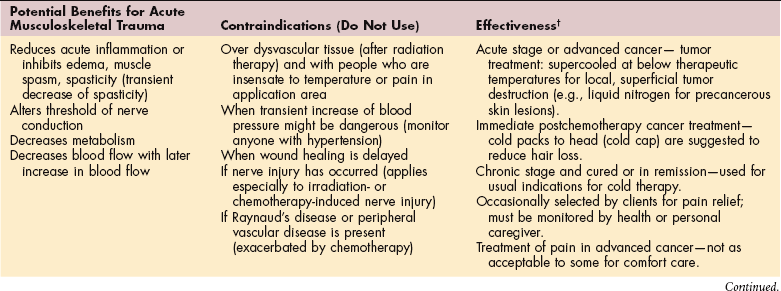
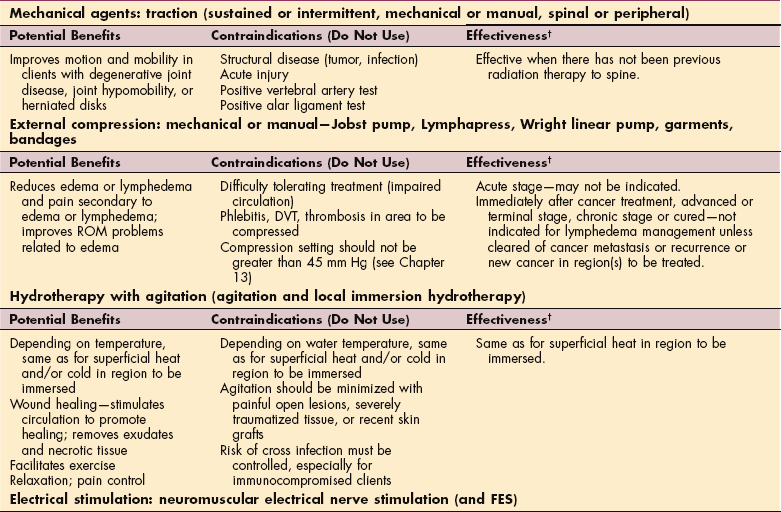
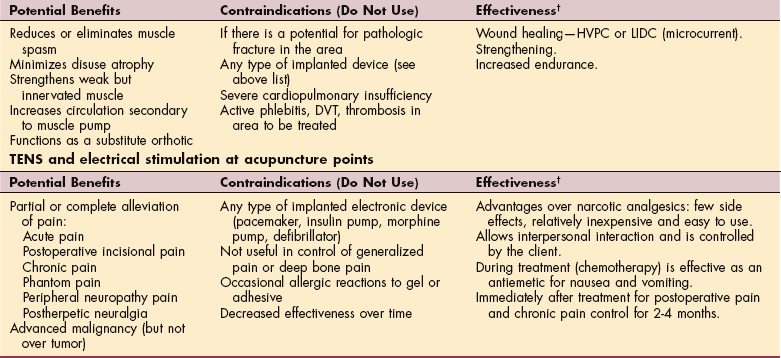
MEDICAL MANAGEMENT OF CANCER
The goal of Healthy People 2010 is to reduce the number of new cancer cases, as well as the illness, disability, and death caused by cancer. Evidence suggests that several types of cancer can be prevented and that the prospects for surviving cancer continue to improve. The ACS estimates that half of all cancer deaths in the United States could be prevented if Americans adopted a healthier lifestyle and made better use of available screening tests. The ability to reduce cancer death rates depends in part on the existence and application of various types of resources.
First, the means to provide culturally and linguistically appropriate information on prevention, early detection, and treatment to the public and to health care professionals are essential. Second, mechanisms or systems must exist for providing people with access to state-of-the-art preventive services and treatment. Third, a mechanism for maintaining continued research progress and for fostering new research is essential. Personalized prevention may become a tool in the future thanks to desktop oncology in the postgenome era of research. Desktop oncology refers to the genomics data produced by high technology. Desktop oncology provides knowledge on demand to anyone regarding cancer-related biomarkers. Combining genetic screening for cancer predisposition in the general population and selecting individualized targeted chemoprevention may dramatically improve cancer rates in the future.97
Studying older adults who do not develop cancer may help identify the genetic changes associated with age-resistant protective mechanisms. Genetic information that can be used to improve disease prevention strategies is emerging for many cancers and may provide the foundation for improved effectiveness in clinical and preventive medicine services.
Primary Prevention
Prevention is the first key to the management of cancer. Primary prevention may include screening to identify high-risk people and subsequent reduction or elimination of modifiable risk factors (e.g., tobacco use, diet high in unsaturated fats and low in fiber, sun or radiation exposure). Physical activity and weight control also can contribute to cancer prevention.
Chemoprevention, the use of agents to inhibit and reverse cancer, has focused on diet-derived agents. More than 40 promising agents and agent combinations (e.g., green and black tea phenols, lycopene, soy isoflavones, vitamins D and E, selenium, and calcium) are being evaluated clinically as chemopreventive agents for major cancer targets, including breast, prostate, colon, and lung cancer.98 In addition, low-dose aspirin intake and nonsteroidal antiinflammatory drug (NSAID) intake have shown promising results in the prevention of gastrointestinal cancers.
Research focusing on a cancer vaccine to wake up the immune system with a warning that cancer is present and stimulate an immune response against cancer cells is being investigated in clinical studies, although currently no known specific immunization prevents cancer in general. The most promising vaccines are for malignant melanoma and prostate cancer; vaccines for cancer viruses (human papillomavirus [HPV] associated with cervical cancer171 and hepatitis B virus [HBV] associated with liver cancer) are already in use.62
The person’s own tumor cells can be obtained during surgery, radiated to inactivate them, and then reinfused. This stimulates the immune system to react and make antibodies against these specific cells. The vaccine specifically evokes the activity of killer T cells to directly target and destroy tumors in all vaccine recipients. A vaccine given on an outpatient basis would be less dangerous than surgery and less toxic than other cancer treatments such as chemotherapy and radiation therapy.
Secondary Prevention
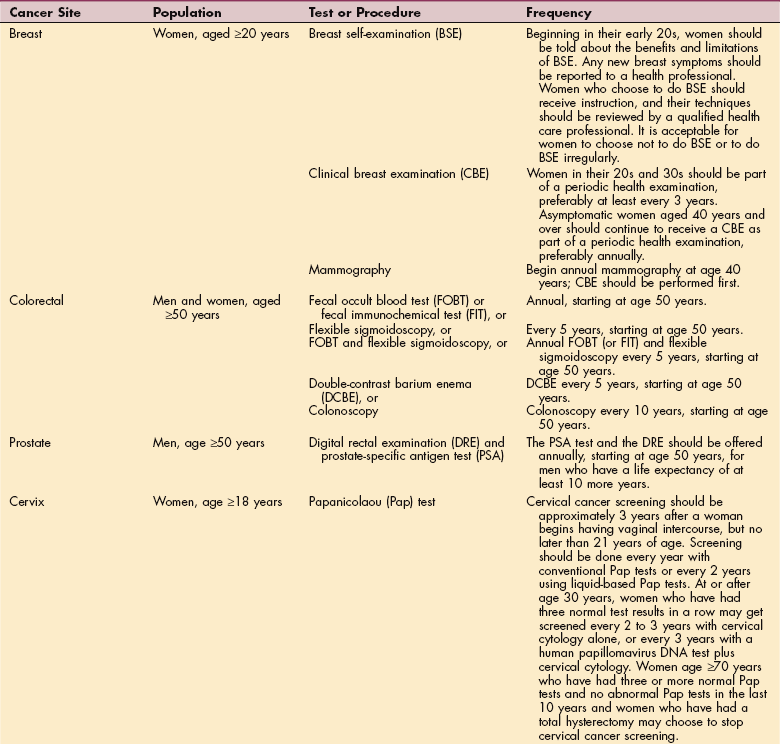
Secondary prevention aimed at preventing morbidity and mortality uses early detection85 and prompt treatment (Table 9-6). Some drugs, such as tamoxifen (Nolvadex), are used in both primary and secondary prevention of breast cancer. Tamoxifen has been approved by the Federal Drug Administration (FDA) as a preventive agent in women who have a high risk for possible development of breast cancer.211 The preliminary results of a randomized trial comparing tamoxifen to a placebo in women considered at high risk for breast cancer suggested that the risk of breast cancer in this group of high-risk women could be decreased by approximately 50% with the administration of tamoxifen.141
Multifactor risk reduction is an important part of secondary prevention for people diagnosed with cancer at risk for recurrence. This is especially true because the adverse effect of several risk factors is cumulative and many risk factors are interrelated.
Diagnosis
Medical history and physical examination are usually followed by more specific diagnostic procedures. Useful tests for the early detection and staging of tumors include laboratory values, radiography, endoscopy, isotope scan, CT scan, mammography, MRI, and biopsy. Advances in nuclear medicine have made it possible to examine images of organs, structures, and physiologic or pathologic processes and detect the distribution of radiopharmaceuticals according to their uptake and metabolism.
Tissue Biopsy
Biopsy of tissue samples is an important diagnostic tool in the study of tumors. Tissue for biopsy may be taken by curettage (Papanicolaou [Pap] smear), fluid aspiration (pleural effusion, lumbar puncture, or spinal tap), fine needle aspiration (breast or thyroid), dermal punch (skin or mouth), endoscopy (rectal polyps), or surgical excision (visceral tumors and nodes).
An open biopsy is performed in the operating room and consists of making an incision and removing a portion of the abnormal tissue. The amount removed depends on the abnormality, but it is usually a piece of tissue about one inch in diameter.
Needle biopsy uses a large diameter needle to take a core or plug of tissue. An incisional biopsy takes a slice or wedge of the lesion but does not attempt to remove the entire pathologic structure. Excisional biopsy (also referred to as a lumpectomy) removes the tumor and a perimeter of normal tissue or “margins.” The goal is to remove enough tissue to get negative margins when the tissue sample is examined under a microscope by a pathologist.
Stereotactic mammotome biopsy of the breast uses digital x-rays of the breast taken from two angles to locate the abnormality seen on the mammogram. A computer then calculates the proper angle and depth of insertion of a core biopsy needle. This needle is inserted into the breast, using local anesthesia, and multiple (a dozen or more) core specimens are removed. Each core is about 2 mm by 15 mm long. These cores are then sent to the pathologist for diagnosis. A second type of stereotactic procedure places a wire into the exact location of an abnormality within the breast. Ultrasound or mammography is used to find the lesion. The surgeon uses the wire to relocate the abnormality within the breast during an open biopsy. The procedure for placing the wire is the same as for taking a core biopsy, but a thin needle is used instead of a core biopsy needle. Once the needle is in place, a thin wire is inserted through the needle, and the needle is removed.
Sentinel lymph node (SLN) biopsy has become a standard diagnostic procedure to assess lymph node status of various tumors (e.g., breast, melanoma, endometrial, valvular, or head and neck) and to assess staging. A blue dye is injected around the cancerous tumor (or the biopsy site if the tumor has been removed). The dye flows through the ducts, and the first node or nodes it reaches is identified as the sentinel or sentinels. An incision is made over the nodes, and the blue-stained sentinel node or nodes (1 to 3) are removed and analyzed. The removal of more than three SLNs is classified as a dissection. Complications of SLN biopsy include allergic reaction to the blue dye (less than 1%), pneumothorax from unintended opening of the parietal pleura, sensory or motor nerve injury (small risk), lymphedema, surgical site infections (less than 1%), and seromas (10%).76
Information on the lymphatic drainage from the cancer can have a direct impact on surgery. SLN biopsy has reduced the number of unnecessary axillary dissections in breast cancer. The status of axillary nodes is the most important prognostic factor in breast cancer and in determining the medical management.
Tumor Markers
Tumor markers, substances produced and secreted by tumor cells, may be found in the blood serum. The level of tumor marker seems to correlate with the extent of disease. A tumor marker is not diagnostic itself but can signal malignancies. Carcinoembryonic antigen (CEA) is one tumor marker that may indicate malignancy of the large bowel, stomach, pancreas, lungs, and breasts. CEA and other serum titers, such as CA 125 (ovarian), CA 27-29 (breast), and prostate-specific antigen (PSA), may be valuable during chemotherapy to evaluate the extent of response and detect tumor recurrence.
Other tumor markers found in the blood (no more specific than CEA) include alpha fetoprotein (AFP), a fetal antigen uncommon in adults and suggestive of testicular cancer. The beta-2 (β2) microglobin is used in the monitoring of lymphomas, and lactic dehydrogenase (LDH) is particularly elevated in fast-growing malignancies. Human chorionic gonadotropin (β subunit) may indicate testicular cancer or choriocarcinoma. PSA helps evaluate prostatic cancer. Because of the lack of specificity of the markers individually (except PSA), test panels are used more frequently rather than just individual tumor marker evaluations.155
Several research institutes have developed a monoclonal antibody that identifies breast cancer and other cancer cells. The monoclonal antibody is used to devise a simple blood test for use in diagnosis and monitoring treatment of breast and ovarian cancers and will be used in the future to diagnose colon cancer. Combining the breast cancer antibody with nuclear medicine scanning techniques will provide a noninvasive means of determining lymphatic spread and guide surgeons in determining the extent of surgery required.10
Treatment
Changes in the health care system have shifted much of cancer care to the ambulatory and home settings. The medical management of cancer may be curative (i.e., with the intent to cure) or palliative (i.e., provides symptomatic relief but does not cure). Major therapies that are the focus of curative cancer treatment at this time include surgery, radiation, chemotherapy, biotherapy (also called immunotherapy or molecularly based therapy), angiogenesis therapy, and hormonal therapy.
New tests called gene profiling assays are now available that can predict fairly accurately what certain tumors will do and how best to treat them. Research has shown that tumors, like any other living tissue, contain genetic information that can be read with increasing accuracy. The goal is to analyze the genetic makeup of the tumor then choose the specific treatment most likely to be effective given that gene profile, while avoiding exposing the person to toxic therapies that might not be helpful or necessary. Two gene-profiling tests are already available for breast cancer; others are being evaluated for non-Hodgkin’s lymphoma, head and neck cancer, prostate cancer, kidney cancer, melanoma, and ovarian cancer.
Many cancers, such as myelodysplasia and hematologic malignancies (e.g., lymphoma, myeloma, leukemia), can be treated effectively in older adults, although advanced age does present many challenges. The future of oncologic care may rest on the model of individualized (tailored) therapy based on a pretreatment assessment of the each individual’s organ reserves, physical condition, and cognitive function. Identifying predictive factors of successful outcome will help assess who could benefit from more aggressive treatment and have the greatest chance for successful outcomes.13,52 When curative measures are no longer possible or available, palliative treatment may include radiation, chemotherapy, physical therapy (e.g., physical agents, exercise, positioning, relaxation techniques, biofeedback, or manual therapy), medications, acupuncture, chiropractic care, alternative medicine (e.g., homeopathic and naturopathic treatment), and hospice care.
Complementary and Alternative Medicine
Many people are seeking help in the cure and palliation of cancer through complementary and alternative medicine (CAM) therapies, such as acupuncture, hypnosis, mind-body techniques, massage, music, yoga, meditation, and other methods, to improve physical and mental well-being.5,41 Conventional treatments do not always relieve symptoms of pain, fatigue, anxiety, and mood disturbance. Some people cannot tolerate the side effects of conventional treatment. CAM has received consumer attention and concern on the part of those who provide conventional or standard medical therapy.
The ACS has published a guide to help consumers make these kinds of treatment decisions5 and provided some direction for health care professionals.203 Major research institutions and universities are beginning to investigate the effectiveness of these types of interventions for cancer. A new movement toward integrative medicine combining the best of complementary modalities with mainstream conventional therapies has been launched.
Major Treatment Modalities
Cancer treatment depends on an understanding of the biology of metastasis and how tumor cells interact with the microenvironment of different organs to design effective therapies.56 Each of the curative therapies described here may be used alone or in combination, depending on the type, stage, localization, and responsiveness of the tumor and on limitations imposed by the person’s clinical status.
Surgery, once a mainstay of cancer treatment, is now used most often in combination with other therapies. Surgery may be used curatively for tumor biopsy and tumor removal or palliatively to relieve pain, correct obstruction, or alleviate pressure. Surgery can be curative in persons with localized cancer, but 70% of clients have evidence of micrometastases at the time of diagnosis, requiring surgery in combination with other treatment modalities to achieve better response rates. Adjuvant therapy used after surgery eradicates any residual cells.
Radiation Therapy.: Radiation therapy (RT or XRT), also known as radiotherapy, plays a vital role in the multimodal treatment of cancer. It is used to destroy the dividing cancer cells by destroying hydrogen bonds between DNA strands within the cancer cells, while damaging resting normal cells as little as possible. Recent advances in RT have primarily involved improvements in dose delivery. The focus of future treatment is on combining RT with targeted therapies such as angiogenesis inhibitors.31
Radiation consists of two types: ionizing radiation and particle radiation. Both types have the cellular DNA as their target; however, particle radiation produces less skin damage. The goal is to ablate as many cancer cells as possible while simultaneously sparing surrounding normal tissues. Radiation is given over a period of weeks to capture cells at each stage of the cell cycle. Radiation is particularly effective at the end of the G2 phase (Fig. 9-3) when the cells are most susceptible to radiation.
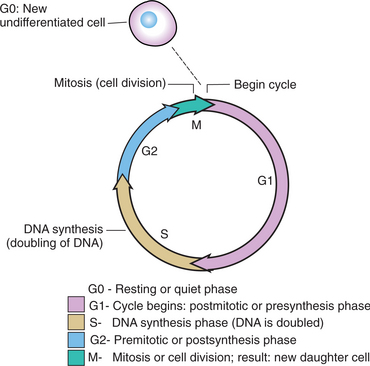
Figure 9-3 Cell cycle. One round of cell division requires duplication of DNA during the S phase and proper segregation of duplicated chromosomes during mitosis (M phase). G0 are the “resting cells” temporarily out of the proliferative cycle; when stimulated, these cells move into the G1 phase and begin to multiply. G1 and G2 are “gap” phases. G1 is the postmitotic period during which time RNA/protein synthesis occurs. G2 is the premitotic period and the last step in the mitotic cycle followed by M (mitosis) when cell division takes place. Most organ cells that are hormonally linked take approximately 19 to 33 days to complete one full cycle. Chemotherapy eliminates up to 95% of cancerous cells in the body; this is called the “kill rate.” Not all cancer cells will be eradicated; the immune system may be able to eliminate the remaining cells but not always. Adjuvant treatment, such as chemotherapy and radiation therapy, is administered in repeated doses over time in an attempt to kill cells in the most susceptible phases. For example, chemotherapy is most effective during DNA synthesis and mitosis. Cells are most sensitive to radiation therapy in the G2 phase. A certain percentage of cells will be unaffected because they are in the G0 or resting phase. Cells in the G0 phase are undifferentiated or stem (mesenchymal) cells waiting until called on by the body to serve a particular (differentiated) need. Stem cells in the G0 phase are resistant to chemotherapy and radiation therapy and the reason chemotherapy and radiation therapy are not 100% effective modalities in the ablation of microcirculation of tumor cells. The repeated or cyclical treatment is designed to catch G0 cells later in the growth cycle. (Modified from Abeloff MD, Armitage JO, Niedruhuber JE, et al: Clinical oncology, ed 3, 2004, London, Churchill Livingstone.)
Radiation treatment approaches include external beam radiation and intracavitary and interstitial implants. Radiation may be used preoperatively to shrink a tumor, making it operable while preventing further spread of the disease during surgery. After the surgical wound heals, postoperative doses prevent residual cancer cells from multiplying or metastasizing.
RT may be delivered externally or internally depending on the type and extent of the tumor by (1) external beam (teletherapy), (2) sealed source (brachytherapy), and (3) unsealed source (systemic therapy). When the distance between the radiation source and the target is short, the term brachytherapy is used. Brachytherapy allows for a rapid falloff in dose away from the target volume. When the radiation source is at a distance from the target, the term teletherapy is used. Teletherapy allows for a more uniform dose across the target volume.
Modern radiology has advanced to include site-specific techniques that take into account complex tissue contours and irregular shapes, visceral movement, digestion, and the effect of respiration on the lungs when the lungs are the target organ. Intensity modulated RT (IMRT) now allows for sculpting the radiation field and dose to match the area being irradiated. Computer optimization techniques help determine the distribution of beam intensities across a treatment volume.20
X-rays, radioactive elements, and radioactive isotopes are most often used in RT. Isotopes implanted in the tumor or a body cavity by external beam sources are delivered in the form of electromagnetic waves (e.g., x-rays or gamma rays) or as streams of particles (e.g., electrons). X-rays generated by linear accelerators and gamma rays generated by radioactive isotopes (e.g., cobalt-60, radium-226, or cesium-137) are referred to as sealed source radiation therapies or brachytherapy. This form of radiation is used for the treatment of visceral tumors because the rays penetrate to great depths before reaching full intensity and thereby spare the skin from toxic effects.
Strontium and yttrium aluminum garnet (YAG) lasers have been administered for the palliation of bone pain related to metastatic bone disease in both prostate and breast cancer.102,192 Electron beam irradiation is most useful in the treatment of superficial tumors, since energy is deposited at the skin and quickly dissipates, sparing the deeper tissues from toxic effects.
Normal and malignant cells respond to radiation differently, depending on blood supply, oxygen saturation, previous irradiation, and immune status. Cells most affected by chemotherapy and radiation have the greatest oxygenation and are the fast producing cells (e.g., hair, skin). Generally, normal cells recover from radiation faster than malignant cells; damaged cancer cells cannot self-repair. Success of the treatment and damage to normal tissue also vary with the intensity of the radiation.
Standard radiation fractionation is a course of 1.8 to 2.0 Gy per day in single daily doses. Accelerated or hypo- fractionation refers to delivering the same total dose over a shortened treatment time (one or just a few treatment sessions). Hyperfractionation refers to the same total delivered dose over the same treatment time but in an increased number of fractions; in other words, smaller fractions are delivered more often than once a day. Although a large single dose of radiation has greater cellular effects than fractions of the same amount delivered sequentially, a protracted schedule allows time for normal tissue to recover in the intervals between individual sublethal doses.20
Challenges with radiation treatment still remain because of the inability to identify microscopic disease with accuracy. Immobilizing patients and keeping them completely still for the duration of treatment is also difficult. Weight loss associated with treatment alters body geometry, requiring further corrections in dosimetry.
The next step in radiation oncology is to account for physiologic movements during irradiation. This may be accomplished with adaptive radiation with daily modulation of prescription and delivery using real-time imaging called four-dimensional (4-D) conformal RT (CRT).20 See Chapter 5 for a more complete discussion of the effects of RT.
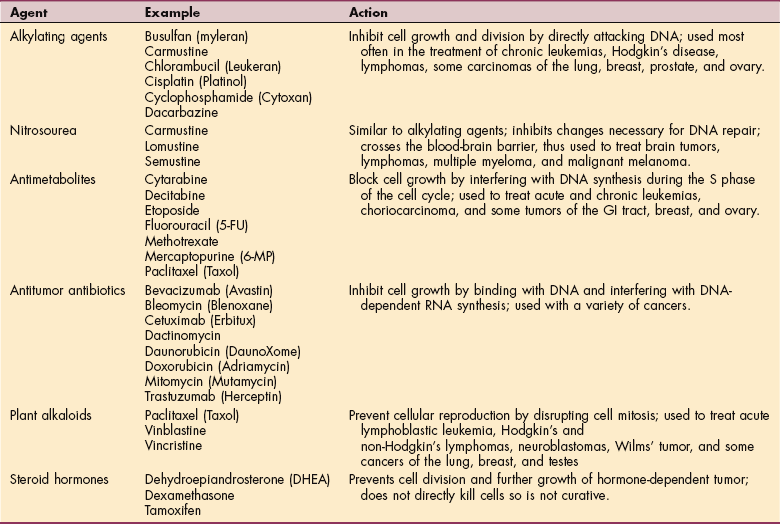
Chemotherapy.: Chemotherapy includes a wide array of chemical agents to destroy cancer cells. It is particularly useful in the treatment of widespread or metastatic disease, whereas radiation is more useful for treatment of localized lesions. Chemotherapy is used in eradicating residual disease, as well as inducing long remissions and cures, especially in children with childhood leukemia and adults with Hodgkin’s disease or testicular cancer. Several major chemotherapeutic agents are listed in Table 9-7. For a more complete discussion of chemotherapy, see Chapter 5.
Chemotherapy (and RT) kills most of the billion or more cells in each cubic centimeter of tumor tissue. However, cytotoxic therapies do not always eradicate every tumor cell for several reasons. Unlike normal cells, cancer cells are genetically unstable and replicate inaccurately. As the tumor grows, multiple subpopulations of cells with different biologic characteristics develop. Some of the cells will be resistant to treatment. After the treatment-sensitive cells are eliminated, the resistant cells may divide rapidly, recreating a tumor that is now resistant to the therapy.194
Almost all chemotherapy agents kill cancer cells by affecting DNA synthesis or function, a process that occurs through the cell cycle. Each drug varies in the way this occurs within the cell cycle. Chemotherapy interferes with the synthesis or function of nucleic acid targeting cells in the growth phase and therefore does not kill all cells (e.g., 5% are in the quiet or quiescent phase and are unaffected by chemotherapy) (see Fig. 9-3).
Combination therapies are often used because some drugs work better during different cell cycles. For example antimetabolites are most effective during the presynaptic (G1) phase, whereas alkylating agents target cells during the synthesis of DNA (S phase) and the postsynthesis (G2) phase. Treatment is designed to capture cell cycles at different phases for optimum cell death.119
Chemotherapeutic drugs can be given orally, subcutaneously, intramuscularly (IM), intravenously (IV), intracavitarily (into a body cavity such as the thoracic, abdominal, or pelvic cavity), intrathecally (through the sheath of a structure, such as through the sheath of the spinal cord into the subarachnoid space), and by arterial infusion, depending on the drug and its pharmacologic action and on tumor location. Administration in any form is usually intermittent to allow for bone marrow recovery between doses.
Although the effects of cancer treatment from chemotherapy, radiation therapy, hormonal therapy, and biotherapy are discussed in Chapter 5, new information about “chemobrain” has been published and updated here. Chemobrain, sometimes called “chemo fog” or “brain fog” refers to problems with memory, attention, and concentration reported by many people who have been treated with chemotherapy.
Not all chemotherapy recipients develop problems with cognitive or mental function but if it does happen, the effects can last several years. MRIs of brain structures have shown temporary shrinkage in the brain structures that are responsible for cognition and awareness. Shrinkage may be a possible physiologic explanation for chemotherapy-related cognitive difficulties.91
Three cancer drugs in particular (cisplatin, carmustine, cytosine arabinoside) have been implicated in laboratory cell cultures with killing brain cells. In fact 70% to 100% of brain cells compared to only 40% to 80% of cancer cells were destroyed by these drugs in animal studies. These cancer drugs may block new cell formation in the hippocampus, a brain structure essential to memory and learning.50
Mediating the Effects of Chemotherapy.: Colony-stimulating factors (CSFs) may be used to support the person with low blood counts related to chemotherapy. CSFs function primarily as hematopoietic growth factors, guiding the division and differentiation of bone marrow stem cells. They also influence the functioning of mature lymphocytes, monocytes, macrophages, and neutrophils.
Currently, erythropoietin (EPO), human granulocyte colony-stimulating factor (G-CSF), granulocyte-macrophage colony–stimulating factor (GM-CSF), and thrombopoietin (oprelvekin) and various interleukins are being used for chemotherapy-induced pancytopenia (deficiency of all cellular components of blood). EPO is used to treat anemia by stimulating bone marrow production of red blood cells. Interleukins are a large group of cytokines sometimes called lymphokines when produced by the T-lymphocytes or monokines when produced by mononuclear phagocytes. Interleukins have a variety of effects, but most interleukins direct other cells to divide and differentiate. Both G-CSF and GM-CSF are very useful in protecting individuals from prolonged neutrophil nadirs (lowest points after neutrophil count has been depressed by chemotherapy). Thrombopoietin (oprelvekin) has been recently identified and has shown promise in promoting elevation in platelet counts.169 In addition, GM-CSF has shown significant antitumor effects that prolong survival and disease-free survival in adults with stage III and IV melanoma who are at high risk for recurrence after surgical resection.190
Biotherapy.: Biotherapy, sometimes referred to as immunotherapy or immune-based therapy, relies on biologic response modifiers (BRMs) to change or modify the relationship between the tumor and host by strengthening the host’s biologic response to tumor cells. Much of the work related to BRMs is still experimental, so the availability of this type of treatment varies regionally within the United States.
The most widely used agents include interferons, which have a direct antitumor effect, and IL-2, one type of cytokine, a protein released by macrophages to trigger the immune response.81 In addition to their desired immune effects, interferons cause a number of significant toxicities, including constitutional, hematologic, hepatic, and prominent effects on the nervous system, especially depression.
Other forms of biotherapy include bone marrow or stem cell transplantation, monoclonal antibodies, CSFs, and hormonal therapy. Bone marrow transplantation (BMT) or peripheral stem cell transplantation (PSCT) is used for cancers that are responsive to high doses of chemotherapy or radiation. These high doses kill cancer cells but are also toxic to bone marrow; BMT provides a method for rescuing people from bone marrow destruction while allowing higher doses of chemotherapy for a better antitumor result.
BMT was a technique developed to restore the marrow to people who had lethal injury to that site because of bone marrow failure, destruction of bone marrow by disease, or intensive chemical or radiation exposure. At first, the source of the transplant was the marrow cells of a healthy donor who had the same tissue type (human leukocyte antigen [HLA]; markers on the white blood cells) as the recipient (usually a sibling or close family relative). Donor programs have been established to identify unrelated donors who have a matching HLA.
The transplant product is a very small fraction of the marrow cells called stem cells. These cells occur in the bone marrow and also circulate in the blood and can be harvested from the blood of a donor by treating the donor with an agent or agents (e.g., G-CSF) that cause a release of larger numbers of stem cells into the blood and collect them by hemapheresis. Since blood (peripheral site), as well as marrow, is a good source of cells for transplantation, the term stem cell transplantation has replaced the general term for these procedures (see the section on Bone Marrow Transplantation in Chapter 21).
Other immune-based strategies currently being evaluated or cancer therapy in preclinical models and clinical trials include vaccines combined with local therapies, the use of antioxidants during radiation therapy, and monoclonal antibodies (mAbs). mAbs are laboratory-engineered copies of proteins produced from a single clone (monoclonal) of B-lymphocytes that can stimulate the immune system to attack cancer cells. mAbs are antibodies that are identical because they were produced by one type of immune cell and are all clones of a single parent cell.
These antibodies are biologic therapies that act specifically against a particular antigen. They can also be bound with radioisotopes and injected into the body to detect cancer by attaching to tumor cells. The antibody may not actually kill target cells, but rather it marks the cells so that other components in the immune system attack it or initiate a signaling mechanism that leads to the target cell’s self-destruction.181 Monoclonal antibodies have been developed to help combat specific cancers, including colorectal cancer and some forms of non-Hodgkin’s lymphoma.
Research is under way to find a way to use these antibodies as a means of destroying specific cancer cells without disturbing healthy cells. Rituximab (Rituxan), trastuzumab (Herceptin), Alemtuzumab (Campath-1H), and Cetuximab (Erbitux) are a few of the monoclonal antibodies currently in use for cancer treatment (e.g., lymphoma, breast, colorectal, chronic leukemia, and head and neck cancer).
Avastin (bevacizumab, formerly known as anti-VEGF) is an antibody used in combination with chemotherapy in the treatment of colon, lung, and breast cancer. This antibody binds to vascular endothelial growth factor (VEGF) made by the tumor cells and prevents it from forming new blood vessels to supply the tumor cells.
Rituximab is used primarily in the treatment of non-Hodgkin’s lymphoma. Rituximab binds to lymphoid cells in the thymus, spleen, lymph nodes, and peripheral blood in order to lyse and destroy specific immune target cells.
Trastuzumab is used in the treatment of metastatic breast cancer in women who have overexpression of the human epidermal growth factor receptor 2 (HER2) protein. It binds with this protein and inhibits proliferation of cells with this protein and also mediates an antibody-mediated destruction of the cancer cells that have the HER2 receptor overexpression.64 Both agents are usually used in combination with or in addition to other chemotherapeutic agents for treatment.
Not all people respond to mAbs, presumably because of differences in the receptors being targeted. Molecular testing will have to become part of designer biologic therapy in which drugs are chosen on an individual basis after genetic profiling and immunoassay. Antibodies also function as carriers of cytotoxic substances, such as radioisotopes, drugs, and toxins, making them a key focus area of cancer research right now.181
Antiangiogenic Therapy.: Antiangiogenic therapy shows promise as a strategy for cancer treatment. Research has shown that the one common area of vulnerability of all cells in any phase of growth is the nonnegotiable need for oxygen. Tumor cells cannot survive without oxygen and other nutrients transported by the blood. In fact, tumor cells cannot survive at distances greater than 150 μm from a blood vessel.194
Antiangiogenic therapy may be able to put a stop to pathologic angiogenesis, the process by which a malignant tumor develops new vessels and is the primary means by which cancer cells spread. Antiangiogenesis factors, their receptors, and the signaling pathways that govern angiogenesis in solid tumors have been discovered. Treatment with antiangiogenesis factors (e.g., endostatin, angiostatin, or calpastatin) approved for use in the United States focuses on blocking the general process of tumor growth by cutting off their blood supply rather than on the destruction of an already formed cancerous mass.59 Scientists expect that combinations of angiogenesis inhibitors or broad-spectrum angiogenesis inhibitors will be needed for long-term use in cancer if tumor cells have or develop multiple molecular signaling pathways, a characteristic called redundancy.60
In the future, antiangiogenic agents may be used as maintenance therapy to control cancer much the same way that medications are used to control hypertension or hyperlipidemia. It is expected that different mutations in cancer will require individualized therapy based on current knowledge of specific tumors, their patterns of resistance, and response to angiogenesis inhibitors.82
Hormonal Therapy.: Hormonal therapy is used for certain types of cancer shown to be affected by specific hormones. For example, tamoxifen, an antiestrogen hormonal agent, is used in breast cancer to block estrogen receptors in breast tumor cells that require estrogen to thrive.
The luteinizing-releasing hormone leuprolide is now used to treat prostate cancer. With long-term use, this hormone inhibits testosterone release and tumor growth. Goserelin acetate (Zoladex) is a newer hormone used in prostate cancer that is a synthetic form of luteinizing hormone–releasing hormone (LH-RH). Goserelin acetate inhibits pituitary gonadotropic secretion, thus decreasing serum testosterone levels.211
Effects of Cancer Treatment
Although it may make more sense to include a discussion of the side effects of cancer treatment in this chapter, we have opted to place that topic in Chapter 5 to help emphasize the point that the long-term effects of cancer treatment are problems that affect multiple systems. The therapist must take this approach when planning intervention and offering patient/client education.
With improved survival rates, we expect to see more delayed reactions and long-term sequelae to today’s cancer treatment modalities. With improved survival and longevity, we may see an increased prevalence of cancer recurrence in the future, too. This may mean worsening of symptoms such as peripheral neuropathy or lymphedema from second and third rounds of treatment. In time, with the identification of genetic traits of cancer, treatment may become more specific to the cancer cells and less toxic to healthy cells and tissue, eventually reducing and maybe even eliminating side effects experienced by many of today’s cancer survivors.
Prognosis
Thirty years ago a cancer diagnosis was often a death sentence; survivors referred to themselves as “victims.” Cancer is no longer considered a death sentence, and many survivors return to the mainstream of family life, community activities, and work. Medical treatment is often provided in outpatient settings, making it possible to work during treatment.86
Today, there are 10 million cancer survivors in the United States; 65% of all people diagnosed with cancer have a 5-year survival rate. In general, this means that the chance of a person recently diagnosed with cancer being alive in 5 years is 65% of the chance of someone not diagnosed with cancer. Such statistics adjust for normal life expectancy (accounting for factors such as diabetes, heart disease, injuries, or dying of old age).3
In general, increased survival rates occur with screening and early detection, especially for cancers that do not have a highly effective treatment such as melanoma. Prognosis is influenced by the type of cancer, the stage and grade of disease at diagnosis, the availability of effective treatment, the response to treatment, and other factors related to lifestyle such as smoking, alcohol consumption, diet, and nutrition. Despite advances in early diagnosis, surgical techniques, systemic therapies, and patient care, the major cause of death from cancer is due to metastases that are resistant to therapy.56
The prognosis is poor for anyone with advanced, disseminated cancer. Researchers continue to search for the mechanisms responsible for cancer metastases and chemotherapeutic failure and develop new strategies to circumvent drug resistance. Generally, the earlier cancers are found, the simpler treatment may be and the greater likelihood of a cure.
The term no evidence of disease (NED) may be used when all signs of the disease have disappeared after treatment but before the end of 5 years occurs. There are no signs of the disease using current tests. If the response is maintained for a long period, the term durable remission may be used (Box 9-4). The person who is alive and without evidence of disease for at least 5 years after diagnosis is considered cured. The terms survival and cure do not always portray the functional status of a cancer survivor. Many people considered cured are left with physical limitations and movement dysfunctions that interfere with their daily lives.
Even without complete remission, cancer can be controlled to provide longer survival time and improved QOL, but these factors are not reflected in survival rates. Cancer statistics reported usually include a lag time so that rates may not reflect the most recent treatment advances. Survival rates for many cancers have increased from 1960 to the present, but not all cancers have been characterized by this increase. For example, while survival rates for Hodgkin’s disease and prostate, testicular, and bladder cancers have increased by at least 25%, the survival rates for cancers of the oral cavity and pharynx, liver, pancreas, esophagus, and colon have decreased or increased less than 5% during the same period.
A significantly lower survival rate in black American men for most cancer classifications has been noted. This difference may be due to a variety of factors, including limited access to health care, little or no insurance, lack of a primary health care provider, limited knowledge of the benefits of early diagnosis and treatment, and greater exposure to carcinogens. Central to these social forces is access to health care, including prevention, information, early detection, and quality treatment.212
In terminally ill individuals, rates of change are more important indicators of survival than absolute measures. Using a modified Barthel Index comprised of 10 activities of daily living (ADLs), each with five levels of dependency (maximum score more than 100 points), can provide important predictions about length of time until death. Half of those individuals with advanced cancer who lose 10 or more points per week die within 2 weeks, and three-fourths are dead at 3 weeks. In contrast, 50% of all cases without declines in score survive for 2 months or more. This may be a useful tool for planning and end-of-life issues in a hospice setting.15
Selected older adults with cancer can benefit from intensive care. Age is associated with higher mortality, especially for adults over 60 years old and when combined with multiple comorbidities.188 A comprehensive geriatric assessment can be helpful in identifying individuals likely to benefit from cytotoxic treatment. Therapies may be adjusted based on renal and cardiac function. Cardiac toxicity and neurotoxicity are common in persons aged 65 years and older.13
CANCER, PHYSICAL ACTIVITY, AND EXERCISE TRAINING
Investigators have begun extensive research in the area of exercise and cancer. As with the prevention and management of heart disease, obesity, osteoporosis, and diabetes, exercise has an important role in relation to cancer. More and more studies of cancer as a prevention strategy, as a means to ameliorate side effects of cancer treatment, and to promote improved health among cancer survivors are being published. The results of studies are varied and wide-ranging and complicated by the fact that exercise can be aerobic, strength training, flexibility, balance training, and conditioning or any combination of these forms. Each type of exercise has its own physiologic and psychologic benefits in the normal, healthy adult population.
The effects of each type of exercise on individuals with cancer are being investigated in many studies. Additionally, not all cancers are alike or affect the body in the same way; cancer exercise benefits may vary based on cancer type, stage, type of treatment, changes made by treatment, and so on. Exercise appears to be safe, but long-term outcomes have not been reported. Some types of exercise have been shown detrimental to the immune system and this must be considered (see discussion in Chapter 7).
Only a composite summary is provided here. Therapists working with the oncology population are encouraged to study each individual cancer encountered to find the best choice of prescriptive exercise published in the literature.
Exercise as a Cancer Prevention Strategy
Physical activity is defined as body movement caused by skeletal muscle contraction that results in quantifiable energy expenditure. Both epidemiologic and laboratory data indicate that the level of physical activity in which an individual engages may affect cancer risk. Exercise is distinguished from other types of physical activity by the fact that the intensity, duration, and frequency of the activity are specifically designed to improve physical fitness.
Based on available data, a role for exercise in specifically reducing cancer risk has been shown for breast and colorectal cancer, with more equivocal evidence for others such as melanoma, lung, and prostate cancers.182 The exact amount of exercise needed to prevent cancer is debatable. It is currently not known what would be most beneficial for which cancers, at which stage of disease, or treatment.89 The ACS advises moderate habitual physical activity as a potentially protective measure against certain types of neoplasms, particularly tumors of the colon and the female reproductive tract. The activity should cause a slight increase in heart rate and breathing lasting 30 minutes, at least 5 days a week.
Exercise-induced changes in the activity of macrophages, natural killer cells, lymphokine-activated killer cells, neutrophils, and regulating cytokines suggests that immunomodulation may contribute to the protective value of exercise (see also the section on Exercise, Physical Activity, and the Immune System in Chapter 7).122,216
Exercise for the Person with Cancer
Exercise programs also appear to have a beneficial influence on the clinical course of cancer, at least in the early stages of the disease. At the present time, cytokine modulation with exercise is receiving considerable research attention. Researchers theorize that exercise can regulate production of certain hormones, which when unregulated, may spur tumor growth.
With 10 million Americans alive today who have been through the cancer experience, it is important to develop interventions to enhance immune function, prevent or minimize muscle wasting thus counteracting the detrimental physiologic effects of cancer and chemotherapy, and maintain QOL after cancer diagnosis. Physical activity and exercise training are interventions that address a broad range of QOL issues, including physical (e.g., muscular strength, body composition, nausea, and fatigue), functional (e.g., functional capacity), psychologic (e.g., coping and mood changes), spiritual, emotional, and social well-being.37
Studies examining the therapeutic value of exercise for people with various cancers during primary cancer treatment suggest that exercise is safe and feasible, improving physical functioning and some aspects of QOL.36,101,175
Screening and Assessment
Medical screening should be conducted with all clients before their participation in an exercise program.1 This type of screening is especially important for people with cancer who receive various levels of treatment that can affect the physiologic response to exercise. For example, fatigue is a common symptom of nearly every form of cancer treatment.
The therapist will need to take a detailed history of treatment administered to date, examine laboratory results, and distinguish between fatigue from deconditioning and fatigue from medical interventions to determine the most effective and efficient approach to rehabilitation. The medical history should also look for conditions not related to cancer, such as hypertension, diabetes, coronary artery disease, and preexisting orthopedic conditions. The person’s current physical condition, condition before disease onset, and age are also important variables.214
A self-reporting survey instrument called the Cancer Rehabilitation Evaluation System (CARES; formerly called the Cancer Inventory of Problem Situations [CIPS]) is a useful tool for evaluating rehabilitation needs and interventions.66,173 The therapist must understand the stages of the disease and know the type and timing of the medical intervention, especially for radiation and chemotherapy. The body’s physiologic response to these agents (e.g., fatigue, neuropathy, or chemo brain) may alter the normal training response and affect tolerance for exercise and compliance with exercise programs. Cognitive rehabilitation techniques may be needed to improve patient/client compliance, function, and QOL.63
Cardiac dysfunction months to years after chemotherapy can result in left ventricular failure, cardiomyopathy, and/or congestive heart failure. These conditions may impact the client’s ability to exercise. Signs and symptoms of subclinical cardiac conditions may develop with the initiation of an exercise program. Careful history taking and clinical assessment may result in early detection and intervention, potentially reducing morbidity.
Auscultation to screen for abnormal lung or heart sounds is important to identify any precautions or contraindications to exercise. The individual is not likely to be able to sustain exercise levels if there are any physiologic abnormalities present. Medical consult may be required before initiating a training program.
Monitoring Vital Signs
Monitoring physiologic responses to exercise is important in the immunosuppressed population. Exercise intensity determined by training heart rate may be difficult to use since some people have inappropriate heart responses to exercise and large physiologic changes on a day-to-day basis from disease and treatment (e.g., changes in medications).
Baseline testing is important to determine safe guidelines and to provide a starting place against which to measure improvement and to identify the individual’s functional exercise level. A hypertensive response to exercise is common among individuals with cancer and undergoing cancer treatment. Starting an aerobic training program is not advised if such a response is observed during testing.163
Exercise intensity can be guided by heart rate ranges based on oxygen consumption or metabolic equivalent (MET) levels. The therapist can use test results to prescribe a program starting at approximately 60% of the individual’s maximum level. The therapist uses prior exercise levels, prior exercise capabilities, baseline function, and individual abilities even when using the predictive formula because each client may respond differently (unpredictably).163
The therapist (or client) should always monitor oxygen saturation with pulse oximetry and monitor heart rate (for arrhythmias), pulse rate, breathing frequency, and blood pressure before, during, and after the treatment session. Borg Ratings of Perceived Exertion (RPE) scale (see Table 12-13) or other scales can be used to determine level of symptom distress or severity. RPE is also used when the client is taking cardiac medications that blunt heart rate response to exercise or when other conditions and comorbidities are present that may prevent the use of target heart rate formulas.
Watch closely for early signs (dyspnea, pallor, sweating, and fatigue) of cardiopulmonary complications of cancer treatment. The activity level of someone with anemia also may require adjustment. This client may have elevated pulse and respiratory rates because of hypoxia, with increased cardiac output resulting from the body’s effort to maintain an adequate oxygen supply.
Exercise During and After Chemotherapy or Radiation Therapy
Bone marrow suppression is a common and serious side effect of many chemotherapeutic agents and can be a side effect of radiation therapy in some instances such as when radiation dosage exceeded 50 Gy in the past or currently when high levels of radiation are used to prevent death despite potential radiotherapy toxicities later. Therefore it is extremely important to take a client history of current or past radiation therapy dosages and to monitor the hematologic values in clients receiving these treatment modalities.
The therapist must review these values before any type of vigorous exercise or activity is initiated. Current guidelines recommend that individuals undergoing chemotherapy or radiation therapy should not exercise within 2 hours of the treatment because increases in circulation may attenuate (alter or change) the effects of the treatment.67 Although this recommendation is not based on evidence-based research, it is a guideline followed by the National Institutes of Health (NIH) because of the physiologic effects of moderate to vigorous physical activity and exercise on the redistribution of cardiac output and blood flow to the working muscle. In the case of both radiation therapy and chemotherapy, there is a potential to enhance treatment toxicity with the shift in blood flow. The suggested 2-hour delay is reasonable given the half-life of most chemotherapeutic agents and the rate of decay of fractionated doses of radiation.151
Moderate intensity aerobic exercise (walking for 20 to 45 minutes, 3 to 5 times per week at 50% to 70% of measured maximum heart rates during 7 weeks of radiation) has been shown to maintain erythrocyte levels during radiation treatment (of breast cancer).49 Physical activity can also improve mood and reduce anxiety and mental stress for people undergoing chemotherapy. Independence and QOL improve as functional ability improves.44,46,161 A helpful guideline to indicate when aerobic exercise is contraindicated (or when reevaluation of the exercise program is indicated) in chemotherapy clients is given in Box 9-6. Keep in mind these values are primarily educated estimates based on clinical consensus; there is a need for further research and stronger evidence to support these values.151
Older adults, especially older adults with bone disease or significant comorbidities and impairments, such as arthritis or peripheral neuropathies, still need help with balance, strength, and coordination to remain safe from falls and injuries.
Exercise for Cancer-Related Fatigue
Fatigue related to cancer treatment is common and disabling for many people. People in cancer treatment are often advised to rest after chemotherapy, but aerobic exercise and physical activity have been shown to help improve energy level and stamina, reduce fatigue, reduce nausea, increase muscle mass, and increase daily activities without increasing fatigue. The use of exercise as an adjunct intervention for CRF is gaining in favor as an effective strategy.191 Traditional (e.g., walking or aerobics) and alternative forms of exercise (e.g., Tai chi or yoga) are being investigated.65,206 Exercise may increase the body’s ability to recover from the effects of chemotherapy.9
Much of the exercise-related research is focused on breast cancer, but aerobic exercise after bone marrow transplantation43,172 and exercise during treatment of cancers, such as multiple myeloma,33 solid tumors, and lymphoma, have been studied.206 Exercise combined with improved nutrition for CRF and deconditioning has also been reported successful in demonstrating significant improvements in the 6-minute walk test distance, the squat test, and fatigue level.142
Clients receiving chemotherapy and radiation therapy experiencing CRF who are already on an exercise program may need to exercise temporarily at a lower intensity and progress at a slower pace; the goal is to remain as active as possible. For sedentary individuals, low intensity activities, such as stretching and brief, slow walks, can be implemented and slowly advanced.20
However, it can be difficult to convince someone who is extremely tired (and especially those who did not exercise before their cancer diagnosis) that exercise will improve his or her symptoms. The therapist may have to begin with discussions over a period of time about the importance of exercise. This is especially important if the person is significantly deconditioned. The therapist can begin with an assessment of previous exercise or activity patterns. Ask the following questions:
• Can you do your normal daily activities?
• Can you participate in a formal or informal exercise program?
• What is your normal amount and frequency of exercise?
• Have you had to modify or change your exercise level or other activity patterns since the development of fatigue?
Symptoms of fatigue, headache, and lethargy begin in most people when hemoglobin falls to 12 g/dl. Mildto-moderate graded exercise is possible for many people at this level. Symptoms become more pronounced when hemoglobin decreases to 10 g/dl, reducing exercise capacity.
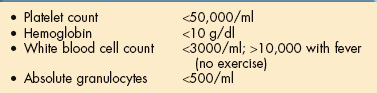
Exercise is not always possible for most people when the hemoglobin level is 8 g/dl or less. Hemoglobin levels should be maintained around 12 g/dl during the administration of chemotherapy,13 but protocols vary from institution to institution and even from one physician to another within the same facility. Bloodless medicine (see discussion in Chapter 14) has made it possible for clients to tolerate lower hemoglobin levels previously thought unacceptable without compromising oxygen delivery. See also exercise guidelines provided in Tables 40-8 and 40-9, keeping in mind that most oncology settings have their own guidelines that may be more liberal. For example, in some oncology settings, exercise is contraindicated when white blood cell count drops below 500/mm3 (compared to 5000/mm3 listed in Table 40-8) and when platelets drop below 5000/mm3 (compared to 20,000/mm3 listed in Table 40-9).
The American College of Sports Medicine (ACSM) guidelines for termination of testing or training may also be consulted.1 People with cancer are advised to contact their physician if any of the abnormal responses listed in Box 9-7 develop.
Exercise and Bone Metastases
See Special Implications for the Therapist: Metastatic Tumors in Chapter 26.
Prescriptive Exercise
Types, limitations, and precautions of prescriptive exercise intervention in the treatment of cancer, especially cancer pain, are being studied. The programs studied vary in length from 6 weeks for individuals who were going through radiation therapy to 6 months for those in chemotherapy and the entire duration of bone marrow transplantation. The exercise interventions vary somewhat but most include progressive programs of 15 to 30 minute sessions, 3 to 5 days a week, at an intensity equal to 60% to 80% of maximum heart rate (RPE 11 to 14). A perceived exertion of no greater than 12 may be used as a guide when exercise testing is not possible (see Table 12-13).
The frequency and duration of exercise are determined by the clinical status of the person. If weight training is prescribed, high-repetition, low-weight circuit programs are recommended that do not exceed RPE of 14.109 Other clinical tools for monitoring and more specific guidelines for exercise are available.45,48,149
Individuals who exercised more than 60 minutes per day were more likely to report higher levels of fatigue, suggesting a maximum effective dose for individuals receiving adjuvant chemotherapy (in this case for breast cancer). No serious adverse events were reported in any of the studies, although anyone in the high-risk category with serious comorbidities was excluded, and most exercise programs were flexible and symptom-limited.
The reported outcomes of these and other studies show that exercise has a powerful effect on CRF, with fatigue levels reported as 40% to 50% lower in exercising participants. Exercise reduces fatigue and emotional distress and improves QOL.
Without exception, all of these studies showed lower levels of fatigue and emotional distress as well as decreased sleep disturbance (if this was studied as an outcome) in people who exercised during treatment compared to controls or to baseline scores in single-group designs. A summary of the studies included is available.132
Not all people with cancer are able to participate in aerobic exercise. People who ambulate less than 50% of the time or who are confined to bed and those who fatigue with mild exertion may not be candidates for aerobic exercise.213 Range of motion and gentle resistive work until tolerance for activity improves are still important. Some people may become easily fatigued with minimal exertion. Energy-conservation techniques and work simplification (Box 9-8) may be necessary for the person with chronic fatigue and for those whose functional status is declining. Therapeutic exercise should be scheduled during periods when the person has the highest level of energy.
Generalized weakness associated with cancer treatment can be more debilitating than the disease itself. Whenever possible, exercise, including strength training and cardiovascular training, is an essential component for many people with cancer. Improving strength and endurance aids in countering the effects of the disease and the effects of medical interventions. Increased physical activity may increase the homeostatic sleep drive to increase nighttime sleep and may help relieve CRF.33
Interval exercise or a bedside exercise program may be preferred at first. Interval exercise may be the only treatment possible in this circumstance. This is performed during frequent but short sessions throughout the day with work-rest intervals beginning at the person’s level of tolerance. This may be no more than 1 minute of exercise activity followed by 1 minute of rest, then 1 minute of exercise, and so on. As the person’s endurance level increases, the duration of work may be increased and the interval of rest decreased. See also Special Implications for the Therapist: Anemia in Chapter 14.
Exercise and Lymphedema
In the past, therapists were cautioned to carefully design a program that did not cause or exacerbate cancer-related complications such as edema. It was advised that repetitive or strenuous exercise would increase the production of lymph fluid and lymphedema would be the result because lymph nodes were removed during surgery, damaged by radiation therapy, or invaded by the tumor, leaving scar tissue that prevented normal lymph drainage. In fact, it is now known that exercise activates muscle groups and joints in the affected extremity and does not induce lymphedema.77,78 Resistance training has not been shown to adversely affect lymphedema.2
Combining a specific exercise program for each individual with the use of sufficient compression will facilitate the process of decongestion by using the natural pumping effect of the muscles to increase lymph flow while preventing limb refilling. Most clinicians experienced in lymphedema treatment agree on basic guidelines for exercise. See further discussion in Chapter 13.
Exercise and Advanced Cancer
With improved detection and treatment, more and more people are living with cancer as a controllable chronic disease or in advanced stages of cancer. There is insufficient research on exercise in such individuals to make specific recommendations for physical activity and exercise. In such cases, therapists are advised to prescribe exercise based on individual needs and abilities.
General training precautions for warmup and cool down should be followed while monitoring for abnormal heart rate or blood pressure responses and observing each individual for pathologic symptomatic responses (e.g., hypertension, chest pain, onset of wheezing, claudication or leg cramps, shortness of breath, and dizziness or fainting). Clients should be encouraged to remain adequately hydrated at all times unless medically directed otherwise.
Compromised skeletal integrity, especially in the presence of muscle wasting, increases the risk of fracture and may prevent weight-bearing activities. Aerobic exercise may have to begin with non–weight-bearing exercise, such as cycling, rowing, or swimming (for those who are not immunocompromised or neutropenic), with a gradual return to weight-bearing activities whenever possible to prevent loss of bone density. People with severe muscle weakness may tolerate cycling better than walking.148 Interval exercise (periods of exercise alternated with periods of rest) may be used with a goal in mind of increasing the exercise time and decreasing the rest.148
Exercise for Cancer Survivors
Being sedentary is a risk factor for several of the most common types of cancer (e.g., breast and colon). Survivors tend to decrease levels of physical activity and exercise during and after completion of their treatment, especially if they were sedentary before their cancer diagnosis.94 Low-intensity exercise can seem like high intensity for these individuals. In addition, some therapies reduce exercise capacity because of cardiopulmonary, neurologic, and musculoskeletal impairments. Until studies verify these findings, it is assumed that the beneficial effects of activity and exercise on cardiovascular health, bone strength, lean body mass, and balance also apply to cancer survivors.
The type, frequency, duration, and intensity of exercise should be individualized based on the survivor’s age, previous fitness level, type of cancer and cancer treatment, and the presence of any additional comorbidities. Some specific guidelines are available in Box 9-9.
Until results of systematic studies are available, the ACS recommends at least 30 to 60 minutes of moderate to vigorous physical activity at least 5 days per week to reduce the risk of cancer, cardiovascular disease, and diabetes.103,111 There is no reason to think these recommendations would not benefit cancer survivors. Any step toward this goal should be encouraged.
Survivors should be educated to understand that any exercise has a linear benefit with increasing health benefit with higher volume of physical activity. Caution should be provided that extremely high levels of exercise might increase the risk for infections and exercise-related injuries.137 See further discussion in the section on Exercise and the Immune System in Chapter 7.
CHILDHOOD CANCER
Each year approximately 8400 children in the United States are diagnosed with cancer; approximately 2000 deaths of children 19 and under are attributed to cancer.96 With recent advances in treatment, 79% of these children will survive 5 years or more (improved from 58% for children diagnosed 30 years ago and an increase of almost 40% since the early 1960s). Cancer is the second leading cause of death among children between 1 and 14 years of age.96,116 Treatment-related deaths have declined as a result of advances in clinical supportive care (e.g., antibiotic therapy, indwelling venous-access lines, blood products, and enteral and parenteral nutrition) that maximize the benefits and minimize the side effects of cancer therapy.
The types of cancers that occur in children vary greatly from those seen in adults. Leukemias, particularly acute lymphocytic leukemia (ALL); lymphomas (almost half of all childhood cancers involve the blood or blood-forming organs); brain tumors; embryonal tumors; and soft tissue sarcomas are the most common pediatric malignancies, whereas adenocarcinomas (e.g., lung, breast, or colorectal) are more common in adults.104,116
Other differences that must be taken into account when treating the child with cancer include the stage of growth and development, stage of psychosocial and cognitive development, and emotional response of the child to the illness and its treatment. The immaturity of the child’s organ systems often has important treatment implications.
Types of Childhood Cancers
The most common pediatric malignancies are ALL, non-Hodgkin’s lymphoma, Hodgkin’s disease, and primary CNS tumors. Neuroblastoma, Wilms’ (kidney) tumor, rhabdomyosarcoma, and retinoblastomas are the types of solid tumors occurring most frequently in children.116
ALL, the most common childhood malignancy, accounts for almost one-third of all pediatric cancers. White males are affected most often, and although the exact cause is unknown, radiation, chromosomal abnormalities, viruses, and congenital immunodeficiencies have all been associated with an increased incidence of leukemia. See also the section on The Leukemias in Chapter 14.
Wilms’ tumor, a malignancy that may affect one or both kidneys, occurs in children under the age of 14 years and is slightly more prevalent in females than males. Epidemiologic research suggests an increased incidence in children of men exposed to lead or hydrocarbons. Recently, an association between Wilms’ tumor and chromosomal abnormalities has been established, specifically deletion of a suppressor gene located on the short arm of chromosome 11. This chromosomal anomaly is an autosomal dominant trait requiring evaluation of other family members.
Neuroblastoma is the most common extracranial solid tumor in children and the most commonly diagnosed neoplasm during the first year of life. Approximately 500 new cases are diagnosed annually in the United States, and the incidence is higher among whites than nonwhites. Neuroblastoma can originate anywhere along the sympathetic nervous system, but more than half the tumors occur in the retroperitoneal area and present as an abdominal mass. Other common sites include the posterior mediastinum, pelvis, and neck. If the bone marrow is involved, bone pain may occur. See the section on Neuroblastoma in Chapter 30.
Rhabdomyosarcoma is the most common soft tissue sarcoma and the seventh leading cause of cancer in children. This tumor, which is more prevalent in males than females, originates from the same embryonic cells that give rise to striated muscle. The peak incidence is between the age of 2 and 5 years and a second peak occurs between 15 and 19 years, with much improved survival rates with early detection and treatment today. The most common tumor sites include the head and neck, genitourinary tract, and extremities. Rhabdomyosarcoma of the head and neck can lead to CNS involvement, including cranial nerve palsies, meningeal symptoms, and respiratory paralysis. See further discussion in Chapter 26.
Other common cancers seen in children are bone cancers, both osteogenic and Ewing’s sarcomas (see Chapter 26), and brain tumors (see Chapter 30).
Late Effects and Prognosis
As advances in cancer therapy improve, the prognosis of children with malignancies continues to improve. Over the past 25 years, there have been significant improvements in the 5-year survival rate for many childhood cancers, especially ALL and acute myeloid leukemia, non-Hodgkin’s lymphoma, and Wilms’ tumor. Between 1974 and 1996, 5-year survival rates among children for all cancer sites combined improved from 56% to 75%.96 With increasing survival rates, there is a growing concern about the late effects of disease and treatment.
The term late effects refers to the damaging effects of surgery, radiation, and chemotherapy on nonmalignant tissues, as well as to the social, emotional, and economic consequences of survival. These effects can appear months to years after treatment and can range in severity from subclinical to clinical to life-threatening. Fortunately, not all children experience such effects, but those who do often end up in the rehabilitation setting.
Late effects have been identified in almost every organ system. Treatment involving the CNS can cause deficits in intelligence, hearing, and vision. Treatment involving the CNS, head and neck, or gonads can cause endocrine abnormalities such as short stature, hypothyroidism, or delayed secondary sexual development. Children treated with anthracyclines (e.g., doxorubicin [Adriamycin]) are at risk for development of cardiomyopathies, especially with increasing cumulative doses.189
Surgery and radiation involving the musculoskeletal system have been associated with defects such as kyphosis, scoliosis, and spinal shortening. Finally, the child who has received radiation or chemotherapy has a tenfold greater chance of developing a second malignancy than a child who has never had cancer.
References
1. ACSM (American College of Sports Medicine). Guidelines for exercise testing and prescription, ed 6. Philadelphia: Lippincott, 2000.
2. Ahmed, RL. Randomized controlled trial of weight training and lymphedema in breast cancer survivors. J Clin Oncol. 2006;24:2765–2772.
3. American Cancer Society (ACS): Cancer facts and figures 2006. Available on-line at http://www.cancer.org Accessed December 9, 2006.
3a. American Cancer Society (ACS). Guidelines on nutrition and physical activity for cancer prevention. CA Cancer J Clin. 2006;56(5):254–281.
4. American Cancer Society (ACS): Guide to cancer drugs. Available on-line at http://www.cancer.org/docroot/cdg/cdg_0.asp Accessed January 22, 2007.
5. American Cancer Society (ACS). Guide to complementary and alternative cancer methods. New York: The Society, 2000.
6. American Cancer Society (ACS): Nutrition and cancer: strategy 2000. CA Cancer J Clin. 1999;49(6):331–361.
6a. American Cancer Society (ACS): Preserving fertility after cancer. CA Cancer J Clin. 2005;55(4):211–228.
7. American Institute for Cancer Research. Food, nutrition and the prevention of cancer: a global perspective. Washington, DC: The Institute, 1997.
8. Anllo, LM. Sexual life after breast cancer. J Sex Marital Ther. 2000;26(3):241–248.
9. Armstrong, D. Easing the unwanted effects of chemotherapy. Johns Hopkins Med Lett. 2007;18(12):4–5.
10. Austin Research Institute: ARI achievements. Available on-line at http://www.ari.unimelb.edu.au/achieve
11. Bach, EC. Cancer rehabilitation in the home care setting. Rehabil Oncol. 2000;18(1):32–33.
12. Bach, PB, Schrag, D, Brawley, OW. Survival of blacks and whites after a cancer diagnosis. JAMA. 2002;287:2106–2113.
13. Balducci, L. Management of cancer in the elderly. Oncology. 2006;20(2):135–152.
14. Baquet, CR, Commiskey, P, Mack, K, et al. Esophageal cancer epidemiology in blacks and whites: racial and gender disparities in incidence, mortality, survival rates and histology. J Natl Med Assoc. 2005;97(11):1471–1478.
15. Bennett, M, Ryall, N. Using the modified Barthel index to estimate survival in cancer patients in hospice: observational study. BMJ. 2000;321:1381–1382.
16. Bernabei, R, Gambassi, M, Lapane, K, et al. Management of pain in elderly patients with cancer. JAMA. 1998;279:1877–1882.
17. Body, JJ. Bisphosphonates for malignancy-related bone disease: current status, future developments. Support Care Center. 2006;14(5):408–418.
18. Brawley, OW. Some perspective on black-white cancer statistics. CA Cancer J Clin. 2002;52(6):322–325.
19. Brown, J, Byers, T, Thompson, K, et al. Nutrition during and after cancer treatment: a guide for informed choices by cancer survivors. CA Cancer J Clin. 2001;51(3):153–187.
20. Bussi, MK. Advances in radiation therapy: conventional to 3D, to IMRT, to 4D, and Beyond. CA Cancer J Clin. 2005;55(2):117–134.
21. Calle, EE. Overweight obesity, and mortality from cancer in a prospectively studied cohort of U.S. adults. N Engl J Med. 2003;348(17):1625–1638.
22. Camphausen, K, Moses, MA, Beecken, WD, et al. Radiation therapy to a primary tumor accelerates metastatic growth in mice. Cancer Res. 2001;61(5):2207–2211.
23. Campisi, J. Cancer, aging and cellular senescence. In Vivo. 2000;14:183–188.
24. Carati, CJ. Treatment of postmastectomy lymphedema with low-level laser therapy: a double blind, placebo-controlled trial. Cancer. 2003;98(6):1114–1122.
25. Cech, T. Life at the end of the chromosome: telomeres and telomerase. Angew Chem Int Ed Engl. 2000;39(1):34–43.
26. Cech, TR, Egan, LW, Doyle, C, et al. The biomedical research bottleneck. Science. 2001;293(5530):573.
27. Chaudhari, BR. Following the TRAIL to apoptosis. Immunol Res. 2006;35(3):249–262.
28. Chen, MS, Jr. Cancer health disparities among Asian Americans: what we do and what we need to do. Cancer. 2005;104(12 Suppl):2895–2902.
29. Cherny, N. The management of cancer pain. CA Cancer J Clin. 2000;50:70–120.
30. Cheson, B. Hodgkin’s disease and the non-Hodgkin’s lymphomas. In: Lenhard R, Osteen R, Gansler T, eds. The American Cancer Society’s clinical oncology. Atlanta: American Cancer Society, 2001.
31. Citrin, D, Menard, C, Camphausen, K. Combining radiotherapy and angiogenesis inhibitors: clinical trial design. Int J Radiat Oncol Biol Phys. 2006;64(1):15–25.
32. Clemons, M, Dranitsaris, G, Cole, D, et al. Too much, too little, too late to start again? Assessing the efficacy of bisphosphonates in patients with bone metastases from breast cancer. Oncologist. 2006;11(3):227–233.
33. Coleman, EA. Feasibility of exercise during treatment of multiple myeloma. Cancer Nursing. 2003;26(5):410–419.
34. Corbin, L. Safety and efficacy of massage therapy for patient with cancer. Cancer Control. 2005;12(3):158–164.
35. Costanza, M, Li, F, Finn, L, et al. Cancer prevention: strategies for practice. In: Lenhard R, Osteen R, Gansler T, eds. The American Cancer Society’s clinical oncology. Atlanta: American Cancer Society, 2001.
36. Courneya, KS. Exercise in cancer survivors: an overview of research. Med Sci Sports Exerc. 2003;35:1846–1852.
37. Courneya, KS, Friedenreich, CM. Physical exercise and quality of life following cancer diagnosis: a literature review. Ann Behav Med. 1999;21(2):171–179.
38. Dalzell MA: Biophysical modalities in oncology: guidelines for the use of electrical stimulation, laser and ultrasound. Proceedings from Cancer rehabilitation: the multidisciplinary integration of traditional and “whole person” care, Montreal, L’Espirit Rehabilitation Centers, January 21-23, 2000.
39. Danaei, G. Causes of cancer in the world: comparative risk assessment of nine behavioural and environmental risk factors. Lancet. 2005;366(9499):1784–1793.
40. DeMichele, A, Glick, J. Cancer-related emergencies. In: Lenhard R, Osteen R, Gansler T, eds. The American Cancer Society’s clinical oncology. Atlanta: American Cancer Society, 2001.
41. Deng, G, Cassileth, BR. Integrative oncology: complementary therapies for pain, anxiety, and mood disturbance. CA Cancer J Clin. 2005;55(2):109–116.
42. Department of Health and Human Services. The health consequences of smoking: a report of the surgeon general. Rockville, MD: Department of HHS, Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health, 2004.
43. Dimeo, F. Aerobic exercise following bone marrow transplantation. Rehabil Oncol. 2000;18(3):6–7.
44. Dimeo, F. Exercise programs for cancer patients during chemo-and radiotherapy. Rehabil Oncol. 2000;18(3):5.
45. Dimeo, F. Strategies in managing cancer fatigue. Rehabil Oncol. 1999;17(3):27–28.
46. Dimeo, F, Rumberger, BG, Keul, J. Aerobic exercise as therapy for cancer fatigue. Med Sci Sports Exerc. 1998;30:475–478.
47. Doyle, C. Nutrition and physical activity during and after cancer treatment: An American Cancer Society guide for informed choices. CA Cancer J Clin. 2006;56(6):323–353.
48. Drouin, J, Pfalzer, LA. Aerobic exercise guidelines for the person with cancer. Acute Care Pers. 2001;10(1&2):18–24.
49. Drouin, JS, Young, TJ, Beeler, J, et al. Random control clinical trial on the effects of aerobic exercise training on erythrocyte levels during radiation treatment for breast cancer. Cancer. 2006;107:2490–2495.
50. Duffner, PK. The long-term effects of chemotherapy on the central nervous system. J Biol. 2006;5(7):21.
51. Eatock, AM, Scatzlein, A, Kayes, L. Tumour vasculature as a target for anticancer therapy. Cancer Treat Rev. 2000;26:191–204.
52. Ershler, WB. Issues of aging and geriatric medicine: relevance to cancer treatment and hematopoietic reconstitution. Biol Blood Marrow Transplant. 2006;12(Suppl 1):100–106.
53. Ershler WB: Working group 7: the biology of aging and cancer. National Institute on Aging, 2005. Available on-line at http://www.nia.nih.gov Accessed April 2, 2006.
54. Feltman B: Query posted on the APTA Oncology Section List Serve, February 2, 2007.
55. Ferlay J: GLOBOCAN 2002. Cancer incidence, mortality and prevalence worldwide. IARC Cancer base No. 5. Version 2.0. Lyon, France, IARC Press, 2004. Available on-line at http://www-dep.iarc.fr/globocan/database.htm Accessed January 30, 2007.
56. Fidler, IJ. Cancer biology is the foundation for therapy. Cancer Biol Ther. 2005;4(9):1036–1039.
57. Fidler, IJ. Metastasis: quantitative analysis of distribution and fate of tumor embolilabeled with 125 I-5-iodo-2′-deoxyuridine. J Natl Cancer Inst. 1970;45(4):773–782.
58. Fischer, SM, Gozansky, WS, Sauaia, A, et al. A practical tool to identify patients who may benefit from a palliative approach: the CARING criteria. J Pain Symptom Manage. 2006;31(4):285292.
59. Folkman, J. Angiogenesis. Annu Rev Med. 2006;57:1–18.
60. Folkman, J. Antiangiogenesis in cancer therapy endostatin and its mechanisms of action. Exp Cell Res. 2006;312(5):594–607.
61. Fong, CS. Recent advance in immunological tests in paraneoplastic neurological syndrome. Acta Neurol Taiwan. 2005;14(1):28–35.
62. Franceschi, S. Strategies to reduce the risk of virus-related cancers. Ann Oncol. 2000;11(9):1091–1096.
63. Gabanelli, P. A rehabilitative approach to the patient with brain cancer. Neurol Sci. 2005;26(Suppl 1):S51–S52.
64. Gahart, B, Nazareno, A. 2001 Intravenous medications, ed 16. St Louis: Mosby, 2001.
65. Galantino, ML. The effects of Tai Chi and walking on fatigue and body mass index in women living with breast cancer. Rehabil Oncol. 2003;21(1):17–22.
66. Ganz, PA. Quality of life and cancer rehabilitation. Rehabil Oncol. 1999;17(3):9–11.
67. Gerber, L, Augustine, E. Rehabilitation management: restoring fitness and return to functional activity. In: Harris J, Lippman M, Morrow M, et al, eds. Diseases of the breast. Philadelphia: Lippincott, 2000.
68. Giovannucci, E. Nutritional factors in human cancers. Adv Exp Med Biol. 1999;472:29–42.
69. Glaves, D. Correlation between circulating cancer cells and incidence of metastases. Br J Cancer. 1983;48(5):665–673.
70. Goodman, CC, Snyder, TE. Differential diagnosis for physical therapists: screening for referral, ed 4. Philadelphia: WB Saunders, 2007.
71. Goudas, LC. The epidemiology of cancer pain. Cancer Invest. 2005;23(2):182–190.
72. Graham, JE. Stress, age, and immune function: toward a lifespan approach. J Behav Med. 2006;29(4):389–400.
73. Gudas, S. The physical therapy challenge in disseminated cancer. APTA Newsletter. 1987;5:3.
74. Guerreiro, AS. IGF-1R: Potential role in antitumor agents. Drug News Perspect. 2006;19(5):261–272.
75. Hansen, PE. Personality traits, health behavior, and risk for cancer: a prospective study of Swedish twin court. Cancer. 2005;103(5):1082–1091.
76. Harlow SP: Lymphatic mapping and sentinel node biopsy. ACS Surgery (Web MD), 2005. Available on-line at http://www.medscape.com/viewarticle/498442. Accessed February 8, 2005.
77. Harris, SR. Challenging myths in physical therapy. Phys Ther. 2001;81(6):1180–1183.
78. Harris, SR, Niesen-Veertommen, SL. Challenging the myth of exercise-induced lymphedema following breast cancer: a series of case reports. J Surg Oncol. 2000;74(2):95–98.
79. Hayflick, L. The limited in vitro lifetime of human diploid cell strains. Exp Cell Res. 1965;37:614–636.
80. Heath, C, Fontham, E. Cancer etiology. In: Lenhard R, Osteen R, Gansler T, eds. The American Cancer’s Society clinical oncology. Atlanta: American Cancer Society, 2001.
81. Herberman, R. Basis for current major therapies for cancer, section E. In: Lenhard R, Osteen R, Gansler T, eds. The American Cancer Society’s clinical oncology. Atlanta: American Cancer Society, 2001.
82. Herbst, RS. Clinical cancer advances 2005: major research advances in cancer treatment, prevention, and screening a report from the American Society of Clinical Oncology. J Clin Oncol. 2006;24(1):190–205.
83. Hertzog, RG. Ancestral telomere shortening: a countdown that will increase life span? Med Hypotheses. 2006;67:157–160.
84. Hill, C, Cleeland, C, Gustein, H. Effective pain treatment in cancer patients. In: Lenhard R, Osteen R, Gansler T, eds. The American Cancer Society’s clinical oncology. Atlanta: American Cancer Society, 2001.
85. Hillner, BE, Ingle, JN, Berenson, JR, et al. American Society of Clinical Oncology guideline on the role of bisphosphonates in breast cancer. J Clin Oncol. 2000;18(6):1378–1391.
86. Hoffman, B. Cancer survivors at work: a generation of progress. CA Cancer J Clin. 2005;55(5):271–280.
87. Hogan, RD, Burke, KM, Franklin, TD. The effect of ultrasound on microvascular haemodynamics in skeletal muscle: effects during ischaemia. Microvasc Res. 1982;23:370–379.
88. Hughes, MK. Sexuality and the cancer survivor: a silent coexistence. Cancer Nurs. 2000;23(6):477–482.
89. Humpel, N, Iverson, DC. Review and critique of the quality of exercise recommendations for cancer patients and survivors. Support Care Cancer. 2005;13:493–502.
90. Hutchins, LF. Under-representation of patients 65 years of age or older in cancer-treatment trials. N Engl J Med. 1999;341:2061–2067.
91. Inagaki, M. Smaller regional volumes of brain gray and white matter demonstrated in breast cancer survivors exposed to adjuvant chemotherapy. Cancer. 2007;109(1):146–156.
92. Institute of Medicine (IOM). IOM Food and Nutrition Board: dietary reference intakes for vitamin C, vitamin E, selenium, and carotenoids. Washington, DC: National Academy Press, 2000.
93. aa Institute of Medicine (IOM): Unequal treatment: confronting racial and ethnic disparities in health care. Available on-line at http://www.iom.edu/CMS/3740/4475.aspx Accessed on April 27, 2006.
94. Irwin, ML. Physical activity levels before and after a diagnosis of breast cancer. HEAL study. Cancer. 2003;97:1746–1757.
95. Iwata, T. Thymectomy for paraneoplastic stiff-person syndrome associated with invasive thymoma. J Thorac Cardiovasc Surg. 2006;132(1):196–197.
96. Jemal, A. Cancer statistics. CA Cancer J Clin. 2007;57(1):43–66.
97. Katoh, M, Katoh, M. Bioinformatics for cancer management in the post-genome era. Technol Cancer Res Treat. 2006;5(2):169–176.
98. Kelloff, GJ, Crowell, JA, Steele, VE, et al. Progress in cancer chemoprevention: development of diet-derived chemopreventive agents. J Nutr. 2000;130:467S–471S. [(suppl)].
99. Kiecolt-Glaser, JK. Psycho-oncology and cancer: psychoneuroimmunology and cancer. Ann Oncol. 2002;13(Suppl 4):165–169.
100. Kiecolt-Glaser, JK, Glaser, R. Psychoneuroimmunology and cancer: fact or fiction? Eur J Cancer. 1999;35(11):1603–1607.
101. Knols, R. Physical exercise in cancer patients during and after medical treatment: a systematic review of randomized and controlled trials. J Clin Oncol. 2005;23:3830–3842.
102. Kraeber-Bodere, F, Champion, L, Rousseau, C, et al. Treatment of bone metastases of prostate cancer with strontium-89 chloride: efficacy in relation to the degree of bone involvement. Eur J Nucl Med. 2000;27(10):1487–1493.
103. Kushi, LH. American Cancer Society guidelines on nutrition and physical activity for cancer prevention: reducing the risk of cancer with healthy food choices and physical activity. CA Cancer J Clin. 2006;56(5):254–281.
104. Landier, W. Childhood acute lymphoblastic leukemia: current perspectives. Oncol Nurs Forum. 2001;28:823–833.
105. Larsen, J, Nordström, Ljungman, P, et al. Symptom occurrence, symptom intensity, and symptom distress in patients undergoing high-dose chemotherapy with stem-cell transplantation. Cancer Nurs. 2004;27(1):55–64.
106. Lawrence, C, Golbert, C, Peters, W. Evaluation of symptom distress in a bone marrow transplant outpatient environment. Ann Pharmacotherapy. 2006;30(9):941–945.
107. Lejbkowicz, F, Zwiran, M, Salzberg, S. The response of normal and malignant cells to ultrasound in vitro. Ultrasound Med Biol. 1993;19:75–82.
108. Lenhard RE, Osteen R, Gansler T, eds. The American Cancer Society’s clinical oncology. Atlanta: American Cancer Society, 2001.
109. Leutholtz, BC, Ripoll, I. Exercise and disease management. Boca Raton, FL: CRC Press, 1999.
110. Lewis, MM, Reilly, JF. Sports tumors. Am J Sports Med. 1987;15:362–365.
111. Lichtenstein, AH. Diet and lifestyle recommendations revision 2006: a scientific statement from the American Heart Association Nutrition Committee. Circulation. 2006;114:82–96.
112. Lim, C-S, Cellular senescence, cancer mortality, and organismal aging: a paradigm shift. Biochem Biophys Res Commun 2006;344:1–2. Available on-line at http://www.sciencedirect.com. Accessed April 28, 2006.
113. Lorusso, L. Immunological features of neurological paraneoplastic syndromes. Int J Immunopathol Pharmacol. 2004;17(2):135–144.
114. Lossi, C. Clinical hypnosis versus cognitive behavioral training for pain management with pediatric cancer patients undergoing bone marrow aspirations. Int J Clin Exp Hypn. 1999;47:104–116.
115. Mackey, KM, Sparling, JW. Experiences of older women with cancer receiving hospice care: significance for physical therapy. Phys Ther. 2000;80(5):459–468.
116. Marcus, K. Pediatric solid tumors. In: Lenhard R, Osteen R, Gansler T, eds. The American Cancer Society’s clinical oncology. Atlanta: American Cancer Society, 2001.
117. Maxwell, L. Therapeutic ultrasound and tumor metastasis. Physiotherapy. 1995;81:272–275.
118. McGarvey, CL, Gergich, NL, Soballe, P, et al. A case report: breast cancer metastasis and implications of bony metastasis on activity and ambulation. Rehabil Oncol. 2006;24(1):4–17.
119. McGarvey, CM. Staging and management of patients with breast cancer. Baltimore: Norton School of Lymphatic Therapy, 2004.
120. Michaud, D, Spiegelman, D, Clinton, S, et al. Fruit and vegetable intake and incidence of bladder cancer in a male prospective cohort. J Natl Cancer Inst. 1999;91:605–613.
121. Molassiotis, A, Van Den Akker, OB, Milligan, DW, et al. Psychological adaptation and symptom distress in bone marrow transplant recipients. Psychooncology. 1996;5:9–22.
122. Moldoveanu, AI, Shepard, RJ, Shek, PN. The cytokine response to physical activity and training. Sports Med. 2001;31(2):115–144.
123. Monier, R. Fundamental aspects, mechanisms of carcinogenesis and dose-effect relationships. C R Acad Sci III. 2000;323:603–610.
124. Mundy, GR. Mechanisms of bone metastasis. Cancer. 1997;80(8 suppl):1546–1553.
125. Mundy, GR. Metastasis to the bone: causes, consequences, and therapeutic opportunities. Nat Rev Cancer. 2002;2:584–593.
126. Munoz, P. Role of the TRF2 telomeric protein in cancer and ageing. Cell Cycle. 2006;5(7):718–721.
127. Murinson, B. Stiff-person syndrome. Neurologist. 2004;10(3):131–137.
128. Nakaya, N. Personality traits and cancer survival: a Danish cohort study. Br J Cancer. 2006;95(2):146–152.
129. National Academy of Science Research Council: Health risks from exposure to low levels of ionizing radiation: BEIR VII-Phase 2, 2005. Available on-line at http://books.nap.edu/catalog/11340.html Accessed September 5, 2006.
130. National Cancer Institute (NCI): Staging: questions and answers. Available on-line at http://www.cancer.gov/cancertopics/factsheet/detection/staging Accessed January 3, 2007.
131. National Comprehensive Cancer Network (NCCN): Adult cancer pain treatment guideline v.1: 2006. Available on-line at http://www.nccn.org Accessed February 10, 2007.
132. National Comprehensive Cancer Network (NCCN): Clinical practice guidelines in oncology: cancer-related fatigue, Table 1. V.1.2007. Available on-line at http://www.nccn.org Accessed on January 29, 2007.
133. National Comprehensive Cancer Network (NCCN): Clinical practice guidelines in oncology: cancer-related fatigue, V.1.2007. Available on-line at http://www.nccn.org Accessed on January 29, 2007.
134. National Comprehensive Cancer Network (NCCN): Clinical practice guidelines in oncology: palliative care, V.1.2006. Available on-line at http://www.nccn.org Accessed February 11, 2007.
135. National Comprehensive Cancer Network (NCCN): Clinical practice guidelines in oncology: pediatric cancer pain, V.1. 2006. Available on-line at http://www.nccn.org Accessed February 10, 2007.
135a. National Hospice and Palliative Care Organization: About NHPCO. Available on-line at http://www.nho.org Accessed March 13, 2007.
136. Nguyen-Huu, BK. Antiamphiphysin-positive stiff-person syndrome associated with small cell lung cancer. Mov Disord. 2006;21(8):1285–1287.
137. Nieman, DC. Is infection risk related to exercise workload? Med Sci Sports Exerc. 2000;32:S406–S411. [(Suppl)].
138. O’Brien, K, et al. Cancer statistics for Hispanics, 2003. CA Cancer J Clin. 2003;53(4):208–226.
139. Olumi, A. A critical analysis of the use of p53 as a marker for management of bladder cancer. Urol Clin North Am. 2000;27:75–82.
140. Onn, A, Isobe, T, Itasaka, S, et al. Development of an orthotopic model to study the biology and therapy of primary human lung cancer in nude mice. Clin Cancer Res. 2003;9(15):5532–5539.
141. Osteen, R. Breast cancer. In: Lenhard R, Osteen R, Gansler T, eds. The American Cancer Society’s clinical oncology. Atlanta: American Cancer Society, 2001.
142. Packel, L. Cancer-related fatigue and deconditioning: a program evaluation. Rehab Oncol. 2006;24(2):3–8.
143. Parkin, DM. Global cancer statistics, 2002. CA Cancer J Clin. 2005;55:74–108.
144. Patel, AV. Obesity, recreational physical activity, and risk of pancreatic cancer in a large U.S. cohort. Cancer Epidemiol Biomarkers Prev. 2005;14(2):459–466.
145. Pendino, F. Telomeres and telomerase: pharmacological targets for new anticancer strategies? Curr Cancer Drug Targets. 2006;6(2):147–180.
146. Penson, RT, Gallagher, J, Gioiella, ME, et al. Sexuality and cancer: conversation comfort zone. Oncologist. 2000;5(4):336–344.
147. Perry, JK. The oncogenic potential of growth hormone. Growth Horm IGR Res. 2006;16(5-6):277–280.
148. Pfalzer, L. Aerobic exercise for patients with disseminated cancer. Clin Manag Phys Ther. 1988;8(2):28–31.
149. Pfalzer L: Clinical monitoring tools and guidelines for exercise. Proceedings from Cancer Rehabilitation: the multidisciplinary integration of traditional and “whole person” care, Montreal, L’Espirit Rehabilitation Centers, January 21-23, 2000.
150. Pfalzer L: Personal communication, 2006.
151. Pfalzer L: Personal communication, 2007.
152. Pfalzer, L. Physical agents/modalities for survivors of cancer. Rehabil Oncol. 2001;19(2):12–24.
153. Pfalzer, L. Research models and tools for physical therapy in cancer rehabilitation. In Proceedings from Cancer Rehabilitation: the multidisciplinary integration of traditional and “whole person” care. Montreal: L’Espirit Rehabilitation Centers; 2000.
154. Pfalzer L, Drouin JS: Examination and screening of individuals with cancer prior to therapeutic interventions. Combined Sections Presentation, San Diego, California, February 2-6, 2006.
155. Pfeifer, J, Wick, M. Pathologic evaluation of neoplastic diseases. In: Lenhard R, Osteen R, Gansler T, eds. The American Cancer Society’s clinical oncology. Atlanta: American Cancer Society, 2001.
156. Phillips, JM, Williams-Brown, S. Cancer prevention among racial and ethnic minorities. Semin Oncol Nurs. 2005;21(4):278–285.
157. Pickle, LW. A new method of estimating United States and state-level cancer incidence counts for the current calendar year. CA Cancer J Clin. 2007;57(1):30–42.
158. Piper, BF, Dodd, MJ, Ream, E, et al. Improving the clinical measurement of cancer treatment-related fatigue. In Better health through nursing research: international state of the science. Washington, DC: American Nurses Association; 1999.
159. Pittock, SJ. Amphiphysin autoimmunity: paraneoplastic accompaniments. Ann Neurol. 2005;58(1):96–107.
160. Plaut, SM, Rutter, MA. Addressing sexual issues in patients with cancer. Acute Care Perspectives. 2001;10(1&2):41–44.
161. Porock, D, Kristjanson, LJ, Tinnelly, K, et al. An exercise intervention for advanced cancer patients experiencing fatigue: a pilot study. J Palliative Care. 2000;16(3):27. [30-36].
162. Renehan, AG. Obesity and cancer risk: the role of the insulin-IGF axis. Trends Endocrinol Metab. 2006;17(8):328–336.
162a. Ries, E. A special place: physical therapy in hospice and palliative care. PT Magazine. 2007;15(3):42–47.
163. Rinehart-Ayres, M, Pfalzer L, Drouin JS: Exercise training guidelines for individuals with cancer, APTA Combined Sections Meeting. The Oncology Section. New Orleans, February 2005.
164. Ringer, D, Schnipper, L. Principles of cancer biology. In: Lenhard R, Osteen R, Gansler T, eds. The American Cancer Society’s clinical oncology. Atlanta: American Cancer Society, 2001.
165. Rizzo, A. CML: lessons learned when the therapist becomes the patient. Rehabil Oncol. 2003;21(3):4–6.
166. Rock, CL. Nutrition and survival after the diagnosis of breast cancer: a review of the evidence. J Clin Oncol. 2002;20:3302–3316.
167. Rodriquez-Nieto, S. Role of alterations in the apoptotic machinery in sensitivity of cancer cells to treatment. Curr Pharm Des. 2006;12(24):4411–4425.
168. Romanov, S, Kozakiewicz, B, Holst, C, et al. Normal human mammary epithelial cells spontaneously escape senescence and acquire genomic changes. Nature. 2001;409:633–637.
169. Rosenthal, P. Complications of cancer and cancer treatment. In: Lenhard R, Osteen R, Gansler T, eds. The American Cancer Society’s clinical oncology. Atlanta: American Cancer Society, 2001.
170. Santiago-Palma, J, Payne, R. Palliative care and rehabilitation. Cancer. 2001;92(suppl 4):1049–1052.
171. Saslow, D. American Cancer Society guideline for human papillomavirus (HPV) vaccine use to prevent cervical cancer and its precursors. CA Cancer J Clin. 2007;57(1):7–29.
172. Sayre, RS, Marcoux, BC. Exercise and autologous bone marrow transplants. Clin Manage Phys Ther. 1992;12(4):78–82.
173. Schag, CAC, Heinrich, RL. Development of a comprehensive quality of life measurement tool: the CARES. Oncology. 1990;4(5):135–138.
174. Schattner, M. Nutrition support of the patient with cancer. In Shils ME, ed.: Modern nutrition in health and disease, ed 10, Philadelphia: Lippincott, Williams & Wilkins, 2006. [2006].
175. Schmitz, KH. Controlled physical activity trials in cancer survivors: a systematic review and meta-analysis. Cancer Epidemiol Biomarkers Prev. 2005;14:1588–1595.
176. Schottenfeld, D, Beebe-Dimmer, J. Chronic inflammation: a common and important factor in the pathogenesis of neoplasia. CA Cancer J Clin. 2006;56(2):69–83.
177. Schover, LR, McKee, AL. Sexuality rehabilitation. Rehabil Oncol. 2000;18(1):16–18.
178. Seffrin, J. An endgame for cancer. CA Cancer J Clin. 2000;50:4–5.
179. Servaes, P, Verhagen, S, Schreuder, HW, et al. Fatigue after treatment for malignant and benign bone and soft tissue tumors. J Pain Symptom Manage. 2003;26:1113–1122.
180. Sexual Information and Education Council: Annotated bibliographies about sexuality with illness and disability, 2001. Available on-line at http://w.ww.siecus.org
180a. Shapiro, CL, Manola, J, Leboff, M. Ovarian failure after adjuvant chemotherapy is associated with rapid bone loss in women with early-stage breast cancer. J Clin Oncol. 2001;19(14):3306–3311.
181. Sharkey, RM, Goldenberg, DM. Targeted therapy of cancer: new prospects for antibodies and immunoconjugates. CA Cancer J Clin. 2006;56(4):226–243.
182. Shepard, RJ, Shek, PN. Cancer, immune function, and physical activity. Can J Appl Physiol. 1995;20(1):1–25.
183. Shin, JS, Hong, A, Solomon, MJ, et al. The role of telomeres and telomerase in the pathology of human cancer and aging. Pathology. 2006;38(2):103–113.
184. Sicard-Rosenbaum, L, Danoff, JV, Guthrie, JA, et al. Effects of energy-matched pulsed and continuous ultrasound on tumor growth in mice. Phys Ther. 1998;78(3):271–277.
185. Siminoff, LA, Ross, L. Access and equity to cancer care in the USA: a review and assessment. Postgrad Med J. 2005;81(961):674–679.
186. Smith, RA, von Eschenbach, AC, Wender, R, et al. American Cancer Society guidelines for the early detection of cancer. CA Cancer J Clin. 2001;51(1):38–75.
187. Smith-Warner, SA. Alcohol and breast cancer in women: a pooled analysis of cohort studies. JAMA. 1998;279:535–540.
188. Soares, M. Effect of age on survival of critically ill patients with cancer. Crit Care Med. 2006;34(3):715–721.
189. Sorensen, K, Levitt, G, Bull, C, et al. Anthracycline dose in childhood acute lymphoblastic leukemia: issues of early survival versus late cardiotoxicity. J Clin Oncol. 1997;15:61–68.
190. Spitler, L, Grossbard, M, Ernstoff, M, et al. Adjuvant therapy of stage III and IV malignant melanoma using granulocyte-macrophage colony stimulating factor. J Clin Oncol. 2000;18:1614–1621.
191. Stasi, R. Cancer-related fatigue: evolving concepts in evaluation and treatment. Cancer. 2003;98:1786–1801.
192. Strohl, P. Radiation therapy. In: Miaskowski C, Buchsel P, eds. Oncology nursing: assessment and clinical care. St Louis: Mosby, 1999.
193. Stummvoll, GH, Aringer, M, Machold, KP, et al. Cancer polyarthritis resembling rheumatoid arthritis as a first sign of hidden neoplasms. Scand J Rheumatol. 2001;30:40–44.
194. Súilleabháin, KO. Despite initial setbacks, researchers are focusing on antiangiogenic therapy more than ever. OncoLog: Report to Physicians. 49(6), 2004.
195. Svendsen, KB. Breakthrough pain in malignant and non-malignant diseases: a review of prevalence, characteristics and mechanisms. Eur J Pain. 2005;9(2):195–206.
196. Tatu, B. Physical therapy intervention with oncological emergencies. Rehabil Oncol. 2005;23(1):4–6.
197. ter Haar, G, Dyson, M, Oakley, S. Ultrasound in physiotherapy in the United Kingdom: results of a questionnaire. Physiother Pract. 1988;4:69–772.
198. Theriault, RL, Biermann, JS, Brown, E. NCCN Task Force Report: Bone health and cancer care. J Natl Compr Canc Net. 2006;4(suppl 2):S1–S20.
199. Thomas, C, Williams, T, Cobos, E. Lung cancer. In: Lenhard R, Osteen R, Gansler T, eds. The American Cancer Society’s clinical oncology. Atlanta: American Cancer Society, 2001.
200. Thun, M. Modifiable risk factors still major cause of cancer deaths worldwide. CA Cancer J Clin. 2006;56(2):64–65.
201. Underwood, W, 3rd., Dunn, RL, Williams, C, et al. Gender and geographic influence on the racial disparity in bladder cancer mortality in the US. J Am Coll Surg. 2006;202(2):284–290.
202. Urquidi, V, Tarin, D, Goodison, S. Role of telomerase in cell senescence and oncogenesis. Annu Rev Med. 2000;51:65–79.
203. Varricchio, CG. American Cancer Society: a cancer source book for nurses, ed 8. Boston: Jones and Bartlett Publishers, 2004.
204. Vasiri, H, Benchimol, S. Alternative pathways for the extension of cellular life span, inactivation of p53/pRb and expression of telomerase. Oncogene. 1999;18:7676–7680.
205. Vitetta, L. Mind-body medicine: stress and its impact on overall health and longevity. Ann NY Acad Sci. 2005;1057:492–505.
206. Watson, T, Mock, V. Exercise and cancer-related fatigue: a review of current literature. Rehabil Oncol. 2003;21(1):23–30.
207. Weihua, Z, Tsan, R, Schroit, AJ, et al. Apoptotic cells initiate endothelial sprouting via electrostatic signaling. Cancer Res. 2005;65(24):11529–11535.
208. Wikipedia: Cancer staging. Available on-line at http://www.en.wikipedia.org/wiki/Cancer_staging Accessed February 9, 2007.
209. Willett, W. Fruit and vegetable intake and risk of major chronic disease. J Natl Cancer Institute. 2004;96(21):1577–1584.
210. Willett, W. Goals for nutrition in the year 2000. CA Cancer J Clin. 1999;49:331–352.
211. Wilson, B, Shannon, M, Stang, C. Nursing drug guide 2001. Upper Saddle River, NJ: Prentice-Hall Health, 2001.
212. Wingo, P, Parkin, D, Eyre, H. Measuring the occurrence of cancer, impact and statistics. In: Lenhard R, Osteen R, Gansler T, eds. The American Cancer Society’s clinical oncology. Atlanta: American Cancer Society, 2001.
213. Winningham, ML. Walking program for people with cancer. Cancer Nurs. 1991;14:270–276.
214. Winningham, ML, MacVicar, MG, Burke, CA. Exercise for cancer patients: guidelines and precautions. Phys Sportsmed. 1986;14:121–134.
215. Women’s Health Initiative (WHI): Findings from the WHI Postmenopausal Hormone Therapy Trials. Available on-line at http://www.nhlbi.nih.gov/whi Accessed May 5, 2006.
216. Woods, J, Lu, Q, Ceddia, MA, et al. Special feature for the Olympics: effects of exercise on the immune system: exercise-induced modulation of macrophage function. Immunol Cell Biol. 2000;78(5):545–553.
217. World Health Organization. Cancer pain and palliative care program. Cancer Pain Release. 1990.
218. Yancik, R, Ries, L. Aging and cancer in America: demographic and epidemiologic perspectives. Hematol Oncol Clin North Am. 2000;14:17–24.
219. Yarbro, C, Frogge, MH, Goodman, M. Cancer symptom management. Sudbury, MA: Jones & Bartlett, 1999.
220. Zhang, SM, Hunter, DJ, Rosner, BA, et al. Intakes of fruits, vegetables, and related nutrients and the risk of non-Hodgkin’s lymphoma among women. Cancer Epidemiol Biomarkers Prev. 2000;9(5):477–485.