The Endocrine and Metabolic Systems
ENDOCRINE SYSTEM
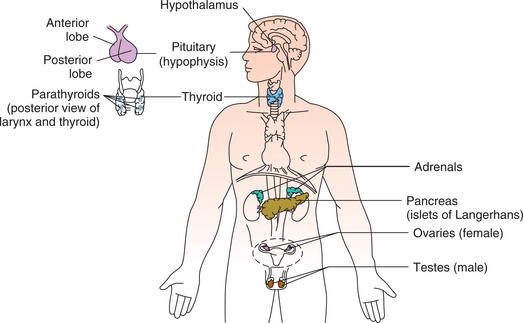
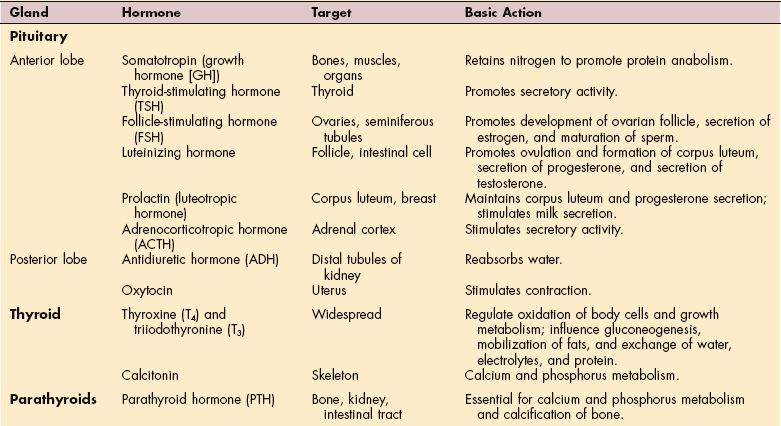
The endocrine system is composed of various glands located throughout the body (Fig. 11-1). These glands are capable of synthesis and release of special chemical messengers called hormones, which are transported by the bloodstream to the cells and organs on which they have a specific regulatory effect (Table 11-1). The endocrine system and the nervous system control and integrate body function to maintain homeostasis. Whereas the nervous system sends its messages along nerve fibers, eliciting swift and selective neural responses, the endocrine system sends its messages in the form of hormones via the bloodstream.
Table 11-1
Endocrine Glands: Secretion, Target, and Action.
When reading a client’s chart, it is important to know basic hormone functions or effects that may have an impact on therapy treatment. At least 30 different hormones have been identified, but only those most common to therapy clients are included here.
Hormonal effects have a slower onset than neural effects, but they maintain a longer duration of action. The actions of the endocrine system may be localized to one area or generalized to all the cells of the body.158 The endocrine system has the following five general functions:
1. Differentiation of the reproductive and central nervous system of the developing fetus.
2. Stimulation of sequential growth and development during childhood and adolescence.
3. Coordination of the male and female reproductive systems.
4. Maintenance of optimal internal environment throughout the lifespan.
5. Initiation of corrective and adaptive responses when emergency demands occur.160
The endocrine system meets the nervous system at the hypothalamic-pituitary interface. The hypothalamus, the main integrative center for the endocrine and autonomic nervous systems, controls the function of endocrine organs by neural and hormonal pathways. Although the communicative and integrative roles of the endocrine and nervous systems are similar, the precise ways in which each system functions differ.
Hypothalamic Control
Neural pathways connect the hypothalamus to the posterior pituitary (or neurohypophysis), providing the hypothalamus direct control over both the anterior and posterior portions of the pituitary gland (Fig. 11-2). Disorders of the hypothalamic-pituitary axis are manifested clinically, usually either by syndromes of hormone excess or deficiency or by visual impairment from optic nerve compression because of the location of the hypothalamus and pituitary.
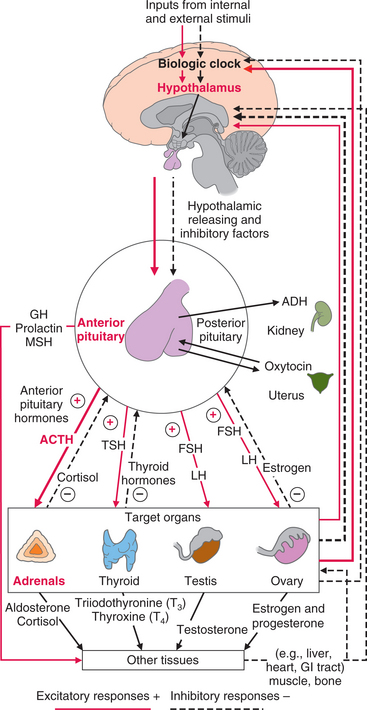
Figure 11-2 Control of the endocrine system by the nervous system. One example of the complex feedback loops described in the text is highlighted here. The hypothalamus controls the pituitary gland through releasing and inhibiting factors. The anterior lobe of the pituitary gland then releases tropic (stimulating) hormones that act on target glands (thyroid, adrenals, gonads). Endocrine pathology occurs when dysfunction occurs in releasing, tropic, or effector hormones, or when defects occur in the target tissue.
Neural stimulation to the posterior pituitary provokes the secretion of two effector hormones: antidiuretic hormone (ADH) and oxytocin. The hypothalamus also exerts hormonal control at the anterior pituitary through releasing and inhibiting factors. Hypothalamic hormones stimulate the pituitary to release tropic (stimulating) hormones, such as adrenocorticotropic hormone (ACTH), thyroid-stimulating hormone (TSH), luteinizing hormone (LH), and follicle-stimulating hormone (FSH) (see Fig. 11-2). At the same time, effector hormones, such as growth hormone (GH) and prolactin, are released or inhibited, affecting the adrenal cortex, thyroid, and gonads. Endocrine pathology develops as a result of dysfunction of releasing, tropic, or effector hormones or when defects occur in the target tissue.
In addition to hormonal and neural controls, a negative feedback system regulates the endocrine system. The mechanism may be simple or complex. Simple feedback occurs when the level of one substance regulates the secretion of a hormone. For example, low serum calcium levels stimulate parathyroid hormone (PTH) secretion; high serum calcium levels inhibit it. Complex feedback loops occur through the hypothalamic-pituitary-target organ axis. For example, after an injury or major stress, secretion of the hypothalamic corticotropin-releasing hormone (CRH) releases pituitary ACTH, which in turn stimulates adrenal cortisol secretion. Subsequently, a rise in serum cortisol inhibits ACTH by decreasing CRH secretion (see Fig. 11-2).
Steroid therapy disrupts the hypothalamic-pituitary-adrenal (HPA) axis by suppressing hypothalamic-pituitary secretion. Such treatment is necessary for some conditions but problems can occur when there is too rapid or abrupt of a withdrawal of exogenous steroid. The result can be life-threatening adrenal insufficiency because the HPA axis does not have enough time to recover sufficiently to stimulate cortisol secretion.
Hormonal Effects
In response to the hypothalamus, the posterior pituitary secretes oxytocin and ADH. Oxytocin stimulates contraction of the uterus and is responsible for the milk letdown reflex in lactating women. ADH controls the concentration of body fluids by alteration of the permeability of the kidney’s distal convoluted tubules and collecting ducts to conserve water. The secretion of ADH depends on plasma volume and osmolality as monitored by hypothalamic neurons. Circulatory shock and severe hemorrhage are the most powerful stimulators of ADH; other stimulators include pain, emotional stress, trauma, morphine, tranquilizers, certain anesthetics, and positive-pressure breathing.
The anterior pituitary secretes prolactin, which stimulates milk production, and human GH (HGH), which affects most body tissues. HGH stimulates growth by increasing protein synthesis and fat mobilization and by decreasing carbohydrate utilization. Hyposecretion of HGH results in dwarfism; hypersecretion causes gigantism in children and acromegaly in adults.
The thyroid gland secretes the iodinated thyroid hormones thyroxine (T4) and triiodothyronine (T3). (For reference values of thyroid hormone levels mentioned throughout this chapter see Table 40-19.) Thyroid hormones, necessary for normal growth and development, act on many tissues to regulate our basal metabolism (i.e., the rate at which we convert food and oxygen into energy) and to increase metabolic activity and protein synthesis. T4 is more abundant in the bloodstream than T3, but T3 is more active in directing the production of proteins vital to cell function.
Deficiency of thyroid hormone causes varying degrees of hypothyroidism, from a mild, clinically insignificant form to the life-threatening extreme, myxedema coma. Congenital hypothyroidism causes a condition in children previously referred to as cretinism (now considered an undesirable term).
Hypersecretion of thyroid hormone causes hyperthyroidism and in extreme cases, thyrotoxic crisis. Excessive secretion of TSH from the pituitary gland causes thyroid gland hyperplasia, resulting in goiter in chronic iodine deficiency states. Other causes of goiter are discussed in this chapter (see the section on Thyroid Gland in this chapter).
The parathyroid glands secrete PTH, which regulates calcium and phosphate metabolism. PTH elevates serum calcium levels by stimulating resorption of calcium and phosphate from bone, reabsorption of calcium and excretion of phosphate by the kidneys, and by combined action with vitamin D, absorption of calcium and phosphate from the gastrointestinal (GI) tract.
Hyperparathyroidism results in hypercalcemia; hypoparathyroidism causes hypocalcemia. Altered calcium levels also may result from nonendocrine causes such as metastatic bone disease. Pathologic changes in calcium affecting bone bring these conditions to the therapist’s attention.
The endocrine pancreas produces glucagon from the alpha-cells and insulin from the beta-cells. Glucagon, the hormone of the fasting state, releases stored glucose to raise the blood glucose level. Insulin, the hormone of the nourished state, facilitates glucose transport, promotes glucose storage, stimulates protein synthesis, and enhances free fatty acid uptake and storage. Insulin deficiency causes diabetes mellitus (DM); insulin excess can be exogenous (i.e., a person with diabetes may receive more insulin than is required) or insulin excess may result from a tumor of the beta-cells called insulinoma. Whatever the cause of excess insulin, hypoglycemia (abnormally low level of glucose in the blood) is the result.
The adrenal cortex secretes mineralocorticoids, glucocorticoids, and sex steroids. Aldosterone, a mineralocorticoid, regulates the reabsorption of sodium and the excretion of potassium by the kidneys and is involved intimately in the regulation of blood pressure. An excess of aldosterone (aldosteronism) can result primarily from hyperplasia or from adrenal adenoma or secondarily from many conditions, such as congestive heart failure or cirrhosis. The adrenal medulla is an aggregate of nervous tissue that produces the catecholamines epinephrine and norepinephrine, which are involved in the fight-or-flight response. (See the section on Neuroendocrine Response to Stress in this chapter.)
The testes and ovaries are also endocrine glands responsible for synthesizing and secreting hormones (see Chapters 19 and 20).
Adipose tissue can be classified as an endocrine gland because it secretes several hormones responsible for metabolism, hunger, vasoconstriction, and cellular growth and development. The concept of adipose tissue as an endocrine organ is quite new, but it is clear that molecules secreted into the bloodstream by fat, such as adiponectin and leptin, act on target organs at distant sites (see the section on Adipose Tissue in this chapter).
Endocrine Pathology
Dysfunctions of the endocrine system are classified as hypofunction and hyperfunction. The source of hypofunction and hyperfunction may be inflammation or tumor originating in the hypothalamus, the pituitary gland, or in other endocrine glands. Inflammation may be acute or subacute but is usually chronic, which results in glandular hypofunction. Chronic endocrine abnormalities (e.g., deficiencies of cortisol, thyroid hormone, or insulin) are common health problems requiring life-long hormone replacement for survival. Rarely, some endocrine gland tumors result in ectopic hormone production and may affect the musculoskeletal system.
Ectopic hormone production is the production and secretion of hormone or hormonelike substances from a source other than the normal source of the hormone. For example, some endocrine gland tumors can metastasize and produce excess hormone from new tumor sites (e.g., some types of thyroid, parathyroid, and adrenal cancers). Some nonendocrine cancers, particularly certain lung cancers, can secrete ACTH and GH. (See Chapter 9 for a discussion of paraneoplastic syndromes associated with this phenomenon.)
Neuroendocrine Response to Stress
The concept that stress of any kind (emotional, physical, psychological, or spiritual) may influence immunity and resistance to disease has been the subject of investigation for many years. The endocrine system, together with the immune system and the nervous system, mounts an integrated response to stressors. Only a brief review of the neuroendocrine response to stress contributing to disease is presented in this section. The reader is referred to a more specific text for detailed description of the endocrine system. Evidence to support a psychoneuroimmunologic basis for disease is discussed elsewhere in this text (see Chapters 3 and 7).178
Hormones of the neuroendocrine system affect components of the immune system,49 and mediators produced by immune components regulate the neuroendocrine response. The sympathetic nervous system is aroused during the stress response and causes the medulla of the adrenal gland to release catecholamines, such as epinephrine, norepinephrine, and dopamine, into the bloodstream. Simultaneously, the pituitary gland releases a variety of hormones, including ADH (from the posterior pituitary gland), prolactin, GH, and ACTH from the anterior pituitary gland.
Catecholamines
Catecholamines are organic compounds that play an important role in the body’s physiologic response to stress. Their release at sympathetic nerve endings increases the rate and force of muscular contraction of the heart, thereby increasing cardiac output; constricts peripheral blood vessels, resulting in elevated blood pressure; elevates blood glucose levels by hepatic and skeletal glycogenolysis; and promotes an increase in blood lipids by increasing the catabolism (breakdown) of fats.
Glycogenesis is the splitting of glycogen, a starch stored primarily in the liver but also in the muscles, yielding glucose. The well-known metabolic effects of adrenal catecholamines prepare the body to take physical action in the fight-or-flight phenomenon. Stressors commonly associated with catecholamine release include exercise, thermal changes, and acute emotional states.
Cortisol
Cortisol is the principal glucocorticoid hormone released from the adrenal cortex and also known as hydrocortisone when synthesized pharmaceutically. Cortisol has multiple functions (Table 11-2), but it primarily regulates the metabolism of proteins, carbohydrates, and lipids to cause an elevation in blood glucose level. These effects on glucose level and fat metabolism result in increased blood glucose and plasma lipid levels and promote the formation of ketone bodies when insulin secretion is insufficient. For this reason, glucocorticoids (including cortisol) are referred to as antiinsulin diabetogenic hormones.20
Table 11-2
Physiologic Effects of Cortisol
| Functions Affected | Physiologic Effects |
| Protein metabolism | Increases protein synthesis in the liver and depresses protein synthesis in muscle, lymphoid tissue, adipose tissue, skin, and bone; increases plasma level of amino acids. |
| Carbohydrate and lipid metabolism | Diminishes peripheral uptake and utilization of glucose; increases output of glucose from the liver; enhances the elevation of blood glucose promoted by other hormones. |
| Lipid metabolism | Breakdown of fat in the extremities (lipolysis) and production of fat in the face and trunk (lipogenesis). |
| Inflammatory effects | Decreases circulating eosinophils, lymphocytes, and monocytes; increases release of polymorphonuclear leukocytes from the bone marrow; decreases accumulation of leukocytes at the site of inflammation; delays healing; essential for vasoconstrictive action of norepinephrine. |
| Digestive function | Promotes gastric secretion. |
| Urinary function | Enhances urinary excretion. |
| Connective tissue function | Decreases proliferation of fibroblasts in connective tissue (and thus delays healing). |
| Muscle function | Maintains normal contractility and maximal work output for skeletal and cardiac muscle. |
| Bone function | Decreases bone formation. |
| Vascular system and myocardial function | Maintains normal blood pressure; permits increased responsiveness of arterioles to the constrictive action of adrenergic stimulation; optimizes myocardial performance. |
| Central nervous system function | Modulates perceptual and emotional functioning (mechanism unknown); essential for normal arousal and initiation of activity. |
Cortisol is essential to norepinephrine-induced vasoconstriction and other physiologic phenomena necessary for survival under stress. The production of glucose promoted by cortisol provides a source of energy for body tissues (nerve cells in particular), and the pooling of amino acids from catabolized proteins may ensure amino acid availability for protein synthesis at sites where replacement is critical such as muscle or cells of damaged tissue.
Another effect of cortisol is that of dampening the body’s inflammatory response to invasion by foreign agents. This antiinflammatory protective mechanism helps preserve the integrity of body cells at the site of the inflammatory response and provides the basis for the major therapeutic use of this steroid. Cortisol also inhibits fibroblast proliferation and function at the site of an inflammatory response and accounts for the poor wound healing, increased susceptibility to infection, and decreased inflammatory response often seen in individuals with chronic glucocorticoid excess. Whether cortisol-induced effects are adaptive or destructive depends on the subsequent concentration and length of cortisol exposure.
Other Hormones
Other hormones, such as endorphins, GH, prolactin, and testosterone, may be released as part of the response to stressful stimuli. Endorphins, a term derived from endogenous and morphine, are a group of opiate-like peptides produced naturally by the body at neural synapses in the central nervous system. These hormones serve to modulate the transmission of pain perceptions by raising the pain threshold and producing sedation and euphoria.
As its name implies, growth hormone (GH) stimulates and controls the rate of skeletal and visceral growth by directly influencing protein, carbohydrate, and lipid metabolism. GH levels increase in the blood after a variety of physically or psychologically stressful stimuli such as surgery, fever, physical exercise, or the anticipation of exhausting exercise, cardiac catheterization, electroshock therapy, or gastroscopy.18
Prolactin stimulates the growth of breast tissue and sustains milk production in postpartum mammals. Prolactin levels in plasma increase with a variety of stressful stimuli, including such procedures as gastroscopy, proctoscopy, pelvic examination, and surgery, but they show little change after exercise. Testosterone, a hormone that regulates male secondary sex characteristics and sex drive (libido), decreases after stressful stimuli such as anesthesia, surgery, marathon running, and acute illness (e.g., respiratory failure, burns, or congestive heart failure). Decreased testosterone during these circumstances restrains growth and reproduction to preserve energy for protective responses.18
Aging and the Endocrine System50,118
The exact effects of aging on the endocrine system are not clear. In particular, the question of whether changes in endocrine function are a cause of aging or a natural consequence of aging remains unresolved. The endocrine system has not been implicated as the direct cause of aging. Coexisting age-related variables, such as acute and chronic nonendocrine disease, use of medications, alterations in diet, changes in body composition and weight, and changes in sleep-wake cycle affecting the endocrine system, confuse the picture. New analytical tools to evaluate the neuroregulation of the endocrine axes are predicted to yield important information in the next decade.
Age-associated declines in physiologic performance of the endocrine system are well documented, and it is accepted that the basis of this decline is a failure of homeostasis. The conventional view is that “normal” aging changes predispose to age-related disease and contribute to the poor recovery of aging adults after illness or severe stresses such as surgery. Equilibrium concentrations of the principal hormones necessary to maintain homeostasis are not necessarily altered with age, but what may differ as we get older is the way we achieve equilibrium hormone levels, which points to changes in regulatory control.
Collectively, available clinical data suggest a general model of early neuroendocrine aging in the human (both males and females) with variable but predictable disruption in the time-delayed feedback and feedforward interconnections among neuroendocrine glands.252 Thus with advancing age, significant alterations in hormone production, metabolism, and action are found.
The continuum of the age-related changes is highly variable and sex-dependent. Whereas only subtle changes occur in the pituitary, adrenal, and thyroid function, changes in glucose homeostasis, reproductive function, and calcium metabolism are more apparent. The role of the thyroid gland in the metabolism of the healthy older person remains unclear. No major defects are apparent in healthy individuals; however, during episodes of ill health, the thyroid’s ability to maintain homeostasis is often limited.50
Aging is associated with a higher incidence of disorders or diseases of the endocrine system, including type 2 DM, hypothyroidism, and an increased incidence of atypical endocrine diseases during later life. Cellular damage associated with aging, genetically programmed cell change, and chronic wear and tear may contribute to endocrine gland dysfunction or alterations in responsiveness of target organs (as a result of changes with aging and disease, the target organs may lose their ability to respond to hormones).
Other endocrine changes that may be associated with aging and especially contribute to the age-associated failure in homeostasis include the neuroendocrine theory of aging. This theory attempts to explain the altered biologic activity of hormones, altered circulating levels of hormones, altered secretory responses of endocrine glands, altered metabolism of hormones, and loss of circadian control of hormone release. These changes are postulated to occur as a result of a genetic program encoded in the brain and then controlled and relayed to peripheral tissues through hormonal and neural agents.160 This theory suggests that cells are programmed to function only for a given time.
Menopause as a result of programmed changes in the reproductive system is an example of this theory. Changes in the neuroendocrine system because of the loss of ovarian function at menopause have an important biologic role for women in the control of reproductive and nonreproductive functions and regulate mood, memory, cognition, behavior, immune function, the locomotor system, and cardiovascular functions.203 It is thought that the temporal patterns of neural signals are altered during middle age, leading to cessation of reproductive cycles, and that the complex interplay of ovarian and hypothalamic/pituitary pacemakers becomes increasingly dysfunctional with aging, ultimately resulting in menopause.203
The relationship between aging and the structure and function of the endocrine system cannot be separated from the changes in the immune system and the central nervous system (CNS). Evidence is increasing in support of an immune–neuroendocrine homeostatic network in humans with the thymus gland playing a key role in the immunoregulation of the nervous and endocrine systems. The early onset of thymus involution may act as a triggering event that initiates the gradual decline in endocrine homeostasis resulting in the aging process.92
Additionally, as the nervous system ages, a progressive reduction takes place in the body’s capacity to maintain homeostasis in the face of environmental stress. The overall effect of the changes in aging in the neuroendocrine system is a progressive resistance to the inhibitory feedback of the end-organ hormonal secretion (see Fig. 11-2). Thus, although the initial response to a stressful stimulus may be appropriate, as the body ages, the response is more likely to be persistent and ultimately inappropriate or even harmful.61
Anatomic Changes with Aging
The pituitary gland undergoes both anatomic and histologic changes associated with aging. By age 80 years, the weight of the anterior pituitary lobe (adenohypophysis) is reduced approximately 75% from its peak during young adulthood. The blood supply is reduced, and a higher incidence of adenomas and cysts is described during later life.
The thyroid gland becomes relatively smaller and fibrotic, and its position becomes lower-lying and retrosternal with age. As with the pituitary gland, blood supply to the thyroid gland is decreased. Secretion of thyroid hormones may diminish with age.
The parathyroid gland demonstrates tissue changes with advancing age, but no major change is apparent in PTH levels. Hyperparathyroidism occurs primarily in persons older than 50 years and most commonly results from a single adenoma. It occasionally occurs with multiple adenomas or hyperplasia of two or more parathyroid glands. It is rarely caused by parathyroid carcinoma.
The adrenal glands have more fibrous tissue with aging, but because of compensatory feedback mechanisms, no relative alteration is apparent in functional cortisol levels. The most common cause of hypercortisolism occurs with the use of corticosteroids for medical conditions. As previously mentioned, because steroid use can suppress the pituitary-adrenal axis, adrenal insufficiency can occur after discontinuation of steroid therapy.
Changes in the reproductive glands have been shown clearly to have physiologic effects, most notably on the cardiovascular system and the skeleton (ovary) and muscle mass and libido (testis).185 These effects are discussed elsewhere (see Chapters 19, 20, and 24).
Hormonal Changes with Aging
The female reproductive system undergoes changes as part of the normal aging process. Menopause leads to changes in the genitourinary tract and accelerates the loss of minerals from bone and leads to an alteration in the lipid composition in the mature woman. Male hormones have been linked to preservation of bone and muscle mass and to an increased tendency toward developing certain diseases (e.g., benign prostatic hypertrophy or liver disease) during later life.
Loss of body hair, changes in the skin’s collagen content and thickness, an increase in the percentage of body fat, a decrease in lean body mass, a decrease in bone mass, and a decrease in protein synthesis are signs of endocrinopathy that may be associated with decreased GH levels.118 With the decline of GH secretion, sleep cycles are disrupted, and the potential for sequelae associated with sleep deprivation (e.g., depression, fibromyalgia) is now recognized.251
As mentioned, interactions between the endocrine and immune systems also influence the aging process. Declining hormonal levels are accompanied by increased activity of tumor-suppressor genes in the aging population unless these genes have been mutated so that suppressor function is lost. In fact, the most common somatic mutation of human cancers is the loss of tumor suppressor genes as a result of exposures to a lifetime of mutagens. In the presence of decreased hormonal levels, loss of tumor suppressor genes accounts for the increased probability of tumors with advancing age, again demonstrating the link between the endocrine and immune systems.118
All of these changes have an increasing effect on humans because the average lifespan has increased, meaning a greater part of women’s lives will be lived in an hypoestrogenic state. Men and women alike will experience a decline in GH secretion, increased exposure to mutagens, and a greater possibility of the loss of tumor suppressor genes.203
Musculoskeletal Signs and Symptoms of Endocrine Disease
Signs and symptoms of endocrine pathology vary, depending on the gland affected and whether the pathology is as a result of an excess (hyperfunction) or insufficiency (hypofunction) of hormonal secretions.89 In a therapy setting, the most common signs and symptoms associated with endocrine pathology observed in the musculoskeletal system are presented here.
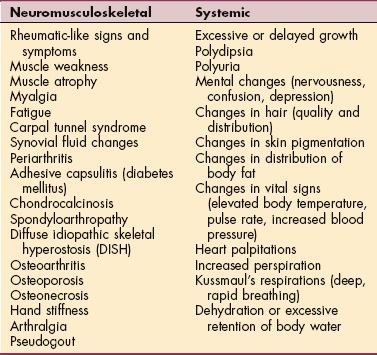
Growth and development of connective tissue structures are influenced strongly and sometimes controlled by various hormones and metabolic processes. When these processes are altered, structural and functional changes can occur in various connective tissues, producing musculoskeletal signs and symptoms in addition to other systemic signs and symptoms of endocrine dysfunction (Table 11-3).
The therapist must be aware that clients with an underlying but undiagnosed endocrine disorder may present initially with a musculoskeletal problem and that clients with established endocrine disorders are not cured by hormonal replacement or suppression. Rather, they may develop progression of musculoskeletal impairment in response to hormone fluctuations.
Rheumatoid arthritis can be an indicator of an underlying endocrine disease. Early rheumatic symptoms, such as myalgias and arthralgias, are seen commonly with a number of endocrine diseases. DM is associated with a variety of rheumatic syndromes such as the stiff-hand syndrome and limited joint motion syndrome. Although rheumatic symptoms can appear suddenly in people with an endocrine disorder, an insidious onset is much more common.
Muscle weakness, atrophy, myalgia, and fatigue that persist despite rest may be early manifestations of thyroid or parathyroid disease, acromegaly, diabetes, Cushing’s syndrome, or osteomalacia. In endocrine disease, most proximal muscle weakness is usually painless and may be unrelated to either the severity or the duration of the underlying disease. However, when true demonstrative weakness occurs (particularly in hyperthyroidism and hyperparathyroid disease), proximal muscle weakness is related to the severity and duration of the underlying endocrine problem. Any compromise of muscle energy metabolism aggravates and perpetuates trigger points such as are associated with myofascial pain syndrome (see Chapter 27) or tender points in muscle associated with fibromyalgia syndrome (see Chapter 7).
Carpal tunnel syndrome (CTS) (see discussion in Chapter 39) resulting from median nerve impairment at the wrist is a common finding in people with certain endocrine and metabolic conditions such as acromegaly, diabetes, pregnancy, and hypothyroidism (see Table 39-5). Any increase in the volume of contents of the carpal tunnel impinges on the median nerve (e.g., neoplasm, calcium, gouty tophi deposits, edema, or tenosynovitis).
In endocrine disorders, CTS is frequently bilateral, which is one characteristic that may distinguish it from overuse syndromes and other causes of CTS. Unreported tarsal tunnel syndrome may also occur, another distinguishing characteristic of an underlying systemic origin of symptoms when present along with CTS.
Tenosynovitis (inflammation of the tendon sheaths) occurs with some infectious processes and many musculoskeletal conditions. Fluid infiltrating the tunnel may soften the transverse carpal ligament, which can make the bony arch flatten and compress the nerve.87 Thickening of the transverse carpal ligament also may occur with systemic disorders such as acromegaly or myxedema.
CTS in persons with diabetes represents one form of diabetic neuropathy caused by ischemia-related microvascular damage of the median nerve. This ischemia then causes increased sensitivity to even minor pressure exerted in the carpal tunnel area.111 Vitamin B6 deficiency, repetitive activities, and obesity may also be factors in the development of CTS for the person with diabetes.2,65
CTS occurring during pregnancy may be caused by extra fluid and/or fat, diabetes (gestational or previously diagnosed), vitamin deficiencies, or other causes unrelated to the pregnancy itself (e.g., rheumatoid arthritis or job-related biomechanical stress). The fact that many women develop CTS at or near menopause may suggest that the soft tissues about the wrist may be affected in some way by hormones.41
Periarthritis (inflammation of periarticular structures including the tendons, ligaments, and joint capsule) and calcific tendinitis occur most often in the shoulders of people who have endocrine disease. Chondrocalcinosis is the deposition of calcium salts in the joint cartilage; when accompanied by attacks of goutlike symptoms, it is called pseudogout. In 5% to 10% of people with chondrocalcinosis, an associated underlying endocrine or metabolic disease occurs such as hypothyroidism, hyperparathyroidism, or acromegaly.78 People diagnosed with fibromyalgia also may have altered thyroid function149 and present with shoulder impingement secondary to chondrocalcinosis (see the section on Fibromyalgia in Chapter 7).
Spondyloarthropathy (disease of joints of the spine) and osteoarthritis occur in individuals with various endocrine or metabolic diseases, including hemochromatosis (disorder of iron metabolism with excess deposition in the tissues; also known as bronze diabetes and iron storage disease), ochronosis (metabolic disorder caused by alkali deposits, resulting in discoloration of body tissues), acromegaly, and DM.
Hand stiffness, hand pain, and arthralgias of the small joints of the hand may occur with endocrine and metabolic diseases. Flexor tenosynovitis with stiffness is a common finding in persons with hypothyroidism. This condition often accompanies CTS.147
SPECIFIC ENDOCRINE DISORDERS
The pituitary gland, or hypophysis, is a small (1 cm in diameter), oval gland located at the base of the skull in an indentation of the sphenoid bone directly posterior to the sphenoid sinus (see Figs. 11-1 and 11-2). It is often referred to as the master gland because of its role in regulating other endocrine glands. It is joined to the hypothalamus by the pituitary stalk (neurohypophyseal tract) and is influenced by the hypothalamus through releasing and inhibiting factors. The pituitary consists of two parts: the anterior pituitary (adenohypophysis) and the posterior pituitary (neurohypophysis) lobes. The anterior pituitary secretes six different hormones (ACTH, TSH, LH, FSH, HGH, and prolactin) (see Fig. 11-2).
The posterior pituitary is a downward offshoot of the hypothalamus and contains many nerve fibers; it produces no hormones of its own. The hormones ADH (also called vasopressin) and oxytocin are produced in the hypothalamus and then stored and released by the posterior pituitary. These hormones pass down nerve fibers from the hypothalamus through the pituitary stalk to nerve endings in the posterior pituitary; they accumulate in the posterior pituitary during less active periods of the body. Transmitter substances, such as acetylcholine and norepinephrine, are thought to activate release of these substances by the posterior pituitary gland when they are stimulated by nerve impulses from the hypothalamus.18
Anterior Lobe Disorders
Disorders of the pituitary gland occur most frequently in the anterior lobe, most often caused by tumors, pituitary infarction, genetic disorders, and trauma. The three principal pathologic consequences of pituitary disorders are hyperpituitarism, hypopituitarism, and local compression of brain tissue by expanding tumor masses.19 (See also Chapter 30.)
Overview.: Hyperpituitarism is an oversecretion of one or more of the hormones secreted by the pituitary gland, especially GH, resulting in acromegaly or gigantism. It is caused primarily by a hormone-secreting pituitary tumor, typically a benign adenoma. Other syndromes associated with hyperpituitarism include Cushing’s disease, amenorrhea, and hyperthyroidism.
Cushing’s disease is one form of Cushing’s syndrome and results from oversecretion of ACTH by a pituitary tumor, which in turn results in oversecretion of adrenocortical hormones (see the section on Cushing’s Syndrome in this chapter). Pituitary tumors produce both systemic effects and local manifestations.
Systemic effects include the following:
1. Excessive or abnormal growth patterns, resulting from overproduction of growth hormone.
2. Hyperprolactinemia (increased prolactin secretion) resulting in amenorrhea, galactorrhea (spontaneous milk flow in women without nursing), and gynecomastia and impotence in men.
3. Overstimulation of one or more of the target glands, resulting in the release of excessive adrenocortical, thyroid, or sex hormones.
Local pituitary tumors produce symptoms as the growing mass expands within the bony cranium. Local manifestations may include visual field abnormalities (pressure on the optic chiasma where the optic nerve crosses over), headaches, and somnolence (sleepiness).
Gigantism and Acromegaly.: Gigantism, an overgrowth of the long bones, and acromegaly, increased bone thickness and hypertrophy of the soft tissues, result from GH–secreting adenomas of the anterior pituitary gland. Although GH–producing tumors that cause these conditions are rare, they are the second most common type of hyperpituitarism. Gigantism develops in children before the age when the epiphyses of the bones close; people who develop gigantism may grow to a height of 9 feet. Gigantism develops abruptly, whereas acromegaly develops slowly.
Acromegaly is a disease of adults and develops after closure of the epiphyses; the bones most affected are those of the face, jaw, hands, and feet. In adults, acromegaly occurs equally among men and women and usually between ages 30 and 50 years.19 Both conditions are characterized by the same skeletal abnormalities because hypersecretion of GH produces cartilaginous and connective tissue overgrowth, resulting in coarsened facial features; protrusion of the jaw (prognathism); thickened ears, nose, and tongue; and broad hands, with spadelike fingers (Fig. 11-3).
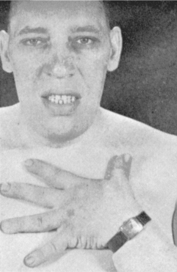
Figure 11-3 Acromegaly (hyperpituitarism). Acromegaly occurs as a result of excessive secretion of growth hormone after normal completion of body growth. The resulting overgrowth of bone in the face, head, and hands is pictured here. (From Jarvis C: Physical examination and health assessment, Philadelphia, 1992, WB Saunders.)
In gigantism, as the tumor enlarges and invades normal tissue, target organ functions are impaired by the loss of other tropic (stimulating) hormones such as TSH, LH, FSH, and ACTH. Clients with acromegaly may experience local manifestations, such as headache, diplopia, blindness, and lethargy, as the tumor compresses brain tissue.
Acromegaly-induced myopathy with muscle weakness and reduced exercise tolerance may be more common than previously appreciated. The pathologic or physiologic reason for this weakness has not been determined. Alterations in muscle size and strength in individuals with acromegaly are an accepted association and may be multifactorial in origin. It could be the result of a com- bination of the direct effects of growth hormone on muscle, the metabolic and mechanical neuropathies present with the condition, the mechanical disadvantage occurring as a result of joint hypermobility, or restriction caused by articular changes and periarticular bone remodeling.162
MEDICAL MANAGEMENT
DIAGNOSIS, TREATMENT, AND PROGNOSIS.
Increased mortality is linked with elevated GH and/or the target growth factor called insulin-like growth factor I (IGF-I).70 Timely diagnosis and appropriate treatment are imperative in reducing this potentially disabling chronic and progressive condition.70 Uncontrolled GH and IGF-I may accelerate the rate of bone turnover; in a small number of people, this long-term exposure may predispose the individual to malignant bone tumor.142 Long-term follow-up of disease activity and comorbidities is recommended with management rather than cure being the primary goal.69 Quality of life is often below reference values for the normal population of the same age.261 Diagnosis is established by documenting autonomous GH hypersecretion and by imaging of the pituitary gland. Pituitary tumors are treated usually by surgical removal, drug therapy, and/or external beam radiation therapy.
Drugs are now available that effectively normalize levels of growth hormone and prolactin and decrease pituitary tumor size.69 Drug therapy has replaced surgery in most cases of prolactin-secreting adenomas, but surgery is still the treatment of choice for pituitary adenomas that cause acromegaly.
Some drug or radiation therapy may be required if levels of GH remain high after surgery. Radiation therapy is also useful when surgery is not curative.170 Frequently, after pituitary surgery, pituitary function is lost and at that time, treatment with thyroid, cortisone, and hormone replacement may be necessary.
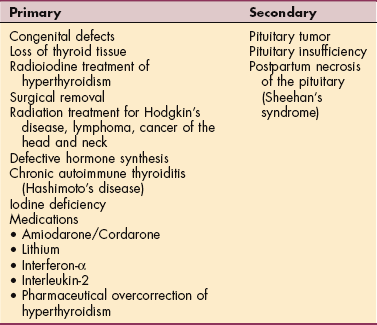
Hypopituitarism.: Hypopituitarism (also panhypopituitarism and dwarfism) results from decreased or absent hormonal secretion by the anterior pituitary gland. Panhypopituitarism refers to a generalized condition caused by partial or total failure of all six of the anterior pituitary’s vital hormones (ACTH, TSH, LH, FSH, HGH, and prolactin).
Hypopituitarism and panhypopituitarism are rare disorders that occur as a result of the following:
1. Hypophysectomy (removal or destruction of the pituitary by surgery, irradiation, or chemical agents).
2. Nonsecreting pituitary tumors.
3. Postpartum hemorrhage (the fall in blood pressure and subsequent hypoxia after delivery causes necrosis of the gland).
4. Reversible functional disorders (such as starvation, anorexia nervosa, severe anemia, and GI tract disorders).
Clinical manifestations are dependent on the age at onset and the hormones affected (Box 11-1). More than 75% of the pituitary must be obliterated by tumors or thromboses before symptoms develop. Specific disorders resulting from pituitary hyposecretion include GH deficiency, with subsequent short stature, delayed growth, and delayed puberty; secondary adrenocortical insufficiency from diminished synthesis of ACTH by the pituitary gland, which in turn causes diminished secretion of adrenocortical hormones by the adrenal cortex; hypothyroidism (thyroid hormone is dependent on TSH secreted by the pituitary); and sexual and reproductive disorders from deficiencies of the gonadotropins (LH and FSH).
Treatment for hypopituitarism involves removal (if possible) of the causative factor, such as tumors, and lifetime replacement of the missing hormones.
Posterior Lobe Disorders
Diabetes Insipidus.: Diabetes insipidus, a rare disorder, involves a physiologic imbalance of water secondary to ADH deficiency. Injury or loss of function of the hypothalamus, the neurohypophyseal tract, or the posterior pituitary gland can result in diabetes insipidus (Box 11-2).
Because the major functions of ADH are to promote water resorption by the kidney and to control the osmotic pressure of the extracellular fluid, when ADH production decreases, the kidney tubules fail to resorb water. The end result is excretion of large amounts of dilute urine. Unlike urine in DM, which contains large amounts of glucose, urine in diabetes insipidus is dilute and contains no glucose. Other clinical manifestations include polydipsia (excessive thirst), nocturia (excessive urination at night), and dehydration (e.g., poor tissue turgor, dry mucous membranes, constipation, muscle weakness, dizziness, and hypotension) (see Box 5-8). Fatigue and irritability may develop secondary to sleep disruption and in association with nocturia.
If a person is conscious and able to respond appropriately to the thirst mechanism, hydration can be maintained. However, if a person is unconscious or confused and unable to take in necessary fluids to compensate for fluid loss, rapid dehydration, shock, and death can occur. Treatment is usually exogenous replacement of ADH with vasopressin or a synthetic derivative, such as Pitressin, along with administration of diuretics. When this condition is caused by tumor, resection of the tumor can affect a cure.
Syndrome of Inappropriate Antidiuretic Hormone Secretion.: Syndrome of inappropriate ADH (SIADH) is a disorder associated with excessive release of ADH, which disturbs fluid and electrolyte balance, resulting in a water imbalance. SIADH has a wide variety of causes, including pituitary damage resulting from infection or trauma.
Tumors can cause unregulated production of ADH leading to severe hyponatremia (sodium depletion, less than 115 mEq/L) with resultant lethargy, nausea, anorexia, and generalized weakness. Mild hyponatremia (125 to 130 mEq/L) causes increased thirst, muscle cramps, and lethargy. Rapid onset of SIADH can result in coma, convulsions, or death.240 SIADH can be triggered by the stress of surgery or many systemic disorders and response to certain medications, including chemotherapy medications such as vincristine and cyclophosphamide195,240 (Box 11-3).
SIADH is the opposite of diabetes insipidus, so treatment of diabetes insipidus with vasopressin can lead to SIADH if excessive amounts are administered. In SIADH, instead of large fluid losses, water intoxication occurs as a result of fluid retention. Under normal circumstances, ADH regulates serum osmolality. Serum osmolality is a measure of the number of dissolved particles per unit of water in serum. In a solution, the fewer the particles of solute in proportion to the number of units of water (solvent), the less concentrated the solution. A low serum osmolality indicates a higher-than-usual amount of water in relation to the amount of particles dissolved in it.
In other words, serum osmolality provides a measure of hydration of cells. For example, a low serum osmolality accompanies overhydration (i.e., edema); an increased serum osmolality is present in a state of fluid volume deficit. Osmolality is proportional with dilutional or depletional states (true for water and sodium). The normal value for serum osmolality is 280 to 300 mOsm/kg of water.39 When serum osmolality falls, a feedback mechanism causes inhibition of ADH, which promotes increased water excretion by the kidneys to raise serum osmolality to normal. When this feedback mechanism fails and ADH levels are sustained, fluid retention results. Ultimately, serum sodium levels fall, resulting in hyponatremia and water intoxication.111
Although fluid retention is the primary symptom, edema is rare unless water overload exceeds 4 L; much of the free water excess is within cellular boundaries. Neurologic and neuromuscular signs and symptoms predominate and are directly related to the swelling of brain tissue and to sodium changes within neuromuscular tissues. CNS dysfunction, characterized by alterations in level of consciousness, seizures, and coma, can occur when serum sodium falls to 120 mEq/L or less. Hyponatremia can result in diminished GI function; this problem is complicated further by the need for fluid restriction.
Correction of life-threatening sodium imbalance is the first aim of treatment followed by correction of the underlying cause. If SIADH is caused by malignancy, success in alleviating water retention may be obtained by surgical resection, irradiation, or chemotherapy. Otherwise, treatment for SIADH is symptomatic and includes restriction of water intake, careful replacement of sodium chloride, and administration of diuretics. Other pharmaceuticals (e.g., demeclocycline and tetracycline or lithium) also may be used to block the renal response to ADH.
Thyroid Gland
The thyroid gland is located in the anterior portion of the lower neck, below the larynx, on both sides of and anterior to the trachea (see Fig. 11-1). The primary hormones produced by the thyroid are thyroxine (T4), triiodothyronine (T3), and calcitonin. Both T3 and T4 regulate the metabolic rate of the body and increase protein synthesis. Calcitonin has a weak physiologic effect on calcium and phosphorus balance in the body. Thyroid function is regulated by the hypothalamus and pituitary feedback controls and by an intrinsic regulator mechanism within the gland.95
Both thyroid hormones travel from the thyroid via the bloodstream to distant parts of the body, including the brain, heart, liver, kidneys, bones, and skin where they activate genes that regulate body functions. When the hypothalamus senses that circulating levels have dropped, it signals the pituitary gland, which sends TSH to the thyroid to trigger the release of thyroid hormones.
Disorders of the thyroid gland may be functional abnormalities leading to hyperfunction or hypofunction of the gland or anatomic abnormalities such as thyroiditis, goiter, and tumor. Enlargement of the thyroid gland or neoplasm may or may not be associated with abnormalities of hormone secretion.
Susceptibility to thyroid disease is largely determined by the interaction of genetic makeup, age, and sex. Approximately 27 million Americans have been diagnosed with thyroid disease; many other people are undiagnosed because the signs and symptoms are so nonspecific. The risk of thyroid disease increases with age but is difficult to detect in adults over 60 because it typically masquerades as other illnesses such as heart disease, depression, or dementia. Women, particularly those with a family history of thyroid disease, are much more likely to have thyroid pathology than men. Although most thyroid conditions cannot be prevented, they respond well to treatment.
Thyroid hormone acts on nearly all body tissues, so excessive or deficient secretion affects various body systems. Alterations in thyroid function produce changes in nails, hair, skin, eyes, GI tract, respiratory tract, heart and blood vessels, nervous tissue, bone, and muscle.95
Women may notice disturbances in mood and in menstrual cycles. Menstrual irregularity, worsening premenstrual syndrome (PMS), new onset of depression later in life, postpartum depression (after pregnancy/birth), anxiety syndromes, and excessive fatigue have been reported by many women with thyroid dysfunction.
Both hyperthyroidism and hypothyroidism can adversely affect cardiac function. Sustained tachycardia in hyperthyroidism and sustained bradycardia with cardiac enlargement in hypothyroidism can result in cardiac failure. Both conditions affect the general rate of metabolism, the muscular system, the nervous system, the GI system, and as mentioned, the cardiovascular system.
Hyperthyroidism
Definition and Overview.: Hyperthyroidism is an excessive secretion of thyroid hormone, sometimes referred to as thyrotoxicosis, a term used to describe the clinical manifestations that occur when the body tissues are stimulated by increased thyroid hormone. Excessive thyroid hormone creates a generalized elevation of body metabolism, the effects of which are manifested in almost every system.
The most common form of hyperthyroidism is the autoimmune condition known as Graves’ disease, which increases T4 production and accounts for 85% of cases of hyperthyroidism. Like most thyroid conditions, hyperthyroidism affects women more than men (4: 1), especially women between ages 20 and 40 years.
Rarely, a person with inadequately treated hyperthyroidism may experience what is called a thyroid storm. This potentially fatal condition is an acute episode of thyroid overactivity characterized by high fever, severe tachycardia, delirium, dehydration, and extreme irritability or agitation. Stress occurring in the presence of undiagnosed or untreated hyperthyroidism may precipitate such an event. Stressors may include surgery, infection, toxemia of pregnancy, labor and delivery, diabetic ketoacidosis (DKA), myocardial infarction, pulmonary embolus, and medication overdose.
Etiologic and Risk Factors.: Hyperthyroidism may result from both immunologic and genetic factors. Graves’ disease, the most common form of hyperthyroidism, is most likely autoimmune in development, and although it is more common in women with family histories of thyroid abnormalities, major risk factors have not been identified. In addition, autoimmune hyperthyroid disease is present in people with other immune-related disorders such as Sjögren’s syndrome,130 rheumatoid arthritis, and psoriatic arthritis.157
Hyperthyroidism also may be caused by the overfunction of the entire gland, such as in Graves’ disease, or less commonly, by hyperfunctioning of a single adenoma or multiple toxic nodules. Rarely, overtreatment of myxedema associated with hypothyroidism (see next section) may result in hyperthyroidism, and more rarely, thyroid cancer can cause glandular hyperfunction.
Pathogenesis.: About 95% of people with Graves’ disease have circulating autoantibodies called thyroid-stimulating immunoglobulins (TSI) that react against thyroglobulin (precursor for thyroid hormones). These autoantibodies may be the result of a defect in suppressor T-lymphocyte function that allows formation of TSIs. Evidently, TSIs in the serum of hyperthyroid Graves’ clients are autoantibodies that react against a component of the thyroid cell membranes, stimulating enlargement of the thyroid gland and secretion of excess thyroid hormone.
Because the action of thyroid hormone on the body is stimulatory, hypermetabolism results with increased sympathetic nervous system activity. The excessive amounts of thyroid hormone stimulate the cardiac system and increase the number of β-adrenergic receptors throughout the body. This excess thyroid hormone secretion, coupled with the increased secretion of catecholamines, leads to tachycardia, increased stroke volume, and increased peripheral blood flow. The increased metabolism also leads to a negative nitrogen balance, lipid depletion, and a resultant state of nutritional deficiency.
Clinical Manifestations.: Because hyperthyroidism is caused by an excess secretion of thyroid hormone, the clinical picture of Graves’ disease is in many ways the opposite of that of hypothyroidism. The classic symptoms of Graves’ disease are mild symmetric enlargement of the thyroid (goiter), nervousness, heat intolerance, weight loss despite increased appetite, sweating, diarrhea, tremor, and palpitations. Hyperthyroidism may induce atrial fibrillation, precipitate congestive heart failure, and increase the risk of underlying CAD for myocardial infarction.
Exophthalmos (abnormal protrusion of the eyes) (Fig. 11-4) is considered most characteristic but is absent in many people with hyperthyroidism and may exacerbate after adequate treatment of the hyperthyroid state. Changes, such as swelling behind the eyes, are mediated by autoimmune production of antibodies to soft tissues (particularly the fibroblasts). Highly specialized ophthalmic surgery (surgical decompression) may be effective for correcting the severe exophthalmos when vision is impaired. Retroorbital radiation has also been shown to be effective.260
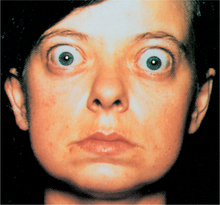
Figure 11-4 Exophthalmos, or protruding eyes. This is a forward displacement of the eyeballs associated with thyroid disease. Because the eyes are surrounded by unyielding bone, fluid accumulation in the fat pads and muscles behind the eyeballs causes protruding eyes and a fixed stare. Without treatment of the underlying cause, the client with severe exophthalmos may be unable to close the eyelids and may develop corneal ulceration or infection, eventually resulting in loss of vision. Note the lid lag; the upper eyelid rests well above the limbus (edge of the cornea where it joins the sclera), and white sclera is visible. This is evident when the person moves the eyes from up to down. Physical therapy is not recommended in these cases until after the endocrine problem is resolved. Then therapeutic intervention with ultrasound, joint mobilization, stretching, and strengthening may be indicated to treat any residual dysfunction. (From Seidel H et al: Mosby’s guide to physical examination, ed 3, St Louis, 1995, Mosby.)
Many other symptoms are commonly present because this condition affects many body systems (Table 11-4). As mentioned, complications, such as thyroid storm and heart disease, can occur. Emotions are adversely affected by the increased metabolic activity within the body. Moods may be cyclic, ranging from mild euphoria to extreme hyperactivity or delirium and depression, which may persist even after successful treatment of hyperthyroidism.29 Excessive hyperactivity may be associated with extreme fatigue.
Table 11-4
Systemic Manifestations of Hyperthyroidism.
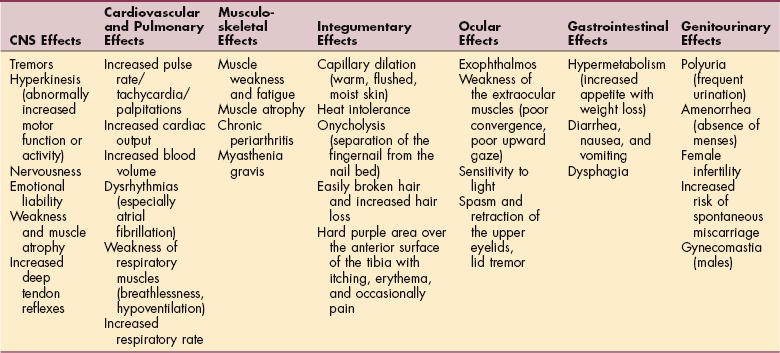
Modified from Goodman CC, Snyder TE: Differential diagnosis for physical therapists: screening for referral, ed 4, Philadelphia, 2007, WB Saunders. CNS, Central nervous system.
Hyperthyroidism in older adults is notorious for presenting with atypical or minimal symptoms.248 Signs and symptoms are not the usual ones and may be attributed to aging. Many older people actually appear apathetic instead of hyperactive. Cardiovascular abnormalities, as described previously, are much more common in older adults.
Neuromuscular Manifestations.: Chronic periarthritis also is associated with hyperthyroidism. Inflammation that involves the periarticular structures, including the tendons, ligaments, and joint capsule, is termed periarthritis. This syndrome is characterized by pain and reduced range of motion. Calcification, whether periarticular or tendinous, may be seen on x-ray studies. Both periarthritis and calcific tendinitis can occur most often in the shoulder in clients who have undiagnosed, untreated, or inadequately treated endocrine disease. The involvement can be unilateral or bilateral and can worsen progressively to become adhesive capsulitis, or frozen shoulder. Acute calcific tendinitis of the wrist also has been described in such clients. Although antiinflammatory agents may be needed for acute symptoms, chronic periarthritis usually responds to treatment of the underlying hyperthyroidism.
Proximal muscle weakness (most marked in the pelvic girdle and thigh muscles) accompanied by muscle atrophy, known as myopathy, can occur in cases of undiagnosed, untreated, or inadequately treated hyperthyroidism. The therapist may first notice problems with coordination or balance or notice weakness of the legs, causing a client difficulty in ambulating, rising from a chair, or climbing stairs.68
Respiratory muscle weakness can present as dyspnea. The pathogenesis of the weakness is still a subject of controversy; muscle strength seems to return to normal in 6 to 8 weeks after medical treatment, with a slower resolution of muscle wasting. In severe cases, normal strength may not be restored for months.
The incidence of myasthenia gravis, which is also an antibody immune disease, is increased in clients with hyperthyroidism, which in turn can aggravate muscle weakness. If the hyperthyroidism is corrected, improvement of the myasthenia gravis usually follows.
Sudden, periodic paralysis while at rest characterized by recurrent episodes of motor weakness of variable intensity can occur in a selective population (more common among people of Asian origin). This phenomenon is precipitated by intracellular shifts of potassium triggered by thyroid overactivity and hyperinsulinemia after ingestion of carbohydrates and increased physical activity. Administration of potassium is required to prevent life-threatening arrhythmias.177,200
MEDICAL MANAGEMENT
There is no way to prevent Graves’ disease. Early screening can help determine if someone is at risk. Two simple blood tests can be conducted, one to measure TSH and the second for antithyroid antibodies. Testing should be done by age 40 years (or perhaps earlier for women who intend to get pregnant), especially in the presence of a positive family history.
DIAGNOSIS.
Diagnosis is based on clinical history, physical presentation, examination findings, and laboratory test results. Hyperthyroidism is almost always associated with suppressed TSH. The very rare exception is that of a TSH-secreting pituitary adenoma. In very mild hyperthyroidism the T4 would be normal, but the measurement of T3 usually would be elevated or at the upper range of normal. This is called T3 toxicosis and almost always precedes Graves’ disease. Diagnostic tests, such as radioactive iodine uptake (RIU), can confirm the presence of hyperthyroidism and differentiate among causes of hyperthyroidism.39 RIU studies are elevated in Graves’ disease and nodular thyrotoxicosis but are very low or negative in thyroiditis-caused hyperthyroidism. TSI is positive in almost all people with Graves’ disease. It is essential to distinguish hyperthyroidism caused by Graves’ disease and nodular thyrotoxicosis from thyroiditis because the treatment for each is different.267
TREATMENT.
The three major forms of therapy are antithyroid medication, radioactive iodine (RAI), and surgery. Most endocrine specialists would now recommend radioactive iodine as first-line therapy in anyone older than 18 years of age who is not pregnant. Some physicians treat as young as the age of 12 years because long-term studies have shown no increased incidence of thyroid cancer or leukemia in people receiving such treatment.255
Iodine-131 therapy takes several months before it is effective, so adrenergic-blocking agents are sometimes given in the interim to control the activity of the sympathetic nervous system. Once the RAI is administered, the iodine concentrates in the thyroid gland, disrupting hormone synthesis. Typically, everyone who receives RAI becomes hypothyroid and requires thyroid hormone replacement for the rest of their lives. Almost everyone treated with radioactive iodine is hypothyroid during the first year of therapy but eventually normalizes with replacement therapy.
Use of antithyroid drugs (propylthiouracil and methimazole) is also effective and is the usual choice of therapy during pregnancy and for children under the age of 12 years. Side effects from drug treatment include rheumatoid-like arthritis and agranulocytosis (serious and potentially fatal) and usually resolve after 10 days of discontinuing the drug. About half of the people treated with antithyroid drugs have a later recurrence of hyperthyroid activity. Again, adrenergic-blocking agents may be used with these drugs.260
Partial or subtotal thyroidectomy is an effective way to treat hyperthyroidism caused by Graves’ disease and single or multinodular thyrotoxicosis. The ideal surgical treatment leaves a small portion of the functioning thyroid gland to avoid permanent hormone replacement. Surgical treatment is effective in most cases, although surgical complications can develop such as vocal cord paralysis (resulting from laryngeal nerve damage) or hypoparathyroidism leading to hypocalcemia (resulting from inadvertent removal of parathyroid gland tissue).19
PROGNOSIS.
Antithyroid drugs may be tapered and discontinued if remission is possible. Remission rates are higher in people with mild degrees of hyperthyroidism, small goiters, and for those who are diagnosed early. Even with remission, life-long follow-up is recommended because many remissions are not permanent. Relapses are most likely to occur in the postpartum period.267
After radioiodine treatment, regular life-long medical supervision is required. Frequently, hypothyroidism develops even as long as 1 to 3 years after treatment. Exophthalmos may not be reversed by intervention. In severe cases, the person may be unable to close the eyelids and must have the lids taped shut to protect the eyes. Without intervention, severe exophthalmos can progress to corneal ulceration or infection and loss of vision.
Hypothyroidism
Definition and Etiologic Factor.: Hypothyroidism (hypofunction) refers to a deficiency of thyroid hormone in the adult that results in a generalized slowed body metabolism; it is the most common disorder of thyroid function in the United States and Canada. More than 50% of cases occur in families in which thyroid disease is present.
Like diabetes, hypothyroidism can be categorized as type I (hormone deficient) and type II (hormone resistant). The condition has traditionally been classified as either primary or secondary. Type I/primary hypothyroidism occurs as a result of reduced functional thyroid tissue mass or impaired hormonal synthesis or release. Type II/secondary hypothyroidism accounts for a small percentage of all cases of hypothyroidism and occurs as a result of inadequate stimulation of the gland because of pituitary or hypothalamic disease (failure to produce TSH and TRH, respectively) (Table 11-5). (See section on Hashimoto’s Thyroiditis in this chapter.)
In the United States and Canada, this disease commonly is caused by congenital autoimmune thyroiditis, thyroid ablation via surgery or RAI therapy, or medication with thiouracil or lithium; rarely it is a result of subacute thyroiditis, iodine deficiency, dietary factors, congenital abnormalities in iodination, or pituitary failure.266
Incidence.: Hypothyroidism is about four times more prevalent in women than in men. Although hypothyroidism may be congenital and therefore present at birth, the highest incidence is between ages 30 and 60 years. More than 95% of all people with hypothyroidism have the primary form of the disease.111
Pathogenesis.: In type I/primary hypothyroidism, the loss of thyroid tissue leads to decreased secretion of thyroid hormone. In response to a decrease in thyroid hormone, TSH secretion is increased from the anterior pituitary gland as the body attempts to stimulate increased production of thyroid hormone. In the normal body, when hormone levels rise sufficiently, the pituitary slows TSH production. With hypothyroidism, the thyroid gland does not respond fully to TSH, so not enough T3 and T4 reach the body organs and body functions begin to slow. Whenever the body perceives an inadequate amount of thyroid hormone, the pituitary releases more and more TSH in an effort to stimulate thyroid hormone production. The result is an elevated TSH level in the blood when thyroid function is low.
Decreased levels of thyroid hormone lead to an overall slowing of the basal metabolic rate. This slowing of all body processes leads to bradycardia, decreased GI tract motility, slowed neurologic functioning, a decrease in body heat production, and achlorhydria (absence of hydrochloric acid from gastric juice). Lipid metabolism also is altered by hypothyroidism with a resultant increase in serum cholesterol and triglyceride levels and a concomitant increase in arteriosclerosis and coronary heart disease. Thyroid hormones also play a role in the production of red blood cells with the potential for the development of anemia.
Type II/secondary hypothyroidism is most commonly the result of failure of the pituitary gland to synthesize and release adequate amounts of TSH.
Clinical Manifestations.: As with all disorders affecting the thyroid and parathyroid glands, clinical signs and symptoms associated with hypothyroidism affect many systems of the body (Table 11-6). Typically, the early clinical features of hypothyroidism are vague and ordinary, so they escape detection (e.g., fatigue, mild sensitivity to cold, mild weight gain resulting from fluid retention [10 to 15 lb], forgetfulness, depression, and dry skin or hair).
Table 11-6
Systemic Manifestations of Hypothyroidism
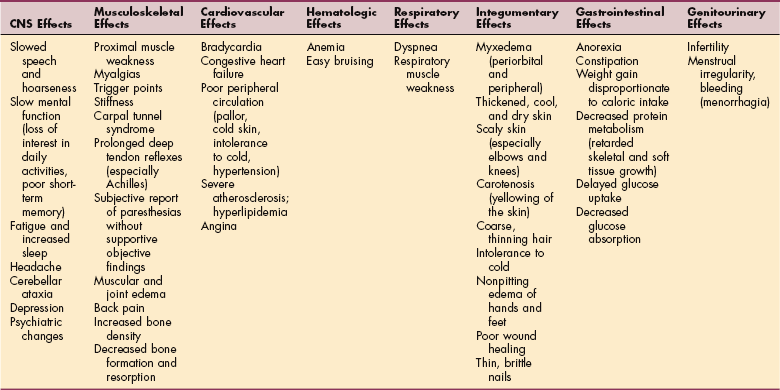
Modified from Goodman CC, Snyder TE: Differential diagnosis for therapists: Screening for referral, ed 4, Philadelphia, 2007, WB Saunders. CNS, Central nervous system.
As the disorder progresses, myxedema and its associated signs and symptoms appear. Myxedema is a result of an alteration in the composition of the dermis and other tissues, causing connective tissues to be separated by increased amounts of mucopolysaccharides and proteins. This mucopolysaccharide-protein complex binds with water, causing a nonpitting, boggy edema, especially around the eyes, hands, feet, and in the supraclavicular fossae. Thickening of the tongue, laryngeal and pharyngeal structures, hoarseness, and slurred speech occur as a result of myxedema.266
Other clinical manifestations associated with hypothyroidism may include decreasing mental stability; dry, flaky, inelastic skin; dry, sparse hair; hoarseness; upper eyelid droop; and thick, brittle nails. Cardiovascular involvement leads to decreased cardiac output, slow pulse rate, and signs of poor peripheral circulation. Other possible effects of hypothyroid function are anorexia, abdominal distention, menorrhagia, decreased libido, infertility, ataxia, intention tremor, and nystagmus.
Neuromuscular symptoms are among the most frequent manifestations of hypothyroidism seen in a therapy practice. Flexor tenosynovitis with stiffness can accompany CTS in persons with hypothyroidism. CTS arising from myxedematous tissue in the carpal tunnel area can develop before other signs of hypothyroidism become evident. Most people with CTS associated with hypothyroidism do not require surgical treatment because symp- toms of median nerve compression respond to thyroid replacement.
A wide spectrum of rheumatic symptoms occurs in people with hypothyroidism. A subset of fibromyalgia with muscle aches and tender points may be seen early; replacement therapy with thyroid hormone eliminates the symptoms, which aids in the diagnosis of the underlying cause of this form of fibromyalgia. Most cases of fibromyalgia fall into the type II (hormone-resistant) category. It is likely acquired as a result of mutated receptors.81
An inflammatory arthritis indistinguishable from rheumatoid arthritis may be seen. The arthritis predominantly involves the small joints of the hands and apparently differs from the viscous noninflammatory effusions observed in large joints of individuals with hypothyroidism. In general, the arthritis resolves with normalization of the thyroid hormone levels.163
Proximal muscle weakness can occur in persons with hypothyroidism, sometimes accompanied by pain. Trigger points are frequently detected on examination, and diffuse muscle tenderness may be the major finding. Muscle weakness is not always related to either the severity or the duration of hypothyroidism; it can be present several months before a medical diagnosis of hypothyroidism is made. Deep tendon reflexes show delayed relaxation time (i.e., prolonged reflexes), especially in the Achilles tendon.68
MEDICAL MANAGEMENT
A substantial delay in diagnosis resulting from the vague onset of symptoms is not uncommon. Specific testing of TSH levels is the most sensitive indicator of primary hypothyroidism. TSH levels are always elevated in primary hypothyroidism. T3 (triiodothyronine) levels do not change dramatically, even in severe hypothyroidism. T4 (thyroxine) levels, however, decrease gradually until they are well below normal in advanced hypothyroidism.
Serum cholesterol, alkaline phosphatase, and triglyceride levels also can be significantly elevated in the presence of hypothyroidism. In addition, the presence of antithyroid antibodies documents the existence of autoimmune thyroiditis resulting in progressive destruction of thyroid tissue by circulating antithyroid antibodies.136,270
TREATMENT.
The goals of treatment for hypothyroidism are to correct thyroid hormone deficiency, reverse symptoms, and prevent further cardiac and arterial damage. If treatment with life-long administration of synthetic thyroid hormone preparations is begun soon after symptoms appear, recovery may be complete. There is some controversy over whether mild hypothyroidism (defined by an elevated serum TSH level with a normal free thyroxine level) should be routinely screened, identified, and treated. Treatment is safe and effective but the question is whether the clinical consequences are enough to justify screening and therapy.136
Proponents of early detection and treatment argue that they lower the risk of atherosclerotic cardiovascular disease (CVD) and prevent progression to overt hypothyroidism, especially in adults who are 65 or older. Older people with underlying heart disease (particularly underlying CAD) can be started on very low doses of thyroxine gradually increased in dosage to ultimately return the TSH to within the normal range. Cardiac complications can occur, including angina severe enough that intervention may be required. Only small doses should be initiated in anyone with preexisting heart problems.
Sometimes, individuals with diagnosed hypothyroidism and taking thyroid medication (e.g., Synthroid, Levothroid, Levoxyl, or Euthyrox) to regulate symptoms will have “normal” levels of TSH when tested but still experience lingering symptoms of the condition. Since there is a broad range of: normal,” it is not always possible to find the exact dosage required for each individual.
Some physicians are reluctant to increase thyroid medication because of possible adverse side effects such as atrial fibrillation or osteoporosis. In some cases, a regimen of T4 along with T3 (Cytomel) works well along with lifestyle changes such as regular exercise and a healthy diet for gastrointestinal and other breakthrough symptoms.
PROGNOSIS.
Severely hypothyroid conditions accompanied by pronounced atherosclerosis (resulting from abnormal lipid metabolism) may cause angina and other symptoms of CAD. Treatment of hypothyroidism-induced angina can be difficult because thyroid hormone replacement increases the heart’s need for oxygen by increasing body metabolism. This increase in metabolism then precipitates angina and aggravates the anginal condition. In severe hypothyroidism, psychiatric abnormalities can occur and are described as “myxedema madness” in the older literature.
Rarely, severe or prolonged hypothyroidism may progress to myxedema coma when aggravated by stress such as surgery, infection, or noncompliance with thyroid treatment. Myxedema coma can be fatal because of the extreme decrease in the metabolic rate, hypoventilation leading to respiratory acidosis, hypothermia, and hypotension.
Goiter
Goiter, an enlargement of the thyroid gland, may be a result of lack of iodine, inflammation, or tumors (benign or malignant). Enlargement also may appear in hyperthyroidism, especially Graves’ disease. Goiter occurs most often in areas of the world in which iodine, which is necessary for the production of thyroid hormone, is deficient in the diet (Fig. 11-5). Factors that inhibit normal thyroid hormone production result in a negative feedback loop, with hypersecretion of TSH. The TSH increase results in the production and secretion of huge amounts of thyroglobulin (colloid) into the glandular follicles and the gland grows in size.

Figure 11-5 Goiter. The enlarged thyroid gland appears as a swelling of the anterior neck. This condition results from a low dietary intake of iodine and is rare in Canada and the United States but may be seen in other parts of the world. (From Thibodeau GA, Patton KT: The human body in health & disease, ed 4, St Louis, 2005, Mosby.)
Thyroglobulin is the large glycoprotein molecule in which thyroid hormones (T3 and T4) are produced in the presence of iodine. When iodine is absent, only the thyroglobulin is made by the gland in response to repeated TSH stimulation. Because the thyroglobulin molecule is large, its increased production causes rapid glandular growth and a marked increase in overall glandular mass occurs called a colloid goiter.95 With the use of iodized salt and iodine-containing binders in commercial foods, this problem almost has been eliminated in the United States and Canada. Although the younger population in the United States may be goiter-free, aging adults may have developed goiter during their childhood or adolescent years and may still have clinical manifestations of this disorder.
Increased neck size may be observed, and when the thyroid increases to a certain point, pressure on the trachea and esophagus may cause difficulty breathing, dysphagia (difficulty swallowing), and hoarseness. Compression of the upper airway can be a fatal complication. Surgical intervention is essential when the trachea is compromised.
Thyroiditis
Thyroiditis, inflammation of the thyroid, may be classified as acute suppurative (pus forming and very rare), subacute granulomatous (uncommon), and lymphocytic or chronic (Hashimoto’s disease). Acute and subacute thyroiditis are uncommon conditions caused by bacterial (Streptococcus pyogenes, Staphylococcus aureus, and Pneumococcus pneumoniae) and viral agents, respectively. Infected glands are painful and associated with systemic symptoms of fever and hyperthyroidism. Several varieties of related autoimmune causes of thyroiditis exist, such as Hashimoto’s (lymphocytic) thyroiditis and postpartum thyroiditis. These types of thyroiditis are generally painless, with only a rare case of Hashimoto’s causing pain. Only the most common form of Hashimoto’s thyroiditis is discussed further.
Hashimoto’s (chronic) thyroiditis affects women more frequently than it does men (10: 1) and is most often seen in the 30-to 50-year age group. The disorder has an autoimmune basis, and genetic predisposition appears to play a role in the etiology. It is associated with HLA-DR3, which is also present in other autoimmune conditions (e.g., Graves’ disease, systemic lupus erythematosus, type 1 DM, pernicious anemia, myasthenia gravis, and rheumatoid arthritis).157 Hashimoto’s thyroiditis causes destruction of the thyroid gland because of the infiltration of the gland by lymphocytes and antithyroid antibodies. This infiltration results in decreased serum levels of T3 and T4, thus stimulating the pituitary gland to increase the production of TSH.
The increased TSH causes hyperfunction of the tissue, and goiter formation (enlargement of the gland) results. In some cases, this increase in function helps maintain a normal hormonal level, but eventually, when enough of the gland is destroyed, hypothyroidism develops. Hashimoto’s thyroiditis is one of the most common causes of hypothyroidism in women older than 50 years.
Signs of chronic thyroiditis usually include painless symmetric or asymmetric enlargement of the gland and an irregular surface, which occasionally causes pressure on the surrounding structures. This pressure may subsequently cause dysphagia and respiratory distress.
Most clients are euthyroid (have a normally functioning thyroid), about 20% are hypothyroid, and fewer than 5% are hyperthyroid, with these people having combined Hashimoto’s and Graves’ disease caused by a genetic component.111 The course of Hashimoto’s thyroiditis varies. Most people see a decrease in the size of the goiter and remain stable for years with treatment.
Treatment is directed toward suppressing the TSH to the lower end of the normal range to decrease TSH stimulation of the gland, and to correct hypothyroidism if present. Tablets containing thyroxine (T4) can help regulate and maintain adequate levels of circulating hormones. Generally, long-term or permanent therapy is advised.
Thyroid Cancer
Although malignant tumors of the thyroid are rare, thyroid cancer makes up more than 90% of all endocrine cancers and accounts for 63% of deaths from endocrine cancer, with an increasing incidence worldwide.22 In the United States, 33,550 new cases of thyroid cancer were diagnosed in 2007 with 1530 deaths reported the same year.113
Thyroid cancer affects women more than men (2: 1 ratio), mainly between the ages of 40 and 60 years. A past medical history of radiation to the head, neck, or chest (e.g., for an enlarged thymus or tonsils, acne, or Hodgkin’s disease) is the most obvious risk factor.
The usual presentation of thyroid cancer is the appearance of a hard, painless nodule on the thyroid gland or a gland that is multinodular. Most palpable nodules of the thyroid are benign adenomas and rarely become malignant or grow to a significant size to cause pressure against the trachea. Red flag symptoms include vocal cord paralysis, ipsilateral cervical lymphadenopathy, and fixation of the nodule to surrounding tissues.45,107
About 5% of palpable nodules are malignant. Of the malignant nodules, most are a variety that seldom metastasizes beyond regional lymph nodes of the neck, resulting in a good prognosis for most people.
Women have more thyroid nodules and more thyroid cancer than men. However, the presence of a thyroid nodule in a man is regarded with greater suspicion for cancer. Thyroid cancer is diagnosed by fine-needle aspiration (FNA) biopsy. More advanced molecular techniques for the diagnosis of thyroid nodules are being developed.164 Molecular diagnostic assays using tumor-specific markers may improve the sensitivity and accuracy of FNA, possibly reducing the number of surgical procedures to remove lesions that later prove to be benign.207
Treatment usually involves removal of all or part of the thyroid. Neck resection of involved lymph nodes may be done for metastases to the neck. Radioactive ablation of remaining thyroid tissue is standard practice for most thyroid cancers. External radiation may be used in some situations. Major postoperative complications may involve damage to the laryngeal nerve, hemorrhage, and hypoparathyroidism.184
Most thyroid cancers are treatable; however, disease recurrence and metastasis may occur in as many as 20% of affected individuals.22 Individuals treated for thyroid cancer require long-term follow-up to detect recurrent disease, which can present years after initial therapy.136
Parathyroid Glands
Two parathyroid glands are located on the posterior surface of each lobe of the thyroid gland. These glands secrete PTH, which regulates calcium and phosphorus metabolism.
PTH exerts its effect by the following:
1. Increasing the release of calcium and phosphate from the bone (bone demineralization).
2. Increasing the absorption of calcium and excretion of phosphate by the kidneys.
3. Promoting calcium absorption in the GI tract.111
Disorders of the parathyroid glands may come to the therapist’s attention because these conditions can cause periarthritis and tendinitis. Both types of inflammation may be crystal-induced, with formation of periarticular or tendinous calcification. Rarely, ruptured tendons resulting from bone resorption at the insertions occur in cases of primary hyperparathyroidism. These complications and problems are seen infrequently because most cases are diagnosed earlier with the advent of blood screening for identification of asymptomatic hypercalcemia.
Hyperparathyroidism
Definition and Incidence.: Hyperparathyroidism is a disorder caused by overactivity of one or more of the four parathyroid glands that disrupts calcium, phosphate, and bone metabolism. Women are affected more than men (2: 1), usually after age 60 years (postmenopausal). Hyperparathyroidism is frequently overlooked in the over-60 population. Symptoms in the early stages for this group are subtle and easily attributed to the aging process, depression, or anxiety. Eventually the symptoms intensify as the level of serum calcium rises, but this situation is accompanied by increased bone damage and other complications.
Etiologic and Risk Factors.: Hyperparathyroidism is classified as primary, secondary, or tertiary. Primary hyperparathyroidism develops when the normal regulatory relationship between serum calcium levels and PTH secretion is interrupted. This occurs when one or more of the parathyroid glands enlarge, increasing PTH secretion and elevating serum calcium levels.
The most common cause is a single adenoma of the parathyroid gland. Hyperplasia of the gland without an identifying injury and multiple adenomas are less common causes. Medications, such as thiazide diuretics for hypertension and lithium carbonate for psychiatric disorders, have also been implicated as a cause or factor that exacerbates hyperparathyroidism.
Secondary hyperparathyroidism occurs when the glands are hyperplastic from malfunction of another organ system. A hypocalcemia-producing abnormality outside the parathyroid gland results in a compensatory response of the parathyroid glands to chronic hypocalcemia. This is usually the result of renal failure (decreased renal activation of vitamin D), but it also may occur with osteogenesis imperfecta, Paget’s disease, multiple myeloma, carcinoma with bone metastasis, laxative abuse, and vitamin D deficiency.
Tertiary hyperparathyroidism is seen almost exclusively in dialysis clients who have long-standing secondary hyperparathyroidism. Hyperplasia occurs and the parathyroid glands ultimately become autonomous in function and unresponsive to serum calcium levels. Parathyroidectomy is required even after successful renal transplantation has resolved the cause of the secondary hyperparathyroidism.73
Pathogenesis and Clinical Manifestations.: The primary function of PTH is to maintain a proper balance of calcium and phosphorus ions within the blood. PTH is not regulated by the pituitary or the hypothalamus and maintains normal blood calcium levels by increasing bone resorption and GI absorption of calcium. It also maintains an inverse relationship between serum calcium and phosphate levels by inhibiting phosphate reabsorption in the renal tubules.
Abnormal PTH production disrupts this balance; symptoms of hyperparathyroidism are related to this release of bone calcium into the bloodstream. Excessive circulating PTH leads to bone damage, hypercalcemia, and kidney damage (Table 11-7). In fact, hyperparathyroidism is the most common cause of hypercalcemia, which can lead to nervous system, musculoskeletal, metabolic, and cardiovascular problems.
Table 11-7
Systemic Manifestations of Hyperparathyroidism
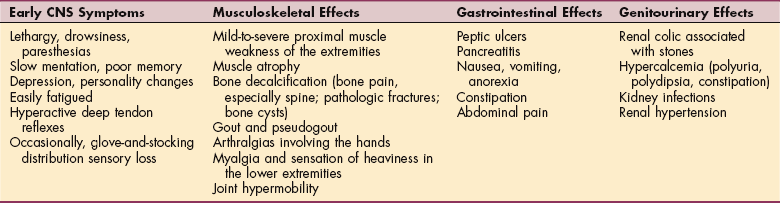
Modified from Goodman CC, Synder TE: Differential diagnosis for physical therapists: Screening for referral, ed 4, Philadelphia, 2007, WB Saunders. CNS, Central nervous system.
Bone Damage.: Oversecretion of PTH causes excessive osteoclast growth and activity within the bones. Osteoclasts are active in promoting resorption of bone, which then releases calcium into the blood, causing hypercalcemia. This calcium loss leads to bone demineralization, and in time, the bones may become so fragile that pathologic fractures, deformity (e.g., kyphosis of the thoracic spine), and compression fractures of the vertebral bodies occur. If uncontrolled, osteoclast proliferation may cause lytic bone lesions (bone disintegrates, leaving holes).
Surgical treatment of hyperparathyroidism (parathyroidectomy) can be expected to result in biochemical cure and increased bone mineral density of the lumbar spine and femoral neck (areas rich in cancellous bone), in both symptomatic and asymptomatic clients. Cortical bone loss, however, is not as readily reversible in either group.225 Early surgical treatment of hyperparathyroidism may assist in the prevention of spine and hip fractures in this population.
Hypercalcemia.: As excessive PTH secretion results in bone resorption and hypercalcemia as just described, hypercalciuria (excessive calcium in the urine) eventually develops because the excessive filtration of calcium overwhelms this renal mechanism. High serum calcium levels also stimulate hypergastrinemia (excess gastrin, a hormone that stimulates secretion of gastric acid and pepsin in the blood), abdominal pain, peptic ulcer disease, and pancreatitis.
Kidney Damage.: As serum calcium levels rise in response to excessive PTH levels, large amounts of phosphorus and calcium are excreted and lost from the body. Excretion of these compounds occurs through the renal system, leaving deposits of calcium phosphate within the renal tubules. This produces a kidney condition called nephrocalcinosis. Because calcium salts are insoluble in urine, kidney stones composed of calcium phosphate develop. Serious renal damage may not be reversible with parathyroidectomy.
Some people with hyperparathyroidism may be completely asymptomatic, but even seemingly asymptomatic clients with elevated serum and PTH levels have been found to have paresthesias, muscle cramps, and loss of pain and vibratory sensation in a stocking-glove distribution. Others suffer from a wide range of symptoms as a result of skeletal disease, renal involvement, GI tract disorders, and neurologic abnormalities.
MEDICAL MANAGEMENT
The diagnosis of hyperparathyroidism depends on measurement of PTH levels in persons found to be hypercalcemic (high serum levels of calcium). Serum calcium and PTH levels are elevated, serum phosphorus may be low normal or depressed, and urine calcium can range from low to high. Radiographic evidence of skeletal damage is important to measure in asymptomatic clients with mild hyperparathyroidism. Skeletal damage can be seen on x-ray as diffuse demineralization of bones, bone cysts, subperiosteal bone resorption, and loss of the laminae durae surrounding the teeth.
TREATMENT AND PROGNOSIS.
Treatment for primary hyperparathyroidism is surgical removal (parathyroidectomy). Minimally invasive parathyroidectomy is advised even for individuals with mild elevation in calcium because of the risk for more serious complications of hyperparathyroidism such as renal failure, osteoporosis, and early death from CVD. Preoperative localization of the adenoma by scanning with technetium (Tc 99m-labeled sestamibi) allows for limited resection whenever possible.
The prognosis is good if the condition is identified and treated early. Untreated, hyperparathyroidism exacerbates many conditions among older adults such as osteoporosis and CAD. Emergency medical management of severe hypercalcemia includes use of drugs to lower serum calcium, such as hydration and loop diuretics, which promote calcium loss through the kidneys, and antiresorption agents, which inhibit calcium release from bone.
Long-term medical management of hypercalcemia with drugs is not as effective as parathyroid surgery, but if needed for short-term treatment, drugs, such as calcimimetics, bisphosphonates, estrogen, and calcitonin, can prevent progressive bone demineralization.230
Hypoparathyroidism
Definition.: Hyposecretion, hypofunction, or insufficient secretion of PTH are ways to describe hypoparathyroidism.19 Because the parathyroid glands primarily regulate calcium balance, hypoparathyroidism causes hypocalcemia and produces a syndrome opposite that of hyperparathyroidism with abnormally low serum calcium levels, high serum phosphate levels, and possible neuromuscular irritability (tetany) (Box 11-4).
Etiologic Factors and Incidence.: Hypoparathyroidism is either iatrogenic, which is most common, or idiopathic. Iatrogenic (acquired) causes include accidental removal of the parathyroid glands during thyroidectomy or anterior neck surgery. Variations in location and color in addition to the minute size of parathyroid glands make identification difficult and may result in glandular damage or accidental removal during thyroid removal or anterior neck surgery. Other iatrogenic causes can include infarction of the parathyroid glands as a result of an inadequate blood supply to the glands during surgery, strangulation of one or more of the glands by postoperative scar tissue, and rarely, massive thyroid irradiation. Other secondary causes of hypoparathyroidism may include hemochromatosis, sarcoidosis, amyloidosis, tuberculosis, neoplasms, or trauma. Idiopathic causes affect children nine times as often as adults and affect twice as many women as men. Like Graves’ disease and Hashimoto’s thyroiditis, idiopathic hypoparathyroidism may be an autoimmune disorder with a genetic basis.
Pathogenesis.: See the section on Pathogenesis of Hyperparathyroidism in this chapter for a description of the regulation of calcium and phosphate by PTH and the parathyroid glands. PTH normally functions to increase bone resorption to maintain a proper balance between serum calcium and phosphate. When parathyroid secretion of PTH is reduced, bone resorption and GI tract absorption slow, serum calcium levels fall, and severe neuromuscular irritability develops. Calcifications may form in various organs such as the eyes and basal ganglia. Serum phosphate levels rise without sufficient PTH because fewer phosphorus ions are secreted by the distal tubules of the kidneys with decreased renal excretion of phosphorus.
Clinical Manifestations.: Mild hypoparathyroidism may be asymptomatic, but it usually produces hypocalcemia and high serum phosphate levels that affect the CNS and other body systems (Table 11-8). The most significant clinical consequence of hypocalcemia associated with hypoparathyroidism is neuromuscular irritability. In people with chronic hypoparathyroidism, this neuromuscular irritability may result in tetany. Hypocalcemia resistant to PTH, called pseudohypoparathyroidism, is determined genetically and is associated with shortened metacarpals and metatarsals.125
Table 11-8
Systemic Manifestations of Hypoparathyroidism
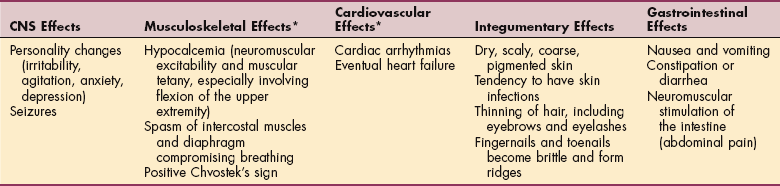
*The therapist should be aware of musculoskeletal and cardiovascular effects, which are the most common and important.
Modified from Goodman CC, Snyder TE: Differential diagnosis for physical therapists, ed 4, St Louis, 2007, WB Saunders.
Acute (overt) tetany begins with a tingling in the fingertips, around the mouth, and occasionally, the feet. This tingling spreads and becomes more severe, producing painful muscle tension, spasms, grimacing, laryngospasm, and arrhythmias. Trousseau’s sign (carpal spasm) and Chvostek’s sign (hyperirritability of the facial nerve, producing a characteristic spasm when tapped) are apparent on examination (see Figs. 5-8 and 5-9). In severe cases, a tracheostomy may be required to correct acute respiratory obstruction secondary to laryngospasm.
MEDICAL MANAGEMENT
Diagnosis of this condition is based on history, clinical presentation, examination, and laboratory values (low serum calcium, high serum phosphate, or low or absent urinary calcium). Radioimmunoassay for PTH demonstrates decreased PTH concentration.
TREATMENT.
Acute hypoparathyroidism, with its major manifestation of acute tetany, is a life-threatening disorder. Treatment is directed toward elevation of serum calcium levels as rapidly as possible with intravenous calcium, prevention or treatment of convulsions, and control of laryngeal spasm and subsequent respiratory obstruction. Treatment of chronic hypoparathyroidism with pharmacologic management is accomplished more gradually than treatment for an acute situation. Surgical intervention is not appropriate and, in fact, is often the cause of this condition (see the section on Etiologic Factors).
PROGNOSIS.
Full recovery from the effects of hypoparathyroidism is possible when the condition is diagnosed early, before the development of serious complications. Unfortunately, once formed, cataracts and brain (basal ganglion) calcifications are irreversible. Death can occur from respiratory obstruction secondary to tetany and laryngospasms if treatment is not initiated early in acute hypoparathyroidism.
Adrenal Glands
The adrenals are two small glands located on the upper part of each kidney (see Fig. 11-1). Each adrenal gland consists of two relatively discrete parts: an outer cortex and an inner medulla. The outer cortex is responsible for the secretion of mineralocorticoids (steroid hormones that regulate fluid and mineral balance), glucocorticoids (steroid hormones responsible for controlling the metabolism of glucose), and androgens (sex hormones).
The centrally located adrenal medulla is derived from neural tissue and secretes epinephrine and norepinephrine, which exert widespread effects on vascular tone, the heart, and the nervous system, and affects glucose metabolism. Together, the adrenal cortex and medulla are major factors in the body’s response to stress.
Glandular hypofunction and hyperfunction characterize the major disorders of the adrenal cortex. Underactivity of the adrenal cortex results in a deficiency of glucocorticoids, mineralocorticoids, and adrenal androgens. Overactivity results in excessive production of these same hormones.
Adrenal Insufficiency
Hypofunction of the adrenal cortex can originate from a disorder within the adrenal gland itself (primary adrenal insufficiency) or it may be due to hypofunction of the pituitary-hypothalamic unit (secondary adrenal insufficiency).230 Adrenocortical insufficiency, whether primary or secondary, can be either acute or chronic.
Primary Adrenal Insufficiency (Addison’s Disease):
Definition and Overview.: Addison’s disease is a condition that occurs as a result of a disorder within the adrenal gland itself, with insufficient cortisol release from the adrenal glands causing a wide range of problems. Addison’s disease was named for the physician who first studied and described the associated symptoms. Adrenal insufficiency affects about 4 adults in 100,000 each year in the United States. Both sexes are affected, but the incidence is slightly higher in women than men. Addison’s disease can occur anytime across the lifespan with a preponderance of cases during middle age (40 to 60 years).
Primary forms of adrenal insufficiency are uncommon; the therapist is most likely to see secondary adrenal insufficiency as a result of suppression of ACTH by steroid therapy or secondary to opportunistic infections related to human immunodeficiency virus (HIV).
Etiologic Factors.: At one time, most causes of Addison’s disease occurred as a complication of tuberculosis, but now most cases are considered idiopathic or autoimmune. Because more than half of all people with idiopathic Addison’s disease have circulating autoantibodies that react specifically against adrenal tissue, this condition is considered to have an autoimmune basis.
Less frequent causes of primary insufficiency include bilateral adrenalectomy, adrenal hemorrhage or infarction, radiation to the adrenal glands, malignant adrenal neoplasm, and infections (e.g., histoplasmosis or cytomegalovirus). Destruction of the adrenal glands by chemical agents has been reported.184 Medications, such as antifungals, adrenolytic agents, etomidate, rifampin, phenytoin, and phenobarbital, can also trigger Addison’s disease.
Risk Factors.: Surgery (including dental procedures); pregnancy (especially with postpartum hemorrhage); accident, injury, or trauma; infection; salt loss resulting from profuse diaphoresis (hot weather or with strenuous physical exertion); or failure to take steroid therapy in persons who have chronic adrenal insufficiency can cause acute adrenal insufficiency.
Pathogenesis and Clinical Manifestations.: This adrenal gland disorder results in decreased production of cortisol (a glucocorticoid) and aldosterone (a mineralocorticoid), two of the primary adrenocortical hormones. Glucocorticoid deficiency causes widespread metabolic disturbances. Consequently, when glucocorticoids become deficient, gluconeogenesis decreases, with resultant hypoglycemia and liver glycogen deficiency. The person grows weak, exhausted, hypotensive, and suffers from anorexia, weight loss, nausea, and vomiting. Emotional disturbances can develop, ranging from mild neurotic symptoms to severe depression. Glucocorticoid deficiency also diminishes resistance to stress.
In anyone who has previously been diagnosed with Addison’s disease, acute symptoms, such as severe abdominal pain, low back or leg pain, severe vomiting, diarrhea, and hypotension, may develop quickly in response to triggers such as trauma, infarction, or infection. The resulting condition, called addisonian crisis, can progress quickly to hypovolemic shock (e.g., hypotension, tachycardia, and loss of consciousness) from rapid fluid loss.
Chronic adrenal insufficiency with chronic cortisol deficiency results in a failure to inhibit anterior pituitary secretion of ACTH. The result is a simultaneous increase in ACTH secretion and melanocyte-stimulating hormone (MSH) (see Fig. 11-2); excessive MSH increases skin and mucous membrane pigmentation. Persons with Addison’s disease may have a bronzed or tanned appearance, which is the most striking physical finding with primary adrenal insufficiency (not present in all people with this disorder). This change in pigmentation may vary in the white population from a slight tan or a few black freckles to an intense generalized pigmentation. The change in pigmentation is most commonly observed over extensor surfaces such as the backs of the hands (metacarpophalangeal joints), elbows, knees, creases of the hands, lips, and mouth. Increased pigmentation of scars formed after the onset of the disease is common. Members of darker-skinned races may develop a slate-gray color that is obvious only to family members.
Aldosterone deficiency causes numerous fluid and electrolyte imbalances. Aldosterone normally promotes conservation of sodium and therefore conserves water and excretion of potassium. A deficiency of aldosterone causes increased sodium excretion, dehydration (see symptoms listed in Box 5-8), hypotension (low blood pressure causing orthostatic symptoms, see Chapter 12), and decreased cardiac output affecting heart size (decrease in size). Eventually, hypotension becomes severe and cardiovascular activity weakens, leading to circulatory collapse, shock, and death. Excess potassium retention (greater than 7 mEq/L) can result in arrhythmias and possible cardiac arrest.
Other clinical effects include decreased tolerance for even minor stress, poor coordination, fasting hypoglycemia (resulting from decreased gluconeogenesis), and a craving for salty food. Addison’s disease may also retard axillary and pubic hair growth in females, decrease the libido (from decreased androgen production), and in severe cases, cause amenorrhea (absence of menstruation).265
MEDICAL MANAGEMENT
Diagnosis of Addison’s disease depends primarily on blood and urine hormonal assays and cortisol response to synthetic ACTH administration. Decreased serum cortisol levels are the hallmark of Addison’s disease. An ACTH stimulation test can help identify the presence of Addison’s disease and the type. In an ACTH stimulation test, baseline measurements of blood and urine cortisol levels are measured. The individual receives an intramuscular injection or intravenous infusion of ACTH to stimulate cortisol secretion. Blood cortisol and aldosterone measurements are repeated 30 to 60 minutes later. These levels should be greater than baseline levels; with adrenal insufficiency, the levels do not rise or rise only slightly.
If the ACTH stimulation test is positive, a CRH stimulation test is conducted to determine if the adrenal insufficiency is primary or secondary. After injection of synthetic CRH, blood cortisol measurements are taken. A high ACTH level without increased cortisol signals primary adrenal insufficiency. An absent or delayed ACTH response without deficient cortisol level indicates secondary adrenal insufficiency.105
Complications from Addison’s disease, such as hyponatremia, hypoglycemia, hyperkalemia, hypercalcemia, and metabolic acidosis, will be apparent in the blood chemistry values obtained.
TREATMENT.
Acute adrenal insufficiency is treated by replacing fluids, electrolytes, glucose, and cortisol while identifying the underlying cause of the problem. Medical management for chronic adrenal insufficiency is primarily pharmacologic, consisting of life-long administration of synthetically manufactured corticosteroids and mineralocorticoids (fludrocortisone). If untreated, Addison’s disease is ultimately fatal. Adrenal crisis requires immediate hospitalization and treatment.
Secondary Adrenal Insufficiency.: Secondary adrenal insufficiency is caused by other conditions outside the adrenals, such as hypothalamic or pituitary tumors, removal of the pituitary, or other causes of hypopituitarism, or too-rapid withdrawal of corticosteroid drugs. Long-term exogenous corticosteroid stimulation suppresses pituitary ACTH secretion and results in adrenal gland atrophy. Untimely discontinuation of adrenocorticosteroid therapy results in acute adrenal insufficiency and can become a life-threatening emergency. Anyone receiving adrenocorticosteroid therapy should be identified through the use of a bracelet or necklace. Steroid therapy must be discontinued gradually so that pituitary and adrenal function can normalize.
Clinical manifestations of secondary disease are somewhat different from symptoms of primary adrenal insufficiency. Whereas most symptoms of primary adrenal insufficiency arise from cortisol and aldosterone deficiency, symptoms of secondary disease are related to cortisol deficiency only. Because the gland is still intact, aldosterone is secreted normally, but the lack of stimulation from ACTH results in deficient cortisol secretion. Arthralgias, myalgias, and tendon calcification can occur, which resolve with treatment of the underlying condition.
Hyperpigmentation is not part of the clinical presentation because ACTH and MSH levels are low. Additionally, because aldosterone secretion may continue at fairly normal levels in secondary adrenal hypofunction this condition does not necessarily cause accompanying hypotension and electrolyte abnormalities.18
As with primary adrenal insufficiency, treatment involves replacement of ACTH and monitoring for fluid and electrolyte imbalances. Too much cortisol replacement can result in the development of Cushing’s syndrome (see next section).
Adrenocortical Hyperfunction
Hyperfunction of the adrenal cortex can result in excessive production of glucocorticoids, mineralocorticoids, and androgens. The three major conditions of adrenocortical hyperfunction are Cushing’s syndrome (glucocorticoid excess), Conn’s syndrome or aldosteronism (aldosterone excess), and adrenal hyperplasia (adrenogenital syndrome). This last condition is rare and congenital and is not discussed further in this text.
Definition and Overview.: Hypercortisolism is a general term for an excess of cortisol in the body. This condition can occur as a result of hyperfunction of the adrenal gland (usually benign or malignant adenomas), an excess of corticosteroid medication, or an excess of ACTH stimulation from the pituitary gland (or other sites). ACTH secreted by the pituitary has a key role in cortisol release. When the hypothalamus senses low ACTH levels in the blood, it sends CRH to the pituitary to stimulate ACTH secretion, which in turn stimulates the adrenal glands to release cortisol. When blood cortisol levels are adequate or elevated, the hypothalamus and pituitary release less CRH and ACTH.
Hypercortisolism resulting from adrenal gland oversecretion or from hyperphysiologic doses of corticosteroid medications is called Cushing’s syndrome. When the hypercortisolism results from oversecretion of ACTH from the pituitary, the condition is called Cushing’s disease. The clinical presentation is the same for both conditions.160
A separate condition called pseudo-Cushing’s syndrome occurs when conditions, such as depression, alcoholism, estrogen therapy, or eating disorders, cause changes similar to those of Cushing’s syndrome. In pseudo-Cushing’s syndrome, the symptoms will go away when the cause is eliminated.
Etiologic Factors and Incidence.: The primary causes of Cushing’s syndrome are hyperphysiologic doses of adrenocorticosteroids and adrenocortical tumors. Cushing’s disease results from pituitary adenomas, which secrete an excess of ACTH causing overstimulation of a normal adrenal gland. A similar condition can occur as a result of ectopic production of ACTH by lung cancer and more rarely, by carcinoid tumors. Tumors that can produce ACTH add to the normal ACTH production by the pituitary and stimulate excessive cortisol secretion.
Therapists are more likely to treat people who have developed medication-induced Cushing’s syndrome. This condition occurs after these individuals have received large doses of cortisol (also known as hydrocortisone) or cortisol derivatives. Exogenous steroids are administered for a number of inflammatory and other disorders such as asthma or rheumatoid arthritis (see Box 5-7). Noniatrogenic Cushing’s syndrome occurs mainly in women, with an average age at onset of 20 to 40 years, although it can be seen in people up to age 60 years.
Pathogenesis and Clinical Manifestations.: Cushing’s syndrome involves the HPA axis causing excess cortisol release from the adrenal glands. When the normal function of the glucocorticoids becomes exaggerated, a wide range of physiologic responses can be triggered, including hyperglycemia, hypertension, proximal muscle wasting, and osteoporosis (Table 11-9; see also Table 5-4).
Table 11-9
Pathophysiology of Cushing’s Syndrome
| Physiologic Effect | Clinical Result |
| Persistent hyperglycemia | “Steroid diabetes” |
| Protein tissue wasting | Weakness as a result of muscle wasting; capillary fragility resulting in ecchymoses; osteoporosis as a result of bone matrix wasting |
| Potassium depletion | Hypokalemia (see Table 5-12), cardiac arrhythmias, muscle weakness, renal disorders |
| Sodium and water retention | Edema and hypertension |
| Hypertension | Predisposes to left ventricular hypertrophy, congestive heart failure, cerebrovascular accidents |
| Abnormal fat distribution | Moon-shaped face; dorsocervical fat pad; truncal obesity, slender limbs, thinning of the skin with striae on the breasts, axillary areas, abdomen, and legs |
| Increased susceptibility to infection; lowered resistance to stress | Absence of signs of infection; poor wound healing |
| Increased production of androgens | Virilism in women (e.g., acne, thinning of scalp hair, hirsutism or abnormal growth and distribution of hair) |
| Mental changes | Memory loss, poor concentration and thought processes, euphoria, depression (“steroid psychosis,” see Chapter 5) |
Cortisol has a key role in glucose metabolism and a lesser part in protein, carbohydrate, and fat metabolism. Cortisol also helps maintain blood pressure and cardiovascular function while reducing the body’s inflammatory responses. Overproduction of cortisol causes liberation of amino acids from muscle tissue with resultant weakening of protein structures (specifically muscle and elastic tissue). The end result may include a protuberant abdomen (Fig. 11-6) with purple striae (stretch marks), poor wound healing, thinning of the skin, generalized muscle weakness, and marked osteoporosis that is made worse by an excessive loss of calcium in the urine. In severe cases of prolonged Cushing’s syndrome, muscle weakness and demineralization of bone may lead to pathologic fractures and wedging of the vertebrae, kyphosis (Fig. 11-7), osteonecrosis (especially of the femoral head), bone pain, and back pain.
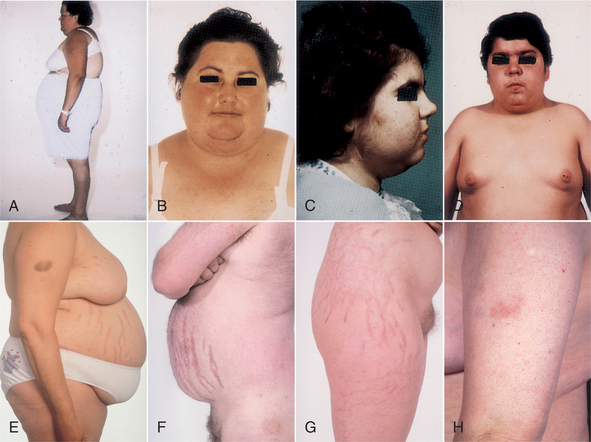
Figure 11-6 Clinical features of Cushing’s syndrome. A, Central and some generalized obesity and dorsal kyphosis in a 30-year-old woman with Cushing’s disease. B, Same woman as in A, showing moon facies (round face), hirsutism (hair growth), and enlarged supraclavicular fat pads. C, Facial rounding, hirsutism, and acne in a 14-year-old girl with Cushing’s. D, Central and generalized obesity and moon facies in a 14-year-old boy with Cushing’s. E and F, Typical central obesity with visible abdominal striae (“stretch marks”) seen in a 41-year-old woman and 40-year-old-man with Cushing’s. G, Striae in a 24-year-old woman with congenital adrenal hyperplasia treated with excessive doses of dexamethasone as replacement therapy. H, Typical bruising and thin skin of Cushing’s syndrome. In this case, the bruising has occurred without obvious injury. (From Larsen RP: Williams textbook of endocrinology, ed 10, Philadelphia, 2003, WB Saunders.)
Figure 11-7 Buffalo hump and hypertrichosis (excessive hairiness; hirsutism) in a male with Cushing’s syndrome. This hump is a painless accumulation of fat that also may occur idiopathically in (usually) women. A familial pattern may exist (i.e., affected women report a similar anatomic change in their mothers). In the case of steroid-induced changes, this condition resolves when the individual stops taking the medication; in such cases, therapeutic intervention by the therapist has no permanent effect. Idiopathic fat deposits and underlying postural changes can be altered with postural correction and soft tissue and joint mobilization techniques. No studies have substantiated whether these changes are long-term. (From Callen JP, Jorizzo J: Dermatological signs of internal disease, ed 2, Philadelphia, 1995, WB Saunders.)
The effect of increased circulating levels of cortisol on the muscles varies from slight to marked. Muscle wasting can be so extensive that the condition simulates muscular dystrophy. Marked weakness of the quadriceps muscle often prevents affected people from rising out of a chair unassisted. Cortisone-induced myopathies are discussed in Chapter 5.
Whenever corticosteroids are administered, the increase in serum cortisol levels triggers a negative feedback signal to the anterior pituitary gland to stop its secretion of ACTH. This decrease in ACTH stimulation of the adrenal cortex results in adrenocortical atrophy during the period of exogenous corticosteroid administration. If these medications are stopped suddenly rather than reduced gradually, the atrophied adrenal gland will not be able to provide the cortisol necessary for physiologic needs. A life-threatening situation known as acute adrenal insufficiency can develop, requiring emergency cortisol replacement (see the discussion on Addisonian Crisis in the Primary Adrenal Insufficiency section in this chapter).230
MEDICAL MANAGEMENT
Although there is a classic cushingoid appearance in persons with hypercortisolism (see Fig. 11-6), diagnostic laboratory studies, including measurement of urine and serum cortisol, are used to confirm the diagnosis. If the initial laboratory tests are positive (elevated cortisol levels), then a dexamethasone suppression test may be done to determine the cause. Dexamethasone, a corticosteroid, signals the pituitary to decrease ACTH secretion. The dexamethasone is administered at night, and serum cortisol levels are measured the next morning. The normal response should be a decrease in blood and urine cortisol levels, signaling ACTH suppression. For the individual with Cushing’s syndrome, cortisol levels remain elevated.104
Serum ACTH levels help determine whether Cushing’s syndrome is ACTH-dependent (e.g., pituitary tumor) or ACTH-independent (adrenal tumor). Further testing (pituitary magnetic resonance imaging [MRI] and abdominal computed tomography [CT] scan) is determined on the basis of these results. X-rays or dual-energy x-ray absorptiometry scans may be needed to assess for fractures or to rule out osteopenia or osteoporosis, respectively. These tests may be conducted to obtain a baseline measurement of bone density or they may be obtained in response to an individual’s report of musculoskeletal symptoms such as bone pain or backache.104
TREATMENT AND PROGNOSIS.
Treatment to restore hormone balance and reverse Cushing’s syndrome or disease may require radiation, drug therapy, or surgery, depending on the underlying cause (e.g., resection of tumors). For individuals with muscle wasting or at risk for muscle atrophy, a high-protein diet may be prescribed. Prognosis depends on the underlying cause and the ability to control the cortisol excess. Cortisol-secreting tumors can recur, thus follow-up screening is advised.
Definition and Overview.: Conn’s syndrome, or primary aldosteronism, occurs when an adrenal lesion results in hypersecretion of aldosterone, the most powerful of the mineralocorticoids. Its primary role is to conserve sodium, and it also promotes potassium excretion. The major cause of primary aldosteronism (an uncommon condition present most often in women aged 30 to 50 years) is a benign, aldosterone-secreting tumor called aldosteronoma.79 Rarely, Conn’s syndrome develops as a consequence of adrenocortical carcinoma.
Secondary hyperaldosteronism also can occur as a consequence of pathologic lesions that stimulate the adrenal gland to increase production of aldosterone. For example, conditions that reduce renal blood flow (e.g., renal artery stenosis), induce renal hypertension (e.g., nephrotic syndrome or ingestion of oral contraceptives), and edematous disorders (e.g., cardiac failure or cirrhosis of the liver with ascites) can cause secondary hyperaldosteronism.
Pathogenesis and Clinical Manifestations.: Aldosterone affects the tubular reabsorption of sodium and water and the excretion of potassium and hydrogen ions in the renal tubular epithelial cells; an excess of aldosterone enhances sodium reabsorption by the kidneys. This leads to the development of hypernatremia (excess sodium in blood, indicating water loss exceeding sodium loss), hypervolemia (fluid volume excess, increase in the volume of circulating fluid or plasma in the body), hypokalemia (low blood levels of potassium), and metabolic alkalosis (see Chapter 5). With the hypervolemia and hypernatremia, the blood pressure increases, often to very high levels, and renin production is suppressed. This hypertension can lead to cerebral infarctions and renal damage.
Without intervention, complications of chronic hypertension develop in the presence of hypertension, hypernatremia, and hypokalemia, heart failure, renal damage, and cerebrovascular accident. Hypokalemia results from excessive urinary excretion of potassium causing muscle weakness; intermittent, flaccid paralysis; paresthesias; or cardiac arrhythmias (see Table 5-12).
This excessive urinary excretion of potassium (hypokalemia) leads to polyuria and resulting polydipsia (excessive thirst). DM is common because hypokalemia interferes with normal insulin transport. Finally, hypokalemia leads to metabolic alkalosis, which can cause a decrease in ionized calcium levels, resulting in tetany and respiratory suppression. However, low serum potassium and alkalosis are not always present at the time of diagnosis.
MEDICAL MANAGEMENT
DIAGNOSIS, TREATMENT, AND PROGNOSIS.
Diagnosis of primary hyperaldosteronism is based on elevations in serum and urine aldosterone studies and CT scanning of the abdomen for evidence of unilateral and sometimes bilateral adenomas of the adrenal gland.79 Radiographic studies can reveal cardiac hypertrophy resulting from chronic hypertension. Radionuclide scanning techniques using radiolabeled substances allow visualization of any tumors present.
The goals of treatment are to reverse hypertension, correct hypokalemia, and prevent kidney damage. Surgical removal of the aldosterone-secreting tumor may also require adrenalectomy, which completely resolves the hypertension within 1 to 3 months. However, without early diagnosis and treatment, renal complications from long-term hypertension may be progressive. Pharmacologic treatment to increase sodium excretion and treat the hypertension and hypokalemia is a nonsurgical alternative.
Adipose Tissue
One does not normally think of fat as endocrine tissue. In fact, adipose tissue can be classified as the largest endocrine organ in the body. This revelation was made only a few years ago. Before that time, fat was viewed as a storage site for energy, with little other function. Now it is clear that neurotransmitters and glucose (along with other molecules) directly act on adipocytes to induce the release of a number of different proteins collectively termed adipokines (or adipocytokines) that can act locally as autocrine hormones or through the blood stream as endocrine hormones.93
Some of the factors released by adipocytes function to maintain the balance of energy. However, others have roles that are beyond the conservation of energy, including the induction of vasoconstriction (angiotensin), inflammation (leptin), or angiogenesis (vascular endothelial growth factor [VEGF]).
Research supporting the role of adipocytes in the secretion of molecules that are transported in the blood has only recently begun. Thus this concept is very new to both researchers and professionals in the health care field. In other portions of this chapter, diseases are discussed according to the altered hormone or organ. Too little is known about the role of adipocytes in disease to present a great deal of detail in this section. Only a few examples of current lines of research are provided.
One of the reasons that the role of adipose tissue in health and disease has been difficult to describe is due to the fact that fat from different parts of the body functions in various ways. In mammals, adipose tissue is divided into two categories: brown and white. Brown fat is a very specialized tissue that is important in thermoregulation, converting energy from food into heat.33 Infants have more brown fat, thus they are more sensitive to temperature changes, especially the cold. The amount of brown fat decreases into adulthood, but some does remain in specific locations through the lifespan. The activity of the adipocytes in brown fat is closely regulated by the sympathetic nervous system.
White fat is the classic adipose tissue responsible for storage of triglycerols to provide a long-term reservoir of energy for the body.247 As an endocrine organ, the secretion of adipokines by white visceral tissue is stressed, yet it is important to remember that the major secregogue (also secretagogue, a stimulating agent) from adipocytes is fatty acids, which are used as a source of energy.
White visceral tissue secretes proteins identified as either “bad adipokines” (tumor necrosis factor [TNF], resistin, interleukin-6 [IL-6], IL-8, acylation-stimulation protein, and angiotensinogen and plasma activator inhibitor-1) or “good adipokines” (leptin and adiponectin).208 These secreted molecules seem to play important regulatory roles in a variety of complex processes, including fat metabolism, feeding behavior, hemostasis, vascular tone, energy balance, and insulin sensitivity.
Obesity
Obesity is the most common nutritional disorder in the Western world and has been categorized as a disease process by the U.S. Social Security Administration.249 Their policy describes obesity as a “complex, chronic disease characterized by excessive accumulation of body fat.”
Obviously there is a direct correlation with adipose cells and obesity—more adipose tissue results in a more obese person. However, obesity is a multifactorial problem related both to behavior and biology. The role of fat cells to send signals to the brain that are translated as feelings of hunger or satiation has only recently been uncovered.
Leptin, the first adipokine identified, acts on the hypothalamus to alter hunger, with increased levels of leptin acting as a hunger depressor. Leptin binds to neurons within the CNS that contain neuropeptide Y, thus confirming its action.119 In humans and in animal models, the level of leptin highly correlates with the levels of adipose tissue.151 Transgenic animals that lack the ability to make leptin are extremely obese and develop diabetes. If the animals are treated with leptin, there is a subsequent reduction in the animal’s food intake and substantial weight loss.187
In human obesity, which has many causes, the role of leptin in hunger is not as clear. As a person becomes more obese, the leptin levels increase, which is the opposite of the expected result. It appears that the target receptors for leptin become less sensitive with increasing amounts of fat.93
It is now thought that leptin’s role in appetite regulation is more important in reduced calorie situations. When calories are restricted and weight loss is occurring, leptin levels decrease. Historically, when humans had to gather their own food, this signal to the brain was important to stave off starvation. Leptin’s role in hunger for persons that have sufficient calorie intake is unclear.
Obesity and Cancer.: The link between obesity and cancer in humans has been apparent for years and continues to be borne out by statistics.86,94 In a major study undertaken in Austria, obesity in men was associated with a high risk of colon cancer and pancreatic cancer. In women, there was a weak positive association between increasing body mass index (BMI) and all cancers combined and strong associations with non-Hodgkin’s lymphomas and uterine cancer.201 In addition, the incidence of breast cancer was positively associated with high BMI but only after 65 years of age. Along with the greater risk of developing cancer in the obese, the outcome for obese cancer patients is significantly worse compared to the lean cancer patient in terms of reoccurrence of the cancer, malignancy, and lifespan.112,196
Although the epidemiologic data strongly link some cancers with obesity, the physiologic mechanisms are only beginning to be elucidated. Leptin has been identified as a major adipokine linking prostate cancer with obesity.14 Along with an increase in leptin levels in obese men, a decrease in the adiponectin levels is also associated with prostate cancer.28
In animal models that develop abdominal obesity, a high likelihood of hyperinsulinemia and precancerous changes in the colon have been observed.47 In studies in men, low plasma adiponectin levels resulted in a higher risk of colorectal cancer.262 At the cellular level, leptin has been shown to cause increased proliferation and precancerous changes in cultured colon epithelial cells.72
Obesity has been associated with an increased risk of breast cancer and a reduced survival rate in women with invasive breast cancer.196 Obese women have higher levels of plasma leptin,112 and high levels of leptin have been found in some types of breast cancer.80 The exact mechanism by which leptin would induce breast cancer growth is unknown.
The number of newly identified adipokines continues to grow with the addition of apelin, visfatin, and others.247 The field of adipose endocrinology is in its infancy and will expand our understanding of the dynamic role of fat in health and disease.
Type 2 Diabetes Mellitus
Although type 2 DM is discussed later in this chapter, the role of adipose tissue is presented here. Although excess white fat in any location may lead to the progression of diabetes, it is the fat surrounding the viscera and the hepatic circulation that is most important. Visceral adipose tissue is best defined as the collection of intraabdominal adipose deposits that includes omental and intrahepatic fat.12
Interestingly, even abdominal obesity has subcategories. MRI and CT scans indicate that some people carry abdominal weight in the subcutaneous region, but the distribution of fat around the organs is minimal. These people appear to have little insulin resistance. In contrast, when intraabdominal fat accumulates around the organs (visceral fat) (Fig. 11-8), insulin resistance quickly follows.76 The molecular link between abdominal fat and insulin resistance has been identified as the adipokine, adiponectin, which is released into the circulation by adipocytes.
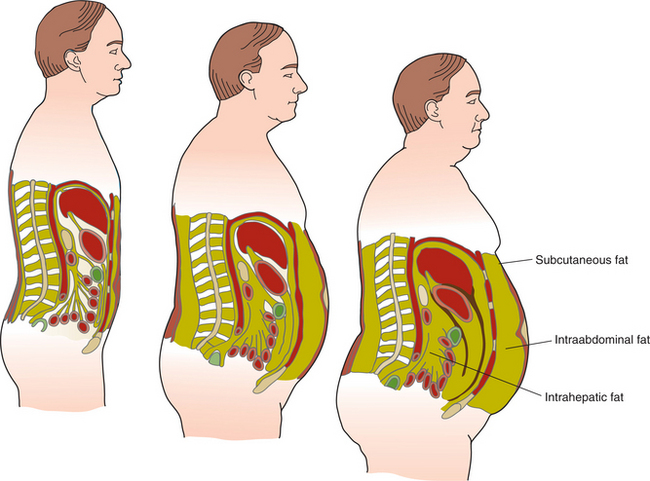
Figure 11-8 Abdominal adipose tissue (fat) can accumulate as subcutaneous, intraabdominal, or intrahepatic (fatty lobules throughout the liver). The body has an almost unlimited capacity to store fat. Central obesity has been linked with serious health consequences (e.g., cardiovascular disease, insulin resistance, diabetes mellitus).
Adiponectin increases insulin sensitivity in muscle and liver and increases free fatty acid oxidation in muscle tissues along with other cell types.93 As the level of adiponectin decreases, the risk for insulin resistance increases. Conversely, if insulin-resistant mice are given extra adiponectin, their glucose tolerance improves and the insulin resistance is reduced.269
Several genetic mutations, including human polymorphisms, of the adiponectin gene are associated with reduced plasma adiponectin concentrations and an increased risk of insulin resistance and type 2 diabetes.97,132 In addition, adiponectin inhibits the formation of atherosclerosis in the large vessels such as the coronary arteries.236
Other adipokines involved in inflammation have been linked to insulin resistance, including IL-6, IL-8, IL-10, and TNF-α.253 These factors are increased with metabolic syndrome or prediabetes conditions.
Inflammation
An interesting observation led to the discovery of an additional role for adipose tissue. Obesity is associated with a systemic low-grade inflammation. This observation led to the idea that adipocytes released compounds into the blood to cause the inflammation. Eventually, it was determined that leptin activates many members of the inflammatory pathway, including CD4+/CD8+ T lymphocytes causing proliferation of the cells.146
Leptin can also activate natural killer cells.270 Within the white fat of obese people, macrophages are known to infiltrate and produce local proinflammatory molecules like TNF-α and IL-6.16 With weight loss there is a decrease in the number of macrophages within adipose tissue and a decrease in the local inflammation. Thus leptin works locally in the adipose tissue.
However, it also has an effect on distant tissues. Leptin links the immune and inflammatory processes to the neuroendocrine system. In addition to playing a key role in modulating T cells, it also is important in autoimmune conditions such as autoimmune encephalomyelitis, type 1 diabetes, bowel inflammation, and rheumatoid arthritis.183
In obesity, leptin is not the only adipokine to induce widespread inflammation. White fat is characterized by an increased production and secretion of a wide range of inflammatory molecules including TNF-α and IL-6, which may have local and systemic effects. Visceral adipose tissue is again the site of much of the inflammatory secretion.
Adipocytes from visceral fat produce large amounts of monocyte chemoattractant protein 1, an adipokine directly involved in cellular remodeling of the heart. One study in women showed a strong correlation between visceral adipose tissue and monocyte chemoattractant protein 1, which resulted in inflammation of the heart and cardiac dysfunction as analyzed by echocardiograms.153
In contrast to leptin, adiponectin has an antiinflammatory action. It has been shown to be protective against atherosclerotic events in large vessels.236 In addition, adiponectin levels rise with long-term physical exercise along with a reduction in inflammatory mediators in the blood.180 However, this antiinflammatory role may be tissue specific as recent evidence suggests that high adiponectin levels may be involved in the chronic inflammation associated with arthritis.62
Pancreas (Islets of Langerhans)
The pancreas lies behind the stomach, with the head and neck of the pancreas located in the curve of the duodenum and the body extending horizontally across the posterior abdominal wall (see Figs. 11-1 and 16-1). It has two functions, acting as both an endocrine gland (secreting the hormones insulin and glucagon) and an exocrine gland (producing digestive enzymes). The cells of the pancreas that function in the endocrine capacity are the islets of Langerhans.
The islets of Langerhans have three major functioning cells: (1) the alpha-cells produce glucagon, which increases the blood glucose levels by stimulating the liver and other cells to release stored glucose (glycogenolysis); (2) the beta-cells produce insulin, which lowers blood glucose levels by facilitating the entrance of glucose into the cells for metabolism; and (3) the delta-cells produce somatostatin, which is believed to regulate the release of insulin and glucagon (Table 11-10).111
Table 11-10
Regulation of Glucose Metabolism.
| Gland | Regulating Function |
| Pancreas | |
| Alpha-cells (islets of Langerhans) | Secrete glucagon; increase blood glucose level |
| Beta-cells (islets of Langerhans) | Secrete insulin (glucose-regulating hormone); decrease blood glucose level; release C-peptide |
| Delta-cells (islets of Langerhans) | Secrete somatostatin; regulate the release of insulin and glucagon |
| Gamma-cells (islets of Langerhans) | Secrete pancreatic polypeptide |
| Adrenal Gland | |
| Medulla: epinephrine | Responds to stress; epinephrine stimulates liver and muscle glycogenolysis to increase the blood glucose level |
| Cortex: glucocorticoids | Increase blood glucose levels by promoting the flow of amino acids to the liver, where they are synthesized into glucose |
| Anterior Pituitary | |
| Adrenocorticotropic hormone (ACTH) | Increases blood glucose levels |
| Human growth hormone (HGH) | Limits storage of fat; favors fat catabolism; inhibits carbohydrate catabolism, raising blood glucose levels |
| Thyroid | |
| T3 and T4 | May raise or lower blood glucose levels |
Diabetes Mellitus
Definition and Overview.: DM is a chronic, systemic disorder characterized by hyperglycemia (excess glucose in the blood) and disruption of the metabolism of carbohydrates, fats, and proteins. Insulin, produced in the pancreas, normally maintains a balanced blood glucose level. DM is characterized as a group of metabolic diseases resulting from defects in the secretion of insulin, action of insulin, or both. The chronic hyperglycemia of DM is associated with long-term damage and dysfunction and impairment of tissues and organs, especially the eyes, kidneys, nerves, heart, and blood vessels.
The majority of cases of DM fall into two large categories: type 1 DM and type 2 DM, related to differences in etiology and pathogenesis of the disease. In type 1 DM (previously called insulin-dependent DM [IDDM] or juvenile-onset DM), the cause of hyperglycemia is an absolute deficiency of insulin production and secretion.
Most individuals with type 1 DM can be identified by serologic evidence showing an autoimmune process occurring in the islet cells of the pancreas along with specific genetic markers. People with type 1 DM are prone to ketoacidosis and specific metabolic derangements associated with hyperglycemia; they require exogenous insulin to maintain life.
Type 2 DM (previously called non–insulin-dependent DM [NIDDM] or adult-onset DM) is a much more prevalent form of diabetes, and the cause is a combination of cellular resistance to insulin action and an inadequate compensatory insulin secretory response. Individuals with type 2 DM are not as likely to exhibit the metabolic derangements common to the person with type 1 DM, and type 2 DM usually can be controlled with diet, exercise, and oral hypoglycemic agents. In some cases, however, people with type 2 DM do require insulin replacement.41 A comparison of the primary differences between the two types of diabetes is shown in Table 11-11.
Table 11-11
Differences Between Types of Diabetes Mellitus*
| Features | Type 1 (Ketosis-Prone) | Type 2 (Not Ketosis-Prone) |
| Age at onset | Usually <20 yr | Usually >40 yr |
| Proportion of all cases | <10% | >90% |
| Type of onset | Abrupt (acute or subacute) | Gradual |
| Etiologic factors | Possible viral/autoimmune, resulting in destruction of islet cells | Obesity-associated insulin resistance |
| HLA association | Yes | No |
| Insulin antibodies | Yes | No |
| Bodyweight at onset | Normal or thin, obesity uncommon | Majority are obese (80%) |
| Endogenous insulin production | Decreased (little or none) | Variable (above or below normal) |
| Ketoacidosis | May occur | Rare |
| Treatment | Insulin, diet, and exercise | Diet, oral hypoglycemic agents, exercise, insulin, and weight control |
*This table does not reflect changes in our understanding of a “hybrid” form of diabetes referred to as types 1.5 or maturity onset diabetes of the young (MODY).
Modified from Goodman CC, Snyder TE: Differential diagnosis in physical therapy, ed 3, Philadelphia, 2000, WB Saunders.
In recent years the lines between type 1 and 2 DM have begun to blur. An autoimmune type of diabetes that begins in middle to late adulthood has been identified. In addition, with increased obesity, type 2 DM is being diagnosed in younger and younger children.250 Recently, autoantibodies typical of type 2 diabetes have been identified in obese people with DM, previously labeled with type 2 DM. The exact classification of these “hybrid” types of diabetes is still being sorted out. Some have used the term type 1.5, or maturity-onset diabetes of the young (MODY). It is important to understand that the characteristics of type 1 and 2 diabetes are not mutually exclusive and should be considered along a spectrum of attributes of the disease.
Prediabetes.: Prediabetes occurs when the body cannot utilize glucose the way it should. After ingesting food, carbohydrates are converted into glucose. The pancreas releases insulin to help move the glucose into the cells to be used for energy. In someone with prediabetes, this process is not completed because either the body cells do not recognize all of the insulin (decreased insulin sensitivity) or the cells stop responding to the action of insulin (increased insulin resistance).
With less glucose moving into the cells, the blood glucose levels start to rise. This is the beginning of a condition referred to as prediabetes. In prediabetes, the blood glucose levels are higher than normal but not quite high enough to be considered diabetes (Table 11-12). Many people with prediabetes have hypertension and dyslipidemia. The trio of comorbidities increases the risk of developing type 2 diabetes and heart disease.
Table 11-12
| Fasting Plasma Glucose Test | 2-Hour Oral Glucose Tolerance Test |
| Normal: <100 mg/dl | Normal: <139 mg/dl |
| Prediabetes: 100-125 mg/dl | Prediabetes: 140-199 mg/dl |
| Diabetes: >125 mg/dl | Diabetes: >200 mg/dl |
Data from the American Diabetes Association (www.diabetes.org), 2007.
According to the American Diabetes Association,41 two categories of hyperglycemia classifications fall between normal and a true diagnosis of diabetes. Impaired glucose tolerance (IGT) and impaired fasting glucose (IFG) refer to intermediate metabolic stages that fall between normal glucose metabolism and diabetes. Both IGT and IFG have been recently called “prediabetes.”41
Individuals with IGT often manifest hyperglycemia only when challenged with the oral glucose load used in the oral glucose tolerance test (OGTT). IFG is diagnosed in people whose fasting plasma glucose levels are equal to or greater than 100 mg/dl but less than or equal to 125 mg/dl. (Normal plasma glucose as defined by the American Dietetic Association (ADA) is less than 100 mg/dl.)41
The terms prediabetes and insulin resistance syndrome are sometimes used interchangeably, and sometimes the terms insulin resistance syndrome and metabolic syndrome are used to describe the same condition. There are slight differences in all three terms with some overlap. For example, since prediabetes represents both a state of decreased insulin sensitivity and increased insulin resistance, it is not strictly the same thing as just insulin resistance syndrome. However, prediabetes is a term often used to refer to the condition called insulin resistance syndrome, and although prediabetes can develop without metabolic syndrome, many people with prediabetes also have metabolic syndrome. Metabolic syndrome includes central obesity, insulin resistance (the “prediabetes component”), and dyslipidemia.
Other Types and Categories of Diabetes Mellitus.: In addition to the main categories of type 1 and type 2 DM, other rare specific types of DM exist, which are associated with a variety of etiologies, including the following:
• Genetic defects in insulin action
• Disorders of the exocrine pancreas such as injury, neoplasm, cystic fibrosis, or infection
• Other endocrinopathies that antagonize insulin secretion or action (e.g., increased growth hormone, cortisol, glucagons, or epinephrine)
• Drug or chemically induced DM (e.g., glucocorticoids, thiazides, thyroid hormone)
• Uncommon forms of immune or genetically associated syndromes (e.g., stiff-man syndrome, Down syndrome, Klinefelter’s syndrome, Turner’s syndrome, and Wolfram syndrome)
• Infections (certain viruses, including rubella, coxsackievirus B, cytomegalovirus, adenovirus, and mumps, have been associated with beta-cell destruction in people with existing genetic markers)
The final category, gestational DM, is defined as any degree of glucose intolerance recognized with the onset of pregnancy. Approximately 6 weeks or more after pregnancy ends, the woman should be reclassified into one of the other categories, depending on whether or not her glucose tolerance resolves. She could be reclassified as normal if no glucose intolerance remains after the pregnancy is completed. Gestational DM accompanies approximately 4% of all pregnancies.
Most women who have gestational DM return to normal glucose metabolism after pregnancy.41 However, with time, these women will likely be diagnosed with type 2 DM.238 In addition, children born to women with gestational DM generally have delayed fine and gross motor skills and a higher prevalence of inattention or hyperactivity compared to children born of women without gestational DM.181
Incidence and Prevalence.: According to the American Diabetes Association, more than 14.6 million Americans have been diagnosed with diabetes, and approximately 6.2 million more people have undiagnosed cases. In addition to the 20.8 million Americans with diabetes (7% of the entire U.S. population), 41 million have prediabetes.
Diabetes, with its severe complications of heart disease, stroke, kidney disease, blindness, and loss of limbs, is the most common endocrine disorder, ranked as a leading cause of death from disease in the United States (mostly because of increased rates of CAD). It is the leading cause of blindness and renal failure in adults.74,169
Black, Native, Hispanic, Mexican, and Asian Americans are 1.5 to 2 times more likely to develop DM than are white Americans, with increasing incidence associated with advancing age. Nearly one-half of all Americans with DM are older than 60 years and nearly one-fourth of the U.S. population over age 65 have diabetes46; males and females are affected equally.
Approximately 90% of all cases of DM are type 2. Type 1 DM and secondary causes (e.g., medications, genetic disease, or hormonal changes) account for the remaining 10%. Since the mid1970s, the incidence of diabetes has steadily increased as a result of prolonged life expectancy; increased incidence of obesity; and reduced mortality resulting in increased live births to people with type 1 DM, whose children are predisposed to future development of type 1 DM.
Etiologic and Risk Factors.: Risk factors for type 1 and type 2 DM have been identified (Box 11-5). Additionally, lifestyle factors, such as watching 2 or more hours of television daily, skipping breakfast, drinking a daily carbonated beverage, and having a waist measurement larger than 35 inches for women and 40 inches (a sign of abdominal fat) for men, may be linked with type 2 diabetes.
More television (sedentary lifestyle) is often linked with less activity, which can lead to weight gain. Eating nonnutritious snacks while watching television and/or drinking soda pop adds extra empty calories, which can also result in weight gain. Eating fast food more than twice a week raises the risk of obesity and the likelihood of becoming resistant to insulin.
High stress can interfere with the body’s ability to make insulin and process glucose; cortisol is a key factor in glucose metabolism and stress is linked with elevated cortisol levels. Stress can also interrupt sleep, and sleep disturbances may be linked with an increased risk of developing insulin resistance. Other lifestyle and risk factors under investigation for diabetes include consuming processed meat (e.g., bacon, hot dogs, and lunch meat) and major depressive disorders.
Type 1 Diabetes Mellitus.: Type 1 DM results from cell-mediated autoimmune destruction of the beta-cells of the pancreas and is a condition of absolute insulin deficiency. This autoimmune process is detectable because markers of cellular destruction called autoantibodies are specific to pancreatic beta-cells. One and sometimes more of these autoantibodies are present and detectable in 85% to 90% of individuals with type 1 DM when the disease initially is diagnosed. People with autoimmune destruction of beta-cells are also prone to other autoimmune disorders such as Graves’ disease, Hashimoto’s thyroiditis, Addison’s disease, vitiligo, autoimmune gastritis, and pernicious anemia.137
Certain human leukocyte antigens (HLA-DR3 and HLA-DR4) on specific chromosomes appear to predispose persons to the development of type 1 DM. For example, there is a fourfold increased prevalence of Down syndrome among people with type 1 diabetes, supporting the theory that genes on chromosome 21 may confer risk for type 1 diabetes.17
In type 1 DM, the rate of beta-cell destruction is rapid in some people (mainly infants and children) and slow in others (mainly adults). Even though immune-mediated diabetes commonly occurs in childhood and adolescence, it can occur at any age, even late in life. Both genetic and environmental factors are associated with autoimmune destruction of the beta cell, although the environmental relationship is still poorly defined.41
In about 10% of cases of type 1 DM, no definable etiology exists. Some individuals in this category (usually of African or Asian origin) have permanent hypoinsulinemia and are prone to ketoacidosis but have no evidence of autoimmunity. Their need for insulin replacement is usually inconsistent. Up to 20% of women with type 1 DM have some kind of eating disorder that predisposes them to further complications with glucose control. Binge eating and use of intense, excessive exercise are common in preteen, teenage, and young women with type 1 DM.44,88 The emphasis on weight control, dietary habits, and food at a time when poor self-esteem, stress, and altered image occur in young women with type 1 DM may contribute to an increased risk for eating disorders.155
Individuals with type 1 DM and eating disorders are more likely to practice insulin omission and reduction, symptoms unique to DM and that increase the risk of DKA and microvascular complications, such as retinopathy, in this population group.88
Type 2 Diabetes Mellitus.: Type 2 DM is a form of diabetes in which individuals have endogenous insulin production but have difficulty with effective insulin action at the cellular level. People with type 2 DM may not need insulin treatment to survive but need other forms of therapy to prevent hyperglycemia and its resulting complications. Although the specific etiologies of type 2 DM are not clear, autoimmune destruction of beta-cells does not occur.
Type 2 DM is associated with obesity. In fact, obesity-dependent diabetes in childhood is now referred to as diabesity and is considered an inflammatory metabolic condition. Both insulin resistance and defective insulin secretion appear very prematurely in obese individuals, and both worsen similarly toward diabetes.218
Most people with type 2 DM are obese and sedentary; these two risk factors cause some degree of insulin resistance. At least 80% of all persons with type 2 DM are obese and the remaining 20%, who are not obese by traditional weight criteria, may have an increased percentage of body fat distribution, particularly in the abdominal area.41
Type 2 diabetes was originally called late- or adult-onset diabetes because it primarily occurred in people 60 years old or older. Starting in the early 1990s, a trend toward the development of type 2 DM in children and adolescents was observed. Excess body fat and sedentary lifestyle are the key risk factors contributing to the development of type 2 DM in younger population groups. Recently, type 2 DM susceptibility genes that lead to insulin resistance in humans were identified.154 Cigarette smoking may also be a risk factor for developing type 2 DM. Smokers exhibit a significantly increased incidence of diabetes compared to people who have never smoked.75
People with this form of diabetes may have normal or elevated insulin levels, but the insulin produced is ineffective because the cells are resistant to attachment to their cellular receptors and subsequent action. Insulin secretion also is impaired, and the beta-cells are unable to secrete increased amounts of insulin when needed. Ketoacidosis seldom occurs in this type of diabetes, but people with type 2 DM are at increased risk for developing macrovascular and microvascular complications. The risk of developing this form of diabetes increases with age, obesity, low cardiorespiratory fitness, and lack of exercise.263 Type 2 DM occurs more frequently in women with prior gestational DM and in individuals with hypertension or dyslipidemia. Its frequency varies in different racial/ethnic groups (see Box 11-3).
Pathogenesis.: Insulin is a hormone secreted by the beta-cells of the pancreas that transports glucose into the cell for use as energy and storage as glycogen: it turns food into energy. It also stimulates protein synthesis and free fatty acid storage in the fat deposits. In DM, insulin is either insufficient in amount (type 1) or ineffective in action (type 2).
Insulin deficiency compromises the body tissues’ access to essential nutrients for fuel and storage.230 When glucose levels are elevated normally (e.g., after eating a meal), beta-cells increase secretion of insulin to transport and dispose of the glucose into peripheral tissues, thereby lowering blood glucose levels and reestablishing blood glucose homeostasis. Defects in the pancreas, liver, or skeletal muscle, singularly or collectively, can contribute to abnormal glucose homeostasis.
Normally, after a meal, the blood glucose level rises. The liver takes up a large amount of this glucose for storage or for use by other tissues such as skeletal muscle and fat. When insulin is deficient or its function is impaired, the glucose in the general circulation is not taken up or removed by these tissues; thus it continues to accumulate in the blood. Because new glucose has not been deposited in the liver, the liver produces more glucose and releases it into the general circulation, which increases the already elevated blood glucose level (Fig. 11-9).230
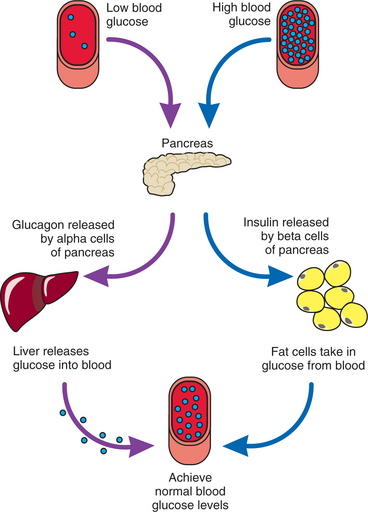
Figure 11-9 Endocrine function of the pancreas. Type 2 diabetes can promote excess sugar release from the liver, render the pancreas incapable of producing sufficient insulin, and dampen the effects of insulin on muscle and fat. Normally after intake of food, the stomach transforms food into glucose, which then enters the blood stream. Rising blood glucose levels signal beta cells in the pancreas to release insulin. The insulin transports glucose into the cell and sets up a cascade of events (e.g., increased rate of glucose utilization and adenosine triphosphate [ATP] generation, conversion of glucose to glycogen, increase in protein and fat synthesis) that eventually results in a decline in blood glucose concentration and restoration of homeostasis. When the blood glucose levels drop (such as occurs in a hypoglycemic state or when fasting), alpha-cells in the pancreas produce glucagon, which increases the blood glucose levels by stimulating the liver and other cells to release stored glucose (a process called glycogenolysis). The blood glucose concentration rises, thus restoring the proper balance and returning the body to a state of homeostasis. Either beta-cell dysfunction or insulin resistance can disrupt this process, resulting in decreased plasma insulin and ultimately hyperglycemia.
When a true deficiency of insulin exists, such as that which occurs in type 1 DM diabetes, the following three major metabolic problems exist: (1) decreased utilization of glucose (as described), (2) increased fat mobilization, and (3) impaired protein utilization. Cells that require insulin for transporting glucose inside the cell, such as in skeletal and cardiac muscle and adipose tissue, are affected most, whereas nerve tissue, erythrocytes, and the cells of the intestines, liver, and kidney tubules, which do not require insulin for glucose transport, are affected the least.
In an attempt to restore balance and normal levels of glucose, the kidney excretes the excess glucose, resulting in glucosuria (sugar in the urine). Glucose excreted in the urine acts as an osmotic diuretic and causes excretion of increased amounts of water. This process results in fluid volume deficit (FVD) (see Chapter 5). The conscious person becomes extremely thirsty and drinks large amounts of water (polydipsia).
Increased fat mobilization occurs because the body can rely on fat stores for energy when glucose is not available. The process of fat metabolism leads to the formation of breakdown products called ketones, which accumulate in the blood and are excreted through the kidneys and lungs. Ketones can be measured in the blood and the urine to indicate the presence of diabetes. Ketones interfere with acid-base balance by producing hydrogen ions. The pH can fall, and the affected person can develop metabolic acidosis (see Chapter 5).
After the renal threshold for ketones is exceeded, the ketones appear in the urine as acetone (ketonuria). When large amounts of glucose and ketones are excreted, osmotic diuresis becomes more severe and fluid and electrolyte loss through the kidneys increases. Sodium, potassium, and other critical electrolytes are lost in the urine, resulting in severe dehydration and electrolyte deficiency and worsening acidosis. When fats are used for a primary source of energy, the body lipid level can rise to five times the normal amount. This elevated level can lead to atherosclerosis and its subsequent cardiovascular complications (see Chapter 12).
Impaired protein utilization occurs because the transport of amino acids (the chief constituent of proteins) into cells requires insulin. Normally, proteins are constantly being broken down and rebuilt. Without insulin to transport amino acids, thereby contributing to protein synthesis, the balance is altered and protein catabolism increases. Catabolism of body proteins and resultant protein loss hamper the inflammatory process and diminish the tissue’s ability to repair itself.
Because the person with type 2 DM continues to produce and use some amount of endogenous insulin, the metabolic problems associated with inappropriate use of fat and protein for energy do not occur as severely. People with type 2 DM are not prone to ketoacidosis and the metabolic derangements associated with type 1 DM. They are, however, still at great risk for hyperglycemic osmotic diuresis, dehydration, shock, and loss of electrolytes.230
Pathophysiology of Diabetic Complications.: The long-term presence of DM affects the large blood vessels (macrovascular), small blood vessels (microvascular), and nerves throughout the body. The chronic hyperglycemia of diabetes results in the accelerated atherosclerosis that leads to macrovascular disease, affecting arteries that supply the heart, brain, and lower extremities. Insulin resistance has also been implicated in the macrovascular changes in diabetes.85
Diabetes is also associated with the development of diabetes-specific microvascular pathology in the retina, renal glomerulus, and peripheral nerve. As a result, diabetes is a leading cause of blindness, kidney failure, and a variety of debilitating neuropathies. The microvascular disease in the retina, glomerulus, and vasa nervorum has similar underlying pathophysiology.
Hyperglycemia causes abnormalities in blood flow and increased vascular permeability (caused by decreased activity of vasodilators, increased activity of vasoconstrictors, and abnormal production of extracellular matrix), and with time, microvascular cell loss and progressive capillary occlusion occur. In addition, hyperglycemia may also decrease production of trophic factors, which are required to maintain healthy endothelial and neuronal cells.
How does hyperglycemia cause these macrovascular and microvascular damages? Four main molecular mechanisms have been proposed: increased polyol pathway flux; increased advanced glycation end-product (AGE) formation; activation of protein kinase C isoforms; and increased hexosamine pathway flux.26 All four mechanisms seem to reflect a single hyperglycemia-induced process of overproduction of superoxide by the mitochondrial electron-transport chain. As an example, neuropathy in diabetes presumably results from the increased polyol pathway flux and is related to the accumulation in the nerve cells of sorbitol, a by-product of improper glucose metabolism. This accumulation then results in abnormal fluid and electrolyte shifts and nerve-cell dysfunction. The combination of this metabolic derangement and the diminished vascular perfusion to nerve tissues contributes to the severe problem of diabetic neuropathy. (See the section on Diabetic Neuropathy in Chapter 39.)
Cardinal Signs and Symptoms.: In type 1 diabetes, symptoms of marked hyperglycemia include polyuria, polydipsia, weight loss with polyphagia, and blurred vision (Table 11-13). These symptoms occur as a result of the inability of the body to use glucose appropriately and the resulting osmotic diuresis, dehydration, and starvation of body tissues. In type 1 DM, the utilization of fats and proteins for energy causes severe hunger, fatigue, and weight loss. The person with this type of diabetes may present initially in DKA.
Table 11-13
Cardinal Signs of Diabetes at Diagnosis
| Clinical Manifestations | Pathophysiologic Bases |
| Polyuria (excessive urination, types 1 and 2) | Water is not reabsorbed from renal tubules because of osmotic activity of glucose in the tubules. |
| Polydipsia (excessive thirst, types 1 and 2) | Polyuria causes dehydration, which causes thirst. |
| Polyphagia (excessive hunger, type 1) | Starvation secondary to tissue breakdown causes hunger. |
| Weight loss (type 1) | Glucose is not available to the cells; body breaks down fat and protein stores for energy; dehydration. |
| Recurrent blurred vision (types 1 and 2) | Chronic exposure of the lenses and retina to hyperosmolar fluids causes blurring of vision. |
| Ketonuria (type 1) | Fatty acids are broken down so ketones are present in urine. |
| Weakness, fatigue, and dizziness (types 1 and 2) | Dehydration leads to postural hypotension; energy deficiency and protein catabolism contribute to fatigue and weakness. |
| Often asymptomatic (type 2) | Physical adaptation often occurs because rise in blood glucose is gradual. |
People with type 2 diabetes also may have some of these cardinal signs and symptoms, but the aging population may not recognize the abnormal thirst or frequent urination as abnormal for their age. The person with type 2 diabetes frequently goes undiagnosed for many years because onset of type 2 DM is often gradual enough that the classic signs of hyperglycemia are not noticed. More commonly, they may experience visual blurring, neuropathic complications (e.g., foot pain), infections, and significant blood lipid abnormalities. Type 2 DM is commonly diagnosed while the client is hospitalized or receiving medical care for another problem. Frequently, the person presents with one of the long-term complications of DM such as CVD, neuropathy, retinopathy, or nephropathy.