Transplantation
Transplantation for the treatment of end-stage organ failure has been one of the major medical advances of the last 20 to 30 years. The success of this form of treatment has improved dramatically with better understanding of the rejection process and the introduction of more effective immunosuppressive medications.
There have been further advances in the prevention and management of bacterial, viral, and fungal infections so that recipients of transplanted organs or cells live longer. A number of people have survived for well over 25 years. Unadjusted survival rates at 5 years range from 90% for recipients of living related kidney transplants to 40% for the small number of heart-lung recipients, with the 5-year survival rate averaging 70%.215,316
The fiftieth anniversary of the first successful live-donor kidney transplantation was celebrated at the 2004 National Kidney Foundation’s U.S. Transplant Games in Minnesota. Dr. Murray and Dr. Harrison performed a live-donor kidney transplantation between identical twins.
By the end of the 1960s, the great advances in surgical techniques combined with immunologic and pharmacologic discoveries led to further successful organ transplantation, including the first heart, liver, and pancreas. With the commercial introduction of cyclosporine in 1983, the world of transplantation has made remarkable strides in becoming an acceptable medical intervention in the treatment of various end-stage organ diseases, including lung transplantation.226
In the past 2 decades we have seen great advances in the preservation of donor organs and surgical techniques to transplant multiple organs. There also have been advances in the detection of early rejection; further advances in immunotherapy and management of infections have made treatment even more successful. Over 350,000 lives have been saved or enhanced by transplantation.316
INCIDENCE
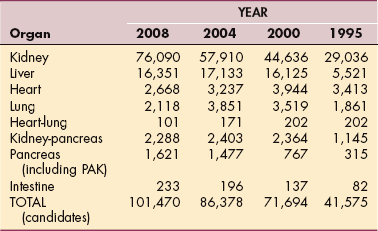
Transplantation remains limited by an acute worldwide shortage of available and suitable human organs. In 2006 there were over 92,000 people waiting for transplants. It is estimated that every day 19 people die while waiting on the United Network for Organ Sharing transplant list. In 2005 an estimated 12,000 to 15,000 deaths in the United States had the potential to yield suitable organs, yet only 7593 deceased individuals donated organs.
Each cadaveric donor can donate up to 25 organs and tissues to help as many as 50 recipients. This means up to 500,000 organs should be available for transplantation each year, but only approximately 25,000 transplantations are performed (Table 21-1).
Table 21-1
National Waiting List for Organ Transplantation
Data from the 2008 United Network for Organ Sharing, an online database system called UNet containing data regarding every organ donation and transplant event in the United States. Available at www.unos.org/data/.
One positive highlight in the incidence of transplantation has come from the increase in living related organ donation. In 2001 the number of live-donor organs recovered was more than the number of deceased-or cadaver-donor organs. In 2005 almost 7000 individuals donated a kidney or a portion of their liver, lung, or pancreas to provide an opportunity for another to survive an end-stage organ disease.316
TYPES OF TRANSPLANTATION
Many types of tissues and organs can be donated and therefore transplanted, including the heart, lungs, liver, pancreas, kidneys, intestines, skin, bone and bone marrow, umbilical cord blood, veins, soft tissues, heart valves, corneas, and eyes.
Ovarian cryopreservation and transplantation is under investigation. Considering that more than 50,000 reproductive-age women are exposed to sterilizing chemotherapy and radiotherapy annually in the United States alone152 and thousands lose their ovarian function due to gynecologic surgery, larger trials of ovarian cryopreservation and transplantation are strongly justified.
Many different terms are used to describe types of transplantations (Box 21-1). Allograft (homograft) transplantations are between individuals of the same species (e.g., human being to human being). Autologous transplantations are within the same individual (e.g., skin graft from leg to hand; blood or bone marrow for own use later).
Xenogeneic (heterograft) transplantations are between individuals of different species (e.g., pig to human being). Allogeneic transplantation is one in which the source comes from a human leukocytic antigen (HLA; see Chapter 7) matched donor (usually a sibling). Syngeneic transplants are between genetically identical members of the same species (identical twins); the syngeneic transplant is also called an isograft.
Orthotopic homologous transplantation refers to the surgical placement (grafting) of the donor organ into the normal anatomic site. In the heterotopic homologous transplantation, the recipient’s diseased organ is left intact and the donor organ is placed in parallel with anastomoses between the two organs.
Combined-Organ Transplantation
Combined-organ transplantations from a single donor are uncommon relative to single-organ transplantations (Box 21-2). Research to date generally suggests that organ rejection is decreased in cases of combined-organ transplantation compared with single-organ transplantation. Short-term survival in combined-organ transplantation seems to be acceptable, but long-term recipient and graft survival remains unknown at this time. No single center has accumulated a significant experience, and as a result long-term results in the current era are unknown.17
Organ Retransplantation
Occasionally, retransplantation is necessary because of acute graft failure, graft rejection or injection, or the recurrence of the primary disease as in the case liver or heart and lung transplantation. When the body mounts a defense against the transplanted organ, a clinical picture of chronic rejection presents itself. This form of rejection leads to a destruction of the donor organ over time.
In many cases the immunosuppressive medication is no longer able to suppress the immune response and rejection persists, which will eventually cause organ failure. One option is for the recipient to undergo another transplantation procedure. Transplant recipients in need of a retransplantation once again become organ candidates and must meet certain criteria for transplantation of that specific organ.
There is typically an increase risk of morbidity and/or mortality for these candidates when compared with outcomes of first-time transplantation procedures. For most organs the 3-and 5-year survival is not as high for recipients who have undergone retransplantation.234,236
A model for end-stage liver disease has been developed and tested to predict the outcome of transplantation for liver recipients with advanced liver disease. A similar model to estimate survival after retransplantation is being developed to help identify individuals with a poor expected outcome; this information could be useful in further refining candidate selection criteria.199 Studies show the model-based allocation system may not benefit candidates who undergo liver retransplantation.234
Ethical issues centered on the availability (i.e., shortage) of organs are always a consideration with retrans plantation. Graft survival after retransplantation is less than for primary transplants, both for immunologic (e.g., rejection) and nonimmunologic (e.g., donor age, donor size, cadaver vs. live donor) reasons, and requires more aggressive monitoring for rejection.
Pediatric Transplantation
Solid-organ transplantation has become accepted therapy for the treatment of end-stage organ dysfunction in children. As with adult organ transplantation, the supply of cadaver pediatric organ transplants is limited. And, like adult organ transplantation, living related donation is on the rise in pediatrics. Close tissue match of the related donor allows a higher compatibility rate and transplantation scheduling before the child is in critical condition improves outcomes.
Children can receive adult organs; in the case of the liver, only a portion of the adult donor liver is needed. Preoperative and postoperative assessment and care are very similar to adult care. Management may be complicated by infections such as hepatitis B and cytomegalovirus. Morbidity and mortality are often attributed to the consequences of long-term immunosuppression and include graft failure, increased incidence of cancer, hypertension, and renal failure or diabetes from overimmunosuppression.
There are known age-related differences in all phases of pharmacokinetics (absorption, distribution, metabolism, elimination); information specifically related to age and differences in the pharmacokinetics of immunosuppressants is very limited at this time. Biologic and psychologic changes common during the transition from childhood to adolescence and adolescence to adulthood present some unique challenges.141
Parent training and education are essential components in the transplantation process. The care team pays special attention to the psychosocial and emotional needs of the child and family. Noncompliance and nonadherence are common behaviors among all age groups but especially among adolescents. The consequences of this behavior include increased rejection, late graft loss, and death. Despite the best 1-year graft survival of any age group, the long-term transplantation outcomes in this age group are not as optimal.82,263
ORGAN PROCUREMENT AND ALLOCATION
With the passage of the National Organ Transplant Act (NOTA) in 1984, the U.S. federal government began the process of establishing a comprehensive framework for the development and administration of a national transplant system.317
The Organ Procurement and Transplant Network (OPTN) (www.optn.org) was created to maintain a national registry to track the process and outcomes related to organ donation and transplantation. During the past 20 years, nearly 350,000 people have received organ transplants at 250 U.S. transplantation centers, and the national waiting list has grown from 8400 people to more than 85,000.207
No further legislative progress was made to amend the NOTA because of the lack of agreement as to the federal government’s authority to set allocation policy, a task assigned to the OPTN. Then in 2004, the Organ Donation and Recovery Improvement Act (ODRIA, Public Law 108-216) was signed with a legislative provision to establish a federal grant program to provide assistance to living donors for travel and other expenses.
Instead of tackling disagreements over how to establish fair and equitable organ allocation policies, the Act focuses on strengthening efforts to increase donation rates, including ways to make live donation an easier and more financially appealing option. Removing financial barriers from living organ donations may help expand access to transplantation for members of lower socioeconomic groups who may not be able to consider living donation.207
This legislation also grants money to states for organ donor awareness, public education, and outreach activities designed to increase the number of organ donors, establish programs coordinating organ donation activities, and conduct studies of long-term effects associated with living organ donation.340
United Network for Organ Sharing
In order to establish a means to procure and distribute donor organs in an appropriate and ethical manner, the United States was divided into 58 local areas, ranging in population from 1 million to 12 million, and those areas were then divided into 11 regions (Fig. 21-1). Each local area has a designated organ procurement organization (OPO) responsible for recovering and transporting organs to transplantation hospitals in their territories. The local OPO also provides a wealth of medical and public education about organ transplantation and the promotion of donation.
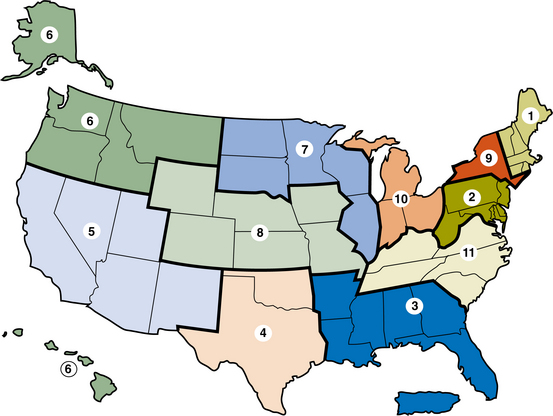
Figure 21-1 The United Network for Organ Sharing (UNOS) is divided into 11 geographic regions. (Courtesy United Network for Organ Sharing, Richmond, VA, 2006.)
On the national level the United Network for Organ Sharing (UNOS; www.unos.org) is a private, nonprofit organization that provides critical services in the area of organ transplantation. UNOS administers the national organ waiting list, coordinates the matching and distribution of donor organs via the local OPO throughout the United States, tracks outcomes, establishes physician training for the medical and surgical management of transplant recipients, and provides public education.316
UNOS is composed of every transplantation center, tissue-matching laboratory, and OPO within the United States, which are required to report a massive amount of data to UNOS. The OPTN is given these data, which analyzes in relation to the transplantation candidates, recipients, and living and decreased donors.
Great detail in the process to match candidates with a donor organ(s) has been developed to ensure equity based on medical need. There are two large processes occurring simultaneously to match and allocate organs. One side involves the identification and management of the potential transplantation candidate (someone waiting for an organ) through the transplantation center; on the other side is the identification and procurement of viable organs for donation.
After a thorough evaluation the transplantation center reports vital medical information about each candidate to UNOS. When a possible deceased organ donor has been identified, the local OPO will obtain and report valuable medical information about the donor to UNOS. The UNOS computer system will search for a suitable match between the donor and candidate.
A list of potential candidates will be provided in a priority order, and the organ is offered to the candidate who has the highest medical need as well as the greatest likelihood of a successful outcome based on the analysis of prior transplantation.
This allocation begins with the transplantation center being contacted at the local level, but if a local candidate does not match the donor organ or has a lower medical need, the organ will be offered to a candidate in the region where the donor organ was procured and then to adjacent regions and then finally nationally, with the goal to utilize every suitable organ.317
Allocation Policy.: The allocation of deceased donor organs has significantly changed over the years, with the goal to ensure that people who have the most urgent medical need will be given priority despite the amount of waiting time a person has on the UNOS list.319 The change in allocation policy is known as transplant benefit.
The policy is intended to balance anticipated duration of survival on the transplantation list with length of benefit from receiving a transplant. Priority for transplanted organs will go to those candidates most urgently needing a transplant and expected to receive the most survival benefit from the transplant. Under earlier organ allocation policy, priority was based on the amount of time candidates had been on the waiting list.
This distribution system is also designed to decrease the disadvantage some people had due to the progressive nature of their disease or the uneven distribution of transplantation centers within the United States. The new policy considers the waiting list urgency and transplantation benefit of each candidate based on individual clinical diagnostic factors. The improved computerized organ-matching system has made these important changes possible.
Organ Donation
Death is usually the circumstance under which organs are procured, either when complete and irreversible loss of all brain and brainstem activity occurs or when the heart stops in the case of cardiac death. The specific neurologic event that has resulted in brain death may include blunt traumatic injury to the head, intracranial hemorrhage, or penetrating traumatic injury.
Deceased organ donation continues to be the primary source for available organs. There has been a collaborative effort (Organ Donation Breakthrough Collaborative) by the U.S. Department of Health and Human Services Health Resources and Services Administration. The goal of the program is to unite the various agencies involved in transplantation and to define their roles and efforts to increase the identification of potential organs.
The transplantation center, donor hospitals, and the local OPO have teamed up to increase their efforts in procuring organs to save and enhance the lives of people who are dying from end-stage organ disease. This collaborative effort, which began in 2003, has resulted in the largest 1-year increase (11%) in deceased organ donor in the past 10 years.316
In the same time period there has been a 3% increase in living donation. The year 2004 was the first year since 2000 that the number of decreased donors exceeded 7000 in number and also exceeded the number of living donors.316
Source of Organ Donations
The characteristics of deceased and living donors have changed over the years, and the effect of these changes on the outcome for the recipient is not fully known. Early on it was typical that the deceased donor was a young individual who was declared brain dead from a traumatic brain injury. Today more deceased donations are from the older person who has suffered a fatal cerebral vascular injury from stroke or aneurysm.316
There is also an increased use of organs harvested from non–heart-beating deceased donors, especially in kidney transplantation. Most transplantation centers are still reluctant to transplant other organs from the non–heart-beating donor due to the risk of ischemic injury to the organs.123
There has also been an increased utilization of organs from what is referred to as an extended donor pool. These are deceased donors that fall outside the general criteria to be an acceptable donor, such as older individuals, individuals with diabetes, those with some forms of cancer, and individuals who are positive for the hepatitis B or C virus. There are reports of promising results when organs from this extended donor pool have been used.
Individuals previously considered unacceptable transplantation candidates may be offered a donor organ from this extended donor pool. Seropositive hepatitis B or C candidates can now receive a transplant from a positive donor. In the past these individuals were not considered acceptable candidates for transplantation.45 Successful kidney transplantations have been completed from deceased donors who have a medical history of diabetes.3
Many so called “marginal” or “suboptimal” organs have been discarded by centers while people die each day while on the waiting list. Marginal organs can provide a viable solution to organ shortage. When used with appropriate surgical techniques and immunosuppression protocols, suboptimal organs can increase the supply of donor organs by 25% to 30%.2
The source of organ donation is also changing in regard to the living donation. In previous years living donation was conducted primarily between relatives. There are currently transplantation centers beginning to perform transplantations using unrelated living donors.316 Finally, transplantation centers are exploring the possibility of ABO-incompatible renal transplantation in candidates who have undergone a splenectomy, plasmapheresis, immunoabsorption therapy, and plasma exchange to remove antibodies, thereby decreasing the antibody-antigen reaction that would cause graft or organ failure.160
Guidelines for Donor Candidates
There are general guidelines or standard criteria that are used when determining the acceptability of a donor, and the criteria will vary slightly based on organ type. These guidelines have been expanded as procedures to procure organs improve and as transplantation centers become more effective at the surgical procedure and medical management of transplant recipients.
Donor age is a consideration, with the majority of donors being under 50 years of age, although this is being extended because of the need for donors, further advancement in postoperative management, and type of organs that are available for procurement. For example, in order for the deceased donor to donate the lungs, the donor must typically be younger than 55 years (or 65 years if there is no history of smoking), with a clear chest x-ray, an absence of chest trauma, no thoracic surgery, clear bronchoscopic examination, and no aspiration or sepsis.
Smoking history must be less than 30 pack-years (see section on lung cancer in Chapter 15), with acceptable oxygenation measured as a PO2 greater than 300 mm Hg. A living related donor must be ABO compatible with the candidate, without a history of lung disease, no previous thoracic surgeries, and larger size than the recipient (weight, height, chest size); the latter requirement is required because only two lung lobes will support the recipient’s full pulmonary function.
Other criteria for donors vary according to the organ being harvested, but overall there must be no evidence of malignancy, human immunodeficiency virus (HIV) or hepatitis B, or sepsis (Box 21-3). Hepatitis C present in the donor is considered a precaution but not a contraindication. If the candidate already has hepatitis C or is critically ill, the risk of developing hepatitis C is not considered a contraindication in the decision to progress with the procedure.
Body weight must be within 20% of the ideal (using the body mass index [BMI] for heart and lung organ donation; see discussion of BMI in Chapter 2) because of the ischemia-perfusion injury associated with obesity.173 Biologically related donors are preferred with clear and altruistic motivation (as opposed to coerced by the family or guilt driven).
Testing will be performed to assess the function for each of the donor organs being considered for procurement. For potential renal donors, urinalysis, creatinine, and blood urea nitrogen will be completed along with tests to assess liver function. To assess the function of the heart, an echocardiogram, 12-lead electrocardiogram, serial arterial blood gas (ABG) measurements, and possibly a right and left catheterization will be ordered. Pancreatic function will be assessed with amylase and lipase studies, serial ABGs, sputum Gram stain, and bronchoscopy to inspect the airways to assess the function of the lungs.236,310,317
Organ Recovery
Hearts and lungs can be preserved for up to 6 hours, livers up to 24 hours, and kidneys up to 72 hours. Lungs cannot be preserved outside the body for any extended period of time. This length of allowable ischemic time helps determine allowable distances between centers.
Efforts are being made to improve organ harvest and preservation techniques and the number of organs harvested. For example, eliminating medical failures before donation through aggressive resuscitation, coagulopathy control, invasive monitoring, and dedicated intensive care unit (ICU) management while implementing a rapid brain death determination protocol has been documented as successfully increasing the number of donor organs available.153,316
Technology to improve organ recovery, maintain organ perfusion, and recover normal cell metabolism is under investigation utilizing a kidney transporter, a portable organ preservation device. The ability to maintain and monitor organ viability over an extended period of time may allow live donors to avoid traveling to the recipient’s location for explant surgery. Eventually, this type of technology may be extended to include transport devices for all other organs.
Efforts to obtain consent for donation continue to improve as part of the Collaborative’s efforts. Public education now includes National Donor Awareness Week and the choice to indicate organ donation on a driver’s license. According to Health Care Financing Administration regulations, hospitals are now required to report every death and impending death to their local OPO in order to continue receiving Medicare benefits.94
Early referral of all imminent deaths to OPOs can result in the OPO conferring with the medical team regarding the best medical plan of care for the recipient, including specific needs of the family as well as procedures necessary for the donor once consent is obtained. These steps will help ensure that care of potential organ donors continues without premature termination.271
A new approach to obtaining family consent is being utilized. Instead of informing the family of the individual’s death and at a later time discussing organ donation (referred to as decoupling), now most families are approached about organ donation at the time when they are making end-of-life decisions. The discussion about organ donation has become a team approach between the OPO staff, physicians, nurses, and clergy. In 2005, 57% of families approached about donation consented, which is up 17% from 2001.316
During this critical period of time assessing a potential donor, personnel from the OPO will be requesting medical evaluation from various specialists (e.g., cardiologist, pulmonologist, nephrologist, gastroenterologist, and surgeon) to determine the viability of the organs that were consented for harvesting and to provide instructions for continued medical care to ensure adequate organ function until the procurement process begins.
Criteria for Organ Candidates
People waiting for transplants (candidates) are listed at one or more of the transplantation centers where they plan to have surgery. A national, computerized waiting list of potential transplantation candidates in the United States is maintained by UNOS with active input from treatment centers. UNOS maintains a 24-hour telephone service to aid in matching donor organs with people on the waiting list and to coordinate efforts with transplantation centers.
Each possible transplantation candidate undergoes various testing, and many of these test results, such as organ(s) needed, blood type, body size, various organ function, walking ability, life support need, virology, and other pertinent comorbidities will be reported to UNOS.
There are criteria for most organs to classify candidates into levels of medical urgency or status based on the medical workup. When a donor becomes available, UNOS is notified and a list of potential organ candidates for that region is identified. UNOS notifies the OPO, which in turn notifies the transplantation center to verify that the candidate is currently medically appropriate and has consented to the transplantation process. Arrangements are then made to transport the donor organ and candidate to the transplantation center and proceed with the surgery.
UNOS has developed a status coding or allocation system to prioritize the candidates waiting for transplantation when a donor organ has been recovered. The goal of these allocation systems is to promote an equitable system that will save as many lives as possible. Each waiting list and the prioritization of possible candidates varies from organ to organ.
In response to a perceived unfairness in organ allocation, Congress issued a “Final Rule” in 1998. The rule called for a more objective ranking of potential recipients on a waiting list and more equality in disease severity among transplant recipients across OPOs. To date, little progress has been made in eliminating geographic inequities. Potential organ candidates in the smallest OPOs continue to receive transplants at a lower level of disease severity.67
The purpose of ranking or assigning the candidate a status is to consider beyond just the waiting time key medical information that has been determined to aid in the prediction of mortality while waiting and to predict the survival posttransplant. The goal is to decrease the number of deaths while waiting on a transplantation list and optimize the utilization of these precious organs.
For example, it is known that an individual who has a cardiac index of less than 1.8 l/min/m2 and who is on a high dose of one or more inotropic medications has a shorter life expectancy than someone who is only on oral medication with compensated heart failure. In this sce- nario the first person would be assigned a status 1A, listed higher on the transplantation listing, and offered an organ before the second person (Box 21-4).
Several factors are taken into consideration in identifying the best-matched candidate or candidates. In general, preference is given to candidates with the most critical status from the same geographic area as the donor because timing is a critical element in the organ procurement process. Waiting for combined-organ transplantation is much more complicated. Once the potential candidate is listed first for transplantation (for either required organ), the second organ must be available and no other higher status candidate must be waiting for that second organ.
The transplantation team bears the responsibility to conserve scarce resources for those who can benefit, requiring careful screening of potential candidates. Transplantation centers follow UNOS guidelines, but criteria may vary from center to center; some centers adhere to a medical evaluation for the acceptance of applicants for transplantation.
Other teams have medical and nonmedical criteria with exclusion criteria for people with severely problem- atic behavior or other psychosocial factors (Box 21-5). There has been a recent trend to recognize the importance of nonmedical issues, such as psychologic stability, family support, and history of compliance or adherence to medical care, when evaluating applicants.
Medical compatibility of the donor and candidate or candidates is determined based on characteristics such as blood type, weight, and age; urgency of need for some organs; and length of time on the waiting list. Any illness that cannot be treated or that will prevent transplantation success must be evaluated carefully. Transplantation is usually not recommended if another illness is predicted to rapidly cause graft failure.
For example, in the case of someone having heart failure who is being considered for heart transplantation, pulmonary arterial pressure will be evaluated. If the pulmonary artery pressure is high and unresponsive to medication, that would dilate the pulmonary vascular bed and reduce pulmonary hypertension; the pressure is considered refractory, or fixed, which can result in failure of the new donor heart.
The donor heart, particularly the right ventricle, will not be able to pump against the high pulmonary pressure and will result in right-sided heart failure. In addition, a previous history of cancer or osteoporosis is considered carefully since postoperative medications can greatly advance these diseases.
A history of problematic behavior, such as adherence to treatment and psychiatric instability, leads to higher posttransplant mortality and morbidity.76,185,276 Compliance issues associated with substance abuse usually include personality disorder, living arrangements, and/or global psychosocial factors. A history of substance abuse requires documentation of abstinence; ongoing drug and/or alcohol abuse can potentially impair the success of the transplantation and requires referral for treatment before placement on a transplantation waiting list.
In the case of live-donor transplantation, psychosocial risk to donors must be taken into consideration. Most published reports have indicated an improved sense of well-being and a boost in self-esteem for living donors, but there have been some reports of depression and disrupted family relationships after donation, and even suicide after a recipient’s death.156
Pretransplant Evaluation
Extensive medical testing is required before someone is placed on the transplantation waiting list (Table 21-2). Blood typing, including Rh factor analysis, is used as one of the first eligibility criteria for donor-candidate matching. Predictor values for acute and chronic rejection are evaluated, such as patent reactive antibodies (a measure of the amount of antibodies circulating in the system), and offer some predictive value of hyperacute rejection.
Table 21-2
Referral to Transplantation Center
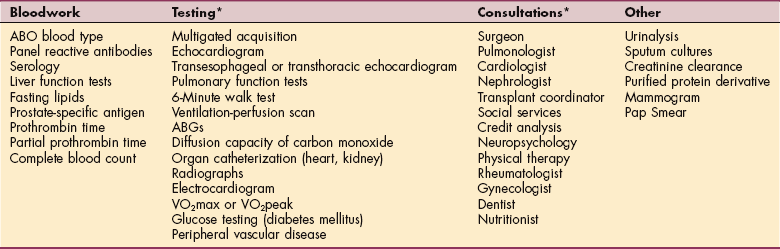
*Specific consultants and the special tests orders are determined by the type of organ transplantation planned.
Other serology testing determines exposure to cytomegalovirus (CMV), Epstein-Barr virus (EBV), herpes simplex viruses (HSVs), and hepatitis because complications can arise related to individual exposure to viral loading (i.e., the greater the viral replication, the higher the incidence of active infection).
CMV infection may contribute to an increased incidence of chronic rejection; HSV has been associated with an increased incidence of necrotizing pneumonitis and cervical cancer; and EBV has been associated with an increase in posttransplantation lymphoproliferative diseases. (See further discussion of these infectious diseases in Chapter 8.) Transplant recipients are placed on appropriate medications to reduce the risk of infection and undergo repeated serologic testing if clinical signs and symptoms suggest infection or infectious disease (see Box 8-1).
Fasting lipids, liver function studies, prostate-specific antigen levels to determine prostate function, and tests specific to the potential organ transplant are carried out. A person with isolated end-stage organ failure with no other complications has a better chance for selection than someone with other complicating factors. In addition to all the testing procedures, the organ candidate meets with a large team of professionals listed in Table 21-2, including, in some centers, rehabilitation staff such as physical and occupational therapists.
ADVANCES AND RESEARCH IN TRANSPLANTATION
Advances in understanding of immunology and organ preservation, surgical technique, pharmacology, and postoperative care have permitted the rapid development of other organ transplantation procedures than just the heart and lung (e.g., liver, pancreas, intestine).
New transplantation is being developed for other organs, such as pancreatic islet cells for the treatment of diabetes and ovarian preservation for later use in cases of cancer requiring removal of the ovaries. The use of hepatic segments for transplantation (either from cadavers or from living related donors) has decreased the number of people (especially children) awaiting liver transplantation.
The first transplantation of skeletal muscle cells to test whether the cells can repair damaged heart muscle took place at Temple University’s heart transplantation program in 2000. Muscle tissue taken from the individual’s arm was transplanted into his own heart during a surgical procedure to implant an assistive device while waiting for a heart transplant.89
Since then, researchers have successfully injected autologous skeletal myoblast cells into myocardium tissue in human beings undergoing concurrent coronary artery bypass grafting or ventricular-assist device implantation. This potential treatment for end-stage heart disease remains under investigation.78
Research is underway to develop transplantable cells for the treatment of human diseases characterized by cell dysfunction or cell death and for which current treatment is inadequate or nonexistent. Scientists are also looking for a way to modulate the human immune system in order to prevent rejection of transplanted cells without the use of immunosuppressive drugs.228
Additional products under investigation include porcine neural cells for stroke, focal epilepsy, and intractable pain; porcine spinal cord cells for spinal cord injury; engineered blood vessels for use as vascular grafts; neurologic cell transfer for Parkinson’s disease or Huntington chorea; human liver cells for cirrhosis; and porcine retinal pigment epithelial cells for macular degeneration.11,228
The diverse research directions being undertaken around the world will continue to change the field of transplantation in the years to come. Gene therapy (including in utero), xenotransplantation, tissue engineering, chimerism, and new fields of study developing daily can only be presented briefly in this text but help represent the overall picture of rapid change in treatment approaches.
Xenotransplantation
Allotransplantation remains the preferred treatment for human organ failure, but shortages of acceptable donor organs and the lack of success in developing suitable artificial organs have led researchers to investigate the use of organs from other species (xenotransplantation). Xenotransplantation is defined more fully as the interspecies transplantation of cells, tissues, and organs or ex vivo interspecies exchange between cells, tissues, and organs.12
Nonhuman primates are now considered an unethical and unsafe source of donor organs, so other species are being considered, in particular the pig. Baboon organs are too small to sustain human beings for long periods. The risk of transmitting deadly infectious agents from nonhuman primates is greater than from other animal species.
Physicians are already successfully using various pig components (e.g., heart valves, clotting factors, islet cells, brain cells) to treat human diseases. Researchers are now breeding genetically manipulated donor pigs whose cells, tissues, and organs could be permanently transplanted into human beings without being destroyed by the human immune system.106
Previously, hyperacute rejection or acute vascular rejection was the biggest disadvantage to xenotransplantation. Circulation of recipient blood through the transplanted organ caused graft failure within 24 hours. Recent scientific progress has eliminated this obstacle.283
Concerns still exist about the potential for transfer of infectious agents from animal to human being, leading to a possible epidemic. Scientists hope that, by using modern biotechnology, it may be possible to generate pigs free of threatening viruses in the future.283
Considerable progress has been made in recent years, and experimental pig-to-primate organ xenotransplantation has resulted in transplant function for days and weeks rather than minutes. Researchers have successfully implanted pig organs as a short-term bridge (up to 10 hours) until a human donor organ can be found and implanted.187
Other hurdles to xenotransplantation include anatomic, physiologic, and biochemical differences. The upright position of human beings is unique in nature. Gravity therefore exerts a different impact on the anatomic location of organs such as lung, heart, liver, and kidney. More pronounced are differences on the humoral and enzymatic basis.
Complex interactions existing in allografts are totally disturbed in xenogeneic situations. Regaining physiologic function of the graft in the foreign environment may be prevented by molecular incompatibilities between the donor and recipient, and there is the possibility of transferring infectious diseases from the animal donor graft to the recipient. Virologists and molecular biologists are concerned about the serious potential for introduction of diseases foreign to the human immune system.254
Experts say that before xenotransplantation can become an everyday reality, safeguards must be developed to ensure the minimization of risk to the recipient and to society. The decision to proceed with clinical application of this technique depends on ethical, regulatory, and legal frameworks established by consensus.106
Issues yet to be resolved include the recipient’s right to privacy, selection of the first recipients of xenografts, concern that the socioeconomically disadvantaged will be used as test subjects for the first xenografts, and animal rights are just a few of the concerns expressed by various interest groups.12,106,265
Tissue Engineering
Tissue engineering, the science of growing living human tissues for transplantation and other therapeutic applications, is a rapidly expanding industry—so much so that biomedical engineering and technology has become a college-degree program designed to develop engineers able to bridge the gap between biology, medicine, and engineering. Tissue engineering applies the principles of biology and engineering toward the development of biologic substitutes that restore, maintain, or improve tissue function.
The science of tissue engineering has given birth to a new clinical discipline called regenerative medicine aimed at restoring the functions of damaged or defective tissues and organs. Aging, associated with a progressive failing of tissues and organs and the leading cause of many diseases, is one of the primary forces behind a branch of regenerative medicine designed to “rejuvenate” the failing, aging body.15,162
Tissue engineering, or the fabrication of functional living tissue, uses cells seeded on highly porous, synthetic, biodegradable, polymer scaffolds as a new approach toward the development of biologic substitutes that may replace lost tissue function. Over the past decade, the fabrication of a wide variety of tissues has been investigated, including both structural and visceral organs.
Bioengineered skin, bone, ligaments, tendons, and articular cartilage are already available in some clinical settings.18 Collagen meniscus implants may be used to regenerate or regrow new meniscus-like tissue, with the goals of slowing down and preventing further degenerative joint disease, enhance joint stability, provide pain relief, and return people to activities at their desired level.
Autologous chondrocyte implantation and osteochondral autologous transplantation take plugs of cartilage or bone from one site, multiply the cells in culture, and place them into a lesion or hole in the native cartilage or bone, respectively.
Functional bone tissue with the necessary strength for load-bearing applications is still under development. Injectable hydrogels have resulted in materials with significantly enhanced compressive strength.277 Several growth factors contained in demineralized bone matrix (e.g., bone morphogenetic protein) are now being used to stimulate bone healing. Tissue engineering for bone healing has great potential for many people who experience nonunion, slow-to-heal bone fractures, or traumatic bone loss associated with war injuries.114
Other research is underway to generate stronger bone substitutes either by increasing osteoblast differentiation and mineralization71 or culturing the engineered tissue for a longer period of time before implantation to allow matrix maturation.50 Learning to tailor the strength of tissue-engineered bone to the person’s need requires further research.73,183
Other examples of current progress in the area of bioengineering include implants filled with islets for people with diabetes to replace insulin injections, a method to generate natural breast tissue to replace saline implants, heart valves, dental tissue (gums, teeth), skeletal muscle tissue isolated from synthetic polymers, and formation of phalanges and small joints from bovine-cell sources (calves).
Laboratory-grown organs are farther off, with the hope for producing donor tissue and organs for transplantation on demand and developing living prosthetics (incorporating living tissue with electronics) for every organ system in the body.184,322 Embryonic and adult stem cells, able to differentiate into all types of cells, remain the hope of many scientists in the treatment of systemic diseases and local tissue defects, as a vehicle for gene therapy, and to generate transplantable tissues and organs in tissue-engineering protocols. There remain many biologic and ethical challenges to overcome before this type of treatment becomes a reality.22,162,232
Medications
Tremendous advancement has been made in the pharmacologic management of transplant recipients. Medications are used with transplant recipients to prevent rejection and treat rejection or infection. Research is ongoing to find ways to reduce or eliminate the long-term use and adverse effects of medications, especially immunosuppressants. New discoveries in cellular immunology have led to a greater understanding of the immune system and its implications for tissue transplantation. Immunosuppressive regimens continue to improve, and newer immunomodulatory strategies are evolving. In particular, new immunosuppressive drugs may allow the recipient to overcome or reduce the early antibody-mediated rejections.42
Most transplant recipients are placed on a three-drug regimen to control the incidence of rejection and minimize the adverse effects that are common if any one drug is given in too high a dosage. This drug cocktail commonly consists of a calcineurin inhibitor, an antimetabolite, and a corticosteroid. Great strides are still being made to decrease the dosage and the number of immunosuppressive medications the transplant recipient is exposed to in order to promote long-term, effective graft function.
There has been a decline in the use of cyclosporine and Neoral over the 5 five years for most organs. These drugs have been replaced with the administration of tacrolimus (Prograf, FK506). Cyclosporine and tacrolimus are classified as calcineurin inhibitors.316 Calcineurin is an enzyme, protein phosphatase, which is responsible for the activating the transcription of interleukin-2 that stimulates the growth and differentiation of T cells. Calcineurin is also linked to the differentiation of fiber types and hypertrophy of muscle fibers.86
The mechanism of action for tacrolimus and cyclosporine is similar in that they both inhibit calcineurin, although tacrolimus is more selective in its action and may be one of the reasons its use has become more popular (Fig. 21-2). Both drugs bind to specific lymphoid tissues and block the production of interleukin-2, which is a critical substance in the growth and proliferation of activated T cells and other immune response cells, such as natural killer cells, macrophages, and lymphocytes.66
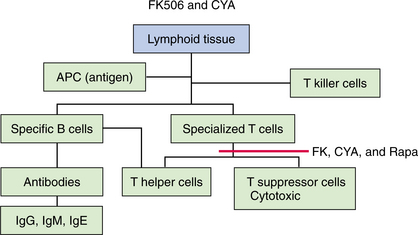
Figure 21-2 Cyclosporin (CsA, CYA, Neoral, Sandimmune) and tacrolimus (FK506, Prograf) block the sensitization of T cells. Cyclosporin and tacrolimus inhibit calcineurin in lymphoid tissues and thus inhibit the production of immune mediators such as interleukin-2. In general, sirolimus (Rapamune) structure is similar to cyclosporin but its action is different. It does not interfere directly with the cytokine production but inhibits the growth and proliferation of T and B lymphocytes by inhibiting the lymphocytes from taking action in response to stimulatory signals from certain cytokines. (Courtesy Chris L. Wells, University of Maryland Medical Center, and James H. Dauber, University of Pittsburgh Medical Center-Health Systems.)
Another classification of drugs commonly used along with a calcineurin inhibiting medication are the antimetabolites. These drugs include azathioprine (Imuran), mycophenolate mofetil (CellCept), and cyclophosphamide (Cytoxan).316 There has been an increased utilization of mycophenolate mofetil over other drugs in this classification. Azathioprine works by suppressing the bone marrow (as exhibited by thrombocytopenia, leukopenia, and anemia), and mycophenolate mofetil inhibits the inflammatory response mediated by the immune system (Fig. 21-3).190,191 Cyclophosphamide, which is typically thought of as an anticancer drug, has been used in transplant recipients. It inhibits the replication of DNA and RNA in the lymphocytes and other key cells involved in mounting an immune response against the transplanted organ.66
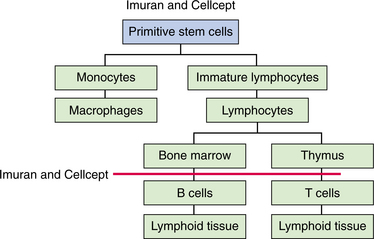
Figure 21-3 Azathioprine (Imuran) and mycophenolate mofetil (CellCept) are theorized to block lymphocytes from maturing into T cells. This inhibits the immune-mediated inflammatory response. (Courtesy Chris L. Wells, University of Maryland School of Medicine, and James H. Dauber, University of Pittsburgh Medical Center-Health Systems, 2000.)
The third class of drugs includes prednisone, a corticosteroid with an effect at the level of the macrophages. Prednisone blocks the production of interleukin-2 in the presence of an antigen to stimulate a major histocompatible complex, thus stimulating both B-cell and T-cell response (Fig. 21-4). Work continues to be done to decrease the exposure and utilization of corticosteroids because of the adverse effects of this medication, including diabetes, osteoporosis, and fat deposition.
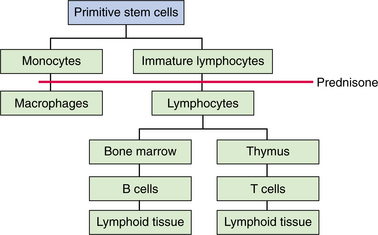
Figure 21-4 Prednisone works at the macrophage level and is theorized to block the production of interleukin-2, thus preventing the formation of major histocompatible complexes that normally stimulate both B-and T-cell response. (Courtesy Chris L. Wells, University of Maryland School of Medicine, and James H. Dauber, University of Pittsburgh Medical Center-Health Systems, 2000.)
Sirolimus (rapamycin, Rapamune) is an immunosuppressant that inhibits cytokine-driven cell proliferation and maturation. It was approved for use with renal trans- plants and was introduced for use with other organ transplants in the late 1990s. Sirolimus is presently being used in a low percentage of transplant recipients. It may be used in combination with a calcineurin inhibiting drug and mycophenolate mofetil. Some centers use sirolimus and mycophenolate mofetil alone, particularly with pancreas kidney recipients.166,316 Recent studies have reported a decrease in chronic rejection in heart transplant recipients with the use of sirolimus.167
Unlike cyclosporine and tacrolimus, which prevent the body from reacting to the transplant, sirolimus “stalls the engine,” disabling the body’s ability to reject the transplanted organ. Because of this effect, sirolimus in combination with cyclosporine and steroids not only lowers the incidence of acute renal allograft rejection, but also permits cyclosporine sparing (reduced amounts or eventual elimination) without an increased risk of rejection.
Medication Reduction and Withdrawal
Among individuals who initially received sirolimus in combination with cyclosporine and steroids, those who had steroid treatment stopped 1 month after transplantation had significantly fewer rejection episodes and were spared the numerous toxic side effects associated with long-term steroid administration.136
Ongoing research continues to explore and support this practice as early as 4 days after transplantation.8,10,338 Steroid withdrawal can increase the risk of acute rejection but reduces the incidence of infection. Maintaining a sufficient immunosuppressive regimen is the key to successful steroid withdrawal.
Withdrawal or marked reduction of corticosteroids is of particular benefit in the case of diabetes mellitus and in the presence of severe osteoporosis or aseptic necrosis of bone. Steroid withdrawal has been shown possible in up to 70% of pancreas transplant candidates who are otherwise maintained on tacrolimus-based immunosuppression.143,157
One other way to reduce the amount of immunosuppression required is through the use of monoclonal antibodies, such as OKT3, a mouse monoclonal antibody to human T cells. OKT3 blocks the ability of the candidate’s T cells to recognize foreign antigens, thus inhibiting both the generation and the function of cytotoxic T cells responsible for graft rejection. T cells are rapidly decreased in the circulation after the drug is given; by the third day there are usually no detectable circulating T cells. Unfortunately, the use of OKT3 for induction therapy has an association with chronic rejection in heart transplant recipients.261
Research groups are working toward identifying the critical components on particular grafts that are seen as foreign to modify them.31 This work will enable the graft to succeed while simultaneously allowing the host immune response to carry out its main tasks. Strategies that teach the immune system to accept the transplanted tissue rather than attack it, a process called chimerism, are under investigation (see the section on Future Trends under Blood and Bone Marrow Transplantation in this chapter).135,175,288
Chimerism involves inducing the donor’s immune system onto the candidate’s so that the candidate’s immune system no longer rejects the organ or tissue. In bone marrow transplantation (BMT) chimerism is achieved when bone marrow and host cells exist compatibly without signs of graft-versus-host disease (GVHD).
Researchers studying how the developing fetus avoids destruction may be able to identify protective biologic pathways and then use this model to develop drugs to interrupt the rejection process and promote tolerance of foreign tissue.147,312
BIOPSYCHOSOCIAL IMPLICATIONS
Many different ethical, social, moral, economic, and legal issues are associated with the procurement and allocation of living or bioengineered tissue. In addition, new information concerning the psychoneuroimmune responses (see the section on Psychoneuroimmunology in Chapter 1) in healthy tissues and organs has added new dimensions to understanding the emotional adjustment for recipients of living organ and tissue transplants.
Legal and Ethical Considerations
Before alternate treatment methods can be fully implemented, scientific and medical communities and the general public will have to seriously consider and attempt to resolve legal and ethical issues. For example, federal law prohibits the sale of human organs in the United States, and violators are subject to fines and imprisonment. However, individuals have taken matters into their own hands and established donor matching services on the Internet (Box 21-6).
In some countries of continental Europe, organ donation is governed by “presumed consent” legislation. Unless legally designated otherwise, organ donation is presumed on the death of each individual. Consent leg- islation has had a proven and positive, sizeable effect on organ donation rates in the last 10 years.1
Although Western opinion is almost universally against the practice of paid organ donation and the use of organs from judicially executed prisoners, similar laws are not in place worldwide. The ethics of both issues continue to be debated.
Bioethical Considerations
Another area of concern involves researchers growing human cells into tissues using stem cells derived from human embryos left over from attempts at artificial fertilization or following abortions. Currently in the United States embryonic stem cell lines are being made in the private sector and in private universities that use private funding. U.S. federal funding currently is restricted to 22 cell lines that were made on or before August 9, 2001.
Legislation in the United States to allow federal funding for research using stem cells derived from embryos originally created for fertility treatments and willingly donated by consenting adults has been introduced and remains a debated issue. Other countries such as Israel, England, and India and in parts of Asia have moved ahead in this area.
For the most part, embryonic stem cells are only used in fundamental research. It is predicted that it will be at least 5 to 8 years before they can be put to use in clinical trials. Since January 2006, stem cell trials for the treatment of stroke, spinal cord injury, leg ischemia, and myocardial infarction have been conducted in India using bone marrow–derived stem cells.
In Canada, the Canadian Stem Cell Network coordinates Canadian stem cell research at over 70 research sites. Research in Canada has historically been on adult stem cells; however, as of June 2005 a small amount of human embryonic stem cell research was underway with two human embryonic stem cell lines.
The Canadian federal government has passed legislation banning human cloning for reproductive or therapeutic purposes. However, the Assisted Human Reproduction Act allows Canadians to derive new human stem cell lines from embryos left over after fertility treatments. The Act also recommended that an authority be set up to license, inspect, and enforce activities controlled under the act and to foster the application of ethical principles in relation to assisted human reproduction. The Assisted Human Reproduction Agency of Canada was established in 2006.127,315
The United Kingdom established a national bank for stem cell lines, called the UK Stem Cell Bank, to work closely with the clinical and research communities to provide qualified stocks of human stem cell lines of adult, embryonic, and fetal origin for both research use and for use in emerging human therapy.128
Many bioethicists and lawmakers still question the appropriateness of this research until the ethical issues and appropriate concerns can be voiced and resolved. Questions about the nature of human life and its protection, the safeguard of human dignity, and the use of genetic material have been raised. However, new discoveries in the rapidly developing field of stem cell research, such as the discovery of master stem cells (see the section on Blood and Bone Marrow Transplantation in this chapter) replacing the use of embryonic tissue, may bypass these bioethical concerns.
Concerns related to animal-derived matrix proteins have also been raised. Some private bioengineering companies are proactively researching ways to develop human tissue with matrix proteins naturally secreted by the cells rather than developing tissue from animal-derived matrix proteins. In the area of animal organ transplantation (xenotransplantation), rules governing the welfare of animals bred for transplants are being formulated. For example, a ban on having children has been placed on all people receiving animal organ transplantation.
Psychoemotional Considerations
Transplant applicants face many challenges, including the obvious physical illness, complex assessment protocols, uncertainties about surgery and outcome, the possibility of relocation to obtain transplant services, and large expenses. Waiting for a transplant can be accompanied by a vast range of changing emotions, such as relief, despair, elation, depression, excitement, and apprehension.
No single attitude is common or expected; each person’s reaction is a valid expression of his or her experience. Most candidates find waiting for surgery a stressful time and, of course, the longer the wait, the greater the stress. The evaluation process itself and the wait for the results after tests and procedures require complex coping strategies, especially if the person is denied for transplantation. Deciding whether to accept a donor organ or wait for a potentially better one can create considerable psychologic and emotional distress.
When death is a possibility, candidates may worry that negative thinking will harm their health. Others are distressed that someone else must die before an organ will be available for transplantation or that receiving an available organ deprives someone else of life. Candidates are encouraged to focus on the desired outcome without completely ignoring the alternatives for themselves and outcomes for others. Counseling and support groups are often recommended.
Anxiety and depression are common complications of medical illness of any kind and may interfere with the candidate’s or recipient’s participation in rehabilitation. Symptoms of posttraumatic stress disorder (PTSD) are not uncommon in the recipient or partner after organ implantation or mechanical assist device implantation followed by heart transplantation.43,214
Clinical symptoms of PTSD, anxiety, or depression can be subtle and mimic the individual’s health condition, requiring candidate or recipient self-awareness and careful screening by all members of the transplantation team to identify and treat early. Attention to the supporting members of the recipient’s family and partners is also advised.43
A condition severe enough to require organ transplantation can sometimes impair concentration, memory, judgment, or ability to process thoughts. In particular, approximately one third of all liver transplant candidates have severe impairment of mental abilities (i.e., hepatic encephalopathy) and may be extremely confused or even delirious at the time of transplantation. Similar mental impairment can occur with heart, lung, and kidney candidates, although it is less common.
Posttransplant
Postoperatively, recipients face a long recovery period, the potential for graft rejection, reintegration into family and work roles, and lifelong changes, such as the need for drug compliance and changes in diet. Adaptation after transplantation is a lifelong process and depends on several factors, such as the success of the transplantation, expectations before the transplantation and perceived outcomes, postoperative complications or side effects of the antirejection drugs, permanent physical changes or changes in appearance, as well as other individual considerations.
Identification of recipients most likely to have compliance and psychiatric problems early after transplant is important in focusing interventions that maximize recipients’ psychosocial status in these areas and thus improve long-term physical health outcomes.76,296 Pretransplant psychiatric disorders, female gender, longer hospitalization, more impaired physical function, and less social support from caregivers and family in the perioperative period are known risk factors for posttransplant anxiety and depressive and psychologic disorders.75
Stress on the family and the need for family support and counseling also affect treatment outcomes. Health care staff may observe a deterioration of family relationships after transplantation (especially between husband and wife or between partners). Older children receiving organ transplantation may face the challenge of parents not allowing or unable to allow the child to grow into adulthood. Support groups can be extremely helpful in these types of situations and should be recommended early by the health care team.
Some recipients experience difficulty accepting the transplanted organ as their own. Others wonder if their new organ carries some donor characteristics. Typical literature available from transplantation centers contains reassurances that a heart, liver, or kidney can be transplanted into another person without the transfer of personal characteristics. Changes reported are often attributed to the normal process associated with overcoming a serious life-threatening illness.
On the other hand, research pioneered by Candace Pert,247 formerly a molecular biologist at the National Institutes of Health, has discovered the biologic basis for emotions. The results of Pert’s research have demonstrated that peptides and various other ligands (information carriers) and their receptors are the physiologic substrates of emotion. This work strongly helped establish the field of psychoneuroimmunology (see the section on Psychoneuroimmunology in Chapter 1) and supports the idea that emotions, personal characteristics, behaviors, and thoughts are biochemically derived—not only being actively present within our tissues, but also making up a bodywide system carrying this type of information across cellular barriers.
Following is a brief summary of Pert’s research findings. The reader is referred to her book Molecules of Emotion: the Science Behind Mind-Body Medicine247 for a more thorough treatise of her findings and similar organ-specific information presented by others.237,245,326 The application of Pert’s findings and their implications for transplantation are merely speculation at this time but raise some interesting and potentially serious psychosocial and legal issues.
For example, many organ recipients report frequent dreams or nightmares. It is not uncommon to have dreams while they are half awake; sometimes disorientation accompanies sleep disturbance or waking dreams commonly labeled delirium or confusion. Others report changes in temperament or mood, sexual libido, food preferences, and personal preferences. The exact etiology of these perceived changes remains unknown; previously, the dreams and dreamlike disturbances have been attributed to postoperative sleep disruption (e.g., anyone waking up often in the night is more likely to remember more dreams) or considered an unpleasant side effect of the medications (cyclosporine, prednisone).
Other researchers suggest that it is reasonable to include the impact of depression, and possibly other psychologic states, among factors that may affect the net state of immunosuppression in transplant recipients.171
New knowledge within the field of psychoneuroimmunology combined with many more case reports of altered dreams, thought processes, memories, and behavior as the number of organ transplants increases may bring to light new understanding of transplantation psychoneuroimmunology in the decade ahead. One of the first carefully documented and published reports to explore this type of phenomenon may be of interest to readers.299
POSTTRANSPLANT COMPLICATIONS
With advances in technology and immunology, transplantation of almost any tissue is feasible, but the clinical use of transplantation to remedy disease is still limited for many organ systems because of the rejection reaction and other posttransplant complications.
Complications following organ transplantation can be classified into three broad categories: (1) complications associated with the procurement and surgical procedure, (2) complications of the transplantation, and (3) complications of the immunosuppressive agents used to prevent rejection. Each type of organ transplantation has its own accompanying surgical risks and complications (see discussion in each section).
Following surgery, two main complications of organ transplantation remain infection and organ rejection. The most serious posttransplant complication is death, which can be caused by a infection, organ toxicity, GVHD, relapse, and various other causes (Fig. 21-5).
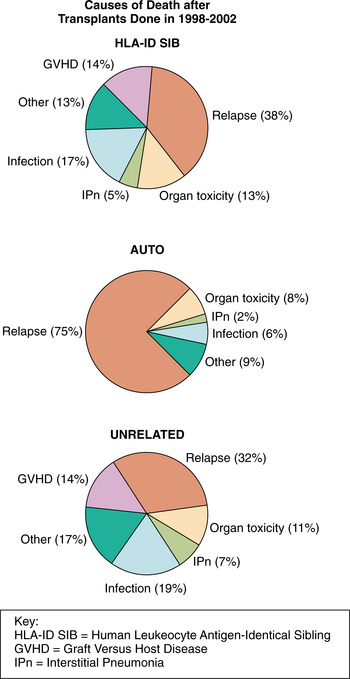
Figure 21-5 Causes of death after transplants done from 1998 to 2002. (Courtesy Center for International Blood & Marrow Transplant Research (CIBMTR). CIBMTR Newsletter 12[1], May 2006.)
Infection and organ rejection are common and treatable, but prevention is the first step. For this reason, pretransplantation serologic testing is done to determine histocompatibility and to avoid transmitting infectious agents (e.g., CMV, vancomycin-resistant enterococci [VRE], HBV, HCV) from donor to recipient. Herpeszoster infection can be a serious complication of organ transplantation, with postherpetic neuralgia occurring in almost half of the recipients affected. Heart and lung recipients have the highest incidence (15%), followed by renal (7.4%) and liver (5.7%).117
As the survival rates improve other complications arise, such as the increase risk of other diseases such as cancer and diabetes.
Ischemic Reperfusion Injury
Ischemic reperfusion injury may occur with any organ transplantation; it results in the onset of the inflammatory response, which has both immediate and long-term effects on the donated organ. The donated organ is very sensitive to the amount of time it is not being perfused or supplied with blood, which can lead to ischemia.62
The presence of ischemia can result in permeability of the endothelium and activation of many inflammatory-associated cells, such as macrophages and neutrophils. The endothelium loses its antiadhesive nature and a thrombogenic environment is established.34
After the organ has been surgical implanted, the clamps are removed to allow blood to once again perfuse the organ. It has been suggested that the abrupt flow of blood may create further trauma to the epithelial lining of the blood vessels because the transplant recipients may be circulating blood at a higher pressure than to what the donor organ is accustomed.
This phenomenon is especially common in heart or lung transplantation in the presence of comorbid pulmonary hypertension. This results in a reperfusion injury, which leads to leukocytes and platelet aggregation, which further causes endothelial permeability and inflammatory cell activation and adherence.
It is common for most transplant recipients to have to recover from a mild ischemic reperfusion injury usually lasting 3 to 5 days. If the injury is significant, organ dysfunction and failure and possible death of the recipient are possible. More recently it has been shown that there is a significant relation between the presence of an ischemic reperfusion injury and development of chronic rejection via the activation of both the innate and adaptive immune responses and organ regeneration.34
Histocompatibility
In all cases of graft rejection, the cause is incompatibility of cell surface antigens. The rejection of foreign or transplanted tissue occurs because the recipient’s immune system recognizes that the surface HLA proteins of the donor’s tissue are different from the recipient’s.
Certain antigens are more important than others for successful transplantation, including ABO and Rh antigens present on red blood cells and histocompatibility antigens, most importantly the HLA. As expected, there is a better chance of graft acceptance with syngeneic or autologous transplants because the cell surface antigens are identical. For all categories of transplantation, minimizing HLA mismatches is associated with a significantly lower risk of graft loss.60
It has been shown that in a person with HLA antibodies, the antibodies are directed against the antigen of the donor kidney and will result in immediate graft failure. It has also been documented that the when specific HLA antibodies are directed against B cells, hyperacute rejection is produced, leading to graft dysfunction and possible failure.302
Rh antigen is more important in heart and kidney transplants than in lung transplantation. In renal transplants HLA cross-matches are routinely performed, whereas this is not commonly done in lungs (unless the candidate has a high patent reactive antibody count). Cross-matching policies may vary by institution.
The process of determining histocompatibility, that is, finding compatible donors and candidates, is called tissue typing. Before transplantation, testing in the laboratory is carried out to determine whether antibodies incompatible with the donor have been formed by the candidate (a positive cross-match). If the cross-match is positive, the transplant will fail; a negative test result is necessary for a successful transplant.
Graft Rejection
Transplant rejection may occur for immunologic or nonimmunologic reasons. When the body recognizes the donor tissue as nonself and attempts to destroy the tissue shortly after transplantation, rejection occurs as an immunologic phenomenon primarily because of histocompatibility.
Nonimmunologic factors can occur as a result of the draining reperfusion process necessary in organ harvest and transplantation. Ischemic injury occurs when normal blood and oxygen supply to the donor organ is stopped at the time of organ harvest, whereas reperfusion injury can occur when blood flow is returned to the organ after transplantation. Ischemic reperfusion injury has been associated with an increase in acute and chronic rejection.34
As residual blood is drained from the transplanted organ into the host’s general circulation, the body recognizes the transplanted tissue cells as foreign invaders (antigens) and immediately sets up an immune response by producing antibodies. These antibodies are capable of inhibiting metabolism of the cells within the transplanted organ and eventually actively causing their destruction.
Research to develop a reliable method to reduce the ischemic reperfusion injury is currently ongoing. Eliminating the occurrence of poor early graft function and consequently reducing the chances for rejection episodes are the primary goals of these investigations.81,84
Types of Graft Rejection
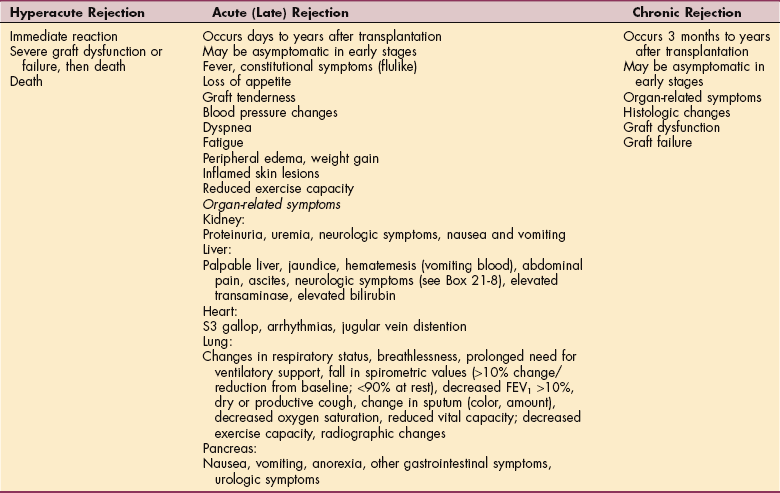
There are three types of transplant rejection—the hyperacute rejection, the acute or late acute rejection, and the chronic rejection—depending on the amount of time that passes between transplantation and rejection (Table 21-3).
Hyperacute rejection (rare with antibody screening and tissue typing) is dominantly mediated by humoral responses of the immune system (natural antibodies, complement cascade) and the activation of coagulation factors. There is an immediate rejection after transplantation when the recipient has preformed antibodies to donor tissue.
This reaction necessitates prompt medical action, which may include surgical removal of the transplanted tissue or the use of life-support devises such as a temporary ventricular assist device (VAD) or extracorporeal membrane oxygenator, in the case of a heart or lung transplantation. These devices can be used to support blood circulation and gas exchange while the patient undergoes such treatment as plasmapheresis and immunoglobulin therapy in an attempt to remove the reactive antibodies. Medical treatment may diminish the hyperacute rejection response and allow the donor organ to recover, or it may allow time for another donor organ to be implanted.190
The acute or late acute rejection can appear days to years after the transplantation. This type of rejection involves a combination of cellular and humoral reactions. Acute antibody-mediated rejection or vascular rejection typically occurs days to weeks posttransplant. There is an interaction between the recipient’s antibody and donor HLA or endothelial cell antigens that leads to graft vessel injury and thrombosis formation. Acute cellular rejection is most common in the first 3 to 6 months posttransplant and involves the proliferation and infiltration of T lymphocytes and macrophages.134,190 Despite the early pattern of acute rejection, both humoral and cellular rejection can occur at any time.
Clinically, there is sudden onset of organ-related symptoms, which may be associated with fever and graft tenderness, fatigue, or decrease exertional tolerance, or the recipient may be totally asymptomatic. Graft rejection must be differentiated from immunosuppressive toxicity.
This form of rejection can be reliably graded using a system of categories of mild, moderate, and severe rejection. Acute rejection, if detected in its early stages, can be reversed with immunosuppressive therapy. With the advancement in immunosuppressive medications and management, there has been a decline in acute rejection, which has lead to an increase in 1-year graft and recipient survival.316
Chronic rejection can occur as early as 3 months, but it is usually months to years before the chronic rejection occurs. This type of rejection develops as a function of both cell-mediated and humoral-mediated reactions and is characterized by slow, progressive organ failure.
Growing evidence indicates that chronic rejection is the aggregate sum of irreversible immunologic and nonimmunologic injuries to the graft over time. Chronic rejection is associated with chronic vascular changes such as arteriopathy or diffuse atherosclerosis with intimal proliferative changes depending on the type of organ. In the presence of a chronic immune/inflammatory process within the donor organ, the intimal lining of the vascular tissue undergoes fibrosis and vascular remodeling. This leads to a decrease in the lumen size and ischemia of the distal tissue and perpetuates the inflammatory reaction.126,190
A history of acute rejection episodes, either asymptomatic or clinically apparent, and inadequate therapeutic level of the immunosuppression medications or poor compliance are among the most recognizable immunologic risk factors for chronic rejection.218
Adherence to immunosuppressive therapy is a key factor contributing to transplant failures that occur within 2 years after surgery. Financial barriers such as the lack of insurance coverage are the most common reason for noncompliance. The Immunosuppressive Medications Act supported by the National Kidney Foundation would extend Medicare coverage of postoperative medications so that organs are not lost because of a lack of insurance coverage.227 Chronic rejection results in irreversible cellular damage within the donor organ and leads to graft dysfunction and eventually failure.126,190 Rarely is chronic rejection responsive to medical therapies.
Graft-Versus-Host Disease
Acute GVHD remains a major obstacle in the curative potential of allogeneic stem cell or organ transplantation. The use of BMT to bone marrow–depleted or immunodeficient people has resulted in the complication of GVHD. People at highest risk include premature infants and others who are immunosuppressed as a result of either congenital or acquired disease or because of the administration of immunosuppressive therapy, as in the case of organ transplant recipients.
GVHD occurs when immunocompetent T lymphocytes in the grafted material recognize foreign antigens in the recipient, initiating a cascade of events mediated by cellular and inflammatory factors and resulting in a reaction against the recipient’s tissues.97 GVHD may be acute, occurring in the first 1 to 2 months after transplantation, or chronic, developing at least 2 to 3 months after transplantation.
GVHD remains the major toxicity of BMT. It does not occur in autologous BMT or peripheral blood stem cell transplantation (PBSCT) but may occur in an allograft or syngeneic transplant, even with a perfect HLA match, because of unidentified and therefore unmatched antigens. Pretransplant immunologic and genetic manipulation using hematopoietic stem cells has minimized GVHD in individuals receiving high-dose chemotherapy.
Key risk factors for the development of GVHD include older age, source of allogeneic stem cells (marrow vs. blood), and gender mismatching. Conditioning regimens containing total body irradiation are associated with higher incidence and severity of GVHD compared with those involving only chemotherapy.7
Signs and symptoms of GVHD are fever, skin rash, hepatitis, diarrhea, abdominal pain, ileus, vomiting, and weight loss. As the disease worsens, the skin rash may progress to skin blistering with severe diarrhea, abdominal pain, and hepatic dysfunction.
Chronic GVHD is also characterized by hardening of organ tissues (connective tissue disorders such as scleroderma) and drying of mucous membranes (mucositis), especially in the gastrointestinal mucosa, liver, respiratory bronchioles (bronchiolitis), and skin (scleroderma or systemic lupus erythematosus) (Box 21-7).
A generalized polyneuropathy coincident with the occurrence of GVHD has been reported. The neuropathy affects proximal and distal muscles and demonstrates hyporeflexia or areflexia. Electrophysiologic studies do not meet strict criteria for demyelination. The signs of neuropathy improve after immunosuppressive treatment or simultaneously with the resolution of GVHD.
Preventing GVHD through the use of prophylactic (cyclosporine or tacrolimus) is advised when appropri- ate. Untreated GVHD is often fatal as a result of hemorrhage and infection. Inhibition of cytotoxic T-lymphocyte–mediated tissue injury in the early stages of GVHD is recommended along with administration of agents to eliminate the donor’s lymphocytes.
High-dose corticosteroids are the mainstay of therapy. Treatment with immunosuppressive therapy including prednisone, cyclosporine, or tacrolimus (FK506, Prograf), thalidomide, or a combination of these agents has improved the long-term outlook for people with chronic GVHD. Even so, the overall fatality rate is about 20%; in the severe form, this rate exceeds 80%. Most people with grade 4 disease do not survive.7 Chronic GVHD may resolve slowly (sometimes taking years) with gradual restoration of cell-mediated and humoral immunity function.
Immunosuppression
The fact that a primary role of the immune system is to distinguish between self and nonself presents a major problem for the transplant recipient: the immunologic response of the recipient to the donor’s tissues. In the person with an intact immune system (immunocompetence), the recipient’s immune system recognizes the transplanted tissue or organ as foreign (nonself) and produces antibodies and sensitized lymphocytes against it.
The ultimate objective of immunosuppressive therapy (see the section on Medications under Advances and Research in Transplantation in this chapter) is to block transplantation candidate reactivity to the donor’s organ while sparing other responses. Since these drugs suppress immunologic reactions, infection is a leading cause of death, particularly within the first postoperative year.192 Infection is still the leading cause of death in some transplant recipients. However, increased understanding of rejection mechanisms has made it possible to suppress specific elements of the immune response and has lead to a decrease in death-related infection and rejection.30,316
Although lower amounts of immunosuppressive drugs are now prescribed, these drugs must be taken for the life of the recipient and physical changes (see Figs. 11-5 11-6 11-7) and other side effects remain a well-known problem (see Table 5-3).
Side Effects of Long-Term Immunosuppression
Long-term immunosuppression can have serious consequences for the recipient, such as diabetes and accelerated hyperlipidemia, with associated atherosclerosis and subsequent cardiovascular disease.
There is a high incidence of musculoskeletal effects that will concern the therapist, such as decreased bone density and osteoporosis. Half of all transplant recipients are diagnosed with osteoporosis and one third have documented vertebral fractures,193 steroid-induced myopathies, as well as avascular necrosis and musculoskeletal injuries.
Within the first 6 months after transplantation, the organ candidate can lose more bone density than any woman during the postmenopausal period. Osteoporosis has become a silent contributor to mortality in organ transplantation.286 Physical therapy intervention must address this concern.
Neurotoxic reactions are manifested by a fine tremor, paresthesias, and occasionally seizures. Sensorimotor demyelinating polyradiculoneuropathy has been reported as a rare side effect in liver transplant recipients receiving tacrolimus (Prograf, FK506).195 Neuropathies and paresthesia can occur with FK506335; quadriplegia is a rare adverse event.
The individual may report difficulty completing fine motor activities of daily living (ADLs), such as poor handwriting and difficulty eating. These changes may be significant enough to stop some people from going out publicly or dining out in restaurants. Reports of memory loss may not be secondary to actual alterations in memory, but rather a decrease in executive function.69,293 Most of these events are dose related and reversible.
Cancers in Solid-Organ Transplant Recipients
Organ recipients have three times the incidence of various cancers, and some specific cancers are 100 times more frequent in the immunosuppressed population after transplantation than in the general population. Cancer incidence is proportional to immunosuppression drug levels.72 Cardiac transplant recipients have a higher incidence of cancer than do other transplant recipients, perhaps because of the higher levels of immunosuppression.
The most common tumors among transplant recipients (40% to 50% incidence) are squamous cell cancers of the lips and skin owing to the enhanced photosensitivity. Squamous cell carcinoma is often more aggressive than in nonimmunosuppressed people, with multiple sites of presentation and frequent recurrence.93
Skin cancers involving papillomavirus are the most frequent cancers observed in transplant recipients, occurring in half of the long-term survivors. The incidence of basal cell carcinoma is 10% higher than the general population, and the incidence of squamous cell carcinoma has been reported to be 250 times greater in orthotopic homologous transplantation recipients.148
Organ recipients are also at increased risk for some malignancies such as Kaposi’s sarcoma, non-Hodgkin lymphomas and other posttransplant lymphoproliferative disorders, soft tissue sarcomas, carcinomas of the vulva and perineum, carcinomas of the kidney, and hepatobiliary tumors.74,309
True incidence of cancer is difficult to determine given the different organ types being transplanted and inconsistent reporting to cancer registries. Most transplant recipients, however, have no increased risk of most cancers common in the general population, such as lung, breast, prostate, or cervical cancers. An increased risk for colon cancer remains controversial.311
Cancer is more common in those receiving kidney transplants than in the general population and more common than in comparable patients on dialysis. Malignant lymphomas occur 11.8 times more often in kidney transplant recipients compared with the general population. The majority of lymphomas occur after the first posttransplant year.235
It has been suggested that immunosuppressive agents may cause DNA damage and interfere with normal DNA repair mechanisms. Immune surveillance, which ordinarily prevents the growth and development of malignancies, may be impaired by certain immunosuppressive medications. Exactly why kidney transplantation is more affected by these factors than other organs remains unknown.161
There have been studies that have reported an increase in lung cancer in heart transplant recipients who have a history of smoking. There is an increased incidence in posttransplant lymphoproliferative disease in heart transplant recipients who underwent OKT3 induction therapy or use of antithymocyte globulin for rejection therapy.
Gastrointestinal Problems
Gastrointestinal complications of solid-organ transplantation have been well described in the literature. Disorders of the colon and rectum are a considerable source of morbidity, especially after heart and lung transplantation. Colorectal problems occur among 7% of lung transplant recipients, 6% of heart-lung transplant recipients, and 4% of heart transplant recipients. Major events include diverticulitis, perforation, and malignancy. More minor complications include polyps, pseudo-obstruction, and benign anorectal disease.113
Wound Healing
Advances in surgical techniques and immunosuppression have led to an appreciable reduction in postoperative complications following transplantation. However, wound complications as one of the most common types of posttransplantation surgical complications can still limit these improved outcomes and result in prolonged hospitalization, hospital readmission, and reoperation.210
Chronic immunosuppressive drug therapy impairs and prolongs wound healing, especially common among those organ recipients with diabetic or neuropathic pedal ulcers. The two most important risk factors for wound complications are immunosuppression and obesity. Other risk factors include surgical and/or technical factors (e.g., type of incision, reoperation, surgeon’s expertise), advancing age, diabetes mellitus, malnutrition, and uremia.210
Therapists should be involved in preventive management of wound complications; identifying and minimizing risk factors whenever possible is important. Therapists involved in wound therapy should inform their clients, members of the clients’ families, employers of clients, and third-party payers to expect longer times in healing plantar ulcers because of long-term immunosuppressive therapy.280
Total-contact casting remains a highly effective and rapid method of healing neuropathic pedal ulcers in diabetic immunosuppressed clients and transplant recipients, although it may take several weeks longer than it would for those individuals who were not immunocompromised. Transplant recipients who are immunocompromised appear to be no more at risk for wound failure complications when using total-contact casting as a treatment modality than those individuals without these additional variables.280
ORGAN TRANSPLANTATION AND EXERCISE, ACTIVITY, AND SPORTS
Whereas some people will have a period of only a few days of physical inactivity before transplantation (e.g., toxic liver failure), the majority of organ candidates will live with their diseased organs for a prolonged period of time, often years. By the time of organ transplantation, candidates usually have experienced a period of long-term ill health leading to end-stage organ failure accompanied by severe deconditioning and exercise intolerance.
Complications of long-term immunosuppressive therapy and the kind of organ that has failed will determine some of the problems an individual may face in relation to exercise, activities, and sports. Most transplantation candidates experience an impaired physical performance level that not only interferes with the ability to perform leisure-time exercise, but also often limits the ability to perform even simple physical tasks, such as climbing stairs.170
Weakness, dyspnea on exertion, and fatigue are often present, and there may be little motivation for exercise and sport. Finding an activity or exercise that the person can do successfully is the first step to initiating regular lifelong exercise.
Whether or not a potential candidate receives a transplant, therapy can be focused toward more function and improved quality of life. Exercise training increases work capacity as measured by increased oxygen consumption (VO2), increases efficiency of oxygen utilization in the muscles, normalizes distribution of muscle fiber types, increases aerobic metabolism with delays in the onset of lactic acid buildup, and promotes modulation of the parasympathetic nervous system with more sensitive baroreceptors.36,246 Exercise training also improves psychologic factors such as depression in pretransplant and posttransplant individuals.57,178,246
An assessment of transplant candidates must take into consideration daily life and daily activities, including potential return to work requirements. For example, a job that requires lifting requires assessment of cardiovascular compliance and hemodynamic stability during lifting, whereas someone at home must be safe in ADLs.
Pretransplant Activity and Exercise
It has been proposed that peripheral skeletal and respiratory (in the case of thoracic involvement) muscle work capacity is reduced before transplantation and contributes to the limitations of exercise seen in the posttransplantation population.329 Preservation of muscle strength before transplantation becomes impossible for some who are acutely ill. Muscular dysfunction attributable to detraining and deconditioning is common.181
While an individual waits on the transplant list, it is important that the candidate participate in an exercise program with the goal to promote functional mobility. Exercise programs should be individualized to focus on the needs of each person required to maintain function, self-control, and esteem.
Exercises should be functional, with an emphasis on strengthening the proximal muscles of the pelvis and the lower extremities, especially the gluteal and quadriceps muscles, as well as muscles of the shoulder girdle and trunk to support upper extremity function and accessory respiratory efficiency.
Weight training to maintain or increase muscular strength may help the candidate counteract the steroid’s targeting effects on muscle and adverse effects of immobility and chronic inflammatory effects.64,233,246 It has been reported by transplantation centers around the United States that transplantation candidates who take part in an exercise program before surgery are likely to recover more rapidly following transplantation. Researchers are beginning to publish data on exercise performance before and after transplantation.26,58,125,250
Posttransplant Activity and Exercise
After organ transplantation, the underlying pathophysiologic process returns to normal if the donor organ is functioning appropriately. For example, exercise perfor mance in individuals with heart transplants increases with respect to pretransplantation performance but remains subnormal and does not improve with time after surgery.26 The extent of recovery depends on the function of the transplanted organ, which in turn is determined by the quality and function of the organ implanted, the presence of any rejection or infection, and the development of other comorbidities.
Despite the pretransplant physical deconditioning and exercise limitations, transplant recipients can progressively return to a normal life with return to work and even safely participate in sporting activity and exercise.170 National and International Transplant Games, a multidisciplinary sporting event started in 1978, illustrates the degree to which organ candidates can return to exercise and sports. At the 2006 National Kidney Foundation sponsored games in Louisville, Ky., transplant recipients from all 50 states, with the oldest recipient being 84 years of age, participated to celebrate the gift of life and experience competition.
Regular exercise enhances quality of life and lowers the risk of cardiovascular disease, hypertension, and diabetes. This is especially important in transplant recipients because many immunosuppressive drugs can be atherogenic and diabetogenic.120,166,333 Standards for how soon after transplantation physical training can or should begin have not been uniformly established. It is recommended that physical therapy should begin on postoperative day 1, with the goal to mobilize out of bed as soon as medically stable. Physical training should begin as soon as the recipient is up and walking.
Despite the restoration of system function that allows most transplant recipients to return to an improved quality of life, including returning to work, having and caring for children, and participating in leisure recreational activities, a persistent limitation in peak aerobic and anaerobic capacity can be appreciated when compared with health-and age-matched normal subjects.329
There is a decrease in maximal and peak oxygen consumption (VO2), decrease in workload, earlier onset of anaerobic threshold, and lower VO2 at the anaerobic threshold. Heart transplant recipients have lower exercise capacity than other transplant recipients.329 There is evidence to suggest that recipients continue to have abnormalities in both central and peripheral chemoreflex mechanism along with the adverse effects of the immunosuppressive medications that contribute to prolonged deficits of exercise capacity.29,64
Exercise training after transplantation increases exercise capacity, improves endurance, and increases muscle strength, contributing to higher quality of life after transplantation.170,233,222,240,329 Physical activity and exercise may reduce or attenuate side effects of immunosuppression. Transplant recipients tolerate progressive exercise training and can achieve near-normal and even normal levels of function.241
Various exercises have been prescribed for transplant recipients. Studies have documented various training programs, including aerobic programs of low to high intensities, muscle endurance, and resistive training. It is difficult to make any specific conclusion about the perfect exercise program. The best recommendation that can be made is to prescribe a comprehensive exercise program that includes muscular strength and endurance training, restoring functional mobility and improving cardiopulmonary endurance.98
Research shows that 6 months of specific resistance exercise training (weight training) restores fat-free mass to levels greater than before treatment and dramatically increases skeletal muscle strength. Resistance exercise, as part of a strategy to prevent steroid-induced myopathy, is safe and should be initiated early after transplantation.
Gaining density in the lumbar spine is especially important because up to 35% of transplant recipients develop lumbar spine bone fractures.39 Resistive training has been shown to restore bone density to pretransplant levels compared with an additional 6% loss in subjects who did not participate in resistance training. Marked increase in muscle mass, strength, and exercise capacity was also observed.291
Guidelines for Activity and Exercise
Whether assessing aerobic, anaerobic ability, or ADLs (Table 21-4), measurements of vitals, including blood pressure, heart rate, oxygen saturation, respiratory rate, and rate-pressure products (heart rate and systolic blood pressure), can provide valuable information and can be used as measurable outcomes of treatment intervention.
Table 21-4
Exercise Guidelines for Organ Candidates and Recipients*
|
• Select an enjoyable activity or exercise and always have a goal! Some centers target a long-term goal by organizing an annual fun run/walk, or join one already organized. • Include adequate warmup, stretching, and cool-down periods. • Progress activity or exercise as described in text. • Include interval training, aerobic activity, strength training, and conditioning. • Combine activities and/or exercise program with energy conservation techniques (see Box 9-8). • Maintain a normal breathing pattern; breath holding may contribute to excessive elevation in blood pressure and produce associated symptoms such as dyspnea and lightheadedness. • Exercise 4 to 5 days a week; allow 24 to 48 hours recovery time after strenuous activity and 48 hours after moderate to vigorous resistance training for those involved muscles. • Aerobic exercise should be completed 5 days a week for 30 minutes at a moderate intensity level. |
| Endurance Training: High Repetitions/Low to Moderate Resistance |
| Weight Training: Low Repetitions/More Resistance |
| Weight training has become an acceptable component of a comprehensive exercise program. It is recommended to begin strength training at low-to moderate-intensity levels along with a progressive aerobic and stretching program. It is important to assess blood pressure response during exercise and evaluate for signs and symptoms of right-sided heart failure in the presence of pulmonary hypertension. The recipients should also be monitored for hemoptysis, overuse injuries, poorly regulated glucose, electrolyte and nutritional imbalances, and surgical precautions. Anyone with documented pulmonary hypertension may have the following associated signs and symptoms (lightheadedness, dizziness, angina-like pain, decreased cardiac output with exercise, or development of abnormal heart sounds with exertion) needs to be supervised during low-level interval exercises. |
| Once the person is medically stable and basic level of function is restored, the therapist can more accurately prescribe a supervised exercise program by completing a symptom-limited aerobic exercise test as well as a 1 or 3 repetition maximum. The therapist must take into account any significant impairments present and the effects of long-term steroid use, especially muscle wasting, osteoporosis, and coagulopathy. Risk for injury is higher in this group when using one repetition maximum. |
|
• Use weights that are 40% to 60% of the one repetition maximum.† • Perform 3 to 8 repetitions per set. • Perform 3 to 5 sets of each exercise per workout. • Perform each exercise through a functional range of motion. |
| Maintain or Monitor | Terminate Exercise if… |
| Allow only minimal dyspnea; respiratory rate should be <30 breaths/minute with minimal rales heard on auscultation | Respiratory rate >40 breaths/minute with increased rales |
| Allow only mild level of fatigue; use rate of perceived exertion (Borg scale; see Table 12-13) | 3/10 on Borg scale |
| Maintain stable vital signs (HR, blood pressure); maintain stable cardiac output (rate/pressure product, pulse pressure) | HR exceeds target zone, decrease in SBP, pulse pressure narrows (SBP –– DBP), decrease in rate/pressure product (RPP = HR × SBP) |
| Maintain stable electrocardiogram | Increased incidence of arrhythmias or perceived palpitations |
| Maintain central venous pressure (CVP) | Monitor in the presence of right-sided heart failure, maintain CVP >20 mm Hg; terminate exercise relative to other symptoms |
| Maintain pulmonary arterial pressure (PAP) | Rest is indicated if PAP rises >5 mm Hg; terminate exercise if PAP rises and persists after rest and/or in the presence of other symptoms |
| Maintain oxygen saturation >90% (this is individually determined by each center according to each person’s medical status) | Oxygen saturation <90% (or saturation below prescription) |
| Monitor for signs of bleeding | See Tables 40-8 and 40-9 |
| Observe client response to exercise; allow moderate level of dyspnea | Change in mental status (e.g., confusion, hostility), onset of pallor or diaphoresis, client request |
HR, Heart rate; SBP, systolic blood pressure; DBP, diastolic blood pressure.
*Guidelines for exercise are modified for the organ recipient but follow the ACSM’s Guidelines for Exercise Testing and Prescription. These are only guidelines; each exercise program must be individually tailored to the organ recipient’s condition and comorbidities. Progression must be according to tolerance.
†Intensities of 80% to 100% have been shown to produce the most rapid gain in muscle strength within the normal population. However, because of the possibility of overtraining or injury in the pretransplant or posttransplant population, caution must be used when overloading a muscle or muscle group.4
In general, as the intensity of activity increases, the heart rate and systolic blood pressure increase, with a concomitant return to baseline with cessation of activity (see Appendix B). The response the transplant recipient has to exercise will depend on the type of transplant, medications taken, and present level of fitness. For example, heart transplant recipients typically have a blunted heart rate response with exercise due to the denervated state of the heart.
Monitoring the recovery may be an objective measure of improvement in physical capacity. Consistent abnormal responses should be reported to the physician for further evaluation. Other considerations are determined according to the underlying pathologic condition (e.g., cardiomyopathy, congestive heart failure, renal failure, diabetes, cirrhosis) and pretransplant treatment (e.g., VADs, medications, dialysis). See Special Implications for the Therapist boxes for each specific diagnosis in this chapter.
For all transplant candidates and recipients, the duration of beginning aerobic exercise should be until fatigue begins; allow for a short recovery period and repeat in an interval manner until the duration is at least 20 minutes of continuous exercise. The goal is to perform at least 30 minutes of nonstop activity daily at a moderate exertional level before reducing exercise frequency to four to five times weekly. Individuals trying to control blood pressure or lose weight should work for a longer duration, 45 to 60 minutes, at a lower intensity (e.g., 50% to 65% of predicted maximal heart rate).98 The exercise program should consider the recipient’s comorbidities and be individualized to the needs and goals of the transplant recipient.52,166,167
Limitations on Activity and Exercise
Transplant trauma is a theoretic possibility, so organ recipients are advised not to participate in contact sports. Except for this general broad precaution, limitations on sporting and exercise must be evaluated on a case-by-case basis and may be determined by the course of the illness. For example, the person who undergoes emergency liver transplantation for acute liver failure will have relatively little secondary damage.
On the other hand, someone with chronic renal failure can develop renal osteodystrophy (see Fig. 18-7 and the section on chronic renal failure in Chapter 18), decreased bone density, osteoporosis, reduced peak cardiac output (because of the arteriovenous fistula required for vascular access), and irreversible neuropathies and myopathies. Anyone experiencing renal failure secondary to diabetes will have multiple other secondary complications (see the section on Diabetes in Chapter 11).
Many other potential limitations on sporting and exercise must be recognized and evaluated, such as the condition of the recipient at the time of transplantation and the type of organ that has failed. For example, people with severe pulmonary disease necessitating heart-lung transplantation often experience malnutrition and muscle wasting before transplantation. Liver failure can cause abnormalities of lung function, including ventilation/perfusion mismatching, pulmonary hypertension, and loss of oxygen-diffusing capacity.
Denervation of the transplanted heart, pancreas, liver, or kidneys results in a loss of sympathetic nerves to the organ (e.g., loss of vagal response in the heart, impaired insulin in the pancreas, and altered renin responses in the kidney) requiring some modifications in the exercise program. In contrast, surgical removal of sympathetic liver nerves does not inhibit hepatic glucose production during exercise, and denervation of the lungs does not impair the ability to increase ventilation during physical exertion.170
The denervated lung will experience reduced tidal volumes and decreased lung compliance. There is a delay in the bronchodilation response requiring an extended warmup period in order to obtain the catecholamine response necessary for organ vasodilation and therefore increased tidal volume during exercise.
There is evidence that reinnervation does occur to some extent for some recipients. For heart transplant recipients who have some degree of autonomic nervous system function restored, there is an increase in heart rate greater than 35 beats/minute with peak exercise levels and also an immediate decrease in heart rate after exercise. This restoration results in an increase in exercise capacity that is closer to healthy age-matched control subjects.255
Besides the usual exercise-related risks anyone faces, recipients have additional medication-related risks associated with the long-term immunosuppressive therapy, including exaggerated hypertensive response, myopathies, neuropathies, osteoporosis, and fractures.
The adverse effects of the immunosuppressive medications on skeletal muscle, including the muscle-wasting effects of glucocorticoids, are well-known. It is documented that quadriceps strength of renal transplant candidates is only 70% of normal, although this side effect can be counteracted by resistance exercise training.137,138,255 Other potential side effects are listed in Tables 5-3 and 5-4; see also the section on Immunosuppression under Posttransplant Complications in this chapter.
Finally, transplanted organs may be exposed to chronic rejection, limiting the function of the organ. With the decline in organ function there is a decrease in exercise tolerance. For example, in heart transplant recipients chronic rejection is associated with a decrease in cardiac output, onset of heart failure, and accelerated atherosclerosis.324
With lung transplantation, the recipient suffering from chronic rejection may present with an impairment in gas exchange, leading to desaturation and increased air trapping and work of breathing. However, despite all these variables, the benefits of exercise in maintaining a healthy lifestyle and sense of well-being are much greater than the risks imposed by organ transplantation. Although it is assumed that transplant recipients will spontaneously increase their physical activity after transplantation, fear of harming the new organ or protective family members may discourage vigorous activity.
As a member of the transplantation team, the therapist should encourage a program of regular activity immediately after transplantation and provide exercise guidelines as a part of the long-term transplantation care plan. It is recommended that the recipient be referred for supervised outpatient services since many studies have documented an increase in recovery with supervised exercise as opposed to a home exercise program.291
Vigorous exercise training for competition is not contraindicated for healthy transplant recipients. However, cardiorespiratory fitness and strength training should progress gradually before the client engages in more strenuous sports participation. Exercise should be reduced in duration and intensity but not necessarily discontinued during rejection episodes.240
HEMATOPOIETIC CELL TRANSPLANTATION
Hematopoietic stem cell transplantation (HSCT) is defined as any transplantation of blood-or marrow-derived hematopoietic stem cells, regardless of transplant type (allogeneic or autologous) or cell source (bone marrow, peripheral blood, or placental or umbilical cord blood).219
HSCT, the collection and engraftment of hematopoietic stem cells from the bone marrow (and now also from the peripheral blood and umbilical cord blood) to cure a series of diseases previously considered incurable, is one of the most dramatic developments in the last 30 years.
Initial attempts to transfer techniques derived from animal studies to human beings failed until an understanding of HLA and tissue typing made it possible to select compatible sibling donors and now unrelated donors. The genes for the HLA, the human major histocompatibility complex, are located on the short arm of chromosome 6; siblings have a 25% chance of being a match.
Nonrelated candidates have less than a 1 in 5000 chance of having identical HLA types. Graft rejection and GVHD rates are inversely related to the degree of HLA compatibility. The National Bone Marrow Donor Registry types people as potential donors for unrelated people who require transplantation.
HSCT is an accepted form of treatment for people who require very high-dose chemotherapy or radiation therapy to treat their disease, usually a type of cancer. The chemotherapy and/or radiation therapy results in severe injury to blood cells formed in the bone marrow (marrow ablation). In order to restore the person’s ability to make blood and immune cells, stem cells from a compatible donor (or from the recipient) can be administered.
Stem cells are very immature cells that can develop into any of the three types of blood cells (red cells, white cells, platelets) (Fig. 21-6). Bone marrow is used for such transplants because it contains these undifferentiated stem cells. Blood also contains stem cells but in such small numbers that the cells cannot be counted or identified in ordinary blood tests. New procedures have made it possible to induce stem cells to leave the marrow and enter the blood, where they are collected or harvested for administration to a candidate in need of HSCT. Stem cells are also collected from umbilical cord and placental blood.
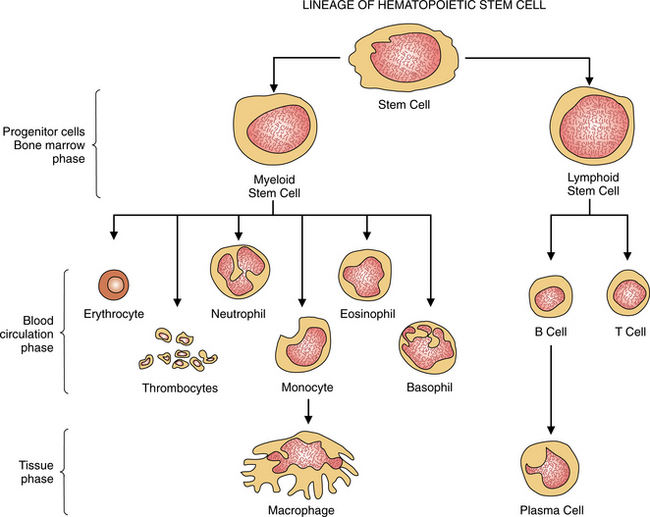
Figure 21-6 A stem cell is any precursor cell or “mother” cell that has the capacity for both replication and differentiation, giving rise to different blood cell lines. This figure shows the origin, development, and structure of thrombocytes, leukocytes, and erythrocytes from pluripotent (able to develop into different cells) stem cells. The process of hematopoiesis (formation and development of blood cells) usually takes place in the bone marrow.
Indications for Hematopoietic Cell Transplantation
Today, transplantation of stem cells from peripheral blood, bone marrow, or cord blood is the treatment of choice for a variety of chemotherapy-sensitive hematologic malignancies, solid tumors, syndromes of bone marrow failure, genetic diseases and, more recently, autoimmune disorders. HSCT (peripheral blood stem cell transplantation and BMT) enables individuals to survive high-dose chemotherapy via “rescue” infusion of new, healthy bone marrow elements or stem cells.109
In 1998 breast cancer was the most common indication for HSCT in North America, accounting for nearly one third of all transplantation procedures. However, subsequent reports have concluded that high-dose chemotherapy plus autologous BMT does not improve survival in women with metastatic breast cancer.24,287
Non-Hodgkin lymphoma was the second most common indication, followed by acute myelogenous leukemia, multiple myeloma, and chronic myelogenous leukemia (Fig. 21-7). Reduced transplant-related mortality rates have led to a widening of indications for HSCT, but based on the experience in the use of stem cell transplantation with metastatic breast cancer, ongoing research is essential to verify the efficacy of this treatment modality for each individual disease entity.208
Figure 21-7 The most common indications for allogeneic and autologous transplantation differ. Allogeneic transplantation is the predominant approach for acute and chronic leukemias; myelodysplasia; and nonmalignant diseases such as aplastic anemia, immune deficiencies, and inherited metabolic disorders. Autologous transplantation is used for ovarian and other solid malignancies, as well as Hodgkin and non-Hodgkin lymphomas and multiple myeloma. Routine use of this transplantation for breast cancer has been discontinued and remains under investigation. (Courtesy Center for International Bone & Marrow Transplant Research (CIBMTR). CIBMTR Newsletter 12[1], May 2006.)
Sources of Hematopoietic Cell Transplant
The three types of peripheral blood or bone marrow transplantation are determined by the source of the donation: syngeneic, autologous, and allogeneic. The major sources of transplantable hematopoietic stem cells come from human bone marrow, peripheral blood, and umbilical cord blood.328 As with all other transplantations, syngeneic involves peripheral blood or bone marrow from an identical twin with identical HLAs on the cell surfaces.
Autologous transplants use the person’s own stem cell sources and must be cancer free by harvesting during remission or after cancer cells have been killed (Fig. 21-8). The person who donates bone marrow for his or her own use does not have cancer in either the bone marrow or the blood cells. This autologous BMT greatly reduces the occurrence of side effects from the transplant.
Figure 21-8 Approximately 90% of autologous transplantations use only hematopoietic progenitor cells collected from blood. The remainder use bone marrow alone or in combination with cells collected from blood. (Courtesy Center for International Bone & Marrow Transplant Research (CIBMTR). CIBMTR Newsletter 12[1], May 2006.)
Allogeneic transplants require a donor with closely matched HLAs (usually a sibling); allogeneic sources for stem cell transplantation are primarily derived from bone marrow, with a smaller but steady increase in the number of allogeneic transplants coming from peripheral blood and cord blood (Fig. 21-9). Autologous blood stem cells are now widely used in place of allogeneic BMT, although allogeneic transplantation remains the only curative therapy for inherited disorders of metabolism and chronic myelocytic leukemia and the most effective treatment for severe aplastic anemia.
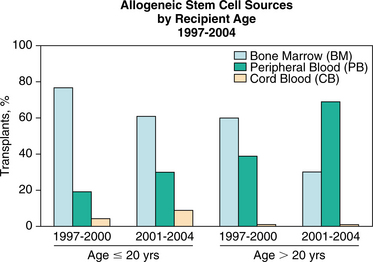
Figure 21-9 Most allogeneic transplants use bone marrow grafts. There has been a steady increase in the number of allogeneic transplantations using cells collected from blood. Although use is increasing, there are still relatively few transplantations using umbilical cord blood cells. (Courtesy Center for International Bone & Marrow Transplant Research (CIBMTR). CIBMTR Newsletter 12[1], May 2006.)
Unlike allogeneic transplantation, autologous BMT can be performed in older people with relative safety owing to the freedom from GVHD as a complication. A primary concern with autologous BMT is the possible presence of viable tumor cells in the graft. Numerous methods have been developed to remove contaminating tumor cells from the graft in a process referred to as purging.
Peripheral blood has largely replaced bone marrow as a source of stem cells for autologous recipients. A benefit of harvesting such cells from the donor’s peripheral blood instead of bone marrow is that it eliminates the need for general anesthesia associated with bone marrow aspiration. The most striking advantage of blood stem cell over bone marrow cell transplants is the shortened period of total aplasia of bone marrow after chemotherapy. This reduction in the period of aplasia may allow repeated marrow ablative treatment cycles in solid tumors, thereby reducing mortality. In addition, blood-derived grafts may contain fewer malignant cells than the bone marrow cells.
Since the late 1980s umbilical cord blood has been harvested to assist people in need of stem cells previously only available through BMTs. Umbilical cord blood is the blood that remains in the umbilical cord and placenta after a baby is born. It is derived from the portion of the umbilical cord that has been cut and is usually thrown away. Like bone marrow, cord blood has been found to be a rich source of stem cells and may provide treatment advantages over bone marrow, especially if it comes from an immediate family member.
Family members wishing to store their newborn’s cord blood for their own potential use can do so for a fee. However, anyone with a family member who has a condition for which stem cells may be a treatment option can store cord blood at no cost through the Cord Blood Registry’s Designated Transplant Program. Contact the Cord Blood Registry at (888) 267-3256, (888) 932-6568, or www.cordblood.com to learn more about banking cord blood. Cord blood banking is the process of collecting the blood immediately after delivery and freezing it for long-term cryogenic storage.
The potential advantages of cord blood stem cell transplantation over BMT include a large potential donor pool, rapid availability since the cord blood has been prescreened and tested, no risk or discomfort to the donor, rare contamination by viruses, and lower risk of GVHD.
Potential disadvantages are primarily related to the many unknown variables with a new, experimental procedure, such as the possibility of genetic or congenital disease transmission by stem cells, uncertain long-term success using cord blood stem cells, and a longer period of time for treatment to be effective compared with cells from adult blood or bone marrow, leaving the recipient at risk for infection. It remains unknown how long cord blood can be stored without losing its effectiveness.
Transplantation Procedure
HSCT involves the intravenous infusion of hematopoietic progenitor cells from the patient (autologous) or an HLA-matched donor (allogeneic). Before transplantation, the recipient undergoes a conditioning regimen with high-dose chemotherapy or radiotherapy (or both) to destroy defective bone marrow or residual cancer cells.268
Allogeneic transplant recipients remain hospitalized until their absolute neutrophil count exceeds 1500/mm3 for 48 hours (normal values are 3000 to 7000/mm3). Autologous transplant recipients may be managed as outpatients. All transplanted individuals are expected to remain geographically near during the first 100 days after transplantation, when the incidence of acute rejection is the greatest, and to use reverse isolation (mask and glove wear) until they reach an acceptable neutrophil level.109
Bone Marrow
The BMT process consists of several phases: conditioning, harvest, marrow infusion, preengraftment, and engraftment. Conditioning refers to the immunosuppression treatment regimen of chemotherapy, radiation therapy, or both used to eradicate all malignant cells, provide a state of immunosuppression, and create space in the bone marrow for the engraftment of the new marrow. Conditioning occurs 7 to 10 days before the actual BMT takes place.
Bone marrow is withdrawn from the donor by needle aspiration from the iliac crest or sternum on an outpatient basis and does not require surgical intervention. The donated marrow is usually infused through an intravenous line 48 to 72 hours after the last dose of chemotherapy or radiation therapy.
Peripheral Blood Stem Cells
Peripheral blood stem cells are harvested from the person’s own stem cells through a process referred to as leukapheresis. Leukapheresis, or hemapheresis, is the process of removing a donor’s blood to extract a specific component and returning the unneeded parts to the donor. The process uses continuous circulation of blood from a donor through an apparatus and back to the donor, making it possible to remove desired elements from large volumes of blood. Besides stem cells, this process can separately harvest platelets, red blood cells, white blood cells, or plasma. The infused cells usually begin producing new blood cells in the marrow within a few weeks, and immune function returns within 1 to 2 years in successful transplants.
Cord Blood
Collection of cord blood takes place immediately after the umbilical cord is clamped after birth. After the baby is removed from the delivery area, a needle is inserted into the umbilical vein and blood is withdrawn and collected by gravity drainage. A standard blood collection bag is used, containing nutrients and an anticoagulant solution to keep blood from clotting.
An alternate method involves collecting the cord blood after the child is delivered but before the placenta is delivered. This collection method may be advantageous because it allows earlier collection before the blood has a chance to clot, and it uses the contractions of the uterus to enhance blood collection. This second technique is more intrusive and may potentially interfere with the mother’s care after delivery.
After collection, the bag of cord blood is immediately transported to a facility for testing (e.g., HLA typing, identification of any viruses) and preservation. The blood is then frozen and held in liquid nitrogen at very low temperatures for future use. At the time of transplantation, the cord blood is thawed and infused through a vein into the recipient.
Umbilical cord blood stem cells are different than other types of stem cells. Umbilical cord blood stem cells are the “youngest” safely available stem cells and the product of a live birth. Freezing these cells essentially stops the clock and prevents aging and damage that may occur to the cells later in life.
Another source of stem cells, embryonic stem cells, has been at the heart of heated debate.186 Currently, embryonic stem cells are not being used to treat human beings. A third category of stem cells is adult stem cells, such as those found in bone marrow as previously discussed. Adult stem cells serve very specialized roles in children and adults and are not as proliferative as those found in cord blood.
Complications of Hematopoietic Cell Transplantation
Many people entering transplantation have already developed significant multisystem depletion, including cardiopulmonary, nutritional, musculoskeletal, and neurologic impairment related to the underlying disease. Bone metastasis, steroid myopathy, polyneuropathies, depleted protein stores, and impaired skin integrity are common comorbidities before HSCT transplantation (Box 21-8).
The high-dose chemotherapy and radiation typically used as the preparative regimen for HSCT produces considerable morbidity and mortality. Virtually all HSCT recipients rapidly lose all T and B lymphocytes after con- ditioning, losing immune memory accumulated through lifetime exposure to infectious agents, environmental antigens, and vaccines.
Transfer of donor immunity to HSCT recipients is variable and cannot be relied on to provide long-term immunity against infectious diseases. Cognitive slowing, impaired memory, and impaired executive functions are also seen in people following interferon-alpha treatments and whole-brain irradiation.323
Neurologic complications occur in accordance with the stage of HSCT. For example, during conditioning, drug-related encephalopathies and seizures or complications secondary to medical procedures are possible. During bone marrow depletion, metabolic and drug-related encephalopathies and seizures, septic cerebral infarctions, and hemorrhages may occur. Chronic immunosuppression results in infections by viruses and opportunistic organisms, and late events such as central nervous system relapses of the original disease, neurologic complications of graft versus host disease, and second neoplasms are observed. The frequency and type of neurologic complication depends on the type of HSCT and the underlying disease.268
Cardiac or pulmonary toxicity also may occur as a result of the irradiation and immunosuppressive drugs used to prepare candidates for the transplantation. Interstitial pneumonitis, an inflammation of the lungs, is a common complication, especially among allogeneic transplantations, which leaves the person susceptible to obstructive small airway disease. Arrhythmias may be the first sign that a chemotherapeutic agent is becoming cardiotoxic.
Infections and hemorrhagic complications during the period of bone marrow anaplasia and emerging immune competence and GVHD are the most life-threatening complications of BMT, and they may be fatal. Rejection of allogeneic transplantation resulting in infection, relapse, or GVHD is often fatal. Infections include early bacterial infections or later opportunistic infections, especially CMV interstitial lung disease. EBV-associated lymphoproliferative disorders (posttransplantation lymphoproliferative disorders) are a significant problem after stem cell transplantation from unrelated donors or mismatched family members.130
Recurrence of malignancy is always a possibility. Other complications of BMT include sterility, cystitis, cataract formation, cardiomyopathy, and veno-occlusive liver disease. Neuromuscular changes, such as peripheral neuropathies, muscle cramping, and steroid myopathies, may also develop.
The complications of PBSCT are essentially the same as those of BMT, but hematologic recovery after PBSCT is much more rapid (10 to 12 days and as early as 7 days), thereby significantly shortening the period of postchemotherapy neutropenia (decreased neutrophils, a type of granular leukocyte used to fight infection) and thrombocytopenia (decreased platelets in peripheral blood). This, in turn, reduces platelet and red blood cell transfusion and facilitates earlier discharge from the hospital. In most cases, PBSCT can be performed on an outpatient basis.
Prognosis
Enormous progress has been made in understanding the biology, therapy, and prophylaxis strategies of transplantation and in extending the range of potential bone marrow donors to include unrelated people. Dramatic advances have occurred in the prevention of serious infection, including CMV infection, formerly a significant cause of mortality.
One-hundred-day mortality, defined as death before 100 days after transplant, is often used as a gauge of procedure-related toxicity. Allogeneic transplants are associated with relatively high risks of GVHD, failure of engraftment, infections, and liver toxicity, resulting in high early mortality. Long-term survival rates are improved with BMT, but this type of transplantation does not confer a normal life span.285,305
Unfortunately, toxicities of conventional transplantation remain a major limitation to successful application of the procedure. Primary disease recurrence continues to account for the majority of deaths in autologous transplant recipients. New cancer, organ failure, and suicide are also cited as causes of death among long-term survivors after BMT. The incidence of suicide is higher among such survivors than among healthy subjects.216
Despite high morbidity and mortality after HSCT, recipients who survive long term are likely to enjoy good health. For those who survive more than 5 years after HSCT, 93% are in good health and 89% return to work or school full time. Eighty-eight percent of those who survive 10 years after HSCT report that the benefits of transplantation outweigh the side effects.289
Although the success rates of transplantation have been improving over time, the prognosis still depends on the underlying disease, delays in transplantation, associated risk of relapse (e.g., leukemia) and current remission state, the development of GVHD based on the level of match between donor and recipient, and the age of the recipient. There is no effective therapy for severe GVHD, and people stricken with it rarely recover.
The success of allogeneic marrow grafting is inversely proportional to the age of the recipient. Most marrow transplant centers do not perform transplantations in anyone older than 50 years, but some groups do make treatment decisions on the basis of a person’s estimated physiologic age rather than chronologic age. These age restrictions do not apply to syngeneic or autologous transplants because these people will not develop GVHD. However, people older than 60 years do not tolerate intensive treatment as well as younger people.
Future Trends
The development of techniques to achieve a state of chimerism and continued research efforts toward improved tolerance and elimination of malignant cells will lead to a wider application of HSCT in the coming decade.304 Infusing donor bone marrow as an adjunct to solid-organ transplantation in order to prevent organ rejection is being investigated with animal studies.249 This would open up an entirely new application of BMT and potentially alter the need for exogenous immunosuppression.
Efforts to reduce acute and long-term side effects of the high-dose conditioning regimens currently required to control the malignancy and prevent graft rejection may extend the use of allografts for people older than 55 years or for younger people with certain preexisting organ damage.56 Transplantation using less toxic preparations may also make it possible to treat autoimmune diseases in the years ahead.
Researchers have located a master (stem) cell in the bone marrow of rats that will convert itself into functioning liver tissue cells under special conditions.248 Preliminary studies from the same laboratory have also isolated a stem cell that converts into pancreatic cells. Other laboratory research has found in bone marrow the stem cells for bone, cartilage, tendon, muscle, and fat.150,253 Although this work has been only demonstrated in laboratory animals, it is a significant step toward learning how to regenerate human organs.
Swedish scientists isolated and identified for the first time the neural stem cell responsible for forming adult brain cells. This finding supports the idea that brain and other neural tissues regenerate despite conventional belief that these tissues do not regenerate.154,155,217 Manipulation of these cells could open the door for replacing damaged neural tissues in spinal cord injuries and other neurologic conditions, such as multiple sclerosis, Huntington chorea, and Parkinson’s disease.
ORGAN TRANSPLANTATION
Overview
The first kidney transplantation was done in 1954 by Joseph Murray, MD, resulting in a successful graft and providing the recipient with an additional 8 years of life. It was the first organ transplantation of any kind ever performed, with the success credited to the live donor (adult twin brother). Kidney transplantation remains the most common type of solid-organ transplant (see Table 21-1).
As people over the age of 65 years become the fastest growing segment of the population, the number of cases of end-stage renal disease (ESRD) requiring kidney transplantation will continue to increase. Diabetes is now the most common cause of ESRD. Almost half of adults undergoing transplantation already have diabetes.
Renal replacement remains the most successful form of treatment for ESRD and, in the case of diabetes, offers an opportunity to eliminate dependence on dialysis and exogenous insulin. Simultaneous kidney-pancreas transplantation has become a safe and effective method to treat advanced diabetic nephropathy and results in stable metabolic function, reduced cholesterol, and improved blood pressure control.297
Until recently, almost all candidates for renal transplantation had been treated for months or years with hemodialysis, but now it is possible to plan a kidney transplant before the complete shutdown of the kidney or kidneys, avoiding dialysis completely. Studies have shown that when dialysis is used, peritoneal dialysis is associated with a lower incidence of delayed graft function and may be preferred over hemodialysis.29,327
Continuous ambulatory peritoneal dialysis is a maintenance system of dialysis in which an indwelling catheter permits fluid to drain into and out of the peritoneal cavity by gravity. The individual remains able to complete this type of dialysis three or four times per day while at home rather than coming to a hemodialysis clinic three or four times per week for 3 or 4 hours at a time while the blood is filtered through a dialysis machine.
Receiving a kidney transplant without a prior history of hemodialysis or peritoneal dialysis presents some problems in adjustment for those people who, never having experienced the intrusion of dialysis, must now learn to live with the side effects of antirejection drugs and potential complications of surgery. Whereas the person on dialysis may see transplantation as a welcome relief, recipients who have never been treated with dialysis may be more likely to view the surgery and recovery as an ordeal and therefore experience more difficulties in adjustment.
Indications and Incidence
The primary indication for renal transplantation is type 1 diabetes with ESRD, which occurs in over one third of all cases of type 1 diabetes.197 Cardiac autonomic neuropathy occurs as a result of uremia and diabetes with severe cardiac dysfunction when these conditions occur at the same time. Both kidney transplantation and kidney-pancreas transplantation result in improved cardiac autonomic function and modulation of heart rate.59
There are now more than 50,000 people on the waiting list for cadaveric kidney transplant, but only 14,000 of these procedures are performed yearly in the United States.321 A more unusual indication would be polycystic kidney disease, especially when combined with polycystic liver disease.
Transplant Candidates
As mentioned, people with type 1 diabetes and ESRD can choose dialysis or transplantation for renal replacement therapy. Those individuals with type 1 diabetes younger than 45 years with little or no atherosclerotic vascular disease are ideal candidates for a combined kidney-pancreas transplantation. The addition of a pancreas transplant is associated with greater morbidity and may require higher levels of immunosuppression but can result in stabilization of neuropathy and improved quality of life.197
Some renal transplant candidates are at high risk for cardiac events, sometimes fatal. Analysis of these clinical risk factors (including age at least 50 years, type 1 diabetes mellitus, and abnormal electrocardiogram) may assist in identifying candidates who may be at risk for cardiac death.188 For those choosing transplantation, a kidney from a living related donor is associated with longer graft and individual survival.
Transplantation Procedure
Kidney grafts may be positioned intraperitoneally, anastomosed to the iliac vessels, and then drained into the bladder (Fig. 21-10); extraperitoneally in the iliac fossa through an oblique lower abdominal incision; or in small children, retroperitoneally with a midline abdominal incision.
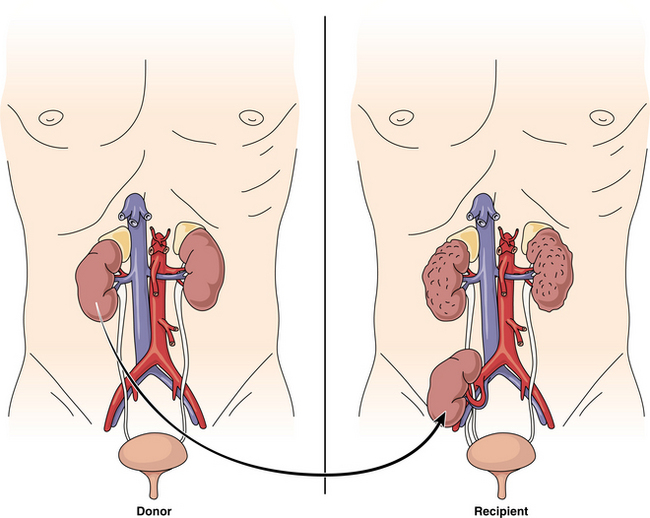
Figure 21-10 In a minimally invasive live-donor kidney transplantation, surgeons remove the donor’s kidney through a 3-to 4-inch incision below the donor’s umbilicus. For the recipient, blood vessels of the donor kidney are attached to the major abdominal blood vessels. The ureter of the donor kidney is attached to the recipient’s bladder. The donor kidney begins working immediately. The recipient’s own malfunctioning kidney is not always removed.
More recently a procedure called laparoscopic nephrectomy has been introduced to remove the live-donor kidney. Four small incisions called ports are made in the abdomen. The ports allow the surgeon to insert the laparoscope and other instruments used in the procedure to clamp off arteries and the ureter and cut the kidney loose.
Complications
As with other solid-organ transplantation, renal recipients experience graft dysfunction, organ-related infection (nephrotoxicity), and graft rejection as the three most common complications. Surgical complications include renal artery thrombosis, urinary leak, and lymphocele, although chronic rejection accounts for most renal allograft losses after the first year after transplantation. Donor organ quality, delayed graft function, and other donor and recipient variables leading to reduced nephron mass are nonimmunologic factors that contribute to the progressive deterioration of renal graft function.218
Cardiovascular and cerebrovascular diseases are major causes of morbidity and mortality after kidney transplantation. Extensive carotid vascular wall abnormalities increase significantly despite kidney and pancreas transplantation in those individuals with type 1 diabetes mellitus and progressive uremia.
Although initiation of plaque development is related to systemic factors, progression of established plaque is largely influenced by local factors within the arterial wall and therefore unaffected by organ transplantation.223 In fact, research has shown an association between CMV infection and atherosclerotic plaque formation in coronary heart disease, a finding that has also been found in posttransplant cardiac complications in kidney recipients with CMV.142
Other complications may include renal dysfunction with prolonged use of cyclosporine, hypertension, lipid disorders, hepatitis, cancer, and osteopenia. Hypertension occurs in up to 80% of renal graft recipients; approximately 15% of graft recipients develop chronic hepatitis. There is a high degree of impaired bone formation associated with renal grafts, resulting in severe osteoporosis when compared with other organ transplantations.
Basal cell and squamous cell carcinoma, Kaposi sarcoma, lymphomas, and posttransplant lymphoproliferative disease are 20 times more frequent in this population. Kidney cancer is 15 times more common after kidney transplantation compare with the general population. Melanoma, leukemia, hepatobiliary tumors, and cervical and vulvovaginal tumors are five times more likely compared with the general population. Testicular and bladder cancers are three times more common.161
Pelvic congestion syndrome can occur in a kidney donor or recipient when removal of the kidney ligates the ovarian vein. Retrograde flow in the ovarian vein causing ovarian varicosities (varicose veins of the ovaries) with venous stasis produces congestion and chronic pelvic pain in some women.20 Imaging studies have verified the fact that there are very few venous valves in the blood vessels of the pelvic area.91,133,300 Any compromise of the valves or blood vessels in the area can result in pelvic congestion syndrome.
Prognosis
Long-term renal transplant survival has improved from 23% (graft) and 36% (recipient) for cadaveric donor in 1986 to 92% and 93%, respectively, in 1996.321 The long-term outcome of simultaneous kidney-pancreas transplant recipients is not well established yet and ranges from 79% for a 1-year graft survival rate to 5-year survival rates for combined kidney-pancreas transplant, kidney transplant, and pancreas transplant reported as 95%, 85%, and 88%, respectively.297
Future Trends
The renal research community has made great strides in improving client outcomes on dialysis and following organ transplantation. However, only a small fraction of individuals with ESRD undergo transplantation because of a lack of donor organs, requiring continued research to find alternative methods of successful treatment. Alternately, preserving donor organs more effectively may ensure better graft survival rates. Since both glomerular and tubular functions are inhibited at temperatures below 18° C, efforts to develop organ preservation techniques at warmer temperatures are underway.294
Current research continues to examine the possibilities of reducing cyclosporine dosage through the concomitant use of sirolimus without increased infections or neoplasms developing and with the potential for reducing the incidence or progression of chronic rejection. Other efforts to reduce the incidence of acute rejection episodes include assessment of a chimeric monoclonal antibody.158
The search for adult renal stem cells for cell-based treatment is ongoing.40 At the same time, a bioartificial (extracorporeal) kidney has been successfully engineered. The unit includes a conventional dialysis filter (cartridge that filters the blood) connected to a renal tubule assist device that contains human tubule cells, which reclaim electrolytes, salt, glucose, and water lost in traditional dialysis. This therapeutic modality may decrease the survival gap between current renal replacement therapies and healthy kidney functions.308
Acute renal failure has a high morality rate despite hemodialysis or continuous renal replacement therapy. The first human trial of a bioartificial kidney was reported in October 2004.145 The hope is that complications of acute renal failure due to excessive inflammation can be delayed or avoided while giving the patient’s own kidney time to recover. Six of the 10 patients who received the unit who were expected to die recovered. Phase I and II clinical trials show that such a device replaces multiple kidney function and improves survival in acute renal failure.307
Bladder Transplantation
Research is underway to engineer bladder tissue substitutes. Tissue grown from the person’s own cells has been use to engineer bladder regeneration in young children. This is a tissue engineering feat more than actual organ transplantation. A number of synthetic and natural materials have been used in experimental settings. The production of functional bladder tissue replacements has not been completed yet.177
Damaged bladder tissue was removed in a small number of children aged 4 to 19 years with bladder impairment associated with myelomeningocele. Normal, healthy bladder (urothelial and muscle) cells were harvested and reproduced in a laboratory setting, then reintroduced into the remaining bladder. Since autogeneic tissue was used, there were no side effects from the grafting procedure and no rejection.16
Liver Transplantation
The first liver transplant in a human being was performed in 1963. Since that time, the success of this procedure has improved so much that 1600 liver transplantations were completed in the United States in 1990. In the following decade, a team of surgeons at the University of Chicago developed the living related transplant that requires only a lobe of a liver or small pieces taken from living donors. This technique has also been applied to cadaveric donor livers by splitting the liver to expand the donor pool.44
Indications
Orthotopic liver transplantation has become an established therapy for end-stage liver disease (e.g., cirrhosis caused by alcoholism, hepatitis C), acute liver failure, and primary biliary cirrhosis or primary sclerosing cholangitis as well as for nonalcoholic cirrhosis and hepatic or biliary malignancy.
Biliary atresia (bile ducts not formed normally) is the most common indication for pediatric liver transplantation. Five hundred pediatric liver transplants are performed annually; more than half are for biliary atresia. Other indications include neonatal cholestasis (children born with liver failure for unknown reasons), metabolic error leading to liver failure, acute liver failure for any reason (e.g., viral infection, drug overdose, tumor, cirrhosis).
Theoretically, anyone with advanced, irreversible liver disease with certain mortality may be considered for a liver transplant provided the disease can be corrected by liver transplantation (Table 21-5).
Hepatitis B is not eliminated by transplantation, but its recurrence and the damage it can cause may be substantially reduced by giving the client hepatitis B globulin. Hepatitis recurs in the transplanted liver in 80% to 90% of cases, but the damage to the new liver is slow, so many years of symptom-free living can occur.
With the exception of metastatic malignancy and hepatic lymphoma, there are few absolute contraindications to liver transplantation. Liver transplantation for large primary liver cancers is very limited; transplantation remains the best treatment for small tumors (less than 5 cm) in a liver that is already cirrhotic.337
People with metastatic disease that has spread to the liver are no longer treated with liver transplantation because of the poor outcome. The exception is a small group of people whose liver cancer is characterized by neuroendocrine tumors that are very slow growing. The results of transplantation in this group are not as good as for those individuals with benign disease but acceptable enough to qualify for transplantation.
Some metabolic disorders (e.g., familial hypercholesterolemia) that arise in the liver but produce damage elsewhere in the body can be cured if the liver is replaced with a liver from a normal individual. Many inborn errors of liver metabolism are benign and are not associated with end-stage liver disease. Acute fulminant liver failure secondary to severe hepatitis owing to a virus, toxin, or poison can be life threatening and requires liver transplantation to prevent death.