Chapter 12 Surgery for adults
INTRODUCTION
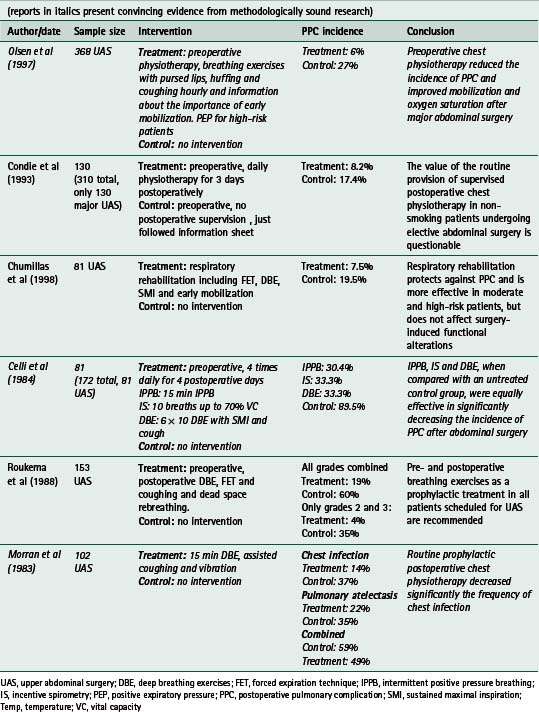
Perioperative physiotherapy aims to prevent or minimize the adverse physiological changes associated with major surgical procedures. In these patients, physiotherapy has played a significant part in minimizing the adverse effects of anaesthesia and surgery on the respiratory system for more than 50 years. This role of the physiotherapist has been supported by evidence from clinical trials reported since 1947. The evidence advocated pre- and postoperative physiotherapy for all patients having major surgery, to reduce the incidence of postoperative pulmonary complications (PPC) and thereby reduce patient morbidity and prolonged hospital admission.
Recent advances in both surgical and pain management, the evolution of new forms of postoperative physiotherapy support and a reduction in the incidence of clinically significant PPC have provided the stimulus for a re-evaluation of the place of physiotherapy in surgery. The incidence of PPC has shown a gradual reduction over time in these patient populations. In part this is due to a change in the method for the measurement of PPC, and also because anaesthetic and surgical techniques have improved. The use of epidural and patient-controlled analgesic techniques on the ward has also had a profound impact on the incidence of PPC. The incidence of PPC is stated to be 5–10% (Brooks-Brunn 1995), whereas earlier it was reported to be 20–30% (King 1933, Wightman 1968). The introduction of ‘fast track’ postoperative management and minimally invasive abdominal, thoracic and cardiac surgery has also impacted on the physiotherapy management in these patient populations, although more research is needed in these areas.
This chapter outlines the effects of the surgical process, pain and pain management on the respiratory system, the development of PPC, evidence for the physiotherapy treatment of different surgical populations, use of a variety of physiotherapy treatment techniques, management of surgical drips and drains, including underwater seal drainage and brief notes on different types of surgical procedures and lung cancer. The chapter has been written to aid both undergraduate physiotherapy students and cardiorespiratory clinicians in better understanding the area of physiotherapy and surgery. The evidence base underpinning the place of physiotherapy in surgery has been reviewed by Denehy & Browning (2007).
THE SURGICAL PROCESS
General anaesthesia
General anaesthesia (GA) provides the patient with sleep, amnesia and analgesia. Constant monitoring of the patient’s vital signs allow these to be kept within physiological limits. General anaesthesia can be divided into three different stages: induction, maintenance and reversal or emergence. Before induction an intravenous (IV) administration of a combination of an anxiolytic drug with amnesic power such as midazolam, together with a narcotic such as fentanyl, is often given. The narcotic given preoperatively helps to prevent nerve impulses, arising from intraoperative events, from sensitizing central neuronal structures and is called pre-emptive analgesia (Katz 1993). There is some evidence that analgesia given before the painful stimulus reduces subsequent pain, but this remains controversial.
Induction of anaesthesia:
Anaesthesia is usually achieved by IV administration of a short-acting, coma-inducing drug such as propofol or thiopental. Intubation may be performed if the surgery requires administration of muscle relaxants to cause paralysis (as is the case in major surgical procedures such as abdominal surgery). Maintenance of anaesthesia is achieved using inhalational agents such as sevoflurane with nitrous oxide or air with a suitably high inspired oxygen concentration (FiO2). Total intravenous anaesthesia using propofol may be used for maintenance and instead of an inhalation agent. During maintenance, muscle relaxants are often used to aid the surgical procedure and narcotics given for both intraoperative and postoperative analgesia. The process of reversal begins well before the surgeon has finished. Inspired anaesthetic concentrations are scaled back and drugs to reverse paralysis such as neostigmine are given. Analgesia is provided using narcotics or regional analgesia and extubation occurs once the patient can protect their airway (gag reflex) (Euliano & Gravenstein 2004).
Induction of anaesthesia causes unavoidable changes in lung mechanics, lung defences and gas exchange. The most profound effect on the lung of a GA is the reduction in lung volumes, particularly functional residual capacity (FRC). These are discussed in a later section.
Management of acute postoperative pain
It has been suggested that pain in the early postoperative period may be the most important factor responsible for ineffective ventilation, poor cough, impaired ability to breath deeply and sigh, atelectasis, hypoxaemia and respiratory distress postoperatively (Sabanathan et al 1999). It is clear that pain is an important factor that can be modified postoperatively to attenuate some of the above physiological changes associated with surgery. For this reason it is critically important that postoperative pain management is optimum. Inadequate analgesia may delay discharge from hospital, cause sleep disturbances and limit early mobilization.
Reduction in acute postoperative pain facilitates improved patient comfort and satisfaction, reduced length of hospital stay and rehabilitation. Acute postoperative pain is the result of local tissue damage with release of algesic substances (prostaglandins, histamine, serotonin, bradykinin) and generation of noxious stimuli, which are transduced by nociceptors and transmitted by A-delta and C nerve fibres to the neuraxis. Complex modulating influences occur in the spinal cord, producing segmental responses including increased sympathetic stimulation, muscle spasm and increased gastrointestinal tone. Other impulses are transmitted to higher centres via the spinothalamic and spinoreticular tracts producing cortical and suprasegmental responses. These result in further increased sympathetic tone increasing metabolism and oxygen consumption (Ready 1985). The major anatomical targets for relief of postoperative pain are the peripheral tissues, nerve axons in peripheral nerves and dorsal nerve roots, the dorsal horn of the spinal cord and the brain. Many different methods of pain relief, directed to these different anatomical sites are available to patients. Several other factors may modify postoperative pain: these include the site and duration of surgery and the extent of the incision and surgical trauma. However, the physiological and psychological makeup of the patient and past pain experience also play a part. Postoperative pain is often accompanied by changes in autonomic activity, which are sympathetically mediated and include hypertension, tachycardia, sweating and decreased gut motility (National Health and Medical Research Council 2005).
Drug management of postoperative pain
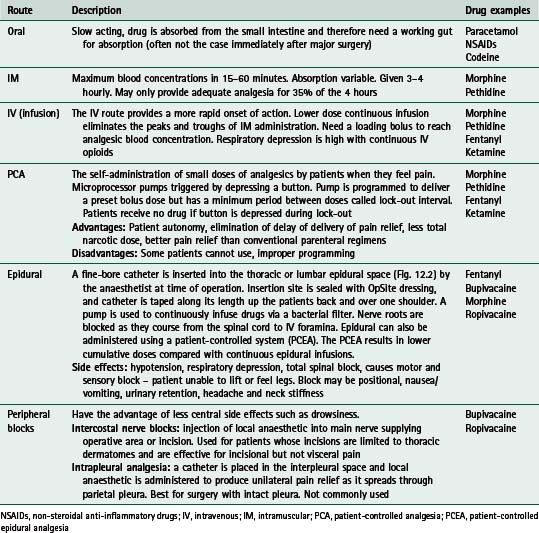
Many different methods of pain relief are available, using several different routes of administration. Opiates and derivatives (such as morphine, pethidine, fentanyl) make up a large proportion of these drugs and morphine arguably remains the benchmark drug (Barrat 1997). Table 12.1 gives a summary of common routes of administration of drugs. In Table 12.2 a list of the common drugs used for postoperative pain is shown, together with their potential side effects.
Table 12.1 Common routes of drug administration for postoperative analgesia
| Route | Description | Drug examples |
|---|---|---|
| Oral | Slow acting, drug is absorbed from the small intestine and therefore need a working gut for absorption (often not the case immediately after major surgery) | Paracetamol NSAIDs Codeine |
| IM | Maximum blood concentrations in 15–60 minutes. Absorption variable. Given 3–4 hourly. May only provide adequate analgesia for 35% of the 4 hours | Morphine Pethidine |
| IV (infusion) | The IV route provides a more rapid onset of action. Lower dose continuous infusion eliminates the peaks and troughs of IM administration. Need a loading bolus to reach analgesic blood concentration. Respiratory depression is high with continuous IV opioids | Morphine Pethidine Fentanyl Ketamine |
| PCA |
Microprocessor pumps triggered by depressing a button. Pump is programmed to deliver a preset bolus dose but has a minimum period between doses called lock-out interval. Patients receive no drug if button is depressed during lock-out
|
Morphine Pethidine Fentanyl Ketamine |
| Epidural |
A fine-bore catheter is inserted into the thoracic or lumbar epidural space (Fig. 12.2) by the anaesthetist at time of operation. Insertion site is sealed with OpSite dressing, and catheter is taped along its length up the patients back and over one shoulder. A pump is used to continuously infuse drugs via a bacterial filter. Nerve roots are blocked as they course from the spinal cord to IV foramina. Epidural can also be administered using a patient-controlled system (PCEA). The PCEA results in lower cumulative doses compared with continuous epidural infusions.
|
Fentanyl Bupivacaine Morphine Ropivacaine |
| Peripheral blocks | Bupivacaine Ropivacaine |
NSAIDs, non-steroidal anti-inflammatory drugs; IV, intravenous; IM, intramuscular; PCA, patient-controlled analgesia; PCEA, patient-controlled epidural analgesia
Opioids are drugs that bind with specific opioid receptors. They mimic endogenous peptide transmitters involved in pain modulation and act principally within the central nervous system. Non-steroidal antiinflammatory drugs (NSAIDs), such as indomethacin, are also used in management of acute postoperative pain and can act as opioid sparing agents. They decrease production of prostaglandins that sensitize nociceptor nerve endings to inflammatory mediators and also have an antipyretic effect (reduce fever). Local anaesthetic agents such as bupivacaine, block the initiation and propagation of action potentials by blocking sodium channels. They are used to produce nerve root blocks and to depress action potentials in sensory neurons, thereby reducing pain (NHMRC 2005).
The use of multimodality analgesia rather than single analgesic administration is more common in the new millennium as is the existence of hospital pain management teams led by anaesthetists and nurses.
Opiates have significant respiratory depression effects (Sabanathan et al 1999) and they are only partially effective in relieving pain. Richardson and Sabanathan (1997) report that since opioids may only relieve pain transmitted by C fibres, where opioid receptors exist, the sharp pain transmitted by A fibres still exists. More recently, use of local anaesthetics (bupivacaine, ropivacaine) by the epidural route has gained more favour in postoperative pain management, often used in combination with opioids. The introduction of these multimodal methods has also seen an increase in the use of NSAIDs such as indometacin. These are used as opioid sparing agents, but have also been shown to improve analgesia by a reduction in inflammation and stress inhibition (Richardson & Sabanathan 1997). There is level 1 evidence that oral administration of NSAIDs is as effective as intravenous (National Health and Medical Research Council 2005). Oral paracetamol or narcotic drugs may also be added to these regimens. Multimodal or balanced analgesia has been developed in response to the commonly associated side effects of monotherapy – nausea, vomiting, paralytic ileus and respiratory depression in the case of opioids and urinary retention, motor block and hypotension in the case of local anaesthetic agents. Balanced analgesia was also developed to allow early postoperative ambulation and enteral feeding, which minimize the respiratory and gut complications associated with use of opioids, leading to earlier patient discharge from hospital. The drugs are used in smaller doses than if used separately and provide effective pain relief as a result of their synergistic actions (Peeters-Asdourian & Gupta 1999).
The most common methods for pain control following abdominal surgery are patient-controlled analgesia (PCA) with intravenous opioids (Fig. 12.1) and continuous epidural analgesia delivering a combination of local anaesthetic and opioids (Fig. 12.2). In a recent systematic review it was concluded that continuous epidural analgesia is superior to PCA in relieving postoperative pain for up to 72 hours in patients undergoing intra-abdominal surgery. Similarly a large review of analgesia reports that all techniques of epidural analgesia provide better postoperative pain relief compared with parenteral opioid administration (NHMRC 2005). However, PCA remains a common method of analgesic delivery, as patients often prefer it (Werawatganon & Charuluxanun 2005). Continuous infusion may be associated with increased risk of respiratory depression compared with bolus IV administration and evidence for improved pain relief with continuous administration is lacking (NHMRC 2005).
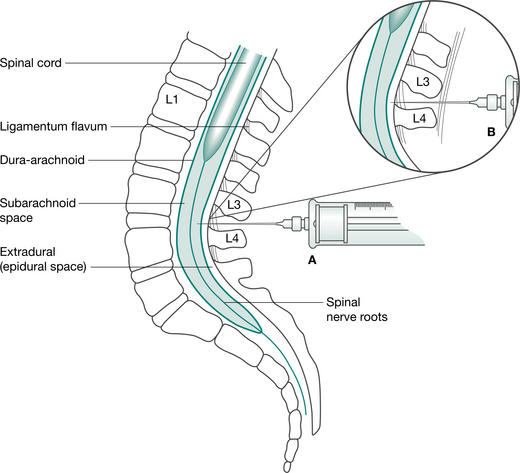
Figure 12.2 Spinal and epidural anaesthesia. (A) Position of needle in subarachnoid space for spinal anaesthesia. (B) Position of needle in epidural space for epidural anaesthesia/analgesia.
Non-pharmacological methods for managing postoperative pain are generally perceived to be adjuncts to pharmacological methods, but there is growing evidence for the value of their contribution (National Health and Medical Research Council 2005). Education including providing procedural information about treatment (such as provided by physiotherapists preoperatively), combined with sensory information describing the sensory experiences a patient may expect and information regarding coping strategies, may be effective in reducing negative affect, pain medication use and improving clinical recovery after surgery (National Health and Medical Research Council 2005). Preoperative education may encourage a more positive attitude toward pain relief, although there is no evidence that preoperative education about pain has any effect on postoperative pain after cardiac surgery (National Health and Medical Research Council 2005). Implementation of an acute pain management service may also improve pain relief.
Measurement of pain
Pain is difficult to measure as it is a purely individual and sensory experience (Dodson 1985). Postoperative pain is acute and initiated by tissue injury during surgery, but reduces with time and the natural healing process. The measurement of pain is often necessary to assess the results of an intervention or to measure intensity of pain, such as postoperative pain. Regular measurement of pain leads to improved acute pain management. Most measures of pain are based upon self-report but can provide sensitive and consistent results if performed properly (National Health and Medical Research Council 2005).
Several different instruments may be used to measure pain, these include:
Except for the McGill pain questionnaire, these methods are unidimensional; that is, they only measure intensity of pain in absolute terms or changes in pain intensity. Verbal scales may use words that have different meanings for different people, such as mild, moderate or severe pain. These categorical scales are quick and simple but less sensitive than numerical rating scales such as the visual analogue scale (VAS) (National Health and Medical Research Council 2005). The McGill questionnaire uses 20 groups of two to six words and the patient is asked which word in each group best describes their pain. While this offers more valid information than VDS, it is very time consuming and it not used extensively for the measurement of acute postoperative pain. Verbal rating scales are commonly used in clinical practice and use of the VAS is the most commonly used method. Visual analogue scales usually consist of a straight line, 10 cm long, the extremes of which are taken to represent the limits of the subjective experience being measured. In the case of pain measurement, one end of the line may be defined as ‘no pain’ and the other as ‘severe pain’ or ‘worst possible pain’. The subject is asked to place a mark on the line corresponding to the severity of their pain. The distance from the mark to the end of the scale is taken to represent pain severity. The most common way to use a VAS in the study of postoperative pain is to ask the patient to score the pain they are experiencing at the time of completion of the VAS. Visual analogue scales may also be used to obtain a pain score that reflects pain or pain relief over the preceding 24 hours. Commonly, physiotherapists ask patients to rate their pain on activity in the postoperative period to provide more meaningful information. The VAS has been shown to be a linear scale for patients with postoperative pain of mild to moderate intensity. Therefore results are equally distributed across the scale so that the difference between each number on the scale is equal. It is reported that values greater than 70 mm are indicative of severe pain while values between 45 and 74 mm represent moderate pain and those between 5 and 44 mm mild pain (National Health and Medical Research Council 2005).
Box 12.1 shows the physiotherapy key points with regard to analgesia.
Box 12.1 Physiotherapy key points with regard to analgesia
 Always check vital signs, particularly respiratory rate and blood pressure, as hypotension is a common side effect of pain management. Most important in position changes.
Always check vital signs, particularly respiratory rate and blood pressure, as hypotension is a common side effect of pain management. Most important in position changes. If a patient has had a spinal block or epidural alwaysassess motor and sensory function of the lower limbs, especially before upright mobilization.
If a patient has had a spinal block or epidural alwaysassess motor and sensory function of the lower limbs, especially before upright mobilization. Ask the patient if they need to use their PCA or PCEA before a physiotherapy treatment session or ask about a bolus dose of analgesia before treatment.
Ask the patient if they need to use their PCA or PCEA before a physiotherapy treatment session or ask about a bolus dose of analgesia before treatment. Always liaise with medical and nursing staff before treating the patient and know the local guidelines for mobilizing patients with an epidural in situ.
Always liaise with medical and nursing staff before treating the patient and know the local guidelines for mobilizing patients with an epidural in situ.PCA, patient-controlled analgesia; PCEA, patient-controlled epidural analgesia
Effects of the surgical process on respiratory function
The intra- and postoperative periods are frequently associated with alterations in pulmonary function (Craig 1981). Furthermore, altered physiological function of the respiratory system is an expected finding, especially after upper abdominal and thoracic surgery (Ford et al 1993). The combined effects of the GA, postoperative pain, recumbency, immobility and administration of drugs after surgery lead to several respiratory abnormalities.
Lung volumes
The characteristic abnormality of respiratory mechanics following major surgery is a restrictive ventilatory defect manifest by changes in vital capacity (VC) and functional residual capacity (FRC) (Wahba 1991). The VC can reduce to 40% of preoperative values, while the FRC may gradually reduce to be 70% of preoperative value at 24 hours postoperatively. These changes may persist for 5–10 days following surgery (Craig 1981). The timing of greatest reduction in FRC, while varying between studies, is generally on the first or second postoperative day. In morbidly and even mildly obese patients there is a significant reduction in FRC compared with patients within the ideal weight range (Jenkins & Moxham 1991). Although most other lung volumes also reduce following major surgery, it is thought that the reductions in FRC represent the most clinically important changes because of the functional consequences. The alteration in lung volumes after lower abdominal and laparoscopic surgery is less pronounced.
Functional residual capacity and closing capacity
FRC may be affected by a number of factors. It is linearly related to height and is 10% less in females for the same body height (Nunn 1993). The FRC is affected by gravity and therefore body position. In supine the abdominal contents push the diaphragm cephalad, reducing intrathoracic volume and FRC. In normal subjects FRC is reduced by approximately 500–1000 ml upon adopting the supine position (Macnaughton 1994). It is highest in standing and reduces with recumbency (Fig. 12.3). A change in body position from bed to sitting in a chair increased the FRC by 17% in 10 patients following upper abdominal surgery.
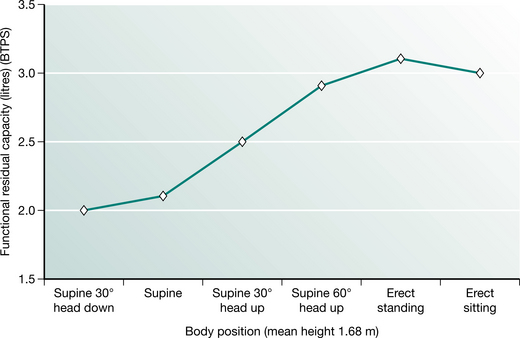
Figure 12.3 Functional residual capacity in different body positions.
(Adapted from Nunn 1993 p 55 with permission from Butterworth-Heinemann, Oxford)
The relationship of FRC with the closing capacity of the lungs explains the functional significance of perioperative reductions in FRC. Closing capacity (CC) is defined as the lung volume at which dependent airways begin to close, or cease to ventilate (Macnaughton 1994). The small airways (less than 1.0 mm diameter) in the periphery of the lung are not supported by cartilage and are therefore influenced by transmitted pleural pressures. Normally the transpulmonary pressure or distending pressure is less than atmospheric, producing a positive pressure, which distends the lungs. Breathing at lower lung volumes produces a higher pressure in gravity-dependent lung regions. This produces a negative distending pressure and causes small, unsupported airways to narrow or close, resulting in reduced ventilation. The relationship between FRC and CC is an important determinant of dependent airway closure. If the CC exceeds the FRC, as it does during tidal breathing in the upright position in people over 70 years of age, then dependent lung regions underventilate, resulting in 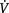 /
/ mismatch and hypoxaemia. The relationship between CC and FRC is responsible for the reduction in arterial oxygen tension (PaO2) with increasing age. Closing capacity increases with age due to loss of elastic lung tissue. It also increases in chronic lung disease and with cigarette smoking, due also to changes in lung elasticity. In combination with these increases in CC, any factor which at the same time reduces FRC will significantly affect the relationship between the two volumes, such that dependent airway closure occurs during normal tidal breathing (Macnaughton 1994) (Fig. 12.4).
mismatch and hypoxaemia. The relationship between CC and FRC is responsible for the reduction in arterial oxygen tension (PaO2) with increasing age. Closing capacity increases with age due to loss of elastic lung tissue. It also increases in chronic lung disease and with cigarette smoking, due also to changes in lung elasticity. In combination with these increases in CC, any factor which at the same time reduces FRC will significantly affect the relationship between the two volumes, such that dependent airway closure occurs during normal tidal breathing (Macnaughton 1994) (Fig. 12.4).
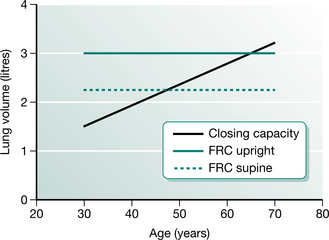
Figure 12.4 The relationship between functional residual capacity (FRC) and closing capacity.
(Adapted from Nunn 1993 p 56 with permission from Butterworth-Heinemann, Oxford)
Anaesthesia, surgery and recumbency reduce FRC. The reduction in FRC together with possible increases in CC in some subjects may account for the regional changes in ventilation that occur perioperatively and lead to reduced compliance, altered ventilation and perfusion (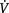 /
/ ), arterial hypoxaemia and atelectasis from absorption of trapped gas behind closed airways. Postoperative hypoxaemia is inevitable, but usually subclinical, and like FRC, is usually lowest on the first and second days after surgery. Therefore supplemental oxygen is routinely given postoperatively (Craig 1981, Fairshter & Williams 1987).
), arterial hypoxaemia and atelectasis from absorption of trapped gas behind closed airways. Postoperative hypoxaemia is inevitable, but usually subclinical, and like FRC, is usually lowest on the first and second days after surgery. Therefore supplemental oxygen is routinely given postoperatively (Craig 1981, Fairshter & Williams 1987).
The mechanisms for the perioperative reductions in FRC and VC in upper abdominal surgery (UAS) are thought to be: mechanical disruption of the thorax and abdomen, absence of spontaneous sighs, shallow breathing, pain and inhibition of diaphragmatic function (Wahba 1991). Abdominal distension, presence of a nasogastric tube and the use of analgesics to control postoperative pain may impact on ventilatory function.
Mucociliary clearance
Mucociliary clearance is a major function of the airway epithelium. This important function depends both on the physicochemical properties of the airway mucus and on the activity of the cilia (Kim 1997). Anaesthesia, intubation, mechanical ventilation reduced lung volumes and reduced cough effectiveness perioperatively present a significant insult to the mucociliary escalator. A summary of the perioperative contributions to altered mucociliary clearance is given in Box 12.2.
Box 12.2 Factors promoting postoperative mucociliary dysfunction
(Adapted from Denehy and van de Leur 2004)
Respiratory muscle function
Diaphragmatic excursion has been shown in research literature to be reduced following abdominal and thoracic surgery and postoperative pain may contribute to diaphragm dysfunction. This is associated with reduced VC, changed pattern of ventilation to predominately rib cage movement rather than abdominal movement and postoperative hypoxaemia. The reduced function may last up to 1 week following surgery. Reflex inhibition of the phrenic nerve may occur after UAS, but research findings are inconsistent. The breathing pattern observed after abdominal surgery may act as a protective mechanism by splinting the abdomen, allowing faster healing of abdominal incisions and reducing the risk of peritoneal infection (Ford et al 1993).
Postoperative pulmonary complications
One of the aims of physiotherapy in the perioperative period is to counteract the adverse pulmonary changes produced as a result of the surgical process (Stiller et al 1994). The factors described above; dependent atelectasis, hypoxaemia and altered V , although occurring in most patients, can lead to clinically significant PPC in some patients after surgery. Two basic theories have been proposed to explain the pathogenesis of PPC following major surgery. Firstly, regional hypoventilation and blockage of airways by mucus and, secondly, absorption of alveolar air distal to a mucus plug in the proximal airways, may lead to eventual collapse unless fresh air enters through collateral channels (Marini 1984). Regional hypoventilation results from reductions in FRC and the altered relationship between FRC and CC together with diaphragmatic dysfunction postoperatively as discussed above. The precise sequence and relative contributions of each of the two mechanisms for developing PPC is unclear. It is possible that they vary among patients and that both alveolar hypoventilation and secretion plugging coexist to contribute to postoperative lung changes (Denehy & Browning 2007). Other risk factors that may predispose to increased risk of mucus plugging may be a history of cigarette smoking, weak cough, prolonged intubation, presence of a nasogastric tube and prolonged postoperative atelectasis (Smith & Ellis 2000).
, although occurring in most patients, can lead to clinically significant PPC in some patients after surgery. Two basic theories have been proposed to explain the pathogenesis of PPC following major surgery. Firstly, regional hypoventilation and blockage of airways by mucus and, secondly, absorption of alveolar air distal to a mucus plug in the proximal airways, may lead to eventual collapse unless fresh air enters through collateral channels (Marini 1984). Regional hypoventilation results from reductions in FRC and the altered relationship between FRC and CC together with diaphragmatic dysfunction postoperatively as discussed above. The precise sequence and relative contributions of each of the two mechanisms for developing PPC is unclear. It is possible that they vary among patients and that both alveolar hypoventilation and secretion plugging coexist to contribute to postoperative lung changes (Denehy & Browning 2007). Other risk factors that may predispose to increased risk of mucus plugging may be a history of cigarette smoking, weak cough, prolonged intubation, presence of a nasogastric tube and prolonged postoperative atelectasis (Smith & Ellis 2000).
The definition of PPC can include atelectasis or pneumonia (atelectasis and collapse are terms that are often used interchangeably). Significant PPCs have been defined as complications that alter the patient’s clinical course (O’Donohue 1992). PPC may be defined by using radiological and bacteriological criteria, clinical signs and symptoms, or a combination of these (Pasquina et al 2006). The definition of PPC impacts on the incidence obtained. Using only radiological evidence of atelectasis for example, gives a higher incidence of complications than using a combination definition. To date, no valid definition has been established and as a result the definitions used are variable.
The clinical signs and symptoms of PPC may include the following:
 arterial desaturation (measured using a pulse oximeter), often defined as <90% on two consecutive days
arterial desaturation (measured using a pulse oximeter), often defined as <90% on two consecutive days radiological evidence of atelectasis or pneumonia (routine chest X-rays are not common after surgery)
radiological evidence of atelectasis or pneumonia (routine chest X-rays are not common after surgery) raised oral temperature (febrile is >37.5°C), often defined as >38°C on more than one consecutive day as a raised temperature on the first day after surgery is a common finding resulting from the surgical insult
raised oral temperature (febrile is >37.5°C), often defined as >38°C on more than one consecutive day as a raised temperature on the first day after surgery is a common finding resulting from the surgical insult abnormal lung auscultation (given that the majority of patients have some dependent atelectasis, reduced breath sounds are commonly found in the first 2 days after surgery)
abnormal lung auscultation (given that the majority of patients have some dependent atelectasis, reduced breath sounds are commonly found in the first 2 days after surgery) prescription of an antibiotic specific for lung infection (many patients are given routine antibiotics immediately postoperatively depending on type of surgery).
prescription of an antibiotic specific for lung infection (many patients are given routine antibiotics immediately postoperatively depending on type of surgery).Many of these signs and symptoms occur in patients after major surgery and a combination of three or four, occurring together, could be considered as clinically significant PPC.
The incidence of postoperative atelectasis is reported to be 70% following UAS but the incidence of clinically significant complications is reported to range from 5% to 20% (Denehy et al 2001). There is a lower reported incidence following cardiac surgery of around 5%–7% (Brasher et al 2003, Pasquina et al 2003). The incidence following thoracic surgery varies but is reported to be 8–32% (Gosselink et al 2000). A higher incidence of 16% (Law & Wong 2006) to 30% (Gosselink et al 2000) is reported in patients following oesophageal surgery.
Risk factors for postoperative pulmonary complications
Advances in operative technique and postoperative patient management have led to surgery of increasing complexity being performed routinely in patient populations with more severe comorbidities. Estimation of surgical risk is therefore important for all health professionals involved in the management of surgical patients. Surgical risk is the probability of morbidity and mortality secondary to the presence of pre-, intra- and postoperative risk factors. This discussion will be limited to the development of PPC that most affects physiotherapists. Assessment of risk of developing PPC is important for the physiotherapist as it allows prioritized respiratory care for high-risk subjects and more appropriate use of often scarce resources in physiotherapy staffing. Surgical complications such as wound breakdown, bleeding, renal failure and other respiratory problems such as development of pulmonary embolus (PE) will not be discussed in detail. However, it is important to consider a PE in differential diagnosis of a respiratory complication such as pneumonia. The symptoms of both may be quite similar and include pleuritic chest pain, moderate to severe hypoxaemia, breathlessness and fever. Anticoagulation with heparin and then warfarin is indicated for PE. The identification of any pain in the calf on assessment is also important as it may indicate a deep vein thrombosis (DVT), although clinical diagnosis of DVT is unreliable with 50% of patients with DVT on venography showing symptoms.
Several patient characteristics are associated with an increased risk of developing complications. There is a large volume of literature published on this topic. In a systematic review (Fisher et al 2002), 40 variables were reported as possible risk factors for patients having non-thoracic surgery. There have been attempts to find a group of risk factors (model) that predict most complications in a particular patient population; several different models currently exist and none provide perfect prediction. The most common patient, operation and postoperative factors considered to increase risks of developing a PPC are described below. In physiotherapy research, a weighted model was developed to predict the risk of patients having abdominal surgery developing PPC (Scholes et al 2006). Five main risk factors (all these risk factors occurring together in one patient) were identified in this model, which predicted 82% of patients who developed a PPC in a population of 268 patients having upper abdominal surgery. Patients predicted as high risk were eight times more likely than those predicted to be at low risk of developing a PPC. The risk factors identified were:
In addition to the risk factor model above, a systematic review of non-cardiopulmonary surgery (Smetana et al 2006) reports good evidence for the following risk factors to increase incidence of PPC:
Procedure-related risk factors that were supported by good evidence were thoracic, abdominal, emergency and prolonged surgery. There is fair evidence for significant intraoperative blood loss, oesophageal surgery and abnormal chest radiograph. However, in this systematic review, which presents the highest level of evidence, there was good evidence that the following were not important risk factors: obesity, asthma, hip and gynaecological surgery (lower abdominal surgery). These results challenge some traditional views, especially that of obesity being considered a risk factor. The results from this systematic review support the risk factors identified in the model by Scholes et al (2006). In patients having oesophageal surgery, age, operation duration and location of tumour in the proximal oesophagus were identified in one study as risk factors for PPC in 421 patients (Law et al 2004).
The ASA score (1–5) divides patients into five groups and collectively rates patient risk from anaesthesia. It was developed as a standardized way for anaesthetists to convey information about the patients’ overall health status and to allow outcomes to be stratified by a global assessment of their severity of illness. In practice, the ASA score may be the only overall documentation of preoperative condition that is used widely. Generally, the attending anaesthetist ascribes a score to each patient upon preoperative assessment.
The classification of physical status recommended by the House of Delegates of the American Society of Anesthesiologists (1963) is:
Box 12.3 shows key points in the surgical process.
Box 12.3 Key points in the surgical process
 Functional residual capacity (FRC) is reduced perioperatively as a result of anaesthesia, surgery and recumbency.
Functional residual capacity (FRC) is reduced perioperatively as a result of anaesthesia, surgery and recumbency. The altered relationship between FRC and closing capacity is an important determinant of dependent airway collapse.
The altered relationship between FRC and closing capacity is an important determinant of dependent airway collapse. Ventilation/perfusion (
Ventilation/perfusion ( /
/ ) mismatch and arterial hypoxaemia commonly occur after major surgery, although increases in CO2 are rare unless patients are narcotized.
) mismatch and arterial hypoxaemia commonly occur after major surgery, although increases in CO2 are rare unless patients are narcotized.TYPES OF SURGERY
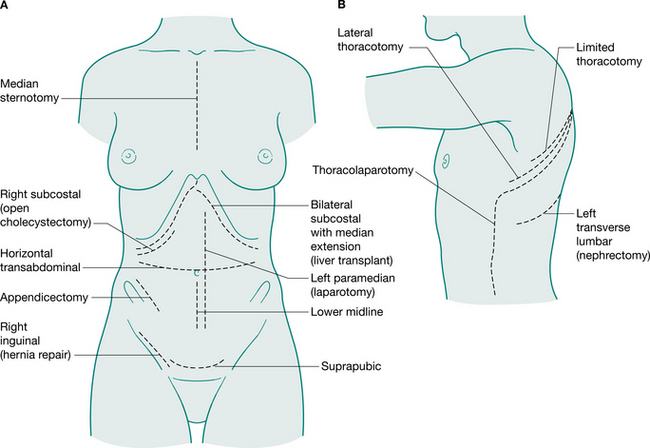
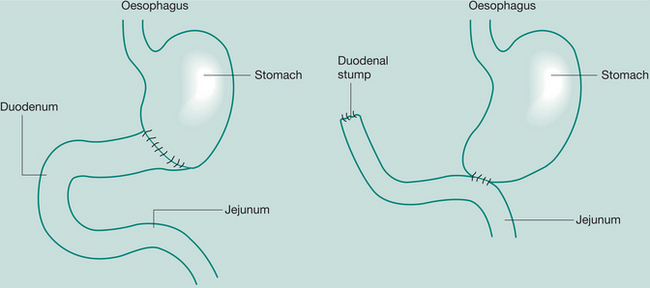
Generally, surgical incisions are placed to optimize access to the target organ (Fig. 12.5). Understanding surgery involves an appreciation of the anatomy of the abdominal and thoracic organs and the muscles and bony structures surrounding them. An appreciation of nomenclature also aids understanding descriptions of operations. The prefix of words can help to locate the surgery: for example, enter- relates to small intestine, gaster- to stomach, pneum- to lung. Surgical procedures may also be named for the person who first performed or reported them: for example, Nissen fundoplication, a wrap of fundus of the stomach around the intra-abdominal oesophagus; a Whipple’s procedure, a pancreaticoduodenectomy; and Hartmann’s procedure, a sigmoid colectomy with a colostomy.
Understanding ‘endings’ of words also helps to work out the type of surgery (Table 12.3).
Table 12.3 Surgical terminology
| Ending | Meaning | Example |
|---|---|---|
| -tomy | Cut, cut out | Thoracotomy |
| -ect | Outside or extra | Gastrectomy (removal of stomach) |
| -stom | Mouth | Colostomy (an opening between the colon and skin) |
| -rraphy | Sew or suture | Herniorraphy (hernia repair) |
| -plasty | Mould or shape | Thoracoplasty (removal of ribs to collapse underlying diseased lung which reshapes the thorax) |
| -plico | To fold | Fundoplication (a wrap or fold of fundus of stomach) |
| -scop | To look at | Mediastinoscopy (look at mediastinum) |
Abdominal surgery
Understanding abdominal surgery requires an appreciation of the anatomy of abdominal organs. The abdomen is a cavity lined by peritoneum and surrounded by muscle and skin. Abdominal organs may be intraperitoneal, and thus nourished via a mesentery (e.g. stomach), or extraperitoneal (e.g. pancreas). Some organs are both (e.g. liver).
Access to abdominal organs may be via their lumen, when there is one. This is called endoscopy (Greek skopein × to look at) where a fibre-optic telescope containing a light source and instruments are inserted. Examples are gastroscopy, colonoscopy and endoscopic retrograde choliangiopancreatography (ERCP). This may be used for diagnosis or therapy.
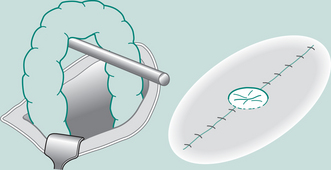
Laparoscopy involves insufflation of the peritoneal cavity with CO2 gas (pneumoperitoneum), insertion of a camera through a 5–10 mm subumbilical incision and inspection of the abdominal contents using the transmitted picture and a monitor. Commonly three ports are used to introduce the instruments and perform the procedure. The technique is performed under general anaesthesia and the most common laparoscopic technique is for the removal of the gall bladder (cholecystectomy), but many other procedures are now performed this way including hernia repair, appendicectomy, splenectomy and oophorectomy (Harris 2006). The term minimal access surgery has been used to reflect the fact that the operations themselves are the same, but the surgical approach is less invasive, which impacts on the postoperative recovery of the patient. It has been well established in the literature that laparoscopic cholecystectomy is associated with a low incidence of PPC (Sharma et al 1996).
Until the publication of a large randomized controlled trial (RCT) in 2004, there was a moratorium placed on laparoscopic cancer surgery owing to concerns regarding the oncological outcomes. This large (1200 cases) RCT reported that that there were no differences found in tumour recurrence using laparoscopic compared with open surgery. The benefits of this minimal access surgery have been reported, in a systematic review, to lead to lower morbidity, reduced postoperative pain, faster recovery of respiratory function, earlier recovery of bowel function and shorter length of hospital stay (laparoscopic patients were discharged a mean of 1.7 days earlier). Laparoscopic surgery, however, took 30% longer to perform (Tjandra & Chan 2006). There is an expanding interest in laparoscopic colorectal surgery but more research on the longer-term outcomes and standardizing surgical expertise are needed. Indeed, the more recent trend toward early postoperative rehabilitation also reduces length of hospital stay with improved patient quality of life after surgery. Therefore the integration of early ‘fast tracking’ rehabilitation with laparoscopic colorectal surgery may be required to fully evaluate the justification of the application of this surgery on a larger scale (Kehlet & Kennedy 2006).
A narrative literature review conducted by Olsen (2000) concluded that routine prophylactic chest physiotherapy is not necessary after laparoscopic upper gastrointestinal surgery such as fundoplication and vertical banded gastroplasty. The efficacy of physiotherapy in other forms of laparoscopic surgery such as colorectal surgery has not been investigated. A survey found that 58% of physiotherapists in Australian hospitals where laparoscopic colorectal surgery is performed routinely assess and treat these patients postoperatively (Browning, personal communication, 2006). However, future research examining the need for physiotherapy in this patient group is recommended.
Where minimally invasive procedures are not appropriate or possible, the abdominal cavity may be opened (laparotomy) through a variety of incisions placed to maximize access to the target organ. A midline incision is the most often used, as it allows access to most areas and may be easily extended to allow access to the whole of the abdominal cavity. The length and site of the incision has important implications for the amount of pain the patient may experience postoperatively and for the methods of controlling pain. Closing the surgical wound using different types of sutures may be achieved using continuous methods as in abdominal wounds or interrupted methods as in sternal wires. Staples are also used – they are faster but more expensive and produce a scar with a poorer cosmetic result than sutures. Many of the surgical complications that may occur are related to the incision, such as wound infection or dehiscence.
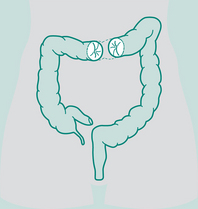
Abdominal surgery includes all operations involving the abdominal viscera. Colorectal and hepatobiliary surgery are most commonly encountered by physiotherapists, since they generally involve an incision above the umbilicus and are considered UAS (Celli et al 1984). Risk of PPC is greater in UAS than in lower abdominal operations such as hysterectomy. Many colorectal procedures are performed to remove cancer, colorectal cancer being the most common cause of cancer death (in non-smokers) in many Western countries. Conditions such as diverticulitis and ulcerative colitis are also common reasons for surgical intervention. Hepatobiliary procedures are performed for both malignant and benign diseases of the biliary tree. These include operations involving the liver, pancreas, spleen, duodenum, bile duct and gall bladder. Box 12.4 describes several commonly encountered abdominal operations.
Box 12.4 Some commonly performed abdominal surgical procedures
Right hemicolectomy
Indications: Ca right colon, terminal ileum
Incision: Right paramedian, midline, right oblique
Continuation restored by: Anastomosis of ileum to transverse colon
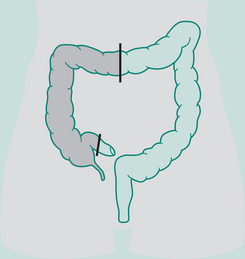
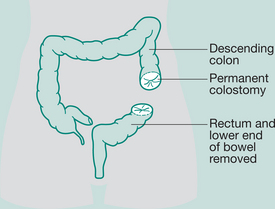
Abdomino-perineal (AP) resection/sigmoid colostomy
Indication: Ca lower portion large bowel and rectum, ulcerative colitis
Ileostomy
Most are permanent (small bowel)
Indications: Ulcerative colitis, Crohn’s disease, Ca of bowel
Incision: Left paramedian or midline
Operation: Continent pouch ileostomy
Reservoir constructed out of distal ileum: removal of large colon and rectum
Outlet from reservoir is arranged as a valve so that fluid cannot escape on to the abdominal wall
Indications: Ca, trauma, Crohn’s disease, to rest bowel
Incision: Depends on site of colostomy
Operation: Stoma formed from colon. Names according to section of colon it is situated in, e.g. ascending
Types of sigmoid colostomy permanent (performed for abdomino-perineal resection – Ca rectum)
Double-barrelled colostomy
Both loop distal and proximal are opened, may be permanent or temporary depending on disease
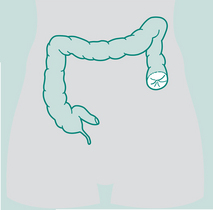
Loop colostomy
Usually formed in transverse colon. Loop of bowel brought out through incision, plastic
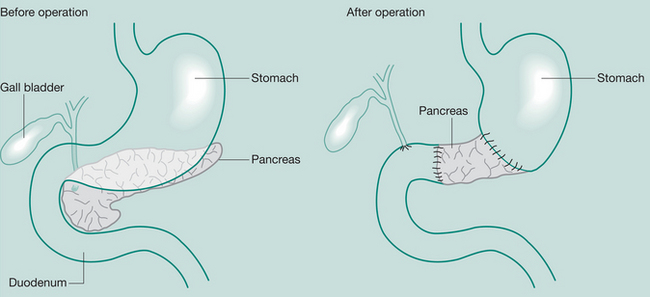
Whipple’s procedure (pancreaticoduodenectomy)
May be required when severe pancreatitis is confined to the head of the gland or in Ca
Resection of the distal stomach, common bile duct, duodenum, gall bladder and the pancreas to the mid-body
Gastrectomy
Removal of portions of the stomach
Indications: (partial) peptic ulcer, Ca distal stomach; (total) Ca
Incisions: Upper vertical – midline, paramedian, transverse or oblique.
If thoracic extension for oesophagogastrectomy is involved, a left thoracotomy is performed.
If upper oesophagus is involved, a right thoracotomy is used
Thoracic surgery
Thoracic surgery encompasses topics related to disorders of the chest wall, pleural space, lungs, oesophagus, mediastinum and chest trauma (Smith 2006). It has developed extensively in the past 50 years and now also includes lung transplantation (Chapter 15), video-assisted thoracoscopic surgery (VATS) and lung volume reduction surgery.
Most commonly, removal of part or all of the lung is performed to remove a carcinoma. Of over 3000 lung resections performed in 27 European centres, two-thirds of these were for lung cancer (Berrisford et al 2005). In Australia, the total number of new cases of lung, tracheal and bronchial cancers in 2005 was 9000 and this is predicted to increase (particularly in females) by 30% in 2011 with an ageing population. Lung cancer was the fourth most common cancer in both men and women in Australia in 2003 and presents a significant disease burden (AIHW & AACR 2007). It is reported to be the leading cause of cancer-related death in men and women worldwide (Hassan 2006).
Carcinoma of the lung
Cigarette smoking is the single most common predisposing factor for lung carcinoma, but other factors such as environmental or occupational exposure to hydrocarbons or asbestos are also implicated. There are several different pathological types of lung carcinoma; these are divided into non-small cell and small cell carcinoma. The non-small cell types are squamous cell, adenocarcinoma, large cell and adenosquamous carcinoma. Of these, squamous cell and adenocarcinoma are most common, making up approximately 80% of all lung cancers (Smith 2006). Small cell carcinomas are the most malignant and make up about 10% of presentations of lung cancers. Extrathoracic spread is common at the time of presentation.
Thoracic clinical signs and symptoms of lung cancer include: cough, haemoptysis, chest pain, hoarse voice (if recurrent laryngeal nerve involved), shortness of breath, arm pain and weakness. There may be other symptoms depending on the extent and site of metastases at presentation, such as bone, liver or central nervous system involvement. Physical findings may include loss of weight, fatigue, finger clubbing, pleural effusion or lung collapse. Several investigations may be undertaken to determine the cell type, extent and location of the carcinoma; these include chest radiograph, pulmonary function tests, computed tomography (CT) scan, needle biopsy of tumour under CT control, bronchoscopy, mediastinoscopy, positron emission tomography (PET) scan, ventilation/perfusion scan, and sputum cytology.
Lung cancer is staged depending on the size and location of the tumour (T), the amount of spread to lymph nodes in the thorax (N), and the presence or absence of metastases (M). Each of these is further divided and scored from 0 to 4 (for example T1N0M0). The TNM classification is used internationally for all non-small cell lung cancers to give information about prognosis and to guide therapy (Hassan 2006). The most prognostic indicator in lung cancer is the extent of the disease. Early-stage cancer lung cancer (T1N0M0 and T2N0M0) have a potentially high curability with surgery (Hassan 2006). The common sites for distant spread are to mediastinal lymph nodes, brain, liver, bones, kidneys and pancreas.
For those presenting with localized tumours and an adequate respiratory reserve, surgical resection offers the only curative treatment. However, only about one-third of presentations are considered for surgery. The 5-year survival (number of patients still surviving at 5 years) from curative surgery is about 30%, but the overall 5-year survival is only 15% (Smith 2006). Radiotherapy may be given and is usually palliative, as is chemotherapy. Both may also be used before and after surgical resection. For small cell carcinoma, multiagent chemotherapy is the only current treatment.
Surgical resection
Thoracotomy allows access to the lung. A full posterolateral thoracotomy involves an extensive incision through the 5th or 6th intercostal space (Fig. 12.5) including muscular division of trapezius, latissimus dorsi, lower rhomboids, serratus anterior, the intercostals and erector spinae. A rib retractor is used to separate the ribs and sometimes a partial rib resection is performed to improve exposure of the lung. This incision is used less commonly but remains the standard approach. A lateral (axillary) thoracotomy is a limited muscle sparing incision made between the anterior and posterior axillary lines and is used when limited access is required as in surgery of the pleura such as pleurectomy. Anterior thoracotomy involves a small incision below the breast and is used for open lung biopsy. The common thoracic surgical procedures are defined in Box 12.5.
Box 12.5 Common thoracic surgical procedures
Minimally invasive surgery using thoracoscopy is now used where possible. This technique offers less surgical trauma and reduced recovery time and is the thoracic equivalent of laparoscopy. Video-assisted thorascopic surgery (VATS) simultaneously uses a light source, camera and telescope through several ports of access made by 2 cm incisions in the chest wall. Commonly three ports are used: one for the telescope, light and camera and two for surgical instruments. This technique is used commonly for pleural surgery such as pleurectomy performed for recurrent pneumothorax, as well as increasingly for smaller lung resection procedures.
Oesophageal surgery
Oesophageal surgery is generally performed for carcinoma and has a high mortality usually due to late disease presentation. Surgery may be performed after combined treatment of chemotherapy and radiotherapy or may be palliative to provide symptom relief. Patients generally present with symptoms of dysphagia (problems with swallowing); other common symptoms include loss of weight, regurgitation and substernal pain. The sites of metastases are similar to the lung and include cervical lymph nodes, lungs, liver, bones and other viscera.
The surgical approach for carcinoma of the middle and lower thirds of the oesophagus is by laparotomy (to mobilize and prepare the stomach) and right thoracotomy (to resect the oesophagus); this is commonly known as an Ivor Lewis oesophagectomy. The stomach is delivered up into the thorax to anastomose with the remaining proximal oesophagus through the diaphragmatic hiatus. In carcinoma of the upper oesophagus, a neck incision is used in conjunction with the laparotomy and thoracotomy to allow anastomosis (Law & Wong 2006).
Placement of a prosthetic tube or now more commonly a stent to restore luminal patency provides symptom relief of dysphagia for carcinoma that is not otherwise suitable for other treatments. Laser therapy to vaporize the tumour is also used.
Surgery of the pleura
Pleural surgery is commonly performed for recurrent pneumothorax, pleural effusion and empyema.
Pneumothorax
Pneumothorax is defined as the presence of air in the pleural space. The common types of pneumothorax are primary spontaneous, secondary spontaneous, tension and traumatic (Smith 2006). Primary spontaneous pneumothorax is the most common and usually results from the rupture of a tiny bleb at the apex of the lung. Clinical signs may include acute chest pain and shortness of breath or shortness of breath on exertion. It commonly occurs in tall thin individuals of either sex. The size of the pneumothorax will usually dictate the management approach. This may include observation and repeat chest radiographs, needle aspiration of air directly from the pleural space or insertion of intercostal drainage. Spontaneous pneumothorax may be recurrent, with figures suggesting that about 30% recur. After a second episode this figure increases to 70%. Surgical management is usually indicated after two pneumothoraces on the same side and this most commonly involves pleurectomy or pleurodesis to allow the visceral pleura to adhere to the parietal, which thereby obliterates the ‘potential’ pleural space.
Secondary pneumothorax occurs as a result of underlying lung disease such as COPD or lung abscess. Traumatic pneumothorax occurs after penetrating trauma such as by a rib fracture, knife or gunshot wound. Traumatic pneumothoraces are usually accompanied by haemothorax, which is defined as an accumulation of blood in the pleural space. Bleeding may be from the chest wall, heart, major vessels or lungs. When it occurs in conjunction with a pneumothorax it is called a haemopneumothorax. Lung contusion is also common in traumatic lung injury and involves injury to lung parenchyma, oedema and blood collecting in the alveoli and an inflammatory reaction to blood components in the lung. Gas exchange may be significantly affected by contusion, which may lead to acute respiratory distress syndrome (Trauma.org 2004).
Tension pneumothorax results when the site of air leak acts as a one-way valve so that air enters the pleural space during inspiration but cannot escape during expiration. The volume of air and pressure in the hemithorax increase, resulting in compression of the ipsilateral lung, mediastinal shift away from the side of pneumothorax including shift of the trachea, and possible kinking of the great vessels if the mediastinal shift is large. Clinical signs include deviation of the trachea, absent breath sounds, acute respiratory distress, raised jugular venous pressure and hypotension, Tension pneumothorax can be life threatening and should be relieved as soon as possible by insertion of a large-bore needle to let the air escape under pressure followed by insertion of an intercostal drain (Smith 2006). Figure 12.6 shows different types of pneumothorax and flail chest.
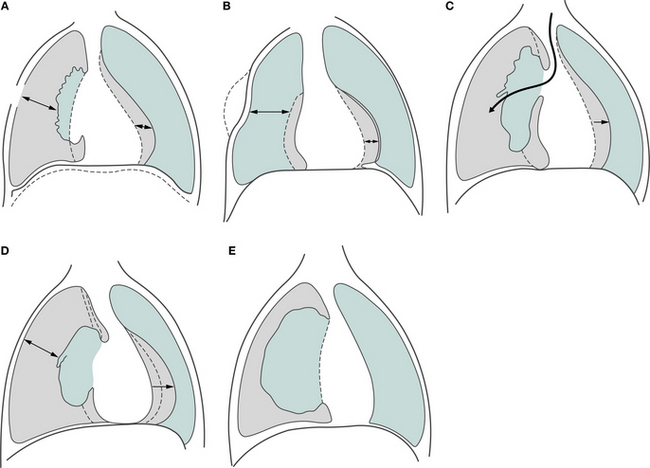
Figure 12.6 (A) Open pneumothorax secondary to chest trauma making respiration totally ineffective as air is sucked in and out of the open wound. (B) Flail chest secondary to multiple anterior and posterior rib fractures, resulting in chest wall instability. (C) Tension pneumothorax allowing air to enter the pleural space with each inspiratory breath. (D) Tension pneumothorax on expiration. The hole in the lung closes on expiration, resulting in a build-up of pressure in the pleural space with mediastinal shift. (E) Partial pneumothorax. Partial collapse of the lung away from the chest wall but not under tension.
Following open thoracic surgical procedures (including VATS), intercostal drainage tube(s) are positioned in the pleural space before surgical closure and connected to a closed drainage system called underwater sealed drainage (UWSD).
Key points for physiotherapy in thoracic surgery are shown in Box 12.6.
Box 12.6 Key points for physiotherapy in thoracic surgery
 Care must be taken with positioning patients after pneumonectomy. In general, patients may be positioned on to their operation side (so that the remaining lung is uppermost). However, the protocol in some units requires patients to remain sitting for the first 3–6 days until the fluid in the hemithorax which replaces the removed lung has become more organized (fibrous). Once this has occurred there is much less risk of fluid entering the anastomosis of the bronchial stump and potentially spilling into the remaining lung. Physiotherapists must check with individual surgeons/units regarding local protocol.
Care must be taken with positioning patients after pneumonectomy. In general, patients may be positioned on to their operation side (so that the remaining lung is uppermost). However, the protocol in some units requires patients to remain sitting for the first 3–6 days until the fluid in the hemithorax which replaces the removed lung has become more organized (fibrous). Once this has occurred there is much less risk of fluid entering the anastomosis of the bronchial stump and potentially spilling into the remaining lung. Physiotherapists must check with individual surgeons/units regarding local protocol. A serious complication of pneumonectomy is pulmonary oedema. Postoperative fluid balance is important since the entire cardiac output is directed through only one lung. A positive fluid balance together with signs of tachypnoea, tachycardia and hypoxaemia should be reported immediately.
A serious complication of pneumonectomy is pulmonary oedema. Postoperative fluid balance is important since the entire cardiac output is directed through only one lung. A positive fluid balance together with signs of tachypnoea, tachycardia and hypoxaemia should be reported immediately. After pneumonectomy, the fluid in the hemithorax is controlled by clamping and releasing the intercostal catheter (ICC). This management is different to that after other thoracic surgery where the ICCs are on gravity drainage ± suction.
After pneumonectomy, the fluid in the hemithorax is controlled by clamping and releasing the intercostal catheter (ICC). This management is different to that after other thoracic surgery where the ICCs are on gravity drainage ± suction. Patients having oesophageal surgery are often undernourished preoperatively and this may affect anastomotic healing and general progress after surgery.
Patients having oesophageal surgery are often undernourished preoperatively and this may affect anastomotic healing and general progress after surgery. The head-down position is usually avoided after oesophageal surgery to prevent gastric reflux which may lead to aspiration or affect the integrity of the anastomosis. Head-up positions (for example on two pillows) are preferred.
The head-down position is usually avoided after oesophageal surgery to prevent gastric reflux which may lead to aspiration or affect the integrity of the anastomosis. Head-up positions (for example on two pillows) are preferred. Positive pressure should be applied with caution and not at all unless a functioning nasogastric tube is in situ and on free drainage.
Positive pressure should be applied with caution and not at all unless a functioning nasogastric tube is in situ and on free drainage.Underwater seal drainage units are a system used specifically to drain air and/or fluid from the thoracic cavity in order to regain and/or maintain re-expansion of the lung by re-establishing normal negative pressure in the pleural space. Effective gas exchange will only occur if the lungs are able to expand sufficiently to allow adequate ventilation. The pleural membranes assist with this vital function. Each lung sits in its own pleural cavity and is attached to the mediastinum. The exterior surface of the lung is covered by a thin, serous membrane called the visceral pleura. At the root of the lung, the visceral pleura become continuous with the parietal pleura, which line the wall of the pleural cavity. Separating the two membranes is approximately 10 ml of serous pleural fluid, produced by the pleural membranes. This fluid acts to lubricate the pleural surfaces and reduces friction between the parietal and visceral pleura during respiration.
A negative pressure exists between the visceral and parietal pleura. It acts to provide a suction between the two membranes, counteracting the tendency of the lungs to recoil. Intrapleural pressure varies slightly throughout the different phases of the ventilatory cycle. Before inspiration, the intrapleural pressure is approximately −5 cmH2O. During inspiration the chest wall expands, decreasing the negative pressure to approximately −8 cmH2O, which allows the lung to expand and air to flow inward. During expiration, the intrapleural pressure decreases to approximately −4 cmH2O, allowing the air to flow from the lung to the atmosphere.
While the intrapleural space remains intact and free of all but a small amount of pleural fluid, the negative pressure required to hold the membranes together will be maintained. However, if air or fluid of any kind is allowed to enter the pleural space, the negative pressure will be lost and the affected lung will partially or fully collapse. In such cases, an underwater seal drain may be indicated to remove the fluid or air from the pleural space, thus restoring the negative pressure necessary to allow lung expansion.
Underwater sealed drainage is indicated in the presence of any surgery or trauma where there has been a significant disruption to the integrity of the pleural space. The most common substances to enter the intrapleural space are air, blood, pus or an excess of pleural fluid. These may appear alone or in combination, and cause an increase in intrapleural pressure from negative to positive, thus terminating the suctioning effect and resulting in collapse of the lung.
Intercostal catheters (chest tubes)
A chest tube is generally made of clear pliable plastic into which a radio-opaque strip may be incorporated. The diameter of the tube will vary depending on the size of the patient and on what is being drained. A smaller drain is usually employed to evacuate air while a larger drain is used for fluids (Miller & Sahn 1987).
The location of the substance to be drained usually determines the placement of the tube. When the patient is upright, fluids in the pleural space will generally gravitate to the lower zones of the thorax, while air will usually rise to the apex. Therefore, for drainage of a pneumothorax the tube is usually inserted anteriorly in the mid clavicular line into the 2nd or 3rd intercostal space or in the mid axillary line in the 3rd to 5th space and is directed towards the apex of the thorax. When fluids are to be drained, the tube is usually inserted slightly lower, in the mid axillary line and the 6th space, and directed basally. Following surgery such as lobectomy or pleurectomy, two intercostal catheters are inserted: one is usually directed apically and one basally.
Principles of underwater seal drainage
The underwater seal
The underwater seal prevents air re-entering the pleural space. Usually, the distal end of the drain tube is submerged 2 cm under the surface level of water in the drainage (or collection) chamber. This creates a hydrostatic resistance of + 2 cmH2O in the drainage chamber.
Creation of a pressure gradient
Normal intrapleural pressure is negative. However, if air or fluid enters the pleural space, intrapleural pressure becomes positive. Air is eliminated from the pleural space into the drainage chamber when intrapleural pressure is greater than + 2 cmH2O. Thus, air moves from a higher (intrapleural) to lower pressure (within the drainage chamber) along a pressure gradient. The drainage chamber has a vent to allow air to escape the chamber, and not build up within the chamber.
Gravity
Fluids will drain by gravity into the drainage chamber, and will not spill back into the pleural space if the bottle is always kept below the level of the patient’s chest. If the bottle needs to be lifted above the chest (e.g. during a patient transfer), the tubing should be briefly double clamped as close to the patient as possible. The movement and unclamping should take place as quickly as possible to minimize clamping time.
Types of underwater seal drainage systems
One-bottle system
The simplest form of UWSD is a one-bottle system. This system can drain both air and fluid. The distal end of the drainage tube must remain under the water surface level. There is always an outlet (vent) to the atmosphere to allow air to escape. A problem with this system is that when fluid starts to fill the chamber it creates a more positive (hydrostatic) pressure due to a rise in the fluid level (Fig. 12.7). Thus it is more difficult for air to escape from the pleural space into the drainage chamber due to a reduced pressure gradient (in this situation, a two-bottle system is preferable). A single-bottle system is suitable for use with a simple pneumothorax, when the vent is left open to the atmosphere, or following a pneumonectomy when the tubing is clamped and released hourly.
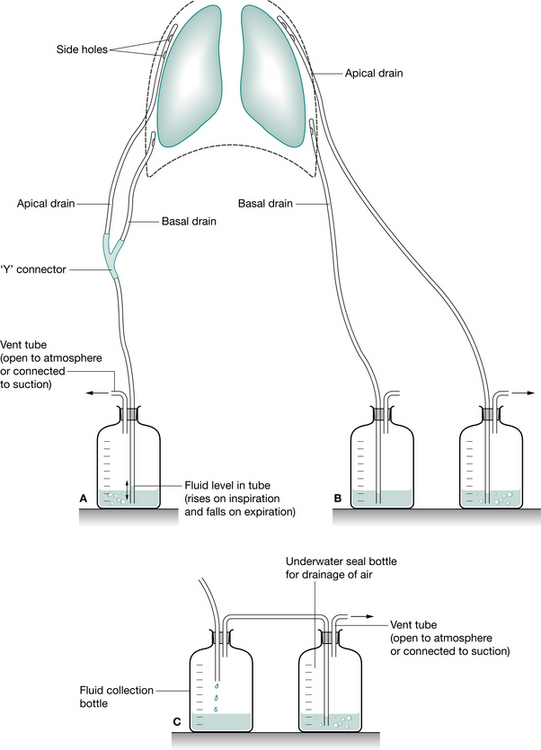
Figure 12.7 Underwater seal chest drainage. (A) Single-bottle system allowing use of one bottle via a ‘Y’ connector to drain fluid and air. (B) Two separate bottles enabling drainage of air from the apical drain and fluid from the basal drain. (C) Two-compartment drainage system where two bottles are connected in series, the first collecting fluid and the second acting as the underwater seal drainage for air.
Two-bottle system
This system is suitable for drainage of air and fluid (Fig. 12.7). The first chamber is for collection of fluid and the second is for the collection of air. As the two are separate, fluid drainage does not adversely affect the pressure gradient for evacuation of air from the pleural space. A separate chamber for fluid collection enables monitoring of volume and expelled matter (e.g. pus, blood clots).
Three-bottle system
When air or fluid needs a greater pressure gradient to move from the pleural space to the collection system (e.g. excess volume of fluid or a large air leak), suction is required. Suction may be applied via a third bottle or suction chamber. (The first two bottles are as described above.) With this system, suction is regulated by the depth of the tube under the water in the suction chamber (rather than the pressure setting at the wall). As the wall suction is applied, air is pulled into the suction chamber from the atmosphere and bubbling occurs. Note: it is the depth of water that regulates the amount of negative pressure (suction) not the wall suction pressure. The difficulties with the systems described are that they are complex and have many connections, all of which must be intact. With these systems there is a danger that the glass bottles will break or tubing becomes disconnected.
Disposable (all-in-one) three-bottle systems
This refers to the combination of the three-bottle system into one device for easier connection and management. There are many different systems used in clinical practice and when examining these systems it is useful to remember that they are simply a three-bottle system: one chamber collects fluid, one chamber collects air and has the underwater seal and the other is the suction control chamber. These systems are easier to move around and carry during patient ambulation as they have carrying handles and hooks.
Waterless ‘dry’ suction systems
There are some systems in use that operate on a waterless suction system. The level of suction is set on a dial; then the wall suction is turned on slowly to the point where an indicator denotes that suction is set at the required level. The column of water is replaced with a calibrated spring mechanism accurate to ±1 cmH2O. If suction from the wall exceeds the level set on the suction dial, a chamber opens allowing excessive sucking to be applied to the atmosphere and not the patient. It has some advantages over water-sealed drains: it is silent, has no water levels to evaporate, a high level of suction is available and it is easy to set up.
Patient assessment and underwater seal drainage systems
As part of a physiotherapy objective assessment, specific examination of a UWSD system should be performed. There are four important aspects of examination – swing, bubbling, drainage, and suction.
Swing.
The intrapleural pressure changes that occur during inspiration and expiration are transmitted to the drainage system. During inspiration, when a more negative pressure is generated, fluid moves up the tube of the drainage collection chamber and during expiration the movement is in the opposite direction. This movement of fluid along the tube during normal breathing is termed ‘wing’. It may be a small movement during quiet breathing or a large movement when the patient is coughing or during an increased inspiratory effort. If the patient is attached to suction, pressure is more regulated and swing is reduced.
Bubbling.
The presence or absence of bubbling in the underwater seal chamber of the system should be determined. Bubbling in the underwater seal chamber indicates an air leak (from the pleural space). It is important not to confuse the bubbling in the underwater seal chamber with the bubbling in the suction chamber. The bubbling in the suction chamber indicates suction is applied to the system at the correct level. The following points should be considered:
When examining the underwater sealed drainage system, ask the patient to take a deep breath and observe for swinging and bubbling. If there is no bubbling at this time, ask the patient to cough and observe for bubbling. Assessment for air leak (from the pleural space) must include observation of the underwater seal chamber during coughing.
Drainage.
It is important to note the pattern of drainage of fluid from the chest drain for two reasons:
Large amounts of haemoserous drainage may also be associated with hypovolaemia, hypotension and low haemoglobin. It is not uncommon for some drainage to occur during patient movement: e.g. transfers and exercise. Generally, drainage of more than 100 ml per hour or a sudden increase in drainage is cause for concern and medical staff should be informed if this occurs.
Suction.
As previously mentioned, it is the level of water in the suction chamber that regulates the amount of suction. When the wall suction is applied, this should result in gentle bubbling only (in the suction chamber). Vigorous bubbling will not increase the amount of suction applied and will cause evaporation of the water in the suction tube. No bubbling in the suction chamber indicates that the wall suction is not sufficiently high and needs to be increased. (It is very important to check that all tubing connections are intact before increasing the suction.) In some cases of persistent air leak, suction may be switched off to facilitate healing. The more common systems used now are integrated all-in-one disposable systems. General criteria for removal of intercostal catheters and underwater sealed drains are outlined in Box 12.7, while Box 12.8 gives key points for underwater sealed drainage.
Box 12.8 Key points for underwater sealed drainage
 The water seal must remain intact at all times. Therefore, the water seal chamber must always be in an upright position. If the water seal chamber is tipped over such that the tip of the tube is above the water surface level, air can re-enter the pleural cavity.
The water seal must remain intact at all times. Therefore, the water seal chamber must always be in an upright position. If the water seal chamber is tipped over such that the tip of the tube is above the water surface level, air can re-enter the pleural cavity. The drainage system must always be kept below the patient’s chest or clamped briefly if it must be raised above this level.
The drainage system must always be kept below the patient’s chest or clamped briefly if it must be raised above this level. If a patient with an air leak needs to lie on the drain tubing, it is important to ensure that air may continue to drain from the pleural space into the UWSD system, and that the weight of the patient does not result in occlusion of the tubing. If the latter were to occur, a tension pneumothorax could result.
If a patient with an air leak needs to lie on the drain tubing, it is important to ensure that air may continue to drain from the pleural space into the UWSD system, and that the weight of the patient does not result in occlusion of the tubing. If the latter were to occur, a tension pneumothorax could result. If the tubing becomes disconnected, it should be clamped by hand as close to the patient as possible. The drain may be immediately reconnected only if the ends of the tubing have avoided contact and contamination. A nurse should be called to help with reconnection. The risk of this occurring can be minimized by checking all connections of the chest drain before moving the patient. When a patient is getting out of bed or moving around in the bed, care should be taken that the patient does not lean on the chest tubing.
If the tubing becomes disconnected, it should be clamped by hand as close to the patient as possible. The drain may be immediately reconnected only if the ends of the tubing have avoided contact and contamination. A nurse should be called to help with reconnection. The risk of this occurring can be minimized by checking all connections of the chest drain before moving the patient. When a patient is getting out of bed or moving around in the bed, care should be taken that the patient does not lean on the chest tubing. If the chest drain falls out, the wound should be covered immediately with a gloved hand and urgent assistance called for.
If the chest drain falls out, the wound should be covered immediately with a gloved hand and urgent assistance called for. If positive pressure is being used (continuous positive airway pressure, bilevel positive airway pressure, intermittent positive pressure breathing and manual hyperinflation) in the presence of an air leak, the air leak needs to be constantly monitored as it may be exacerbated by these techniques. For example, during application, the air leak may be present on expiration when previously it was observed only on coughing.
If positive pressure is being used (continuous positive airway pressure, bilevel positive airway pressure, intermittent positive pressure breathing and manual hyperinflation) in the presence of an air leak, the air leak needs to be constantly monitored as it may be exacerbated by these techniques. For example, during application, the air leak may be present on expiration when previously it was observed only on coughing. The pain from a chest drain can be quite severe and may limit the patient’s ability to cooperate with physiotherapy treatment.
The pain from a chest drain can be quite severe and may limit the patient’s ability to cooperate with physiotherapy treatment. Patients should be able to move around with their chest drain in situ. They should be encouraged to keep the shoulder of the affected side moving, and generally discouraged from adopting protective postures.
Patients should be able to move around with their chest drain in situ. They should be encouraged to keep the shoulder of the affected side moving, and generally discouraged from adopting protective postures. Patients may be disconnected from suction and taken for a walk, but communication with the medical and nursing team is essential regarding unit policy. It is important to ensure that the underwater sealed drainage system is kept below the level of the chest at all times.
Patients may be disconnected from suction and taken for a walk, but communication with the medical and nursing team is essential regarding unit policy. It is important to ensure that the underwater sealed drainage system is kept below the level of the chest at all times. If the patient cannot be disconnected from suction, walking within the confines of the suction tubing or marching on the spot may be attempted.
If the patient cannot be disconnected from suction, walking within the confines of the suction tubing or marching on the spot may be attempted. If a patient is being disconnected from suction, it is essential that the tubing from the collection chamber to the suction is disconnected rather than just switching the wall suction off. (If the patient is left connected to the wall suction with the suction switched off, there is no vent to the atmosphere through which air may escape from the pleural space and a tension pneumothorax may occur).
If a patient is being disconnected from suction, it is essential that the tubing from the collection chamber to the suction is disconnected rather than just switching the wall suction off. (If the patient is left connected to the wall suction with the suction switched off, there is no vent to the atmosphere through which air may escape from the pleural space and a tension pneumothorax may occur).Cardiac surgery
Cardiac surgery has been performed since the 1960s following the introduction of cardiopulmonary bypass. It is performed for both congenital (Chapter 10) and acquired heart diseases and can be closed surgery (without the need for cardiopulmonary bypass) or open-heart surgery (OHS). The most common adult cardiac surgery is performed for ischaemic heart disease. Table 12.4 lists common types of congenital abnormalities that require cardiac surgery. This chapter will focus on the management of coronary artery disease and valve pathology.
Table 12.4 Common congenital abnormalities of the heart
| Abnormality | Description |
|---|---|
| Patent ductus arteriosus (PDA) | Persistent large PDAs at birth allow shunting of blood from the aorta back into the pulmonary circulation. The ductus is closed using endovascular techniques or thoracotomy |
| Coarctation of the aorta | A narrowing of the aorta, most commonly distal to the left subclavian artery. Treated by angioplasty or resection with grafting |
| Atrial septal defects (ASDs) and ventricular septal defects (VSDs) | Incomplete or failure of development of the interatrial (ASD) or interventricular (VSD) septa. The defects are closed with a prosthetic patch during open heart surgery |
Coronary artery disease
Coronary atherosclerosis is a significant disease, particularly of Western countries. Atherosclerosis is the hardening and narrowing of arteries due to atheroma (from the Greek word meaning ‘porridge’). It develops in larger vessels (>2 mm internal diameter) mainly as a result of longer-term injury to the endothelium. An initial ‘fatty streak’ lesion is caused by increased permeability of the endothelial cells. Following this, accumulation of oxidized low-density lipoprotein by macrophages and infiltration of T lymphocytes leads to the development of a white fibrolipid plaque. This plaque may ulcerate and a thrombus can form and may embolize. The risk of rupture and haemorrhage of the plaque (fissuring) is high. Coronary vessels commonly develop lesions proximally at the origins of the main coronary branches or at major branch points. A narrowing (>50%) of coronary arteries causes reduced blood flow to the area supplied by that vessel. Common sites of obstruction are the left anterior descending artery, the right coronary artery and the circumflex artery.
Angina occurs when the heart muscle is underperfused and lacks the required oxygen supply as a result of obstruction of a coronary vessel. Angina pectoris is acute pain described as ‘crushing, vice-like, tight, or squeezing pain’ it may be retrosternal or radiate to the jaw, left arm or neck. The reason for this distribution of pain is that during embryonic development the heart originates in the neck, as do the arms. Therefore, these structures receive pain nerve fibres from the same spinal cord segments. Angina commonly occurs on exertion/exercise and is relieved by rest and medication such as sublingual glycerol trinitrate. The myocardial perfusion is reversible and does not cause permanent damage.
Myocardial infarction (MI) occurs with prolonged or permanent occlusion, causing severe chest pain at rest (which may be ‘silent’), which lasts several hours and is accompanied by autonomic responses such as sweating, nausea and vomiting. Myocardial infarction involves necrosis of cardiac muscle followed by inflammatory cell infiltration and eventual fibrous repair. The classical cause of infarction is atheromatous plaque with thrombosis and vasospasm leading to complete occlusion of the vessel lumen. Myocardial infarction can vary from mild to fatal and may lead to arrhythmias. Typical diagnostic changes associated with MI are ST segment elevation on electrocardiograph (Chapter 3) and raised levels of serum proteins released from disrupted myocardial cells, particularly the isoenzyme of creatine kinase (CK) more specific to cardiac muscle (CK-MB). This is raised from about 4 hours to 72 hours following infarction. More specific markers, troponin T and I, are found only in myocardial cells and are the gold standard diagnostic test for MI; the blood levels peak at 14–20 hours following MI (Noble et al 2005).
Frequent or unstable angina (angina at rest) commonly requires the coronary artery occlusion to be either dilated or bypassed. There are several approaches to coronary revascularization: these are by immediate fibrinolysis using pharmacological agents such as streptokinase or alteplase (within 6–12 hours of the event), using percutaneous transluminal coronary angioplasty (PTCA) with or without stenting the vessel(s) or by bypassing the occlusion during OHS. The use of thrombolytic therapy positively promotes activation of the fibrinolytic system, helping to break down clots. Greatest benefits are reported if the drugs are administered (intravenously) within 70 minutes of the onset of pain. PTCA is used for discreet lesions in one or two vessels. A balloon-tipped catheter is placed at the blockage guided by a fine wire and the balloon is inflated. This pushes the plaque against the artery wall, enlarging the lumen. This procedure has a high rate of restenosis. A stent (expandable wire cage) may be placed during PTCA to keep the lumen patent and this has been more successful in reducing the rate of restenosis (Noble et al 2005). Investigations used include chest X-ray, 12-lead ECG, stress test, thallium scans, positive emission tomography (PET) scan with coronary angio-graphy being the definitive diagnostic investigation (Chapter 3).
Coronary artery bypass surgery
Several severe coronary stenoses associated with unstable or uncontrolled angina may be treated with OHS and bypass grafting. This operation involves access via a median sternotomy, requires cardiopulmonary bypass (although ‘off-pump’ operations are now performed) and uses conduits, commonly the left and right internal thoracic arteries, the radial artery and saphenous vein, to bypass the coronary occlusion. The operative mortality is 1%, with perioperative stroke, myocardial ischaemia, arrhythmias, cardiovascular instability, sternal infection and haemorrhage being the most common complications. Respiratory complications include low lung volumes, basal atelectasis and pleural effusion. Long-term survival is excellent with 90–95% alive at 5 years and 80–85% at 10 years but is significantly influenced by age at surgery, diabetes, left ventricular function and risk factor modification after surgery (Tatoulis & Smith 2006).
Cardiopulmonary bypass
Cardiopulmonary bypass (‘heart–lung machine’) was developed to allow operations on a still and empty heart. Cardiopulmonary bypass (CPB) involves the use of a mechanical pump and an oxygenating device to supply oxygenated blood to the body while the heart is empty. Blood is removed from the right atrium (using gravitational flow), oxygenated across a membrane and returned to the ascending aorta. A heat exchanger cools and rewarms blood and a centripetal pump or roller propels the blood (at 4–6 l/min) back into the aorta. A vent may be placed in the left ventricle to remove blood collecting from the coronary sinus and operative field. Complete arrest of the heart requires cross clamping of the aorta, during which the myocardium is protected with hypothermia to reduce oxygen requirements. This is achieved by infusion of cold (4°C) cardioplegic solution (usually potassium with oxygenated blood and other products) into the coronary arteries, topical cold solution and using the heat exchanger on the CPB machine. The cardioplegic solution arrests the heart in diastole. The duration of CPB is usually between 1 and 2 hours, although up to 3 hours is possible. After 2 hours, problems such as blood cell destruction by the roller pump, coagulation problems, neurological, hepatic and renal dysfunction may occur.
The preferred conduits for grafting are arteries, since they have a higher patency. The right internal thoracic artery is commonly anastomosed to the important, left anterior descending artery. The patency of the right internal thoracic artery at 5 years is 96% and at 10 years is still 95% compared with the saphenous vein (75%, reducing to 50% at 10 years) (Tatoulis & Smith 2006). Once surgery is completed, the patient is rewarmed and sinus rhythm restored. The sternum is wired together, and drains are placed behind the sternum and into the pleural cavities (with UWSD) if necessary. A typical operation usually takes 3–4 hours and the average number of vessels bypassed is three or four. The postoperative length of hospital stay is approximately 5–7 days for uncomplicated surgery and many patients are referred to outpatient (Phase III) cardiac rehabilitation after discharge following hospital (Chapter 14).
Acquired valvular heart disease
Acquired valvular heart disease is limited to the aortic, mitral and occasionally tricuspid valves. The most common form of valvular disease in Western countries is stenosis (narrowing), which is generally calcific and incompetence/regurgitation as a result of degeneration and ageing. The two forms may occur together. The incidence of rheumatic heart disease (caused by rheumatic fever) has declined in Western countries but is still high in developing regions such as China, India and South America. The two most common presentations of valvular dysfunction are aortic stenoses and mitral regurgitation (Tatoulis & Smith 2006). The main symptom of valve disease is dyspnoea, but it may manifest late in the disease process because of adequate ventricular reserve. Angina and syncope may occur in aortic stenosis and cerebral emboli or endocarditis may also complicate valve lesions. Echocardiography is the investigation and transoesophageal echocardiography provides anatomical detail of pathology and may be used intraoperatively. It is common for patients to also have investigations for coronary artery disease since both valve and coronary artery bypass surgery occur together in about 30% of patients (Tatoulis & Smith 2006).
Aortic valve surgery involves OHS via a median sternotomy and CPB. A prosthetic valve replaces the diseased one and the operation takes approximately 3 hours with 80 minutes of CPB.
Mitral valve surgery.
Mitral stenosis may be treated by valvuloplasty, where a balloon is passed via catheterization of the femoral vein into the left atrium and mitral valve and then inflated. However, OHS is required for severe disease and may involve debridement of calcification, resection of portions of the thickened chordae or may require replacement with a prosthetic valve. Mitral regurgitation is usually due to elongated and ruptured chordea, leading to a prolapsed and flail valve. In severe disease, valve replacement is necessary.
Cardiac valve prostheses.
Both tissue and mechanical valves are used for valve replacement surgery. Mechanical valves (such as St Jude) are durable but the patient needs to continue, for life, on anticoagulation therapy. Tissue valves (bioprostheses) may be human allografts or xenografts from specially treated porcine or bovine valves. With these valves no anticoagulation is necessary and they have long durability.
Operative mortality is 1–3% in valve surgery and 3–5% for combined valve and coronary artery surgery, and long-term results are excellent. Morbidity of valve surgery is relatively high, with problems of anticoagulant-related haemorrhage, thromboembolism, endocarditis and perivalvular leaks occurring in 1% of patients annually.
Monitoring following cardiac surgery
Pacing wires may be inserted at the time of the operation and the patient is returned to the cardiothoracic intensive care with ECG, blood pressure, right atrial pressure, pulmonary artery pressure and cardiac and urine output monitoring. The patient is initially mechanically ventilated (4–12 hours), then weaned and supported with oxygen therapy. Some patients require inotropic circulatory support including dopamine and afterload reducing agents such as nitroprusside. In cases where the function of the left ventricle is very poor postoperatively, an intra-aortic balloon pump (IABP) may be used to support the heart.
The IABP is a circulatory device providing support for the left ventricle. It consists of a single or multi-chamber balloon attached to an external pump console via a large lumen catheter. The balloon is inflated during diastole and deflated during systole. Inflation and deflation of the balloon is synchronized to the patient’s cardiac rhythm via the ECG trace. The physiological aims of the IABP are to improve myocardial perfusion and reduce afterload, resulting in a reduction in myocardial oxygen demand, improved cardiac contractility and decreased risk of ongoing myocardial ischaemia. These aims are achieved by increasing the blood flow through the coronary arteries from the counterpressure exerted by the inflated balloon. This in turn reduces the volume of blood in the aortic arch, thus decreasing the work of the left ventricle to eject the cardiac output. The large lumen catheter is inserted into the femoral artery with the balloon positioned in the aorta between the subclavian and renal arteries. Patients usually begin on a cycle of 1 : 1 (i.e. inflation/deflation every heart contraction) and are weaned off to 1 : 2, and then 1 : 3, usually over a period of several days before the pump is removed. Patients with the IABP in situ should be treated by physiotherapists as indicated following discussion with the ICU consultant. Their myocardial dysfunction may result in an inability of oxygen supply to meet demand, a condition known as circulatory shock. Situations that put these patients under greater physiological stress should therefore be avoided (i.e. activities that increase the work of their myocardium). If the patient is repositioned, great care should be taken not to kink the IABP catheter. Aggressive anticoagulation is required while the IABP is in situ and therefore patients should remain resting in bed for 24 hours following removal of the pump. The ICU physiotherapist must alert ward physiotherapists as to when mobilization is allowed, as often the pump will be removed and the patient transferred to the ward during this 24- hour bed rest period (School of Physiotherapy 2006b). Ventricular assist devices (VADs), which perform the work of the left ventricle, are used when inotropes and balloon pump are insufficient to maintain adequate circulation.
Newer concepts in coronary artery surgery
Performance of surgery on a beating heart without the use of cardiopulmonary bypass (CPB) is called ‘off pump’ surgery and its use is becoming more common in many cardiac units worldwide. Minimally invasive techniques using an endoscope and robotics (avoiding a median sternotomy) are being utilized in some centres, and endoscopic harvesting of the saphenous vein to minimize incisions and pain is being performed. The use of these newer techniques will impact on the physiotherapy management of this patient population, as it is probable that these patients will require less physiotherapy intervention after surgery. However, this means that those patients who ultimately undergo open-heart surgery with CPB in the future may represent a much sicker population who will need increased physiotherapy resources. Research into physiotherapy management and outcomes for patients having heart surgery needs to continue as the populations being treated change with the introduction of new surgical techniques.
Box 12.9 shows key points in cardiac surgery
Box 12.9 Key points in cardiac surgery
 There is no evidence to support the effectiveness of physiotherapy treatment while the patient is intubated and ventilated in the intensive care unit after return from open-heart surgery (OHS).
There is no evidence to support the effectiveness of physiotherapy treatment while the patient is intubated and ventilated in the intensive care unit after return from open-heart surgery (OHS). Level 1 evidence from systematic reviews does not support the use of prophylactic respiratory physiotherapy treatment to reduce the incidence of postoperative pulmonary complications after OHS.
Level 1 evidence from systematic reviews does not support the use of prophylactic respiratory physiotherapy treatment to reduce the incidence of postoperative pulmonary complications after OHS. There is insufficient evidence to support or refute the role of thoracic mobilizing exercises in improving function or range of movement after open-heart surgery; therefore these exercises are still recommended.
There is insufficient evidence to support or refute the role of thoracic mobilizing exercises in improving function or range of movement after open-heart surgery; therefore these exercises are still recommended. Patients with the intra-aortic balloon pump (IABP) in situ should be treated by physiotherapists as indicated only following discussion with the intensive care consultant.
Patients with the intra-aortic balloon pump (IABP) in situ should be treated by physiotherapists as indicated only following discussion with the intensive care consultant.Common surgical intravenous access and drains
Postoperatively, surgical patients have several different intravenous lines and drains. These are:
Surgical wound or cavity drains to remove blood or serous fluid from around the operative site. These drains may be open and drain into gauze or dressing, such as a drain placed in an abscess cavity, or closed and drain into a collection bag with or without added suction to enhance drainage. The drains are removed when minimal fluid is being drained, by cutting the suture that anchors them and withdrawing the tube from the patient (Tjandra 2006).
Intravenous (IV) cannulation is used for administration of fluids and drugs such as analgesics, usually into the forearm or hand. Thrombophlebitis develops at the insertion site after about 3 days and IV cannulas should be resited if infusions are required for longer periods.
Central venous catheterization is used for longer-term venous access or for administration of total parenteral nutrition or chemotherapy and also for short-term monitoring of central venous pressure (CVP). The cannula may be inserted into the subclavian or jugular veins. The catheter tip sits in the superior vena cava.
Infusion pumps are used to deliver drugs with mechanical assistance. Three types are commonly seen:
patient-controlled analgesia (PCA)
‘IMED’ (see Fig. 12.1).
Catheterization of the bladder involves introducing a tube through the urethra and into the bladder. An indwelling catheter is most commonly seen in the hospital setting. These catheters are equipped with a balloon, which is inflated following induction to anchor the catheter. It remains in situ until a patient is able to void completely and voluntarily. A suprapubic catheter is placed directly into the bladder through an incision in the abdominal wall. It is indicated for long-term management of urinary tract dysfunction.
Nasogastric tubes may be used either to deliver enteral feeds, medications or fluid or to drain/aspirate gastric contents. Fine-bore or wide-bore tubing is passed nasally down the oesophagus into the stomach. If a nasogastric tube is being used for the purpose of feeding, it will be attached to feed or will be spigotted. If it is being used as a drain, a drain bag will be connected to the end of the tubing.
Enteral feeding is feeding directly into the digestive system. It requires a functioning gastrointestinal system and is preferred over parenteral nutrition as it maintains gut mucosal barrier integrity, mucosal structure and function and gut hormones. Feeding tubes may be inserted into several structures. In the stomach (nasogastric tube), the duodenum (gastrostomy) or the small intestine (jejunostomy).
Total parenteral nutrition (TPN) is delivered via a central line and nutrients are given intravenously. IV feeding can be supplementary or complete.
Stoma.
A stoma is an artificial orifice or opening surgically created between a hollow organ, e.g. bowel, and the abdominal wall. A bag is attached to the external opening. Its purpose is to replace the function of a non-functioning or excised natural orifice. Two types of stoma are commonly seen:
 A colostomy is the formation of a stoma by fixing a portion of the colon to the abdominal wall. Its purpose is to act as an artificial anus, evacuating the bowel of its contents.
A colostomy is the formation of a stoma by fixing a portion of the colon to the abdominal wall. Its purpose is to act as an artificial anus, evacuating the bowel of its contents. An ileostomy is the formation of a stoma by fixing a part of the ileum to the abdominal wall. This is usually performed when the entire colon is resected. Its purpose is to evacuate the contents of the small bowel.
An ileostomy is the formation of a stoma by fixing a part of the ileum to the abdominal wall. This is usually performed when the entire colon is resected. Its purpose is to evacuate the contents of the small bowel.A stoma can be temporary or permanent depending on the pathology. Temporary stomas are created to relieve acute bowel obstruction, rest an inflamed distal bowel or to prevent bowel contents escaping into the peritoneal cavity following cases of bowel perforation. When the underlying problem has been corrected, the stoma is usually surgically closed and normal bowel evacuation is resumed. Permanent stomas are indicated when the bowel distal to the stomas has been removed. A stomal therapist or specialist nurse usually performs stomal care and extensive education of the patient (School of Physiotherapy 2006b).
Box 12.10 shows key points for the physiotherapist in managing drips and drains.
Box 12.10 Key points for the physiotherapist in managing drips and drains
 Note dosage/rate of drugs being delivered especially by infusion pumps. For patient controlled analgesia (PCA) note if the system is in patient ‘lock-out mode’. Patients will be unable to use delivery button until the lockout signal is cleared.
Note dosage/rate of drugs being delivered especially by infusion pumps. For patient controlled analgesia (PCA) note if the system is in patient ‘lock-out mode’. Patients will be unable to use delivery button until the lockout signal is cleared. All pumps can run on battery but ensure cords are plugged into wall supply when patients are non ambulant.
All pumps can run on battery but ensure cords are plugged into wall supply when patients are non ambulant. All pumps can be transferred to mobile intravenous poles. Nursing staff may need to assist with some types of PCA pump transfer, as they need to be unlocked with keys.
All pumps can be transferred to mobile intravenous poles. Nursing staff may need to assist with some types of PCA pump transfer, as they need to be unlocked with keys. Care and consultation is required if you plan to use the head down position with a patient who is being enterally fed.
Care and consultation is required if you plan to use the head down position with a patient who is being enterally fed. For patients with a colostomy or ileostomy observe for leakages before moving the patient. If full of air or fluid, the bag will require emptying before mobilizing or rolling the patient.
For patients with a colostomy or ileostomy observe for leakages before moving the patient. If full of air or fluid, the bag will require emptying before mobilizing or rolling the patient. Ensure a nasogastric tube is always supported after positioning a patient so there is no traction on the patients nose to reduce the risk of disconnecting the tubing. Dislodging a nasogastric tube may lead to aspiration and may not be possible to replace the tube for example following oesophageal surgery.
Ensure a nasogastric tube is always supported after positioning a patient so there is no traction on the patients nose to reduce the risk of disconnecting the tubing. Dislodging a nasogastric tube may lead to aspiration and may not be possible to replace the tube for example following oesophageal surgery.PHYSIOTHERAPY FOR PATIENTS HAVING SURGERY
Evidence
In many countries physiotherapists see patients awaiting major surgical procedures (upper abdominal, thoracic, cardiac, oesophageal) prophylactically with a ‘blanket’ referral system from medical staff. The physiotherapist may assess and educate the patient preoperatively and then treat postoperatively, using a variety of techniques. However, in the past 15 years research evidence has challenged components of this traditional prophylactic approach. Advances in surgery and pain management, together with a reduction in the incidence of clinically significant postoperative pulmonary complications, have also impacted on physiotherapy management. In this section, the evidence for physiotherapy intervention will be outlined with reference specifically to abdominal and cardiothoracic surgery. In reviewing the evidence for physiotherapy there are three strands of knowledge necessary to consider:
Sackett et al (2000) and Herbert et al (2005) describe in depth the steps of evidence-based practice.
From the previous discussion of the surgical process it is clear that the physiological changes occurring in the lungs following major surgery and the proposed theories of pathogenesis of PPC provide empirical support for the place of physiotherapy intervention to counteract these changes (Denehy & Browning 2007).
To review evidence from clinical trials an extensive database search is needed. A review of literature can be undertaken using electronic searches including MEDLINE, CINAHL, ISI Web of Science, PEDro and evidence-based medicine reviews (Cochrane, DARE). The search terms that could be entered include pulmonary complications, atelectasis, pneumonia and surgery, respiratory therapy, chest physiotherapy, chest physical therapy, deep breathing exercises (DBE), early mobilization, early ambulation, continuous positive airway pressure (CPAP), incentive spirometry (IS) and positive expiratory pressure (PEP), together with the type of surgery of interest.
Evidence in abdominal surgery
Using the above search terms, six randomized controlled trials (with a no-treatment control group) can be accessed from 1980 to 2006 in upper abdominal surgery (UAS). These provide Levels 1b and 2b evidence (Sackett et al 2000) for the place of physiotherapy management and are summarized in Table 12.5. Older data were not included because it was thought that these would no longer reflect current practice either in physiotherapy or pain and surgical management. Five of these six research papers are of moderate methodological quality but only three represent convincing evidence in support of the use of physiotherapy treatment to reduce the incidence of PPC following abdominal surgery (these are presented in italics in Table 12.5). Based on these trials, the provision of physiotherapy treatment to reduce the incidence of PPC in patients having UAS will on average prevent one respiratory complication in every four or five patients treated.
The evidence from these trials is further supported by three systematic reviews (Lawrence et al 2006, Overend et al 2001, Thomas & McIntosh 1994). Systematic reviews provide combined evidence from several trials using systematic and explicit methodology and therefore offer the highest level of evidence while narrative reviews may introduce bias in interpretation. One systematic review (Pasquina et al 2006) examines the evidence for the place of respiratory physiotherapy to prevent PPC. Of the 35 papers available between 1952 and 2005, only 13 included a no-treatment control group and of these, six included both upper and lower abdominal surgery populations. The authors concluded that routine physiotherapy following abdominal surgery was not justified. However several major shortcomings of this review were evident: the inclusion of papers from the 1950s, inclusion of lower abdominal surgery (routine physiotherapy is not performed for patients after lower abdominal surgery) and the omission of a large randomized controlled trial by Olsen et al (1997). The 368 subjects in the Olsen study represent more than half of the number of patients reported in the six RCTs with an endpoint of pneumonia. Inclusion of this study may have altered the conclusions of this review, which are in contrast to the findings of another review by Lawrence and co-workers (2006). In summary, on the basis of the above evidence the place of physiotherapy treatment in reducing PPC after upper abdominal surgery is supported.
Evidence in cardiac surgery
Following cardiac surgery, the evidence obtained from a large body of research clearly challenges the continued traditional approach of prophylactic physiotherapy intervention. A search of the physiotherapy evidence database (PEDro) using ‘cardiac surgery’ reveals four systematic reviews regarding the place of respiratory physiotherapy for patients having cardiac surgery (Brooks et al 2001, Jonasson & Timmermans 2001, Moore 1997, Pasquina et al 2003) in addition to numerous clinical trials. The most recent systematic review concluded that there is no clear evidence that prophylactic respiratory physiotherapy reduces the incidence of PPC following cardiac surgery (Pasquina et al 2003). In a randomized clinical trial, Stiller and colleagues (1994) found no difference in the incidence of PPC between a group of cardiac surgery patients receiving physiotherapy and a group receiving no physiotherapy intervention. Based on this evidence, it is recommended that postoperative treatment that aims to reduce PPC, such as breathing exercises, is not routinely included in physiotherapy management of this patient population. A Swedish paper (Westerdahl et al 2005) presented evidence that deep breathing exercises may reduce the degree of atelectasis following cardiac surgery. However, the authors commented that the value of this short-term improvement in influencing the patient’s clinical recovery and length of stay was not the aim of the study. Based on the current evidence, in many centres the assessment and monitoring of mobility is the only intervention. Provision of rehabilitation for these patients is supported by a large body of evidence and is discussed in detail in Chapter 14. The place and effectiveness of shoulder and thoracic mobilizing exercises has not been studied extensively in this population and presents opportunity for research. Stiller et al (1997) reported that active range of movement exercises of the trunk and upper limb in the early postoperative period did not affect the incidence of musculoskeletal problems up to 10 weeks after surgery. The incidence of musculoskeletal complications in this study was 30%. Others report a relatively high incidence of musculoskeletal problems after cardiac surgery, particularly after internal mammary artery grafts (El-Ansary et al 2000, LaPier & Schenk 2002).
Evidence in thoracic and oesophageal surgery
Literature pertaining to the place of physiotherapy for patients having thoracic and oesophageal surgery is scant and inconclusive. A randomized clinical trial examining physiotherapy intervention is much needed in these patient populations. Currently, given the lack of definite evidence either way, it is recommended that patients undergoing these operations are treated both pre- and postoperatively. In general the site (both a thoracotomy and abdominal incision for oesophageal surgery) and extent of these operations, the positioning of the stomach within the thorax after oesophageal resection, together with patient factors such as cachexia, particularly in patients presenting for oesophageal surgery, mean that they represent a high-risk group for developing PPC. The incidence of PPC in these patients has been reported to be between 8 to 30% (Gosselink et al 2000).
Box 12.11 shows key points in evidence for physiotherapy for the surgical patient.
Box 12.11 Key points in evidence for physiotherapy for the surgical patient
 There is a large body of evidence that supports the place of physiotherapy treatment to reduce the incidence of postoperative pulmonary complication (PPC) after upper abdominal surgery.
There is a large body of evidence that supports the place of physiotherapy treatment to reduce the incidence of postoperative pulmonary complication (PPC) after upper abdominal surgery.Physiotherapy treatment techniques
The main presenting problems found on assessment of patients who have had a major surgical procedure are reduced lung volumes, which may lead to impaired gas exchange, and less commonly impaired airway clearance. The research described above presents a variety of different treatment techniques that are aimed at improving these problems in patients undergoing surgery. The definition and uses of these different techniques are described in Chapters 4, 5 and 6. In the management of surgical patients the physiotherapy techniques commonly used include preoperative education, deep breathing exercises (DBE), incentive spirometry (IS), positive expiratory pressure (PEP), continuous positive airway pressure (CPAP), early positioning and mobilization and less commonly intermittent positive pressure breathing (IPPB). The current research into the comparative efficacy of these techniques in preventing PPC after major surgery provides Level 3 evidence (Sackett et al 2000) that no one particular physiotherapy technique appears to be most effective. This is supported by a meta-analysis and systematic review (Thomas & McIntosh 1994), which concluded that there were no significant differences in the incidence of PPC using either IS, DBE or IPPB after upper abdominal surgery. Furthermore both IS and DBE were found to be more effective than no treatment. A second systematic review (Overend et al 2001) examined the effect of IS in preventing PPC and reported that the balance of evidence from the best available studies (10 out of 46 studies) failed to support the use of IS for reducing the incidence of PPC following UAS.
In this section the use of these techniques specifically for surgical patients will be presented. In the research discussed in this section it is difficult to isolate the effects of individual techniques, since most results are confounded by the effects of body positioning and mobilization. It is only recently that attention has been paid to mobilization as a treatment technique in its own right, despite the early work of Dean & Ross (1992). The inclusion of early mobilization is important since it is also beneficial in treating deconditioning following surgery.
Preoperative assessment and education
With the advent of same-day surgery and preadmission clinics it has become more difficult for the physiotherapist to see patients before surgery. However, patients undergoing higher-risk operations such as those having liver transplantation, upper abdominal, ear, nose and throat, aortic aneurysm repairs, thoracic or oesophageal surgery should be treated preoperatively.
The aims of preoperative physiotherapy treatment are to:
Following a thorough assessment of the patient (Chapter 1), including documentation of patient risk factors:
Explain:
 presence of drips/drains/oxygen therapy/catheter/intercostal catheter (if uncertain, be non-specific or ask if the surgeon or anaesthetist has explained these)
presence of drips/drains/oxygen therapy/catheter/intercostal catheter (if uncertain, be non-specific or ask if the surgeon or anaesthetist has explained these)Demonstrate/practise:
 recommend the frequency and number of times for the deep breathing exercises, FET, supported cough and foot and ankle exercises
recommend the frequency and number of times for the deep breathing exercises, FET, supported cough and foot and ankle exercises provide the patient with a written practice routine or information booklet, taking into consideration the language skills of the patient
provide the patient with a written practice routine or information booklet, taking into consideration the language skills of the patient for surgery such as cardiothoracic, upper limb and trunk exercises should be included in addition to education regarding underwater sealed drainage and discussion of rehabilitation on discharge
for surgery such as cardiothoracic, upper limb and trunk exercises should be included in addition to education regarding underwater sealed drainage and discussion of rehabilitation on discharge for patients having ear, nose and throat surgery, information about tracheostomy care, particularly oxygen therapy and suction, should be provided.
for patients having ear, nose and throat surgery, information about tracheostomy care, particularly oxygen therapy and suction, should be provided.Note: If the patient exhibits respiratory symptoms that require physiotherapy for airway clearance, for example patients with bronchiectasis, full treatment is imperative preoperatively. Patients with acute exacerbations of respiratory disease rarely undergo general anaesthesia until they are in a stable condition.
Postoperative management
Assessment.
All patients should be assessed thoroughly following surgery by the attending physiotherapist (Chapter 1), and a prioritized problem list and treatment plan developed. A brief overview of possible treatment techniques that can be used follows. More information on these techniques can be found in Chapters 4, 5 and 6.
Deep breathing exercises to improve lung volumes.
Application of breathing exercises after upper abdominal and thoracic surgery is an integral part of most physiotherapy treatments. The use of inconsistent terminology is a feature of the literature describing breathing exercises. Traditionally, deep breathing exercises may include bilateral basal expansion, thoracic expansion exercises, diaphragmatic breathing and sustained maximal inspiration. The aim of a deep breath is to produce a large and sustained increase in transpulmonary pressure, which distends the lungs and reinflates collapsed lung units (Duggan & Kavanagh 2005), and deep breathing exercises have been the mainstay of physiotherapy treatment for these patient groups for over 50 years.
Most deep breathing exercises that aim to improve lung volume are performed from functional residual capacity (FRC) to total lung capacity (TLC). As a result of the effect of body position on lung volume they are most often performed in erect sitting, but may also be used in combination with gravity-assisted drainage and forced expiratory manoeuvres. If airway clearance is the main aim of treatment then an appropriate airway clearance technique, such as the active cycle of breathing techniques (ACBT), should be used. It is important to note that when using ACBT in surgical populations, the emphasis would be focused more on the deep breathing component to improve ventilation and less on the forced expiratory manoeuvres such as FET when compared with patients with excess secretion production. For example, in surgical patients it may be more effective to perform three sets of DBE before the FET. Variations in inspiratory flow rates are thought to alter the distribution of ventilation; so, to improve ventilation to the dependent lung regions that are most affected following major surgery, a slow inspiratory flow rate is recommended (Tucker & Jenkins 1996).
The importance of periodic deep breaths in people with normal lungs has been evaluated and has shown that regular large breaths to TLC are essential to maintain inflation. Furthermore, it was reported that five consecutive breaths to TLC were needed for effective inflation of alveoli (Ferris & Pollard 1960). In addition, a sustained maximal inspiration (SMI) mimics a sigh or yawn and also aims to increase transpulmonary pressure (Bakow 1977). Sustained maximal inspirations have been reported to redistribute gas into areas of low lung compliance, thus enhancing lung expansion through lung interdependence and by using collateral ventilation pathways (Menkes & Traystman 1977). It may also allow time for alveoli with slow time constants to fill and the addition of a 3-second SMI at TLC is therefore recommended (Denehy & Browning 2007). Based on this research, the common treatment regimen used for deep breathing exercises is five deep breaths, with a 3-second inspiratory hold to maintain increased transpulmonary pressure, once every waking hour (Platell & Hall 1997).
Physiotherapists may choose to perform deep breathing exercises by using tactile stimulation over the patient’s lower ribs, or anteriorly on the costal margin. The addition of resistance through the hands and application of a quick stretch is thought to encourage a maximal inspiration (Levenson 1982). Studying diaphragmatic motion using ultrasonography showed a significant increase in diaphragmatic excursion following surgery when tactile or ‘hands-on’ breathing techniques were used (Blaney & Sawyer 1997).
The regimen of breathing exercises used was found to be reasonably uniform across studies included in a meta-analysis of the effectiveness of physiotherapy techniques in the prevention of PPC (Thomas & McIntosh 1994) indicating that the treatment regimen, based upon physiological principles and developed in the 1960s, is still common in clinical practice today. The lack of current research evidence to support the method of implementation of breathing exercises means that this technique may be used suboptimally by physiotherapists, and this in turn may affect treatment efficacy. It is not clear whether the depth of inspiration achieved during deep breathing exercises is more important than dose and frequency of breaths. Additionally, while there is good evidence that lung expansion techniques are important as part of the postoperative treatment (Fig. 12.8), it is not yet clear whether lung expansion can be achieved just as well by mobilizing the patient.
Figure 12.8 Deep breathing exercises 2 days following right hemicolectomy. The physiotherapist facilitates basal expansion with her hands.
Incentive spirometry (IS) was developed to stimulate the patient to perform deep breathing exercises under supervision or independently. The additional effects of IS compared with deep breathing exercises have been widely discussed in the literature. The use of IS has been evaluated in different patient diagnostic groups such as adult cardiothoracic surgery, UAS and paediatrics (Overend et al 2001). The additional value of IS over other forms of postoperative prophylaxis has not been supported in these patient groups. It was reported that the addition of IS to physiotherapy, including deep breathing exercises and early mobilization, did not significantly alter the incidence of PPC following thoracic and oesophageal surgery (Gosselink et al 2000). Based on the current available evidence, use of IS is therefore not supported when deep breathing exercises and mobilization are possible.
Positive pressure techniques for postoperative patients
PEP and IR-PEP.
Research evidence reporting the clinical efficacy of positive expiratory pressure (PEP) mask physiotherapy is conflicting and most of the research has been undertaken in patients with chronic sputum production rather than in surgical patients. Conclusions from these data are hampered by use of different treatment regimens, and the lack of equivalence in frequency of the other physiotherapy techniques with which PEP was compared. The physiological effects of PEP are thought to be that application improves gas mixing in the lungs leading to improved lung function and oxygenation, together with improved forced expiratory flow (Darbee et al 2004). In the treatment of patients undergoing UAS, PEP has been compared with conventional pre- and postoperative physiotherapy. The effect of adding PEP to conventional physiotherapy was measured in 71 patients following elective abdominal surgery and the incidence of PPC was found to be 31% after conventional physiotherapy and 22% after physiotherapy plus PEP (Campbell et al 1986). The PEP device used in this study is now commonly known as ‘bubble’ PEP as the end-expiratory pressure is maintained by the height of a column of water in a plastic bottle (Chapter 5 ). In this study no manometer was added to the circuit so that accurate PEP pressures could not be recorded. Comparison of PEP with continuous positive airway pressure (CPAP) and IS in a further study in UAS reported that both CPAP and PEP were more effective than IS in maintaining gas exchange and lung volumes postoperatively and lowering the incidence of atelectasis (Ricksten et al 1986). However, PEP and inspiratory resistance (IR) PEP have been used in previous research in patients having UAS with good effect, although comparison of techniques was not the main aim of the study (Olsen et al 1997). In patients undergoing oesophageal surgery a comparison of the use of IR-PEP and CPAP in the postoperative period showed that CPAP more effectively reduced the need for reintubation. The treatment with IR-PEP comprised 30 breaths every 2 hours and CPAP was applied for 30 minutes every 2 hours. Dosage and frequency of PEP application vary but commonly PEP is applied with physiotherapy treatments once or twice daily for 8–10 breaths in each of six cycles with a pressure of 10–20 cmH2O.
Continuous positive airway pressure (CPAP).
The use of CPAP by physiotherapists is generally in the form of intermittent or periodic application by mouthpiece or mask (Fig. 12.9). The decision of whether to implement CPAP treatment in postoperative patients is frequently made by the physiotherapist in conjunction with the medical team. In some centres, physiotherapists do not use CPAP on the wards as a treatment technique, giving a variable profile of use internationally.
Figure 12.9 A patient receiving continuous positive airway pressure (CPAP) via (A) nasal mask with Ambu® positive expiratory pressure valve and Velcro® head straps, (B) Dräger CPAP unit.
The effects of CPAP application on lung volumes have been well documented. These include an increase in VC, a reduction in respiratory rate, reduced minute ventilation (MV) and increased FRC. The application of CPAP following UAS has been demonstrated to increase FRC when compared with other forms of respiratory prophylaxis (Lindner et al 1987). There is also support for the improvement of atelectasis with CPAP application after UAS (Andersen et al 1980).
The indications for the use of CPAP will to some extent depend on the unit and team structures of different centres and the role of the physiotherapist within them. Indications are derived from the physiological benefits of CPAP (Keilty & Bott 1992), but CPAP is generally considered a second-line treatment intervention for patients following major surgery. Indications include a significant fall in FRC and a significant deterioration in respiratory function and gas exchange as a result of atelectasis, most particularly acute hypoxaemia. The types of patient conditions seen therefore may include upper abdominal, thoracic, or cardiac surgery, especially for those high-risk patients not responding to voluntary lung expansion techniques such as mobilization and deep breathing exercises. Uncooperative or confused patients who develop significant respiratory problems after surgery may also benefit from application of CPAP as it requires no cooperation from the patient. In patients admitted to intensive care with acute hypoxaemia, the application of CPAP reduced the requirement for intubation and the incidence of severe complications (Squadrone et al 2005). The use of CPAP in intensive care units is generally considered to be a medical intervention and therefore physiotherapists may not be involved in the decision-making process or application of the technique.
To date, no specific treatment regimens have been compared to establish efficacy. Therefore the pressures used, length of treatment and frequency of application varies in different centres. In general, following surgery, CPAP should be applied as soon as a deterioration in the patient’s condition is evident and used until clinical improvement is achieved. A suggested protocol is application of 7.5–10 cmH2O CPAP for 30 minutes every 2–4 hours initially. Three continuous hours of application daily has also been associated with benefits (Denehy et al 2001).
Positive pressure devices in patients having thoracic or oesophageal surgery should be applied with caution. Flow rates and pressures used should be as low as possible to achieve effectiveness. Precautions with CPAP treatment may include an unstable cardiovascular system and recent oesophageal surgery. Swallowing air may overdistend the oesophagus at the site of the anastomosis, affecting its integrity. The presence of a nasogastric tube during CPAP administration to decompress the oesophagus is recommended. In a study of patients having oesophageal surgery, no adverse effects of CPAP application were reported (Olsen et al 2002).
Bilevel positive airway pressure.
This can also be used in the management of surgical patients but generally is used in the treatment of hypercapnia rather than acute hypoxaemia. The evidence for its use in treating hypercapnia is strong (Chapter 11). In surgical patients bilevel devices may be used with the main aim of treatment being to improve lung volumes and gas exchange, particularly hypoxaemia.
Intermittent positive pressure breathing (IPPB).
In postoperative patients the efficacy of IPPB has been compared with IS, breathing strategies and respiratory physiotherapy. The results from this extensive research were equivocal, with IPPB conferring no added benefit when compared with other methods of postoperative prophylaxis. Intermittent positive pressure breathing may be a useful tool in the management of postoperative patients who are unable or unwilling to inspire maximally (O’Donohue 1992). For patients with reduced lung volumes, improvement in vital capacity may provide the patient with a more effective cough, thereby improving secretion clearance. The aims of IPPB need to be directed toward improving volumes; the pressure and flow rate settings must therefore be set accordingly for individual patients and will depend on their body position and underlying lung pathology (O’Donohue 1992).
Positioning and mobilization
The cardiovascular and respiratory effects of immobility and bed rest have been well documented (Dean 1985, 1993). These include reduced lung volumes and capacities especially FRC, reduced PaO2, decreased maximal oxygen consumption (V.O2max), cardiac output (CO) and stroke volume, increased heart rate (HR) and orthostatic intolerance. The goal of mobilization of postoperative patients is to exercise at a sufficient level to increase minute ventilation and CO while still being within safe physiological limits.
Mobilization is a term used to describe a change from the supine or slumped posture in bed to sitting out of bed, standing or walking, although physiotherapists often use the terms positioning (sitting, standing) and ambulating (walking). A systematic review supports the benefits of upright positioning compared with the supine posture in improving FRC, oxygen saturation, PaO2 and reducing PaCO2 (Nielsen et al 2003). In surgical patients, sitting upright out of bed is an important component of postoperative management (Fig. 12.10). This technique is used by both nursing staff and physiotherapists. In a study of 15 subjects it was shown that ambulation on the third to fifth day after surgery increased tidal volume and respiratory rate, but that this increase was not significant once the effects of change in body position from bed to standing had been taken into account (Orfanos et al 1999).

Figure 12.10 A postoperative patient who has just been sat out of bed in a chair to facilitate improvement in lung volumes and mobility.
Early mobilization is an important and widely practised component of postoperative patient care following surgery. In the early postoperative period, the ability of patients to mobilize is limited by the presence of intravenous lines and surgical drains, pain, decreased arousal, anxiety, surgical incisions and medication and a large proportion of patients become newly dependent on assistance to mobilize at this time (Denehy & Browning 2007). At two large Australian hospitals it was found that 52% of patients required at least standby assistance in order to mobilize safely on the fifth postoperative day (Mackay et al 2005). The same study reported that patients who received significantly higher numbers of physiotherapy treatments including more physiotherapy-assisted mobilization had better mobility outcomes, faster rates of resolution of PPC, and that a greater proportion of these patients were discharged earlier from hospital.
The benefits of mobilization in cardiac surgery patients have been established and research supports the benefits of mobilization without the additional need for deep breathing exercises or IS (Jenkins et al 1989). For patients having open UAS the addition of deep breathing exercises to a physiotherapist-directed program of early mobilization was found to have no effect on further reducing the incidence of PPC in 45 patients (Mackay et al 2005).
Despite the established important place of mobilization following surgery, little research has been aimed at identifying the intensity or frequency of mobilization that is most effective. Prescription of exercise such as walking a patient after surgery should be based on scientific principles. More research is needed to measure the intensity at which physiotherapists can safely mobilize patients and how often this should be undertaken. Close monitoring is essential during and immediately following mobilization, especially at the first treatment session. Specific exercise guidelines must be based upon the individual patient’s assessment findings and regular reassessment by the physiotherapist is important. Use of physiological outcomes could be included such as SpO2, Borg rating of perceived exertion and heart and respiratory rate. It is possible that greater improvements in acute outcomes such as length of stay and incidence of PPC may be achieved with increased intensity and frequency of mobilization. In research conducted at a large tertiary hospital in Australia it was found that the average time that patients were standing or walking on the first day after open UAS was only 3 minutes (Browning et al 2005).
With advances in anaesthesia, surgical techniques and perioperative care, older people are having surgery that may not have been possible in the past. Not only are these patients at a higher risk of developing PPC but they are also at risk of a significant decline in physical function for some time following surgery. The functional recovery of patients aged over 60 years following abdominal surgery may be significantly reduced at 3–6 months. Prevention of PPC and improving functional status of older patients before surgery may enhance recovery (Lawrence et al 2004). In addition the use of ‘fast-track’ pathways, pioneered by Kehlet & Mogensen (1999), where attention is paid to nutrition and early mobilization, are being implemented more frequently around the world as they have been shown to reduce length of stay and hospital costs. Physiotherapy plays an important part in the early mobility of all major surgical patients (Fig. 12.11) and physiotherapists need to investigate the use of intensive rehabilitation both in the pre-, early and later postoperative periods to provide sound evidence upon which to direct treatment in the future.
Box 12.12 shows key points in physiotherapy treatment techniques.
Box 12.12 Key points in physiotherapy treatment techniques
 Preoperative treatment allows physiotherapists to assess patient risk factors and may be an effective treatment in its own right.
Preoperative treatment allows physiotherapists to assess patient risk factors and may be an effective treatment in its own right. The additional value of incentive spirometry over other forms of postoperative prophylaxis has not been supported in surgical patients.
The additional value of incentive spirometry over other forms of postoperative prophylaxis has not been supported in surgical patients. Continuous positive airway pressure is most commonly used as a second-line treatment intervention in the management of surgical patients.
Continuous positive airway pressure is most commonly used as a second-line treatment intervention in the management of surgical patients. Mobilization of patients has been included in most of the research assessing effectiveness of physiotherapy treatment as a whole. The effectiveness of mobilization alone after abdominal, thoracic and oesophageal surgery needs further investigation.
Mobilization of patients has been included in most of the research assessing effectiveness of physiotherapy treatment as a whole. The effectiveness of mobilization alone after abdominal, thoracic and oesophageal surgery needs further investigation.Specialist surgery
In this section several other types of surgery encountered by physiotherapists will be briefly described; these are chest trauma, head and neck surgery, adult liver transplantation and vascular surgery.
Chest trauma
Sternal and rib fractures are commonly sustained after road traffic accidents. Sternal fractures are generally managed conservatively, but may require internal fixation. Effective pain management is one of the most important aims of treatment for both sternal and rib fractures. When several ribs are fractured both anteriorly and posteriorly a ‘flail’ segment of chest wall may be evident. This creates paradoxical movement of the ribs such that the flail segment moves inward on inspiration, asynchronously with the chest wall. The underlying lung is therefore not ventilated adequately. Patients with a flail segment are at high risk for developing atelectasis and subsequent respiratory failure unless managed well. This means that pain control needs to be optimal and the physiotherapist should consider use of CPAP (after discussion with medical staff) to splint the flail segment and allow it to move in synchrony with the chest wall. The level of positive pressure is titrated so that synchrony is achieved. Underlying pneumothorax should be looked for on the chest radiograph before instituting positive pressure. Serial chest X-rays may be necessary in cases where a small untreated pneumothorax exists. Patients with chest trauma may also have underlying lung contusion and they can deteriorate quickly on the ward if not assessed and treated vigilantly.
Head and neck surgery
Most surgery performed in the head and neck region is for malignancy, predominately squamous cell carcinoma. Metastases to cervical lymph nodes are common and significantly impact prognosis. There has been a shift away from radical surgery such as total laryngectomy with the emphasis of treatment on organ preservation (particularly the larynx) where possible (Judson 2006). Multimodality treatments including chemotherapy and radiotherapy are used together with more selective surgery that spares sternomastoid, the internal jugular vein and the accessory nerve. Additionally, endoscopic laser techniques are now used for endo-laryngeal and hypopharyngeal disease. Free tissue transfer techniques (flaps) allow tissue with a better blood supply to be used for reconstruction after more radical surgery. Where flaps have been used there may be precautions with specific movements and liaison with medical staff is essential. The use of these techniques leads to a better functional and cosmetic outcome for patients (Judson 2006). Following some surgical procedures, patients may have a temporary tracheostomy in situ to protect the airway from swelling and compression. For radical laryngectomy a permanent tracheostomy is necessary and speech therapists will be involved in the management of speech and swallowing problems. The physiotherapist will need to use suction to remove secretions from patients with tracheostomies and should understand how to care for patients with tracheostomies (Chapter 8).
Vascular surgery
Atherosclerosis accounts for most peripheral arterial disease and is usually coexistent with coronary and cerebrovascular disease. For physiotherapists, the most commonly encountered patients have undergone aortic aneurysm repair. An aneurysm is a permanent localized dilatation of an artery to one and a half times the normal diameter. When an artery dilates it may rupture or a thrombus may form. The incidence of aneurysms is higher in smokers, patients with hypertension and in Caucasians, and presentation can be asymptomatic until pending rupture. Abdominal aortic aneurysms are usually resected when they are larger than 5 cm in diameter as seen on ultrasound or computed tomography (CT) scan (as an incidental finding). Open repair involves an extensive midline laparotomy, clamping of the aorta and sewing in a prosthetic arterial graft to replace the area of aneurysm either using a straight or bifurcated method. Endoluminal repair involves insertion of a graft through the common femoral artery but not all patients are suitable to be treated using this method, as narrow and calcified arteries can pose technical problems. Further refinement of this technique in the future may overcome these problems (Harris 2006). Perioperative mortality is less with the endoluminal approach than open. Thoracic aortic aneurysms are less common than infrarenal aneurysms.
Liver transplantation
Liver transplantation is indicated in irreversible end-stage liver diseases caused by infections such as viral hepatitis A, B, C and E, immune or metabolic disorders, alcohol or drug abuse. Potential recipients are referred to a multidisciplinary transplant unit and are admitted to a waiting list for a suitable donor. While on the transplant list, clinical stability is maintained and nutritional deficiencies identified and treated in order to increase the probability of a successful outcome of the transplant. Patients who have liver failure may have a number of complications, including jaundice, portal hypertension, coagulopathy, varices, ascites, hepatopulmonary syndrome causing hypoxaemia, muscle wasting, deconditioning and fatigue. Sometimes the patients are in the intensive care unit (ICU) before transplantation. It is important to remember that they have a coagulopathy and treatment should be performed carefully and only when indicated.
Transplant surgery is a difficult procedure due to the premorbid problems associated with liver failure. The operation usually lasts about 8–10 hours, but may be as long as 23 hours. Large amounts of blood replacement products are usually needed but cell saving is also used. The liver is accessed via a reverse ‘L’ incision; clamping of the inferior vena cava below the liver reduces cardiac output by 50% and is tolerated for 10–15 minutes. Veno-venous bypass via the femoral vein, or inferior vena cava may be used if the patient is very sick or cardiovascularly unstable. Split liver transplants are performed where the left lobe of the liver of a donor may be used for a child and the right for an adult.
Postoperatively patients are admitted to the ICU for mechanical ventilation, haemodynamic monitoring and monitoring of liver function tests. If the patient’s temperature increases to >37.5°C a full septic work-up is performed. The most common postoperative complications are rejection, infection (respiratory, wound, biliary tract, blood), renal failure and graft ischaemia. Antirejection and antiviral drugs are given postoperatively.
Physiotherapy management of the respiratory status of the patient is important because of prolonged anaesthesia, poor premorbid patient condition and since the liver is under the right hemidiaphragm, right lower lobe atelectasis and effusion are common. Musculoskeletal management is also important with the aims of increasing muscle bulk, strength and endurance. Back pain is common in this patient group and education about posture and lifting should be included in a comprehensive rehabilitation programme postoperatively (Stockton 2001). Research has shown that patients can improve their 6-minute walking distance and V.O2 with exercise programmes. In the 1996 United States Transplant Games, 16 athletes with liver transplants competed. Their exercise capacity was 101% of age-matched controls, demonstrating that they are capable of achieving high levels of physical functioning (J Luke, Personal Communication, 2006).
Box 12.13 shows key physiotherapy points for specialist surgery.
Box 12.13 Physiotherapy key points for specialist surgery
 The same principles of physiotherapy management are indicated for special surgery: to prevent or minimize respiratory complications and rehabilitate the patient.
The same principles of physiotherapy management are indicated for special surgery: to prevent or minimize respiratory complications and rehabilitate the patient. Ensure patients with rib and sternal fractures have effective pain management and are able to take deep breaths.
Ensure patients with rib and sternal fractures have effective pain management and are able to take deep breaths. Be vigilant with patients who have a flail segment as their respiratory status can deteriorate rapidly.
Be vigilant with patients who have a flail segment as their respiratory status can deteriorate rapidly.Acknowledgement
I wish to acknowledge the help of several physiotherapists in the preparation of this chapter: Sarah Ridley for her diagrams carried through from the third edition; and current PhD students Laura Browning and Annemarie Lee for their suggestions on content. Additionally, I would like to acknowledge the clinicians of the University of Melbourne Clinical Schools who jointly contributed the information presented from the cardiorespiratory practical manuals of the School of Physiotherapy.
AIHW (Australian Institute of Health and Welfare) & AACR (Australasian Association of Cancer Registries), 2007. Cancer in Australia: an overview, 2006. Cancer series no. 37. Cat. no. CAN 32. (www.aihw.gov.au/publications/can/ca06/ca06.pdf) (Accessed July 23 2007)
American Society of Anesthesiologists (ASA). New classification of physical status. Anesthesiology. 1963;24:111.
Andersen J, Olesen B, Eikhard B, et al. Periodic continuous positive airway pressure, CPAP, by mask in the treatment of atelectasis. European Journal of Respiratory Disease. 1980;61:20-25.
Bakow E. Sustained maximal inspiration - a rationale for its use. Respiratory Care. 1977;22:379-382.
Barrat S. Advances in acute pain management. International Anesthesiology Clinics. 1997;35:27-33.
Berrisford R, Brunelli A, Rocco G, et al. The European Thoracic Surgery Database project: modelling the risk of in-hospital death following lung resection. European Journal of Cardiothoracic Surgery. 2005;28:306-311.
Blaney F, Sawyer T. Sonographic measurement of a diaphragmatic motion after upper abdominal surgery: a comparison of three breathing manoeuvres. Physiotherapy Theory and Practice. 1997;13:207-215.
Brasher P, McClelland K, Denehy L, et al. Does removal of deep breathing exercises from a physiotherapy program including pre-operative education and early mobilization after cardiac surgery alter patient outcomes? Australian Journal of Physiotherapy. 2003;49:165-173.
Brooks D, Crowe J, Kelsey C, et al. A clinical practice guideline on peri-operative physical therapy. Physiotherapy Canada. 2001;53:9-25.
Brooks-Brunn JA. Postoperative atelectasis and pneumonia. Heart and Lung. 1995;24:94-115.
Browning L, Denehy L, Scholes R. Quantitative measurement of mobility following upper abdominal surgery. Australian Journal of Physiotherapy. 2005;52:S8.
Campbell T, Ferguson N, McKinlay R. The use of a simple self-administered method of positive expiratory pressure (PEP) in chest physiotherapy after abdominal surgery. Physiotherapy. 1986;72:498-500.
Celli B, Rodriguez K, Snider G. A controlled trial of intermittent positive pressure breathing incentive spirometry, and deep breathing exercises in preventing pulmonary complications after abdominal surgery. American Review of Respiratory Disease. 1984;130:12-15.
Chumillas S, Ponce J, Delgado F, et al. Prevention of postoperative pulmonary complications through respiratory rehabilitation: a controlled clinical study. Archives of Physical Medicine and Rehabilitation. 1998;79:5-9.
Condie E, Hack K, Ross A. An investigation of the value of routine provision of post-operative chest physiotherapy in non-smoking patients undergoing elective abdominal surgery. Physiotherapy. 1993;79:547-552.
Craig DB. Postoperative recovery of pulmonary function. Anesthesia and Analgesia. 1981;60:46-52.
Darbee J, Ohtake P, Grant B, et al. Physiologic evidence for the efficacy of positive expiratory pressure as an airway clearance technique in patients with cystic fibrosis. Physical Therapy. 2004;84:524-536.
Dean E. Effect of body position on pulmonary function. Physical Therapy. 1985;65:613-618.
Dean E. Bedrest and deconditioning. Neurology Report. 1993;17:6-9.
Dean E, Ross J. Discordance between cardiopulmonary physiology and physical therapy: towards a rational basis for practice. Chest. 1992;101:1694-1698.
Denehy L, Browning L. Abdominal surgery: the evidence for physiotherapy intervention. In: Partridge C, editor. Recent advances in physiotherapy. Chichester: John Wiley and Sons, 2007.
Denehy L, Carroll S, Ntoumenopoulos G, et al. A randomized controlled trial comparing periodic mask CPAP with physiotherapy after abdominal surgery. Physiotherapy Research International. 2001;6:236-250.
Denehy L, van de Leur J. Postoperative mucus clearance. In: Rubins B, van der Schans C, editors. Therapy for mucus-clearance disorders. New York: Marcel Dekker, 2004.
Dodson M. The management of postoperative pain. London: Edward Arnold Ltd, 1985.
Duggan M, Kavanagh B. Pulmonary atelectasis - a pathogenic perioperative entity. Anesthesiology. 2005;102:838-854.
El-Ansary D, Adams R, Ghandi A. Musculoskeletal and neurological complications following coronary artery bypass graft surgery: a comparison between saphenous vein and internal mammary artery grafting. Australian Journal of Physiotherapy. 2000;46:19-25.
Euliano T, Gravenstein J. Essential anaesthesia - from science to practice. Cambridge: Cambridge University Press, 2004.
Fairshter R, Williams J. Pulmonary physiology in the postoperative period. Critical Care Clinics. 1987;3:286-306.
Ferris B, Pollard D. Effect of deep and quiet breathing on pulmonary compliance in man. Journal of Clinical Investigation. 1960;39:143-149.
Fisher B, Majumdar S, McAllister F. Predicting pulmonary complications after non-thoracic surgery: a systematic review of blinded studies. American Journal of Medicine. 2002;112:219-225.
Ford GT, Rosenal TW, Clergue F, et al. Respiratory physiology in upper abdominal surgery. Clinics in Chest Medicine. 1993;14:237-252.
Gosselink R, Schrever K, Cops P, et al. Incentive spirometry does not enhance recovery after thoracic surgery. Critical Care Medicine. 2000;28:679-683.
Hassan I, 2006 Lung cancer: staging. www.emedicine.com/radio/topic807.htm (Last updated 28 September 2006, accessed 16 July 2007)
Harris J. Disorders of the arterial system. In: Tjandra J, Clunie G, Kaye A, Smith J, editors. Textbook of surgery. Massachusetts: Blackwell Publishing, 2006.
Herbert R, Jamtvedt G, Mead J, et al. Practical evidence based physiotherapy. London: Elsevier Butterworth-Heinemann, 2005.
Jenkins S, Moxham J. The effects of mild obesity on lung function. Respiratory Medicine. 1991;85:309-311.
Jenkins S, Soutar S, Loukota J, et al. Physiotherapy after coronary artery surgery: are breathing exercises necessary? Thorax. 1989;44:634-639.
Jonasson B, Timmermans C. The effect of incentive spirometry on postoperative pulmonary complications: a systematic review. Chest. 2001;120:971-978.
Judson R. Tumours of the head and neck. In Tjandra J, Clunie G, Kaye A, Smith J, editors: Textbook of surgery, 3rd edn., Massachusetts: Blackwell Publishing, 2006.
Katz J. Preoperative analgesia for postoperative pain. Lancet. 1993;342:65-66.
Kehlet H, Kennedy R. Laparoscopic colonic surgery - mission accomplished or work in progress. Colorectal Disease. 2006;8:514-517.
Kehlet H, Mogensen T. Hospital stay of 2 days after open sigmoidectomy with a multimodal rehabilitation programme. British Journal of Surgery. 1999;86:227-230.
Keilty S, Bott J. Continuous positive airways pressure. Physiotherapy. 1992;78:90-92.
Kim WD. Lung mucus: a clinician’s view. European Respiratory Journal. 1997;10:1914-1917.
King D. Postoperative pulmonary complications. Surgery, Gynecology and Obstetrics. 1933;56:43-47.
LaPier T, Schenk R. Thoracic musculoskeletal considerations following open-heart surgery. Cardiopulmonary Physical Therapy Journal. 2002;13:16-20.
Law S, Wong J. Tumours of the oesophagus. In: Tjandra J, Clunie G, Kaye A, Smith J, editors. Textbook of surgery. Massachusetts: Blackwell Publishing, 2006.
Law S, Wong K, Kwok K, et al. Predictive factors for postoperative pulmonary complications and mortality after esophagectomy for cancer. Annals of Surgery. 2004;240:791-800.
Lawrence V, Cornell J, Smetana G. Strategies to reduce postoperative pulmonary complications after noncardiothoracic surgery: systematic review for the American College of Physicians. Annals of Internal Medicine. 2006;144:596-608.
Lawrence V, Hazuda H, Cornell J, et al. Functional independence after major abdominal surgery in the elderly. Journal of the American College of Surgeons. 2004;199:762-772.
Levenson C. Breathing exercises. In: Pulmonary management in physical therapy. New York: Churchill Livingstone; 1982.
Lindner K, Lotz P, Ahnefeld F. Continuous positive airway pressure effect on functional residual capacity, vital capacity and its subdivisions. Chest. 1987;92:66-70.
Mackay M, Ellis E, Johnston C. Randomized clinical trial of physiotherapy after open abdominal surgery in high risk patients. Australian Journal of Physiotherapy. 2005;51:151-159.
Macnaughton PD. Posture and lung function in health and disease. International Journal of Intensive Care. 1994;Winter:133-137.
Marini JJ. Postoperative atelectasis: pathophysiology, clinical importance and principles of management. Respiratory Care. 1984;29:516-528.
Menkes H, Traystman R. Collateral ventilation. American Review of Respiratory Disease. 1977;116:287-309.
Miller K, Sahn S. Chest tubes. Chest. 1987;91:258-264.
Moore S. Effects of interventions to promote recovery in coronary artery bypass surgical patients. Journal of Cardiovascular Nursing. 1997;12:59-70.
Morran C, Finlay I, Mathieson M, et al. Randomized controlled trial of physiotherapy for postoperative pulmonary complications. British Journal of Anaesthesia. 1983;55:1113-1116.
National Health and Medical Research Council, 2005 Acute pain management: scientific evidence www.nhmrc.gov.au/publications/synopses/cp104syn.htm (accessed 16 July 2007)
Nielsen K, Holte K, Kehlet H. Effects of posture on postoperative pulmonary function. Acta Anaesthesiologica Scandinavica. 2003;47:1270-1275.
Noble A, Johnson R, Thomas A, et al. The cardiovascular system. Basic science and clinical conditions. London: Elsevier Churchill Livingstone, 2005.
Nunn J. Nunn’s applied respiratory physiology. Oxford: Butterworth-Heinemann, 1993.
O’Donohue W. Postoperative pulmonary complications. Postgraduate Medicine. 1992;91:167-175.
Olsen M. Chest physiotherapy in open and laparoscopic abdominal surgery. Physical Therapy Reviews. 2000;5:125-130.
Olsen MF, Hahn I, Nordgren S, et al. Randomized controlled trial of prophylactic chest physiotherapy in major abdominal surgery. British Journal of Surgery. 1997;84:1535-1538.
Olsen MF, Wennberg E, Johnsson E, et al. Randomized clinical study of the prevention of pulmonary complications after thoracoabdominal resection by two different breathing techniques. British Journal of Surgery. 2002;89:1228-1234.
Orfanos P, Ellis E, Johnston C. Effects of deep breathing exercises and ambulation on pattern of ventilation in postoperative patients. Australian Journal of Physiotherapy. 1999;45:173-182.
Overend T, Anderson C, Lucy S, et al. The effect of incentive spirometry on postoperative pulmonary complications: a systematic review. Chest. 2001;120:971-978.
Pasquina P, Tramer M, Walder B. Prophylactic respiratory physiotherapy after cardiac surgery: systematic review. British Medical Journal. 2003:1379-1381.
Pasquina P, Tramer M, Granier J, et al. Respiratory physiotherapy to prevent pulmonary complications after abdominal surgery: a systematic review. Chest. 2006;130:1887-1899.
Peeters-Asdourian C, Gupta S. Choices in pain management following thoracotomy. Chest. 1999;115:122S-124S.
Platell C, Hall J. Atelectasis after abdominal surgery. Journal of the American College of Surgeons. 1997;185:584-592.
Ready L. Acute postoperative pain. Anesthesia. 1985;69:2135-2143.
Richardson J, Sabanathan S. Prevention of respiratory complications after abdominal surgery. Thorax. 1997;52:S35-S40.
Ricksten S, Bengtsson A, Soderberg C, et al. Effects of periodic positive airway pressure by mask on postoperative pulmonary function. Chest. 1986;89:774-781.
Roukema J, Carol E, Prins J. The prevention of pulmonary complications after upper abdominal surgery in patients with non-compromised pulmonary status. Archives of Surgery. 1988;123:30-34.
Sabanathan S, Shah R, Tsiamis A, et al. Oesophagogastrectomy in the elderly high-risk patients: role of effective regional analgesia and early mobilization. Journal of Cardiovascular Surgery. 1999;40:153-156.
Sackett D, Strauss S, Richardson W, et al. Evidence-based medicine. Edinburgh: Churchill Livingstone, 2000.
Scholes R, Denehy L, Sztendur E, et al. Development of a risk assessment model to predict pulmonary risk following upper abdominal surgery. Australian Journal of Physiotherapy. 2006;52:S26.
School of Physiotherapy University of Melbourne. Cardiorespiratory physiotherapy 1 student clinical Manual. Parkville, 2006.
School of Physiotherapy University of Melbourne. Cardiorespiratory physiotherapy 2 student clinical manual. Parkville, 2006.
Sharma K, Brandstetter R, Brensilver J, et al. Cardiopulmonary physiology and pathophysiology as a consequence of laparoscopic surgery. Chest. 1996;110:810-815.
Smetana G, Lawrence V, Cornell J. Preoperative pulmonary risk stratification for non-cardiothoracic surgery: systematic review for the American college of physicians. Annals of Internal Medicine. 2006;144:581-595.
Smith J. Common topics in thoracic surgery. In Tjandra J, Clunie G, Kaye A, Smith J, editors: Textbook of surgery, 3rd edn., Massachusetts: Blackwell Publishing, 2006.
Smith MCL, Ellis E. Is retained mucus a risk factor for the development of postoperative atelectasis and pneumonia? - Implications for the physiotherapist. Physiotherapy Theory and Practice. 2000;16:69-80.
Squadrone V, Coha M, Cerutti E, et al. Continuous positive airway pressure for treatment of postoperative hypoxaemia. Journal of the American Medical Association. 2005;293:589-595.
Stiller K, McInnes M, Huff N, et al. Do exercises prevent musculoskeletal complications after cardiac surgery? Physiotherapy Theory and Practice. 1997;13:117-126.
Stiller K, Montarello J, Wallace M, et al. Efficacy of breathing and coughing exercises in the prevention of pulmonary complications after coronary artery surgery. Chest. 1994;105:741-747.
Stockton K. Exercise training in patients with chronic liver disease. Physiotherapy Theory and Practice. 2001;17:29-38.
Tatoulis J, Smith J. Principles and practice of cardiac surgery. In Tjandra J, Clunie G, Kaye A, Smith J, editors: Textbook of surgery, 3rd edn., Massachusetts: Blackwell Publishing, 2006.
Thomas J, McIntosh J. Are incentive spirometry, intermittent positive pressure breathing, and deep breathing exercises effective in the prevention of postoperative pulmonary complications after upper abdominal surgery? A systematic overview and meta-analysis. Physical Therapy. 1994;74:3-16.
Tjandra J. Colorectal cancer and adenoma. In Tjandra J, Clunie G, Kaye A, Smith J, editors: Textbook of surgery, 3rd edn., Massachusetts: Blackwell Publishing, 2006.
Tjandra J, Chan M. Systematic review of the short-term outcome of laparoscopic resection for colon and rectosigmoid cancer. Colorectal Disease. 2006;8:375-388.
Trauma.org, 2004 Pulmonary contusion. www.trauma.org/index.php?/main/article/398/ (Accessed 16 July 2007)
Tucker B, Jenkins S. The effect of breathing exercises with body positioning on regional lung ventilation. Australian Journal of Physiotherapy. 1996;42:219-227.
Wahba R. Perioperative functional residual capacity. Canadian Journal of Anaesthesia. 1991;38:384-400.
Werawatganon T, Charuluxanun S, 2005 Patient-controlled intravenous opioid analgesia versus continuous epidural analgesia for pain after intra-abdominal surgery. Cochrane Database of Systematic Reviews 2005, Issue 1. Art. No: CD004088. DOI: 10.1002/14651858.CD004088.pub2
Westerdahl E, Lindmark B, Eriksson T, et al. Deep-breathing exercises reduce atelectasis and improve pulmonary function after coronary bypass surgery. Chest. 2005;128:3482-3488.
Wightman JAK. A prospective survey of the incidence of postoperative pulmonary complications. British Journal of Surgery. 1968;55:85-91.