HYPERSENSITIVITY DISORDERS
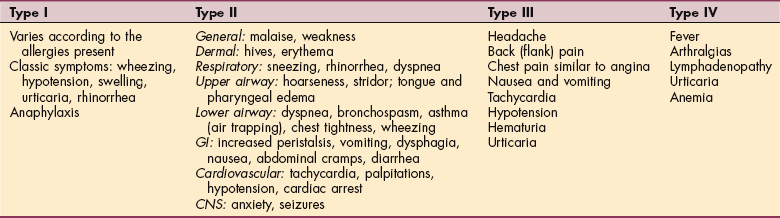
An exaggerated or inappropriate immune response may lead to various hypersensitivity disorders. Such disorders are classified as types I, II, III, or IV, although some overlap exists (Table 7-4).
Overreaction to a substance, or hypersensitivity, is often referred to as an allergic response, and although the term allergy is widely used, the term hypersensitivity is more appropriate. Hypersensitivity designates an increased immune response to the presence of an antigen (referred to as an allergen) that results in tissue destruction. The damage and suffering come predominantly from the immune response itself rather than from the substances that provoke it.
The several types of hypersensitivity reactions include immediate, late phase, and delayed, based on the rapidity of the immune response. Immediate hypersensitivity reactions usually occur within minutes of exposure to an allergen. If the skin is affected, blood vessels dilate and fluid accumulates, causing redness and swelling. In the eyes and nose, increased fluid and mucous secretions cause tearing and a runny nose.
Late-phase inflammation and symptoms persist for hours to days after the allergens are removed and can cause cumulative damage (e.g., progressive lung disease) if they persist. Delayed hypersensitivity reaction occurs after sensitization to certain drugs or chemicals (e.g., penicillin, poison ivy). These reactions often take several days to cause symptoms.
Type I Hypersensitivity (Immediate Hypersensitivity, Allergic Disorders, Anaphylaxis)
Type I hypersensitivity reactions include hay fever, allergic rhinitis, urticaria, extrinsic asthma, and anaphylactic shock. In this type of immediate hypersensitivity response, IgE, instead of IgG, is produced in response to a pathogen (allergen).
The term atopy is often used to describe IgE-mediated diseases. People with atopy have a hereditary predisposition to produce IgE antibodies against common environmental allergens and have one or more atopic diseases.
Allergens are a special class of antigens that cause an allergic response. These normally harmless substances are inhaled (e.g., mold spores, animal dander, dust mites, grasses, weeds), eaten (e.g., nuts, fruits, shellfish, eggs), or injected (e.g., venom from fire ants, wasps, bees, hornets), or they come in contact with the skin or mucous membranes (e.g., plants, cosmetics, metals, drugs, dyes, latex).
IgE resides on mast cells in connective tissue, especially the upper respiratory tract, GI tract, and dermis. When IgE meets the pathogen again, an immediate response occurs with histamine release, along with other inflammatory mediators (e.g., chemotactic factors, prostaglandins, and leukotrienes) that enhance and prolong the response initiated by histamine (Fig. 7-20).
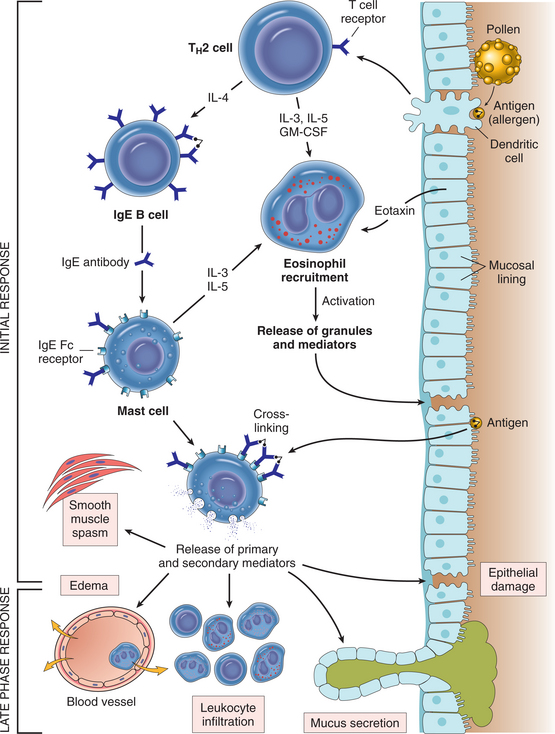
Figure 7-20 Pathogenesis of immediate (type I) hypersensitivity reaction. The late-phase reaction is dominated by leukocyte infiltration and tissue injury. TH2, T-helper type 2 CD4 cells. (Reprinted from Kumar V: Robbins and Cotran: pathologic basis of disease, ed 7, Philadelphia, 2005, Saunders.)
If this response becomes systemic, widespread release of histamine (rather than just local tissue response) results in systemic vasodilation, bronchospasm, increased mucus secretion, and edema, referred to as anaphylaxis.
Classic associated signs and symptoms are wheezing, hypotension, swelling, urticaria, and rhinorrhea (clear, runny nose often accompanied by sneezing) (Fig. 7-21). Anaphylaxis is a life-threatening emergency and requires immediate intervention with injected epinephrine to restore blood pressure, strengthen the heartbeat, and open the airways. Bee stings remain the number one cause of anaphylaxis; other triggers include penicillin, foods, animal dander, children, semen, and latex.
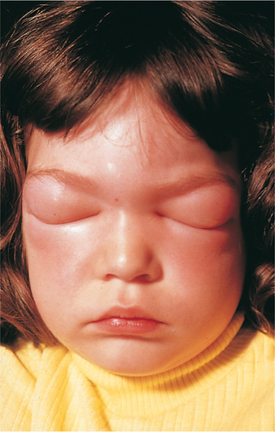
Figure 7-21 Type I hypersensitivity reaction. Severe swelling of the eyelids in this child is the result of an allergic response to a bee sting. In some children and adults, difficulty breathing may be the first symptom of anaphylaxis. Intervention can be delayed until it is too late because there is no visible sign of narrowed airways. (Reprinted from Zitelli BJ, Davis HW: Atlas of pediatric physical diagnosis, ed 4, St Louis, 2002, Mosby.)
A marked increase in the prevalence of atopic disease has occurred in the United States during the last 2 decades, indicating the importance of environmental influences. The mechanisms for this action are outlined in greater detail elsewhere.130
Type II Hypersensitivity (Cytotoxic Reactions to Self-Antigens)
When the body’s own tissue is recognized as foreign or nonself, activation of complement occurs with subsequent agglutination (clumping together) and phagocytosis of the identified pathogens. This means the cellular membrane of normal tissues (e.g., red blood cells [RBCs], leukocytes, and platelets) is disrupted and ultimately destroyed. Self-antigen disorders include blood transfusion reactions, hemolytic disease of the newborn, autoimmune hemolytic anemia, and myasthenia gravis (Fig. 7-22).
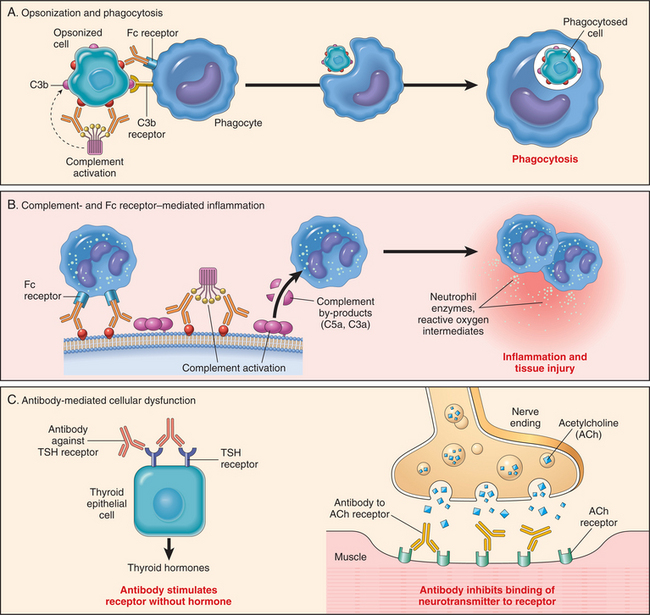
Figure 7-22 Schematic illustration of the three major mechanisms of antibody-mediated injury in type II hypersensitivity reactions. A, Opsonization of cells by antibodies and complement components and ingestion by phagocytes. B, Inflammation induced by antibody binding to Fc receptors of leukocytes and by complement breakdown products. C, Antireceptor antibodies disturb the normal function of receptors. In these examples, antibodies against the thyroid-stimulating hormone (TSH) receptor activate thyroid cells in Graves’ disease, and acetylcholine (ACh) receptor antibodies impair neuromuscular transmission in myasthenia gravis. (Reprinted from Kumar V: Robbins and Cotran: pathologic basis of disease, ed 7, Philadelphia, 2005, Saunders.)
A second type of hypersensitivity response occurs when there is a cross-reaction between exogenous pathogens and endogenous body tissues, such as in rheumatic fever. For example, group A hemolytic streptococci (the exogenous pathogen) are attacked by the immune system, but the body also misinterprets the mitral valve (endogenous body tissue) as a foreign microorganism (i.e., as streptococcus) and attacks normal, healthy tissue in the same way it attempts to destroy the true pathogenic microorganisms. Another example of this type of cross-reaction is an exogenous virus causing the immune system to attack the peripheral nervous system as nonself, such as in acute Guillain-Barré syndrome.
Type III Hypersensitivity (Immune Complex Disease)
Normally, excessive circulating antigen-antibody complexes called immune complexes are effectively cleared by the reticuloendothelial system. When circulating immune complexes (antigen-antibody complexes) successfully deposit in tissues around small blood vessels, they activate the complement cascade and cause acute inflammation and local tissue injury (Fig. 7-23).
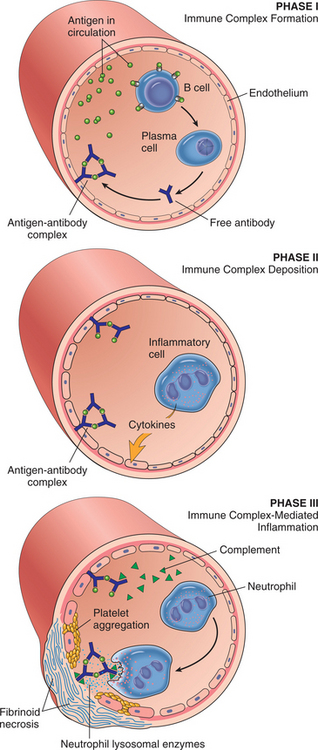
Figure 7-23 Schematic illustration of the three sequential phases in the induction of systemic immune complex–mediated disease (type III hypersensitivity). (Reprinted from Kumar V: Robbins and Cotran: pathologic basis of disease, ed 7, Philadelphia, 2005, Saunders.)
The subsequent vasculitis most commonly affects the skin, causing urticaria (wheals); joints, causing synovitis, such as in rheumatoid arthritis; kidneys, causing nephritis; pleura, causing pleuritis; and pericardium, causing pericarditis.
Systemic lupus erythematosus (SLE) is the classic picture of vasculitis, occurring in various organ systems. The antigen is the individual’s own nucleus of cells; antinuclear antibodies (ANAs) are made, which in turn form a complex with the antigen and are deposited in the skin, joints, and kidneys, causing acute immune injury. Other examples of this hypersensitivity reaction occur in association with infections such as HBV and bacterial endocarditis, malignancies, or after drug or serum therapy.
Type IV Hypersensitivity (Cell-Mediated Immunity)
Type IV is a delayed hypersensitivity response such as the reaction that occurs in contact dermatitis after sensitization to an allergen (commonly a cosmetic, adhesive, topical medication, or plant toxin such as poison ivy), latex sensitivity, or the response to a TB skin test present 48 to 72 hours after the test.
In this type of reaction, the antigen is processed by macrophages and presented to T cells. The sensitized T cells then release lymphokines, which recruit other lymphocytes, monocytes, macrophages, and PMNs (Fig. 7-24). Graft-versus-host disease and transplant rejection are also examples of type IV reactions (see Chapter 21).
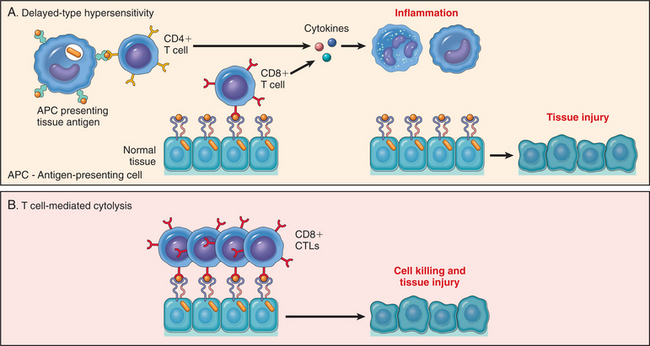
Figure 7-24 Mechanisms of T cell–mediated (type IV) hypersensitivity reactions. A, In delayed-type hypersensitivity reactions, CD4+ T cells (and sometimes CD8+ cells) respond to tissue antigens by secreting cytokines that stimulate inflammation and activate phagocytes, leading to tissue injury. B, In some diseases, CD8+ cytolytic T lymphocytes (CTLs) directly kill tissue cells. (Reprinted from Kumar V: Robbins and Cotran: pathologic basis of disease, ed 7, Philadelphia, 2005, Saunders.)
AUTOIMMUNE DISEASES
Autoimmune diseases fall into a category of conditions in which the cause involves immune mechanisms directed against self-antigens. More specifically, the body fails to distinguish self from nonself, causing the immune system to direct immune responses against normal (self) tissue and become self-destructive.
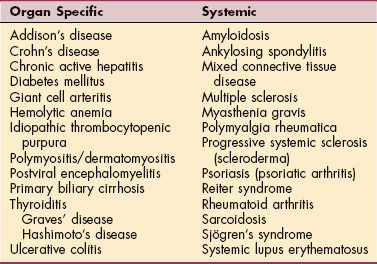
More than 56 autoimmune diseases have been identified, affecting everything from skin and joints to vital organs. Autoimmune diseases can be viewed as a spectrum of disorders, some of which are systemic and others of which involve a single organ. A portion of the known diseases most likely to be seen in a rehabilitation setting is listed in Table 7-5.
At one end of the continuum are organ-specific diseases, in which localized tissue damage occurs, resulting from the presence of specific autoantibodies. An example is Hashimoto’s disease of the thyroid, characterized by a specific lesion in the thyroid gland with production of antibodies with absolute specificity for certain thyroid constituents.
In the middle of the continuum are disorders in which the lesion tends to be localized in one organ, but the antibodies are not organ specific. An example is primary biliary cirrhosis, in which inflammatory cell infiltration of the small bile ductule occurs, but the serum antibodies are not specific to liver cells.
At the other end of the spectrum are non–organ-specific diseases, in which lesions and antibodies are widespread throughout the body and not limited to one target organ. SLE is an example of this type of autoimmune disease. Identification of ANAs that attack the nucleic acids (DNA and RNA) and other components of the body’s own tissues established SLE as an autoimmune disease.
In this book, with the few exceptions included in this chapter (e.g., fibromyalgia, CFS, SLE), autoimmune disorders are discussed individually in the most appropriate chapter. For example, Reiter’s syndrome, rheumatoid arthritis, and Sjögren’s syndrome are discussed in Chapter 27. Polymyositis, dermatomyositis, and progressive systemic sclerosis are discussed in Chapter 10. Giant cell arteritis is discussed in Chapter 12, sarcoidosis in Chapter 15, and so on. More information is available on autoimmune-related diseases.6
Etiologic and Risk Factors
Although the autoimmune disorders are regarded as acquired diseases, their causes often cannot be determined. Autoimmunity is believed to result from a combination of factors, including genetic, hormonal (women are affected more often than men by autoimmune diseases), and environmental influences (e.g., exposure to chemicals, other toxins, or sunlight and drugs that may destroy suppressor T cells).
Although no single gene has been identified as responsible for autoimmune diseases, clusters of genes seem to increase susceptibility. In most autoimmune disorders, a known or suspected genetic susceptibility is evident, and certain HLA types show increased risk, such as ankylosing spondylitis with HLA-B27 (see Table 40-20).
The influence of hormonal factors is confusing since some autoimmune diseases occur among women in their 20s and 30s, when estrogen is high, and others develop after menopause or before puberty, when estrogen levels are low. During pregnancy, many women with rheumatoid arthritis or MS experience complete remission, whereas pregnant women with SLE often experience exacerbations.
Other factors implicated in the development of immunologic abnormalities resulting in autoimmune disorders include viruses, stress, cross-reactive antibodies, and various autoimmune diseases occurring in women who have had silicone gel breast implants. This organ-specific autoimmune disease has been associated with musculoskeletal problems.
Pathogenesis
Autoimmune disorders involve disruption of the immunoregulatory mechanism, causing normal cell-mediated and humoral immune responses to turn self-destructive, resulting in tissue damage.
The exact pathologic mechanisms for this process remain unknown. The importance of the innate immune system in determining whether T cells become activated and functional in autoimmune disorders has been shown.168 Researchers suspect that more than one part of the immune system must be involved for autoimmune disease to develop.
Some autoimmune diseases affect a single organ (e.g., pancreas in type 1 diabetes), whereas others affect a large system or more than one system (e.g., MS). In some cases the autoimmune process overstimulates organ function, as in Graves’ disease, in which excess thyroid hormone is produced.
Gene-mapping studies have demonstrated that allergy and autoimmunity must involve not only the recognition of antigen by T cells, but also the very important immunoregulatory effects of cytokines, inhibitory receptors, and survival factors. Linkage analysis of the human genome has revealed candidate loci for susceptibility to MS, type 1 diabetes, SLE, and Crohn’s disease. Continued genetic analysis is ongoing to identify the specific genetic link in hopes of finding a more specific treatment.123
Although antibodies and T-cell receptors can accurately distinguish between closely related antigens, they sometimes cross-react with apparently unrelated antigens, either because the two antigens happen to share an identical epitope (see Fig. 7-2) or because two different epitopes have similar shapes and charges. Such cross-reactions may be the underlying pathogenesis of some autoimmune diseases.61
Many autoimmune diseases are associated with characteristic autoantibodies. In other words, the body begins to manufacture antibodies directed against the body’s own cellular components or specific organs. These antibodies are known as autoantibodies, in this case, producing autoimmune diseases.
For example, SLE is associated with anti-DNA and anti–splice sosomal (Sm) antigen, Sjögren’s syndrome is associated with anti-ribonucleoproteins (SS-A and SS-B), progressive systemic sclerosis is associated with anticentromere and anti–Scl-70 (DNA topoisomerase), psoriasis and psoriatic arthritis are associated with HLA-B13, and mixed connective tissue disease is associated with anti-ribonucleoprotein without anti-DNA.
Antibodies specific to hormone receptors on the surface of cells have been found and determined to be partially responsible for some conditions. Examples include myasthenia gravis, in which antiacetylcholine receptor antibodies are involved; Graves’ disease, in which antibodies against components of thyroid cell membranes, including the receptors for thyroid-stimulating hormone, are responsible; and certain cases of insulin-resistant diabetes mellitus, in which the antibodies affect insulin receptors on cells.
Other diseases involving autoimmune mechanisms include rheumatic fever, rheumatoid arthritis, autoimmune hemolytic anemia, idiopathic thrombocytopenic purpura, and postviral encephalomyelitis.
Clinical Manifestations
Autoimmune disorders share certain clinical features, and differentiation among them is often difficult because of this. Common findings include synovitis, pleuritis, myocarditis, endocarditis, pericarditis, peritonitis, vasculitis, myositis, skin rash, alterations of connective tissues, and nephritis. Constitutional symptoms such as fatigue, malaise, myalgias, and arthralgias are also common.
MEDICAL MANAGEMENT
Diagnosis can be difficult because autoimmune diseases are poorly understood, mimic one another, and often consist of vague symptoms such as lethargy or migratory joint pain. Laboratory tests may reveal thrombocytopenia, leukopenia, immunoglobulin excesses or deficiencies, ANAs, rheumatoid factor, cryoglobulins, false-positive serologic tests, elevated muscle enzymes, and alterations in serum complement. The Coombs’ test is positive when hemolytic anemia is present.
Some of the laboratory alterations that occur in autoimmune diseases (e.g., false-positive serologic tests, rheumatoid factor) occur in asymptomatic people. These changes may also be demonstrated in certain asymptomatic relatives of people with connective tissue diseases, in older individuals, those taking certain medications, and people with chronic infectious diseases.
TREATMENT.
Treatment of autoimmune diseases varies with the specific disease. Treatment must maintain a delicate balance between adequate suppression of the autoimmune reaction to avoid continued damage to the body tissues and maintenance of sufficient functioning of the immune mechanism to protect the person against foreign invaders. In general, autoimmune diseases are treated by the administration of corticosteroids to produce an antiinflammatory effect and salicylates to provide symptomatic relief.
The wealth of new information gleaned from research in the last decade has been used to improve immunization strategies and hopefully will lead to new approaches to the reinduction of immune tolerance. The development of an effective vaccine is under close scrutiny,123 as is the use of intense immunosuppression (immunoablation) followed by stem cell transplantation for the treatment of autoimmune diseases.
Since autoimmune disease is the result of genetic dysregulation, gene therapy may become a viable alternative in the future. Scientists have been involved in developing new drugs aimed at the mechanism of autoimmunity rather than treating its effects. Based on new information about the function of Fc receptors, which bind antibodies that are instructing the immune system in the destructive inflammation characteristic of autoimmune diseases, scientists are looking for blocking compounds to prevent this interaction.
Systemic Lupus Erythematosus
Lupus erythematosus, sometimes referred to as lupus, is a chronic inflammatory autoimmune disorder that appears in several forms, including discoid lupus erythematosus (DLE), which affects only the skin (usually face, neck, scalp) (see Chapter 10), and systemic lupus erythematosus (SLE), which can affect any organ or system of the body.
The clinical picture of SLE presents on a continuum with different combinations of organ system involvement. The most common of these presentations are latent lupus, drug-induced lupus, antiphospholipid antibody syndrome, and late-stage lupus. Latent lupus describes a constellation of features suggestive of SLE but does not qualify as classic SLE (Box 7-7). Many people with latent lupus persist with their clinical presentation of signs and symptoms over many years without ever developing classic SLE.
Drug-induced lupus may be diagnosed in people without prior history suggestive of SLE in whom the clinical and serologic manifestations of SLE develop while the person is taking a drug (most often hydralazine used to treat hypertension or procainamide used to treat arrhythmia). The symptoms cease when the drug is stopped, with gradual resolution of serologic abnormalities.
Antiphospholipid antibody syndrome describes the association between arterial and venous thrombosis, recurrent fetal loss, and immune thrombocytopenia with a variety of antibodies directed against cellular phospholipid (lipids in cell membranes containing phosphorus) components. This syndrome may be part of the clinical manifestations seen in SLE, or it may occur as a primary form without other clinical features of lupus.
Late-state lupus is defined as chronic disease duration of greater than 5 years. In such cases, morbidity and mortality are affected by long-term complications of SLE that result either from the disease itself or as a consequence of its therapy. These late complications may include end-stage renal disease, atherosclerosis, pulmonary emboli, and avascular necrosis. In late-stage lupus, when no evidence of active disease exists and the client is on low-dose or no corticosteroids, cognitive disabilities are a common manifestation.
Incidence
SLE is primarily a disease of young women; it is rarely found in older people. It usually develops in young women of childbearing years, but many men and children also develop lupus. Lupus is three times more common in African American women than in Caucasian women and is also more common in women of Hispanic, Asian, and Native American descent.
SLE also appears in the first-degree relatives of individuals with lupus more often than it does in the general population, which indicates a strong hereditary component. However, most cases of SLE occur sporadically, indicating that both genetic and environmental factors play a role in the development of the disease.
Etiologic and Risk Factors
The cause of SLE remains unknown, but evidence points to interrelated immunologic, environmental, hormonal, and genetic factors. Whether SLE represents a single pathologic entity with variable expression or a group of related conditions remains unknown. Immune dysregulation in the form of autoimmunity is thought to be the prime causative mechanism. SLE shows a strong familial link, with a much higher frequency among first-degree relatives. Evidence for genetic susceptibility is present, and linkage studies in conjunction with genome scans may delineate this more specifically in the future.270
Genetically determined immune abnormalities may be triggered by both exogenous and endogenous factors. Although the predisposition to disease is hereditary, it is likely to involve different sets of genes in different individuals. As the human genome becomes more extensively mapped, a susceptibility gene may be found, although it remains possible that the differences in disease course among ethnic groups relate solely to their environment and other social factors.
Other factors predisposing to SLE may include physical or mental stress, which can provoke neuroendocrine changes affecting immune cell function; streptococcal or viral infections; exposure to sunlight or ultraviolet light, which can cause inflammation and tissue damage; immunization; pregnancy; and abnormal estrogen metabolism.
Whether pregnancy induces lupus flare-ups has not been established; existing data suggest both that it does and does not. More studies are needed to further determine the effects of pregnancy on this condition.
A higher incidence of SLE exacerbation occurs among women taking even low-dose estrogen contraceptives. Since an increased risk of thrombosis is possible in young women with SLE, estrogen-containing contraceptives are avoided or used at the lowest effective dose.
No evidence exists that postmenopausal estrogen replacement therapy is associated with SLE flare-ups, and since women in this age range are at increased risk for coronary artery disease and osteoporosis, estrogen replacement therapy can be taken. For all women with SLE who have been treated with cyclophosphamide, an increased risk of gynecologic malignancy is evident.190
The role of the Epstein-Barr virus as a possible risk factor for SLE remains under investigation.117 SLE may also be triggered or aggravated by treatment with certain drugs (e.g., hydralazine, anticonvulsants, penicillins, sulfa drugs, and oral contraceptives), which could modify both cellular responsiveness and immunogenicity of self-antigens.
Pathogenesis
The central immunologic disturbance in SLE is autoantibody production. The body produces antibodies (e.g., ANAs) against its own cells. Deposition of the formed antigen-antibody complexes at various tissue sites can suppress the body’s normal immunity and damage tissues.
In fact, one significant feature of SLE is the ability to produce antibodies against many different tissue components such as RBCs, neutrophils, platelets, lymphocytes, or almost any organ or tissue in the body. This wide range of antigenic targets has resulted in SLE being classified as a disease of generalized autoimmunity. Given the clinical diversity of SLE, the disease may be mediated by more than one autoantibody system and several immunopathogenic mechanisms.
Specific pathologic findings are organ dependent; for example, repeat biopsies of the kidney show inflammation; cellular proliferation; basement membrane abnormalities; and immune complex deposition comprised of IgM, IgG, and IgA.
Skin lesions demonstrate inflammation and degeneration at the dermal-epidermal junction, with the basal layer being the primary site of injury. Other organ systems affected by SLE are usually studied only at autopsy. Although these tissues may show nonspecific inflammation or vessel abnormalities, pathologic findings are sometimes minimal, suggesting a mechanism other than inflammation as the cause of organ damage or dysfunction.
Clinical Manifestations
Generally, SLE is more severe than discoid lupus, and no two people with SLE will have identical symptoms. For some people, only the skin and joints will be involved. For others, joints, lungs, kidneys, blood, or other organs and/or tissues may be affected.
Musculoskeletal.: Arthralgias and arthritis constitute the most common presenting manifestations of SLE, but the onset of SLE may be acute or insidious and may produce no characteristic clinical pattern. Other early symptoms may include fever, weight loss, malaise, and fatigue.
Acute arthritis can involve any joint but typically affects the small joints of the hands, wrists, and knees. It may be migratory or chronic; most cases are symmetrical, but asymmetrical polyarthritis is not uncommon.
Unlike rheumatoid arthritis, the arthritis of SLE is not usually erosive or destructive of bone, and symptoms are not usually severe enough to cause joint deformities, but pain can cause temporary functional impairment. When deformities do occur, ulnar deviation, swan-neck deformity, or fixed subluxations of the fingers often occur as well. Tenosynovitis and tendon ruptures may occur.
Cutaneous and Membranous Lesions.: The skin rash occurs most commonly in areas exposed to sunlight (ultraviolet rays) and may be exacerbated by the use of cosmetic products containing alpha hydroxy acids. The classic butterfly rash over the nose and cheeks is common (Fig. 7-25; see also Fig. 10-22).
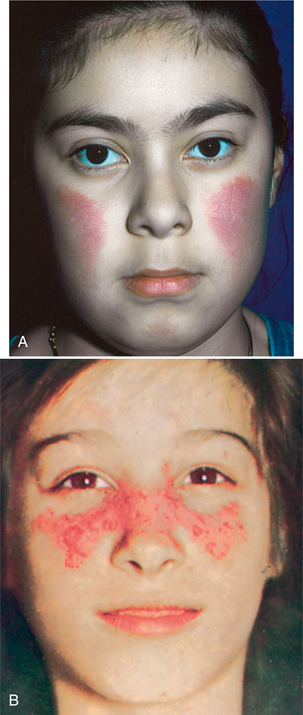
Figure 7-25 The butterfly rash of SLE. The rash can vary from an erythematous blush (A) to thickened epidermis to scaly patches (B). (Reprinted from Kliegman R, et al: Nelson textbook of pediatrics, ed 18, Philadelphia, 2007, Saunders.)
Discoid lesions associated with DLE are raised, red, scaling plaques with follicular plugging and central atrophy (see Fig. 10-20). This raised edging and sunken center gives them a coin-like appearance (see further discussion, Chapter 10).
Vasculitis (inflammation of cutaneous blood vessels) involving small-and medium-size vessels may cause other skin lesions, including infarctive lesions of the digits (Fig. 7-26), splinter hemorrhages, necrotic leg ulcers, or digital gangrene. Raynaud’s phenomenon occurs in about 20% of people.
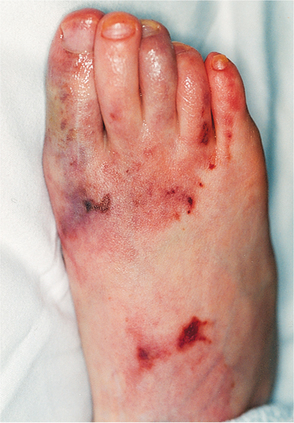
Figure 7-26 A 12-year-old girl with SLE and antiphospholipid antibodies with painful cutaneous vasculitis of the right foot. Arterial thrombosis documented by angiography resulted in cyanosis of the large toe. Symptoms resolved with treatment with heparin and corticosteroids. (Reprinted from Kliegman R, et al: Nelson textbook of pediatrics, ed 18, Philadelphia, 2007, Saunders.)
Diffuse or patchy alopecia (hair loss) may be temporary, with hair regrowth once the disease is under control. However, permanent hair loss can occur from the extensive scarring of discoid lesions. Painless ulcers of the mucous membranes are common involving the mouth, vagina, and nasal septum.
Cardiopulmonary System.: Signs of cardiopulmonary abnormalities may develop, such as pleuritis, pericarditis, and dyspnea. Myocarditis, endocarditis, tachycardia, and pneumonitis (acute or chronic) may also occur. Pulmonary hypertension and congestive heart failure are less common and usually secondary to a combination of factors. Anyone with SLE with the antiphospholipid antibody syndrome is at a high risk of thrombosis (see the section on Collagen-Vascular Disease in Chapter 12).
Central Nervous System.: A significant number of people with SLE will have CNS involvement at some point in their illness, sometimes referred to as neuropsychiatric manifestations. Clinical manifestations may be related to specific autoantibodies that react with nervous system antigens and/or cytokine-mediated brain inflammation and include headaches, irritability, and depression (most commonly).
Emotional instability, psychosis, seizures, cerebrovascular accidents, cranial neuropathy, peripheral neuropathy, and organic brain syndrome can also occur. Return to the previous level of intellectual function may follow remission of the neuropsychiatric flare, or permanent cognitive impairment may occur.
The pattern of cognitive dysfunction is diverse; intensity can vary within the same person and can be affected by mood.62 The person may have difficulties with verbal memory, attention, language skills (verbal fluency, productivity), and psychomotor speed. Progressive cognitive impairment, sometimes subtle and sometimes obvious, may develop even in the absence of clinically diagnosed episodes of neuropsychiatric disease.63 People with SLE may or may not have other signs of lupus when they experience neurologic symptoms.
Renal System.: Pathologic changes may also occur in the kidneys, where the glomerulus is the usual site of destruction; other renal effects may include hematuria and proteinuria, progressing to kidney failure. Antiphospholipid antibodies are a significant cause of morbidity and mortality in cases of renal involvement as a result of thrombosis and the development of thrombotic microangiopathy.121
Other Systems.: Anemia from decreased erythrocytes is a common finding, with associated amenorrhea (cessation of menstrual flow) among women. Sometimes the spleen and cervical, axillary, and inguinal nodes are enlarged; hepatitis may also develop. Nausea, vomiting, diarrhea, and abdominal pain may occur with GI involvement. All symptoms mentioned in this section can occur at the onset or at any time during the course of lupus. Nearly all people with SLE experience fluctuations in disease activity with exacerbations and remissions.
MEDICAL MANAGEMENT
There is no known way to prevent SLE, but preventive measures can reduce the risk of flare-ups. For photosensitive people, avoidance of (excessive) sun exposure and/or the regular application of sunscreen usually prevents rashes. Regular exercise helps prevent muscle weakness and fatigue. Immunization protects against specific infections.
Support groups; counseling; and reliance on family members, friends, and health care professionals can help alleviate the effects of stress. Lifestyle choices and personal behavior are very important for people with SLE. These include smoking, excessive consumption of alcohol, too much or too little of prescribed medication, or postponing regular medical checkups.144
DIAGNOSIS.
Diagnosis of SLE is difficult because it often mimics other diseases and the symptoms are often vague, varying greatly from individual to individual. The American Rheumatism Association has issued a list of criteria for classifying SLE to be used primarily for consistency in epidemiologic surveys. Usually four or more of these signs are present at some time during the course of the disease (see Box 7-7).
In addition to the routine history and physical examination, laboratory findings are an important part of the diagnosis of SLE and subsequent monitoring of clinical disease activity. Specific test procedures and their significance are available.143 ANAs are present in all cases of SLE, but their presence does not make a definitive diagnosis. However, if ANAs are absent, SLE is probably not present. Lupus anticoagulant testing and immunologic antiphospholipid establish the presence of antiphospholipid syndrome.
Magnetic resonance imaging scans of the head are usually ordered for all people experiencing new episodes of focal neurologic deficits, seizures, altered consciousness, or psychosis. Neuropsychologic assessment may be helpful for identifying subtle, clinically latent sequelae of CNS events, such as stroke, and in monitoring the response to drug treatment.62
TREATMENT.
The objectives of medical intervention are to reverse the autoimmune and inflammatory processes and prevent exacerbations and complications. At the present time, pharmacologic interventions are the primary means of accomplishing these goals.
Mild symptoms can be managed with NSAIDs to relieve muscle and joint pain while reducing tissue inflammation. Corticosteroid-sparing agents (e.g., methotrexate) used earlier preserve bone and offer protection from premature cardiovascular disease. Anticoagulants for individuals who have antiphospholipid antibody syndrome and coagulopathies ensure a more favorable outcome.
Antimalarial agents (e.g., chloroquine [Aralen], hydroxychloroquine [Plaquenil]) are useful against the dermatologic, arthritic, and renal symptoms of this disease. Immunomodulating drugs (e.g., azathioprine [Imuran], cyclophosphamide [Cytoxan]) are immunosuppressive drugs used to suppress inflammation and subsequently the immune system. These are used only with active disease, especially with severe kidney involvement. Corticosteroids and cytotoxic drugs are given in more severe disease that has not responded to these other types of drug therapy.
Treatment in the future may be more specific as knowledge of genes that participate in the predisposition, pathogenesis, pharmacogenetics of, and protection against this disease come to light. Better understanding of the role of sex hormones has allowed trials of weak androgens or prolactin inhibitors.
New immunomodulators or immunosuppressants, immune ablation with subsequent stem cell transplantation, and more precise immunoregulation (e.g., tolerance-induction strategies; intervention at the level of T-cell costimulation; targeting of cytokines, complement, and FcγR) may become standard intervention tools.23,134
PROGNOSIS.
The prognosis improves with early detection and intervention that prevents organ damage and improves life expectancy. The overall reduction in the use of large doses of corticosteroids over the past 2 decades has significantly reduced morbidity and mortality rates.
People with SLE have an increased prevalence of valvular and atherosclerotic heart disease, apparently because of factors related to the disease itself and to drug therapy necessary in severe cases. Symptomatic large-vessel occlusive disease in SLE, occurring several years after the diagnosis of the disease, is associated with a relatively poor short-term outcome. There is an increased risk of certain cancers in SLE; the risk appears to be most heightened for lymphoma.19
Prognosis is less favorable for those who develop cardiovascular, renal, or neurologic complications or severe bacterial infections. High stress, poor social support, and psychologic distress are modifiable factors associated with health outcomes for people with SLE.66,261
Fibromyalgia
Fibromyalgia or fibromyalgia syndrome (FMS), formerly mislabeled or misdiagnosed as fibrocytis, fibromyositis, myofascial pain, CFS, or SLE, is a chronic muscle pain syndrome. It is considered a syndrome and not a disease and has now been defined by the American College of Rheumatology as pain that is widespread in at least 11 of 18 tender points (see Clinical Manifestations and Diagnosis in this section).
FMS currently falls under the auspices of rheumatology, having originally been determined to have no known organic basis. However, with the recent advances in understanding of FMS with documented objective biochemical, endocrine, and physiologic abnormalities, it may be best characterized as a biologic (organic) disorder associated with neurohormonal dysfunction of the ANS.
It is commonly associated with many other conditions (e.g., hypothyroidism, rheumatoid arthritis, connective tissue disease, SLE, CFS); the link between these disorders is under investigation.
Fibromyalgia has been differentiated from myofascial pain (see the section on Myofascial Pain Syndrome in Chapter 27) in that fibromyalgia is considered a systemic problem with widespread multiple tender points as one of the key symptoms.
Myofascial pain is a localized condition specific to a muscle and may involve as few as one or as many as several areas with characteristic trigger points that are painful and refer pain to other areas when pressure is applied. The person with FMS may have both tender points and trigger points requiring specific treatment interventions for each.
The person with myofascial pain syndrome does not exhibit other associated constitutional or systemic signs or symptoms unless palpation elicits a painful enough response to elicit an ANS response with nausea and/or vomiting, increased blood pressure, and increased pulse.
It has been proposed that fibromyalgia and CFS are two names for the same syndrome, with CFS being an early form of FMS, but at present CFS is thought to differ by the greater degree of fatigue. People with fibromyalgia tend to experience more pain. In contrast to CFS, fibromyalgia is associated with a variety of initiating or perpetuating factors such as psychologically distressing events, primary sleep disorders, inflammatory rheumatic arthritis, and acute febrile illness.
Fibromyalgia and CFS have similar disordered sleep physiology, and evidence suggests a reciprocal relationship of the immune and sleep-wake systems. Interference with either system has effects on the other and will be accompanied by the symptoms of CFS.173 A significant number of people with FMS meet the criteria for CFS and vice versa.
Incidence
Fibromyalgia occurs in over 6 million Americans. It has now surpassed rheumatoid arthritis as the most common musculoskeletal disorder in the United States. Women are affected more often than men (90% are women), with symptoms appearing between the ages of 20 and 55 years, although it has been diagnosed in children as young as 6 years and adults as old as 85 years.
Risk Factors
Risk factors or triggering events for the onset of fibromyalgia may include prolonged anxiety and emotional stress, trauma (e.g., motor vehicle accident, work injury, surgery), rapid steroid withdrawal, hypothyroidism, and viral and nonviral infections.
Fibromyalgia may also develop with no obvious precipitating events or illnesses. It is more prevalent in minimally to moderately physically fit persons and is not usually found in highly trained athletes; a strong correlation exists between fibromyalgia and anxiety or depression (it remains unclear whether these factors are contributory or a result of this condition).
Women with extracapsular silicone (silicone gel outside of the fibrous scar that forms around breast implants) as a result of rupture are more likely to report having fibromyalgia, but more data are needed to confirm this association.24,25
Etiologic Factors
Research is now ongoing to determine the cause of fibromyalgia; most likely the initiation of this condition is multifactorial (Fig. 7-27). Debate continues over whether fibromyalgia is even an organic disease and, if so, whether it is caused by abnormal biochemical, metabolic, or immunologic pathology.
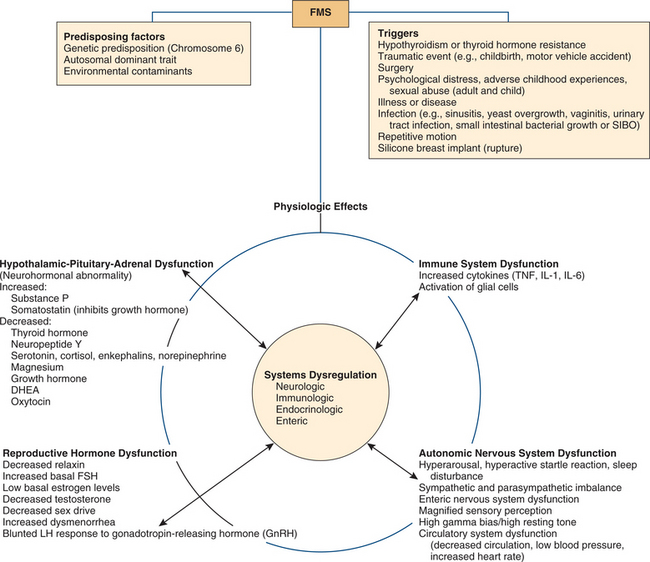
Figure 7-27 Multifactorial causes of FMS. There are many hypotheses and models of how multiple factors contribute to the development of FMS. This model represents data thus far to support FMS as a biologic (organic) disorder caused by neurohormonal dysfunction of the ANS. The physiologic effects of four primary systems dysfunction are listed.
Possible etiologic theories include diet; viral origin; sleep disorder; occupational, seasonal, or environmental influences; psychologic distress; adverse childhood experiences, including sexual abuse73,163; and a familial or hereditary link.
Pathogenesis
Both central and peripheral mechanisms may operate in the pathophysiology of impaired muscle function and pain in fibromyalgia. A disturbance in four regulatory systems of the body has been identified in people with FMS: (1) the hypothalamic-pituitary-adrenal axis (HPA), (2) the ANS, (3) the reproductive hormone axis (RHA), and (4) the immune system. Although these systems function independently, each system influences the other, and each helps regulate cellular function. Disruption of one system can influence the other systems, disrupting cellular function.112
Hypothalamic-Pituitary-Adrenal Axis.: The HPA axis is considered the stress system of the body, affecting the body’s ability to cope with stress, both psychoemotional and biologic, including dysfunction in metabolic and physiologic processes controlling blood pressure, blood sugar levels, infection control, and so on. The hypothalamus, pituitary, and adrenal glands produce chemical messengers that modulate pain, sleep, mood, sex drive, appetite, energy, and circulation. Many of the HPA axis hormones are found to be at abnormal levels in FMS (see Fig. 7-27).112
Substance P, a neurotransmitter for pain, may play a role in the transmission of nociceptive information. The inhibitory system acts to lessen or filter out some of the painful signals transmitted to the brain. These pain stimuli are usually transmitted by substance P.
Increased activity of substance P may explain an abnormally decreased pain threshold in fibromyalgia.271 Elevated levels of substance P have been found in the cerebrospinal fluid of fibromyalgia clients, resulting in an exaggerated response to normal stimuli and an amplified effect on pain. People with FMS are not just sensitive to pain, they also find loud noises, odors, and bright lights aversive.
The role of other pain-inhibiting neurotransmitters, such as serotonin, γ-aminobutyric acid, enkephalins, epinephrine, and norepinephrine has been studied.93 Although substance P is elevated, decreased levels of all of these neurotransmitters in the cerebrospinal fluid have been observed.
Serotonin, a CNS neurotransmitter that is made from tryptophan (an essential amino acid obtained from the diet), is necessary for restorative sleep and appears to play a role in pain control, immune system function, vascular constriction and dilation, and even emotions that may contribute to such feelings as depression or anxiety. Earlier studies found the concentration of serotonin end products (metabolites) to be lower than normal in clients with fibromyalgia, supporting a hypothesis of aberrant pain perception resulting from a deficiency of serotonin.217-219
However, clinical trials showed that conventional FMS interventions such as tricyclic antidepressants used to increase the amount of serotonin at synapses were no more effective than placebo in improving or alleviating FMS symptoms.32 This suggests that it is unlikely FMS results primarily from a serotonin deficiency, but perhaps another mechanism that causes serotonin deficiency and the other features of this condition.
Available new evidence suggests that impaired metabolism caused by inadequate thyroid hormone regulation at the cellular level may be the underlying pathogenesis. The inadequate regulation of cell function may result from a thyroid hormone deficiency or from cellular resistance to normal levels of thyroid hormone.158,159 Whether this inadequate thyroid hormone regulation is triggered by genetic mutations208 and/or can be attributed to environmental contaminants167 remains unproved.
Abnormalities in the function of the HPA-kidney-bladder axis may account for the irritable bladder syndrome and the female urethral syndrome characterized by urinary frequency and urgency. Low blood pressure and blood volume (ANS dysfunction) may also contribute to this condition. Studies in this area are very limited at this time.
Autonomic Nervous System.: The activity of the skeletal muscles, heart, stomach, intestines, blood vessels, and sweat glands during daily stress tends to be excessive in fibromyalgia. These organs overactivate, resulting in the heart beating faster, the stomach secreting excessive digestive juices and contracting erratically, the smooth muscles of the intestines and bowel contracting abnormally, breathing becoming rapid and shallow, and blood vessels constricting, which decreases blood flow to body parts. These and other ANS responses may occur in response to a relatively mild life stressor and linger even after cognitive memory of the event is gone.
People who do not have fibromyalgia experience these changes, but the autonomic responses occur in smaller amplitude and for a shorter period before returning to normal levels. In FMS the nervous system’s ability to modulate and return to normal is fragile and lacks the subtle ability to respond quickly; responses are more exaggerated and the return to normal takes more time.112,271
The enteric system (autonomic nervous control of the digestive system) is often significantly disrupted in fibromyalgia. Digestion is often compromised, and the absorption of nutrients into the bloodstream where they can be used by the body for cell function is often inadequate for healthy daily function. The enteric system’s interaction with other systems (e.g., brain, immune system) links effects of nutritional deficits to other functions as well.112
Sleep disturbances may contribute to fibromyalgia symptoms; researchers are investigating alterations of the neuroimmunoendocrine systems that accompany disordered sleep physiology, resulting in the nonrestorative sleep, pain, fatigue, and cognitive and mood symptoms that people with fibromyalgia (and CFS) experience.
People affected do not enter restorative sleep (phase IV sleep) or rapid eye movement (REM) sleep. Deficiency of non-REM sleep also contributes to sleep disturbance by reducing the amount of time the muscles enter a state of resting muscle tone. Eighty percent of the body’s growth hormone is secreted by the pituitary gland (under hypothalamic control) during deep sleep, and it is crucial for normal muscle metabolism and tissue repair. Substantial nighttime decreases in growth hormone have been reported in FMS.145
These types of sleep disturbances are not unique to fibromyalgia but have been observed in many people with rheumatoid arthritis, osteoarthritis, and other painful rheumatic diseases.
The Reproductive Hormone Axis.: Reproductive hormones help regulate the HPA axis in a bidirectional feedback loop (see Fig. 11-2). During chronic stress, a decrease in function occurs in both the HPA and the RHA, with diminished reproductive capability, fatigue, sleep disruption, and illness or exacerbation of FMS. Female reproductive hormones, especially estrogen and progesterone, exert influence over menstrual cycle, bowel and bladder function, blood pressure, sleep cycles, endorphins, serotonin levels, thyroid function, digestive activity, sex drive, sense of well-being, and much more.
The onset or exacerbations of fibromyalgia often occur around or during the time of sex hormone–related events (e.g., menses, pregnancy, childbirth, perimenopause, menopause), but few studies exist to study the relationship between these cycles and fibromyalgia. The possibility of inadequate thyroid hormone regulation of the hypothalamic-pituitary-gonadal axis for men and women has been suggested.158
Immune System.: Finally, a model for pathologic pain syndromes such as FMS and CFS has been formulated based on pain facilitory effects produced by the immune system. Immune cells, activated in response to infection, inflammation, or trauma, release proinflammatory cytokines that signal the CNS to release glia within the brain and spinal cord. Pain has been classically viewed as being mediated solely by neurons, but the discovery that spinal cord glia (microglial and astrocytes) amplify pain has changed this view.
When glial cells become activated by sensory signals arriving from the periphery, they can release a variety of substances known to be involved in chronic pain (e.g., nerve growth factor, excitatory amino acids, nitric oxide), and they can also control the release of neurotransmitters (e.g., substance P).
Once activated, such as when viruses and bacteria enter the CNS, glial cells cause prolonged release of proinflammatory cytokines (e.g., TNF, IL-1, IL-6), creating an exaggerated pain state. Glia may be the key driving force for the pain created by tissue inflammation and nerve injury because they can increase the release of pain transmitters and cytokines from the neurons in the surrounding area, and they are connected to large networks that allow activation of glia at distant sites.
This pain model emphasizes again the need for anyone with FMS to minimize pain-generating aggravants such as infectious agents, trauma, and inflammation or other triggers (see Fig. 7-27).262
Clinical Manifestations
Fibromyalgia is characterized by muscle pain as the major symptom, often described as aching or burning, a “migraine headache of the muscles.” Diffuse pain or tender points are present on both sides of the body in many muscle groups, including the neck, back, arms, legs, jaw, feet, and hands (Fig. 7-28).
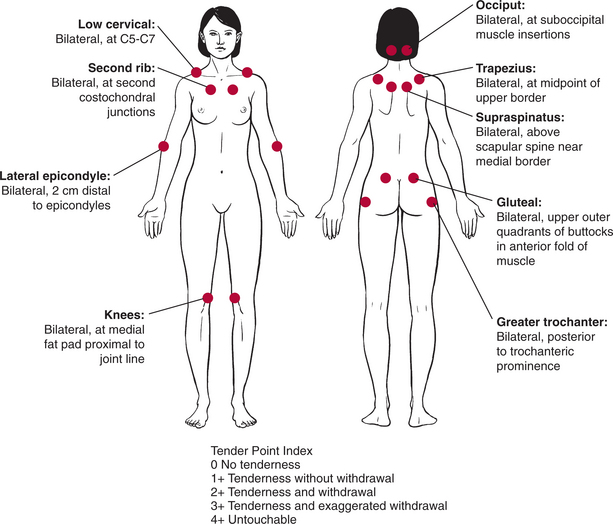
Figure 7-28 Anatomic locations of tender points associated with fibromyalgia. According to the literature, digital palpation should be performed with an approximate force of 4 kg (enough pressure to indent a tennis ball), but clinical practice suggests much less pressure is required to elicit a painful response. For a tender point to be considered positive, the subject must state that the palpation was “painful.” A reply of “tender” is not considered a positive response. Counting the number of points as part of the clinical diagnosis of FMS has been discounted266; however, the presence of multiple tender points is still a key feature of FMS. (Reprinted from Goodman CC, Snyder T: Differential diagnosis for physical therapists: screening for referral, ed 4, Philadelphia, 2007, Saunders.)
Sleep disturbances result in fatigue and exhaustion, even after a night’s sleep. Men with fibromyalgia typically have fewer symptoms and milder tender points (less “hurt all over” reports), less fatigue, and fewer incidences of irritable bowel syndrome compared with women who have FMS.272
Other symptoms or associated problems occur with a high frequency (Table 7-6), sometimes more incapacitating than the pain and tender points. Symptoms are often exacerbated by stress; overloading physical activity, including overstretching; damp or chilly weather; heat exposure or humidity; sudden change in barometric pressure; trauma; or another illness.
Table 7-6
Clinical Manifestations of Fibromyalgia
| Sign/Symptom | Incidence (%)* |
| Muscle pain (myalgia), tender points | 99† |
| Visual problems (e.g., blurring, double vision, bouncing images) | 95 |
| Mental and physical fatigue | 85 |
| Sleep disturbance/morning fatigue | 80 |
| Morning stiffness (persists >30 min) | 75 |
| Mitral valve prolapse | 75 |
| Global anxiety | 72 |
| Cognitive (memory) problems (e.g., decreased attention span, impaired short-term memory, decreased concentration, increased distractibility) | 71 |
| Irritable bowel syndrome | 70 |
| Inflammatory bowel disease (Crohn’s disease, ulcerative colitis) | 50-60 |
| Headaches | 70 |
| Hypersensitivity to noise, odors, heat, or cold (cold intolerance) | 50-60 |
| Paresthesias | 50 |
| Swollen feeling (joint or soft tissues) | 50 |
| Muscle spasms or nodules | 50 |
| Reactive hypoglycemia (e.g., weakness, irritability, disorientation) | 45-50 |
| Pelvic pain | 43 |
| Irritable bladder syndrome, female urethral syndrome | 40 |
| Hypotension (low blood pressure, elevated heart rate); NMH or vasopressor syncope | 40 |
| Raynaud’s phenomenon | 38 |
| Sicca syndrome (dry eyes/mouth) | 33 |
| Respiratory dysfunction (e.g., dyspnea, erratic breathing patterns during exertion) | 33 |
| Restless leg syndrome, nocturnal myoclonus, periodic leg movement disorder | 30-60 |
| Auditory problems | 31 |
| Temporomandibular dysfunction | 25 |
| Depression | 20 |
| Allergies | Unknown |
| Lack of libido | Unknown |
| Skin discoloration | Unknown |
| Sciatica | Unknown |
*These figures were compiled from a variety of sources but represent a fairly accurate clinical perspective.
†Although the American College of Rheumatology requires the identification of at least 11 out of 18 tender points to qualify for a diagnosis of FMS, some clinicians report isolated individuals without pain but characterized by the physiologic effects and manifestations of FMS or patients with fewer than 11 tender points.
Modified from Hulme J: Fibromyalgia: a handbook for self-care and treatment, ed 3, Missoula, MT, 2000, Phoenix.
Those people with fibromyalgia who are aerobically fit manifest fewer symptoms than those who remain physically deconditioned and aerobically unfit. Biofeedback specialists have shown that blood circulation to the affected areas is often significantly decreased while at rest, and a noticeable decrease in circulation occurs with changes in barometric pressure.
During exercise, when circulation should normally increase to muscles and the brain, in fibromyalgia just the opposite happens, and circulation is decreased significantly.112 Real-time ultrasonography has confirmed the lower magnitude of muscle vascularity following dynamic and during static exercise. The immediate flow response to muscular activity was lower in magnitude and of a shorter duration in people with fibromyalgia compared to healthy controls.71
The diaphragm is significantly affected in fibromyalgia to the point that it ceases to function as the major breathing muscle, and accessory muscles of the neck and upper chest take over. This overwork results in tender points or tightness of the neck and chest muscles.
In general, the level of muscular activity in fibromyalgia is high, even when the body is sitting or reclining. During daily activities such as cleaning, cooking, typing, and even socializing, the muscles used for these activities are at a higher level of activity than the muscles of a normal person doing the same tasks.
When the activity is over and the person with fibromyalgia is resting, those same muscles continue to repeat the activity over and over at a lower intensity so that no outward movement is apparent. This factor, combined with increased central pain processing, may lower tender point thresholds.17
MEDICAL MANAGEMENT
No definitive test is currently available to determine the presence of fibromyalgia, and usually the organs involved are not the cause but merely the messenger of a problem originating elsewhere in the body.
According to the American College of Rheumatology (ACR), two criteria must be met before a medical diagnosis of FMS can be made: (1) widespread (four-quadrant) pain both above and below the waist present for at least 3 months and (2) subjective report of pain when pressure is applied to 11 of the 18 common FMS tender points on the body (see Fig. 7-28).
Subjective assessment of tender points can be elicited by the use of an instrument called a dolorimeter, which distributes pressure equally over a discrete point. With a dolorimeter, the pressure required to produce pain in a given area can be recorded.
Controversy also exists regarding current use of the ACR’s criteria for tender point count in clinical diagnosis of FMS. In fact, the original author of the ACR criteria has suggested that counting the tender points was “perhaps a mistake” and has advised against using it in clinical practice as the only means of diagnosis.266 A proposed survey method that does not require physical examination may be more accurate.129
Often, the diagnosis is determined as a process of elimination by ruling out other conditions based on clinical presentation and past medical history (Box 7-8). In addition to the presence of tender points, skin fold tenderness, increased reactive skin hyperemia, and low tissue compliance (in the trapezius and paraspinal regions) provide further diagnostic information.
No special laboratory or radiologic testing is necessary for making a diagnosis of FMS; routine testing for rheumatoid factor or ANAs is not recommended. While routine inexpensive tests, including complete blood count, basic chemistry (blood urea nitrogen, creatinine, hepatic enzymes, serum calcium), and thyroid function, should be undertaken (if not done in the past year); any other test to rule out other conditions, unless clinically indicated (by both symptoms and physical examination), is a waste of time and resources.
A routine complete blood count test may demonstrate anemia caused by medications or another disease, which may contribute to fatigue, and cytopenia; a baseline chemistry test is useful in monitoring various medication side effects. Because spinal pain in FMS and pain caused by pathologic changes in the spine (e.g., osteoarthritis and osteopenia with vertebral compression fracture) cannot always be distinguished clinically, a spinal x-ray may be necessary for a middle-aged or older adult, especially considering other risk factors for these conditions. It is important not to miss these diagnoses, because the management approach is likely to be different from FMS alone. A sleep study should be considered only when history suggests a primary sleep disorder.273
TREATMENT.
Despite the chronicity and complexity of FMS, there are pharmacological and nonpharmacological interventions available that have clinical benefit. Helping clients with fibromyalgia must be holistic and multidisciplinary, including education and support, stress management, nutrition and lifestyle training (e.g., coping strategies, applying work simplification and ergonomic principles, and psychotherapy), medications (tricyclic antidepressants, selective serotonin reuptake inhibitors, serotonin-norepinephrine reuptake inhibitors, muscle relaxants, analgesics, and anticonvulsants), local modalities and techniques for muscle pain (e.g., relaxation techniques, biofeedback, Physiologic Quieting, or soft tissue techniques), and conditioning and aerobic exercise.
Cognitive behavioral therapy aimed at altering sensory, affective, cognitive, and behavioral aspects of chronic pain (e.g., pain severity, emotional distress, depression, anxiety, pain behavior) has been shown to be effective over a long period, even when the disease process cannot be controlled and symptoms worsen. Based on current evidence, a stepwise program emphasizing education, certain medications, exercise, cognitive therapy, or all four should be recommended.92
Alternative and complementary intervention (e.g., acupuncture, herbal or vitamin supplements, chiropractic, hypnotherapy, and others) is available and often provides palliative relief from symptoms for periods. Like most interventions (medical or unconventional), no single intervention is effective all the time, and the person with FMS may cycle through various modalities over time.
PROGNOSIS.
Many people with mild symptoms are managed without a specialist and have an expected good long-term outcome, but most people experience persistent symptoms of fibromyalgia for many years or a lifetime. Good therapy must be instituted early in the client’s course if there is to be any chance of achieving substantial improvement or a remission.
ISOIMMUNE DISEASE
Organ and Tissue Transplantation
With recent advances in technology and immunology, organ and tissue transplantation is becoming commonplace. In fact, transplantation of almost any tissue is feasible, but the clinical use of transplantation to remedy disease is still limited for many organ systems because of the rejection reaction. Transplant rejection, an isoimmune phenomenon, occurs in response to transplantation because the body usually recognizes the donor tissue as nonself and attempts to destroy the tissue shortly after transplantation.
In all cases of graft rejection, the cause is incompatibility of cell surface antigens. The rejection of foreign or transplanted tissue occurs because the recipient’s immune system recognizes that the surface HLA proteins of the donor’s tissue are different from the recipient’s.
For this reason, HLA matching of donor and recipient greatly enhances the probability of graft acceptance. Certain antigens are more important than others for a successful transplant, including ABO and Rh antigens present on RBCs and histocompatibility antigens, most importantly the HLA. As expected, a better chance of graft acceptance is evident with syngeneic or autologous transplants because the cell surface antigens are identical.
For a complete discussion of histocompatibility, graft rejection (acute versus chronic), graft-versus-host disease, and immunosuppression, see Chapter 21.
References
1. Aboulafia, DM, Pantanowitz, L, Dezube, BJ. AIDS-related non-Hodgkin lymphoma: still a problem in the era of HAART. AIDS Read. 2004;14(11):605–617.
2. Academy for Educational Development: Attitudes and strategies: a comprehensive approach to preventing blood-borne infections among IDUs. Available on-line at http://www.cdc.gov/idu/idu.htm Accessed April 26, 2008.
3. Aderem, A, Underhill, DM. Mechanisms of phagocytosis in macrophages. Ann Rev Immunol. 1999;17:593–623.
4. Albano, SA, Wallace, DJ. Managing fatigue in patients with SLE. J Musculoskelet Med. 2001;18(3):149–152.
5. Almeida, TF, Roizenblatt, S, Benedito-Silva, AA, et al. The effect of combined therapy (ultrasound and interferential current) on pain and sleep in fibromyalgia. Pain. 2003;104(3):665–672.
6. American Autoimmune Related Diseases Association. Available on-line at http://www.aarda.org
7. Arnold, DM, Julian, JA, Walker, IR. Mortality rates and causes of death among all HIV-positive individuals with hemophilia in Canada over twenty-one years of follow-up. Blood. 2006;108(2):160–164.
8. Assis, MR, Silva, LE, Alves, AM, et al. A randomized controlled trial of deep water running: clinical effectiveness of aquatic exercise to treat fibromyalgia. Arthritis Rheum. 2006;55:57–65.
9. Balbin, EG, Ironson, GH, Solomon, GF. Stress and coping: the psychoneuroimmunology of HIV/AIDS. Baillieres Best Pract Res Clin Endocrinol Metabol. 1999;13(4):615–633.
10. Barroso, PF, et al. Effect of antiretroviral therapy on HIV shedding in semen. Ann Intern Med. 2000;133(4):280–284.
11. Bauer, ME, et al. Chronic stress in caregivers of dementia patients is associated with reduced lymphocyte sensitivity to glucocorticoids. J Neuroimmunol. 2000;103(1):84–92.
12. Bedimo, R, Chen, RY, Westfall, AO, et al. Sustained HIV viral suppression following treatment interruption: an observational study. AIDS Res Hum Retroviruses. 2006;22(1):40–44.
13. Befus, AD, Mathison, R, Davison, J. Integration of neuro-endocrine immune responses in defense of mucosal surfaces. Am J Trop Med Hyg. 1999;60(4):26–34.
14. Benca, RM, Quintas, J. Sleep and host defenses: a review. Sleep. 1997;20(11):1027–1037.
15. Bennett, K. Exercise in fibromyalgia and chronic fatigue syndrome. In: Hall CM, Brody LT, eds. Therapeutic exercise. Philadelphia: Lippincott-Raven, 1998.
16. Bennett, R. A balanced view on exercise. Fibromyalgia Net. 1997:5.
17. Bennett, RM. Emerging concepts in the neurobiology of chronic pain: evidence of abnormal sensory processing in fibromyalgia. Mayo Clin Proc. 1999;74:385–398.
18. Berin, MC, McKay, DM, Perdue, MH. Immune-epithelial interactions in host defense. Am J Trop Med Hyg. 1999;60(4):16–25.
19. Bernatsky, S, Ramsey-Goldman, R, Clarke, A. Malignancy and autoimmunity. Curr Opin Rheumatol. 2006;18(2):129–134.
20. Borderi, M, et al. Metabolic complications of HIV-1 antiretroviral therapy: the lipodystrophy syndrome. New Microbiol. 2001;24(3):303–315.
21. Bower, M, Palmieri, C, Dhillon, T. AIDS-related malignancies: changing epidemiology and the impact of highly active antiretroviral therapy. Curr Opin Infect Dis. 2006;19(1):14–19.
22. Branson, BM, Handsfield, HH, Lampe, MA, et al. Revised recommendations for HIV testing of adults, adolescents, and pregnant women in health-care settings. MMWR. 2006;55(RR-14):1–24.
23. Brodsky, RA, Petri, M, Jones, RJ. Hematopoietic stem cell transplantation for systemic lupus erythematosus. Rheum Dis Clin North Am. 2000;26(2):377–387.
24. Brown, SL, Duggirala, HJ, Pennello, G. An association of silicone-gel breast implant rupture and fibromyalgia. Curr Rheumatol Rep. 2002;4(4):293–298.
25. Brown, SL, et al. Silicone gel breast implant rupture, extracapsular silicone, and health status in a population of women. J Rheumatol. 2001;28:996–1003.
26. Buckley, RH. Primary cellular immunodeficiencies. J Allergy Clin Immunol. 2002;109(5):747–757.
27. Cade, WT, Peralta, L, Keyser, RE. Aerobic exercise dysfunction in human immunodeficiency virus: a potential link to physical disability. Phys Ther. 2004;84(7):655–664.
28. Cairns, R, Hotopf, M. A systematic review describing the prognosis of chronic fatigue syndrome. Occup Med. 2005;55:20–31.
29. Calabrese, LH. Changing patterns of morbidity and mortality in HIV disease. Cleve Clin J Med. 2001;68(2):105. [109-110].
30. Calabrese, LH, Kirchner, E, Shrestha, R. Rheumatic complications of human immunodeficiency virus infection in the era of highly active antiretroviral therapy: emergence of a new syndrome of immune reconstitution and changing patterns of disease. Semin Arthritis Rheum. 2005;35(3):166–174.
31. Cambier, J. Immunosenescence: a problem of lymphopoiesis, homeostasis, microenvironment, and signaling. Immunol Rev. 2005;205:5–6.
32. Carette, S. What have clinical trials taught us about the treatment of fibromyalgia? J Musculoskelet Pain. 1995;3:133–140.
33. Celentano, DD, Sifakis, F, Hylton, J, et al. Race/ethnic differences in HIV prevalence and risks among adolescent and young adult men who have sex with men. J Urban Health. 2005;82(4):610–621.
34. Centers for Disease Control and Prevention: HIV/AIDS among women. Available on-line at: http://www.cdc.gov/hiv/topics/women/resources/factsheets/women.htm Accessed May 29, 2006.
35. Centers for Disease Control and Prevention: Cases of HIV and AIDS infection in the United States, 2004. Available at http://www.cdc.gov/hiv/topics/surveillance/resources/reports/2004report Accessed May 29, 2006.
36. Centers for Disease Control and Prevention: Trials of pre-exposure prophylaxis for HIV prevention. Available on-line at www.cdc.gov/hiv/PUBS/TenofovirFactSheet.htm Accessed on March 30, 2006.
37. Centers for Disease Control and Prevention: HIV/AIDS surveillance report, 16:1-40, 2004.
38. Centers for Disease Control and Prevention: Chronic fatigue syndrome: the revised case definition, 2006. Available on-line at http://www.cdc.gov/cfs/ Accessed April 26, 2008.
39. Centers for Disease Control and Prevention: Guidelines for the prevention of opportunistic infections (OIs) among HIV-infected persons-2002. Available at http://aidsinfo.nih.gov/contentfiles/OIpreventionGL.pdf
40. Centers for Disease Control and Prevention. Guidelines for prevention of HIV and hepatitis B virus transmission to health care and public safety workers. MMWR. 1989;38:S1–S6.
41. Centers for Disease Control and Prevention. Public Health Service guidelines for the management of health care worker exposures to HIV and recommendations for postexposure prophylaxis. MMWR. 1998;47(RR-7):1–33.
42. Chaitow, L. Fibromyalgia & muscle pain, ed 2. London: Thorson’s Health Series, 1999.
43. Chaitow, L. Fibromyalgia syndrome: a practitioner’s guide to treatment. Philadelphia: Churchill Livingstone, 2000.
44. Chang, TL, Chang, CH, Simpson, DA, et al. Inhibition of HIV infectivity by a natural human isolate of Lactobacillus jensenii engineered to express functional two-domain DC4. Proc Natl Acad Sci U S A. 2003;100(20):167–11677.
45. Chaudhuri, A, Behan, PO. Fatigue in neurological disorders. Lancet. 2004;363:978–988.
46. Chinniah, K, Mody, GM, Bhimma, R, et al. Arthritis in association with human immunodeficiency virus infection in Black African children: causal or coincidental? Rheumatology (Oxford). 2005;44(7):915–920.
47. Chinnock, L, et al. Is thumb nailbed blanching consistent with 4 kg of pressure when performing the Manual Tender Point Survey? Phys Ther. 2001;81(5):A25.
48. Citak-Karakaya, I, Akbayrak, T, Demirturk, F, et al. Short and long-term results of connective tissue manipulation and combined ultrasound therapy in patients with fibromyalgia. J Manipulative Physiol Ther. 2006;29(7):524–528.
49. Clapp, LL, et al. Acute effects of thirty minutes of light-intensity, intermittent exercise on patients with chronic fatigue syndrome. Phys Ther. 1999;79(8):749–756.
50. Clark S: Health care professionals teaming up. Available on-line at http://www.fmnetnews.com. Accessed March 3, 2007.
51. Clark, SR, et al. Exercise and patient outcome in fibromyalgia. Arthritis Rheum. 1991;34:S190.
52. Clarke, JN, James, S. The radicalized self: the impact on the self of the contested nature of the diagnosis of chronic fatigue syndrome. Soc Sci Med. 2003;57:1387–1395.
53. Cleare, AJ. The neuroendocrinology of chronic fatigue syndrome. , Endocr Rev. 2003;24:236–252.
54. Cleare, AJ. The HPA axis and the genesis of chronic fatigue syndrome. Trends Endocrinol Metab. 2004;15:55–59.
55. Currier, JS. How to manage metabolic complications of HIV therapy: what to do while we wait for answers. AIDS Read. 2000;10(3):162–169.
56. Davies, C. The trigger point therapy workbook: your self-treatment guide for pain relief. Oakland, CA: New Harbinger Publishers, 2001.
57. Dean, E. Oxygen transport deficits in systemic disease and implications for physical therapy. Phys Ther. 1997;77(2):187–202.
58. De Becker, P, et al. Exercise capacity in chronic fatigue syndrome. Arch Intern Med. 2000;160(21):3270–3277.
59. de Becker, P, McGregor, N, de Meirleir, K. Possible triggers and mode of onset of chronic fatigue syndrome. J Chronic Fatigue Syndr. 2002;10:3–18.
60. Delves, PJ, Roitt, IM. The immune system (I). N Engl J Med. 2000;343(1):37–49.
61. Delves, PJ, Roitt, IM. The immune system (II). N Engl J Med. 2000;343(2):108–117.
62. Denburg, SD, Carbotte, RM, Denburg, JA. Identifying cognitive deficits in systemic lupus erythematosus. J Musculoskelet Med. 1999;16(6):356–363.
63. Denburg, SD, Denburg, JA. Cognitive dysfunction and antiphospholipid antibodies in systemic lupus erythematosus. Lupus. 2003;12(12):883–890.
64. Des Jarlais, DC, Perlis, T, Arasteh, K, et al. HIV incidence among injection drug users in New York City, 1990 to 2002: use of serologic test algorithm to assess expansion of HIV prevention services. Am J Public Health. 2005;95(8):1439–1444.
65. Diamond, C, Taylor, TH, Aboumrad, T, et al. Increased incidence of squamous cell anal cancer among men with AIDS in the era of highly active antiretroviral therapy. Sex Transm Dis. 2005;32(5):314–320.
66. Dobkin, PL, et al. Psychosocial contributors to mental and physical health in patients with systemic lupus erythematosus. Arthritis Care Res. 1998;11(1):23–31.
67. Donenberg, GR, et al. Understanding AIDS-risk behavior among adolescents in psychiatric care: links to psychopathology and peer relationships. J Am Acad Child Adolesc Psychiatry. 2001;40(6):642–653.
68. Donoghoe, MC, Lazarus, JV, Matic, S. HIV/AIDS in the transitional countries of Eastern Europe and Central Asia. Clin Med. 2005;5(5):487–490.
69. Dudgeon, WD, Phillips, KD, Bopp, CM, et al. Physiological and psychological effects of exercise interventions in HIV disease. AIDS Patient Care STDS. 2004;18(2):81–98.
70. Eichner, ER. Infection, immunity, and exercise: what to tell patients? Physician Sportsmed. 1993;21(1):125–135.
71. Elvin, A, Siosteen, AK, Nilsson, A, et al. Decreased muscle blood flow in fibromyalgia patients during standardized muscle exercise. Eur J Pain. 2006;10(2):137–144.
72. Evans, WJ, Roubenoff, R, Shevitz, A. Exercise and the treatment of wasting: aging and human immunodeficiency virus infection. Semin Oncol. 1998;25(2 Suppl 6):112–122.
73. Finestone, HM, et al. Chronic pain and health care utilization in women with a history of childhood sexual abuse. Child Abuse Negl. 2000;24(4):547–556.
74. Fleshner, M. Exercise and neuroendocrine regulation of antibody production: protective effect of physical activity on stress-induced suppression of the specific antibody response. Int J Sports Med. 2000;21(Suppl 1):S14–S19.
75. Frankel, SS, et al. Replication of HIV-1 in dendritic cell-derived syncytia at the mucosal surface of the adenoid. Science. 1996;272:115–117.
76. Fransen, J, Russell, IJ. The fibromyalgia help book: practical guide to living better with fibromyalgia. St Paul, MN: Smith House Press, 1997.
77. Franzen, C, Salzberger, B, Fatkenheuer, G. Avascular necrosis of both femoral heads in an HIV-infected patient receiving protease inhibitors. Eur J Med Res. 2001;6(2):83–84.
78. Fulcher, KY, White, PD. Chronic fatigue syndrome: a description of graded exercise treatment. Physiotherapy. 1998;84:223–226.
79. Fulcher, KY, White, PD. Randomised control led trial of graded exercise in patients with the chronic fatigue syndrome. BMJ. 1997;314(7095):1647–1652.
80. Fulcher, KY, White, PD. Strength and physiological response to exercise in patients with chronic fatigue syndrome. J Neurol Neurosurg Psychiatry. 2000;69(3):302–307.
81. Gaab, J, Engert, V, Heitz, V, et al. Associations between neuroendocrine response to insulin tolerance test and patient characteristics in chronic fatigue syndrome. J Psychosom Res. 2004;56:419–424.
82. Galantino, ML. Human immunodeficiency virus (HIV) infection: living with a chronic illness. In Humphred D, ed.: Neurological rehabilitation, ed 4, St Louis: Mosby, 2001.
83. Galantino, ML, Eke-Okoro, ST, McGinnis, PQ, et al. Exploration of balance difficulties in AIDS neuropathy. Rehab Oncol. 2002;20(2):6–9. [17].
84. Galantino, ML, et al. Use of noninvasive electroacupuncture for the treatment of HIV-related peripheral neuropathy: a pilot study. J Alt Complement Med. 1999;5(2):135–142.
85. Galantino, ML, et al. Physical therapy management for the patient with HIV. Lower extremity challenges. Clin Podiatr Med Surg. 1998;15(2):329–346.
86. Galantino, ML, Shepard, K, Krafft, L, et al. The effect of group aerobic exercise and t’ai chi on functional outcomes and quality of life for persons living with acquired immunodeficiency syndrome. J Altern Complement Med. 2005;11(6):1085–1092.
87. Gao, X, et al. Effect of a single amino acid change in MHC class I molecules on the rate of progression to AIDS. N Engl J Med. 2001;344(22):1668–1675.
88. Garg, A. Lipodystrophies. Am J Med. 2000;108:143–152.
89. George, SL, Varmaz, D. What you need to know about GB virus C. Curr Gastroenterol Rep. 2005;7(1):54–62.
90. Gianotti, N, Lazzarin, A. Sequencing antiretroviral drugs for long-lasting suppression of HIV replication. New Microbiol. 2005;28(4):281–297.
91. Global HIV/AIDS pandemic., 2006. MMWR. 2006;55(31):841–844.
92. Goldenberg, DL, Burckhardt, C, Crofford, L. Management of fibromyalgia syndrome. JAMA. 2004;292(19):2388–2395.
93. Goldstein JA: What important brain neurotransmitters does TRH alter? Available on-line at http://www.fmnetnews.com. Accessed March 19, 2007.
94. Gomez, CR, Boehmer, ED, Kovacs, EJ. The aging innate immune system. Curr Opin Immunol. 2005;17(5):457–462.
95. Gowans, SE, deHueck, A. Effectiveness of exercise in management of fibromyalgia. Curr Opin Rheumatol. 2004;16(2):138–142.
96. Gowans, SE, deHueck, A. Exercise for fibromyalgia: benefits and practical advice. J Musculoskelet Med. 2006;23(9):614–622.
97. Grant, RM, Buchbinder, S, Cates, W, Jr., et al. AIDS. Promote HIV chemoprophylaxis, don’t prevent it. Science. 2005;309(5744):2170–2171.
98. Grant, RM, Buchbinder, S, Cates, W, Jr., et al. AIDS. Promote HIV chemoprophylaxis, don’t prevent it [letter]. Science. 2006;311(5758):175–176.
99. Gray, AB, et al. Anaerobic exercise causes transient changes in leukocyte subsets and IL-2R expression. Med Sci Sports Exerc. 1992;24:1332.
100. Green, EC, Halperin, DT, Nantulya, V, et al. Uganda’s HIV prevention success: the role of sexual behavior change and the national response. AIDS Behav. 2006;10(4):335–346.
101. Grulich, AE, Li, Y, McDonald, A, et al. Rates of non-AIDS-defining cancers in people with HIV infection before and after AIDS diagnosis. AIDS Read. 2002;16:1155–1216.
102. Gusi, N, Tomas-Carus, P, Hakkinen, A, et al. Exercise in waist-high warm water decreases pain and improves health-related quality of life and strength in the lower extremities in women with fibromyalgia. Arthritis Rheum. 2006;55:66–73.
103. Hanson, K, Hicks, C. New antiretroviral drugs. Curr HIV/AIDS Rep. 2006;3(2):93–101.
104. Hatcher, S, House, A. Life events, difficulties and dilemmas in the onset of chronic fatigue syndrome: a case-control study. Psychol Med. 2003;33:1185–1192.
105. Headley, B. When movement hurts: a self-help manual for treating trigger points. Longmont, CO: Innovative Systems, 1997.
106. Healthy People 2010: Objectives: HIV/AIDS. Available on-line at http://www.healthypeople.gov/document/html/volume1/13HIV.htm Accessed May 27, 2006.
107. Hirschhorn, L. Women, HIV, and HAART: closing the gender gap. Int Med. 1999;20(1):32–39.
108. Hobbs J: HIV/AIDS and the guide to physical therapist practice: the integumentary system. Presented at the American Physical Therapy Association Combined Sections, San Diego, CA, February 2006.
109. Honeyman-Lowe G: Ultrasound treatment of the fibromyalgia patient. Presented at the conference of the French Fibromyalgia Association, Grenoble, France, May 2000. Available by contacting the Fibromyalgia Research Foundation at P.O. Box 396, Tulsa, OK 74101 or www.drlowe.com.
110. Honeyman-Lowe, G, Ultrasound treatment of trigger points: differences in techniques for myofascial pain syndrome and fibromyalgia patients. Myalgia ′99 1999;2:12–15. Available by contacting the Fibromyalgia Research Foundation at P.O. Box 396, Tulsa, OK 74101 or www.drlowe.com.
111. Hooper, E, Hamilton, WD. The river: a journey to the source of HIV and AIDS. Boston: Back Bay Press, 2000.
112. Hulme, J. Fibromyalgia: a handbook for self-care and treatment, ed 3. Missoula, MT: Phoenix, 2000.
113. Iloeje, UH, Yuan, Y, L’italien, G, et al. Protease inhibitor exposure and increased risk of cardiovascular disease in HIV-infected patients. HIV Med. 2005;6(1):37–44.
114. Irwin, KL, et al. Potential for bias in studies of the influence of human immunodeficiency virus infection on the recognition, incidence, clinical course, and microbiology of pelvic inflammatory disease. Obstet Gynecol. 1994;84:463–469.
115. Irwin, K, Moorman, AC, O’Sullivan, MJ, et al. Influence of human immunodeficiency virus infection on pelvic inflammatory disease. Obstet Gynecol. 2000;95(4):525–534.
116. Jaffe, HW, Janssen, RS. Incorporating HIV prevention into the medical care of persons living with HIV. MMWR. 2003;52(RR 12):1–24.
117. James, JA, Harley, JB, Scofield, RH. Epstein-Barr virus and systemic lupus erythematosus. Curr Opin Rheumatol. 2006;18(5):462–467.
118. Janssen, RS, Onorato, IM, Valdiserri, RO, et al. Advancing HIV prevention: new strategies for a changing epidemic. MMWR. 2003;52(15):329–332.
119. Jason, LA, Richman, JA, Rademaker, AW, et al. A community-based study of chronic fatigue syndrome. Arch Intern Med. 1999;159:2129–2137.
120. Jones, SG. Taking HAART: how to support patients with HIV/AIDS. Nursing2004. 2004;34(6):6–12.
121. Joseph, RE, Radhakrishnan, J, Appel, GB. Antiphospholipid antibody syndrome and renal disease. Curr Opin Nephrol Hypertens. 2001;10(2):175–181.
122. Jubrias, SA, Bennett, RM, Klug, GA. Increased incidence of a resonance in the phosphodiester region of 31P nuclear magnetic resonance spectra in the skeletal muscle of fibromyalgia patients. Arthritis Rheum. 1994;37:801–807.
123. Kamradt, T, Mitchison, NA. Tolerance and autoimmunity. N Engl J Med. 2001;344(9):655–664.
124. Kapasi, ZF, et al. The role of endogenous opioids in exercise-induced enhancement of the secondary antibody response in mice. Phys Ther. 2001;81(11):1801–1809.
125. Kapasi, ZF, et al. The effects of intense physical exercise on secondary antibody response in young and old mice. Phys Ther. 2000;80(11):1076–1086.
126. Kapasi, ZF, Catlin, PA, Adams, MA, et al. Effect of duration of a moderate exercise program on primary and secondary immune responses in mice. Phys Ther. 2003;83(7):638–6347.
127. Kapasi, ZF, McRae, ML, Ahmed, R. Suppression of viral specific primary T-cell response following intense physical exercise in young but not old mice. J Appl Physiol. 2005;98(2):663–671.
128. Kapasi, ZF, Ouslander, JG, Schnelle, JF, et al. Effects of an exercise intervention on immunologic parameters in frail elderly nursing home residents. J Gerontol A Biol Sci Med Sci. 2003;58(7):636–643.
129. Katz, RS, Wolfe, F, Michaud, K. Fibromyalgia diagnosis: a comparison of clinical, survey, and American College of Rheumatology criteria. Arthritis Rheum. 2006;54(1):169–176.
130. Kay, AB. Allergy and allergic disease. N Engl J Med. 2001;344(1):30–37.
131. Keller, P. Physical therapy management of patients with HIV-positive/AIDS. Adv Phys Therapists. 1994;5:6–7. [17].
132. Kenny, PE. The changing face of AIDS. Nursing2004. 2004;34(8):54–63.
133. Kietrys, D, Gillardon, P, Galantino, ML. Contemporary issues in rehabilitation of patients with HIV disease. Part III: the effects of exercise on individuals with HIV disease. Rehab Oncol. 2002;20(2):10–14.
134. Kirou, KA, Salmon, JE, Crow, MK. Soluble mediators as therapeutic targets in systemic lupus erythematosus: cytokines, immunoglobulin receptors, and the complement system. Rheum Dis Clin North Am. 2006;32(1):103–119.
135. Kirsch, DL, Smith, RB. The use of cranial electrotherapy stimulation in management of chronic pain: a review. Neuro Rehabil. 2000;14(2):85–94.
136. Klein, J, Sato, A. The HLA system (I). N Engl J Med. 2000;343(10):702–709.
137. Korber, B, et al. Timing the ancestor of the HIV-1 pandemic strains. Science. 2000;288(5472):1789–1796.
138. Kordi, R, Wallace, WA. Blood borne infections in sport: risks of transmission, methods of prevention, and recommendations. Br J Sports Med. 2004;38(6):678–684.
139. Koulil, SV, Effting, M, Kraaimaat, FW, et al. A review of cognitive behaviour therapies and exercise programmes for fibromyalgia patients: state of the art and future directions. Ann Rheum Dis. 2006;66(5):171–181.
140. Krishnaswamy, G, et al. The cardiovascular and metabolic complications of HIV infection. Cardiol Rev. 2000;8(5):260–268.
141. Krsnich-Shriwise, S. Fibromyalgia syndrome: an overview. Phys Ther. 1997;77(1):68–75.
142. Kuhne L: Improving functional mobility for fibromyalgia patients with aquatic exercise. Presented at the Aquatic Therapy Rehabilitation Institute Conference, Las Vegas, August 1995.
143. Lahita RG: Laboratory tests used in the diagnosis of lupus. Available on-line at http://www.lupus.org/webmodules/webarticlesnet/templates/new_aboutdiagnosis.aspx?articleid=364&zoneid=15; http://www.lupus.org/webmodules/webarticlesnet/templates/new_newsroomnews.aspx?articleid=364&zoneid=59; http://www.lupus.org/webmodules/webarticlesnet/templates/new_aboutdiagnosis.aspx?a=364&z=15&page=3 Accessed April 26, 2008.
144. Lahita RG: Treatment of lupus. Available on-line at http://www.lupus.org/webmodules/webarticlesnet/templates/new_aboutdiagnosis.aspx?articleid=81&zoneid=15 April 26, 2008.
145. Landis, CA, et al. Decreased nocturnal levels of prolactin and growth hormone in women with fibromyalgia. J Clin Endocrinol Metab. 2001;86(4):1672–1678.
146. Lanier, LL. NK cell recognition. Ann Rev Immunol. 2005;23:225–274.
147. Lavreys, L. Effect of circumcision on incidence of human immunodeficiency virus type 1 and other sexually transmitted diseases. J Infect Dis. 1999;180(2):330–336.
148. Leonard, JT, Roy, K. The HIV entry inhibitors revisited. Curr Med Chem. 2006;13(8):911–934.
149. Leuthotlz, BC, Ripoll, I. Exercise and disease management. Boca Raton, FL: CRC Press, 1999.
150. Liang, TS, Erbelding, E, Jacob, CA, et al. Rapid HIV testing of clients of a mobile STD/HIV clinic. AIDS Patient Care STDS. 2005;19(4):253–257.
151. Lichtbroun, AS, Raicer, MMC, Smith, RB. The treatment of fibromyalgia with cranial electrotherapy stimulation. J Clin Rheum. 2001;7(2):72–78.
152. Lim, ST, Levine, AM. Recent advances in acquired immunodeficiency syndrome (AIDS)-related lymphoma. CA Cancer J Clin. 2005;55(4):229–241.
153. Lloyd, AR. Postinfective fatigue. In: Jason LA, Fennell PA, Taylor RR, eds. Handbook of chronic fatigue syndrome. Hoboken, NJ: John Wiley & Sons; 2003:108–123.
154. Lopes, RA, Benatti, MC, de Zollner, RL. A review of latex sensitivity related to the use of latex gloves in hospitals. AORN J. 2004;80(1):64–71.
155. Lopez, OL, Wess, J, Sanchez, J, et al. Neurobehavioral correlates of perceived mental and motor slowness in HIV infection and AIDS. J Neuropsych Clin Neurosci. 1998;10(3):343–350.
156. Lowder, T, Padgett, DA, Woods, JA. Moderate exercise protects mice from death due to influenza virus. Brain Behav Immunol. 2005;19(5):377–380.
157. Lowe, JC. Revised edition of the trigger point manual, vol 1: fibromyalgia. J Bodywork Move Ther. 2000;4(3):197–201.
158. Lowe, JC. The metabolic treatment of fibromyalgia. Boulder, CO: McDowell, 2000.
159. Lowe, JC, Honeyman-Lowe, G. Facilitating the decrease in fibromyalgic pain during metabolic rehabilitation: an essential role for soft tissue therapies. J Bodywork Move Ther. 1998;2(4):208–217.
160. Lowe, JC, Honeyman-Lowe, G. The metabolic rehabilitation of fibromyalgia patients. In: Chaitow L, ed. Fibromyalgia syndrome: a practitioner’s guide to treatment. Philadelphia: Churchill Livingstone, 2000.
161. Lynch, FA, Kirov, SM. Changes in blood lymphocyte populations following surgery. J Clin Lab Immunol. 1986;20:75.
162. Mandell, GL. Principles and practice of infectious diseases, ed 5. Philadelphia: Churchill Livingstone, 2000.
163. McBeth, J, et al. The association between tender points, psychologic distress, and adverse childhood experiences: a community-based study. Arthritis Rheum. 1999;42(7):1397–1404.
164. McCain, GA, et al. A controlled study of the effects of a supervised cardiovascular fitness training program on the manifestations of primary fibromyalgia. Arthritis Rheum. 1988;31:1135–1141.
165. McCarthy, DA, Dale, MM. The leukocytosis of exercise: a review and model. Sports Med. 1988;6:333–363.
166. McGowan I, Cheng A, Coleman S, et al: Assessment of bone mineral density (BMD) in HIV-infected antiretroviral-therapy-naive patients. Presented at the 8th Conference on Retroviruses and Opportunistic Infections, Chicago, February 2001.
167. McKinney, JD, Pedersen, LG. Do residue levels of polychlorinated biphenyls (PCBs) in human blood produce mild hypothyroidism? J Theoret Biol. 1987;129:231–241.
168. Medzhitov, R, Janeway, C. Innate immunity. N Engl J Med. 2000;343(5):338–344.
169. Mehta, N, Reilly, M. Atherosclerotic cardiovascular disease risk in the HAART-treated HIV-1 population. HIV Clin Trials. 2005;6(1):5–24.
170. Merchant, RC, Keshavarz, R. Human immunodeficiency virus postexposure prophylaxis for adolescents and children. Pediatrics. 2001;108(2):E38.
171. Mire-Sluis AR, Thorpe R, eds. Cytokines. San Diego: Academic Press, 1998.
172. Mok, CC, Wong, RW. Pregnancy in systemic lupus erythematosus. Postgrad Med J. 2001;77(905):157–165.
173. Moldofsky, H. Fibromyalgia, sleep disorder and chronic fatigue syndrome. Ciba Found Symp. 1993;173:262–271.
174. Moore, RD, Keruly, JC, Chaisson, RE. Differences in HIV disease progression by injecting drug use in HIV-infected persons in care. J Acquir Immune Defic Syndr. 2004;35(1):46–51.
175. MMWR. HIV and AIDS: United States, 1981-2000. MMWR. 2001;50(21):430–434.
176. Murasko, DM, Jiang, J. Response of aged mice to primary virus infections. Immunol Rev. 2005;205:285–296.
177. Nichols, DS, Glenn, TM. Effects of aerobic exercise on pain perception, affect, and level of disability in individuals with fibromyalgia. Phys Ther. 1994;74:327–332.
178. Nieman, DC, et al. Effects of brief, heavy exertion on circulating lymphocyte subpopulations and proliferative response. Med Sci Sports Exerc. 1992;24:1339.
179. Nieman, DC, et al. Infectious episodes in runners before and after the Los Angeles Marathon. J Sports Med Phys Fitness. 1990;30:316.
180. Nieman, DC, Pedersen, BK. Nutrition and exercise immunology. Boca Raton, FL: CRC Press, 2000.
181. Nixon, S, O’Brien, K, Glazier, RH, et al. Aerobic exercise interventions for people with HIV/AIDS. Cochrane Database Syst Rev. 2001;1:CD001796.
182. Pabst, R, Westerman, J, Binns, RM, Lymphocytic trafficking. Delves, PJ, Roitt, JM, eds. Encyclopedia of immunology, ed 2, 3. London: Academic Press, 1998.
183. Panlilio, AL, Cardo, DM, Grohskopf, LA, et al. Updated U.S. Public Health Service guidelines for management of occupational exposures to HIV and recommendations for postexposure prophylaxis. MMWR. 2005;54(RR09):1–17.
184. Paroli, M, Propato, A, Accapezzato, D. The immunology of HIV-infected long-term non-progressors-a current view. Immunol Lett. 2001;79(1-2):127–129.
185. Paton, NI, Macallan, DC, Griffin, GE, et al. Bone mineral density in patients with human immunodeficiency virus infection. Calcif Tissue Int. 1997;61:30–32.
186. Pedersen, BK, et al. Cytokines in aging and exercise. Int J Sports Med. 2000;21(Suppl 1):S4–S9.
187. Pedersen, BK, Hoffman-Goetz, L. Exercise and the immune system: regulation, integration, and adaptation. Physiol Rev. 2000;80(3):1055–1081.
188. Pedersen, BK, Saltin, B. Evidence for prescribing exercise as therapy in chronic disease. Scand J Med Sci Sports. 2006;16(Suppl 1):3–63.
189. Petersen, AM, Pedersen, BK. The anti-inflammatory effect of exercise. J Appl Physiol. 2005;98(4):1154–1162.
190. Petri, M. Is estrogen therapy safe in women with lupus? J Musculoskelet Med. 1994;11(12):11.
191. Phaneuf, S, Leeuwenburgh, C. Apoptosis and exercise. Med Sci Sports Exerc. 2001;33(3):393–396.
192. Phoenix, Inc: Thermister product available on-line at http://www.phoenixpub.com.
193. Plug, I, Van Der Bom, JG, Peters, M, et al. Mortality and cause of death in patients with hemophilia, 1992-2001: a prospective cohort study. J Thromb Haemost. 2006;4(3):510–516.
194. Porter, K, Babiker, A, Bhaskaran, K. Determinants of survival following HIV-1 seroconversion after the introduction of HAART. Lancet. 2003;362(9392):1267–1274.
195. Poulsen, HE, Weimann, A, Loft, S. Methods to detect DNA damage by free radicals: relation to exercise. Proc Nutr Soc. 1999;58(4):1007–1114.
196. Powell, P, et al. Randomised controlled trial of patient education to encourage graded exercise in chronic fatigue syndrome. BMJ. 2001;322(7283):387–390.
197. Pratley, R, et al. Strength training increases resting metabolic rate and norepinephrine levels in healthy 50 to 65 year old men. J Appl Physiol. 1994;76(1):133–137.
198. Prevention and control of tuberculosis in correctional and detention facilities. recommendations from CDC. MMWR. 2006;55(RR-09):1–44.
199. Price, JR, Couper, J. Cognitive behaviour therapy for adults with chronic fatigue syndrome. Cochrane Database Syst Rev. 2000;2:CD001027.
200. Prins, JB, van der Meer, JW, Bleijenberg, G. Chronic fatigue syndrome. Lancet. 2006;28:346–355. [367(9507)].
201. Pusch, O, Boden, D, Hannify, S, et al. Bioengineering lactic acid bacteria to secret the HIV-1 virucide cyanovirin. J Acquir Immune Defic Syndr. 2005;40(5):512–520.
202. Qaqish, RB, Sims, KA. Bone disorders associated with the human immunodeficiency virus: pathogenesis and management. Pharmacotherapy. 2004;24(10):1331–1346.
203. Quin, TC, et al. Viral load and heterosexual transmission of human immunodeficiency virus type 1. N Engl J Med. 2000;342(13):921–929.
204. Rachlin ES, ed. Diagnosis and comprehensive management of myofascial pain. St Louis: Mosby, 1994.
205. Ramsey-Goldman, R. Assessing disease activity in SLE patients during pregnancy. Lupus. 1999;8(8):677–684.
206. Ramsey-Goldman, R, Schilling, E. Immunosuppressive drug use during pregnancy. Rheum Dis Clin North Am. 1997;23(2):149–167.
207. Redwine, L, et al. Effects of sleep deprivation on interleukin-6, growth hormone, cortisol, and melatonin levels in humans. J Clin Endocrinol Metab. 2000;85(10):3597–3603.
208. Refetoff, S, Weiss, RE, Usala, SJ. The syndromes of resistance to thyroid hormone. Endocrine Rev. 1993;14:348–399.
209. Reid, S, Chalder, T, Cleare, A, et al. Extracts from “Clinical Evidence”: chronic fatigue syndrome. BMJ. 2000;320:292–296.
210. Reyes, M, Nisenbaum, R, Hoaglin, DC, et al. Prevalence and incidence of chronic fatigue syndrome in Wichita, Kansas. Arch Intern Med. 2003;163:1530–1536.
211. Rigsby, LW. Effects of exercise training on men seropositive for HIV-1. Med Sci Sports Exerc. 1992;24(1):6–12.
212. Rodriguez-Merchan, EC. HIV and HCV coinfected hemophilia patients: what are the best options of orthopaedic treatment? Haemophilia. 2006;12(Suppl 3):90–101.
213. Roe, DL, Lewis, RE, Cruse, JM. Association of HLA-DQ and-DR alleles with protection from or infection with HIV-1. Exp Mol Pathol. 2000;68(1):21–28.
214. Roubenoff, R. Acquired immunodeficiency syndrome wasting, functional performance, and quality of life. Am J Manage Care. 2000;6(9):1003–1016.
215. Roubenoff, R, Abad, LW, Lundgren, N. Effect of acquired immune deficiency syndrome wasting on the protein metabolic response to acute exercise. Metabolism. 2001;50(3):288–292.
216. Roubenoff, R, et al. A pilot study of exercise training to reduce trunk fat in adults with HIV-associated fat distribution. AIDS. 1999;12(11):1373–1375.
217. Russell, IJ. Neurohormonal aspects of fibromyalgia syndrome. Rheum Dis North Am. 1989;15:149–168.
218. Russell, IJ, et al. Platelet 3H-imipramine uptake receptor density and serum serotonin levels in patients with fibromyalgia/fibrositis syndrome. J Rheumatol. 1992;19:104–109.
219. Russell, IJ, Vaeroy, H. Cerebrospinal fluid (CSF) biogenic amines in fibromyalgia syndrome. ACR Sci Abstracts. 1990:S55.
220. Sacky, K, Shankle, D, Hobbs, J. Just sweat it out: physical therapy’s role in the HIV pandemic. Res Initiat Treat Action. 1998;4(4):8–10.
221. Salit, IE. Precipitating factors for the chronic fatigue syndrome. J Psychiatr Res. 1997;31:59–65.
222. Salvato PD: HIV update: treatment therapies for the millennium. Presented at the American Physical Therapy Association Combined Sections Meeting, New Orleans, February 2000.
223. Sanchez, T, Finlayson, T, Drake, A. Risk, prevention, and testing behaviors-United States, National HIV Behavioral Surveillance System: men who have sex with men. MMWR. 2006;55(SS-6):1–20.
224. Sande, MA, Volberding, PA. The medical management of AIDS, ed 6. Philadelphia: W.B. Saunders, 1999.
225. Scribner, AN, et al. Osteonecrosis in HIV: a case control study. J Acquir Immune Defic Syndr. 2000;25(1):19–25.
226. Sebasta D, Laplante MP: Disability Statistics Center. HIV/AIDS, disability, and employment. Available on-line at http://dsc.ucsf.edu/main.php Accessed May 17, 2006.
227. Senneff, JA. Numb toes and aching soles: coping with peripheral neuropathy. San Antonio, TX: Medpress, 1999.
228. Serhal, D, Calabrese, LH. Diagnosing and managing rheumatic disorders in HIV-infected persons. J Musculoskelet Med. 2000;17(10):606–620.
229. Shehan, DA, LaLota, M, Johnson, DF, et al. HIV/STD risks in young men who have sex with men who do not disclose their sexual orientation. MMWR. 2003;52(05):81–85.
230. Shephard, RJ. Chronic fatigue syndrome. A brief review of functional disturbances and potential therapy. J Sports Med Phys Fitness. 2005;45(3):381–392.
231. Shephard, RJ, Shek, PN. Effects of exercise and training on natural killer cell counts and cytolytic activity: a meta-analysis. Sports Med. 1999;28(3):177–195.
232. Sheriff, S, Antibody-antigen complexes, three-dimensional structures. Delves, PJ, Roitt, IM, eds. Encyclopedia of immunology, ed 2, 1. London: Academic Press, 1998.
233. Shevach, EM. CD4+ CD25+ suppressor T cells. more questions than answers. Nat Rev Immunol. 2002;2(6):389–400.
234. Shinkai, S, et al. Effects of 12 weeks of aerobic exercise plus dietary restriction on body composition, resting energy expenditure, and aerobic fitness in mildly obese middle-aged women. Eur J Appl Physiol Occup Physiol. 1994;68:258–265.
235. Simons, DG, Travell, JG, Simons, LS. Travell and Simons’ myofascial pain and dysfunction, the trigger point manual. Baltimore: Williams & Wilkins; 1999;1 and 2.
236. Smith, BA, Neidig, JL, Nickel, JT, et al. Aerobic exercise: effects on parameters related to fatigue, dyspnea, weight composition in HIV-infected adults. AIDS. 2001;15(6):693–701.
237. Smith, RJ. Adherence to antiretroviral HIV drugs: how many doses can you miss before resistance emerges? Proc Biol Sci. 2006;273(1586):617–624.
238. Smythe, H. Examination for tenderness: learning to use 4 kg force. J Rheumatol. 1998;25(1):149–151.
239. Sonnenberg, P, Murray, J, Glynn, JR, et al. HIV-1 and recurrence, relapse, and reinfection of tuberculosis after cure. Lancet. 2001;358(9294):1687–2163.
240. Spence, DW, et al. Progressive resistance exercise: effect on muscle function and anthropometry of a select AIDS population. Arch Phys Med Rehabil. 1990;71(9):644–648.
241. Stapleton, JT, Xiang, J, Williams, CF. HIV and GB virus C coinfection. Lancet Infect Dis. 2006;6(4):187–189.
242. Starlanyl, DJ, Copeland, M. Fibromyalgia and chronic pain syndrome: a survival manual, ed 2. Oakland, CA: New Harbinger, 2001.
243. Starz, TW, et al. Putting the finger on fibromyalgia: the manual tender point survey. J Musculoskelet Med. 1997;14(1):61–67.
244. Steinbrook, R, Drazen, JM. AIDS: will the next 20 years be different? N Engl J Med. 2001;344(23):1781–1782.
245. Straub, RH, et al. Neuropeptide Y cotransmission with norepinephrine in the sympathetic nerve-macrophage interplay. J Neurochem. 2000;75(6):2464–2471.
246. Stringer, WW. Mechanisms of exercise limitation in HIV+ individuals. Med Sci Sports Exerc. 2000;32:412–421. [(Suppl)].
247. Sutton, MY. Lower genital tract infection and HIV in women. Curr Infect Dis Rep. 2000;2(6):539–545.
248. Swica, Y, Breitbart, W. Treating pain in patients with AIDS and a history of substance use. West J Med. 2002;176(1):33–39.
249. Taussig, J, Shouse, RL, LaMarre, M, et al. HIV transmission among male inmates in a state prison system-Georgia, 1992-2005. MMWR. 2006;55(15):421–426.
250. Taylor-Robbins, C. Continuous ultrasound in the treatment of trigger points. J Myofascial Ther. 1994;1(4):12.
251. Terry, L, Sprinz, E, Stein, R, et al. Exercise training in HIV-1-infected individuals with dyslipidemia and lipodystrophy. Med Sci Sports Exerc. 2006;38(3):411–417.
252. Theorell, T, Blomkvist, V, Lindh, G, et al. Critical life events, infections, and symptoms during the year preceding chronic fatigue syndrome (CFS): an examination of CFS patients and subjects with a nonspecific life crisis. Psychosom Med. 1999;61:304–310.
253. Trends in HIV/AIDS diagnoses-33 states, 2001-2004. MMWR. 2005;54(45):1149–1153.
254. Twenty-five years of HIV/AIDS-United States, 1981-2006. MMWR. 2006;55(21):1–34.
255. Ungvarski, PJ, Flaskerud, JH. HIV/AIDS: a guide to primary care management, ed 4. Philadelphia: W.B. Saunders, 1999.
256. Vesosky, B, Turner, J. The influence of age on immunity to infection with Mycobacterium tuberculosis. Immunol Rev. 2005;205:229–243.
257. Viner, R, Hotopf, M. Childhood predictors of self reported chronic fatigue syndrome/myalgic encephalomyelitis in adults: national birth cohort study. BMJ. 2004;329:941.
258. von Andrian, UH, MacKay, CR. T-cell function and migration. N Engl J Med. 2000;343(14):1020–1034.
259. Walker BD: Immunopathogenesis of HIV infection. Available on-line at http://www.medscape.com/Medscape/HIV/AnnualUpdate/2001.
260. Wallace, DJ, Wallace, JB. All about fibromyalgia: a guide for the patients and their families. Oxford, UK: Oxford University Press, 2002.
261. Ward, MM, et al. Psychosocial correlates of morbidity in women with systemic lupus erythematosus. J Rheumatol. 1999;26(10):2153–2158.
262. Watkins, LR, Maier, SF. Immune regulation of central nervous system functions: from sickness responses to pathological pain. J Intern Med. 2005;257(2):139–155.
263. Wearden, AJ, et al. Randomised double-blind, placebo-controlled treatment trial of fluoxetine and graded exercise for chronic fatigue syndrome. Br J Psychiatry. 1998;172:485–490.
264. White, PD. What causes chronic fatigue syndrome? BMJ. 2004;329:928–929.
265. Whiting, P, Bagnall, A, Sowden, A, et al. Interventions for the treatment and management of chronic fatigue syndrome: a systematic review. JAMA. 2001;286:1360–1368.
266. Wolfe, F. Stop using the American College of Rheumatology criteria in the clinic. J Rheumatol. 2003;30(8):1671–1672.
267. Woods, JA. Exercise and neuroendocrine modulation of macrophage function. Int J Sports Med. 2000;21(Suppl 1):S24–S30.
268. Workowski, KA, Berman, SM. Sexually transmitted disease treatment guidelines 2006. MMWR. 2006;55(RR-11):1–100.
269. Yang, EV, Glaser, R. Stress-induced immunomodulation and the implications for health. Int Immunopharmacol. 2002;2(2-3):315–324.
270. Yim, YS, Wakeland, EK. The genetics of lupus. Curr Opin Nephrol Hypertens. 2001;10(3):437–443.
271. Yunus, MB. Towards a model of pathophysiology of fibromyalgia: aberrant central pain mechanisms with peripheral modulation. J Rheumatol. 1992;19:846–850.
272. Yunus, MB. Gender differences in fibromyalgia and other related syndromes. J Gend Specif Med. 2002;5(2):42–47.
273. Yunus, MB. A comprehensive medical evaluation of patients with fibromyalgia syndrome. Rheum Dis Clin North Am. 2002;28(2):201–217.