity toward viruses, fungi, and bacteria by releasing defensin, which are peptides with natural antibiotic activity.
If the inflammatory stimulus subsides, the neutrophils rapidly die out because their lifespan (after extrusion from the circulation) is approximately 24 hours; they are replaced by monocytic/macrophage cells responsible for cleaning up the cellular debris left after neutrophils have done their job. Certain inflammatory stimuli can induce a sustained neutrophil response (e.g., first defense against pyogenic bacteria), a predominantly lymphocytic response (e.g., fight tumor cells or respond to viruses), or an eosinophilic response (e.g., plays a role in asthma and allergies or attacks parasites).
In addition to the types of WBCs present, the total and differential counts of the leukocytes in the circulating blood are also very important diagnostic tools (see Table 40-11). An increased number of circulating leukocytes (leukocytosis) is often an indication of an active inflammatory reaction (typically to an infection or tissue injury). A decreased WBC count (leukopenia) can, for example, be seen in certain types of infections and is an indicator of grave prognosis in severe systemic infections (sepsis).
The main function of the leukocytes recruited to the affected tissue is to remove or eliminate the injurious stimulus. Leukocytes achieve this function by releasing enzymes and toxic substances that kill, inactivate, and degrade microbial agents, foreign antigens, or necrotic tissue. Leukocytes also take up these materials by phagocytosis and release growth factors necessary for healing or regeneration (see Fig. 6-17).
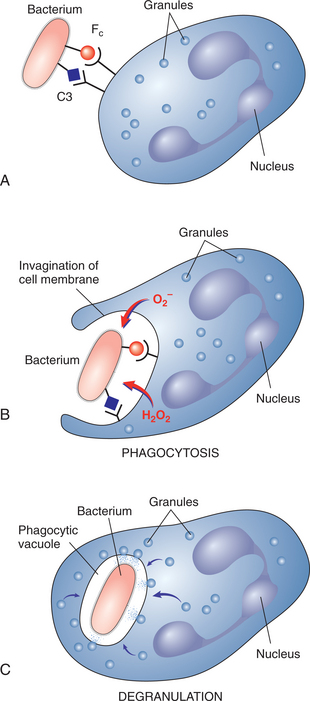
Figure 6-17 Phagocytosis of bacteria. A, The bacterium that was opsonized (coated with IgG and complement [C3]) binds to the Fc and complement receptors on the surface of the leukocytes. B, Engulfment of the bacterium into an invagination of surface membrane is associated with an oxygen burst and formation of oxygen radicals that are bactericidal and thus kill the bacterium. C, Inclusion of the bacterium into a phagocytic vacuole is associated with the fusion of the vacuole with lysosomes and specific granules of the leukocyte. The contents of the lysosomes and specific granules are bactericidal and contribute to final inactivation and degradation of the bacterium. The cytoplasm of the leukocyte becomes devoid of granules in a process referred to as degranulation of leukocytes. (From Damjanov I: Pathology for the health-related professions, ed 3, Philadelphia, 2006, WB Saunders.)
In addition to the role played by blood vessels in inflammation, a contribution is made from a system of thin-walled channels formed by endothelial cells with loose junctions. These channels are called the lymphatics and ultimately drain into the subclavian vein via the thoracic duct (see Fig. 13-8). These channels in physiologic conditions help drain fluid and protein from the interstitium, thereby reducing edema. They also serve as a conduit for the removal of certain leukocytes and inflammatory stimuli.141
The movement of the phagocytic cells into the lymphatic vessels allows presentation of the engulfed material to immunocompetent cells located in the lymph nodes. Hyperplasia of immunocompetent cells (T and B lymphocytes) in the lymph nodes leads to an enlargement of the nodes called lymphadenopathy. During the process of removing infectious agents, lymphatics and their lymph nodes may become actively inflamed. Clinically, the inflamed lymphatics may appear as red streaks under the epidermis and may be painful to palpation; this condition is called lymphangitis (see Chapter 13).
Chemical Mediators of Inflammation.: A large number of chemical mediators are responsible for the vascular and leukocyte responses generated by the cells involved in an acute inflammatory response. These mediators are either released from inflammatory cells (cell-derived) or are generated by the action of plasma protease (plasma-derived). Mediators of inflammation are multifunctional and have numerous effects on blood vessels, inflammatory cells, and other cells in the body. Some of their primary effects in the inflammatory response include vasodilation or vasoconstriction, modulation of vascular permeability, activation of inflammatory cells, chemotaxis, cytotoxicity, degradation of tissue, pain, and fever. These mediators include histamine, serotonin, bradykinin, the complement system, platelet-activating factors, arachidonic acid derivatives (e.g., prostaglandins, leukotrienes), and cytokines (Table 6-5).
Table 6-5
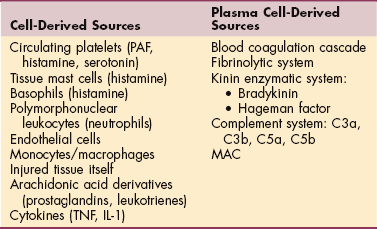
PAF, Platelet-activating factor; MAC, membrane attack complex; TNF, tumor necrosis factor; IL-1, interleukin-1.
Histamine.: Histamine is synthesized and stored in granules (for quick availability and release) of mast cells, basophils, and platelets. Histamine causes endothelial contraction leading to the formation of gaps, which increase blood vessel permeability and allow fluids and blood cells to exit into the interstitial spaces (vascular leak). Histamine’s effect occurs quickly but is short lived because it is inactivated in less than 30 minutes. Histamine is also a potent vasodilator and bronchoconstrictor. Serotonin is another mediator released from platelets; it induces vasoconstriction, but its effect is usually overridden by the vasodilator action of histamine.
Platelet-Activating Factor.: Leukocytes and other cells on stimulation also synthesize three classes of inflammatory mediators that are derived from phospholipids (the major lipids present in cell membranes). The first of these mediators is an acetylated lysophospholipid named platelet-activating factor (PAF). The other two classes of mediators are derived from a fatty acid (arachidonic acid) of membrane phospholipids and are called prostaglandins and leukotrienes. All three of these lipid mediators have potent and wide-ranging inflammatory activities. In addition, these mediators have hormonelike functions that modulate physiologic responses and induce pathology in a variety of organ systems.
PAF was so-named because it was first found to induce platelet activation and secretion. PAF is now known to be a potent activator of cells, such as smooth muscle cells, endothelial cells, and leukocytes, by receptor binding and intracellular signaling mechanisms. As a consequence, PAF can induce the aggregation of leukocytes and leukocyte infiltration in tissues and can profoundly affect vasomotor tone and permeability.135 PAF can potentiate (increase or strengthen) the activity of other inflammatory mediators.