Chapter 6 Patients’ problems, physiotherapy management and outcome measures
INTRODUCTION
This chapter discusses the problems commonly encountered by the physiotherapist when working with patients who have respiratory or cardiovascular dysfunction. The patient problems identified in this chapter are those developed following assessment of the patient and are most likely to respond to physiotherapy treatment. This chapter will assist the physiotherapist to utilize clinical reasoning skills by linking and interpreting information (subjective and objective findings) to develop an analysis that is based on the patient’s problems (Chapter 1). The presence of pathology affecting the respiratory and cardiovascular systems affects normal physiological functioning and the signs and symptoms produced are the clinical manifestations of this pathophysiology. The physiotherapist therefore requires a thorough knowledge of normal physiology as well as the pathology and pathophysiology of the respiratory and cardiovascular systems. In addition, an understanding is required of the possible sequelae of the pathological process, the clinical presentations of the disorder(s), the likely impairments, activity limitations and participation restrictions, impact on quality of life (QoL) and the anticipated prognosis for the patient.
Patient assessment, problem solving and physiotherapy management should be tailored to the individual. Individuals with common problems, such as dyspnoea and reduced exercise tolerance, resulting from respiratory or cardiovascular disease may be managed in groups, for example, in cardiopulmonary rehabilitation programmes.
PROBLEM SOLVING
The key to the effective physiotherapy management of a patient is the accurate identification of the patient’s problems. The assessment will reveal clinical features that the physiotherapist considers important and these are used to determine the patient’s main problem(s). The problems commonly encountered are:
 musculoskeletal dysfunction - postural abnormalities, decreased compliance or deformity of the chest wall.
musculoskeletal dysfunction - postural abnormalities, decreased compliance or deformity of the chest wall.The patient problems identified in this chapter are particular to the physiotherapist and are not based on pathologies or derived in the same manner as the medical problem list. For example, a patient may be admitted to hospital with a diagnosis of chest infection associated with excess sputum production, cough and fever. The physiotherapist is not able to treat infection per se but is able to manage the problem of impaired airway clearance. In order to determine the patient’s problem(s) it is essential to identify the significant information gained from the subjective and objective examination. For example, increased production of sputum may be reported by the patient or objectively inspected and measured. Colonization of mucus, evident from microculture, and coarse inspiratory crackles on auscultation are common clinical features that indicate impaired airway clearance.
Some clinical features may provide evidence for a number of patient problems; for example, reduced chest expansion may be a feature of airflow limitation as a result of lung hyperinflation or reduced lung volume (e.g. resulting from significant atelectasis or interstitial lung disease (ILD)). It is necessary to make a judgement about the clinical features collectively to determine which problem(s) they provide evidence for. Once the problem(s) has been identified, the physiotherapist needs to consider the likely pathophysiological basis for the problem(s) so that an appropriate intervention plan can be determined. Patients often present with more than one problem that is amenable to physiotherapy. In this situation, the intervention plan should focus on strategies that address as many patient problems as possible, using best evidence and practice, and should be determined in collaboration with the individual and with a focus on self-management where appropriate.
Some of the clinical features revealed during the assessment may not be features of any of the cardiopulmonary problems listed in this chapter (e.g. poor self-management skills, the presence of risk factors for postoperative pulmonary complications (PPCs) such as cigarette smoking, incorrect use of an inhaler or immobility in the early postoperative period). Patient assessment will also identify the presence of any important factors that must be considered when applying the principles of physiotherapy management for a particular problem to an individual patient. Examples of such factors include the presence of comorbid conditions (e.g. diabetes mellitus, osteoporosis), psychosocial barriers or special communication needs (e.g. language or cultural requirements).
Case studies
The following case studies provide examples of how to formulate a patient problem list. The reasoning for identifying each problem and the considerations for patient intervention are given. Assessment of the patient may reveal many features. The clinical features reported in these case studies include only those considered to be important in formulating the problem list for each case.
CASE STUDY 6.1
A 57-year-old woman is admitted to a tertiary hospital via the emergency department.
History of presenting condition
Has been feeling unwell for 4 days with increasing cough and breathlessness and difficulty clearing her sputum.
Chronic obstructive pulmonary disease (COPD)
Gastro-oesphageal reflux disorder (GORD)
Osteoarthritis affecting both knees.
Long-acting anticholinergic and combination therapy (long-acting β2−agonist and corticosteroid) administered via a dry powder inhaler
Angiotensin-converting enzyme (ACE) inhibitor for hypertension
Proton pump inhibitor for GORD
Statin for raised cholesterol.
Lives at home with her husband but is finding it increasingly difficult to manage
Independent in self-care when her condition is stable (e.g. showering, dressing), but now requires significant help from her husband.
Pulmonary function tests were performed at a recent clinic visit at a time when her condition was stable Table 6.1.
Current medical investigations
Respiratory rate 28 breaths/min
Chest radiograph reveals hyperinflation of lungs with an increase in the retrosternal space; low, flattened diaphragms; hyperlucent lung fields with paucity of vascular markings in the periphery but prominent hilar markings and narrow heart silhouette
Arterial blood gas results on admission: pH 7.28, PaO2 8.7 kPa (65 mmHg), PaCO2 9.3 kPa (70 mmHg), HCO3− 29mmol/l, base excess −1 and SaO2 92% on 28% oxygen via a Venturi oxygen mask
Sputum microculture result Pseudomonas aeruginosa.
Physiotherapy subjective examination
Normally has a productive cough. Presently coughing throughout the day especially in the morning and when moving about the bed
Sputum is usually scant and clear. Over last week sputum has become yellow and cough is productive all day
Reports being progressively more short of breath over the last 5 years and now reports being breathless when mobilizing around the bed. Sleeps with two pillows at night to relieve breathlessness and symptoms of heartburn
Although limited by breathlessness, patient normally able to walk about 150 metres and is independent in self-care when condition is stable (e.g. showering, dressing) but now requires significant help and is breathless walking to the toilet
Smoked 25 cigarettes per day for 30 years (ceased 10 years ago).
Physiotherapy objective examination
Patient appears thin and frail
Barrel-shaped chest with increased anteroposterior diameter and thoracic kyphosis
Obvious respiratory distress with prominent use of accessory muscles, elevated shoulder girdle and intercostal recession. Increased inspiratory : expiratory (I : E) ratio (prolonged expiration)
Increased use of abdominal muscles during expiration
Using pursed-lip breathing (PLB) during conversation
Chest expansion symmetrical and poor in all zones
Auscultation reveals decreased breath sounds with coarse inspiratory crackles in lower lobes and a prolonged expiratory phase and generalized expiratory wheeze
Sputum productive of 20 ml thick, purulent mucus (P2) expectorated in past 12 hours
Cough effective and tight but having significant difficulty expectorating sputum Table 6.2.
Table 6.1 Pulmonary function tests, case study 1
| Test | Observed | Predicted |
|---|---|---|
| FVC (l) | 2.76 | 2.90 |
| FEV1 (l) | 1.04 | 2.30 |
| FEV1/FVC (%) | 38.00 | 77.00 |
| FRC (l) | 4.67 | 2.62 |
| RV (l) | 3.24 | 1.96 |
| TLC (l) | 6.16 | 5.02 |
| TLCO (mmol/min/kPa) | 4.16 | 7.59 |
FVC, forced vital capacity; FEV1, forced expiratory volume in 1 second; FRC, functional residual capacity; RV, residual volume; TLC, total lung capacity; TLCO, transfer factor of the lung for carbon monoxide
Table 6.2 Analysis of problems from case study 1
Note: respiratory muscle dysfunction may be present (clinical features include orthopnoea, CO2 retention and altered breathing pattern) and further assessment may be warranted
FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; TLC, total lung capacity; FRC, functional residual capacity; PLB, pursed-lip breathing; TLCO, transfer factor of the lung for carbon monoxide; GORD, gastro-oesophageal reflux disorder; 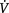 /
/ , ventilation/perfusion ratio; VT, tidal volume
, ventilation/perfusion ratio; VT, tidal volume
CASE STUDY 6.2
A 65-year-old man was admitted to hospital for emergency surgery to treat an acute perforated duodenal ulcer. A suture and omentoplasty were performed via a midline upper abdominal incision. On the third postoperative day the patient’s condition deteriorated and assessment revealed the following features.
History of presenting condition
Sudden onset of abdominal pain and vomiting.
Diabetes mellitus controlled with diet
Ischaemic heart disease (IHD).
Home medications: aspirin, nitrate, ACE inhibitor, β−adrenoreceptor blocker, statin and diuretic
Current medications: postoperative pain managed with an opioid (morphine) administered via patient-controlled analgesia (PCA), antiemetic for nausea.
Lives at home with wife and is independent
Smoked 20 cigarettes a day for 40 years (ceased 2 years ago).
Current medical investigations
Respiratory rate 28 breaths/min
Nasogastric tube aspirate 300 ml over 24 hours.
Urine output 1700 ml over 24 hours
White blood cell count (WCC) 14 × 109/l
Chest radiograph revealed right middle and lower lobe infiltrate consistent with collapse and consolidation
Arterial blood gas results: pH 7.48, PaO2 7.8 kPa (59 mmHg), PaCO2 4.5 kPa (34 mmHg) and SaO2 91% on room air.
Physiotherapy subjective examination
Reports that he has coughed very little overnight and today
No sputum produced either overnight or today
Only ambulating with assistance, as a result of persistent nausea
Reports that he forgets to use the PCA.
Physiotherapy objective examination
Appears ill and is pale and clammy
Chest expansion poor lower zones: right < left
Abdominal splinting on inspiration
Cough is painful, weak, moist and ineffective
Auscultation reveals decreased breath sounds left lower lobe, right middle and lower lobes
Table 6.3 Analysis of problems from case study 2
| Current cardiopulmonary problems | Evidence for each problem based on clinical features | Most likely pathophysiological basis for each problem |
|---|---|---|
| Reduced lung volume | Atelectasis resulting from marked decrease in FRC, postoperative diaphragmatic dysfunction, reduced function of surfactant, airway obstruction from mucus plugging and abdominal incisional pain | |
| Impaired gas exchange | Acute hypoxaemia: PaO2 7.8 kPa (59 mmHg) and SaO2 91% on room air | Mixed causes for hypoxaemia including 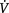 / / mismatch caused by a decrease in FRC mismatch caused by a decrease in FRC |
| Impaired airway clearance | Patient is febrile, elevated WCC, moist cough | Possible colonization of sputum, possible systemic dehydration, impaired MCC from opioid, ineffective cough |
| Considerations for treatment | ||
FRC, functional residual capacity; MCC, mucociliary clearance; WCC, white blood cell count; PPC, postoperative pulmonary complication; IHD, ischaemic heart disease; PCA, patient-controlled analgesia, 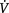 /
/ , ventilation/perfusion ratio; PaO2, partial pressure of oxygen in arterial blood; SaO2, arterial oxygen saturation
, ventilation/perfusion ratio; PaO2, partial pressure of oxygen in arterial blood; SaO2, arterial oxygen saturation
The problem-solving approach to determining patient management not only assists with the identification of existing problems but also enables recognition of potential patient problems. For example, a high-risk surgical patient will develop reduced lung volume and has the added potential to develop problems of impaired airway clearance but if active treatment, such as early ambulation, is started during the at-risk period, these problems may be prevented. Some problems are not amenable to physiotherapy intervention or physiotherapy intervention may be detrimental.
For the patient with more than one problem, it is essential to prioritize the problem list and to establish the short and long-term goals of the patient and, where appropriate, their relatives and/or caregiver and any other service providers (e.g. other allied health professionals, nurses and medical practitioners). Some problems may only be short term, for example, reduced lung volume in the immediate postoperative period. Developing and prioritizing the problem list and developing the intervention should, whenever possible, take place in consultation with the patient. Some interventions may be determined by taking into consideration other factors such as the availability of resources and the model of service delivery.
Once the short- and long-term goals of the patient and, where appropriate, their relatives and/or caregiver and any other service providers have been established, the next stage is to identify the means of achieving these goals through physiotherapy intervention and the time frame over which they are to be achieved. The appropriate intervention requires selecting the optimal physiotherapy management strategy and this should be evidence based where possible. When a patient has several physiotherapy problems, the physiotherapy techniques selected should ideally address more than one of the high-priority problems. When selecting a treatment approach, the potential risks to the patient (e.g. the possibility of causing adverse physiological responses) and methods to minimize such risks must be taken into consideration. Other factors to be considered include ensuring that the intervention is appropriate for the patient’s age, occupation, ability to communicate, cultural beliefs, level of understanding and motivation and the presence of any psychosocial factors which may interfere with the treatment approach (e.g. fear, anxiety or depression). It is important also to determine the patient’s likes and dislikes; for example, patient preferences for types of activities are vital considerations when developing an exercise programme.
Common to the management of most problems is the education of the patient by the physiotherapist. This is essential to ensure that the patient takes responsibility for their own management and becomes actively involved in the management of their problem and the prevention of associated problems. If the problem is amenable to physiotherapy, treatment should be started. Conditions that are not amenable to physiotherapy intervention or that require the expertise of a specialist physiotherapist (e.g. a physiotherapist specializing in continence problems) should be referred appropriately. With some problems, a stage will be reached when the natural rate of recovery will no longer be augmented by physiotherapy intervention and treatment should then be discontinued.
The selection and use of appropriate outcome measures are fundamental to the evaluation of physiotherapy intervention. Healthcare fundholders increasingly require data demonstrating the effects of physiotherapy intervention, using instruments that are reliable and valid. Other parties requiring outcome data include the patient, the patient’s relatives and caregivers, employers of physiotherapists, clinicians, patient support groups and associations of patients with particular conditions (e.g. cystic fibrosis, heart failure), members of other healthcare professions and insurers. Thus, when selecting which outcome data to monitor it is important to consider the relevant stakeholders.
In this chapter the problems commonly encountered by the physiotherapist when managing patients with respiratory or cardiovascular dysfunction are discussed. The underlying pathophysiology for each problem is outlined and the clinical features that assist in the identification of the problem are described. The physiotherapy management is listed alongside each problem and discussed in greater detail in other chapters. The discussion of each problem concludes with guidelines for the choice of clinical outcome measures to be used to evaluate physiotherapy intervention.
PROBLEM - IMPAIRED AIRWAY CLEARANCE
Impaired airway clearance is an important physiotherapy problem because of the potential for the patient to develop an overwhelming infection, major atelectasis and other associated problems such as impaired gas exchange and airflow limitation. Further, untreated persistent infections may predispose to the development of chronic lung disease such as bronchiectasis.
Normal airway clearance depends upon two mechanisms - mucociliary clearance (MCC) and effective cough. Alveolar clearance may also contribute to the clearance of secretions from the peripheral airways (Houtmeyers et al 1999a).
Secretions and debris in the small airways are transported toward the large airways by the mucociliary blanket or escalator and eventually swallowed or cleared by a cough. The mucociliary escalator consists of cilia and a mucus layer. Impurities are caught in the mucus layer and the cilia beat synchronously to move the mucus towards the upper airway. In health, airway mucus is composed mostly of water and the daily volume of mucus in a healthy adult is up to 100 ml (Clarke 1990).
When the volume of secretions reaching the larynx and pharynx has increased to the extent that an individual becomes conscious of the presence of secretions on coughing or ‘clearing the throat’ the mucus is defined as sputum; the presence of sputum is abnormal.
While mucociliary transport is the major mechanism for clearing secretions in healthy subjects, cough is an important mechanism, especially in people with lung disease. The effectiveness of a cough is related to the volume and viscosity of secretions and the velocity of airflow through the airway lumen. An effective cough requires a high flow rate and a small cross-sectional area of the airway. Dynamic compression of the airways starts downstream from the equal pressure point (Chapter 5) where intraluminal and extraluminal pressures around the bronchial wall are equal (Irwin & Widdicombe 2000). This compression will increase airflow velocity by decreasing the cross-sectional diameter of the airways.
Vigorous coughing can cause a number of adverse effects including abnormal cardiovascular responses (e.g. systemic hypotension and hypertension, rhythm disturbances), abnormalities of the genitourinary tract (e.g. urinary incontinence), gastrointestinal symptoms (e.g. gastro-oesophageal reflux, inguinal hernia), musculoskeletal problems (e.g. rupture of rectus abdominis, rib fractures), neurologic features (e.g. cough syncope, headache, stroke, seizures) and respiratory complications (e.g. airflow limitation, laryngeal trauma, pneumothorax, tracheobronchial trauma). These effects are largely due to the high intrathoracic pressures and expiratory velocities associated with vigorous coughing (Irwin & Widdicombe 2000).
Abnormalities in the normal airway clearance system (i.e. MCC and cough) will result in an accumulation of secretions causing airway obstruction and possibly lead to atelectasis. The subsequent inhomogeneity of ventilation may adversely affect gas exchange. Airway obstruction and the presence of excess secretions also increase the risk of infection. Inflammatory responses to infection cause the release of chemical mediators such as proteases and elastases that can destroy the airway epithelium. This leads to unstable, overcompliant airways that contribute to impaired airway clearance (Barker 2002).
Table 6.4 lists the pathophysiological basis of impaired airway clearance and includes clinical examples (Clarke 1990, Houtmeyers et al 1999a, Irwin & Widdicombe 2000).
Table 6.4 Pathological basis of impaired airway clearance and clinical examples
| Pathophysiological basis | Comment and clinical examples | |
|---|---|---|
| Increased or altered composition of mucus | Bronchiectasis, chronic bronchitis, cystic fibrosis, asthma, pneumonia Presence of an artificial airway increases mucus secretion Changes viscosity and increases amount of secretions, thereby slowing MCC Leads to viscous secretions which are difficult to mobilize and expectorate May occur postoperatively if fluid restriction imposed Excess fluid loss due to prolonged very high respiratory rate |
|
| Abnormalities in cilial structure or function | Primary ciliary dyskinesia Damage to ciliated epithelium from excessive endotracheal suctioning |
|
| Impaired MCC | Rate of MCC decreases with age Decreases MCC e.g. Tobacco smoke, NOx – may decrease MCC Some general anaesthetics and narcotics depress MCC May cause a loss of ciliated epithelium causing mucus retention and slowing MCC Slows MCC Coughing and expectoration may be avoided due to embarrassment |
|
| Abnormal cough reflex | 1. Decreased | Decreased level of consciousness, general anaesthesia, narcotic analgesics Inhibition due to pain, e.g. postoperatively, chest trauma, pleurisy Damage to vagal or glossopharyngeal nerves Laryngectomy Paralysed vocal cords Denervated lungs (heart-lung or lung transplantation) |
| 2. Increased | Bronchial hyperreactivity Poorly controlled asthma Viral infections may increase sensitivity |
|
| Ineffective cough due to the inability to generate sufficient expiratory flow | Severe reduction in VC Respiratory muscle weakness Airflow limitation may cause cough to be weak and/or ineffective Decreased airflow through dilated bronchiectatic airways |
|
| Abnormal cough | Stimulates the cough reflex May lead to chronic cough and microaspirations of gastric contents |
MCC, mucociliary clearance; NOx nitrogen oxides; VC, vital capacity; GORD, gastro-oesophageal reflux disorder
Special case - postoperative patient with impaired airway clearance
Many factors either present preoperatively or arising in the perior postoperative period increase mucus secretion and/or impair MCC and may be responsible for the development of PPC. It is therefore important for the physiotherapist to identify patients who have an increased risk of developing PPCs (Chapter 12).
Clinical features
The clinical features of impaired airway clearance are usually those resulting from excess or retained secretions. The history of usual daily sputum production obtained from the patient may reveal a chronic productive cough. Changes in the normal pattern of sputum production, such as an increase in the amount or a change in the colour or consistency of the sputum, are likely. Some patients report difficulty expectorating secretions. Further questioning may reveal an increase in the number of chest infections or hospitalizations for their illness compared with previous years. Patients with chronic lung disease may report signs of stress incontinence on coughing.
Examination of the patient may reveal an altered breathing pattern due to increased work of breathing (WOB). The presence of infection may produce fever and tachycardia. When the secretions cause marked airflow limitation, wheezing may be audible (see Problem - airflow limitation). Auscultatory findings include diminished or absent breath sounds, bronchial breath sounds, crackles or wheezes. The cough may be moist or dry and hacking, effective and productive or ineffective and weak. Some patients have a paroxysmal cough with associated adverse effects such as dizziness, syncope or exhaustion. The examination of any sputum expectorated may reveal an increase in the volume or weight compared with the patient’s normal expectorant. The colour of the sputum may have changed to yellow, green or brown and there may be blood present (haemoptysis). Also the consistency of the sputum may have altered and microculture may reveal colonization (e.g. with bacteria) (Chapter 1).
Chest radiographs sometimes show signs of lung collapse and/or consolidation or abnormalities reflecting the underlying disease process; for example bronchiectatic changes.
The clinical features of impaired airway clearance in the postoperative patient may include an increased volume of sputum expectorated compared with the patient’s usual expectorant; a weak, ineffective moist cough; possible bacterial contamination of expectorated sputum; fever; and chest radiographic changes consistent with atelectasis or pneumonia (Chapter 12).
Physiotherapy management
Physiotherapy is an integral part of the management of patients with impaired airway clearance but bronchial secretions only become a physiotherapy problem when they are excessive, retained or difficult to eliminate. Some patients expectorate a small amount of foul-smelling, tenacious sputum postoperatively but this is not a problem if the patient is conscious, able to cough effectively and self-ambulating.
Airway clearance techniques comprise a range of physiotherapy interventions used for the management of impaired airway clearance (Chapter 5). These techniques aim to promote clearance of excessive secretions from the distal airways and thereby prevent the consequences of obstruction and thus improve ventilation homogeneity and gas exchange. Airway clearance techniques may incorporate positive pressure or oscillation applied at the mouth or chest wall (manual or mechanical) and/or breathing strategies to aid the movement of secretions to the central airways. From the central airways, forced expiratory manoeuvres such as coughing or huffing are used to facilitate expectoration. Such manoeuvres aim to use high expiratory flow rates to shear secretions from the airway walls.
The physiotherapy management of impaired airway clearance is influenced by the underlying cause and acuity of the patient’s condition. For patients with chronic hypersecretory lung disease who regularly produce excess bronchial secretions, the use of daily airway clearance techniques is recommended (Jones & Rowe 1998, van der Schans et al 2000). The rationale for daily treatment is to reduce stagnation of secretions in an attempt to avoid contamination with pathogens and thereby reduce the destruction of airway walls caused by the inflammatory response. This may slow the cycle of progressive tissue damage. The physiotherapist’s role in this case is to prescribe and teach a daily airway clearance regimen that is individually tailored and acceptable to the patient. Factors to be considered when choosing an airway clearance technique include:
The physiotherapist’s role may change in a patient with hypersecretory lung disease when the patient experiences a worsening of their condition such as during a chest infection. During a hospital admission for an acute illness the physiotherapist may take a more active role and the frequency and duration of treatments may increase. It may be that a change of technique is indicated and it is the physiotherapist’s role, in consultation with the patient, to select a technique that addresses the changing condition.
A number of additional measures are available that have been shown to enhance or improve airway clearance. Table 6.5 outlines these measures (Conway et al 1992, Elkins et al 2006, Houtmeyers et al 1999b, Jones et al 2003, Wark et al 2005, Wills & Greenstone 2006).
Table 6.5 Additional measures to enhance airway clearance
| Measure | Examples and uses |
|---|---|
| Humidification | |
| Nebulization | MCC may be improved by hypertonic saline, amiloride, recombinant human deoxyribonuclease and β-adrenergic agonists |
| Analgesia | Patients in whom pain is inhibiting an effective cough |
| Physical activity | Increased respiratory rate and VT increase expiratory flow rates and sputum clearance |
NIV, non-invasive ventilation; MCC, mucociliary clearance; VT, tidal volume
In the postoperative patient it is essential to establish whether the patient has excess secretions and whether they have difficulty managing their own airway clearance. This is one of the factors influencing the risk of the patient developing PPCs. The techniques to assist sputum clearance in the postoperative patient aim to increase alveolar ventilation and expiratory flow rates using, for example, upright positioning and ambulation at an adequate intensity with encouragement to take deep breaths. Expectoration of secretions can be facilitated by supported coughing or huffing (Chapter 12). If the patient has large amounts of secretions, techniques such as the active cycle of breathing techniques (ACBT) and vibrations may be used.
For patients who are reluctant or unable to cough, a spontaneous cough may be elicited by physical activity or a change of position. The cough reflex may be elicited using a tracheal rub or suctioning. Strengthening of the abdominal muscles and assisted cough techniques (e.g. abdominal support with an upward pressure) or a mechanical insufflation- exsufflation device may be helpful for patients with impaired cough due to weakness of the abdominal muscles (Chapter 16). For the intubated and ventilated patient, improved alveolar ventilation and increased expiratory flow rates can be achieved by positioning and manual hyperinflation, and secretions cleared by suctioning (Chapter 8).
Outcome measures
Short-term outcomes can be monitored by a change in sputum expectorated, as measured by weight, volume or rate of expectoration. Ease of sputum expectoration can be measured using a categorical scale or visual analogue scale (VAS). In acute conditions, chest radiographs and auscultatory findings may provide evidence for a change in the patient’s condition. Radio-aerosol clearance may be used as an outcome measure in studies of airway clearance techniques (van der Schans et al 1999).
Long-term outcomes in patients with hypersecretory lung disease may be assessed by the number of exacerbations, courses of antibiotics, hospitalizations and days lost from work/study per year. Quality of life scales, such as the St George’s Respiratory Disease Questionnaire (SGRQ), include a section that quantifies symptoms of cough and sputum (Jones et al 1992). Pulmonary function, in particular spirometry, has been used to evaluate the effects of airway clearance techniques but may be relatively insensitive to the intervention (van der Schans et al 1999).
In the high-risk postoperative patient with excess bronchial secretions, outcomes from physiotherapy intervention may be measured by the prevention of PPCs.
Improved cough or huff technique may be associated with a reduction in associated problems such as fatigue, dyspnoea, syncope, airflow limitation, arterial oxygen desaturation or stress incontinence.
PROBLEM - DYSPNOEA
Dyspnoea is the term generally applied to the sensations experienced by individuals complaining of unpleasant or uncomfortable respiration (Ambrosino & Scano 2001). In clinical practice, the terms breathlessness and dyspnoea are used interchangeably. However ‘breathlessness’ is one of many descriptors used by patients to convey their experience of dyspnoea. Other common terms used by patients suggest unrewarded inspiration (i.e. ‘can’t get the air in’) and chest tightness. It is possible these different descriptors originate from different pathological processes. For example, individuals with COPD frequently use terms that reflect an increase in the effort of breathing or WOB (Scano et al 2005).
Dyspnoea is a common and distressing symptom experienced by patients with respiratory and cardiovascular disease and is frequently the symptom that causes the patient to seek medical care. On occasions, it may be difficult to distinguish from the patient’s account whether the symptoms are of respiratory or cardiovascular origin as in both the patient may report breathlessness on exertion, when lying supine, causing waking during the night and acute episodes of breathlessness at rest.
Many healthy individuals become aware of their breathing when exercising at a moderate or high intensity and report that their breathing is rapid and that they are puffing. These changes in breathing reflect the increased ventilation required during exercise and are appropriate for the situation. In contrast, individuals with respiratory or cardiovascular disease may become aware of unpleasant breathing sensations at very low levels of physical activity and even at rest or in response to emotional or stressful situations. In such situations the appropriate term for these respiratory sensations is dyspnoea. Dyspnoea is not tachypnoea, hyperventilation or hyperpnoea. These three terms all describe ventilation in response to different stimuli and may represent normal physiological responses. Although hypoxaemia and hypercapnia increase ventilatory response, the severity of hypoxaemia and hypercapnia are not directly linked to the perception of dyspnoea. The sensation of dyspnoea appears to originate with the activation of sensory systems within the lung, chest wall and respiratory muscles that give rise to an awareness of breathing discomfort (American Thoracic Society (ATS) 1999, Schwartzstein & Parker 2006).
The sensation of dyspnoea is influenced by many factors including the patient’s psychological status and their experience and memory. The presence of fear, anxiety, depression and anger heighten the perception of dyspnoea (ATS 1999). In some patients, dyspnoea may be perceived as life threatening. A patient’s ability to describe and quantify the unpleasant sensation of dyspnoea is also very variable. This is not unlike the variability seen when patients report pain. These factors may in part explain why the intensity of dyspnoea for a given level of impairment in lung function, or exercise capacity, can vary greatly among individuals.
Although it is generally contended that dyspnoea arises as a consequence of multiple complex and varied interactions, the precise mechanisms responsible for the sensation of dyspnoea are poorly understood and the management of dyspnoea poses considerable difficulties. Clinically, dyspnoea results from several different pathophysiological mechanisms and in some patients more than one mechanism will be responsible (Table 6.6) (ATS 1999, Scano et al 2005, Schwartzstein & Parker 2006).
Table 6.6 Pathophysiological basis for dyspnoea in respiratory and cardiovascular disease and clinical examples
| Pathophysiological basis | Clinical examples | |
|---|---|---|
| 1. Increase in elastic load due to: | a. Decrease in lung compliance | Increases the inspiratory muscle work required to overcome the elastic recoil of the lungs. Increases in 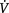 E are achieved mainly by increasing respiratory rate, e.g. ILD, breathing at low lung volumes, pulmonary congestion E are achieved mainly by increasing respiratory rate, e.g. ILD, breathing at low lung volumes, pulmonary congestionHyperinflation (e.g. COPD, cystic fibrosis, asthma) increases the WOB |
| b. Decrease in chest wall compliance and /or compliance of the abdominal compartment | Obesity, kyphoscoliosis, ankylosing spondylitis | |
| 2. Increase in airways resistance | Increases expiratory muscle work to effect airflow through narrowed airways (e.g. COPD, asthma) | |
| 3. Weakness or fatigue of the respiratory muscles | See Problem – respiratory muscle dysfunction | |
| 4. Increase in metabolic rate | Increases ventilatory requirements, e.g. fever, exercise | |
| 5. Low cardiac output / ischaemia | Inadequate cardiac output causes reflex medullary ventilatory stimulation when the oxygen supply to the exercising muscle is inadequate to meet metabolic needs, e.g. IHD, heart failure or in the presence of ventricular arrhythmias, valvular problems or cardiomyopathy | |
| 6. Blood gas abnormalities | Hypoxaemia or hypercapnia | |
| 7. Deconditioning | Lactate accumulates at low levels of exercise causing an increase in ventilation | |
| 8. Anaemia | When severe causes dyspnoea on exertion | |
| 9. Acute changes in permeability of pulmonary capillaries | Pulmonary oedema | |
| 10. Perfusion limitation | The presence of a large 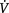 / / mismatch or shunt invariably causes dyspnoea, e.g. pulmonary embolus, pulmonary infarction, cyanotic heart disease, pulmonary congestion mismatch or shunt invariably causes dyspnoea, e.g. pulmonary embolus, pulmonary infarction, cyanotic heart disease, pulmonary congestion |
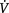 E, minute ventilation; ILD, interstitial lung disease; COPD, chronic obstructive pulmonary disease; WOB, work of breathing;
E, minute ventilation; ILD, interstitial lung disease; COPD, chronic obstructive pulmonary disease; WOB, work of breathing; 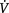 /
/ , ventilation/perfusion ratio
, ventilation/perfusion ratio
Special case - chronic lung disease
There are several pathophysiological causes of dyspnoea in patients with chronic lung disease. In patients with moderate to severe COPD, the increase in airway resistance is associated with lung hyperinflation and gives rise to an increase in the WOB (see Problem - airflow limitation). In patients with ILD, a greater than normal inspiratory effort is required to overcome the increased lung elastic recoil and may give rise to dyspnoea. Peripheral muscle dysfunction and deconditioning are common in patients with chronic lung disease and the associated increase in lactic acid accumulation during submaximal levels of exercise stimulates ventilation. Respiratory muscle dysfunction may also be a contributory factor to dyspnoea especially in patients with COPD (see Problem - respiratory muscle dysfunction). The presence of hypoxaemia may contribute to the WOB by stimulating ventilation. Psychosocial factors (e.g. anxiety) may heighten the perception of dyspnoea.
Some patients with moderate or severe disease, especially those with COPD, report marked dyspnoea when performing activities of daily living (ADL) that involve the use of the upper limbs, especially when the upper limbs are unsupported. Performing activities that involve unsupported upper limb movements leads to a loss of these arm trunk muscles as elevators of the rib cage, thereby reducing their contribution to the generation of the intrapleural pressure needed for inspiration. The breathing pattern during unsupported upper limb exercise in patients who report dyspnoea with upper limb movements is often rapid and irregular, and dyssynchronous thoraco-abdominal movements and breath holding may occur (Celli 1994). A further workload is imposed when activities involve raising the arms above the head. This arm position gives rise to the early onset of lactate accumulation in the upper limbs leading to an increase in carbon dioxide (CO2) production, which stimulates ventilation.
Clinical features
The time course for the onset of dyspnoea (e.g. acute vs insidious) gives important information as to the likely aetiology and is obtained from the subjective assessment. Most commonly, the patient’s account will reveal that dyspnoea is elicited by physical activity, for example during ADL or when walking on the flat or up inclines. The patient may also report that adopting certain body positions causes dyspnoea, for example, when attempting to lie flat to sleep. Many patients with chronic lung disease will report day-to-day variability in dyspnoea intensity and this may include variation depending on the time of day and climatic conditions. Extremes of temperature and humidity and high levels of atmospheric pollution tend to heighten the perception of dyspnoea in most individuals. In chronic hyperventilation disorder, dyspnoea may occur at rest and is often accompanied by an excessive frequency of sighs (Chapter 17).
Some patients may seek medical care when they become very breathless playing sports, for example golf or bowls, but many individuals attribute breathlessness to ageing or a poor level of fitness and fail to seek help until breathlessness occurs during ADL. Patients with respiratory disease or heart failure may also report feeling fatigued; this may be a generalized symptom or felt predominantly in the legs during physical activity.
On examination, the patient may display an altered breathing pattern. Abnormalities in the rate and depth of breathing, inspiratory to expiratory ratio and symmetry of chest movements are often observed. Clinical features indicative of an increase in the WOB associated with airflow limitation include the adoption of an upper chest breathing pattern with shoulder girdle fixation to enable the accessory muscles to assist with inspiration. Other signs commonly seen in patients with severe airflow limitation who report dyspnoea include an increase in abdominal effort during expiration, paradoxical breathing and PLB. Characteristically, expiratory time will be prolonged in the presence of airflow limitation (see Problem - airflow limitation). The patient may complain of feeling hot and appear sweaty if the WOB is excessive and dyspnoea is associated with fear or panic.
Assessment may reveal signs and symptoms of other problems, most commonly airflow limitation, impaired gas exchange and, in patients with cardiovascular disease, angina may accompany dyspnoea. Exercise tolerance will usually be reduced as a result of dyspnoea on exertion. The chest radiograph will often show signs consistent with the underlying pathology (e.g. lung hyperinflation, effusion, pneumothorax, areas of collapse or consolidation). A laboratory-based incremental exercise test with continuous measurement of ventilatory and cardiovascular variables can be used to differentiate between dyspnoea arising from cardiovascular or respiratory origin. If respiratory muscle weakness is suspected to be a contributing factor this can be confirmed by measuring maximum inspiratory and expiratory mouth pressures (PiMax and PeMax) (Chapter 3 and see Problem - respiratory muscle dysfunction).
Special case - problems with bladder and bowel function
The problem of urinary or faecal incontinence may arise in the presence of dyspnoea, decreased exercise tolerance or impaired airway clearance (White et al 2000). Patients who are breathless are often sedentary and have a reduced appetite and fluid intake, and difficulty preparing food. The breathless patient who is also constipated may have difficulty breath holding and assuming an adequate position to enable defecation (Markwell & Sapsford 1995). Reduced fluid intake and frequent or ‘just in case’ toileting, to prevent stress or urge urinary incontinence, can result in the bladder becoming accustomed to accommodating smaller volumes of urine. This leads to an increased frequency to void. Dyspnoea is likely to intensify when attempting to walk quickly to reach the toilet in time and may also become worse with functional tasks requiring the upper limbs, such as undressing, making urgency worse or resulting in incontinence.
Physiotherapy management
In patients with airflow limitation, bronchodilators may reduce dyspnoea and improve exercise tolerance; therefore, inhaler technique and timing of bronchodilators should be optimized.
Positioning, breathing control and relaxation techniques are used in an attempt to decrease the WOB and eliminate unnecessary muscular activity (Gosselink 2004, O’Neill & McCarthy 1983). Positions such as the forward lean position with the arms supported or high side lying may be useful for patients who are severely distressed, especially when the cause of their breathlessness is COPD. In these patients, the forward lean position increases transdiaphragmatic pressure, improves thoraco-abdominal movements and reduces activity of the scalenes and sternomastoid muscles. In addition, the forward lean position with the arms supported allows the pectoral muscles to significantly contribute to rib cage elevation (Gosselink 2004). Relaxation techniques that do not involve breath holding or the contraction of large muscle groups may be useful for relieving dyspnoea in those who are anxious (Gosselink 2004). Symptomatic relief may be achieved by increasing the movement of cold air onto the patient’s face (e.g. sitting by an open window, use of a fan). This stimulates mechanoreceptors on the face and the decrease in skin temperature may alter afferent feedback to the brain and the perception of dyspnoea (ATS 1999).
Pursed-lip breathing is often spontaneously adopted by patients with COPD and may be very effective in reducing the discomfort associated with dyspnoea. This breathing strategy aims to prevent airway closure and increase expiratory time. This may lead to a decrease in respiratory rate, an increase in tidal volume (VT) and, in turn, may improve gas exchange at rest. Pursed-lip breathing appears to be most effective when used by patients with COPD who adopt the technique spontaneously (Gosselink 2004). Recovery from dyspnoea following physical activity can be assisted with positioning (e.g. forward lean with the arms supported) together with breathing strategies such as breathing control or PLB. Some patients have a tendency to breath-hold when performing physical tasks and encouraging exhalation during effort is another breathing strategy that may be helpful. For selected patients with severe dyspnoea, education on energy conservation techniques during ADL is important. Conversely, many patients with respiratory or cardiovascular disease adopt a very sedentary lifestyle and, in addition to starting an exercise programme, such patients require encouragement to participate in a greater range of ADL with adequate rests as required to recover from breathlessness. Exercise training is an effective method of relieving dyspnoea in patients with stable chronic lung disease and in those with cardiac failure (ATS /European Respiratory Society (ERS) 2006, Lacasse et al 2006, Rees et al 2004). The underlying mechanisms responsible for the reduction in dyspnoea following exercise training are varied and for the individual may include:
 Physiologic training effect with an increased aerobic capacity of the peripheral muscles associated with decreased lactate production and decreased ventilation at a given submaximal workload
Physiologic training effect with an increased aerobic capacity of the peripheral muscles associated with decreased lactate production and decreased ventilation at a given submaximal workload Decreased oxygen consumption (VO2) and ventilation for a given level of physical activity as a result of improved mechanical efficiency (e.g. increased stride length when walking)
Decreased oxygen consumption (VO2) and ventilation for a given level of physical activity as a result of improved mechanical efficiency (e.g. increased stride length when walking) Reduced anxiety as a result of improved self-confidence and desensitization to the intensity of dyspnoea from repeated controlled exposure to a stimulus (e.g. as may occur with regular participation in supervised exercise classes).
Reduced anxiety as a result of improved self-confidence and desensitization to the intensity of dyspnoea from repeated controlled exposure to a stimulus (e.g. as may occur with regular participation in supervised exercise classes).Walking aids (e.g. rollator/wheeled walker, gutter frame/pulpit frame) that facilitate the forward lean position and arm support may reduce dyspnoea, increase exercise tolerance and may limit the extent of oxygen desaturation in some patients with COPD (Probst et al 2004, Solway et al 2002). Ambulatory oxygen may be beneficial for patients who are dyspnoeic on exercise and demonstrate oxygen desaturation. Oxygen therapy is only indicated if shown to produce benefit in terms of increased exercise tolerance and reduced breathlessness. The application of non-invasive ventilation (NIV) during exercise may reduce dyspnoea and improve exercise endurance and has been shown to be effective in patients with COPD (van’t Hul et al 2002). However, the use of NIV during exercise is cumbersome and often difficult in clinical practice.
For patients with COPD, high-intensity inspiratory muscle training (IMT) has been shown to reduce dyspnoea but the effects of such training on measures of whole body exercise capacity are less convincing (Geddes et al 2005, Hill et al 2006, Lotters et al 2002). Indications for IMT in COPD patients include the presence of severe musculoskeletal problems that prevent participation in a whole body exercise training programme and severe intractable dyspnoea persisting following whole body exercise training.
Outcome measures
The relevant outcome measures include assessment of dyspnoea intensity at rest, during ADL and when exercising. Dyspnoea intensity is most easily quantified using the modified Borg (0-10) Category Ratio Scale (Borg 1982). Changes in exercise tolerance are usually assessed in the clinical setting using a field walking test (Chapter 3). Scales that quantify the functional limitation due to dyspnoea are useful outcome measures of physiotherapy intervention and include the Medical Research Council (MRC) Scale and the New York Heart Association (NYHA) Scale, the University of California San Diego Shortness of Breath Questionnaire, the London Chest Activity of Daily Living Scale and the Pulmonary Functional Status and Dyspnea Questionnaire (Meek 2004). A health-related QoL questionnaire that includes quantification of dyspnoea during ADL is a useful outcome measure for patients who present with dyspnoea and participate in a pulmonary or heart failure rehabilitation programme (Chapters 13 and 14).
PROBLEM - DECREASED EXERCISE TOLERANCE
Exercise tolerance in patients with respiratory or cardiovascular disease is invariably limited by dyspnoea, pain (chest or legs) or fatigue (general or local). This section outlines the pathophysiological basis for exercise limitation occurring in respiratory and cardiovascular diseases. In addition to the pathophysiologic abnormalities that adversely affect exercise tolerance, depression and anxiety often accompany chronic respiratory or cardiovascular disease and may decrease an individual’s confidence or motivation to exercise.
Chronic lung disease
Patients with moderate to severe respiratory disease usually terminate exercise due to the development of intolerable symptoms (i.e. dyspnoea or leg fatigue) and fail to reach maximal heart rate (HR) and oxygen consumption. Leg fatigue is more likely to be a limiting factor when cycling is performed as compared with walking. In patients with moderate to severe COPD and chest wall abnormalities (e.g. kyphoscoliosis), respiratory muscle dysfunction may also be present and may contribute to dyspnoea and decreased exercise tolerance (see Problem - respiratory muscle dysfunction) (ATS/ERS 2006). Patients with moderate or severe respiratory disease, especially those with COPD, may experience marked dyspnoea when performing ADL that involve the use of the upper limbs, especially when the upper limbs are unsupported (see Problem - dyspnoea).
A respiratory impairment to exercise may be due to dysfunction of any or all components of the respiratory system. Normally, the ventilatory demands of exercise are met by an increase in VT and respiratory rate. Physiological abnormalities present in respiratory disease limit the ability to increase VT during exercise and thus increases in minute ventilation (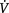 E) occur as a result of a disproportionate increase in respiratory rate. This occurs in diseases characterized by airflow limitation such as COPD, ILD, chest wall impairments and respiratory muscle weakness. Thus, for a given level of VE during exercise, VT tends to be lower and respiratory rate higher than in healthy individuals (Roca & Rabinovich 2005). The excessive increase in respiratory rate is very costly in terms of the oxygen required by the respiratory muscles because of the much larger number of muscle contractions and the increase in deadspace ventilation. In effect, the respiratory muscles may use oxygen at the expense of other skeletal muscles.
E) occur as a result of a disproportionate increase in respiratory rate. This occurs in diseases characterized by airflow limitation such as COPD, ILD, chest wall impairments and respiratory muscle weakness. Thus, for a given level of VE during exercise, VT tends to be lower and respiratory rate higher than in healthy individuals (Roca & Rabinovich 2005). The excessive increase in respiratory rate is very costly in terms of the oxygen required by the respiratory muscles because of the much larger number of muscle contractions and the increase in deadspace ventilation. In effect, the respiratory muscles may use oxygen at the expense of other skeletal muscles.
In many patients with moderate to severe COPD, functional residual capacity (FRC) is elevated compared with normal even at rest (i.e. the lungs are hyperinflated). Peripheral airway narrowing and loss of lung elastic recoil lead to gas trapping during expiration and are responsible for the increase in FRC. During exercise, the increase in respiratory rate limits the time available for expiration, resulting in a further increase in end-expiratory lung volume. Dynamic hyperinflation is the term used for the increase in gas trapping that occurs in patients with COPD during exercise and acute exacer- bations (see Problem - airflow limitation). The associated lung hyperinflation serves to improve ventilation by decreasing airway resistance and increasing expiratory flow rates. However, the main disadvantage of this compensatory mechanism is the altered mechanics of the respiratory muscles that increase WOB. The associated increase in deadspace ventilation and reduction in alveolar ventilation contribute to the impairments in gas exchange observed during exercise.
Healthy subjects maintain normal levels of partial pressure of oxygen in arterial blood (PaO2) unless extreme levels of exercise are undertaken and partial pressure of carbon dioxide in arterial blood (PaCO2) does not change from resting values until acidosis occurs as a result of high blood lactate levels. When this occurs, ventilation is further increased and PaCO2 falls (ATS/American College of Chest Physicians (ACCP) 2003). In contrast, many patients with chronic lung disease experience oxygen desaturation on exercise especially when the transfer factor of the lung for carbon monoxide (TLCO) is markedly impaired (Hadeli et al 2001, Roca & Rabinovich 2005). A low TLCO is characteristic of ILD and also occurs in some patients with COPD who have predominantly emphysematous changes. In such individuals, hypoxaemia contributes to exercise intolerance both directly through increased chemoreceptor activity and indirectly via stimulation of lactic acid production (ATS/ERS 2006).
In patients with asthma, exercise capacity may be limited by bronchoconstriction and exercise-induced asthma (EIA) may be the only symptom of asthma especially in young patients with mild disease. Usually with exercise there is a mild bronchodilator effect thought to be due to the increase in sympathetic stimulation and withdrawal of vagal control. With EIA, a rapid fall in FEV1 occurs immediately after the cessation of exercise. The likelihood of developing EIA in susceptible individuals is greater when their asthma control is poor. Exercise performed when an individual has an upper respiratory tract infection or has recently had a cold is more likely to provoke EIA. The types of exercise and environmental conditions most likely to cause EIA are exercise that is continuous and is associated with a high ventilatory demand, for example long distance running or cycling, especially when such exercise takes place in cold and dry conditions or in the presence of high levels of air pollution or known allergens. Following a warm-up, or short bout of exercise, there is a refractory period when protection against EIA occurs (Storms 1999, 2005).
Cardiovascular impairment
Multiple factors contribute to exercise intolerance in patients with cardiovascular disease. These factors include inadequate oxygen delivery, abnormalities in HR response, systolic and diastolic function, abnormal pulmonary vascular responses and peripheral muscle dysfunction (see below) (ATS /ACCP 2003). Compared with healthy subjects, in patients with cardiovascular disease the increase in stroke volume with exercise is often reduced and there is a greater reliance on the HR response to achieve the levels of cardiac output required to meet the metabolic requirements. However, the HR response to exercise may be blunted compared with normal. In more severe disease (e.g. especially chronic heart failure (CHF)), the peak HR achieved declines and during submaximal exercise the HR response is greater for a given level of VO2 compared with normal (Pina et al 2003). Ventilatory responses to exercise are often abnormal and are characterized by a greater VE at submaximal VO2. In the absence of coexisting lung disease, arterial desaturation during exercise is not observed in patients with stable cardiovascular disease (ATS/ACCP 2003).
Peripheral muscle dysfunction in chronic lung disease and heart failure
Peripheral muscle dysfunction occurs in patients with chronic lung disease and heart failure and contributes to reduced exercise tolerance. Most of the evidence for peripheral muscle dysfunction has been obtained from studies of the quadriceps muscles in patients with COPD and CHF. The documented abnormalities include muscle wasting, decreased strength, a reduction in the proportion of type I fibres (i.e. endurance fibres), a reduction in the number of capillary contacts to muscle fibre cross-sectional area and a decreased concentration of mitochondrial enzymes (ATS/ERS 2006, Gosker et al 2003). Further, in patients with CHF, blood flow to the exercising muscles is reduced as a result of a lower than normal cardiac output and impairment in the endothelium-dependent vasodilatory pathways in these muscles (Pina et al 2003). The changes described above are responsible for the reduced capacity of the peripheral muscles for aerobic metabolism observed in patients with chronic lung disease and CHF. As a consequence, there is a greater increase in lactic acidosis for a given exercise work rate and this contributes to the development of leg fatigue and in turn increases the ventilatory requirements. The aetiology of the peripheral muscle dysfunction is multifactorial and may include inactivity-induced deconditioning, systemic inflammation, oxidative stress, blood gas abnormalities and corticosteroid use (ATS/ERS 2006, Gosker et al 2003).
Peripheral arterial disease
In peripheral arterial disease, the oxygen supply to the exercising muscles is inadequate to meet the metabolic requirements. This oxygen deficiency leads to a build-up of lactic acid and claudication pain occurs during low-intensity weight bearing exercise (i.e. walking).
Marked deconditioning
Marked deconditioning is a common sequela of the prolonged bed rest and decreased mobility that occurs in patients with a protracted recovery following pneumonia, major surgery and time spent in an intensive care unit. Deconditioning is also commonly found in patients with respiratory or cardiovascular disease who avoid exercise because of dyspnoea or fatigue. In a deconditioned individual, the oxygen cost of exercise is higher at any given submaximal exercise intensity than in an individual who is physically fit. This is due to central and peripheral mechanisms that include an increased HR response to exercise, increase in cardiac afterload and a decrease in the aerobic capacity of the peripheral muscles. This leads to the early onset of aerobic metabolism. Further, the avoidance of physical activity and exercise may decrease the skill and efficiency of physical movements.
Other conditions that limit exercise tolerance
Obesity is a common problem in developed countries and is associated with the morbidity and mortality of many conditions including coronary artery disease, type II diabetes and obstructive sleep apnoea. The presence of obesity decreases exercise capacity due to the increase in the metabolic requirements at rest, and the greater respiratory and cardiac work required by an obese individual during exercise.
Many individuals with respiratory or cardiovascular disease are elderly. In addition to the age-related decline in physical work capacity, such individuals may have neurological or musculoskeletal conditions that limit their ability to exercise.
Clinical features
Subjectively the patient with reduced exercise tolerance will report that their performance of ADL or the ability to exercise is limited as a result of breathlessness, fatigue or pain. Patients with EIA will report that respiratory symptoms, i.e. cough, chest tightness or wheeze, occur during or immediately following exercise. If peripheral arterial disease is present, the patient will report pain in the calf and often also in the buttock limiting their ability to walk. The intensity of pain will be increased when walking up inclines, hurrying and walking on uneven ground and barefoot.
On examination, functional exercise capacity, which is most commonly assessed in the clinical setting with a field walking test, will be reduced, abnormal physiological responses (e.g. oxygen desaturation, blunted or increased HR response) may be present and the patient will report symptoms limiting exercise (i.e. dyspnoea, fatigue or pain). When EIA is present, there will be a reduction in FEV1 and peak expiratory flow rate (PEFR) measured immediately following exercise. In patients with claudication pain, a marked limp may be noticeable prior to the patient terminating a walking test. In the obese individual, exercise tolerance is usually limited due to breathlessness occurring with low-intensity exercise.
A cardiopulmonary exercise test, including assessment of the physiologic responses, is required to identify the pathophysiological limitation to exercise (Chapters 3, 13 and 14 on assessment, pulmonary rehabilitation and cardiac rehabilitation). The physiologic variables measured usually include VO2, carbon dioxide output (VCO2), VE, VT, and respiratory rate, oxygenation (measured via oximetry, SpO2), HR and rhythm (electrocardiogram (ECG)) and systemic blood pressure. In many clinical settings, a full cardiopulmonary exercise test may not be available; however, when a cardiopulmonary exercise test is performed, the data obtained can assist with exercise prescription.
On examination of the patient there may be signs of peripheral muscle wasting and a reduction in muscle strength found on testing. In some patients, respiratory muscle strength may be reduced compared with normal values (Chapter 3).
For the patient with acute cardiopulmonary dysfunction an exercise test is inappropriate. Information regarding the likely responses to interventions aimed at improving oxygen transport (e.g. transferring from bed to chair, standing, ambulating) will be obtained from knowledge of responses to nursing and medical interventions and observation of subjective and objective data over time. Chapter 4 details the therapeutic effects of positioning and mobilization for the patient with acute cardiopulmonary dysfunction.
Physiotherapy management
Identification and optimal medical management of the underlying cause of reduced exercise tolerance (e.g. COPD, IHD, valvular heart disease, CHF) and of comorbid conditions (e.g. osteoarthritis, diabetes, obesity, malnutrition) are essential before starting an exercise programme. Individuals with known or suspected EIA should use an inhaled short-acting β2−agonist or non-steroidal anti-inflammatory medication before exercise.
There is strong evidence to support the benefits of exercise training for people with respiratory and cardiovascular disease (Lacasse et al 2006, Leng et al 2001, Rees et al 2004, Smart & Marwick 2004). Regular physical activity is also effective in preventing cardiovascular disease (Thompson et al 2003). The following section outlines the broad principles of exercise training. The reader is also referred to Chapters 4, 8, 12-14 for further information on the physiotherapy management of specific patient populations.
An exercise programme should be designed to meet the specific requirements of the patient. Supervised exercise training, involving a group of patients with the same or similar condition, can be very helpful. Such training provides peer support that may assist patients to overcome anxiety and improve their motivation to exercise. Further, the supervised setting ensures that the exercise prescription is safe and enables the physiotherapist to adjust the prescription on a regular basis. Concurrent with starting an exercise programme, many patients need encouragement to engage in more physical activity during their everyday life.
The frequency, intensity, duration and modes of exercise should be individually selected for each patient based on assessment findings and established goals. In general, the programme should consist of a warm-up, stretches, an aerobic component, resistive training, when appropriate, and a cool-down. Postural correction is also important (Chapter 5 and see Problem - musculoskeletal dysfunction: postural abnormalities, decreased compliance or deformity of the chest wall). Unsupported upper limb exercises are especially important for patients who have respiratory disease and complain of dyspnoea during ADL that involve the upper limbs. Although it is recognized that endurance training is beneficial for most patients, it may be more important for some patients to improve their speed of ambulation over a short distance (e.g. to cross a road or to improve their ability to reach the toilet because of urinary urgency) rather than focusing on improving the distance the patient can walk. Resistance training is an important component of a programme for many patients, as weakness of the peripheral muscles is commonly found in patients with respiratory or cardiovascular disease and may contribute to exercise intolerance (Chapters 13 and 14).
Intermittent exercise and interval training are useful to improve the exercise endurance of patients who are severely deconditioned, extremely dyspnoeic, excessively fatigued or have claudication pain, and are essential when rehabilitating patients in the acute care setting. When marked oxygen desaturation occurs with exercise, the inclusion of frequent rests can assist in maintaining oxygen saturation at an acceptable level.
An adequate warm-up before exercise and including intermittent exercise to provide and make use of the refractory period is very important for those with EIA (Storms 2005).
In the dyspnoeic patient, the use of breathing strategies may assist with improving exercise tolerance and may accelerate the rate of recovery following exercise (see Problem - dyspnoea). Walking endurance may be increased with the use of a walking aid that provides arm support and facilitates a forward lean position (see Problem - dyspnoea). Ambulatory oxygen therapy during exercise is essential for patients who are receiving long-term oxygen therapy and should be provided in accordance with local guidelines. The flow rate may need to be increased especially during exercise that involves large muscle groups (i.e. walking, step-ups); however, care must be taken to monitor signs of carbon dioxide retention in susceptible patients. In patients who markedly desaturate on exercise but have adequate oxygen saturation levels at rest, formal assessment of the benefits of ambulatory oxygen for exercise is required. A walking test (field test or treadmill test) should be used in preference to a cycle ergometry test to identify exercise-induced hypoxaemia as oxygen desaturation is more marked with walking than cycling (Poulain et al 2003, Turner et al 2004).
In selected patients, the application of non-invasive ventilation (NIV) during exercise may enable exercise to be performed at a greater intensity or duration.
Patients should, whenever possible, exercise in an appropriate environment. Extremes of temperature, very high humidity, very windy conditions and environments with high levels of airborne irritants or pollutants should be avoided. These recommendations are especially important for individuals with EIA.
Other medical conditions
Regular exercise and an overall increase in physical activity are important components of an obesity treatment regimen that also includes dietary control. Regular exercise in the obese individual may improve body image, self-esteem and mood, and assist with more rigorous adherence to a dietary regimen. Even in the absence of weight loss, regular exercise can help blood lipid profiles and reduce the risk of hypertension and mortality from cardiovascular disease (Paffenbarger et al 1983, Sandvik et al 1993). An exercise programme for obese patients should include an aerobic component, and non-weight bearing exercise at a low to moderate intensity to avoid the development of musculoskeletal problems. A component of resistance training is also important. The duration and frequency of exercise should be gradually increased with the overall aim of achieving a high-energy expenditure.
Exercise training in patients with type II diabetes has been shown to improve glycaemic control, decrease body fat and improve plasma lipids even in the absence of weight loss (American Diabetes Association 2004). Patients with type I and type II diabetes should be able to safely participate in exercise following careful screening and providing they have good control of their blood glucose level. This requires regular blood glucose monitoring before, during and following exercise. Screening of patients with diabetes before starting an exercise programme is required to identify the presence of any complications that may be exacerbated by exercise. These complications comprise cardiovascular abnormalities, including autonomic neuropathy, as these abnormalities may be associated with silent ischaemia and hypotension or hypertension following exercise. Patients with diabetes who are at a high-risk for underlying cardiovascular disease should undergo a graded exercise test with ECG monitoring before starting an exercise programme. Screening for the presence of peripheral arterial disease, retinopathy, ne-phropathy and peripheral neuropathy should also be undertaken in patients with diabetes. The presence of retinopathy requires individuals to avoid exercise that involves straining or Valsava-like manoeuvres. Peripheral neuropathy and peripheral arterial disease are more common in people with diabetes and precautionary measures for the feet, such as special socks and proper footwear, are essential to avoid trauma to the area. Adequate hydration is essential to avoid any adverse effects of exercise on the cardiovascular system or glucose levels; thus, fluid replacement during exercise is important to compensate for fluid loss from sweating (American Diabetes Association 2004).
Outcome measures
Commonly used measures for evaluating the effects of an exercise programme include the assessment of anthropometric variables (e.g. weight, body mass index, waist : hip ratio) in selected patients, resting blood pressure, peripheral muscle strength, functional exercise capacity including assessment of HR response to exercise and symptoms (dyspnoea, fatigue, pain) and QoL using disease-specific questionnaires where available. The ability to perform ADL may be assessed via patient self-report or quantified using validated questionnaires. The economic benefits of an exercise programme for people with CHF or COPD may be evaluated by recording hospital admissions and length of stay before and following rehabilitation.
Chapters 13 and 14 detail specific outcome measures for patients participating in pulmonary or cardiac rehabilitation programmes.
PROBLEM - REDUCED LUNG VOLUME
Reduced lung volume refers to the inability to expand lung tissue and includes a reduction of residual volume (RV), VT, expiratory reserve volume (ERV) and/or a reduction in inspiratory reserve volume (IRV). There are two main clinical consequences of these reduced lung volumes: a reduction in total lung capacity (TLC) and vital capacity (VC), as a patient may be unable to increase inhaled volume sufficiently to expand lung tissue, and a reduction in FRC, as a patient may be unable to sustain alveolar inflation. A reduction in lung volume occurs in a variety of situations and may be short-lived (e.g. when FRC is reduced following major surgery) or chronic (e.g. when all lung volumes and hence TLC are reduced as a consequence of ILD). On occasions the cause is a disease process affecting the lung parenchyma but in many situations the reduction in lung volume arises from processes affecting other structures, such as the chest wall, respiratory muscles or the pleura. A decrease in FRC is an almost universal finding following upper abdominal surgery or cardiothoracic surgery.
Many patients with reduced lung volumes and capacities present with the problems of dyspnoea and decreased exercise tolerance, due to the inability to meet the ventilatory demands of physical activity (for example, patients with ILD may present with dyspnoea on ADL). Alternatively, they may present with the problem of impaired airway clearance due to the inability to take a sufficiently deep breath resulting in reduced expiratory flow and an ineffective cough. Impaired gas exchange and orthopnoea may also be present in patients with reduced lung volume.
The pathophysiological basis for a reduction in lung volume and clinical examples are given in Table 6.7.
Table 6.7 Pathophysiological basis of a reduction in lung volume and clinical examples
| Pathophysiological basis | Clinical examples | |
|---|---|---|
| Decrease in compliance | 1. Lung | ILD |
| 2. Chest wall | Kyphoscoliosis, ankylosing spondylitis, spinal cord injury – quadriplegia, disruption to the integrity of the chest wall due to trauma, e.g. flail chest | |
| Atelectasis | 1. Postoperative period: normal consequence of UAS and cardiothoracic surgery | Due to the general anaesthetic, surgery and changes occurring in the postoperative period including a lack of periodic deep breaths, absence of sighs, diaphragm dysfunction, supine position |
| 2. Reduced function of surfactant | ARDS, smoke inhalation, high FiO2, effect of general anaesthesia and opioids | |
| 3. Airway obstruction | Foreign body, mucus plugging, hilar adenopathy, mediastinal masses | |
| 4. Negative airway pressure | Endobronchial suctioning | |
| Compression of lung tissue | 1. Pleural space encroachment | Effusion, empyema |
| 2. Mediastinal structures | Tension pneumothorax causing mediastinal shift | |
| 3. Cardiomegaly | Decreases ventilation to left lower lobe when supine, e.g. CHF | |
| 4. Abdominal distension | Obesity, ascites, following abdominal surgery, running-in phase of peritoneal dialysis, pregnancy | |
| Pain | Causes shallow breathing, e.g. postoperatively, chest trauma, rib fractures | |
| Decreased ability of respiratory muscles to generate sufficient negative pressure | Respiratory muscle weakness causing an ineffective cough (see Problem – respiratory muscle dysfunction) | |
ILD, interstitial lung disease; UAS, upper abdominal surgery; ARDS, acute respiratory distress syndrome; FiO2, fraction of inspired oxygen; CHF, chronic heart failure
A reduction in lung volumes is only problematic when it significantly affects gas exchange, the ability to clear the airways or causes dyspnoea. The main consequences of a decrease in lung volume are:
 Impaired oxygenation due to V/Q mismatching. This occurs because the small airways in the dependent lung regions may close during normal tidal volume breathing. In some cases, wasted perfusion, where blood passes through consolidated, collapsed or damaged lung without being oxygenated (intrapulmonary shunt), may also contribute to impaired oxygenation, especially if hypoxic vasoconstriction is ineffective. Low tidal volume breathing may be associated with a failure to clear the anatomic dead space, resulting in impaired gas exchange.
Impaired oxygenation due to V/Q mismatching. This occurs because the small airways in the dependent lung regions may close during normal tidal volume breathing. In some cases, wasted perfusion, where blood passes through consolidated, collapsed or damaged lung without being oxygenated (intrapulmonary shunt), may also contribute to impaired oxygenation, especially if hypoxic vasoconstriction is ineffective. Low tidal volume breathing may be associated with a failure to clear the anatomic dead space, resulting in impaired gas exchange. Ineffective cough due to the reduction in VC, which reduces the ability to generate an adequate expiratory airflow.
Ineffective cough due to the reduction in VC, which reduces the ability to generate an adequate expiratory airflow.Clinical features
Subjectively the patient may report that breathing requires effort. On examination there will be an adaptive breathing pattern characterized by a small VT and increased respiratory rate. In the presence of pain, or fear of pain (e.g. in the patient with a surgical incision, fractured ribs or pleuritic pain), there will be an absence of periodic sighs. Chest expansion will be reduced overall such as with ILD or may be a localized finding, for example in the area overlying a collapsed lobe.
On auscultation there will be absent, diminished or bronchial breath sounds. The cough will be weak, due mainly to the inability to achieve an adequate inhaled volume and expiratory airflow (e.g. in spinal cord injury, quadriplegia). Chest radiograph findings may identify the cause of the reduction in lung volumes such as a pleural disorder, ILD, lobar or lung collapse.
Clinical features resulting from acute lobar collapse depend on the extent of the collapse, the abruptness of onset and the underlying respiratory impairment. A slowly developing segmental or lobar collapse may produce few symptoms if the patient has otherwise normal lungs. If a similar magnitude of lung collapse occurs acutely in a patient with chronic lung disease, severe respiratory distress may develop in which case the clinical features of hypoxaemia will be present (see Problem - impaired gas exchange). If however, there is a decrease in perfusion to the collapsed lung as a result of hypoxic pulmonary vasoconstriction, hypoxaemia may be minimal or absent.
Lung function test results show a decrease in lung volumes (RV) and capacities (FRC, VC and TLC). Hypoxaemia may be present, particularly when there is an inability to expand a large portion of lung tissue, primarily as a result of V/Q mismatching arising from changes in the FRC/closing volume relationship. Hypercapnia is often absent but will occur if there is associated hypoventilation or chronic lung disease.
Physiotherapy management
When gas exchange is reduced or WOB is increased, physiotherapy management is largely focused on the optimization of lung volumes achieved by upright positioning. As upright positions increase FRC and therefore alveolar inflation, high sitting, sitting out of bed and ambulation are encouraged. The side-lying position is preferred to slumped or supine positions and may be modified by tilting the patient towards prone to further decrease compression on lung tissue. This is especially so in patients with abdominal distension (Jenkins et al 1988). Functional residual capacity may also be increased with the use of continuous positive airway pressure (CPAP).
Short-lived increases in VT and inspiratory capacity may occur with the use of breathing techniques (e.g. thoracic expansion exercises, sustained maximal inspirations with or without the use of an incentive spirometer, intermittent positive airway pressure (IPPB)) and manual hyperinflation in the intubated patient. These breathing techniques assist in re-expansion of lung tissue and will be more efficient if performed in upright positions. In addition, upright positioning will assist patients to increase inhaled volume sufficiently to generate adequate expiratory airflow and produce an effective cough.
For patients with fixed and irreversible chest wall or lung pathology (e.g. kyphoscoliosis, ILD), physiotherapy is unable to influence lung volume; however, the associated problems of dyspnoea and reduced exercise tolerance can be treated (see Problem - dyspnoea and Problem - decreased exercise tolerance).
Ambulation increases VE (Chapter 4) and when ambulating, patients should be encouraged to take frequent deep breaths to ensure that the increased VE is not solely due to an increase in respiratory rate (Orfanos et al 1999, Zafiropoulos et al 2004) and to assist in lung re-expansion.
Patients at increased risk of developing clinically significant atelectasis following surgery should be identified and prophylactic physiotherapy started (Chapter 12).
Obese patients may benefit from exercise programmes designed to achieve weight reduction provided that exercise is accompanied by dietary control (see Problem - decreased exercise tolerance).
Outcome measures
Following physiotherapy intervention there may be a change in cough effectiveness. Auscultatory findings may reflect changes in lung volume. Chest radiographs may reflect resolution of the underlying problem but are not always good indicators of clinical progress; for example, following coronary artery surgery, small pleural effusions or atelectasis may persist for a considerable time after the patient has recovered clinically. Pulmonary function tests will reflect changes in lung volume if the underlying lung pathology is reversible. Improvements in associated problems such as impaired gas exchange, reduced exercise tolerance and dyspnoea may be evident. In the high-risk surgical patient, the outcome will be prevention of PPCs and reduced length of hospitalization.
PROBLEM - IMPAIRED GAS EXCHANGE
Impaired gas exchange is common in patients with respiratory or cardiovascular disease. In some patients, abnormalities may only become evident when increased demands are imposed on the respiratory and cardiovascular systems such as during physical activity, an infection or when changes in ventilation occur as a normal consequence of sleep (Chapter 11). Gas exchange abnormalities rarely occur in the absence of one or more of the other problems described in this chapter. Impaired gas exchange presents as hypoxaemia, hypercapnia or hypocapnia (West 2005). When the respiratory system is unable to provide adequate gas exchange for metabolic requirements, respiratory failure occurs.
Although changes in ventilation arise in response to hypoxaemia and hypercapnia, dyspnoea is not necessarily present. The physiotherapist does not always have a role in the management of impaired gas exchange; for example, in the patient with acute pulmonary embolus.
Hypoxaemia
Hypoxaemia may be acute or chronic and is seen in a wide range of conditions. Type I respiratory failure is present in a patient who is awake and at rest when PaO2 is below 8 kPa (60 mmHg); values at sea level. The pathophysiological basis of hypoxaemia and clinical examples are given in Table 6.8.
Table 6.8 Pathophysiological basis of hypoxaemia and clinical examples
| Pathophysiological basis | Clinical examples | |
|---|---|---|
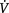 / / mismatch mismatch |
Commonest cause in respiratory disease | |
| 1. Decrease in FRC | Reduced lung volumes secondary to UAS or cardiothoracic surgery, obesity, ascites, atelectasis, supine position, ILD | |
| 2. Increase in CV | Small airway closure due to airflow limitation (e.g. cigarette smoking, COPD), pulmonary oedema, increased age | |
| Wasted ventilation (perfusion limitation) | Occurs when a perfusion defect prevents inspired oxygen from reaching arterial blood e.g. pulmonary embolus | |
| Wasted perfusion (intrapulmonary shunt) | Occurs when blood is shunted through non-ventilated lung tissue (e.g. atelectasis or consolidation), somewhat attenuated by hypoxic pulmonary vasoconstriction | |
| Cardiac shunt | Atrial septal defect, ventricular septal defect | |
| Hypoventilation | Site of abnormality: | |
| 1. Respiratory centre | Depression of hypercapnic and hypoxic ventilatory drives by drugs, general anaesthesia, as a normal consequence of sleep | |
| 2. Medulla | Trauma, neoplasm | |
| 3. Spinal cord | Trauma, neoplasm | |
| 4. Anterior horn cell | Trauma, neoplasm, poliomyelitis | |
| 5. Innervation of the respiratory muscles | Phrenic nerve paralysis | |
| 6. Disease of the myoneural junction | Myasthenia gravis | |
| 7. Respiratory muscles | Weakness or fatigue (see Problem –respiratory muscle dysfunction) | |
| 8. Upper airway obstruction | Foreign body, obstructive sleep apnoea | |
| 9. Excessive WOB | When an added load on the mechanics of breathing occurs such as in the patient with acute severe asthma who is exhausted, acute exacerbation of COPD | |
| Diffusion limitation | Decrease in alveolar–capillary surface area Decrease in diffusion gradient Increased thickness of alveolar–capillary membrane Decreased transit time of red blood cell in pulmonary capillary |
Emphysema Low FiO2 as occurs at high altitude Scarring or fluid in the interstitial space, e.g. pulmonary oedema, ILD May cause hypoxaemia on exercise in the presence of another cause of diffusion limitation |
| Decrease in FiO2 | High altitude | |
| Malfunctioning of respiratory equipment | Disconnection of gas supply | |
| Endotracheal suctioning | ||
| Mixed causes | Combination of 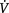 / / mismatch, diffusion limitation, shunt and hypoventilation mismatch, diffusion limitation, shunt and hypoventilation |
Seen in severe chronic lung disease |
Imbalance between 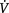 O2 and DO2 (i.e. oxygen consumption and delivery) O2 and DO2 (i.e. oxygen consumption and delivery) |
This causes a reduction in PvO2. Low PvO2 magnifies the effects of  / / mismatch and shunt on a patient’s level of oxygenation mismatch and shunt on a patient’s level of oxygenation |
Low cardiac output states, severe anaemia |
WOB, work of breathing; COPD, chronic obstructive pulmonary disease;  /
/ , ventilation/perfusion ratio; FRC, functional residual capacity;
, ventilation/perfusion ratio; FRC, functional residual capacity;
CV, closing volume; UAS, upper abdominal surgery; ILD, interstitial lung disease; ARDS, acute respiratory distress syndrome; FiO2, fraction of inspired oxygen;  O2, oxygen consumption, PvO2, mixed venous oxygen tension; DO2, oxygen delivery
O2, oxygen consumption, PvO2, mixed venous oxygen tension; DO2, oxygen delivery
Hypercapnia
Hypoxaemia always occurs in the presence of hypercapnia in the spontaneously breathing patient unless the patient is receiving oxygen therapy. For the patient who is awake and at rest, a raised PaCO2 is the hallmark of type II respiratory failure, which is present when PaCO2 exceeds 6.7 kPa (50 mmHg) and is accompanied by a PaO2 of less than 8 kPa (60 mmHg); values at sea level. Type II respiratory failure can be acute or chronic. The pathophysiological basis of hypercapnia and clinical examples are given in Table 6.9 (Haslet et al 2002).
Table 6.9 Pathophysiological basis of hypercapnia and clinical examples
| Pathophysiological basis | Clinical examples | |
|---|---|---|
| Hypoventilation | 1. Reduced central drive | Obesity-hypoventilation syndrome, depression of the respiratory centre due to reduced conscious state, general anaesthesia, narcotics, barbiturates |
| 2. Respiratory muscle dysfunction | See Problem – respiratory muscle dysfunction | |
| 3. Added load on the mechanics of breathing | Changes in compliance of the lung or chest wall, e.g. chest wall trauma, pulmonary oedema, large pleural effusion, ILD Increase in airways resistance, e.g. severe COPD |
|
Increased  CO2 CO2 |
Increased metabolism, e.g. fever, sepsis, trauma, burns, exercise Metabolic acidosis |
|
| Increased dead space as a fraction of VT | COPD, pulmonary embolus, low lung volume breathing, e.g. with pain, respiratory muscle weakness |
ILD, insterstitial lung disease; COPD, chronic obstructive pulmonary disease;  CO2, carbon dioxide production; VT, tidal volume
CO2, carbon dioxide production; VT, tidal volume
Under normal conditions, PaCO2 is an important factor in the chemical control of ventilation and the relationship between PaCO2 and VE is linear (Schwartzstein & Parker 2006). The ventilatory response to PaCO2 is reduced during sleep, in elderly individuals and there is a wide variation in the ventilatory response to PaCO2 among individuals. When the PaCO2 is normal, there is little increase in ventilation until PaO2 has fallen below 8 kPa (60 mmHg). When hypercapnia is present, the ventilatory response to hypoxaemia is heightened. When PaCO2 is chronically elevated, for example in some patients with COPD, the ventilatory response to PaCO2 is significantly decreased and hypoxaemia becomes the chief stimulus to ventilation (Lumb 2005, West 2005). When high levels of inspired oxygen are administered to such patients to relieve the hypoxaemia (i.e. those with type II respiratory failure), ventilation may become depressed resulting in a decrease in VE and a subsequent worsening of gas exchange.
Hypocapnia
In clinical practice, a low PaCO2 is a far less common occurrence than a raised PaCO2. In an individual who is spontaneously breathing an increase in the rate or depth of breathing may not result in hypocapnia. For example, a large VT accompanied by a low respiratory rate may not reduce PaCO2 below normal levels. Conversely, a small VT and high respiratory rate, such as when panting, may not lower PaCO2 or may even raise PaCO2 if the VT fails to clear the anatomic dead space. The underlying mechanism involved in alveolar hyperventilation and thus hypocapnia is an increased respiratory drive mediated through a behavioural or metabolic respiratory control system (Phillipson 2005).
Clinical features
Patients may adapt to chronic changes in arterial blood gas tensions whereas acute hypoxaemia and hypercapnia are less well tolerated.
There are few clinical features associated with mild hypoxaemia; however, if associated with severe anaemia or impaired cardiac output, mild hypoxaemia may be poorly tolerated. At moderate degrees of hypoxaemia (PaO2 between 4.5 and 8 kPa (34 and 60 mmHg) ventilation may be increased to twice the normal level (Schwartzstein & Parker 2006). The features of moderate or severe hypoxaemia that develop acutely are tachypnoea, restlessness, confusion, sweating, tachycardia, hypertension, skin pallor and cyanosis. With severe hypoxaemia, bradycardia, arrhythmias and hypotension may develop. Worsening neurologic signs associated with acute severe hypoxaemia include blurred vision, tunnel vision, loss of coordination, impaired judgement, convulsions, coma and permanent brain damage. Hypoxaemia exacerbates cardiac arrhythmias and angina in patients with IHD and may predispose to heart failure. The long-term cardiovascular consequences of chronic hypoxaemia are pulmonary hypertension and cor pulmonale.
The clinical features of hypercapnia are determined more by the onset of the raised PaCO2 rather than the severity. Chronic hypercapnia may not be clinically manifest until a more rapid rise is induced by infection, sedation, or oxygen administration. Signs and symptoms associated with hypercapnia include those consistent with vasodilatation, for example warm peripheries, the appearance of flushed skin and a full and bounding pulse. The cardiac system may respond with episodes of extrasystoles. The resulting increase in cerebral blood flow is responsible for headache (this occurs especially on waking and can occur during exercise), a raised cerebrospinal fluid pressure and sometimes papilloedema (oedema of the optic disc). Impaired concentration and drowsiness may also be present. Rapid accumulation of CO2 and the accompanying acidosis may quickly lead to convulsions and coma. The clinical features of hypercapnia may be less obvious in patients with COPD who have chronic respiratory failure because metabolic adaptations may occur in response to chronically elevated PaCO2.
The signs and symptoms of hypocapnia are many and varied (Gardner 2003). They include paraesthesia in the hands, face and trunk and tetany. A reduction in central nervous system and cerebral blood flow may be responsible for dizziness, loss of consciousness, visual disturbances, headache, tinnitus, ataxia and tremor. With acute hypocapnia, systemic blood pressure falls and HR increases. Peripheral vasoconstriction is thought to be responsible for the complaint of cold hands.
Impaired gas exchange will be reflected in abnormalities in arterial blood gases, pulse oximetry and transcutaneous measures of PaCO2.
Physiotherapy management
Physiotherapy management is determined by the underlying pathophysiological cause(s) and may be limited. Oxygen therapy is indicated whenever tissue oxygenation is impaired and should be prescribed by the medical practitioner in accordance with established guidelines. Physiotherapists should ensure the correct application of oxygen therapy including the delivery device and flow rate/concentration (Chapter 5). For patients with type II respiratory failure susceptible to oxygen-induced respiratory depression, controlled oxygen therapy should be administered via a fixed performance device (Chapter 5). For these patients, careful administration of oxygen therapy is required when a nebulizer is used (Chapter 5).
Gas exchange may by optimized by positioning patients in an upright position to increase FRC. In spontaneously breathing adults with unilateral lung pathology, V/Q matching may be improved by positioning in side lying with the unaffected lung dependent. However, hypoxaemia due to hypoventilation may be worsened by positioning in supine lying due to the increased load on the respiratory muscles.
Physical activity may improve gas exchange by enhancing oxygen transport or may result in further impairment in gas transfer (i.e. identified by a fall in oxygen saturation) in some patients with severe cardiopulmonary dysfunction. Oxygen therapy during ambulation may be indicated in some patients and this should be assessed using a walking test (Chapter 3) (Poulain et al 2003, Turner et al 2004). In other patients, NIV applied during ambulation may improve gas exchange. Non-invasive ventilation is also a useful treatment for exacerbations of COPD, some causes of acute respiratory failure and nocturnal hypoventilation (Mehta & Hill 2001). Specifically, CPAP may improve oxygenation in patients with acute cardiogenic pulmonary oedema (Cooper 2004).
Special case - positioning the ventilated patient
Prone positioning may improve oxygenation in acutely ill patients who are mechanically ventilated and sedated (e.g. patients with severe acute respiratory failure or acute respiratory distress syndrome) (Stiller 2000, Wong 1999). However, turning critically ill patients who are mechanically ventilated into the side-lying position may acutely increase VO2 and cause transient hypoxaemia evoking exercise and stress-like responses (Horiuchi et al 1997).
Outcome measures
Arterial blood gases, oximetry and transcutaneous end-tidal measures of PaCO2 will reflect changes in gas exchange. Short-term changes in cognitive function and symptoms such as headache may be evident when abnormalities in gas exchange are corrected.
In patients with long-term abnormalities in gas exchange, changes in QoL may occur in response to interventions such as NIV or long-term oxygen therapy. In this patient group, a reduction in healthcare utilization and improved survival may also occur.
PROBLEM - AIRFLOW LIMITATION
Airflow limitation can be described as an abnormal resistance or obstruction to airflow. This problem generally occurs together with other physiotherapy problems such as dyspnoea, decreased exercise tolerance or impaired airway clearance. Airflow limitation can be caused by factors inside the airway, changes in the airway wall and factors outside the airway. The pathophysiological basis for airflow limitation is presented in Table 6.10 together with clinical examples (West 2003). Airflow limitation may be reversible (e.g. asthma without fixed airflow obstruction), partially reversible (e.g. chronic bronchitis) or irreversible (e.g. emphysema).
Table 6.10 Pathophysiological basis for airflow limitation and clinical examples
| Pathophysiological basis | Clinical examples | |
|---|---|---|
| Causes inside the airway | Partial or total occlusion of the airway lumen | Mucus, e.g. chronic bronchitis, cystic fibrosis, bronchiectasis, asthma Inhaled foreign body Bronchial tumour |
| Changes in the airway wall | Smooth muscle contraction | Asthma |
| Smooth muscle hypertrophy and hyperplasia | Asthma | |
| Inflammation of the mucosa | Asthma, infective exacerbation of COPD | |
| Hypertrophy of mucus glands | Chronic bronchitis | |
| Thickening of the bronchial wall | Chronic bronchitis, asthma | |
| Dilation and destruction of airway wall | Cystic fibrosis, bronchiectasis | |
| Infiltration of the bronchial mucosa with eosinophils and lymphocytes | Asthma | |
| Changes in the osmolarity of normal airway fluid produced by cooling | Exercise-induced asthma – see Problem – decreased exercise tolerance | |
| Causes outside the airway | Loss of radial traction due to a decrease in elastic recoil secondary to increases in lung compliance | Emphysema |
| Compression | Enlarged lymph nodes, tumour |
COPD, chronic obstructive pulmonary disease
Special case - lung hyperinflation in COPD
Lung hyperinflation is an abnormal increase in the volume of air remaining in the lungs at the end of relaxed expiration. In patients with COPD, static lung hyperinflation, which develops over time, results from an increase in expiratory airflow resistance and a reduction in elastic recoil giving rise to air trapping (Ferguson 2006).
Dynamic hyperinflation is related to the degree of airflow limitation and the time available for exhalation. The increased metabolic demands of exercise, for example, require an increase in VE which is achieved by an increase in VT and respiratory rate. The increase in respiratory rate reduces the time available to exhale. Exhalation is already prolonged due to the increased resistance to expiratory airflow. The resultant incomplete exhalation causes air trapping and an increase in end-expiratory lung volume, which leads to the development of intrinsic positive end-expiratory pressure (PEEP). As end-expiratory lung volume increases, there is a decrease in inspiratory capacity. With limited inspiratory capacity, increases in VE can only be achieved by an increase in respiratory rate. This further reduces expiratory time and a vicious cycle of air trapping and progressive hyperinflation occurs.
During an acute exacerbation of COPD there is an increase in expiratory airflow resistance due to inflammation and mucus plugging and so dynamic hyperinflation may occur at a lower level of physical activity than usual. Similarly, when a patient becomes emotional or anxious, respiratory rate often increases and the cycle of air trapping and progressive hyperinflation may occur.
Dynamic hyperinflation increases the WOB in order to initiate inspiratory airflow, the inspiratory muscles are required to generate a pleural pressure in excess of the intrinsic PEEP. This imposes an additional threshold load at the start of inspiration because the inspiratory muscles have to overcome this positive pressure. As lung hyperinflation increases there is a decrease in lung compliance leading to a further increase in the WOB on inspiration. As a result of lung hyperinflation, the diaphragm fibres are shortened and the altered length-tension relationship may decrease the ability to generate muscle tension and inspiratory pressure.
Some patients with severe hyperinflation, especially during exercise, may use the abdominal release mechanism to decrease the work of the diaphragm while still maintaining its output. To effect this mechanism, the patient contracts the abdominal muscles at the end of expiration, thus pushing the contents of the abdomen up against the diaphragm and improving its length-tension relationship. The increase in lung volume during the subsequent inspiration occurs by a sudden release of the abdominal pressure that acts to passively pull the diaphragm downwards (McCarren 1992).
Clinical features
The patient with airflow limitation may report chest tightness or wheeze, cough and breathlessness, limiting their ability to undertake ADL and exercise. In asthma, cough and breathlessness may be particularly evident at night or early morning and may lead to sleep disruption. Patients may report that their cough is ‘tight’ and they have difficulty clearing secretions.
On examination, patients may display signs of an increased WOB to overcome the increase in airway resistance. Respiratory rate may be increased and expiration may be active, with contraction of the abdominal muscles, and prolonged expiration (increased I : E ratio). Recruitment of the accessory respiratory muscles may be evident and indrawing of the intercostal spaces and supraclavicular fossae visible. Pursed-lip breathing is adopted by some patients with severe airflow limitation (e.g. COPD).
With long-standing disease, examination of the chest may reveal signs of hyperinflation such as a barrel-shaped chest with an increase in the anteroposterior diameter and a raised shoulder girdle. Paradoxical movement of the lower chest wall can occur in patients with severe hyperinflation as the diaphragm is unable to descend and contraction of the diaphragm during inspiration leads to indrawing of the lower ribs (Hoover’s sign).
Wheezing may be audible. Auscultatory findings often reveal widespread, polyphonic wheezes; however, patients with hyperinflation may have reduced breath sounds.
The chest radiograph may show signs of hyperinflation as well as signs consistent with the underlying condition; for example, the presence of emphysematous bullae or bronchiectatic changes.
Abnormalities in pulmonary function indicative of airflow limitation comprise a reduction in FEV1, FEV1/FVC, PEFR and forced expiratory flow at 25-75% of forced expiratory flow (FEF25–75). Characteristic patterns can be seen in the flow-volume loop and may help with identifying the cause and site of the airflow limitation (Chapter 3). Functional residual capacity, RV and TLC are often increased and the ratio of RV to TLC may be elevated.
Physiotherapy management
Where airflow limitation is reversible, optimal medical management with medication is essential. Patient education is an important aspect of the management of airflow limitation, particularly in asthma, and may be undertaken by a physiotherapist in some settings. A simple explanation about the mechanism(s) of airflow limitation, the importance of avoiding trigger factors including cigarette smoke and allergens, and the use and effects of medications are essential. Self-management should be encouraged and, in particular for patients with asthma, the physiotherapist’s role includes reinforcing the patient’s action plan that has been developed by a medical officer to manage their changing symptoms (Gibson et al 2002).
Education and instruction in the correct method of using inhaled medications to reduce airflow limitation is necessary to ensure maximal deposition of the medication. A large range of devices is available for the delivery of inhaled respiratory medications and the physiotherapist should be familiar with these (Chapter 5). Airway clearance techniques with prior, effective delivery of an inhaled bronchodilator may help to overcome airflow limitation due to retained bronchial secretions and bronchospasm. Where necessary airway clearance techniques should be adapted to ensure that no increase in airflow limitation occurs. Increasing the length of periods of breathing control between huffs in the forced expiration technique (FET) may help to prevent an increase in airflow limitation. For patients with COPD it may be necessary to modify the huff to prevent dynamic airway collapse by decreasing expiratory force or increasing inspiratory volume (van der Schans 1997). Alternatively, positive expiratory pressure (PEP) therapy has been suggested as an appropriate airway clearance technique for patients with COPD as it provides positive mouth pressure to splint open collapsible airways (Holland & Button 2006). Further details on these airway clearance techniques are provided in Chapter 5 on physiotherapy techniques.
Breathing strategies such as PLB may be encouraged in patients who spontaneously adopt the technique (Gosselink 2004). This technique aims to splint the airways open and increase expiratory time, thereby reducing dyspnoea. Other breathing strategies may be directed towards the management of dyspnoea associated with airflow limitation (see physiotherapy management in Problem - dyspnoea). For patients with asthma a number of breathing ‘retraining’ strategies (such as the Buteyko technique; Chapter 17) have been advocated although there is no consistent evidence for improved asthma control (Holloway & Ram 2004).
For the management of EIA, see Problem - decreased exercise tolerance.
Where the cause of airflow limitation is a tumour or enlarged lymph nodes, physiotherapy management is not indicated.
Outcome measures
Changes in symptoms related to airflow limitation, particularly in asthma, can be measured with a health-related QoL questionnaire. A number of widely used questionnaires exist, including:
For patients with COPD, the SGRQ includes a section that quantifies symptoms of wheeze/chest tightness (Jones et al 1992).
Short-term changes in airflow limitation, as reflected by wheeze, may be evident on auscultation.
Pulmonary function tests may reflect changes in the degree of airflow limitation depending upon the underlying aetiology and whether the condition is reversible.
PROBLEM - RESPIRATORY MUSCLE DYSFUNCTION
Respiratory muscle dysfunction (i.e. reduced strength or endurance) may be present in a range of clinical conditions including neuromuscular disorders (e.g. motor neuron disease, multiple sclerosis, Guillain-Barré syndrome, myasthenia gravis, muscular dystrophy, tetraplegia), connective tissue diseases (e.g. systemic lupus erythematosus), chronic lung disease (e.g. COPD, cystic fibrosis), chest wall disorders (e.g. kyphoscoliosis) and CHF. The problem may also occur in patients treated with oral corticosteroids and in those who are mal-nourished (Troosters et al 2005). Respiratory muscle dysfunction becomes a physiotherapy problem when the patient presents with problems such as dyspnoea, impaired airway clearance or inability to wean from mechanical ventilation.
Clinical features
Mild respiratory muscle dysfunction is often difficult to detect using simple clinical measures. Weakness of the respiratory muscles is often advanced before clinical symptoms are present because the pressure required to initiate inspiratory flow represents only a small proportion of the maximum force-generating capacity of the inspiratory muscles (Troosters et al 2005). The main clinical features and patient problems associated with respiratory muscle weakness are an unexplained reduction in VC, abnormal breathing pattern, nocturnal hypoxaemia and hypercapnia in the absence of chronic lung disease, dyspnoea, decreased exercise tolerance and impaired airway clearance.
Subjectively, the patient may report breathlessness, especially when supine or standing in water up to their chest, for example when entering the sea or a swimming pool. The weight of water causes pressure on the chest and the abdominal wall and thus the load on the inspiratory muscles, especially the diaphragm, is increased. Marked breathlessness during ADL that involve unsupported upper limb activities may be reported by patients with diaphragm weakness or bilateral phrenic nerve palsy. Daytime somnolence, early morning headaches and impaired mental function may be present if hypoxaemia and hypercapnia occur during sleep. Abnormalities in breathing pattern may be present and include increased respiratory rate, decreased VT, reduced chest expansion, use of accessory muscles and respiratory alternans (periods of breathing using only chest wall muscles alternating with periods of breathing using the diaphragm). Profound diaphragm weakness or paralysis gives rise to paradoxical movement of the abdomen occurring during inspiration and is most easily seen with the patient supine (Moxham 1999). This occurs when the diaphragm is unable to match the negative intrapleural pressure generated by the other inspiratory muscles, resulting in passive transmission of this pressure during inspiration and causing the abdominal contents to be pulled upwards. When upright, recruitment of the abdominal muscles may occur during expiration in order to elevate the diaphragm so that gravity and chest wall and lung recoil pressure can assist diaphragm descent during inspiration. The patient may have a weak cough due to an inadequate inspired volume or weakness of the expiratory muscles resulting in a reduction in expiratory airflow. The weak cough may in turn lead to the problem of impaired airway clearance.
The plain chest radiograph is a useful diagnostic tool in unilateral diaphragm paralysis as the affected dome is elevated. In addition to respiratory muscle weakness there may also be signs of generalized muscle wasting and assessment may reveal a reduction in peripheral muscle strength. The ability to perform ADL and participate in exercise will be impaired as a result of dyspnoea reflecting the difficulties of the inspiratory muscles to cope with the required increase in ventila- tion. Depending on the underlying pathological process, peripheral muscle dysfunction may also contribute to exercise intolerance.
Lung function
Lung function may be normal in the absence of marked weakness. The most frequently noted abnormality in lung function is a marked reduction in VC. In the presence of severe bilateral diaphragm weakness, the VC is low when the patient is upright and typically falls by more than 50% when supine, whereas in normal subjects the reduction in VC when supine is less than 10% (ATS/ERS 2002). The fall in VC is due to the weight of the abdominal contents in supine exerting an upward pressure on the diaphragm. Measurement of VC is especially useful in the management of progressive disorders such as Guillain-Barré syndrome. The pattern of abnormality of other lung capacities and volumes is less consistent. Residual volume is normal or may be increased, especially if there is marked expiratory muscle weakness. As a consequence, TLC is reduced to a lesser extent than VC and the RV/TLC and FRC/TLC ratios are often increased (ATS/ERS 2002). In the absence of lung disease, airway resistance to expiratory airflow is normal as indicated by a normal FEV1/FVC ratio. Gas transfer (i.e. TLCO) is usually normal or only mildly reduced as a result of the inability to fully distend the lungs at TLC and thus enable the entire alveolar surface to be exposed to carbon monoxide (ATS/ERS 2002). Depending on the underlying aetiology, inspiratory and/or expiratory muscle strength (i.e. PiMax and PeMax) will be reduced compared with normal values (Chapter 3). Assessment of respiratory muscle endurance is more complex and is rarely performed outside of specialized laboratories.
Gas exchange abnormalities
In the awake patient with chronic muscle weakness, even when severe, only a mild reduction in PaO2 is present and PaCO2 is usually normal. When respiratory muscle weakness occurs acutely, a more marked reduction is PaO2 is seen. In the absence of lung disease, daytime hypercapnia is unlikely unless VC is reduced to less than 50% predicted normal value and respiratory muscle strength is reduced to less than 40% predicted normal value (ATS/ERS 2002). During sleep, dips in oxygen saturation occur during periods of rapid eye movement (REM) sleep when the work of the diaphragm is increased due to the reduction in the tone of the intercostals and accessory muscles. In the patient with slowly progressive weakness of the respiratory muscles, hypercapnia often develops insidiously at night and over time leads to persistent daytime hypercapnia and thus type II respiratory failure (Chapter 11).
Physiotherapy management
Targeted respiratory muscle training may be an effective means of increasing the strength and endurance of the respiratory muscles (Chapter 5). Most of the evidence for the benefits of respiratory muscle training has been gained from studies of IMT in patients with COPD. In this patient population, IMT performed using a threshold loading device or a target-flow resistive device has been shown to increase inspiratory muscle strength and endurance and reduce dyspnoea (Geddes et al 2005, Lotters et al 2002). The optimal IMT protocol has not been determined; however, high-intensity interval-based protocols, which include regular rest periods, appear to be more efficient at improving respiratory muscle function than protocols that do not permit rest periods (Hill et al 2004, 2006).
Although unsupported upper limb exercise training increases the reliance on the diaphragm to generate inspiratory flow, such training does not appear to improve diaphragm function. Patients may benefit from whole body exercise training as a result of improvements in peripheral muscle function. For patients with excess bronchial secretions, assistance with airway clearance including the use of assisted coughing techniques is often required (Chapters 5 and 16).
Non-invasive ventilation provided during sleep may be required to rest the respiratory muscles and correct blood gas abnormalities. Some patients may also require periods of NIV during the daytime.
In some patients, the underlying cause of respiratory muscle dysfunction is progressive and the main role of physiotherapy is the successful management of associated and potential problems (e.g. impaired airway clearance).
Outcome measures
The following outcome measures should be considered when evaluating the response to physiotherapy intervention. The patient’s perception of dyspnoea during ADL and exercise can be assessed using the Borg Category Ratio Scale or a VAS (see Problem - dyspnoea). Perception of dyspnoea during ADL can also be quantified using standardized questionnaires, for example, the dyspnoea domain of the Chronic Respiratory Disease Questionnaire (Guyatt et al 1987). Measurement of PiMax and PeMax will determine whether changes in respiratory muscle strength have occurred. Observation of breathing pattern and evaluation of cough effective- ness are important outcomes to evaluate. Measurement of VC is a useful outcome measure and simple to perform clinically. Changes in exercise tolerance can be assessed using a field walking test (Chapter 3).
In the mechanically ventilated patient who is having problems weaning, changes in respiratory muscle function may be associated with changes in the tolerance to periods of spontaneous breathing (i.e. duration of periods, arterial blood gas tensions).
In situations where the cause of respiratory muscle dysfunction is a progressive condition, the outcome measures for evaluating physiotherapy intervention often relate to the management of associated problems (such as Problem - impaired airway clearance) and may include such measures as the frequency of chest infection.
PROBLEM - DYSFUNCTIONAL BREATHING PATTERN
Dysfunctional breathing pattern rarely occurs in isolation in the physiotherapy problem list but is more commonly associated with other patient problems, some of which are amenable to physiotherapy intervention. Examples of these associated problems include dyspnoea and airflow limitation. Resolution of the associated problems may be accompanied by a return to a more normal breathing pattern. Some breathing patterns result from neurological abnormalities (e.g. Cheyne-Stokes respiration occurring in a brainstem cerebrovascular accident) or metabolic dysfunction. These breathing abnormalities are not amenable to physiotherapy intervention.
Observation of breathing pattern and respiratory effort may provide reliable clues about a patient’s underlying problem. For example, the characteristic breathing pattern of patients with reduced lung volume is a low VT and increased respiratory rate. When ventilatory demand is increased, patients with marked airflow limitation recruit their accessory respiratory muscles and signs of abdominal paradox and intercostal recession may be observed. Patients with airflow limitation may also use PLB (see Problem - airflow limitation). The changes in respiratory mechanics and breathing pattern give rise to an increased WOB in patients with reduced lung volume and airflow limitation but are necessary adaptations for the maintenance of adequate ventilation and gas exchange and are not dysfunctional.
Dysfunctional breathing mainly occurs when the WOB is exaggerated such that patients overbreathe or have an abnormally high respiratory rate (e.g. hyperventilation syndrome) for their metabolic requirements. The pathophysiology of dysfunctional breathing is complex and may arise due to psychological (e.g. anxiety) or physiological stress. The problem may also occur during exercise (Gardner 1996). This syndrome is often referred to as ‘hyperventilation syndrome’ and is discussed in detail in Chapter 17. Dysfunctional breathing is also commonly found in patients with asthma (Morgan 2006). In the severely breathless patient, a dysfunctional breathing pattern may become evident when the patient is recovering from breathlessness elicited by physical activity. In such instances, the dysfunctional breathing pattern may be due to anxiety.
Dysfunctional breathing may also result from abnormal functioning of the chest wall (e.g. flail chest or from spinal cord injury) where the normal respiratory mechanics are altered.
Clinical features
These include abnormalities in respiratory rate and depth, including excessive sighing or breath holding, and changes in the I : E ratio. Observation and palpation may reveal asymmetrical chest wall movement, paradoxical chest wall movement, asynchronous movements or respiratory alternans. The patient may overuse their accessory respiratory muscles, and may actively elevate their shoulders during inspiration. Associated excessive muscle activity (e.g. facial grimacing, gripping objects) may also occur.
Arterial blood gas analysis may demonstrate abnormalities consistent with an underlying problem (e.g. hypocapnia in hyperventilation syndrome or hypoxaemia and hypercapnia in spinal cord injury). Diagnostic tests may be used to assess hyperventilation including the voluntary hyperventilation provocation test, Nijmegen questionnaire and breath-holding time (Chapter 17).
Physiotherapy management
Breathing pattern retraining is focused on changing dysfunctional breathing patterns when they are not associated with strategies employed by the patient to reduce other problems such as dyspnoea or airflow limitation. Thus, physiotherapy generally aims to eliminate the exaggerated muscle activity associated with increased WOB or associated responses. Retraining may focus on changing VT, flow rate and strategies such as relaxation and breathing control are encouraged (Chapter 17).
Dysfunctional breathing patterns associated with abnormalities of the chest wall or spinal cord injury may be managed with positive pressure ventilation if these are associated with abnormal gas exchange.
Outcome measures
Physiotherapy intervention can be evaluated using the Nijmegen questionnaire, breath-holding time, diaries recording disability/distress/symptoms and QoL measures (Thomas et al 2003) (Chapter 17).
Arterial blood gas analysis, transcutaneous CO2 and oximetry may reflect concomitant changes in gas exchange.
PROBLEM - PAIN
This section outlines pain of respiratory and cardiovascular origin as well as other causes of pain located in the chest. Information regarding claudication pain and the physiotherapy management is included in the Problem - decreased exercise tolerance.
Chest pain of respiratory origin
The origin and characteristic features of pain due to respiratory causes, together with clinical examples, are given in Table 6.11 (Murray & Gebhart 2000). Pain of respiratory origin arises from the parietal pleura and from stimulation of the mucosa of the trachea and main bronchus. The lung parenchyma and visceral pleura are insensitive to pain. However, inflammatory processes in peripheral regions of the lung that involve the overlying visceral pleura often gives rise to pain as a result of involvement of the adjacent parietal pleura. Chest wall pain is often reported by patients who have chronic cough or dyspnoea as a result of the associated musculoskeletal pathology.
Chest pain of cardiovascular origin
Table 6.12 outlines the main causes and characteristic features of pain due to cardiovascular causes.
Table 6.12 Causes and characteristic features of chest pain due to cardiovascular causes
| Cause | Characteristic features | Stimuli |
|---|---|---|
| Myocardial ischaemia | Myocardial ischaemia does not always cause pain | |
| 1. Stable angina pectoris | ||
| 2. Unstable angina | As for stable angina | |
| 3. Acute myocardial infarction | Pain is similar to that of angina but is generally more severe and of longer duration | Usually requires large doses of opiates to control the pain |
| Pericarditis due to inflammation of parietal pericardium from a variety of causes – bacterial, viral, neoplasm, post-myocardial infarction | ||
| Diseases of the aorta | ||
RPP, rate pressure product (systolic blood pressure × heart rate)
Chest pain which may be unrelated to respiratory or cardiovascular disease
Neural, muscular or skeletal pain.
Examples of causative factors are disc degeneration, bony metastases, muscle injuries, inflammation of soft tissues and disorders of the costal cartilages. Pain in the chest wall also occurs following insertion of an intercostal catheter and following cardiothoracic surgery, fractured ribs, musculoskeletal disorders and tumours involving the ribs or soft tissues. The pain is usually localized to the affected area and the area is usually tender on palpation. The patient usually reports that the pain is a dull ache or may be described as a sharper pain. The pain is usually increased on respiratory movements including deep inspiration and cough, and is exacerbated by trunk and shoulder movements.
Oesophageal pain.
The causes of pain arising from the oesophagus are:
Peptic ulceration and gallbladder disease.
Diseases of the stomach, duodenum or biliary system may give rise to pain felt in the chest although this pain is more commonly confined to the abdomen.
With peptic ulceration the pain is burning in nature, occurs following meals and is relieved by antacids.
Pain of biliary origin is usually colicky in nature and felt on the right side of the abdomen, the front and back of the chest. The pain may be related to the ingestion of certain foods; for example, fatty foods.
Clinical features
The clinical features associated with pain of respiratory or cardiovascular origin are outlined in Tables 6.11 and 6.12. Subjective assessment will identify the clinical features of the pain and factors that elicit pain or increase the intensity of the pain.
Quantification of the pain intensity can be made using a VAS, a verbal rating of pain severity on a 0-10 or 0-5 scale. On examination of the patient there may be signs of an abnormal breathing pattern and systemic signs such as sweating, pallor and tachycardia. Chest expansion may be reduced over the painful area and there may be an associated reduction in breath sounds on auscultation and pleural rub.
Physiotherapy management
Diagnosis and management of the underlying cause are essential. Anti-inflammatory agents or analgesics are used for musculoskeletal, pleuritic and pericardial pain.
Direct methods of pain management used by physiotherapists include heat modalities, interferential, transcutaneous electrical nerve stimulation, Entonox, acupuncture and manual therapy. Knowledge of pain management, for example medications and their onset and duration of action, route of administration, is required so that physiotherapy interventions can be provided when pain management is optimal.
The physiotherapy management of patients with stable angina is described in Chapter 14.
Outcome measures
These may include assessment of pain and function obtained via subjective questioning and objectively using a VAS or other pain scale. The dose and frequency of analgesics or anti-inflammatory medications required by the patient should be recorded. Outcome measures for evaluating the effects of physiotherapy for the patient with angina are given in Chapter 14.
PROBLEM - MUSCULOSKELETAL DYSFUNCTION
Chest wall stiffness and abnormal posture are commonly seen in patients with neuromuscular disease or chronic lung disease especially when this is associated with lung hyperinflation. Also at risk are patients following sternotomy or thoracotomy and patients who receive prolonged mechanical ventilation. Changes in muscle length, strength and endurance will occur as a result of chest wall and postural abnormalities. Further, in the elderly, there are age-related changes affecting the musculoskeletal system. With increased age, there is a decrease in the range of movement of the costovertebral joints and a decrease in the elasticity of the cartilage in the thoracic spine. These changes increase thoracic kyphosis.
Patients with severe chronic lung disease who adopt the forward-lean position to relieve breathlessness may develop a stiff, kyphotic thoracic spine and abnormal posture of the neck. This in turn will limit the range of movement possible in the cervical and thoracic spine and shoulder girdle.
Neuromuscular disorders lead to progressive muscle weakening, which in turn may cause postural abnormalities, for example, scoliosis or kyphoscoliosis, further limiting chest wall muscle strength and mobility.
Clinical features
The patient may present with an abnormal posture, reduced range of movement of the cervical spine, thoracic spine and glenohumeral joint and may report pain or stiffness resulting in decreased function. The assessment of pain, associated functional limitation, posture, muscle length, strength and endurance and joint range of movement are covered elsewhere (Chapter 5).
Physiotherapy management
Physiotherapy management should include, where appropriate, postural correction, stretching techniques (for example, hold-relax), of tight muscles, mobilizations to the cervical spine and thoracic spine, costotransverse, costochondral and sternochondral joints, to the ribs and to the glenohumeral joint (Bray et al 1995, Vibekk 1991) and muscle strengthening exercises. Postural correction and stretches to improve chest wall mobility should be incorporated into other active exercises and ADL. Where possible, especially when chronic lung disease is present, patients should be taught to manage their own condition with clear instructions on appropriate stretches and exercises to carry out daily at home (Chapter 5).
The patient’s position during treatment will need to be carefully selected as many patients will not be able to lie prone or supine due to dyspnoea and mobilizations will have to be performed in sitting or forward lean sitting.
Following a sternotomy or thoracotomy, advice should be given on postural correction and upper limb exercises (Chapter 12).
For patients with neuromuscular disorders, physiotherapy management is targeted to the underlying condition.
Clinical outcomes
Range of movement of the cervical spine, thoracic spine and glenohumeral joints can be measured before and following physiotherapy intervention. Muscle length and strength can also be measured where appropriate. Short-term changes in pain can be measured on a VAS. Longer-term changes can be measured with a generic QoL scale such as the Short Form-36 Health Survey (SF-36) (Ware & Gandek 1998).
Adams RJ, Ruffin RE, Smith BJ. Validity of a modified version of the Marks Asthma Quality of Life Questionnaire. Journal of Asthma. 2000;37(2):131-143.
Ambrosino N, Scano G. Measurement and treatment of dyspnoea. Respiratory Medicine. 2001;95:539-547.
American Diabetes Association. Physical activity/exercise and diabetes. Diabetes Care. 2004;27:S58-S62.
American Thoracic Society. Dyspnea. Mechanisms, Assessment, and Management: A Consensus Statement. American Journal of Respiratory and Critical Care Medicine. 1999;159:321-340. www.thoracic.org/sections/publications/statements/pages/respiratory-disease-adults/dyspnea1-20.html (Accessed 9 July 2007)
American Thoracic Society/American College of Chest Physicians. ATS/ACCP statement on cardiopulmonary exercise testing. American Journal of Respiratory and Critical Care Medicine. 2003;167:211-277. www.thoracic.org/sections/publications/statements/pages/pfet/cardioexercise.html (Accessed 9 July 2007)
American Thoracic Society/European Respiratory Society. ATS/ERS Statement on respiratory muscle testing. American Journal of Respiratory and Critical Care Medicine. 2002;166:516-624. www.thoracic.org/sections/publications/statements/pages/respiratory-disease-adults/respmuscle.html (Accessed 9 July 2007)
American Thoracic Society/European Respiratory Society Statement on Pulmonary Rehabilitation. American Journal of Respiratory and Critical Care Medicine 2006;173:1390-1413. www.thoracic.org/sections/publications/statements/pages/respiratory-disease-adults/atserspr0606.html www.thoracic.org/sections/publications/statements/pages/respiratory-disease-adults/respmuscle.html.
Barker AF. Bronchiectasis. New England Journal of Medicine. 2002;346(18):1383-1393.
Borg GAV. Psychophysical basis of perceived exertion. Medicine and Science in Sports and Exercise. 1982;14:377-381.
Bray CE, Partridge JE, Banks SK, 1995 Thoracic mobilisation in the management of respiratory and cardiac patients. Proceedings of the Australian Physiotherapy Association Cardiothoracic Special Group, 4th National Conference, 22-24 April, Melbourne
Celli BR. Physical reconditioning of patients with respiratory diseases: legs, arms, and breathing retraining. Respiratory Care. 1994;39(5):481-495.
Clarke S. Physical defences. In: Brewis RAL, Gibson GJ, Geddes DM, editors. Respiratory medicine. London: Baillière Tindall; 1990:176-189.
Conway JH, Fleming JS, Perring S, et al. Humidification as an adjunct to chest physiotherapy in aiding tracheo-bronchial clearance in patients with bronchiectasis. Respiratory Medicine. 1992;86(2):109-114.
Cooper CB. Respiratory failure. Medicine. 2004;32(1):95-98.
Elkins M, Robinson M, Rose BR, et al. A controlled trial of long-term hypertonic saline in patients with cystic fibrosis. New England Journal of Medicine. 2006;354(3):229-240.
Ferguson GT. Why does the lung hyperinflate? Proceedings of the American Thoracic Society. 2006;3:176-179.
Gardner WN. The pathophysiology of hyperventilation disorders. Chest. 1996;109:515-534.
Gardner WN. Hyperventilation: a practical guide. Medicine. 2003;31(11):7-8.
Geddes EL, Reid WD, Crowe J, et al. Inspiratory muscle training in adults with chronic obstructive pulmonary disease: a systematic review. Respiratory Medicine. 2005;99:1440-1458.
Gibson PG, Powell H, Coughlan J 2002 Self-management education and regular practitioner review for adults with asthma. Cochrane Database of Systematic Reviews Issue 3. Art. No.: CD001117. DOI: 10.1002/14651858.CD001117. Aug 2006
Gosker HR, Lencer NHM, Franssem FME, et al. Striking similarities in systemic factors contributing to decreased exercise capacity in patients with severe chronic heart failure or COPD. Chest. 2003;123:1416-1424.
Gosselink R. Breathing techniques in patients with chronic obstructive pulmonary disease. Chronic Respiratory Disease. 2004;1:163-167.
Guyatt GH, Berman LB, Townsend M, et al. A measure of quality of life for clinical trials. Thorax. 1987;42:773-778.
Hadeli KO, Siegel EM, Sherrill DL, et al. Predictors of oxygen desaturation during submaximal exercise in 8,000 patients. Chest. 2001;120:88-92.
Haslet C, Chilvers ER, Corris PA. Respiratory Disease. In: Haslet C, Chilvers ER, Boon NA, et al, editors. Davidson’s principles and practice of medicine. Edinburgh: Churchill Livingstone, 2002., ch 13
Hill K, Jenkins SC, Hillman DR, et al. Dyspnoea in COPD: can inspiratory muscle training help? Australian Journal of Physiotherapy. 2004;50:169-180.
Hill K, Jenkins SC, Philippe D, et al. High-intensity inspiratory muscle training in chronic obstructive pulmonary disease. European Respiratory Journal. 2006;27:1119-1128.
Holland AE, Button BM. Is there a role for airway clearance techniques in chronic obstructive pulmonary disease? Chronic Respiratory Disease. 2006;3:83-91.
Holloway E, Ram FSF, 2004 Breathing exercises for asthma. Cochrane Database of Systematic Reviews Issue 1. Art. No.: CD001277. DOI: 10.1002/14651858.CD001277. pub2. Aug 2006
Horiuchi K, Jordan D, Cohen D, et al. Insights into the increased oxygen demand during chest physiotherapy. Critical Care Medicine. 1997;25(8):1347-1351.
Houtmeyers E, Gosselink R, Gayan-Ramirez G, et al. Regulation of mucociliary clearance in health and disease. European Respiratory Journal. 1999;13:1177-1188.
Houtmeyers E, Gosselink R, Gayan-Ramirez G, et al. Effects of drugs on mucus clearance. European Respiratory Journal. 1999;14:452-467.
Irwin RS, Widdicombe J. Cough. In Murray JF, Nadel JA, editors: Textbook of respiratory medicine, 3rd edn., Philadelphia: WB Saunders, 2000. 1, ch 21
Jenkins SC, Soutar SA, Moxham J. The effects of posture on lung volumes in normal subjects and in patients pre- and post-coronary artery surgery. Physiotherapy. 1988;74(10):492-496.
Jones AP, Rowe BH, 1998 Bronchopulmonary hygiene physical therapy for chronic obstructive pulmonary disease and bronchiectasis. Cochrane Database of Systematic Reviews Issue 4. Art. No.: CD000045. DOI: 10.1002/14651858.CD000045. Jul 2006
Jones AP, Wallis CE, Kearney CE, 2003 Recombinant human deoxyribonuclease for cystic fibrosis. Cochrane Database of Systematic Reviews Issue 3. Art. No.: CD001127. DOI: 10.1002/14651858.CD001127. Jul 2006
Jones PW, Quirk FH, Baveystock CM, et al. A self-complete measure of health status for chronic airflow limitation. The St. George’s Respiratory Questionnaire. American Review of Respiratory Disease. 1992;145:1321-1327.
Juniper EF, Guyatt GH, Ferrie PJ, et al. Measuring quality of life in asthma. American Review of Respiratory Disease. 1993;147:832-838.
Juniper EF, Guyatt GH, Cox FM, et al. Development and validation of the Mini Asthma Quality of Life Questionnaire. European Respiratory Journal. 1999;14:32-38.
Juniper EF, O’Byrne PM, Gyatt GH, et al. Development and validation of a questionnaire to measure asthma control. European Respiratory Journal. 1999;14:902-907.
Lacasse Y, Brosseau L, Milne S etal 2006 Pulmonary rehabilitation for chronic obstructive pulmonary disease. Cochrane Database of Systematic Reviews Issue 4. Art. No.: CD003793. DOI: 10.1002/14651858.CD003793 pub. 2 15 Sept 2007
Leng GC, Fowler B, Ernst E, 2001 Exercise for intermittent claudication. Cochrane Database of Systematic Reviews Issue 2. Art. No.: CD000990. DOI: 10.1002/14651858.CD000990 3 Aug 2006
Lotters F, van Tol B, Kwakkel G, et al. Effects of controlled inspiratory muscle training in patients with COPD: a meta-analysis. European Respiratory Journal. 2002;20:570-578.
Lumb AB. Nunn’s applied respiratory physiology, 6th edn., Philadelphia: Elsevier Butterworth-Heinemann, 2005.
Marks GB, Dunn SM, Woolcock AJ. An evaluation of an asthma quality of life questionnaire as a measure of change in adults with asthma. Journal of Clinical Epidemiology. 1993;46(10):1103-1111.
Markwell S, Sapsford R. Physiotherapy management of obstructed defaecation. Australian Journal of Physiotherapy. 1995;41(4):279-283.
McCarren B. Dynamic pulmonary hyperinflation. Australian Journal of Physiotherapy. 1992;38(3):175-179.
Meek PM. Measurement of dyspnea in chronic obstructive pulmonary disease. Chronic Respiratory Disease. 2004;1:29-37.
Mehta S, Hill NS. State of the art: noninvasive ventilation. American Journal of Respiratory and Critical Care Medicine. 2001;163:540-577.
Morgan MDL. Dysfunctional breathing in asthma: is it common, identifiable and correctable? Thorax. 2006;57(Suppl II):ii31-ii35.
Moxham J. Respiratory muscles. Medicine. 1999:126-129.
Murray JF, Gebhart GF. Chest pain. In Murray JF, Nadel JA, editors: Textbook of respiratory medicine, 3rd edn., Philadelphia: WB Saunders, 2000. 1, ch 22
O’Neill S, McCarthy DS. Postural relief of dyspnoea in severe chronic airflow limitation: relationship to respiratory muscle strength. Thorax. 1983;38:595-600.
Orfanos P, Ellis ER, Johnston C. Effects of deep breathing exercises and ambulation on pattern of ventilation in post-operative patients. Australian Journal of Physiotherapy. 1999;45:173-182.
Paffenbarger R, Wing A, Hyde R, et al. Physical activity and incidence of hypertension in college alumni. American Journal of Epidemiology. 1983;117:245-247.
Phillipson EA. Disorders of ventilation. In Kasper DL, Braunwald E, Fauci AS, et al, editors: Harrisons’s principles of internal medicine, 16th edn., New York: McGraw-Hill, 2005., ch 246
Pina IL, Apstein CS, Balady GJ, et al. Exercise and heart failure. A statement from the American Heart Association Committee on exercise, rehabilitation, and prevention. Circulation. 2003;107:1210-1225.
Poulain M, Durand F, Palomba B, et al. 6-minute walk testing is more sensitive than maximal incremental cycle testing for detecting oxygen desaturation in patients with COPD. Chest. 2003;123:1401-1407.
Probst VS, Troosters T, Coosemans I, et al. Mechanisms of improvement in exercise capacity using a rollator in patients with COPD. Chest. 2004;126:1102-1107.
Rees K, Taylor RS, Singh S et al. 2004 Exercise based rehabilitation for heart failure. Cochrane Database of Systematic Reviews Issue 3. Art. No.: CD003331. DOI: 10.1002/14651858.CD00331. pub2 30 Jul 2006
Roca J, Rabinovich R, 2005 Clinical exercise testing. European Respiratory Monograph 31: 146-165
Sandvik L, Erikssen J, Thaulow E, et al. Physical fitness as a predictor of mortality among healthy middle aged Norweigan men. New England Journal of Medicine. 1993;328:533-537.
Scano G, Stendardi L, Grazzini M. Understanding dyspnoea by its language. European Respiratory Journal. 2005;25:380-385.
Schwartzstein RM, Parker MJ. Respiratory physiology. In: A clinical approach. Philadelphia: Lippincott Williams & Wilkins; 2006.
Smart N, Marwick TH. Exercise training for patients with heart failure: a systematic review of factors that improve mortality and morbidity. American Journal of Medicine. 2004;116:693-706.
Solway S, Brooks D, Lau L, et al. The short-term effect of a rollator on functional exercise capacity among individuals with severe COPD. Chest. 2002;122:56-65.
Stiller K. Physiotherapy in intensive care: towards an evidence-based practice. Chest. 2000;118:1801-1813.
Storms WW. Exercise induced asthma: diagnosis and treatment for the recreational or elite athlete. Medicine and Science in Sports and Exercise. 1999;31(1):S33-S38. (Suppl)
Storms WW. Asthma associated with exercise. Immunology and Allergy Clinics of North America. 2005;25:31-43.
Thomas M, McKinley RK, Freeman E, et al. Breathing retraining for dysfunctional breathing in asthma: a randomised controlled trial. Thorax. 2003;58:110-115.
Thompson PD, Buchner D, Pina IL, et al. Exercise and physical activity in the prevention and treatment of atherosclerotic cardiovascular disease. Circulation. 2003;107:3109-3116.
Troosters T, Gosselink R, Decramer M, 2005 Respiratory muscle assessment. European Respiratory Monograph 31: 51-71
Turner SE, Eastwood PE, Cecins NM, et al. Physiologic responses to incremental and self-paced exercise in COPD. A comparison of three tests. Chest. 2004;126:766-773.
van der Schans CP. Forced expiratory manoeuvres to increase transport of bronchial mucus: a mechanistic approach. Monaldi Archives of Chest Disease. 1997;52(4):367-370.
van der Schans CP, Postma DS, Koeter GH, et al. Physiotherapy and bronchial mucus transport. European Respiratory Journal. 1999;13:1477-1486.
van der Schans C, Prasad A, Main E, 2000 Chest physiotherapy compared to no chest physiotherapy for cystic fibrosis. Cochrane Database of Systematic Reviews Issue 2. Art. No.: CD001401. DOI: 10.1002/14651858.CD001401. Jul 2006
van’t Hul A, Kwakkel G, Gosselink R. The acute effects of noninvasive ventilatory support during exercise on exercise endurance and dyspnea in patients with chronic obstructive pulmonary disease. A systematic review. Journal of Cardiopulmonary Rehabilitation. 2002;22:290-297.
Vibekk P. Chest mobilization and respiratory function. In: Pryor JA, editor. Respiratory care. Edinburgh: Churchill Livingstone; 1991:103-119.
Ware JE, Gandek B. Overview of the SF-36 health survey and the international quality of life assessment (IQOL) project. Journal of Clinical Epidemiology. 1998;51(11):903-912.
Wark PAB, McDonald V, Jones AP, 2005 Nebulised hypertonic saline for cystic fibrosis. Cochrane Database of Systematic Reviews, Issue 3. Art. No.: CD001506. DOI: 10.1002/14651858.CD001506. pub2. Jul 2006
West JB. Pulmonary pathophysiology - the essentials, 6th edn., Baltimore: Lippincott Williams & Wilkins, 2003.
West JB. Respiratory physiology: the essentials, 7th edn., Philadelphia: Lippincott Williams and Wilkins, 2005.
White D, Stiller K, Roney F. The prevalence and severity of symptoms of incontinence in adult cystic fibrosis patients. Physiotherapy Theory and Practice. 2000;16:35-42.
Wills P, Greenstone M, 2006 Inhaled hyperosmolar agents for bronchiectasis. Cochrane Database of Systematic Reviews Issue 2. Art. No.: CD002996. DOI: 10.1002/14651858.CD002996.pub2. Jul 2006
Wong WP. Use of body positioning in the mechanically ventilated patient with acute respiratory failure: application of Sackett’s rules of evidence. Physiotherapy Theory and Practice. 1999;15:25-41.
Zafiropoulos B, Alison JA, McCarren B. Physiological responses to the early mobilisation of the intubated, ventilated abdominal surgery patient. Australian Journal of Physiotherapy. 2004;50:95-100.