Cystic Fibrosis
PREFERRED PRACTICE PATTERNS
4A:
Primary Prevention/Risk Reduction for Skeletal Demineralization
4C:
Impaired Muscle Performance
6C:
Impaired Ventilation, Respiration/Gas Exchange, and Aerobic Capacity/Endurance Associated with Airway Clearance Dysfunction
6E:
Impaired Ventilation and Respiration/Gas Exchange Associated with Ventilatory Pump Dysfunction or Failure
6F:
Impaired Ventilation and Respiration/Gas Exchange Associated with Respiratory Failure
Airway Clearance Techniques
The therapist must always be aware that anyone with CF is susceptible to infections, in particular Burkholderia cepacia. Care must be taken to avoid transmission via equipment, other patients, or oneself. Handwashing is essential, and high alcohol hand rubs may be more effective.163 The therapist will be involved with airway clearance techniques carried out several times per day or as often as the person is able to tolerate it without undue fatigue. Airway clearance techniques should not be performed before or immediately after meals, so treatment must be scheduled to avoid mealtimes.
Aerosol therapy to deliver medication to the lower respiratory tract should be administered just before airway clearance techniques to maximize the effectiveness of both treatments. Breathing exercises, improving posture, mobilizing the thorax through active exercise, and manual therapy are part of promoting good breathing patterns and improving inspiratory muscle endurance. Specifics of airway clearance techniques for this population are beyond the scope of this text; the reader is referred to more detailed materials available.102,103,105,140
The many difficulties surrounding percussion and postural drainage (e.g., poor compliance, time consuming, and requiring the assistance of a trained individual) have resulted in the development of alternative airway clearance techniques that can be accomplished without the assistance of another caregiver.
Each of these techniques (e.g., autogenic drainage, active cycle breathing, positive expiratory pressure [PEP], Flutter valve therapy, Acapella, and Quake) requires a certain level of compliance, motivation, understanding, neuromuscular function, and breath control. The therapist is very instrumental in evaluating each individual’s needs, motivation, abilities, resources, and preferences in determining the best intervention or interventions to use.
Autogenic drainage, active cycle breathing, and PEP help the individual to move the mucus up to the larger airways where it can be coughed out more easily. Autogenic drainage comprises a series of sequential breathing exercises designed to clear the small, medium, and large airways in that order. The PEP device maintains pressure in the lungs, keeping the airways open and allowing air to get behind the mucus (Fig. 15-15). This device has been shown to be effective in increased sputum production, improved lung function, and improved oxygenation.107

Figure 15-15 PARI PEP. The positive expiratory pressure (PEP) device maintains pressure in the lungs, keeping the airways open and allowing air to get behind the mucus to improve airway clearance, lung volume capacity, and oxygenation. The device can be used by children (A) and adults (B). (PARI PEP is a registered trademark of PARI GmbH. Used with permission. Please note: the authors have no commercial gain from inclusion of this product.)
The Flutter valve is similar to the PEP device but utilizes a stainless steel ball that vibrates, alternately opening and closing the device’s air hole, pulsing air back into the airways (Fig. 15-16). Current practice is to use one of the devices (PEP or Flutter valve) followed by autogenic breathing techniques. The total treatment time is about 15 minutes and can be carried out independently by some children and most adolescents and adults with mild-to-moderate disease who can follow the directions and control their breathing.
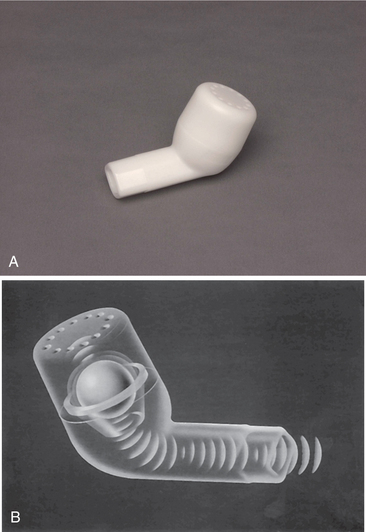
Figure 15-16 The Flutter mucus clearance device. A, Flutter device can be used in anyone with chronic obstructive pulmonary disease (COPD). B, Flutter valve shown in this schematic utilizes a stainless steel ball that vibrates, alternately opening and closing the device’s air hole, pulsing air back into the airways. (Courtesy Axcan Pharma, Birmingham, AL, 2007. Used with permission.)
Preliminary studies on the Flutter device suggest that Flutter valve therapy is an acceptable alternative postural drainage and percussion and may be more effective than postural drainage in prolonging the ability to raise secretions. Sputum production increased significantly 30 minutes after the end of treatment, and 1 hour after the end of treatment, it was significantly greater than the amount produced after postural drainage.26,170 In a comparison study lasting 1 year, PEP was superior to the Flutter in maintaining pulmonary function, reducing hospitalizations, and reducing antibiotic use.291
A high-frequency chest wall oscillation vest can provide mucus clearance for those individuals who lack the ability to perform the simpler techniques and is especially helpful with children (although the cost may seem prohibitive, it is less than a single hospitalization for a pulmonary exacerbation). The device consists of an inflatable vest (The Vest) attached to an air pulse generator (Fig. 15-17). The generator, a compressor-like device, rapidly inflates and deflates the vest, gently compressing and releasing the chest wall to create airflow within the lungs. This device treats all lobes simultaneously for the duration of the time it is activated. This process moves mucus toward the larger airways where it can be cleared by coughing.
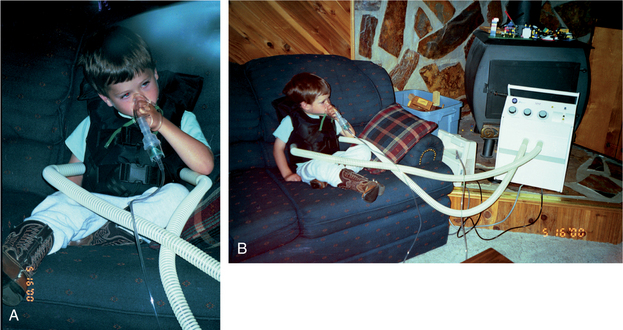
Figure 15-17 A, 4-year-old wearing American Biosystems, Inc (ABI) Vest to self-administer high-velocity oscillations. The vest can accommodate a child as young as 2 years old and is worn 30 minutes twice each day. Medications used to open airways and relax bronchospasms are administered through a nebulizer during the first 10 minutes of treatment, and the machine automatically shuts off every 10 minutes to allow the person to clear secretions. B, A foot-pad control mechanism (not shown) can be used to manually stop the machine anytime to allow the client to cough and clear mucus. Any position can be assumed, and at this age, the child can do everything himself. This device promotes compliance and is accompanied by reduced use of medications and infections. (Courtesy Kerry Resch, Missoula, MT, 2001.)
The effectiveness of this mechanized device has been supported by the limited research published to date. Results from The Vest are becoming quite well documented, demonstrating decreased hospitalizations and slower rate of FEV1 decline.255,377 The Vest Clearance System web site (http://www.thevest.com/research/bibliography.asp) has an extensive bibliography. Future research should determine optimal compression frequencies and wave forms.255,299,377 The Vest is comparable to PEP in effectiveness, but arterial oxygen saturation may drop during its use, so this must be monitored during exacerbations.107
Nutrition
Malnutrition and deterioration of lung function are closely interrelated and interdependent in the person with CF. Each affects the other, leading to a spiral decline in both. The occurrence of malnutrition during childhood seems to be associated with impaired growth and repair of the airway walls. In children, when growth in body length occurs, good nutrition is associated with better lung function. When adequate nutrition is combined with physical training and aerobic exercise, improved body weight, respiratory muscle function, lung function, and exercise tolerance occur with increases in both respiratory and other muscle mass.192
Exercise
Increasingly, exercise and sport are being advanced as core components of treatment for individuals with CF of all ages. A large portfolio of exercise literature has already established that supervised exercise programs enhance fitness (and thereby improve survival), increase sputum clearance, delay the onset of dyspnea, delay declines in pulmonary function, prevent decrease in bone density,444 enhance cellular immune response,46 and increase feelings of well-being, thereby potentially improving self-image, self-confidence, and quality of life for the person with CF. Both short-and long-term aerobic and anaerobic training have positive effects on exercise capacity, strength, and lung function.55
Reduced systemic oxygen extraction is an important factor, limiting exercise in many individuals with CF, but the specific parameters of this limitation remain unknown and probably vary from person to person.312,325 Even unsupervised programs produce a training effect and pulmonary benefits.306 Inspiratory muscle training alone in individuals with CF has shown improved lung function and increased work capacity, as well as improved psychosocial status.113,127
The therapist can be very instrumental in providing client and family education about the importance of combining good nutrition and exercise/activity. The therapist helps each individual develop an exercise routine that includes strengthening, stretching, aerobic, and endurance components with special attention to breathing exercises to aerate all areas of the lungs. Weight loss with exercise is of special concern in this population, especially for the individual with CF and diabetes mellitus.
Energy expenditure is higher than usual for individuals with CF and diabetes during periods of recovery from mild exercise or activity because of increased work of breathing (WOB) consistent with higher ventilatory requirements.441 This requires careful collaboration among client/family, therapist, and nutritionist. In addition, systemic inflammatory response to exercise may be greater for individuals with CF, potentially exacerbating the disease.213
As longevity increases with CF, quality of life issues are more important, thus these issues, such as posture, arthropathies, and neuromuscular control, are becoming more important for the physical therapist.121,285 When treating anyone with CF who has sustained trauma, a multisystem approach is critical for optimal outcomes in physical therapy.411
Individuals with CF awaiting a transplant must remain as active as possible; whenever possible, the therapist can design a safe but effective exercise program. If significant oxygen desaturation or severe breathlessness limits activity, then exercise on a treadmill, stationary bike, or even a stationary device for seated pedaling is recommended with supplemental oxygen supplied at sufficient flow to match minute ventilatory requirements.444
Studies of exercise performance in lung transplant recipients with end-stage CF report that exercise performance improves after transplantation but remains well below normal.325 In a study of 12 individuals 8 to 95 months after lung transplant, the diaphragm and abdominal muscle strength was preserved relative to healthy controls, but quadriceps strength was significantly diminished and affected exercise performance. Corticosteroid use partly contributed to this strength deficit.342
Athletes with Cystic Fibrosis
For those individuals who are able and interested in participating in sports (Fig. 15-18), special considerations must be addressed. Calorie intake and maintaining weight, nutrition, and fluid and electrolyte balance are major concerns. Each individual must be assessed, evaluated, and monitored carefully. The therapist, family, and nutritionist can work together to minimize exercise and nutrition-induced complications. The information presented here is only a general guideline and may need to be modified for individual needs, metabolism, personal health, level and type of sports participation, and so on.

Figure 15-18 A, 18-month-old shortly after diagnosis; face-mask is a nebulizer (device designed to create and throw an aerosol) that is delivering albuterol (bronchodilator). B, This same individual in 1999 at age 15 years (6 feet tall; 145 lb) competing in a regional soccer tournament. C, Same young man at 23 years (6 feet 2 inches, 175 lb), still actively hiking, cycling, running, and playing intramural sports, while enrolled in graduate school. (Courtesy Kevin Hanson, Helena, MT, 2007. Used with permission.)
During the off-training season, the individual (especially children and adolescents) will need to eat one and one-half times the protein and calories of an athlete who does not have CF in order to maintain weight. During the sport season, calorie intake must be increased with the goal of maintaining weight.
Hyponatremia (deficiency of sodium in the blood; see discussion in Chapter 5) can be a serious problem for athletes with CF who excrete three to five times the sodium (in sweat) of an athlete without CF during sports participation. This situation combined with increased intake of water further dilutes the sodium levels in the body. Sodium loss combined with losses of potassium and magnesium can result in lifethreatening situations for these individuals.
Some guidelines for these athletes include weighing before and after exercise and considering the loss as a loss of fluids accompanied by electrolytes; replacing fluid loss with electrolytes (e.g., drinks such as Gatorade or Recharge) instead of water; taking an appropriate number of salt tablets; and eating a meal with sodium-, potassium-, and magnesium-rich foods.
Sporting activities should not be undertaken during infective exacerbations. Sports that carry a medical risk for people with CF (e.g., bungee and parachute jumping, skiing, or scuba diving) should be avoided. Individuals with CF who have portal hypertension with significant enlargement of the spleen and liver should be advised against contact sports, such as rugby and football, in addition to bungee and parachute jumping.
Skiing for anyone with CF who is already hypoxic is not advised; episodes of acute right-sided heart failure brought on by a combination of altitude and unaccustomed strenuous anaerobic and aerobic exercise have been reported. Scuba diving is contraindicated for anyone with lung disease if there is any evidence of air trapping. On ascent, the air expands, increasing the risk of developing a pneumothorax.444
Transition to Adult Care
CF centers and other centers providing life-long care for clients with CF have realized the need for a transition phase between pediatric and adult care and the provision of counseling for parents and the young adult with CF to assist them with this marked change in approach to the young adult’s care.
The physical therapist can and should have an integral role in preparing families and clients for various phases in care from pediatrics to adolescent care to a more independent model of adult care. If such a program does not exist in your current facility, materials and resources are available to help get such a program started and established.40,85,289,458
Many families receive care for their child for years at a pediatric center and develop life-long relationships with health care agencies and staff. Every effort should be made to accomplish a smooth transition for the client and the family, since the move from the well-established pediatric facility to a new facility or medical team can be a stressful transition for all.
Families in rural settings or who travel distances to benefit from centralized services in CF clinics or centers have some unique needs that should be addressed as well. For example, maintaining a complete medical record in more than one facility is not always possible. Without a good systems coordinator, gaps in the medical record from one clinic to another become all too common.
Transition to adult care should be a planned process over time. Every effort should be made to avoid an abrupt transfer. The pediatric team has the responsibility to “set the stage” for the transition. The process can be started early with expectations for an eventual adult transition reinforced in intervals. Written information about the transition, setting an actual time frame, and planning each step in the process are important.289
Three guiding principles are suggested, regardless of the format chosen for transition. First, there must be an adult team that is both interested and able to provide care. Second, there needs to be a very well planned, coordinated approach to the transition. Third, there must be excellent communication and interaction between the pediatric and adult teams.289 Transition programs should be flexible enough to meet the needs of a wide range of young people, health conditions, and circumstances. The actual transfer of care should be individualized to meet the specific needs of young people and their families.362 Family involvement, including parents, guardians, and/or partner and the client, is essential to the success of any transition phase. The omission of any key people from the transition team can result in frustration, feelings of abandonment, and miscommunication, which could ultimately lead to compromised care for the individual with CF.
Adults With Cystic Fibrosis
As individuals with CF survive longer into adulthood, the unique needs of this population group are being considered. Health care in the adult setting encourages independence and increased self-reliance.459 Achieving an ideal nutritional status is an integral part of management of people with CF, but how these requirements change as the individual ages remains unknown. Emphasis is continually placed on dietary intake and weight; the effects of this on eating behavior and self-perceptions are under investigation.1,424,446
Other concerns include the effects of long-term use of pancreatic enzymes, osteoporosis associated with late-stage CF and its complications of increased fracture rates and severe kyphosis,19,20,191 the effects of hormonal changes in relation to the menstrual cycle on lung function,224 psychosocial-spiritual issues,200 and infertility issues.209
The origin of bone disease in CF is multifactorial and not completely understood. However, glucocorticoid therapy, delayed pubertal maturation, malabsorption of vitamin D, poor nutritional status, inactivity, and hypogonadism are all potential contributing factors.105a In addition, chronic pulmonary inflammation causes increased circulating levels of cytokines that augment bone resorption and suppress bone formation. Decreases in BMD resulting from these factors can lead to osteoporosis, fragility fractures, and possible exclusion from lung transplant candidacy.105a
Although a number of therapeutic options are now available for osteoporosis, it is likely that prevention, prompt recognition, and early treatment will be far more effective in achieving bone health than intervention at later stages of the problem. An in-depth discussion of bone health and disease in CF is available.105a
Stress urinary incontinence has also been shown to affect many girls and women with CF, probably caused by the forceful and prolonged coughing bouts characteristic of the disease.94,294 Interventions aimed at improving pelvic floor muscle strength and coordination may be appropriate for these individuals.