Infectious Diseases of the Musculoskeletal System
Although chronic disease accounts for the majority of morbidity and mortality in the United States, infectious diseases of the musculoskeletal system are still common in the general population.23 Fortunately, most of the microorganisms that humans encounter do not produce disease. In fact, staphylococcus is part of the natural flora found in healthy individuals. The interaction between host and organism is complex but is responsible for determining whether an infectious disease ensues.
The method by which a microorganism attaches itself to a host cell is not well understood, but it is the important first step in establishing an infection (see Chapters 6 and 7). Once the methods of adhesion of the microorganism to the host are better understood, new methods of prevention with vaccines that block linkage to the host may offer better control of infectious diseases.
The body has a number of defense systems in place to prevent an infection from becoming established. These are important in understanding musculoskeletal infections. The skin serves as an effective barrier to invasion by microorganisms but can be easily breached by trauma, invasive procedures, or the bites from insects108 and animals.82 Even the best defense systems can fail or be overwhelmed by the shear numbers of invading microorganisms. When bacteria become established in the host, the multiplication of the organism is what triggers the inflammatory response (see Chapter 6).
The presence of the foreign microorganism also initiates the immune response (see Chapter 7). This includes the phagocytic system, which consists of neutrophils, monocytes, and macrophages that attempt to ingest invading microorganisms. This occurs as a general response to any invasion. Lymphocytes and immunoglobulins deliver a more specific response. Depending on the location of the infection, the functioning of the hematologic, cardiopulmonary, gastrointestinal (GI), endocrine, and genitourinary systems may be impaired.
The use of temporary and permanent implants and prosthetic materials, including bioprosthetic implants, is now commonplace. Therapists often treat clients with biomaterial implants. Joint replacements, heart valves, vascular prostheses, artificial disk replacements, shunts, dental implants, baclofen/insulin/pain pumps, sutures, catheters, and allografts are a few of the devices that can harbor infections. Musculoskeletal infections can require drastic measures to treat and prevent morbidity. For example, infection after total joint replacement often requires removal of the hardware, and joint sepsis may require extensive surgical debridement.100
Understanding the epidemiology, pathogenesis, and treatment of musculoskeletal infections will allow the clinician to play an active role in all phases of diagnosis and intervention. From early detection of signs and symptoms that identify those clients who are at risk and need of further medical evaluation to the rehabilitation of those clients who undergo surgical intervention for musculoskeletal infections, the role of therapists and their impact on the outcome of treatment cannot be underestimated.
Only the most common infectious diseases seen in a therapy practice are included in this chapter (e.g., osteomyelitis, diskitis, infectious arthritis, and tuberculosis [TB]). Necrotizing fasciitis, streptococcal myositis, and soft tissue infection leading to gangrene are discussed in Chapter 8; cellulitis is discussed in Chapters 8 and 10.
OSTEOMYELITIS
Overview
Osteomyelitis is an inflammation of bone caused by an infectious organism such as bacteria, but fungi, parasites, and viruses can also cause skeletal infections (see Chapter 8). The pathophysiology of osteomyelitis is complex and poorly understood. Key factors include the virulence of the infecting organism; the individual’s immune status; any underlying disease; and the type, location, and vascularity of the involved bone.
Acute osteomyelitis is the clinical term for a new infection in bone that can develop into a chronic reaction when intervention is delayed or inadequate. It is a rapidly destructive pyogenic infection often seen in children, older adults, and intravenous (IV) drug abusers. The infectious agent enters the body through an open wound or the GI tract. The infection has the capability to spread quickly through the bloodstream, resulting in septicemia or a septic infectious joint.
In adults, osteomyelitis is usually a subacute or chronic infection that develops secondary to an open injury to bone and surrounding soft tissue. Chronic osteomyelitis is often the result of a relapse, persistent bone infection, or acute disease remaining undiagnosed.
Incidence
Acute hematogenous osteomyelitis is a relatively uncommon but potentially serious disease occurring more often in children than adults and affecting boys more often than girls. Acute hematogenous osteomyelitis is the most common type and is usually seen in children. Chronic osteomyelitis is more common in adults and immunocompromised people. With the use of antibiotics the incidence of osteomyelitis was expected to decline. However, with the presence of drug-resistant organisms, the number of IV drug abusers, and the increased use of implantable prosthetic devices, osteomyelitis is actually becoming more common.100
Etiologic Factors
Staphylococcus aureus is the usual causative agent of acute osteomyelitis.94 It has the ability to bind to cartilage, produce a protective glycocalyx, and stimulate the release of endotoxins. Glycocalyx is the glycoprotein and polysaccharide covering that surrounds many cells; in bacterial cells the glycocalyx forms masses of fibers that extend from the bacterial cell and are the means by which the bacteria adhere to surfaces.
Other organisms such as group B streptococcus, pneumococcus, Pseudomonas aeruginosa, Haemophilus influenzae, and Escherichia coli also produce bone infections.29,44,100 Among people with sickle cell anemia, Salmonella infection is associated with osteomyelitis. Osteomyelitis can be acquired from exogenous or hematogenous sources.
Exogenous osteomyelitis is acquired by invasion of the bone by direct extension from the outside as a result of inoculation into the bone by a penetrating or puncture wound, extension from an overlying abscess or burn, or other trauma such as an open fracture.8,18 Surgical procedures, open fractures, and implanted orthopedic devices are common sources of acute osteomyelitis infection. These examples of osteomyelitis secondary to a contiguous area of infection are common in immunocompromised people and in those with diabetes or severe vascular insufficiency.11
Hematogenous osteomyelitis is acquired from spread of organisms from preexisting infections such as occurs in impetigo; furunculosis (persistent boils); infected lesions of varicella (chickenpox); and sinus, ear, dental, soft tissue, respiratory (through alveoli when an upper respiratory infection is present), and genitourinary infections. Vaginal, uterine, ovarian, bladder, and intestinal infections can lead to iliac or sacral osteomyelitis.80
Osteomyelitis of the arm and hand bones may occur in drug abusers, and vertebral osteomyelitis is seen in adults from hematogenous spread from pelvic or urinary tract infections. The lumbar spine is the most commonly involved area. In children the infection is spread hematogenously and usually develops in the metaphyseal regions of the long bones, adjacent to the growth plates (e.g., distal femur, proximal tibia, humerus, and radius).100
Risk Factors
In general, anyone who is chronically ill (e.g., diabetes or alcoholism) or who receives large doses of steroids or immunosuppressive drugs is particularly susceptible to osteomyelitis (Box 25-1). Older adults with additional preexisting medical conditions, such as malignancy, malnutrition, and renal or hepatic failure, are also at a greater risk.66 Spinal cord injury with complete motor and sensory paralysis at the paraplegic level appears to be a risk factor for vertebral osteomyelitis.41
Nutritional status is an often overlooked variable in acute and chronic illness. Anyone who is at risk for malnutrition or is compromised by a poor nutritional status is at risk for infection, slowed tissue healing, and an increased incidence of postsurgical complications.93
Infection is the second most common cause of prosthetic joint failure, after mechanical loosening. Some cases of prosthetic joint infection may be misclassified as aseptic loosening. The incidences of prosthetic joint infection is higher after a revision.102
Pathogenesis
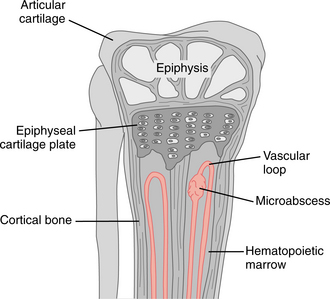
The pathophysiology of osteomyelitis is complex and poorly understood. Key factors include the virulence of the infecting organism; the person’s immune status; any underlying disease; and the type, location, and vascularity of the involved bone. Regardless of the source of the pathogen, the pathogenesis of bone infection initially involves an inflammatory response. Acute osteomyelitis may develop in the metaphysis of long bones because of the decreased amount of phagocytosis and/or slower rate of blood flow in the terminal arterioles. The vascular loop in growing bone is a common site of bacterial seeding because the arterioles form a loop and then drain into the medullary cavity without establishing a capillary bed (Fig. 25-1). Trauma, including microtrauma, may also increase susceptibility to infection by slowing the blood flow.
The metaphysis of long bones is very porous, allowing exudate from the infection to spread easily. As the organ- isms grow and form pus within the bone, tension builds within the rigid medullary cavity, forcing pus through the haversian canals. Haversian canals are anastomosing channels that constitute the basic unit of structure in compact bone. These canals contain blood, lymph vessels, and nerves.
Once bacteria gain access to these channels, they are able to proliferate unimpeded, forming a subperiosteal abscess that deprives the bone of its blood supply and eventually may cause necrosis. Necrotic cells then become a fertile bed for the organisms to multiply. Because sensory nerve endings are absent in cancellous bone, this process can progress without pain. Necrosis then stimulates the periosteum (osteoblasts) to create new bone.
The sheath of new bone, called an involucrum, forms around the sequestrum, necrotic tissue that has become separated, which works its way out through an abscess or the sinuses (Fig. 25-2). By the time the sequestrum forms, the osteomyelitis is chronic. In adults, this complication is rare because the periosteum is firmly attached to the cortex and resists displacement. Instead, infection disrupts and weakens the cortex, which predisposes the bone to pathologic fracture.
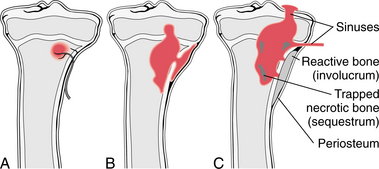
Figure 25-2 Osteomyelitis. A, Initial infection. The bacteria reach the metaphysis through the nutrient artery. B, First stage: bacterial growth results in bone destruction and formation of an abscess. C, Second stage: from the abscess cavity, the pus spreads between the trabeculae into the medulla, through the cartilage into the joint, or through the haversian canals of the compact bones to the outside. These sinuses traversing the bone persist for a long time and heal slowly. The pus destroys the bone and sequesters part of it in the abscess cavity. Reactive new bone is formed around the focus of inflammation. (From Damjanov I: Pathology for the health-related professions, ed 3, Philadelphia, 2006, WB Saunders.)
In vertebral osteomyelitis the infection is first found in the metaphysis or cartilaginous endplates and quickly spreads to the intervertebral disk. Abscess formation is common through direct extension of the infection to adjacent tissues. The abscesses can advance posteriorly to involve the epidural area or anteriorly to produce a paravertebral abscess that can extend to the psoas muscle, producing hip pain.
Clinical Manifestations
The primary manifestations of osteomyelitis vary between adults and children. Back pain is typically the chief complaint in adults, but once the infection becomes systemic (as opposed to an abscess) a low-grade fever may be present. Children are more likely to present with acute, severe complaints, such as high fever and intense pain, but in some cases, local manifestations will predominate such as edema, erythema, and tenderness. These signs are easier to detect in the extremities, unlike vertebral osteomyelitis where the infected structures lie much deeper.
In the initial phases of the infection, pain may not be a factor because of the lack of pain fibers in cancellous bone. This makes diagnosis and intervention difficult because of the potential rapid spread of the infectious agent, which is facilitated by the delay in the administration of appropriate antibiotics. When the infection extends into the periosteum, increased joint pain; diminished function; and systemic signs, such as fever, swelling, and malaise; may rapidly develop.
The pain will likely be described as deep and constant, increasing with weight bearing when the infection is anywhere in the lower extremity. Clients presenting with chronic osteomyelitis complain of local pain and swelling but otherwise are often asymptomatic. The clinical sign of “sausage toe” has been used to detect underlying pedal osteomyelitis. This clinical sign has been demonstrated to have good sensitivity and specificity in clients with diabetes.85
Once present, spinal osteomyelitis can produce intermittent or constant back pain. The pain can be aggravated by motion but is also present regardless of activity level in some individuals and throbbing at rest. It may radiate in a radicular distribution and is commonly accompanied by spinal tenderness and rigidity; accessory motions of the spine are often difficult to perform. As mentioned, pyogenic vertebral osteomyelitis may result in a psoas abscess causing painful hip extension and/or an antalgic limp. Cervical abscess formation may lead to torticollis or dysphagia.101
Radiculopathy, myelopathy, or even complete paralysis can occur with neural compression as a result of abscess, instability, or spinal deformity associated with vertebral osteomyelitis. Direct spread of the infection into the epidural space can cause meningitis.101
Sacroiliac osteomyelitis is usually characterized by severe, local pain with tenderness and an antalgic gait. The pain may radiate to the buttocks or abdomen. The history will be a recent onset of localized pain. Besides pain, other symptoms may include fever, local tenderness, and swelling. Any unexplained cellulitis should be considered a sign of osteomyelitis in children, even if no other contributing signs or symptoms are evident.100
MEDICAL MANAGEMENT
Since chronic osteomyelitis is also recognized as a complication of treatment of open fractures, prevention of infection is important. The risks can be minimized if the wound is thoroughly debrided, irrigated, and left open for delayed primary closure. Delayed primary closure allows the wound bed to be inspected, and further debridement can be carried out if necessary.
Clients with any of the conditions listed in Box 25-1 or with any additional risk factors should be taught proper preventive measures and be aware of early warning signs. As mentioned, nutritional status plays a critical role in the prevention of infection and the body’s ability to combat infection once it occurs. Anyone with biomaterial implants will have an increased risk of infection, especially in the immediate postoperative period. Although rare, late infections occurring 1 year postoperatively have been reported.50
SCREENING.
Although diagnosing infectious diseases is outside the scope of the therapist’s practice, screening is still appropriate, using a thorough history and a review of systems as presented in the Guide to Physical Therapist Practice2 to help identify pathologies that require further medical evaluation.
Signs and symptoms of systemic disease can sometimes be easily detected. For screening purposes, the presence of a fever, unexplained weight loss, history of cancer, and failure to respond to adequate treatment are good indicators of more serious pathology.34,48 Sometimes the history alone is quite informative.
Disturbances in the sleep pattern, such as awakening with pain, requiring sleep medications, or inability to fall asleep, along with symptoms that increase with walking, have been found to be associated with serious back problems.86 In other areas of the body, localized pain in the presence of other risk factors may raise suspicion.
DIAGNOSIS.
The diagnosis of infection in total joint revisions is challenging. An aspirate with a white blood cell (WBC) count over 50,000 is considered infected without an implant. A total knee replacement (TKR) is considered infected with 2500 WBC/ml.68 Medical diagnosis is often delayed because of the lack of specific signs and symptoms, especially in chronic osteomyelitis. Signs and symptoms that are generally associated with infection may be masked by (or mistaken for) normal postoperative changes.
Diagnosis and antimicrobial and surgical treatment of prosthetic joint infection are complex, and the recovery can be arduous and prolonged.102 A careful history and a thorough physical examination are important. Laboratory values and radiographs are often negative in the early stages. Positive cultures are obtained in only 50% to 80% of cases; however, this is improving because of advancements in molecular techniques.20,94,102 Any unexplained cellulitis in children is suggested to be considered as a possible indicator of underlying osteomyelitis.100
Radiographs may not detect bony abnormality in infections of less than 10 days’ duration. Lytic lesions may be demonstrable on radiographs within 2 weeks of onset of the disease. A periosteal reaction develops later (Fig. 25-3). Magnetic resonance imaging (MRI) and isotope bone scans are the procedures of choice in delineating the disease’s anatomic extent.100 Imaging tests are often used to localize or confirm the presence of infection. MRI is sensitive and provides valuable detail of septic arthritis, spinal osteomyelitis, and diabetic foot infections.77 Radionuclide bone scan can detect early stage disease and is helpful in detection and identifying multiple sites of involvement.17 This procedure may be used as an alternative when MRI cannot be performed or as an adjunct diagnostic tool in people with an uncertain diagnosis.
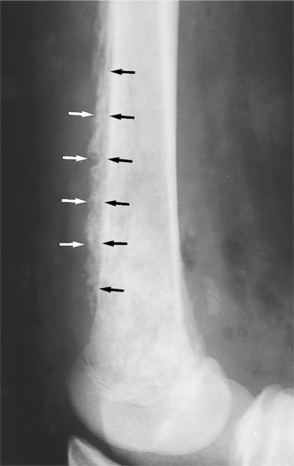
Figure 25-3 Chronic osteomyelitis. A lateral view of the knee shows the periosteal reaction (arrows) suggestive of chronic osteomyelitis. The bone of the distal femur has a mottled appearance as a result of the infection. (From Mettler FA: Essentials of radiology, ed 2, Philadelphia, 2005, W.B. Saunders.)
Newer nuclear medicine imaging techniques, such as fluorine-18-fluorodeoxyglucose positron emission tomography (FDG-PET) scans, provide accurate localization of infection and/or source of fever of undetermined origin (FUO), thereby guiding additional testing. FDG-PET helps diagnose spinal osteomyelitis and can be useful in measuring the extent of disease and monitoring response to treatment.64,76,106
Identification of the infectious pathogen is of utmost importance because the type of medication used often depends on the infecting microorganism. Specimens are obtained for culture or stains by aspiration, needle biopsy, or swab. Accurate identification is often difficult due to the technical problems of obtaining an acceptable sample. Image-enhanced needle biopsy can improve the specimen quality.
Early acute postoperative prosthetic joint infection in most cases occurs in the first 3 months after surgery. Chronic, postoperative prosthetic joint infection is characterized by more subtle signs of inflammation, chronic persistent postoperative pain, and/or early loosening of the implant. Differentiating late postoperative prosthetic joint infection from aseptic implant failure can be challenging.102
TREATMENT.
Immediate treatment is called for, especially in acute osteomyelitis. The successful use of sequential IV and high-dose antibiotic therapy is now an accepted modality and has lessened the role of surgery in these infections. The choice of antibiotics is based on the culture results. Other factors to consider in choosing an antibiotic are the client’s age and health status, site of infection, and previous antimicrobial therapy.
Antibiotics are delivered intravenously to hasten their effect when faced with serious infection that can progress rapidly. Evaluation of response to treatment by monitoring C-reactive protein levels has decreased the average duration of therapy to 3 to 4 weeks with few relapses.58,94
Surgery is indicated if the infection has spread to the joints. In fact, this is considered an orthopedic emergency. Articular cartilage can be damaged in a matter of hours. The goal of surgery is to drain exudate or pus from the bone or joint. Often, extensive debridement of both bone and surrounding soft tissue is required. Various reconstructive procedures may be considered once the infection is eliminated. These include soft tissue procedures to provide well-vascularized soft tissue coverage of defects and revascularized bone for stabilization of the affected area. In adults, both surgery and antibiotics are often required.
The goals for treatment of chronic osteomyelitis are to eliminate the infection by use of antibiotics if possible or by surgically removing infected tissue. If surgery is indicated, then the current trend is toward more radical surgery rather than serial debridements. In general, chronic osteomyelitis is more difficult to treat than the acute type because it is difficult to eradicate completely. Exacerbations may respond well to treatment with rest and antibiotics, only to flare-up again months later.
In the spine, surgery may be necessary to treat the infection and to address spinal deformity. Deformity of the spine from the infection or subsequent surgery may lead to pain or neurologic compromise. If surgery is not indicated, the person may be treated with short-term bed rest or the use of a brace for immobilization.
The use of appropriate antibiotics prophylactically is standard for some procedures, such as total joint replacements, and in open wounds that are contaminated. Antibiotic bead chains can be implanted in the infected area to achieve concentrated levels of antibiotics in the local tissue without raising serum levels to toxic ranges. These bead chains can be an effective method of prophylaxis and treatment of established infections.59,96
PROGNOSIS.
Fifty years ago, acute osteomyelitis carried a mortality risk of 20%. The risk of death in the majority of the population is now negligible, but treatment remains a challenge to the orthopedic surgeon, radiologist, infectious disease specialist, and therapist.100 With early medical interventions, an infection arrest rate of 90% can be expected, even in chronic cases.79
If the process is unattended for a week to 10 days, some permanent loss of bone structure almost always occurs, as does the possibility of growth abnormality in the pediatric population. When osteomyelitis is diagnosed in the early clinical stages and treated with antibiotics, the prognosis is excellent. However, it is more often the case that the infection remains undetected for months, during which time it establishes itself in the affected bone and surrounding soft tissues.
When osteomyelitis persists for a long period of time, infected necrotic bone serves as an isolated reservoir for infection that will not respond well to systemic antibiotics. Reduced blood flow will facilitate the likelihood that an infection will be established and the pharmacologic agent will be prevented from reaching the locus of infection. The emergence of antibiotic resistance, particularly resistance to methicillin and vancomycin by S. aureus organisms, contributes to long-term sequelae and morbidity.94
Chronic osteomyelitis has a poor prognosis, even when treated surgically. People with chronic osteomyelitis are often in great pain, require prolonged medical care, and may, although rarely, require amputation of an extremity. In the older adult population, the reoccurrence of chronic osteomyelitis ranges from 3% to 40% and the mortality rate is reported as being less than 5%.66 The bottom line is that the best way to minimize the mortality and morbidity observed in osteomyelitis is to practice preventive measures and reduce the time between the onset of the infection and the initiation of an appropriate intervention.
INFECTIONS OF PROSTHESES AND IMPLANTS
Overview
Over the past decades, joint replacement surgery has become commonplace, which is largely attributed to the success of these procedures in restoring function to people with disabling arthritis. People receiving total joint replacements number in the hundreds of thousands each year worldwide, and virtually millions of people have indwelling prosthetic joints. Likewise, during the past 30 years, many new instrumentation systems for internal fixation of the spine have been developed.14
Implant infection remains the primary cause of prosthetic failure, occurring either acutely within the first month postoperatively or months to years after the joint replacement.6 Nearly 80% of these infections are caused by staphylococcus organisms, which enter by perioperative, hematogenous, or contiguous means. Perioperative infections occur around the time of surgery and are probably caused by contaminated hands at the surgical site. Hematogenous infections occur as a result of a primary infection somewhere else in the body. Contiguous infections occur secondary to a nearby infection.105
Other types of prostheses or implants susceptible to infection include breast implants, penile implants, dental implants, cardiac implants, other orthopedic devices and hardware, shunts, and even contact lenses (external to epithelial surfaces that can give rise to serious lifethreatening infections).
Incidence
The successful development of synthetic materials and the introduction of artificial devices into nearly all body systems have been accompanied by the adaptation of microorganisms to the opportunities these devices provide for eluding defenses and invading the host. With improvements in surgical procedures and prophylactic antibiotics, the incidence of infection has been reduced to less than 1.5%. The incidence of infection does increase with longer procedures and revisions.78 However, as the number of people undergoing replacements has grown, reoperations have become increasingly common. Bioprostheses, implanted in large numbers in the 1970s and early 1980s, have now gone into the second decade of life since implantation, a time when biodegradation becomes more common. Multiple reoperations carry a higher risk of infection. Likewise, as the population ages, an increasing number of total hip, knee, shoulder, elbow, wrist, and finger arthroplasties are coming up for revision.
Infection of a prosthetic joint causes loosening of the prosthesis and sepsis with significant mortality and morbidity. Two-thirds of prosthetic joint infections occur within 1 year of surgery and are to the result of intraoperative inoculations of bacteria into the new joint or postoperative bacteremias. Early infections have been substantially reduced by preoperative use of antibiotics, the use of laminar flow in operating rooms, and improved surgical technique.78
Risk Factors
Certain groups have been identified as predisposed toward infection of their prosthetic joints, including those with prior surgery at the site of the prosthesis, rheumatoid arthritis, corticosteroid therapy, diabetes mellitus, poor nutritional status, low albumin, obesity, and extremely advanced age.14
Any factor that delays or impairs wound healing increases the risk of infection. Psoriasis, steroids, diabetes mellitus, and immunodeficiency increase the risk of prosthetic infection. Immunodeficiency can be either local (e.g., wear debris from the implant) or systemic (e.g., Candida albicans, rheumatoid arthritis, or immunosuppressive medications). Risk factors for infection of spinal instrumentation may include IV drug use, paraplegia with neurogenic bladder, and pyelonephritis secondary to renal calculi.
Certain factors or events can enhance the ability of bacteria to multiply rapidly and increase the risk of infection (e.g., wound hematomas, seromas, hemarthroses, fresh operative wounds, ischemic wounds, and tissues in diabetic and steroid-treated people). In the early postimplantation period the fascial layers have not yet healed, and the deep, periprosthetic tissue is not protected by the usual physical barriers. Any superficial infection that develops, such as an infected wound hematoma, wound infection, or suture abscess, can become a preceding event for joint prosthesis infection.
Joint replacement has been shown to be responsible for reactivation of quiescent infections that occurred earlier in a person’s life. Evaluation of a client for history of osteomyelitis, tuberculosis, human immunodeficiency virus (HIV), or other infection caused by bacteria with latent capabilities is important before joint replacement surgery.33
Etiologic Factors and Pathogenesis
The four major pathologic processes that may arise in response to any biomedical implant are infection, inflammation, thrombosis, and neoplasia. Only infection is discussed in this section. Prosthetic joints and other implants become infected by two different pathogenic routes: the hematogenous route and the locally introduced route. Any bacterium can induce infection of a total joint replacement by the hematogenous route, which accounts for 30% of prosthetic joint infections. Hematogenous spread may occur from dental, skin, respiratory tract, and urinary tract infections or procedures.
Locally introduced forms of infection account for about 70% of all prosthetic joint infections and occur as a result of wound infection next to the prosthesis, or to operative contamination. Operative contamination may be a result of direct implantation at the time of the operation by the operating team, from environmental sources, or from contaminated implant materials. Holes in the implants (e.g., press fit acetabular cups) are potential pathways through which debris can gain access to the implant-bone interface, resulting in infection, creating periprosthetic bone loss, and potentially initiating loosening.74 Generally, these infections are caused by a single pathogen, but polymicrobial sepsis can occur.
Staphylococci (coagulase-negative staphylococci and S. aureus) are the principal causative agents of joint prosthesis infection (approximately 40% and 20%, respectively), aerobic streptococci and gram-negative bacilli are each responsible for 10% and 5% of cases, mixed flora constitutes 10%, and anaerobes are involved in about 5% of these infections. No organism is found in about 10% of cases.110 Rarely, latent foci of chronic, nonactive osteomyelitis are reactivated by the disruption of tissue associated with prosthetic surgery. Previous S. aureus and Mycobacterium tuberculosis infections can recur postoperatively.14
Bacteria adhere tightly to the implant surface by adhesins, which recognize specific host proteins absorbed on the material surface. They also use biofilms, a type of slime that protects bacterial colonies from destruction by phagocytes. Multiple layers of biofilm allow staphylococci to cling to the surface of the implant and avoid both immune system defenses and antibiotic diffusion. The slimes can even alter the host’s immune capability and increase antibiotic resistance. Bacteria can lie dormant on the prosthetic device for years until the person becomes immunocompromised from aging or other health issues.6
Prostheses that are cemented into place with polymethyl methacrylate can develop infection at the bone-cement interface, whereas cementless prostheses are more likely to develop sepsis in the bone contiguous with the metallic alloy. As foreign bodies, these prostheses facilitate local sepsis by decreasing the quantity of bacteria necessary to establish infection and by permitting pathogens to persist on their avascular surface, sequestered from circulating immunologic defenses (leukocytes, antibodies, and complement) and from systemic antibiotics.14
In the presence of prosthetic or implantable devices, many bacteria form a fibrous material called glycocalyx. Organisms can reproduce within this matrix and form a thick biofilm that is protected in part from host defense mechanisms. Biofilms are an important issue for surgery involving prosthetic or implantable devices. For example, only 32% of infections caused by slime-producing staphylococci resolved with antibiotics compared with 100% recovery in non–slime-producing strains.
The implant and its adherent biofilm must be removed for the infection to resolve since recurrent, acute infections or disseminated, persistent infection may develop if these reservoirs are allowed to continue. Meticulous protocols for sterilization of implants should be followed, since biofilms may be an important mechanism of antibiotic resistance.81
Clinical Manifestations
Persistent joint pain may be the only symptom with no clinical signs of infection at all. Staphylococcus infections are usually characterized by symptoms similar to wound infection such as edema, hematoma, fever, and local pain. Late-onset or delayed infection usually present as increasing joint pain followed by rapid onset of systemic symptoms.104
Prosthetic joint infections can be divided into the following three categories: early (infection that develops less than 3 months after surgery), delayed (3 to 24 months after surgery), and late (more than 24 months).88
Early infections typically present with acute symptoms such as fever, joint pain, warmth, and redness. These individuals may form a sinus tract from the prosthesis to the skin with purulent drainage. Virulent organisms, such as S. aureus or gram-negative bacilli, are usually responsible for early infections.
People who present with delayed infections often lack systemic symptoms, making diagnosis difficult. These individuals display joint pain and/or joint loosening. Organisms responsible for these infections are less virulent such as coagulase-negative staphylococci (particularly S. epidermidis) and Propionibacterium acnes. Early and delayed infections are typically acquired at the time of surgery, whereas late infections develop from hematogenous seeding. In one study of 63 infected hip prosthesis, 29% were early infections, 41% delayed, and 30% were late infections.45
When a bloodborne infection arises in a prosthetic joint several months or years after implantation surgery, the fully healed connective tissue often is capable of restricting the septic process to a relatively small focus at the bone-cement interface. Joint pain is the principal symptom of deep tissue infection, irrespective of mode of presentation and suggests either acute inflammation of the periarticular tissue or loosening of the prosthesis caused by subacute erosion of bone at the bone-cement interface.
MEDICAL MANAGEMENT
Sterilization and attention to infection control guidelines reduces the risk of infection.6 Proper handwashing remains the key to reducing the transmission of pathogens to others and the spread of antimicrobial resistance. Despite this well-known fact, adherence by surgeons and other important health care workers remains low.103 Researchers are working to design self-sterilizing materials and more effective infection-resistant materials. New coatings with an antiadhesive or biocide capabilities are the next step.
DIAGNOSIS.
Clinical manifestations of joint pain, swelling, erythema, and warmth all reflect an underlying inflammatory process in the surrounding tissues but are not specific for infection. When a painful prosthesis is accompanied by fever or purulent drainage from overlying cutaneous sinuses, infection is likely. The physician must differentiate infection from aseptic and mechanical problems (e.g., hemarthrosis, mechanical loosening, or dislocation). Constant joint pain is suggestive of infection, whereas mechanical loosening commonly causes pain only with motion or weight bearing.
The diagnosis of joint prosthesis infection is dependent on isolation of the pathogen by aspiration of joint fluid or by obtaining tissue at arthrotomy. Gram stain and culture will typically identify the responsible organism in 65% to 94% of cases.110 Special media may be required if fungus or atypical organisms are suspected. Culture of sinus tract drainage or overlying skin should be avoided. Elevated serum leukocyte count, erythrocyte sedimentation rate (ESR), and C-reactive protein level are suggestive but not diagnostic of joint infection. Ultrasound-guided (ultrasonography) aspiration in suspected sepsis of arthroplasty has been developed to facilitate this process.
Radiologic abnormalities may be helpful when changes are noted over serial radiographs, but some changes can lag behind symptoms 3 to 6 months. When both the distal and proximal components of a prosthetic joint demonstrate pathology on radiography, infection is more likely than simple mechanical loosening. However, such radiographic changes are not specific for infection and may also be seen with aseptic processes.
Typical radioisotope scans demonstrate increased uptake in areas of bone with enhanced blood supply or increased metabolic activity (a normal finding during the first 6 months postimplantation). Positive scans at 6 months after implantation are abnormal but do not differentiate among inflammation, possible loosening, and infection. However, newer technetium scans using labeled monoclonal antibodies appear to be more accurate for detecting prosthetic joint infection.55
A series of tests before, during, and after surgery are needed for early detection of implant infection. Periimplant infection does not respond well to oral antibiotic therapy because of antibiotic resistance from widespread clinical use of broad-spectrum antibiotics. The biofilm-forming strains of staphylococcus seem to have an even higher degree of resistance to antibiotics. The person is at risk for implant failure and possible death.6
Successful treatment requires a combination of surgical intervention (usually removal and replacement of the entire implant) and long-term antimicrobial therapy.104,105 A temporary implant coated with antibiotics may be put in place while the person is treated with IV antibiotics. When there are no further signs of infection, a permanent replacement prosthesis can be implanted.
TREATMENT.
Prosthesis removal accompanied by extensive and meticulous surgical debridement of surrounding tissue and effective antimicrobial therapy are usually necessary to treat deep infections, especially infections involving the interface between prosthesis and bone. Surgical debridement with retention of the prosthesis, followed by a course of antibiotics may be appropriate for a limited and select group or people.67
For more predictably effective treatment of prosthetic joint replacement sepsis, complete removal of all foreign materials (metallic prosthesis, cement, and any accompanying biofilm) is essential. This can be done in a one-or two-stage exchange. The most successful protocol incorporates standardized antimicrobial therapy with a two-stage surgical procedure: (1) removal of prosthesis and cement and placement of an antibiotic impregnated cement spacer, followed by a 6-week course of bactericidal antibiotic therapy and (2) reimplantation of a new prosthesis using cement impregnated with an antibiotic at the conclusion of the 6-week antibiotic course.27,75 Cementless two-stage hip procedures may result in infection rates similar to total hip arthroplasties done with cemented components, but studies are pending.60,107
Sometimes, surgical intervention is not possible because of a medical or surgical condition or refusal on the part of the affected individual. In such cases, life-long oral antibiotic treatment may be required to suppress the infection and retain function of the joint. Serial radiographs are needed to monitor progressive bone resorption at the bone-cement interface. In such cases, the localized septic process may still extend into adjacent tissue compartments or become a systemic infection, or the person may develop side effects of chronic antibiotic administration.
PROGNOSIS.
Infection associated with prostheses and implants can produce significant morbidity and occasionally death. Early recognition and prompt therapy for infection in any location is critical to reducing the risk of seeding the joint implant hematogenously. Situations likely to cause bacteremia should be avoided.14
The American Dental Association and the American Academy of Orthopedic Surgeons have jointly advised that a single dose of prophylactic antibiotic be given to selected individuals undergoing dental procedures associated with significant bleeding and potential hematologic bacterial contamination. The selected populations include people with inflammatory arthropathies, immunosuppression, diabetes mellitus, malnutrition, hemophilia, or previous prosthetic joint infection and anyone undergoing these dental procedures within 2 years after joint replacement.
Perioperative antibiotic prophylaxis has been shown to reduce deep wound infection effectively in total joint replacement surgery. Cephalosporins continue to be the antibiotic of choice for orthopedic surgeons because of the broad spectrum of activity against the most common pathogens. The antibiotic is given within 60 minutes before the incision is made (120 minutes if vancomycin or a fluoroquinolone is added), and if the procedure is long, another dose is administered during surgery. The medication is then continued for less than 24 hours after surgery.13
Although only 2% to 3% of prostheses become infected within 10 years after implantation, there is considerable risk of morbidity (e.g., hospitalization, amputation, or disability) and even death.4
DISKITIS
Overview and Incidence
Although spinal infections are rare, for both children and adults, the disk is the most common site. Diskitis can range from a self-limiting inflammatory process to a pyogenic infection. It may involve the intervertebral disk or vertebral end plates, or both. The infection rate in adults 2 to 8 weeks postdiskectomy is less than 3%.
Etiologic and Risk Factors
A bacterial origin is usually the cause of the infection. S. aureus is commonly found, but in some cases, no organism can be isolated. M. tuberculosis is also detected in disk infections. Children as young as 2 years old can develop diskitis, which may be confused with vertebral osteomyelitis. The origin of the infection in children may be traumatic, but the source of infection is more likely to be the hematogenous spread of a bacterial infection preceding the diskitis, such as in the upper respiratory or urinary tract. Fungal/yeast infections can also lead to diskitis.26
In adults the disk is relatively avascular, contributing to diskitis as the most common postoperative complication after diskectomy. The infection may involve the adjacent vertebrae and spread to the disk through the cartilaginous endplates. Other procedures capable of directly inoculating the disk, such as diskography, also carry the risk of infection. Direct inoculation is the only method by which an infection can arise from within the disk. As with children, urinary tract infections as the result of catheterization or cystoscopy may also be the underlying source of disk space infections.
Pathogenesis
If the infection arises within the disk itself, formation of a peridural abscess is not common. However, an infection extending to the disk from the cartilaginous plate can spread to cause an epidural abscess posteriorly or a paravertebral abscess anteriorly.47 The pathogenesis of infection formation is described in this chapter (see the Pathogenesis section under Infectious Arthritis in this chapter).
Clinical Manifestations
Diskitis presents in different ways at different ages, but fever and spinal pain are classic symptoms in children. In the very young child, back pain or a refusal to walk and pain with hip extension may be the first symptoms and must be taken seriously. Abdominal pain and weight loss may occur, and the child may not be able to flex the lower back.16 In children presenting with diskitis the concern is whether the diagnosis is actually vertebral osteomyelitis. Clinically, either condition will result in children refusing to walk, limping, or complaining of back pain. However, those with vertebral osteomyelitis often appear more ill and febrile.15,40
In adults, disk infection after spinal procedures usually is noted within a few days, whereas those developing from an infection at a distant site may not be evident for months. Spinal pain will be common and sometimes severe with radiation of the pain into the lower extremities. The lower extremity pain is not usually radicular; instead, it may involve multiple nerve levels. People may present with unusual posture and movement patterns that could be erroneously labeled pain of psychogenic origin. In both children and adults the back pain may range from mild to severe. The client will often report that the pain is made worse with activity and that rest does not relieve the pain.
MEDICAL MANAGEMENT
In children, routine radiographs are positive and often diagnostic but may not become so until 2 weeks have passed. Disk space narrowing, endplate irregularity, and a loss of lumbar lordosis are noted. Bone scans are also used for initial evaluation. Inflammatory markers may be elevated, but often laboratory tests are unhelpful and cultures of blood and disk tissue are negative. In an 18-year retrospective study, plain x-rays were found to provide adequate diagnostic information in 75% of the cases reviewed. However, MRI is essential in providing the differential diagnosis when vertebral osteomyelitis is a possibility; MRI reduces diagnostic delay and may help avoid the requirement for a biopsy.16,26,40
In adults the use of MRI in conjunction with bone scans using gadolinium enhancement has been found to be useful in differentiating normal postoperative disk space changes from those caused by infection. MRI is most useful in determining the extent of the infection and the required duration of oral antibiotic therapy after initial IV antibiotics but less valuable in demonstrating the type of infection (e.g., pyogenic versus tuberculous). The sclerosis present later in the disease course may be confused with a benign degenerative process or even with metastatic disease. MRI and biopsy may be used to differentiate chronic cases.
TREATMENT.
Treatment for the younger person may consist of bed rest, hip spica casting, and bracing. Antibiotics are used but do not appear to radically alter the natural course of the infection in children.16 In adults a removable body jacket can be used along with specific or empiric antibiotic therapy. The use of antibiotics prophylactically is common after spinal surgery in adults to prevent this condition from developing. Antifungal agents may first be tried for Candida diskitis. Surgery may be necessary.26
PROGNOSIS.
In both adults and children the prognosis is good, although pain may persist for several months up to several years. In children, long-term follow-up care has shown a resolution of the pain in spite of persistent radiographic changes.30
Late radiographic changes in adults include vertebral collapse, kyphosis, and eventually, bony ankylosis, which can take up to 2 years to run its course. Adults can expect similar spontaneous healing to occur, especially in those individuals with a strong immune response.
Complications can arise when the infection spreads or when an abscess forms. An epidural abscess can result in paralysis and is noted in older people, in those who have involvement of the cervical region, and in those with associated medical problems, such as diabetes.
INFECTIOUS (SEPTIC) ARTHRITIS
Overview and Incidence
Infectious causes of fever and arthritis can be divided into four groups by causative agent (Box 25-2). This section is confined to the discussion of bacterial arthritis (also called septic or infectious arthritis), which differs from reactive arthritis (see Chapter 27) in several ways. Bacterial arthritis may be a local response with joint destruction and sepsis, whereas reactive arthritis is defined as the occurrence of an acute, aseptic, and inflammatory arthropathy arising after an infectious process but at a site remote from the primary infection. Some of the other infectious causes of arthritis are discussed in other chapters (see Chapter 8 for Lyme disease and Epstein-Barr virus [EBV], Chapter 12 for rheumatic fever, and see Chapter 7 for HIV).
Etiologic and Risk Factors
Bacteria, viruses, and fungi are all capable of infecting a joint, by invading and inflaming the synovial membrane.54 S. aureus and Neisseria gonorrhoeae are the most common organisms responsible for infectious arthritis. Predisposing factors for development of septic arthritis are listed in Box 25-3. Microorganisms can be introduced into the joint by direct inoculation, direct extension, or by hematogenous (through the bloodstream) spread, which is the most common route (Box 25-4). In addition, direct penetrating trauma, joint arthroplasty, and chronic joint damage as seen in diseases, such as rheumatoid arthritis, are also considered to put a joint at risk.39
The primary risk factor for septic arthritis is a preexisting abnormal joint; up to 47% of affected individuals have underlying joint disease.35 A history of alcohol abuse, IV drug abuse, HIV infection, or other infectious disease increases the likelihood of having an infectious joint that becomes septic. Although nongonococcal infectious arthritis can affect individuals across the lifespan, children and older adults are at greatest risk.9
Pathogenesis
After being directly inoculated into the joint cavity, bacteria rapidly multiply in the liquid culture medium of the synovial fluid and are phagocytosed by synovial lining cells. Bacteria are either killed by the synovial cells or form microabscesses within the synovial membrane. Organisms that reach the synovium through the bloodstream multiply in enlarging microabscesses of the synovium until they break into the articular cavity.
Bacterial products, such as endotoxins and cell wall fragments, stimulate synovial cells to release tumor necrosis factor (TNF) and interleukin 1 (IL-1). These cytokines up-regulate expression of adhesion ligands in synovial membrane vessel endothelial cells, resulting in leukocyte attachment and migration into synovial fluid and articular tissues. Bacterial fragments form antigen-antibody complexes that activate the classic pathway of complement, and bacterial toxins activate the alternative complement pathway to produce proinflammatory products C3a and C5a (see Chapter 6).
The phagocytosis of bacteria also results in autolysis of neutrophils with release of lysosomal enzymes into the joint, which causes synovial, ligament, and cartilage damage. Cellular immune mechanisms also appear to play a role in acute joint infection. After 48 hours of synovial infection, T-lymphocytes infiltrate the synovium, IL-6 levels are increased, and B-cell activation results in immunoglobulin G (IgG) antibody production.
Bacterial toxins also activate the coagulation system, causing intravascular thrombosis in the subsynovial vessels and fibrin deposition on the surface of the synovium and articular cartilage. This layer of fibrin provides a gelatinous nidus for bacterial replication. Microvascular obstruction leads to ischemia and necrosis, further permitting abscess formation destroying the cartilage matrix.
Finally, after the acute necrotic inflammatory synovitis, the synovial membrane proliferates, forming an inflammatory exudate called pannus that erodes articular cartilage of the joint capsule and subchondral bone. All of this can take place in 17 days, quickly destroying a joint in the process. This underscores the need for urgency in detection and intervention in septic arthritis.
A chronic inflammatory synovitis may persist even after antibiotics have eradicated the infection. The threshold for starting empiric therapy with antibiotics for those individuals with acute joint pain and swelling should therefore be low.35
Clinical Manifestations
People with infectious arthritis can be any age and can present with an acute onset of joint pain, swelling, tenderness, and loss of motion. Fever, chills, and other systemic symptoms depend on the stage of the illness. Physical examination may reveal the classic signs of infection such as increased temperature of the joint, swelling, redness, and loss of function. Pus may drain outside through a sinus formed from the joint to the outside. Only the severity and the nature of these signs will differentiate the septic joint from more mundane causes such as tendinitis and other noninfectious inflammatory diseases.
A child with a septic joint will often refuse to bear weight and be extremely tender to palpation at the joint and along the metaphysis. Destruction of the joints can proceed rapidly and have long-lasting effects.19 In addition to the infection, the WBCs that enter the joint to combat the infection release enzymes that have a deleterious effect on articular cartilage. In a series of young children studied under the age of 2 years with septic knees, 24% had a varus or valgus deformity at long-term follow-up care.98
In adults, S. aureus produces a monarticular sepsis, usually at the hip or knee. In children, the ankle and elbow are also common sites. Although not as common, polyarticular septic infectious arthritis has been reported. Gonococcus affects mostly women and may produce skin lesions, tenosynovitis, and polyarthralgias, in addition to systemic symptoms. Prosthetic joints are also sites of infection, which is probably introduced at the time of surgery. S. epidermidis is often the cause.
In general the coexistence of fever and the signs and symptoms of an acute exacerbation of arthritis must arouse suspicions of a septic joint and be managed as a medical emergency until proved otherwise.39,53
MEDICAL MANAGEMENT
Besides a detailed history and physical examination, the confirmation of the diagnosis is made by analysis of the joint fluid obtained by aspiration. The decision to aspirate the joint, however, is made on the basis of history and physical examination. The method to obtain a sample of the aspirate depends on the joint that is involved. Needle aspiration when possible is often considered the method of choice.66 Aspiration of the sacroiliac and hip joints is difficult, and fluoroscopy is sometimes used. Decisions on treatment and appropriate antibiotics are aided by the results of cultures, stains, and laboratory studies such as the WBC count and ESR.
With the development of advanced deoxyribonucleic acid (DNA) analysis techniques, identification of traces of bacterial genomes may eventually make it possible to develop specific vaccines or pharmacologic agents to prevent or treat septic arthritis.
TREATMENT.
As already mentioned, any joint infection is considered a medical emergency. Admission to the hospital for treatment with specific IV antibiotics is required. Continued treatment with oral medication for an additional 2 to 3 weeks is standard.
Aspiration of the joint is critical. Besides needle aspiration, more aggressive techniques of tidal irrigation, arthroscopy, or arthrotomy may be used depending on the situation.66 Open drainage is indicated for hip joint infections. In prosthetic joints the infection may require removal of the hardware and cement, along with a more prolonged course of antibiotics.
Early in the course of intervention, the joint should be rested. This may be accomplished by splinting, traction, or casting. Care in application of the splint will preserve function, and the splint should be removed periodically for ROM exercise. The importance of these simple ROM exercises cannot be overlooked because the risk for joint contracture as a complication of immobilization is a concern, especially in the older adult. More vigorous types of exercise and aggressive mobilization activities are performed when signs of infection have resolved.54,66
The aggressiveness of intervention is dictated by the specific organism, the joint involved, duration of symptoms, and the health of the individual. Surgical drainage is often required to preserve function and prevent complications. In some cases, joint instability (e.g., chronic or repeated hip subluxation) may require more extensive surgical intervention.
PROGNOSIS.
As for other infectious diseases, prompt treatment is the key to a successful outcome. If treatment is initiated within 5 to 7 days of onset, a good or excellent long-term result can be expected.66,99 Currently, the mortality rate has dropped considerably for infections caused by nongonococcal agents; however, sequelae in the form of destructive changes in the bone or joint can result in significant functional limitations.
Mortality is higher in the older adult (increases after age 65 years) even with quick and correct interventions.66 Overall mortality from septic arthritis ranges from 10% to 25%, and permanent joint disability occurs in 25% to 50% of survivors. Septic arthritis of the knee is associated with better outcomes than that of the hip.35 The more common complications include osteomyelitis, abscess formation, and permanent loss of joint motion and joint instability. If the infection is not controlled, toxemia and septicemia can cause death.
INFECTIOUS (INFLAMMATORY) MUSCLE DISEASE
Overview and Incidence
Myositis is a general term used to describe inflammation of the muscles that can be an autoimmune condition or directly caused by viral, bacterial, and parasitic agents. Infection-induced myositis is most often caused by S. aureus and parasites such as trichinella and the tapeworm larva, Taenia solium.
When affecting skeletal muscle, these infectious agents result in inflammatory changes with sequela, ranging from significant functional losses to a minor self-limiting condition. Autoimmune conditions can be activated or aggravated by infections, which may explain the link between myositis, infections, and autoimmune processes.
The most common forms are dermatomyositis (DM), polymyositis (PM), and inclusion body myositis (IBM) (Box 25-5). DM appears to occur more often in children and older adults. Inflammatory myopathies are discussed further in Chapter 27; PM and DM are included in Chapter 10.
IBM is the most common acquired muscle disease in adults over 50 years and often misdiagnosed. This form is often progressive and debilitating and often does not respond to available treatments.12,37
Incidence.: Myositis is diagnosed in 1 in 100,000 people a year, although some experts suspect that many cases may go unidentified because it is so often mistaken for the symptoms of aging or in women, depression.71
Etiology and Pathogenesis
Myositis can be the first sign of a malignancy. Doctors have been aware of cancer-associated myositis since 1916. Recent studies have quantified the risk, finding that people with DM face a threefold risk of cancer, whereas those individuals with PM face a 40% increase in risk.62
Some experts theorize that the autoimmune rheumatic diseases that occur in people over age 40 years may reflect an anticancer immune response in a large number of people. In effect, these people may be cancer survivors without knowing it because the muscle cells are accidentally harmed during the immune system’s assault on the tumor.21,62
The antigens that produce the immune response are present in normal muscle tissue but at low levels. They are much more prevalent in myositis cells of individuals with autoimmune myositis and in muscle cells that are regenerating such as occurs after an injury. It is hypothesized that a feed-forward loop occurs when damaged muscle cells start to repair themselves. These cells express higher amounts of the antigens, causing the immune system to respond; the immune response causes further damage to the muscle, which in turn repairs itself, its regenerating cells expressing even more antigens, and continuing the feed-forward cycle.22
The IBM form of myositis appears to be autoimmune-mediated by cytotoxic T cells and deposits of amyloid-related proteins. There is a strong association of IBM with human leukocyte antigens (HLA) I and II. IBM tends to develop in individuals with other autoimmune disorders or immunodeficiency.32,72 A small number of IBM cases may be hereditary (h-IBM) but most are “sporadic” (s-IBM), meaning there is not a direct genetic link.71
Inflammation is a major cause of muscle damage. Numerous drugs may induce myopathies, including cholesterol-lowering statins. Lipid-lowering drugs associated with myotoxicity can cause symptoms ranging in severity from myalgias to rhabdomyolysis, resulting in renal failure and death (discussed in Chapter 27).7,28,69
A study sponsored by Scripps Health, called Imposter-16 (“Is Myopathy Part of Statin Therapy?”), is trying to determine whether muscle diseases, including myositis, are linked to the cholesterol-lowering medications. The study is currently ongoing, but preliminary results suggest some people with certain metabolic abnormalities are predisposed to statin-induced rhabdomyolysis.5
Myositis caused by parasites is considered a relatively uncommon condition, however, the parasitic infection trichinosis is reported to affect up to 4% of the population.
Clinical Manifestations
The commonly observed symptoms of this family of conditions are as would be expected for any inflammatory process. These nonspecific symptoms include malaise, fever, muscle swelling, pain, tenderness, and lethargy. Specifically, the inflammatory response found in PM and DM is in connective tissue and muscle fibers.
Other than the symptoms associated with these infections, there is a risk of tissue necrosis and extensive muscle tissue damage with atrophy and weakness, especially if left untreated. Other clinical features of myositis are dysphagia, decreased esophageal motility, vasculitis, Raynaud’s phenomenon, cardiomyopathy, and interstitial pulmonary fibrosis. A purple skin rash and eyelid edema are often associated with DM. The distribution of the rash includes the eyelids, face, chest, and extensor surfaces of the extremities. In adults, subcutaneous calcium deposits are a sign of severe long-term DM.
In most cases, IBM progresses slowly over months or years and is characterized by frequent falling episodes, trouble climbing stairs or standing from a seated position. Drop foot and subsequent tripping may be reported. Weak grip, difficulty swallowing, and muscle atrophy and weakness are often accompanied by functional decline and pain or discomfort secondary to weakness.71
MEDICAL MANAGEMENT
In addition to careful and thorough history, a muscle biopsy is the primary diagnostic tool. Muscle aches and pains associated with a bout of influenza often may actually be a subacute viral myopathy. A differential diagnosis requires muscle biopsy, electromyography, and laboratory values.
The muscle biopsy will make the differentiation between PM, DM, and IBM and exclude other myotonic disease. Electromyography will demonstrate muscle irritability and myopathic changes. Because of the associated release of creatine kinase (CK) into the blood with skeletal muscle damage, this enzyme can be a useful measure of the extent of the infection (see Table 40-15). CK levels are five to ten times higher than normal in PM, but only mildly increased in IBM.12
TREATMENT AND PROGNOSIS.
Aggressive early treatment of any of these conditions will lead to an improved prognosis. Trichinosis can be very successfully treated with pharmacologic agents. The treatment of PM and DM often includes immunosuppressive therapy and corticosteroids. There is no established treatment that improves, arrests, or slows the progression of IBM; it is resistant to treatment with antiinflammatory, immunosuppressant, or immunomodulating agents.49 Because of the resulting muscle weakness and possible extensive skeletal muscle damage associated with myositis, the client must be prepared for an aggressive and prolonged rehabilitative process.
The role of the physical and occupational therapist should not be underestimated in the attainment of a successful outcome. Submaximal exercise has been shown to be effective, although eccentric or intense exercise is not recommended.12,51
Other clinical trials are testing new drugs to add to the arsenal of corticosteroids, immunosuppressants, and IV immunoglobulin, a plasma product. Scientists are conducting trials of rituximab, an artificial antibody used to treat certain types of cancer; infliximab, which blocks the effect of TNF, which is a protein associated with inflammation; and etanercept, which blocks TNF-α, also involved in inflammation.71
For people with PM and DM, existing medications work well, although many of the drugs have serious side effects and may cease being effective over time.
Infections of Bursae and Tendons
Acute infections affecting the bursae and tendons are uncommon and must be treated appropriately to avoid complications. The hand is very susceptible to scratches, bites, and subsequent infections. Hand infection can range from cellulitis to tenosynovitis. Because of the superficial nature of these tissues and the potential for dysfunction, hand infections are given special attention in this section.
Etiologic and Risk Factors
The bursae and tendons that lie close to the skin surface are most susceptible to infection from direct contact with microorganisms. Trauma to the elbow and knee is common, especially in sports such as wrestling. Anaero bic bacteria are more commonly seen in wounds from bites and in people with diabetes. The bacteria enter the body by direct inoculation through a local skin abrasion or with common procedures such as a cortisone injection into the inflamed bursae. S. aureus is the most common organism isolated and may cause up to 80% of infections.
Hand infections often develop from untreated injuries. Up to 60% of hand infections are related to trauma, 25% are caused by human bites, and 10% are a result of animal bites.15 As with all infections, people with diabetes or who are immunocompromised have a greater risk of developing an infection in the hand. In addition the risk of osteomyelitis is also of concern due to the close proximity of bone.
Pathogenesis
Infection in the hand can spread along synovial sheaths, fascial planes, and via lymphatic channels. Bursae are lined with a membrane similar to synovium and are therefore subject to the same pathologic processes, namely, inflammatory conditions caused by acute or chronic infections. (See Pathogenesis section under Infectious Arthritis in this chapter.)
Clinical Manifestations
The olecranon and prepatellar bursae can be sites of localized infection. An olecranon bursal infection will cause pain, loss of function, and swelling, which may be accompanied by cellulitis. Infections of other bursae, such as the prepatellar and subdeltoid bursae, have similar presentations.
Tendon sheaths of the extremities can also become infected. As mentioned earlier, the hand is a common site because of its susceptibility to minor trauma. The anatomy of the hand determines the nature and presentation of the infection. For example, the tendon sheaths of the thumb and small finger extend proximally to the wrist, whereas the sheaths of the index, long, and ring fingers stop at the proximal pulley.
An infection of the flexor tendon of the thumb could rapidly spread to the small finger. Common signs associated with an infectious tenosynovitis include a finger maintained in slight flexion; fusiform (spindle-shaped) swelling; pain on extension (passive or active); and tenderness along the tendon sheath into the palm.
MEDICAL MANAGEMENT
DIAGNOSIS, TREATMENT, AND PROGNOSIS.
In most joints, examination will identify a localized swelling not a joint effusion. Aspiration of fluid for laboratory analysis is performed before treatment. The use of antibiotics is often adequate, but surgical incision and drainage are sometimes required; occasionally, bursectomy is required. Prompt treatment with drainage, irrigation, and antibiotics is crucial.
Appropriate treatment of hand infections is based on an accurate identification by culture of the organism causing the infection. In addition to the appropriate antibiotic, the hand is immobilized and elevated, and often, surgical drainage is necessary. Necrotic tissue is cautiously debrided, and the wound is left open to drain.
Early and aggressive rehabilitation is essential, especially for a structure as complicated and integrated as the hand. Both physical and occupational therapists may play a role in this process. Given the potential complications of surgery and immobilization and the potential for tissue loss, a comprehensive rehabilitation program is necessary to maximize function.
EXTRAPULMONARY TUBERCULOSIS
TB is an acute or chronic infection caused by M. tuberculosis that can affect multiple organ systems via lymphatic and hematogenous spread during the initial pulmonary infection (see Chapter 15). Disseminated or miliary TB involves not only the lungs but also most other organ systems. Systems involved may include the pulmonary, genitourinary, musculoskeletal, and lymphatic systems. Of these, the lymphatic system is most commonly involved in immunocompromised hosts such as those with HIV.
Extrapulmonary TB is more difficult to diagnose and treat than pulmonary TB. This is due in part to clinicians’ lack of familiarity with the condition. In addition, extrapulmonary TB is often in inaccessible areas, which makes aspiration or biopsy and therefore diagnosis, more difficult. Also, smaller numbers of bacilli can cause extensive damage to joints but are harder to detect. TB involving the bone is usually transferred hematogenously from some other organ, usually the lung. Only one-fourth of people with skeletal TB have a known history of TB.
Skeletal Tuberculosis
Because skeletal TB is relatively uncommon, delays in diagnosis are frequent. After a 40-year decline, the incidence of TB has increased over the past several decades. The World Health Organization (WHO) estimates that more than 8 million new cases of TB occur annually and approximately 3 million individuals die from TB and associated complications every year.3 More than 15 million people in the United States are estimated to be infected with TB. This increase is due in part to the presence of acquired immune deficiency syndrome (AIDS). The causes for this change in number of active cases of TB are discussed at length in Chapter 15. Although TB is found in all 50 states, New York, California, and Hawaii have had the highest incidence. Skeletal TB is uncommon, but infection rates have held constant over the years. About 10% to 15% of TB is extrapulmonary and only 10% of extrapulmonary TB is skeletal.
Pathogenesis and Clinical Manifestations
Extrapulmonary TB is spread hematogenously from other organs. In adults, the onset of skeletal TB is insidious, developing 2 to 3 years after the primary infection. Early signs and symptoms include pain and stiffness; the pain may be localized or referred. The lower thoracic and lumbar spine is commonly involved (Pott’s disease), but other sites (e.g., weight-bearing joints or elbows) have been reported.31 Systemic signs, such as fever, chills, weight loss, and fatigue, are not common in the early phase. Joint effusion often occurs with TB arthritis and has been shown to affect muscles and nerves around the joint.25,109
In the case of spine involvement (occurring 5% in the cervical spine, 25% in the thoracic spine, and 20% in the lumbar and lumbosacral spine), infection begins in the cancellous bone of the vertebral body and eventually spreads to the intervertebral disk and adjacent vertebrae. As the disease progresses, nerve root irritation, pressure from abscess, and collapse of the vertebral body will cause a progressive increase in pain and protective spasm with cord compression and possible paraplegia.38,70 The abscess may extend from the lumbar region to the psoas muscle, producing hip pain (see Figs. 16-14 and 16-15).
MEDICAL MANAGEMENT
Early diagnosis is very helpful in preserving articular cartilage and the joint space but is often delayed for several months to years after the initial presentation because there are no symptoms pathognomonic of extrapulmonary TB. Treatment may be delayed because symptoms are consistent with chronic sciatica, when the true cause of the radiating pain is tuberculous sacroiliitis with an anterior synovial cyst.24
Conventional radiographs are important in the initial detection, and computed tomography (CT) and MRI can assist in further evaluation. Confirmation of skeletal TB requires microbiologic assessment with smear and culture. In the spine, this confirmation can be accomplished with fine-needle aspiration. Tissue biopsy is more often required for extrapulmonary disease.
TREATMENT AND PROGNOSIS.
Treatment does not differ for pulmonary and extrapulmonary TB. Although surgical debridement is sometimes necessary, usually pharmacologic treatment is sufficient. (See Chapter 15 for discussion of medical management of TB.) Chemotherapy is the mainstay in the management of TB spondylitis, but decompressive surgery may be required in the presence of Pott’s paraplegia.70
Rehabilitation after surgery for TB of the spine or extremities follows standard orthopedic principles. Medical intervention and subsequent rehabilitation are individualized based on the extent of the infection. Surgical treatment of joint infection may include arthrotomy, synovectomy, and treatment of articular erosions.
Extraarticular infections can sometimes be treated with curettage and bone grafting. For more advanced infections, resection of bones and joints, arthrodesis, and limb salvage or amputation may be indicated. Factors to be considered include the affected bone, extent of surgical excision, and involvement of soft tissue, articular cartilage, or bone. Weight bearing is often limited, but active movement is often encouraged.
In the spine, surgery is more often needed to address nerve compression or deformity secondary to collapse of the vertebral body rather than the infection. The resultant deformity often includes a marked kyphotic curve with a gibbus formation (Fig. 25-4). Paralysis can be a serious complication of vertebral TB and can be a result of the disease process or a secondary spinal deformity. Paralysis persisting longer than 6 months is unlikely to improve, and late paralysis with inactive disease and significant kyphosis is much less responsive to treatment.70
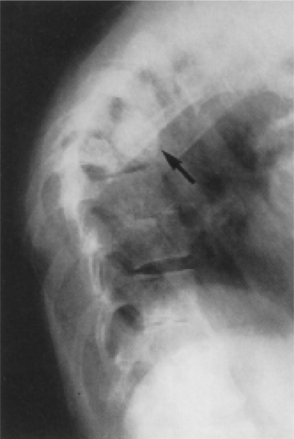
Figure 25-4 Tuberculous spondylitis. Involvement at multiple levels. Gibbus deformity is seen in the upper thoracic region (arrow). (From Yao D, Sartoris D: Musculoskeletal tuberculosis, Radiol Clin North Am 33:681, 1995.)
In the joint, if TB is diagnosed early when the infection is confined to the synovium, rest, medication, and joint protection may be adequate. In advanced disease with caseation, fibrosis, and scarring, the vascularity is reduced (Fig. 25-5), which makes medications less effective. Surgical excision or curettage of the affected areas may be necessary. In joints the granulomatous tissue acts to separate the articular cartilage from the underlying bone.
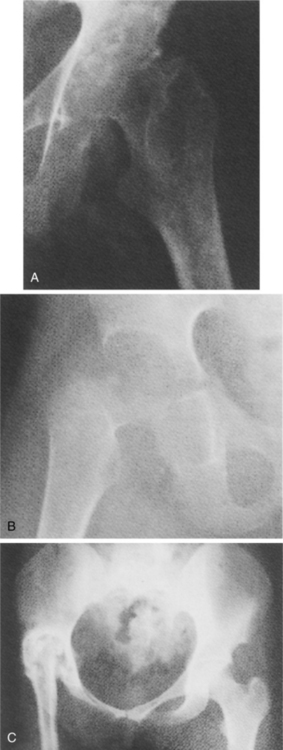
Figure 25-5 Tuberculous arthritis. A, Bony erosion of acetabulum and femoral head with joint space loss. There also is evidence of periarticular osteopenia. B, Similar findings to A but with further destruction of the femoral head and acetabulum. C, Advanced TB of the hip with superior displacement and ankylosis of the right hip joint. (From Yao D, Sartoris D: Musculoskeletal tuberculosis, Radiol Clin North Am 33:687, 1995.)
SUMMARY OF SPECIAL IMPLICATIONS FOR THE THERAPIST
Rehabilitation after medical intervention for infectious diseases must proceed in a comprehensive and coordinated fashion. The pathology of each type of infectious disease process and every medical decision made in treatment will have some bearing on rehabilitation and outcome. When should ROM exercise begin? Should it be active or passive? What is the client’s weight-bearing status? What is the general health of the client? What are the client’s goals and expectations? Many questions such as these must be addressed when planning the rehabilitation program.
The hip joint is commonly affected and has been alluded to in several chapters as a site of infectious processes of many origins and will be used here again to illustrate some factors that should be considered in planning rehabilitation. The client and therapist may be anxious for an exercise program to begin, but this must be done in concert with medical treatment and consideration of the stage of recovery.
Early in the course of intervention, rest may be a predominant feature but even this calls for therapy intervention. All health care providers must be made aware of the potentially adverse effects of apparently simple movements. For example, using a bedpan and performing isometric exercises produce acetabular contact pressure close to that of walking.52 Therefore clinicians should not assume that a client who is on bed rest is not producing elevated joint compressive forces. Clients must be instructed in proper methods of moving, transferring, and positioning themselves.
Active ROM exercise is often the first type of supervised exercise permitted. Even simple movements must be done while noting limits set by pain, spasm, or apprehension. Active hip flexion has been found to increase acetabular contact pressure similar to that of full weight bearing.97 However, passive ROM exercise has been found to have a beneficial effect on healing joints.43,87
Continuous passive ROM as a means of early mobilization in a postoperative rehabilitation regimen has been investigated and found to have no long-term benefit compared with daily standardized exercises.10,62,65 However, the data on the effect of continuous passive motion on overall analgesic use and prevalence of deep vein thrombosis remain under investigation.
It may be worth remembering the motion requirements of certain positions so that functional goals can be set; for example, ascending stairs requires just over 65 degrees of hip flexion, whereas sitting in a chair requires about 105 degrees.57 Restoring full ROM may not be as realistic or as necessary as obtaining the ROM required to restore function.
Once the client is ambulatory, his or her weight-bearing status must be determined. This again depends on the type of infection, surgical procedure, stage of recovery, and extent of joint destruction. Articular cartilage heals very slowly, and the cartilage on weight-bearing surfaces should be protected from undue compressive or shear forces. In addition, immobilization has deleterious effects on articular cartilage and other periarticular tissue1,38 (see Chapter 6 and Table 6-7).
Exercise after prolonged immobilization should take these factors into consideration. Often, a non–weight-bearing status is used with the intention of minimally loading the joint. In fact, the compressive forces may actually increase when compared with those of a touch-down, weight-bearing gait pattern.84 Other factors, such as the weight of the limb and muscular contraction, must be considered. Long-term use of an ambulatory aid may be indicated even as much as a 2-to 3-year period.46 The use of a cane in the contralateral hand can reduce weight-bearing forces by up to 15%.36
References
1. Akeson, W, Amiel, D, La Violette, D. The connective tissue response to immobility: a study of the chondroitin-4 and-6 sulfate; dermatan sulphate changes in periarticular connective tissue of control and immobilized knees of dogs. Clin Orthop. 1967;51:183–197.
2. American Physical Therapy Association. Guide to physical therapist practice. Phys Ther. 2001;81(suppl 1):1–699.
3. American Thoracic Society. Diagnostic standards and classification of tuberculosis in adults and children. Am J Crit Care Med. 2000;161:1376–1395.
4. An, YH, Friedman, RJ. Prevention of sepsis in total joint arthroplasty. J Hosp Infect. 1996;33:93–108.
5. Antons, KA. Clinical perspectives of statin-induced rhabdomyolysis. Am J Med. 2006;119(5):400–409.
6. Arciola, CR, Alvi, FI, An, YH, et al. Implant infection and infection resistant materials: a mini review. Int J Artif Organs. 2005;28(11):1119–1125.
7. Baer, AN. Myotoxicity associated with lipid-lowering drugs. Curr Opin Rheumatol. 2007;19(1):67–73.
8. Barret, JP, Desai, MH, Herndon, DN. Osteomyelitis in burn patients requiring skeletal fixation. Burns. 2000;26(5):487–489.
9. Bass, AR, Schoen, RT. Bacterial arthritis: aggressive diagnosis, effective therapy. J Musculoskel Med. 1996;13(8):13–21.
10. Beaupre, LA, et al. Exercise combined with continuous passive motion or slider board therapy compared with exercise only: a randomized controlled trial of patients following total knee arthroplasty. Phys Ther. 2001;81(4):1029–1037.
11. Bennett, OM, Namnyak, SS. Acute septic arthritis of the hip joint in infancy and childhood. Clin Orthop. 1992;281:123–132.
12. Boon, A. Inclusion body myositis masquerading as polymyositis: a case study. Arch Phys Med Rehab. 2000;81(8):1123–1126.
13. Bratzler, DW, Houck, PM, Surgical Infection Prevention Guideline Writers Workgroup. Antimicrobial prophylaxis for surgery an advisory statement from the National Surgical Infection Prevention Project. Am J Surg. 2005;189(4):395–404.
14. Brause, B. Infections with prostheses in bones and joints. In Mandell G, Bennett J, Dolin R, eds.: Principles and practice of infectious diseases, ed 6, Philadelphia: Churchill Livingstone, 2005.
15. Brown, D, Young, L. Hand infections. South Med J. 1993;86:56–66.
16. Brown, R, et al. Discitis in young children. J Bone Joint Surg. 2001;83(1):106–111.
17. Buhl, T. Bone infection in patients suspected of complicating osteomyelitis: the diagnostic value of dual isotope bone-granulocyte scintigraphy. Clin Physiol Funct Imaging. 2005;25(1):20–26.
18. Bullough, PG. Bullough and Vigorita’s orthopaedic pathology, ed 3. London: Mosby-Wolfe, 1997.
19. Çaksen, H, et al. Septic arthritis in childhood. Pediatr Intern. 2000;42(5):534–540.
20. Carek, PJ, Dickerson, LM, Sack, JL. Diagnosis and management of osteomyelitis. Am Fam Phys. 2001;63(12):2413–2420.
21. Casciola-Rosen, L. Autoimmune myositis: new concepts for disease initiation and propagation. Curr Opin Rheumatol. 2005;17(6):699–700.
22. Casciola-Rosen, L. Enhanced autoantigen expression in regenerating muscle cells in idiopathic inflammatory myopathy. J Exp Med. 2005;201(4):591–601.
23. Centers for Disease Control and Prevention. CDC fact book 2000/2001. Atlanta: Department of Health and Human Services, 2000.
24. Chen, WS. Chronic sciatica caused by tuberculous sacroiliitis. Spine. 1995;20:1194–1196.
25. Chen, WS, Eng, HL. Posterior interosseous neuropathy associated with tuberculous arthritis of the elbow joint: report of two cases. J Hand Surg (Am). 1994;19:611–613.
26. Chia, SL. Candida spondylodiscitis and epidural abscess: management with shorter courses of anti-fungal therapy in combination with surgical debridement:. J Infect. 2005;51:17–23.
27. Chohfi, M, et al. Pharmacokinetics, uses, and limitations of vancomycin-loaded bone cement. Int Orthop. 1998;22(3):171–177.
28. Christopher-Stine, L. Adult inflammatory myopathies. Best Pract Res Clin Rheumatol. 2004;18(3):331–344.
29. Colson, P, La Scola, B, Champsaur, P. Vertebral infections caused by haemophilus aphrophilus: case report and review. Clin Microbiol Infect. 2001;7(3):107–113.
30. Crawford, A, et al. Discitis in children. Clin Orthop. 1991;266:70–79.
31. Dahl, C. Physical therapy management of tuberculous arthritis of the elbow. Phys Ther. 2001;81(6):1253–1259.
32. Dalakas, MC. Inflammatory, immune, and viral aspects of inclusion-body myositis. Neurology. 2006;66(suppl 1):33–38.
33. DeBraun, BJ. Prevention of infection in the orthopedic surgery patient. Nurs Clin North Am. 1998;33(4):671–684.
34. Deyo, R, Rainville, J, Kent, D. What can the history and physical examination tell us about low back pain? JAMA. 1992;268:760–765.
35. Drees, M. Septic arthritis: treat early to minimize morbidity. J Musculoskel Med. 22(4), 2005.
36. Ely, DD, Schmidt, GI. Effect of cane on variables of gait for patients with hip disorders. Phys Ther. 1977;57:507–516.
37. Engel, WK. Inclusion-body myositis: clinical, diagnostic, and pathologic aspects. Neurology. 2006;66(suppl 1):20–29.
38. Enneking, WF, Horowitz, M. The intraarticular effects of immobilization on the human knee. J Bone Joint Surg. 1972;54A:973–985.
39. Erkan, D, Yazici, Y, Paget, SA. Fever and arthritis: narrowing the diagnosis. J Musculoskel Med. 2000;17(11):676–687.
40. Fernandez, M, Carroll, CL, Baker, CJ. Discitis and vertebral osteomyelitis in children: an 18-year review. Pediatrics. 2000;105(6):1299–1304.
41. Frisbie, JH, et al. Vertebral osteomyelitis in paraplegia: incidence, risk factors, clinical picture. J Spinal Cord Med. 2000;23(1):15–22.
42. Gabriel, S, et al. Complications leading to surgery after breast implant. N Engl J Med. 1997;336(10):677–682.
43. Gelberman, RH, et al. Effects of early intermittent passive mobilization on healing canine tendons. J Hand Surg. 1982;7:170–175.
44. Gentry, LO. Management of osteomyelitis. Int J Antimicr Agents. 1997;9(1):37–42.
45. Giulieri, SG. Management of infection associated with total hip arthroplasty according to a treatment algorithm. Infection. 2004;32:222–228.
46. Givens-Heiss, D, et al. In vivo acetabular contact pressures during rehabilitation. II. Postacute phase. Phys Ther. 1992;72:7007–7010.
47. Goel, V, Young, JB, Patterson, CJ. Infective discitis as an uncommon but important cause of back pain in older people. Age Ageing. 2000;29(5):454–456.
48. Goodman, CC, Snyder, TE. Differential diagnosis in physical therapy. Philadelphia: WB Saunders, 2000.
49. Griggs, RC. The current status of treatment for inclusion-body myositis. Neurology. 2006;66(2 suppl 1):30–32.
50. Heggeness, M, et al. Late infection of spinal instrumentation by hematogenous seeding. Spine. 1993;18:492–496.
51. Heikkillä, S. Rehabilitation in myositis. Physiother. 2001;87(6):301–309.
52. Hodge, WA, et al. Contact pressure from an instrumented hip endoprosthesis. J Bone Joint Surg. 1989;71A:1378–1386.
53. Ignacio, EM. Pediatric septic arthritis. Trauma. 2001;5:67–81.
54. Issa, N. Diagnosing and managing septic arthritis: a practical approach. J Musculoskel Med. 2003;20(2):161–211.
55. Ivancevic, V. Imaging of low-grade bone infection with a technetium-99m labeled monoclonal anti-NCA-90 Fab’ fragment in patients with previous joint surgery. Eur J Nucl Med Mol Imaging. 2002;29:547–551.
56. Janowsky, E, Kupper, L, Hulka, B. Meta-analyses of the relation between silicone breast implants and the risk of connective tissue diseases. N Engl J Med. 2000;342(11):781–790.
57. Johnston, RC, Schmidt, GL. Hip motion measurements for selected activities of daily living. Clin Orthop. 1970;72:205–215.
58. Khan, MH. Serum C-reactive protein levels correlate with clinical response in patients treated with antibiotics for wound infections after spinal surgery. Spine J. 2006;6(3):311–315.
59. Klemm, K. The use of antibiotic-containing bead chains in the treatment of chronic bone infections. Clin Microbiol Infect. 2001;7(1):28–31.
60. Kraay, MJ, Goldberg, VM, Fitzgerald, SJ, et al. Cementless two-staged total hip arthroplasty for deep periprosthetic infection. Clin Orthop Relat Res. 2005;441:243–249.
61. Lachiewicz, PF. The role of continuous passive motion after total knee arthroplasty. Clin Orthop. 2000;380:144–150.
62. Levine, SM. Cancer and myositis: new insights into an old association. Curr Opin Rheumatol. 2006;18(6):620–624.
63. Lipworth, L, Tarone, RE, McLaughlin, JK. Silicone breast implants and connective tissue disease: an updated review of the epidemiologic evidence. Ann Plast Surg. 2004;52(6):598–601.
64. Love, C, Tomas, MB, Tronco, GG, et al. FDG PET of infection and inflammation. Radiographics. 2005;25(5):1357–1368.
65. MacDonald, SJ, et al. Prospective randomized clinical trial of continuous passive motion after total knee arthroplasty. Clin Orthop. 2000;380:30–35.
66. Mader, JT, et al. Bone and joint infections in the elderly: practical treatment guidelines. Drugs Aging. 2000;16(1):67–80.
67. Marculescu, CE, Berbari, EF, Hanssen, AD, et al. Outcome of prosthetic joint infections treated with debridement and retention of components. Clin Infect Dis. 2006;42(4):471–478.
68. Mason, A. The value of white blood cell counts before revision total knee arthroplasty. J Arthroplasty. 2003;18(8):1038–1043.
69. McClure, DL. Statin and statin-fibrate use was significantly associated with increased myositis risk in a managed care population. J Clin Epidemiol. 2007;60(8):812–818.
70. Moon, MS. Tuberculosis of the spine: controversies and a new challenge. Spine. 1997;22(15):1791–1797.
71. Myositis Association About myositis. Available on-line at: www.myositis.org Accessed August 2, 2007
72. Needham, M. Genetics of inclusion-body myositis. Muscle Nerve. 2007;35(5):549–551.
73. Nyren, O, et al. Risk of connective tissue disease and related disorders among women with breast implants: a nationwide retrospective cohort study in Sweden. BMJ. 1998;316(7129):417–422.
74. Obenaus, C. Extra-large press-fit cups without screws for acetabular revision. J Arthroplasty. 2003;18(3):271–277.
75. Pagnano, M, et al. Blood management in two-stage revision knee arthroplasty for deep prosthetic infection. Clin Orthop. 1999;367:238–242.
76. Palestro, CJ. Diagnostic imaging tests and microbial infections. Cell Microbiol. 2007. [Epub ahead of print].
77. Palestro, CJ. Infection and musculoskeletal conditions: imaging of musculoskeletal infections. Best Pract Res Clin Rheumatol. 2006;20(6):1197–1218.
78. Palmer, L. Management of the patient with a total joint replacement: the primary care practitioner’s role. Lippincott’s Prim Care Pract. 1999;3(4):419–427.
79. Patzakis, M, Mader, J. Symposium: Current concepts in the management of osteomyelitis. Contemp Orthop. 1994;28:157–185.
80. Perry, CR. Bone and joint infections. London: Martin Dunitz, 1996.
81. Petri, WA, Mann, BJ, Huston, CD. Microbial Adherence. In Mandell G, Bennett J, Dolin R, eds.: Principles and practice of infectious diseases, ed 6, Philadelphia: Churchill Livingstone, 2005.
82. Phillips, NI, Robertson, JA. Osteomyelitis of the skull vault from a human bite. Brit J Neurosurg. 1997;11(2):168–169.
83. Pittet, B, Montandon, D, Pittet, D. Infection in breast implants. Lancet Infect Dis. 2005;5(8):462–463.
84. Radin, EL, et al. Practical biomechanics for the orthopedic surgeon, ed 2. Philadelphia: Churchill Livingstone, 1992.
85. Rajbhandari, SM, Sutton, M, Davies, C. “Sausage toe,” a reliable sign of underlying osteomyelitis. Diabetic Med. 2000;17(1):74–77.
86. Roach, K, et al. The use of patient symptoms to screen for serious back problems. J Orthop Sports Phys Ther. 1995;21:2–6.
87. Salter, RB, et al. The biological effect of continuous passive motion on the healing of full thickness defects in articular cartilage. J Bone Joint Surg. 1980;62A:1232–1251.
88. Schafroth, M. Infections. In: Ochsner PE, ed. Total hip replacement. Berlin: Springer-Verlag, 2003.
89. Shanklin, D, Smalley, D. Dynamics of wound healing after silicone device and implantation. Exp Mol Pathology. 1999;67(1):26–39.
90. Shanklin, D, Smalley, D. The immunopathology of siliconosis: history, clinical presentation and relation to silicosis and the chemistry of silicon and silicone. Immunol Res. 1998;18(3):125–173.
91. Singh, NK. Reconstructive breast implantation after mastectomy for breast cancer-invited critique. Arch Surg. 2005;140:1160–1161.
92. Smalley, D, et al. Lymphocyte response to silica among offspring of silicone breast implant recipients. Immunobiology. 1996;196(5):567–574. [1997].
93. Smith, TK. Nutrition: its relationship to orthopedic infections. Orthop Clin North Am. 1991;22:373–377.
94. Song, KM, Sloboda, JF. Acute hematogenous osteomyelitis in children. J Am Acad Orthop Surg. 2001;9(3):166–175.
95. Stein, Z. Silicone breast implants: Epidemiological evidence of sequelae. Am J Public Health. 1999;89(4):484–487.
96. Stengel, D, Bauwens, K, Sehouli, J, et al. Systematic review and meta-analysis of antibiotic therapy for bone and joint infections. Lancet Infect Dis. 2001;1(3):175–188.
97. Strickland, E, et al. In vivo acetabular contact pressures during rehabilitation. I. Acute phase. Phys Ther. 1992;72:691–699.
98. Strong, M, et al. Sequelae from septic arthritis of the knee during the first two years of life. J Pediatr Orthop. 1994;14:745–751.
99. Studahl, M, et al. Septic arthritis of the knee: a 10 year review and long-term follow-up using a new scoring system. Scand J Infect Dis. 1994;26:85–93.
100. Tavakoli, M, et al. Diagnosis and management of osteomyelitis: decision, analytic, and pharmacoeconomic considerations. Pharmacoeconomics. 1999;16(6):627–647.
101. Tay, BK, Deckey, J, Hu, SS. Spinal infections. J Am Acad Orthop Surg. 2002;10(3):188–197.
102. Trampuz, A. Molecular and antibiofilm approaches to prosthetic joint infection. Clin Orthopaedics. 2003;414:69–88.
103. Trampuz, A, Widmer, AF. Hand hygiene: a frequently missed lifesaving opportunity during patient care. Mayo Clin Proc. 2004;79(1):109–116.
104. Trampuz, A, Zimmerli, W. New strategies for the treatment of infections associated with prosthetic joints. Curr Opin Investig Drugs. 2005;6(2):185–190.
105. Trampuz, A, Zimmerli, W. Prosthetic joint infections: update in diagnosis and treatment. Swiss Med Wkly. 2005;135(17-18):243–251.
106. Vicente, AG, Diagnosis of orthopedic infection in clinical practice using Tc-99m sulesomab (antigranulocyte monoclonal antibody fragment). Clin Nucl Med. 2004(12):781–785.
107. Volin, SJ, Hinrichs, SH, Garvin, KL. Two-stage reimplantation of total joint infections: a comparison of resistant and non-resistant organisms. Clin Orthop Relat Res. 2004;427:94–100.
108. Warrell, DA. Clinical challenge of venomous bites and stings in the Asia-Pacific area. Toxicon. 1997;35(4):474.
109. Yao, DC, Sartoris, DJ. Musculoskeletal tuberculosis. Radiol Clin North Am. 1995;33:679–689.
110. Zimmerli, W. Prosthetic joint infections. N Engl J Med. 2004;351(16):1645–1652.
111. Zinberg, E. Chronic osteomyelitis. In Nickel V, Botte M, eds.: Orthopaedic rehabilitation, ed 2, New York: Churchill Livingstone, 1992.