MEDICAL MANAGEMENT
The physician must distinguish between the symptoms of arteriosclerotic PVD, such as intermittent claudication and coldness of the feet, and symptoms of venous disease, because occlusive arterial disease usually contraindicates the operative management of varicosities below the knee. When the two conditions coexist, the reduced blood flow caused by the atherosclerosis may even improve the varicosities by reducing blood flow through the veins.
Visual inspection and palpation identify varicose veins of the legs, and Doppler ultrasonography or the duplex scanner is useful in detecting the location of incompetent valves. Endoscopy or radiographic diagnosis identifies esophageal varices, rectal examination or proctoscopy is used to diagnose hemorrhoids, and palpation identifies varicocele (scrotal swelling).
TREATMENT.
Treatment of mild varicose veins is conservative, consisting of periodic daily rest periods with feet elevated slightly above the heart. Client education as to the importance of promoting circulation is stressed, including instructions to make frequent changes in posture, a daily exercise program, and the appropriate use of properly fitting elastic stockings.
When varicosities have progressed past the stage at which conservative care is helpful, surgical intervention and compression sclerotherapy may be considered. In the past, surgical treatment of varicose veins consisted of removing the varicosities and the incompetent perforating veins (ligation and stripping), a procedure sometimes referred to as stripping the veins or miniphlebectomy.
Other procedures for varicose veins have been developed, including radiofrequency (radio waves used to seal off the vein), sclerotherapy (injections of a hardening, or sclerosing, solution; over several months’ time, the injected veins atrophy and blood is channeled into other veins), and laser therapy (noninvasive use of near-infrared wavelengths). Ligation and stripping of the greater saphenous vein prevent its use as a source for future CABGs, motivating researchers to develop effective intervention techniques that salvage large veins.
Oral dietary supplementation has been adopted by some individuals as an addition to traditional management of varicose veins. The loss of vascular integrity associated with the pathogenesis of both hemorrhoids and varicose veins may be aided by several botanical extracts shown to improve microcirculation, capillary flow, and vascular tone while strengthening the connective tissue of the perivascular substrate.204
PROGNOSIS.
Good results with relief of symptoms are usually possible in the majority of cases. Early conservative care for varicose veins during initial stages may help prevent the condition from worsening, but advanced disease may not be prevented from recurring, even with surgical intervention or sclerotherapy. Although surgery for varicose veins can improve appearance, it may not reduce the physical discomfort, suggesting that most lower limb symptoms may have a nonvenous cause. A high mortality is associated with ruptured, bleeding esophageal varices (see Chapter 16).
Chronic Venous Insufficiency
Definition and Incidence.: Chronic venous insufficiency (CVI), also known as postphlebitic syndrome and venous stasis, is defined as inadequate venous return over a long period of time. This condition follows most severe cases of DVT, although it is possible to develop CVI without prior episodes of DVT. CVI may also occur as a result of leg trauma, varicose veins, and neoplastic obstruction of the pelvic veins. The long-term sequelae of CVI may be chronic leg ulcers, accounting for the majority of vascular ulceration; incidence is expected to continue rising with the aging of America.235
Etiologic Factors and Pathogenesis.: CVI occurs when damaged or destroyed valves in the veins result in decreased venous return, thereby increasing venous pressure and producing venous stasis. Without adequate valve function and in the absence of the calf muscle pump, blood flows in the veins bidirectionally, causing high ambulatory venous pressures in the calf veins (venous hypertension). Superficial veins and capillaries dilate in response to the venous hypertension. Red blood cells, proteins, and fluids leak out of the capillaries into interstitial spaces, producing edema and the reddish brown pigmentation characteristic of CVI.
Chronic pooling of blood in the veins of the lower extremities prevents adequate cellular oxygenation and removal of waste products. Any trauma, especially pressure, further lowers the oxygen supply by reducing blood flow into the area. Cell death occurs, and necrotic tissue develops into venous stasis ulcers. The cycle of reduced oxygenation, necrosis, and ulceration prevents damaged tissue from obtaining necessary nutrients, causing delayed healing and persistent ulceration. Poor circulation impairs immune and inflammatory responses, leaving venous stasis ulcers susceptible to infection.
Other contributing factors may include poor nutrition, immobility, and local trauma (past or present). A previous history of burns requiring skin grafts predisposes the individual to venous insufficiency. The area of the graft usually lacks superficial veins, properly functioning capillaries, or both, resulting in blood pooling in these areas. As a result previously burned areas and skin grafts in the lower extremity are susceptible to vascular ulceration.
Clinical Manifestations.: CVI is characterized by progressive edema of the leg; thickening, coarsening, and brownish pigmentation of skin around the ankles; and venous stasis ulceration (see Table 12-20). Venous insufficiency ulcers constitute approximately 80% of all lower extremity ulcers, occurring most often above the medial malleolus where venous hypertension is greatest.
These ulcers characteristically are shallow wounds with a white creamy to fibrous slough over a base of good granulation tissue. They can be very painful with a moderate to large amount of drainage. The wounds typically have irregular borders and are partial to full thickness, often with signs of reepithelialization (e.g., pink or red granulation base). Frequently, moderate to severe edema is present in the limb; in longstanding cases, this edema becomes hardened to a dense, woody texture. The skin of the involved extremity is usually thin, shiny, dry, and cyanotic. Dermatitis and cellulitis may develop later in this condition.
MEDICAL MANAGEMENT
The physician will differentiate between CVI and other causes of edema and ulceration of the lower extremities using client history, clinical examination, and diagnostic tests to rule out or confirm superimposed acute phlebitis. Arterial and venous insufficiency may coexist in the same person.
Treatment goals and techniques are as for varicose veins (increase in venous return, reduction of edema). Conventional methods of compression and rest and elevation (e.g., more frequent periods of leg elevation above the level of the heart are encouraged throughout the day with the foot of the bed elevated 6 inches at night) have been augmented by surgical intervention.
Rapid progress in endovascular procedures with angioplasty and stenting has made it possible for the development of techniques to relieve obstruction and repair reflux in the deep veins. Venous stasis ulcers require ongoing treatment, usually involving the therapist (e.g., primary intervention for edema reduction and topical ulcer and wound care). More detailed information is available.155,181,321,322 Researchers are developing bioengineered skin, a living human dermal replacement for the management of venous ulcers. See the section on Skin Transplantation in Chapter 21.
The prognosis is poor for resolution of CVI, with chronic venous stasis ulcers causing loss of function and progressive disability. Recurrent episodes of acute thrombophlebitis may occur, and noncompliance with the treatment program is common.
Vasomotor Disorders
Vasomotor disorders of the blood vessels causing headaches and reflex sympathetic dystrophy (now classified as complex regional pain syndrome) are discussed in Chapters 37 and 39, respectively.
Raynaud’s Disease and Raynaud’s Phenomenon:
Definition and Overview.: Intermittent episodes of small artery or arteriole constriction of the extremities causing temporary pallor and cyanosis of the digits (fingers more often than toes) and changes in skin temperature are called Raynaud’s phenomenon. These episodes occur in response to cold temperature or strong emotion, such as anxiety or excitement. When this condition is a primary vasospastic disorder it is called (idiopathic) Raynaud’s disease. If the disorder is secondary to another disease or underlying cause, the term Raynaud’s phenomenon is used.
Incidence and Etiologic Factors:
RAYNAUD’S DISEASE.: Eighty percent of persons with Raynaud’s disease are women between the ages of 20 and 49 years. The exact etiology of Raynaud’s disease remains unknown, but it appears to be caused by hypersensitivity of digital arteries to cold, release of serotonin, and genetic susceptibility to vasospasm. Raynaud’s disease accounts for 65% of all people affected by this condition. Raynaud’s disease is usually experienced as more annoying than medically serious.
RAYNAUD’S PHENOMENON.: Epidemiologists estimate that Raynaud’s phenomenon is a problem for 10% to 20% of the general population; it affects women 20 times more frequently than men, usually between the ages of 15 and 40 years. Risk factors for Raynaud’s phenomenon are different between men and women. The Framingham Offspring Study reports that age and smoking are associated with Raynaud’s phenomenon in men only, whereas an association with marital status and alcohol use was observed in women only. These findings suggest that different mechanisms influence the expression of Raynaud’s phenomenon in men and women.112
Raynaud’s phenomenon as a condition secondary to another disease is often associated with Buerger’s disease or connective tissue disorders (collagen vascular diseases), such as Sjögren’s syndrome, scleroderma, polymyositis and dermatomyositis, mixed connective tissue disease, SLE, and rheumatoid arthritis (see Box 12-17). Raynaud’s phenomenon can be a sign of occult (hidden) neoplasm, especially suspected when it presents unilaterally.
Raynaud’s phenomenon may also occur with change in temperature, such as occurs when going from a warm outside environment to an air-conditioned room. In addition, Raynaud’s phenomenon may be associated with occlusive arterial diseases and neurogenic lesions, such as thoracic outlet syndrome, or with the effects of long-term exposure to cold (occupational or frostbite), trauma, or use of vibrating equipment such as jackhammers. Injuries to the small vessels of the hands may produce Raynaud’s phenomenon. The trauma can be a result of repetitive stress that comes from using crutches for extended periods, typing on a computer keyboard, or even playing the piano.
Several medications (e.g., β-blockers, ergot alkaloids prescribed for migraine headaches, antineoplastics used in chemotherapy) have also been implicated. Because nicotine causes small blood vessels to constrict, smoking can trigger attacks in persons who are predisposed to this phenomenon.
Pathogenesis and Clinical Manifestations.: Scientists theorize that Raynaud’s phenomenon is associated with a disturbance in the control of vascular reflexes. Although the causes differ for Raynaud’s disease and Raynaud’s phenomenon, the clinical manifestations are the same, based on a pathogenesis of arterial vasospasm in the skin.
It begins with the release of chemical messengers, which cause blood vessels to constrict and remain constricted. The flow of oxygenated blood to these areas is reduced, and the skin becomes pale and cold. The blood in the constricted vessels, which has released its oxygen to the tissues surrounding the vessels, pools in the tissues, producing a bluish or purplish color.
In the case of fibromyalgia-associated Raynaud’s phenomenon, symptoms may be the result of cold-induced spasms of the arteries caused by a problem in the autonomic nervous system control of the blood supply to the extremities. Altered or reduced numbers of α2-adrenergic receptors on the platelets correlate with Raynaud’s phenomenon in fibromyalgia-associated Raynaud’s.35 These receptors are involved in the functioning of the autonomic nervous system. This could explain why the cold-induced pain is significant but without skin color changes in this population.
In most cases, the skin color progresses from blue to white to red. First, ischemia from vasospastic attacks causes cyanosis, numbness, and the sensation of cold in the digits (thumbs usually remain unaffected). The affected tissues become numb or painful. For unknown reasons, the flow of chemical that triggered the process eventually stops. The vessels relax, and blood flow is restored. The skin becomes white (characterized by pallor) and then red (characterized by rubor) as the vasospasm subsides and the capillaries become engorged with oxygenated blood. Oxygen-rich blood returns to the area, and as it does so, the skin becomes warm and flushed. The person may experience throbbing, paresthesia, and slight swelling as this occurs.
Sensory changes, such as numbness, stiffness, diminished sensation, and aching pain, often accompany vasomotor manifestations. Initially, no abnormal findings are present between attacks, but over time, frequent, prolonged episodes of vasospasm causing ischemia interfere with cellular metabolism, causing the skin of the fingertips to thicken and the fingernails to become brittle.
In severe, chronic Raynaud’s phenomenon, the underlying condition may have produced scars in the vessels, reducing the vessel diameter and therefore blood flow. When attacks occur, they are often more severe, resulting in prolonged loss of blood to fingers and toes, which can produce painful skin ulcers; rarely, gangrene may develop. Episodes of Raynaud’s disease are often bilateral, progressing distally to proximally along the digits. Raynaud’s phenomenon may be unilateral, involving only one or two fingers, but this clinical presentation warrants a physician’s differential diagnosis, since it can be associated with cancer (Fig. 12-35).
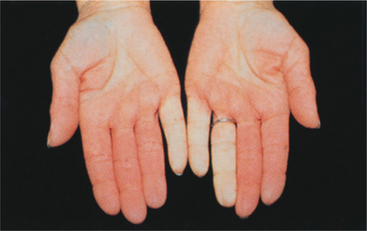
Figure 12-35 Raynaud’s disease or phenomenon. White color (pallor) from arteriospasm and resulting deficit in blood supply may initially involve only one or two fingers, as shown here. Cold and numbness or pain may accompany the pallor or cyanosis stage. Subsequent episodes may involve the entire finger and may include all the fingers. Toes are affected in 40% of cases. (From Jarvis C: Physical examination and health assessment, ed 4, Philadelphia, 2004, Saunders.)
MEDICAL MANAGEMENT
Diagnosis is usually made by clinical presentation and past medical history. Raynaud’s disease is diagnosed by a history of symptoms for at least 2 years with no progression and no evidence of underlying cause. Raynaud’s disease must be differentiated from the numerous possible disorders associated with Raynaud’s phenomenon. Untreated and uncontrolled Raynaud’s may damage or destroy the affected digits. Rarely, necrosis, ulceration, and gangrene result. Even with intervention, the person with Raynaud’s disease or phenomenon may experience disability and loss of function.
PREVENTION AND TREATMENT.
Treatment for Raynaud’s disease is limited to prevention or alleviation of the vasospasm because no underlying cause or condition has been discovered, although pharmacologic agents for primary and secondary Raynaud’s phenomenon are under investigation. Clients are encouraged to avoid stimuli that trigger attacks, such as cool or cold temperatures, changes in temperature, and emotional stress, and to eliminate use of nicotine, which has a constricting effect on blood vessels.
Physical or occupational therapy is often prescribed and should include client education about managing symptoms through protective skin care and cold protection (see Box 12-14), biofeedback, stress management and relaxation techniques, whirlpool or other gentle heat modalities, and exercise. Large movement arm circles in a windmill fashion can restore circulation in some people. The individual will have to experiment with the speed at which to move the arms; some people benefit from slow, gentle movement, whereas others find greater success with fast rotations.
Calcium channel blockers are the treatment of choice. Pharmacologic management may also include nonaddicting analgesics for pain. Therapists can be instrumental in teaching physiologic modulation starting with hand warming. A hand-held device to measure fingertip temperature combined with self-guided or audio-guided relaxation can be very effective and is available.156
When conservative care fails to relieve symptoms and the condition progresses clinically, sympathetic blocks followed by intensive therapy may be helpful. Sympathectomy may be necessary for persons who only temporarily benefit from the sympathetic blocks.
Treatment for Raynaud’s phenomenon consists of appropriate treatment for the underlying condition or removing the stimulus causing vasospasm. The clinical care described for Raynaud’s disease may also be of benefit. In addition, the use of antioxidants as an effective treatment of Raynaud’s phenomenon as well as the role of therapeutic angiogenesis (regeneration of vessels) remains under investigation.
Vascular Neoplasms
Malignant vascular (i.e., involving the blood vessels) neoplasms are extremely rare and include angiosarcoma, hemangiopericytoma, and Kaposi’s sarcoma. Angiosarcomas (hemangiosarcomas) can occur in either gender and at any age, most commonly appearing as small, painless, red nodules on the skin, soft tissue, breast, bone, liver, and spleen. Almost one half of all people with angiosarcoma die of the disease.
Hemangiopericytoma arises from the smooth muscle cells that are external to the walls of capillaries and arterioles. Most commonly located on the lower extremities and retroperitoneum (space between the peritoneum lining the walls of the abdominal and pelvic cavities and the posterior abdominal wall), these tumors are composed of spindle cells with a rich vascular network. Metastasis to the lungs, bone, liver, and lymph nodes occurs in 10% to 50% of cases, but the majority of hemangiopericytomas are removed surgically without having invaded or metastasized.
Kaposi’s sarcoma in association with AIDS most likely occurs as a result of loss of immunity. One form of the tumor resembles a simple hemangioma with tightly packed clusters of capillaries, most often visible on the skin. Although Kaposi’s sarcoma is malignant and may be widespread in the body, it is not usually a cause of death.
Arteriovenous Malformations
Arteriovenous malformations (AVMs) are congenital vascular malformations of the cerebral vasculature. AVMs are the result of localized maldevelopment of part of the primitive vascular plexus consisting of abnormal arteriovenous communications without intervening capillaries. There is a central tangled mass of fragile, abnormal blood vessels called the nidus that shunts blood from cerebral feeding arteries directly into cerebral veins. The loss of the normal capillary network between the high-pressure arterial system and the low-pressure venous system results in a faster flow and elevated pressure within the delicate vessels of the AVM. The lack of a gradient pressure system predisposes the lesion to rupture.
AVMs vary in size, ranging from massive lesions that are fed by multiple vessels to lesions too small to identify. Perfusion to adjacent brain tissue may be impaired because blood flow is diverted to the AVM, a phenomenon referred to as vascular stealing. AVMs may occur in any blood vessel, but the most common sites include the brain, GI tract, and skin. Approximately 10% of cases present with aneurysms. Small AVMs are more likely to bleed than large ones, and once bleeding occurs, repeated episodes are likely.
Clinical presentation depends on the location of the malformation and may relate to hemorrhage from the malformation or an associated aneurysm or to cerebral ischemia caused by diversion or stasis of blood. Seizures, migrainelike headaches unresponsive to standard therapy, and progressive neurologic deficits may develop.
Diagnostic testing and planned intervention rely on cerebral angiography to show the AVM size, location, feeding vessels, nidus, and venous outflow vessels. Other tests may include MRI, x-rays, ultrasound, electroencephalogram (EEG), and arteriogram. Treatment options are individualized depending on the size and location of the lesion as well as any other surgical risks present.
In the last 15 years, endovascular embolization and stereotactic radiosurgery (delivery of extremely precise doses of radiation to destroy abnormal blood vessels) have increased survival outcomes, especially for lesions previously considered inoperable or in cases of high surgical risk factors. Prognosis is guarded, since there is a 2% to 4% chance of hemorrhage with the concomitant risk of permanent neurologic deficit or even death.
The Lymphatic Vessels
The lymphatic system (see also the section on The Lymphatic System in Chapter 13) is part of the circulatory system that collects excess tissue fluid and plasma that has leaked out of capillaries into the interstitial space and returns it to the bloodstream.
The lymphatic system consists of lymphatic vessels and lymph nodes and functions to remove impurities from the circulatory system and to produce cells of the immune system (lymphocytes) that are vital in fighting bacteria and viruses. The lymph nodes are also part of the lymphoid system, the organs and tissues of the immune system. All the lymphoid organs link the hematologic and immune systems in that they are sites of residence, proliferation, differentiation, or function of lymphocytes and mononuclear phagocytes (mononuclear phagocyte system).
Disorders of the lymphatic system may result from inflammation of a lymphatic vessel (lymphangitis), inflammation of one or more lymph nodes (lymphadenitis), an increased amount of lymph (lymphedema), or enlargement of the lymph nodes (lymphadenopathy). There are also three forms of lymph vascular insufficiency that can occur.
The first, dynamic insufficiency, occurs when the lymphatic load exceeds the lymphatic transport capacity. In this situation, the anatomy of the lymphatic system and its function are normal but are overwhelmed. A second form of insufficiency of the lymph vascular system is caused by a reduction of the lymphatic transport capacity below the level of a normal lymphatic protein load. This reduction results in low lymph flow failure called mechanical insufficiency.
A third form of lymph vascular insufficiency occurs when the lymphatic system has a reduced transport capacity, leading to an overflowing of lymph. This form is called safety valve insufficiency. For a complete discussion of the lymphatic system, see Chapter 13.
OTHER CARDIAC CONSIDERATIONS
Despite the success of new immunosuppressive regimens and better results with transplantation, few people who are dying of heart failure will actually have the opportunity to receive a heart transplant. The mechanical technology may eventually allow selected persons to receive long-term (permanent) support as a substitute for cardiac transplantation. The recipient is often home in 6 weeks with follow-up home health care. In the future, studies may be done to determine the efficacy of removing the cardiac assistive device after prolonged heart rest provides cardiac recovery. Survival with the natural recovered heart may be possible in some people.
Researchers are actively pursuing tissue engineering to replace transplantation and mechanical devices (e.g., artificial heart, implanted cardiac assistive devices or other bridges to transplantation) to help keep people alive while they await heart transplant or as a replacement intervention for transplantation.
The Cardiac Client and Surgery
Persons with previously diagnosed cardiac disease undergoing general or orthopedic surgery are at risk for additional postoperative complications. Anesthesia and surgery are often associated with marked fluctuations of heart rate and blood pressure, changes in intravascular volume, myocardial ischemia or depression, arrhythmias, decreased oxygenation, and increased sympathetic nervous system activity. In addition, changes in medications, surgical trauma, wound healing, infection, hemorrhage, and pulmonary insufficiency may overwhelm the diseased heart. All these factors place an additional stress on the cardiac client during the perioperative period.
Cardiac surgery via median sternotomy requires a longitudinal incision and disruption of the sternum. During the operative procedure, the bone is rewired with stainless steel wire and fixed with low separation strength and security of closure approximately 5% of normal (this increases to 90% of normal strength at 6 weeks for most people).214
A single-lung transplantation requires a posterolateral thoracotomy, whereas a double-lung transplant requires bilateral anterior thoracotomies referred to as a “clam shell.” In this latter procedure, the rib cage and sternum are lifted perpendicularly as the hood of a car would be lifted. The heart-lung procedure is still done by open sternotomy.
Complications following a sternotomy include mediastinitis, poor wound healing, chronic pain, posttraumatic stress disorder, and, more rarely, brachial plexus injury. Risk factors for these complications may include obesity, osteoporosis, diabetes or other comorbidities, large breasts in women (the weight of both breasts puts additional traction on sutures), and client noncompliance or poor compliance.
Development of a less invasive means of performing cardiac surgery may be possible with recent advances in technology, especially videoscopic visualization and the ability to provide myocardial protection. Surgeons are examining alternate techniques in hopes of reducing operative stress, postoperative pain, and postoperative recovery time.
New procedures are being developed that eliminate the use of a sternotomy, such as the minithoracotomy or “keyhole” thoracotomy via a small incision that allows surgeons to operate on a beating heart. These alternate surgical techniques involve passing instruments through small incisions in the skin and muscle and between the ribs. Surgeons can suture bypass vessels around blocked coronary arteries without shutting down the heart and rerouting the blood through a bypass machine.
Cardiogenic Shock
Shock is acute, severe circulatory failure associated with a variety of precipitating conditions. Regardless of the cause, shock is associated with marked reduction of blood flow to vital organs, eventually leading to cellular damage and death. See Table 14-1 for categories and causes of shock.
The therapist may see a client in one of three stages of shock. Stage 1, compensated hypotension, is characterized by reduced cardiac output that stimulates compensatory mechanisms that alter myocardial function and peripheral resistance. During this stage, the body tries to maintain circulation to vital organs such as the brain and the heart and clinical symptoms are minimal. Blood pressure may remain normotensive.
In stage 2, compensatory mechanisms for dealing with the low delivery of nutrients to the body are overwhelmed, and tissue perfusion is decreased. Early signs of cerebral, renal, and myocardial insufficiency are present. Cardiogenic shock (inadequate cardiac function) may result from disorders of the heart muscle, valves, or electrical pacing system. Shock associated with MI or other serious cardiac disease carries a high mortality rate. The therapist is only likely to see this type of client in a CCU setting.
Stage 3 is characterized by severe ischemia with damage to tissues by toxins and antigen-antibody reactions. The kidneys, liver, and lungs are especially susceptible; ischemia of the GI tract allows invasion by bacteria with subsequent infection.
Clinical manifestations of shock may include (in early stages) tachycardia, increased respiratory rate, and distended neck veins. In early septic shock (vascular shock caused by infection), there is hyperdynamic change with increased circulation, so that the skin is warm and flushed and the pulse is bounding rather than weak.
In the second phase of shock (late septic shock) hypoperfusion (reduced blood flow) occurs with cold skin and weak pulses, hypotension (systolic blood pressure of 90 mm Hg or less), mottled extremities with weak or absent peripheral pulses, and collapsed neck veins. This phase is usually irreversible; the client is unresponsive, and cardiovascular collapse eventually occurs. The therapist should be aware that some healthy adults may have blood pressure levels this low without ill effects or with only minor symptoms of orthostatic hypotension when changing positions quickly.
Treatment is directed toward both the manifestations of shock and its cause.
The Cardiac Client and Pregnancy
Normal physiologic changes during pregnancy can exacerbate symptoms of underlying cardiac disease, even in previously asymptomatic individuals. The most common cardiovascular complications of pregnancy are peripartum cardiomyopathy, aortic dissection, and pregnancy-related hypertension.
Peripartum cardiomyopathy or cardiomyopathy of pregnancy is discussed briefly earlier in the chapter (see the section on Cardiomyopathy). Pregnancy predisposes to aortic dissection, possibly because of the accompanying connective tissue changes. Dissection usually occurs near term or shortly postpartum in the arteries (including coronary arteries) or the aorta, and special implications are the same as for aneurysm.
The Heart in Collagen Vascular Diseases
Collagen vascular diseases (now more commonly referred to as diffuse connective tissue disease) (Box 12-17) often involve the heart, although cardiac symptoms are usually less prominent than other manifestations of the disease.
Lupus Carditis
SLE is a multisystem clinical illness (see Chapter 7) characterized by an inflammatory process that can target all parts of the heart, including the coronary arteries, pericardium, myocardium, endocardium, conducting system, and valves. Lupus cardiac involvement may include pericarditis, myocarditis, endocarditis, or a combination of the three. Cardiac disease can occur as a direct result of the autoimmune process responsible for SLE or secondary to hypertension, renal failure, hypercholesterolemia (excess serum cholesterol), drug therapy for SLE, and, more rarely, infection (infective carditis).
Pericarditis is the most frequent cardiac lesion associated with SLE, presenting with the characteristic substernal chest pain that varies with posture, becoming worse in recumbency and improving with sitting or bending forward. In some people, pericarditis may be the first manifestation of SLE.
Myocarditis (see also the section on Myocardial Disease) is a serious complication reported to occur in less than 10% of people with SLE. The simultaneous involvement of cardiac and skeletal muscle may occur more commonly than previously suspected. More sensitive diagnostic techniques now make early detection of occult myocarditis possible. Myocarditis in association with SLE occurs most often as left ventricular dysfunction and conduction abnormalities with varying degrees of heart block.
Lupus endocarditis occurs in up to 30% of persons affected by SLE. Major lesions associated with lupus endocarditis include the formation of multiple noninfectious wartlike elevations (verrucae) around or on the surface of the cardiac valves, most commonly the mitral and tricuspid valves. Other types of valvular disease associated with SLE include mitral and aortic regurgitation or stenosis.
Rheumatoid Arthritis
On rare occasions, the heart is involved as a part of rheumatoid arthritis, a chronic, systemic, inflammatory disorder that can affect various organs but predominantly involves synovial tissues of joints (see Chapter 27). When the heart is affected, rheumatoid granulomatous inflammation with fibrinoid necrosis may occur in the pericardium, myocardium, or valves. Involvement of the heart in rheumatoid arthritis does not compromise cardiac function.
Ankylosing Spondylitis
Ankylosing spondylitis is a chronic, progressive inflammatory disorder affecting fibrous tissue primarily in the sacroiliac joints, spine, and large peripheral joints (see Chapter 27). A characteristic aortic valve lesion develops in as many as 10% of persons with longstanding ankylosing spondylitis. The aortic valve ring is dilated, and the valve cusps are scarred and shortened. The functional consequence is aortic regurgitation (see the section on Aortic Regurgitation [Insufficiency]).
Scleroderma
Scleroderma or systemic sclerosis is a rheumatic disease of the connective tissue characterized by hardening of the connective tissue (see Chapter 10). Involvement of the heart in persons with scleroderma is second only to renal disease as a cause of death in scleroderma. The myocardium exhibits intimal sclerosis (hardening) of small arteries, which leads to small infarctions and patchy fibrosis. As a result, CHF and arrhythmia are common. Cor pulmonale may occur secondary to interstitial fibrosis of the lungs, and hypertensive heart disease may occur as a result of renal involvement.
Polyarteritis Nodosa
Polyarteritis refers to a condition of multiple sites of inflammatory and destructive lesions in the arterial system; the lesions consist of small masses of tissue in the form of nodes or projections (nodosum) (see previous discussion in this chapter). The heart is involved in up to 75% of cases of polyarteritis nodosa. The necrotizing lesions of branches of the coronary arteries result in MI, arrhythmias, or heart block. Cardiac hypertrophy and failure secondary to renal vascular hypertension occur.
Cardiac Complications of Cancer and Cancer Treatment227
Many treatments for cancers are also known to be cardiotoxic. People with cancer experience all the usual cardiac problems that occur in the general population in addition to complications of cancer and its therapy. Tumor masses can cause compression of the heart and great vessels resulting in pericardial effusions and tamponade. Certain tumors can cause arrhythmias and may secrete mediators that are directly toxic to the heart. Pericardial effusions and tamponade can follow surgery, radiation, or chemotherapy.
Cardiac toxicity may occur following chest irradiation, especially when combined with the administration of many chemotherapeutic agents. Chest radiation for any type of cancer (e.g., Hodgkin’s disease, non-Hodgkin’s lymphoma, esophageal cancer, lung cancer, breast cancer) exposes the heart (and lungs) to varying degrees and doses of radiation. Previous mediastinal radiation and increasing cumulative doses of chemotherapy or irradiation are known risk factors for the development of cardiotoxicity.
Radiation exposure can cause considerable scarring within the subendocardial adipose tissue, endocardial thickening, and interstitial fibrosis.284 Collectively the latter three defects would make the heart less capable of expanding during systole. Pericardial effusion is the most common manifestation of radiation heart disease, but coronary arteries are known to become fibrotic and undergo luminal narrowing, resulting in hypertension, angina, and MI.
Doxorubicin (Adriamycin) was identified as being cardiotoxic during the early drug trials in the 1970s, but it took 5 to 6 years of actual use for the full extent of cardiac damage to become obvious. Today, cumulative doses of doxorubicin are limited to approximately 550 mg/m2 because the drug can cause fatal CHF in doses above this amount. Often these effects are not seen until years or decades after treatment with the drug has been completed. Many other chemotherapeutic agents are cardiotoxic but not to the extent of doxorubicin, and these effects tend be more acute than chronic.366
Chemotherapy agents may prompt acute and chronic heart failure (e.g., anthracycline antibiotics, mitoxantrone, doxorubicin combined with paclitaxel used in the treatment of breast cancer)262 or coronary spasm leading to angina, MI, arrhythmias, or sudden death (e.g., 5-fluorouracil). Anthracycline effects on the heart reduce exercise tolerance. Endocarditis also occurs in cancer clients with vascular access devices and immune compromise.
Recombinant technology has resulted in the development of biologic response modifiers, including the interferons, interleukins, and tumor necrosis factor, which also have some adverse cardiovascular effects. Hypotension and tachycardia are the most common problems, although there have been some reports of myocardial ischemia and infarction. These adverse effects appear to be caused by significant alterations in fluid balance rather than any dysrhythmic or cardiotoxic properties of the drugs. Fortunately, many of the cardiac complications associated with chemotherapeutic agents and biologic response modifiers are transient and reversible.150
The most common manifestations of cardiotoxicity are cardiac arrhythmias or acute or chronic pericarditis. Other cardiac problems that may develop include blood pressure changes, thrombosis, ECG changes, myocardial fibrosis with a resultant restrictive cardiomyopathy, conduction disturbances, CHF, accelerated and radiation-induced CAD, and valvular dysfunction. These may occur during or shortly after treatment or within days or weeks after treatment; or they may not be apparent until months and sometimes years after completion of chemotherapy.251
Although only a small percentage of persons develop serious problems or obvious symptoms of cardiotoxicity, many people have functional limitations that are not clinically apparent because they are physically inactive. A number of risk factors may predispose someone to cardiotoxicity, including total daily dose, increasing cumulative dose, schedule of administration, concurrent administration of cardiotoxic agents, prior chemotherapy, mediastinal radiation, age (younger than 18 years or older than 70 years), female gender, history of preexisting cardiovascular disorders or other comorbidities such as diabetes, and presence of electrolyte imbalances (e.g., hypokalemia, hypomagnesemia).251
References
1. Abela, GS, Aziz, K. Cholesterol crystals rupture biological membranes and human plaques during acute cardiovascular events a novel insight into plaque rupture by scanning electron microscopy. Scanning. 2006;28(1):1–10.
2. Abidov, A, Rozanski, A, Hachamovitch, R, et al. Prognostic significance of dyspnea in patients referred for cardiac stress testing. N Engl J Med. 2005;353(18):1889–1898.
3. Abou-Agag, LH, Aikens, ML, Tabengwa, EM, et al. Polyphenols increase t-PA and u-PA gene transcription in cultured human endothelial cells. Alcohol Clin Exp Res. 2001;25(2):155–162.
4. Agency for Health Care Policy and Research Cardiac rehabilitation. Columbia, MD: The Agency; 1995. Clinical guideline no. 17, Copies are available for $8.00 from the U.S. Government Printing Office, (202) 512-1800. Order stock no. 01702600154-9. Also available on-line at http://text.nlm.nih.gov/
5. Albert, CM, Mittleman, MA, Chae, CU, et al. Triggering of sudden death from cardiac causes by vigorous exertion. N Engl J Med. 2000;343(19):1409–1411.
6. Albert, MA, Ridker, PM. The role of C-reactive protein in cardiovascular disease risk. Curr Cardiol Rep. 2000;1(2):99–104.
7. Aldrich, D, Hunt, D. When can the patient with deep venous thrombosis begin to ambulate. Phys Ther. 2004;84(3):268–273.
8. American College of Cardiology/American Heart Association (ACC/AHA). 2005 Practice guidelines for the management of patients with peripheral arterial disease (lower extremity, renal, mesenteric, and abdominal aortic): a collaborative report from the American Association for Vascular Surgery/Society for Vascular Surgery, Society for Cardiovascular Angiography and Interventions, Society for Vascular Medicine and Biology, Society of Interventional Radiology, and the ACC/AHA Task Force on Practice Guidelines. Circulation. 2006;113:463–654.
9. American College of Sports Medicine (ACSM). Guidelines for exercise testing and prescription, ed 7. Baltimore: Williams & Wilkins, 2005.
10. American College of Sports Medicine (ACSM). Position stand: the recommended quantity and quality of exercise for developing and maintaining cardiorespiratory and muscular fitness, and flexibility in healthy adults. Med Sci Sports Exerc. 1998;30(6):975–991.
11. American College of Sports Medicine (ACSM). Resource manual for guidelines for exercise testing and prescription, ed 5. Baltimore: ACSM, 2005.
12. American Heart Association (AHA) 2001 Heart and stroke statistical update. Dallas: The Association; 2000. Available on-line at http://www.americanheart.org/statistics/index.html Accessed April 25, 2008.
13. American Heart Association (AHA): Angioplasty and cardiac revascularization statistics (2007). Available on-line at http://www.americanheart.org/presenter.jhtml?identifier=4439 Accessed May 30, 2007.
14. American Heart Association (AHA) Coronary heart disease and stroke remain the leading causes of death of women in America. Dallas: The Association; 2000. Available on-line at www.americanheart.org Accessed April 25, 2008.
15. American Heart Association (AHA): Mitral valve prolapse. Available on-line at http://www.americanheart.org/presenter.jhtml?identifier=7273 Accessed May 31, 2007.
16. American Heart Association (AHA) Statement on exercise: benefits and recommendations for physical activity programs for all Americans. Dallas: The Association; 2000. Available on-line at http://www.americanheart.org/Scientific/statements/ Accessed April 25, 2008.
17. American Heart Association (AHA) Women, heart disease, and stroke statistics. Dallas: The Association; 2000. Available on-line at http://www.americanheart.org Accessed April 25, 2008.
18. American Physical Therapy Association (APTA): House of Delegates policy no. 06-80-19-55, program 32, 2001. Available on-line at http://www.apta.org Accessed April 25, 2008.
19. Anand, SS, Wells, PS, Hunt, D, et al. Does this patient have a DVT? JAMA. 1998;279(14):1094–1099.
20. Anand, SS, Wells, PS, Hunt, D, et al. Does this patient have a DVT? JAMA. 1998;279(14):1094–1099.
21. Anderson, DR. Combined use of clinical assessment and d-dimer to improve the management of patients presenting to the emergency department with suspected deep vein thrombosis. J Thromb Haemost. 2003;1(4):645–651.
22. Anderson, DR. Thrombosis in the emergency department: use of a clinical diagnosis model to safely avoid the need for urgent radiological investigation. Arch Intern Med. 1999;159(5):477–482.
23. Andraws, R. Effects of antibiotic therapy on outcomes of patients with coronary artery disease: a meta-analysis of randomized controlled trials. JAMA. 2005;293(21):2641–2647.
24. Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT) Officers and Coordinators for the ALLHAT Collaborative Research Group. Major outcomes in high-risk hypertensive patients randomized to angiotensin-converting enzyme inhibitor or calcium channel blocker vs diuretic. JAMA. 2002;288:2981–2997.
25. Antman, EM. Use of nonsteroidal antiinflammatory drugs: an update for clinicians: a scientific statement from the American Heart Association. Circulation. 2007;115(12):1634–1642.
26. Aram, V. The Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure. JAMA. 2003;289:2560–2571.
27. Aspinall, W. Clinical testing for the craniovertebral hypermobility syndrome. J Orthop Sports Phys Ther. 1989;12:180–181.
28. Autar, R. Calculating patients’ risk of deep vein thrombosis. Br J Nurs. 1998;7(1):7–12.
29. Autar, R. Nursing assessment of clients at risk of deep vein thrombosis (DVT): the Autar DVT scale. J Adv Nurs. 1996;23(4):763–770.
30. Bahcelioglu, M. The diagonal ear-lobe crease: a sign of some diseases. Saudi Med J. 2005;26(6):947–951.
31. Bairey, M. Insights from the NHLBI-Sponsored Women’s Ischemia Syndrome Evaluation (WISE) study: Part II: gender differences in presentation, diagnosis, and outcome with regard to gender-based pathophysiology of atherosclerosis and macrovascular and microvascular coronary disease. J Am Coll Cardiol. 2006;47(3 suppl):S21–29.
32. Barsky, AJ. Palpitations and cardiac awareness after heart transplantation. Psychosom Med. 1998;60(5):557–562.
33. Beckman, JA. The United States preventive services task force recommendation statement on screening for peripheral arterial disease: more harm than benefit? Circulation. 2006;114(8):861–866.
34. Bellew, JW. Geriatric fitness: effects of aging and recommendations for exercise in older adults. Cardiopulm Phys Ther J. 2005.
35. Bennett, R, FMS and Raynaud’s phenomenon. Fibromyalgia Network Newsletter 1996. Available from P.O. Box 31750, Tucson, AZ 85751-1750. Phone: (800) 853-2929. Available on-line at www.fmnetnews.com Accessed April 25, 2008.
36. Benton, RE, Sale, M, Flockhart, DA, et al. Greater quinidine-induced QTc interval prolongation in women. Clin Pharmacol. 2000;67(4):413–418.
37. Bergholm, R, Makimattila, S, Valkonen, M, et al. Intense physical training decreases circulating antioxidants and endothelium-dependent vasodilation in vivo. Atherosclerosis. 1999;145:341–349.
38. Blackwood, AM, Sagnella, GA, Cook, DG, et al. Urinary calcium excretion, sodium intake and blood pressure in a multi-ethnic population: results of the Wandsworth Heart and Stroke Study. J Hum Hypertens. 2001;15(4):229–237.
39. Blankstein, R. Female gender is an independent predictor of operative mortality after coronary artery bypass graft surgery: contemporary analysis of 31 Midwestern hospitals. Circulation. 2005;112(9 suppl):I323–I327.
40. Blattler, W. Leg compression and ambulation is better than bed rest for the treatment of acute deep venous thrombosis. Int Angiol. 2003;22(4):393–400.
41. Bleasdale-Barr, KM, Mathias, CJ. Neck and other muscle pains in autonomic failure: their association with orthostatic hypotension. J R Soc Med. 1998;91(7):344–359.
42. Booth, GL. Relation between age and cardiovascular disease in men and women with diabetes compared with non-diabetic people: a population-based retrospective cohort study. Lancet. 2006;368(9529):29–36.
43. Braith, RW. Exercise training in patients with CHF and heart transplant recipients. Med Sci Sports Exerc. 1998;30(10):S367–S378.
44. Braith, RW, Vincent, KR. Resistance exercise in the elderly person with cardiovascular disease. Am J Geriatr Cardiol. 1999;8(2):63–79.
45. Braith, RW, Welsch, MA, Feigenbaum, MS, et al. Neuroendocrine activation in heart failure is modified by endurance exercise training. J Am Coll Cardiol. 1999;34(4):1170–1175.
46. Brandsma, JW, Robeer, BG, van den Heuvel, S, et al. The effect of exercises on walking distance of patients with intermittent claudication: a study of randomized clinical trials. Phys Ther. 1998;78(3):278–288.
47. Braun, S, Schrotter, H, Reynen, K, et al. Myocardial infarction as complication of left atrial myxoma. Int J Cardiol. 2005;101(1):115–121.
48. Braunwald E, Zipes DP, Libby P, eds. Heart disease: a textbook of cardiovascular medicine, ed 6, Philadelphia: Saunders, 2001.
49. Brendle, DC, Joseph, LJ, Corretti, MC, et al. Effects of exercise rehabilitation on endothelial reactivity in older patients with peripheral arterial disease. Am J Cardiol. 2001;87(3):324–329.
50. Brickner, ME, Hillis, LD, Lange, RA. Congenital heart disease in adults. I. N Engl J Med. 2000;342(4):256–263.
51. Brickner, ME, Hillis, LD, Lange, RA. Congenital heart disease in adults. II. N Engl J Med. 2000;342(5):334–342.
52. Brooks G: Physiologic monitoring of patients during exercise. Presentation at Combined Sections Pre-conference Seminar, New Orleans, February 2, 2000.
53. Brown, S, Norris, J, Kraus, W, et al. Effects of moderate exercise training in the absence of weight loss on cardiovascular risk factors in mildly obese subjects. Clin Exerc Physiol. 2000;2(1):25–31.
54. Brown, WV. The benefit of aggressive lipid lowering. Atherosclerosis. 2000;1(2):15–19.
55. Burkman, RT. Oral contraceptives: current status. Clin Obstet Gynecol. 2001;44(1):62–72.
56. Butany, J, Nari, V, Naseenuddin, A, et al. Cardiac tumours: diagnosis and management. Lancet Oncol. 2005;6(4):219–228.
57. Cahalin, LP. Cardiac muscle dysfunction. In: Hillegass E, Sadowsky S, eds. Essentials of cardiopulmonary physical therapy. ed 2. Philadelphia: Saunders; 2001:106–181.
58. Cahalin, LP. Exercise training in heart failure: inpatient and outpatient considerations. AACN Clin Issues. 1998;9(2):225–243.
59. Cahalin, LP. Heart failure. Phys Ther. 1996;76(5):516–533.
60. Cahalin, LP, Review of the effects of resistance training in patients with chronic heart failure: potential effects upon the muscle hypothesis. Cardiopulm Phys Ther J 2006. Available on-line at http://findarticles.com/p/articles/mi_qa3953. Accessed May 31, 2007.
61. Campbell, CL. Aspirin dose for the prevention of cardiovascular disease: a systematic review. JAMA. 2007;297(18):2018–2024.
62. Cappuccio, FP, Kalaitzidis, R, Duneclift, S, et al. Unraveling the links between calcium excretion, salt intake, hypertension, kidney stones, and bone metabolism. J Nephrol. 2000;13(3):169–177.
63. Carabello, PJ, Heit, JA, Atkinson, EJ, et al. Long-term use of oral anti-coagulants and the risk of fracture. Arch Intern Med. 1999;159:1750–1756.
64. Cariski, AT. Cilostazol: a novel treatment option in intermittent claudication. Int J Clin Pract Suppl. 2001;119:11–18.
65. Carpenter, A. Valvular heart disease in women: the surgical perspective. J Thorac Cardiovasc Surg. 2004;127(1):4–6.
66. Carr, DB. Intra-abdominal fat is a major determinant of the National Cholesterol Education Program Adult Treatment Panel III criteria for the metabolic syndrome. Diabetes. 2004;53:2087–2094.
67. Cassady, SL. Peripheral arterial disease: a review of epidemiology, clinical presentation, and effectiveness of exercise training. Cardiopulm Phys Ther J. 2004.
68. Cassar, K. Intermittent claudication. BMJ. 2006;333(7576):1002–1005.
69. Centers for Disease Control and Prevention (CDC): Health statistics, 2000. Available on-line at www.cdc.gov Accessed April 25, 2008.
70. Chae, CU, Pfeffer, MA, Glynn, RJ, et al. Increased pulse pressure and risk of heart failure in the elderly. JAMA. 1999;281(7):634–639.
71. Chakraborti, T, Mandal, A, Mandal, M, et al. Complement activation in heart diseases: role of oxidants. Cell Signal. 2000;12(9-10):607–617.
72. Chandler, JM, Duncan, PW. Balance and falls in the elderly: issues in evaluation and treatment. In Guccione AA, ed.: Geriatric physical therapy, ed 2, St Louis: Mosby, 2000.
73. Chandola, T. Chronic stress at work and the metabolic syndrome: prospective study. BMJ. 2006;332(7540):521–525.
74. Chilton, R. New trends in cardiovascular disease. Galveston, TX: University of Texas Medical Branch, 2001.
75. Chobanian, AV, National High Blood Pressure Education Program Coordinating Committee. The Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure. JAMA. 2003;289:2560–2571.
76. Ciccone, CD. Pharmacology in rehabilitation, ed 3. Philadelphia: FA Davis, 2001.
77. Ciesla, N. State of the heart: cardiopulmonary PT in the ’90s. Phys Ther. 1996;4(5):64–71.
78. Cohen, JC. Sequence variations in PCSK9, low LDL, and protection against coronary heart disease. N Engl J Med. 2006;354(12):1264–1272.
79. Cole, CR, Blackstone, EH, Pashow, FJ, et al. Heart-rate recovery immediately after exercise as a predictor of mortality. N Engl J Med. 1999;341(18):1351–1357.
80. Cole, CR, Foody, JM, Blackstone, EH, et al. Heart rate recovery after submaximal exercise testing as a predictor of mortality in a cardiovascularly healthy cohort. Ann Intern Med. 2000;132(7):552–555.
81. Colman, JM, Sermer, M, Seaward, PG, et al. Congenital heart disease in pregnancy. Cardiol Rev. 2000;8(3):166–173.
82. Colucci, WS. Nesiritide for the treatment of decompensated heart failure. J Card Fail. 2001;7(1):92–100.
83. Commandeur, S. Polymers, drug release, and drug-eluting stents. J Interv Cardiol. 2006;19(6):500–506.
84. Cooke, GA. A mechanistic investigation of ACE inhibitor dose effects on aerobic exercise capacity in heart failure patients. Eur Heart J. 2002;23(17):1360–1368.
85. Cooper, WO. Major congenital malformations after first-trimester exposure to ACE inhibitors. N Engl J Med. 2006;354(23):2443–2451.
86. Cote, P, Kreitz, BG, Cassidy, JD, et al. The validity of the extension-rotation test as a clinical screening procedure before neck manipulation: a secondary analysis. J Manipulative Physiol Ther. 1996;19(3):159–164.
87. Criqui, MH, Denenberg, JO, Langer, RD, et al. The epidemiology of peripheral arterial disease: importance of identifying the population at risk. Vasc Med. 1997;2(3):221–226.
88. Crumlish, CM, Bracken, J, Hand, MM, et al. When time is muscle. Am J Nurs. 2000;100(1):26–34.
89. Curvers, J, Nienhuis, SJ, Nap, AW, et al. Activated protein C resistance during in vitro fertilization treatment. Eur J Obstet Gynecol Reprod Biol. 2001;95(2):222–224.
90. Darbee, JC. Outcome measures in cardiopulmonary physical therapy: Medical Research Council (MRC) Dyspnea Scale. Cardiopulm Phys Ther J. 2006.
91. De Oliveira, E, Silva, ER, Foster, D, et al. Alcohol consumption raises HDL cholesterol levels by increasing the transport rate of apolipoproteins A-I and A-II. Circulation. 2000;102(19):2347–2352.
92. Dean, E. Preferred practice patterns in cardiopulmonary physical therapy: a guide to physiologic measures. Cardiopulm Phys Ther. 1999;10(4):124–134.
93. Deckelbaum, RJ, Fisher, EA, Winston, M. AHA Conference Proceedings: summary of a scientific conference on preventive nutrition: pediatrics to geriatrics. Circulation. 1999;100(4):450–456.
94. Del Rosario, JD, Strong, WB. The preparticipation cardiovascular evaluation of young athletes. J Musculoskel Med. 1999;16(8):445–459.
95. Dennison, AD. Exercise for individuals with Marfan syndrome. Cardiopulm Phys Ther J. 2006.
96. Denollet, J, Brutsaert, DL. Personality, disease severity, and the risk of long-term cardiac events in patients with a decreased ejection fraction after myocardial infarction. Circulation. 1998;97(2):128–129.
97. Devereux, RB, Brown, WT, Kramer-Fox, R, et al. Inheritance of mitral valve prolapse: effect of age and sex on gene expression. Ann Intern Med. 1982;97:826–832.
98. Devereux, RB, Kramer-Fox, R, Brown, WT, et al. Relation between clinical features of mitral valve prolapse syndrome and echocardiographically documented mitral valve prolapse. J Am Coll Cardiol. 1986;8:763–772.
99. Dhalla, NS, Temsah, RM, Netticadan, R. Role of oxidative stress in cardiovascular diseases. J Hypertens. 2000;18(6):655–673.
100. Dublin, S. Risk of new-onset atrial fibrillation in relation to body mass index. Arch Intern Med. 2006;166(21):2322–2328.
101. Durand, P, Prost, M, Loreau, N, et al. Impaired homocysteine metabolism and atherothrombotic disease. Lab Invest. 2001;81(5):645–672.
102. Egan, BM. Sleep and hypertension: burning the candle at both ends really is hazardous to your health. Hypertension. 2006;47(5):816–817.
103. Everson, SA, Kaplan, GA, Goldberg, DE, et al. Hypertension incidence is predicted by high levels of hopelessness. Hypertension. 2000;35(2):561–567.
104. Fall risk: predicting vs. preventing, 2001. Available on-line at www.onbalance.com Accessed April 25, 2008.
105. Fardy, PS, Azzollini, A, Magel, JR, et al. Gender and ethnic differences in health behaviors and risk factors for coronary disease among urban teenagers: the PATH program. J Gend Specif Med. 2000;3(2):59–68.
106. Federman, DG, Kirsner, RS. An update on hypercoagulable disorders. Arch Intern Med. 2001;161(8):1051–1056.
107. Fishbein, I. Bisphosphonate-mediated gene vector delivery from the metal surfaces of stents. Proc Natl Acad Sci U S A. 2006;103(1):159–164.
108. Fletcher, GF. Exercise standards for testing and training: a statement for healthcare professionals. From the American Heart Association. Circulation. 2001;104:1694–1740.
109. Ford, E, Newman, J, Deosaransingh, K. Racial and ethnic differences in the use of cardiovascular procedures: findings from the California Cooperative Cardiovascular Project. Am J Public Health. 2000;90(7):1128–1134.
110. Forsum, E. Calculation of energy expenditure in women using the MET system. Med Sci Sports Exerc. 2006;38(8):1520–1525.
111. Fowlkes, FG, Housley, E, Cawood, EH, et al. Edinburgh Artery Study: prevalence of asymptomatic and symptomatic peripheral arterial disease in the general population. Int J Epidemiol. 1991;30:384–392.
112. Fraenkel, L, Zhang, Y, Chaisson, CE, et al. Different factors influencing the expression of Raynaud’s phenomenon in men and women. Arthritis Rheum. 1999;42(2):306–310.
113. Francis, GS. Pathophysiology of chronic heart failure. Am J Med. 2001;110(suppl 7A):37–46.
114. Frownfelter D, Dean E, eds. Cardiovascular and pulmonary physical therapy, ed 4, St Louis: Mosby, 2006.
115. Fuchs, FD, Chambless, LE, Whelton, PK, et al. Alcohol consumption and the incidence of hypertension: the atherosclerosis risk in communities study. Hypertension. 2001;37(5):1242–1250.
116. Fukuda, K. Development of regenerative cardiomyocytes from mesenchymal stem cells for cardiovascular tissue engineering. Artif Organs. 2001;25(3):187–193.
117. Fuster, V, et alAmerican College of Cardiology, American Heart Association Task Force, European Society of Cardiology Committee for Practice Guidelines, European Heart Rhythm Association. 2006 Guidelines for the management of patients with atrial fibrillation. Circulation. 2006;114(7):e257–e354.
118. Gan, SC, Beaver, SK, Houck, PM, et al. Treatment of acute myocardial infarction and 30-day mortality among women and men. N Engl J Med. 2000;343(1):8–15.
119. Gangwisch, JE, Heymsfield, SB, Boden-Albala, B, et al. Short sleep duration as a risk factor for hypertension: analyses of the First National Health and Nutrition Examination Survey. Hypertension. 2006;47:833–839.
120. Gardner, AW, Forrester, L, Smith, GV. Altered gait profile in subjects with peripheral arterial disease. Vasc Med. 2001;6(1):31–34.
121. Gardner, AW, Katzel, LI, Sorkin, JD, et al. Improved functional outcomes following exercise rehabilitation in patients with intermittent claudication. J Gerontol A Biol Sci Med Sci. 2000;55(10):M570–M577.
122. Gardner, AW, Poehlman, ET. Exercise rehabilitation programs for the treatment of claudication pain. JAMA. 1995;274:975–980.
123. Gherpelli, JLD, Azeka, E, Riso, A, et al. Choreoathetosis after cardiac surgery with hypothermia and extracorporeal circulation. Pediatr Neurol. 1998;19:113–118.
124. Gill, TM, DiPietro, L, Krumholz, HM. Role of exercise stress testing and safety monitoring for older persons starting an exercise program. JAMA. 2000;284(3):342–349.
125. Gillum, LA, Mamidipudi, SK, Johnston, SC. Ischemic stroke risk with oral contraceptives: a meta-analysis. JAMA. 2000;284(1):72–78.
126. Giri, S, Thompson, PD, Kiernan, FJ, et al. Clinical and angiographic characteristics of exertion-related acute myocardial infarction. JAMA. 1999;282(18):1731–1736.
127. Glynn, RJ, Chae, GU, Guralnik, JM, et al. Pulse pressure and mortality in older people. Arch Intern Med. 2000;160(18):2765–2772.
128. Goldberg, JA. Aerobic and resistive exercise modify risk factors for coronary heart disease. Med Sci Sports Exerc. 1989;21:669–674.
129. Goodman, CC, Snyder, TE. Differential diagnosis for physical therapists: screening for referral, ed 4. Philadelphia: Saunders, 2007.
130. Goraya, TY, Jacobsen, SJ, Pellikka, PA, et al. Prognostic value of treadmill exercise testing in elderly persons. Ann Intern Med. 2000;132(11):862–870.
131. Gorelick, PB, Sacco, RL, Smith, DB, et al. Prevention of a first stroke: a review of guidelines and a multidisciplinary consensus statement from the National Stroke Association. JAMA. 1999;281(12):1112–1120.
132. Gottlieb, S. Short, sharp bouts of exercise good for the heart. BMJ. 2000;321(7261):589.
133. Gray, JC. Case report: diagnosis of intermittent vascular claudication in a patient with a diagnosis of sciatica. Phys Ther. 1999;79(6):582–590.
134. Grubb, BP, Kosinski, DJ. Syncope resulting from autonomic insufficiency syndromes associated with orthostatic intolerance. Med Clin North Am. 2001;85(2):457–471.
135. Grundy, SM. Diagnosis and management of the metabolic syndrome: an American Heart Association/National Heart, Lung, and Blood Institute Scientific Statement. Circulation. 2005;112(17):2735–2752.
136. Grundy, SM, Balady, GJ, Criqui, MH, et al. Guide to primary prevention of cardiovascular diseases: a statement for healthcare professionals from the task force on risk reduction. Circulation. 1997;95:2329–2331.
137. Gulbins, H. Homografts: a review. Expert Rev Cardiovasc Ther. 2003;1(4):533–539.
138. Gutersohn, T, Scheidegger, EP. Is baldness bad for the heart? Dermatology. 2005;211(1):72–74.
139. Hall, CM, Brody, LT. Therapeutic exercise: moving towards function, ed 2. Philadelphia: Lippincott Williams & Wilkins, 1998.
140. Harris, KA, Holly, RG. Physiological response to circuit weight training in borderline hypertensive subjects. Med Sci Sports Exerc. 1987;19:246–252.
141. Harvard Women’s Health Watch (HWHW). New view of heart disease in women. Harv Womens Health Watch. 2007;14(6):1–3.
142. Harvard Women’s Health Watch (HWHW). Varicose veins. Harv Womens Health Watch. 2000;8(11):5.
143. Hayase, M, Woodbum, KW, Perlroth, J, et al. Photoangioplasty with local motexafin luteum delivery reduces macrophages in a rabbit post-balloon injury model. Cardiovasc Res. 2001;49(2):449–455.
144. Hayek, E. Mitral valve prolapse. Lancet. 2005;365(9458):507–518.
145. Hayward, CS, Webb, CM, Collins, P. Effect of sex hormones on cardiac mass. Lancet. 2001;357(9265):1354–1356.
146. Healthy People 2010: Healthy people objectives, 2001. Available on-line at www.health.gov/healthypeople/ Accessed April 25, 2008.
147. Heiat, A, Vaccarino, V, Krumholz, HM. An evidence-based assessment of federal guidelines for overweight and obesity as they apply to elderly persons. Arch Intern Med. 2001;161(9):1194–1203.
148. Helgason, CM, Wolf, PA. American Heart Association Prevention Conference IV: prevention and rehabilitation of stroke: executive summary. Circulation. 1997;96:701–707.
149. Hillegass, EA. Cardiovascular diagnostic tests and procedures. In: Hillegass EA, Sadowsky HS, eds. Essentials of cardiopulmonary physical therapy. ed 2. Philadelphia: Saunders; 2001:336–379.
150. Hillegass EA, Sadowsky HS, eds. Essentials of cardiopulmonary physical therapy, ed 2, Philadelphia: Saunders, 2001.
151. Hines, LM, Stampfer, MJ, Ma, J, et al. Genetic variation in alcohol dehydrogenase and the beneficial effect of moderate alcohol consumption on myocardial infarction. N Engl J Med. 2001;344(8):549–555.
152. Hingorani, A, Ascher, E, Marks, N, et al. Morbidity and mortality associated with brachial vein thrombosis. Ann Vasc Surg. 2006;20(3):297–300.
153. Hirsh, J, Hoak, J. Management of deep vein thrombosis and pulmonary embolism: a statement for healthcare professionals. Council on Thrombosis (in consultation with the Council on Cardiovascular Radiology), American Heart Association. Circulation. 1996;96(6):2099.
154. Hu, FB, Stampfer, MJ, Colditz, GA, et al. Physical activity and risk of stroke in women. JAMA. 2000;283(22):2961–2967.
155. Hudson, KD, Long, L. Management of chronic venous ulcers. Phys Ther Case Rep. 2000;3(2):57–63.
156. Hulme, J. Fibromyalgia: a handbook for self care and treatment, ed 3. Missoula, MT: Phoenix, 2000.
157. Humphrey, R. Abdominal obesity and metabolic syndrome. Acute Care. 2004;13(4):2–4.
158. Humphrey, R, Arena, R. Surgical innovations for chronic heart failure in the context of cardiopulmonary rehabilitation. Phys Ther. 2000;80(1):61–69.
159. Humphrey, R, Bartels, MN. Exercise, cardiovascular disease, and chronic heart failure. Arch Phys Med Rehabil. 2001;82(3 suppl 1):S76–S81.
160. Irion, GL. Development of an inpatient cardiac rehabilitation program. Acute Care Perspect. 2000;9(1):21–22.
161. Irion, GL. Effect of upper extremity movement on sternal skin stress. Acute Care Perspect. 2006;15(3):1–6.
162. Irwin, S, Tecklin, JS. Cardiopulmonary physical therapy, ed 4. St Louis: Mosby, 2003.
163. Izquierdo-Porrera, AM, Gardner, AW, et al. Effects of exercise rehabilitation on cardiovascular risk factors in older patients with peripheral arterial occlusive disease. J Vasc Surg. 2000;31(4):670–677.
164. Jairath, N. Implications of gender differences on coronary artery disease risk reduction in women. AACN Clin Issues. 2001;12(1):17–28.
165. Johns Hopkins Medical Letter Health After 50. Exercising toward recovery after a heart attack. Johns Hopkins Med Lett Health After. 2000;50 12(4):4–5.
166. Johns Hopkins Medical Letter Health After 50. Peripheral vascular disease: walking toward a cure. Johns Hopkins Med Lett Health After. 2000;50 12(1):3.
167. Jolliffe, JA, Rees, K, Taylor, RS, et al. Exercise-based rehabilitation for coronary heart disease (Cochrane Review). Cochrane Database Syst Rev. 2001;1:CD001800.
168. Jones, EC, Devereux, RB, Roman, MJ, et al. Prevalence and correlates of mitral regurgitation in a population-based sample (the Strong Heart Study). Am J Cardiol. 2001;87(3):298–304.
169. Jones, JW, Richman, BW, Crigger, NA, et al. Technique of transmyocardial revascularization: avoiding complications in high-risk patients. J Cardiovasc Surg. 2001;42(3):353–357.
170. Jover, JA, Hernandez-Garcia, C, Morado, IC, et al. Combined treatment of giant-cell arteritis with methotrexate and prednisone: a randomized, double-blind, placebo-controlled trial. Ann Intern Med. 2001;134(2):106–114.
171. Junger, M. Mobilization versus immobilization in the treatment of acute proximal deep venous thrombosis: a prospective, randomized, open, multicentre trial. Curr Med Res Opin. 2006;22(3):593–602.
172. Kainulainen, K. Location on chromosome 15 of the gene defect causing Marfan syndrome. N Engl J Med. 1990;323(14):935–939.
173. Kamath, NV, Warner, MR, Camisa, C. Infective endocarditis: cutaneous clues to the diagnosis. Consultant. 1999;39(11):3085–3097.
174. Kannel, WB. The Framingham Study: its 50-year legacy and future promise. J Athleroscler Thromb. 2000;6(2):60–66.
175. Kannel, WB, Wolf, PA, Benjamin, EJ, et al. Prevalence, incidence, prognosis, and predisposing conditions for atrial fibrillation: population-based estimates. Am J Cardiol. 1998;82(8A):2N–9N.
176. Kavanagh, T. Cardiac rehabilitation. In: Goldberg L, Elliot DL, eds. Exercise for prevention and treatment of illness. Philadelphia: FA Davis; 1994:48–82.
177. Kelly, J, Rudd, AG. Giant cell arteritis presenting with arm claudication. Age Ageing. 2001;30(2):167–169.
178. Kenny, RA, Dey, AB. Syncope. In: Tallis RC, Fillit HM, Brocklehurst JC, eds. Brocklehurst’s textbook of geriatric medicine and gerontology. ed 5. New York: Churchill Livingstone; 1998:455–473.
179. Kim, JR, Oberman, A, Fletcher, GF, et al. Effect of exercise intensity and frequency on lipid levels in men with coronary heart disease: Training Level Comparison Trial. Am J Cardiol. 2001;87(8):942–946.
180. Kinney, MR, Packa, DR. Andreoli’s comprehensive cardiac care, ed 8. St Louis: Mosby, 1995.
181. Kloth, L, McCullough, JM. Wound healing: alternatives in management, ed 3. Philadelphia: FA Davis, 2001.
182. Koch, S. Cerebral fat microembolism and cognitive decline after hip and knee replacement. Stroke. 2007;38(3):1079–1081.
183. Koppes, LL, Twisk, JW, Van Mechelen, W, et al. Cross-sectional and longitudinal relationships between alcohol consumption and lipids, blood pressure, and body weight indices. J Stud Alcohol. 2005;66(6):713–721.
184. Kouchoukos, NT, Dougenis, D. Surgery in the thoracic aorta. N Engl J Med. 1997;336(26):1876–1888.
185. Kraaijenhagen, RA. Simplification of the diagnostic management of suspected deep vein thrombosis. Arch Intern Med. 2002;162(8):907–911.
186. Lakatta, EG. Age-associated cardiovascular changes in health: impact on cardiovascular disease in older persons. Heart Fail Rev. 2002;7(1):29–49.
187. Lamont, D, Parker, L, White, M, et al. Risk of cardiovascular disease measured by carotid intima-media thickness at age 49-51: life course study. BMJ. 2000;320(7230):273–278.
188. Lane, GJ. Simultaneous bilateral versus unilateral total knee arthroplasty: outcomes analysis. Clin Orthop. 1997;345:106–112.
189. Lansky, AJ. Comparison of differences in outcome after percutaneous coronary intervention in men versus women 40 years of age. Am J Cardiol. 2004;93(7):916–919.
190. Lansky, AJ. Percutaneous coronary intervention and adjunctive pharmacotherapy in women: a statement for healthcare professionals from the American Heart Association. Circulation. 2005;111:940–953.
191. LaPier, TK, Howell, D. Functional limitations in older patients recovering from coronary artery bypass. Cardiopulm Phys Ther J. 2003.
192. Lauer, M. Exercise testing in asymptomatic adults: a statement for professionals from the American Heart Association Council on Clinical Cardiology, Subcommittee on Exercise, Cardiac Rehabilitation, and Prevention. Circulation. 2005;112(5):771–776.
193. Lavie, CJ, Milani, RV. Cardiac rehabilitation and preventive cardiology in the elderly. Cardiol Clin. 1999;17(1):233–240.
194. Legato, MJ. Gender and the heart: sex-specific differences in normal anatomy and physiology. J Gend Specif Med. 2000;3(7):15–18.
195. Legato, MJ. Gender specific aspects of human biology for the practicing physician. Armonk, NY: Futura, 1997.
196. Leng, GC. Exercise for intermittent claudication. Cochrane Database Syst Rev. 2000;2:CD000990.
197. Leung, DYM, Meissner, HC. The many faces of Kawasaki syndrome. Hosp Pract. 2000;35(1):77–81.
198. Levy, D, Wilson, PWF. Atherosclerotic cardiovascular disease: an epidemiologic perspective. In: Topol EJ, ed. Comprehensive cardiovascular medicine. Philadelphia: Lippincott-Raven; 1998:27–43.
199. Lichtenstein, AH, et althe American Heart Association Nutrition Committee. Diet and lifestyle recommendations revision 2006: a scientific statement from the American Heart Association Nutrition Committee. Circulation. 2006;114(1):82–96. [[Epub June 19, 2006.]].
200. Loeys, BL. Aneurysm syndromes caused by mutations in the TGF-beta receptor. N Engl J Med. 2006;355(8):788–798.
201. Luft, FC. Molecular genetics of salt-sensitivity and hypertension. Drug Metab Dispos. 2001;29(4 pt 2):500–504.
202. Luskin, FM. A review of mind-body therapies in the treatment of cardiovascular disease: I. Implications for the elderly. Altern Ther Health Med. 1998;4(3):46–61.
203. Ma, H. Arterial wall cholesterol content is a predictor of development and severity of arterial thrombosis. J Throm Thrombolysis. 2006;22(1):5–11.
204. MacKay, D. Hemorrhoids and varicose veins: a review of treatment options. Altern Med Rev. 2001;6(2):126–140.
205. Maddux, JT, Wink, O, Messenger, JC, et al. Randomized study of the safety and clinical utility of rotational angiography versus standard angiography in the diagnosis of coronary artery disease. Catheter Cardiovasc Interv. 2004;62(2):167–174.
206. Magee, DJ. Orthopedic physical assessment, ed 4. Philadelphia: Harcourt Health Sciences, 2001.
207. Maisch, B, Ristic, AD, Seferovic, PM. New directions in diagnosis and treatment of pericardial disease: a project of the Taskforce on Pericardial Disease of the World Heart Federation. Herz. 2000;25(8):794–798.
208. Maisel, AS, Koon, J, Krishnaswamy, P, et al. Utility of B-natriuretic peptide as a rapid, point-of-care test for screening patients undergoing echocardiography to determine left ventricular dysfunction. Am Heart J. 2001;141(3):267–374.
209. Manson, JAE, Hu, FB, Rich-Edwards, JW, et al. Brisk exercise reduces coronary risk in women. N Engl J Med. 1999;341:650–658.
210. Marder, VJ, Mellinghoff, IK. Cocaine and Buerger disease: is there a pathogenetic association? Arch Intern Med. 2000;160(13):2057–2060.
211. Marinella, MA, Kathula, SK, Markert, RJ. Spectrum of upper-extremity deep venous thrombosis in a community teaching hospital. Heart Lung. 2000;29(2):113–117.
212. Maron, BJ, Towbin, JA, Thiene, G, et al. Contemporary definitions and classification of the cardiomyopathies: an American Heart Association scientific statement from the Council on Clinical Cardiology, Heart Failure and Transplantation Committee; Quality of Care and Outcomes Research and Functional Genomics and Translational Biology Interdisciplinary Working Groups; and Council on Epidemiology and Prevention. Circulation. 2006;113(14):1807–1816.
213. Maron, BJ, Thompson, PD, Puffer, JC, et al. Cardiovascular preparticipation screening of competitive athletes: a statement for health professionals from the Sudden Death Committee, American Heart Association. Circulation. 1996;94(4):850–856.
214. Maxwell M: Personal communication, International Heart Institute, Missoula, MT, 2001.
215. McCann, ME. Sexual healing after heart attack. Am J Nurs. 1989;89(9):1133–1138.
216. McKhann, GM, Borowicz, LM, Goldsborough, MA, et al. Depression and cognitive decline after coronary artery bypass grafting. Lancet. 1997;349(9061):1282–1284.
217. Meier, CR. The possible role of infections in acute myocardial infarction. Biomed Pharmacother. 1999;53(9):397–404.
218. Meisler, P. Sternum support harness for treatment of sternotomy pain and the prevention of sternal instability. Cardiopulm Phys Ther J. 2003.
219. Menasche, P, Hagege, AA, Scorsin, M, et al. Myoblast transplantation for heart failure. Lancet. 2001;357(9252):279–280.
220. Meyer, K, Samek, L, Schwaibold, M, et al. Interval training in patients with severe chronic heart failure: analysis and recommendations for exercise procedures. Med Sci Sports Exerc. 1997;29(3):306–312.
221. Michelena, HI, Ezekowitz, MD. Atrial fibrillation: are there gender differences? J Gend Specif Med. 2000;3(6):44–49.
222. Mieres, JH, Shaw, LJ, Arai, A, et al. Role of noninvasive testing in the clinical evaluation of women with suspected coronary artery disease: consensus statement from the Cardiac Imaging Committee, Council on Clinical Cardiology, and the Cardiovascular Imaging and Intervention Committee, Council on Cardiovascular Radiology and Intervention, American Heart Association. Circulation. 2005;111:682–696.
223. Miller, DT. Atherosclerosis: the path from genomics to therapeutics. J Am Coll Cardiol. 2007;49(15):1589–1599.
224. Mohler, ER. Peripheral arterial disease: identification and implications. Arch Intern Med. 2003;163(19):2306–2314.
225. Moliterno, DJ. No association between plasma lipoprotein (a) concentrations and the presence or absence of coronary atherosclerosis in African-Americans. Arterioscler Thromb Vasc Biol. 1995;15(7):850–855.
226. Morbidity and Morbidity Weekly Report. Mortality from coronary heart disease and acute myocardial infarction United States, 1998. MMWR Morb Mortal Wkly Rep. 2001;50(06):90–93.
227. Morris GS: Chemotherapy-induced cardiotoxicity. Adapted from presentation at American Physical Therapy Association Combined Sections Meeting, San Diego, February 2, 2006.
228. Mosca, L. Evidence-based guidelines for cardiovascular disease prevention in women. Circulation. 2004;109(5):672–693.
229. Mosca, L. Evidence-based guidelines for cardiovascular disease prevention in women: 2007 update. Circulation. 2007;115:1481–1501.
230. Mueller, MJ. Invited commentary: the effect of exercises on walking distance of patients with intermittent claudication. Phys Ther. 1998;78(3):286–288.
231. Mulcare, JA, Jackson, K, Petersen, DR, et al. Physiological responses during unweighted ambulation of patients with transtibial amputation: a pilot study. Phys Ther Case Rep. 1999;2(3):99–103.
232. Muller, JE. Triggering myocardial infarction by sexual activity: low absolute risk and prevention by regular physical exertion: determinants of Myocardial Infarction Onset Study investigators. JAMA. 1996;275(18):1405–1409.
233. Nagir, GV, Chaput, LA, Vittinghoff, E, et al. Pulse pressure and cardiovascular events in postmenopausal women with coronary heart disease. Chest. 2005;127(5):1498–1506.
234. Najjar, SS, Scuteri, A, Lakatta, EG. Arterial aging: is it an immutable cardiovascular risk factor? Hypertension. 2005;46(3):454–462.
235. National Center for Health Statistics (NCHS): Surveys and data: vital statistics, 2001. Available on-line at www.cdc.gov/nchs/default.htm Accessed April 25, 2008.
236. National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) final report. Circulation. 2002;106:3143–3421.
237. National Heart Attack Alert Program (NHAAP): Available on-line at www.nhlbi.nih.gov/health/prof/heart/index.htm#mi April 25, 2008.
238. National Institute of Diabetes and Digestive Kidney Diseases (NIDDKD). Overweight, obesity, and health risk: National Task Force on the Prevention and Treatment of Obesity. Arch Intern Med. 2000;160(7):898–904.
239. National Institutes of Health: National Heart, Lung, and Blood Institute detection, evaluation, and treatment of high blood cholesterol in adults: Adult Treatment Panel III (ATPIII) guidelines, 2001. Available on-line at www.nhlbi.nih.gov Accessed April 25, 2008.
240. Nichol, KL. Influenza vaccination in the elderly: impact on hospitalization and mortality. Drugs Aging. 2005;22(6):495–515.
241. Nichol, KL, Nordin, J, Mullooly, J, et al. Influenza vaccination and reduction in hospitalization for cardiac disease and stroke among the elderly. N Engl J Med. 2003;348(14):1322–1332.
242. Nieto, FJ, Young, TB, Lind, BK, et al. Association of sleep-disordered breathing, sleep apnea, and hypertension in a large community-based study: Sleep Heart Healthy Study. JAMA. 2000;283(14):1829–1836.
243. Nikol, S, Huehns, TY. Preclinical and clinical experience in vascular gene therapy: advantages over conservative/standard therapy. J Invasive Cardiol. 2001;13(4):333–338.
244. Nolan, J, Fox, KA. Heart rate variability and cardiac failure. Heart. 1999;81(5):561–562.
245. Ohtsuka, T, Hamada, M, Hiasa, G, et al. Effect of beta-blockers on circulating levels of inflammatory and anti-inflammatory cytokines in patients with dilated cardiomyopathy. J Am Coll Cardiol. 2001;37(2):412–417.
246. O’Kane, PD, Jackson, G. Erectile dysfunction: is there silent obstructive coronary artery disease? Int J Clin Pract. 2001;55(3):219–220.
247. Orlic, D, Kajstura, J, Chimenti, S, et al. Bone marrow cells regenerate infarcted myocardium. Nature. 2001;410(6829):640–641.
248. Owen, PJ, Rajiv, C, Vinereanu, D, et al. Subclinical hypothyroidism, arterial stiffness, and myocardial reserve. J Clin Endocrinol Metab. 2006. [[Epub ahead of print.]].
249. Oxenham, H, Sharpe, N. Cardiovascular aging and heart failure. Eur J Heart Fail. 2003;5(4):427–434.
250. Paepe, A. Bleeding and bruising in patients with Ehlers-Danlos syndrome and other collagen vascular disorders. Br J Haematol. 2004;127(5):491–500.
251. Pai, VB, Nahata, MC. Cardiotoxicity of chemotherapeutic agents: incidence, treatment, and prevention. Drug Saf. 2000;22(4):263–302.
252. Panagiotakos, DB, Kromhout, D, Menotti, A, et al. The relation between pulse pressure and cardiovascular mortality in 12,763 middle-aged men from various parts of the world: a 25-year follow-up of the seven countries study. Arch Intern Med. 2005;165(18):2142–2147.
253. Pandey, DK, Labarthe, DR, Goff, DC, et al. Community-wide coronary heart disease mortality in Mexican Americans equals or exceeds that in non-Hispanic whites: the Corpus Christi Heart Project. Am J Med. 2001;110(2):147–148.
254. Papamichael, CM, Lekakis, JP, Stamatelopoulos, KS, et al. Ankle-brachial index as a predictor of the extent of coronary atherosclerosis and cardiovascular events in patients with coronary artery disease. Am J Cardiol. 2000;86(6):615–618.
255. Partsch, H. Compression and walking versus bed rest in the treatment of proximal deep venous thrombosis with low molecular weight heparin. J Vasc Surg. 2000;32:861–869.
256. Partsch, H. Immediate mobilisation in acute vein thrombosis reduces post-thrombotic syndrome. Int Angiol. 2004;23(3):206–212.
257. Partsch, H. Therapy of deep vein thrombosis with low molecular weight heparin, leg compression and immediate ambulation. Vasa. 2001;30:195–204.
258. Pasternak, RC. Cardiovascular disease prevention: I. What works what doesn’t? Consultant. 1999;39(11):2957–2963.
259. Pasternak, RC. Cardiovascular disease prevention: II. What works what doesn’t? Consultant. 1999;39(11):2973–2984.
260. Pellicano, R. Updated review (2006) on Helicobacter pylori as a potential target for the therapy of ischemic heart disease. Panminerva Med. 2006;48(4):241–246.
261. Pellicia, A, Maron, BJ. Outer limits of the athlete’s heart: the effect of gender and relevance to the differential diagnosis with primary cardiac diseases. Cardiol Clin. 1997;15:381–396.
262. Perez, EA. Doxorubicin and paclitaxel in the treatment of advanced breast cancer: efficacy and cardiac considerations. Cancer Invest. 2001;19(2):155–164.
263. Pescatello, LS. Physical activity, cardiometabolic health and older adults: recent findings. Sports Med. 1999;28(5):315–323.
264. Pescatello, LS, Miller, B, Danias, PG, et al. Dynamic exercise normalizes resting blood pressure in mildly hypertensive premenopausal women. Am Heart J. 1999;138(5 pt 1):916–921.
265. Pihlstrom, BL, Michalowicz, BS, Johnson, NW. Periodontal diseases. Lancet. 2005;366(9499):1809–1820.
266. Piña, IL. Exercise and heart failure: a statement from the American Heart Association Committee on Exercise, Rehabilitation, and Prevention. Circulation. 2003;107:1210–1225.
267. Pladevall, M. A single factor underlies the metabolic syndrome: a confirmatory factor analysis. Diabetes Care. 2006;29(1):113–122.
268. Pollock, ML, Franklin, BA, Balady, GJ, et al. AHA science advisory: resistance exercise in individuals with and without cardiovascular disease: benefits, rationale, safety, and prescription. An advisory from the Committee on Exercise, Rehabilitation, and Prevention, Council on Clinical Cardiology, American Heart Association; position paper endorsed by the American College of Sports Medicine. Circulation. 2000;101(7):828–833.
269. Prandoni, P. Below-knee elastic compression stockings to prevent the post-thrombotic syndrome: a randomized, controlled trial. Ann Intern Med. 2004;141(4):249–256.
270. Public Access Defibrillation League (PADL): Cardiac arrest survivor’s act, 2001. Available on-line at www.padl.org Accessed April 25, 2008.
271. Quyyumi, AA. Women and ischemic heart disease: pathophysiologic implications from the Women’s Ischemia Syndrome Evaluation (WISE) study and future research steps. J Am Coll Cardiol. 2006;47(3 suppl):S66–S71.
272. Rangarajan, U, Kochar, MS. Hypertension in women. WMJ. 2000;99(3):65–70.
273. Ray, JG, Mamdani, M, Tsuyuki, RT, et al. Use of statins and the subsequent development of deep vein thrombosis. Arch Intern Med. 2001;161(11):1405–1410.
274. Reynen, K. Cardiac myxomas. N Engl J Med. 1995;333(24):1610–1617.
275. Reynen, K, Kockeritz, U, Strasser, RH. Metastases to the heart. Ann Oncol. 2004;15(3):375–381.
276. Rich, MW. Heart failure in the twenty-first century: a cardiogeriatric syndrome. J Gerontol A Biol Sci Med Sci. 2001;56(2):M88–M96.
277. Riddle, DL, Preliminary validation of clinical assessment for deep vein thrombosis in orthopaedic outpatients. Clin Orthop Relat Res. 2005(432):252–257.
278. Riddle, DL, Wells, PS. Diagnosis of lower-extremity deep vein thrombosis in outpatients. Phys Ther. 2004;84(8):729–735.
279. Riding, G. Paradoxical cerebral embolization: an explanation for fat embolism syndrome. J Bone Joint Surg Br. 2007;86(1):95–98.
280. Ridker, PM. C-reactive protein and the prediction of cardiovascular events among those at intermediate risk. J Am Coll Cardiol. 2007;49(21):2129–2138.
281. Ridker, PM. Development and validation of impoved algorithms for the assessment of global cardiovascular risk in women: the Reynolds Risk Score. JAMA. 2007;297(6):611–619.
282. Rivett, DA. The premanipulative vertebral artery testing protocol: a brief review. Physiotherapy. 1995;23:9–12.
283. Rivett, DA, Sharples, KJ, Milburn, PD. Effect of premanipulative tests on vertebral artery and internal carotid artery blood flow: a pilot study. J Manipulative Physiol Ther. 1999;6:368–375.
284. Roberts, WC. Neoplasms involving the heart, their stimulators, and adverse consequences of their therapy. Proc (Bayl Univ Med Cent). 2001;14(4):358–376.
285. Rocca, JD. Responding to atrial fibrillation. Nursing2007. 2007;37(4):37–44.
286. Rosen, RC. Erectile dysfunction and cardiac disease: recommendations of the Second Princeton Conference. Curr Urol Rep. 2006;7(6):490–496.
287. Rosendorff, C, Black, HR, Cannon, CP, et al. Treatment of hypertension in the prevention and management of ischemic heart disease: a scientific statement from the American Heart Association Council for High Blood Pressure Research and the Councils on Clinical Cardiology and Epidemiology and Prevention. Circulation. 2007. [[Epub ahead of print.]].
288. Rosenfeld, AG. Women’s risk of decision delay in acute myocardial infarction: implications for research and practice. AACN Clin Issues. 2001;12(1):29–39.
289. Salem, RO. Effects of alcohol on hemostasis. Am J Clin Pathol. 2005;123:S96–S105. [(suppl)].
290. Sapico, FL, Liquete, JA, Sarma, RJ. Bone and joint infections in patients with infective endocarditis: review of a 4 year experience. Clin Infect Dis. 1996;22:783–787.
291. Scherer, S. Disability and quality of life in people with peripheral arterial disease and intermittent claudication. Cardiopulm Phys Ther J. 2003.
292. Scherer, S. Exercise in the patient with claudication. Cardiopulm Phys Ther. 1999;10(2):45–48.
293. Schiele, F, Batur, MK, Seronde, MF, et al. Cytomegalovirus, Chlamydia pneumoniae, and Helicobacter pylori IgG antibodies and restenosis after stent implantation: an angiographic and intravascular ultrasound study. Heart. 2001;85(3):304–311.
294. Schroll, M, Munck, O. Estimation of peripheral arteriosclerotic disease by ankle blood pressure measurements in a population study of 60-year-old men and women. J Chronic Dis. 1981;34:261–269.
295. Schwartz, GG. Myocardial Ischemia Reduction with Aggressive Cholesterol Lowering (MIRACL) Study Investigators.: Effects of atorvastatin on early recurrent ischemic events in acute coronary syndromes: the MIRACL study: a randomized controlled trial. JAMA. 2001;285(13):1711–1718.
296. Schwartz, JB. The electrocardiographic QT interval and its prolongation in response to medications: differences between men and women. J Gend Specif Med. 2000;3(5):25–28.
297. Scott-Okafor, HR, Silver, KK, Parker, J, et al. Lower extremity strength deficits in peripheral arterial occlusive disease patients with intermittent claudication. Angiology. 2001;52(1):7–14.
298. Seals, DR. The aging cardiovascular system: changes in autonomic function at rest and in response to exercise. Int J Sport Nutr Exerc Metab. 2001;11:S189–S195. [(suppl)].
299. Selnes, OA, Goldsborough, MA, Borowicz, LM, et al. Determinants of cognitive change after coronary artery bypass surgery: a multifactorial problem. Ann Thorac Surg. 1999;67(6):1669–1676.
300. Selnes, OA, Goldsborough, MA, Borowicz, LM, et al. Neurobehavioural sequelae of cardiopulmonary bypass. Lancet. 1999;353(9164):1601–1606.
301. Selnes, OA, Royall, RM, Grega, MA, et al. Cognitive changes 5 years after coronary artery bypass grafting: is there evidence of late decline? Arch Neurol. 2001;58(4):598–604.
302. Shaffer, RG. Effect of acute exercise on endothelial progenitor cells in patients with peripheral arterial disease. Vasc Med. 2006;11(4):219–226.
303. Shankar, K. Exercise prescription. Philadelphia: Hanley & Belfus, 1999.
304. Shaw, DK, Deutsch, DT, Bowling, RJ. Efficacy of shoulder range of motion exercise in hospitalized patients after coronary artery bypass graft surgery. Heart Lung. 1990;19(3):321–322.
305. Shaw, LJ. Insights from the NHLBI-Sponsored Women’s Ischemia Syndrome Evaluation (WISE) study: Part I: gender differences in traditional and novel risk factors, symptom evaluation, and gender-optimized diagnostic categories. J Am Coll Cardiol. 2006;47(3 suppl):S4–S20.
306. Shaw, LJ, Lewis, JF, Hlatky, MA, et al. Women’s Ischemic Syndrome Evaluation: current status and future research directions: report of the National Heart, Lung and Blood Institute Workshop: October 2-4, 2002: Section 5: Gender-Related Risk Factors for Ischemic Heart Disease. Circulation. 2004;109(6):e56–e58.
307. Sherwood, A, May, CW, Siegel, WC, et al. Ethnic differences in hemodynamic responses to stress in hypertensive men and women. Am J Hypertens. 1995;8:552–557.
308. Shumway-Cook, A, Brauer, S, Woollacott, M. Predicting the probability for falls in community-dwelling older adults using the Timed Up & Go Test. Phys Ther. 2000;80(9):896–903.
309. Silberstein, L, Davies, A, Kelsey, S, et al. Myositis, polyserositis, and constrictive pericarditis as manifestations of chronic graft-versus-host disease after peripheral stem cell transplantation. Bone Marrow Transplant. 2001;27(2):231–233.
310. Skinner, JS. Exercise testing and exercise prescription for special cases, ed 2. Philadelphia: Lea & Febiger, 1993.
311. Smit, AA, Halliwill, JR, Low, PA, et al. Pathophysiological basis of orthostatic hypotension in autonomic failure. J Physiol. 1999;519(pt 1):1–10.
312. Society for Vascular Surgery (SVS): VascularWeb. Available on-line at http://www.vascularweb.org/ Accessed June 4, 2007.
313. Srivastava, D. Genetic assembly of the heart: implications for congenital heart disease. Annu Rev Physiol. 2001;63:451–469.
314. Stamou, SC, Corso, PJ. Coronary revascularization without cardiopulmonary bypass in high-risk patients: a route to the future. Ann Thorac Surg. 2001;71(3):1056–1061.
315. Stewart, KJ, McFarland, LD, Weinhofer, JJ, et al. Safety and efficacy of weight training soon after acute myocardial infarction. J Cardiopulm Rehabil. 1998;18:37–44.
316. Stone, PH, Krantz, DS, McMahon, RP, et al. Relationship among mental stress-induced ischemia and ischemia during daily life during exercise: the psychophysiologic investigations of myocardial ischemia (PIMI) study. J Am Coll Cardiol. 1999;33(6):1476–1484.
317. Strong, JP, Malcolm, GT, McMahan, CA, et al. Prevalence and extent of atherosclerosis in adolescents and young adults: implications for prevention from the Pathobiological Determinants of Atherosclerosis in Youth Study. JAMA. 1999;282(8):727–735.
318. Stump, DA. Noncardiac surgery brain injury: etiologic factors and prevention. Heart Surg Forum. 2003;6(4):196–197.
319. Summaries for patients. can compression stockings prevent the post-thrombotic syndrome? Ann Intern Med. 2004;141(4):I12.
320. Sundquist, J, Winkleby, MA, Pudaric, S. Cardiovascular disease risk factors among older black, Mexican-American, and white women and men: an analysis of NHANES III, 1988-1994: Third National Health and Nutrition Examination Survey. J Am Geriatr Soc. 2001;49(2):109–116.
321. Sussman, C. The wound care patient education and resource manual. Gaithersburg, MD: Aspen, 1999.
322. Sussman C, Bates-Jensen B, eds. Wound care: a collaborative practice manual for physical therapists and nurses, ed 2, Gaithersburg, MD: Aspen, 2001.
323. Szombathy, T, Janoskuti, L, Szalai, C, et al. Angiotensin II type 1 receptor gene polymorphism and mitral valve prolapse syndrome. Am Heart J. 2000;139(1 pt 1):101–105.
324. Takazoe, K, Ogawa, H, Yasue, H, et al. Increased plasminogen activator inhibitor activity and diabetes predict subsequent coronary events in patients with angina pectoris. Ann Med. 2001;33(3):206–212.
325. Takeyama, J, Itoh, H, Kato, M, et al. Effects of physical training on the recovery of the autonomic nervous activity during exercise after coronary artery bypass grafting: effects of physical training after CABG. Jpn Circ J. 2000;64(11):809–813.
326. Tasso, KH. Gross motor development of a child with multiple congenital heart defects. Phys Ther Case Rep. 2000;3(2):71–77.
327. Temizhan, A, Dincer, I, Pamir, G, et al. Is there any effect of chronobiological changes on coronary angioplasty? J Cardiovasc Risk. 2001;8(1):15–19.
328. Tepper S: Management of deep venous thrombosis. Adapted from presentation at American Physical Therapy Association Combined Sections Meeting, San Diego, February 2, 2006.
329. Tepper, SH, McKeough, DM. Deep venous thrombosis: risks, diagnosis, treatment interventions and prevention. Acute Care Perspect. 2000;9(1):1–7.
330. Thiel, H, Wallace, K, Donat, J, et al. Effect of various head and neck positions on vertebral artery blood flow. Clin Biomech. 1994;9:105–110.
331. Thomas, CM, Pierzga, JM, Kenney, WL. Aerobic training and cutaneous vasodilation in young and older men. J Appl Physiol. 1999;86(5):1676–1686.
332. Tikkanen, MJ. Erectile dysfunction as a risk factor for coronary heart disease: implications for prevention. Int J Clin Pract. 2007;61(2):265–268.
333. Tonetti, MS. Treatment of periodontitis and endothelial function. N Engl J Med. 2007;356(9):911–920.
334. Trujillo-Santos, J. Bed rest or ambulation in the initial treatment of patients with acute deep vein thrombosis or pulmonary embolism: findings from the RIETE registry. Chest. 2005;127:1631–1636.
335. Tsai, MW. Effects of exercise training on heart rate variability after coronary angioplasty. Phys Ther. 2006;86(5):626–635.
336. Vainio, H, Bianchini, F. Prevention of disease with pharmaceuticals. Pharmacol Toxicol. 2001;88(3):111–118.
337. Van Belle, E, Ketelers, R, Bauters, C. Patency of percutaneous transluminal coronary angioplasty sites at 6-month angiographic follow-up: a key determinant of survival in diabetics after coronary balloon angioplasty. Circulation. 2001;103(9):1218–1224.
338. VanSwearingen, JM, Brach, JS. Making geriatric assessment work: selecting useful measures. Phys Ther. 2001;81(6):1233–1252.
339. Varma, C, Camm, AJ. Pacing for heart failure. Lancet. 2001;357(9264):1277–1283.
340. Vaughan, CJ, Casey, M, He, J, et al. Identification of a chromosome 11q23.2-q24 locus for familial aortic aneurysm disease, a genetically heterogeneous disorder. Circulation. 2001;103(20):2469–2475.
341. Vliet, EL. Screaming to be heard: hormonal connections women suspect … and doctors ignore. New York: M. Evans, 1995.
342. Waksman, R. Biodegradable stents: they do their job and disappear. J Invasive Cardiol. 2006;18(2):70–74.
343. Walsh, JP, Bremner, AP, Bulsara, MK, et al. Subclinical thyroid dysfunction as a risk factor for cardiovascular disease. Arch Intern Med. 2005;165(21):2467–2472.
344. Wannamethee, SG, Shaper, AG. Physical activity in the prevention of cardiovascular disease: an epidemiological perspective. Sports Med. 2001;31(2):101–114.
345. Watchie, J. Cardiopulmonary complications of cancer treatment. Clin Manage. 1992;12:92–95.
346. Watchie, J. Cardiopulmonary implications of specific diseases. In: Hillegass EA, Sadowsky HS, eds. Essentials of cardiopulmonary physical therapy. ed 2. Philadelphia: Saunders; 2001:285–335.
347. Waters, DD. Women’s Ischemic Syndrome Evaluation: current status and future research directions: report of the National Heart, Lung and Blood Institute Workshop: October 2-4, 2002: Section 4: Lessons from Hormone Replacement Trials. Circulation. 2004;109:e53–e55.
348. Weaver, WD, Cerqueira, M, Hallstrom, AP, et al. Prehospital-initiated vs. hospital-initiated thrombolytic therapy: the Myocardial Infarction Triage and Intervention Trial. JAMA. 1993;270(10):1211–1216.
349. Weise, Shelly. Women and coronary heart disease: exercise, wellness and risk modification. Cardiopulm Phys Ther J. 2002.
350. Wells, PS. A simple clinical model for the diagnosis of deep-vein thrombosis combined with impedance plethysmography: potential for an improvement in the diagnostic process. J Intern Med. 1998;243(1):15–23.
351. Wells, PS. Advances in the diagnosis of venous thromboembolism. J Thromb Thrombolysis. 2006;21(1):31–40.
352. Wells, PS. Diagnosis of deep vein thrombosis. Biomed Pharmacother. 1996;50(6-7):235–242.
353. Welsch, MA. Heart failure syndrome: implications for exercise training. Cardiopulm Phys Ther J. 2004.
354. Wenger, NK. Coronary heart disease in women: highlights of the past 2 years stepping stones, milestones and obstructing boulders. Nat Clin Pract Cardiovasc Med. 2006;3:194–202.
355. Wenger, NK. Lipid management and control of other coronary risk factors in the postmenopausal woman. J Womens Health Gend Based Med. 2000;9(3):235–243.
356. White, RH. Incidence and time course of thromboembolic outcomes following total hip or knee arthroplasty. Arch Intern Med. 1998;158(14):1525–1531.
357. Whitelaw, GP. Evaluation of intermittent pneumatic compression devices. Orthopedics. 2001;24(3):257–261.
358. Whitworth, JA, World Health Organization, International Society of Hypertension Writing Group. 2003 World Health Organization (WHO)/International Society of Hypertension (ISH) statement on management of hypertension. J Hypertens. 2003;21(11):1983–1992.
359. Wiley, RL, Dunn, CL, Cox, RH, et al. Isometric exercise training lowers resting blood pressure. Med Sci Sports Exerc. 1992;24:749–754.
360. Willems, R. Non-pharmacologic treatment of atrial fibrillation in elderly persons. Am J Geriatr Cardiol. 2005;14(2):68–72.
361. Williams, JE, Paton, CC, Siegler, IC, et al. Anger proneness predicts coronary heart disease risk: prospective analysis from the atherosclerosis risk in communities (ARIC) study. Circulation. 2000;101(17):2034–2039.
362. Williams, SG, Cooke, GA, Wright, DJ, et al. Disparate results of ACE inhibitor dosage on exercise capacity in heart failure: a reappraisal of vasodilator therapy and study design. Int J Cardiol. 2001;77(2-3):239–245.
363. Wilson, PW. Metabolic risk factors for coronary heart disease: current and future prospects. Curr Opin Cardiol. 1999;14:176–185.
364. Wilson, PW. The myocardial ischemia reduction with aggressive cholesterol lowering study summary for patients. Curr Cardiol Rep. 2002;4(6):485.
365. Womack, CJ, Ivey, FM, Gardner, AW, et al. Fibrinolytic response to acute exercise in patients with peripheral arterial disease. Med Sci Sports Exerc. 2001;33(2):214–219.
366. Yeh, ETH, Ton, AT, Lenihan, DJ, et al. Cardiovascular complications of cancer therapy: diagnosis, pathogenesis and management. Circulation. 2004;109(25):3122–3131.
367. Zambetti, M, Moliterni, A, Materazzo, C, et al. Long-term cardiac sequelae in operable breast cancer patients given adjuvant chemotherapy with or without doxorubicin and breast irradiation. J Clin Oncol. 2001;19(1):37–43.
368. Zieske, AW, Malcolm, GT, Strong, JP. Pathobiological determinants of atherosclerosis in youth (PDAY) cardiovascular specimen and data library. J La State Med Soc. 2000;152(6):296–301.
369. Zoldhelyi, P, Chen, ZQ, Shelat, HS, et al. Local gene transfer of tissue factor pathway inhibitor regulates intimal hyperplasia in atherosclerotic arteries. Proc Natl Acad Sci U S A. 2001;98(7):4078–4083.